Abstract
Importance
Assessment of the causative association between the COVID‐19 and cause of death has been hampered by limited availability of systematically performed autopsies. We aimed to present autopsy‐confirmed causes of death in patients who died with COVID‐19 and to assess the association between thrombosis and diffuse alveolar damage consistent with COVID‐19 (DAD).
Methods
Consecutive forensic (n = 60) and clinical (n = 42) autopsies with positive post‐mortem SARS‐CoV‐2 PCR in lungs (age 73 ± 14 years, 50% men) were included. The cause of death analysis was based on a review of medical records and histological reports. Thrombotic phenomena in lungs were defined as pulmonary thromboembolism (PE), thrombosis in pulmonary artery branches or microangiopathy in capillary vessels.
Results
COVID‐19 caused or contributed to death in 71% of clinical and 83% of forensic autopsies, in whom significant DAD was observed. Of the patients with COVID‐19 as the primary cause of death, only 19% had no thrombotic phenomena in the lungs, as opposed to 38% amongst those with COVID‐19 as a contributing cause of death and 54% amongst patients whose death was not related to COVID‐19 (p = 0.002). PE was observed in 5 patients. Two patients fulfilled the criteria for lymphocyte myocarditis.
Conclusions
Vast majority of all PCR‐positive fatalities, including out‐of‐hospital deaths, during the SARS‐CoV‐2 pandemic were related to DAD caused by COVID‐19. Pulmonary artery thrombosis and microangiopathy in pulmonary tissue were common and associated with the presence of DAD, whilst venous PE was rarely observed. Histology‐confirmed lymphocyte myocarditis was a rare finding.
Keywords: COVID‐19, forensic autopsy, pathology

Introduction
The SARS‐CoV‐2 infection outbreak that led to the ongoing pandemic has been associated with significant morbidity and mortality. However, assessing the causative association between SARS‐CoV‐2 infection and cause of death has been problematic and due in part to limited availability of systematically collected unselected post‐mortem data, which has been reported as small series [1]. In particular, deaths occurring out of hospital without confirmed SARS‐CoV‐2 infection prior to death may have been missed from reporting in the COVID‐19 context [2] and contributed to excess mortality during the pandemic [3, 4. Thromboembolic events and pulmonary thromboembolism (PE) in particular have been proposed as important and potentially lethal complications associated with COVID‐19 [5] that can lead to unaccounted COVID‐19‐related deaths in out‐of‐hospital settings. This, however, has not been supported by systematic pathology data since, to the best of our knowledge, studies that would specifically address out‐of‐hospital deaths with post‐mortem‐confirmed COVID‐19 are generally lacking.
In Russia, a special procedure aimed to distinguish cases in which death was related to or not causally associated with COVID‐19 using post‐mortem examinations was used since March 2020 and also included forensic autopsies performed for all deaths that occurred out of hospital. The authors participated in an expert assessment of fatal COVID‐19 cases in St. Petersburg submitted for review in March–May 2020. The study had two aims: 1) to present clinical characteristics and causes of death in patients who died with COVID‐19, and 2) to assess the prevalence of thrombotic events during the course of COVID‐19 and their association with the presence of diffuse alveolar damage (DAD) consistent with COVID‐19.
Methods
According to the procedure for handling death cases during the COVID‐19 pandemic implemented in Russia in March 2020, all in‐hospital deaths of patients with the COVID‐19 disease in Russia (either suspected or confirmed using real‐time polymerase chain reaction (PCR) assay) required a clinical autopsy aimed at establishing the patients’ cause of death. All deaths occurring at home and cases when violence could not be ruled out as a cause of death were submitted for forensic autopsy. The background COVID‐19‐positive cohort was therefore comprised of all fatal cases that occurred in St. Petersburg between 25 March 2020 and 31 May 2020 where SARS‐CoV‐2 tissue PCR in the trachea, bronchi or pulmonary tissue samples was positive. All COVID‐19‐positive fatalities were submitted for systematic centralized evaluation by the expert commission appointed by the St. Petersburg City Committee on Health Protection in order to establish the cause of death and its possible relation to the COVID‐19 infection.
A routine autopsy was initially performed either at a local clinical pathology laboratory affiliated with the hospital where the death occurred (clinical autopsy) or at the city forensic pathology laboratory (forensic autopsy). Tissue samples collected from pre‐specified locations (trachea, segmental bronchi and their branches of the 1st‐ and 2nd‐order, adjacent pulmonary parenchyma and subpleural regions) were submitted for histological assessment using light microscopy and haematoxylin–eosin and van Gieson’s staining and SARS‐CoV‐2 real‐time PCR using AmpliSens® CoV‐Bat‐FL Kit (Russia). In patients with visible macroscopic alterations, additional pulmonary tissue specimens selected for histological verification of the nature of the pulmonary damage were taken from lung segments that expressed the most distinguishable macroscopical abnormalities. Histological assessments of tissues collected from vital organs such as heart, liver, kidney, pancreas, spleen and brain were also performed.
After initial evaluation at the local pathology laboratory, all histological specimens from patients who had PCR‐confirmed COVID‐19 disease along with the autopsy report and available medical documentation were forwarded to the central pathology laboratory (MR and VK) for independent review of histological specimens, validation of consistency of observed findings with COVID‐19 and assessment of its possible causative association with death.
