Abstract
Background
Aspirin use reduces colorectal cancer (CRC) incidence, but there is limited evidence regarding associations of aspirin and non-aspirin non-steroidal anti-inflammatory drugs (NSAIDs) with CRC-specific survival.
Methods
This prospective analysis includes women and men from the Cancer Prevention Study-II Nutrition Cohort who were cancer free at baseline (1992 or 1993) and diagnosed with CRC during incidence follow-up through 2015. Detailed information on aspirin and non-aspirin NSAID use was self-reported on questionnaires at baseline, in 1997, and every 2 years thereafter. Pre- and postdiagnosis data were available for 2686 and 1931 participants without distant metastases, respectively, among whom 512 and 251 died from CRC during mortality follow-up through 2016. Secondary analyses examined associations between prediagnosis aspirin use and stage at diagnosis (distant metastatic vs localized or regional). All statistical tests were 2-sided.
Results
Long-term regular use of aspirin (>15 times per month) before diagnosis was associated with lower CRC-specific mortality (multivariable-adjusted hazard ratio [HR] = 0.69, 95% confidence interval [CI] = 0.52 to 0.92). Postdiagnosis regular aspirin use was not statistically significantly associated with risk of CRC-specific mortality overall (HR = 0.82, 95% CI = 0.62 to 1.09), although participants who began regular aspirin use only after their diagnosis were at lower risk than participants who did not use aspirin at both the pre- and postdiagnosis periods (HR = 0.60, 95% CI = 0.36 to 0.98). Long-term aspirin use before diagnosis was also associated with lower odds of diagnosis with distant metastases (multivariable-adjusted odds ratio = 0.73, 95% CI = 0.53 to 0.99).
Conclusions
Our results suggest that long-term aspirin use before a diagnosis of nonmetastatic colorectal cancer may be associated with lower CRC-specific mortality after diagnosis, consistent with possible inhibition of micrometastases before diagnosis.
Colorectal cancer (CRC) patients seek guidance on lifestyle factors to improve their prognoses (1). Evidence from randomized trials and observational studies convincingly demonstrate that relatively long-term regular aspirin use lowers risk of CRC incidence, even at low doses (2). Recently, however, the Aspirin in Reducing Events in the Elderly (ASPREE) study, a randomized trial of aspirin compared with placebo in people age 70 years or older, followed for an average of 4.7 years, reported an unexpected but statistically significant increase in CRC-specific mortality (hazard ratio [HR] = 1.77, 95% confidence interval [CI] = 1.02 to 3.06) with the low-dose intervention (3,4). This result may be because of chance (5) but nonetheless raises potential concerns about the relatively short-term effects of aspirin initiation on CRC mortality risk in the elderly.
There is currently no evidence from randomized trials on aspirin use and mortality outcomes specifically from studies of CRC survivors. Several observational studies, however, have examined the association between postdiagnosis aspirin use and CRC-specific mortality (6‐15). Some of these studies observed a reduction in mortality associated with postdiagnosis aspirin use (8‐12,14); others did not (6,7,13,15). The association between prediagnosis aspirin use and CRC mortality is also inconsistent across studies (6,8,11,16‐21). Most of these studies did not adjust their aspirin and CRC mortality estimates for potential confounding from non-aspirin non-steroidal anti-inflammatory drug (NSAID) use, an important limitation that bears consideration.
There is evidence that non-aspirin NSAIDs may inhibit colorectal carcinogenesis (22,23). Randomized clinical trials support the hypothesis that cyclooxygenase (COX)-2 selective inhibitors (ie, celecoxib and rofecoxib) are effective in preventing colorectal adenomas (24‐27). In a systematic review and meta-analysis, non-aspirin NSAIDS were associated with a reduced risk of incident CRC (28), but their association with CRC-specific mortality has been investigated in only a handful of studies with conflicting findings (14,16,17,20,21).
Both aspirin and non-aspirin NSAIDs block the metabolism of arachidonic acid through the prostaglandin H synthase or Cox pathways (29). Aspirin irreversibly inactivates COX-1 and COX-2 through acetylation, whereas non-aspirin NSAIDs reversibly inhibit the activity of these enzymes through competitive binding (30,31). Non-aspirin NSAIDs may inhibit colorectal carcinogenesis by inhibiting COX-2 (22,23). Aspirin, however, appears unlikely to inhibit carcinogenesis by inactivating COX-2; instead, it appears to inhibit carcinogenesis by inhibiting platelet activation through COX-1 and reducing thromboxane A expression (31). Mature platelets lack a nucleus and cannot replace inactivated COX-1, so even low-dose aspirin effectively inhibits platelet activation (31). Inhibition of platelet activation is thought to inhibit metastases (32,33) and may mediate the cancer-preventive properties of aspirin (31).
It is important to inform clinicians and CRC survivors about the potential benefits and harms of aspirin and non-aspirin NSAIDs. We used data from the Cancer Prevention Study-II Nutrition cohort (CPS-II) to examine the associations of pre- and postdiagnosis use of aspirin and non-aspirin NSAIDs with CRC-specific mortality among CRC survivors. Because inhibition of distant metastases is a potential mechanism of action for aspirin, we also explored associations of long-term aspirin use among case patients with and without distant metastases.
