Abstract
An outbreak of a novel coronavirus (COVID‐19 or 2019‐CoV) infection has posed significant threats to international health and the economy. Patients with COVID‐19 are at risk of cytokine storm, acute respiratory distress syndrome (ARDS), reduced blood oxygenation, mechanical ventilation, and a high death rate. Although recent studies have shown remdesivir and dexamethasone as treatment options, there is an urgent need to find a treatment to inhibit virus replication and to control the progression of the disease. Essential biometal zinc has generated a lot of excitement as one of the promising candidates to reduce the severity of COVID‐19 infection. Several published observations outlined in the review are the reasons why there is a global enthusiasm that zinc therapy could be a possible therapeutic option. However, the biggest challenge in realising the therapeutic value of zinc is lack of optimal treatment modalities such as dose, duration of zinc supplementation and the mode of delivery. In this review, we discuss the regulatory mechanism that hinges upon the bioavailability of zinc. Finally, we propose that intravenous zinc could circumvent the confounding factors affecting the bioavailability of zinc and allow zinc to achieve its therapeutic potential. If successful, due to advantages such as lack of toxicity, low cost and ease of availability, intravenous zinc could be rapidly implemented clinically.
Keywords: bioavailability, COVID‐19, immunity, intravenous, oral, SARS‐COV‐2, zinc
1. INTRODUCTION
The latest outbreak of coronavirus disease (COVID‐19) is caused by severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2). This enveloped virus with non‐segmented, single‐stranded, positive‐strand RNA virus belonging to a group of highly pathologic human coronaviruses (Hcov) like SARS and MERS infects the lower respiratory tract and causes severe pneumonia and acute respiratory distress syndrome (ARDS). 1 The symptoms of patients infected with SARS‐CoV‐2 range from minimal symptoms to severe respiratory failure with multiple organ failure. Its high transmissibility and increased morbidity have made COVID‐19 a serious public health threat and burden. The cornerstone of COVID‐19 management is patient isolation and supportive medical care where necessary, including pulmonary ventilation. There are limited treatment options or therapy for COVID‐19 infection, although recent studies have shown remdesivir and dexamethasone as treatment options. 2 , 3 , 4
Zinc is an essential biometal and the second most abundant trace metal in the human body. Zinc deficiency is associated with a range of pathological conditions, including retarded growth, and delayed wound healing and tissue repair. 5 , 6 Zinc is critical for cell proliferation, cell cycle regulation, differentiation and apoptosis. It plays an essential role for DNA and RNA synthesis, protein synthesis and a functional role in regulating protein–DNA interactions of zinc‐finger proteins, including many transcription factors and steroid hormone receptor. 7 , 8 Numerous studies have shown a correlation between zinc deficiency and critical illness. 9 , 10
Numerous studies have demonstrated zinc's ability to inhibit viral replication. These viruses include the common cold (often a type of coronavirus), 11 respiratory syncytial virus infections, 12 cytomegalovirus infections 13 and herpes labialis. 14 More importantly, zinc can prevent the growth of SARS‐coronavirus (SARS‐CoV) and equine arteritis virus (EAV) in cells grown in the lab. 15 In the later stages of infection, zinc could be beneficial by mitigating the impact of dysregulation of the immune system, inflammation and hypoxia‐induced oxidative stress. 16 , 17 , 18 , 19 , 20 , 21 , 22 Numerous studies have demonstrated that zinc modulates immune response, is anti‐inflammatory, improves recovery of patients with severe pneumonia and is protective against hypoxia‐induced ischaemia reperfusion injury. Therefore, this possibility that zinc can target multiple pathways driving the complex pathogenesis of COVID‐19 infection has led to the hypothesis that zinc supplementation would be beneficial in COVID‐19 patients. 16 , 17 , 18
However, the biggest challenge in realising zinc's therapeutic value is lack of optimal treatment modalities such as dose, duration of zinc supplementation, and the mode of delivery. In this paper, we review the current literature on zinc supplementation challenges and highlight the importance of using an effective zinc delivery method to achieve its potential as a therapeutic agent against COVID‐19 infection.
2. METHODS
We searched the Pubmed database for articles of relevance published in the English language up until 22 July 2020. We searched for the following keywords: “COVID‐19”, “SARS‐COV‐2”, “zinc”, “cytokine storm”, “immunity”, “oral” and “intravenous”. We performed a title and abstract review of articles focusing on the pathophysiology of COVID‐19, potential therapies for COVID‐19, reports on zinc and COVID‐19, articles on the mechanism of zinc absorption and articles on the supplementation of zinc for a diverse range of acute illnesses.
3. PATHOGENESIS OF COVID‐19 INFECTION
Spike protein (S) on the SARS‐COV‐2 virus enables viral entry into cells by binding to the angiotensin‐converting enzyme 2 (ACE 2) membrane protein, the host cell. 23 , 24 Infection of the respiratory epithelial cells triggers the macrophages and dendritic cells of the innate immune system. 1 , 18 Pathology of COVID‐19 involves an aggressive inflammatory response with the release of a large amount of pro‐inflammatory cytokines including interleukin 6 (IL‐6), interleukin 10 (IL‐10), granulocyte‐colony‐stimulating factor (G‐CSF), monocyte chemoattractant protein 1 (MCP1), macrophage inflammatory protein 1 alpha (MIP1α), and tumour necrosis factor‐alpha (TNFα) an event known as “cytokine storm”. 25 , 26 This exaggerated cytokine storm induces apoptosis of airway and alveolar epithelial cells, endothelial and epithelial cells of pulmonary microvasculature, causing alveolar oedema and subsequent hypoxia. 1 , 27 , 28 , 29 The prolonged and excessive cytokine/chemokine response leads to ARDS, multiple organ dysfunction and physiological deterioration. 1
Extrapulmonary organ injuries are also associated with COVID‐19 infection, as there is an increased incidence of cardiac injury in critically ill COVID‐19 patients. 2 , 24 , 30 Similarly, the incidence rate of acute kidney injury (AKI) is between 3% and 23% in COVID‐19 patients, which increases to 20–40% in patients admitted to ICU. 30 , 31 , 32 The mechanisms for these extrapulmonary complications are multifactorial. Proposed mechanisms include damage to cardiac and renal cells triggered by the virus's entry into the cells via ACE 2. The cytokine storm mediates secondary damage to the cells within the tissue in the later stages of infection. A recent study reported acute proximal tubular injury and peritubular erythrocyte aggregation, the ischaemic collapse of glomeruli with thrombi, endothelial damage, and inflammation in the kidneys of COVID‐19 patients. 33 In summary, COVID‐19 has complex pathogenesis, which is not just limited to an episode of respiratory infection but leads to dysfunction and failure of multiple organs. To counter a disease with such complex pathogenesis will require a therapeutic that can simultaneously target these multiple pathological pathways.