The presence of DAD was considered significant if typical histological alterations [6] were present in the alveolar–capillary unit, such as intra‐alveolar oedema, with multiple macrophages in the alveolar lumen and hyaline membranes lining the alveolar septa. DAD was considered minimal if no macroscopic abnormalities were observed at autopsy and alveolar patency was preserved. Typical examples of pulmonary parenchyma exhibiting signs of significant and minimal DAD are presented in Fig. 1.
Fig. 1.
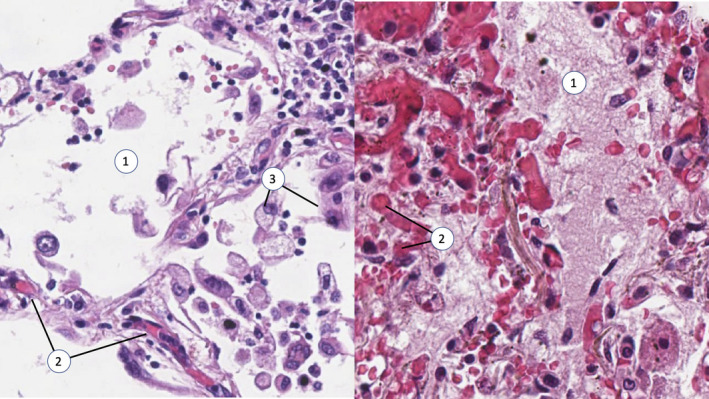
Minimal (left panel) and significant (right panel) diffuse alveolar damage. Note the viral transformation of desquamated alveolocytes, the early signs of microangiopathy with detectable interalveolar capillary occluded by erythrocyte clots and preserved alveolar patency on the left panel. On the right panel, alveolar space is filled with exudate, and capillaries are massively occluded with microthrombi. Numbers in the figure indicate alveolar space (1), microthrombi in alveolar capillaries (2) and desquamated alveolocytes (3). Haematoxylin–eosin staining, magnification ×200.
As a rule, death was categorized as COVID‐19‐related (COVID‐19 death) if patients demonstrated significant DAD. The presence and the characteristics of other comorbidities as potential death causes were considered in order to then categorize COVID‐19 as the primary or the contributing cause of death. In all other cases with positive SARS‐CoV‐2 tissue PCR but with no or minimal DAD, COVID‐19 was considered a concomitant condition not causally associated with death (non‐COVID‐19 death).
The final decision with regard to case classification was made by expert commission members (ER, VV, GS, VK and MR) based on the original local laboratory pathology report and the central pathology laboratory evaluation. In the clinical autopsy cohort, the review also included clinical documentation from the patient’s latest hospital admission. In case of disagreement or uncertainty, the decision was reached by a consensus‐based discussion.
Thrombotic phenomena reported during post‐mortem studies were classified as either pulmonary thromboembolism (PE), thrombosis in the branches of the pulmonary artery (PA) or microangiopathy at the level of alveolar–capillary unit. As distinguishing between PE and in situ thrombi may be problematic, especially in the medium‐size pulmonary vessels, we applied a conservative approach and diagnosed PE only in cases with identifiable embolic source, and all other cases of thrombi observed in the medium‐ and small‐size pulmonary vessels were classified as thrombosis in PA branches. Microangiopathy was defined as a complex of characteristic phenomena observed in the microcirculatory bed [6], including microthrombi in the arterioles, erythrocyte stasis and erythrocyte aggregates in capillary vessels, and capillary congestion.
In a subset of consecutive 42 cases of in‐hospital deaths in which clinical autopsies were performed (clinical autopsy cohort), the cause of death analysis was performed using medical record review for detailed analysis of the COVID‐19 disease course, therapy, clinical manifestations and histological findings. Similarly, a detailed review of histological and autopsy findings was performed in all 60 tissue PCR‐positive forensic autopsy cases.
All post‐mortem examinations, tissue collection and preparation, and information processing were performed as a part of standard routine. The study complies with the Declaration of Helsinki and meets local ethical and regulatory requirements.
Continuous data are presented as the mean ± standard deviation or the median [interquartile range]. The chi‐square test or the Fisher exact test was used to compare categorical variables, and the t‐test was used to compare continuous variables. Statistical analysis was performed using IBM SPSS Statistics release 26.0.0.0.
Results
Background COVID‐19‐positive cohort
In total during the study period, the SARS‐CoV‐2 tissue PCR of trachea and/or pulmonary tissue was positive in 799 cases (mean age 72 ± 14, 52% men), and in 502 (63%) of these cases, death was considered COVID‐19‐related (Figure S1). In this group, men died at a significantly younger age than women (69 ± 15 vs. 75 ± 13, p < 0.001).
Of all 799 SARS‐CoV‐2 tissue PCR‐positive autopsies, 60 were forensic autopsies. Age at death in the forensic autopsy cohort tended to be higher (75 ± 14 vs. 72 ± 14 years, p = 0.087), but gender distribution was similar to the patients for whom clinical autopsies were performed (48% vs. 52% men, respectively, p = 0.790). The cause of death was more likely to be COVID‐19‐related in the forensic autopsy cohort (83%) than in the patients for whom clinical autopsies were performed (62%, p < 0.001).
Forensic autopsy cohort
Patient characteristics and the list of primary causes of death in the forensic autopsy cohort are presented in Table 1 and Fig. 2. Death was identified as COVID‐19‐related in 50 (83%) patients, all of whom presented with significant DAD.
