Abstract
This case report of a 38-year-old man with bilateral Best vitelliform macular dystrophy (BVMD) presents bilateral quiescent type 1 neovascularizations (NV) detected by optical coherence angiography (OCTA) and their multimodal imaging characteristics. It was emphasized that this kind of quiescent and asymptomatic NV may be present in nearly every stage of BVMD and it was concluded that OCTA is a noninvasive, easy, and rapid method that is superior to other imaging methods in detecting them.
Keywords: Best vitelliform macular dystrophy, optical coherence tomography angiography, type 1 neovascularization
Introduction
Best vitelliform macular dystrophy (BVMD), or Best disease, is a bilateral hereditary disease that has autosomal dominant inheritance and is characterized by a typical “egg-yolk” appearance in the macula. The disease emerges in childhood and young adulthood and has 5 stages based on clinical appearance.1,2 It generally progresses slowly, with a decrease in visual acuity that is minimal and inconsistent with the fundus appearance in the early stages. There is often asymmetry in visual acuity between the eyes, and central scotomas and metamorphopsia appear at later stages.3,4
Reports indicate that sudden vision loss during the natural course of BVMD is caused by secondary neovascularization (NV) and is a relatively common occurrence.3,4,5,6,7 Optical coherence tomography angiography (OCTA) is a fairly new, rapid, and noninvasive imaging method currently used to investigate the presence of all types of NV and polypoidal choroidal vasculopathy (PCV) in the retinal and choroidal layers. In addition, it has been reported in various studies that OCTA may be useful in various retinal diseases for the early detection of inactive and quiescent NV structures that do not yet show exudative symptoms.8
To the best of our knowledge, this article is the first to present asymptomatic and quiescent type 1 NV lesions detected by OCTA and their multimodal imaging features in both eyes of a patient diagnosed with bilateral BVMD.
Case Report
A 38-year-old man presented to our clinic with complaints of low vision in both eyes. He reported that he had been diagnosed with BVMD 10 years earlier, his visual acuity had been stable for a long time, and his brother also had the same disease.
On examination, his best corrected visual acuity was 7/10 in the right and 5/10 in the left eye, intraocular pressures were 12 mmHg bilaterally, and anterior segment examination was normal. Biomicroscopic fundus examination revealed bilateral round, well-defined lesions 2-3 disc diameters in size containing yellowish subretinal material. It was observed that the subretinal material in the right eye had descended to the inferior part of the lesion to form a pronounced level (pseudohypopyon, stage 3), while in the left eye most of the material had been resorbed and had a scrambled egg appearance (vitelliruptive, stage 4). In addition, the presence of a round, gray nodular formation surrounded by a dark halo just below the macular center was also noted in the left eye.
Color photographs (Topcon 3D-OCT 2000 Corporation, Tokyo, Japan) and infrared photographs were obtained, and fundus autofluorescence (FAF) images (Heidelberg Spectralis HRA + OCT, Heidelberg, Germany) revealed hyperautofluorescent subretinal fluid in both eyes (Figure 1a-c and 2a-c), while spectral domain optical coherence tomography (SD-OCT) images acquired with the same device also clearly demonstrated subretinal fluid under the macula in both eyes and the presence of vitelliform material forming a level that was less pronounced in the left eye. In addition to these findings, SD-OCT sections passing through the nodular structure in the left eye showed that this lesion appeared to be a slightly irregular, pointed, hyperreflective pigment epithelial detachment (PED) containing moderately reflective material (Figures 1d-f and 2d-f).
Figure 1.
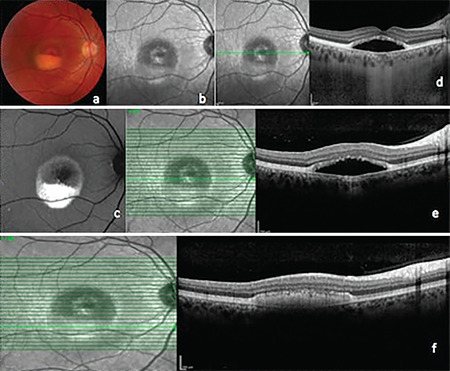
Right eye. a) Color fundus photograph, pseudohypopyon stage; b) Infrared photograph; c) Fundus autofluorescence image; d) Enhanced depth imaging optical coherence tomography (EDI-OCT); e, f) Spectral domain OCT
Figure 2.