4. THERAPEUTIC PROPERTIES OF ZINC
Based on zinc's ability to inhibit viral replication, mitigate the impact of dysregulation of the immune system, protect hypoxia‐induced oxidative stress, and its anti‐inflammatory properties, it is hypothesised that zinc supplementation would be beneficial in patients with COVID‐19 infection. 16 , 17 , 18 , 19 , 20 , 21 , 22
4.1. Zinc and viral replication
Zinc and zinc ionophores (e.g. zinc‐pyrithione) have been known to inhibit virus entry, fusion, replication, protein translation and viral dissemination. Zinc has been shown to inhibit replication of many RNA viruses in vitro, including influenza virus, respiratory syncytial viruses, picornaviruses and some forms of coronavirus. 7 , 11 , 15 , 34
When administered intranasally, zinc gluconate shortened the common cold duration when administered within 24 hours of the onset of symptoms. 11 Inhibitory effect of zinc salts on viral replication was observed in vitro for several picornaviruses such as rhinoviruses, foot‐and‐mouth disease virus, coxsackievirus, and mengovirus. 15 Combination of Zn2+ ions and pyrithione at low concentrations (2 μM Zn2+ and 2 μM PT) inhibited the replication of SARS‐coronavirus (SARS‐CoV) and equine arteritis virus (EAV) in cell culture. 15 In coronaviruses, Zn2+ inhibits both the proteolytic processing of replicase polyproteins and the RNA‐dependent RNA polymerase (RdRp) activity. Although mechanisms of action of Zn are unknown, several possibilities exist. Firstly, DNA and RNA polymerases use divalent metal ions like Mg2+ as a co‐factor, and one possible mechanism is that Zn2+ displaces Mg2+ and subsequently inhibits RdRp activity. In support is the observation that various divalent metals ions (Cu2+, Ni2+, Mn2+) sustained the activity of poliovirus RdRp. 34 In contrast, Zn2+ is incapable of sustaining RdRp catalysed nucleotide incorporation. 34 Secondly, a Zn‐binding pocket has been identified in the Dengue virus and SARS‐coronavirus RdRp. Therefore, there is a possibility that Zn binding may induce a structural change in the conformation of RdRp, which no longer enables RdRp to catalyse nucleotide incorporation. Finally, adding high concentrations of Zn2+ to cells impairs viral polyprotein processing integral to virus replication. 35 A review concluded that zinc supplementation was the most effective systemic treatment for cutaneous viral warts compared to other available options. 7
Antiviral properties of zinc can be used firstly to enhance the body's endogenous antiviral response and systemic immunity in zinc‐deficient patients and, secondly, as a therapeutic agent to inhibit virus replication directly. Although zinc's possible therapeutic use has been shown in in vitro studies, reliable and convincing clinical data is lacking for it to be used as a therapeutic against COVID‐19.
4.2. Zinc and its role in immunity and inflammation
Zinc regulates innate and adaptive immunity by influencing the proliferation and maturation of immune cells, and zinc acts as a modulator of immune response and inflammation. 36 , 37 , 38 The function of T cell and balance between T cell subsets are susceptible to changes in zinc status. Interestingly, patients with severe COVID‐19 disease showed lymphopenia, particularly a reduction in peripheral blood T cells. 25 Impaired zinc homeostasis increases the risk of an excessive inflammatory response. 23 , 37
During the acute phase of an infection, a decrease in plasma zinc has been reported. Hence, decreased plasma zinc levels in the setting of critical illness may not be a true zinc deficiency, but an inflammatory response where hepatic metallothionein increases in response to cytokine release associated with inflammation sequester zinc in the liver. 9 , 39 , 40 Furthermore, the inflammatory response triggers increased excretion of zinc, further lowering plasma zinc levels. 9
Zinc supplementation can reverse the negative effects of zinc deficiency, including impaired immune cell development, compromised T‐cell‐mediated immune response, and decreased oxidative stress. 36 , 37 Zinc supplementation decreases oxidative stress and expression of inflammatory proteins in the elderly and reduces infection incidence. 41 , 42 When elderly subjects were supplemented with 45 mg elemental zinc for 6 months, it decreased plasma inflammatory cytokines by inhibiting nuclear factor‐kB (NF‐kB) a transcription factor critical in the expression of inflammatory cytokines. 41 , 42
Given that the COVID‐19 infection and associated extrapulmonary complications are driven mostly by pro‐inflammatory cytokines, some critically ill patients could become zinc deficient. Therefore, increasing plasma zinc concentrations in these patients will restore the zinc levels and help improve immunity and reduce inflammation in COVID‐19 patients. At the same time, it is unclear if increasing zinc plasma concentration in zinc‐replete patients would have a similar therapeutic effect.
4.3. Zinc and acute respiratory distress syndrome
COVID‐19 patients who develop acute respiratory distress syndrome (ARDS) may require mechanical ventilation as a supportive treatment. 43 , 44 Plasma zinc levels are reduced significantly more in patients who develop ARDS than healthy controls and non‐ARDS intensive care unit (ICU) controls. 45 Furthermore, although mechanical ventilation is necessary to support ARDS patients, it exacerbates lung injury through mechanical stress‐activated signalling pathways. 45 The study showed that zinc and its downstream target protein metallothionein (MT) play a critical role in enhancing tolerance against mechanical ventilation‐induced lung injury. 45 In vivo studies in mice have shown that zinc deficiency augments lung injury and decreases MT levels. 45
4.4. Zinc and pneumonia
Inadequate plasma zinc levels have been postulated as a risk factor for pneumonia, and several trials have shown that zinc can reduce the duration of pneumonia. A double‐blind placebo‐controlled trial by Brooks et al., in which 20 mg zinc was supplemented daily for 7 days showed a reduction in the recovery time and improvement in symptoms in patients with severe pneumonia. 46 Srinivasan et al. reported that zinc supplementation significantly decreased the fatalities from severe pneumonia, although it did not affect recovery time. 47 A study by Roscioli et al. investigated the impact of zinc and its regulatory effect on airway epithelium, postulating that zinc is critical for airway epithelium's functional integrity, and zinc deficiency promotes apoptosis of airway epithelial cells. 48 Overall, zinc could have a role in minimising airway damage and the inflammation associated with severe infection and mechanical ventilation, and zinc supplementation may alleviate airway inflammation and cellular damage.
4.5. Zinc and its protective role in hypoxia and ischaemia reperfusion injury
In 2019, the Nobel prize in medicine was awarded for the discovery of hypoxia‐inducible transcription factors (HIFs). Under hypoxic conditions, cells upregulate HIFs, which induces expression of over 200 diverse genes involved in cell survival; a key mechanism for cells and tissue to adapt to hypoxia. 49 , 50
Animal studies have shown that zinc protects against renal ischaemia and AKI. 21 , 22 O'Kane et al., using a preclinical sheep animal model, showed that intravenous zinc therapy reduced the ischaemic burden, substantiated by a statistically significant reduction in serum creatinine and urea, two markers of renal injury. In vitro experiments showed that zinc increased the survival of normal human tubular kidney HK‐2 cells and embryonic HEK293 cells subjected to cellular stress, including hypoxia, hydrogen peroxide‐induced oxidative stress, and nutrient starvation. 21 There is a strong basis that zinc could mitigate the effects of hypoxia and ischaemic injury on heart, kidney and liver due to COVID‐19.
Overall, this review provides a strong correlation between the pathogenesis of COVID‐19 infection and zinc's biochemical and biological properties. This leads to the hypothesis that zinc therapy could be beneficial both in early and later stages of COVID‐19 disease via its inhibitory effect on viral entry and replication, its ability to enhance immunity, mitigate inflammation, and its protective effects against hypoxia. However, the treatment parameters required to establish zinc's therapeutic value against COVID‐19 such as dose and delivery method (oral vs intravenous), and zinc supplementation duration are lacking.
5. CHALLENGES IN THE THERAPEUTIC USE OF ZINC AGAINST COVID‐19
To evaluate zinc's potential as a therapeutic agent against COVID‐19, it is necessary to increase the availability of free Zn2+ ions at the site of injury or infection. In this review, we have highlighted factors that may affect such an undertaking.
