Abstract
Background
Sodium zirconium cyclosilicate (SZC; formerly ZS-9) is an oral potassium binder for the treatment of hyperkalemia in adults. SZC acts in the gastrointestinal tract and additionally binds hydrogen ions in acidic environments like the stomach, potentially transiently increasing gastric pH and leading to drug interactions with pH-sensitive drugs. This study assessed potential pharmacokinetic (PK) interactions between SZC and nine pH-sensitive drugs.
Methods
In this single-dose, open-label, single-sequence cross-over study in healthy adults, amlodipine, atorvastatin, clopidogrel, dabigatran, furosemide, glipizide, levothyroxine, losartan or warfarin were each administered alone and, following a washout interval, with SZC 10 g. Maximum plasma concentration (Cmax), area under the plasma concentration–time curve from 0 to the last time point (AUC0–t) and AUC extrapolated to infinity (AUCinf) were evaluated. No interaction was concluded if the 90% confidence interval for the geometric mean ratio (SZC coadministration versus alone) of the PK parameters was within 80–125%.
Results
During SZC coadministration, all PK parameters for amlodipine, glipizide, levothyroxine and losartan showed no interaction, while reductions in clopidogrel and dabigatran Cmax, AUC0–t and AUCinf (basic drugs) were <50% and increases in atorvastatin, furosemide and warfarin Cmax (acidic drugs) exceeded the no-interaction range by ˂2-fold.
Conclusions
SZC coadministration was associated with small changes in plasma concentration and exposure of five of the nine drugs evaluated in this study. These PK drug interactions are consistent with transient increases in gastric pH with SZC and are unlikely to be clinically meaningful.
Keywords: drug interactions, gastric pH-sensitive drugs, hyperkalemia, pharmacokinetic analysis, sodium zirconium cyclosilicate
INTRODUCTION
Hyperkalemia is defined as elevated potassium (K+) concentrations (generally >5.0 mmol/L) that increase the likelihood of adverse outcomes, including life-threatening cardiac arrhythmias [1–3]. Patients with chronic kidney disease (especially end-stage renal disease), heart failure and/or diabetes are at an increased risk of developing hyperkalemia [1, 4]. Medications such as renin–angiotensin–aldosterone system inhibitors, mineralocorticoid receptor antagonists and nonsteroidal anti-inflammatory drugs can also increase the likelihood of hyperkalemia, whereas some diuretics decrease hyperkalemia risk [1]. As patients with hyperkalemia are often treated for multiple comorbidities with numerous concomitant medications, the potential for drug–drug interactions should be considered prior to the prescription of hyperkalemia treatment.
Potassium (K+) binders such as sodium zirconium cyclosilicate (SZC, formerly ZS-9), patiromer and sodium polystyrene sulfonate (SPS) are available options for hyperkalemia treatment [5–7]. These compounds are not systemically absorbed, remaining in the gastrointestinal tract until excreted [1, 8], thereby reducing the possibility of metabolic enzyme– or active transporter–based interactions. Nevertheless, these binding agents may directly or indirectly interact with coadministered medications in the gastrointestinal tract. SPS and patiromer are nonspecific, polymeric, cation-binding resins that bind medications such as warfarin, metoprolol, phenytoin, furosemide, amlodipine and amoxicillin (with SPS) [5] or reduce systemic exposure of coadministered ciprofloxacin, levothyroxine and metformin (with patiromer) [7, 9]. Therefore the prescribing information for SPS and patiromer recommend separating the administration of these K+ binders from other oral medications by 3 h to minimize potential drug–drug interactions [5, 7]. In contrast, SZC is an inorganic, crystalline compound with an electrostatic pocket of 0.3-nm diameter that selectively binds monovalent cations, particularly K+ and ammonium () ions [10], but may also bind hydrogen (H+) ions at low pH. SZC in vitro studies showed no evidence of uptake or direct binding to concomitant medications [11]; however, H+ binding in the stomach may transiently increase gastric pH and alter the absorption and bioavailability of oral drugs with pH-dependent solubility [6, 12, 13]. It is hypothesized that the peak rise in gastric pH with SZC may be analogous to that of proton-pump inhibitors (PPIs), but acting as a localized effect lasting ≤2 h. Although transient in nature, increased gastric pH with SZC may still result in drug–drug interactions. Thus there is a need to examine the effect of SZC on the pharmacokinetic (PK) profiles of commonly used medications with gastric pH-dependent absorption.
This clinical pharmacology study was conducted to assess the effect of concomitant SZC administration on the PK profiles of nine weakly acidic or basic drugs.
MATERIALS AND METHODS
Study design
A single-center, single-dose, open-label, single-sequence cross-over study assessed the effect of SZC on the PK profiles of nine coadministered drugs (amlodipine, atorvastatin, clopidogrel, dabigatran, furosemide, glipizide, losartan, levothyroxine and warfarin). These drugs were identified as being potentially affected by SZC following in vitro screening.
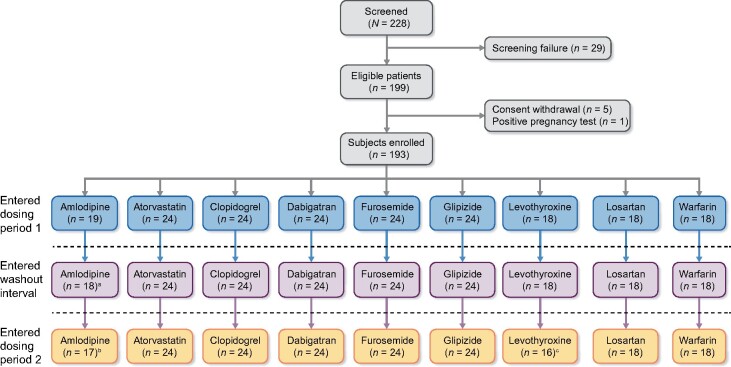
In two 9-day dosing periods, participants first received the drug alone and then concomitantly with a single dose of SZC 10 g (Figure 1). A prespecified washout interval, based on the half-life of each administered drug (i.e. ≥5 half-lives between doses), separated the two dosing periods.
