Abstract
Objectives
To report a case of povidone-iodine (PVP-I, Iso-Betadine®) disinfection of lower leg fasciotomy wounds resulting in iodide absorption and possibly contributing to hypothyroidism and prolonged kidney injury.
Design
Case report. Setting. Pediatric intensive care unit (PICU), university hospital. Patients. A 13-year-old patient presenting with prolonged oligoanuric kidney failure and unexplained primary hypothyroidism three weeks after severe abdominal sepsis with multiple organ dysfunction and major rhabdomyolysis due to bilateral lower leg compartment syndrome, necessitating moderate size fasciotomies, disinfected daily with PVP-I. Interventions. Interruption of PVP-I exposure and initiation of thyroid hormone substitution. Measurements and Main Results. Hypothyroidism was revealed during diagnostic work-up for persistent hypertriglyceridemia. Thyroxine (T4) (4.0 mg/L) and tri-iodothyronine (T3) (64 ng/L) were moderately low, yet thyroid stimulating hormone (TSH) (16.8 mIU/L) was fourfold the maximal normal range value. This pattern, atypical for prolonged critical illness-related hypothyroidism, prompted interruption of PVP-I exposure and initiation of thyroid hormone substitution. Urinary production and creatinine clearance recovered during the following days, and one week later, intermittent renal replacement therapy could be terminated, suggesting that PVP-I toxicity and/or hypothyroidism may have contributed to the persistent renal failure three weeks after resolved septic shock and rhabdomyolysis. Elevated serum and urinary anion gap normalized simultaneously, but this evolution of rather nonspecific indices could be multifactorial.
Conclusion
PVP-I is a commonly used broad-spectrum antimicrobial agent for prevention and treatment of wound infections. Toxic complications due to PVP-I absorption, after disinfection of extended thermal injuries larger than 20% of the body surface, have been described. In critically ill children, however, toxic effects of PVP-I may occur due to repeated disinfection of less extended wounds. Proposed screening strategies include: monitoring of the volumes of PVP-I applied daily; of the thyroid function, the serum, and/or urinary anion gap and the urinary iodide concentrations. These strategies, however, remain to be validated. This case report should be a wake-up call for daily integration of wound management in the clinical evaluation of critically ill patients.
1. Introduction
PVP-I (Iso-Betadine®) is a broad-spectrum antimicrobial agent commonly used for prevention and treatment of wound colonization and infections. It is a chemical complex of iodine with microbicidal activity and polyvinylpyrrolidone (povidone), a synthetic polymer. The antiseptic is widely used as a 10% solution in water, containing 1% iodine [1, 2]. Despite its potential beneficial effects on wound healing [3], there are several reports published on metabolic and toxic complications due to absorption of the therapeutic agent [4, 5], especially after disinfection of thermal injuries larger than 20% of the body surface [6].
We here present the atypical case of PVP-I disinfection of moderate size fasciotomy wounds resulting in unexpected intoxication and hypothyroidism and possibly contributing to delayed recovery of acute kidney failure in the context of resolved septic shock.
2. Case Report
We present the case of a 13-year-old boy who was hospitalized at the PICU of the University Hospitals Leuven after acute laparotomy for small bowel obstruction resulting in an ischemic bowel and a 30 cm enterectomy. Postoperatively, the patient suffered severe septic shock with acute septic cardiomyopathy and multiple organ failure including liver failure and disseminated intravascular coagulation (DIC). Within hours, he developed a compartment syndrome of the lower extremities and concurrent rhabdomyolysis with a level of creatine kinase (CK) reaching to 250000 U/L (reference ≤190 U/L), requiring urgent medial and lateral fasciotomy of both lower legs on ICU day 1. CK levels declined promptly to 40000 U/L on ICU day 3, 4834 U/L on ICU day 6, and 351 U/L by ICU day 7. Acute kidney injury (AKI) initially required continuous venovenous hemofiltration (CVVH) and, after 10 days, intermittent hemodialysis (IHD) therapy. Fasciotomy wounds were initially marginally perfused with important edema of the lower legs and feet, necessitating postponing the closure of the fasciotomy wounds until after a month. In order to prevent infection in these high-risk wounds, the subcutaneous tissue and the surrounding skin was disinfected daily with antiseptic PVP-I solution at a concentration of 10%. Wound cultures remained sterile during the entire open wound treatment interval, and from ICU day 12 onward, daily wound care was skipped on several occasions allowing the patient some rest, given the good clinical wound evolution (see Table 1).
