Abstract
Differences in pancreatic anatomy, size, and function exist in men and women. The anatomical differences could contribute to the increase in complications associated with pancreatic surgery in women. Although diagnostic criteria for pancreatitis are the same in men and women, major sex differences in etiology are reported. Alcohol and tobacco predominate in men, whereas idiopathic and obstructive etiologies predominate in women. Circulating levels of estrogens, progesterone, and androgens contribute significantly to overall health outcomes; premenopausal women have lower prevalence of cardiovascular and pancreatic diseases suggesting protective effects of estrogens, whereas androgens promote growth of normal and cancerous cells. Sex chromosomes and gonadal and nongonadal hormones together determine an individual’s sex, which is distinct from gender or gender identity. Human pancreatic disease etiology, outcomes, and sex-specific mechanisms are largely unknown. In rodents of both sexes, glucocorticoids and estrogens from the adrenal glands influence pancreatic secretion and acinar cell zymogen granule numbers. Lack of corticotropin-releasing factor receptor 2 function, a G protein–coupled receptor whose expression is regulated by both estrogens and glucocorticoids, causes sex-specific changes in pancreatic histopathology, zymogen granule numbers, and endoplasmic reticulum ultrastructure changes in acute pancreatitis model. Here, we review existing literature on sex differences in the normal exocrine pancreas and mechanisms that operate at homeostasis and diseased states in both sexes. Finally, we review pregnancy-related pancreatic diseases and discuss the effects of sex differences on proposed treatments in pancreatic disease.
Keywords: Adrenal Steroids, Corticotropin-Releasing Factor Receptor 2, Estradiol, Exocrine Pancreas, Sex Differences, Sex Hormones
Abbreviations used in this paper: AP, acute pancreatitis; COVID-19, coronavirus disease 2019; CP, chronic pancreatitis; CRF, corticotropin-releasing factor; ER, estrogen receptor; GI, gastrointestinal; GnRH, gonadotropin-releasing hormone; GPER, G protein estrogen receptor; HPA, hypothalamic-pituitary-adrenal; HPG, hypothalamic-pituitary-gonadal; IPMN, intraductal papillary mucinous neoplasm; PDAC, pancreatic ductal adenocarcinoma; PR, progesterone receptor; RER, rough endoplasmic reticulum; ROS, reactive oxygen species; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; UCN1, urocortin 1
Summary.
Here, we review existing literature on sex differences in normal and diseased exocrine pancreas. We discuss known factors that influences sex-biased (patho)physiological processes and general principles to consider while addressing sex differences in the pancreas.
There is growing recognition that sex-biased factors can modify both physiologic and disease responses involving the gastrointestinal tract. As a reflection of the importance of this concept, the National Institutes of Health announced a policy to include “sex as a biological variable” that requires investigators to explicitly consider the impact of sex in research design and data interpretation. Sex chromosome or autosome genes, sex hormones, and the environment together determine an individual’s sex. This is distinct from gender or gender identity, which is an individual’s perception of self and by society as male, female, or both. Thus, only humans have gender, as we recognize it, whereas both humans and animals have sex.
Although sex-dependent differences in pancreatic disease etiology and outcomes are recognized, the sex-specific mechanisms that affect these responses are largely unknown. Most mechanistic studies focused on the effects of estrogen and glucocorticoid metabolites on acinar cell function, whereas data on the roles of androgens and progesterone and their respective metabolites is nonexistent. Side-by-side comparisons of mechanistic studies or disease outcomes in animal models inclusive of both sexes are rarely performed. Similarly, few clinical studies that have considered sex as a variable in analysis of pancreatic disease outcomes, as highlighted in this review. This emphasizes the need for a more comprehensive and deliberate inclusion of sex as a variable in clinical diagnosis, study designs, and data analyses of pancreatic diseases. Sex-dependent differences are first reviewed in the context of the normal exocrine pancreas. Next, we delineate proposed mechanisms driving sex differences in exocrine pancreas disease states, focusing on hormonal signaling pathways. Finally, we summarize the reported effects of sex steroid hormones and their targets on biologic responses and (patho)physiological outcomes.
Sex Hormonal Axis and Distribution
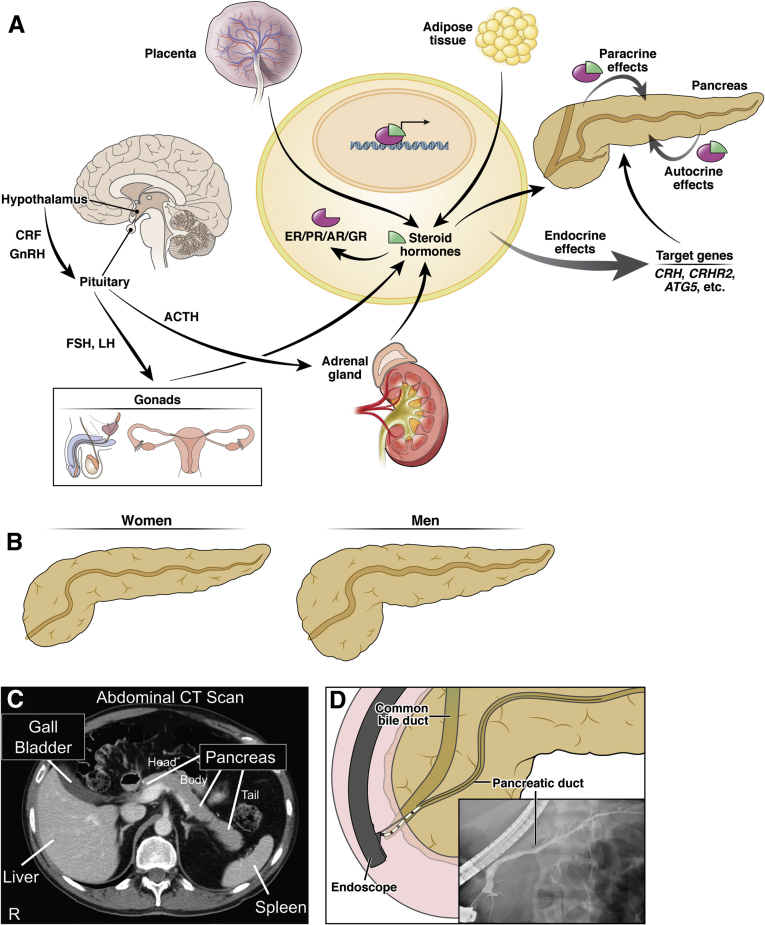