5.1. Effect of zinc deficiency on zinc uptake
A meta‐analysis of seven trials by Hemilä determined that zinc lozenges shortened the common cold duration by 33%. 51 Therapeutically, oral zinc has been shown to be beneficial against a large number of dermatological conditions. 52 Oral zinc supplementation has no effect on morbidity from falciparum malaria in children in rural west Africa, but it did reduce morbidity associated with diarrhoea. 53 In contrast, a meta‐analysis of five randomised controlled trials determined that oral zinc sulphate does not prevent or reduce the incidence, severity or pain intensity of chemotherapy‐induced oral mucositis in cancer patients. 54 Multiple trials using oral Zn supplementation have failed to demonstrate improvement against either diarrhoea, infection rates or related mortality 55 , 56 or ulcer resolution. 57 Currently lack of appropriately blinded randomised control trials does not favour zinc's use despite many studies showing its positive effect. More importantly, meta‐analysis of 80 randomised controlled trials with 205 401 eligible participants determined that the benefits of oral zinc supplementation is dependent on the prevalence of underlying zinc deficiency. 58 A placebo‐controlled trial by Chang et al. showed that there was no clinical benefit of zinc supplementation (40 mg zinc sulphate orally for 5 days) in terms of time to resolution of fever, tachypnoea and duration of hospitalisation in the management of acute lower respiratory tract infection among indigenous Australian children. 59 The study determined that at baseline, zinc levels were in the normal reference range (10–18 μmol/L), and zinc supplementation had no significant effect on serum zinc levels (median, 16.5 μmol/L zinc supplementation vs 14 μmol/L placebo; P = .31). 59 The authors suggest that zinc supplements may not be useful, and their effect may depend on the prevalence of underlying zinc deficiency in a particular population. 59 Therefore, reduced bioavailability following oral delivery may be the reason some trials of oral zinc supplementation have failed to demonstrate improvement against either diarrhoea, infection rates or mortality. 55 , 56 We postulate that it may be that the beneficial effect of zinc supplementation seen in previous trials 46 conducted in developing countries stems from the correction of zinc deficiency.
There are reports of the prevalence of zinc deficiency in the western and developed world. For example, in an Australian cohort, while 15% of all men had low serum Zn, none aged less than 40 had serum zinc below the cut‐off value for zinc deficiency (10.7 μmoL/L). 60 We know that Australians with an average age of 70.6 ± 7 yrs are zinc sufficient as their serum zinc levels are 12.7 ± 2.5 μmol/L. 61 A German study demonstrated an 18.7% prevalence of zinc deficiency in an older population ranging from 60 to 84 years. 62 Older adults are at greater risk of requiring hospitalisation or dying if diagnosed with COVID‐19, and oral zinc supplementation may increase serum zinc levels and prove beneficial. However, in light of published evidence that the majority of the population (>80%) in the older age group (over 60 yrs) are zinc sufficient in the developed world, any trial investigating oral zinc's therapeutic value against COVID‐19 may be negative due to reduced or no zinc uptake in the absence of underlying zinc deficiency.
5.2. Factors that affect zinc bioavailability
In this review, we discuss various regulatory mechanisms that influence zinc bioavailability. As shown in Figure 1, the regulatory mechanisms that affect zinc bioavailability are divided into four steps. Step 1: Dietary factors that affect zinc bioavailability; Step 2: Factors affecting intestinal zinc absorption; Step 3: Binding of zinc to blood albumin and red blood cells; Step 4: Cytosolic zinc buffering and muffling.
FIGURE 1.
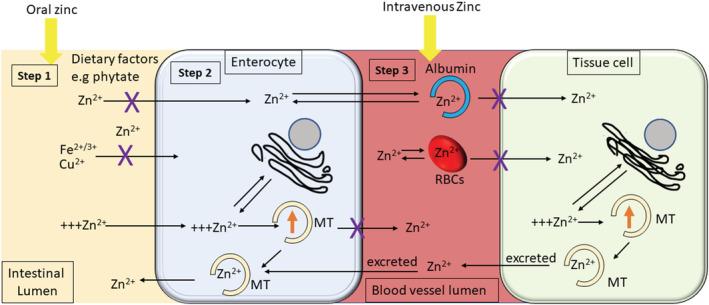
Regulatory mechanisms governing zinc absorption, transport and distributionZinc absorption, transport and distribution is regulated at four steps as described here. Step 1: The oral absorption of zinc at the intestinal lumen is influenced by several dietary factors, e.g. phytates, iron (Fe2+) and copper (Cu2+). Factors like phytate has a chelating effect on zinc impairing the bioavailability of oral zinc. Step 2: The absorbed Zn2+ ions are transported from the intestinal lumen into the enterocyte. Zn2+ ion uptake into the enterocyte is controlled by zinc‐dependent protein metallothionein (MT). The zinc‐dependent MT controls zinc absorption within the enterocyte. As zinc regulates MT gene expression, when body zinc is replete, or a high dose of zinc is taken orally, there is a stimulation of metallothionein synthesis. Metallothionein then sequesters excess zinc and ultimately excretes back into the intestinal lumen. This rate‐limiting step at the enterocytes is the first regulatory mechanism that regulates zinc uptake. Excess zinc may also be sequestered into cellular organelles such as mitochondria, endoplasmic reticulum and the Golgi apparatus. Step 3: Once the enterocytes release the absorbed Zn2+ ions into the blood vessel lumen, they bind to blood albumin. This regulatory mechanism, involving albumin, helps distribute Zn2+ ion around the body and regulates the availability of free Zn2+ ions for uptake by cells within the tissue. Furthermore, red blood cells (RBCs) are highly permeable to zinc, and reversible binding of zinc to RBC membranes plays a vital role in regulating the availability of free Zn2+ ions. Step 4: Finally, the regulatory mechanism that governs the availability of intracellular free Zn2+ ions within tissue cells is the cytosolic zinc buffering and muffling by various cytoplasmic proteins, including MT and cellular organelles. As seen in the figure, when zinc ions are delivered orally, it has to overcome all four regulatory mechanisms before they can reach cells within a tissue. When zinc is administered, intravenously directly into the vessel lumen, it bypasses the two intestinal regulatory steps 1 and 2. We reason that serum zinc and intracellular zinc within the cells affected by the virus or injury will be higher following administration of intravenous zinc than oral zinc
5.2.1. Step 1: Dietary factors that affect zinc bioavailability
Zinc absorption is affected by several factors such as luminal amino acids, ligands secreted by the pancreas, phosphates, phytates, iron, copper, calcium, endotoxins, and corticosteroids affect oral and systemic zinc absorption. 63 , 64 , 65 Phytates are the most significant dietary factor that impairs oral zinc bioavailability due to its chelating effect on zinc in the small intestine. Dietary phytate resulted in more than 80% of the variance in zinc absorption from oral supplements. 66 In summary, dietary zinc in the intestinal lumen is mainly complexed by food components influencing the actual available and absorbable zinc concentration. These factors make oral zinc an unpredictable delivery method.
5.2.2. Step 2: Factors affecting intestinal zinc absorption
If zinc was to be supplemented orally, then zinc absorption becomes a key factor. An elegant review by Maares and Haase provides an overview of the current knowledge on human intestinal zinc absorption, including the major cellular processes and nutritional aspects. 67 Briefly, zinc uptake takes place at the intestinal brush border membrane, where it is transported from the lumen into the enterocytes. It is subsequently released from the enterocytes into the portal blood, where it is predominantly bound to albumin, which distributes the metal in the body.