FIGURE 1:
Study design. aThe washout interval was 7 days for the clopidogrel, dabigatran, glipizide, losartan and furosemide cohorts; 14 days for the atorvastatin, amlodipine and warfarin cohorts; and 35 days for the levothyroxine cohort.
The study was conducted at Riverside Clinical Research (Edgewater, FL, USA) and in accordance with the US Food and Drug Administration’s guidance for drug–drug interaction studies [14].
Study participants
Healthy adults 18–60 years of age with a body mass index (BMI) of 18–35 kg/m2 were eligible for study entry (Supplementary data, Table S1). Participants were required to have no clinically significant clinical laboratory results or electrocardiograms (determined by the investigator) and to be able to undergo repeated blood sampling or venous catheterization.
Major exclusion criteria were significant cardiovascular, respiratory, hepatic, renal, gastrointestinal or neurological disorders; a history of diabetes; sitting systolic blood pressure >150 mmHg or diastolic blood pressure >90 mmHg; a positive result for hepatitis B surface antigen or hepatitis C or human immunodeficiency virus antibodies; a history of excessive methylxanthine use within 30 days (determined by the investigator); regular use of drugs of abuse and/or positive findings on urinary drug screening; current tobacco use and/or positive findings on urinary cotinine screening; and alcohol consumption >28 U/week.
Concomitant drug therapy, including nonprescription medications, vitamins, minerals and dietary supplements, PPIs or antacids, was not permitted. Prescription medication within 14 days (except contraceptives in women with childbearing potential) and/or nonprescription medication within 7 days prior to dosing was not permitted.
In the clopidogrel, dabigatran and warfarin cohorts, use of concomitant medication that affected coagulation was not permitted and those with a significant active hematological disease, history of coagulopathy, bleeding disorders or a family history of premature cerebral hemorrhage, abnormal clotting test results at screening, head injury within the last 2 years or actual or potential hemorrhagic conditions were excluded.
Treatments and dosing
The assessed drugs are summarized in Table 1. A single dose of each drug alone was administered on study Day 1 of dosing period 1 and a single dose of the drug was coadministered with a single dose of SZC 10 g on study Day 1 of dosing period 2 with breakfast. Levothyroxine was administered 30 min before breakfast (per label) and SZC 10 g was administered with breakfast on study Day 1 of dosing period 2.
Table 1.
Chemical characteristics of the assessed drugs
| Drug | Evaluated dose | Recommended starting dosea | Dosage form | BCS classb | pKac | Acid/base |
|---|---|---|---|---|---|---|
| Amlodipine | 5 mg | 5 mg QD | Tablet | I [15] | 9.4 | Base |
| Atorvastatin | 10 mg | 10 or 20 mg QD | Tablet | II [16] | 4.3 | Acid |
| Clopidogrel | 75 mg | 75 mg QDd | Tablet | II [17] | 5.3 | Acid |
| Dabigatran | 75 mg | 75 or 150 mg BIDe | Capsule | II [18] | 4.0 and 6.7f | Base |
| Furosemide | 20 mg | 20–80 mg BID | Tablet | IV [15] | 3.9 | Acid |
| Glipizide | 5 mg | 5 mg QD | Tablet | II [19] | 5.9 | Acid |
| Levothyroxine | 50 µg | 100–125 µg/dayg | Tablet | III [15] | 2.2h | Acid |
| Losartan | 25 mg | 50 mg QD | Tablet | III [17] | 5.5 | Acid |
| Warfarin | 5 mg | Individualizedi | Tablet | I [15] | 5.1 | Acid |
BCS Class I defined as high permeability, high solubility; Class II as high permeability, low solubility; Class III as low permeability, high solubility and Class IV as low permeability, low solubility (https://www.ddfint.net/search.cfm).
300 mg loading dose given in patients with non-ST-segment elevation acute coronary syndrome.
75 mg QD in patients with creatinine clearance 15–30 mL/min.
PRADAXA (dabigatran etexilate)—Australian Package Insert.
Full replacement dose (1.7 µg/kg/day for 70 kg adult).
Individualized according to international normalized ratio and condition being treated.
BCS: biopharmaceutics classification system; BID: twice daily; pKa: the negative log of the acid dissociation constant or Ka value; QD: once daily.
Participants were instructed to swallow the tablets or capsules whole with water while standing upright and not to lie supine for 2 h after dosing. Staff prepared each SZC dose (slurry/suspension of two 5-g sachets in 40-mL water) immediately prior to its supervised administration.
Assessments
Participants stayed at the clinic for 24 h from morning of study Day 1 until after collection of the 24-h post-dose blood sample on study Day 2 and subsequently returned on study Days 2–9 of each dosing period for blood sampling. For each cohort, 2- to 6-mL blood samples were obtained at specified time points during each dosing period (Supplementary data, Table S2). Physical examination, evaluation of vital signs, electrocardiogram, laboratory testing and monitoring of adverse events (AEs) were also performed.
