Highlights
Neurological manifestations constitute a serious cause of severe coronavirus disease 2019 (COVID‐19).
Microglial hyperactivation by the two‐hit process.
Severe COVID‐19 patients carry pre‐activated microglia following a first immune challenge.
Exaggerated response of sensitized microglia to COVID‐19, the second immune challenge.
1.
By the end of February 2021, approximately 2.5 million individuals worldwide have died of the novel coronavirus disease 2019 (COVID‐19), caused by the severe acute respiratory syndrome coronavirus (SARS‐CoV‐2). The number of severe and critically ill patients is also substantial. In some studies, such as one in China, enrolling 77 314 individuals (including 44 662 confirmed cases), 14% of participants experienced severe symptoms, with 5% being critically ill. 1 While the general mortality rate is estimated to be around 2%, the rate of death among the critically ill patients can reach up to 47%. 2 Therefore, early prognosis of individuals at risk would offer timely intervention options that may save more patient lives.
Although COVID‐19 is primarily a respiratory disease, neurological manifestations have emerged as a serious cause of morbidity and mortality. In a retrospective study from Wuhan, China, enrolling 214 hospitalized patients, 36.4% exhibited neurological manifestations including headache, ataxia, dizziness, seizure, and impaired consciousness. 3 Results from a UK‐wide surveillance study 4 enrolling 153 patients showed that altered mental state was the second most common condition (31% of subjects, of whom 6% had encephalitis). In all, 18% of patients presented with a neuropsychiatric diagnosis and 62% with a cerebrovascular event (e.g., ischemic stroke), highlighting that acute neurological complications are commonly encountered, even in persons younger than 60 years of age. 4 At even a higher rate, in a French study involving 58 hospitalized patients, 67% presented corticospinal tract signs, and 69% developed agitation. 5
Strong sickness response related to the hyper‐inflammatory state, that is, a “cytokine storm,” including severe effects in the central nervous system (CNS), mostly occurs in older patients (>65 of age), though surely not in all individuals. A possible explanation for such interindividual differences is that younger individuals have a stronger immune response of certain components such as interleukin‐17 (IL‐17) that may aid in preventing a more long‐lasting drastic infection, 6 or that already higher levels of circulating oxidative stress and pro‐inflammatory cytokines in the elderly make them more prone for such a hyper‐inflammatory state. 7 It is also suggested that individuals who carry elevated levels of pre‐existing immunity against seasonal human coronaviruses (HCoVs), which cross‐react with SARS‐CoV‐2, are somewhat protected from severe and even symptomatic COVID‐19. 8
Another explanation for interindividual susceptibly to COVID‐19 is related to microglia, also referred to as the brain's immune cells. These cells are derived from macrophages and constitute the most prominent immune and cytokine‐producing cells in the brain. 9 The reactivity of these innate immune cells of the CNS has been frequently pointed out to vary markedly between individuals, especially with regard to age, health status, and stress, including early‐life encountered immune challenges, such as traumatic physiological events, early infection, and exposure to pollutants (reviewed in Hanamsagar and Bilbo 9 ), among others. Recent data suggested that these non‐neuronal cells could play a pivotal role in COVID‐19 infections and the severity of the illness. 3 , 4 , 5 , 10 , 11 , 12 , 13
In accordance with the increasing evidence, we propose that microglia reactivity constitutes an additional plausible explanation for the observed interindividual differences in response to COVID‐19 and also for the severity of symptoms observed in COVID‐19 patients, in particular in older patients or those with comorbidity. This would be in line with the “two‐hit” hypothesis, which suggests that an initial stress encountered earlier in life (the first hit) sensitizes microglial cells that later results in an exaggerated elevation of microglial activity to subsequent stressor events (the second hit). This first triggering produces so‐termed “primed microglia” (Figure 1), which can then, following a second insult, more readily be activated to a detrimental hyper‐activated state. 7 , 14 Microglial priming (or sensitization) is characterized by numerous transcriptional and morphological modifications that occur as stress responses, including altered synaptic connections, epigenetic changes including histone modifications and hypomethylation of inflammatory genes, upregulation of antigens at the cellular surface and multiplied numbers of microglia itself (reviewed in Norden et al. 7 and Niraula et al. 14 ). Primed microglia does not refer to an elevated immune response at baseline, but rather to an exaggerated and uncontrolled inflammatory reaction to a secondary immune challenge/stimulus that may be even of comparably mild nature. 14
Figure 1.