Zinc absorption is a saturable process, whereby zinc uptake by the enterocytes is a rate‐limiting step, and the body's zinc status regulates its net absorption. Such that dietary zinc absorption in zinc‐replete humans is typically in the range of 16–50%; in contrast, in zinc‐deficient humans, the dietary zinc absorption is as high as 92%. 67 The zinc‐dependent protein metallothionein (MT) controls zinc absorption within the enterocyte. As zinc regulates MT gene expression, 68 when body zinc is replete, there is a stimulation of metallothionein synthesis, which in turn inhibits zinc absorption. 64 Secondary to the regulation by MT, cellular organelles such as mitochondria, endoplasmic reticulum and the Golgi apparatus have been shown to sequester excess zinc. These zinc‐storing organelles help maintain a low steady‐state level of free Zn2+ ions and play a vital role in cellular zinc homeostasis. 69 Most currently registered zinc trials against COVID‐19 use oral delivery. The idea in some of those trials is that a high oral dose of zinc may overcome the rate‐limiting step within the enterocyte. However, high oral zinc causes a rapid and significant increase in intracellular zinc, which then upregulates expression of metallothioneins which ultimately decreases subsequent gut zinc absorption. 67 , 70
It is known that ionophores such as chloroquine (CQ), its analogue hydroxychloroquine (HCQ) and pyrithione increase zinc uptake. For example, in the presence of pyrithione, zinc crosses the plasma membrane in greater amounts compared with zinc alone, 71 and zinc uptake is increased nearly 10‐fold. 72 The rapid accumulation of intracellular zinc within the enterocyte in the presence of ionophores may lead to an immediate and significant upregulation of intestinal metallothioneins which will subsequently decrease gut zinc absorption and increases zinc excretion. 67 , 70 , 73 Therefore, it may be that ionophores may increase intracellular zinc within the enterocyte. Still, the robust activation of the MT regulatory mechanism may prevent the enterocyte from releasing the excess zinc into the blood. Previously, for children infected with Plasmodium falciparum when supplemented orally with 25 mg elemental zinc combined with chloroquine (10 mg/kg/d) for 3 days, their plasma zinc increased from a baseline of 8.54 ± 3.93 to 10.95 ± 3.63 μmol/L compared to chloroquine‐only treatment, which increased the plasma zinc from a baseline of 8.34 ± 3.25 to 10.16 ± 3.25 μmol/L. 74 Such a small and biologically ineffective 74 increase in the plasma zinc in the presence of ionophores such as CQ could be due to the MT‐based zinc regulatory mechanism present in the human intestine.
5.2.3. Step 3: Zinc binding to blood albumin and red blood cells
The second regulatory mechanism involves blood protein albumin. Various in vitro studies have shown that serum not only reduced intracellular uptake of zinc but has convincingly demonstrated that serum reduces the zinc‐mediated biological effects. 75 , 76 , 77 The reduced zinc uptake has been attributed to serum albumin. 77 Serum albumin effectively acts as an extracellular “zinc buffer” that controls the unwarranted increase in intracellular free Zn2+ ions within tissue‐specific cells, blood cells and the endothelial cells lining the blood vessels. Albumin acts as a defence mechanism against a sudden increase in blood zinc levels.
Furthermore, high albumin in the range of 600 μM in the human blood and at least six zinc‐binding sites per each albumin molecule specifies a large zinc buffering capacity in the human blood. 78 Additionally, the plasma/serum pharmacokinetics of zinc is complicated by the fact that human erythrocytes are highly permeable to zinc. It has been shown that red blood cells (RBCs) sequester more than 90% of the extracellular zinc and once zinc is inside the RBCs, the zinc efflux rate is less than 2% of that of the influx rate. 79 Such a regulatory mechanism within the blood will counter any attempt to increase free Zn2+ ions inside the tissue cells at the site of injury.
5.2.4. Step 4: Cytosolic zinc buffering and muffling
Finally, even if we increase plasma zinc to a level that can increase free intracellular Zn2+ ions within the cells at the site of injury, the third regulatory mechanism that poses a significant barrier is the intracellular zinc homeostasis mechanism termed “cytosolic zinc buffering and muffling”, details of which are reviewed elsewhere. 80 Briefly, under steady‐state conditions, the cell's repertoire of homeostatic zinc molecules includes cytosolic zinc‐binding proteins, transporters localised to cytoplasmic and organellar membranes, and sensors of cytoplasmic free zinc ion maintain the concentration of free Zn2+ ions in the picomolar range. Furthermore, when cells are challenged by a high concentration of extracellular zinc, the resultant influx of Zn2+ ions is dampened by muffling reactions ultimately restoring the cytosolic Zn2+ ion concentration to its original value by either shuttling Zn2+ ions into subcellular stores or by removing Zn2+ ions from the cell.
The biological significance of such a regulatory mechanism has been demonstrated using various biological readouts. 21 , 75 For example, zinc concentrations starting from 1 μmol/L up to 25 μmol/L did not induce gastrin gene expression in SW480 colon cancer cells. In contrast, 50 μmol/L zinc caused a sudden increase in the expression of gastrin by nearly 20‐fold. Further increase in zinc to 100 μmol/L had only a marginal effect on gastrin expression. 75 A similar dose‐dependent effect of zinc was observed in HK‐2 cells undergoing glucose oxygen deprivation such that no effect was observed until a level of 25 μmol/L zinc was reached. However, 50 μmol/L increased survival by nearly 200% and a further increase in zinc to 75 μmol/L led to diminished survival. 21 These results suggest that low doses of extracellular zinc (i.e. < 25 μmol/L) raise the level of intracellular free Zn2+ ions that can be quickly reversed by the cytosolic zinc buffering and muffling mechanisms. However, when extracellular zinc concentration increases to greater than 25 μmol/L, the cytosolic regulatory mechanisms are overwhelmed, leading to a biologically meaningful and non‐hazardous increase in intracellular free Zn2+ ions.
To have a beneficial effect against COVID‐19, the aim is to develop a zinc treatment modality that can increase the free intracellular Zn2+ ions at the infection or injury site. All the registered trials assessing the beneficial effect of zinc supplementation against COVID use oral delivery. Here we have reviewed the published knowledge regarding the robust regulatory mechanisms that control zinc bioavailability. We believe that if zinc was supplemented orally, it would have to overcome all the four regulatory mechanisms, as shown in Figure 1. In contrast, IV zinc administration will bypass Steps 1 and 2, resulting in higher bioavailability and may allow us to achieve optimal serum zinc levels. Further IV zinc delivery will enable precise control on the dosage and timing of administration. In summary, variability in the absorption of oral zinc and the published knowledge regarding the robust intestinal zinc regulatory mechanisms suggest that compared to oral zinc, IV zinc delivery has a better prospect at increasing plasma zinc to therapeutic levels required to achieve meaningful clinical outcomes. However, further studies are warranted.
6. VARIATION IN ZINC BIOAVAILABILITY
Zinc has been given intranasally in cases of influenza infection. 11 Interestingly, localised intranasal delivery of zinc led to anosmia or loss of smell. 81 No doubt when the threshold of cells of various origin to resist the toxic effect of zinc is in the low μmol/L range, the adverse anosmia manifestation may have been eventuated due to very high concentration of zinc, in the range of 30 mmol/L, used in those nasal sprays.