Most studies addressing sex differences in (patho)physiological outcomes in the pancreas have focused on the role of circulating levels of sex steroid hormones, mainly estradiol and corticosterone; thus, the data on understanding the role of different sex steroids on pancreatic acinar cells are limited. The perception of estrogens/progesterone as female and testosterone as male hormones is misleading. It is important to remember that both sexes and all genders have all classes of steroid hormones including estrogens, progesterone, and androgens, albeit at different levels that vary depending on the age in both humans and rodents.1,2 Sex hormone synthesis and secretion is regulated by the hypothalamic-pituitary-gonadal (HPG) axis in which gonadotropin-releasing hormone secreted from the hypothalamus into the portal circulation reaches the anterior pituitary, ultimately leading to the secretion of follicle-stimulating hormone and luteinizing hormone. Pulsatile release of these hormones supports continuous steroidogenesis and sperm production in males3 and ova development, ovulation, and stimulation of estradiol and progesterone from the ovaries in females.1 Gonads, adrenals, brain, and the placenta are the primary steroidogenic tissues that express P450scc (CYP11A1) to initiate steroidogenesis by converting cholesterol to pregnenolone, the precursor for all steroid hormones.4,5 In addition, many nonsteroidogenic tissues (liver, kidney, adipose fat, breast, heart) express other steroidogenic enzymes that modify steroid precursors taken up from the circulation.6, 7, 8 Adipose tissue, particularly in postmenopausal women and older men, can contribute significantly to the systemic bioactive circulation of estradiol.9 Circulating sex steroid hormones (endocrine actions) or those secreted locally (paracrine or autocrine actions) bind their cognate receptors—estrogen receptors (ERα, Erβ) and G protein–coupled estrogen receptor 1 (GPER [also known as GPR30]), progesterone receptor (PR), and androgen receptors. With the exception of GPER, each is in the nuclear receptor family and these hormone-receptor complexes bind to specific promoter regions to regulate a range of transcriptional responses (Figure 1A).10
Figure 1.
Sex-specific differences in pancreatic function, morphology, and anatomy. (A) The gonads, adrenal gland, brain, and the placenta are primary steroidogenic tissues in humans capable of synthesizing all steroid hormones from cholesterol. Peptide hormones such as gonadotropin-releasing hormone (GnRH), FSH, and LH also modulate release of sex steroid hormones from the gonads, whereas CRF/ACTH actions result in synthesis and release of glucocorticoids from the adrenal glands. Adipose tissues are also known to perform steroidogenesis and synthesize estrogens; pancreas and pancreatic fat may also be capable of synthesizing and releasing estrogens, androgens, and progesterone. These steroid hormones bind to their cognate receptors and translocate to the nucleus, where the hormone-bound receptor complex binds to their respective hormone-response elements on the DNA to influence transcription of several target genes. The endocrine, paracrine, and autocrine actions of these hormones activate or repress several autosomal genes, such as CRH, CRHR2, ATG5, and other unknown ones (represented with etc.), which then modulate sex-specific signaling or pathways in the pancreas at baseline and in diseased state. (B) Schematic representation of human pancreatic size and volume with women on an average displaying less volume and a smaller pancreatic duct than men. (C) Radiograph imaging of the human abdomen showing pancreatic head, body, and tail in relation to other organs. (D) Radiocontrast imaging depicting cannulation of the pancreatic duct during an endoscopic retrograde cholangiopancreatography procedure. These anatomical differences in the pancreas may explain risk factors for different pancreatic diseases, including an increased risk of postendoscopic retrograde cholangiopancreatography pancreatitis in women compared with men. FSH, follicle-stimulating hormone; LH, luteinizing hormone; R, right side of patient.
The human and rodent pancreata are highly innervated and along with hormones (endocrine and gut), neural inputs regulate secretion from the exocrine pancreas. Included are peptide hormones, such as gonadotropins and their associated cognate hormone receptors, which differ in localization throughout the body. For example, gonadotropin-releasing hormone is found in hypothalamic and peripheral tissues, such as the placenta, gonads, mammary glands, and the pancreas.11 Normal human pancreatic tissue expresses ERα, ERβ, GPER, PR, and androgen receptors.10 Several other peptide hormone/receptor systems are expressed in the mammalian pancreas, but the precise role and function of many of these systems remains unclear.
The HPG axis and sex steroid hormones also interact with the components of the stress hypothalamic-pituitary-adrenal (HPA) axis, the mediator of stress responses, which can impact gastrointestinal disease processes.12 For example, although best known for their role in mediating “flight or fight” stress responses through the HPA axis, peptide hormones of the corticotropin-releasing-factor (CRF) system, including CRF, 3 urocortins (UCN1–3), and their 2 G protein–coupled receptors, CRF type 1 (CRF1) and type 2 (CRF2), are expressed in both the exocrine and the endocrine pancreas, where they regulate acinar cell secretion, organelle stress, and glucose homeostasis.13, 14, 15, 16, 17, 18 CRF, UCN1-3, and CRF receptors, are widely distributed, including in the gastrointestinal (GI) system,19, 20, 21, 22, 23 although CRF1 is predominantly found in the brain and CRF2 in the peripheral tissues. On the one hand, CRF and UCN1 bind to CRF1 but only hypothalamic CRF, not UCN1, stimulates ACTH synthesis and secretion from the anterior pituitary, which in turn stimulates the synthesis and release of glucocorticoids from the adrenal glands (cortisol in humans and corticosteroid in rodents). On the other hand, binding of UCN1-3 to CRF2, facilitates negative feedback to the HPA axis and recovery from the stress responses.24 The expression and function of CRF family members at baseline and in various disease states in both sexes is reviewed elsewhere.14 No study has evaluated the levels of sex steroid hormones in the mammalian pancreas.
