Abstract
Electronic cigarettes (e-cigarettes) were introduced in the United States in 2007 and by 2014 they were the most popular tobacco product amongst youth and had overtaken use of regular tobacco cigarettes. E-cigarettes are used to aerosolize a liquid (e-liquid) that the user inhales. Flavorings in e-liquids is a primary reason for youth to initiate use of e-cigarettes. Evidence is growing in the scientific literature that inhalation of some flavorings is not without risk of harm. In this review, 67 original articles (primarily cellular in vitro) on the toxicity of flavored e-liquids were identified in the PubMed and Scopus databases and evaluated critically. At least 65 individual flavoring ingredients in e-liquids or aerosols from e-cigarettes induced toxicity in the respiratory tract, cardiovascular and circulatory systems, skeletal system, and skin. Cinnamaldehyde was most frequently reported to be cytotoxic, followed by vanillin, menthol, ethyl maltol, ethyl vanillin, benzaldehyde and linalool. Additionally, modern e-cigarettes can be modified to aerosolize cannabis as dried plant material or a concentrated extract. The U.S. experienced an outbreak of lung injuries, termed e-cigarette, or vaping, product use-associated lung injury (EVALI) that began in 2019; among 2,022 hospitalized patients who had data on substance use (as of January 14, 2020), 82% reported using a delta-9-tetrahydrocannabinol (main psychoactive component in cannabis) containing e-cigarette, or vaping, product. Our literature search identified 33 articles related to EVALI. Vitamin E acetate, a diluent and thickening agent in cannabis-based products, was strongly linked to the EVALI outbreak in epidemiologic and laboratory studies; however, e-liquid chemistry is highly complex, and more than one mechanism of lung injury, ingredient, or thermal breakdown product may be responsible for toxicity. More research is needed, particularly with regard to e-cigarettes (generation, power settings, etc.), e-liquids (composition, bulk or vaped form), modeled systems (cell type, culture type, and dosimetry metrics), biological monitoring, secondhand exposures and contact with residues that contain nicotine and flavorings, and causative agents and mechanisms of EVALI toxicity.
Keywords: e-cigarettes, Vaporizers, Flavorings, e-liquids, Delta-9-tetrahydrocannabinol (Δ9-THC), Toxicity
1. Summary
Electronic nicotine delivery systems such as electronic cigarettes (e-cigarettes) are used to heat an e-liquid composed of humectants and sometimes flavorings and nicotine. The heated e-liquids forms an aerosol (mixture of liquid droplets and gas phase substances) that is inhaled by the user. Some e-cigarette users inhale this aerosol to mimic tobacco smoking without tobacco combustion. Electronic devices intended for cannabis such as personal vaporizers are used to heat the dried plant material or its extracts to deliver aerosolized cannabinoids in a form that can be inhaled without combustion. Additionally, special interchangeable coil head adapters have enabled the use of e-cigarettes to aerosolize cannabis as dried plant material or a concentrated extract, either by itself or dissolved in an e-liquid. Since the introduction of e-cigarettes in the United States in 2007, these devices rapidly gained popularity, especially amongst U.S. youth, and by 2014, were the most popular tobacco product for this demographic, overtaking use of regular tobacco cigarettes. The rise in popularity of e-cigarettes has raised important public health questions, including: 1) given the attraction of flavorings on e-cigarette use, what is our understanding of the toxicity of flavored e-liquids?, and 2) given the ease in which e-cigarettes can be used to aerosolize substances, what is our understanding of the toxicity of substances underlying the outbreak of lung injury related to consumption of cannabis and nicotine products that occurred predominantly in the United States termed e-cigarette, or vaping, product use-associated lung injury (EVALI)? With regard to the first question, many flavorings used in e-liquids fall under the “generally recognized as safe” (GRAS) provision in the Federal Food, Drug, and Cosmetic Act of the U.S. Food and Drug Administration; however, their GRAS status applies only to use in ingested foods, not for exposure via the inhalation pathway. In surveillance studies, flavorings were cited by youth as a primary reason for use of e-cigarettes. As such, there is growing concern about toxicity from inhalation of aerosolized flavorings in e-liquids and whether e-cigarettes pose a risk for dependence or addiction to nicotine for a new generation of youth. To better understand the state of knowledge on the toxicity of flavored e-liquids, we reviewed literature in the PubMed and Scopus databases and identified 67 original articles that evaluated toxicity of flavored e-liquids using cellular in vitro, rodent in vivo, and human models. Whether as bulk liquid or aerosol from an e-cigarette, some flavored e-liquids induced toxicity in the respiratory tract (cytotoxicity, generation of reactive oxygen species, and impairment of clearance mechanisms), cardiovascular and circulatory systems (impaired nitric oxide (NO) signaling and other effects related to endothelial dysfunction), skeletal system (altered gene expression in osteoblasts, toxicity in the oral cavity), and skin (cytotoxicity). In general, embryonic cells were more sensitive to flavored e-liquids compared with adult cells, which indicated a possible indirect pathway for developmental effects. Additionally, some flavorings in e-liquids were immune sensitizers, irritants, or genotoxic. At least 65 individual flavoring ingredients in flavored e-liquids were observed to contribute to reported toxic effects. Cinnamaldehyde was most frequently reported to be cytotoxic, followed by vanillin, menthol, ethyl maltol, ethyl vanillin, benzaldehyde and linalool. With regards to our second question, among EVALI patients, 82% reported using a delta-9-tetrahydrocannabinol (Δ9-THC, the main psychoactive component in cannabis) containing e-cigarette, or vaping, product, 33% reported only using a Δ9-THC-containing product, 57% reported using any nicotine-containing product, and 14% used only a nicotine-containing product. Our literature search identified 33 articles of interest related to EVALI. Five of the 33 articles of interest on EVALI contained information on in vitro or in vivo pulmonary toxicity. Vitamin E acetate (VEA), a diluent and thickening agent in cannabis-based products, is strongly linked to the EVALI outbreak in epidemiologic and laboratory studies, and VEA has been found to produce a similar syndrome in mice. However, e-liquid chemistry is highly complex, and more than one mechanism of lung injury, ingredient, or thermal breakdown product may be responsible for toxicity. From our review, a total of 13 research gaps and opportunities were identified related to considerations for e-cigarettes (generation, power settings, etc.), e-liquids (composition, bulk or vaped form), modeled systems (cell type, culture type, and dosimetry metrics), biological monitoring, secondhand exposures and contact with residues that contain nicotine and flavorings, and causative agents and mechanisms of EVALI toxicity.
2. Introduction
Electronic delivery systems are devices that heat a liquid solution (e-liquid) or dry material to volatilize its constituents, which are inhaled by the user in the form of an aerosol. Devices intended for nicotine delivery are referred to as electronic nicotine delivery systems (ENDS) and include electronic cigarettes (e-cigarettes or e-cigs), e-cigars, e-pipes, and e-hookahs. Early versions of e-cigarettes were intended to mimic the tobacco smoking experience but without tobacco combustion (Grana, Benowitz, & Glantz, 2014); newer generations of e-cigarettes no longer mimic the smoking experience (e.g., the size, shape, and design does not mimic regular tobacco cigarettes). The e-liquid for e-cigarettes contains humectants and usually nicotine and flavorings. When heated in an e-cigarette, the e-liquid is volatilized and forms a mixture of liquid droplets and gas-phase compounds in air. Though technically an aerosol, the mixture that is inhaled by the user is colloquially referred to as “vapor” and the experience is termed “vaping.” In 2016, the U.S. Food and Drug Administration (FDA), under authority of the Family Smoking Prevention and Tobacco Control Act, began to regulate e-cigarettes as tobacco products, which includes the use of flavors in products. Many flavorings used in e-liquids for e-cigarettes fall under the “generally recognized as safe” (GRAS) provision in the Federal Food, Drug, and Cosmetic Act under the jurisdiction of the FDA. Under FDA regulation, any substance that will be added to food is subject to premarket approval, unless it is generally recognized, by scientific experts, as safe under the conditions of its intended use. FDA determines the safety of the substance if it is subject to premarket approval whereas qualified experts outside of government can submit a GRAS notification to the FDA for their approval. The Flavor and Extract Manufacturers Association of the United States maintains a program of expert reviewers for GRAS status of flavorings to be used in foods (as defined in Section 201(f) of the Act). Flavor and Extract Manufacturers Association nominations of flavorings for GRAS status only assesses safety for exposure through ingestion. Approval of their nominations by FDA does not provide regulatory authority for the use of a flavoring in e-liquids where exposure is via inhalation from vaping. The use of flavorings with GRAS status in e-liquids has raised concern by public health experts and the Flavor and Extract Manufacturers Association alike of possible toxicity (FEMA, 2020). Herein, when discussing e-liquids, the terms “flavor” or “flavored” refer to a taste sensation (e.g., fruity) of an e-liquid and the term “flavoring” refers to the specific chemical that imparts a taste (e.g., α-ionone is a flavoring for raspberry). When discussing flavored e-liquids, general flavor categories are in lowercase (e.g., fruity) whereas names of commercial products are capitalized (e.g., Mango).
“Vaporizers” are devices used for drug delivery via inhalation (Giroud et al., 2015; Meier & Hatsukami, 2016; Varlet et al., 2016). Cannabis plant is typically referred to as “marijuana” when the plant contains more than 0.3% (by dry weight) of delta-9-tetrahydrocannabinol (Δ9-THC), the main psychoactive cannabinoid, and it is referred to as “hemp” when the plant contains less 0.3% of Δ9-THC. Though hemp has lower Δ9-THC content, it will often have a higher concentration of cannabidiol (CBD). Vaporizers include table top devices to heat dried cannabis without combustion at moderate temperatures to create an aerosol that is inhaled and or portable, pocket pen-vaporizers to heat its wax and oil extracts at higher temperatures to deliver aerosolized cannabinoids in a form that can be inhaled (Giroud et al., 2015; Varlet et al., 2016). In addition to electronic products specifically designed for cannabis, other devices, including e-cigarettes that are designed to deliver nicotine, can be modified to deliver various substances. For example, e-cigarettes can be used after-market to inhale alcohol, cannabis, amphetamines, cocaine, and heroin (Breitbarth, Morgan, & Jones, 2018; Giroud et al., 2015; MacLean, Valentine, Jatlow, & Sofuoglu, 2017; Meier & Hatsukami, 2016). For cannabis extracts, the e-liquid contains the drug, thickening agents, diluents/thinning agents, and may be flavored based on user preference or to mask the odor of cannabis and make it less detectable (Blount et al., 2020; Giroud et al., 2015). In 2017, He et al. first reported a case of acute respiratory failure in a person that inhaled aerosolized cannabis oil (He, Oks, Esposito, Steinberg, & Makaryus, 2017). In late 2019, Schier et al. first reported an outbreak of lung injuries in the United States later termed e-cigarette, or vaping, product use-associated lung injury (EVALI) among persons that reported using an e-cigarette or vaping product to inhale Δ9-THC, only using a Δ9-THC-containing product, using any nicotine-containing product, or only using a nicotine-containing product, which renewed concerns of toxicity from drug use by electronic delivery systems (Schier et al., 2019).
This targeted review is intended to provide students, public health departments, regulators, educators, researchers, and clinicians with: 1) background on the design and basic functioning of electronic delivery systems, 2) an understanding of the composition of e-liquids that contain flavorings or drugs, 3) a perspective on trends in usage of electronic delivery systems for nicotine and drug delivery, 4) a focused review of current knowledge on flavorings- and cannabis-induced toxicity related to their use in e-liquids and electronic delivery systems, and 5) a summary of knowledge gaps and research opportunities. Previously, Kaur et al. reviewed the literature on flavorings-related toxicity specific to lung cells and discussed potential biomarkers (Kaur, Muthumalage, & Rahman, 2018). The current review article extends the work of Kaur et al. to include more recent literature on lung toxicity and provides a general overview of all organs and systems currently known to be impacted by flavored e-liquids. For a review of the clinical aspects of EVALI, see Cherian et al. (Cherian, Kumar, & Estrada, 2020).
3. What are electronic delivery systems?
Electronic delivery systems are devices used to heat a substance to generate an aerosol that is inhaled by the user. Substances can include an e-liquid that that may contain flavorings and nicotine, Δ9-THC, or CBD or the device can heat dried cannabis plant material or its concentrated wax and oil extracts. Among electronic delivery systems, e-cigarettes are relatively new devices that are intended to aerosolize e-liquids that contain nicotine, but they are also used to aerosolize e-liquids that contain cannabis extracts. In contrast, vaporizers for cannabis plant material or its extracts are available in states with a legal retail market and have preceded e-cigarettes (Varlet, 2016).
3.1. E-cigarettes for nicotine delivery
Since their introduction in the early 2000s (and appearance in the U.S. marketplace in 2007), the internal design and external appearance of e-cigarettes for nicotine delivery has evolved continuously, with each subsequent change referred to as a “generation” (Bhatnagar et al., 2014; Schmidt, 2020; Schraufnagel et al., 2014). All e-cigarettes have four basic components: 1) a battery (rechargeable or non-rechargeable) used to heat a coil, 2) a cartridge to store the e-liquid, 3) an atomizer (i.e., heating coil that converts e-liquid to aerosol) chamber, and 4) a mouthpiece through which the user inhales (Breitbarth et al., 2018; Giroud et al., 2015). Figure 1 shows schematics depicting four generations of e-cigarette designs.
First generation e-cigarettes were intended to mimic the appearance (both in size and shape) of regular tobacco cigarettes and therefore have been referred to as “cigalikes” (Schmidt, 2020). First generation e-cigarettes came pre-assembled in various nicotine concentrations with and without flavorings. Upon inhalation through the mouthpiece, the battery would be activated to heat the e-liquid and a light would illuminate at the tip to simulate burning tobacco. These devices were discarded after the e-liquid was consumed.
Second generation e-cigarettes were typically larger than regular tobacco cigarettes, had medium-sized rechargeable batteries, evolved to contain a powerful atomizer to deliver greater energy that enhanced nicotine delivery, and large refillable cartridges for e-liquids (Schmidt, 2020). Variations of atomizers in second (and third) generation e-cigarettes included “cartomizers,” which were similar in design to atomizers but utilized a synthetic filler material that was wrapped around the heating coil to absorb e-liquid, and “clearomizers” composed of a clear tank but no filler material. Some second and future generation e-cigarettes had a manual switch that allowed the user to modulate puff length and frequency (referred to as topography) and therefore have been referred to as “personal vaporizers” (Protano et al., 2018). Other designs allowed the user to automatically puff by inhaling through the e-cigarette mouthpiece without the need to depress a switch and had adjustable voltages.
Third generation e-cigarettes had high capacity batteries and were designed to give the user more options to modify the applied voltage and resistance of the coil, which varied the coil temperature, and in turn, affected the characteristics of aerosol produced according to a user’s preferences. The sub-ohm level lower resistance coil contained in third generation devices is reported to be powerful enough to emit higher concentration of aerosols compared to earlier generation e-cig devices (Protano et al., 2018). Compared to their predecessors, third generation devices were more modifiable in external design to attach larger tanks that permitted even higher volume e-liquid storage and in functional capacity that allowed the user to customize vaping experiences (Schmidt, 2020). These models may also be referred to as “tank-style” e-cigarettes (Bhatnagar et al., 2014) or “juice monsters” (Talih et al., 2017).
Fourth generation e-cigarettes or “pod mods” feature a replaceable pre-filled or refillable cartridge that contains e-liquid referred to as a “pod” in combination with a modifiable (“mod”) system. One example of a replaceable pod e-cigarette is JUUL® brand devices, which operate at 3.7 V, the heating coil has resistance of 1.6 Ohm, and the maximum power is 8.1 W (Talih et al., 2019). An example of a refillable pod e-cigarette is Suorin brand devices, such as their Air series and Drop models. Among the major differences of pod mods in comparison with earlier generation e-cigarettes were their e-liquid formulation that contains nicotine in salt form (discussed below), coil electronics, and their external appearance. Pod mods come in many different shapes and colors but are typified by the JUUL® brand design that resembles a computer USB memory stick (Ramamurthi, Chau, & Jackler, 2019). Many pod mods are designed to have similar appearance to everyday items and the e-liquids are formulated to produce little odor or visible cloud when used (see Section 4.1), which enables “stealth vaping” among adolescents, whereby they are not detected by family members, teachers, etc. (Fadus, Smith, & Squeglia, 2019; Huang et al., 2019; Leavens et al., 2019; Mallock et al., 2020; Ramamurthi et al., 2019).
Fig. 1.
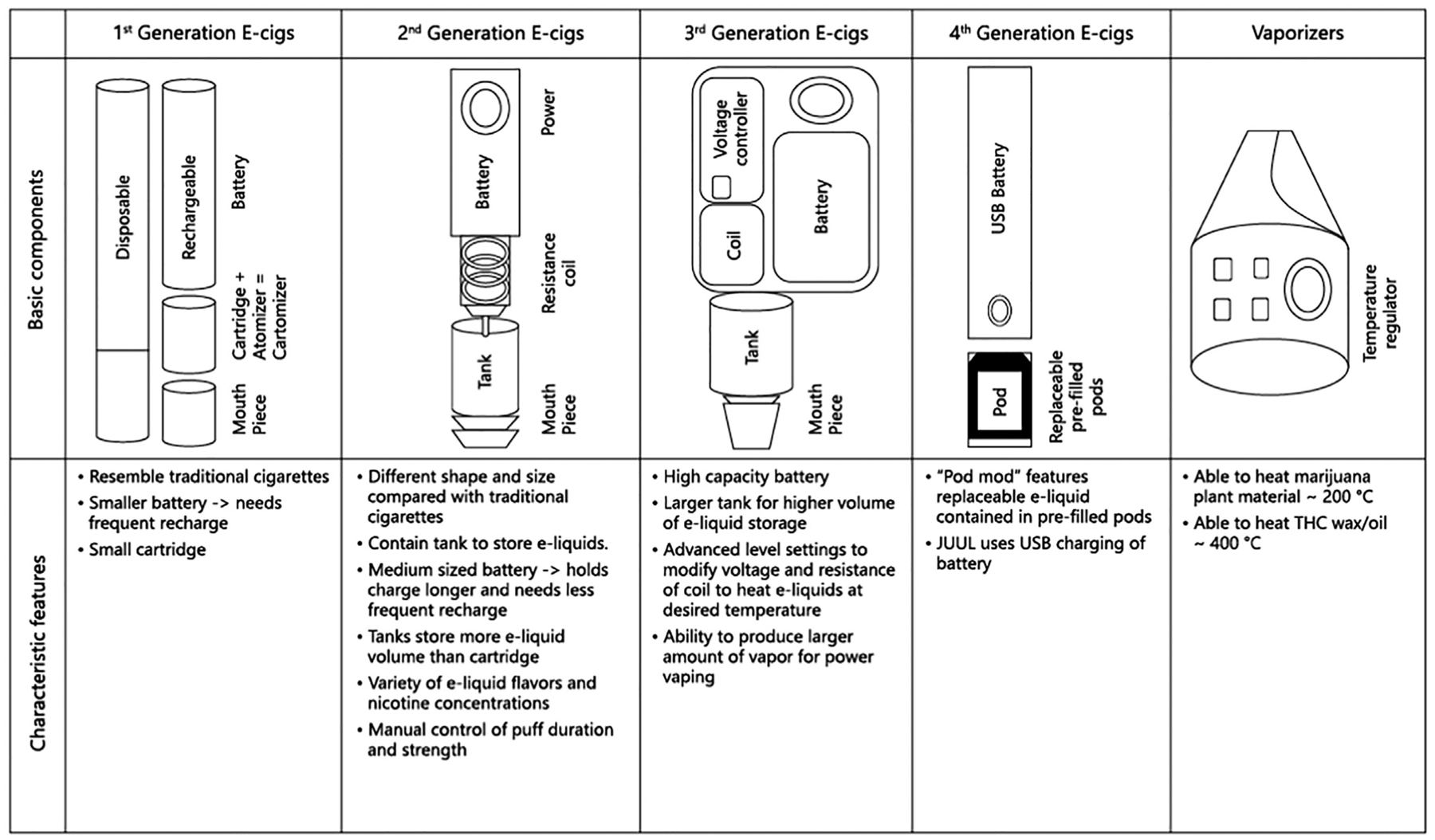
Schematics of e-cigarette generations. For more information on electronic delivery systems for nicotine or cannabis, e-liquids, and practices in altering devices to change delivery see the “E-Cigarette, or Vaping, Products Visual Dictionary” freely available at https://www.cdc.gov/tobacco/basic_information/e-cigarettes/pdfs/ecigarette-or-vaping-products-visual-dictionary-508.pdf
Finally, it is worth mentioning that some users of later generation e-cigarette engage in a practice known as “dripping” in which the e-liquid is applied directly on the atomizer rather than utilizing the cartridge with filler that is normally wrapped around the heating coil.
3.2. Vaporizers and e-cigarettes for cannabis delivery
Electronic delivery systems for cannabis include vaporizers (see Fig. 1) and e-cigarettes. Vaporizers are tabletop or personal portable devices used to heat dried cannabis plant material to about 200 °C without combustion to volatilize the active ingredients (Breitbarth et al., 2018). Vaporizers are one means for “dabbing”, the practice by which a small amount of concentrated Δ9-THC extract in the form of thick waxes or oils (e.g., butane hashish oil [BHO]) is heated and the user inhales the aerosol. BHO is a viscous, sticky, wax-like concentrate that may have up to 30 times the Δ9-THC concentration of dried cannabis plant material (Breitbarth et al., 2018;Varlet, 2016; Varlet et al., 2016). Ramamurthi et al. provided an insightful review of stealth vaping of cannabis using portable electronic devices and pointed to the commercial availability of products designed to hide the device by taking on the appearance of common items such as an ink pen, travel coffee mug, electronic car key fob, small electronics such as a remote control or an iPod®, a small mobile phone, a candy dispenser (vaporizer placed inside a package of Tic Tac® breath mints), and integrated into clothing such as hooded sweatshirts and backpacks (Ramamurthi et al., 2019).
Changes in atomizers for second and third generation e-cigarettes, the ability to modulate applied power and coil temperature, and special interchangeable coil head adapters have enabled the use of e-cigarettes to vape cannabis as dried plant material or a concentrated extract, either by itself or dissolved in an e-liquid (Breitbarth et al., 2018; Giroud et al., 2015). The practice of vaping cannabis compounds dissolved in an e-liquid was dubbed “cannavaping” by one research group (Varlet, 2016; Varlet et al., 2016). In addition to natural Δ9-THC or CBD, synthetic cannabinoids dissolved in an e-liquid or sprayed onto aromatic herbs, can be aerosolized using an e-cigarette fitted with a special coil head adapter (Breitbarth et al., 2018; Giroud et al., 2015).
4. What is an e-liquid?
E-liquids are solutions that contain humectants and usually nicotine and flavorings. E-liquids for cannabis delivery contain diluents/thickeners, cannabis extracts, and sometimes flavorings.
4.1. E-liquids for nicotine delivery
The basic constituents of an e-liquid for an e-cigarette are humectants, which are hygroscopic substances that help retain moisture (primarily propylene glycol [PG] and/or vegetable glycerin [VG]), water, ethanol, and usually nicotine and flavorings. The proportions of PG and VG in an e-liquid may be tailored to the user’s personal experiences and preferences. For example, PG has a lower density than VG, and when used at a higher proportion in an e-liquid, it contributes to an experience referred to as “throat hit”, which is a sensation produced at the back of a user’s throat upon inhalation of nicotine that may range from pleasant to harsh (Smith, Heckman, Wahlquist, Cummings, and Carpenter, 2020). VG is used at a higher proportion than PG if a user seeks a denser exhaled cloud and is popular among “power vapers” or “cloud chasers” who perform tricks such as creation of exhaled shapes (Schmidt, 2020).
E-liquids contain nicotine in free-base (basic pH ~8 to 10) or salt (acidic pH) form (El-Hellani et al., 2015). The form of nicotine in an e-liquid and resultant aerosol influences its bioavailability, which has varied with e-cigarette generation. E-liquids used in many third-generation and prior e-cigarettes contained 18 to 95% of their total nicotine in free-base form, which tended to have a more harsh throat hit (El-Hellani et al., 2015). The free-base form of nicotine differs from the acidic salt form used in fourth generation e-cigarettes (discussed below). Use of nicotine in a salt form has permitted manufacturers to increase the nicotine concentration of e-liquids used in e-cigarettes (Romberg et al., 2019). It has been reported that once an e-liquid refill container is opened, nicotyrine is formed via oxidization and the ratio of nicotyrine to nicotine in the e-liquid and aerosol generated by an e-cigarette increased over time. Nicotyrine is potentially toxic but also inhibits the in vivo metabolism of nicotine, hence, it is hypothesized to be a potentially useful smoking cessation aid (Martinez, Dhawan, Sumner, & Williams, 2015).
Trace levels of ethanol and water are added to e-liquids to enhance the experience for a variety of flavorings that are a major selling point of all ENDS (Berg, 2016). The presence of flavorings may add to the addictive effects of e-cigarettes (Soule, Lopez, Guy, & Cobb, 2016). For example, both vanillin and ethylvanillin have been shown to be monoamine oxidase inhibitors, which are substances present in tobacco smoke that enhance smokers’ addiction to nicotine by delaying the catalytic degradation of neurotransmitters by monoamine oxidase enzymes (Truman, Stanfill, Heydari, Silver, & Fowles, 2019). The exact number of flavored e-liquids currently available is unknown. In 2014, it was reported that there were 7,764 different flavored e-liquids available for use in first-and second-generation e-cigarettes (Zhu et al., 2014). Five years later there were over 19,000 commercial e-liquids on the market. Of these, 16,300 e-liquids could be classified into a flavor category; the most prominent flavors were fruit (34%), tobacco (16%), and dessert (10%) (Havermans et al., 2021). At least 210 different flavorings chemicals were used to create these flavored e-liquids and the mean number of flavorings per e-liquid was 10 (Krusemann et al., 2021). Table 1 is a list of 65 flavoring chemicals present in flavored e-liquids that have been reported to induce in vitro or in vivo toxicity in at least one study. These 65 flavorings were a subset of all flavorings that were tested in the studies listed in Table 1; some flavorings did not induce toxicity in some of the reviewed studies.
Table 1.
Sixty-five flavoring ingredients in non-cannabis e-liquids demonstrated to induce toxic effects on cells.
Ketones [D = Diketone, α-D = α-Diketone]
Terpenoids [A = alcohol, E = ether, K = ketone]
E-liquids for pod mods contain humectants, water, and usually flavorings but differ from e-liquids used in previous generation e-cigarettes in two important ways that maximize nicotine uptake to blood: 1) they contain an acid additive, and 2) nicotine in this matrix is in the form of a protonated salt (Gotts, 2019; Harvanko, Havel, Jacob, & Benowitz, 2020; Jackler & Ramamurthi, 2019; Ramamurthi et al., 2019; Talih et al., 2019). A regular tobacco cigarette contains approximately 1.5–2% nicotine which is equivalent to 1.5–2 mg nicotine/mL by volume. JUUL® brand is the most popular pod mod style device in the United States and in 2017, accounted for nearly 40% of all e-cigarette sales and over 70% of retail (excluding vape shop and internet sales) e-cigarette sales (Huang et al., 2019; Ramamurthi et al., 2019). JUUL® brand pods contain up to 5% nicotine by weight, which is equivalent to 5.9% by volume or 59 mg nicotine/mL (Jackler & Ramamurthi, 2019; Ramamurthi et al., 2019). According to Jackler and Ramamurthi, JUUL® company claimed that each 0.7 mL e-liquid pod delivers about 200 puffs or the equivalent to the amount of nicotine in a pack of 20 tobacco cigarettes (Jackler & Ramamurthi, 2019). Harvanko et al. measured acids in 23 e-liquids for second and third generation e-cigarettes and pods for fourth generation devices. The frequency of detected acids (how many of the 23 products contained an acid) in order from most to least was as follows: lactic, benzoic, levulinic, salicylic, malic, and tartaric (Harvanko et al., 2020). The use of acids in the e-liquid formulation for pod mod (and some earlier generation) e-cigarettes marks a critical evolution in e-cigarette technology. Nicotine has two basic nitrogen groups in its chemical structure and depending on the pH of the e-liquid, will exist in free-base (basic pH) or salt (acidic pH) form. Addition of a weak acid to the formulation yields an e-liquid that contains nicotine in the salt form that has a lower pH (~4.9) than free-base nicotine (~8 to 10), thereby allowing high levels of nicotine to be inhaled (and absorbed into blood) more easily and with less irritation or harsh throat hit compared with free-base nicotine in regular tobacco cigarettes and earlier generation e-cigarettes (Gotts, 2019; Harvanko et al., 2020; Jackler & Ramamurthi, 2019; Schmidt, 2020; Talih et al., 2019). JUUL® brand flavored e-liquids once included Cool Mint, Classic Menthol, Mango, Fruit Medley, Cool Cucumber, Crème Brulee, Classic Tobacco, and Virginia Tobacco. As of November 2019, JUUL® only sells menthol and tobacco flavored e-liquids. Note that other manufacturers have developed flavor enhancement pods that attach to the mouthpiece of JUUL® and other brand pod mod devices to mix flavorings with the user’s nicotine salt e-liquid (Cwalina, Leventhal, & Barrington-Trimis, 2020).
4.2. E-liquids for cannabis delivery
E-cigarettes are used to vape Δ9-THC, CBD, and synthetic cannabinoids (made in the laboratory) dispersed in e-liquids. Δ9-THC extracts, because of their unique physiochemical properties, are difficult to disperse in PG/VG humectants. Δ9-THC extracts are hydrophobic, highly viscous, semi-solid materials that are usually mixed with diluents, which might include vitamin-E acetate (VEA), medium chain triglycerides, coconut oil, squalane, or terpenes to form an e-liquid (Blount et al., 2020; Chand, Muthumalage, Maziak, & Rahman, 2019; Duffy et al., 2020; Giroud et al., 2015; Varlet, 2016). Among these diluents, in some cases VEA was used prior to its being strongly linked to EVALI because it has similar viscosity to pure Δ9-THC extract oil and was preferred by manufacturers because it is tasteless and odorless, making it difficult for consumers to visually differentiate a product composed of pure Δ9-THC oil compared with one that has been diluted with VEA (Blount et al., 2020; Duffy et al., 2020). Some e-liquids for Δ9-THC extracts were reported to contain pure PG as a diluent (Giroud et al., 2015; Peace, Stone, Poklis, Turner, & Poklis, 2016; Varlet et al., 2016). When heated at temperatures used to vape cannabis oils, PG can form acetaldehyde and formaldehyde (Troutt & DiDonato, 2017). Polar synthetic cannabinoids readily dissolve in the same e-liquid formulations that are used for nicotine delivery (Apirakkan et al., 2020; Breitbarth et al., 2018). Similarly, CBD can be dispersed in the same e-liquid formulations used for nicotine delivery (Grafinger, Kronert, Broillet, & Weinmann, 2020; Peace, Butler, Wolf, Poklis, & Poklis, 2016).
5. Who uses electronic delivery systems for nicotine and cannabis delivery?
E-cigarettes have rapidly gained popularity amongst youth for nicotine delivery, and by approximately 2014, were the most popular tobacco product for this demographic, overtaking use of regular tobacco cigarettes in the United States and the United Kingdom (de Lacy, Fletcher, Hewitt, Murphy, & Moore, 2017; Singh et al., 2016). By 2020, in the United States, 19.6% of high school students and 4.7% of middle school students were current e-cigarettes users (Wang et al., 2020). Globally, the use of ENDS is one of the most popular ways to inhale cannabis (Breitbarth et al., 2018).
5.1. E-cigarettes for nicotine delivery
Flavorings are a primary reason for use of any type of ENDS (Ambrose et al., 2015; Corey, Ambrose, Apelberg, & King, 2015; Cullen et al., 2019; Harrell et al., 2017; Okawa, Tabuchi, & Miyashiro, 2020; Pang et al., 2020; Rostron, Cheng, Gardner, & Ambrose, 2020; Tsai et al., 2018; Villanti et al., 2017). Specifically, from wave 1 of the U.S. Population Assessment of Tobacco and Health (PATH) study, among youth (age 12–17 years) that ever-used any type of ENDS, 81.5% reported that product flavoring was a reason for use (Ambrose et al., 2015). Further, “comes in flavors that I like” was the most highly ranked reason among youth who were ENDS users (Villanti et al., 2017). In wave 4 of the PATH study, flavor use among current (within past 30-days) ENDS users was 97.0% among youth, 96.8% among young adults (age 18–24 years), and 81.2% among adults (age ≥25 years) (Rostron et al., 2020). The 2020 National Youth Tobacco Survey (NYTS) revealed that among current e-cigarette users, 84.7% of high school students and 73.9% of middle school students used flavored e-liquids; fruit, menthol, mint, and candy, desserts, or other sweets were the most commonly reported flavors (Wang et al., 2020). Recently invented and popularized single-pod style devices such as JUUL® brand e-cigarette represent a unique form of e-cigarette that utilize nicotine salts (previously described in Sections 3 and 4) and prevalence of their use is described separately in Section 5.1.4. In general, prevalence estimates for current use of e-cigarettes for nicotine delivery follow the rank order (from highest to lowest): high school and college students > middle school students > adults, which indicate that these devices pose a widespread public health problem; data are briefly summarized herein for each demographic in order of decreasing prevalence.
5.1.1. High school and college students
E-cigarette use is highly prevalent amongst U.S. high school and college students. From 2011 to 2020, the prevalence of e-cigarette usage among U.S. high school students increased more than 13-fold from 1.5% to 19% (Singh, Arrazola, et al., 2016; Wang et al., 2020). In comparison with other countries, the prevalence of ever current (prior 30 days) e-cigarette use among high school students in Poland was 8.2%, and among high school students in Greece it was 2.8% (Goniewicz & Zielinska-Danch, 2012; Soteriades et al., 2020).