Methods
Study Participants
The analytic cohort for this analysis began with 4701 participants in the CPS-II Nutrition cohort who were free from colorectal cancer at baseline and who were subsequently diagnosed with invasive colon (International Classification of Diseases [ICD]-10: C18) or rectal cancer (ICD-10: C19-C20) after baseline enrollment (1992 or 1993) and before the end of incidence follow-up (June 30, 2015). Participants were followed for mortality outcomes through December 31, 2016. Exclusions were made for the following reasons: case participants identified through the National Death Index who could not be verified with cancer registries (n = 295), cases with a cancer diagnosed prior to enrollment into the CPS-II Nutrition cohort in 1992 or 1993 other than nonmelanoma skin cancer (n = 495), cases with an implausible diagnosis date (n = 16), cases with an unknown cancer stage at diagnosis (n = 164), cases with a nonadenocarcinoma CRC histology (n = 81), cases who were diagnosed with CRC and died on the same day (n = 4), and cases missing aspirin and/or non-aspirin NSAID data on the prediagnosis questionnaire (n = 532). We also excluded persons diagnosed with distant metastatic disease (n = 428) in our primary analyses because we anticipate the poor prognosis associated with distant metastatic disease would not be materially influenced by aspirin or non-aspirin NSAIDs, similar to our previous observations with other lifestyle and behavioral factors in this cohort (34‐39). We had 2686 participants for prediagnosis analyses, among whom 1568 deaths occurred (512 from CRC, 366 from cardiovascular disease [CVD], and 690 from all other causes). A total of 2036 participants returned a valid postdiagnosis questionnaire that included aspirin and non-aspirin NSAID information. In postdiagnosis analyses, the first year of follow-up after completion of the postdiagnosis questionnaire was excluded to avoid bias from reverse causation, because participants may stop using aspirin when they are close to death. This resulted in the exclusion of an additional 105 participants who died within 1 year of returning their postdiagnosis questionnaire. We had 1931 participants for postdiagnosis analyses that included 1043 deaths (251 from CRC, 274 from CVD, and 518 from all other causes).
Assessment of Aspirin and Non-Aspirin NSAID Use
Both pre- and postdiagnosis aspirin and non-aspirin NSAID use were categorized as none, less than regular (1-14 pills per month), or regular (≥15 pills per month). Prediagnosis use was defined from the self-reported questionnaire returned immediately prior to participant diagnosis (mean = 1.5 years before diagnosis).
Prediagnosis aspirin use and non-aspirin NSAID use durations were categorized into 5 categories based on the combination of reported use on the prediagnosis questionnaire and on the questionnaire returned immediately prior to that questionnaire (eg, for a participant who was diagnosed in 2000, her or his 1999 and 1997 questionnaires would be used to derive this variable). These categories were defined as follows: 1) no use on both the prediagnosis and the preceding questionnaires, 2) regular use on the prediagnosis but not on the preceding questionnaires (ie, shorter duration use), 3) regular use on both the prediagnosis and the preceding questionnaires (ie, longer duration use), 4) regular use on the prediagnosis questionnaire and unknown use on the preceding questionnaire (ie, unknown duration), and 5) less than regular use on the prediagnosis survey and/or mixed levels of use on the preceding questionnaire.
Analyses of postdiagnosis use were based on aspirin and non-aspirin NSAID use reported on the postdiagnosis questionnaire, defined as the first questionnaire returned after the diagnosis date (mean = 1.2 years), and used categories analogous to the above. Specifically, analyses of duration were based on combinations of use reported on the pre- and postdiagnosis questionnaires. Although some individuals had repeated questionnaire assessments of postdiagnosis aspirin use, we used only the first assessment after CRC diagnosis to reduce the potential for bias from reverse causation with questionnaires collected closer to the time of death.
Vital status, cause of death, and date of death were determined through linkage to the National Death Index through December 31, 2016. Cause of death was obtained for 99.3% of all known deaths in the cohort. The outcome of interest was defined as CRC-specific mortality (ICD-10: codes C18 to C20). We decided a priori not to examine overall mortality or CVD-specific mortality outcomes because of plausible confounding by CVD risk, the primary indication for daily aspirin use.
Statistical Analysis
Hazard ratios were estimated using Cox proportional hazards models. The validity of the proportional hazards assumption was tested by creating multiplicative interaction terms between NSAID use and time and comparing Cox models with and without interaction terms using the likelihood ratio test; no violations were observed. For analyses of prediagnosis use, each participant contributed person-time starting on their diagnosis date. For analyses of postdiagnosis use, each participant contributed person-time starting on a date 1 year after the completion date of his or her postdiagnosis questionnaire (using delayed-entry Cox model procedures) to reduce potential biases from reverse causation. Follow-up continued until death or the administrative end of follow-up on December 31, 2016.
All Cox models included age at diagnosis; sex; calendar year of diagnosis; education; Surveillance, Epidemiology, and End Results (SEER) disease stage; body mass index (BMI); cigarette smoking status; and personal history of CVD. We also examined the following as potential confounders: use of cholesterol-lowering drugs, history of CRC screening, self-reported history of diabetes, hypertension, and other cancers after baseline, but these were not included in the final multivariable models as the hazard ratios did not materially change. In addition, we examined prediagnosis aspirin and non-aspirin NSAID use as confounders of postdiagnosis aspirin and non-aspirin NSAID use and vice versa. Missing data for covariates were modelled as an “unknown or missing” category for categorical variables.
We examined whether associations of pre- and postdiagnosis total aspirin and non-aspirin NSAID use with CRC mortality were modified by age at diagnosis (younger than 70 vs 70 years or older), sex (male vs female), SEER stage (local vs regional), site (colon vs rectum), BMI (<30 vs ≥30 kg/m2), personal history of CVD (none vs present, updated using a time-dependent variable), and smoking (never vs ever) via multiplicative interaction terms and the likelihood ratio test.
We conducted sensitivity analyses of postdiagnosis aspirin use to assess potential reverse causation. These analyses excluded the first 2 years of follow-up after the postdiagnosis questionnaire and excluded participants whose postdiagnosis questionnaire was completed more than 18 months after diagnosis to reduce bias from measurement error.
