Abstract
Introduction
Patients with COVID‐19 frequently exhibit a hypercoagulable state with high thrombotic risk, particularly those admitted to intensive care units (ICU). Thromboprophylaxis is mandatory in these patients; nevertheless, thrombosis still occurs in many cases. Thus, the problem of assessing an adequate level of anticoagulation in ICU patients becomes evident during the COVID‐19 pandemic. The aim of this study was to evaluate the heparin resistance and the efficacy of heparin monitoring using an anti‐Xa activity assay.
Methods
Thirty‐seven heparin‐treated patients admitted to ICU for SARS‐CoV‐2 pneumonia were retrospectively studied for antifactor Xa activity (anti‐Xa), activated partial thromboplastin time (APTT), Antithrombin, Fibrinogen, D‐Dimer, Factor VIII, von Willebrand Factor, and the total daily amount of heparin administered. The correlation between APTT and anti‐Xa was evaluated for unfractionated heparins (UFH). The correlations between the daily dose of UFH or the dosage expressed as IU/kg b.w. for low molecular weight heparin (LMWH) and anti‐Xa were also evaluated.
Results
Twenty‐one patients received calcium heparin, 8 sodium heparin, and 8 LMWH. A moderate correlation was found between APTT and anti‐Xa for UFH. APTT did not correlate with coagulation parameters. 62% of UFH and 75% of LMWH treated patients were under the therapeutic range. About 75% of patients could be considered resistant to heparin.
Conclusions
SARS‐COV2 pneumonia patients in ICU have frequently heparin resistance. Anti‐Xa seems a more reliable method to monitor heparin treatment than APTT in acute patients, also because the assay is insensitive to the increased levels of fibrinogen, FVIII, and LAC that are common during the COVID‐19 inflammatory state.
Keywords: activated partial thromboplastin time, anticoagulant monitoring, anti‐Xa, COVID‐19, critical care, heparin resistance
1. INTRODUCTION
Patients with COVID‐19, especially those admitted to intensive care units (ICU), are at high thrombotic risk. 1 , 2 , 3 Thrombosis in patients with COVID‐19 is multifactorial and may result from a hypercoagulable state due to endothelial damage, hypoxic injury, cytokine release, inflammatory cell infiltration, and increased platelet activity. 4
Although without evidence from randomized controlled studies, some guidelines suggest to prevent thrombosis in COVID‐19 ICU patients by increasing the prophylactic anticoagulation dosage using either unfractionated (UFH) or low molecular weight (LMWH) heparins. 5 , 6 , 7 , 8 , 9 , 10 , 11 Despite such changes, a number of COVID‐19 patients on heparin regimens still experience thrombotic events, 1 , 12 , 13 , 14 and when the UFH dosage exceeds 35 000 units/day, a condition of heparin resistance can be defined. 15
Heparin resistance entails the need of huge UFH dosages to reach the therapeutic target. This occurs with UFH only, since LMWH displays a more predictable dose‐response relationship. 16 The factors that may favor heparin resistance in acute inflammatory states are antithrombin deficiency, elevated levels of factor VIII and/or fibrinogen, increased levels of heparin‐binding proteins, and increased heparin clearance. 16 , 17
It is still not clear which are the best tests to monitor and guide anticoagulation in acutely ill patients, especially when UFH is used in the presence of high levels of disease‐related procoagulant factors. 18 Moreover, falsely lowered APTT secondary to high level of factor VIII and fibrinogen may artificially indicate an apparent heparin resistance. 18 , 19
For the above‐mentioned reasons, anti‐Xa seems the more appropriate way to monitor heparin based anticoagulation regimens and to prevent a possible increased bleeding risk, especially when high dosages of heparin are administered. 7 , 17 , 20
The aim of our retrospective study was to evaluate the monitoring of anticoagulation with anti‐Xa activity in COVID‐19 ICU patients receiving heparin and the correlation between APTT and anti‐Xa in patients treated with UFH. The rate of heparin resistance during the ICU stay and its possible determining factors were also evaluated.