Table 1.
Evolution from ICU day 0 until day 54 of serum levels of TSH, T3, T4, and TBG and of urinary iodine concentration. Wound care applied on these ICU days.
| ICU day | TSH (mIU/L) | T3 total (ng/dL) | T4 total (mg/dL) | TBG (mg/L) | Urinary iodine (mg/L) | Wound care fasciotomy wounds |
|---|---|---|---|---|---|---|
| Reference | 0.27-4.20 | 80-200 | 5.1-14.1 | 14.0-24.0 | ≤100 | |
| 0 | ||||||
| 1 | ||||||
| 2 | NaCl 0.9%+Iso-Betadine® gel | |||||
| 3 | NaCl 0.9% | |||||
| 4 | ||||||
| 5 | ||||||
| 6 | NaCl 0.9%+Iso-Betadine® tulle | |||||
| 7 | ||||||
| 8 | ||||||
| 9 | NaCl 0.9%+Dakin Cooper®+Iso-Betadine® tulle | |||||
| 10 | NaCl 0.9%+Dakin Cooper® | |||||
| 11 | NaCl 0.9%+Dakin Cooper®+Iso-Betadine® tulle | |||||
| 12 | NaCl 0.9%+Dakin Cooper®+Iso-Betadine® tulle | |||||
| 13 | ||||||
| 14 | NaCl 0.9%+Dakin Cooper®+Iso-Betadine® dressing | |||||
| 15 | NaCl 0.9%+Braunol® 7.5% | |||||
| 16 | NaCl 0.9%+Dakin Cooper® | |||||
| 17 | NaCl 0.9% | |||||
| 18 | NaCl 0.9% | |||||
| 19 | ||||||
| 20 | ||||||
| 21 | NaCl 0.9%+Braunol® 7.5% | |||||
| 22 | ||||||
| 23 | 16.8 | 64 | 4.0 | 22.1 | ||
| 24 | NaCl 0.9% | |||||
| 25 | 13.3 | 59 | 3.9 | 20.6 | ||
| 26 | ||||||
| 27 | ||||||
| 28 | 5.35 | 44 | 6.4 | 23.8 | ||
| 29 | >10000 | NaCl 0.9%+chlorhexidine digluconate 0.5% in ethanol 70%+Flaminal Forte® | ||||
| 30 | ||||||
| 31 | >10000 | |||||
| 32 | NaCl 0.9%+Flamigel® | |||||
| 33 | ||||||
| 34 | ||||||
| 35 | 5.03 | 59 | 4.5 | 19.7 | ||
| 36 | NaCl 0.9%+Flaminal Forte® | |||||
| 37 | ||||||
| 38 | >10000 | NaCL 0.9%+Dakin Cooper® | ||||
| 39 | ||||||
| 40 | ||||||
| 41 | NaCl 0.9% | |||||
| 42 | 3.51 | 82 | 7.8 | 19.0 | ||
| 43 | ||||||
| 44 | 3413 | NaCl 0.9%+Flaminal Forte® | ||||
| 45 | NaCl 0.9%+Flaminal Forte® | |||||
| 46 | NaCl 0.9%+Flaminal Forte® | |||||
| 47 | NaCl 0.9% | |||||
| 48 | NaCl 0.9%+Flaminal Forte® | |||||
| 49 | 4.28 | 71 | 5.3 | 17.2 | NaCl 0.9%+Flaminal Forte® | |
| 50 | NaCl 0.9%+Flaminal Forte® | |||||
| 51 | NaCl 0.9%+chlorhexidine digluconate 0.5% in ethanol 70%+Flaminal Forte | |||||
| 52 | NaCl 0.9%+Flaminal Forte® | |||||
| 53 | 7.82 | |||||
| 54 |
Abbreviations: T3: tri-iodothyronine; T4: thyroxine; TSH: thyroid stimulating hormone; TBG: thyroid binding globulin.