Sex differences in Anatomy and Function of Normal Pancreas
Human Studies
The human pancreas is compact retroperitoneal organ and is located juxtaposed to greater curvature of the stomach and proximal duodenum. The exocrine pancreas secretes a bicarbonate-rich fluid and digestive enzymes in response to a meal; the major role of the endocrine pancreas is insulin-mediated glucose homeostasis. Differences in pancreatic anatomy, size, and function exist in men and women; from birth until <90 years of age, women consistently have average smaller pancreas volume (∼15%–25%) than age- and body mass index–matched men.25 Women have a smaller pancreatic head and total volume than men but no difference in body or tail pancreatic volume (Figure 1B and C).26 Smaller pancreatic volumes, with proportional reductions in the size of other anatomical structures such as duct diameter (Figure 1B and D), could contribute to the increase in complications associated with pancreatic surgery in women compared with men.25,27 For example, 4 risk factors associated with pancreatic duct stent occlusion in chronic pancreatitis are (1) stent diameter, (2) long stent length, (3) female sex, and (4) pancreatic exocrine insufficiency requiring oral enzyme supplementation.28 Female sex is also an independent predictor for both development of exocrine insufficiency and a pancreatic fistula after patients have undergone a distal pancreatectomy.29,30 Interestingly, male sex is associated with increased risk of pancreaticoduodenal artery aneurysms rupture regardless of size of aneurysm.31 Thus, the increased risk for women over men of complications of endoscopic interventions and surgery is likely due in part to the reduced pancreatic duct diameter on average (Figure 1B and D).
Women have greater pancreatic echogenicity by ultrasonography than men (ratios 1.52:1), whereas fat-to-parenchyma ratio is similar in both sexes until <39 years of age; thereafter, women have ∼20%–29% less ratio than men as fat mass remains largely constant in women, but increases in men with age.25 Women are more likely to develop a normal pancreatic variant with less fatty involution of ventral pancreatic tissue during embryogenesis.32 Screening ultrasonography has shown that men have more subclinical morphological changes including main pancreatic duct dilation and age-dependent calcification, whereas women have more cystic lesions.33 Pancreatic biological sensitivity to secretin, as defined by an increase in serum immunoreactive cationic trypsinogen after secretin challenge, can vary by sex; male sex, smoking, and alcohol intake are significantly associated with increased pancreatic sensitivity to secretin.34,35 Among men and women over 45 years of age, women have lower exocrine pancreas sensitivity to secretin and lower concentrations of bicarbonate in pancreatic juice; this is reversed with short-term estradiol (valerianate) administration in menopausal women.35,36 These findings suggest that estradiol can enhance the efficiency of pancreatic bicarbonate secretion concentration and output volume.
Animal Studies
Mouse and rat pancreas are compact in the splenic side but diffusely dispersed in the mesentery on the duodenal side37; rats but not mice lack a gall bladder. Estradiol administration in male rats increases acinar cell enzyme content but decreased amylase secretion in vivo.38 While gonadectomy has no perceptible effect on acinar cell morphology or zymogen granule numbers, when combined with adrenalectomy it results in marked reduction in zymogen granule numbers and morphology.39,40 Administration of estradiol or triamcinolone, a glucocorticoid, restores zymogen granule numbers to sham control levels within 9 hours of treatment; zymogen granules have decreased amylase levels that do not respond to estradiol treatment. The effect of estradiol is mediated by estradiol-binding protein, which localizes to the rough endoplasmic reticulum (RER) and periphery of the Golgi apparatus, where the estradiol-protein complex might regulate secretory process, possibly at the site of granule formation41; these effects of estrogen are distinct from the actions mediated by ER. In ovariectomized and adrenalectomized female rats, pancreatic acini show wider intralobular and interlobular spaces and decreased acinar lumen size that does not resolve with estradiol treatment.42 In castrated and adrenalectomized male rats, acinar cells show both increased peri- and interlobular spaces that are normalized with estradiol treatment. Swollen Golgi apparatus and increased numbers of condensing vacuoles with the Golgi and zymogen granules are reversed with estradiol treatment.39 Glucocorticoids and progesterone, but not estrogen treatment, in the AR42J rat pancreatic acinar–derived tumor cell line increase number of secretory granules within cells and amylase content.43 Estradiol administration to isolated rat acinar cells doubled intracellular amylase and trypsin contents compared with unstimulated controls as a result of increase in translation of these secretory proteins, but unlike cholecystokinin, did not affect secretion.44 In male rats with obstructive jaundice, plasma estradiol and cholecystokinin levels were increased compared with controls and both levels were positively associated with pancreatic weight and plasma bilirubin levels.45 These findings suggest that both glucocorticoids and estrogens from the adrenals, rather than from the gonads, exert influence on pancreatic secretory apparatus.41 Exocrine pancreas function is only partially reversed by estradiol; steroid hormones exert their effects by acting directly on pancreatic acini, although downstream target genes and mechanisms are largely unknown.
Sex Differences in Pancreatic Diseases
Few studies have considered sex as a biological variable in clinical outcomes in health or disease; instead, sex is accounted for as a confounder, thereby resulting in paucity of data in this area.
Acute Pancreatitis
Human Studies
Prominent sex-dependent differences in the features, including the impact of acute pancreatitis (AP) risk factors are described. In Chinese patients, higher mean waist circumference is associated with risk of AP, and mean waist circumference is higher in men than in women; smoking, alcohol consumption, abdominal adiposity, and diabetes were other factors that contributed to sex differences in increased AP risk.46 Similarly, in Danish patients, the incidence of AP is higher in men than in women.47 These factors (smoking, alcohol, abdominal obesity, and diabetes) appear to account for the overall increased risk of developing AP in males worldwide. In a multiethnic prospective study in the United States, moderate alcohol consumption (<2 glasses/d) was protective against AP in women.48 Female sex is also independently associated with greater risk of biliary pancreatitis and postendoscopic retrograde cholangiopancreaticography pancreatitis.49,50 These differences are independent of morphological changes and anatomical variations of the pancreaticobiliary ductal system and the mechanism(s) accounting for the this difference remains unclear.51 Differences in the clinical manifestation of autoimmune pancreatitis also varies in a sex-dependent manner. Type I autoimmune pancreatitis (AIP), or lymphoplasmacytic sclerosing pancreatitis, is characterized by a fibroinflammatory process and increased IgG4-positive plasma cells. Type II AIP with prominent neutrophilic infiltrates in the lobules and ducts is confined to the pancreas, exhibits normal IgG4 levels, and is difficult to diagnose. Type I AIP predominantly affects elderly men, whereas there are no sex differences in the incidence of type II AIP.52 In Japanese patients with type I AIP, the sex ratio was 3.7 (men to women).53 Men are at greater risk for developing type I AIP differs, whereas women are at greater risk for developing systemic autoimmune diseases. This suggests that the immune mechanisms of AIP may differ in a fundamental way from other autoimmune diseases.