2. MATERIALS AND METHODS
We conducted a retrospective study using the data of 37 patients admitted for severe COVID‐19 pneumonia to two ICUs of ASST Ovest Milanese (Legnano and Magenta tertiary care general Hospitals, in northern Italy) from 30th March, to 4th June, 2020. All patients were treated with heparin—either UFH or LMWH—at intermediate or therapeutic dosages, depending on the established policies. As a rule, before COVID‐19 pandemics, ICUs used UFH at prophylactic dosage (ie, 0.2 U three times/day). After the COVID‐19 first outbreak, the high rate of thromboses prompted an increase of such dosage by ICU professionals, whereas the additional ICUs built up to tackle the large patient flow chose LMWH for the easier usage, at intermediate or therapeutic dosage. No ordinary prophylactic doses of heparin were used. We identified patients searching COVID‐19 subjects in whom anti‐Xa level was measured and plasma aliquots stocked frozen. We excluded patients with concomitant hematological disorders or cancer.
Demographic data, comorbidities, Sequential Organ Failure Assessment (SOFA) score, and PaO2/FiO2 ratio were evaluated at ICU admission. We retrieved the therapeutic schedules during the stay in ICU and the results of blood coagulation tests—as specified below—from the first hospital admission until the discharge from ICU.
Patients were treated with calcium UFH subcutaneously at intermediate (25 000 IU in two daily doses) and therapeutic dosages (up to 52 500 IU in maximum three daily doses). Sodium UFH was used intravenously for therapeutic purposes until the APTT target was reached. LMWH (Enoxaparin in any instance) was used at intermediate (4000 IU twice daily) and therapeutic (200 IU/Kg b.w. in two daily doses) dosages. The addition of anti‐Xa activity measurement in our routine was prompted after we evidenced the first thrombosis cases in patients with low APTT levels despite apparently adequate heparin treatment, and this policy was then extended to all ICU COVID‐19 patients.
The study protocol was approved by the Institutional Ethical Committee of Milano Area 3 and conducted in compliance with the Helsinki Declaration.
2.1. Blood collection and storage
Blood samples were collected in 2.7 mL evacuated blood tubes containing 3.2% (109 mmol/L) buffered sodium citrate, with a 1:9 ratio between anticoagulant and blood. The blood tubes were immediately centrifuged at 1500 g for 15 minutes at room temperature. The samples were tested for anti‐Xa, APTT, and AT, and two platelet‐poor plasma aliquots of at least 0.5 mL were stored in cryovials at −80°C for further analyses (ie, FVIII).
2.2. Laboratory testing
All tests were performed using the same analyzer (ACL TOP 750 CTS; Werfen) and using proprietary reagents (Instrumentation Laboratory; Werfen) as follows: Anti‐Xa activity, HemosIL Liquid anti‐Xa; PT, HemosIL RecombiPlasTin 2G; APTT, HemosIL SynthASil; D‐Dimer (DD), HemosIL HS‐DD; Antithrombin (AT), HemosIL Liquid Antithrombin, and Fibrinogen (FIB), HemosIL QFA Thrombin. The Factor VIII (FVIII) activity was measured by the clotting time one‐stage method using the HemosIL FVIII deficient plasma. Von Willebrand Antigen (HemosIL Von Willebrand Factor Antigen) and Willebrand Factor Ristocetin Cofactor activity (R:CoF) assay (HemosIL Von Willebrand Factor Ristocetin Cofactor Activity, latex assay with beads coated with wild‐type GPIb in the presence of ristocetin classifiable as VWF:GPIbR) was also performed.
The same lot of reagents was used for all analytical sessions throughout the study. All frozen samples not immediately tested were thawed at 37°C for 5 minutes in a water bath before being submitted to further analytical rounds.
Two internal quality control samples were run at the beginning of each analytical session.
All processed sample passed both the visual and instrumental preanalytical checkings.
2.3. Anticoagulation monitoring
According to standard practice, the monitoring scheme of intravenous sodium‐UFH treatment includes a blood sampling after 6 hours since the treatment onset and additional samplings at 4‐6 hours following any dose change. The monitoring of subcutaneous calcium‐UFH treatment is performed after 6 hours from the injection if administrations are set every 12 hours, or after 4 hours from injection if set every 8 hours. 21
To monitor LMWH administration, all blood samplings for anti‐Xa level were taken after 4 hours from injections administered every 12 hours. Samplings were taken after at least 2‐3 doses to ensure the achievement of a functional drug steady state, and when heparin levels are expected at their highest. 22
The expected heparin level range for UFH, as measured with the anti‐Xa, is 0.1‐0.2 UI/mL for prophylaxis and 0.3‐0.7 UI/mL for therapy. The expected heparin level range for LMWH is 0.2‐0.4 UI/mL for prophylaxis and 0.6‐1.0 UI/ mL for therapy. 16 , 22
We have taken into account just one anti‐Xa measurement for each patient in ICU, choosing the sampling taken at the maximum dosage of heparins.