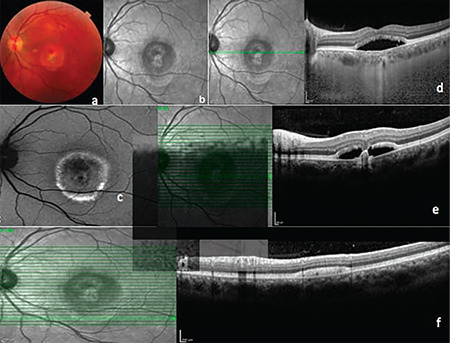
Left eye. a) Color fundus photography, vitelliruptive stage; b) Infrared photograph; c) Fundus autofluorescence image; d) Enhanced depth imaging optical coherence tomography (EDI-OCT); e, f) Spectral domain OCT
OCTA imaging (RTVue-XR Avanti OCT system, Optovue, Fremont, CA) revealed vascular networks originating from an NV structure at the choriocapillary level in both eyes (forming a ring in the left eye) and cross-sectional OCTA images clearly showed increased flow signals in these regions (Figure 3a, b and 4a, b). The patient underwent fluorescein angiography (FA) and indocyanine green angiography (ICGA) (Heidelberg Spectralis HRA + OCT, Heidelberg, Germany) to determine the activity of the NV lesions and investigate the possibility that the nodular lesion was PCV. Although FA examination demonstrated irregular hyperfluorescent staining patterns in both maculas, there was no dye leakage until the late phases (Figure 3c, d and 4c, d. On ICGA, there were hypercyanotic areas of punctate staining around the macula of both eyes starting in the early phases, although no hot-spot or plaque-style staining pattern was observed in the late phases. The nodular structure in the left eye was significantly hypocyanotic in the early phases of angiography, after which it showed a hypercyanotic character starting with mild staining at minute 13 and continuing to the end of angiography (Figure 3e, f and 4e, f).
Figure 3.
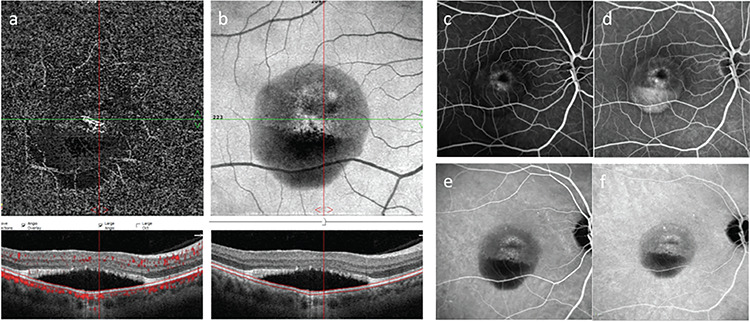
Right eye. a) En face optical coherence tomography angiography (OCTA); b) Structural OCTA; c, d) Fluorescein angiography early and late phase; e, f) Indocyanine green angiography, early and late phase
Figure 4.
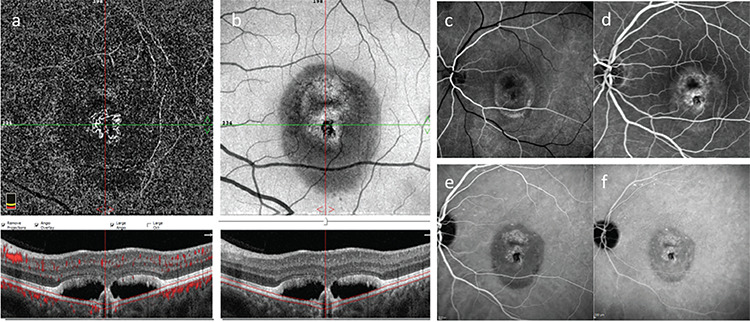
Left eye. a) En face optical coherence tomography angiography (OCTA); b) Structural OCTA; c, d) Fluorescein angiography early and late phase; e, f) Indocyanine green angiography, early and late phase
Discussion
In this article, we present asymptomatic and quiescent type 1 NV lesions detected only by OCTA in both eyes of a patient diagnosed with bilateral BVMD and their characteristics on multimodal imaging with OCTA as well as color and infrared photography, FAF, SD-OCT, FA, and ICGA. To the best of our knowledge, this article is the first to describe asymptomatic and quiescent type 1 NV and its multimodal imaging features in a patient with BVMD.