Potential effects of sex hormones on the natural history of AP have been described in human studies. Although mortality from severe AP requiring intensive care unit admission does not differ between sexes, elevated estradiol levels are correlated with increased mortality.54 Elevated estradiol levels (≥102 pg/mL) are more predictive of mortality compared with typical pancreatitis mortality scores including Sequential Organ Failure Assessment score, multiple organ dysfunction score, and APACHE II score.54 The increased risk of death with serum estradiol elevations is seen with other forms of acute injuries in both men and women.55,56 Whether this is due to a direct effect of estradiol in exacerbating or ameliorating injury or it just a biomarker with no direct role in injury remains to be studied. Estrogens, but not anabolic androgen steroids, are associated with both an increased serum triglyceride levels and pancreatitis,57 but the source of these steroid hormones remains to be determined. Case reports describe development of hypertriglyceridemic AP in both men and women in settings of elevated estradiol levels, such as during treatment for in vivo fertilization, birth control pills, and hormonal therapies.58, 59, 60 In 2 patients with lipoprotein lipase deficiency, a genetic disease caused by deficiency in the rate-limiting protein associated with plasma triglyceride catabolism, developed their first episodes of AP after initiation of oral contraceptive pills.58,61 Case reports document recurrent cyclical hereditary pancreatitis induced by menstruation and inhibited by ovarian function suppression. Given the association between estradiol and the exocrine pancreas, the estradiol surge during the menstruation cycle may increase the pancreas sensitivity to pancreatitis development or modulate severity when accompanied by other insults.62
Distribution of intraabdominal/omental fat and intrapancreatic fat is associated with increased risk of severe acute pancreatitis.63,64 Although risk for severe acute pancreatitis is similar in men and women with the same intraabdominal fat content, men have an increased propensity for developing intraabdominal fat compared with women.65 Risk for severe acute pancreatitis increases with greater intrapancreatic fat mass,63 and while not studied in the context of sex differences, intraabdominal vs intrapancreatic fat mass distributions may contribute to exacerbated acute pancreatitis through distinct mechanisms.64 Intrapancreatic fat increases pancreatic parenchymal necrosis, whereas intra-abdominal fat is associated with organ failure with minimal initial pancreatic parenchymal necrosis.64 Future studies should assess whether the association between different types of adipose tissue and pancreatic severity is mediated by sex steroids or glucocorticoids, or both.
Animal Studies
The effects of estrogens in the setting of AP are complex and vary with sex. In the pancreaticobiliary duct-ligated AP model in mice of both sexes, several ER agonists decrease oxidative pancreatic and pulmonary damage.66 In caerulein-induced AP model in male rats, estradiol attenuates lung injury through AKT pathway.67 In a choline-deficient ethionine diet model of pancreatitis, young female but not male mice develop severe acute hemorrhagic pancreatic necrosis. However, when male mice are treated with exogenous estradiol, they develop similar levels of pancreatic injury as do females.68 Thus, whether at baseline or in diseased state, glucocorticoids and estrogens from various sites, including the adrenals and gonads, modulate pancreatic histology and secretion, and probably act on transcriptional regulation of downstream genes in the target tissues.
Estrogen, androgen, and glucocorticoid binding elements are present in the promoter regions human CRHR2 gene; its transcription is influenced by steroid hormones.69 Components of the CRF system are expressed in a sexually dimorphic manner in the bed nucleus of the stria terminalis, a brain region that regulates sexual behaviors across all animals. Not surprisingly, the CRF system modulates sex-dependent differences in pancreatitis and other gut-associated diseases.13,18,21 The CRF system is uniquely positioned to orchestrate a myriad of functions. Its initial function was to regulate nutritional stress in fruit flies and worms and acquired osmo/vasoregulation along with HPA axis modulation to its functions during the course of vertebrate evolution14; all of these processes show sex-biased responses and outcomes. In the caerulein-induced AP model, although both male and female mice develop pancreatitis when given identical doses of caerulein, C57BL/6 female mice show less necrosis and vacuolization than male mice do but have similar levels of neutrophil infiltration as male mice13; baseline edema is higher in female than in male mice. Lack of CRF2 (Crhr2–/–) rendered female more susceptible than male mice to caerulein-induced AP; necrosis and vacuolization were greater than in C57BL/6 mice.13 Lack of CRF2 additionally resulted in greatly distorted RER and atypical autophagic bodies in male Crhr2-/- and heterozygous mice, whereas RER disruptions were less pronounced in female Crhr2-/- mice compared with C57BL/6 mice of either sex. UCN1, a high-affinity ligand for CRF2, decreased endoplasmic reticulum stress and unfolded-protein responses in male but not in female mice.13 Additionally, UCN1 treatment induced different patterns of extracellular signal-regulated kinase signaling. These findings suggest a protective role for CRF2 in pancreatic secretion and organelle stress13; this effect is similar to that seen after estradiol treatment in gonadectomized animals. Thus, autosomal genes that are subject to modulation by steroid hormones of both gonadal and nongonadal origins may mediate sex differences in AP.
