Abstract
Background: Appropriate empiric antibiotic therapy for ventilator-associated pneumonia improves outcomes. Controversy exists regarding the inclusion of aminoglycosides in empiric therapeutic regimens.
Methods: The Ovid and Cochrane databases were searched for relevant literature regarding the use of aminoglycosides in combination therapy for ventilator-associated pneumonia. The data supporting the use of aminoglycosides in certain populations and strategies to limit toxicity are summarized.
Results: In patients at high risk of infection with antibiotic-resistant gram-negative bacilli and in those with severe illness, aminoglycosides improve clinical outcomes. In critically ill populations, short-duration therapy and high-dose extended-interval dosing of aminoglycosides can improve therapeutic efficacy while limiting nephrotoxicity.
Conclusions: In selected populations using appropriate dosing strategies, aminoglycosides should be considered for empiric treatment of ventilator-associated pneumonia.
Aminoglycosides as an antibiotic class maintain broad-spectrum activity against both gram-negative bacilli and gram-positive cocci, with the rate of resistance to this class remaining relatively stable despite decades of use [1]. Their mechanism of action is believed to be interruption of protein synthesis at the ribosomal level, demonstrating both in vitro synergism with β-lactam antibiotics for many gram-negative bacilli and gram-positive cocci and substantial post-antibiotic effect [1,2]. Thus, two separate rationales may support the use of aminoglycosides for empiric therapy: (1) Improvement of the coverage of resistant gram-negative bacilli and (2) obtaining better outcomes as a result of therapeutic synergy with other agents. The routine use of aminoglycosides commonly is limited by their potential to induce or potentiate nephrotoxicity. However, when dosed appropriately and de-escalated when culture and sensitivity data show they are not appropriate, nephrotoxicity should be limited.
An appropriate risk/benefit analysis for the use of empiric aminoglycosides requires knowledge of the relatively complex data regarding appropriate empiric antibiotic therapy (i.e., pathogens sensitive to empiric regimen), the likelihood of resistant pathogens, mortality risk stratification, aminoglycoside dosing strategies, and the risk of nephrotoxicity. An argument for the use of aminoglycoside agents in combination therapy for the empiric therapy for ventilator-associated pneumonia (VAP) in selected patients is presented here, as well as a strategy to limit nephrotoxicity.
Increasing the Adequacy of Empiric Coverage of Resistant GNB
In certain patient populations, aminoglycosides may improve substantially the likelihood of appropriate empiric coverage of VAP caused by gram-negative bacilli. Observational data demonstrate the importance of correctly treating causative pathogens empirically in VAP, with inadequate treatment significantly increasing the risk of death [1,3–7]. In hospital-acquired pneumonia, resistance among gram-negative pathogens may limit the ability to cover all likely pathogens with a single agent or class [1,7–9]. Thus, combination therapy, including aminoglycosides, may increase the likelihood of adequate empiric therapy and improve outcomes.
The importance of adequate empiric coverage of pathogens in those patients suspected of having VAP was suggested in the late 1980s by Celis et al. [10] and was established in the mid-1990s through a series of observational studies by various authors [3–6]. Four studies demonstrated roughly a doubling of both crude and attributable death if empiric therapy did not provide adequate coverage of pathogens [3,5,6,10]. These findings were supported by a subsequent prospective observational study by Kollef et al. in 1999 [4]. Those authors followed all critically ill patients admitted to either of two surgical intensive care units (ICUs). Six hundred fifty-five patients had infections (either community- or hospital-acquired), and 26% received inadequate empiric therapy. Those latter patients had a greater than a two-fold increase in the risk of death. Of the gram-negative pathogens isolated in this study, a quarter were antibiotic resistant.
The importance of adequate empiric coverage for serious infections, including VAP, continues to be confirmed in recent observational studies [7,11–13]. Table 1 outlines 17 studies examining the outcome of VAP with and without appropriate antibiotic therapy. All studies demonstrate greater numbers of deaths with inadequate empiric coverage, the differences in 13 of these studies reaching statistical significance [3–7,10,12–22]. A delay in appropriate antibiotic therapy is associated with more deaths even if antibiotics are adjusted when sensitivity data are returned after 48 to 72 h [5,19,23]. Multivariable analysis of independent risk factors for the 30-day mortality rate in patients with gram-negative VAP demonstrates that inappropriate initial antibiotic therapy is strongly and significantly associated with the risk of death (adjusted odds ratio [OR] 11.7) [12].