E-cigarette use has increased dramatically among college students in the last decade, with some estimates that one in four students are current users. Among students at eight colleges in North Carolina, the prevalence of current e-cigarette usage in 2009 was 1.5% (Sutfin, McCoy, Morrell, Hoeppner, & Wolfson, 2013). By 2011, the prevalence of current e-cigarette usage was 3.1% among Texas college students and by 2013, the prevalence of current e-cigarette usage was 14.9% among students at four New York State colleges (Saddleson et al., 2015). Roberts et al. followed students at a U.S. college from their year of entry through their third year and reported that current e-cigarette usage increased from 5.9 to 24.4% (Roberts, Keller-Hamilton, Ferketich, & Berman, 2020).
5.1.2. Middle school students
E-cigarette use among middle school students is a public health problem; an estimated 1 in 10 students could be current users. From the NYTS, between 2011 and 2015, the prevalence of current e-cigarette usage by U.S. middle school (grades 6–8) students increased 9-fold to 5.3% (Singh, Arrazola, et al., 2016). By 2018, it was estimated that the prevalence of e-cigarette use among U.S. middle school students was 10.6% (Fite, Cushing, & Ortega, 2020). In an update of the NYTS, Wang et al. reported that the prevalence of e-cigarette usage among middle school students in 2020 was 4.7% or 550,000 students (Wang et al., 2020).
5.1.3. Adults (18 years or older)
E-cigarette use is popular among adults and is common in many occupations, which indicates that these devices present a public health and workplace health concern. The prevalence of e-cigarette use by adults in the United States (18 years or older) is up 5.5% (Bao, Liu, Du, Snetselaar, & Wallace, 2020; Coleman et al., 2017; Delnevo et al., 2016; Jaber et al., 2018; Kava, Hannon, & Harris, 2020; Mirbolouk et al., 2018; Mirbolouk et al., 2019; Pearson, Richardson, Niaura, Vallone, & Abrams, 2012; Schoenborn & Gindi, 2015; Syamlal, Jamal, King, & Mazurek, 2016; Wang et al., 2019). For comparison, the prevalence of e-cigarette use was 4.3% in Japan (among older adolescents and young adults), 11.6% in Myanmar (among tobacco smokers), and 1.2% (women) and 3.7% (men) in Estonia (Okawa et al., 2020; Reile & Parna, 2020; Soteriades et al., 2020). Among U.S. adults, prevalence of current e-cigarette usage were consistently highest for the 18 to 24-year age group (5.1 to 40%) and more than half of current e-cigarette users (51.2%) were under 35 years (Mirbolouk et al., 2018; Mirbolouk et al., 2019; Rostron et al., 2020; Schoenborn & Gindi, 2015). E-cigarette usage among adults by industry in the United States were highest in the accommodation and food services industry (6.9%). By occupation, prevalence of e-cigarette use was highest in food preparation and serving-related jobs (6.8%) (Syamlal et al., 2016). Current e-cigarette use among adults in the United States varies by geographic location, e.g., one study reported prevalence that ranged from 2.4% (Washington, DC) to 6.7% (Oklahoma) (Hu et al., 2019).
5.1.4. Pod mod fourth generation e-cigarettes
Pod mod devices are a rapidly growing public health concern amongst youth and adults. Specifically, among fourth generation devices, JUUL® brand is the most popular pod mod style device with youth and adults in the United States, which in 2017, accounted for nearly 40% of all e-cigarette sales and over 70% of retail (excluding vape shops and internet sales) e-cigarette sales (Huang et al., 2019; Ramamurthi et al., 2019). Among current U.S. high school students who use e-cigarettes, 59.1% reported JUUL® as their usual brand in the last 30 days, whereas for middle school students who used e-cigarettes, 54.1% reported using only JUUL® (Cullen et al., 2019). At one U.S. college, the prevalence of current exclusive JUUL® usage was 21% among students (Roberts, Keller-Hamilton, Ferketich, & Berman, 2020). Among adults surveyed who tried JUUL®, 26% reported being exclusive JUUL® users (Leavens et al., 2019). Vallone et al. recently reported on 2018 – 2019 data from the Truth Longitudinal Cohort survey: for persons aged 15 to 34 years, the prevalence of current JUUL® users was 6.1%; from 2018 to 2019, JUUL® use increased among every age group in the survey but was highest (12.8%) for persons aged 18 to 20 years, though brand preferences vary over time (Vallone et al., 2020) as do flavor and device preferences (Ali et al., 2020). More recently, use of JUUL® brand devices among youth has declined in favor of Puff Bar, Pop Vapes, and other brands of disposable devices that come in a variety of flavors (Dai & Hao, 2020; Delnevo, Giovenco, & Hrywna, 2020; Miech et al., 2021).
5.2. Vaporizers and e-cigarettes for cannabis delivery
Early trends in e-cigarette usage for nicotine delivery among youth raised concerns about the potential use of these devices for consuming cannabis and other drugs (Giroud et al., 2015; Morean, Kong, Camenga, Cavallo, & Krishnan-Sarin, 2015). These concerns were supported by a survey of over 12,000 youth aged 16 to 19 years in Canada, the United States, and England which reported that use of e-cigarettes to aerosolize e-liquids was associated with their use to aerosolize cannabis (Smith et al., 2020). Multiple surveys reported that use of electronic delivery systems to inhale cannabis was more popular among high school (about 14–18 years old) students compared with middle school students and adults (Breitbarth et al., 2018; Dai, 2020; Dai & Siahpush, 2020; Morean et al., 2015).
Breitbarth et al. evaluated in detail the literature of surveys conducted from 2013 to 2017 on use of electronic delivery systems for inhalation of cannabis (Breitbarth et al., 2018). They reported that among high school students in the United States, 5.4–11.5% reported ever using an electronic delivery system to inhale cannabis and in Canada, that prevalence was 8%. Additionally, in the United States, the percentage of high school age cannabis users that used an electronic delivery system to inhale cannabis was higher if they resided in a state with legal medical cannabis compared to a state without legal medical cannabis (50.8% compared with 35.6%). Results from the NYTS conducted from 2017 to 2018 indicated that the percentage of high school students that used e-cigarettes to inhale cannabis increased from 16.1 to 21.7% (Dai, 2020). More recently, it was reported that 4.4 to 5.0% of high school students who responded to the Monitoring the Future survey conducted in the United States during 2017 reported that they used e-cigarettes to inhale cannabis (Dai & Siahpush, 2020).
From Breitbarth et al., in 2015, 3.4% of middle school students in Florida used an electronic delivery system to inhale cannabis (Breitbarth et al., 2018). Using data from the NYTS, Dai reported that from 2017 to 2018 the percent of middle school students that used an e-cigarette to inhale cannabis increased from 4.5 to 5.5% (Dai, 2020). Of middle school students who participated in the Monitoring the Future survey, 1.7% reported that they used an e-cigarette to inhale cannabis (Dai & Siahpush, 2020).
Cannabis is estimated to be used by 3.5% of adults worldwide, though usage varies within and between countries (Breitbarth et al., 2018). Breitbarth et al. summarized that in one study of 2016 data, 22.5% of college students in the United States had used an electronic delivery system to inhale cannabis. In a 2013–2014 survey, the percentages of adults that used an electronic delivery system to inhale cannabis was reported to be 5.8% in Australia, 11.2% in the United States, and 13.3% in Canada. In a 2017 survey, the percentages of adults that used an electronic delivery system to inhale cannabis was 6.2% in the United Kingdom. In states where non-medical adult use of cannabis was legal in the United States, 53.8% of adults reported that they used an electronic delivery system to inhale cannabis. The EVALI outbreak in the United States began in 2019 and as of January 14, 2020 there were 2,022 hospitalized patients who had data on substance use; 82% reported using Δ9-THC-containing products, 33% reported exclusive use of Δ9-THC-containing products; 57% reported using nicotine-containing products, and 14% reported exclusive use of nicotine-containing products. As of February 20, 2020 there were at least 2,807 hospitalized cases of EVALI reported to the Centers for Disease Control and Prevention (www.cdc.gov/EVALI).
5.3. Timeliness of this review article
The preceding sections outlined the components and evolution of e-cigarettes and personal vaporizers, compositions of e-liquids and cannabis extracts, and trends in e-cigarette use for nicotine delivery and cannabis delivery. Based on this background information, persons who span ¢a wide range of ages (from middle school students to adults) use e-cigarettes to aerosolize substances. A major driver for e-cigarette use, especially among youth, is the availability of flavored e-liquids. Many flavorings used in e-liquids for e-cigarettes fall under the FDA GRAS safety assessment program; however, GRAS status applies only to flavorings in foods for exposure through ingestion and does not provide regulatory authority for the use of a flavoring in e-liquids where exposure is via inhalation from vaping. There is precedent that flavorings intended for ingestion can cause significant bodily damage if inhaled. Notably, some workers that handled mixtures that contained the flavoring diacetyl during microwave popcorn production developed bronchiolitis obliterans, a devastating and sometimes fatal lung disease (Kreiss et al., 2002). Given past experiences, the widespread use of flavorings in e-liquids, and paucity of data, there is a clear need to critically evaluate the current state of knowledge on possible toxic effects from inhalation of flavorings and identify research gaps and opportunities. Further, the recent EVALI outbreak has brought attention to the trend of using e-cigarettes and the chemical complexity of e-liquids for cannabis delivery.
6. Methods
Fig. 2 summarizes the methods used to identify articles for this review. Peer-reviewed literature in English were sought in the PubMed and Scopus databases using the following keyword strings: (ENDS OR e-cigarette OR electronic cigarette OR electronic nicotine OR vaporizer OR EVALI), (flav* OR aroma), and (e-juice OR refill solution OR e-liquid) for publications as of April 30, 2020. For PubMed, results of these search strings were merged with the Boolean operator “AND”, which resulted in 190 articles. Abstracts of these 190 articles were screened and any that were non-English or outside our scope (i.e., policy, analytical method, exposure assessment, aerosol or e-liquid characterization, heated tobacco or similar products, safety reports or case studies of nicotine poisoning) were eliminated, which yielded 43 possible articles for inclusion. For Scopus, the merged search strings returned 641 citations, of which 514 were eliminated because they were non-English or outside of our scope, leaving 127 articles to consider for inclusion in this review. Next, the 170 citations (43 from PubMed + 127 from Scopus) were merged and we eliminated 19 that were duplicates citations then removed another 19 that were review/editorial articles, two risk assessments, three clinical reports, three studies of biomarkers or characterization of exposure only, and one smoking cessation trial, which resulted in N = 123 articles for consideration in final review. The third-level review involved detailed evaluation by one author (A.B.S.). Studies that did not evaluate toxicology of flavorings in e-liquids or flavored e-liquids or contain relevant information on EVALI were eliminated, which resulted in 61 publications on flavorings-related toxicology and 26 publications related to EVALI included in the review. In July 2020, the literature review was updated, which identified thirteen additional citations that met the criteria for inclusion in this review, bringing the totals to 67 publications on flavorings-related toxicology and 33 publications on EVALI. The articles included in this review were primarily in vitro studies (Table 2), with fewer publications available on in vivo rodent and human studies (Table 3).
Fig. 2.
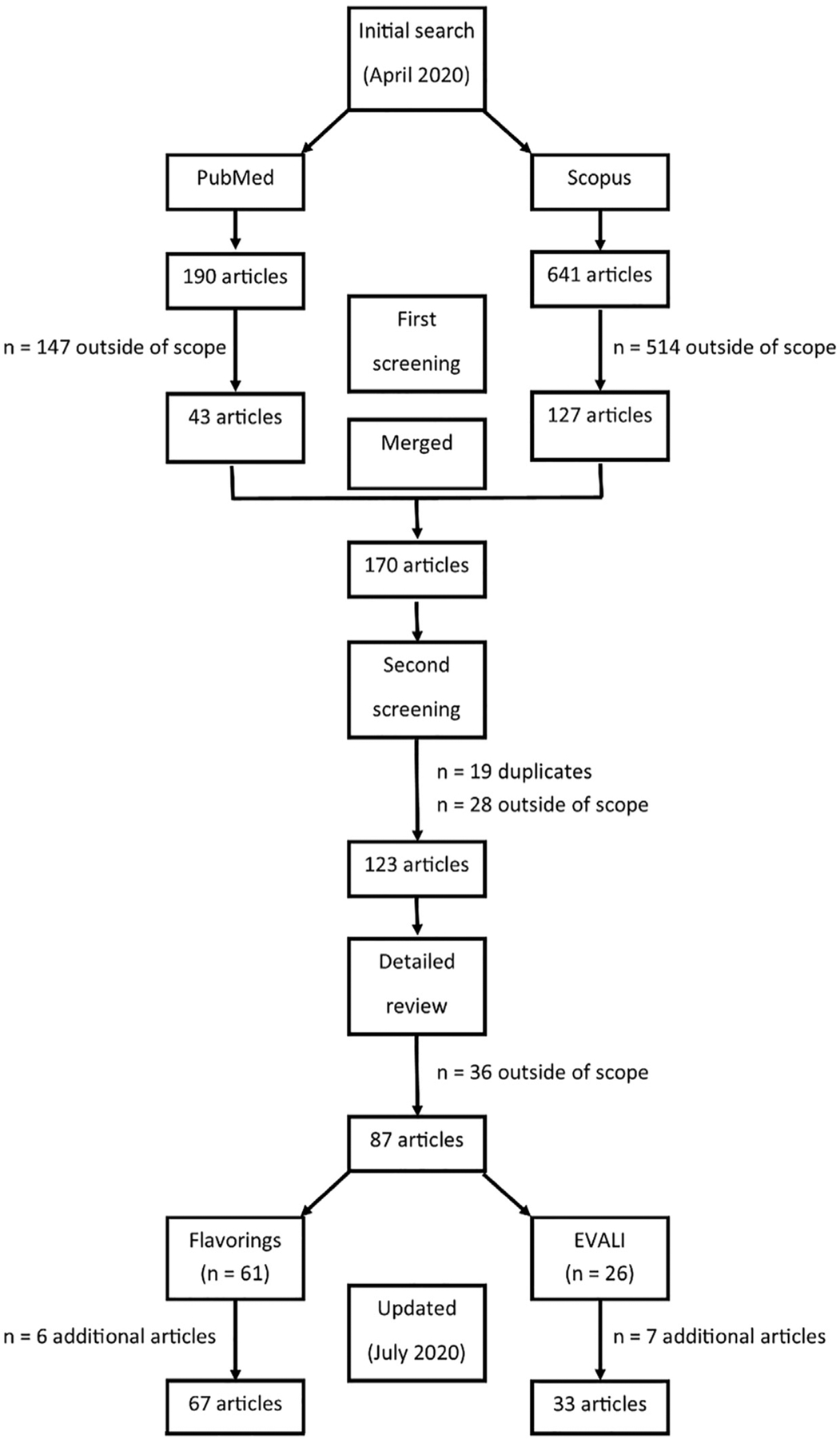
Flow diagram of literature search conducted in April 2020 and again in July 2020
Table 2.
Chronological summary of flavoring-induced in vitro toxicity literature. Unless otherwise noted, all flavored e-liquids are commercial products.
| Study | Typea | Cells | System Modeled | Source | Summaryb |
|---|---|---|---|---|---|
| Bahl et al. (2012) | Submerged | HESC MNSC HPF |
Developmental Developmental Respiratory |
Human – embryonic stem cells Mouse – primary, neural stem cells Human – primary, pulmonary fibroblasts |
|
| Farsalinos et al. (2013) | Submerged | H9c2 | Cardiovascular | Rat – myoblasts |
|
| Romagna et al. (2013) | Submerged | BALB/3T3 fibroblasts | Respiratory | Mouse – embryonic fibroblasts |
|
| Behar, Davis, Wang, et al. (2014) | Submerged | HESC HPF |
Developmental Respiratory |
Human – embryonic stem cells Human – primary, pulmonary fibroblasts |
|
| Cervellati et al. (2014) | Submerged | HaCaT A549 |
Skin Respiratory |
Human – primary, epidermal keratinocytes Human – lung, epithelial |
|
| Misra et al. (2014) | Submerged | A549 CHO-K1 |
Respiratory Genotoxicity |
Human – lung, epithelial Hamster – ovarian |
|
| Lerner et al. (2015) | Cell-free Submerged |
N/A HFL-1 |
Respiratory Respiratory |
N/A Human – normal, fibroblasts |
|
| Behar et al. (2016) | Submerged | HESC HPF A549 |
Developmental Respiratory Respiratory |
Human – embryonic stem cells Human – primary, pulmonary fibroblasts Human – lung, epithelial |
|
| Leigh et al. (2016) | ALI | H292 | Respiratory | Human – epithelial |
|
| Putzhammer et al. (2016) | Submerged | HUVEC | Cardiovascular | Human – umbilical/vascular endothelial |
|
| Sherwood and Boitano (2016) | Submerged ALI |
16HBE14o-MTE | Respiratory Respiratory |
Human – bronchial epithelial Mouse – primary tracheal |
|
| Singh, Arrazola, et al. (2016) | Submerged | Beas-2B | Respiratory | Human – lung bronchus |
|
| Sundar et al. (2016) | ALI | HPdLF HGEPp EpiGingival™ (Gin 100) |
Respiratory Respiratory Respiratory |
Human – ligament fibroblasts Human – primary, gingival epithelial Human – normal, oral epithelial |
|
| Welz et al. (2016) | Submerged | Oropharyngeal mucosa tissue | Respiratory | N/A |
|
| Bengalli et al. (2017) | Submerged | A549 ABB co-culture (NCI-H441 + HPMEC-ST1.6R) |
Respiratory Respiratory |
Human – lung, epithelial Human – lung, epithelial + Human – pulmonary microvascular endothelial |
|
| Clapp et al. (2017) | Submerged | Macrophage Neutrophil Natural Killer |
Respiratory Respiratory Circulatory |
Human – primary from lavage Human – primary from blood collection Human – primary from blood collection |
|
| Gerloff et al. (2017) | Submerged | Beas-2B H292 HFL-1 HBE |
Respiratory Respiratory Respiratory Respiratory |
Human – lung bronchus Human – epithelial Human – normal, fibroblasts Human – bronchial epithelial |
|
| Leslie et al. (2017) | Submerged | Beas-2B IB3–1 C38 CALU3 Wi-38 J774 THP-1* |
Respiratory Respiratory Respiratory Respiratory Respiratory Respiratory Respiratory |
Human – lung bronchus Human – bronchial Human – bronchus Human – lung epithelial Human – diploid lung fibroblasts Mouse – monocyte macrophage Human – monocyte peripheral blood |
|
| Rowell et al. (2017) | Submerged | CALU3 | Respiratory | Human – lung epithelial |
|
| Ween et al. (2017) | Submerged | THP-1* | Respiratory | Human – monocyte peripheral blood |
|
| Behar, Luo, et al. (2018) | Submerged | HPF A549 |
Respiratory Respiratory |
Human – primary, pulmonary fibroblasts Human – lung, epithelial |
|
| Behar, Wang, and Talbot (2018) | Submerged | HESC HPF A549 |
Developmental Respiratory Respiratory |
Human – embryonic stem cells Human – primary, pulmonary fibroblasts Human – lung, epithelial |
|
| Bitzer et al. (2018) | Cell-free | N/A | Respiratory | N/A |
|
| Fetterman et al. (2018) | Submerged | HAEC | Cardiovascular | Human – aortic endothelial |
|
| Kim et al. (2018) | Submerged | Streptococcus mutans | Skeletal | Bacteria |
|
| Muthumalage et al. (2018) | Submerged Cell free | MM6 U937 N/A |
Respiratory Respiratory Respiratory |
Human – blood monocyte Human – pleura/pleural lymphocyte N/A |
|
| Otreba et al. (2018) | Submerged | A549 | Respiratory | Human – lung, epithelial |
|
| Sassano et al. (2018) | Submerged HAM |
HEK-293T** A549 |
Respiratory Respiratory HASMC Human – primary, aorta monocyte Respiratory |
Human – kidney epithelial Human – lung, epithelial Cardiovascular Human – primary alveolar macrophage |
|
| ALI Song et al. (2018) |
HBE Submerged |
HMEEC | Respiratory Skin |
Human – bronchial epithelial Human – middle ear epithelial |
|
| Zhao et al. (2018) | Cell-free | N/A | Respiratory | N/A |
|
| Berkelhamer et al. (2019) | Submerged Isolated tissue |
PASMC Pulmonary arteries Bronchial rings |
Cardiovascular Cardiovascular Respiratory |
Ovine – pulmonary artery smooth muscle Ovine – neonatal and adult pulmonary arteries Ovine – neonatal and adult intrapulmonary bronchial rings |
|
| Bishop et al. (2019) | ALI | MucilAir™ | Respiratory | Human – 3D airway epithelial |
|
| Clapp et al. (2019) | Submerged | HBE Beas-2B |
Respiratory Respiratory |
Human – bronchial epithelial Human – lung bronchus |
|
| Czekala et al. (2019) | ALI | EpiAirway™ | Respiratory | Human – 3D respiratory model |
|
| Erythropel et al. (2019) | Submerged | HEK-293T** | Respiratory | Human – embryonic kidney |
|
| Hickman et al. (2019) | Submerged | Neutrophil | Respiratory | Human – primary lung |
|
| Iskandar, Zanetti, Kondylis, et al. (2019) | ALI | SmallAir™ EpiOral™ |
Respiratory Respiratory |
Human – small airway model Human – oral mucosal model |
|
| Iskandar, Zanetti, Marescotti, et al. (2019) | ALI Submerged |
SmallAir™ EpiOral™ HBE |
Respiratory Respiratory Respiratory |
Human – small airway model Human – oral mucosal model Human – bronchial epithelial |
|
| Lee et al. (2019) | Submerged | iPSC-EC | Cardiovascular | Human – induced pluripotent stem-cell derived endothelial |
|
| Nystoriak et al. (2019) | Submerged | hiPSC-CM | Cardiovascular | Human – induced pluripotent stem-cell |
|
| Omaiye et al. (2019) | Submerged | Beas-2B | Respiratory | Human – lung bronchus |
|
| Otero et al. (2019) | Submerged | MG-63 Saos-2 |
Skeletal Skeletal |
Human – bone fibroblast Human – osteosarcoma |
|
| Park et al. (2019) | ALI | HBE | Respiratory | Human – bronchial epithelial |
|
| Stevenson et al. (2019) | Submerged | SenzaCell | Immune | Human – myeloid leukemia-derived cell line |
|
| Ween et al. (2020) | Submerged | THP-1* HBE |
Respiratory Respiratory |
Human – monocyte peripheral blood Human – bronchial epithelial |
|
| Al-Saleh et al. (2020) | Submerged | TK6 CHO |
Genotoxicity Genotoxicity |
Human – spleen lymphoblast Hamster – ovarian |
|
| Go et al. (2020) | Submerged | HMEEC | Skin | Human – middle ear epithelial |
|
| Gómez et al. (2020) | Submerged | THP-1* | Respiratory | Human – monocyte peripheral blood |
|
| Kerasioti et al. (2020) | Submerged | EA.hy926 | Cardiovascular | Human – umbilical/vascular endothelial |
|
| Lamb et al. (2020) | Submerged | Beas-2B | Respiratory | Human – lung bronchus |
|
| Lucas et al. (2020) | Submerged | HFL-1 | Respiratory | Human – normal, fibroblasts |
|
| Marescotti et al. (2020) | Submerged | HBE | Respiratory | Human – bronchial epithelial |
|
| Noël et al. (2020) | Submerged | HUVEC/Tert2 | Cardiovascular | Human – umbilical/vascular endothelial |
|
| Pearce et al. (2020) | Submerged | HBE | Respiratory | Human – bronchial epithelial |
|
| Rowell et al. (2020) | Submerged | HBE CALU3 HEK-293T |
Respiratory Respiratory Respiratory |
Human – bronchial epithelial Human – lung epithelial Human – kidney |
|
| Tsai et al. (2020) | Submerged | Ca9-22 CAL-27 |
Respiratory Respiratory |
Human – gingival epithelial Human – tongue epithelial |
|
| Wavreil and Heggland (2020) | Submerged | MG-63 | Skeletal | Human–bone fibroblasts |
|
| Zahedi et al. (2019) | Submerged | C17.2 | Developmental | Mouse – embryonic stem cell |
|
ALI = air-liquid interface
AO = acridine orange; AxV = annexin V; CCK = cell counting assay; DCFH-DA = 2’−7’-dichlorodihydrofluorescein diacetate; DHE = dihydroethidium; ESR = electron spin resonance; IC50 = half maximal inhibitory concentration; LC50 = lethal concentration, 50%; MTS = 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium; MTT = 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide; NRU = Neutral Red Uptake; PCR = polymerase chain reaction; PI = propidium iodide; PG = propylene glycol; ROS = reactive oxygen species; RTCA = real-time cell analysis; TEER = transepithelial electrical resistance; TUNEL = (terminal deoxynucleotidyl transferase dUTP nick-end labeling; VG = vegetable glycerin; WST-1 = 4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzene disulfonate; WST-8 = water-soluble tetrazolium-8 dye
THP-1 = blood circulatory cells used as a model for the respiratory system
HEK-293T = renal cells used as a model for the respiratory system
Table 3.
Summary of flavoring-induced in vivo toxicity literature. Unless otherwise noted, all flavored e-liquids are commercial products.
| Study | Speciesa | Route | Organ system | Summaryb |
|---|---|---|---|---|
| Walele et al. (2016) | Humans (M) | Inhalation | Circulatory |
|
| Werley et al. (2016) | Crl:CD(SD) rats (F) | Nose-only inhalation | Circulatory Respiratory |
|
| St Helen et al. (2017) | Humans (M/F) | Inhalation | Circulatory |
|
| Conklin et al. (2018) | C57BL6/J mice (M) | Whole body inhalation | Circulatory |
|
| Chapman et al. (2019) | Balb/c mice (M/F) | Whole body inhalation | Respiratory |
|
| Rao et al. (2020) | Sprague-Dawley rats (M/F) | Nose-only inhalation | Cardiovascular |
|
| Szostak et al. (2020) | ApoE−/− mice (F) | Whole body inhalation | Cardiovascular |
|
| Reumann et al. (2020) | ApoE−/− mice (F) | Whole body inhalation | Skeletal |
|
| Szafran et al. (2020) | C57BL6/J mice (F) | Whole body inhalation | Respiratory |
|
F = female; M = male
ALP = alkaline phosphatase; AUC = area under concentration-time curve; BALF = bronchoalveolar lavage fluid; 3-HPMA = 3-hydroxypropyl mercapturic acid; LDH = lactate dehydrogenase
7. Toxicology of flavored e-liquids used in e-cigarettes
Fig. 3 depicts the relationship between vaped flavored e-liquids and target organs within the body. Examples of known toxic responses and potential adverse health effects by target organ are also given based on our literature review. Table 1 lists 65 flavorings used in e-liquids that were shown to induce toxicity. Cinnamaldehyde was most frequently reported to be cytotoxic, followed by vanillin, menthol, ethyl maltol, ethyl vanillin, benzaldehyde and linalool, and the remaining chemicals. Table 2 (in vitro studies) and Table 3 (in vivo and human studies) summarize the main findings of the 67 identified publications on the toxicity of flavored e-liquids and flavorings. Studies to date have focused primarily on the respiratory tract. Aerosolized flavorings in e-liquids are not fully metabolized in the lung and a portion of the inhaled dose is absorbed into the bloodstream and distributed throughout the body to the cardiovascular, developmental, skeletal, and immune systems. Many flavorings were genotoxic or mutagenic in multiple cell types of the body. Additionally, aerosolized flavorings may adversely affect the skin. Based on toxicological data such as those presented in this section and other considerations, the U.S. FDA announced on January 2, 2020 their finalized enforcement policy on unauthorized flavored cartridge (pod)-based e-cigarettes (e.g., JUUL® pod mod devices) that appeal to children (FDA, 2020). Under this policy, companies must cease the manufacture, distribution and sale of unauthorized flavored cartridges, though tobacco and menthol flavorings were exempted from the policy. This ban applied only to flavored cartridges for use in pod mod devices and does not apply to manufacturers of flavor enhancement pods that attach to the mouthpiece of JUUL® and other brand pod mod devices, nor does it limit the availability of flavored e-liquids for use in disposable (first generation) and tank-style (second and third generation) e-cigarettes.
Fig. 3.
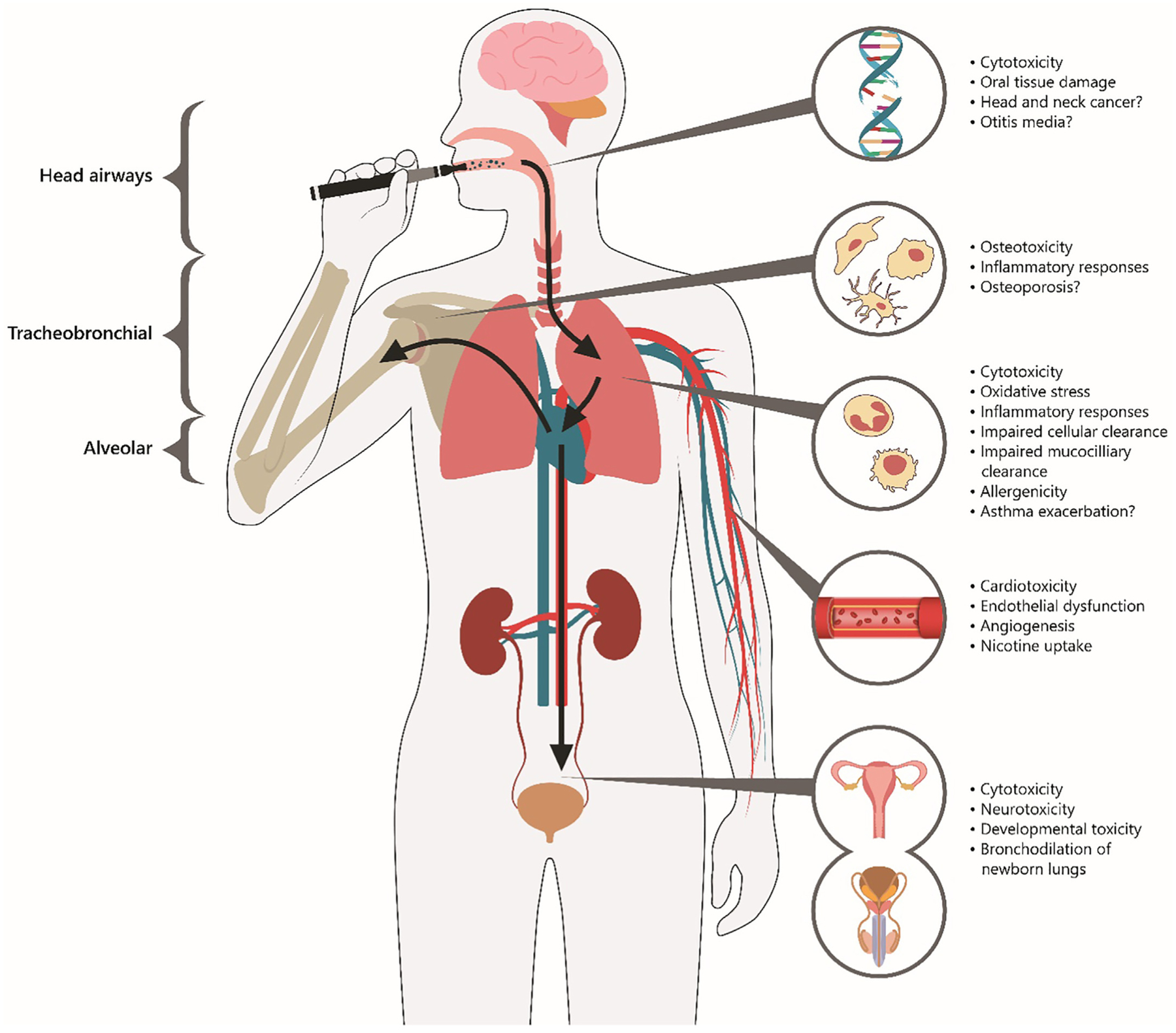
Aerosolized flavored e-liquids and target organs/systems within the body including known toxic responses and potential adverse health effects.
It is important to note that various methods were used to expose cells to a bulk flavored e-liquid or aerosol generated from a flavored e-liquid that was heated using an e-cigarette or other means. This lack of standardization in methods used to expose cells makes inter-comparison of study results difficult, and as noted in Section 9, standardization of many experimental parameters is likely to reduce inter-study variability. For studies that exposed cells to bulk e-liquids, the most common approach was to dilute the e-liquid in cell culture medium. For studies that exposed cells to aerosolizede-liquid, there is much variability in methods used to generate aerosol such as the generation of e-cigarette used, the device settings (voltage, power, coil resistance), puff topography, and coil temperature as well as in methods used to collect aerosols. For example, Romagna et al. were the first to expose cells to constituents of aerosolized flavored e-liquid. In their study, an e-cigarette was connected via tubing to a flask that contained culture medium. The other end of the tubing was positioned just above the culture medium; a vacuum pump was used to draw aerosol from the e-cigarette into the flask and over the medium and the fraction of aerosol that dissolved in the culture medium was used to expose cells in a submerged culture system (Romagna et al., 2013). Another method employed to expose cells in some studies was the use of a smoking machine to puff on an e-cigarette. Once generated by a smoking machine, aerosol can be trapped in a condenser filled with culture medium (Bengalli, Ferri, Labra, & Mantecca, 2017), passed through a liquid impinger (Bitzer et al., 2018), or collected by other means and used to expose cells in a submerged culture system. Another experimental design is to directly expose cells to aerosol generated from an e-cigarette by a smoking machine using an air-liquid interface (ALI) system (Leigh, Lawton, Hershberger, & Goniewicz, 2016).