2.4. Thrombotic events
The recorded thrombotic events included both venous (deep vein thrombosis [DVT], catheter‐related thrombosis [CRT], and pulmonary embolism [PE]) and arterial thrombosis (myocardial infarction [MI], ischemic stroke, and other systemic thromboembolism).
Ultrasonography was used to detect DVT or CRT as part of standard monitoring in our COVID‐19 ICU patients, while PE was diagnosed by computed tomography pulmonary angiography (CTPA) or echocardiography on the basis of clinical suspicion, hemodynamic parameters, and respiratory stability of the patient.
2.5. Definition of heparin resistance
Heparin resistance is defined as a daily dose in excess of 35 000 units/day for UFH in therapeutic regimens. 18 In addition, patients that received a total UFH dose >15 000 units/day (a prophylactic dosage) without reaching the minimum anti‐Xa level of 0.2 IU/mL were considered as resistant. However, since similar criteria are lacking for LMWH, we have arbitrarily defined as LMWH‐“resistant” those patients that did not reach the expected anti‐Xa ranges (Table 2).
TABLE 2.
Heparin resistance: Definition criteria
| Type of heparin | Heparin dose | Anti‐Xa (IU/mL) | |
|---|---|---|---|
| Sodium or calcium UFH | >35 000 IU/day* | AND | ≤0.7 |
| Calcium UFH | >15 000 IU/day | AND | ≤0.2 |
| LMWH | >200 IU/Kg b.w. | AND | ≤0.6 |
| LMWH | >4000 IU/day | AND | ≤0.2 |
Patients who exceeded the therapeutic heparin dosage without reaching the expected anti‐Xa level and those exceeding the prophylactic heparin dosage without reaching the anti‐Xa prophylactic range were considered resistant. Asterisk denotes criteria included in Ref. 18, while the other listed criteria are defined in‐house.
Abbreviations: b.w., body weight; LMWH, low molecular weight heparin; UFH, unfractioned heparin.
2.6. Statistical analysis
Data are presented as mean and standard deviation (±SD) or median and interquartile range [IQR] for continuous variables and percentage (%) or proportion (ratio) for categorical variables.
Normal distribution was assessed with Shapiro‐Wilk test. Ordinary least square regression after log transformation of both variables was performed. Data were analyzed using ibm spss software (version 20). P < .05 was considered statistically significant.
3. RESULTS
Baseline data are shown in Table 1. Patients were predominantly aged males, frequently overweight, with various comorbidities, mainly arterial hypertension and diabetes.
TABLE 1.
Baseline data at admission to Emergency Department
| Parameters | Values |
|---|---|
| Patients, n | 37 |
| Age, years – mean (SD) | 63 (8) |
| Gender, M/F (ratio) | 30/7 (4.3) |
| Pts with comorbidities, n/total n (%) | 25/37 (67.6) |
| Hypertension, n | 22 |
| Cardiovascular disease, n | 4 |
| Cerebrovascular disease, n | 2 |
| Diabetes, n | 6 |
| CKD, n | 1 |
| Respiratory disease, n | 4 |
| Single comorbidity, n | 15 |
| Multiple comorbidities (2 or more of the above‐listed), n | 10 |
| BMI, Kg/m2 – mean (SD) | 29.0 (5.1, 19‐40) |
| BMI ≥30, n (%) | 18 (48.6) |
|
PT ratio, median [IQR] a PT normal range: 0.8‐1.20 Ratio |
1.1 [1.1‐1.2] |
|
APTT ratio, median [IQR] a APTT normal range: 0.8‐1.20 Ratio |
1.0 [0.9‐1.0] |
|
D‐dimer, ng/mL – median [IQR] b D‐dimer normal range: <270 ng/mL DDU |
304 [136‐640] |
|
Platelets, cells × 103/µL‐ mean (SD) Platelets normal range: 140‐440x103 |
192 (66) |
Abbreviations: APTT, activated partial thromboplastin time; BMI, body mass index; CKD, chronic kidney disease; DDU, D‐dimer results using a D‐dimer unit; IQR, interquartile range; PT, prothrombin time; pts, patients; SD, standard deviation.