Several studies published in recent years have reported that quiescent NV lesions can be found in many retinal diseases, especially age-related macular degeneration, and that they may remain asymptomatic for years.8,9 Histologically, NVs are divided into three types: type 1 NV, which is located under the retinal pigment epithelium (RPE) (angiographically occult); type 2, which originates from the choroid and passes through Bruch’s membrane and RPE to extend into the subretinal space (angiographically classic); and type 3, which develops within the neurosensory retina (retinal angiomatous proliferation).10 In our case, the NV lesion was seen at the choriocapillaris level on OCTA and was therefore classified as type 1. NV lesions in both eyes were considered inactive (quiescent) due to the absence of macular hemorrhage on clinical examination, no findings of subretinal, intraretinal, or sub-RPE fluid on SD-OCT, and no leakage in late FA.11,12 The absence of fibrosis on clinical examination and imaging ruled out the possibility of scar tissue. The fact that the patient’s eyes had very similar visual acuity, as well as being compatible with stage 3 and 4 BVMD and the patient having no complaints of a sudden decrease in vision also suggested that the NV was asymptomatic.
Using OCTA, Batıoğlu et al.3 detected NV networks with associated polypoidal dilations at the choriocapillaris level in both eyes of a pregnant BVMD patient with complaints of reduced vision, and described the case as pachychoroid neovasculopathy. In our patient, a nodular formation was detected just below the macular center in the left eye and resembled a pointed PED on SD-OCT. We thought that it may be associated with a polypoidal structure and performed ICGA, but this diagnosis was discounted because its staining properties on ICGA were not typical for PCV.
There are many articles in the literature describing secondary NV lesions and their OCTA imaging features in eyes with BVMD. Patel et al.5 used OCTA to examine eyes with 4 different retinal dystrophies and secondary NV development and emphasized that the morphological structure of the NV lesions could be clearly demonstrated on OCTA despite distortion of the retinal anatomy. Shahzad and Siddiqui6 were able to visualize type 2 NV lesions secondary to BVMD with OCTA in one eye of a pediatric patient presenting with a sudden decline in vision. Guduru et al.4 examined the vascular structure of the retina with OCTA and FA in 19 eyes with secondary NV. They reported that OCTA was superior to FA in measuring NV and that the ring-shaped NV pattern was rare. Stattin et al.7 presented the features of multimodal imaging using FAF, SD-OCT, FA, ICGA, and OCTA in a BVMD patient with metamorphopsia and sudden decline in vision in one eye. Researchers have detected NV lesions at the outer retinal and choriocapillary levels with OCTA while emphasizing that conventional angiography techniques such as FA and ICGA were inconclusive. In our case, we were only able to visualize the presence of a quiescent type 1 NV lesion at the choriocapillary level in both eyes with OCTA.
In summary, we concluded that OCTA is a noninvasive, easy, rapid, and reliable imaging method superior to other modalities for detecting secondary NV lesions in eyes with BVMD, even if quiescent and asymptomatic.
Footnotes
Ethics
Informed Consent: Obtained.
Peer-review: Externally peer reviewed.
Authorship Contributions
Surgical and Medical Practices: J.M., Concept: J.M., Design: J.M., Data Collection or Processing: M.E.B., Analysis or Interpretation: J.M., M.E.B., Literature Search: M.E.B., Writing: J.M.
Conflict of Interest: No conflict of interest was declared by the authors.
Financial Disclosure: The authors declared that this study received no financial support.