Table 1.
Influence of Adequate Initial Antibiotic Therapy on Mortality Rate in Ventilator-Associated Pneumonia
| Series | Year | No. of patients | Adequate treatment death/pts (%) | Inadequate treatment death/pts (%) | p |
|---|---|---|---|---|---|
| Celis et al. [10] | 1988 | 118 | 33/118 (28) | 11/12 (92) | <0.001 |
| Alvarez-Lerma [3] | 1996 | 430 | 46/284 (16) | 36/146 (25) | 0.03 |
| Luna et al. [5] | 1997 | 50 | 6/16 (38) | 31/34 (91) | <0.01 |
| Rello et al. [6] | 1997 | 113 | 13/86 (15) | 10/27 (37) | <0.05 |
| Sanchez-Nieto et al. [22] | 1998 | 38 | 6/24 (25) | 6/14 (43) | NS |
| Kollef et al. [4] | 1999 | 411 | 48/106 (45) | 51/305 (17) | <0.01 |
| Bercault & Boulain [14] | 2001 | 135 | 37/92 (40) | 18/34 (53) | NS |
| Dupont et al. [16] | 2001 | 111 | 21/55 (38) | 27/57 (47) | NS |
| Iregui et al. [19] | 2002 | 107 | 21/74 (28) | 23/33 (70) | <0.01 |
| Hanes et al. [18] | 2002 | 163 | 9/79 (11) | 21/84 (25) | 0.02 |
| Dupont et al. [17] | 2003 | 322 | 37/220 (17) | 21/92 (23) | NS |
| Leroy et al. [20] | 2003 | 132 | 42/106 (40) | 16/26 (62) | 0.04 |
| Clec'h C et al. [15] | 2004 | 70 | 2/27 (7) | 16/43 (37) | <0.01 |
| Mueller et al. [21] | 2005 | 82 | 1/28 (4) | 12/54 (22) | <0.01 |
| Kollef et al. [12] | 2008 | 76 | 8/59 (14) | 11/17 (65) | <0.01 |
| Rello et al. [11] | 2011 | 484 | (35) | (48) | 0.01 |
| Tseng et al. [13] | 2012 | 131 | 20/71 (28) | 37/60 (62) | 0.0225 |
NS = non-significant.
The presence of multi-drug-resistant (MDR) pathogens greatly increases the risk of inadequate empiric antibiotic therapy. As noted above in the study by Kollef et al., antibiotic resistance is common, with a quarter of the isolated gram-negative pathogens being antibiotic resistant [4]. Multi-drug-resistant gram-negative pathogens within ICUs are a serious concern. In a prospective observational study of nosocomial infections in 24 ICUs in Belgium from 2006–2007, 49.6% of the gram-negative pathogens were MDR, and in 36% of all nosocomial infections, antibiotic therapy was classified as inadequate [24]. Prior antibiotic exposure within the last 90 d or during the hospital stay appears to be among the strongest risk factors for the presence of MDR bacteria [24–26]. Other risk factors include exposure to healthcare environments in the previous 90 d, current hospital length of stay greater than five d, and chronic illness or immunosuppression.
Unless highly effective antibiotic stewardship programs are in place, and efforts to reduce resistant pathogens have been highly successful within the local environment, the risk of MDR pathogens as a cause of hospital-acquired infection remains significant. Thus, to improve the adequacy of empiric antibiotic treatment for patients at risk of MDR pathogens, existing guidelines recommend combination therapy [25]. The use of local data to establish guidelines can increase compliance with adequate empiric therapy for VAP by targeting therapy to the most common pathogens historically isolated within an institution or specific ICU and their antibiotic sensitivities [8,27]. In a before and after study of implementing such a locally developed guideline, Ibrahim et al. demonstrated an increase in compliance with appropriate empiric therapy to greater than 90% coverage with the combination of a β-lactam and fluoroquinolone [27]. In contrast, in a similar study by Beardsley et al., the combination of a β-lactam and fluoroquinolone achieved appropriate empiric coverage in only 70% of episodes of late-onset pneumonia, whereas a combination including an aminoglycoside achieved appropriate coverage in 93% of cases [8].