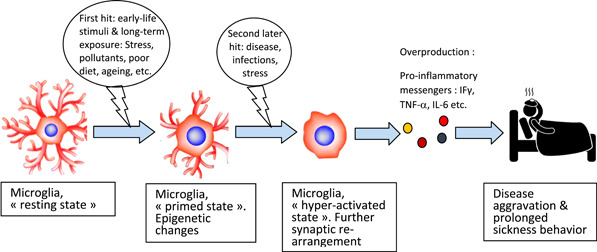
Microglial hyperactivation by the two‐hit process and its contribution to disease aggravation such as COVID‐19. COVID‐19, coronavirus disease 2019; IFγ, interferon gamma; IL, interleukin; TNF‐α, tumor necrosis factor‐α
Therefore, our hypothesis is that many patients experiencing severe COVID‐19 carry already pre‐activated microglia. Thus, these primed guardians will be hyperactive when facing an ensuing immunological challenge (here the SARS‐Co‐V‐2 infection: the “second‐hit”). Consequently, this second insult potentiates the response of microglia that will produce an array of neurotoxic agents, including pro‐inflammatory cytokines, reactive oxygen (ROS), and reactive nitrogen species (RNS). As a consequence, the cytokine storm in the brain and the periphery ensues in excessive strong sickness symptoms, a maladaptive brain mediated‐response, contributing to health deterioration; whereas in general, normal sickness behavior is rather adaptive and reversible, facilitating recovery from disease. 15
Indeed, high levels of various pro‐inflammatory cytokines are a typical observation in patients with severe illness. 16 Already during early disease stages, significantly higher levels of IL‐6 and IL‐10 were detected in patients with severe COVID‐19 compared to those with mild illness. 2 It is well admitted that systemic inflammation can adversely affect the central nervous system, leading to aberrant brain‐resident immune‐cell activation. Typically systemic inflammation, including the potentially fatal “systemic cytokine storm,” may even result in organ failure, including pulmonary, kidney, and heart failure in severe COVID‐19. However, a neuroinflammation scenario, characterized by the activation of glial cells, would provoke rather an encephalopathy, including mild neurological manifestations (such as headache, anosmia, and ageusia) and also severe symptoms (such as delirium, acute ischemic stroke, meningitis/encephalitis, acute necrotizing hemorrhagic encephalopathy, and seizure) that can be potentially life‐threatening in up to two‐thirds of the hospitalized patients. 3 , 4 , 5
In a prospective autopsy cohort study performed on 21 patients who passed away from COVID‐19, a massive activation of microglia with the formation of nodules was found. 10 In a post‐mortem study carried out on 25 COVID‐19 patients, 80% presented modifications markedly in the microglial immune activation; 48% exhibited a moderate‐to‐severe degree of microgliosis and microglial nodules. 11 Significant axonal damage was observed in the cohort, which was more pronounced in patients with microglial nodules. In addition, several microglial clusters displaying high immune activation and expression of microglial functional markers were identified in patients who succumbed to COVID‐19. 11 In a U.S. study, from 19 COVID‐19 autopsy cases, 13 exhibited perivascular‐activated microglia; in 5 patients activated microglia were detected adjacent to neurons, which is suggestive for neuronophagia in several brain areas. 12 In a German post‐mortem study, an increased phagocytic activity was inferred from the microglia phenotype (higher expression of the lysosomal marker CD68). In this study, it was hypothesized that anosmia, a common neurological symptom in COVID‐19 patients, was related to pronounced astrogliosis and microgliosis in the olfactory bulb. 13
During the ongoing COVID‐19 pandemic, the mortality rate in older patients is approaching 10%, and has been stated recently to be up to 62 times higher in people over 65 compared to those under 54 years of age. 17 Aging has been reported to be associated with higher levels of ROS and RNS, and inflammatory markers such as C‐reactive protein (CRP) and circulating cytokines such as IL‐6. Interestingly, in a cohort study, an average of 10‐year equivalent decline in global cognitive performance was found in people who were hospitalized and ventilated and then had recovered from COVID‐19. 18 However, it has also been proposed that aging can by itself act as priming or sensitizing stimulus for microglia, setting the base for the “first‐hit.” 14 Priming of microglia was also proposed to occur during neurodegenerative diseases, as well as by psychological stress across life span, as reviewed previously. 14
Indeed, chronic diseases such as obesity and type‐2 diabetes, also more likely to co‐occur with age, and which can lead to chronic low‐grade inflammation, have been associated with microglial activation in several animal model studies. 19 Interestingly, it has been stated that once microglia are activated, they may then take on a primed profile. 7 , 14 In addition to the connection between the “two‐hit” on microglia and the severity of COVID‐19, of course other host‐related factors predisposing for a higher risk of severe COVID‐19 disease do exist, such as recently highlighted for the genomic segment on chromosome 3 that has been inherited from Neanderthals, constituting a risk locus for respiratory failure in people from Asia and to a lesser extent also for Europeans. 20
2.