Last, to examine the association between aspirin use and distant metastases at the time of diagnosis, we conducted a separate post hoc case-case analysis that included the main cohort of 2686 participants in the primary analyses and an additional 428 participants who had distant metastases at diagnosis (ie, cancer has spread to distant parts of the body such as the liver, lungs, or distant lymph nodes). In this group of 3114 participants, we modeled diagnosis with distant metastatic CRC as the outcome variable in a case-case logistic regression model that included prediagnostic aspirin use as the main exposure of interest and age, sex, diagnosis year, education, BMI, smoking, and history of CVD as co-variables. We also examined the associations of prediagnosis aspirin use with CRC-specific mortality among patients with distant metastatic disease. All statistical tests were 2-sided; P values less than .05 are considered statistically significant.
Results
Baseline characteristics for the prediagnosis period according to total aspirin and total non-aspirin NSAID use are shown in Table 1. Mean age at CRC diagnosis was 73.5 (SD = 7.3 ) years. Regular aspirin users were more likely to have a history of CVD and/or diabetes and to use cholesterol medications (all P < .001), as anticipated. Regular non-aspirin NSAID users were more likely to be women and to have a BMI of 30 kg/m2 or more. Associations of characteristics with postdiagnosis use were similar (Supplementary Table 1, available online). Most regular aspirin users reported use of 1 tablet per day for each day of the month (72.3% of prediagnosis users and 78.3% of postdiagnosis users), consistent with use for CVD prevention.
Table 1.
Descriptive characteristics of women and men diagnosed with nondistant metastatic colorectal cancer in the Cancer Prevention Study-II by strata of prediagnostic use of aspirin and non-aspirin NSAIDsa
| Characteristic | Aspirin |
Non-aspirin NSAIDS |
||||
|---|---|---|---|---|---|---|
| None | 1 to <15 pills per mo | ≥15 pills per mo | None | 1 to <15 pills per mo | ≥15 pills per mo | |
| No. (%) | No. (%) | No. (%) | No. (%) | No. (%) | No. (%) | |
| Total No. | 1328 | 234 | 1124 | 2056 | 271 | 359 |
| Age at diagnosis, y | ||||||
| <65 | 173 (13.0) | 53 (22.6) | 82 (7.3) | 234 (11.4) | 43 (15.9) | 31 (8.6) |
| 65 to <70 | 272 (20.5) | 44 (18.8) | 192 (17.1) | 388 (18.9) | 49 (18.1) | 71 (19.8) |
| 70 to <75 | 350 (26.4) | 53 (22.6) | 242 (21.5) | 471 (22.9) | 67 (24.7) | 107 (29.8) |
| 75 to <80 | 286 (21.5) | 50 (21.4) | 331 (29.4) | 522 (25.4) | 62 (22.9) | 83 (23.1) |
| 80 to <85 | 163 (12.3) | 26 (11.1) | 188 (16.7) | 297 (14.4) | 34 (12.5) | 46 (12.8) |
| ≥85 | 84 (6.3) | 8 (3.4) | 89 (7.9) | 144 (7.0) | 16 (5.9) | 21 (5.8) |
| Sex | ||||||
| Female | 682 (51.4) | 117 (50.0) | 443 (39.4) | 912 (44.4) | 128 (47.2) | 202 (56.3) |
| Male | 646 (48.6) | 117 (50.0) | 681 (60.6) | 1144 (55.6) | 143 (52.8) | 157 (43.7) |
| Race | ||||||
| White | 1294 (97.4) | 228 (97.4) | 1100 (97.9) | 2004 (97.5) | 265 (97.8) | 353 (98.3) |
| Black | 17 (1.3) | 3 (1.3) | 11 (1.0) | 25 (1.2) | 3 (1.1) | 3 (0.8) |
| Other/Unknown | 17 (1.3) | 3 (1.3) | 13 (1.2) | 27 (1.3) | 3 (1.1) | 3 (0.8) |
| Education | ||||||
| <High school | 86 (6.5) | 17 (7.3) | 68 (6.0) | 137 (6.7) | 19 (7.0) | 15 (4.2) |
| High school graduate | 381 (28.7) | 64 (27.4) | 282 (25.1) | 585 (28.5) | 56 (20.7) | 86 (24.0) |
| Some college | 416 (31.3) | 79 (33.8) | 321 (28.6) | 605 (29.4) | 88 (32.5) | 123 (34.3) |
| College graduate | 438 (33.0) | 73 (31.2) | 449 (39.9) | 720 (35.0) | 107 (39.5) | 133 (37.0) |
| Unknown | 7 (0.5) | 1 (0.4) | 4 (0.4) | 9 (0.4) | 1 (0.4) | 2 (0.6) |
| Stage | ||||||
| Local | 645 (48.6) | 112 (47.9) | 590 (52.5) | 1047 (50.9) | 128 (47.2) | 172 (47.9) |
| Regional | 683 (51.4) | 122 (52.1) | 534 (47.5) | 1009 (49.1) | 143 (52.8) | 187 (52.1) |
| BMI (kg/m2) | ||||||
| <18.5 | 27 (2.0) | 0 (0) | 19 (1.7) | 38 (1.8) | 5 (1.8) | 3 (0.8) |
| 18.5 to <25 | 538 (40.5) | 92 (39.3) | 439 (39.1) | 831 (40.4) | 120 (44.3) | 118 (32.9) |
| 25 to <30 | 501 (37.7) | 98 (41.9) | 460 (40.9) | 823 (40.0) | 89 (32.8) | 147 (40.9) |
| ≥30 | 246 (18.5) | 43 (18.4) | 199 (17.7) | 345 (16.8) | 55 (20.3) | 88 (24.5) |
| Unknown | 16 (1.2) | 1 (0.4) | 7 (0.6) | 19 (0.9) | 2 (0.7) | 3 (0.8) |
| Smoking | ||||||
| Never | 573 (43.1) | 104 (44.4) | 398 (35.4) | 846 (41.1) | 106 (39.1) | 123 (34.3) |
| Current | 85 (6.4) | 13 (5.6) | 40 (3.6) | 111 (5.4) | 10 (3.7) | 17 (4.7) |
| Former | 663 (49.9) | 116 (49.6) | 685 (60.9) | 1092 (53.1) | 154 (56.8) | 218 (60.7) |
| Unknown | 7 (0.5) | 1 (0.4) | 1 (0.1) | 7 (0.3) | 1 (0.4) | 1 (0.3) |
| CVD | 191 (14.4) | 25 (10.7) | 414 (36.8) | 477 (23.2) | 62 (22.9) | 91 (25.3) |
| Diabetes | 162 (12.2) | 23 (9.8) | (20.4) | 334 (16.2) | 33 (12.2) | 47 (13.1) |
| Hypertension | 603 (45.4) | 98 (41.9) | 659 (58.6) | 1034 (50.3) | 126 (46.5) | 200 (55.7) |
| Cholesterol medication | ||||||
| Never | 998 (75.2) | 175 (74.8) | 598 (53.2) | 1380 (67.1) | 172 (63.5) | 219 (61.0) |
| Former | 59 (4.4) | 8 (3.4) | 57 (5.1) | 93 (4.5) | 10 (3.7) | 21 (5.8) |
| Current | 271 (20.4) | 51 (21.8) | 469 (41.7) | 583 (28.4) | 89 (32.8) | 119 (33.1) |
Based on status at time of prediagnosis questionnaire. BMI = body mass index; CVD = cardiovascular disease; NSAIDs = non-steroidal anti-inflammatory drugs.