PK endpoints
Evaluated PK parameters were the observed maximum plasma or serum drug concentration (Cmax), area under the plasma concentration–time curve from 0 to the last measurable time point (AUC0–t), AUC extrapolated to infinity (AUCinf), time to Cmax (tmax) and terminal elimination half-life (t1/2). Only samples with concentrations greater than or equal to the lower limit of quantification (LLOQ) were used in the analysis. PK profiles with predose concentrations both ≥LLOQ and ≥5% of Cmax were excluded from the analyses.
Plasma concentrations of amlodipine, atorvastatin (parent and o-OH and p-OH atorvastatin metabolites), clopidogrel (parent and clopidogrel acid metabolite), levothyroxine (T3 and T4), losartan (parent and losartan acid) and the R- and S-isomers of warfarin were measured by Worldwide Clinical Trials (Austin, TX, USA). Plasma concentrations of furosemide, glipizide and dabigatran were measured by inVentiv Health (Québec City, Québec, Canada).
Statistical analysis
A sample size of 18 or 24 participants for each cohort was selected based on the within-subject coefficient of variation (WSCV) for Cmax or AUC. The atorvastatin, clopidogrel, dabigatran and furosemide cohorts, which had a high WSCV for Cmax (>30%), used an additive design in which 24 patients were initially enrolled, and if a data review indicated insufficient precision of the 90% CIs, the WSCV was used to calculate a sample size for a second cohort to provide the required precision when combined with the original cohort.
PK parameters were estimated by noncompartmental methods. Treatment comparison of parameters in the absence versus presence of SZC was performed by an analysis of variance model (SAS PROC GLM; SAS Institute, Cary, NC, USA) using treatment and participant as the classification variables and natural logarithmic-transformed parameters. Natural log-transformed parameters and a two one-sided t-test was used to determine 90% CIs for the least-squares geometric mean ratios (GMRs) comparing the Cmax and AUC0–t of each drug administered alone versus with SZC; least-squares GMRs and 90% CIs were subsequently converted back to the original scale. An absence of any interaction was concluded if the 90% CI for the GMRs fell within 80–125% for each parameter.
Statistical analysis of PK parameters was performed using SAS version 9.4 (SAS Institute). Descriptive statistics, including the number of participants, arithmetic mean, geometric mean and coefficient of variation and minimum, median and maximum values, were calculated for plasma or serum concentrations and PK parameters. Safety results were summarized using descriptive statistics.
Research involving human participants
This study was conducted in accordance with US Code of Federal Regulations (Title 21) and the International Conference on Harmonization E6 (R1) Guidelines of Good Clinical Practice and was approved by the institutional review board. The Declaration of Helsinki and its most recent updates (Seoul, 2008) were observed.
Informed consent
All participants provided written informed consent.
Data sharing statement
Data underlying the findings described in this article may be obtained in accordance with AstraZeneca’s data sharing policy described at https://astrazenecagrouptrials.pharmacm.com/ST/Submission/Disclosure.
RESULTS
Participant disposition and baseline demographics
The study enrolled 193 participants between December 2015 and February 2016 and 189 (97.9%) completed the study (Figure 2). Participant demographics are summarized in Table 2. The majority of participants were female in the clopidogrel, glipizide, furosemide and amlodipine cohorts and male in the dabigatran, losartan, atorvastatin, warfarin and levothyroxine cohorts. In most cohorts, the majority were white, except for the levothyroxine cohort, which had equal proportions of White and Black/African American participants.
FIGURE 2:
Participant disposition. aOne participant from the amlodipine cohort was lost to follow-up during dosing period 1. bOne participant from the amlodipine cohort was unable to return on the final day of dosing period 2 and discontinued on Day 8. cTwo participants from the levothyroxine cohort who were unable to return for dosing period 2 were excluded based on the sponsor’s decision during the washout interval.
Table 2.
Participant demographics by drug cohort
| Patient demographic | Drug cohort |
||||||||
|---|---|---|---|---|---|---|---|---|---|
| Clopidogrel (n = 24) | Dabigatran (n = 24) | Glipizide (n = 24) | Losartan (n = 18) | Furosemide (n = 24) | Atorvastatin (n = 24) | Amlodipine (n = 19) | Warfarin (n = 18) | Levothyroxine (n = 18) | |
| Age (years), mean (SD) | 35.0 (11.5) | 35.7 (11.3) | 40.4 (12.0) | 35.0 (12.5) | 33.9 (12.7) | 33.4 (13.1) | 34.7 (12.0) | 40.8 (12.3) | 35.0 (11.4) |
| Gender, n (%) | |||||||||
| Female | 19 (79.2) | 11 (45.8) | 20 (83.3) | 1 (5.6) | 13 (54.2) | 11 (45.8) | 10 (52.6) | 8 (44.4) | 2 (11.1) |
| Male | 5 (20.8) | 13 (54.2) | 4 (16.7) | 17 (94.4) | 11 (45.8) | 13 (54.2) | 9 (47.4) | 10 (55.6) | 16 (88.9) |
| Race, n (%) | |||||||||
| White | 19 (79.2) | 16 (66.7) | 17 (70.8) | 10 (55.6) | 17 (70.8) | 16 (66.7) | 14 (73.7) | 13 (72.2) | 8 (44.4) |
| Black/African American | 3 (12.5) | 8 (33.3) | 5 (20.8) | 8 (44.4) | 5 (20.8) | 8 (33.3) | 5 (26.3) | 4 (22.2) | 8 (44.4) |
| Asian | 1 (4.2) | 0 | 1 (4.2) | 0 | 0 | 0 | 0 | 0 | 1 (5.6) |
| Other | 1 (4.2) | 0 | 1 (4.2) | 0 | 2 (8.3) | 0 | 0 | 1 (5.6) | 1 (5.6) |
| Ethnicity, n (%) | |||||||||
| Hispanic | 4 (16.7) | 2 (8.3) | 2 (8.3) | 3 (16.7) | 1 (4.2) | 4 (16.7) | 1 (5.3) | 0 | 1 (5.6) |
| Non-Hispanic | 20 (83.3) | 22 (91.7) | 22 (91.7) | 15 (83.3) | 23 (95.8) | 20 (83.3) | 18 (94.7) | 18 (100.0) | 17 (94.4) |
| Weight (kg), mean (SD) | 75.0 (13.9) | 77.1 (15.4) | 78.4 (16.3) | 88.0 (11.3) | 76.5 (14.7) | 74.5 (12.8) | 78.0 (11.6) | 85.3 (19.0) | 85.4 (14.5) |
| BMI (kg/m2), mean (SD) | 26.6 (3.8) | 25.5 (4.0) | 27.0 (4.1) | 27.1 (2.9) | 25.8 (4.4) | 25.8 (3.5) | 26.5 (4.1) | 27.6 (4.4) | 27.8 (3.8) |
SD, standard deviation.