In summary, microglial hyper‐activation is associated with several diseases and serious complications that could be fatal for ill humans. Microglial priming or sensitization, which could occur in response to several inflammatory conditions including normal and pathological aging, psychological stress, or other stressor events encountered early in life and also chronic diseases, characterized by low‐grade inflammation, is an important process for vulnerability to subsequent immune challenges. According to the recent evidence, the “two‐hit” hypothesis provides an additional explanation for the vulnerability of certain patients to severe COVID‐19. Furthermore, a better understanding of this process may pave the route toward developing appropriate interventions, preventing an excessive microglial cascade activation.
AUTHOR CONTRIBUTIONS
Jaouad Bouayed created the outline of the article and wrote the majority of the manuscript. Torsten Bohn contributed with additional aspects and edited the document.
CONFLICT OF INTERESTS
The authors declare that there are no conflict of interests.
REFERENCES
- 1. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID‐19) outbreak in China summary of a report of 72 314 cases from the Chinese center for disease control and prevention. JAMA. 2020;323:1239‐1242. [DOI] [PubMed] [Google Scholar]
- 2. Dayarathna S, Jeewandara C, Gomes L, et al. Similarities and differences between the 'cytokine storms' in acute dengue and COVID‐19. Sci Rep. 2020;10:19839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Mao L, Jin H, Wang M, et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683‐690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Varatharaj A, Thomas N, Ellul MA, et al. Neurological and neuropsychiatric complications of COVID‐19 in 153 patients: a UK‐wide surveillance study. Lancet Psychiatry. 2020;7:875‐882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Helms J, Kremer S, Merdji H, et al. Neurologic features in severe SARS‐CoV‐2 infection. N Engl J Med. 2020;382:2268‐2270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Pierce CA, Preston‐Hurlburt P, Dai Y, et al. Immune responses to SARS‐CoV‐2 infection in hospitalized pediatric and adult patients. Sci Transl Med. 2020;12:eabd5487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Norden DM, Muccigrosso MM, Godbout JP. Microglial priming and enhanced reactivity to secondary insult in aging, and traumatic CNS injury, and neurodegenerative disease. Neuropharmacology. 2015;96(Pt A):29‐41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Guthmiller JJ, Wilson PC. Remembering seasonal coronaviruses. Science. 2020;370(6522):1272‐1273. 10.1126/science.abf4860 [DOI] [PubMed] [Google Scholar]
- 9. Hanamsagar R, Bilbo SD. Environment matters: microglia function and dysfunction in a changing world. Curr Opin Neurobiol. 2017;47:146‐155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Schurink B, Roos E, Radonic T, et al. Viral presence and immunopathology in patients with lethal COVID‐19: a prospective autopsy cohort study. Lancet Microbe. 2020;1:e290‐e299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bengsch B, Schwabenland M, Salié H, et al. Deep spatial profiling of COVID19 brains reveals neuroinflammation by compartmentalized local immune cell interactions and targets for intervention. Research Square. 2021. 10.21203/rs.3.rs-63687/v1 [DOI] [Google Scholar]
- 12. Lee M‐H, Perl DP, Nair G, et al. Microvascular injury in the brains of patients with Covid‐19. N Engl J Med. 2020;384:481‐483. 10.1056/NEJMc2033369 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Matschke J, Lütgehetmann M, Hagel C, et al. Neuropathology of patients with COVID‐19 in Germany: a post‐mortem case series. Lancet Neurol. 2020;19:919‐929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Niraula A, Sheridan JF, Godbout JP. Microglia priming with aging and stress. Neuropsychopharmacol Rev. 2017;42:318‐333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Bouayed J, Bohn T. Adapted sickness behavior—why it is not enough to limit the COVID‐19 spread? Brain Behav Immun. 2021;93:4‐5. 10.1016/j.bbi.2020.12.028 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Leisman DE, Ronner L, Pinotti R, et al. Cytokine elevation in severe and critical COVID‐19: a rapid systematic review, meta‐analysis, and comparison with other inflammatory syndromes. Lancet Respir Med. 2020;8(12):1233‐1244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Yanez ND, Weiss NS, Romand J‐A, Treggiari MM. COVID‐19 mortality risk for older men and women. BMC Public Health. 2020;20:1742. 10.1186/s12889-020-09826-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Hampshire A, Trender W, Chamberlain SR, et al. Cognitive deficits in people who have recovered from COVID‐19 relative to controls: an N = 84,285 online study. MedRxiv Preprint. 2020. 10.1101/2020.10.20.20215863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Maldonado‐Ruiz R, Montalvo‐Martínez L, Fuentes‐Mera L, Camacho A. Microglia activation due to obesity programs metabolic failure leading to type two diabetes. Nutr Diabetes. 2017;7:e254. 10.1038/nutd.2017.10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Zeberg H, Pääbo S. The major genetic risk factor for severe COVID‐19 is inherited from Neanderthals. Nature. 2020;587(7835):610‐612. [DOI] [PubMed] [Google Scholar]