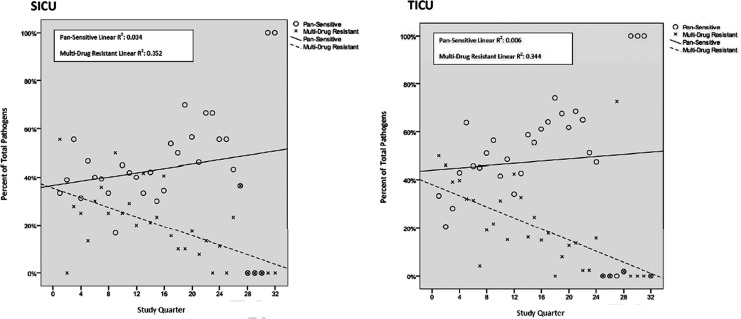
At the author's institution, empiric antibiotic therapy for VAP using combination therapy with aminoglycosides is based on risk stratification utilizing local unit-specific data to ensure adequate coverage, a system employed for more than 10 years [28]. Additionally, aggressive antibiotic stewardship and infection-reduction programs have been employed during the same period. Stewardship protocols include limiting antibiotic prophylaxis to evidence-supported indications with the narrowest appropriate coverage for 24 h or less, quantitative cultures for the diagnosis of VAP, and de-escalation of therapy based on culture results. In 2005, a program to maintain antibiotic heterogeneity while eliminating an antibiotic class on a rotating quarterly schedule was instituted. Antibiotic resistance to one or multiple classes of drugs among local gram-negative pathogens has declined significantly over time in the trauma and surgical ICUs (Fig. 1). During the 8-y period from 2001 to 2008, these ICUs have had a 77% relative reduction in MDR gram-negative pathogens and a 56% relative increase in the percentage of pansensitive pathogens [28].
FIG. 1.
Percentage of gram-negative pathogens that were either pansensitive or multi-drug resistant by quarter after initiation of an antibiotic stewardship program in 2001. Trend lines demonstrate significant increase in percentage of pathogens that were pansensitive and decrease in those that were multi-drug resistant in both the trauma (TICU) and the surgical (SICU) intensive care units. (Reprinted with permission from Dortch et al. [28]).
Despite these positive effects of antibiotic stewardship initiatives, combination empiric coverage of potential gram-negative pathogens, including aminoglycosides, have still proved to be beneficial. Analysis of our empiric combination therapy for VAP over a 5-y period demonstrated that aminoglycoside use was required to achieve adequate coverage in 20% of cases. Adequate empiric therapy was achieved for 94% and 88% of pathogens, respectively, for the trauma and surgical ICUs. Additionally, no pathogens resistant to aminoglycosides were sensitive to fluoroquinolones during this time. However, the majority of pathogens resistant to fluoroquinolones were sensitive to aminoglycosides (63%). Thus, the addition of aminoglycosides to our empiric regimen has improved the frequency of adequate empiric coverage substantially.
Improved Outcomes Secondary to Therapeutic Synergy
As noted above, the combination of an aminoglycoside with a β-lactam antibiotic demonstrates a synergistic bacteriocidal effect for both gram-negative bacilli and gram-positive cocci in vitro [1]. However, demonstrating a therapeutic benefit from this combination therapy in the clinical setting has been much more complex, perhaps because of an offsetting detrimental effect of aminoglycoside nephrotoxicity. On the basis of an analysis of both observational and interventional studies, the combination of an aminoglycoside and a β-lactam appears to offer therapeutic benefit in more severely ill populations while potentially being detrimental in those who are less ill [1,11,29,30].
Aminoglycosides have been studied extensively in both pneumonia and sepsis and several meta-analyses published. Most studies published prior to 2010 have not demonstrated a clear benefit to combination therapy for pneumonia or sepsis as long as empiric therapy appropriately covered the pathogen [26,31–34]. However, most of these studies either did not include the most severely ill patient population or did not perform analyses based on severity of illness stratification [35]. In a prospective study of 200 consecutive cases of P. aeruginosa bacteremia, combination therapy resulted in a significantly lower mortality than monotherapy (27% vs 47%; p < 0.02) [36]. These findings were confirmed in a meta-analysis of combination therapy for gram-negative bacilli bacteremia by Safdar et al., demonstrating a clear benefit to the use of combination therapy in patients with P. aeruginosa bacteremia with roughly a 50% reduction in the number of deaths [33]. Combination therapy improves outcomes in patients with severe community-acquired pneumonia (i.e., those with septic shock or requiring mechanical ventilation) even when the β-lactam therapy appropriately covers the pathogen [35–37]. Therapeutic benefit for combination therapy with an aminoglycoside has been demonstrated in patients with bacteremia who present in septic shock or who are neutropenic [11]. In a meta-analysis of studies of combination therapy versus monotherapy for the treatment of serious infections associated with sepsis and septic shock, Kumar et al demonstrated that combination therapy improves survival and clinical response in high-risk, life-threatening infections but may be detrimental in those patients who are at low risk [30].