One participant in the amlodipine cohort was lost to follow-up after study Day 2 of dosing period 1 and another in the amlodipine cohort was discontinued on study Day 8 of dosing period 2. Two participants in the levothyroxine cohort were discontinued during the washout interval. Seven participants were excluded from the PK analyses due to having predose drug concentrations that were ≥LLOQ and ≥5% Cmax (one in the furosemide cohort and two in the warfarin cohort), insufficient predose concentrations ≥LLOQ (one in the losartan cohort and two in the atorvastatin cohort) and no predose concentrations ≥LLOQ (one in the clopidogrel cohort).
Effect of SZC coadministration on PK profile
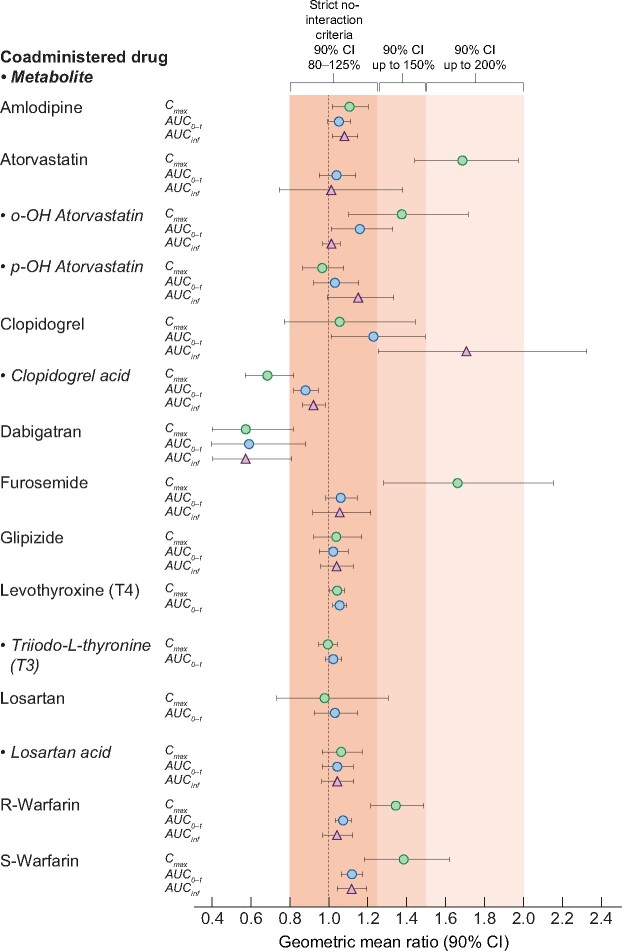
SZC coadministration had no apparent effect on the PK profiles of amlodipine 5 mg, glipizide 5 mg, levothyroxine 50 µg (or its triiodo-L-thyroxine metabolite) or losartan 25 mg (or its acid metabolite). Criteria for no interaction (GMR 90% CIs within 80–125%) were not met with some PK parameters for clopidogrel 75 mg, dabigatran 75 mg, atorvastatin 10 mg, furosemide 20 mg or warfarin 5 mg during SZC coadministration (Figure 3). The parameters of each drug alone or with SZC are summarized in Supplementary data, Table S3.
FIGURE 3:
Forest plot of the least-squares GMR (90% CI) for SZC + drug versus drug alone. Dark shading indicates strict no-interaction criteria (90% CI of GMR within 80–125%), medium shading indicates 90% CI up to 150% and light shading indicates 90% CI up to 200%. AUC0–t, area under the plasma concentration-time curve from 0 to the final time with a concentration ≥LLOQ; AUCinf, area under the plasma concentration-time curve extrapolated to infinity.