Table 1.
Clinical characteristics and the primary cause of death of the 102 patients with positive SARS‐CoV‐2 tissue PCR at autopsy
| Clinical autopsies | Forensic autopsies | |||||
|---|---|---|---|---|---|---|
|
COVID‐19‐related n = 30 |
Non‐COVID‐19‐related n = 12 |
p‐value |
COVID‐19‐related n = 50 |
Non‐COVID‐19‐related n = 10 |
p‐value | |
| Male, n (%) | 17 (57) | 5 (46) | 0.499 | 23 (46) | 6 (60) | 0.500 |
| Age, years | 67 ± 15 | 76 ± 13 | 0.056 | 79 ± 10 | 60 ± 21 | 0.017 |
| Age <60 years, n (%) | 10 (33) | 2 (17) | 0.453 | 2 (4) | 5 (50) | 0.001 |
| SARS‐CoV‐2 PCR positive prior to death, n | 22 of 26 tested | 3 of 6 tested | 0.101 | n.a. | n.a. | ‐ |
| Pre‐existing comorbidities, n (%) | ||||||
| Hypertension | 27 (90) | 11 (92) | 1.0 | 44 (80) | 7 (70) | 0.163 |
| Diabetes | 8 (27) | 2 (15) | 0.696 | n.d. | n.d. | ‐ |
| Ischaemic heart disease | 13 (43) | 8 (67) | 0.306 | 34 (68) | 4 (50) | 0.149 |
| COPD | 8 (27) | 0 | 0.128 | 4 (8) | 1 (10) | 1.000 |
| Obesity | 11 (37) | 1 (8) | 0.070 | n.d. | n.d. | ‐ |
| CHF | 4 (13) | 6 (50) | 0.020 | 5 (10) | 1 (10) | 1.000 |
| Ischaemic stroke | 6 (20) | 5 (42) | 0.243 | 6 (12) | 1 (10) | 1.000 |
| Atrial fibrillation | 8 (28) | 4 (33) | 0.704 | n.d. | n.d. | ‐ |
| No risk factors | 1 (3) | 1 (8) | 0.495 | 3 (6) | 3 (30) | 0.052 |
| Significant/minimal/no DAD by autopsy, n (%) | 30/0/0 | 1/1/10 | <0.001 | 50/0/0 | 1/4/5 | <0.001 |
| Primary cause of death | DAD – 23 | CHF – 4 | DAD – 43 | Ruptured aortic aneurism – 1 | ||
| PE – 2 | PE – 2 | Lymphocyte myocarditis – 1 | Cancer – 3 | |||
| Ischaemic stroke – 1 | Bacterial pneumonia – 2 | Myocardial infarction – 1 | Ruptured intracranial aneurism – 1 | |||
| ICH – 1 | Sepsis – 1 | Ischaemic stroke – 1 | Bacterial pneumonia – 2 | |||
| CMV pneumonia – 1 | Ischaemic stroke – 2 | Head trauma – 1 | Head trauma – 3 | |||
| Cancer – 1 | Tuberculosis – 1 | ICH – 1 | ||||
| Sepsis – 1 | Cancer – 2 | |||||
CHF, congestive heart failure; CMV, cytomegalovirus; DAD, diffuse alveolar damage; DAD, diffuse alveolar damage; ICH, intracranial haemorrhage; n.a., not applicable; n.d., no data; PCR, polymerase chain reaction.
Fig. 2.
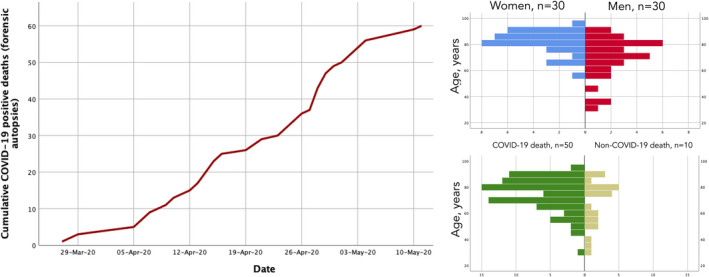
Cumulative number of deaths with positive SARS‐CoV‐2 real‐time PCR on trachea/pulmonary parenchyma referred from the forensic pathology unit (left panel), age‐gender distribution (upper right‐hand panel) and the determined relationship between COVID‐19 and the cause of death (lower right‐hand panel).
In 10 cases, COVID‐19 infection was considered to be a background concomitant condition (non‐COVID‐19 deaths). One patient died of a ruptured aortic aneurism, and despite the presence of typical morphological manifestations of SARS‐CoV‐2 pneumonia with significant DAD, SARS‐CoV‐2 was judged as unrelated to the cause of death. In the other 9 patients, no DAD or minimal DAD was observed in the pulmonary parenchyma.
Pre‐existing cardiovascular comorbidities were observed in the vast majority of patients. A detailed description of primary vs. contributing cause of deaths in the forensic autopsy cohort is presented in Table S1.
The amplification cycle threshold for positive PCR was reported for 9 tracheal (median Ct 33 [27–36]) and 13 parenchymal (median Ct 33 [28–35]) specimens.
Clinical autopsy cohort
Clinical characteristics in the subgroup of 42 consecutive clinical autopsy cases in which the cause of death analysis was performed using medical record review are presented in Table 1 and Table S2. The patients included in the subgroup did not differ from the rest of the patients for whom clinical autopsy was performed in either age (69 ± 15 vs 72 ± 15 years, p = 0.205) or gender (52% vs 59% males, p = 0.342).