35 pts.
28 pts.
All patients had a normal APTT at admission to the emergency department (ED), except for one patient in Dabigatran therapy, and all had normal APTT prior to initiation of heparin therapy.
Prothrombin time ratio was abnormal in 10/37 (27.0%) patients in ED, and nobody was on antivitamin‐k antagonists. The D‐Dimer was higher than normal in 20/28 cases (71.4%).
At the time of sampling for this study, 21 patients were treated with calcium heparin (at a median 25 000 IU/day), eight with sodium heparin (at a median 48 000 IU/day), and eight with LMWH (at a median 10 000 IU/day).
A global moderate correlation (R 2 = .643, P < .001) was found between APTT and anti‐Xa level in the 29 patients on UFH (Figure 1). However, in this skewed distribution, a weaker correlation seems evident as much as the anti‐Xa level increases.
FIGURE 1.
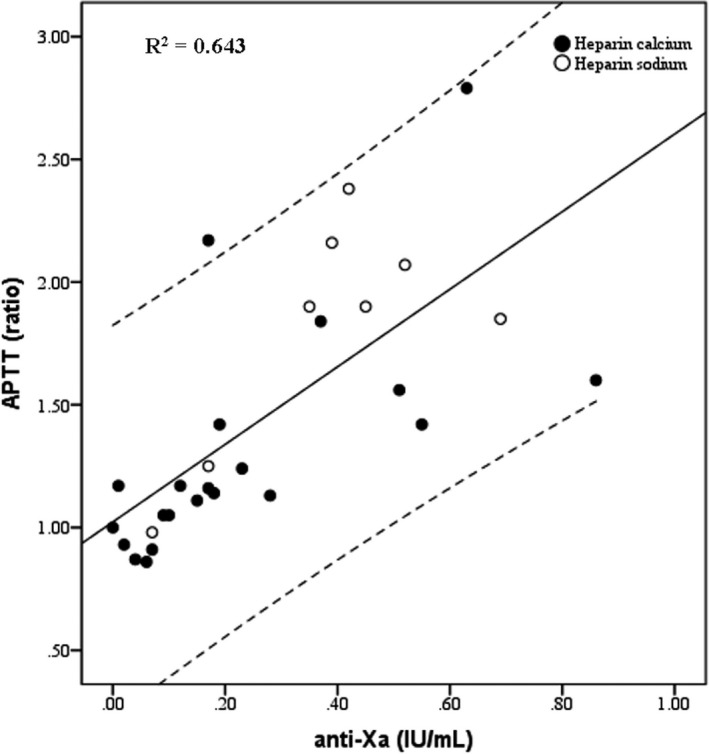
Correlation between activated partial thromboplastin time (APTT) and anti‐Xa heparin levels. Area between dashed lines denotes 95% CI
Looking at the UFH dosage and the consequent anti‐Xa level, 18/29 (62.1%) patients were under the therapeutic range (ie, anti‐Xa from 0.3 and 0.7 IU/mL). Concerning these 18 pts, 11 (61.1%) were treated with an intermediate dosage, from 15 000 to 35 000 IU/day (Figure 2, lower left quadrant) and 7 (38.9%) received a therapeutic dosage of >35 000 IU/day (Figure 2, upper left quadrant). Only 10 out of 29 patients (34.5%) were on therapeutic range (one was over‐range), and 9 of them received more than 35 000 IU/day (Figure 2, upper central quadrant). Overall, 16/29 (55.2%) patients were treated with >35 000 IU of UFH.
FIGURE 2.