7.1. Respiratory system
The human respiratory tract can be divided into three main regions: head-airways, tracheobronchial, and alveolar (see Fig. 3). The head-airways region extends from the nose and mouth to the larynx, the tracheobronchial region from the trachea to the bronchioles, and the alveolar region includes the terminal and respiratory bronchioles and the alveoli. When aerosolized e-liquid is inhaled, it will travel throughout these successive regions of the respiratory tract and interact with various cell types and induce different effects. Herein, we critically review publications related to toxicity associated with flavored e-liquids. For more information on respiratory hazards of e-cigarettes, including health impacts of non-flavored e-liquids, the reader is referred to several recent review articles (Chun, Moazed, Calfee, Matthay, & Gotts, 2017; Gotts, Jordt, McConnell, & Tarran, 2019; Tzortzi, Kapetanstrataki, Evangelopoulou, & Beghrakis, 2020).
7.1.1. Cytotoxicity
From Table 2, flavored e-liquids and flavoring constituents were shown to be cytotoxic to cells encountered in the head (e.g., oropharyngeal mucosa tissue model), tracheobronchial (e.g., human lung bronchus Beas-2B cell line), and alveolar (e.g., mouse macrophage J774 cell line) regions of the respiratory tract. The Talbot laboratory performed the seminal work on in vitro respiratory toxicity of flavored e-liquids. They first suggested that cytotoxicity of e-liquids was related to flavorings, not nicotine. In their study, twelve out of 36 butterscotch, caramel, coffee, fruit, chocolate, menthol, tobacco, and cinnamon flavored e-liquid products were highly cytotoxic to human primary pulmonary fibroblast (HPF) cells; Cinnamon Ceylon product was the most potent (Bahl et al., 2012). Shortly thereafter, Romagna et al. published the first study that evaluated cytotoxicity of aerosolized flavored e-liquids. A second-generation e-cigarette was used to aerosolize 21 flavored e-liquids; only Coffee flavored e-liquid was cytotoxic in BALB/3T3 fibroblasts at the highest tested concentration (Romagna et al., 2013). Some caution is warranted in generalizing the results from Romagna et al. to the human respiratory system because the cells used were mouse embryonic cells. Cervalleti et al. reported that a Balsamic flavored e-liquid was cytotoxic to human lung epithelial A549 cells (Cervellati et al., 2014) whereas Misra et al. reported that Classic Tobacco and Magnificent Menthol flavored e-liquids were not cytotoxic to this cell line (Misra, Leverette, Cooper, Bennett, & Brown, 2014). The Talbot laboratory continued their line of inquiry by asking whether Cinnamon Ceylon e-liquid specifically, or cinnamon flavored e-liquids in general, were cytotoxic. In an interesting study design, HPF cells were plated in a cross pattern and a single dose of a cinnamon-flavored e-liquid was added to the center culture well of the cross. Of the eight cinnamon-flavored e-liquids (including Cinnamon Ceylon), five were volatile and induced cytotoxicity in adjacent cell culture wells. Next the authors analyzed the chemical composition of e-liquids that exhibited cytotoxicity and identified four common flavorings: cinnamaldehyde, 2-methoxycinnamaldehyde, dipropylene glycol, and vanillin. HPF cells were exposed to authentic standards of each flavoring and all were cytotoxic; cinnamaldehyde and 2-methoxycinnamaldehyde were the most potent (Behar et al., 2014). Results of this publication sparked a debate with the Farsalinos laboratory on whether it was appropriate to test diluted e-liquids since, when aerosolized, they are heated and the characteristics of the aerosol might differ from the bulk liquid (Behar, Davis, Bahl, Lin, & Talbot, 2014; Farsalinos, Romagna, & Voudris, 2014). The Talbot laboratory exposed A549 and HPF cells to aerosolized Cinnamon Ceylon flavored e-liquid and aerosolized cinnamaldehyde flavoring. Aerosols produced with a fixed voltage second generation e-cigarette and a variable voltage third-generation e-cigarette were cytotoxic. For the third-generation e-cigarette, the cytotoxicity of aerosolized Cinnamon Ceylon flavored e-liquid and cinnamaldehyde flavoring increased with applied voltage from 3 V (4.2 W) to 5 V (11.9 W). Additionally, the authors reported the formation of new substances in aerosol, including 2,3-butanedione (diacetyl), at 5 V but not 3 V (Behar et al., 2016). In subsequent studies, the Talbot laboratory reaffirmed that both the e-cigarette generation and applied voltage influenced aerosol production (including the formation of new substances), which in turn affected cytotoxicity (Behar, Luo, McWhirter, Pankow, & Talbot, 2018) and that observed cytotoxicity from exposure to diluted e-liquids and aerosolized e-liquids agreed 74% of the time, which indicated that bulk liquids have utility to screen for cytotoxicity (Behar, Wang, & Talbot, 2018). Other researchers, including Otreba et al. have independently confirmed that cytotoxicity of aerosolized flavored e-liquids increased with applied e-cigarette voltage (Otreba, Kosmider, Knysak, Warncke, & Sobczak, 2018).
Within a few years of the first publication on cytotoxicity of flavored e-liquids, there were advances in the complexity of study designs of respiratory toxicity, including the first uses of an ALI system and a tissue model, development of a high capacity screening method, and an in vivo study. Leigh et al. noted that a submerged cell culture, to which diluted e-liquid was added, does not accurately model inhalation exposure of an aerosol. The authors exposed H292 human lung epithelial cells to aerosolized Tobacco, Piña Colada, Menthol, Coffee, and Strawberry flavored e-liquids generated using a tank-style e-cigarette in an ALI system and reported that all flavored e-liquids were cytotoxic; Strawberry was the most potent (Leigh et al., 2016). Keeping with the theme of trying to more accurately mimic the complex conditions in the respiratory tract, Welz et al. employed an oropharyngeal mucosa tissue model to evaluate the cytotoxicity of Apple, Cherry, and Tobacco flavored e-liquids and base humectant mixtures (free of nicotine and flavors). All flavored e-liquids were cytotoxic, though Apple and Cherry were more cytotoxic compared with Tobacco (Welz et al., 2016). The sheer number of flavored e-liquids available and number of flavorings used in these products makes individual screening a formidable challenge. Sherwood and Boitano adopted a high-capacity real-time cell analysis technique to screen multiple flavorings using human bronchial epithelial (HBE) cells. Seven flavoring chemicals were screened and 2,5-dimethypyrazine, damascenone, linalool, α-ionone, and ethyl maltol were all cytotoxic (Sherwood & Boitano, 2016). Singh et al. evaluated 18 flavored e-liquids using Beas-2B human lung bronchus cells and confirmed earlier reports that menthol, tobacco, and butterscotch flavored e-liquids were among the most cytotoxic (Singh, Luquet, Smith, Potgieter, & Ragazzon, 2016).
Bengalli et al. compared the cytotoxicity of aerosolized Mint and Cinnamon flavored e-liquids in a monoculture (submerged A549 lung cells) and an alveolar-blood barrier (ABB) co-culture system (NCI-H441 human lung epithelial cells + HPMEC-ST1.6R human pulmonary microvascular endothelial cells). Both aerosols were cytotoxic in the mono- and co-culture systems, though the monoculture was generally more sensitive to cytotoxic effects (Bengalli et al., 2017). Several more studies were published in the literature that reported (sometimes conflicting) results on the cytotoxic potential of flavored e-liquids and flavorings to various cell types in the respiratory system (Gerloff et al., 2017; Gómez et al., 2020; Lucas et al., 2020; Rowell et al., 2017; Ween et al., 2020; Ween, Whittall, Hamon, Reynolds, & Hodge, 2017). Interestingly, results of an in vitro study with HFL-1 pulmonary fibroblasts indicated that in addition to cytotoxicity, an e-liquid that contained tobacco, coconut, vanilla and cookie flavors increased senescence-associated beta-galactosidase (SA-β-gal) activity and inhibited transforming growth factor-β1 (TGF-β1) (Lucas et al., 2020). An increase in SA-β-gal is indicative of cellular senescence (alterations in cellular homeostasis consistent with pre-mature aging). TGF-β1 controls differentiation of fibroblast cells into myofibroblasts, and inhibition of this growth factor indicated compromised wound healing responses in cells.
To clarify the role of cell type in observed cytotoxicity results, Leslie et al. systematically compared responses in multiple types of lung cells. The authors evaluated the influence of 10 aerosolized flavored e-liquids on seven cell types: four human-derived bronchial epithelial cell lines (Beas-2B, IB3-1, C38, and CALU-3), one mouse macrophage cell line (J774), one human monocyte cell line (THP-1), and one human fibroblast cell line (Wi-38). These cell lines were selected to test the effects of aerosolized flavored e-liquids on multiple respiratory cell types that would encounter inhaled e-cigarette aerosol: bronchial epithelial cells that line the upper respiratory tract, underlying fibroblast cells, and macrophages, which are immune cells that function to remove foreign material from lung surfaces. As expected, different cell types exhibited different sensitivity to aerosolized flavored e-liquids. In general, Beas-2B lung epithelial cells were most sensitive and aerosolized Strawberry and Cherry flavored e-liquids were most cytotoxic. Based on their data, the authors concluded that the chosen cell line can influence cytotoxicity study results and there was a need for a standardized in vitro test protocol to evaluate respiratory cytotoxicity of e-liquids (Leslie et al., 2017).
Subsequent research evolved from testing single flavorings to exploring the effects of complex mixtures of flavoring chemicals used in e-liquids, the use of ALI systems became more common, more high-throughput screening (HTS) and systems toxicology approaches were reported, and the first study of flavored e-liquids for JUUL® brand pod mod e-cigarettes was reported. Muthumalage et al. was the first to systematically evaluate and compare the cytotoxicity of individual flavoring chemicals and flavoring mixtures used in e-liquids; they reported that the mixtures were more cytotoxic to respiratory tract cells than the individual constituents (Muthumalage et al., 2018). In a subsequent study, Marescotti et al. evaluated 28 flavoring chemicals both independently and in mixtures using laboratory prepared e-liquids and HBE cells. The authors reported that individually 2-acetylthiazole, allyl hexanoate, α-pinene, citronellol, guaiacol, linalool, methyl anthranilate, 3-methyl-2,4-nonanedione, 3-(methylthio) propionaldehyde, and phenethyl alcohol e-liquids exhibited increased cytotoxicity; citronellol and α-pinene were the most cytotoxic. When they evaluated the cytotoxicity of flavoring mixtures, the cytotoxicity of mixtures differed from that of the individual flavorings and citronellol was the main driver of toxicity while other flavorings contributed to synergistic effects (Marescotti et al., 2020). Tissue models better mimic in vivo conditions than submerged monocultures because they contain differentiated cell types that are present in the respiratory epithelium. Using ALI systems, aerosolized Blueberry flavored e-liquid was not cytotoxic in the EpiAirway™ 3D tissue model (Czekala et al., 2019) but an aerosolized e-liquid that contained cinnamaldehyde flavoring was cytotoxic in the MucilAir™ 3D tissue model (Bishop et al., 2019).
Sassano et al. reported an open source three-phase HTS approach. This HTS approach permitted screening of cytotoxicity for 148 commercial flavored e-liquids, more than any single study to date. The authors reported that liquid and aerosolized Arctic Tobacco, Pumpkin Pie, Chocolate Banana, Cherry Kola, Kola, Hot Cinnamon Candies, Mojito, Green Gummies, Vanilla Bean, and Menthol Tobacco flavored e-liquids were most cytotoxic to HEK-293T cells. It is important to note that, though often used in toxicology studies because of their robustness, HEK are human kidney epithelial cells, not respiratory cells. The authors further evaluated a subset of 14 flavored e-liquids and reported that they were cytotoxic in A549 human lung epithelial, HBE, and primary alveolar macrophage cells. Their data revealed a weak correlation between the presence or absence of flavorings in e-liquids and cytotoxicity; however, there was a correlation between cytotoxicity and the concentration of vanillin and cinnamaldehyde flavorings in e-liquids and vanillin was identified as a major driver of cytotoxicity. Aside from product-specific cytotoxicity data, this HTS approach revealed two important findings. Firstly, cytotoxicity was consistent regardless of whether cells were exposed to the flavored e-liquid itself or the aerosolized flavored e-liquid, which supports the utility of bulk e-liquid screening that was debated in the earlier literature. Secondly, the authors observed that the more flavoring chemicals in an e-liquid product, the more cytotoxic it was to respiratory cells, which was consistent with prior studies using embryonic stem cells (Bahl et al., 2012). Overall, their results suggested that an HTS approach to evaluate the cytotoxicity of flavored e-liquids may be feasible (Sassano et al., 2018).
In a series of studies by the tobacco cigarette industry, which has developed its own e-cigarette products, a three-tier systems toxicology framework was proposed to evaluate toxicity of e-liquids (Iskandar et al., 2019; Marescotti et al., 2020). The first tier of this framework was intended to screen e-liquids for potential toxicity, the second tier to determine the mechanism of toxicity for e-liquids, and the third tier to determine the mechanism of toxicity for the aerosolized e-liquids. Using their approach, Iskander et al. reported no difference in cytotoxicity of a flavored (not specified) e-liquid compared with an unflavored e-liquid of the same composition using a submerged monoculture of HBE cells or an ALI system with SmallAir™ (human small airway) and EpiOral™ (human mucosal) 3D tissue models (Iskandar, Zanetti, Marescotti, et al., 2019). In a follow-on tobacco industry study, Marescotti et al. applied this systems toxicology approach but presented a computationally derived scoring system for each tier to create a single summary score of all observed toxic effects of an e-liquid. Their scoring system was applied to 28 flavoring chemicals alone or in mixtures, and as noted earlier in this section, cytotoxicity of mixtures differed from that of the individual flavoring constituents (Marescotti et al., 2020). Based, in part, on the results of these studies, the tobacco cigarette industry touted their systems toxicology approach as a valuable tool to screen single flavoring substances and rank them based on their toxicity “so that manufacturers can develop and/or produce e-liquids with nontoxic flavor composition and doses” (Marescotti et al., 2020). As noted in Section 5.1, use of flavorings in e-liquids is a known major attractant for youth to begin e-cigarette use.
The Talbot laboratory extended their work on cytotoxicity of aerosolized flavored e-liquids and flavorings produced by early generation e-cigarettes to fourth generation pod mod devices. Omaiye et al. evaluated the cytotoxicity of all eight JUUL® brand flavored e-liquids. All e-liquids were cytotoxic to Beas-2B human lung bronchus cells and five of eight aerosolized flavored e-liquids were cytotoxic to these cells. Cytotoxicity of aerosolized flavored e-liquids was highly correlated with ethyl maltol flavoring concentration and weakly correlated with menthol and vanillin flavoring concentrations. The authors noted that the U.S. FDA has raised concerns that JUUL® use may pose risk of addiction to nicotine for a new generation of adolescents and serve as a gateway to use of regular tobacco cigarettes. They also noted that their data raised a new concern that the high levels of flavorings in JUUL® e-liquids can damage or kill lung cells (Omaiye, McWhirter, Luo, Pankow, & Talbot, 2019). As noted in Section 4.1, as of November 2019, JUUL® only sells Menthol, Classic Tobacco, and Virginia Tobacco flavored e-liquids. Recently, Lamb et al. evaluated the effects of aerosolized JUUL® brand Menthol and Virginia Tobacco e-liquids on mitochondrial function. They reported that aerosolized Menthol flavored e-liquid caused mitochondrial dysfunction in Beas-2B lung epithelial cells (Lamb, Muthumalage, & Rahman, 2020), which may lead to a variety of diseases. Note that since JUUL stopped selling fruity and other flavored e-liquids, some manufacturers have developed flavor enhancement pods that attach to the mouthpiece of JUUL® and other brand pod mod devices to mix flavorings with the user’s nicotine salt e-liquid (Cwalina et al., 2020).
Secondhand exposure to regular tobacco smoke is associated with development of otitis media, an infection characterized by pain, inflammation, and flow of fluid out of the middle ear cavity (Song et al., 2018). Epithelial cells help to maintain homeostasis and sterility of the middle ear and abnormalities in these cells can lead to the development of otitis media. Given the association with regular tobacco smoking, the Chang laboratory asked whether the use of e-cigarettes may negatively impact the middle ear. In their initial study, human middle ear epithelial cells were exposed to flavored e-liquids without nicotine (Song et al., 2018). A total of 73 e-liquids grouped into five flavor categories (tobacco, coffee, fruit, mint/menthol, and “other” such as caramel and honey) were tested for cytotoxic potential. Among specific flavored e-liquid products, Tobacco, Coffee, Mango, and Chocolate-Menthol were cytotoxic with Chocolate-Menthol being the most potent. Among flavor categories, mint/menthol was the most cytotoxic. In a follow-on study, these researchers evaluated the effect of Menthol and Tobacco flavored e-liquids on middle ear epithelial cells (Go, Mun, Chae, Chang, & Song, 2020). Both flavored e-liquids increased release of mRNA of inflammatory cytokines and mucin production and induced apoptosis and autophagy; Menthol flavored e-liquid was more cytotoxic compared with Tobacco flavored e-liquid. The authors concluded that flavored e-liquids were cytotoxic and could cause otitis media in middle ear epithelial cells via reduced cell viability and stimulation of inflammatory cytokines and mucin production.
Regular tobacco cigarettes generate secondhand smoke via smoldering of the cigarette when not puffed and exhalation of smoke. E-cigarettes do not smolder like a regular tobacco cigarette, so the only secondhand exposure potential is the aerosol that is exhaled by a user. The composition of mainstream aerosol (what is inhaled by the user) differs from that of the secondhand aerosol (Marco & Grimalt, 2015; Papaefstathiou, Bezantakos, Stylianou, Biskos, & Agapiou, 2020; Samburova et al., 2018). In the studies of otitis media, cells were exposed to diluted e-liquids even though development of this disease has been associated with secondhand tobacco smoke exposure. These studies have provided a valuable foundation for understanding possible effects of flavored e-liquids, though future studies of otitis media should also consider exposure to secondhand (exhaled) aerosol constituents.
7.1.2. Oxidative stress and inflammatory responses
The toxic effects of flavored e-liquids and flavoring constituents in the respiratory tract extends beyond cytotoxicity. Several researchers have explored respiratory oxidative stress and inflammatory responses as key events in the pathogenesis of chronic respiratory system diseases. The generation of reactive oxygen species (ROS) and the effects on cell signaling may result from a respiratory or oxidative burst response to cellular contact with a foreign body. Generation of ROS may lead to the stimulation of inflammatory processes such as secretion of chemotactic factors, proteolytic enzymes, lipoxygenases, and the release of signaling proteins (Leonard, Harris, & Shi, 2004). There is a critical balance between oxidants and antioxidant defenses (Ho, Magnenat, Gargano, & Cao, 1998) and if cells are unable to maintain this redox balance, it may result in a chronic inflammatory state in the respiratory system. This inflammatory state may result in damage to the cells involved and to the surrounding tissue via activation of signaling pathways, inflammatory cytokine production, altered gene expression, and other cellular modifications.
Several research groups have screened flavored e-liquids for capacity to produce ROS using cell-free and cellular systems. Lerner et al. evaluated the capacity of 22 flavored e-liquids to generate ROS using a cell-free fluorescent probe. In this study, aerosol was generated using an e-cigarette attached to a smoking machine; the aerosol was passed through a bubbler that contained 2’,7’dichlorofluorescein dye solution then analyzed for oxidized dichlorofluorescein fluorescence using a spectrophotometer. All flavored e-liquids generated ROS, though amounts differed by product; sweet or fruit flavored e-liquids were stronger oxidizers than tobacco flavored e-liquids. The authors concluded that ROS generated by flavored e-liquids was dependent on the presence of flavoring chemicals. The authors also explored factors that could influence generation of ROS in aerosolized flavored e-liquids. Their results identified ROS in aerosolized Magnificent Menthol and Classic Tobacco flavored e-liquids, which indicated that ROS may be inhaled directly into the lung during e-cigarette use. Further, generation of ROS in aerosolized e-liquids was impacted by the age of the e-cigarette heating coil (see e-cigarette schematics in Fig. 1): more ROS were produced from a new coil compared with coils that were previously used at least 50 times (Lerner et al., 2015). Muthumalage et al. also evaluated cell-free ROS generation from flavored e-liquids aerosolized with new and aged heating coils using the 2’,7’dichlorofluorescein dye method. When aerosolized with a new atomizer, American Tobacco, Mystery Mix, and Mixed Flavors e-liquids generated ROS and when aerosolized with a used atomizer, Café Latte, Cinnamon Roll, and Cotton Candy flavored e-liquids generated ROS. In this study, hydrogen peroxide (H2O2) equivalents (a relatively stable ROS species) produced with new and used heating coils were similar (Muthumalage et al., 2018). Unfortunately, the same flavored e-liquids were not tested with both conditions of coils (new or used), which precluded inference as to the influence of coil age on ROS generation. Zhao et al. characterized cell-free ROS generation from two flavored e-liquids. These authors used Trolox, a water-soluble form of vitamin E to measure ROS production. Trolox is oxidized to its Trolox quinone in the presence of ROS. Briefly, aerosol from an e-cigarette generated using a smoking machine was pulled through a bubbler that contained Trolox solution. The Trolox solution was split and horseradish peroxidase added to one sample. Liquid chromatography with tandem mass spectrometry analysis was used to analyze samples for Trolox quinone, i.e., the sample with no horseradish peroxidase was used to quantify short-lived ROS species and the among of hydrogen peroxide equivalents generated was quantified as the difference the samples with and without horseradish peroxidase. Zhao et al. reported that aerosolized Fruit flavored e-liquid generated three times more total ROS and H2O2 compared with aerosolized Tobacco flavor e-liquid. Based on these and other experimental results presented, the authors concluded that aerosolized e-liquids may contain ROS precursors that can generate more ROS upon deposition in the lung and phagocytosis by macrophages (J. Zhao et al., 2018). Iskander et al. reported no difference in oxidative stress from exposure to an aerosolized flavored (not specified) e-liquid compared with an unflavored e-liquid using a submerged monoculture of HBE cells and an ALI system with SmallAir™ (human small airway) 3D tissue model (Iskandar, Zanetti, Marescotti, et al., 2019). Bitzer et al. screened 49 laboratory-prepared flavored e-liquids for oxidative capacity and reported that a vanilla flavored e-liquid generated less ROS compared with an unflavored PG/VG humectant mixture in a cell-free system (collected in alpha phenyl-N-tert butyl nitrone [PBN] spin trap followed by electron paramagnetic resonance analysis), but 20 flavored e-liquids generated significantly higher ROS compared with the humectant mixture (Bitzer et al., 2018). A recent study characterized the metals content and oxidative capacity of aerosolized JUUL® Fruit Medley flavored e-liquid (no longer sold) and two nicotine-containing e-liquids from first generation e-cigarettes (Pearce et al., 2020). Aerosol from Mistic and Logic Power first generation e-cigarette nicotine e-liquids had higher concentrations of total metals and generated more ROS in HBE cells compared with aerosolized JUUL® Fruit Medley e-liquid. At the highest dose tested (25 puffs), all aerosolized e-liquids caused oxidative stress (measured as reduction in HBE cellular glutathione levels). Results were not compared with aerosolized humectant-only e-liquid exposures nor aerosolized nicotine-free e-liquid exposures, as such, it is difficult to disentangle the relative influence of flavorings, metals, and nicotine on reported results from this study.
Studies of flavoring chemicals used in e-liquids have demonstrated the capacity of individual flavorings and mixtures of flavorings to generate ROS. As noted above, Bitzer et al. measured ROS production of 10 flavorings in a cell-free system. Each flavoring was dissolved in PG/VG and aerosolized using an e-cigarette. Ethyl vanillin PG acetal, ethyl vanillin, β-damascone, and δ-tetradecalactone were negatively correlated with ROS levels, which suggested an inhibitive effect. Six flavorings (γ-decalactone, citral, ethyl maltol, piperonal, d-limonene, and linalool) were positively correlated with ROS levels, which indicated that they contributed to increased radical production. Further, linalool, piperonal, and citral caused significant increases of lipid peroxidation products (Bitzer et al., 2018). Muthumalage et al. measured ROS generation from seven flavoring chemicals using a cell-free system (2’,7’dichlorofluorescein dye method). Diacetyl, cinnamaldehyde, maltol, o-vanillin, and coumarin flavorings generated ROS in a concentration-dependent manner whereas acetoin and 2,3-pentanedione only generated ROS at highest tested concentration (Muthumalage et al., 2018). As noted earlier in this section, the oxidative burst capacity of immune cells such as neutrophils is a powerful component of the body’s first line of defense against inhaled foreign material. Hickman et al. measured the impact of flavoring chemicals on the oxidative burst capacity of primary human neutrophils isolated from venous blood. They reported that cinnamaldehyde and ethyl vanillin most attenuated oxidative burst, benzaldehyde and benzaldehyde PG acetal attenuated the response at higher tested concentrations, and isoamyl acetate did not affect the response (Hickman, Herrera, & Jaspers, 2019). As described in Section 7.1.1, the tobacco cigarette industry has proposed a three-tiered systems toxicology approach to evaluation of e-liquids. The second step of their approach was evaluation of the mechanisms of toxicity of e-liquids. Marescotti et al. evaluated the impact of 28 flavoring chemicals, singly or in mixtures, using laboratory prepared e-liquids and human bronchial epithelial cells. Of the 28 flavorings evaluated singly, two, citronellol and α-pinene were reported to induce increased oxidative stress (Marescotti et al., 2020).
Smoking tobacco cigarettes is associated with tooth loss and destruction of connective tissue and matrix, which may lead to risk of development of periodontitis (Sundar, Javed, Romanos, & Rahman, 2016). According to Sundar et al., upon inhalation, aerosolized e-liquid will first contact tissue in the oral cavity and head-airways region so they evaluated whether flavored e-liquids would adversely impact human periodontal ligament fibroblast (HPdLF) cells and human gingival fibroblasts and primary gingival epithelial tissue (EpiGingival™ 3D tissue model) using an ALI system (Sundar et al., 2016). They reported that aerosolized Magnificent Menthol and Classic Tobacco flavored e-liquids increased secretion of the proinflammatory cytokine IL-8 in HPdLF cells, which indicated that oxidants overwhelmed cellular antioxidant defenses. Further, aerosolized Magnificent Menthol flavored e-liquid increased release of the inflammatory markers COX-2, S100A8, and RAGE and aerosolized Classic Tobacco flavored e-liquid increased release of S100A8 in HPdLF cells. When aerosolized, both flavored e-liquids increased secretion of PGE2, an inflammatory marker, in EpiGingival™ tissue. Based on their data, the authors highlight the pathologic role of aerosolized flavored e-liquids on cells and tissue in the oral cavity (Sundar et al., 2016); however, some caution is warranted in this interpretation because nicotine levels differed between flavored e-liquids. Iskander et al. reported no difference in oxidative stress from exposure to a aerosolized flavored (not specified) e-liquid compared with an unflavored e-liquid using an ALI system with EpiOral™ (human oral mucosal) 3D tissue model (Iskandar, Zanetti, Marescotti, et al., 2019). Lipid peroxidation is implicated in the pathogenesis of periodontal disease. In the only study to evaluate the oxidative capacity of aerosolized flavored e-liquids in humans, Menicagli et al. measured salivary malondialdehyde as a marker of lipid peroxidation. The authors reported that levels of salivary malonidialdehyde were significantly higher in e-cigarette users who aerosolized a tobacco-flavored e-liquid without nicotine, which suggested oxidative damage (Menicagli, Marotta, and Serra, 2020). Collectively, the results of most studies indicate that flavored e-liquid aerosol can disrupt cell function via oxidative stress pathways, which may result in oral diseases.
As inhaled aerosol from an e-cigarette penetrates past the head-airway region and into the tracheobronchial and alveolar regions, there is further opportunity for adverse impacts on oxidant homeostasis in lung cells. IL-8 functions as a chemoattractant for inflammatory cells such as neutrophils and is a well-established biomarker of oxidativestress-mediated inflammation and tissue damage in the lung. The literature on IL-8 secretion in response to exposure to flavored e-liquids and flavoring chemicals is conflicting. Misra et al. observed that IL-8 levels in A549 human lung epithelial cells were lower following exposure to flavored e-liquids compared with other tobacco product extracts and that IL-8 was released by cells exposed to aerosolized flavored e-liquids (Misra et al., 2014). Clapp et al. reported that Sini-cide (cinnamon flavored) e-liquid significantly suppressed IL-8 secretion by human alveolar macrophage cells (Clapp et al., 2017). In another study, 36 flavored e-liquids and seven flavoring chemicals were evaluated, and it was reported that specific flavored e-liquids and acetoin flavoring suppressed IL-8 secretion by human pleura/pleural lymphocyte (U937) cells (Muthumalage et al., 2018). Czekala et al. did not observe any change in IL-8 levels in EpiAirway™ (human lung) 3D tissue model following exposure to aerosolized Blueberry flavored e-liquid compared with an unflavored e-liquid (Czekala et al., 2019). In contrast, it has been reported that IL-8 release was increased in human lung fibroblast (HFL-1) cells exposed to Cinnamon Roll and other flavored e-liquids (Lerner et al., 2015) as well as an e-liquid that contained a mixture of tobacco, coconut, vanilla, and cookie flavors (Lucas et al., 2020); human lung epithelial (A549) cells exposed to aerosolized Cinnamon flavored e-liquid (Bengalli et al., 2017); human neutrophils isolated from venous blood exposed to Kola, Hot Cinnamon Candies, Banana Pudding, Menthol Tobacco and Banana flavored e-liquids (Clapp et al., 2017); human lung bronchus (Beas-2B) cells exposed to acetoin, diacetyl, maltol, and o-vanillin flavorings and HFL-1 cells exposed to acetoin, pentanedione, maltol, and o-vanillin (Gerloff et al., 2017); THP-1 human monocytes from isolated peripheral blood exposed to three Apple flavored e-liquids (Ween et al., 2017); U937 cells exposed to cinnamaldehyde, diacetyl, 2,3-pentanedione, o-vanillin, maltol, and coumarin flavorings; and, human blood monocyte (MM6) cells exposed to acetoin, cinnamaldehyde, and o-vanillin flavorings (Muthumalage et al., 2018). In addition to IL-8, other proinflammatory molecules that are secreted or suppressed in response to exposure to flavored e-liquids or flavoring chemicals include IL-1β and IL-6, IL-10, chemokine (C-X-C motif) ligand (CXCL) 1, CXCL2 and CXCL10 (Clapp et al., 2017; Czekala et al., 2019; Gómez et al., 2020; Leigh et al., 2016; Ween et al., 2017; Ween et al., 2020); monocyte chemotactic protein (MCP)-1 (Bengalli et al., 2017; Ween et al., 2017); and tumor necrosis factor (TNF)-α, macrophage inflammatory protein (MIP)-1a, and MIP-1b (Ween et al., 2017; Ween et al., 2020).
Collectively, the available body of literature has demonstrated that ROS generation and secretion of proinflammatory signaling molecules following exposure to flavored e-liquids and flavoring chemicals is substance-specific and may be suppressed, unchanged, or increased depending upon the e-liquid composition or flavoring chemical. Additionally, the same flavoring did not induce cytokine secretion in different cell types, which indicated that the choice of cell line is an important consideration in study design.
7.1.3. Impairment of mucociliary clearance
Mucociliary clearance refers to the movement of foreign material that is deposited in the mucous layer that covers the respiratory tract epithelium via the beating of cilia. This mechanism is sometimes referred to as the “mucociliary escalator.” The ciliated portion of the respiratory tract extends from the nose through the tracheobronchial region but excludes the alveolar region. Each cilia of the respiratory tract beats in a coordinated fashion at the same frequency but in a phase-shifted manner with its neighbors, which has the net effect of generating a wave that travels across the epithelium and propels the overlying mucus layer (Bustamante-Marin & Ostrowski, 2017). In the nose, the mucus layer is propelled by ciliary action toward the pharynx whereas in the tracheobronchial region the mucus layer is propelled by cilium toward the larynx, where it is swallowed and excreted via the gastrointestinal tract. Mucociliary clearance is a key respiratory defense and impairment of ciliary beating could increase the risk of respiratory infections in e-cigarette users.
Histological analysis of EpiAirway™ human lung 3D tissue exposed to aerosolized Blueberry flavored e-liquid and aerosolized unflavored e-liquid using an ALI system revealed no effect on cilia morphology (Czekala et al., 2019). Consistent with this observation, Iskander et al. reported that exposure to aerosolized flavored (unspecified) e-liquid and aerosolized unflavored e-liquid did not impact cilia beating frequency in SmallAir™ (human small airway) and EpiOral™ (human mucosal) 3D tissue models using an ALI system (Iskandar et al., 2019).
Sherwood and Boitano evaluated the impact of 2,5-dimethylpyrazine flavoring on HBE cells with a high-capacity real-time cell analysis approach. Cells were treated with non-cytotoxic concentrations of 2,5-dimethylpyrazine, followed by exposure to either forskolin (to raise cyclic adenosine monophosphate) or exogenous adenosine triphosphate (to raise intracellular Ca2+ concentration). Exposure to both compounds resulted in a concentration-dependent reduction in physiological response, which indicated a change in signaling molecules important for maintenance of mucociliary clearance (Sherwood & Boitano, 2016). Aldehydes in regular tobacco cigarette smoke are known to reduce ciliary beat frequency, and in turn, diminish mucociliary clearance (Clapp et al., 2019). To evaluate whether the same effects would be seen from aerosolized flavorings used in e-liquids, Clapp et al. exposed HBE cells to Kola, Hot Cinnamon Candies, and Sini-cide flavored e-liquids that contained cinnamaldehyde flavoring. All bulk and aerosolized flavored e-liquids transiently suppressed cilia beat frequency (Clapp et al., 2019). One research group took a transcriptomic approach to evaluate the influence of flavorings on mucociliary clearance. The authors exposed HBE cells to authentic standards of diacetyl or 2,3-pentanedione using an ALI system and identified 163 and 568 differentially expressed genes, respectively. Of these genes, 142 were common to both flavorings; expression of several genes significantly downregulated production of cilia. Further, exposure to these flavoring chemicals significantly decreased the number of ciliated cells, which indicated the potential to impair mucociliary clearance (Park et al., 2019).