Septic shock, liver failure, and DIC resolved within a week, and the patient was weaned from the ventilator by day 14. Acute kidney injury however did not recover (see Table 2), and triglycerides were rising without apparent reason, contributing potentially to acute pancreatitis. Diagnostic work-up revealed plasma TSH levels at day 23 that were highly increased to 16.8 mU/L (reference range 0.27-4.20 mU/L), levels of T4 4.0 mg/L (reference range 5.1 -14.1) and T3 64 ng/dL (reference range 80-200 ng/dL) was diminished, whereas reverse T3 was slightly increased, and reverse T3 was slightly increased (42 ng/dL). There was a high suspicion of hypothyroidism secondary to absorption of povidone iodine after 3 weeks of extensive wound care (see wound pictures taken on day 22 in Figure 1) with PVP-I. Significant systemic iodine absorption was documented by very high levels of urinary iodine excretion (>10,000 μg/L). Disinfection with PVP-I was formally stopped at ICU day 23 (last application on ICU day 21) and thyroid hormone suppletion was initiated. There was an elevated urinary anion gap of 42.2 mmol/L. At days 29 and 31, urinary iodine level was at a toxic concentration of more than 10,000 μg/L with a urinary anion gap of 40.4 mmol/L. Once the exposure was interrupted, urinary iodine values gradually decreased and urinary output and measured creatine clearance increased as soon as 4 days later, in the absence of diuretics and with increasing urinary creatinine excretion (Tables 2 and 3 and Figure 2). IHD could be interrupted at day 30.
Table 2.
Evolution from ICU day 0 until 54 of measured creatinine clearance, serum creatinine, 24 h urine output volumes, and urinary iodine concentrations during ICU stay.
| ICU day | Creatinine clearance, BSA corrected (mL/min) | Creatinine (mg/dL) | Diuresis 24 h (mL) | Urinary iodine (mg/L) |
|---|---|---|---|---|
| Reference | 0.46-0.77 | ≤100 | ||
| 0 | 0.97 | 182 | ||
| 1 | CVVH | Start CVVH | 140 | |
| 2 | CVVH | CVVH | 20 | |
| 3 | CVVH | CVVH | 25 | |
| 4 | CVVH | CVVH | ||
| 5 | CVVH | CVVH | 17 | |
| 6 | CVVH | CVVH | 8 | |
| 7 | CVVH | CVVH | 2 | |
| 8 | CVVH | CVVH | 4 | |
| 9 | CVVH | CVVH | ||