Death was identified as COVID‐19‐related in 30 (71%) patients, all of whom presented with significant DAD. In 12 cases, COVID‐19 infection was classified as unrelated to the mechanisms causing death. One of these 12 patients died in hospital of massive pulmonary thromboembolism developed on the background of ischaemic cardiomyopathy and severe congestive heart failure, and despite the presence of significant DAD that patient’s death cause was categorized as not related to COVID‐19. In the other 11 patients, no DAD or minimal DAD was observed.
The details regarding the clinical course prior to death can be found in Table S3. Patients were admitted to the hospital with a median of 6 [2–9] days from onset of symptoms with a median duration of 6 [3–14] days of hospital stay until death. The vast majority of patients were admitted to the intensive care unit and were treated with invasive ventilation support.
The amplification cycle threshold for positive PCR was reported for 11 tracheal (median Ct 27 [23–30]) and 17 parenchymal (median Ct 33 [24–35]) samples. Results of PCR tests performed using nasopharyngeal swabs pre‐death in hospital were available in 32 cases, 25 of which were positive. Of the patients with observed significant DAD, 27 had PCR tests done on nasopharyngeal swabs before death. Of the 27 patients, 23 tested positive.
Thrombotic phenomena
Of all 102 post‐mortem studies included in the cause of death analysis, five patients had PE. Thrombosis in the PA branches associated with microangiopathy was observed in 38 patients (37%), and 28 patients (27%) had microangiopathy without notable thrombosis in the PA branches.
Of the five patients with documented PE, a forensic autopsy was performed in one patient who died at home of massive DAD and in whom PE was not deemed to have contributed significantly to death. In four patients, the source of embolism was identified as deep vein thrombosis. In one patient, multiple thrombi were found in the right chambers of the heart and were deemed to be the source of the massive PE that caused death. None of the patients were treated with anticoagulants prior to hospital admission. Two patients were hospitalized with PE following several days of pneumonia symptoms. In two other patients, PE developed between one and two weeks after hospital admission despite a prophylactic dose heparin regimen. We did not observe an association between PE and DAD: PE was present in 1 of 20 patients who had no DAD or minimal DAD (5%) and in 4 of 82 patients (4.9%) who had significant DAD (p = 1.0).
Thrombotic phenomena in lungs were related to the cause of death (Fig. 3). Of the patients with COVID‐19 as the primary cause of death, only 19% had no thrombotic phenomena in the lungs, as opposed to 38% amongst those with COVID‐19 as a contributing cause of death and 54% amongst patients whose death was not related to COVID‐19 (p = 0.002).
Fig. 3.
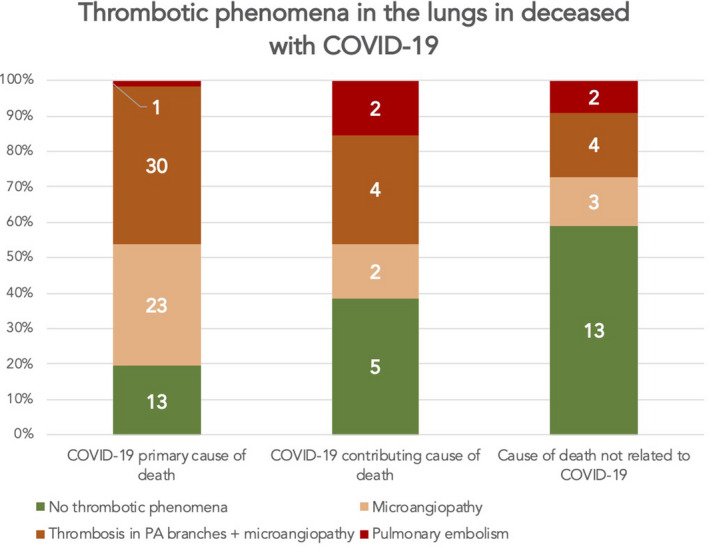
Prevalence of pulmonary embolism, thrombosis in the pulmonary artery (PA) branches and microangiopathy with regard to the association between COVID‐19 and the cause of death. Thrombotic phenomena were less prevalent in patients who died from causes not related to COVID‐19 (p = 0.002). The numbers in the bar diagram correspond to the absolute count of patients in each group.
Thrombotic manifestations were associated with significant DAD (Fig. 4) and were observed in 78% of the patients with significant DAD, whereas just 35% of the patients who had no DAD or minimal DAD had these signs present in the lungs (p = 0.002).
Fig. 4.