Regular use of aspirin on both the prediagnosis and immediately preceding questionnaire was associated with statistically significant lower CRC-specific mortality (multivariable-adjusted HR = 0.69, 95% CI = 0.52 to 0.92; Table 2). Other measures of aspirin use in the prediagnostic period generally resulted in hazard ratios that were below 1 but were not statistically significant. Overall, postdiagnosis use of aspirin or non-aspirin NSAIDs were not associated with CRC-specific mortality during a mean follow-up of 8.4 years (HR = 0.82, 95% CI = 0.62 to 1.09). However, current regular use of aspirin postdiagnosis, but not regular use for the prediagnosis questionnaire (eg, de novo use of aspirin in the postdiagnosis period), was associated with lower CRC-specific mortality (multivariable-adjusted HR = 0.60, 95% CI = 0.36 to 0.98). In sensitivity analyses that included distant metastatic patients alone, a null association between prediagnostic aspirin use and CRC-specific mortality was observed (fewer than 15 pills per month compared with nonuse: multivariable-adjusted HR = 0.94, 95% CI = 0.63 to 1.41, and ≥15 pills per month compared with nonuse: multivariable-adjusted HR = 1.00, 95% CI = 0.76 to 1.33).
Table 2.
Associations of pre- and postdiagnosis aspirin and non-aspirin NSAID use with colorectal cancer-specific mortality among women and men diagnosed with nondistant metastatic colorectal cancer in the Cancer Prevention Study-II cohort.
| Aspirin and non-aspirin NSAID categories | Aspirin |
Non-aspirin NSAIDs |
||
|---|---|---|---|---|
| Deaths per person to years | Multivariable HR (95% CI)a | Deaths per person to years | Multivariable HR (95% CI)a | |
| Prediagnosis | ||||
| Use on prediagnosis questionnaire | ||||
| Nonuse | 272/13 053 | 1.00 (Referent) | 397/19 420 | 1.00 (Referent) |
| 1 to <15 pills per month (less than regular use) | 39/2637 | 0.71 (0.50 to 1.01) | 53/2549 | 1.03 (0.77 to 1.39) |
| ≥15 pills per month (regular use) | 201/9666 | 0.84 (0.69 to 1.02) | 62/3387 | 0.86 (0.65 to 1.13) |
| Use on prediagnosis questionnaire and preceding questionnaire | ||||
| No use at prediagnosis questionnaire and no use at preceding questionnaire | 229/11 199 | 1.00 (Referent) | 349/17 165 | 1.00 (Referent) |
| Regular use at prediagnosis questionnaire but not at preceding questionnaire | 37/1831 | 0.84 (0.58 to 1.21) | 22/1092 | 0.80 (0.51 to 1.24) |
| Regular use at prediagnosis questionnaire and at preceding questionnaire | 85/4198 | 0.69 (0.52 to 0.92) | 18/1073 | 0.73 (0.45 to 1.19) |
| Regular use at prediagnosis questionnaire, no data from preceding questionnaire | 79/3637 | 1.02 (0.78 to 1.34) | 22/1221 | 1.09 (0.70 to 1.70) |
| Less than regular use on prediagnosis questionnaire and other less common patterns of use | 82/4491 | 0.87 (0.67 to 1.13) | 101/4803 | 1.03 (0.82 to 1.30) |
| Postdiagnosisb | ||||
| Use on postdiagnosis questionnaire | ||||
| None | 144/8932 | 1.00 (Referent) | 199/12 409 | 1.00 (Referent) |
| 1 to <15 pills per month (less than regular use) | 14/1374 | 0.78 (0.45 to 1.37) | 23/1952 | 0.81 (0.52 to 1.26) |
| ≥15 pills per month (regular use) | 93/5929 | 0.82 (0.62 to 1.09) | 29/1874 | 1.01 (0.68 to 1.51) |
| Use on postdiagnosis questionnaire and prediagnosis questionnaire | ||||
| No use at postdiagnosis questionnaire and no use at prediagnosis questionnaire | 104/6681 | 1.00 (Referent) | 162/10 541 | 1.00 (Referent) |
| Regular use at postdiagnosis questionnaire, but not at prediagnosis questionnaire | 20/1764 | 0.60 (0.36 to 0.98) | 13/945 | 0.96 (0.54 to 1.72) |
| Regular use at postdiagnosis questionnaire and at prediagnosis questionnaire | 73/4165 | 0.91 (0.66 to 1.27) | 16/929 | 1.11 (0.65 to 1.89) |
| Less than regular use on postdiagnosis questionnaire and other less common patterns of use | 54/3624 | 0.92 (0.66 to 1.29) | 60/3820 | 1.00 (0.73 to 1.35) |
Adjusted for age at diagnosis, diagnosis year category, sex, education, stage, body mass index, smoking and history of cardiovascular disease. CI = confidence interval; HR = hazard ratio; NSAIDs = non-steroidal anti-inflammatory drugs.