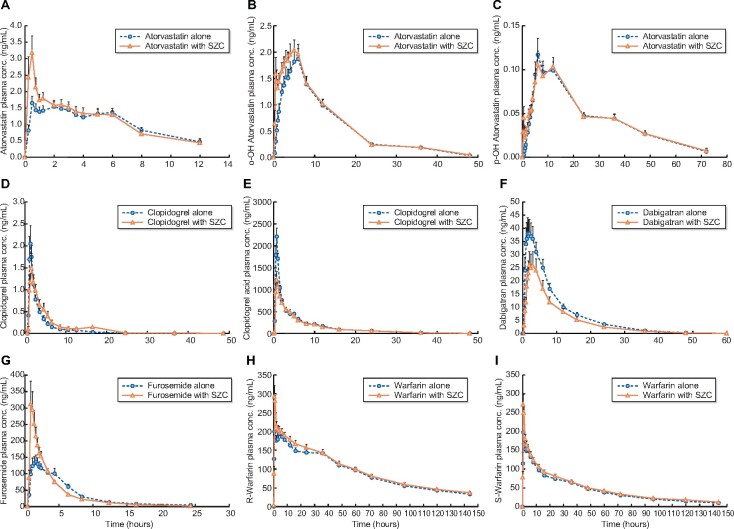
Following SZC coadministration, the Cmax and AUC0–t of clopidogrel and dabigatran were reduced but the Cmax of atorvastatin, furosemide and warfarin was increased. Plasma concentration–time profiles for these five drugs and their metabolites administered alone or with SZC are shown in Figure 4.
FIGURE 4:
Plasma concentration-time curves for (A) atorvastatin, (B) o-OH atorvastatin, (C) p-OH atorvastatin, (D) clopidogrel, (E) clopidogrel acid, (F) dabigatran, (G) furosemide, (H) R-warfarin and (I) S-warfarin with and without SZC coadministration.
The Cmax of atorvastatin and o-OH atorvastatin were increased with SZC coadministration, with GMRs of 169 and 137%, respectively (Figure 3), and median tmax decreased by 0.38 and 1.00 h, respectively. The effect of SZC on the PK profile of atorvastatin and o-OH atorvastatin was transient, lasting <1 h (Figure 4). For p-OH atorvastatin, higher plasma concentrations were observed in the first hour following SZC coadministration, but there was minimal change in Cmax or tmax. AUC0–t and AUCinf were not markedly affected by SZC coadministration for parent atorvastatin and its two metabolites (Figure 3).
SZC coadministration with clopidogrel was associated with GMRs for AUC0–t and AUCinf for parent clopidogrel of 123 and 171%, respectively, and for its acid metabolite of 88 and 92%, respectively, while Cmax of the acid metabolite decreased (GMR of 68%) (Figure 3). Median tmax was also increased by 0.25 h for parent clopidogrel but unchanged for its acid metabolite.
SZC coadministration with dabigatran led to reduced dabigatran exposure, evident as GMRs for Cmax, AUC0–t and AUCinf of 57, 59 and 57%, respectively (Figure 3). The median tmax of dabigatran increased by 0.37 h.
Following SZC coadministration, the GMR for Cmax of furosemide was 166% (Figure 3) and the median tmax decreased by 1.00 h, while the geometric mean of t½ decreased by 1.42 h; AUC0–t and AUCinf showed minimal change compared with furosemide alone. Similarly, after SZC coadministration with warfarin, the GMRs for Cmax of R- and S-warfarin were 134 and 138%, respectively (Figure 3), although the median tmax remained unchanged for either warfarin isomer. GMRs for AUC0–t and AUCinf of S-warfarin were both 112%, while these AUC parameters showed minimal change (GMRs of 107 and 104%, respectively) for R-warfarin.
Safety
No AEs were reported in the clopidogrel and losartan cohorts in the presence and absence of SZC (Supplementary data, Table S4). Participants in the atorvastatin, dabigatran, furosemide and warfarin cohorts also did not report AEs during dosing period 1 (drug administered alone). Headache and AEs associated with gastrointestinal disorders (e.g. nausea or vomiting) were the most common events reported during the study, and these types of AEs occurred when drugs were administered alone or with SZC. All reported AEs were mild or moderate in severity. During dosing period 2, three participants (one each in the dabigatran, furosemide and atorvastatin cohorts) experienced AEs possibly related to either the coadministered drug and/or SZC; all AEs resolved within the same day. No deaths, serious AEs or study drug discontinuations due to an AE were reported.
DISCUSSION
This study examined nine commonly used weakly acidic or basic drugs for potential drug–drug interactions with coadministered SZC in healthy adults. The PK profiles of four drugs (amlodipine, glipizide, levothyroxine and losartan) met predefined no-interaction criteria (i.e. 90% CIs of systemic exposure ratios falling within 80–125%). Changes in PK profiles were observed for atorvastatin, clopidogrel, dabigatran, furosemide and warfarin following SZC coadministration; however, the effect on Cmax was >50% for only two drugs (atorvastatin and furosemide) and none of the parameters exceeded the no-interaction 90% CI range by >2-fold. Therefore these changes were not considered clinically meaningful and SZC coadministration is not expected to affect the clinical activity of these pH-sensitive drugs.
Based on preclinical studies in dogs, as well as studies in humans, SZC is not systemically absorbed, with undetectable plasma zirconium concentrations following administration [11, 20]. SZC binds K+ ions with high specificity throughout the gastrointestinal tract, with bound K+ being excreted in the feces [10, 21]. Clinical studies of adults with hyperkalemia demonstrated a rapid reduction in serum K+ concentrations, with an onset of action within 1 h of SZC administration [21], and the majority of patients achieved significant K+ reductions within 24 h in the 48-h correction period and maintenance of normokalemia for up to 28 days [22, 23]. Although in vitro studies indicate that direct binding of SZC to other drugs is unlikely [11], the partial protonation of SZC (i.e. Na+–H+) may alter the absorption of concomitantly administered drugs by acting as a buffer when suspended in aqueous media and transiently increasing gastric pH [10]. In this study, small changes in systemic exposure were observed, consistent with increased absorption of weak acids (atorvastatin, clopidogrel acid, furosemide and warfarin) or decreased absorption of weak bases (dabigatran), which is expected due to pH-dependent changes in their ionization and solubility [12, 13]. Furthermore, because SZC traps H+ ions in acidic environments and releases them in more basic environments with an asymptote of pH ∼5.5, the maximal theoretical gastric fluid pH increase associated with SZC is pH ∼5.5. PPIs are appropriate comparators for potential SZC drug interactions since they maintain gastric pH between 6 and 7 [24, 25], which is a similar or slightly higher gastric pH than would be observed with SZC. Therefore the concomitant use of PPIs with SZC is not expected to raise gastric pH higher than PPIs alone.