Zinc is primarily supplemented orally. Solomons et al. demonstrated that different formulations of oral zinc had different bioavailability; for example, consumption of 30 mg of zinc as Nutriset tablet (ZinCfant®) resulted in lower plasma zinc when compared to an aqueous solution of zinc sulphate which contained the same amount of elemental zinc. 82 When 30 mg of elemental zinc was given orally to humans, it resulted in an only 1.8‐fold peak increase in plasma zinc to 27 ± 12 μmol/L from a baseline of 15 ± 5.1 μmol/L over 4 hours. 82 Feillet‐Coudray et al. showed oral supplementation of 30 mg zinc per day over 6 months resulted in a marginal increase in the plasma zinc from 14 ± 1.7 μmol/L in the placebo group to 17 ± 3.5 μmol/L in the zinc group. 83
Using a preclinical large animal model of sheep, which closely resemble the pathophysiology observed in humans, a study by O'Kane et al. showed that plasma zinc levels needed to be raised by 7‐fold from a baseline concentration of 11 ± 0.4 μmol/L to 70 ± 5.8 μmol/L to protect the sheep against renal ischaemia reperfusion injury. 50 A study by Castro et al. determined that such an increase in plasma zinc in humans can only be achieved using intravenous zinc delivery. The study determined that when 37 mg of zinc was administered orally, it raised the peak serum zinc levels to only 25.5 ± 0.4 μmol/L from a baseline of 13 ± 1.5 μmol/L over 300 minutes. 68 In contrast, when a lower 20 mg dose of zinc was administered intravenously, it raised the serum zinc levels by nearly 6‐fold from a baseline of 13 ± 1.5 μmol/L to 101 ± 18.6 μmol/L over 100 minutes. 68
Based on pharmacokinetic modelling, a study by Cvijanovich et al. showed that 500 μg/kg/d intravenous zinc supplementation restored the plasma zinc levels within the normal range in zinc‐deficient children in intensive care without inducing a prolonged period of zinc accumulation and related toxicity. 9
In summary, various regulatory mechanisms affect the uptake of zinc as shown in Figure 1; different formulations of zinc, different route of administration and zinc status of the trial cohorts, can all influence zinc bioavailability.
7. THERAPEUTIC USE OF ZINC IN COVID‐19
Hydroxychloroquine (HCQ) has been shown to act as a zinc ionophore, increasing zinc uptake and inducing apoptosis in malignant cells. 18 , 84 Therefore, increased zinc uptake was one of the possible mechanisms based on which hydroxychloroquine was theorised to be a potential treatment of COVID‐19. However, a recent trial by Boulware et al. showed that hydroxychloroquine was ineffective for prevention of COVID‐19. 84 Further, it may be that for HCQ to be effective, it needed to be administered in combination with zinc. However, the trial determined that zinc, in combination with HCQ, was ineffective as there was no evidence that HCQ, in combination with zinc, reduced the incidence of COVID‐19 after a high‐risk exposure. 84 However, the study cannot be considered conclusive as it was limited by the small sample size of the zinc cohort exacerbated by lack of details regarding zinc formulation, dose and treatment duration. Further data showing whether HCQ, in combination with zinc, did indeed increase plasma zinc concentration is also lacking. There is an urgent need for well‐conducted clinical trials to investigate the effects of zinc as a therapeutic agent in patients hospitalised with COVID‐19 infection.
8. SAFETY OF INTRAVENOUS ZINC IN HUMANS
Zinc toxicity is characterised non objectively by vomiting, diarrhoea, fever, lethargy, muscle pain and stiffness 85 , 86 and objectively by anaemia, copper deficiency 9 and kidney injury. 21 The lethal dose is unknown for intravenous zinc in humans. 85 The upper safe daily oral intake limit for elemental zinc is 40 mg/d as defined by the National Institute of Health (USA) and a lower 25 mg/d as defined by the European Food Safety Authority. However, no such recommendation exists for intravenous zinc. Previously in the treatment of burns, when elemental zinc was administered intravenously at doses ranging from 26.4 to 37.5 mg/d for eight consecutive days, it did not produce any side effects in humans. 87 , 88 , 89 Moreover, zinc at doses ranging from 5 to 22 mg/d has been administered routinely as a parenteral nutrition component without any reported side effects. 90
Furthermore, a published phase I clinical trial in critically ill children with suspected zinc deficiency administered zinc intravenously at an elemental dose of 0.75 mg/kg/d for 7 days without producing any adverse effects. 9 Therefore, safety concerns for a clinical trial using intravenous zinc within the dose ranges mentioned above can be assuaged. 9 , 87 , 88 , 89
An analysis by Hambidge et al. indicated that the maximal absorption (A max) for zinc via oral delivery is only ≈7 mg zinc/d. 66 This low A max is due to the molecular complexities of oral zinc absorption into and across the enterocyte and basolateral membrane, as discussed in the current review. In summary, we suggest that intravenous zinc may overcome low A max of oral zinc supplementation and will have a better prospect of increasing the levels of free Zn2+ ions inside the tissue cells at the injury site.
9. RANDOMISED, PLACEBO‐CONTROLLED TRIAL OF HIGH DOSE INTRAVENOUS ZINC IN SARS‐COV‐2‐POSITIVE PATIENTS
Cheap, safe and easily administered interventions to improve outcomes associated with COVID‐19 infection remain an important goal. Zinc and its salts inhibit viral infections in clinical and experimental settings, 11 , 12 , 13 , 14 , 15 , 34 , 35 , 91 , 92 , 93 including replication of SARS‐coronavirus (SARS‐CoV). 15 Thus, zinc supplementation might be beneficial in patients with COVID‐19. 16 , 17 , 18 , 19 , 20 , 21 , 22 Clinical trials have been registered to test the efficacy of zinc against COVID, mostly with oral zinc supplementation. 94 However, the bioavailability of enterally administered zinc is low. 20 Thus, logically, intravenous zinc should be the preferred mode of administration. 20 Unfortunately, no studies have assessed the feasibility, safety and impact on serum zinc levels of high‐dose intravenous zinc (HDIVZn). We have initiated a phase IIa pilot double‐blind randomised controlled trial (RCT) to determine the feasibility, safety and biological efficacy of HDIVZn in subjects with COVID‐19 (Trial registration No. ACTRN12620000454976). A detailed clinical trial protocol has been published previously. 95 Briefly, we will randomise consenting COVID‐19 confirmed hospitalised adults with oxygen saturation (SpO2) of 94% or less while on ambient air to either daily HDIVZn or saline placebo. Colourless pharmaceutical grade zinc chloride (ZnCl2) stock solution will be diluted in 250 mL of normal saline and infused via peripheral intravenous access over 3 hours at a dose of 0.5 mg/kg/d (elemental zinc concentration 0.24 mg/kg/d) for a maximum of 7 days, or until hospital discharge or death.
10. CONCLUSION
No doubt, there is much published data on the potential use of zinc as a therapeutic against COVID‐19. 16 , 17 For zinc to have its intended therapeutic effect, one of the requisites is to increase the intracellular concentration of free zinc in the cells and tissue impacted by COVID pathology. To date, there is no definitive knowledge regarding the amount of zinc that may be required to have a therapeutic effect on COVID‐19 patients. Factors such as the presence of pre‐existent zinc deficiency, the variance in zinc bioavailability caused by different formulation, dose and delivery methods, especially the issues affecting oral zinc absorption, may all influence the clinical outcomes.
Recently published studies using the large animal model have shown that intravenous zinc is effective at reducing ischaemia reperfusion injury (IRI) and permanent organ dysfunction. 21 The scientific basis behind the effectiveness of intravenous zinc is its ability to deliver a pharmacologic zinc dose and eliminate the factors that limit the bioavailability of zinc when given orally. Further, the fact that the drug dose correction factor (K m) for humans (K m = 37) and sheep (K m = 36) is almost identical suggests a direct carryover of human equivalent elemental zinc dosage from sheep to humans. 96 Concerns regarding the safety of intravenous zinc can be assuaged based on published reports where humans were treated with higher doses of zinc 9 , 87 , 88 , 89 than would eventuate from the sheep study.