Postdiagnosis duration models do not include the “regular use at postdiagnosis questionnaire, no data from prediagnosis questionnaire” category because inclusion in the analysis required known prediagnosis use.
Our finding that long-term prediagnosis aspirin use was associated with lower CRC-specific mortality in participants with nondistant metastatic cancer motivated us to test the hypothesis that prediagnosis aspirin use might inhibit the development of distant metastases that are detectable at diagnosis. Consistent with our hypothesis, we found statistically significantly lower odds of distant metastases at the time of diagnosis with regular aspirin use on the prediagnosis questionnaire (multivariable-adjusted odds ratio = 0.77, 95% CI = 0.61 to 0.97) and with use on both the prediagnosis and preceding questionnaires (multivariable-adjusted odds ratio = 0.73, 95% CI = 0.53 to 0.99; Table 3).
Table 3.
Prediagnosis aspirin use and odds of distant metastatic colorectal cancer at time of diagnosis
| Prediagnosis aspirin use | Non-metastatic CRC, No. | Metastatic CRC, No. | OR (95% CI)a |
|---|---|---|---|
| Use on prediagnosis questionnaire | |||
| No use | 1328 | 235 | 1.00 (Referent) |
| 1 to <15 pills per month (less than regular use) | 234 | 44 | 1.06 (0.74 to 1.51) |
| ≥15 pills per month (regular use) | 1124 | 149 | 0.77 (0.61 to 0.97) |
| Use on prediagnosis questionnaire and preceding questionnaire | |||
| No use at prediagnosis questionnaire and no use at preceding questionnaire | 1097 | 196 | 1.00 (Referent) |
| Regular use on prediagnosis questionnaire but not at preceding questionnaire | 205 | 21 | 0.56 (0.35 to 0.92) |
| Regular use at prediagnosis questionnaire and at preceding questionnaire | 559 | 72 | 0.73 (0.53 to 0.99) |
| Regular use at prediagnosis questionnaire, no data from preceding questionnaire | 360 | 56 | 0.91 (0.65 to 1.27) |
| Less than regular use on prediagnosis questionnaire and other less common patterns of use | 465 | 83 | 0.98 (0.74 to 1.30) |
Adjusted for age at diagnosis, diagnosis year category, sex, education, body mass index, smoking, and history of cardiovascular disease. CI = confidence interval; CRC = cardiovascular disease; OR = odds ratio.
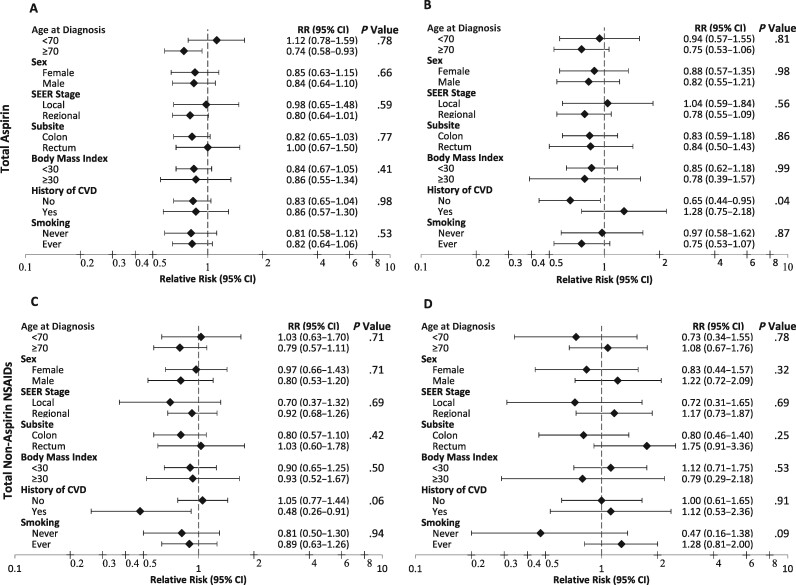
When evaluating potential effect modifiers, only personal history of CVD statistically significantly modified the association between regular aspirin use and CRC-specific mortality (Figure 1). Regular postdiagnosis aspirin use was associated with a lower risk of CRC mortality among persons without a history of CVD but not among persons with a history of CVD (Pinteraction = 0.04).
Figure 1.
Forest plots depicting potential effect modifiers of the association between aspirin and other non-steroidal anti-inflammatory drugs (NSAIDs) use pre- (A, C, respectively) and postdiagnosis (B, D, respectively) and colorectal cancer–specific mortality. The diamonds represent hazard ratios, and the error bars represent 95% confidence intervals (CIs). P values were 2-sided and were calculated via multiplicative interaction terms and the likelihood ratio test. CVD = cardiovascular disease; RR = relative risk; SEER = Surveillance, Epidemiology, and End Results.
Results for aspirin combined with non-aspirin NSAID use (ie, total NSAID use) were like those for aspirin alone (Supplementary Tables 2 and 3 and Supplementary Figure 1, available online). When analyses were performed using a 2-year lag period, associations were similar overall (data not shown).