This study showed that SZC is unlikely to affect low-density lipoprotein cholesterol (LDL-C) reductions by atorvastatin. Among participants receiving atorvastatin, SZC coadministration increased the atorvastatin and o-OH atorvastatin Cmax by 69 and 37%, respectively, but had no impact on AUC0–t or AUCinf. No changes in Cmax or AUC were noted for p-OH metabolite. Of note, parent atorvastatin and its hydroxyl (o-OH and p-OH) metabolites are all pharmacologically active [26]. Food also alters atorvastatin Cmax and AUC >24 h (AUC0–24), with decreases of 25 and 9%, respectively, without impacting LDL-C reductions [27]. Similarly, morning versus evening administration reduces atorvastatin exposure by ∼30% without impacting its activity [28]. Given that SZC coadministration did not affect the atorvastatin AUC and the Cmax effects were transient, it is unlikely that SZC affects atorvastatin activity.
The observed changes in clopidogrel exposure with SZC coadministration are not expected to affect clopidogrel efficacy. As clopidogrel is extensively metabolized following absorption, systemic clopidogrel exposure is variable, with very small amounts of parent drug being detectable in plasma [29]. In general, the AUC of clopidogrel acid (predominant inactive metabolite) is >1000-fold greater than that of the parent drug and is therefore the most clinically relevant PK parameter for determining clopidogrel absorption. The observed decreases in the clopidogrel acid AUC0–t and AUCinf, as well as the delayed tmax, are consistent with reduced clopidogrel solubility and a slight decrease in absorption caused by increased gastric pH with SZC. A previous study observed similar changes when clopidogrel was administered with food [30]. In fed versus fasted studies, clopidogrel acid Cmax decreased by 10–16% without altering the antiplatelet activity of clopidogrel [30]. During coadministration of clopidogrel with PPIs that do not inhibit cytochrome P450 (e.g. dexlansoprazole, lansoprazole and pantoprazole), the clopidogrel active metabolite AUC was reduced by up to 14% [31, 32]. Similar to this study, coadministration of clopidogrel with pantoprazole led to a 19% increase in clopidogrel parent drug AUC0–24 and a 14% decrease in active metabolite AUC0–24 [31]. As PPIs also increase gastric pH after administration, the study results indicate that SZC has a pH-based effect on clopidogrel absorption.
The anticoagulant activity of dabigatran is not expected to be altered by SZC coadministration. In this study there was a >40% decrease in dabigatran Cmax, AUC0–t and AUCinf with SZC coadministration. A possible explanation for decreased dabigatran exposure may be reduced stability of the ester prodrug due to higher gastric pH with SZC. There is a very shallow exposure–response relationship between dabigatran plasma concentrations and activated partial thromboplastin time (aPTT). A study of the effect of pantoprazole coadministration with dabigatran in healthy volunteers showed that pantoprazole itself does not affect the exposure–response relationship between dabigatran plasma concentrations and aPTT [33]. Decreases in dabigatran steady-state AUC (18–19%) and Cmax (20–23%) were observed after pantoprazole coadministration, but there was no change in the anticoagulant activity of dabigatran [33]. Since the SZC interaction with dabigatran is also gastric acid pH-mediated, it is reasonable to conclude that SZC also does not alter this exposure–response relationship.
The diuretic effects of furosemide are not expected to change during SZC coadministration. Furosemide Cmax increased by 66% with SZC coadministration; however, the AUC0–t and AUCinf were unchanged. Simulated pharmacodynamic modeling using Chennavasin et al.’s method [34] indicated that the magnitude of furosemide Cmax change was associated with a 15% increase in 24-h Na+ excretion, which is within the normal variability for Na+ excretion after furosemide administration. Therefore the change in furosemide Cmax is considered not clinically relevant.
No special measures are considered necessary to manage drug–drug interactions during SZC coadministration with warfarin. SZC coadministration was associated with a 1.3- and 1.4-fold increase in the R- and S-warfarin isomer Cmax, respectively. However, the AUC0–t and AUCinf were within the no-interaction 90% CI range for both isomers. Since ≥3 days of warfarin administration are required to achieve stable concentrations and anticoagulation [35], warfarin AUC (rather than Cmax) is the driving PK parameter for anticoagulant effects. As the AUCs of both warfarin isomers were unaffected by SZC coadministration, SZC is not expected to alter the anticoagulation activity of warfarin. Any potentially relevant change in warfarin anticoagulation can be addressed during routine monitoring [36].
No new safety issues were observed during SZC coadministration in these drug–drug interaction studies. Commonly reported AEs (≤2 participants per cohort) were headache, nausea and vomiting.
Study limitations were the use of an additive design in some drug cohorts (atorvastatin, clopidogrel, dabigatran and furosemide) due to a requirement for a relatively large number of participants and the lack of pharmacodynamic endpoint assessments.