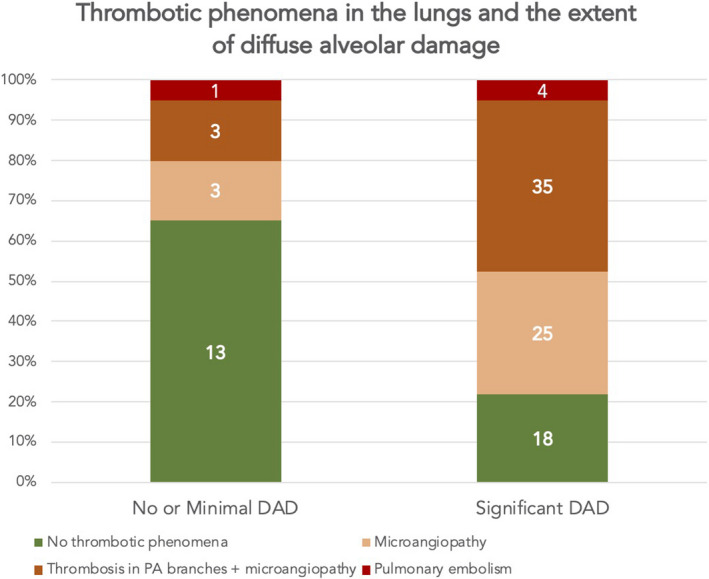
Prevalence of pulmonary embolism, thrombosis in the pulmonary artery (PA) branches and microangiopathy related to the presence and extent of diffuse alveolar damage consistent with SARS‐CoV‐2 infection. Thrombotic phenomena were less prevalent in patients with no diffuse alveolar damage or with minimal diffuse alveolar damage (p = 0.002). The numbers in the bar diagram correspond to the absolute number of patients in each group.
Extrapulmonary thrombotic events observed in our study were ischaemic stroke (n = 4), acute myocardial infarction (n = 3) and critical lower limb ischaemia (n = 1). All of these eight patients had pre‐existing advanced atherosclerotic plaques in cerebral, coronary and peripheral arteries. Significant DAD was observed in two patients who developed ischaemic stroke, two patients with myocardial infarction and one patient with critical lower limb ischaemia.
In total, 11 patients developed either PE or extrapulmonary thrombotic events. Of these 11 patients, 73% (n = 8) had significant DAD as compared to 81% (n = 74) of the patients who did not develop thrombotic events (p = 0.447).
Inflammatory reaction in the myocardium
Interstitial oedema and microangiopathy with erythrocyte aggregates occluding the capillary vessels were observed in 35 of 102 reviewed autopsies, and in 12 others, it was associated with mononuclear infiltration of myocardium. The extent of inflammatory reaction fulfilled the morphological criteria for lymphocyte myocarditis [7] in two patients and in one of whom myocarditis was associated with extensive noncoronary necroses and was considered the primary cause of death. Neither of the two patients in whom quantitative criteria for lymphocyte myocarditis were fulfilled had any notable amount of pericardial effusion. We did not observe a significant association between the presence of DAD in the lungs and lymphomononuclear infiltration in the myocardium, which was present in 11 of 82 patients with significant DAD and in 1 of 20 patients with no DAD or minimal DAD (p = 0.452). Figure 5 illustrates the structural alterations observed in the myocardium in patients with COVID‐19.
Fig. 5.
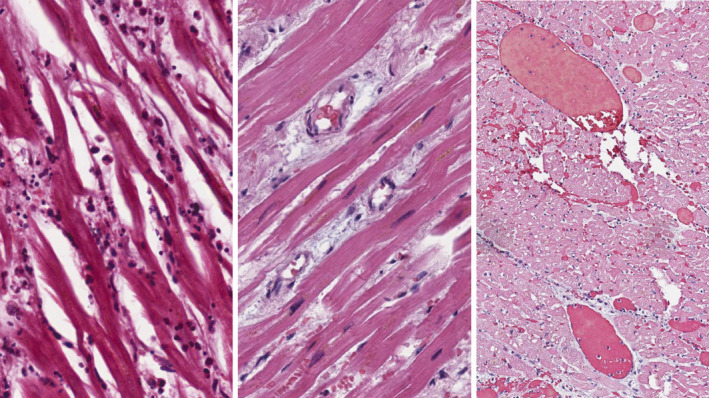
The myocardium in patients with COVID‐19. Left panel: interstitial oedema and diffuse lymphomononuclear infiltration (haematoxylin–eosin stain, ×200). Middle panel: prominent interstitial oedema (haematoxylin–eosin stain, ×150). Right panel: massive thrombosis in myocardial vessels of variable diameter (haematoxylin–eosin stain, ×100).
Discussion
Generalizability of study findings
Our data provide the most representative autopsy‐verified spectrum of causes of death during the COVID‐19 pandemic in a city with a population exceeding 5,000,000. According to the Russian Federal State Statistic Service (ROSSTAT), during the March–May 2020 study period, 2,016 COVID‐19‐positive patients died in St. Petersburg, of whom 1,290 patients (64%) died of COVID‐19‐related causes [8]. The excess mortality calculated with regard to the average number of deaths in St. Petersburg during the same months of 2016‐2019 was +12,3% or 1918 in the absolute count. The similarity between excess mortality figures and number of reported deaths with COVID‐19 indicates that ROSSTAT methodology accurately reflects the expected contribution of the pandemic to the increased mortality, with a low probability of substantially undercounting COVID‐19 amongst the deceased. The study population of 799 autopsy‐verified SARS‐CoV‐2‐positive deaths represents 39% of all deaths that occurred during the study period in patients with positive PCR. The proportion of death causatively related to COVID‐19 infection in our background population is remarkably similar to the ROSSTAT report and is thus likely to be representative of the population as a whole.
Out‐of‐hospital deaths and forensic autopsies
Our nonselected cohort also makes it possible to determine the relative contribution of undiagnosed COVID‐19 to death during the pandemic. To the best of our knowledge, our forensic autopsy cohort is the largest reported so far, as previous reports were limited to case presentations [9] or limited case series [10, 11. In St. Petersburg, forensic autopsies are centralized and are performed at a single location at the Bureau of Forensic Pathology Expertise. All cases in which disease affecting lungs could be suspected during forensic examination underwent tissue PCR and were referred to the central laboratory for independent review.