Dosing Strategies and Nephrotoxicity
The main concern about using aminoglycosides in empiric therapy for pneumonia is the fear of nephrotoxicity. However, if aminoglycosides are dosed appropriately and de-escalated as indicated when sensitivity data become available, nephrotoxicity should rarely, if ever, occur. Nephrotoxicity from aminoglycosides is correlated most strongly with trough levels and total time of exposure [38]. Extended dosing regimens should be employed for aminoglycosides, maximizing concentration-dependent killing and limiting toxicity by allowing troughs to fall below 0.5 mcg/mL [1]. The majority of the therapeutic effect from combination therapy is believed to occur early in the course of treatment, and appropriate de-escalation should limit aminoglycoside exposure to 3 d or less. Meta-analysis of extended versus routine dosing regimens clearly demonstrated reduced nephrotoxicity of extended regimens, with an overall rate of acute kidney injury of 5.5% for patients receiving full therapeutic courses of aminoglycosides [39]. However, in one large observational study of more than 2,000 patients with a median therapeutic course of 3 d of aminoglycoside, the overall rate of a creatinine concentration ≥0.5 mg/dL above the baseline was 1.2% [40]. Thus, the overall rate of severe renal dysfunction as a result of empiric therapy should be well under 1%. At our institution, all cases of renal failure developing in our surgical ICUs are reviewed as part of our quality assurance and process improvement programs. No cases of acute renal failure have been attributed to empiric exposure to aminoglycosides during the period of the program.
In summary, the addition of an aminoglycoside to β-lactam therapy for empiric coverage of pathogens in patients suspected of having VAP should be considered when the risk of gram-negative bacilli resistance is significant or when patients are more severely ill, particularly in the presence of septic shock. The need to cover the involved pathogens empirically has substantial support on the literature. Even in settings with aggressive antibiotic stewardship and infection reduction programs, aminoglycosides increase the percentage of gram-negative pathogens that are adequately covered empirically. If quantitative techniques for the diagnosis of pneumonia, extended dosing of aminoglycosides, short therapeutic courses, and appropriate de-escalation are all employed, nephrotoxicity should be eliminated. Extending the use of aminoglycosides to double cover pathogens for longer therapeutic courses is not supported in the literature.
Author Disclosure Statement
The author has no conflicts of interest in relation to this manuscript.
References
- 1.Boyer A, Gruson D, Bouchet S, et al. Aminoglycosides in septic shock: An overview, with specific consideration given to their nephrotoxic risk. Drug Saf 2013;36:217–230 [DOI] [PubMed] [Google Scholar]
- 2.Davis BD. Mechanism of bactericidal action of aminoglycosides. Microbiol Rev 1987;51:341–350 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Alvarez-Lerma F. Modification of empiric antibiotic treatment in patients with pneumonia acquired in the intensive care unit. ICU-Acquired Pneumonia Study Group. Intensive Care Med 1996;22:387–394 [DOI] [PubMed] [Google Scholar]
- 4.Kollef MH, Sherman G, Ward S, Fraser VJ. Inadequate antimicrobial treatment of infections: A risk factor for hospital mortality among critically ill patients. Chest 1999;115:462–474 [DOI] [PubMed] [Google Scholar]
- 5.Luna CM, Vujacich P, Niederman MS, et al. Impact of BAL data on the therapy and outcome of ventilator-associated pneumonia. Chest 1997;111:676–685 [DOI] [PubMed] [Google Scholar]
- 6.Rello J, Gallego M, Mariscal D, et al. The value of routine microbial investigation in ventilator-associated pneumonia. Am J Respir Crit Care Med 1997;156:196–200 [DOI] [PubMed] [Google Scholar]