CONCLUSIONS
SZC coadministration was associated with small changes in the absorption of atorvastatin, clopidogrel, furosemide and warfarin (drugs with known pH-sensitive gastric absorption) that are unlikely to represent clinically meaningful interactions. The decreases in exposure of basic drugs (i.e. dabigatran) and increases in Cmax of acidic drugs (i.e. atorvastatin, clopidogrel acid, furosemide and warfarin) were consistent with an SZC-related transient increase in gastric pH.
SUPPLEMENTARY DATA
Supplementary data are available at ckj online.
Supplementary Material
ACKNOWLEDGEMENTS
The authors would like to thank Henrik S. Rasmussen and Adam Tomasi, both formerly of ZS Pharma, Inc., for contributions to the study design and execution. Medical writing support was provided by Sarah Greig, PhD (Auckland, New Zealand) and Mary Falcone, PhD (Philadelphia, PA, USA), of inScience Communications, Springer Healthcare and funded by AstraZeneca.
FUNDING
This study was funded by ZS Pharma, Inc., a subsidiary of AstraZeneca.
AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to the analysis and interpretation of study data, contributed to the drafting of the manuscript and provided their final approval of the version to be published. W.G.K. contributed to the study design and performed the PK and associated statistical analyses. All authors agreed to be accountable for all aspects of the work.
CONFLICT OF INTEREST STATEMENT
M.N. and D.W.B. are shareholders and full-time employees of AstraZeneca. W.G.K. is the principal member of Kramer Consulting LLC and was a consultant to ZS Pharma, Inc. when this study was conducted.
REFERENCES
- 1. Sarwar CMS, Bhagat AA, Anker SD. et al. Role of hyperkalemia in heart failure and the therapeutic use of potassium binders. Handb Exp Pharmacol 2017; 243: 537–560 [DOI] [PubMed] [Google Scholar]
- 2. An JN, Lee JP, Jeon HJ. et al. Severe hyperkalemia requiring hospitalization: predictors of mortality. Crit Care 2012; 16: R225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Kovesdy CP. Updates in hyperkalemia: outcomes and therapeutic strategies. Rev Endocr Metab Disord 2017; 18: 41–47 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Luo J, Brunelli SM, Jensen DE. et al. Association between serum potassium and outcomes in patients with reduced kidney function. Clin J Am Soc Nephrol 2016; 11: 90–100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.US Food and Drug Administration. Kayexalate (sodium polystyrene sulfonate) Powder for Suspension, for Oral or Rectal Use: Prescribing Information. 2017. http://www.accessdata.fda.gov/drugsatfda_docs/label/2017/011287s026lbl.pdf (17 July 2020, date last accessed)
- 6.US Food and Drug Administration. Lokelma (sodium zirconium cyclosilicate) for Oral Suspension: Prescribing Information. 2018. http://www.accessdata.fda.gov/drugsatfda_docs/label/2018/207078s000lbl.pdf (17 July 2020, date last accessed)
- 7.US Food and Drug Administration. Veltassa (patiromer) for Oral Suspension: Prescribing Information. 2018. http://www.accessdata.fda.gov/drugsatfda_docs/label/2018/205739s016lbl.pdf (17 July 2020, date last accessed)
- 8. Meaney CJ, Beccari MV, Yang Y. et al. Systematic review and meta-analysis of patiromer and sodium zirconium cyclosilicate: a new armamentarium for the treatment of hyperkalemia. Pharmacotherapy 2017; 37: 401–411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Lesko LJ, Offman E, Brew CT. et al. Evaluation of the potential for drug interactions with patiromer in healthy volunteers. J Cardiovasc Pharmacol Ther 2017; 22: 434–446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Stavros F, Yang A, Leon A. et al. Characterization of structure and function of ZS-9, a K+ selective ion trap. PLoS One 2014; 9: e114686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.European Medicines Agency. Lokelma: Assessment Report.2018. http://www.ema.europa.eu/en/documents/assessment-report/lokelma-epar-public-assessment-report_en.pdf (17 July 2020, date last accessed)
- 12. Mitra A, Kesisoglou F.. Impaired drug absorption due to high stomach pH: a review of strategies for mitigation of such effect to enable pharmaceutical product development. Mol Pharm 2013; 10: 3970–3979 [DOI] [PubMed] [Google Scholar]
- 13. Buxton ILO, Benet L.. Pharmacokinetics: the dynamics of drug absorption, distribution, metabolism and elimination. In: Brunton LL, Chabner BA, Knollman BCS (eds). Goodman & Gilman’s: The Pharmacological Basis of Therapeutics. Beijing, China: McGraw-Hill, ; 2011: 17–40 [Google Scholar]
- 14.US Food and Drug Administration. Guidance for Industry. Clinical Drug Interaction Studies—Study Design, Data Analysis, and Clinical Implications. 2017. http://www.fda.gov/downloads/drugs/guidances/ucm292362.pdf (17 July 2020, date last accessed)
- 15.World Health Organization. Proposal to Waive in vivo Bioequivalence Requirements for the WHO Model List of Essential Medicines Immediate Release, Solid Oral Dosage Forms. https://www.who.int/medicines/services/expertcommittees/pharmprep/QAS04_109Rev1_Waive_invivo_bioequiv.pdf (17 July 2020, date last accessed)