Chronic Pancreatitis
Human Studies
Sex-dependent differences are also described in chronic pancreatitis (CP). In epidemiological studies, CP occurs more often in men than in women (ratio of 4.5).70 Among patients with CP, alcohol and tobacco are associated with CP in men (24:1) but not in women.71,72 Male sex is independently associated with requiring hospital readmission among patients with CP.73 Older men are at increased risk of exocrine pancreatic insufficiency compared with older women.74 Men are more likely to transition from acute to CP than women.75 It is possible that this increased risk of developing CP in men after AP is related to the increased rates of smoking in males, a recognize factor for CP development.21 Pancreatic fat accumulation is a significant risk factor for CP,76 but whether pancreatic fat mass can perform steroidogenesis in the setting of pancreatitis and contributes to altered specific estrogen metabolite levels in a sex-specific manner needs to be investigated. In patients with CP, men are more sensitive to deep, generalized hyperalgesia, even with opioid therapy, than women. It is hypothesized that women have a stronger inhibitory neural response than men, leading to reduced hyperalgesia compared with men.77
Animal Studies
Animal studies of CP share findings with human epidemiological studies. The effects of estradiol in animal studies varies among strains. Only male Wistar Bonn/Kobori rats spontaneously develop CP; treatment with estradiol partially attenuates acinar cell apoptosis and CP, independent of corticosterone and testosterone levels or T-cell mediated changes.78 Animal models and select studies of human tissues have implicated excess reactive oxygen species (ROS) in the pathogenesis of CP. Other studies suggest that the ability to reduce ROS levels may be sex dependent. For example, increased ROS accumulation, apoptosis, fibrosis, and inflammation are much greater in male compared with female autophagy-deficient (Atg5 floxed) mice.79 However, tamoxifen treatment of Atg5Fl/Fl mice might have confounded sex-specific results in this study.80
Pancreatic Lesions
Human Studies
Several studies have also described associations between sex and incidence of benign, premalignant, and malignant pancreatic neoplasms. Nonfunctional pancreatic endocrine and neuroendocrine tumors occur more often in women than in men.81,82 Similarly, insulinomas are generally rare, but female sex is associated with negative preoperative ultrasound, as they are isoechoic to healthy tissue among a small study of 29 patients.83 In Chinese patients, women have a higher incidence of pancreatic cystic lesions compared with men (1.11% vs 1.5%; P < .001).84 The pathophysiological mechanism explaining the increased incidence of cysts in women compared with men is unknown. While this study did not find sex differences when examining pancreatic cysts with high-risk features predisposing to development of malignancy, whether sex differences contribute to the development of pancreatic cysts resulting in malignancy in a larger cohort is also unknown. Solid pseudopapillary neoplasms are low-grade malignant tumors and represent 0.9%–2.7% of all pancreatic exocrine neoplasms.85 Premenopausal women develop these malignancies more often than men (male-to-female ratio of 1:9) and present with larger tumors than postmenopausal women and men.86,87 Men have a higher rate of calcification and percentage of solid tumor compared with women.88 Sex differences in pseudopapillary neoplasms are consistent with estradiol effects. ERα, ERβ, and PR expression is present in solid pseudopapillary neoplasms.89 Estradiol increases proliferation of pseudopapillary neoplasms and treatment with antiestrogen tamoxifen is associated with improved survival in patients with pseudopapillary neoplasm and unresectable liver metastatic disease.86
Women have an increased incidence of mucinous cystic neoplasms.87 Middle-aged and elderly women particularly have an increased risk of developing mucinous cystic neoplasms, which often have ovarian-type stroma within the cystic walls and share common features with ovarian mucinous cystic neoplasms.90 Case studies have described the development of mucinous cystic neoplasm while on continuous hormone replacement therapy.91 These neoplasms can be alpha inhibin positive (a marker ovarian sex cord-stromal cells and placental cells), PR positive, ER positive, and occasionally, human chorionic gonadotropin positive.92,93 The association between inhibin positive mucinous cystic neoplasms suggests a potential hormonal component within the pathophysiology of these lesions.94 The preponderance associated with many pancreatic neoplasms in women does not hold for all tumor types. For example, the ratio of men to women incidence of intraductal papillary mucinous neoplasms (IPMNs) specific to the main duct is 3, 1.1, and 1.5, whereas in branch duct the ration is 1.8, 0.76, and 0.66 in Asia, the United States, and Europe, respectively.95,96 Because main duct IPMNs have a much more aggressive behavior than do branch duct tumors, men have a worse overall poorer prognosis for this tumor. Case studies have described IPMNs that induces cyclical AP during menstruation that may be due to activin A or inhibin A.97 Though there is no difference in ER expression in IPMNs compared with normal tissue, IPMNs had greater androgen receptor expression compared with normal tissue.98
Pancreatic Cancer
Human Studies
Men have a higher incidence and lower survival of pancreatic ductal adenocarcinoma than do women.99, 100, 101, 102, 103 When looking at specific pancreatic cancer histological types, women developed cystadenocarcinoma104 and papillary histology105 more often than men, with sex difference attributed to differential exposures to risk factors such as tobacco.106 Body mass index in early adulthood is associated with increased pancreatic cancer in men but not in women.107,108 Whether this difference is due to variations in fat distribution in men vs women remains unclear. Premenopausal women, compared with postmenopausal women, have an odds ratio 2.4 higher of pancreatic cancer development.109 Female sex is also associated with better responses to FOLFIRINOX in unresectable pancreatic cancer.110 After pancreatic disease, including acute and CP and pancreatic cancer, men have a 40% higher risk of developing diabetes compared with women.111 Men with pancreatic ductal adenocarcinoma (PDAC) have a significantly lower serum testosterone/dihydrotestosterone ratio than men with CP or nonpancreatic GI tumors.112 This ratio is normalized after resection of stage I pancreatic cancer.113 PDAC patients compared with normal controls have higher levels of estradiol and lower levels of progesterone and testosterone, irrespective of sex.114 Male PDAC patients with low testosterone levels or female PDAC patients with high estrogen levels exhibit worse survival than normal their eugonadal counterparts.115 These studies suggest the possibility of bidirectional signaling between PDAC and sex hormones or dysfunctional HPG axis in patients with PDAC.