7.1.4. Impairment of cell-mediated clearance
Macrophages, neutrophils, and Natural Killer (NK) cells form part of the body’s first line of defense against foreign material that is deposited throughout the respiratory tract, including the non-ciliated alveolar region. Failure of these innate immune cells to perform their functions may leave a person susceptible to infection and/or contribute to impaired resolution of inflammation (Clapp et al., 2017). For example, airway macrophages and neutrophils from regular tobacco cigarette smokers and persons with chronic obstructive pulmonary disease have impaired ability to phagocytize bacteria, which can make them susceptible to development of pneumonia (Hickman et al., 2019; Ween et al., 2017). In one study, Kola and Sini-cide flavored e-liquids impaired macrophage phagocytic capacity (Clapp et al., 2017). Aerosolized Apple and Irish Cream flavored e-liquids reduced macrophage (THP-1 human monocytes from peripheral blood)-mediated phagocytosis of bacteria (Gómez et al., 2020; Ween et al., 2017), an effect that was demonstrated to be related to reduced expression of the bacteria phagocytosis recognition receptor (SR)-A1 on macrophage cell surfaces (Ween et al., 2017). Clapp et al. reported that Hot Cinnamon Candies, Banana Pudding, Menthol Tobacco, Banana, and Sini-cide flavored e-liquids (but not Kola or Solid Menthol flavored e-liquids) reduced neutrophil phagocytosis in a dose-dependent manner (Clapp et al., 2017). Hickman et al. evaluated the influence of common aldehyde e-liquid flavorings on neutrophil functioning and reported that cinnamaldehyde (cinnamon), ethyl vanillin (vanilla), and benzaldehyde and benzaldehyde PG acetal (cherry or almond) impaired phagocytosis but isoamyl acetate (banana) had no effect (Hickman et al., 2019). Clapp et al. isolated NK cells from venous blood and exposed them to flavored e-liquids and reported that Cinnamon flavored e-liquids suppressed NK cell killing of target cells, which suggested that failure of NK cells in the lung to perform their immune functions may leave a person susceptible to infection (Clapp et al., 2017). However, caution is warranted in this conclusion for NK cells isolated from the peripheral blood because these cells constituted a subset referred to as conventional NK cells that exhibited numerous functional and phenotypical differences from resident NK cells in lung tissue (Cong & Wei, 2019). In the only in vivo study to evaluate the impact of aerosolized e-liquids on innate immune cells in the lung, Werley et al. exposed female Crl:CD(SD) rats for 90 days via nose-only inhalation to aerosol produced using a tobacco cigarette industry prototype e-cigarette with a flavored e-liquid (specific flavor not identified) and an unflavored e-liquid with the same composition. Rats in the high flavored e-liquid exposure group had higher alkaline phosphatase, lactate dehydrogenase, and total protein values compared with rats in the low- and mid- flavored e-liquid exposure groups, which suggested general cytotoxicity to lung cells. Levels of these parameters in the high flavored e-liquid exposure group did not differ from the high-exposure vehicle control group. The authors reported at 28 and 90 days, the high flavored e-liquid exposure group had higher proportions of neutrophils than the low- and mid-exposure groups; however, there was no difference in total and differential cell counts of alveolar macrophages, neutrophils, lymphocytes, eosinophils, or total cell count compared with control animals, which suggested no impact on cellular clearance (Werley et al., 2016).
Apoptosis, or programmed cell death, is a normal physiological function whereby cells turnover and are cleared away by alveolar macrophages to prevent build-up of apoptotic debris, which can cause pulmonary inflammation. It has been reported that regular tobacco smokers and persons with chronic obstructive pulmonary disease have a higher rate of apoptosis of airway cells and reduced ability of alveolar macrophages to clear apoptotic debris (a process sometimes referred to as “efferocytosis”) (Ween et al., 2020). Using the same Apple flavored e-liquids from their prior study, Ween et al. reported that all aerosolized e-liquids increased necrosis and apoptosis (in HBE cells), decreased phagocytosis of apoptotic debris (in THP-1 macrophage cells), and reduced surface expression of CD36 receptor, a key apoptotic cell recognition receptor on macrophages, which suggested that, like regular tobacco cigarettes, aerosolized e-liquids could also impair efferocytosis (Ween et al., 2020). In a series of studies by the regular tobacco cigarette industry to evaluate their own e-cigarette products using a three-tiered systems toxicology approach, results were reported on the mechanism of toxicity for e-liquids (Iskandar, Zanetti, Marescotti, et al., 2019; Marescotti et al., 2020). Iskander et al. reported there was no difference in protein markers of autophagy for aerosolized flavored (unspecified) e-liquid compared with aerosolized unflavored e-liquid in SmallAir™ and EpiOral™ 3D tissue models. Marescotti et al. evaluated the influence of specific flavoring chemicals on HBE cells and reported that citronellol, α-pinene, and linalool in e-liquids triggered signs of apoptosis via activated caspase 3 and caspase 7 activity.
Under normal conditions, respiratory tract epithelial cells form junctions that provide a protective barrier to prevent penetration of foreign material into blood (Gerloff et al., 2017). Bengalli et al. used an in vitro reconstructed ABB co-culture composed of alveolar lung epithelial cells (NCI-H441) which are directly exposed to e-cigarette aerosol and basal lung microvascular endothelial cells (HPMEC). This co-culture was grown while measuring transepithelial electrical resistance, and when it reached a maximum value, which indicated that cell junctions were fully differentiated and the epithelial barrier was fully functional, cells were exposed to aerosolized Cinnamon, Tobacco (2 brands), and Menthol (2 brands) flavored e-liquids. ABB integrity was significantly affected by exposure to aerosolized Cinnamon and one brand of Menthol flavored e-liquids. The aerosolized Cinnamon flavored e-liquid appeared to affect barrier integrity via reduced viability of ABB cells, whereas the aerosolized Menthol flavored e-liquid appeared to have acted via loss of cell junction integrity (Bengalli et al., 2017). In contrast, Czekala et al. exposed EpiAirway™ human lung 3D tissue model to aerosolized Blueberry flavored e-liquid and reported that it did not alter tissue barrier function compared with aerosolized unflavored e-liquid (Czekala et al., 2019). Other investigators have focused on the impact of flavoring chemicals on epithelial barrier function. Results of one study indicated that diacetyl, coumarin, acetoin, maltol, and cinnamaldehyde significantly impaired HBE cell barrier function over time (Gerloff et al., 2017). Another study reported that 2,5-dimethylpyrazine disrupted mouse tracheal epithelial cell barrier function (Sherwood & Boitano, 2016).
Cellular Ca2+ levels help to maintain epithelial lung cell homeostasis (and modulate immune cell activation) and control the initiation and persistence of inflammation. Levels of Ca2+ are modulated using cellular pumps, receptors, and channels but activation of these Ca2+ signaling pathways by exogenous substances such as flavored e-liquids could alter cell function, which may result in chronic inflammation or abnormal cell growth (Rowell et al., 2020). To evaluate the effects of flavored e-liquids on Ca2+ signaling, Clapp et al. exposed CALU3 human lung epithelial cells to 100 flavored e-liquids; 42 of these products elicited a cellular Ca2+ response. Modeling indicated that the response to e-liquids was associated with the number of flavoring in products; ethyl maltol, ethyl vanillin, and vanillin were major contributors to responses. One product, Banana Pudding flavored e-liquid was selected as a representative for further testing and it increased cytosolic Ca+2, induced endoplasmic reticulum Ca+2 release, and increased total inositol phosphate production in CALU3, HBE, and HEK cells. Next, CALU3 lung epithelial cells were directly exposed to aerosolized Banana Pudding flavored e-liquid that caused a persistent increase in cytosolic Ca2+. Based on their data, the authors concluded that: 1) multiple flavored e-liquids affect cellular Ca2+ homeostasis independent of nicotine; and, 2) the more chemicals that a flavored e-liquid contained, the more it affected Ca2+ signaling. This latter conclusion is consistent with other studies that observed increased toxicity responses were associated with the number of flavorings in e-liquids (Bahl et al., 2012; Sassano et al., 2018).
Szafran et al. exposed female C57BL/6 mice to filtered air, 70%/30% VG/PG humectant mixture, or 70%/30% VG/PG mixture with vanilla flavoring. In that study, mice exposed to VG/PG with vanilla flavoring exhibited (at a methacholine challenge dose of 25 mg/mL) increased lung tidal volume and minute volume and increased maximum tissue damping (indicator of lung tissue resistance). Exposure to the VG/PG humectant mixture and VG/PG mixture with vanilla flavoring did not yield significantly different numbers of innate immune alveolar macrophage, interstitial macrophage, or neutrophil cells compared with air exposed controls, though exposed mice had increased counts of adaptive immune response dendritic, CD4+ T, and CD19+ B cells. Levels of IgG1 and IgG2b were not altered in serum or BALF from animals exposed to the VG/PG mixture; however, there was a significant increase in IgG1 levels in the BALF of animals exposed to VG/PG mixture with vanilla flavoring compared with air controls. There was an increase in the lung immune lipid mediators 2-arachidonoylglycerol and in 12-hydroxyeicosatetraenoic acid independent of vanilla flavoring. Collectively, their results suggested that PG/VG humectant, with and without vanilla flavoring, can disrupt immune homeostasis (Szafran et al., 2020).
7.2. Cardiovascular and circulatory systems
Among the target organs covered in this review, only the cardiovascular and circulatory systems have been studied using in vitro, in vivo, and human models. In the first study to investigate the effects of flavored e-liquids on the cardiovascular system, Farsalinos et al. screened 20 products for cytotoxic potential in vitro using H9c2 myocardial fibroblast cells and an early generation low voltage e-cigarette. At 6.2 W power, only aerosolized Cinnamon and Cookies flavored e-liquid was cytotoxic at the highest tested concentration. Subsequently, a subset of the flavored e-liquids was aerosolized using an e-cigarette and the authors reported that aerosols produced at 6.2 W or 9.2 W were not cytotoxic to myocardial fibroblasts (Farsalinos et al., 2013).
Smoking regular tobacco cigarettes is known to cause endothelial dysfunction, which is a predictor of increased cardiovascular disease risk (Fetterman et al., 2018). Endothelial dysfunction is an umbrella term that includes dysregulation of endothelial cell functions, alterations in cellular tube formation, and impacts on blood vessel formation (angiogenesis). Investigators have begun to ask whether the use of e-cigarettes may also be associated with endothelial cell damage leading to endothelial dysfunction. Putzhammer et al. evaluated the toxicity of 11 different flavored e-liquids that were aerosolized by first and second generations of e-cigarettes. Using human umbilical vein endothelial cells, the authors reported that aerosolized Berry and Herbal flavored e-liquids were highly cytotoxic, significantly reduced cellular proliferation, caused morphological alterations, and disrupted the endothelial monolayer, but did not generate appreciable intracellular ROS. The authors concluded that e-cigarette device was an important factor in observed outcomes because the highest toxicity of all tested e-liquids was from refillable second generation e-cigarettes and first generation disposable e-cigarette products seemed to be less toxic (Putzhammer et al., 2016). Unfortunately, it was unclear if the same e-liquid formulations were used in all devices, which partially obscures this possible relationship between e-cigarette device and toxicity. Nystoriak et al. exposed human induced pluripotent stem cell (hiPSC)-derived cardiac myocytes (muscle cells) in vitro to liquid cinnamaldehyde flavoring or to heated cinnamaldehyde flavoring and its thermal degradation products at conditions intended to mimic use in e-cigarettes. Liquid cinnamaldehyde flavoring altered hiPSC-MC contraction-dependent signal amplitude, beating rate, and cell morphology at non-cytotoxic concentrations, and with prolonged exposure caused time-dependent dysregulation of cell membrane potential. Interestingly, when hiPSC-MC were exposed to heated cinnamaldehyde aerosol, the observed effects were attenuated. Based on these observations, the authors suggested that heating cinnamaldehyde by itself did not directly lead to the formation of products with greater cardiotoxicity and therefore, testing a diluted e-liquid in vitro may not be as informative as testing the aerosolized form (Nystoriak et al., 2019). Nitric oxide (NO) is a signaling molecule in the cardiovascular system. The loss or impairment of NO signals and release of IL-6 may yield inflammatory conditions that result in vascular dysfunction and atherosclerotic plaque formation. Fetterman et al. evaluated the in vitro toxicity of nine flavorings (vanillin, menthol, cinnamaldehyde, eugenol, dimethylpyrazine, diacetyl, isoamyl acetate, eucalyptol, and acetylpyrazine) on human aortic epithelial cells (Fetterman et al., 2018). When heated, all tested flavorings impaired NO production, possibly via ROS scavenging of NO, which suggested they could induce endothelial cell dysfunction. Additionally, many of the flavorings upregulated IL-6, a proinflammatory cytokine. Lee et al. used iPSC-derived endothelial cells (ECs) to assess the potential impact of six flavored e-liquids on endothelial integrity (Lee et al., 2019). In this study, Menthol and Cinnamon flavored e-liquids significantly decreased iPSC-EC viability via increased caspase 3 and caspase 7 activity and shortened tube formation, the latter which is relevant to angiogenesis. Butterscotch, Menthol, and Cinnamon flavored e-liquids generated the highest levels of intracellular ROS, a widely implicated risk factor in endothelial injury. Cinnamon and Caramel/Vanilla flavored e-liquids increased uptake of low-density lipoproteins and free fatty acids by iPSCECs, which demonstrated a link between the onset of cellular inflammation and impaired endothelial function. Noël et al. reported that consumers can purchase “e-concentrates” that consist of concentrated PG or flavorings to dilute and mix their own desired flavored e-liquid at home. The authors analyzed 34 flavored e-concentrates and 21 flavored e-liquids for their ingredients and tested the cytotoxicity for subset of identified flavoring ingredients using human umbilical vein endothelial cells. Cinnamaldehyde, cinnamaldehyde PG acetal, vanillin, limonene, eugenol, estragole, and anethole flavorings were all cytotoxic, with cinnamaldehyde being the most potent (Noël, Rainer, Gstir, Rainer, & Bonn, 2020). Collectively, these studies indicate that certain flavored e-liquids and flavoring chemicals may cause endothelial dysfunction similar to smoking regular tobacco cigarettes.
One study attempted to reveal the effects of e-cigarettes on the redox state of a human endothelial cell line (EA.hy926 cells) using three Tobacco flavored, two Apple/Mint flavored, and two Vanilla flavored e-liquids (Kerasioti et al., 2020). All flavored e-liquids were cytotoxic to EA.hy926 cells. Next, the authors evaluated the effects of these e-liquids on redox biomarkers: one Vanilla flavored and one Apple/Mint flavored e-liquid increased GSH (reduced form of glutathione) levels; all Tobacco and one Vanilla flavored e-liquid increased ROS production and thiobarbituric acid reactive substance levels; none of the flavored e-liquids influenced total antioxidant capacity or protein carbonyl levels. Overall, the results demonstrated an alteration in cellular redox balance in favor of free radicals in Tobacco and Vanilla flavored e-liquids whereas the Apple/Mint flavored e-liquids appeared to activate cellular antioxidant defenses to protect cells.
Berkelhamer et al. evaluated the effect of flavored e-liquids on fetal, neonatal, and adult lung artery smooth muscle cells. This in vitro study revealed that Menthol and Strawberry flavored e-liquids induced cytotoxicity in all smooth muscle cells; neonatal cells were most sensitive to Menthol flavoring-induced cytotoxicity. The authors also evaluated changes in vasoreactivity using isolated ovine pulmonary arteries – Menthol flavored e-liquid induced relaxation of adult, but not neonatal, pulmonary artery cells (Berkelhamer et al., 2019).
The cardiovascular effects of flavored e-liquids have been studied in vivo using mouse and rat models (Table 3). In the only in vivo study to evaluate JUUL® e-liquids, Rao et al. exposed male and female Sprague-Dawley rats via nose-only inhalation to aerosol from a JUUL® pod mod e-cigarette with Virginia Tobacco flavored e-liquid (nicotine salt), aerosol from an earlier generation tank-style e-cigarette with unflavored e-liquid (freebase nicotine), and smoke from a regular tobacco cigarette. All exposures impaired endothelial function, assessed as arterial flow-mediated dilation, which is a measure for overall cardiovascular health; differences were not significant among exposures. Rats exposed to JUUL® aerosol had higher serum nicotine compared with rats exposed to the same number of puffs from a tank-style e-cigarette or tobacco cigarette. Serum cotinine levels were comparable between rats exposed to JUUL® aerosol and tobacco cigarette smoke (Rao, Liu, & Springer, 2020). The JUUL® e-liquid differed in PG/VG content and the amount and form of nicotine compared with the tank-style e-cigarette, whichprecludedanydefinitiveconclusiononthespecificroleofflavoring in explaining the observed responses. Werley et al. exposed Crl:CD(SD) rats via nose-only inhalation to aerosolized flavored e-liquid (specific flavor not identified) and to aerosolized unflavored e-liquid with the same composition. The authors reported there were no differences in plasma nicotine and cotinine levels between exposures, which suggested little influence of e-liquid flavor on nicotineuptake (Werley et al., 2016); however, missing information on the specific flavoring(s) in the e-liquid limits the certainty of this conclusion. In another in vivo study, Conklin et al. investigated potential biomarkers that could be used to differentiate e-cigarette exposure from other tobacco product exposure such as regular tobacco cigarette smoke. The authors exposed male C57BL6/J mice via whole body inhalation to aerosolized flavored e-liquids and their thermal aldehyde degradation products. One-hour post exposure, urinary excretion of the acrolein metabolite 3-hydroxypropyl mercapturic acid and nicotine alkaloids were significantly higher for mice that inhaled aerosolized Magnificent Menthol flavored e-liquid compared with aerosolized Classic Tobacco flavored e-liquid (Conklin et al., 2018). Szostak et al. exposed female ApoE−/− mice via whole body inhalation to aerosolized laboratory prepared e-liquid that contained guaiacol and other unspecified flavorings and to aerosolized non-flavoring e-liquid constituents. There was no difference in atherosclerosis progression, cardiovascular function, and molecular changes in the heart and aorta for the aerosolized e-liquid that contained guaiacol compared with the aerosolized non-flavoring constituents (Szostak et al., 2020).
Flavorings in e-liquids may influence nicotine pharmacokinetics from e-cigarettes and enhance the reward sensation from vaping. Given these potential impacts of flavor, the effect of flavored e-liquids on nicotine uptake in humans has been evaluated by researchers using a cross-over study design with diverging results. In the first study, 24 male human volunteers aerosolized a Menthol flavored e-liquid or an unflavored e-liquid with the same humectant and nicotine composition using a first-generation e-cigarette. Participants that aerosolized the unflavored e-liquid had significantly higher maximum plasma nicotine concentration and nicotine uptake (area under the curve) compared with those who aerosolized the Menthol flavored e-liquid, which indicated that the presence of Menthol decreased nicotine uptake or elimination from body (Walele, Sharma, Savioz, Martin, & Williams, 2016). In the other cross-over study, 11 males and three females aerosolized Strawberry or Tobacco flavored e-liquids. For a defined puff regimen, participants that aerosolized Strawberry flavored e-liquid had higher nicotine uptake and increased heart rate compared with those who aerosolized Tobacco flavored e-liquid. When permitted to vape ad libitum, participants that aerosolized Strawberry flavored e-liquid had higher plasma nicotine concentration and nicotine uptake compared with those who aerosolized Tobacco flavored e-liquid. Based on these findings, the authors suggested that differences in nicotine uptake were attributable to the less basic pH of the Strawberry flavored e-liquid (pH 8.3) compared with the Tobacco flavored e-liquid (pH 9.1) (St Helen, Dempsey, Havel, Jacob 3rd, & Benowitz, 2017). Given the limited number of flavors tested to date, a firm conclusion cannot yet be made with regards to the impact of aerosolized flavorings on nicotine pharmacokinetics of e-cigarette users. Buchanan et al. have reviewed preclinical and clinical data on the cardiovascular effects of e-cigarettes and the reader is referred to that article for more information on the topic (Buchanan et al., 2020).
7.3. Developmental effects
Smoking regular tobacco cigarettes is a preventable cause of adverse pregnancy outcomes such as increased risk of low birth weight, underdevelopment of organs, congenital anomalies, preterm birth, and other effects (Greene & Pisano, 2019). Pregnant women and women of reproductive age are increasingly using e-cigarettes as an alternative to regular tobacco cigarettes based on the premise that they pose less risk for adverse developmental effects (Greene & Pisano, 2019). A recent National Academies of Sciences, Engineering, and Medicine report concluded that there was “no available evidence” as to whether or not e-cigarettes affect pregnancy outcomes and there was “insufficient evidence” with regard to whether or not maternal e-cigarette use affects fetal development (Greene & Pisano, 2019). These conclusions were based on available literature at the time of their report and future research findings may necessitate revisiting these conclusions.
At the time of our literature search, mainly in vitro studies were available that implied risk of developmental toxicity of flavorings in e-liquids. Several in vitro studies indicated that flavored e-liquids and specific flavoring ingredients in the form of bulk e-liquid or aerosol generated by an e-cigarette were cytotoxic to neural stem cells and that cytotoxicity was higher for stem cells compared with adult differentiated cells (Bahl et al., 2012; Behar, Davis, Wang, et al., 2014; Behar et al., 2016; Behar, Wang, & Talbot, 2018; Atena Zahedi et al., 2019). It is important to note that cytotoxicity to stem cells is only an indirect indicator of possible developmental toxicity. Further, exposure pathway is an important study design consideration because embryonic cells would not be exposed directly to diluted e-liquid or inhaled aerosol.
One study evaluated the developmental impact of flavored e-liquids on bronchodilation of neonate and adult lung bronchial ring tissue (Berkelhamer et al., 2019). The authors reported that Menthol, Strawberry, Tobacco, and Vanilla flavored e-liquids induced bronchodilation of neonatal but not adult bronchial rings and suggested that newborns and infants that inhale secondhand flavored e-cigarette aerosols could be at risk of exaggerated exposure to a higher concentration of aerosol constituents compared with adults because of their increased delivery to the alveoli through dilated airways. Note that Berkelhamer et al. exposed bronchial ring tissue to diluted e-liquid that has chemical properties that differ from primary aerosol inhaled by an e-cigarette user, which in turn, differs from secondhand aerosol exhaled by a user (Marco & Grimalt, 2015; Pankow, Kim, Luo, & McWhirter, 2018; Papaefstathiou, Bezantakos, et al., 2020; Samburova et al., 2018) so any definitive link between induced bronchodilation and secondhand exposure to newborns and infants is yet to be determined.
One in vivo study reported that pregnant and neonate C57BL/6 mice exposed via whole-body inhalation to Tobacco flavored e-liquids with and without nicotine experienced significant reductions in hippocampal gene expression as well as in serum levels of IL-1b, IL-2, and IL-6 cytokines, which indicated risk to the developing nervous system; however, mice were not exposed to a flavoring-free e-liquid so it was unknown if flavorings influenced the observed effects (Zelikoff et al., 2018). More information on neurotoxicity of e-cigarettes in general can be found in a recent review (Ruszkiewicz et al., 2020).
Finally, it is important to note that the current review is focused on flavored e-liquids though so studies that focused only on unflavored e-liquids were beyond our scope. As reviewed by Greene & Pisano, there is evidence that pregnant mice exposed to aerosolized e-liquid with and without nicotine had increased pro-inflammatory cytokines in the lungs of their offspring and altered neurodevelopment, which suggested that they were induced by other components of the aerosol than nicotine. Another study reported that offspring of mice exposed to aerosol generated from an e-liquid without nicotine had abnormal levels of neuroregulatory gene expression, which suggested disturbancesin central metabolic regulation in offspring (Greene & Pisano, 2019). Whether the non-nicotine components responsible for these reported developmental effects were from exposure to flavorings is unknown. More information on the developmental toxicity of e-cigarettes in general can be found in a recent review article (Greene & Pisano, 2019).
7.4. Skeletal system
Use of e-cigarettes with flavored e-liquids is highly prevalent among middle and high school students and adults under 35 years (see Section 5.1). This age range is a critical time for skeletal development because more than half the human skeleton is formed during teenage years and peak bone mass is reached by the late teens to mid-thirties (Otero et al., 2019; Wavreil & Heggland, 2020). Osteoblasts are bone-forming cells that play an essential role in the mineralization of bone and the production of collagen type I, a major structural component of bone extracellular matrix. Inhalation of regular tobacco smoke is a risk factor for osteoporosis, and the Heggland laboratory questioned whether flavored e-liquids would also impair bone health. In their first study, they treated human MG-63 and Saos-2 osteoblast-like cells with diluted flavored e-liquids with or without nicotine (Otero et al., 2019). Exposure to e-liquids induced dose-dependent cytotoxicity independent of nicotine content; unflavored e-liquids were the least cytotoxic, followed by coffee and fruit flavored, menthol, and cinnamon flavored e-liquids (most cytotoxic). Next, the authors evaluated mRNA expression of two key osteoblast genes, RUNX2 and Col1a1, in MG-63 cells. Exposure to Irish Latte, Mango Blast, and Sweet Melon flavored e-liquids upregulated the expression of Col1a1 mRNA (but not RUNX2), which demonstrated the ability of some flavored e-liquids to alter osteoblast gene function. Mango Blast flavored e-liquid with or without nicotine significantly increased collagen type I protein expression. Next, the Heggland laboratory evaluated in more detail the capacity of diluted and aerosolized cinnamon flavored (Napalm, Cinn Candy) e-liquids to induce osteotoxicity (Wavreil & Heggland, 2020). Exposure to aerosolized Napalm and Cinn Candy flavored e-liquids induced significant cytotoxicity in MG-63 cells but did not alter collagen type I protein expression. MG-63 cells exposed to cinnamon flavored e-liquids or aerosolized cinnamon flavored e-liquids exhibited significantly increased ROS production. Collectively, results from these studies indicated that coffee, fruit, menthol, and cinnamon flavored e-liquids were cytotoxic and/or had capacity to alter Col1a1 gene expression in osteoblast-like cells. The capacity of cinnamon flavored e-liquids to induce cytotoxicity may be related to oxidative stress. In a study funded by the tobacco cigarette industry, Reumann et al. reported the only in vivo data on the effects of aerosolized flavored e-liquids on skeletal health. In this study, female mice were exposed via whole body inhalation to tobacco smoke, aerosolized humectants, aerosolized humectants with acids and nicotine, and aerosolized humectants with acids and nicotine and flavorings (not specified). Exposure to tobacco smoke and all variations of e-liquid constituents caused development of microcracks in cortical areas of bones, which suggested ongoing bone remodeling and potential reduction of bone stability (Reumann et al., 2020). Whether these findings have implications for people is unknown.
Another potential impact of sweet flavored e-liquids on the skeletal system is their cariogenic (cavity) potential in teeth. Kim et al. prepared e-liquids composed of humectants and nicotine with ethyl butyrate (pineapple), ethyl maltol (cotton candy), hexyl acetate (apple), and triacetin (velvety/smoky) flavorings or without flavoring chemicals. Streptococcus mutans (UA159) were exposed to aerosolized e-liquids on tooth enamel from donor teeth, which led to a two-fold increase in biofilm formation and up to a 27% decrease in enamel hardness compared with aerosolized unflavored e-liquids. Additionally, aerosolized flavored e-liquids that contained ethyl butyrate, hexyl acetate, and triacetin were associated with bacteria-initiated demineralization of enamel and ethyl maltol inhibited Streptococcus mutans growth and adhesion. The authors concluded that aerosolized e-liquids with these sweet flavoring chemicals had similar physiochemical properties as high-sugar gelatinous candies and acidic drinks and could increase cariogenic potential (Kim et al., 2018).
7.5. Allergenicity and irritation
The exact number of flavoring chemicals used in e-liquids is unknown, though one study reported that at least 210 distinct flavorings were used to create over 16,000 flavored e-liquids (Krusemann et al., 2021). Given the vast number of flavorings that are inhaled into the body and the potential for dermal exposure among e-cigarette users and those who work in vape shops and e-liquid production that handle e-liquids, it was somewhat surprising that only one study was identified that evaluated the allergenicity of e-liquids. In a study by the tobacco cigarette industry, Stevenson et al. used the Genomic Allergen Rapid Detection (GARD) testing strategy to predict and compare the potential of two commercial flavored e-liquids (Blu Cherry and an unspecified flavored product) and laboratory-prepared unflavored e-liquids to induce respiratory or skin sensitization (Stevenson et al., 2019). This testing strategy consisted of assays that measured changes in the transcriptional profiles of 200 genomic biomarkers that were relevant to respiratory (type I immediate IgE-mediated hypersensitivity) or dermal (type IV delayed cell-mediated type hypersensitivity) sensitization and a predictive model to classify substances. Based on the GARD assays, none of the flavored e-liquids were classified as respiratory sensitizers; however, both the Blu Cherry and the unspecified flavored e-liquids were classified as weak dermal sensitizers under European regulatory requirements. Some flavorings are known allergens; however, it is largely unknown if these pure flavoring chemicals can induce sensitization as a component of an e-liquid mixture (Stevenson et al., 2019). An important outcome of the research by Stevenson et al. was the demonstration that the GARD assays, originally developed to assess the sensitization potential of pure chemicals, showed promise to differentiate and broadly classify flavored e-liquid mixtures with regards to their capacity to induce sensitization. Additionally, in one study, it was reported that the average number of flavoring chemicals per flavored e-liquid was 10 (Krusemann et al., 2021), which indicated the potential for mixture effects and possible cross-reactivity among chemicals that share similar molecular structures. Hence, while application of the GARD assay to two flavored e-liquids showed initial promise, much work is needed to fully understand its applicability for widespread screening of complex e-liquids for sensitization capacity.
Asthmatics who smoke regular tobacco cigarettes are susceptible to the effects of inhaled substances during smoking and are known to have worse asthma control, need more unscheduled healthcare visits, and require greater medication (Chapman et al., 2019). Despite this association, little information exists on the effect of e-cigarette use on asthmatics. To address this knowledge gap, Chapman et al. exposed male and female Balb/c mice to house dust mite allergen and aerosolized flavored e-liquids over a period of 21 days. The number of macrophages in bronchial alveolar lavage fluid (BALF) was elevated in mice exposed to house dust mite and aerosolized Black Licorice flavored e-liquid (but not Kola, Banana Pudding, or Cinnacide aerosolized e-liquids) without nicotine. Exposure to aerosolized Black Licorice, Banana Pudding, and Cinnacide flavored e-liquids with 12 mg/mL nicotine suppressed macrophage and eosinophil cell counts compared with control (Kola product not tested). House-dust-mite-induced airway hyperresponsiveness was not altered by exposure to any aerosolized flavored e-liquid regardless of the presence or absence of nicotine. Only inhalation of aerosolized nicotine-free Banana Pudding flavored e-liquid altered airway remodeling as determined by measurement of soluble lung collagen content. Collectively, these results indicate that aerosolized flavored e-liquids without nicotine had significant but varied effects on features of allergic airways disease. This conclusion was important given the appeal of flavored e-liquids to youth (see Section 5.1) and the association between ever or current e-cigarette use and asthma in teenagers (Wills, Choi, & Pagano, 2020).
Non-immunologic responses to flavored e-liquids or flavoring chemicals may include respiratory irritation, which often serves as a sentinel warning of potential toxicity of an inhaled chemical. Irritation is manifest as responses from chemical activation of chemosensory receptors in airway-innervating nerves (Erythropel et al., 2019). The transient receptor potential ion channels, TRPA1 and TRPV1, are receptors for irritant aldehydes in the airways. TRPA1 is the major aldehyde-activated receptor that is activated by acrolein (an abundant aldehyde in tobacco cigarette smoke) as well as flavoring aldehydes such as cinnamaldehyde, benzaldehyde, and vanillin used in e-liquids. TRPV1, the vanilloid receptor, may also contribute to irritant effects from flavorings because it is activated by vanillin-related compounds such as capsaicin but responds poorly to free aldehydes (Erythropel et al., 2019). In one study, human embryonic kidney cells (HEK-293T) were transiently transfected with either human TRPA1 or TRPV1 plasmid DNA to express these respiratory cell irritation receptors. Next, commercial Vanilla (contained vanillin and ethylvanillin) and Cherry (contained benzaldehyde) flavored e-liquids were characterized for in situ formation of their PG acetals over time and laboratory prepared mixtures of benzaldehyde, cinnamaldehyde, citral, ethylvanillin, and vanillin were reacted with PG to form acetals. The flavored e-liquids and flavoring/PG mixtures were aerosolized using a first-generation e-cigarette and the PG acetals were observed to be stable, which indicated that a significant proportion of aldehyde PG acetals can reach the airways of e-cigarette users. Further characterization revealed that the PG acetals were stable in simulated lung fluid for days. All flavorings and their PG acetals activated the aldehyde-sensitive TRPA1 irritant receptors on HEK-293T cells. As expected, the free aldehydes benzaldehyde, ethylvanillin, and vanillin only weakly activated TRPV1 receptors at higher tested concentrations. Interestingly, their corresponding PG acetals provoked a robust activation of TRPV1 receptors at lower concentrations. Based on these data, the authors concluded that 1) aldehyde flavoring PG acetals are formed in situ in flavored e-liquids and persist when an e-liquid is aerosolized; 2) these PG acetals have the capacity to induce a stronger irritation response compared with the free parent aldehyde alone; 3) PG acetals may produce stronger sensory irritation effects than the free aldehydes alone; and 4) that the toxicological properties of PG acetals differed from the parent aldehyde flavorings and e-liquid constituents. The authors advocated for a standard approach to detect and characterize newly formed compounds in flavored e-liquids and their aerosol to assess toxicological effects (Erythropel et al., 2019).