| 10 | CVVH on hold | CVVH on hold | 8 | |
| 11 | CVVH on hold | CVVH on hold | 22 | |
| 12 | IHD | IHD | 30 | |
| 13 | 67 | |||
| 14 | IHD | IHD | 44 | |
| 15 | 35 | |||
| 16 | IHD | IHD | 30 | |
| 17 | 25 | |||
| 18 | 40 | |||
| 19 | IHD | IHD | 42 | |
| 20 | 74 | |||
| 21 | IHD | IHD | 56 | |
| 22 | 147 | |||
| 23 | IHD | IHD | 130 | |
| 24 | 200 | |||
| 25 | IHD | IHD | 150 | |
| 26 | 200 | |||
| 27 | 475 | |||
| 28 | IHD | IHD | 400 | |
| 29 | 550 | >10000 | ||
| 30 | IHD | IHD | 550 | |
| 31 | 6.44 | 2.60 | 900 | >10000 |
| 32 | 8.6 | 3.15 | 1000 | |
| 33 | 9.22 | 3.19 | 1200 | |
| 34 | 15.3 | 2.96 | 1100 | |
| 35 | 16.07 | 2.56 | 1400 | |
| 36 | 18.6 | 2.36 | 1800 | |
| 37 | 23.04 | 2.12 | 2050 | |
| 38 | 33.11 | 1.68 | 1750 | >10000 |
| 39 | 39.75 | 1.32 | 1900 | |
| 40 | 53.02 | 0.96 | 1600 | |
| 41 | 55.83 | 0.82 | 1500 | |
| 42 | 65.71 | 0.70 | 1700 | |
| 43 | 67.83 | 0.64 | 1600 | |
| 44 | 67.52 | 0.57 | 1350 | 3413 |
| 45 | 44.67 | 0.52 | 1600 | |
| 46 | 81.7 | 0.48 | 1500 | |
| 47 | 90.03 | 0.49 | 1600 | |
| 48 | 99.95 | 0.50 | 1300 | |
| 49 | 90.62 | 0.48 | 1500 | |
| 50 | 68.30 | 0.64 | 1300 | |
| 51 | 94.12 | 0.46 | 1500 | |
| 52 | 82.71 | 0.47 | ||
| 53 | 0.41 | |||
| 54 | 0.44 |
Figure 1.
Fasciotomy wounds on both sides of both lower legs.
Table 3.
Evolution from ICU day 0 until day 54 of serum anion gap and electrolytes, arterial lactate, urine iodine concentration, anion gap, and electrolytes.
| ICU day | Serum | Arterial | Urinary | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Anion gap (mEq/L) | Cl− (mmol/L) | HCO3− (mmol/L) | Lactate (mmol/L) | Iodine (mg/L) | Anion gap (mEq/L) | Cl− (mmol/L) | K+ (mmol/L) | Na+ (mmol/L) | |
| Reference | Sep-20 | 98.0-107.0 | 22.0-29.0 | 0.5-2.2 | ≤100 | ||||
| 0 | 30.7 | 101.2 | 14.2 | 8.9 | |||||
| 1 | 34.7 | 98.7 | 15.7 | 3.6 | |||||
| 2 | 19.7 | 102.3 | 21.9 | 3.5 | |||||
| 3 | 18.1 | 102.6 | 21.6 | 2.5 | |||||
| 4 | 20.4 | 101.9 | 20.5 | 3.6 | |||||
| 5 | 19.0 | 100.9 | 21.6 | 2.2 | |||||
| 6 | 17.5 | 102.9 | 22.3 | 1.0 | |||||
| 7 | 15.5 | 104.6 | 21.6 | 0.8 | |||||
| 8 | 18.2 | 101.6 | 20.3 | 0.9 | |||||
| 9 | 20.5 | 96.2 | 22.8 | 0.8 | |||||