Sex hormones may directly signal to pancreatic cancer cells. ERα and ERβ expression is elevated in PDAC cells in culture, particularly during the exponential growth phase.116 This suggests potential benefits of antihormonal drugs, such as estradiol derivatives, in management of pancreatic cancer.117 For example, 2-methoxyestradiol inhibits tumor growth in vivo and in human multidrug resistant pancreatic cancer cells118,119 through mitochondria-dependent apoptosis signaling.120,121 The effects of estrogen metabolites on PDAC are complicated; however, reports suggest that methoxyestradiol exhibits a biphasic effect on pancreatic tumorigensis—both very high and low doses of 2-methoxyestradiol increase cell proliferation and VEGF-A levels through ERα, whereas doses in the middle lead to the opposite effects.122 Treatment of PDAC and neuroendocrine cell lines with high concentrations of progesterone decreased cell proliferation by negatively regulating cell cycle progression through PR isoform A.123,124 Though androgen receptor expression is seen in pancreatic cancer cell lines, only flutamide, an antiandrogen agent, as monotherapy has been reported to have an antitumor effect in humans.117
Animal Studies
Reflecting human epidemiology, male mice are typically more sensitive to PDAC development than female mice in many animal pancreatic cancer models. For example, female mice are less sensitive to corn oil–induced PDAC than male mice, as evidenced by less dramatic increases in acinar hyperplasia and adenoma formation.125 Male rats develop exocrine pancreatic adenomas more often than female rats after injection with another PDAC-inducer 4-hydroxyaminoquinoline-1-oxide. These findings were conserved in castrated male mice with or without testosterone propionate injections.126 In azaserine-treated rat model of PDAC, estrogens inhibit early stages of pancreatic carcinomatosis, whereas androgens may promote pancreatic cancer development.127, 128, 129 Interestingly, in the N-nitroso(2-hydroxypropyl)(2-oxopropyl)amine subcutaneous implantation model of PDAC in Syrian hamsters, female hamsters had a higher incidence, number, and size of PDACs compared with male hamsters. Male hamsters had greater acute liver injury.130 Male mice compared with female mice had more frequent metastatic events after plasma transfusion from human donors.131 These findings highlight species- and strain-dependent differences in animal models, each of which recapitulates some unique feature of human disease.
Human Pregnancy and Pancreatic Diseases
The risk of certain pancreatic diseases increases during pregnancy and could relate to the effect of hormonal fluctuations on factors that affect pancreatic function. Acute pancreatitis among pregnant women is uncommon, occurring in 3 in 10,000 pregnancies but leading to increased mortality risk for the mother and fetus.132 Several factors link pregnancy and the increased risk of pancreatic disease. During pregnancy, biliary cholesterol levels increase and bile acids, including chenodeoxycholic acid and deoxycholic acid, are reduced, making bile more lithogenic. In addition, increased progesterone levels lead to reduced gallbladder smooth muscle wall contraction by inhibiting intracellular calcium through tyrosine kinase and PKA/cAMP pathways. Together, increased bile lithogenicity and bile status increases the risk of gallbladder calculi formation during pregnancy.133 During the third trimester, levels of serum triglycerides increase 2- to 4-fold due to increasing levels of estrogens and placental lactogen.133 This phenomenon is hypothesized to be due to increased fat accumulation and breakdown during pregnancy. Although levels of triglycerides often do not reach >300 mg/dL or levels commonly associated with hypertriglyceridemic pancreatitis, women with preexisting lipid metabolism abnormalities are at increased risk of developing AP.134 Though mucinous cystic neoplasms are commonly premalignant, pregnant women with pancreatic cystic lesions commonly present with malignant lesions that are hormone responsive and may enlarge during pregnancy.135,136 Additionally, there is an increased risk for pancreatic cystic neoplasms to rupture during pregnancy. These risk factors together suggest the importance of individualized treatment and resection timing among pregnant patients with cystic neoplasms.137
Finally, the data associating reproductive factors with decreased risk of PDAC is controversial. Some studies suggest that women with increased parity also have an increased risk of developing pancreatic cancer,109 other studies have shown that women with increased parity (≥7 times) or increased lactation time (≥144 months) had decreased risk of PDAC138 or no relationship between reproductive factors and pancreatic cancer risk.139 It is possible that differences among these studies may be due differences in study populations, exposure quantification, analysis approach, confounders, and design bias. Oral contraceptive pills use and ≥3 pregnancies is associated with reduced PDAC risk, whereas later age of full-term pregnancy increases risk.140,141 A meta-analysis of 10 cohort and 10 case-control studies found that women who gave birth to 2 children had lower risk of pancreatic cancer (relative risk, 0.86), and there was a general decreased risk of pancreatic cancer among women who have given birth and nulliparous women.142 These findings suggest that greater estrogen exposure history is associated with reduced risk of pancreatic cancer development.
Sex differences in Coronavirus Disease 2019
Coronavirus disease 2019 (COVID-19) also exhibits sex differences; although the association between COVID-19 and pancreatic disease is yet to be established, early reports suggest that GI symptoms are more common in women than men.143, 144, 145 Early reports also suggest that men with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection have more severe COVID-19, worse prognosis, and worse outcomes than women.145, 146, 147 The reason for sex difference in COVID-19 outcomes and symptoms is puzzling. Though symptoms associated with COVID-19 are varied, 61.3% of the patients reported at least 1 GI symptom: 33.7% reported diarrhea in a multicenter cohort study across 9 hospitals in the United States.148 Another questionnaire-based study of 1940 nonhospitalized patients in Poland with mild COVID-19 found that self-reported frequency of GI symptoms and olfactory or taste disorders during COVID-19 was higher in women than men.149 However, ascertainment bias in this study cannot be ruled out, and the study used only 4 questions pertaining to COVID-19 symptoms. Several other studies have suggested that mild pancreatic injury may be a rare manifestation of COVID-19150, 151, 152; 17% had elevated amylase or lipase, and pancreatic injury was associated with more severe features of COVID-19.150,153 Other studies have found that between 8.5% and 17.3% of COVID-19 patients present with pancreatic injury.154 The point prevalence of pancreatitis among COVID-19 patients was 0.27%, most commonly due to idiopathic pancreatitis.155 Diabetes, a disease of the endocrine pancreas, also increases the risk of COVID-19 severity and progression, suggesting a potential association between pancreatic diseases and COVID-19.77 In another study, 7% of patients with pancreatic injury also had changes within pancreatic tissue on imaging.156 There is high expression of angiotensin-converting enzyme 2, the protein associated with SARS-CoV-2 entry, within pancreatic tissue, suggesting a possible mechanism for pancreatic injury in SARS-CoV-2 infection.156 Whether sex differences exist in pancreatic manifestations of COVID-19 and its potential mechanisms are yet to be fully determined.