In the vast majority of forensic autopsy cases, DAD was the main cause of death, which confirms earlier reports based on observations made during forensic autopsies of sudden out‐of‐hospital deaths [9, 10, 11. When assessed with regard to the total number of COVID‐19‐related deaths reported during the study period, the autopsy‐confirmed out‐of‐hospital COVID‐19‐related deaths appear to represent 10% of all COVID‐19‐related mortality.
Thrombosis and thromboembolism in COVID‐19
Previous studies indicated a spectrum of thrombotic phenomena associated with DAD caused by COVID‐19, including venous thromboembolism, thrombosis in segmental and subsegmental pulmonary artery vessels [12], and microangiopathy [13]. Thrombi in PA branches are commonly reported [14] and appear to be present in a greater proportion of fatalities during the COVID‐19 pandemic than during the ‘Spanish flu’ of 1918‐1919, as recently reported [15]. Thrombotic microangiopathy attributable to viral endothelial damage [13] and incorporating capillary or arteriolar thrombosis appears to be an even more prevalent finding – it was previously reported in the majority of autopsy studies in small‐size series [1, 6, 14, 16 and was also confirmed by our data. Moreover, thrombi in the PA branches in our cohort were a very common finding and were strongly associated with the DAD (Fig. 4) and the strength of causative association between COVID‐19 and death (Fig. 3). Our findings therefore further confirm the role coagulation alterations play in the pathogenesis of alveolar damage in patients with COVID‐19.
PE was suggested as a relatively common finding in patients who died of COVID‐19, although conflicting results are reported in literature. PE was documented as the cause of death in 4 of the 12 patients in one study [20], but was not observed in another similar‐size cohort [21]. In our data, only a small number of patients were found to have PE, and a few patients had extrapulmonary thrombotic events. Our findings therefore question the contribution of thromboembolic complications as the cause of death to out‐of‐hospital mortality during the COVID‐19 pandemic, and indicate no association with DAD; however, the number of observed events was low.
Anticoagulation has been suggested as a valuable component of COVID‐19 management and was associated with lower mortality and lower risk of intubation in retrospective studies [22, 23. However, data from randomized controlled trials that evaluate the use of oral anticoagulants in COVID‐19 have not yet become available, and nearly one third of thromboembolic complications confirmed at autopsy occurred in patients on therapeutic anticoagulation [22]. It is currently unknown whether or not oral anticoagulation is effective in preventing or treating microangiopathy affecting the capillary bed, which is the major component of COVID‐19‐related organ damage, as also observed in our study.
Myocardial injury
Myocardial injury in the context of COVID‐19 has been proposed as a component of the broader term of acute COVID‐19 cardiovascular syndrome [24], which may manifest by a variety of clinical syndromes that include cardiac arrhythmias, heart failure and myocardial ischaemia with a spectrum of electrocardiographic manifestations [25]. The mechanisms of myocardial injury can be related to either noncoronary lesions attributed to a complex interplay between inflammatory reaction and microangiopathy with interstitial oedema and microcirculatory stasis with erythrocyte aggregates leading to myocardia hypoperfusion [26] or ischaemic lesions due to thrombosis in an epicardial coronary artery.
Although ischaemic heart disease has been considered a risk factor for a severe course of COVID‐19, there is only limited information concerning the prevalence of myocardial infarction as the cause of death in patients with COVID‐19. In a recent analysis based on data from the Swedish national SWEDEHEART registry, a 20% drop in admissions with myocardial infarction was noted during the pandemic nationwide and did not appear to be related to delayed admissions or interrupted chain of acute care delivery [27]. In our cohort, the majority of patients had pre‐existing ischaemic heart disease with autopsy‐confirmed atherosclerotic plaques in the coronary arteries (Table 1), but only three patients (3%) developed acute myocardial infarction during the course of COVID‐19, for two of these three patients, this was the primary cause of death. Circumstances related to myocardial infarction were known for one patient, who then developed inferior myocardial infarction twelve days after hospital admission with COVID‐19, underwent primary percutaneous coronary intervention and died six days later due to progressive cardiorespiratory failure.
By contrast, noncoronary myocardial injury associated with COVID‐19 appears to be a relatively common finding reported in 45% of patients in one cohort study using cardiac magnetic resonance imaging [28]. This prevalence is in striking agreement with our study, according to which 46% of cases showed a histological picture of an inflammatory reaction in the myocardium. In another post‐mortem study, mild lymphocytic myocarditis was observed in one third of cases [21]. In our cohort, however, lymphomononuclear infiltration was less common and was similar to the findings made in the series of 21 post‐mortem examinations performed by cardiovascular pathologists, in which the presence of lymphocytic myocarditis was reported in 3 cases (14%) [26]. Recent meta‐analysis of 277 reported autopsies indicated that the true prevalence of COVID‐19‐associated myocarditis is likely to be even lower – 1.4% of fatal cases [29].
Nevertheless, acute myocardial injury associated with COVID‐19 and manifested with cardiogenic shock was reported [30]. In our cohort, lymphocyte myocarditis with diffuse noncoronary necroses in the ventricular myocardium was considered the primary cause of death in the presence of bilateral pneumonia in only one patient who died at home and in whom a forensic autopsy was later performed.