- 16. Prabhu P, Patravale V.. Dissolution enhancement of atorvastatin calcium by co-grinding technique. Drug Deliv Transl Res 2016; 6: 380–391 [DOI] [PubMed] [Google Scholar]
- 17. Ramirez E, Laosa O, Guerra P. et al. Acceptability and characteristics of 124 human bioequivalence studies with active substances classified according to the biopharmaceutic classification system. Br J Clin Pharmacol 2010; 70: 694–702 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.US Food and Drug Administration. Application Number: 22-512 Chemistry Review(s), 2010. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2010/022512Orig1s000ChemR_Corrrected%203.11.2011.pdf (17 July 2020, date last accessed)
- 19. Jamzad S, Fassihi R.. Development of a controlled release low dose class II drug - glipizide. Int J Pharm 2006; 312: 24–32 [DOI] [PubMed] [Google Scholar]
- 20. Stavros F, Rasmussen HS, Singh B. et al. Assessment of systemic absorption of sodium zirconium cyclosilicate (ZS-9): blood and urine Zr concentration in dogs during a 9-month oral toxicity study [abstract SA-PO927]. J Am Soc Nephrol 2015; 26: 846A [Google Scholar]
- 21. Ash SR, Singh B, Lavin PT. et al. A phase 2 study on the treatment of hyperkalemia in patients with chronic kidney disease suggests that the selective potassium trap, ZS-9, is safe and efficient. Kidney Int 2015; 88: 404–411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Kosiborod M, Rasmussen HS, Lavin P. et al. Effect of sodium zirconium cyclosilicate on potassium lowering for 28 days among outpatients with hyperkalemia: the HARMONIZE randomized clinical trial. JAMA 2014; 312: 2223–2233 [DOI] [PubMed] [Google Scholar]
- 23. Packham DK, Rasmussen HS, Lavin PT. et al. Sodium zirconium cyclosilicate in hyperkalemia. N Engl J Med 2015; 372: 222–231 [DOI] [PubMed] [Google Scholar]
- 24. Hatlebakk JG. Review article: gastric acidity – comparison of esomeprazole with other proton pump inhibitors. Aliment Pharmacol Ther 2003; 17: 10–15 [DOI] [PubMed] [Google Scholar]
- 25. Miehlke S, Madisch A, Kirsch C. et al. Intragastric acidity during treatment with esomeprazole 40 mg twice daily or pantoprazole 40 mg twice daily – a randomized, two-way crossover study. Aliment Pharmacol Ther 2005; 21: 963–967 [DOI] [PubMed] [Google Scholar]
- 26. Lins RL, Matthys KE, Verpooten GA. et al. Pharmacokinetics of atorvastatin and its metabolites after single and multiple dosing in hypercholesterolaemic haemodialysis patients. Nephrol Dial Transplant 2003; 18: 967–976 [DOI] [PubMed] [Google Scholar]
- 27. Whitfield LR, Stern RH, Sedman AJ. et al. Effect of food on the pharmacodynamics and pharmacokinetics of atorvastatin, an inhibitor of HMG-CoA reductase. Eur J Drug Metab Pharmacokinet 2000; 25: 97–101 [DOI] [PubMed] [Google Scholar]
- 28. Cilla DD Jr, Gibson DM, Whitfield LR. et al. Pharmacodynamic effects and pharmacokinetics of atorvastatin after administration to normocholesterolemic subjects in the morning and evening. J Clin Pharmacol 1996; 36: 604–609 [DOI] [PubMed] [Google Scholar]
- 29. Sangkuhl K, Klein TE, Altman RB.. Clopidogrel pathway. Pharmacogenet Genomics 2010; 20: 463–465 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Brvar N, Lachance S, Levesque A. et al. Comparative bioavailability of two oral formulations of clopidogrel: determination of clopidogrel and its carboxylic acid metabolite (SR26334) under fasting and fed conditions in healthy subjects. Acta Pharm 2014; 64: 45–62 [DOI] [PubMed] [Google Scholar]
- 31. Angiolillo DJ, Gibson CM, Cheng S. et al. Differential effects of omeprazole and pantoprazole on the pharmacodynamics and pharmacokinetics of clopidogrel in healthy subjects: randomized, placebo-controlled, crossover comparison studies. Clin Pharmacol Ther 2011; 89: 65–74 [DOI] [PubMed] [Google Scholar]
- 32. Frelinger AL 3rd, Lee RD, Mulford DJ. et al. A randomized, 2-period, crossover design study to assess the effects of dexlansoprazole, lansoprazole, esomeprazole, and omeprazole on the steady-state pharmacokinetics and pharmacodynamics of clopidogrel in healthy volunteers. J Am Coll Cardiol 2012; 59: 1304–1311 [DOI] [PubMed] [Google Scholar]
- 33. Stangier J, Stahle H, Rathgen K. et al. Pharmacokinetics and pharmacodynamics of the direct oral thrombin inhibitor dabigatran in healthy elderly subjects. Clin Pharmacokinet 2008; 47: 47–59 [DOI] [PubMed] [Google Scholar]
- 34. Chennavasin P, Seiwell R, Brater DC.. Pharmacokinetic-dynamic analysis of the indomethacin-furosemide interaction in man. J Pharmacol Exp Ther 1980; 215: 77–81 [PubMed] [Google Scholar]
- 35. Holford NH. Clinical pharmacokinetics and pharmacodynamics of warfarin. Understanding the dose-effect relationship. Clin Pharmacokinet 1986; 11: 483–504 [DOI] [PubMed] [Google Scholar]
- 36. Hirsh J, Fuster V, Ansell J. et al. American Heart Association/American College of Cardiology foundation guide to warfarin therapy. Circulation 2003; 107: 1692–1711 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.