7.5. Genotoxicity
Tobacco cigarette smoking is associated with various cancers of the body (U.S. Department of Health and Human Services, 2014). Based on this association, multiple research groups have evaluated the genotoxic potential of flavored e-liquids (Al-Saleh et al., 2020; Behar et al., 2016; Misra et al., 2014; Menicagli et al., 2020; Welz et al., 2016). Misra et al. were the first to evaluate genotoxicity and they used an in vitro micronucleus assay. This assay measures chromosome damage based on the cytokinesis-block technique. Briefly, this assay quantifies inhibition of actin filaments by cytochalasin B during cytokinesis and the formation of “daughter” cells to distinguish between undivided cells and cells that completed nuclear division (Kirsch-Volders et al., 2011). In the study by Misra et al., neither Classic Tobacco nor Magnificent Menthol flavored e-liquids were genotoxic in Chinese hamster ovary (CHO) cells. Behar et al. evaluated the genotoxicity of aerosolized cinnamaldehyde flavoring in a laboratory prepared e-liquid using the Comet assay. In this assay, cells are lysed, suspended in gel, and placed in an electrophoresis chamber. When an electrical field is applied to the chamber, intact cellular DNA remains stationary but damaged DNA migrates in the gel, which results in a figure shaped like a comet (i.e., undamaged DNA in the head and damaged DNA forming the tail). The extent of migration can be characterized as the percentage of DNA in the tail and olive tail moments (Welz et al., 2016). The percentage of tail DNA in the gel is a measure of the relative fluorescent intensity in the head compared with the tail. Olive tail moments represent the product of the relative amount of DNA that migrated in the gel and the median migration distance. The percentage DNA in the tail is a more robust indicator that enables inter-comparison of study data, whereas olive tail moments may not be comparable between studies (Welz et al., 2016). Results of Comet assays revealed that the percentage of cells with comet tails, comet tail length, and olive tail moments were significantly increased from exposure to aerosolized cinnamaldehyde flavoring at non-cytotoxic concentrations in HESC and HPF cells, but not A549 lung cells. When the cell culture media that contained cinnamaldehyde was replaced with fresh media, levels of genotoxic markers in HESC and HPF cells returned to baseline levels within 24 hours (Behar et al., 2016). Welz et al. noted that head and neck squamous cell cancer (HNSCC) is the seventh most common cancer worldwide and regular tobacco smoking was an important risk factor for developing HNSCC. Hence, this group evaluated the mutagenicity of two fruit flavored (Apple, Cherry) e-liquids and one Tobacco flavored e-liquid in oropharyngeal mucosa tissue cultures. DNA fragmentation assessed using the Comet assay was significantly increased in oropharyngeal tissue upon incubation with Apple and Cherry, but not Tobacco, flavored e-liquids. Based on their mutagenicity data, they concluded that some flavored e-liquids may present risk for development of HNSCC (Welz et al., 2016). In another study of HNSCC, Tsai et al exposed the Ca9-22 oral gingival squamous carcinoma cell line and CAL-27 human tongue squamous cell line to Cinnamon Red Hots (contains cinnamaldehyde) or Apple Juice flavored e-liquids with and without nicotine (Tsai et al., 2020). For Ca9-22 oral gingival cells, exposure to Cinnamon Red Hots flavored e-liquid increased invasiveness (measured as cell movement through a microporous membrane toward a chemoattractant in a Boyden chamber) with or without nicotine whereas exposure to Apple Juice flavored e-liquid decreased cell invasion independent of nicotine. For CAL-27 tongue cells, Cinnamon Red Hots flavored e-liquid decreased cell invasion with or without nicotine but for Apple Juice flavored e-liquid there was no difference with or without nicotine compared with control. Overall, these data indicated a flavor-dependent effect on the regulation of cell invasion in different squamous cell lines. RAGE, a pattern-recognition cell-surface receptor thought to be involved in the invasion of oral squamous cell carcinoma, was measured using immunofluorescence. Briefly, cells are incubated with an antibody against RAGE, the nuclei counterstained, and the cells evaluated by fluorescence microscopy. RAGE was increased in Ca9-22 cells and CAL-27 exposed to both flavored e-liquids and further potentiated by the presence of nicotine. For Ca9-22 oral gingival cells, both flavored e-liquids increased secretion of IL-1α levels but only Apple Juice flavored e-liquid increased IL-8 secretion; the presence of nicotine attenuated cytokine levels. For CAL-27 cells, both flavored e-liquids increased secretion of IL-1α, but IL-8 levels were only increased by Apple Juice flavored e-liquid.
Al-Saleh et al. evaluated 33 brands of flavored e-liquids, nearly all of which contained quantifiable levels of menthol flavoring. Several brands of e-liquids that contained menthol flavoring induced DNA damage as measured by tail movement (Comet assay) in CHO and TK6 (spleen) cells. The concentration of menthol flavoring in the e-liquids was positively correlated with DNA damage in CHO cells. Additionally, several brands of e-liquids that contained menthol flavoring induced chromosome breakage in TK6 cells (micronucleus assay) (Al-Saleh et al., 2020). Pearce et al. characterized the metals content and evaluated the genotoxicity of aerosolized JUUL® Fruit Medley flavored e-liquid (no longer sold) and two nicotine-containing e-liquids from first generation e-cigarettes. All aerosolized e-liquids induced single strand DNA breaks in HBE cells; aerosolized JUUL Fruit Medley and aerosolized first generation Logic Power e-cigarette nicotine e-liquids induced highest levels of DNA damage (Pearce et al., 2020). As noted in Section 7.1.2, effects were not compared with aerosolized humectant-only e-liquid exposures nor aerosolized nicotine-free e-liquid exposures, which precludes a clear relationship between any effects of flavorings on genotoxicity. Tang et al. exposed male FVB/N mice via whole-body inhalation to an e-liquid composed of PG/VG with nicotine; compared with controls (PG/VG vehicle only or filtered room air) mice exposed to the aerosolized e-liquid developed lung adenocarcinoma and bladder urothelial hyperplasia, which indicated that the e-liquid was genotoxic and acted as a lung carcinogen and a potential bladder carcinogen (Tang et al., 2019). Whether flavorings can influence the genotoxicity associated with development of lung adenocarcinoma and bladder urothelial hyperplasia is yet to be evaluated. In the only human study of genotoxicity, Menicagliet al. reported that volunteers who used a tobacco-flavored e-liquid without nicotine had significantly higher levels of micronucleus formation in oral buccal cells compared with air-exposed controls, which suggested a possible pathway for risk of oral cancers (Menicagli et al., 2020).
Collectively, these in vitro studies suggest a role for flavored e-liquids and cinnamaldehyde and menthol flavorings in genotoxic or mutagenic responses in several cell models. Only one study, that of Behar et al. (2016), exposed cells (HPF and HESC) to aerosolized flavoring, whereas the other investigators exposed cells to diluted flavored e-liquids. The potential for most cells to be directly exposed to bulk e-liquid is likely low (with the exceptions of accidental or intentional ingestion and contact with the skin). While screening for toxicity using diluted e-liquids has predictive value (Behar, Wang, & Talbot, 2018), future studies should also include exposure pathways that more closely reflect actual e-cigarette use conditions and expand the literature beyond fruit, tobacco, menthol, and cinnamon flavors to include other categories in the e-liquid flavor wheel such as dessert, coffee/tea, spices, etc. (Krüsemann, Boesveldt, de Graaf, & Talhout, 2019).
7.6. Skin toxicity
Dermal exposure to tobacco cigarette smoke is associated with decreased wound healing, skin cancer, psoriasis, eczema, and premature skin aging (Prieux, Eeman, Rothen-Rutishauser, & Valacchi, 2020); however, the effects of e-cigarette aerosol on the skin is largely unknown. In one of the earliest toxicology studies of e-cigarettes, Cervellati et al. investigated the effects of aerosolized Balsamic flavored e-liquid on HaCaT skin cells. Exposure increased LDH release in a time-dependent manner, which indicated cell damage; aerosolized unflavored e-liquid showed no effects. The morphology of cells exposed to aerosolized Balsamic flavored e-liquid exhibited increased vacuolization and alteration of cytoplasmic membrane that was not observed for aerosolized unflavored e-liquid. Finally, exposure to aerosolized Balsamic flavored e-liquid increased release of IL-8 and IP-10 but suppressed release of IL-6 (Cervellati et al., 2014). Note that in this study, the authors directly exposed skin cells to aerosolized flavored e-liquids that was reflective of mainstream aerosol inhaled into the respiratory tract, not secondhand aerosol that would contact the external layer of the skin or metabolized aerosol that would interact with the internal layers of the skin.
8. Toxicology of e-cigarette, or vaping, product use associated lung injury (EVALI)
The United States experienced an outbreak of lung injuries, later termed EVALI, that began in 2019, and as of February 20, 2020 had resulted in 2,807 hospitalizations and caused 68 deaths. The median age of patients (based on data as of January 14, 2020) was 24 years and nearly two thirds were male (www.cdc.gov/EVALI). There were 2,022 hospitalized patients who had data on substance use as of January 14, 2020; 82% reported using Δ9-THC-containing products, 33% reported exclusive use of Δ9-THC-containing products; 57% reported using nicotine-containing products, and 14% reported exclusive use of nicotine-containing products (www.cdc.gov/EVALI). Our literature searches for this review identified 33 peer-reviewed articles on EVALI for inclusion, though the vast majority (22/33) lacked toxicological data. Six articles provided results of e-liquid or constituent characterization or hypothesized an underlying toxicological mechanism for EVALI (Attfield et al., 2020; Blount et al., 2020; Chand et al., 2019; Muthumalage et al., 2020; Narimani & da Silva, 2020; Wu & O’Shea, 2020). Four articles evaluated the toxicity of common cannabis extract diluents on lung cells (Bhat et al., 2020; Jiang et al., 2020; Matsumoto et al., 2020; Muthumalage et al., 2020) and one article reported an EVALI-like condition in rat lungs from exposure to an e-liquid that did not contain Δ9-THC, VEA, or nicotine (Kleinman et al., 2020). For a detailed review of in vivo studies of EVALI, the reader is referred to a recent review article (Feldman, Stanton, & Suelzer, 2021).
8.1. Possible causative agents
Approximately two years prior to the EVALI outbreak, Troutt and DiDonato heated four thinning agents used for cannabis e-liquids (PG, VG, polyethylene glycol 400, and medium chain triglycride oils [MCT]) to 230 °C and assessed formation of toxic carbonyls. Polyethylene glycol 400 and PG produced formaldehyde and/or acetaldehyde at higher levels compared with VG and MCT (Troutt & DiDonato, 2017). None of these aldehydes produce the pathology observed with EVALI, but the data provided valuable insights during the outbreak because it suggested that other ingredients could be involved in the toxic response. As part of the EVALI investigation, Blount et al. obtained BALF from 51 EVALI patients in 16 states and measured the concentrations of several possible toxic substances, including Δ9-THC and the e-liquid constituents VEA, plant oils (identified by measuring long-chain triglycerides), medium-chain triglyceride oil, coconut oil (identified by measuring medium-chain triglycerides), petroleum distillates, and terpenes such as limonene (Blount et al., 2020). The authors reported that 94% of EVALI patients had BALF samples with detectable Δ9-THC or its metabolites or reported they used an e-cigarette, or vaping product that contained Δ9-THC within 90 days of the onset of symptoms. VEA, coconut oil, and limonene were quantified in 94%, 2%, and 3% of EVALI patient BALF samples, respectively but not in BALF samples from a control group. The authors postulated that the general absence of other toxicants (plant oils, medium-chain triglyceride oil, coconut oil, petroleum distillates, and terpenes) in BALF of EVALI patients discounted the role of these constituents as a primary cause of EVALI.
VEA is strongly linked to EVALI; however, the mechanism or mechanisms by which VEA causes EVALI is currently unclear. Blount et al. hypothesize that the aliphatic tail of VEA could penetrate a layer of lung surfactant to align the molecule in parallel with phospholipids, thereby interfering with surfactant function (Blount et al., 2020). Wu and O’Shea reported that VEA, when aerosolized using a third generation e-cigarette, released ethenone gas, a type of ketene gas and respiratory irritant, which they hypothesized could be a contributing factor to EVALI (Wu & O’Shea, 2020). Attfield et al. also hypothesized that thermal degradation of VEA may be important in EVALI and suggested that acetate moieties are precursors for the formation of ethenone gas and that the reaction occurs at temperatures in excess of 300 °C in the presence of catalytic metals and/or ceramic surfaces present in heating coils of e-cigarettes (Attfield et al., 2020). According to Wu & O’Shea, additional thermal transformation products produced from aerosolized VEA included benzene, butadiene, and formaldehyde, all of which are respiratory irritants and categorized by NIOSH as potential occupational carcinogens, and trace amounts of tetrahydrofuran (respiratory irritant) (NIOSH, 2018); none of these additional transformation products yield the same pathology observed with EVALI (though whether they have effects as mixtures or modify other chemical reactions is unknown). Naramani and da Silva used a computational modeling approach and their results suggested that under “typical” vaping coil temperatures, VEA was unlikely to produce ketene gases at harmful levels; however, at coil temperatures above 700 °C, such as might be encountered in a ‘dry hit’, formation of trimethyl quinone methide acetate would dominate, with lesser amounts of ketene gases produced. The authors note that even with a low yield of ketene gas, concentrations were predicted to be acutely toxic in the lungs at coil temperatures above 700 °C (Narimani & da Silva, 2020).
Muthumalage et al. obtained 38 e-liquid cartridges from EVALI patients and characterized chemical constituents in the bulk liquids and aerosol. The authors reported the presence of VEA and Δ9-THC as well as hydrocarbons, siloxanes, terpenes (including limonene), flavorings, cannabinoids, pesticides, plasticizers, polycaprolactones, and low levels of metals in bulk e-liquids. Terpenes, pesticides, solvents, and carbonyl compounds were quantified in aerosolized e-liquids (Muthumalage, Friedman, et al., 2020). VEA is strongly linked to EVALI, whether these other identified aerosolized substances, as mixtures or through interactions, contributes to the pathophysiology observed for EVALI is unclear.
Another study characterized the chemical composition of bulk liquid diluents (PG, VG, MCT, squalane, vitamin E, VEA, and triethyl citrate) and their aerosol condensates (Jiang et al., 2020). The authors reported formation of new carbonyls (PG, VG, MCT, squalane, vitamin E, VEA), alkyl alcohols (MCT, squalane), long chain alcohols (vitamin E, VEA), short chain esters (MCT, squalane, triethyl citrate), carboxylic acids (triethyl acetate), short chain alkanes (MCT, squalane), and quinones (vitamin E, VEA) in the aerosolized condensates. Aerosolized vitamin E and VEA yielded the transformation product duroquinone and aerosolized VEA produced durohydroquinone; both compounds have capacity to generate ROS. Human lung bronchus epithelial (Beas-2B) cells were exposed in vitro to aerosolized condensates of each diluent and all but triethyl citrate induced significant decreases in cell viability compared with their respective bulk liquid. LDH release was significantly increased for cells exposed to aerosolized condensates of squalane, vitamin E, and MCT (Jiang et al., 2020). Muthumalage et al. investigated effects of MCT and VEA using in vitro and in vivo models. HBE (human bronchial epithelial cells), Beas-2B (human lung bronchus epithelial cells), and MM6 (human blood leukemia derived monocytes) were exposed to aerosolized diluents under ALI conditions in vitro and wild type C57BL/6 mice were exposed via inhalation (Muthumalage, Lucas, et al., 2020). Exposure to MCT and VEA induced cellular generation of ROS in Beas-2B cells; levels were significantly higher compared with air controls, which indicated capacity to initiate and propagate oxidation of biological molecules. Exposure to VEA induced a non-significant increase in IL-8 compared with air exposed control for Beas-2B cells and a significant increase in IL-6 compared with air control for MM6 cells. Both MCT and VEA significantly reduced barrier function in HBE cells; damage to tight junctions between epithelial cells and disruption of the epithelial barrier can drive pathogenesis by promoting inflammatory signaling pathways. Exposure to MCT, but not VEA, resulted in formation of lipid-laden MM6 cells. In wild type C57BL/6 mice exposed to VEA, harvested BALF had increased IL-6 (a biomarker of lung injury) and eotaxin. Exposure to MCT and VEA significantly suppressed levels of MCP-1, RANTES, IL-17A, IL-12p40, and IL-4. There was no difference in total cells, neutrophil cells, and T-helper cells in mice exposed to MCT and VEA compared with control. Exposure to VEA caused changes in eicosanoids, including 6kPGF1a, LTB4, LTC4, LTD4, LTE4, and 5HETE and a reduction in surfactant protein SP-A levels, the latter which plays an important role in lipid homeostasis and innate immune defense. Finally, lipidomic profiling revealed that mice exposed to VEA had significantly higher levels of diradylglycerols, cholesterol ester, and glycerophosphocholines in BALF compared with controls.
Two research groups have reported lung pathologies similar to EVALI were produced in a murine model following inhalation of aerosolized VEA (Bhat et al., 2020; Matsumoto et al., 2020). Using C57BL/6 mice (sex not specified), Bhat et al. demonstrated that exposure to aerosolized VEA caused an increase in BALF VEA and protein (albumin) levels compared with exposure to an aerosolized mixture of PG/VG humectants or air. Total leukocyte cell counts in the lungs of mice exposed to VEA were significantly higher compared with mice exposed to PG/VG or air. Further, BALF of mice exposed to VEA contained many lipid-laden macrophages, which was consistent with clinical observations in EVALI patients, whereas mice exposed to PG/VG contained fewer macrophages and no evidence of lipid accumulation (Bhat et al., 2020). In another study using C57BL/6 mice (female), Matsumoto et al. reported that exposure to VEA increased BALF protein levels and plasma surfactant protein-D levels (which indicated alveolar epithelial injury) compared with air-exposed controls. VEA exposure also increased total BALF cell counts and neutrophil cell counts compared with air-exposed controls and mice exposed to aerosolized JUUL® brand flavored e-liquids; many large vacuolated macrophages contained multiple nuclei (which is a sign of activation) and stained positive for intracellular lipid droplets. Further, mice exposed to VEA had significantly increased concentrations of pro-inflammatory neutrophil chemoattractant KC (murine homologue of IL-8) and monocyte chemokine MCP-3 in lung airspaces. Histological analysis revealed that mice had monocytic and neutrophilic alveolar and interstitial inflammation with increased foamy macrophages in the airspaces, similar to that observed in EVALI patients (Matsumoto et al., 2020). Collectively, these studies indicated that aerosolized VEA, but not PG/VG, caused lung injury consistent with EVALI in C57BL/6 mice.
The widespread association of VEA in BALF of EVALI patients reported by Blount et al. has focused attention on VEA as the primary causative agent. The importance of VEA in the EVALI outbreak was bolstered by three important observations. Firstly, Taylor et al. analyzed 46 Δ9-THC-containing e-cigarette or vaping products obtained in 2019 from EVALI patients for the presence of VEA and other ingredients. For comparison, the authors tested 20 products seized by law enforcement in 2019 (during the outbreak) and ten products seized in 2018 (prior to the EVALI outbreak). Of the 46 products from EVALI patients and the 20 seized products obtained in 2019, 24 (52%) and 20 (100%), respectively, contained VEA. Among the products seized in 2018, none contained VEA (Taylor et al., 2019), which suggested VEA might have been introduced to products just prior to the outbreak. Secondly, emergency department visits related to possible EVALI cases increased sharply during August 11 to September 8, 2019, peaked during the week of September 8, 2019, then decreased thereafter. This peak and subsequent decline followed reports of the strong link between EVALI and VEA, which in part may have contributed to the removal of VEA from products or modifications in consumer behavior and the observed waning trend of new disease cases (Krishnasamy et al., 2020). Thirdly, using a C57BL/6 mouse model, two research groups independently reported that inhalation of VEA, but not PG/VG humectants, caused inflammatory and histological changes in lungs consistent with that observed in EVALI patients (Bhat et al., 2020; Matsumoto et al., 2020).
VEA is strongly implicated in the EVALI outbreak; however, evidence is not sufficient to rule out the contribution of other chemicals of concern, including chemicals in either THC or non-THC products, in some of the reported EVALI cases. As noted previously, 14% of hospitalized EVALI patients reported exclusive use of nicotine-containing products (www.cdc.gov/EVALI), which should not have contained VEA as a constituent. Additionally, Harnett et al. reported syndromic surveillance data, which indicated that low numbers of cases of EVALI were occurring before the outbreak in the summer of 2019 (Hartnett et al., 2020). Recently, an EVALI-like acute condition was reported in rats exposed to aerosol generated using an e-cigarette with nickel-chromium atomizer at high power and an e-liquid composed of PG/VG humectants and tobacco flavoring but no Δ9-THC, VEA, or nicotine (Kleinman et al., 2020). E-liquids are chemically complex mixtures so the potential for unexpected chemistries to occur within an aerosolized mixture is high. Other toxic substances could have been eliminated from the body prior to BALF collection from EVALI patients, and MCT and terpenes have capacity to induce oxidative stress and pulmonary inflammation (Chand et al., 2019; Lozier et al., 2019; Muthumalage, Friedman, et al., 2020; Wu & O’Shea, 2020).
8.2. Possible mechanisms of toxicity
Initially, acute exogenous lipoid pneumonia was given as a diagnosis for patients and it was proposed that inhaled oil droplets could deposit in the alveoli and incite a localized inflammatory response that impaired gas exchange (Davidson et al., 2019). However, subsequent clinical reviews of EVALI cases did not show histologic or radiological evidence of exogenous lipoid pneumonia (Butt et al., 2019; Mukhopadhyay et al., 2020). Based, in part on these reports, it was hypothesized that EVALI was the result of chemical pneumonitis from one or more inhaled toxic substances (Butt et al., 2019). Chand et al. described a model of chemical pneumonitis with involvement of innate immune mechanisms (Chand et al., 2019). Constituents of aerosolized e-liquids that reach the alveoli will contact Type I and Type II epithelial cells, as well as macrophages and polymorphonuclear leukocyte cells such as neutrophils, eosinophils, and basophils. These innate immune cells are part of the first line of defense against exogenous exposures and any changes in their function will alter airway homeostasis (Hickman et al., 2019). Chand et al. proposed the following sequence of events: 1) aerosolized e-liquid constituents (oils, lipids, VG, and VEA) are deposited in the alveoli, 2) this chemical insult provokes cell death and the resulting cellular debris is engulfed by macrophages via efferocytosis leading to accumulation of lipid-laden macrophages, 3) polymorphonuclear cells are recruited to the alveoli and neutrophils release extracellular traps (NETs; extracellular fibers to trap extracellular irritants), and 4) alveolar Type II epithelial and other cells secrete pro-inflammatory cytokines. Oxidative damage may also contribute to the accumulation of oxidative derivatives of cellular lipids and lung surfactant in the alveolar region of the lung (Chand et al., 2019). It is also hypothesized that the aliphatic tail of VEA could penetrate a layer of lung surfactant, which would alter surfactant function in the alveolar region of the lung (Blount et al., 2020). This proposed mechanism is consistent with 1) reports that many EVALI patients had intense acute respiratory inflammation and increased influx of inflammatory cells in the lung (Chand et al., 2019), 2) lipid-laden macrophages observed in BALF from EVALI cases were most similar in morphology to those observed in endogenous lipoid pneumonia (Mukhopadhyay et al., 2020), 3) observations that e-liquid constituents for nicotine delivery can alter surfactant activity (Davies, Birkett, Kotwa, Tomlinson, & Woldetinsae, 2017; Sosnowski, Jablczynska, Odziomek, Schlage, & Kuczaj, 2018), 4) an enrichment of NET-related proteins in sputum (although NETs are beneficial for responding to exogenous pathogens, an accumulation of NETs may cause tissue damage in the host) (Reidel et al., 2018), and 5) elevated release of pro-inflammatory cytokines by lung cells (Bengalli et al., 2017; Czekala et al., 2019; Gómez et al., 2020; Lee et al., 2019; Leigh et al., 2016; Lerner et al., 2015; Misra et al., 2014).
9. Knowledge gaps and research opportunities
This review presented an overview of progress to understand the toxicity of flavored e-liquids used in e-cigarettes and cannabis-containing e-liquids used in vaporizers and e-cigarettes. As highlighted in the preceding sections, there is a lack of standardization with respect to methods for exposing cells for in vitro studies (as well as for in vivo studies). This lack of standardization includes methods used to generate exposures as well as methods used to collect exposures. For exposure to bulk flavored e-liquids, there is variability in the amount of dilution used to prepare e-liquids for exposures to submerged cell cultures. For exposure to aerosolized e-liquids, there is great variability in the methods used to generate aerosol (smoking machine or other device), generation of e-cigarette, the e-cigarette settings (voltage, power, coil resistance), puff topography, and coil temperature. There is also variability in methods used to collect aerosols such as passing across or through cell culture medium, though the collection efficiencies of these methods appear to be poorly quantified. While, as discussed in Sections 7 and 8, much has been learned to date, many knowledge gaps still exist in our understanding of e-cigarette-and vaporizer-induced toxicity. This section briefly discusses 13 research gaps and opportunities identified during our review.
9.1. Considerations for e-cigarettes
As highlighted in Section 3.1 and Figure 1, e-cigarette designs have evolved over time. First generation e-cigarettes permitted little, if any, modifications by the user. With second and subsequent generation e-cigarettes there was a shift toward greater user control over settings. This evolution of e-cigarette designs has created research gaps/opportunities related to device settings and puffing topography for toxicology studies.
9.1.1. Research gap/opportunity 1
What e-cigarette generations should be tested?
First generation e-cigarettes appeared on the market in the early 2000s as simple devices with a battery and coil and have little resemblance to the sophistication of more recent generations of e-cigarettes. Though all four generations of e-cigarettes are still on the market, only a few studies have attempted to evaluate the influence of e-cigarette generation on toxicity of flavored e-liquids. Putzhammer et al. evaluated flavored e-liquids that were aerosolized using first and second generation e-cigarettes and suggested that aerosol generated by first generation e-cigarettes seemed less toxic (Putzhammer et al., 2016); however, the e-liquid formulation was not controlled for in their study design so the impact of device is not clearly known. In another study, Behar et al. aerosolized Cinnamon Ceylon flavored e-liquid with cartomizer- and tank-style e-cigarettes and reported that cytotoxicity was independent of device (Behar et al., 2016). As noted in Section 7, though the FDA has prioritized removing pre-filled flavored e-liquid cartridge-based products (except menthol and tobacco flavors) because they are not authorized for sale. Their production and sale for use in earlier generation devices remains legal in the United States. Additionally, consumers can still purchase concentrated PG or flavorings to dilute and mix their own desired flavored e-liquid (Noël et al., 2020) then fill their pods at home. Given the paucity of data on the impact of device generation and the greater user control over settings such as applied voltage, coil resistance, and power setting, there is a clear research gap/opportunity in our understanding of the influence of e-cigarette generation on the toxicity of aerosolized flavored e-liquids. The U.S. National Institute of Drug Abuse has developed a Standardized Research E-Cigarette (SREC) based on second generation e-cigarette technology that is available for purchase through NJOY, LLC (NIDA, 2017). To our knowledge, there are no third and fourth generation reference e-cigarettes. Note that consumer preferences for e-cigarette devices change over time and understanding current trends of e-cigarette sales is critical for generation of data that will be useful for informing risk minimization strategies (Ali et al., 2020). Finally, there is little understanding of the impact on toxicity of practices such as dripping and power vaping (Kong et al., 2020) and stealth vaping (Fadus et al., 2019) that are popular with youth and other users of later generation e-cigarettes.
9.1.2. Research gap/opportunity 2
What e-cigarette voltage settings should be tested?
Only a few studies have evaluated the impact of e-cigarette voltage settings on toxicity of aerosolized flavored e-liquids (Behar et al., 2016; Behar, Luo, et al., 2018; Farsalinos et al., 2013; Otreba et al., 2018). Except for the report by Fasalinos et al., available data generally supported the conclusion that toxicity was increased with greater applied voltage (resulting in greater power). For example, Otreba et al. demonstrated that cytotoxicity of aerosolized flavored e-liquids increased with increased ENDS voltage from 3.2 V to 4.0 V to 4.8 V. These studies bring into question how to assess the release of different toxins when they depend, in part, on parameters such as device and settings. Given the sheer number of second and third generation e-cigarette models available and the range of possible settings, it is not feasible or practical to test all possible combinations. At a minimum, the type of e-cigarette generation, device model and contextual information such as electronic settings should be included in all reports. A broader research gap/opportunity exists with regards to whether one or more standard device/voltage setting combinations should be included in all toxicity studies to serve as a benchmark to facilitate intra- and inter-comparison of results within and among studies, respectively.
9.1.3. Research gap/opportunity 3
What e-cigarette puffing topography should be tested?
This article presented a detailed toxicological review of flavored e-liquids and various devices that may be grouped into one of four different generations of e-cigarettes. Unfortunately, findings from one research study were not always directly comparable with other(s) due to dissimilarity in experimental parameters such as the e-cigarette device, puff topography, e-liquid, etc. To better integrate available knowledge and improve generalizability of research results, it is important that future studies are conducted in a manner that facilitates inter-comparison of data. Study designs should include parameters that consider uniformity in testing without overly constricting development of novel testing approaches and study designs. The Cooperation Centre for Scientific Research Relative to Tobacco (CORESTA), International Organization for Standardization (ISO), and Association Française de Normalisation (AFNOR) methods for e-cigarettes are example protocols but their limitations cannot be overlooked for certain applications. For example, CORESTA research methods incorporate e-cigarette puff topography according to a standard profile; however, an actual user’s profile will vary based on several factors such as: whether they are an inexperienced user compared with an experienced user; demographic parameters such as male compared with female; age, because teenagers, adults, and older adults have different lung capacities and breathing behaviors; and, it is also important to capture variations in topography among target populations of the study, as they can differ between previous tobacco smokers, never tobacco smokers, dual tobacco smoker and e-cigarette user, etc. ISO 20768:1018 Vapour Products — Routine Analytical Vaping Machine — Definitions and Standard Conditions is intended to define and specify the requirements of smoking machines used in laboratories to draw air through e-cigarettes to generate aerosol for testing but does not define standard conditions for toxicology assessments. As such, there is an important research gap/opportunity to identify a means to permit inter-comparison of toxicology study results to account for puff topography. CORESTA and ISO protocols are not designed to account for all use scenarios so one possible solution is that studies include one of their basic common puff topography scenarios as a benchmark to which results of all other variations of puff topography, device characteristics, and experimental parameters can be compared (e.g., ratio of tested variation to basic or benchmark scenario) within their study and between studies.
9.1.4. Research gap/opportunity 4
How do the heating conditions of these devices influence the aerosol and gas-phase chemistry, and, in turn, toxicity?
The preceding gaps/opportunities relate to properties of the e-cigarette device and topography. Individually, each of these gaps (as well as the composition of the e-liquid, which is addressed below) represent a short-coming in our knowledge of aerosolized e-liquid toxicity; however, collectively, the properties of the device and topography affect the heating conditions of the e-liquid, and hence aerosol and gas-phase chemistry (Saliba et al., 2018; Zhao, Shu, Guo, & Zhu, 2016). In turn, the aerosol and gas-phase chemistry will influence toxicity. While some attention has been given to the influence of voltage setting, more data is needed to understand the influence of heating conditions, including coil composition and variation in coil temperature on e-liquid chemistry and aerosol and gas-phase chemistry (Chen et al., 2018; Saliba et al., 2018; Zhao et al., 2016).
9.2. Considerations for e-liquids
The composition of flavored e-liquids is complex and based on the literature reviewed, we identified several research gaps/opportunities related to e-liquid composition, experimental controls, and the utility of testing bulk liquids. Further compounding investigations of the composition of e-liquids is that they can be made in a commercial setting or by consumers so there is great variability in types and proportions of ingredients, especially flavorings and nicotine.
9.2.1. Research gap/opportunity 5
Is a reference material or a benchmark e-liquid formulation needed?
E-liquid constituents contributed to data variability in toxicological findings (see Section 7). Studies to date generally utilized commercial flavored e-liquid products or laboratory-prepared flavoring chemicals in a matrix intended to mimic e-liquid formulations. Evaluation of commercial flavored e-liquids composed of complex mixtures of different ratios of PG/VG, nicotine, and multiple flavoring chemicals as well as impurities and in situ oxidation products provided the most relevant model of real-world products. Evaluation of single flavoring chemicals in a humectant matrix provided an ideal model to evaluate the specific effect of flavoring chemicals on toxicity but was not representative of real-world commercial e-liquids. Moreover, there can be significant variability within and between different brands for stated versus actual values of e-liquid constituents such as the amount of flavoring and nicotine content. These differences are often not reported or documented in toxicological evaluations, which further makes it difficult to interpret results. Hence, an important research gap/opportunity is to identify a means to permit inter-comparison of study results to account for e-liquid variability. Various research groups have proposed standard e-liquid formulations for testing purposes (Kim et al., 2017; Soulet, Duquesne, Toutain, Pairaud, & Lalo, 2019). One option is that toxicology studies include a standardized reference or benchmark e-liquid formulation to which all other variations of flavored e-liquids can be compared (e.g., ratio of tested variation to basic or benchmark scenario) within their study and between studies. Careful consideration is needed on the composition of a standardized or benchmark e-liquid formulation as consumer preferences for flavors change over time (Ali et al., 2020).
9.2.2. Research gap/opportunity 6
What experimental controls should be included for comparison of flavored e-liquid and flavorings-induced toxicity endpoints?