| 10 | 19.9 | 97.0 | 24.1 | 0.9 | |||||
| 11 | 19.1 | 96.1 | 22.6 | 1.3 | |||||
| 12 | 20.8 | 96.8 | 18.5 | 0.8 | |||||
| 13 | 23.8 | 97.0 | 16.2 | 0.8 | |||||
| 14 | 27.2 | 93.4 | 18.1 | 0.6 | |||||
| 15 | 33.3 | 88.4 | 21.3 | 0.7 | |||||
| 16 | 25.5 | 93.5 | 25.1 | 1.0 | |||||
| 17 | 22.3 | 95.5 | 27.7 | 1.0 | |||||
| 18 | 23.6 | 91.7 | 29.8 | 0.8 | |||||
| 19 | 21.0 | 93.3 | 21.5 | 0.8 | |||||
| 20 | 27.0 | 92.3 | 19.6 | 1.0 | |||||
| 21 | 28.1 | 88.7 | 22.2 | 1.0 | |||||
| 22 | 25.2 | 94.2 | 19.7 | 0.7 | |||||
| 23 | 28.1 | 91.5 | 19.0 | 0.7 | |||||
| 24 | 23.3 | 99.2 | 17.5 | 0.7 | 42.2 | 53 | 28 | 67.2 | |
| 25 | 24.1 | 95.4 | 19.9 | 1.0 | |||||
| 26 | 20.2 | 102.5 | 17.7 | 0.7 | |||||
| 27 | 20.2 | 98.7 | 21.0 | 0.8 | |||||
| 28 | 24.0 | 96.6 | 23.7 | 0.7 | |||||
| 29 | 21.3 | 101.6 | 19.2 | 1.0 | >10000 | ||||
| 30 | 22.6 | 102.2 | 24.6 | 1.6 | |||||
| 31 | 19.9 | 104.3 | 22.1 | 0.9 | >10000 | 40.4 | 54 | 24 | 70.4 |
| 32 | 21.3 | 101.1 | 25.7 | 0.9 | |||||
| 33 | 22.7 | 102.0 | 28 | 0.8 | |||||
| 34 | 20.8 | 102.7 | 25.8 | 0.7 | |||||
| 35 | 21.2 | 109.4 | 21.2 | 0.7 | |||||
| 36 | 22.2 | 110.8 | 16.7 | 0.8 | |||||
| 37 | 22.2 | 113.1 | 16.1 | 0.8 | |||||
| 38 | 15.6 | 123.9 | 16.8 | >10000 | |||||
| 39 | 17.8 | 116.1 | 17.1 | ||||||
| 40 | 19.6 | 112.1 | 16.8 | ||||||
| 41 | 18.7 | 113.1 | 15.0 | ||||||
| 42 | 14.2 | 111.7 | 17.6 | ||||||
| 43 | 15.5 | 107.6 | 18.6 | ||||||
| 44 | 19.7 | 104.0 | 16.0 | 3413 | |||||
| 45 | 19.3 | 104.6 | 17.5 | ||||||
| 46 | 15.9 | 105.4 | 18.9 | ||||||
| 47 | 17.6 | 105.3 | 19.5 | ||||||
| 48 | 21.2 | 98.3 | 18.4 | ||||||
| 49 | 22.1 | 98.9 | 19.6 | 20.4 | 26 | 24 | 22.4 | ||
| 50 | 18.7 | 102.9 | 21.4 | ||||||
| 51 | 16.3 | 101.2 | 24.2 | ||||||
| 52 | 16.3 | 99.1 | 26.9 | ||||||
| 53 | 15.9 | 102.1 | 25.5 | ||||||
| 54 | 16.1 | 103.8 | 23.7 | ||||||
Figure 2.
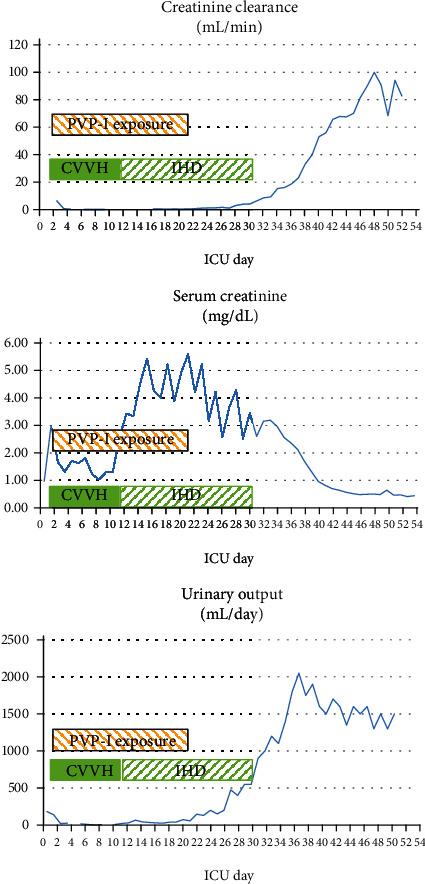
Spontaneous recovery of measured creatinine clearance, normalization of serum creatinine, and urinary output in the days following interruption of PVP-I exposure on ICU day 21. CVVH: continuous venovenous hemofiltration; IHD: intermittent hemodialysis; PVP-I: povidone-iodine disinfection.