Future directions and challenges facing sex differences research
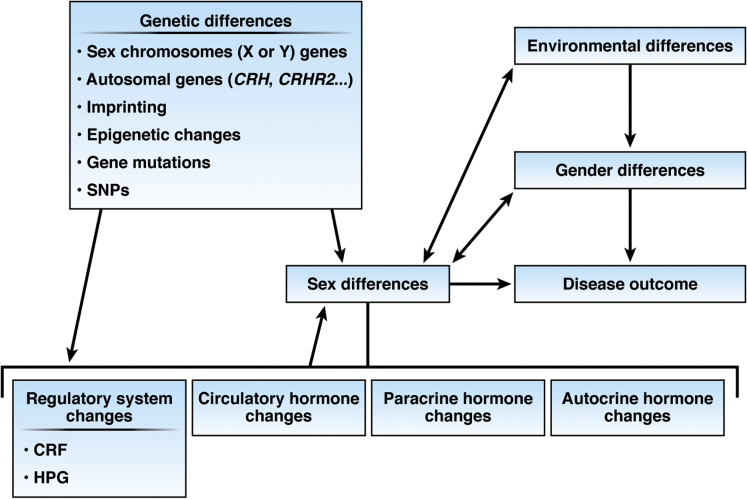
Sex differences in both the normal and diseased exocrine pancreas are evident and should not be ignored in clinical and basic science studies. Sex differences also exist in other GI diseases, most notably inflammatory and functional bowel diseases.157 Sex-dependent differences are due to several major factors: anatomical differences, etiology, differential gene expression, steroid hormone actions, the environment, all of which contribute to underlying (patho)physiological mechanisms (Figure 1). Sex differences research in pancreatic disease has largely focused on etiology, clinical symptoms, circulating sex hormone effects on disease outcomes, and sex hormone derivative effects on pancreatic disease treatment, but few clinical and basic studies have examined mechanisms and outcomes side by side while considering sex as an independent variable. The mechanisms of sex difference in normal and diseased pancreas likely involve an interplay of several factors, not just circulating gonadal hormone levels (Figure 2). Steroid hormones that include both glucocorticoids and estrogens appear to influence acinar cell secretion at both baseline and in pancreatic diseases; however, the role of androgen, progesterone, as well as their metabolites has not been examined. Estrogen and its metabolites mediate their effect by binding to ER and other ER-binding proteins that in turn function as transcription factors to modulate downstream target genes, such as Crh, Crhr2/CRHR2, and Atg5. Use of tandem mass spectrometry allows for simultaneous detection of multiple steroid hormones in plasma and tissue samples and it can specifically distinguish between different steroid metabolites (eg, estradiol vs estrone or estriol and testosterone vs androsterone).2,158 Traditional immunosorbent assays typically report higher steroid levels than mass spectrometry studies due to cross-reactivity; this must be considered in evaluating studies and experimental design. In tissue-specific animal models that use tamoxifen, an estrogen receptor modulator, effects similar to those after estrogen administration can be expected and may not be suitable for studies elucidating sex differences. Because the initiating insults or etiologies can be different for pancreatic diseases between the sexes, distinct and shared mechanisms may be due to expression of genes from sex chromosomes or autosomes and regulated by 1 or more steroid hormones that may originate from the adrenals, gonads, or the target tissue itself. We also acknowledge that cultural and societal differences as well as belonging to a different gender than that assigned at birth also affect health and disease outcomes; these factors have not been considered in any of the literature published thus far on pancreatic diseases but moving forward should be proactively considered. As evidenced by many single studies referenced in this review and the paucity of major clinical studies, sex and gender differences are often ignored in clinical diagnosis, clinical studies, and data analysis. Thus, we find that even when data on sex differences are collected, the data often are not analyzed or reported. Taken together, existing data suggest that sex differences exist, and that a better, more systematic analysis should be considered in future studies. Additionally, a careful analysis at baseline in gonadally intact animals or in healthy humans that sets the foundation for experimental manipulations is much needed for the pancreas and all GI systems.
Figure 2.
Factors that influence sex differences in disease outcomes. Sex differences in pancreatic disease maybe caused by many factors that include genetic differences due to differences in X and Y sex chromosomes, modulation of autosomes by sex hormones, imprinted genes, epigenetic changes, genetic mutations or single nucleotide polymorphisms, environmental differences, gender differences, and differential regulation of the endocrine release of the hormones.
Acknowledgments
The authors thank Caroline Taylor, MD, at Yale School of Medicine and VAMC CT and Thiruvengadam Muniraj, MD, MRCP, at Yale School of Medicine for generously providing radiographic images used in Figure 1C and D.
Footnotes
Conflicts of interest The authors disclose no conflicts.
Funding This work was supported by a National Institutes of HealthNational Institute of Diabetes and Digestive and Kidney Diseases Medical Student Fellowship DK007017 to Melinda Wang; a VA Merit Award, Department of Defense Cancer award (CA180514) to Fred Gorelick; and a P30DK063720 University of California, San Francisco Diabetes Center subaward to Aditi Bhargava.
Contributor Information
Fred Gorelick, Email: fred.gorelick@yale.edu.
Aditi Bhargava, Email: aditi.bhargava@ucsf.edu.