Ideally, all studies of flavored e-liquids and flavorings would compare toxicity results to an appropriately matched control. As described in Section 7 and summarized in Table 2, many in vitro toxicity assessments compared results with unexposed cells, cells exposed to culture media only, cells exposed to air, or cells exposed to a similar e-liquid but not exactly matched for PG/VG or nicotine concentration. As summarized in Table 3, many in vivo studies compared results with air exposed controls. Regardless of study type, few investigators have compared outcomes from exposure to a flavored e-liquid with an identical formulation that only differed in the absence of flavoring chemical, which has obscured the true influence of flavor on toxicity. Hence, a research gap/opportunity related to e-liquids is the standardization of controls for comparison of flavored e-liquid and flavoring chemical toxicity endpoints.
9.2.3. Research gap/opportunity 7
Is there utility in the assessment of bulk flavored e-liquid toxicity?
As detailed in Section 7.1.1, since the initial reports of flavored e-liquid cytotoxicity were published, there was debate as to whether researchers should evaluate the bulk e-liquid or its aerosolized form (Behar, Davis, Bahl, et al., 2014; Farsalinos et al., 2014). Several investigators have reported that toxic endpoints were consistent whether cells were exposed to flavored e-liquid or its aerosolized form, which suggested value in simple screening of liquids as a first step in a toxicology evaluation (Behar et al., 2016; Behar, Wang, & Talbot, 2018; Sassano et al., 2018). On the other hand, characteristics of aerosolized flavored e-liquid often differed from that of the bulk liquid. For example, several investigators have reported that heating an e-liquid resulted in formation of new substances such as aldehydes, acetals, and flavorings, which suggested that testing the bulk liquid may provide an incomplete assessment of exposure (Behar et al., 2016; Erythropel et al., 2019; Noël et al., 2020). Collectively, these results indicated that another research gap/opportunity is the need for a testing approach that begins with screening of the bulk flavored e-liquid and progresses to more detailed evaluation of the aerosolized form under realistic exposure conditions. One example of such an approach was proposed by the tobacco cigarette industry and begins with screening an e-liquid for potential cytotoxicity followed by mechanistic toxicity studies of the liquid and aerosolized forms (Iskandar, Zanetti, Marescotti, et al., 2019).
9.3. Considerations for modeled system
Numerous types of experimental systems are available to evaluate the toxicity of flavored e-liquids, including in vitro submerged monocultures, co-cultures, and tissue; ALI with 3D tissue models; in vivo animal; and, humans. The choice of experimental system is an important one because, as reviewed in Section 7.1, studies have documented differential responses to the same e-liquid among cell types and across developmental stages (embryonic compared with mature).
9.3.1. Research gap/opportunity 8
What exposure system should be used for toxicological assessments of flavored e-liquids?
The choice of exposure system is an important aspect of any experimental design and many factors need to be taken into consideration. Firstly, what exposure are you modeling? Inhaled droplets from aerosolized e-liquids will deposit throughout the respiratory tract, though modeling indicates that particle deposition densities will be highest in the upper bronchial generations with maximum values at the lobar bronchi (Manigrasso, Buonanno, Fuoco, Stabile, & Avino, 2015), so the exposure system should reflect the intended biological compartment. In the case of gases, deposition may be throughout the respiratory tract. Secondly, the desired endpoints, which are required to be measured also play an important role in choosing the exposure model. Hence, study plans for e-cigarette, or vaping, products aerosols need to consider several alternative and complementary models.
Cellular models can use submerged cell lines that are easy to culture and offer the ability to test numerous samples in a high throughput system. Although this type of system has its advantages, some may question its validity due to the method of exposure not resembling actual aerosol interacting with the airway surface. Immortal cells also make culturing easier; however, their altered state compared with primary cells raises questions of how measured endpoints might be affected by the cells themselves. Several studies have used submerged cells and offer important results, including information on cell type sensitivity, dosages, nicotine contribution to toxicity, ROS activation, cytokine signaling and basic chemical composition effects on toxicity (Leslie et al., 2017; Scott et al., 2018; Ween et al., 2017; Zahedi, Phandthong, Chaili, Remark, & Talbot, 2018).
ALI models offer a more complex exposure where the cells can react with the actual aerosol generated from ENDS and not the aerosol captured in liquid culture media. This approach allows a more representative model and offers real-time measurements for exposure. Other factors such as temperature, surface chemistry and aerosol droplet sizes are now allowed to affect the cell, which is not possible with a simple submerged monoculture system. A 3D ALI model allows the cells to differentiate into multi-layered surfaces that more accurately model, e.g., the lung airways (Acosta, et al., 2016; STEMCELL Technologies Inc, 2019; Zscheppang et al., 2018). Further, the complexity and crosstalk between cells is increased and cell-cell effects of the ENDS exposures can be measured more accurately using ALI models with 3D cultures compared with submerged monocultures.
In vivo exposures to ENDS aerosol offer a whole system view where multiple reactions and crosstalk can take place without limitations of systems that attempt to mimic real-life. Exposures can take the form of instillations where a bolus of e-liquid is introduced into the lung and results measured after time. This dose delivery approach offers precise control of the amount of e-liquid introduced to the animal; however, a bolus is not the best model of real-world exposure scenarios. Whole body exposures can be carried out where the ENDS aerosol is introduced into the animal’s breathing space and inhaled. Controls can be set on concentration in the atmosphere, time of exposure and duration. While closer to real-life, whole body exposure does not allow exposure to primary aerosol because what is produced by a smoking machine will interact with air (undergo evaporation, change in particle size, etc.) after it is generated and before it is inhaled by the animal. There is also the possible side effect of animal grooming where the animal may be exposed via ingestion to the condensed ENDs aerosol. In contrast, nose-only exposure studies allow direct exposure to primary ENDS aerosol to the animal and control of dose administered without the concern of co-exposure from grooming (Crotty Alexander et al., 2018; Lee et al., 2018; Smith et al., 2015). Additional advantages of nose-only inhalation systems include exposure flow rates can be specified and more easily monitored, less exposure material is needed since exposure is direct to the nose, easier containment of the test material, animals can be removed mid-exposure without effecting other animals, and nose-only is suitable for repeated dosing. However, nose-only exposures also present disadvantages in experimental procedures. Research has shown a possible change in average minute ventilation, probably due to stress, which could affect exposure amounts if baseline, unstressed, rates are used in the calculations; clearance patterns and uniformity of distribution have been demonstrated to be different depending on the route of exposure whether whole body, nose-only or oral pharyngeal; animals can becomes stressed because of restraint and no food or water during the exposure; and, if exposure tubes are used, there is potential for heat and moisture buildup and animals may try to turn around, which can lead to suffocation (Oberdörster, Castranova, Asgharian, & Sayre, 2015; Pauluhn & Thiel, 2007; Wong, 2007).
In summary, the choice of exposure system is an important and complex choice in any study design. An important research gap/opportunity is the need for better rationale for the choice of exposure system so that the exposure is representative of real-world pathways and the measured endpoints representative of human responses to using ENDS.
9.3.2. Research gap/opportunity 9
What are the characteristics of the delivered dose in the gas and droplet phases?
Aerosolized e-liquids are a mixture of gas phase chemicals and liquid phase droplets that contain chemicals. Equilibrium of constituents between these two phases is based on their partitioning constant or coefficient. Depending on the constituents of an e-liquid, their volatility, and their partitioning constant a significant portion of the inhaled aerosol may be deposited in respiratory tissues (Pankow et al., 2018). Pankow proposed to predict the liquid phase droplet and gas phase distribution of e-liquid constituents as a function of the mass concentration of the aerosol droplets, the composition of the droplets, temperature, and the vapor pressure of the compound (Pankow, 2017). In addition to the relative phase distribution of toxic constituents, it is also important to consider the size distribution of aerosolized e-liquids and the role of particle size in respiratory tract deposition. Manigrasso et al. estimated size-specific doses from e-cigarette aerosols as a function of lung deposition. The authors reported total regional doses were greater in the right lung lobes than left lobes using the Multi-Path Particle Dosimetry software model (Manigrasso et al., 2015). It is important to understand that lung deposition modeling predictions can differ with the puff profile and e-liquid characteristics. Feng et al. used an experimentally validated computational fluid-particle dynamics model to show that several major factors (ambient humidity, initial droplet diameter, and initial water mass fraction) influenced aerosolized e-liquid droplet-growth, which in turn, influenced their lung deposition patterns through enhanced inertial impaction and reduced Brownian motion. These authors and others report that because aerosolized e-liquid droplets were more hygroscopic, they tended to grow larger compared with regular tobacco cigarette smoke in a humid environment, which has implications for dose predictions (Feng, Kleinstreuer, & Rostami, 2015; Sosnowski & Kramek-Romanowska, 2016). While particle aerodynamic size is important for understanding regional deposition throughout the respiratory tract, other metrics of exposure such as particle number, mass, and surface area concentration may also be important parameters in dosimetry. Hence, knowledge of delivered dose is very limited and critical research gaps/opportunities exist on the need to improve understanding of the partitioning of toxic constituents between the gas and liquid phase droplets that were actually delivered to an experimental system as well as identification of informative dosimetry characteristics (number, mass, size, surface area) that are most relevant to toxicity in an experimental system, whether it be a submerged cell culture, ALI system, animal system, or humans.
9.4. Considerations for biological monitoring
Table 3 summarizes published studies that reported biomarkers of exposure in vivo. Markers of exposure to aerosolized e-liquids have generally been limited to monitoring a few substances or metabolites.
9.4.1. Research gap/opportunity 10
What biomarkers of exposure, effect, and disease are available to assess impacts from aerosolized flavored e-liquids?
The influence of flavored e-liquids on plasma nicotine uptake as a marker of exposure is inconclusive. For studies of e-liquids that contain nicotine in free-base form, some reported no difference in plasma nicotine levels from aerosolized flavored e-liquids compared with aerosolized unflavored e-liquids (Walele et al.,2016; Werley et al.,2016), whereas one reported that aerosolized flavor influenced plasma nicotine levels (St Helen et al., 2017). One study evaluated the influence of flavored e-liquids that contained nicotine in salt form and reported that rats exposed to aerosolized JUUL® brand Virginia Tobacco flavored e-liquid generated by a pod mod device yielded higher serum nicotine and cotinine levels compared with a aerosolized unflavored e-liquid generated by a tank-style e-cigarette (Rao et al., 2020). As detailed in Section 7.2, the JUUL® flavored e-liquid differed in PG/VG content and the amount and form of nicotine compared with thee-liquid used in the tank-style e-cigarette, which precluded any definitive conclusion on the specific role of flavoring in explaining the observed responses. Some research groups are focused on identification of biomarkers of exposure that can differentiate smoking habits such as use of flavored e-liquids or between regular tobacco cigarettes and e-cigarettes. For example, Smith et al. reported that exclusive e-cigarette users who used only fruity-flavored e-liquids had significantly higher urinary concentrations of N-acetyl-S-(2-cyanoethyl)-l-cysteine (biomarker of exposure for acrylonitrile) compared with users of any other single e-liquid flavor, but concentrations of biomarkers of exposure to nicotine (cotinine), benzene, and acrolein did not significantly differ among categories of flavored e-liquids (Smith et al., 2019). Many flavorings are aldehydes (see Table 1) and when heated, flavored e-liquids can produce secondary aldehydes dependent upon flavoring ingredients, e-cigarette device characteristics, and topography. Conklin et al. reported that the crotonaldehyde urinary metabolite 3-hydroxy-1-methylpropylmercapturic acid was increased after exposure to tobacco cigarette smoke but not e-cigarette aerosol. Further, exposure to aerosolized Menthol-flavored e-liquid elevated urinary levels of the acrolein metabolite 3-hydroxypropyl mercapturic acid and the sum of markers of nicotine exposure (nicotine, cotinine, trans-3’-hydroxycotinine) compared with exposure to aerosolized Classic Tobacco flavored e-liquid (Conklin et al., 2018). In another study, it was reported that the mercapturic acids N-acetyl-S-(3-hydroxy-1-methylpropyl)-L-cysteine (crotonaldehyde metabolite), N-acetyl-S-(3-hydroxypropyl)-L-cysteine (acrolein metabolite), and N acetyl-S-(3,4-dihydroxybutyl)-L-cysteine (1,3-butadiene metabolite) were significantly higher in urine of regular tobacco smokers compared with e-cigarette users (Frigerio et al., 2020). Salivary cytokine concentrations are being investigated as markers of effect to differentiate between regular tobacco smokers compared with e-cigarette users for endpoints such as gingival health (Faridoun, Sultan, Jabra-Rizk, Weikel, Varlotta, and Meiller, 2021). Another possible matrix for biological monitoring is exhaled breath. Recently, it was reported that e-cigarette users could be differentiated from regular tobacco cigarette smokers based on presence of esters (e.g. ethyl acetate), terpenes (e.g. α-pinene, β-pinene, d-limonene, p-cymene, etc.) and oxygenated compounds (e.g. 3-hexen-1-ol, benzaldehyde, hexanal, decanal, etc.) in their exhaled breath (Papaefstathiou, Stylianou, Andreou, & Agapiou, 2020). Based on the paucity of available data, important research gaps/opportunities that relate to biomonitoring include: 1) what unique markers of exposure for flavoring chemicals that have been shown to induce toxicity are needed for e-cigarettes? and, 2) what specific markers of effect and disease need to be identified, validated, and utilized to assess toxicity, and ideally differentiate between e-cigarette and tobacco cigarette exposures? Research to date is limited to a few studies of biomarkers in blood, urine, saliva, and exhaled breath though opportunities may exist for monitoring biomarkers in sweat and other biological fluids (Zhao et al., 2020).
9.5. Toxicity from passive exposures
In 2010, New Jersey was the first state to prohibit e-cigarette use and vaping in indoor areas of restaurants, bars, and worksites (Marynak et al., 2017). As of December 2019, 14 states, the District of Columbia (DC), and Puerto Rico have enacted similar laws (www.cdc.gov/statesystem). E-cigarettes do not generate aerosol unless the user inhales through the mouthpiece (first generation) or manually activates a battery (second and subsequent generations). Hence, the only source of secondhand aerosol from e-cigarettes is that which is exhaled by the user. One study used a smoking machine to generate aerosol from an e-cigarette in a 30 m3 chamber as an indicator of secondhand exposure; peak PG concentrations were 1400–2200 μg/m3, peak VG concentrations were 60–136 μg/m3, and peak nicotine concentrations were 0.2–0.6 μg/m3 (Geiss, Bianchi, Barahona, & Barrero-Moreno, 2015). As inhaled aerosol travels into and out of the body, particles and gases will deposit in the successive regions of the respiratory tract. Hence, the composition of what is inhaled as mainstream aerosol differs from what is exhaled as secondhand aerosol (Marco & Grimalt, 2015; Papaefstathiou, Bezantakos, et al., 2020; Samburova et al., 2018). Several studies have evaluated concentrations in exhaled breath of e-cigarette users as indicators of potential for passive exposure. For example, one study reported that the average PG, VG, and nicotine levels exhaled by an e-cigarette user into a room were 4980, 82,860, and 13.1 μg/m3, respectively (Breiev et al., 2016). Another study reported that levels of formaldehyde exhaled by an e-cigarette user into a room ranged from 5 to 8 μg/m3 (Melstrom et al., 2017). Schripp et al. sampled for 20 VOCs exhaled by e-cigarette users into a room, though only 2-butanone (2 μg/m3), acetic acid (11–14 μg/m3), acetone (17–25 μg/m3), isoprene (6–10 μg/m3), formaldehyde (8–16 μg/m3), and acetaldehyde (2–3 μg/m3) were above analytical detection limits (Schripp, Markewitz, Uhde, & Salthammer, 2013). Czogala et al. reported that air concentrations of nicotine emitted by e-cigarette users into a chamber had average of 3.3 μg/m3 (range: 0.65–6.23 μg/m3) and O’Connell et al. reported in their study that nicotine concentrations detected in the exhaled breath of e-cigarette users ranged from 11.9 to 11850 μg/m3 (Czogala et al., 2014; O’Connell, Colard, Cahours, & Pritchard, 2015). Other investigators have reported that e-cigarette users exhale large numbers of compounds into room air. Papaefstathiou et al. detected over 70 different VOCs in exhaled breath of e-cigarette users, Saffari et al. quantified 22 metals, 16 alkanes, and 19 organic acids in their exhaled breath, and Samburova et al. quantified 8 aldehydes in exhaled breath (Papaefstathiou, Bezantakos, et al., 2020; Saffari et al., 2014; Samburova et al., 2018). Finally, it was reported that maximum total VOC concentrations were 435–492 ppb when an e-cigarette user puffed in a room (Tzortzi et al., 2020). These studies demonstrated potential for secondhand exposure but did not assess whether by standers were actually exposed to emissions. In the first study of its kind, Flouris et al. directly measured exposure of volunteers who were passively exposed to e-cigarette emissions exhaled by a user and reported they had mean serum cotinine level of 2.4 ng/ml, thereby proving that passive exposure occurs (Flouris et al., 2013). Subsequently, Melstrom et al. measured passive exposure to nicotine in the breathing zone of volunteers (0.2–1 μg/m3 air), Gallart-Mateau et al. reported that passively exposed volunteers had average nicotine concentration in oral fluid of 14 μg/ml, and Ballbè et al. Reported that the geometric mean nicotine concentration in homes of e-cigarette users was 0.13 μg/m3 and passively exposed persons in these homes had salivary cotinine levels of 0.19 ng/ml (Ballbè et al., 2014; Gallart-Mateu, Elbal, Armenta, & de la Guardia, 2016; Melstrom et al., 2017). Van Drooge et al. measured organic chemicals in exhaled breath of passively exposed persons and detected seven VOCs (highest average was for formaldehyde at 14 μg/m3) and nicotine (average of 0.20 μg/m3) (van Drooge, Marco, Perez, & Grimalt, 2019). Tzortzi et al. reported that for passively exposed persons, changes in VOC concentrations from e-cigarette emissions in a room were positively associated with reported nasal and throat-respiratory symptoms (Tzortzi, Teloniatis, et al., 2020). E-cigarette conventions can attract hundreds to thousands of attendees. Johnson et al. reported that non-smokers who attended several e-cigarette conventions had elevated urinary cotinine (0.38–1.1 μg/g), salivary cotinine (0.08–0.17 ng/mL) and urinary trans-3′-hydroxycotinine (0.25–0.85 μg/g), S-(3-hydroxypropyl)-N-acetylcysteine (199–635 μg/g), S-carboxyethyl-N-acetylcysteine (83–117 μg/g), and 8-Isoprostane (260–466 ng/g) immediately after attending these events (Johnson et al., 2019).
In spaces where e-cigarettes are used or e-liquids are handled, exhaled constituents may be deposited onto surfaces and clothing, which can create opportunity for additional exposures, including through absorption or ingestion of particulate matter and other residue that is left on the surface. In a laboratory study, e-cigarette aerosol released into an experimental chamber resulted in nicotine levels up to 500 μg/m2 on surfaces (Goniewicz & Lee, 2015). One study reported the average nicotine concentration on surfaces in e-cigarette user homes was 7.7 μg/m2 (Bush & Goniewicz, 2015). Khachatoorian et al. measured settled residues on samplers placed on surfaces in a residential home and an e-cigarette retail store. The authors reported that in the residential setting, concentrations of nicotine were 2000–3000 ng/sample, concentrations of cotinine were approximately 25–125 ng/sample, concentrations of n-formylnornicotine were approximately 50–150 ng/sample, and myosmine was approximately 25 ng/sample. In the retail store, the maximum concentrations of nicotine, cotinine, and n-formylnoricotine were 284,000, 200, and 1300 ng/sample, respectively (Khachatoorian et al., 2019). It has also been reported that exhaled constituents from a vape shop traveled to an adjacent business in a multi-tenant building, thereby creating an unrecognized exposure (Khachatoorian, Jacob Iii, Benowitz, & Talbot, 2019). In another study, following e-cigarette use in a room, levels of nicotine on surfaces were 2.1–4.0 ng/100 cm2/h and levels of nicotine on cloth samples worn by users and bystanders were 44.4 ng/100 cm2/h to 69.6 ng/100 cm2/h (Melstrom et al., 2017). Liu et al. reported low levels of nicotine, PG, and VG on surfaces in a room after use of e-cigarettes (Liu et al., 2017). For more detailed information on passive exposures to e-cigarette emissions, the reader is referred to a previously published review article (Hess, Lachireddy, & Capon, 2016). The paucity of laws that prohibit indoor use of e-cigarettes indicate a high potential for secondhand exposure and additional exposure from residues on surfaces among workers in a wide range of occupations as well as children and other susceptible populations that reside or spend time in a space (or possibly an adjacent work space) occupied by someone who uses an e-cigarette. Much research to date has focused on the characteristics of mainstream aerosol that is inhaled by the e-cigarette user, with considerably less attention given to passive (secondhand and surface residue) exposures.
9.5.1. Research gap/opportunity 11
Does secondhand aerosol exposure result in toxicity to receptors?
Studies of secondhand aerosol exposure potential can be placed into two general categories: 1) user-generated secondhand aerosol in a room, and 2) mathematical modeling approaches. In studies where a user generated secondhand aerosol, levels of contaminant concentrations were measured at a distance away from the source (user) in a room (Avino et al., 2018; Maloney et al., 2016; Melstrom et al., 2017; O’Connell et al., 2015; Protano, Cattaruzza, Osborn, & Vitali, 2014; Saffari et al., 2014; Zhao et al., 2017) or biomarkers of exposure were measured in urine or saliva of bystanders (Flouris et al., 2013; Gallart-Mateu et al., 2016; Johnson et al., 2019; van Drooge et al., 2019). In one study, persons that were passively exposed to secondhand aerosol completed a questionnaire and some reported transient ocular, nasal, throat-respiratory irritation symptoms (Tzortzi, Teloniatis, et al., 2020). Mathematical modeling of secondhand exposures have included residential and occupational exposure scenarios to aldehydes and ultrafine particles (Avino et al., 2018; Colard, O’Connell, Verron, Cahours, & Pritchard, 2014; Logue et al., 2017; Rostami, Agyemang, & Pithawalla, 2018; Rostami et al., 2016). The importance of understanding toxicity in susceptible populations from passive exposures was raised by possible adverse health effects such as bronchodilation of neonatal bronchial rings (see Section 7.3). Additionally, adults exposed to secondhand aerosol experienced respiratory irritation and reviewed literature documented activation of respiratory irritation signals (see Section 7.5). Unfortunately, the influence of flavored e-liquids and flavoring chemicals on passive exposures has not been thoroughly explored to date, in part because generation of a meaningful secondhand aerosol exposure poses technical challenges. For example, it is infeasible for a user to reproducibly generate secondhand aerosol to expose cells, experimental animals, or humans, especially for sub-chronic and chronic inhalation studies. A smoking machine can be used to reproducibly generate aerosol, but the produced aerosol represents mainstream aerosol. Hence, a critical research gap/opportunity is how to generate a realistic secondhand aerosol for toxicity testing of flavored e-liquids. A possible solution may be to reproducibly generate mainstream aerosol using a smoking machine and pass it through a tube coated with surfactant to mimic aerosol interactions with lung airway lining fluid (Davies et al., 2017; Sosnowski et al., 2018) to obtain a surrogate secondhand aerosol. In the future, it may be possible to use 3-dimensional printed lung models to better mimic aerosol interactions with the respiratory tract compared with a simple tube model.
9.5.2. Research gap/opportunity 12
Does exposure to exhaled e-cigarette aerosols that form residues on surfaces result in toxicity to receptors?
Studies of exposure potential have documented accumulation of nicotine on surfaces in homes and vape shops as well as on clothing (Bush & Goniewicz, 2015; Khachatoorian, Jacob Iii, et al., 2019; Melstrom et al., 2017). Dermal uptake via exposure to residues (or bulk e-liquids from spills, drips, etc.) on surfaces can be modeled with knowledge of skin permeation rates. Frasch and Barbero (2017) have reported skin permeation rates for nicotine differed between Ice Cold Menthol flavored (menthol flavoring) and Lemon-Lime flavored (limonene flavoring) e-liquids (Frasch & Barbero, 2017). In general, there is a lack of understanding of the impact of flavorings in e-liquids on skin permeation of nicotine and other toxicants present in e-liquids. Additionally, skin permeation studies should include determination of rates using skin that is representative of susceptible races and populations such as children and the elderly to account for age-specific differences in elasticity and barrier characteristics.
9.6. Toxicology of EVALI
The EVALI outbreak began in 2019 and spread predominantly across the United States and encompassed patients who inhaled aerosolized Δ9-THC, CBD, or nicotine e-liquids from various legal and illegal sources. The number of cases and deaths attributed to the EVALI outbreak has declined substantially due to several factors, including the identification of VEA as the primary cause and the removal of it from e-cigarette, or vaping, products and modifications in consumer behavior; however, the exact mechanism of action of VEA and if other ingredients played a role remains unknown (Krishnasamy et al., 2020; Taylor et al., 2019). In addition to the research gaps and opportunities described herein, Crotty Alexander et al. has recently reported research priorities related to EVALI that were identified at a workshop convened by the U.S. National Heart, Lung, and Blood Institute (Crotty Alexander et al., 2020).
9.6.1. Research gap/opportunity 13
What additional information will further delineate the causative agent (s)s and underlying mechanism(s) of EVALI?
Several lines of evidence suggest VEA is strongly linked to EVALI (as detailed in Section 8.1). Besides VEA, it may be difficult to define mechanisms or additional potential causative agents responsible for EVALI using in vitro or in vivo methods because of the inherent variability in products (whether legitimate or illicit), product use characteristics of users, and the limited availability of the actual products responsible for eliciting disease for toxicity testing. Further complicating the search for a causative agent is the potential for thinning agents to form toxic aldehyde gases when heated in an electronic delivery system and their reactivity, which may necessitate use of trapping molecules for accurate measurement (Muthumalage, Friedman, et al., 2020; Troutt & DiDonato, 2017; Wu & O’Shea, 2020). Research gaps/opportunities related to identification of potential contributors to EVALI include, but are not limited to: 1) selection of an appropriate test system, 2) creation of surrogate e-liquids with reasonable similarity to those that elicited a toxic response in cases, 3) characterization of a meaningful vaping topography that is representative of cases, 4) generation and characterization of gas and liquid phase constituents for representative puff topographies, and 5) development of representative dose estimates to recreate conditions that yielded EVALI cases. Toward this last gap, De Jesús et al. recently published analytical methods to determine terpenes and petroleum distillates in BALF, which will enable improved dose modeling for EVALI (De Jesús et al., 2020; De Jesús, Silva, Newman, & Blount, 2020).
Mechanisms such as lipoid pneumonia and chemical pneumonitis were postulated to explain observed clinical features of EVALI cases (Chand et al., 2019; Davidson et al., 2019). To understand EVALI requires elucidation of the physical, chemical, and toxicological properties of e-liquids implicated in cases of disease. Researchers have reported chemical characterization of bulk e-liquid obtained from EVALI cases or analysis of BALF (see Section 8); however, no assessment of the physical characteristics of aerosolized e-liquids that contain Δ9-THC or CBD have been conducted to date. Characterization of the physical properties of aerosolized e-liquids in EVALI cases are needed to understand regional lung deposition of liquid droplets. Characterization of chemical thermal degradation products in aerosolized e-liquids from EVALI cases is important to understand the formation of secondary reaction products that may be harmful or react to form harmful compounds. Additionally, the partitioning of hazardous substances between the gas and liquid droplet phases is important for understanding dose. Mechanistic toxicology studies are needed to better understand this disease process, identify biomarkers of effect, and reduce future morbidity and, in some cases, mortality.
10. Conclusions
Available scientific evidence indicates that use of e-cigarettes is not without risk of harm. Flavorings in e-liquids as well as new substances formed during heating and aerosolization of e-liquids can induce adverse effects in several organ systems:
Respiratory system–Some flavored e-liquids and flavoring constituents were cytotoxic to cells encountered throughout the respiratory tract; mixtures of flavorings were more cytotoxic compared with individual flavoring constituents. Flavored e-liquids induced respiratory oxidative stress and inflammatory responses. Further, cinnamaldehyde, ethyl vanillin, benzaldehyde, and benzaldehyde PG acetal flavorings attenuated the oxidative burst capacity of neutrophils. ROS generation and secretion of proinflammatory signaling molecules following exposure to flavored e-liquids and flavoring chemicals is substance-specific. Exposure to e-liquids can also alter signaling molecules that are important for maintenance of mucociliary clearance. Exposure to aerosolized flavored e-liquids reduced the phagocytic capacity of macrophages and neutrophils and suppressed NK cells. Aerosolized flavored e-liquids impaired respiratory tract epithelial cell junctions and compromised this protective barrier. Collectively, exposure to flavored e-liquids and their flavoring ingredients inhibits many powerful components of the body’s respiratory defense mechanisms against inhaled foreign materials.
Cardiovascular and circulatory systems – Some aerosolized flavored e-liquids were cytotoxic to myocardial fibroblast cells and lung artery smooth muscle cells. Exposure to some flavored e-liquid aerosols disrupted umbilical vein endothelial cell monolayer junctions, induced time-dependent dysregulation of cell membrane potential in cardiac myocytes and suppressed NO production by human aortic epithelial cells. Collectively, these data support the conclusion that certain flavored e-liquids and flavoring chemicals may cause endothelial dysfunction similar to smoking regular tobacco cigarettes.
Developmental effects – Specific flavored e-liquids were more cytotoxic to human embryonic stem cells compared with differentiated adult HPF cells; among flavorings, cinnamaldehyde and 2-methoxycinnamaldehyde were the most cytotoxic flavoring constituents. Additionally, some flavored e-liquids induced bronchodilation of neonatal but not adult bronchial rings, which suggested that newborns and infants exposed to e-cigarette aerosol could be at risk of developmental effects.
Skeletal system – Some aerosolized flavored e-liquids increased biofilm formation and decreased enamel hardness of teeth compared with aerosolized unflavored e-liquids. Aerosolized flavored e-liquids that contained ethyl butyrate, hexyl acetate, and triacetin were associated with bacteria-initiated demineralization of enamel and ethyl maltol inhibited Streptococcus mutans growth and adhesion. Once in the body, some flavored e-liquids and/or aerosolized flavored e-liquids can impair bone health via cytotoxicity, ROS production, and alteration of osteoblast gene function via upregulation of Col1a1 mRNA and cause development of microcracks in cortical areas of bones in mice. Hence, inhalation of certain flavored e-liquids can induce negative cariogenic and skeletal effects.
Allergenicity and irritation – One study measured changes in the transcriptional profiles of genomic biomarkers relevant to respiratory or dermal sensitization; two flavored e-liquids were classified as weak dermal sensitizers. One mouse in vivo study indicated that aerosolized flavored e-liquids without nicotine had varied effects on allergic airways disease. Flavored e-liquids and flavoring ingredients can also induce irritation responses. In vitro, HEK-293T cells were transfected with either human TRPA1 or TRPV1 plasmid DNA to express these respiratory cell irritation receptors; all flavorings evaluated and their PG acetals activated the aldehyde-sensitive TRPA1 irritant receptors and the free aldehydes benzaldehyde, ethylvanillin, and vanillin only weakly activated TRPV1 vanilloid receptors. Hence, exposure to flavored e-liquids can induce allergenic and irritative responses in the respiratory tract.
Genotoxicity – Certain aerosolized flavorings such as cinnamaldehyde induce genotoxicity at non-cytotoxic concentrations. Exposure of oropharyngeal mucosa tissue, Ca9-22 oral gingival squamous carcinoma cells, and CAL-27 human tongue squamous cells to certain e-liquids indicated potential risk for development of HNSCC. Menthol flavoring in e-liquids was shown to induce cellular DNA damage and chromosomal damage in vitro. In a human study, volunteers who used a tobacco-flavored e-liquid without nicotine had significantly higher levels of micronucleus formation in oral buccal cells compared with air-exposed controls. In summary, exposure to certain flavored e-liquids can induce genotoxic or mutagenic responses in several cell models and in humans, which raises the possibility for cancers.
Skin – One in vitro study reported that skin cells exposed to aerosolized Balsamic flavored e-liquid exhibited cell damage, increased vacuolization, alteration of cytoplasmic membrane changes, and release of the inflammatory cytokine IL-8. Hence, as with regular tobacco cigarette smoke, exposure to certain flavorings used in e-liquids can have negative impacts on the skin.
EVALI – VEA is strongly linked to EVALI; however, the mechanism or mechanisms by which VEA causes EVALI is currently unclear. One hypothesis is that the aliphatic tail of VEA could penetrate a layer of lung surfactant to align the molecule in parallel with phospholipids, thereby interfering with surfactant function. Another hypothesis is that ethenone gas, which is released when VEA is heated and aerosolized using an e-cigarette, could be a contributing factor. One evaluation of e-liquid cartridges obtained from EVALI patients reported the presence of VEA and Δ9-THC as well as hydrocarbons, siloxanes, terpenes, flavorings, cannabinoids, pesticides, plasticizers, polycaprolactones, and low levels of metals in bulk e-liquids; terpenes, pesticides, solvents, and carbonyl compounds were quantified in aerosolized e-liquids. Another study of e-liquids used for cannabis delivery reported formation of new carbonyls, alkyl alcohols, long chain alcohols, short chain esters, carboxylic acids, short chain alkanes, and quinones. Recently, research groups have reported development of a murine model for EVALI, which will help understand exposure and mechanisms of toxicity.
Acknowledgements
The authors would like to thank P. Erdely and D. Weissman at NIOSH and J. Fowles at the California Department of Public Health for critical review of this manuscript prior to its submission to the journal. The authors also thank JR Wells and ED Arnold at NIOSH for helpful discussions on carbonyl and flavorings chemistry.
Funding Source
Financial support for this review was from NIOSH intramural funds. The funding source was not involved in the conduct or preparation of the review or in the decision to submit the paper for publication.