Based on the scientific knowledge discussed in this review, we have initiated a clinical trial that uses intravenous zinc supplementation (Trial registration No. ACTRN12620000454976). We believe that intravenous zinc could circumvent the confounding factors affecting the bioavailability of zinc and allow zinc to achieve its therapeutic potential. A positive outcome of our trial will have an enormous impact on the health outcomes of patients with COVID‐19 infection and at high risk of developing ARDS. If successful, due to advantages such as lack of toxicity, low cost and ease of availability, intravenous zinc could be rapidly implemented clinically.
COMPETING INTERESTS
There are no conflicting interests to declare.
CONTRIBUTORS
V.C. performed the literature review, collected and analysed the data and wrote the paper. H.E. contributed to the collection and analysis of the date and wrote the paper. M.P. performed data analysis, wrote and reviewed the paper. R.B. and D.J. helped conceive the analysis, contributed to analysis tools, provided expert consultation and provided review of the paper. D.B. helped conceive and designed the analysis, reviewed the paper, and provided expert consultation. J.I. helped conceive and designed the analysis, performed the analysis, wrote and reviewed the paper, and provided expert consultation. O.P. helped conceive and designed the analysis, performed the analysis, and wrote and reviewed the paper.
Chinni V, El‐Khoury J, Perera M, et al. Zinc supplementation as an adjunct therapy for COVID‐19: Challenges and opportunities. Br J Clin Pharmacol. 2021;87(10):3737–3746. 10.1111/bcp.14826
REFERENCES
- 1. Ye Q, Wang B, Mao J. The pathogenesis and treatment of the “Cytokine Storm” in COVID‐19. J Infect. 2020;80(6):607‐613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Feng G, Zheng KI, Yan QQ, et al. COVID‐19 and liver dysfunction: current insights and emergent therapeutic strategies. J Clin Transl Hepatol. 2020;8(1):18‐24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. McMahon JH, Udy A, Peleg AY. Remdesivir for the treatment of COVID‐19 – preliminary report. N Engl J Med. 2020;383(10):992‐993. [DOI] [PubMed] [Google Scholar]
- 4. The RECOVERY Collaborative Group , Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with COVID‐19 – preliminary report. N Engl J Med. 2021;384(8):693‐704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Maret W. Zinc biochemistry: from a single zinc enzyme to a key element of life. Adv Nutr (Bethesda, Md). 2013;4:82‐91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Fukada T, Yamasaki S, Nishida K, Murakami M, Hirano T. Zinc homeostasis and signaling in health and diseases: zinc signaling. J Biol Inorg Chem. 2011;16(7):1123‐1134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Read SA, Obeid S, Ahlenstiel C, Ahlenstiel G. The role of zinc in antiviral immunity. Adv Nutr (Bethesda, Md). 2019;10:696‐710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Vale SH, Leite LD, Alves CX, et al. Zinc pharmacokinetic parameters in the determination of body zinc status in children. Eur J Clin Nutr. 2014;68(2):203‐208. [DOI] [PubMed] [Google Scholar]
- 9. Cvijanovich NZ, King JC, Flori HR, Gildengorin G, Vinks AA, Wong HR. Safety and dose escalation study of intravenous zinc supplementation in pediatric critical illness. JPEN J Parenter Enteral Nutr. 2016;40(6):860‐868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Cvijanovich NZ, King JC, Flori HR, Gildengorin G, Wong HR. Zinc homeostasis in pediatric critical illness. Pediatr Crit Care Med. 2009;10(1):29‐34. [DOI] [PubMed] [Google Scholar]
- 11. Hulisz D. Efficacy of zinc against common cold viruses: an overview. J Am Pharm Assoc. 2004;44(5):594‐603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Suara RO, Crowe JE Jr. Effect of zinc salts on respiratory syncytial virus replication. Antimicrob Agents Chemother. 2004;48(3):783‐790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Li D, Wen LZ, Yu H. Observation on clinical efficacy of combined therapy of zinc supplement and jinye baidu granule in treating human cytomegalovirus infection. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2005;25(5):449‐451. [PubMed] [Google Scholar]
- 14. Femiano F, Gombos F, Scully C. Recurrent herpes labialis: a pilot study of the efficacy of zinc therapy. J Oral Pathol Med. 2005;34(7):423‐425. [DOI] [PubMed] [Google Scholar]
- 15. te Velthuis AJ, van den Worm SH, Sims AC, Baric RS, Snijder EJ, van Hemert MJ. Zn(2+) inhibits coronavirus and arterivirus RNA polymerase activity in vitro and zinc ionophores block the replication of these viruses in cell culture. PLoS Pathog. 2010;6(11):e1001176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Wessels I, Rolles B, Rink L. The potential impact of zinc supplementation on COVID‐19 pathogenesis. Front Immunol. 2020;11:1712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Skalny AV, Rink L, Ajsuvakova OP, et al. Zinc and respiratory tract infections: perspectives for COVID19 (Review). Int J Mol Med. 2020;46(1):17‐26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Rahman MT, Idid SZ. Can Zn be a critical element in COVID‐19 treatment? Biol Trace Elem Res. 2021;199:550‐558. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Jayawardena R, Sooriyaarachchi P, Chourdakis M, Jeewandara C, Ranasinghe P. Enhancing immunity in viral infections, with special emphasis on COVID‐19: a review. Diabetes Metab Syndr. 2020;14(4):367‐382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Ischia J, Bolton DM, Patel O. Why is it worth testing the ability of zinc to protect against ischaemia reperfusion injury for human application. Metallomics. 2019;11(8):1330‐1343. [DOI] [PubMed] [Google Scholar]
- 21. O'Kane D, Gibson L, May CN, et al. Zinc preconditioning protects against renal ischaemia reperfusion injury in a preclinical sheep large animal model. Biometals. 2018;31(5):821‐834. [DOI] [PubMed] [Google Scholar]
- 22. Rao K, Sethi K, Ischia J, et al. Protective effect of zinc preconditioning against renal ischemia reperfusion injury is dose dependent. PLoS ONE. 2017;12(7):e0180028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Wang C, Horby PW, Hayden FG, Gao GF. A novel coronavirus outbreak of global health concern. Lancet. 2020;395(10223):470‐473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Zhu H, Rhee JW, Cheng P, et al. Cardiovascular complications in patients with COVID‐19: consequences of viral toxicities and host immune response. Curr Cardiol Rep. 2020;22(5):32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Yuki K, Fujiogi M, Koutsogiannaki S. COVID‐19 pathophysiology: a review. Clin Immunol. 2020;215:108427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, transmission, diagnosis, and treatment of coronavirus disease 2019 (COVID‐19): a review. JAMA. 2020;324(8):782‐793. [DOI] [PubMed] [Google Scholar]