- 7.Rello J, Ulldemolins M, Lisboa T, Koulenti D, et al. Determinants of prescription and choice of empirical therapy for hospital-acquired and ventilator-associated pneumonia. Eur Respir J 2011;37:1332–1339 [DOI] [PubMed] [Google Scholar]
- 8.Beardsley JR, Williamson JC, Johnson JW, et al. Using local microbiologic data to develop institution-specific guidelines for the treatment of hospital-acquired pneumonia. Chest 2006;130:787–793 [DOI] [PubMed] [Google Scholar]
- 9.Bhat S, Fujitani S, Potoski BA, et al. Pseudomonas aeruginosa infections in the Intensive care unit: Can the adequacy of empirical beta-lactam antibiotic therapy be improved? Int J Antimicrob Agents 2007;30:458–462 [DOI] [PubMed] [Google Scholar]
- 10.Celis R, Torres A, Gatell JM, et al. Nosocomial pneumonia: A multivariate analysis of risk and prognosis. Chest 1988;93:318–324 [DOI] [PubMed] [Google Scholar]
- 11.Martinez JA, Cobos-Trigueros N, Soriano A, et al. Influence of empiric therapy with a beta-lactam alone or combined with an aminoglycoside on prognosis of bacteremia due to gram-negative microorganisms. Antimicrob Agents Chemother 2010;54:3590–3596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kollef KE, Schramm GE, Wills AR, et al. Predictors of 30-day mortality and hospital costs in patients with ventilator-associated pneumonia attributed to potentially antibiotic-resistant gram-negative bacteria. Chest 2008;134:281–287 [DOI] [PubMed] [Google Scholar]
- 13.Tseng CC, Liu SF, Wang CC, et al. Impact of clinical severity index, infective pathogens, and initial empiric antibiotic use on hospital mortality in patients with ventilator-associated pneumonia. Am J Infect Control 2012;40:648–652 [DOI] [PubMed] [Google Scholar]
- 14.Bercault N, Boulain T. Mortality rate attributable to ventilator-associated nosocomial pneumonia in an adult intensive care unit: A prospective case-control study. Crit Care Med 2001;29:2303–2309 [DOI] [PubMed] [Google Scholar]
- 15.Clec′h C, Timsit JF, De LA, et al. Efficacy of adequate early antibiotic therapy in ventilator-associated pneumonia: Influence of disease severity. Intensive Care Med 2004;30:1327–1333 [DOI] [PubMed] [Google Scholar]
- 16.Dupont H, Mentec H, Sollet JP, Bleichner G. Impact of appropriateness of initial antibiotic therapy on the outcome of ventilator-associated pneumonia. Intensive Care Med 2001;27:355–362 [DOI] [PubMed] [Google Scholar]
- 17.Dupont H, Montravers P, Gauzit R, et al. Outcome of postoperative pneumonia in the Eole study. Intensive Care Med 2003;29:179–188 [DOI] [PubMed] [Google Scholar]
- 18.Hanes SD, Demirkan K, Tolley E, et al. Risk factors for late-onset nosocomial pneumonia caused by Stenotrophomonas maltophilia in critically ill trauma patients. Clin Infect Dis 2002;35:228–235 [DOI] [PubMed] [Google Scholar]
- 19.Iregui M, Ward S, Sherman G, et al. Clinical importance of delays in the initiation of appropriate antibiotic treatment for ventilator-associated pneumonia. Chest 2002;122:262–268 [DOI] [PubMed] [Google Scholar]
- 20.Leroy O, Meybeck A, d′Escrivan T, et al. Impact of adequacy of initial antimicrobial therapy on the prognosis of patients with ventilator-associated pneumonia. Intensive Care Med 2003;29:2170–2173 [DOI] [PubMed] [Google Scholar]
- 21.Mueller EW, Hanes SD, Croce MA, et al. Effect from multiple episodes of inadequate empiric antibiotic therapy for ventilator-associated pneumonia on morbidity and mortality among critically ill trauma patients. J Trauma 2005;58:94–101 [DOI] [PubMed] [Google Scholar]
- 22.Sanchez-Nieto JM, Torres A, Garcia-Cordoba F, et al. Impact of invasive and noninvasive quantitative culture sampling on outcome of ventilator-associated pneumonia: A pilot study. Am J Respir Crit Care Med 1998;157:371–376 [DOI] [PubMed] [Google Scholar]
- 23.Luna CM, Blanzaco D, Niederman MS, et al. Resolution of ventilator-associated pneumonia: Prospective evaluation of the clinical pulmonary infection score as an early clinical predictor of outcome. Crit Care Med 2003;31:676–682 [DOI] [PubMed] [Google Scholar]