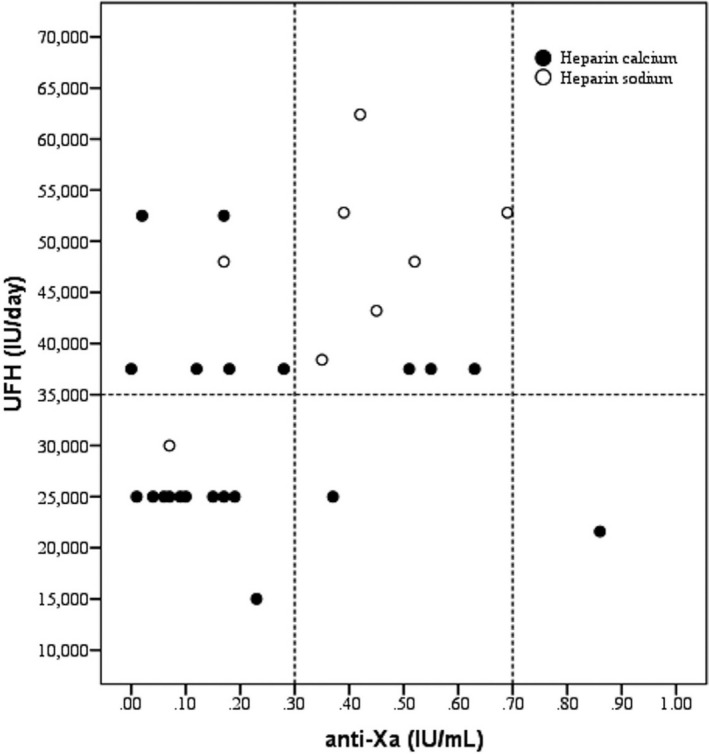
Relationship between anti‐Xa levels and amount of heparin in patients treated with unfractionated heparin. Vertical dashed lines denote the anti‐Xa therapeutic range. Horizontal dashed line defines the heparin resistance threshold. Abbreviations: UFH, unfractionated heparin
Among the eight patients receiving LMWH, six (75.0%) were underdosed despite receiving intermediate or high doses (ie, >200 IU/Kg b.w./day) (Figure S1).
According to the criteria defined in Table 2, 28/37 patients (75.7%) were considered as heparin “resistant.”
The coagulation parameters of patients collected during the ICU stay are summarized in Table 3. Antithrombin was above 54% in all patients, and no AT supplements were administered. FIB, DD, FVIII, and von Willebrand Factor (vWF) were largely above the upper level of the normal range in most patients. An overall significant increase of D‐dimer was observed from baseline to ICU (P = .027) with large individual variations. However, stratifying patients into heparin‐resistant and responsive, no significant differences were observed in DD values (P = .935).
TABLE 3.
Patients' data and coagulation parameters collected during the stay in ICU (SOFA score and PaO2/FiO2 ratio were collected at ICU admission)
| Parameter | Value in patients (N = 37) | Reference range |
|---|---|---|
| SOFA score – median [IQR] | 4 [3‐6] | — |
| PaO2/FiO2 ratio‐ median [IQR] | 140 [100‐160] | — |
| ICU LOS – media (SD) | 27 (17) | — |
| Anti‐Xa – median [IQR] | 0.28 [0.12‐0.45] | — |
| APTT, ratio – median [IQR] | 1.7 [1.0‐1.7] | 0.80‐1.20 |
| AT, % – mean (±SD) | 83 (17) | 80‐120 |
| Fibrinogen, mg/dL, mean (±SD) | 723 (262) | 180‐400 |
| D‐dimer, ng/mL DDU – median [IQR] | 788 [423‐1945] | <270 |
| FVIII, % – median [IQR] a | 213 [189‐253] | 50‐150 |
| vWF‐Ag, % – mean (±SD) a | 541 (184) | 42‐176 b |
| vWF‐Ricof, % – mean (±SD) a | 447 (151) | 49‐163 b |
All the coagulation parameters were measured in the same sample used for anti‐Xa level.
Abbreviations: APTT, activated partial thromboplastin time; AT, antithrombin; DDU, D‐dimer results using a D‐dimer unit; FVIII, Factor VIII; IQR, interquartile range; LOS, length of stay; PaO2/FiO2, arterial oxygen partial pressure /fractional inspired oxygen; SD, standard deviation; SOFA, Sequential Organ Failure Assessment Score; vWF‐Ag, von Willebrand Factor Antigen; vWF‐Ricof, von Willebrand Factor Ristocetin Activity.
36 patients.
Reference ranges: vWF‐Ag, 42%‐141% in blood group 0 patients and 66%‐176% in non‐0 blood group subjects; vWF‐Ricof, 40%‐126% in blood group 0 subjects and 49%‐163% in non‐0 blood groups.
No significant correlation was observed between APTT and FIB, FVIII, and vWF (data not shown).
Nineteen patients (51.3%) experienced thromboembolic events: five had PE, twelve had DVT, two had arterial thrombosis (one lower limb and one cerebellar ischemia), and five had central venous CRT. Five patients had more than one thrombotic event.