Thyroid hormone (intravenous thyroxin-L-sodium, initially started at 100 μg daily and reduced to 50 μg daily after 3 days) was substituted from day 25 until day 45; thereafter, the thyroid function recovered. The fasciotomy wounds on the left side were closed on ICU day 30. The fasciotomy on the right leg could not be approximated because of excessive of traction and healed spontaneously after one month. Finally, at day 54, the patient could be discharged from the intensive care unit. The patient was evaluated 4 months later in outpatient pediatric clinic and was doing very well; kidney function and renal ultrasound were reassuring.
3. Discussion
PVP-I is considered a very safe and effective topical antiseptic. However, cases of systemic toxicity have been reported before. As described by Pietsch and Meakins [6], disinfection of extensive thermal injuries with PVP-I has occasionally been associated with acidosis, thyroid dysfunction, and renal failure, due to absorption of the therapeutic agent [6, 7]. In this case, PVP-I absorption possibly has aggravated sepsis-induced AKI and acidosis. Iodine and povidone can both be systemically absorbed, and it is still uncertain whether iodine or rather the acidic complex povidone-iodine (pH = 2.43) causes the different toxic effects [6, 7]. Pietsch and Meakins [6] recommend against the topical use of PVP-I in thermal injuries greater than 20% body surface or in the presence of renal failure, as absorption of the agent may be enhanced in nonintact skin and greater wound areas [7]. In this report, we describe a case of systemic toxicity after disinfection of less extended wounds after fasciotomy. Toxicity manifestations were a combination of metabolic and electrolyte abnormalities, renal impairment, and thyroid dysfunction. Cases of thyroid dysfunction induced by PVP-I have been reported before [8]. This phenomenon may be induced by the Wolff-Chaikoff effect, as elevated iodine plasma levels temporally block the synthesis of thyroid hormones [9]. In particular, newborns and patients with underlying thyroid disorders seem to be more prone to this toxicity [8, 9].
Besides thyroid dysfunction, blood tests revealed metabolic and electrolyte abnormalities such as high anion gap metabolic acidosis, hyperchloremia, and elevated urinary anion gap. In most cases, it is difficult to reveal the cause of metabolic acidosis because of concurrent disorders [6, 10]. In this patient, metabolic acidosis could have been the result of septic shock, MODS, and AKI (Table 3). Typically, serum chloride levels could be falsely high, because of the interference by iodine, a “chloride look-alike” halogen, resulting in an overestimation of the actual chloride amount [10–12]. Iodine is renally excreted by glomerular filtration; consequently, systemic accumulation of iodine is seen more often in patients with renal impairment [13]. Filtered iodine molecules may have direct toxic effects on the tubular cells, as two patients with AKI and povidone-iodine intoxication had necrosis of tubular cells and interstitial nephritis on kidney biopsy [13, 14]. Although we did not perform a kidney biopsy in our patient, the favorable evolution of the renal function upon discontinuation of PVP-I application suggests that iodine toxicity was possibly a contributing factor to kidney injury and delayed recovery. In addition, the elevated urine anion gap often reflects a renal acidification defect [15], which may possibly result from renal toxicity of iodine.
Besides the evidence of direct toxic effect of iodine molecules, we should also consider indirect effects on renal function. Hypothyroidy can be associated with acute and chronic kidney disease, with improved renal function upon thyroid hormone supplementation [16–18]. The initiation of thyroid hormones maybe an additional contributing factor to enhanced recovery of kidney function in this case. Finally, we cannot exclude that the recovery of renal function shortly after interruption of PVP-I exposure, and initiation of thyroid hormone was a chance finding, given the uncontrolled nature of the data presented. Estimation of the true contribution of both PVP-I exposure and hypothyroidism to occurrence and persistence of AKI would require comparative studies such as randomized animal experiments.