- 24.Vogelaers D, De BD, Foret F, et al. Patterns of antimicrobial therapy in severe nosocomial infections: Empiric choices, proportion of appropriate therapy, and adaptation rates—A multicentre, observational survey in critically ill patients. Int J Antimicrob Agents 2010;35:375–381 [DOI] [PubMed] [Google Scholar]
- 25.Guidelines for the management of adults with hospital-acquired, ventilator-associated, and healthcare-associated pneumonia. Am J Respir Crit Care Med 2005;171:388–416 [DOI] [PubMed] [Google Scholar]
- 26.Eggimann P, Revelly JP. Should antibiotic combinations be used to treat ventilator-associated pneumonia? Semin Respir Crit Care Med 2006;27:68–81 [DOI] [PubMed] [Google Scholar]
- 27.Ibrahim EH, Ward S, Sherman G, et al. Experience with a clinical guideline for the treatment of ventilator-associated pneumonia. Crit Care Med 2001;29:1109–1115 [DOI] [PubMed] [Google Scholar]
- 28.Dortch MJ, Fleming SB, Kauffmann RM, et al. Infection reduction strategies including antibiotic stewardship protocols in surgical and trauma intensive care units are associated with reduced resistant gram-negative healthcare-associated infections. Surg Infect 2011;12:15–25 [DOI] [PubMed] [Google Scholar]
- 29.Kumar A, Zarychanski R, Light B, et al. Early combination antibiotic therapy yields improved survival compared with monotherapy in septic shock: A propensity-matched analysis. Crit Care Med 2010;38:1773–1785 [DOI] [PubMed] [Google Scholar]
- 30.Kumar A, Safdar N, Kethireddy S, Chateau D. A survival benefit of combination antibiotic therapy for serious infections associated with sepsis and septic shock is contingent only on the risk of death: A meta-analytic/meta-regression study. Crit Care Med 2010;38:1651–1664 [DOI] [PubMed] [Google Scholar]
- 31.Garnacho-Montero J, Sa-Borges M, Sole-Violan J, et al. Optimal management therapy for Pseudomonas aeruginosa ventilator-associated pneumonia: An observational, multicenter study comparing monotherapy with combination antibiotic therapy. Crit Care Med 2007;35:1888–1895 [DOI] [PubMed] [Google Scholar]
- 32.Paul M, Silbiger I, Grozinsky S, et al. Beta lactam antibiotic monotherapy versus beta lactam-aminoglycoside antibiotic combination therapy for sepsis. Cochrane Database Syst Rev 2006;(1):CD003344. [DOI] [PubMed] [Google Scholar]
- 33.Safdar N, Handelsman J, Maki DG. Does combination antimicrobial therapy reduce mortality in gram-negative bacteraemia? A meta-analysis. Lancet Infect Dis 2004;4:519–527 [DOI] [PubMed] [Google Scholar]
- 34.Aarts MA, Hancock JN, Heyland D, et al. Empiric antibiotic therapy for suspected ventilator-associated pneumonia: A systematic review and meta-analysis of randomized trials. Crit Care Med 2008;36:108–117 [DOI] [PubMed] [Google Scholar]
- 35.Salva S, Borgatta B, Rello J. Pneumonia in immunocompetent patients: Combination antibiotic therapy. Minerva Anestesiol 2014;80:495–503 [PubMed] [Google Scholar]
- 36.Hilf M, Yu VL, Sharp J, Zuravleff JJ, et al. Antibiotic therapy for Pseudomonas aeruginosa bacteremia: Outcome correlations in a prospective study of 200 patients. Am J Med 1989;87:540–546 [DOI] [PubMed] [Google Scholar]
- 37.Pereira JM, Paiva JA, Rello J. Severe sepsis in community-acquired pneumonia—Early recognition and treatment. Eur J Intern Med 2012;23:412–419 [DOI] [PubMed] [Google Scholar]
- 38.Mueller EW, Boucher BA. The use of extended-interval aminoglycoside dosing strategies for the treatment of moderate-to-severe infections encountered in critically ill surgical patients. Surg Infect 2009;10:563–570 [DOI] [PubMed] [Google Scholar]
- 39.Barza M, Ioannidis JP, Cappelleri JC, Lau J. Single or multiple daily doses of aminoglycosides: A meta-analysis. BMJ 1996;312:338–345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nicolau DP, Freeman CD, Belliveau PP, et al. Experience with a once-daily aminoglycoside program administered to 2,184 adult patients. Antimicrob Agents Chemother 1995;39:650–655 [DOI] [PMC free article] [PubMed] [Google Scholar]