Post‐mortem findings in patients with minimal alveolar damage
Determining the cause of death in patients who were deemed to have died from causes other than COVID‐19 was generally not problematic. Of 22 patients in whom COVID‐19 was not associated with death, only 2 patients had significant DAD, and it is debatable whether or not SARS‐CoV‐2 infection would have contributed to their death. In one of these two cases, death occurred out of hospital as a result of a ruptured aortic aneurism, whilst the other death occurred in hospital where the patient was brought after suffering from a massive PE and myocardial infarction. In these two cases, autopsy findings were such that the extent of DAD was not considered sufficient to justify the conclusion regarding a causal relationship between SARS‐CoV‐2 infection and death. However, one cannot completely rule out the possibility that infection could have triggered cardiovascular complications and contributed to the fatal outcome.
Limitations
Although autopsy remains the gold standard for determining the cause of death, decisions regarding the causal relationships between autopsy findings and death mechanisms remain subjective, especially when autopsies are performed at different locations or when limited information regarding the clinical course of the disease is available, such as for out‐of‐hospital deaths. In our study, we attempted to minimize subjectivity by having the central laboratory review all histological specimens, with the final categorization reached by expert group consensus. This approach, however, cannot completely eliminate a certain degree of subjectivity in classifying morphological patterns, such as those observed in histological specimens, or in establishing causative associations between observed comorbidities and mechanisms of death.
Furthermore, due to the high number of autopsies performed during the pandemic, the extent of examinations that were feasible to perform on a large scale was limited to a meaningful minimum and thus did not include immunohistochemistry or virus identification tissues that could have provided valuable complementary information for the study.
Conclusion
Based on the largest to date systematic review of consecutive clinical and forensic autopsies performed during the first wave of the SARS‐CoV‐2 pandemic, a causal relationship between COVID‐19 and death was confirmed in the vast majority of PCR‐positive fatalities and was strongly associated with extensive DAD. Our data further support the important role of thrombotic phenomena involving microcirculatory bed in the development of COVID‐19‐related organ damage. Cardiac manifestations in the reviewed series were relatively mild and fulfilled the criteria for lymphocyte myocarditis in less than 2% of all cases. Finally, our findings highlight the importance of forensic autopsies, which revealed 10% of all COVID‐19‐related deaths.
Conflict of interests
None declared.
Supporting information
Figure S1. Background cohort.
Table S1. Primary and contributing causes of death in 60 consecutive SARS‐CoV2 tissue PCR positive forensic autopsies.
Table S2. Primary and contributing causes of death among the subgroup of 42 consecutive SARS‐CoV2 tissue PCR positive clinical autopsies.
Table S3. Characteristics of the clinical course and interventions during hospital admission in 42 consecutive patients with positive SARS‐CoV2 tissue PCR at clinical autopsy.
Acknowledgements
Pyotr Platonov was supported by ALF funding (Sweden) and the Swedish Heart–Lung Foundation.
Romanova ES, Vasilyev VV, Startseva G, Karev V, Rybakova MG, Platonov PG. Cause of death based on systematic post‐mortem studies in patients with positive SARS‐CoV‐2 tissue PCR during the COVID‐19 pandemic. J Intern Med 2021; 290: 655–665.
References
- 1. Maiese A, Manetti AC, La Russa R, Di Paolo M, Turillazzi E, Frati P, Fineschi V. Autopsy findings in COVID‐19‐related deaths: a literature review. Forensic Sci Med Pathol. 2020. 10.1007/s12024-020-00310-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Cattaneo C. Forensic medicine in the time of COVID 19: An Editorial from Milano. Italy. Forensic Sci Int. 2020;312:110308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Cusack DA. COVID‐19 pandemic: Coroner's database of death inquiries with clinical epidemiology and total and excess mortality analyses in the District of Kildare March to June 2020. J Forensic Leg Med. 2020;76:102072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Nogueira PJ, Nobre MA, Nicola PJ, Furtado C, Vaz Carneiro A. Excess mortality estimation during the COVID‐19 pandemic: Preliminary data from Portugal. Acta Med Port. 2020;33(6):376–83. [DOI] [PubMed] [Google Scholar]
- 5. Wang T, Chen R, Liu C, Liang W, Guan W, Tang R, et al. Attention should be paid to venous thromboembolism prophylaxis in the management of COVID‐19. Lancet Haematol. 2020;7(5):e362–e363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Sanguedolce F, Zanelli M, Ascani S, Zizzo M, Tortorella S, Soriano A. SARS‐CoV‐2‐related lung pathology: macroscopic and histologic features and their clinical implications. Panminerva Med. 2020. [DOI] [PubMed] [Google Scholar]
- 7. Caforio AL, Pankuweit S, Arbustini E, Basso C, Gimeno‐Blanes J, Felix SB. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013;34(33):2636–48, 2648a–2648d. [DOI] [PubMed] [Google Scholar]
- 8. Natural population change in the subjects of Russian Federation for the period January‐June 2020. In: Federal State Statistic Service, Russian Federation; 2020.