Discussion
In this large prospective cohort study, postdiagnosis use of aspirin and non-aspirin NSAIDs was largely not associated with CRC-specific mortality among women and men diagnosed with CRC, albeit with a few exceptions for new aspirin users and persons without a history of CVD, which should be explored in future studies. However, regular long-term aspirin use before diagnosis was associated with lower CRC-specific mortality. These findings for prediagnosis aspirin use suggest that it might reduce CRC mortality in the overall population, in part, by limiting metastatic spread of colorectal tumors before diagnosis. Our findings of a reduced odds of distant metastatic cancer at diagnosis with prolonged aspirin use supports this hypothesis. Collectively, these results point toward a novel and potentially important model through which aspirin may prevent distant metastases at the time of diagnosis with colon or rectal cancer. This finding is particularly important because treatment options are usually quite good for localized and regional disease, whereas those options have limited success for patients diagnosed with distant metastases.
Our generally null findings for postdiagnosis aspirin use are in line with other studies that reported null associations between postdiagnosis aspirin use and CRC-specific mortality (6,7,13,15). However, other studies reported a statistically significant reduction in CRC-specific mortality with postdiagnosis aspirin use (8‐12,14). Among studies that examined both pre- and postdiagnosis use separately, 2 reported a reduction in CRC-specific mortality only with prediagnosis use (6,7), and 4 reported a reduction in CRC mortality only with postdiagnosis use (8‐11). These inconsistencies are not entirely understood. However, our analyses excluded participants with distant metastatic disease and prior studies did not, a potential source of bias. If patients with distant metastatic disease, near end of life, discontinued aspirin use in previous studies, this could have biased postdiagnosis aspirin use into appearing protective.
The 2 exceptions to our null findings for postdiagnosis aspirin use (ie, new users and when restricted to persons with no history of CVD) were unexpected. We are unaware of a clear biological explanation for these findings, and chance may play a role. Future studies of postdiagnosis aspirin use and CRC-specific mortality should determine if these intriguing findings can be replicated.
Because aspirin use before diagnosis was associated with lower CRC mortality in our study population, we hypothesized that prediagnosis aspirin use might inhibit the dissemination or survival of clinically occult (ie, micro-) metastases from the primary tumor, given that aspirin inhibits platelet activation and that activated platelets may play an important role in promoting metastasis (32,33). Long-term regular aspirin use before diagnosis was associated with lower odds of clinically apparent distant metastases at the time of diagnosis in this study, thus supporting our hypothesis. This finding is consistent with the reduced odds of distant metastasis among CRC cases observed in a pooled analysis of randomized trials of aspirin for CVD prevention (40).
Among studies that have examined prediagnosis aspirin use and CRC-specific mortality (6,8,11,16‐21), 3 studies reported statistically significant reductions in CRC mortality with prediagnosis aspirin use (6,16,21). Two of these studies reported stronger reductions with longer duration of use before diagnosis (16,21), consistent with our findings.
One limitation of our analysis is the inability to examine associations stratified by tumor molecular features. Some studies suggest the association between aspirin use and CRC risk and/or mortality may be modified by various molecular features of the tumor (8,10,15,18,19,41‐43).However, these results are inconsistent, and large pooled-data efforts are needed (44). Other limitations include incomplete medication use and unadjusted multiple comparisons.
An important strength of our study is the ability to examine non-aspirin NSAIDs separate from aspirin. Nearly all previous studies on aspirin and CRC were unable to do this. However, this is a line of research that could shed light on preventive mechanisms by distinguishing between platelet-mediated effects likely limited to aspirin (31) and other mechanisms involving direct inhibition of COX-2 that are shared with non-aspirin NSAIDs and higher doses of aspirin. In agreement with our findings, 3 studies reported no association between prediagnosis use of non-aspirin NSAIDs and CRC-specific mortality (16,17,20). No previous study has reported on non-aspirin NSAID use in the postdiagnosis period and CRC-specific mortality, adding to the clinical impact of these findings.
Given that this is an observational study, we cannot rule out the possibility that the observed associations are confounded by unaccounted risk factors. We also cannot dismiss the possibility that associations with postdiagnosis use could have been biased by reverse causation. Such bias could have occurred if cancer progression caused some individuals to stop, or not start, using aspirin or non-aspirin NSAIDs. However, such bias would have been expected to result in an inverse association between postdiagnosis use and CRC mortality, an association we largely did not observe. In addition, we reduced the potential for reverse causation by assessing use relatively soon after diagnosis with CRC (restricted to local or regional stage disease only) and excluding CRC deaths occurring within 1-2 years of assessment of postdiagnosis use.
Altogether, these data suggest that long-term aspirin use before diagnosis may be associated with lower risk of CRC-specific mortality following a diagnosis of nonmetastatic CRC. This association may be driven, partly, by suppression of clinically occult micrometastases around the time of diagnosis. Additional knowledge about the effect of postdiagnosis use will be gained from the ongoing randomized controlled trials studying the effect of aspirin on cancer mortality being conducted globally: Add-Aspirin trial (45), Adjuvant Aspirin for Colon Cancer (NCT02467582), ALASCCA trial (NCT02647099), ASCOLT trial (NCT00565708), and ASPIRIN Trial (NCT02301286).
Funding
American Cancer Society funds the creation, maintenance, and updating of the Cancer Prevention Study-II cohort.
Notes
Role of the funder: The funder had no role in the design of the study; the collection, analysis, and interpretation of the data; the writing of the manuscript; and the decision to submit the manuscript for publication.
Disclosures: Dr William Cance has a relationship with FAKnostics, LLC, but does not have any current products or services related to this manuscript. All other authors report that they have no disclosures or potential conflicts of interest.