Two episodes of gastrointestinal bleeding were observed during the study. One patient taking UFH 25 000 IU/day, with normal APTT, anti‐Xa under range, and emergency viscoelastic tests indicating hypercoagulability had the rupture of esophageal varices; the other patient receiving LMWH at therapeutic dosage, with normal APTT and anti‐Xa in range, had duodenal ulcers that were treated endoscopically.
4. DISCUSSION
When UFH is used, the monitoring is usually performed by APTT, since it is a widely available and inexpensive parameter. However, the laboratory method used to evaluate APTT greatly influences the setting of therapeutic ranges, since a marked reagent‐to‐reagent variability is described. 23 APTT can be also influenced by increased levels of FVIII or FIB, that are frequently higher in critical patients, 18 and especially in Sars‐CoV‐2 infected subjects. 1
Conversely, monitoring UFH treatment with anti‐Xa takes advantage from a narrower reagent variability. This monitoring strategy is insensitive to the increases of FVIII and FIB 24 , 25 and is also unaffected by the presence of Lupus anticoagulant (LAC), a further possible complication in COVID‐19 patients with prolonged APTT. 1 , 26 However, anti‐Xa is a more expensive assay (about 4 times more than APTT in our country), it is not always available in ordinary laboratories, especially on the 24 hours, and ordering physicians are usually less familiar with anti‐Xa than with APTT.
The overall superiority of anti‐Xa over APTT in UFH therapeutic monitoring is, however, still controversial, 25 , 27 , 28 but the current evidence seems to indicate a better reliability of anti‐Xa for the clinical monitoring of critical patients. 29 , 30
A series of factors associated with a procoagulant state was demonstrable in our patient series, like increased DD, vWF, and FVIII, as reported in the literature. 1 , 31 , 32 The hypercoagulable multifactorial state that develops in acutely infected COVID‐19 patients has prompted the practice of thromboprophylaxis at heparin doses higher than usual 1 and therapeutic levels are used in some severe cases. However, a high rate of thrombosis with low anti‐Xa levels was observed. 12 , 13 , 14
A bedside routine screening using Doppler ultrasound of lower limbs and upper chest vessels may be of help for a timely diagnosis of thrombotic events and can lead to a prompt heparin dosage adjustment.
The correlation between anti‐Xa and APTT values in our patients receiving UFH was slightly better than that reported in the study by White et al 33 using the same reagents. However, the correlation found in our series was similar as compared to a study conducted before the Sars‐CoV‐2 epidemics. 29 In our and in the above‐mentioned studies, the relationship between the two parameters worsens when UFH is used at therapeutic dosages (Figure 1).
In our study cohort, patients receiving UFH who reach and steadily maintain therapeutic ranges of anti‐Xa are relatively few, and most of them require very high heparin dosages. Moreover, a sizable fraction of our patients fails to reach therapeutic ranges of anti‐Xa despite being treated with large amounts of heparin. Some patients on intermediate UFH dosages (ie, >15 000 IU and <35 000 IU) also show very low anti‐Xa levels (ie, <0.1 IU/mL) (Figure 2). The same seems to occur with LMWH also, which is surprising, since we expected a linear dose‐response behavior in our study group. The small patient cohort receiving LMWH may require more extensive observations to corroborate this preliminary finding (Figure S1).
A possible cause of heparin resistance is also antithrombin deficiency. Notably, all patients had levels of antithrombin (ie, >54%) that our ICU clinicians did not considered requiring therapeutic supplementation. To our knowledge, sound guidelines on moderate AT deficit levels indicating the need of replacement are lacking. Moreover, in critical patients, heparin itself can contribute to lower AT levels. Our anti‐Xa assay without the addition of exogenous antithrombin fully reflects the actual clinical condition in vivo. However, testings that include the addition of exogenous antithrombin may overestimate the anti‐Xa and result in a suboptimal heparin administration. 34
Acute‐phase proteins and the increased UFH clearance associated with the inflammatory COVID‐19 state surely play a role in generating heparin resistance. 25 In this complex scenario, we think that using anti‐Xa offers some advantages over APTT as a monitoring method to establish the net effect of heparin treatment, due to its relative insensitivity to acute‐phase proteins and LAC. According to a recent study, this approach can also be of help in reaching more rapidly the therapeutic range. 35 At the time of this writing, such a policy seemed successful in treating patients affected by the second epidemic wave of COVID‐19 (data not shown), which allowed the usage of lower doses of heparin, as described. 15
Limitations of the present study are its retrospective nature, the small patient cohort in a single hospital consortium, and the relatively small fraction of patients receiving LMWH.