- 9. Suess C, Hausmann R. Gross and histopathological pulmonary findings in a COVID‐19 associated death during self‐isolation. Int J Legal Med. 2020;134(4):1285–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Pell R, Fryer E, Manek S, Winter L, Roberts ISD. Coronial autopsies identify the indirect effects of COVID‐19. Lancet Public Health. 2020;5(9):e474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Tombolini A, Scendoni R. SARS‐CoV‐2‐related deaths in routine forensic autopsy practice: histopathological patterns. Int J Legal Med. 2020;134(6):2205–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Lax SF, Skok K, Zechner P, Kessler HH, Kaufmann N, Koelblinger C, et al. Pulmonary arterial thrombosis in COVID‐19 with fatal outcome : results from a prospective, single‐center. Clinicopathologic Case Series. Ann Intern Med. 2020;173(5):350–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, et al. Endothelial cell infection and endotheliitis in COVID‐19. Lancet. 2020;395(10234):1417–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Chmielik E, Jazowiecka‐Rakus J, Dyduch G, Nasierowska‐Guttmejer A, Michalowski L, Sochanik A, Ulatowska‐Bialas M. COVID‐19 autopsies: A case series from Poland. Pathobiology. 2021;88(1):78–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Burkhard‐Koren NM, Haberecker M, Maccio U, Ruschitzka F, Schuepbach RA, Zinkernagel AS. Higher prevalence of pulmonary macrothrombi in SARS‐CoV‐2 than in influenza A: autopsy results from 'Spanish flu' 1918/1919 in Switzerland to Coronavirus disease 2019. J Pathol Clin Res. 2021;7:135–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Sadegh Beigee F, Pourabdollah Toutkaboni M, Khalili N, Nadji SA, Dorudinia A, Rezaei M, et al. Diffuse alveolar damage and thrombotic microangiopathy are the main histopathological findings in lung tissue biopsy samples of COVID‐19 patients. Pathol Res Pract. 2020;216(10):153228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Vasquez‐Bonilla WO, Orozco R, Argueta V, Sierra M, Zambrano LI, Muñoz‐Lara F, et al. A review of the main histopathological findings in coronavirus disease 2019. Hum Pathol. 2020;105:74–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Falasca L, Nardacci R, Colombo D, Lalle E, Di Caro A, Nicastri E, et al. Postmortem findings in italian patients with COVID‐19: A descriptive full autopsy study of cases with and without comorbidities. J Infect Dis. 2020;222(11):1807–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Schurink B, Roos E, Radonic T, Barbe E, Bouman CSC, de Boer HH, et al. Viral presence and immunopathology in patients with lethal COVID‐19: a prospective autopsy cohort study. Lancet Microbe. 2020;1(7):e290–e299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Wichmann D, Sperhake JP, Lutgehetmann M, Steurer S, Edler C, Heinemann A, et al. Autopsy findings and venous thromboembolism in patients with COVID‐19: A prospective cohort study. Ann Intern Med. 2020;173(4):268–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Schaller T, Hirschbuhl K, Burkhardt K, Braun G, Trepel M, Märkl B, Claus R. Postmortem examination of patients with COVID‐19. JAMA. 2020;323(24):2518–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Nadkarni GN, Lala A, Bagiella E, Chang HL, Moreno PR, Pujadas E, et al. Anticoagulation, bleeding, mortality, and pathology in hospitalized patients with COVID‐19. J Am Coll Cardiol. 2020;76(16):1815–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Tang N, Bai H, Chen X, Gong J, Li D, Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J Thromb Haemost. 2020;18(5):1094–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Hendren NS, Drazner MH, Bozkurt B, Cooper LT Jr. Description and proposed management of the acute COVID‐19 cardiovascular syndrome. Circulation. 2020;141(23):1903–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Haseeb S, Gul EE, Cinier G, Bazoukis G, Alvarez‐Garcia J, Garcia‐Zamora S, et al. Value of electrocardiography in coronavirus disease 2019 (COVID‐19). J Electrocardiol. 2020;62:39–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Basso C, Leone O, Rizzo S, De Gaspari M, van der Wal AC, Aubry M‐C, et al. Pathological features of COVID‐19‐associated myocardial injury: a multicentre cardiovascular pathology study. Eur Heart J. 2020;41(39):3827–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Mohammad MA, Koul S, Olivecrona GK, Gӧtberg M, Tydén P, Rydberg E, et al. Incidence and outcome of myocardial infarction treated with percutaneous coronary intervention during COVID‐19 pandemic. Heart. 2020;106(23):1812–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Knight DS, Kotecha T, Razvi Y, Chacko L, Brown JT, Jeetley PS, et al. COVID‐19: myocardial injury in survivors. Circulation. 2020;142(11):1120–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Halushka MK, Vander Heide RS. Myocarditis is rare in COVID‐19 autopsies: cardiovascular findings across 277 postmortem examinations. Cardiovasc Pathol. 2020;50:107300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Tavazzi G, Pellegrini C, Maurelli M, Belliato M, Sciutti F, Bottazzi A, et al. Myocardial localization of coronavirus in COVID‐19 cardiogenic shock. Eur J Heart Fail. 2020;22(5):911–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. Background cohort.
Table S1. Primary and contributing causes of death in 60 consecutive SARS‐CoV2 tissue PCR positive forensic autopsies.
Table S2. Primary and contributing causes of death among the subgroup of 42 consecutive SARS‐CoV2 tissue PCR positive clinical autopsies.
Table S3. Characteristics of the clinical course and interventions during hospital admission in 42 consecutive patients with positive SARS‐CoV2 tissue PCR at clinical autopsy.