Acknowledgments: We sincerely appreciate all Cancer Prevention Study-II participants and each member of the study and biospecimen management group. We acknowledge the contributions to this study from central cancer registries through the Centers for Disease Control and Prevention’s National Program of Cancer Registries and cancer registries supported by the National Cancer Institutes’ SEER Program.
Author contributions: JCF: Conceptualization, Methodology, Formal Analysis, Investigation, Writing Original Draft Preparation, Writing – Review & Editing. EJJ: Conceptualization, Methodology, Data curation, Formal Analysis, Investigation, Writing – Original Draft Preparation, Writing – Review & Editing. CCN: Conceptualization, Methodology, Data curation, Formal Analysis, Investigation, Visualization, Writing – Original Draft Preparation, Writing – Review & Editing. MAG: Conceptualization, Formal Analysis, Investigation, Writing – Review & Editing. WGC: Conceptualization, Formal Analysis, Investigation, Writing – Review & Editing. PTC: Conceptualization, Methodology, Data curation, Formal Analysis, Investigation, Supervision, Writing – Original Draft Preparation, Writing – Review & Editing.
Data Availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Supplementary Material
References
- 1. Siegel RL, Miller KD, Fedewa SA, et al. Colorectal cancer statistics, 2017. CA Cancer J Clin. 2017;67(3):177–193. [DOI] [PubMed] [Google Scholar]
- 2. Drew DA, Cao Y, Chan AT.. Aspirin and colorectal cancer: the promise of precision chemoprevention. Nat Rev Cancer. 2016;16(3):173–186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. McNeil JJ, Nelson MR, Woods RL, et al. Effect of aspirin on all-cause mortality in the healthy elderly. N Engl J Med. 2018;379(16):1519–1528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. McNeil JJ, Gibbs P, Orchard SG, et al. Effect of aspirin on cancer incidence and mortality in older adults. J Natl Cancer Inst. 2020;10.1093/jnci/djaa114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Chan AT, McNeil J.. Aspirin and cancer prevention in the elderly: where do we go from here? Gastroenterology. 2019;156(3):534–538. [DOI] [PubMed] [Google Scholar]
- 6. Bains SJ, Mahic M, Myklebust TA, et al. Aspirin as secondary prevention in patients with colorectal cancer: an unselected population-based study. J Clin Oncol. 2016;34(21):2501–2508. [DOI] [PubMed] [Google Scholar]
- 7. Cardwell CR, Kunzmann AT, Cantwell MM, et al. Low-dose aspirin use after diagnosis of colorectal cancer does not increase survival: a case-control analysis of a population-based cohort. Gastroenterology. 2014;146(3):700–708.e2. [DOI] [PubMed] [Google Scholar]
- 8. Chan AT, Ogino S, Fuchs CS.. Aspirin use and survival after diagnosis of colorectal cancer. JAMA. 2009;302(6):649–658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Goh CH, Leong WQ, Chew MH, et al. Post-operative aspirin use and colorectal cancer-specific survival in patients with stage I-III colorectal cancer. Anticancer Res. 2014;34(12):7407–7414. [PubMed] [Google Scholar]
- 10. Liao X, Lochhead P, Nishihara R, et al. Aspirin use, tumor PIK3CA mutation, and colorectal-cancer survival. N Engl J Med. 2012;367(17):1596–1606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. McCowan C, Munro AJ, Donnan PT, et al. Use of aspirin post-diagnosis in a cohort of patients with colorectal cancer and its association with all-cause and colorectal cancer specific mortality. Eur J Cancer. 2013;49(5):1049–1057. [DOI] [PubMed] [Google Scholar]
- 12. Frouws MA, Bastiaannet E, Langley RE, et al. Effect of low-dose aspirin use on survival of patients with gastrointestinal malignancies: an observational study. Br J Cancer. 2017;116(3):405–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Gray RT, Coleman HG, Hughes C, et al. Low-dose aspirin use and survival in colorectal cancer: results from a population-based cohort study. BMC Cancer. 2018;18(1):228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Hua X, Phipps AI, Burnett-Hartman AN, et al. Timing of aspirin and other nonsteroidal anti-inflammatory drug use among patients with colorectal cancer in relation to tumor markers and survival. J Clin Oncol. 2017;35(24):2806–2813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Hamada T, Cao Y, Qian ZR, et al. Aspirin use and colorectal cancer survival according to tumor CD274 (programmed cell death 1 ligand 1) expression status. J Clin Oncol. 2017;35(16):1836–1844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Coghill AE, Newcomb PA, Campbell PT, et al. Prediagnostic non-steroidal anti-inflammatory drug use and survival after diagnosis of colorectal cancer. Gut. 2011;60(4):491–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Din FV, Theodoratou E, Farrington SM, et al. Effect of aspirin and NSAIDs on risk and survival from colorectal cancer. Gut. 2010;59(12):1670–1679. [DOI] [PubMed] [Google Scholar]
- 18. Kothari N, Kim R, Jorissen RN, et al. Impact of regular aspirin use on overall and cancer-specific survival in patients with colorectal cancer harboring a PIK3CA mutation. Acta Oncol. 2015;54(4):487–492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Gray RT, Cantwell MM, Coleman HG, et al. Evaluation of PTGS2 expression, PIK3CA mutation, aspirin use and colon cancer survival in a population-based cohort study. Clin Transl Gastroenterol. 2017;8(4):e91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Veitonmaki T, Murtola TJ, Talala K, et al. Non-steroidal anti-inflammatory drugs and cancer death in the Finnish prostate cancer screening trial. PLoS One. 2016;11(4):e0153413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Zell JA, Ziogas A, Bernstein L, et al. Nonsteroidal anti-inflammatory drugs: effects on mortality after colorectal cancer diagnosis. Cancer. 2009;115(24):5662–5671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Chan AT, Arber N, Burn J, et al. Aspirin in the chemoprevention of colorectal neoplasia: an overview. Cancer Prev Res. 2012;5(2):164–178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Thun MJ, Henley SJ, Patrono C.. Nonsteroidal anti-inflammatory drugs as anticancer agents: mechanistic, pharmacologic, and clinical issues. J Natl Cancer Inst. 2002;94(4):252–266. [DOI] [PubMed] [Google Scholar]