The value of an elevated urinary anion gap, unexplained hypothyroidism, and hyperchloremia as efficacious parameters to screen for PVP-I toxicity needs to be evaluated in prospective studies. For the time being, we recommend to consider potential iodine intoxication in all critically ill patients treated for several days with PVP-I, particularly when AKI persists or when unexplained hypothyroidism occurs. After weighing the potential systemic/toxic impact of PVP-I disinfection against the extent of wound infection or bacterial colonization, the treating multidisciplinary team should consider substituting PVP-I disinfection with other antiseptics, such as chlorhexidine (with or without cetrimide) and/or substitution PVP-I topical ointment with, for example, silver-based ointments (such as silver sulfadiazine cream).
Patient perspective: prior to sending the informed consent form, we contacted the parents of the patient by phone, more than six months after ICU discharge. The child was doing very well and enjoyed going back to school, but strength in the lower part of the legs was still suboptimal as compared to the premorbid status.
4. Conclusion
The potential for toxic effects after PVP-I disinfection of moderate size wounds might be underestimated and thus go unrecognized. Timely recognition of iodine toxicity during the topical PVP-I exposure and awareness of its potentially severe complications may contribute to optimal management and a favorable outcome. More generally, this case report pleads for daily integration of wound evolution and wound management in the clinical evaluation of critically ill patients.
Acknowledgments
The authors thank the patient, the parents, and the entire PICU team who cared for this child during a prolonged ICU stay. MPC receives funding from the Research Foundation Flanders (FWO) (Grant No. 1832817N) and Onderzoeksraad, KU Leuven (Grant No. C24/17/070) and from the Private Charity Organization “Help Brandwonden Kids.” DD receives funding from the Clinical Research and Education Council (KOOR), University Hospitals Leuven.
Data Availability
The underlying data are part of the patient's medical file and have been consulted with permission of the patient's parents.
Ethical Approval
No case-by-case ethics approval is required for publication of case reports at UZ Leuven when operating within the institution ethical guidelines.
Consent
Written informed consent was obtained from the patient's parent for publication of this case report as per UZ Leuven Ethical Committee policy. A copy of the written consent is available for review by the editor in chief of this journal.
Conflicts of Interest
None of the authors have a competing interest regarding this publication.
Authors' Contributions
YV and MPC wrote the first draft of the paper; all authors reviewed the manuscript and approved the final version.
References
- 1.Sheila A. K. Effect of povidone-iodine on wound healing: a review. Journal of Vascular Nursing. 1999;17(1):17–23. doi: 10.1016/s1062-0303(99)90004-3. [DOI] [PubMed] [Google Scholar]
- 2.Burks R. I. Povidone-iodine solution in wound treatment. Physical Therapy. 1998;78(2):212–218. doi: 10.1093/ptj/78.2.212. [DOI] [PubMed] [Google Scholar]
- 3.Bigliardi P. L., Alsagoff S. A., el-Kafrawi H. Y., Pyon J.-K., Wa C. T., Villa M. A. Povidone iodine in wound healing: a review of current concepts and practices. International Journal of Surgery. 2017;44:260–268. doi: 10.1016/j.ijsu.2017.06.073. [DOI] [PubMed] [Google Scholar]
- 4.Scoggin C., McClellan J. R., Cary J. M. Hypernatriaemia and acidosis in association with topical treatment of burns. The Lancet. 1977;1(8081):p. 959. doi: 10.1016/s0140-6736(77)92263-2. [DOI] [PubMed] [Google Scholar]