- 27. Herold S, Steinmueller M, von Wulffen W, et al. Lung epithelial apoptosis in influenza virus pneumonia: the role of macrophage‐expressed TNF‐related apoptosis‐inducing ligand. J Exp Med. 2008;205(13):3065‐3077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Hogner K, Wolff T, Pleschka S, et al. Macrophage‐expressed IFN‐beta contributes to apoptotic alveolar epithelial cell injury in severe influenza virus pneumonia. PLoS Pathog. 2013;9(2):e1003188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Rodrigue‐Gervais IG, Labbe K, Dagenais M, et al. Cellular inhibitor of apoptosis protein cIAP2 protects against pulmonary tissue necrosis during influenza virus infection to promote host survival. Cell Host Microbe. 2014;15(1):23‐35. [DOI] [PubMed] [Google Scholar]
- 30. Batlle D, Soler MJ, Sparks MA, et al. Acute kidney injury in COVID‐19: emerging evidence of a distinct pathophysiology. J Am Soc Nephrol. 2020;31(7):1380‐1383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Khouchlaa A, Bouyahya A. COVID‐19 nephropathy: probable mechanisms of kidney failure. J Nephropathol. 2020;9(4):e35. [Google Scholar]
- 32. Ronco C, Reis T, Husain‐Syed F. Management of acute kidney injury in patients with COVID‐19. Lancet Respir Med. 2020;8(7):738‐742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Su H, Yang M, Wan C, et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID‐19 in China. Kidney Int. 2020;98(1):219‐227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Arnold JJ, Ghosh SK, Cameron CE. Poliovirus RNA‐dependent RNA polymerase (3Dpol): divalent cation modulation of primer, template, and nucleotide selection. J Biol Chem. 1999;274(52):37060‐37069. [DOI] [PubMed] [Google Scholar]
- 35. Butterworth BE, Korant BD. Characterization of the large picornaviral polypeptides produced in the presence of zinc ion. J Virol. 1974;14(2):282‐291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Maares M, Haase H. Zinc and immunity: an essential interrelation. Arch Biochem Biophys. 2016;611:58‐65. [DOI] [PubMed] [Google Scholar]
- 37. Bonaventura P, Benedetti G, Albarede F, Miossec P. Zinc and its role in immunity and inflammation. Autoimmun Rev. 2015;14(4):277‐285. [DOI] [PubMed] [Google Scholar]
- 38. Vasto S, Mocchegiani E, Candore G, et al. Inflammation, genes and zinc in ageing and age‐related diseases. Biogerontology. 2006;7(5‐6):315‐327. [DOI] [PubMed] [Google Scholar]
- 39. Liuzzi JP, Lichten LA, Rivera S, et al. Interleukin‐6 regulates the zinc transporter Zip14 in liver and contributes to the hypozincemia of the acute‐phase response. Proc Natl Acad Sci U S A. 2005;102(19):6843‐6848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. King JC. Zinc: an essential but elusive nutrient. Am J Clin Nutr. 2011;94(2):679S‐684S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Prasad AS. Zinc: role in immunity, oxidative stress and chronic inflammation. Curr Opin Clin Nutr Metab Care. 2009;12(6):646‐652. [DOI] [PubMed] [Google Scholar]
- 42. Prasad AS. Zinc is an antioxidant and anti‐inflammatory agent: its role in human health. Front Nutr. 2014;1:14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Meng L, Qiu H, Wan L, et al. Intubation and ventilation amid the COVID‐19 outbreak: Wuhan's experience. Anesthesiology. 2020;132(6):1317‐1332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Mohlenkamp S, Thiele H. Ventilation of COVID‐19 patients in intensive care units. Herz. 2020;45(4):329‐331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Boudreault F, Pinilla‐Vera M, Englert JA, et al. Zinc deficiency primes the lung for ventilator‐induced injury. JCI Insight. 2017;2(11):e86507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Brooks WA, Yunus M, Santosham M, et al. Zinc for severe pneumonia in very young children: double‐blind placebo‐controlled trial. Lancet. 2004;363(9422):1683‐1688. [DOI] [PubMed] [Google Scholar]
- 47. Srinivasan MG, Ndeezi G, Mboijana CK, et al. Zinc adjunct therapy reduces case fatality in severe childhood pneumonia: a randomized double blind placebo‐controlled trial. BMC Med. 2012;10(1):14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Roscioli E, Hamon R, Lester S, Murgia C, Grant J, Zalewski P. Zinc‐rich inhibitor of apoptosis proteins (IAPs) as regulatory factors in the epithelium of normal and inflamed airways. Biometals. 2013;26(2):205‐227. [DOI] [PubMed] [Google Scholar]
- 49. Semenza GL. Hypoxia‐inducible factors in physiology and medicine. Cell. 2012;148(3):399‐408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. O'Kane D, Baldwin GS, Bolton DM, Ischia JJ, Patel O. Preconditioning against renal ischaemia reperfusion injury: the failure to translate to the clinic. J Nephrol. 2019;32(4):539‐547. [DOI] [PubMed] [Google Scholar]
- 51. Hemilä H. Zinc lozenges and the common cold: a meta‐analysis comparing zinc acetate and zinc gluconate, and the role of zinc dosage. JRSM Open. 2017;8:2054270417694291. 10.1177/2054270417694291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Gupta M, Mahajan VK, Mehta KS, Chauhan PS. Zinc therapy in dermatology: a review. Dermatol Res Pract. 2014;2014:709152. 10.1155/2014/709152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Muller O, Becher H, van Zweeden AB, et al. Effect of zinc supplementation on malaria and other causes of morbidity in west African children: randomised double blind placebo controlled trial. BMJ. 2001;322(7302):1567. 10.1136/bmj.322.7302.1567 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Tian X, Liu XL, Pi YP, Chen H, Chen WQ. Oral zinc sulfate for prevention and treatment of chemotherapy‐induced oral mucositis: a meta‐analysis of five randomized controlled trials. Front Oncol. 2018;8:484. 10.3389/fonc.2018.00484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Heyland DK, Jones N, Cvijanovich NZ, Wong H. Zinc supplementation in critically ill patients: a key pharmaconutrient? JPEN J Parenter Enteral Nutr. 2008;32(5):509‐519. [DOI] [PubMed] [Google Scholar]
- 56. Lazzerini M, Wanzira H. Oral zinc for treating diarrhoea in children. Cochrane Database Syst Rev. 2016;12:CD005436. 10.1002/14651858.CD005436.pub5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Wilkinson EA, Hawke CI. Does oral zinc aid the healing of chronic leg ulcers? A systematic literature review. Arch Dermatol. 1998;134(12):1556‐1560. [DOI] [PubMed] [Google Scholar]
- 58. Mayo‐Wilson E, Junior JA, Imdad A, et al. Zinc supplementation for preventing mortality, morbidity, and growth failure in children aged 6 months to 12 years of age. Cochrane Database Syst Rev. 2014;CD009384. 10.1002/14651858.CD009384.pub2 [DOI] [PubMed] [Google Scholar]
- 59. Chang AB, Torzillo PJ, Boyce NC, et al. Zinc and vitamin A supplementation in Indigenous Australian children hospitalised with lower respiratory tract infection: a randomised controlled trial. Med J Aust. 2006;184(3):107‐112. [DOI] [PubMed] [Google Scholar]
- 60. Beckett JM, Ball MJ. Zinc status of northern Tasmanian adults. J Nutr Sci. 2015;4:e15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Rembach A, Hare DJ, Doecke JD, et al. Decreased serum zinc is an effect of ageing and not Alzheimer's disease. Metallomics. 2014;6(7):1216‐1219. [DOI] [PubMed] [Google Scholar]
- 62. Jung A, Spira D, Steinhagen‐Thiessen E, Demuth I, Norman K. Zinc deficiency is associated with depressive symptoms—results from the Berlin Aging Study II. J Gerontol A Biol Sci Med Sci. 2017;72:1149‐1154. [DOI] [PubMed] [Google Scholar]
- 63. Lonnerdal B. Dietary factors influencing zinc absorption. J Nutr. 2000;130(5):1378S‐1383S. [DOI] [PubMed] [Google Scholar]