CONFLICT OF INTEREST
Dr Chiara Novelli has received lecture fees and consultancy fees from Instrumentation Laboratory‐Werfen, outside the scope of the submitted work. The other authors have no competing interests.
Supporting information
Figure S1
ACKNOWLEDGEMENTS
We thank Dr Matteo Vidali for his expert guidance in data management and statistical analysis.
Novelli C, Borotto E, Beverina I, Punzi V, Radrizzani D, Brando B. Heparin dosage, level, and resistance in SARS‐CoV2 infected patients in intensive care unit. Int J Lab Hematol. 2021;43:1284–1290. 10.1111/ijlh.13543
DATA AVAILABILITY STATEMENT
Data available on request due to privacy/ethical restrictions.
REFERENCES
- 1. Helms J, Tacquard C, Severac F, et al. High risk of thrombosis in patients with severe SARS‐CoV‐2 infection: a multicenter prospective cohort study. Intensive Care Med. 2020;46:1089‐1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Cui S, Chen S, Li X, et al. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J Thromb Haemost. 2020;18:1421‐1424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Klok FA, Kruip MJHA, van Der Meer NJM, et al. Incidence of thrombotic complications in critically ill ICU patients with COVID‐19. Thromb Res. 2020;191:145‐147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Connors JM, Levy JH. Thromboinflammation and the hypercoagulability of COVID‐19. J Thromb Haemost. 2020;18(7):1559‐1561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Cohoon KP, Mahè G, Tafur AJ, et al. Emergence of institutional antithrombotic protocols for coronavirus 2019. Res Pract Thromb Haemost. 2020;4:510‐517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Susen S, Tacquard CA, Godon A, et al. Prevention of thrombotic risk in hospitalized patients with COVID‐19 and hemostasis monitoring. Crit Care. 2020;24:364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Barnes GD, Burnett A, Allen A, et al. Thromboembolism and anticoagulant therapy during the COVID‐19 pandemic: interim clinical guidance from the anticoagulation forum”. J Thromb Thrombolysis. 2020;50:72‐81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Spyropoulos AC, Levy JH, Ageno W, et al. Scientific and standardization committee communication: clinical guidance on the diagnosis, prevention and treatment of venous thromboembolism in hospitalized patients with COVID‐19. J Thromb. 2020;18:1859‐1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Lemos ACB, Espirito Santo DA, Salvetti MC, et al. Therapeutic versus prophylactic anticoagulation for severe COVID‐19: a randomized phase II clinical trial (HESACOVID). Thromb Res. 2020;196:359‐366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Nadkarni GN, Lala A, Bagiella E, et al. Anticoagulation, bleeding, mortality, and pathology in hospitalized patients with COVID‐19. J Am Coll Cardiol. 2020;76(16):1815‐1826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Shi C, Tingting W, Li JP, et al. Comprehensive landscape of heparin therapy for COVID‐19. Carbohyd Polym. 2021;254:e117232. 10.1016/j.carbpol.2020.117232 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Lodigiani C, Iapichino G, Carenzo L, et al. Venous and arterial thromboembolic complications in COVID‐19 patients admitted to an academic hospital in Milan. Italy. Thromb Res. 2020;191:9‐14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Middeldorp S, Coppens M, van Haaps TF, et al. Incidence of venous thromboembolism in hospitalized patients with COVID‐19. J Thromb Haemost. 2020;18:1995‐2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Llitjos JF, Leclerc M, Chochois C, et al. High incidence of venous thromboembolic events in anticoagulated severe COVID‐19 patients. J Thromb Haemost. 2020;18:1743‐1746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Levine MN, Hirsh J, Gent M, et al. A randomized trial comparing activated thromboplastin time with heparin assay in patients with acute venous thromboembolism requiring large daily doses of heparin. Arch Intern Med. 1994;154(1):49‐56. [PubMed] [Google Scholar]