- 5.Kanakiriya S., De Chazal I., Nath K. A., Haugen E. N., Albright R. C., Juncos L. A. Iodine toxicity treated with hemodialysis and continuous venovenous hemodiafiltration. American Journal of Kidney Disease. 2003;41(3):702–708. doi: 10.1053/ajkd.2003.50134. [DOI] [PubMed] [Google Scholar]
- 6.Pietsch J., Meakins J. L. Complications of povidone-iodine absorption in topically treated burn patients. The Lancet. 1976;307(7954):280–282. doi: 10.1016/S0140-6736(76)91406-9. [DOI] [PubMed] [Google Scholar]
- 7.Zamora J. L. Chemical and microbiologic characteristics and toxicity of povidone-iodine solutions. The American Journal of Surgery. 1986;151(3):400–406. doi: 10.1016/0002-9610(86)90477-0. [DOI] [PubMed] [Google Scholar]
- 8.Chabrolle J. P., Rossier A. Goitre and hypothyroidism in the newborn after cutaneous absorption of iodine. Archives of Disease in Childhood. 1978;53(6):495–498. doi: 10.1136/adc.53.6.495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wolff J., Chaikoff I. L., Goldberg R. C., Meier J. R. The temporary nature of the inhibitory action of excess iodine on organic iodine synthesis in the normal thyroid. Endocrinology. 1949;45(5):504–513. doi: 10.1210/endo-45-5-504. [DOI] [PubMed] [Google Scholar]
- 10.Dela Cruz F. D., Brown D. H., Leikin J. B., Franklin C., Hryhorczuk D. Iodine absorption after topical administration. The Western Journal of Medicine. 1987;146:43–45. [PMC free article] [PubMed] [Google Scholar]
- 11.Aronoff G. R., Friedman S. J., Doedens D. J., Lavelle K. J. _Case Report_ Increased serum iodide concentration from iodine absorption through wounds treated topically with povidone-iodine. The American Journal of Medical Sciences. 1980;279(3):173–176. doi: 10.1097/00000441-198005000-00007. [DOI] [PubMed] [Google Scholar]
- 12.Kenrick B. Review of the diagnostic evaluation of normal anion gap metabolic acidosis. Kidney Diseases. 2017;3:149–159. doi: 10.1159/000479279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Manfro R. C., Comerlato L., Berdichevski R. H., et al. Nephrotoxic acute renal failure in a renal transplant patient with recurrent lymphocele treated with povidone-iodine irrigation. American Journal of Kidney Diseases. 2002;40(3):655–657. doi: 10.1053/ajkd.2002.34930. [DOI] [PubMed] [Google Scholar]
- 14.Kim C. S., Kim S. S., Bae E. H., Ma S. K., Kim S. W. Acute kidney injury due to povidone-iodine ingestion: a case report. Medicine. 2017;96(48, article e8879) doi: 10.1097/MD.0000000000008879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Batlle D., Ba Aqeel S. H., Marquez A. The urine anion gap in context. Clinical Journal of the American Society of Nephrology. 2018;13(2):195–197. doi: 10.2215/CJN.13791217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chang Y. C., Chang C., Yeh Y. C., Chuang L. M., Tu Y. K. Subclinical and overt hypothyroidism is associated with reduced glomerular filtration rate and proteinuria: a large cross-sectional population study. Scientific Reports. 2018;8(1):p. 2031. doi: 10.1038/s41598-018-19693-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Connor A., Taylor J. E. Renal impairment resulting from hypothyroidism. NDT Plus. 2008;1(6):440–441. doi: 10.1093/ndtplus/sfn158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shin D. H., Lee M. J., Kim S. J., et al. Preservation of renal function by thyroid hormone replacement therapy in chronic kidney disease patients with subclinical hypothyroidism. Journal of Clinical Endocrinology and Metabolism. 2012;97(8):2732–2740. doi: 10.1210/jc.2012-1663. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The underlying data are part of the patient's medical file and have been consulted with permission of the patient's parents.