References
- 1.Parodi MB, Iacono P, Campa C, Turco CD, Bandello F. Fundus autofluorescence patterns in Best vitelliform macular dystrophy. Am J Ophthalmol. 2014;158:1086–1092. doi: 10.1016/j.ajo.2014.07.026. [DOI] [PubMed] [Google Scholar]
- 2.Qian CX, Charran D, Strong CR, Steffens TJ, Jayasundera T, Heckenlively JR. Optical coherence tomography examination of the retinal pigment epithelium in Best vitelliform macular dystrophy. Ophthalmology. 2017;124:456–463. doi: 10.1016/j.ophtha.2016.11.022. [DOI] [PubMed] [Google Scholar]
- 3.Batıoğlu F, Yanık Ö, Demirel S, Çağlar Ç, Özmert E. Pakikoroid neovaskülopatinin eşlik ettiği bir Best hastalığı olgusu. Turk J Oftalmol. 2019;49:226–229. [Google Scholar]
- 4.Guduru A, Gupta A, Tyagi M, Jalali S, Chhablani J. Optical coherence tomography angiography characterisation of Best disease and associated choroidal neovascularisation. Br J Ophthalmol. 2018;102:444–447. doi: 10.1136/bjophthalmol-2017-310586. [DOI] [PubMed] [Google Scholar]
- 5.Patel RC, Gao SS, Zhang M, Alabduljalil T, Al-Qahtani A, Weleber RG, Yang P, Jia Y, Huang D, Pennesi ME. Optical coherence tomography angiography of choroidal neovascularization in four inherited retinal dystrophies. Retina. 2016;36:2339–2347. doi: 10.1097/IAE.0000000000001159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shahzad R, Siddiqui MAR. Choroidal neovascularization secondary to Best vitelliform macular dystrophy detected by optical coherence tomography angiography. J AAPOS. 2017;1:68–70. doi: 10.1016/j.jaapos.2016.08.018. [DOI] [PubMed] [Google Scholar]
- 7.Stattin M, Ahmed D, Glittenberg C, Krebs I, Ansari-Shahrezaei S. Optical coherence tomography angiography for detection of secondary choroidal neovascularization in vitelliform macular dystrophy. Retin Cases Brief Rep. 2020;14:49–52. doi: 10.1097/ICB.0000000000000626. [DOI] [PubMed] [Google Scholar]
- 8.Menteş J, Karaca I, Sermet F. Multimodal imaging characteristics of quiescent type 1 neovascularization in an eye with angioid streaks. Am J Ophthalmol Case Rep. 2018;10:132–136. doi: 10.1016/j.ajoc.2018.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Menteş J, Yıldırım Ş. Noneksudatif tip yaşa bağlı maküla dejeneresanslı gözlerde sessiz tip 1 neovaskülarizasyonların spektral domain optik koherens tomografi özellikleri. Turk J Oftalmol. 2019;49:84–88. [Google Scholar]
- 10.Spaide RF, Jaffe GJ, Sarraf D, Freund KB, Sadda SR, Staurenghi G, Waheed NK, Chakravarthy U, Rosenfeld PJ, Holz FG, Souied EH, Cohen SY, Querques G, Ohno-Matsui K, Boyer D, Gaudric A, Blodi B, Baumal CR, Li X, Coscas GJ, Brucker A, Singerman L, Luthert P, Schmitz-Valckenberg S, Schmidt-Erfurth U, Grossniklaus HE, Wilson DJ, Guymer R, Yannuzzi LA, Chew EY, Csaky K, Monés JM, Pauleikhoff D, Tadayoni R, Fujimoto J. Consensus Nomenclature for Reporting Neovascular Age-Related Macular Degeneration Data: Consensus on Neovascular Age-Related Macular Degeneration Nomenclature Study Group. Ophthalmology. 2020;127:616–636. doi: 10.1016/j.ophtha.2019.11.004. [DOI] [PubMed] [Google Scholar]
- 11.Querques G, Srour M, Massamba N, Georges A, Ben Moussa N, Rafaeli O, Souied EH. Functional characterization and multimodal imaging of treatment-naive “quiescent” choroidal neovascularization. Invest Ophthalmol Vis Sci. 2013;54:6886–6892. doi: 10.1167/iovs.13-11665. [DOI] [PubMed] [Google Scholar]
- 12.Querques G, Srour M, Massamba N, Georges A, Ben Moussa N, Rafaeli O, Souied EH. Functional characterization and multimodal imaging of treatment-naive “quiescent” choroidal neovascularization. Invest Ophthalmol Vis Sci. 2013;54:6886–6892. doi: 10.1167/iovs.13-11665. [DOI] [PubMed] [Google Scholar]