- 16. Garcia DA, Baglin TP, Weitz JI, et al. Parenteral anticoagulants. Chest. 2012;141(2S):E24‐E43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Moores LK, Tritschler T, Brosnahan S, et al. Prevention, diagnosis, and treatment of VTE in patients with COVID‐19. Chest. 2020;158(3):1143‐1163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Smythe MA, Priziola J, Dobesh PP, et al. Guidance for the practical management of the heparin anticoagulants in the treatment of venous thromboembolism. J Thromb Thrombolysis. 2016;41:165‐186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Mitsuguro M, Okamoto A, Shironouchi Y, et al. Effects of factor VIII levels on the APTT and anti‐Xa activity under a therapeutic dose of heparin. Int J Hematol. 2015;101:119‐125. [DOI] [PubMed] [Google Scholar]
- 20. Connors JM, Levy JH. COVID‐19 and its implications for thrombosis and anticoagulation. Blood. 2020;135(23):2033‐2040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Jourdi G, Mansour A, Vayne C, et al. Anticoagulation therapy in France: state‐of‐the‐art in 2020. Ann Blood. 2020;5(3):1‐14. [Google Scholar]
- 22. Kearon C, Kahn SR, Agnelli G, et al. Antithrombotic therapy for venous thromboembolic disease. Chest. 2008;133(6S):454S‐545S. [DOI] [PubMed] [Google Scholar]
- 23. Kitchen S, Jennings I, Woods TAL, et al. Wide variability in the sensitivity of APTT reagents for monitoring of heparin dosage. J Clin Pathol. 1996;49:10‐14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Lehman CM, Frank EL. Laboratory monitoring of heparin therapy: partial thromboplastin time or anti‐Xa assay? Lab Med. 2009;40(1):47‐51. [Google Scholar]
- 25. Uprichard J, Manning RA, Laffan MA. Monitoring heparin anticoagulation in the acute phase response: heparin anticoagulation in the acute phase response. Br J Haematol. 2010;149(4):613‐619. [DOI] [PubMed] [Google Scholar]
- 26. Bowles L, Platton S, Yartey N, et al. Lupus anticoagulant and abnormal coagulation tests in patients with Covid‐19. N Engl J Med. 2020;383:288‐290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. McLaughlin K, Rimsans J, Sylvester KW, et al. Evaluation of antifactor‐Xa heparin assay and activated partial thromboplastin time values in patients on therapeutic continuous infusion unfractionated heparin therapy. Clin Appl Thromb Hemost. 2019;25:1‐6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Whitman‐Purves E, Coons JC, Miller T, et al. Performance of anti‐factor Xa versus activated partial thromboplastin time for heparin monitoring using multiple nomograms. Clin Appl Thromb Hemost. 2018;24(2):310‐316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Cuker A, Ptashkin B, Konkle BA, et al. Interlaboratory agreement in the monitoring of unfractionated heparin using the anti‐factor Xa‐correlated activated partial thromboplastin time. J Thromb Haemost. 2008;7:80‐86. [DOI] [PubMed] [Google Scholar]
- 30. Van Roessel S, Middeldorp S, Cheung YW, et al. Accuracy of aPTT monitoring in critically ill patients treated with unfractionated heparin. Neth J Med. 2014;72(6):305‐310. [PubMed] [Google Scholar]
- 31. O'Sullivan JM, Mc Gonagle D, Ward SE, et al. Endothelial cells orchestrate COVID‐19 coagulopathy. Lancet Haematol. 2020;7(8):E553‐E555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. White D, MacDonald S, Bulll T, et al. Heparin resistance in COVID‐19 patients in the intensive care unit. J Thromb Thrombolysis. 2020;50:287‐291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Bell R, Zini G, d'Onofrio G, et al. The hematology laboratory's response to the COVID‐19 pandemic: a scoping review. Int JLabHematol. 2020;00:1‐12. [DOI] [PubMed] [Google Scholar]
- 34. Croles FN, Lukens MV, Mulder R, et al. Monitoring of heparins in antithrombin‐deficient patients. Thromb Res. 2019;175:8‐12. [DOI] [PubMed] [Google Scholar]
- 35. Beun R, Kusadasi N, Sikma M, et al. Thromboembolic events and apparent heparin resistance in patients infected with SARS‐CoV‐2. Int JLabHematol. 2020;42(Suppl 1):19‐20. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1
Data Availability Statement
Data available on request due to privacy/ethical restrictions.