- 24. Arber N, Eagle CJ, Spicak J, et al. Celecoxib for the prevention of colorectal adenomatous polyps. N Engl J Med. 2006;355(9):885–895. [DOI] [PubMed] [Google Scholar]
- 25. Baron JA, Sandler RS, Bresalier RS, et al. A randomized trial of rofecoxib for the chemoprevention of colorectal adenomas. Gastroenterology. 2006;131(6):1674–1682. [DOI] [PubMed] [Google Scholar]
- 26. Bertagnolli MM, Eagle CJ, Zauber AG, et al. Celecoxib for the prevention of sporadic colorectal adenomas. N Engl J Med. 2006;355(9):873–884. [DOI] [PubMed] [Google Scholar]
- 27. Thompson PA, Ashbeck EL, Roe DJ, et al. Celecoxib for the prevention of colorectal adenomas: results of a suspended randomized controlled trial. J Natl Cancer Inst. 2016;108(12):djw151. doi: 10.1093/jnci/djw151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Tomic T, Dominguez-Lopez S, Barrios-Rodriguez R.. Non-aspirin non-steroidal anti-inflammatory drugs in prevention of colorectal cancer in people aged 40 or older: a systematic review and meta-analysis. Cancer Epidemiol. 2019;58:52–62. [DOI] [PubMed] [Google Scholar]
- 29. Smyth E, Grosser T, FitzGerald G.. Goodman & Gillman’s: The Pharmacological Basis of Therapeutics. New York: McGraw-Hill; 2011:937–958. [Google Scholar]
- 30. Patrono C, Garcia Rodriguez LA, Landolfi R, et al. Low-dose aspirin for the prevention of atherothrombosis. N Engl J Med. 2005;353(22):2373–2383. [DOI] [PubMed] [Google Scholar]
- 31. Thun MJ, Jacobs EJ, Patrono C.. The role of aspirin in cancer prevention. Nat Rev Clin Oncol. 2012;9(5):259–267. [DOI] [PubMed] [Google Scholar]
- 32. Gay LJ, Felding-Habermann B.. Contribution of platelets to tumour metastasis. Nat Rev Cancer. 2011;11(2):123–134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Labelle M, Begum S, Hynes RO.. Direct signaling between platelets and cancer cells induces an epithelial-mesenchymal-like transition and promotes metastasis. Cancer Cell. 2011;20(5):576–590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Campbell PT, Newton CC, Dehal AN, et al. Impact of body mass index on survival after colorectal cancer diagnosis: the Cancer Prevention Study-II Nutrition Cohort. J Clin Oncol. 2012;30(1):42–52. [DOI] [PubMed] [Google Scholar]
- 35. Campbell PT, Patel AV, Newton CC, et al. Associations of recreational physical activity and leisure time spent sitting with colorectal cancer survival. J Clin Oncol. 2013;31(7):876–885. [DOI] [PubMed] [Google Scholar]
- 36. Guinter MA, McCullough ML, Gapstur SM, et al. Associations of pre- and postdiagnosis diet quality with risk of mortality among men and women with colorectal cancer. J Clin Oncol. 2018;36(34):3404–3410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. McCullough ML, Gapstur SM, Shah R, et al. Association between red and processed meat intake and mortality among colorectal cancer survivors. J Clin Oncol. 2013;31(22):2773–2782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Yang B, Jacobs EJ, Gapstur SM, et al. Active smoking and mortality among colorectal cancer survivors: the Cancer Prevention Study II nutrition cohort. J Clin Oncol. 2015;33(8):885–893. [DOI] [PubMed] [Google Scholar]
- 39. Yang B, McCullough ML, Gapstur SM, et al. Calcium, vitamin D, dairy products, and mortality among colorectal cancer survivors: the Cancer Prevention Study-II Nutrition Cohort. J Clin Oncol. 2014;32(22):2335–2343. [DOI] [PubMed] [Google Scholar]
- 40. Rothwell PM, Fowkes FG, Belch JF, et al. Effect of daily aspirin on long-term risk of death due to cancer: analysis of individual patient data from randomised trials. Lancet. 2011;377(9759):31–41. [DOI] [PubMed] [Google Scholar]
- 41. Amitay EL, Carr PR, Jansen L, et al. Association of aspirin and nonsteroidal anti-inflammatory drugs with colorectal cancer risk by molecular subtypes. J Natl Cancer Inst. 2019;111(5):475–483. [DOI] [PubMed] [Google Scholar]
- 42. Nishihara R, Lochhead P, Kuchiba A, et al. Aspirin use and risk of colorectal cancer according to BRAF mutation status. JAMA. 2013;309(24):2563–2571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Frouws MA, Reimers MS, Swets M, et al. The influence of BRAF and KRAS mutation status on the association between aspirin use and survival after colon cancer diagnosis. PLoS One. 2017;12(1):e0170775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Coyle C, Cafferty FH, Langley RE.. Aspirin and colorectal cancer prevention and treatment: is it for everyone? Curr Colorectal Cancer Rep. 2016;12(1):27–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Coyle C, Cafferty FH, Rowley S, et al. ADD-ASPIRIN: a phase III, double-blind, placebo controlled, randomised trial assessing the effects of aspirin on disease recurrence and survival after primary therapy in common non-metastatic solid tumours. Contemp Clin Trials. 2016;51:56–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data underlying this article will be shared on reasonable request to the corresponding author.