- 64. Jeejeebhoy K. Zinc: an essential trace element for parenteral nutrition. Gastroenterology. 2009;137(5):S7‐S12. [DOI] [PubMed] [Google Scholar]
- 65. Kondaiah P, Yaduvanshi PS, Sharp PA, Pullakhandam R. Iron and zinc homeostasis and interactions: does enteric zinc excretion cross‐talk with intestinal iron absorption? Nutrients. 2019;11(8):1885. 10.3390/nu11081885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Hambidge KM, Miller LV, Westcott JE, Sheng X, Krebs NF. Zinc bioavailability and homeostasis. Am J Clin Nutr. 2010;91(5):1478S‐1483S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Maares M, Haase H. A guide to human zinc absorption: general overview and recent advances of in vitro intestinal models. Nutrients. 2020;12(3):762. 10.3390/nu12030762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Castro AV, Mendonca BB, Bloise W, Shuhama T, Brandao‐Neto J. Effect of zinc administration on thyrotropin releasing hormone‐stimulated prolactinemia in healthy men. Biometals. 1999;12(4):347‐352. [DOI] [PubMed] [Google Scholar]
- 69. Lu Q, Haragopal H, Slepchenko KG, Stork C, Li YV. Intracellular zinc distribution in mitochondria, ER and the Golgi apparatus. Int J Physiol Pathophysiol Pharmacol. 2016;8(1):35‐43. [PMC free article] [PubMed] [Google Scholar]
- 70. Hempe JM, Carlson JM, Cousins RJ. Intestinal metallothionein gene expression and zinc absorption in rats are zinc‐responsive but refractory to dexamethasone and interleukin 1 alpha. J Nutr. 1991;121(9):1389‐1396. [DOI] [PubMed] [Google Scholar]
- 71. Chowanadisai W, Graham DM, Keen CL, Rucker RB, Messerli MA. Neurulation and neurite extension require the zinc transporter ZIP12 (slc39a12). Proc Natl Acad Sci U S A. 2013;110(24):9903‐9908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Law W, Kelland EE, Sharp P, Toms NJ. Characterisation of zinc uptake into rat cultured cerebrocortical oligodendrocyte progenitor cells. Neurosci Lett. 2003;352(2):113‐116. [DOI] [PubMed] [Google Scholar]
- 73. Lamore SD, Wondrak GT. Zinc pyrithione impairs zinc homeostasis and upregulates stress response gene expression in reconstructed human epidermis. Biometals. 2011;24(5):875‐890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Zinc Against Plasmodium Study Group . Effect of zinc on the treatment of Plasmodium falciparum malaria in children: a randomized controlled trial. Am J Clin Nutr. 2002;76(4):805‐812. [DOI] [PubMed] [Google Scholar]
- 75. Marshall KM, Laval M, Estacio O, et al. Activation by zinc of the human gastrin gene promoter in colon cancer cells in vitro and in vivo. Metallomics. 2015;7(10):1390‐1398. [DOI] [PubMed] [Google Scholar]
- 76. Haase H, Hebel S, Engelhardt G, Rink L. The biochemical effects of extracellular Zn(2+) and other metal ions are severely affected by their speciation in cell culture media. Metallomics. 2015;7(1):102‐111. [DOI] [PubMed] [Google Scholar]
- 77. Blindauer CA, Harvey I, Bunyan KE, et al. Structure, properties, and engineering of the major zinc binding site on human albumin. J Biol Chem. 2009;284(34):23116‐23124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Handing KB, Shabalin IG, Kassaar O, et al. Circulatory zinc transport is controlled by distinct interdomain sites on mammalian albumins. Chem Sci. 2016;7(11):6635‐6648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Kruckeberg WC, Brewer GJ. The mechanism and control of human erythrocyte zinc uptake. Med Biol. 1978;56(1):5‐10. [PubMed] [Google Scholar]
- 80. Colvin RA, Holmes WR, Fontaine CP, Maret W. Cytosolic zinc buffering and muffling: their role in intracellular zinc homeostasis. Metallomics. 2010;2(5):306‐317. [DOI] [PubMed] [Google Scholar]
- 81. Davidson TM, Smith WM. The Bradford Hill criteria and zinc‐induced anosmia: a causality analysis. Arch Otolaryngol Head Neck Surg. 2010;136(7):673‐676. [DOI] [PubMed] [Google Scholar]
- 82. Solomons NW, Romero‐Abal ME, Weiss G, Michalke B, Schumann K. Bioavailability of zinc from NutriSet zinc tablets compared with aqueous zinc sulfate. Eur J Clin Nutr. 2011;65(1):125‐131. [DOI] [PubMed] [Google Scholar]
- 83. Feillet‐Coudray C, Meunier N, Rambeau M, et al. Long‐term moderate zinc supplementation increases exchangeable zinc pool masses in late‐middle‐aged men: the Zenith Study. Am J Clin Nutr. 2005;82(1):103‐110. [DOI] [PubMed] [Google Scholar]
- 84. Boulware DR, Pullen MF, Bangdiwala AS, et al. A randomized trial of hydroxychloroquine as postexposure prophylaxis for COVID‐19. N Engl J Med. 2020;383(6):517‐525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Brocks A, Reid H, Glazer G. Acute intravenous zinc poisoning. Br Med J. 1977;1(6073):1390‐1391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Gallery ED, Blomfield J, Dixon SR. Acute zinc toxicity in haemodialysis. Br Med J. 1972;4(5836):331‐333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Berger MM, Baines M, Raffoul W, et al. Trace element supplementation after major burns modulates antioxidant status and clinical course by way of increased tissue trace element concentrations. Am J Clin Nutr. 2007;85(5):1293‐1300. [DOI] [PubMed] [Google Scholar]
- 88. Berger MM, Binnert C, Chiolero RL, et al. Trace element supplementation after major burns increases burned skin trace element concentrations and modulates local protein metabolism but not whole‐body substrate metabolism. Am J Clin Nutr. 2007;85(5):1301‐1306. [DOI] [PubMed] [Google Scholar]
- 89. Berger MM, Spertini F, Shenkin A, et al. Trace element supplementation modulates pulmonary infection rates after major burns: a double‐blind, placebo‐controlled trial. Am J Clin Nutr. 1998;68(2):365‐371. [DOI] [PubMed] [Google Scholar]
- 90. Stehle P, Stoffel‐Wagner B, Kuhn KS. Parenteral trace element provision: recent clinical research and practical conclusions. Eur J Clin Nutr. 2016;70(8):886‐893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Korant BD, Kauer JC, Butterworth BE. Zinc ions inhibit replication of rhinoviruses. Nature. 1974;248(5449):588‐590. [DOI] [PubMed] [Google Scholar]
- 92. Lanke K, Krenn BM, Melchers WJ, Seipelt J, van Kuppeveld FJ. PDTC inhibits picornavirus polyprotein processing and RNA replication by transporting zinc ions into cells. J Gen Virol. 2007;88(4):1206‐1217. [DOI] [PubMed] [Google Scholar]
- 93. Si X, McManus BM, Zhang J, et al. Pyrrolidine dithiocarbamate reduces coxsackievirus B3 replication through inhibition of the ubiquitin‐proteasome pathway. J Virol. 2005;79(13):8014‐8023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Pal A, Squitti R, Picozza M, et al. Zinc and COVID‐19: basis of current clinical trials. Biol Trace Elem Res. 2020;1‐11. 10.1007/s12011-020-02437-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Perera M, El‐Khoury J, Vidyasagar C, et al. A randomised controlled trial for high‐dose intravenous zinc as adjunctive therapy in SARS‐CoV‐2 (COVID‐19) positive critically ill patients: trial protocol. BMJ Open. 2020;10(12):e040580. 10.1136/bmjopen-2020-040580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Nair AB, Jacob S. A simple practice guide for dose conversion between animals and human. J Basic Clin Pharm. 2016;7(2):27‐31. [DOI] [PMC free article] [PubMed] [Google Scholar]