List of abbreviations
- ABB
alveolar-blood barrier
- ALI
air-liquid interface
- BALF
bronchoalveolar lavage fluid
- BHO
butane hashish oil
- CBD
cannabidiol
- CXCL
chemokine (C-X-C motif) ligand
- CHO
Chinese hamster ovary cells
- CORESTA
Cooperation Centre for Scientific Research Relative to Tobacco
- Δ9-THC
delta-9-tetrahydrocannabinol
- ENDS
electronic nicotine delivery system
- EVALI
e-cigarette, or vaping, product use-associated lung injury
- FDA
U.S. Food and Drug Administration
- GARD
Genomic Allergen Rapid Detection testing strategy
- GRAS
generally recognized as safe
- H2O2
hydrogen peroxide
- HBE
human bronchial epithelial cells
- HESC
human embryonic stem cells
- hiPSC-MC
human induced pluripotent stem cell-derived lung epithelial cells
- hiPSC-MC
human induced pluripotent stem cell-derived cardiac myocyte cells
- HNSCC
head and neck squamous cell cancer
- HPF
human primary pulmonary fibroblast cells
- HTS
high-throughput screening
- IL
interleukin
- ISO
International Organization for Standardization
- MCT
medium chain triglyceride oils
- mg
milligram
- mL
milliliter
- MM6
human blood monocyte cells
- NET
neutrophil extracellular trap
- NO
nitric oxide
- NYTS
National Youth Tobacco Survey
- PATH
Population Assessment of Tobacco and Health
- PG
propylene glycol
- ROS
reactive oxygen species
- VEA
vitamin-E acetate
- VG
vegetable glycerin
- V
volt
- W
watt
Footnotes
Declaration
The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention. Mention of any company or product does not constitute endorsement by the U.S. Government, National Institute for Occupational Safety and Health, or Centers for Disease Control and Prevention.
Conflict of Interest
The authors declare that there are no conflicts of interest.
References
- Acosta MF, Muralidharan P, Meenach SA, Hayes D, S MB, & Mansour HM (2016). In vitro pulmonary cell culture in pharmaceutical inhalation aerosol delivery: 2-D, 3-D, and in situ bioimpactor models. Current Pharmaceutical Design 22, 2522–2531. [DOI] [PubMed] [Google Scholar]
- Ali FRM, Diaz MC, Vallone D, Tynan MA, Cordova J, Seaman EL, … King BA (2020). E-cigarette unit sales, by product and flavor type - United States, 2014–2020. MMWR. Morbidity and Mortality Weekly Report 69, 1313–1318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al-Saleh I, Elkhatib R, Al-Rajoudi T, Al-Qudaihi G, Manogarannogaran P, Eltabache C, … Almugbel S (2020). Cytotoxic and genotoxic effects of e-liquids and their potential associations with nicotine, menthol and phthalate esters. Chemosphere 249, 126153. [DOI] [PubMed] [Google Scholar]
- Ambrose BK, Day HR, Rostron B, Conway KP, Borek N, Hyland A, & Villanti AC (2015). Flavored tobacco product use among US youth aged 12–17 years, 2013–2014. Journal of the American Medical Association 314, 1871–1873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apirakkan O, Frinculescu A, Denton H, Shine T, Cowan D, Abbate V, & Frascione N (2020). Isolation, detection and identification of synthetic cannabinoids in alternative formulations or dosage forms. Forensic Chemistry 18, 100227. [Google Scholar]
- Attfield K, Chen W, Cummings KJ, Jacob P, O’Shea DF, Wang P, & Fowles J (2020). Potential of ethenone (ketene) to contribute to e-cigarette, or vaping, product use-associated lung injury. American Journal of Respiratory and Critical Care Medicine 202, 1187–1189. [DOI] [PubMed] [Google Scholar]
- Avino P, Scungio M, Stabile L, Cortellessa G, Buonanno G, & Manigrasso M (2018). Second-hand aerosol from tobacco and electronic cigarettes: Evaluation of the smoker emission rates and doses and lung cancer risk of passive smokers and vapers. Science of the Total Environment 642, 137–147. [DOI] [PubMed] [Google Scholar]
- Bahl V, Lin S, Xu N, Davis B, Wang YH, & Talbot P (2012). Comparison of electronic cigarette refill fluid cytotoxicity using embryonic and adult models. Reproductive Toxicology 34, 529–537. [DOI] [PubMed] [Google Scholar]
- Ballbè M, Martínez-Sánchez JM, Sureda X, Fu M, Pérez-Ortuño R, Pascual JA, … Fernández E (2014). Cigarettes vs. E-cigarettes: Passive exposure at home measured by means of airborne marker and biomarkers. Environmental Research 135, 76–80. [DOI] [PubMed] [Google Scholar]
- Bao W, Liu B, Du Y, Snetselaar LG, & Wallace RB (2020). Electronic cigarette use among young, middle-aged, and older adults in the United States in 2017 and 2018. JAMA Internal Medicine 180, 313–314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behar RZ, Davis B, Bahl V, Lin S, & Talbot P (2014). Commentary in response to the letter from farsalinos et al. Regarding our publication entitled: “Identification of toxicants in cinnamon-flavored electronic cigarette refill fluids”. Toxicology In Vitro 28, 1521–1522. [DOI] [PubMed] [Google Scholar]
- Behar RZ, Davis B, Wang Y, Bahl V, Lin S, & Talbot P (2014). Identification of toxicants in cinnamon-flavored electronic cigarette refill fluids. Toxicology In Vitro 28, 198–208. [DOI] [PubMed] [Google Scholar]
- Behar RZ, Luo W, Lin SC, Wang Y, Valle J, Pankow JF, & Talbot P (2016). Distribution, quantification and toxicity of cinnamaldehyde in electronic cigarette refill fluids and aerosols. Tobacco Control 25, ii94–ii102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behar RZ, Luo W, McWhirter KJ, Pankow JF, & Talbot P (2018). Analytical and toxicological evaluation of flavor chemicals in electronic cigarette refill fluids. Scientific Reports 8, 8288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Behar RZ, Wang Y, & Talbot P (2018). Comparing the cytotoxicity of electronic cigarette fluids, aerosols and solvents. Tobacco Control 27, 325–333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bengalli R, Ferri E, Labra M, & Mantecca P (2017). Lung toxicity of condensed aerosol from e-cig liquids: Influence of the flavor and the in vitro model used. International Journal of Environmental Research and Public Health 14, 1254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berg CJ (2016). Preferred flavors and reasons for e-cigarette use and discontinued use among never, current, and former smokers. International Journal of Public Health 61, 225–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berkelhamer SK, Helman JM, Gugino SF, Leigh NJ, Lakshminrusimha S, & Goniewicz ML (2019). In vitro consequences of electronic-cigarette flavoring exposure on the immature lung. International Journal of Environmental Research and Public Health 16, 3635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhat TA, Kalathil SG, Bogner PN, Blount BC, Goniewicz ML, & Thanavala YM (2020). An animal model of inhaled vitamin e acetate and evali-like lung injury. The New England Journal of Medicine 382, 1175–1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhatnagar A, Whitsel LP, Ribisl KM, Bullen C, Chaloupka F, Piano MR, … Benowitz N (2014). Electronic cigarettes: A policy statement from the American Heart Association. Circulation 130, 1418–1436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop E, Haswell L, Adamson J, Costigan S, Thorne D, & Gaca M (2019). An approach to testing undiluted e-cigarette aerosol in vitro using 3D reconstituted human airway epithelium. Toxicology In Vitro 54, 391–401. [DOI] [PubMed] [Google Scholar]
- Bitzer ZT, Goel R, Reilly SM, Elias RJ, Silakov A, Foulds J, … Richie JP Jr. (2018). Effect of flavoring chemicals on free radical formation in electronic cigarette aerosols. Free Radical Biology & Medicine 120, 72–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blount BC, Karwowski MP, Shields PG, Morel-Espinosa M, Valentin-Blasini L, Gardner M, … Pirkle JL (2020). Vitamin E acetate in bronchoalveolar-lavage fluid associated with EVALI. The New England Journal of Medicine 382, 697–705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breiev K, Burseg KM, O’Connell G, Hartungen E, Biel SS, Cahours X, … Sulzer P (2016). An online method for the analysis of volatile organic compounds in electronic cigarette aerosol based on proton transfer reaction mass spectrometry. Rapid Communications in Mass Spectrometry 30, 691–697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breitbarth AK, Morgan J, & Jones AL (2018). E-cigarettes-an unintended illicit drug delivery system. Drug and Alcohol Dependence 192, 98–111. [DOI] [PubMed] [Google Scholar]
- Buchanan ND, Grimmer JA, Tanwar V, Schwieterman N, Mohler PJ, & Wold LE (2020). Cardiovascular risk of electronic cigarettes: A review of preclinical and clinical studies. Cardiovascular Research 116, 40–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bush D, & Goniewicz ML (2015). A pilot study on nicotine residues in houses of electronic cigarette users, tobacco smokers, and non-users of nicotine-containing products. The International Journal on Drug Policy 26, 609–611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bustamante-Marin XM, & Ostrowski LE (2017). Cilia and mucociliary clearance. Cold Spring Harbor Perspectives in Biology 9, a028241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butt YM, Smith ML, Tazelaar HD, Vaszar LT, Swanson KL, Cecchini MJ, … Larsen BT (2019). Pathology of vaping-associated lung injury. The New England Journal of Medicine 381, 1780–1781. [DOI] [PubMed] [Google Scholar]
- Cervellati F, Muresan XM, Sticozzi C, Gambari R, Montagner G, Forman HJ, … Valacchi G (2014). Comparative effects between electronic and cigarette smoke in human keratinocytes and epithelial lung cells. Toxicology In Vitro 28, 999–1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chand HS, Muthumalage T, Maziak W, & Rahman I (2019). Pulmonary toxicity and the pathophysiology of electronic cigarette, or vaping product, use associated lung injury. Frontiers in Pharmacology 10, 1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman DG, Casey DT, Ather JL, Aliyeva M, Daphtary N, Lahue KG, … Irvin CG (2019). The effect of flavored e-cigarettes on murine allergic airways disease. Scientific Reports 9, 13671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen W, Wang P, Ito K, Fowles J, Shusterman D, Jaques PA, & Kumagai K (2018). Measurement of heating coil temperature for e-cigarettes with a “top-coil” clearomizer. PLoS One 13, Article e0195925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cherian SV, Kumar A, & Estrada YMRM (2020). E-cigarette or vaping- product associated lung injury: A review. The American Journal of Medicine 133, 657–663. [DOI] [PubMed] [Google Scholar]
- Chun LF, Moazed F, Calfee CS, Matthay MA, & Gotts JE (2017). Pulmonary toxicity of e-cigarettes. American Journal of Physiology. Lung Cellular and Molecular Physiology 313, L193–L206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clapp PW, Lavrich KS, van Heusden CA, Lazarowski ER, Carson JL, & Jaspers I (2019). Cinnamaldehyde in flavored e-cigarette liquids temporarily suppresses bronchial epithelial cell ciliary motility by dysregulation of mitochondrial function. American Journal of Physiology. Lung Cellular and Molecular Physiology 316, L470–L486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clapp PW, Pawlak EA, Lackey JT, Keating JE, Reeber SL, Glish GL, & Jaspers I (2017). Flavored e-cigarette liquids and cinnamaldehyde impair respiratory innate immune cell function. American Journal of Physiology. Lung Cellular and Molecular Physiology 313, L278–L292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colard S, O’Connell G, Verron T, Cahours X, & Pritchard JD (2014). Electronic cigarettes and indoor air quality: A simple approach to modeling potential bystander exposures to nicotine. International Journal of Environmental Research and Public Health 12, 282–299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coleman BN, Rostron B, Johnson SE, Ambrose BK, Pearson J, Stanton CA, … Hyland A (2017). Electronic cigarette use among us adults in the population assessment of tobacco and health (PATH) study, 2013–2014. Tobacco Control 26, e117–e126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cong J, & Wei H (2019). Natural killer cells in the lungs. Frontiers in Immunology 10, 1416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conklin DJ, Ogunwale MA, Chen Y, Theis WS, Nantz MH, Fu XA, … Srivastava S (2018). Electronic cigarette-generated aldehydes: The contribution of e-liquid components to their formation and the use of urinary aldehyde metabolites as biomarkers of exposure. Aerosol Science and Technology 52, 1219–1232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corey CG, Ambrose BK, Apelberg BJ, & King BA (2015). Flavored tobacco product use among middle and high school students–United States, 2014. MMWR. Morbidity and Mortality Weekly Report 64, 1066–1070. [DOI] [PubMed] [Google Scholar]
- Crotty Alexander LE, Drummond CA, Hepokoski M, Mathew D, Moshensky A, Willeford A, … Breen EC (2018). Chronic inhalation of e-cigarette vapor containing nicotine disrupts airway barrier function and induces systemic inflammation and multiorgan fibrosis in mice. American Journal of Physiology. Regulatory, Integrative and Comparative Physiology 314, R834–R847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crotty Alexander LE, Ware LB, Calfee CS, Callahan SJ, Eissenberg T, Farver C, … Tarran R (2020). E-cigarette or vaping product use-associated lung injury: Developing a research agenda. An NIH workshop report. American Journal of Respiratory and Critical Care Medicine 202, 795–802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cullen KA, Gentzke AS, Sawdey MD, Chang JT, Anic GM, Wang TW, … King BA (2019). E-cigarette use among youth in the United States, 2019. Journal of the American Medical Association 322, 2095–2103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cwalina SN, Leventhal AM, & Barrington-Trimis JL (2020). E-cigarette flavour enhancers: Flavoured pod attachments compatible with JUUL and other pod-based devices. Tobacco Control 29, e127–e128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czekala L, Simms L, Stevenson M, Tschierske N, Maione AG, & Walele T (2019). Toxicological comparison of cigarette smoke and e-cigarette aerosol using a 3D in vitro human respiratory model. Regulatory Toxicology and Pharmacology 103, 314–324. [DOI] [PubMed] [Google Scholar]
- Czogala J, Goniewicz ML, Fidelus B, Zielinska-Danch W, Travers MJ, & Sobczak A (2014). Secondhand exposure to vapors from electronic cigarettes. Nicotine & Tobacco Research 16, 655–662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai H (2020). Self-reported marijuana use in electronic cigarettes among US youth, 2017 to 2018. Journal of the American Medical Association 323, 473–474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai H, & Hao J (2020). Online popularity of JUUL and Puff Bars in the USA: 2019–2020. Tobacco Control. 10.1136/tobaccocontrol-2020-055727 E-print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai H, & Siahpush M (2020). Use of e-cigarettes for nicotine, marijuana, and just flavoring among U.S. youth. American Journal of Preventive Medicine 58, 244–249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davidson K, Brancato A, Heetderks P, Mansour W, Matheis E, Nario M, … Fox D (2019). Outbreak of electronic-cigarette-associated acute lipoid pneumonia - North Carolina, July-August 2019. MMWR. Morbidity and Mortality Weekly Report 68, 784–786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies MJ, Birkett JW, Kotwa M, Tomlinson L, & Woldetinsae R (2017). The impact of cigarette/e-cigarette vapour on simulated pulmonary surfactant monolayers under physiologically relevant conditions. Surface and Interface Analysis 49, 654–665. [Google Scholar]
- De Jesús VR, Chambers DM, Reese C, Braselton M, Espinosa P, Corstvet J, & Blount BC (2020). Novel methods for the analysis of toxicants in bronchoalveolar lavage fluid samples from e-cigarette, or vaping, product use-associated lung injury cases: Selected petroleum distillates. Rapid Communications in Mass Spectrometry 34, Article e8898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Jesús VR, Silva LK, Newman CA, & Blount BC (2020). Novel methods for the analysis of toxicants in bronchoalveolar lavage fluid samples from e-cigarette, or vaping, product use associated lung injury (EVALI) cases: Terpenes. Rapid Communications in Mass Spectrometry 34, Article e8879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delnevo C, Giovenco DP, & Hrywna M (2020). Rapid proliferation of illegal pod-mod disposable e-cigarettes. Tobacco Control 29, e150–e151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delnevo CD, Giovenco DP, Steinberg MB, Villanti AC, Pearson JL, Niaura RS, & Abrams DB (2016). Patterns of electronic cigarette use among adults in the United States. Nicotine & Tobacco Research 18, 715–719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Drooge BL, Marco E, Perez N, & Grimalt JO (2019). Influence of electronic cigarette vaping on the composition of indoor organic pollutants, particles, and exhaled breath of bystanders. Environmental Science and Pollution Research International 26, 4654–4666. [DOI] [PubMed] [Google Scholar]
- Duffy B, Li L, Lu S, Durocher L, Dittmar M, Delaney-Baldwin E, … Spink D (2020). Analysis of cannabinoid-containing fluids in illicit vaping cartridges recovered from pulmonary injury patients: Identification of vitamin e acetate as a major diluent. Toxics 8, 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- El-Hellani A, El-Hage R, Baalbaki R, Salman R, Talih S, Shihadeh A, & Saliba NA (2015). Free-base and protonated nicotine in electronic cigarette liquids and aerosols. Chemical Research in Toxicology 28, 1532–1537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erythropel HC, Jabba SV, DeWinter TM, Mendizabal M, Anastas PT, Jordt SE, & Zimmerman JB (2019). Formation of flavorant-propylene glycol adducts with novel toxicological properties in chemically unstable e-cigarette liquids. Nicotine & Tobacco Research 21, 1248–1258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fadus MC, Smith TT, & Squeglia LM (2019). The rise of e-cigarettes, pod mod devices, and JUUL among youth: Factors influencing use, health implications, and downstream effects. Drug and Alcohol Dependence 201, 85–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faridoun A, Sultan AS, Jabra-Rizk MA, Weikel D, Varlotta S, & Meiller TF (2021). Salivary biomarker profiles in e-cigarette users and conventional smokers: A cross-sectional study. Oral Diseases 27, 277–279. [DOI] [PubMed] [Google Scholar]
- Farsalinos KE, Romagna G, Allifranchini E, Ripamonti E, Bocchietto E, Todeschi S, … Voudris V (2013). Comparison of the cytotoxic potential of cigarette smoke and electronic cigarette vapour extract on cultured myocardial cells. International Journal of Environmental Research and Public Health 10, 5146–5162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farsalinos KE, Romagna G, & Voudris V (2014). Cytotoxicity of cinnamon-flavored electronic cigarette refills: Are the results truly applicable to electronic cigarette use? Toxicology In Vitro 28, 1016–1017. [DOI] [PubMed] [Google Scholar]
- FDA (2020). FDA finalizes enforcement policy on unauthorized flavored cartridge-based e-cigarettes that appeal to children, including fruit and mint. https://www.fda.gov/news-events/press-announcements/fda-finalizes-enforcement-policy-unauthorized-flavored-cartridge-based-e-cigarettes-appeal-children/ (accessed May 7, 2020).
- Feldman R, Stanton M, & Suelzer EM (2021). Compiling evidence for EVALI: A scoping review of in vivo pulmonary effects after inhaling vitamin E or vitamin E acetate. Journal of Medical Toxicology. 10.1007/s13181-021-00823-w E-print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FEMA (2020). Safety and regulatory authority to use flavors – focus on vaping products. https://www.femaflavor.org/safety-regulatory-authority-use-flavors-focus-vaping-products/ (accessed March 31, 2020).
- Feng Y, Kleinstreuer C, & Rostami A (2015). Evaporation and condensation of multicomponent electronic cigarette droplets and conventional cigarette smoke particles in an idealized G3–G6 triple bifurcating unit. Journal of Aerosol Science 80, 58–74. [Google Scholar]
- Fetterman JL, Weisbrod RM, Feng B, Bastin R, Tuttle ST, Holbrook M, … Hamburg NM (2018). Flavorings in tobacco products induce endothelial cell dysfunction. Arteriosclerosis, Thrombosis, and Vascular Biology 38, 1607–1615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fite PJ, Cushing C, & Ortega A (2020). Three year trends in e-cigarettes among mid-western middle school age youth. Journal of Substance Use 25, 430–434. [Google Scholar]
- Flouris AD, Chorti MS, Poulianiti KP, Jamurtas AZ, Kostikas K, Tzatzarakis MN, … Koutedakis Y (2013). Acute impact of active and passive electronic cigarette smoking on serum cotinine and lung function. Inhalation Toxicology 25, 91–101. [DOI] [PubMed] [Google Scholar]
- Frasch HF, & Barbero AM (2017). In vitro human epidermal permeation of nicotine from electronic cigarette refill liquids and implications for dermal exposure assessment. Journal of Exposure Science & Environmental Epidemiology 27, 618–624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frigerio G, Mercadante R, Campo L, Polledri E, Boniardi L, Olgiati L, … Fustinoni S (2020). Urinary biomonitoring of subjects with different smoking habits. Part II: An untargeted metabolomic approach and the comparison with the targeted measurement of mercapturic acids. Toxicology Letters 329, 56–66. [DOI] [PubMed] [Google Scholar]
- Gallart-Mateu D, Elbal L, Armenta S, & de la Guardia M (2016). Passive exposure to nicotine from e-cigarettes. Talanta 152, 329–334. [DOI] [PubMed] [Google Scholar]
- Geiss O, Bianchi I, Barahona F, & Barrero-Moreno J (2015). Characterisation of mainstream and passive vapours emitted by selected electronic cigarettes. International Journal of Hygiene and Environmental Health 218, 169–180. [DOI] [PubMed] [Google Scholar]
- Gerloff J, Sundar IK, Freter R, Sekera ER, Friedman AE, Robinson R, … Rahman I (2017). Inflammatory response and barrier dysfunction by different e-cigarette flavoring chemicals identified by gas chromatography-mass spectrometry in e-liquids and e-vapors on human lung epithelial cells and fibroblasts. Applied In Vitro Toxicology 3, 28–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giroud C, de Cesare M, Berthet A, Varlet V, Concha-Lozano N, & Favrat B (2015). E-cigarettes: A review of new trends in cannabis use. International Journal of Environmental Research and Public Health 12, 9988–10008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Go YY, Mun JY, Chae SW, Chang J, & Song JJ (2020). Comparison between in vitro toxicities of tobacco- and menthol-flavored electronic cigarette liquids on human middle ear epithelial cells. Scientific Reports 10, 2544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gómez AC, Rodríguez-Fernández P, Villar-Hernández R, Gibert I, Muriel-Moreno B, Lacoma A, … Domínguez J (2020). E-cigarettes: Effects in phagocytosis and cytokines response against mycobacterium tuberculosis. PLoS One 15, Article e0228919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goniewicz ML, & Lee L (2015). Electronic cigarettes are a source of thirdhand exposure to nicotine. Nicotine & Tobacco Research 17, 256–258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goniewicz ML, & Zielinska-Danch W (2012). Electronic cigarette use among teenagers and young adults in Poland. Pediatrics 130, e879–e885. [DOI] [PubMed] [Google Scholar]
- Gotts JE (2019). High-power vaping injures the human lung. American Journal of Physiology. Lung Cellular and Molecular Physiology 316, L703–L704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gotts JE, Jordt SE, McConnell R, & Tarran R (2019). What are the respiratory effects of e-cigarettes? BMJ (Clinical Research Edition) 366, l5275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grafinger KE, Kronert S, Broillet A, & Weinmann W (2020). Cannabidiol and tetrahydrocannabinol concentrations in commercially available CBD e-liquids in Switzerland. Forensic Science International 310, 110261. [DOI] [PubMed] [Google Scholar]
- Grana R, Benowitz N, & Glantz SA (2014). E-cigarettes: A scientific review. Circulation 129, 1972–1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greene RM, & Pisano MM (2019). Developmental toxicity of e-cigarette aerosols. Birth Defects Research 111, 1294–1301. [DOI] [PubMed] [Google Scholar]
- Harrell MB, Weaver SR, Loukas A, Creamer M, Marti CN, Jackson CD, … Eriksen MP (2017). Flavored e-cigarette use: Characterizing youth, young adult, and adult users. Preventive Medicine Reports 5, 33–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartnett KP, Kite-Powell A, Patel MT, Haag BL, Sheppard MJ, Dias TP, … Adjemian J (2020). Syndromic surveillance for e-cigarette, or vaping, product use-associated lung injury. The New England Journal of Medicine 382, 766–772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harvanko AM, Havel CM, Jacob P, & Benowitz NL (2020). Characterization of nicotine salts in 23 electronic cigarette refill liquids. Nicotine & Tobacco Research 22, 1239–1243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Havermans A, Krusemann EJZ, Pennings J, de Graaf K, Boesveldt S, & Talhout R (2021). Nearly 20 000 e-liquids and 250 unique flavour descriptions: An overview of the Dutch market based on information from manufacturers. Tobacco Control 30, 57–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He T, Oks M, Esposito M, Steinberg H, & Makaryus M (2017). “Tree-in-bloom”: Severe acute lung injury induced by vaping cannabis oil. Annals of the American Thoracic Society 14, 468–470. [DOI] [PubMed] [Google Scholar]
- Hess IM, Lachireddy K, & Capon A (2016). A systematic review of the health risks from passive exposure to electronic cigarette vapour. Public Health Research and Practice 26, 2621617. [DOI] [PubMed] [Google Scholar]
- Hickman E, Herrera CA, & Jaspers I (2019). Common e-cigarette flavoring chemicals impair neutrophil phagocytosis and oxidative burst. Chemical Research in Toxicology 32, 982–985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho YS, Magnenat JL, Gargano M, & Cao J (1998). The nature of antioxidant defense mechanisms: A lesson from transgenic studies. Environmental Health Perspectives 106 (Suppl. 5), 1219–1228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu SS, Homa DM, Wang T, Gomez Y, Walton K, Lu H, & Neff L (2019). State-specific patterns of cigarette smoking, smokeless tobacco use, and e-cigarette use among adults - United States, 2016. Preventing Chronic Disease 16, Article E17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang J, Duan Z, Kwok J, Binns S, Vera LE, Kim Y, … Emery SL (2019). Vaping versus JUULing: How the extraordinary growth and marketing of JUUL transformed the US retail e-cigarette market. Tobacco Control 28, 146–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iskandar AR, Zanetti F, Kondylis A, Martin F, Leroy P, Majeed S, … Hoeng J (2019). A lower impact of an acute exposure to electronic cigarette aerosols than to cigarette smoke in human organotypic buccal and small airway cultures was demonstrated using systems toxicology assessment. Internal and Emergency Medicine 14, 863–883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iskandar AR, Zanetti F, Marescotti D, Titz B, Sewer A, Kondylis A, … Hoeng J (2019). Application of a multi-layer systems toxicology framework for in vitro assessment of the biological effects of classic tobacco e-liquid and its corresponding aerosol using an e-cigarette device with MESHTM technology. Archives of Toxicology 93, 3229–3247. [DOI] [PubMed] [Google Scholar]
- Jaber RM, Mirbolouk M, DeFilippis AP, Maziak W, Keith R, Payne T, … Nasir K (2018). Electronic cigarette use prevalence, associated factors, and pattern by cigarette smoking status in the United States from NHANES (National Health and Nutrition Examination Survey) 2013–2014. Journal of the American Heart Association 7, Article e008178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackler RK, & Ramamurthi D (2019). Nicotine arms race: JUUL and the high-nicotine product market. Tobacco Control 28, 623–628. [DOI] [PubMed] [Google Scholar]
- Jiang H, Ahmed CMS, Martin TJ, Canchola A, Oswald IWH, Garcia JA, … Lin YH (2020). Chemical and toxicological characterization of vaping emission products from commonly used vape juice diluents. Chemical Research in Toxicology 33, 2157–2163. [DOI] [PubMed] [Google Scholar]
- Johnson JM, Naeher LP, Yu X, Sosnoff C, Wang L, Rathbun SL, … Wang JS (2019). A biomonitoring assessment of secondhand exposures to electronic cigarette emissions. International Journal of Hygiene and Environmental Health 222, 816–823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaur G, Muthumalage T, & Rahman I (2018). Mechanisms of toxicity and biomarkers of flavoring and flavor enhancing chemicals in emerging tobacco and non-tobacco products. Toxicology Letters 288, 143–155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kava CM, Hannon PA, & Harris JR (2020). Use of cigarettes and e-cigarettes and dual use among adult employees in the US workplace. Preventing Chronic Disease 17, Article E16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kerasioti E, Veskoukis AS, Skaperda Z, Zacharias A, Poulas K, Lazopoulos G, & Kouretas D (2020). The flavoring and not the nicotine content is a decisive factor for the effects of refill liquids of electronic cigarette on the redox status of endothelial cells. Toxicology Reports 7, 1095–1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khachatoorian C, Jacob P 3rd, Sen A, Zhu Y, Benowitz NL, & Talbot P (2019). Identification and quantification of electronic cigarette exhaled aerosol residue chemicals in field sites. Environmental Research 170, 351–358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khachatoorian C, Jacob Iii P, Benowitz NL, & Talbot P (2019). Electronic cigarette chemicals transfer from a vape shop to a nearby business in a multiple-tenant retail building. Tobacco Control 28, 519–525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JJ, Sabatelli N, Tutak W, Giuseppetti A, Frukhtbeyn S, Shaffer I, … Ondov JM (2017). Universal electronic-cigarette test: Physiochemical characterization of reference e-liquid. Tobacco Induced Diseases 15, 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim SA, Smith S, Beauchamp C, Song Y, Chiang M, Giuseppetti A, … Kim JJ (2018). Cariogenic potential of sweet flavors in electronic-cigarette liquids. PLoS One 13, Article e0203717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirsch-Volders M, Decordier I, Elhajouji A, Plas G, Aardema MJ, & Fenech M (2011). In vitro genotoxicity testing using the micronucleus assay in cell lines, human lymphocytes and 3D human skin models. Mutagenesis 26, 177–184. [DOI] [PubMed] [Google Scholar]
- Kleinman MT, Arechavala RJ, Herman D, Shi J, Hasen I, Ting A, … Kloner RA (2020). E-cigarette or vaping product use-associated lung injury produced in an animal model from electronic cigarette vapor exposure without tetrahydrocannabinol or vitamin E oil. Journal of the American Heart Association 9, Article e017368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kong G, Morean ME, Bold KW, Wu R, Bhatti H, Simon P, & Krishnan-Sarin S (2020). Dripping and vape tricks: Alternative e-cigarette use behaviors among adolescents. Addictive Behaviors 107, 106394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreiss K, Gomaa A, Kullman G, Fedan K, Simoes EJ, & Enright PL (2002). Clinical bronchiolitis obliterans in workers at a microwave-popcorn plant. The New England Journal of Medicine 347, 330–338. [DOI] [PubMed] [Google Scholar]
- Krishnasamy VP, Hallowell BD, Ko JY, Board A, Hartnett KP, Salvatore PP, … Ellington S (2020). Update: Characteristics of a nationwide outbreak of e-cigarette, or vaping, product use-associated lung injury - United States, August 2019-January 2020. MMWR. Morbidity and Mortality Weekly Report 69, 90–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krüsemann EJZ, Boesveldt S, de Graaf K, & Talhout R (2019). An e-liquid flavor wheel: A shared vocabulary based on systematically reviewing e-liquid flavor classifications in literature. Nicotine & Tobacco Research 21, 1310–1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krusemann EJZ, Havermans A, Pennings JLA, de Graaf K, Boesveldt S, & Talhout R (2021). Comprehensive overview of common e-liquid ingredients and how they can be used to predict an e-liquid’s flavour category. Tobacco Control 30, 185–191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Lacy E, Fletcher A, Hewitt G, Murphy S, & Moore G (2017). Cross-sectional study examining the prevalence, correlates and sequencing of electronic cigarette and tobacco use among 11–16-year olds in schools in Wales. BMJ Open 7, Article e012784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamb T, Muthumalage T, & Rahman I (2020). Pod-based menthol and tobacco flavored e-cigarettes cause mitochondrial dysfunction in lung epithelial cells. Toxicology Letters 333, 303–311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leavens ELS, Stevens EM, Brett EI, Hebert ET, Villanti AC, Pearson JL, & Wagener TL (2019). JUUL electronic cigarette use patterns, other tobacco product use, and reasons for use among ever users: Results from a convenience sample. Addictive Behaviors 95, 178–183. [DOI] [PubMed] [Google Scholar]
- Lee KM, Hoeng J, Harbo S, Kogel U, Gardner W, Oldham M, … McKinney WJ Jr. (2018). Biological changes in C57BL/6 mice following 3 weeks of inhalation exposure to cigarette smoke or e-vapor aerosols. Inhalation Toxicology 30, 553–567. [DOI] [PubMed] [Google Scholar]
- Lee WH, Ong SG, Zhou Y, Tian L, Bae HR, Baker N, … Wu JC (2019). Modeling cardiovascular risks of e-cigarettes with human-induced pluripotent stem cell-derived endothelial cells. Journal of the American College of Cardiology 73, 2722–2737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leigh NJ, Lawton RI, Hershberger PA, & Goniewicz ML (2016). Flavourings significantly affect inhalation toxicity of aerosol generated from electronic nicotine delivery systems (ENDS). Tobacco Control 25, ii81–ii87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leonard SS, Harris GK, & Shi X (2004). Metal-induced oxidative stress and signal transduction. Free Radical Biology & Medicine 37, 1921–1942. [DOI] [PubMed] [Google Scholar]
- Lerner CA, Sundar IK, Yao H, Gerloff J, Ossip DJ, McIntosh S, … Rahman I (2015). Vapors produced by electronic cigarettes and e-juices with flavorings induce toxicity, oxidative stress, and inflammatory response in lung epithelial cells and in mouse lung. PLoS One 10, Article e0116732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leslie LJ, Vasanthi Bathrinarayanan P, Jackson P, Mabiala Ma Muanda JA, Pallett R, Stillman CJP, & Marshall LJ (2017). A comparative study of electronic cigarette vapor extracts on airway-related cell lines in vitro. Inhalation Toxicology 29, 126–136. [DOI] [PubMed] [Google Scholar]
- Liu J, Liang Q, Oldham MJ, Rostami AA, Wagner KA, Gillman IG, … Sarkar M (2017). Determination of selected chemical levels in room air and on surfaces after the use of cartridge- and tank-based e-vapor products or conventional cigarettes. International Journal of Environmental Research and Public Health 14, 969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Logue JM, Sleiman M, Montesinos VN, Russell ML, Litter MI, Benowitz NL, … Destaillats H (2017). Emissions from electronic cigarettes: Assessing vapers’ intake of toxic compounds, secondhand exposures, and the associated health impacts. Environmental Science & Technology 51, 9271–9279. [DOI] [PubMed] [Google Scholar]
- Lozier MJ, Wallace B, Anderson K, Ellington S, Jones CM, Rose D, … Mikosz CA (2019). Update: Demographic, product, and substance-use characteristics of hospitalized patients in a nationwide outbreak of e-cigarette, or vaping, product use-associated lung injuries - United States, December 2019. MMWR. Morbidity and Mortality Weekly Report 68, 1142–1148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucas JH, Muthumalage T, Wang Q, Friedman MR, Friedman AE, & Rahman I (2020). E-liquid containing a mixture of coconut, vanilla, and cookie flavors causes cellular senescence and dysregulated repair in pulmonary fibroblasts: Implications on premature aging. Frontiers in Physiology 11, 924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacLean RR, Valentine GW, Jatlow PI, & Sofuoglu M (2017). Inhalation of alcohol vapor: Measurement and implications. Alcoholism, Clinical and Experimental Research 41, 238–250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mallock N, Trieu HL, Macziol M, Malke S, Katz A, Laux P, … Luch A (2020). Trendy e-cigarettes enter europe: Chemical characterization of JUUL pods and its aerosols. Archives of Toxicology 94, 1985–1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maloney JC, Thompson MK, Oldham MJ, Stiff CL, Lilly PD, Patskan GJ, … Sarkar MA (2016). Insights from two industrial hygiene pilot e-cigarette passive vaping studies. Journal of Occupational and Environmental Hygiene 13, 275–283. [DOI] [PubMed] [Google Scholar]
- Manigrasso M, Buonanno G, Fuoco FC, Stabile L, & Avino P (2015). Aerosol deposition doses in the human respiratory tree of electronic cigarette smokers. Environmental Pollution 196, 257–267. [DOI] [PubMed] [Google Scholar]
- Marco E, & Grimalt JO (2015). A rapid method for the chromatographic analysis of volatile organic compounds in exhaled breath of tobacco cigarette and electronic cigarette smokers. Journal of Chromatography. A 1410, 51–59. [DOI] [PubMed] [Google Scholar]
- Marescotti D, Mathis C, Belcastro V, Leroy P, Acali S, Martin F, … Hoeng J (2020). Systems toxicology assessment of a representative e-liquid formulation using human primary bronchial epithelial cells. Toxicology Reports 7, 67–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez RE, Dhawan S, Sumner W, & Williams BJ (2015). On-line chemical composition analysis of refillable electronic cigarette aerosol-measurement of nicotine and nicotyrine. Nicotine & Tobacco Research 17, 1263–1269. [DOI] [PubMed] [Google Scholar]
- Marynak K, Kenemer B, King BA, Tynan MA, MacNeil A, & Reimels E (2017). State laws regarding indoor public use, retail sales, and prices of electronic cigarettes - U.S. States, Guam, Puerto Rico, and U.S. Virgin Islands, September 30, 2017. MMWR. Morbidity and Mortality Weekly Report 66, 1341–1346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto S, Fang X, Traber MG, Jones KD, Langelier C, Hayakawa Serpa P, … Gotts JE (2020). Dose-dependent pulmonary toxicity of aerosolized vitamin E acetate. American Journal of Respiratory Cell and Molecular Biology 63, 748–757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meier E, & Hatsukami DK (2016). A review of the additive health risk of cannabis and tobacco co-use. Drug and Alcohol Dependence 166, 6–12. [DOI] [PubMed] [Google Scholar]
- Melstrom P, Koszowski B, Thanner MH, Hoh E, King B, Bunnell R, & McAfee T (2017). Measuring PM2.5, ultrafine particles, nicotine air and wipe samples following the use of electronic cigarettes. Nicotine & Tobacco Research 19, 1055–1061. [DOI] [PubMed] [Google Scholar]
- Menicagli R, Marotta O, & Serra R (2020). Free radical production in the smoking of e-cigarettes and their possible effects in human health. International Journal of Preventive Medicine 11, 53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miech R, Leventhal A, Johnston L, O’Malley PM, Patrick ME, & Barrington-Trimis J (2021). Trends in use and perceptions of nicotine vaping among US youth from 2017 to 2020. Journal of American Medical Association Pediatrics 175, 185–190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirbolouk M, Charkhchi P, Kianoush S, Uddin SMI, Orimoloye OA, Jaber R, … Blaha MJ (2018). Prevalence and distribution of e-cigarette use among U.S. adults: Behavioral risk factor surveillance system, 2016. Annals of Internal Medicine 169, 429–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirbolouk M, Charkhchi P, Orimoloye OA, Uddin SMI, Kianoush S, Jaber R, … Blaha MJ (2019). E-cigarette use without a history of combustible cigarette smoking among U.S. Adults: Behavioral risk factor surveillance system, 2016. Annals of Internal Medicine 170, 76–79. [DOI] [PubMed] [Google Scholar]
- Misra M, Leverette RD, Cooper BT, Bennett MB, & Brown SE (2014). Comparative in vitro toxicity profile of electronic and tobacco cigarettes, smokeless tobacco and nicotine replacement therapy products: E-liquids, extracts and collected aerosols. International Journal of Environmental Research and Public Health 11, 11325–11347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morean ME, Kong G, Camenga DR, Cavallo DA, & Krishnan-Sarin S (2015). High school students’ use of electronic cigarettes to vaporize cannabis. Pediatrics 136, 611–616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukhopadhyay S, Mehrad M, Dammert P, Arrossi AV, Sarda R, Brenner DS, … Ghobrial M (2020). Lung biopsy findings in severe pulmonary illness associated with e-cigarette use (vaping). American Journal of Clinical Pathology 153, 30–39. [DOI] [PubMed] [Google Scholar]
- Muthumalage T, Friedman MR, McGraw MD, Ginsberg G, Friedman AE, & Rahman I (2020). Chemical constituents involved in e-cigarette, or vaping product use-associated lung injury (EVALI). Toxics 8, 25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muthumalage T, Lucas JH, Wang Q, Lamb T, McGraw MD, & Rahman I (2020). Pulmonary toxicity and inflammatory response of e-cigarette vape cartridges containing medium-chain triglycerides oil and vitamin E acetate: Implications in the pathogenesis of EVALI. Toxics 8, 46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muthumalage T, Prinz M, Ansah KO, Gerloff J, Sundar IK, & Rahman I (2018). Inflammatory and oxidative responses induced by exposure to commonly used e-cigarette flavoring chemicals and flavored e-liquids without nicotine. Frontiers in Physiology 8, 1130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Narimani M, & da Silva G (2020). Does ‘dry hit’ vaping of vitamin E acetate contribute to EVALI? Simulating toxic ketene formation during e-cigarette use. PLoS One 15, Article e0238140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NIDA (2017). NIDA Drug Supply Program. https://www.drugabuse.gov/research/research-data-measures-resources/nida-drug-supply-program/ (accessed June 16, 2020).
- NIOSH (2018). Pocket Guide to Chemical Hazards. http://www.cdc.gov/niosh/npg/default.html (accessed June 16, 2020).
- Noël JC, Rainer D, Gstir R, Rainer M, & Bonn G (2020). Quantification of selected aroma compounds in e-cigarette products and toxicity evaluation in HUVEC/Tert2 cells. Biomedical Chromatography 34, Article e4761. [DOI] [PubMed] [Google Scholar]
- Nystoriak MA, Kilfoil PJ, Lorkiewicz PK, Ramesh B, Kuehl PJ, McDonald J, … Conklin DJ (2019). Comparative effects of parent and heated cinnamaldehyde on the function of human ipsc-derived cardiac myocytes. Toxicology In Vitro 61, 104648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oberdörster G, Castranova V, Asgharian B, & Sayre P (2015). Inhalation exposure to carbon nanotubes (CNT) and carbon nanofibers (CNF): Methodology and dosimetry. Journal of Toxicology and Environmental Health. Part B, Critical Reviews 18, 121–212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Connell G, Colard S, Cahours X, & Pritchard JD (2015). An assessment of indoor air quality before, during and after unrestricted use of e-cigarettes in a small room. International Journal of Environmental Research and Public Health 12, 4889–4907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okawa S, Tabuchi T, & Miyashiro I (2020). Who uses e-cigarettes and why? E-cigarette use among older adolescents and young adults in Japan: JASTIS study. Journal of Psychoactive Drugs 52, 37–45. [DOI] [PubMed] [Google Scholar]
- Omaiye EE, McWhirter KJ, Luo W, Pankow JF, & Talbot P (2019). High-nicotine electronic cigarette products: Toxicity of JUUL fluids and aerosols correlates strongly with nicotine and some flavor chemical concentrations. Chemical Research in Toxicology 32, 1058–1069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otero CE, Noeker JA, Brown MM, Wavreil FDM, Harvey WA, Mitchell KA, & Heggland SJ (2019). Electronic cigarette liquid exposure induces flavor-dependent osteotoxicity and increases expression of a key bone marker, collagen type i. Journal of Applied Toxicology 39, 888–898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otreba M, Kosmider L, Knysak J, Warncke JD, & Sobczak A (2018). E-cigarettes: Voltage- and concentration-dependent loss in human lung adenocarcinoma viability. Journal of Applied Toxicology 38, 1135–1143. [DOI] [PubMed] [Google Scholar]
- Pang RD, Goldenson NI, Kirkpatrick M, Barrington-Trimis JL, Cho J, & Leventhal AM (2020). Sex differences in the appeal of flavored e-cigarettes among young adult e-cigarette users. Psychology of Addictive Behaviors 34, 303–307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pankow JF (2017). Calculating compound dependent gas-droplet distributions in aerosols of propylene glycol and glycerol from electronic cigarettes. Journal of Aerosol Science 107, 9–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pankow JF, Kim K, Luo W, & McWhirter KJ (2018). Gas/particle partitioning constants of nicotine, selected toxicants, and flavor chemicals in solutions of 50/50 propylene glycol/glycerol as used in electronic cigarettes. Chemical Research in Toxicology 31, 985–990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papaefstathiou E, Bezantakos S, Stylianou M, Biskos G, & Agapiou A (2020). Comparison of particle size distributions and volatile organic compounds exhaled by e-cigarette and cigarette users. Journal of Aerosol Science 141, 105487. [Google Scholar]
- Papaefstathiou E, Stylianou M, Andreou C, & Agapiou A (2020). Breath analysis of smokers, non-smokers, and e-cigarette users. Journal of Chromatography B 1160, 122349. [DOI] [PubMed] [Google Scholar]
- Park HR, O’Sullivan M, Vallarino J, Shumyatcher M, Himes BE, Park JA, … Lu Q (2019). Transcriptomic response of primary human airway epithelial cells to flavoring chemicals in electronic cigarettes. Scientific Reports 9, 1400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pauluhn J, & Thiel A (2007). A simple approach to validation of directed-flow nose-only inhalation chambers. Journal of Applied Toxicology 27, 160–167. [DOI] [PubMed] [Google Scholar]
- Peace MR, Butler KE, Wolf CE, Poklis JL, & Poklis A (2016). Evaluation of two commercially available cannabidiol formulations for use in electronic cigarettes. Frontiers in Pharmacology 7, 279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peace MR, Stone JW, Poklis JL, Turner JB, & Poklis A (2016). Analysis of a commercial marijuana e-cigarette formulation. Journal of Analytical Toxicology 40, 374–378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearce K, Gray N, Gaur P, Jeon J, Suarez A, Shannahan J, … Watson-Wright C (2020). Toxicological analysis of aerosols derived from three electronic nicotine delivery systems using normal human bronchial epithelial cells. Toxicology In Vitro, 69 104997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson JL, Richardson A, Niaura RS, Vallone DM, & Abrams DB (2012). E-cigarette awareness, use, and harm perceptions in US adults. American Journal of Public Health 102, 1758–1766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prieux R, Eeman M, Rothen-Rutishauser B, & Valacchi G (2020). Mimicking cigarette smoke exposure to assess cutaneous toxicity. Toxicology In Vitro 62, 104664. [DOI] [PubMed] [Google Scholar]
- Protano C, Avino P, Manigrasso M, Vivaldi V, Perna F, Valeriani F, & Vitali M (2018). Environmental electronic vape exposure from four different generations of electronic cigarettes: Airborne particulate matter levels. International Journal of Environmental Research and Public Health 15, 2172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Protano C, Cattaruzza MS, Osborn JF, & Vitali M (2014). Indoor particulate matter and secondhand smoke: Simulation of an exposure scenario. Annali di Igiene 26, 186–189. [DOI] [PubMed] [Google Scholar]
- Putzhammer R, Doppler C, Jakschitz T, Heinz K, Förste J, Danzl K, … Bernhard D (2016). Vapours of US and EU market leader electronic cigarette brands and liquids are cytotoxic for human vascular endothelial cells. PLoS One 11, Article e0157337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramamurthi D, Chau C, & Jackler RK (2019). JUUL and other stealth vaporisers: Hiding the habit from parents and teachers. Tobacco Control 28, 610–616. [DOI] [PubMed] [Google Scholar]
- Rao P, Liu J, & Springer ML (2020). JUUL and combusted cigarettes comparably impair endothelial function. Tobacco Regulatory Science 6, 30–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reidel B, Radicioni G, Clapp PW, Ford AA, Abdelwahab S, Rebuli ME, … Kesimer M (2018). E-cigarette use causes a unique innate immune response in the lung, involving increased neutrophilic activation and altered mucin secretion. American Journal of Respiratory and Critical Care Medicine 197, 492–501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reile R, & Parna K (2020). E-cigarette use by smoking status in Estonia, 2012–2018. International Journal of Environmental Research and Public Health 17, 519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reumann MK, Schaefer J, Titz B, Aspera-Werz RH, Wong ET, Szostak J, … Nussler AK (2020). E-vapor aerosols do not compromise bone integrity relative to cigarette smoke after 6-month inhalation in an ApoE−/− mouse model. Archives of Toxicology 94, 2163–2177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts ME, Keller-Hamilton B, Ferketich AK, & Berman ML (2020). JUUL and the upsurge of e-cigarette use among college undergraduates. Journal of American College Health. 10.1080/07448481.2020.1726355 E-print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Romagna G, Allifranchini E, Bocchietto E, Todeschi S, Esposito M, & Farsalinos KE (2013). Cytotoxicity evaluation of electronic cigarette vapor extract on cultured mammalian fibroblasts (ClearStream-LIFE): Comparison with tobacco cigarette smoke extract. Inhalation Toxicology 25, 354–361. [DOI] [PubMed] [Google Scholar]
- Romberg AR, Miller Lo EJ, Cuccia AF, Willett JG, Xiao H, Hair EC, … King BA (2019). Patterns of nicotine concentrations in electronic cigarettes sold in the United States, 2013–2018. Drug and Alcohol Dependence 203, 1–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rostami AA, Agyemang S, & Pithawalla YB (2018). A distributed computational model for estimating room air level of constituents due to aerosol emission from e-vapor product use. Food and Chemical Toxicology 116, 114–128. [DOI] [PubMed] [Google Scholar]
- Rostami AA, Pithawalla YB, Liu J, Oldham MJ, Wagner KA, Frost-Pineda K, & Sarkar MA (2016). A well-mixed computational model for estimating room air levels of selected constituents from e-vapor product use. International Journal of Environmental Research and Public Health 13, 828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rostron BL, Cheng YC, Gardner LD, & Ambrose BK (2020). Prevalence and reasons for use of flavored cigars and ends among US youth and adults: Estimates from Wave 4 of the PATH study, 2016–2017. American Journal of Health Behavior 44, 76–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowell TR, Keating JE, Zorn BT, Glish GL, Shears SB, & Tarran R (2020). Flavored e-liquids increase cytoplasmic Ca2+ levels in airway epithelia. American Journal of Physiology. Lung Cellular and Molecular Physiology 318, L226–L241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rowell TR, Reeber SL, Lee SL, Harris RA, Nethery RC, Herring AH, … Tarran R (2017). Flavored e-cigarette liquids reduce proliferation and viability in the CALU3 airway epithelial cell line. American Journal of Physiology. Lung Cellular and Molecular Physiology 313, L52–L66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruszkiewicz JA, Zhang Z, Gonçalves FM, Tizabi Y, Zelikoff JT, & Aschner M (2020). Neurotoxicity of e-cigarettes. Food and Chemical Toxicology 138, 111245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saddleson ML, Kozlowski LT, Giovino GA, Hawk LW, Murphy JM, MacLean MG, … Mahoney MC (2015). Risky behaviors, e-cigarette use and susceptibility of use among college students. Drug and Alcohol Dependence 149, 25–30. [DOI] [PubMed] [Google Scholar]
- Saffari A, Daher N, Ruprecht A, De Marco C, Pozzi P, Boffi R, … Sioutas C (2014). Particulate metals and organic compounds from electronic and tobacco-containing cigarettes: Comparison of emission rates and secondhand exposure. Environmental Science. Processes & Impacts 16, 2259–2267. [DOI] [PubMed] [Google Scholar]
- Saliba NA, El Hellani A, Honein E, Salman R, Talih S, Zeaiter J, & Shihadeh A (2018). Surface chemistry of electronic cigarette electrical heating coils: Effects of metal type on propylene glycol thermal decomposition. Journal of Analytical and Applied Pyrolysis 134, 520–525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Samburova V, Bhattarai C, Strickland M, Darrow L, Angermann J, Son Y, & Khlystov A (2018). Aldehydes in exhaled breath during e-cigarette vaping: Pilot study results. Toxics 6, 46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sassano MF, Davis ES, Keating JE, Zorn BT, Kochar TK, Wolfgang MC, … Tarran R (2018). Evaluation of e-liquid toxicity using an open-source high-throughput screening assay. PLoS Biology 16, Article e2003904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schier JG, Meiman JG, Layden J, Mikosz CA, VanFrank B, King BA, … Meaney-Delman D (2019). Severe pulmonary disease associated with electronic-cigarette-product use - interim guidance. MMWR. Morbidity and Mortality Weekly Report 68, 787–790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt S (2020). Vaper, beware: The unique toxicological profile of electronic cigarettes 128. (pp. 052001), 052001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoenborn CA, & Gindi RM (2015). Electronic cigarette use among adults: United States, 2014. NCHS Data Brief 217, 1–8. [PubMed] [Google Scholar]
- Schraufnagel DE, Blasi F, Drummond MB, Lam DC, Latif E, Rosen MJ, … Van Zyl-Smit R (2014). Electronic cigarettes. A position statement of the forum of international respiratory societies. American Journal of Respiratory and Critical Care Medicine 190, 611–618. [DOI] [PubMed] [Google Scholar]
- Schripp T, Markewitz D, Uhde E, & Salthammer T (2013). Does e-cigarette consumption cause passive vaping? Indoor Air 23, 25–31. [DOI] [PubMed] [Google Scholar]
- Scott A, Lugg ST, Aldridge K, Lewis KE, Bowden A, Mahida RY, … Thickett DR (2018). Pro-inflammatory effects of e-cigarette vapour condensate on human alveolar macrophages. Thorax 73, 1161–1169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sherwood CL, & Boitano S (2016). Airway epithelial cell exposure to distinct e-cigarette liquid flavorings reveals toxicity thresholds and activation of CFTR by the chocolate flavoring 2,5-dimethypyrazine. Respiratory Research 17, 57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh J, Luquet E, Smith DPT, Potgieter HJ, & Ragazzon P (2016). Toxicological and analytical assessment of e-cigarette refill components on airway epithelia. Science Progress 99, 351–398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh T, Arrazola RA, Corey CG, Husten CG, Neff LJ, Homa DM, & King BA (2016). Tobacco use among middle and high school students–United States, 2011–2015. MMWR. Morbidity and Mortality Weekly Report 65, 361–367. [DOI] [PubMed] [Google Scholar]
- Smith D, Aherrera A, Lopez A, Neptune E, Winickoff JP, Klein JD, … McGrath-Morrow SA (2015). Adult behavior in male mice exposed to e-cigarette nicotine vapors during late prenatal and early postnatal life. PLoS One 10, Article e0137953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith DM, Miller C, O’Connor RJ, Kozlowski LT, Wadsworth E, Fix BV, & Hammond D (2020). Modes of delivery in concurrent nicotine and cannabis use (“co-use”) among youth: Findings from the International Tobacco Control (ITC) survey. Substance Abuse. 10.1080/08897077.2019.1709603 E-print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith DM, Schneller LM, O’Connor RJ, & Goniewicz ML (2019). Are e-cigarette flavors associated with exposure to nicotine and toxicants? Findings from Wave 2 of the population assessment of tobacco and health (PATH) study. International Journal of Environmental Research and Public Health 16, 5055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith TT, Heckman BW, Wahlquist AE, Cummings KM, & Carpenter MJ (2020). The impact of e-liquid propylene glycol and vegetable glycerin ratio on ratings of subjective effects, reinforcement value, and use in current smokers. Nicotine & Tobacco Research 22, 791–797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song JJ, Go YY, Mun JY, Lee S, Im GJ, Kim YY, … Chang J (2018). Effect of electronic cigarettes on human middle ear. International Journal of Pediatric Otorhinolaryngology 109, 67–71. [DOI] [PubMed] [Google Scholar]
- Sosnowski TR, Jablczynska K, Odziomek M, Schlage WK, & Kuczaj AK (2018). Physicochemical studies of direct interactions between lung surfactant and components of electronic cigarettes liquid mixtures. Inhalation Toxicology 30, 159–168. [DOI] [PubMed] [Google Scholar]
- Sosnowski TR, & Kramek-Romanowska K (2016). Predicted deposition of e-cigarette aerosol in the human lungs. Journal of Aerosol Medicine and Pulmonary Drug Delivery 29, 299–309. [DOI] [PubMed] [Google Scholar]
- Soteriades S, Barbouni A, Rachiotis G, Grevenitou P, Mouchtouri V, Pinaka O, … Hadjichristodoulou C (2020). Prevalence of electronic cigarette use and its determinants among 13-to-15-year-old students in Greece: Results from the 2013 Global Youth Tobacco Survey (GYTS). International Journal of Environmental Research and Public Health 17, 1671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soule EK, Lopez AA, Guy MC, & Cobb CO (2016). Reasons for using flavored liquids among electronic cigarette users: A concept mapping study. Drug and Alcohol Dependence 166, 168–176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soulet S, Duquesne M, Toutain J, Pairaud C, & Lalo H (2019). Experimental method of emission generation calibration based on reference liquids characterization. International Journal of Environmental Research and Public Health 16, 2262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- St Helen G, Dempsey DA, Havel CM, Jacob P 3rd, & Benowitz NL (2017). Impact of e-liquid flavors on nicotine intake and pharmacology of e-cigarettes. Drug and Alcohol Dependence 178, 391–398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- STEMCELL Technologies Inc (2019). Air-liquid interface culture for respiratory research. https://www.stemcell.com/air-liquid-interface-culture-respiratory-research-lp.html/ (accessed May 22, 2020).
- Stevenson M, Czekala L, Simms L, Tschierske N, Larne O, & Walele T (2019). The use of genomic allergen rapid detection (GARD) assays to predict the respiratory and skin sensitising potential of e-liquids. Regulatory Toxicology and Pharmacology 103, 158–165. [DOI] [PubMed] [Google Scholar]
- Sundar IK, Javed F, Romanos GE, & Rahman I (2016). E-cigarettes and flavorings induce inflammatory and pro-senescence responses in oral epithelial cells and periodontal fibroblasts. Oncotarget 7, 77196–77204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sutfin EL, McCoy TP, Morrell HE, Hoeppner BB, & Wolfson M (2013). Electronic cigarette use by college students. Drug and Alcohol Dependence 131, 214–221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Syamlal G, Jamal A, King BA, & Mazurek JM (2016). Electronic cigarette use among working adults - United States, 2014. MMWR. Morbidity and Mortality Weekly Report 65, 557–561. [DOI] [PubMed] [Google Scholar]
- Szafran BN, Pinkston R, Perveen Z, Ross MK, Morgan T, Paulsen DB, … Noël A (2020). Electronic-cigarette vehicles and flavoring affect lung function and immune responses in a murine model. International Journal of Molecular Sciences 21, 6022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szostak J, Wong ET, Titz B, Lee T, Wong SK, Low T, … Hoeng J (2020). A 6-month systems toxicology inhalation study in ApoE−/− mice demonstrates reduced cardiovascular effects of e-vapor aerosols compared with cigarette smoke. American Journal of Physiology. Heart and Circulatory Physiology 318, H604–H631. [DOI] [PubMed] [Google Scholar]
- Talih S, Salman R, El-Hage R, Karam E, Karaoghlanian N, El-Hellani A, … Shihadeh A (2019). Characteristics and toxicant emissions of JUUL electronic cigarettes. Tobacco Control 28, 678–680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talih S, Salman R, Karaoghlanian N, El-Hellani A, Saliba N, Eissenberg T, & Shihadeh A (2017). “Juice monsters”: Sub-ohm vaping and toxic volatile aldehyde emissions. Chemical Research in Toxicology 30, 1791–1793. [DOI] [PubMed] [Google Scholar]
- Tang MS, Wu XR, Lee HW, Xia Y, Deng FM, Moreira AL, … Lepor H (2019). Electronic-cigarette smoke induces lung adenocarcinoma and bladder urothelial hyperplasia in mice. Proceedings of the National Academy of Sciences of the United States of America 116, 21727–21731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor J, Wiens T, Peterson J, Saravia S, Lunda M, Hanson K, … Holzbauer S (2019). Characteristics of e-cigarette, or vaping, products used by patients with associated lung injury and products seized by law enforcement - Minnesota, 2018 and 2019. MMWR. Morbidity and Mortality Weekly Report 68, 1096–1100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Troutt WD, & DiDonato MD (2017). Carbonyl compounds produced by vaporizing cannabis oil thinning agents. Journal of Alternative and Complementary Medicine 23, 879–884. [DOI] [PubMed] [Google Scholar]
- Truman P, Stanfill S, Heydari A, Silver E, & Fowles J (2019). Monoamine oxidase inhibitory activity of flavoured e-cigarette liquids. NeuroToxicology 75, 123–128. [DOI] [PubMed] [Google Scholar]
- Tsai J, Walton K, Coleman BN, Sharapova SR, Johnson SE, Kennedy SM, & Caraballo RS (2018). Reasons for electronic cigarette use among middle and high school students - national youth tobacco survey, United States, 2016. MMWR. Morbidity and Mortality Weekly Report 67, 196–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai KYF, Hirschi Budge KM, Lepre AP, Rhees MS, Ajdaharian J, Geiler J, … Reynolds PR (2020). Cell invasion, rage expression, and inflammation in oral squamous cell carcinoma (oscc) cells exposed to e-cigarette flavoring. Clinical and Experimental Dental Research 6, 618–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzortzi A, Kapetanstrataki M, Evangelopoulou V, & Beghrakis P (2020). A systematic literature review of e-cigarette-related illness and injury: Not just for the respirologist. International Journal of Environmental Research and Public Health 17, 2248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzortzi A, Teloniatis S, Matiampa G, Bakelas G, Tzavara C, Vyzikidou VK, … Fernandez E (2020). Passive exposure of non-smokers to e-cigarette aerosols: Sensory irritation, timing and association with volatile organic compounds. Environmental Research 182, 108963. [DOI] [PubMed] [Google Scholar]
- U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health (2014). The health consequences of smoking – 50 years of progress: A report of the surgeon general.
- Vallone DM, Cuccia AF, Briggs J, Xiao H, Schillo BA, & Hair EC (2020). Electronic cigarette and JUUL use among adolescents and young adults. Journal of American Medical Association Pediatrics 174, 277–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varlet V (2016). Drug vaping: From the dangers of misuse to new therapeutic devices. Toxics 4, 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Varlet V, Concha-Lozano N, Berthet A, Plateel G, Favrat B, De Cesare M, … Giroud C (2016). Drug vaping applied to cannabis: Is “cannavaping” a therapeutic alternative to marijuana? Scientific Reports 6, 25599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Villanti AC, Johnson AL, Ambrose BK, Cummings KM, Stanton CA, Rose SW, …Hyland A (2017). Flavored tobacco product use in youth and adults: Findings from the first wave of the PATH study (2013–2014). American Journal of Preventive Medicine 53, 139–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walele T, Sharma G, Savioz R, Martin C, & Williams J (2016). A randomised, crossover study on an electronic vapour product, a nicotine inhalator and a conventional cigarette. Part A: Pharmacokinetics. Regulatory Toxicology and Pharmacology 74, 187–192. [DOI] [PubMed] [Google Scholar]
- Wang TW, Gentzke AS, Creamer MR, Cullen KA, Holder-Hayes E, Sawdey MD, … Neff LJ (2019). Tobacco product use and associated factors among middle and high school students - United States, 2019. MMWR Surveillance Summaries 68, 1–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang TW, Neff L, Park-Lee E, Ren C, Cullen KA, & King B (2020). E-cigarette use among middle and high school students — United States, 2020. MMWR. Morbidity and Mortality Weekly Report 69, 1310–1312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wavreil FDM, & Heggland SJ (2020). Cinnamon-flavored electronic cigarette liquids and aerosols induce oxidative stress in human osteoblast-like mg-63 cells. Toxicology Reports 7, 23–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ween MP, Hamon R, Macowan MG, Thredgold L, Reynolds PN, & Hodge SJ (2020). Effects of e-cigarette e-liquid components on bronchial epithelial cells: Demonstration of dysfunctional efferocytosis. Respirology 25, 620–628. [DOI] [PubMed] [Google Scholar]
- Ween MP, Whittall JJ, Hamon R, Reynolds PN, & Hodge SJ (2017). Phagocytosis and inflammation: Exploring the effects of the components of e-cigarette vapor on macrophages. Physiological Reports 5, Article e13370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welz C, Canis M, Schwenk-Zieger S, Becker S, Stucke V, Ihler F, & Baumeister P (2016). Cytotoxic and genotoxic effects of electronic cigarette liquids on human mucosal tissue cultures of the oropharynx. Journal of Environmental Pathology, Toxicology and Oncology 35, 343–354. [DOI] [PubMed] [Google Scholar]
- Werley MS, Kirkpatrick DJ, Oldham MJ, Jerome AM, Langston TB, Lilly PD, … McKinney WJ Jr. (2016). Toxicological assessment of a prototype e-cigaret device and three flavor formulations: A 90-day inhalation study in rats. Inhalation Toxicology 28, 22–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wills TA, Choi K, & Pagano I (2020). E-cigarette use associated with asthma independent of cigarette smoking and marijuana in a 2017 national sample of adolescents. The Journal of Adolescent Health 67, 524–530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong BA (2007). Inhalation exposure systems: Design, methods and operation. Toxicologic Pathology 35, 3–14. [DOI] [PubMed] [Google Scholar]
- Wu D, & O’Shea DF (2020). Potential for release of pulmonary toxic ketene from vaping pyrolysis of vitamin E acetate. Proceedings of the National Academy of Sciences of the United States of America 117, 6349–6355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahedi A, Phandthong R, Chaili A, Leung S, Omaiye E, & Talbot P (2019). Mitochondrial stress response in neural stem cells exposed to electronic cigarettes. iScience 16, 250–269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahedi A, Phandthong R, Chaili A, Remark G, & Talbot P (2018). Epithelial-to-mesenchymal transition of A549 lung cancer cells exposed to electronic cigarettes. Lung Cancer 122, 224–233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zelikoff JT, Parmalee NL, Corbett K, Gordon T, Klein CB, & Aschner M (2018). Microglia activation and gene expression alteration of neurotrophins in the hippocampus following early-life exposure to e-cigarette aerosols in a murine model. Toxicological Sciences 162, 276–286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao D, Aravindakshan A, Hilpert M, Olmedo P, Rule AM, Navas-Acien A, & Aherrera A (2020). Metal/metalloid levels in electronic cigarette liquids, aerosols, and human biosamples: A systematic review. Environmental Health Perspectives 128, 36001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao J, Zhang Y, Sisler JD, Shaffer J, Leonard SS, Morris AM, … Demokritou P (2018). Assessment of reactive oxygen species generated by electronic cigarettes using acellular and cellular approaches. Journal of Hazardous Materials 344, 549–557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao T, Nguyen C, Lin CH, Middlekauff HR, Peters K, Moheimani R, … Zhu Y (2017). Characteristics of secondhand electronic cigarette aerosols from active human use. Aerosol Science and Technology 51, 1368–1376. [Google Scholar]
- Zhao T, Shu S, Guo Q, & Zhu Y (2016). Effects of design parameters and puff topography on heating coil temperature and mainstream aerosols in electronic cigarettes. Atmospheric Environment 134, 61–69. [Google Scholar]
- Zhu SH, Sun JY, Bonnevie E, Cummins SE, Gamst A, Yin L, & Lee M (2014). Four hundred and sixty brands of e-cigarettes and counting: Implications for product regulation. Tobacco Control 23(Suppl. 3), iii3–iii9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zscheppang K, Berg J, Hedtrich S, Verheyen L, Wagner DE, Suttorp N, … Hocke AC (2018). Human pulmonary 3D models for translational research. Biotechnology Journal 13, 1700341. [DOI] [PMC free article] [PubMed] [Google Scholar]