References
- 1.Becker M., Hesse V. Minipuberty: Why does it happen? Horm Res Paediatr. 2020;93:76–84. doi: 10.1159/000508329. [DOI] [PubMed] [Google Scholar]
- 2.Nilsson M.E., Vandenput L., Tivesten A., Norlén A.-K., Lagerquist M.K., Windahl S.H., Börjesson A.E., Farman H.H., Poutanen M., Benrick A., Maliqueo M., Stener-Victorin E., Ryberg H., Ohlsson C. Measurement of a comprehensive sex steroid profile in rodent serum by high-sensitive gas chromatography-tandem mass spectrometry. Endocrinology. 2015;156:2492–2502. doi: 10.1210/en.2014-1890. [DOI] [PubMed] [Google Scholar]
- 3.Morris J.A., Jordan C.L., Breedlove S.M. Sexual differentiation of the vertebrate nervous system. Nat Neurosci. 2004;7:1034–1039. doi: 10.1038/nn1325. [DOI] [PubMed] [Google Scholar]
- 4.Mellon S.H., Griffin L.D. Neurosteroids: biochemistry and clinical significance. Trends Endocrinol Metab. 2002;13:35–43. doi: 10.1016/s1043-2760(01)00503-3. [DOI] [PubMed] [Google Scholar]
- 5.Miller W.L., Bose H.S. Early steps in steroidogenesis: intracellular cholesterol trafficking. J Lipid Res. 2011;52:2111–2135. doi: 10.1194/jlr.R016675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Arlotto M.P., Trant J.M., Estabrook R.W. Measurement of steroid hydroxylation reactions by high-performance liquid chromatography as indicator of P450 identity and function. Methods Enzymol. 1991;206:454–462. doi: 10.1016/0076-6879(91)06114-i. [DOI] [PubMed] [Google Scholar]
- 7.Charni-Natan M., Aloni-Grinstein R., Osher E., Rotter V. Liver and steroid hormones-can a touch of p53 make a difference? Front Endocrinol (Lausanne) 2019;10:374. doi: 10.3389/fendo.2019.00374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Deslypere J.P., Verdonck L., Vermeulen A. Fat tissue: a steroid reservoir and site of steroid metabolism. J Clin Endocrinol Metab. 1985;61:564–570. doi: 10.1210/jcem-61-3-564. [DOI] [PubMed] [Google Scholar]
- 9.Nelson L.R., Bulun S.E. Estrogen production and action. J Am Acad Dermatol. 2001;45:S116–S124. doi: 10.1067/mjd.2001.117432. [DOI] [PubMed] [Google Scholar]
- 10.Robles-Diaz G., Duarte-Rojo A. Pancreas: a sex steroid-dependent tissue. The Israel Med Assoc J. 2001;3:364–368. [PubMed] [Google Scholar]
- 11.Wang L., Xie L.P., Huang W.Q., Yao B., Pu R.L., Zhang R.Q. Presence of gonadotropin-releasing hormone (GnRH) and its mRNA in rat pancreas. Mol Cell Endocrinol. 2001;172:185–191. doi: 10.1016/s0303-7207(00)00369-5. [DOI] [PubMed] [Google Scholar]
- 12.Peyrot C., Brouillard A., Morand-Beaulieu S., Marin M.F. A review on how stress modulates fear conditioning: let's not forget the role of sex and sex hormones. Behav Res Ther. 2020;129:103615. doi: 10.1016/j.brat.2020.103615. [DOI] [PubMed] [Google Scholar]
- 13.Kubat E., Mahajan S., Liao M., Ackerman L., Ohara P.T., Grady E.F., Bhargava A. Corticotropin-releasing factor receptor 2 mediates sex-specific cellular stress responses. Mol Med. 2013;19:212–222. doi: 10.2119/molmed.2013.00036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vuppaladhadiam L., Ehsan C., Akkati M., Bhargava A. Corticotropin-releasing factor family: a stress hormone-receptor system's emerging role in mediating sex-specific signaling. Cells. 2020;9:839. doi: 10.3390/cells9040839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yuan J., Hasdemir B., Tan T., Chheda C., Rivier J., Pandol S.J., Bhagava A. Protective effects of urocortin 2 against caerulein-induced acute pancreatitis. PLoS One. 2019;14 doi: 10.1371/journal.pone.0217065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li C., Chen P., Vaughan J., Lee K.F., Vale W. Urocortin 3 regulates glucose-stimulated insulin secretion and energy homeostasis. Proc Natl Acad Sci U S A. 2007;104:4206–4211. doi: 10.1073/pnas.0611641104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen A., Brar B., Choi C.S., Rousso D., Vaughan J., Kuperman Y., Kim S.N., Donaldson C., Smith S.M., Jamieson P., Li C., Nagy T.R., Shulman G.I., Lee K.-F., Vale W. Urocortin 2 modulates glucose utilization and insulin sensitivity in skeletal muscle. Proc Natl Acad Sci U S A. 2006;103:16580–16585. doi: 10.1073/pnas.0607337103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Paruthiyil S., Hagiwara S.I., Kundassery K., Bhargava A. Sexually dimorphic metabolic responses mediated by CRF2 receptor during nutritional stress in mice. Biol Sex Differ. 2018;9:49. doi: 10.1186/s13293-018-0208-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chang J., Hoy J.J., Idumalla P.S., Clifton M.S., Pecoraro N.C., Bhargava A. Urocortin2 expression in the rat gastrointestinal tract under basal conditions and in chemical colitis. Peptides. 2007;28:1453–1460. doi: 10.1016/j.peptides.2007.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.D'Costa S., Ayyadurai S., Gibson A.J., Mackey E., Rajput M., Sommerville L.J., Wilson N., Li Y., Kubat E., Kumar A., Subramanian H., Bhargava A., Moeser A.J. Mast cell CRF2 suppresses mast cell degranulation and limits the severity of anaphylaxis and stress-induced intestinal permeability. J Allergy Clin Immunol. 2019;143:1865–1877.e4. doi: 10.1016/j.jaci.2018.08.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hasdemir B., Mhaske P., Paruthiyil S., Garnett E.A., Heyman M.B., Matloubian M., Bhargava A. Sex- and corticotropin-releasing factor receptor 2- dependent actions of urocortin 1 during inflammation. Am J Physiol Regul Integr Comp Physiol. 2016;310:R1244–R1257. doi: 10.1152/ajpregu.00445.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.la Fleur S.E., Wick E.C., Idumalla P.S., Grady E.F., Bhargava A. Role of peripheral corticotropin-releasing factor and urocortin II in intestinal inflammation and motility in terminal ileum. Proc Natl Acad Sci U S A. 2005;102:7647–7652. doi: 10.1073/pnas.0408531102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mahajan S., Liao M., Barkan P., Takahashi K., Bhargava A. Urocortin 3 expression at baseline and during inflammation in the colon: corticotropin releasing factor receptors cross-talk. Peptides. 2014;54:58–66. doi: 10.1016/j.peptides.2014.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Neufeld-Cohen A., Tsoory M.M., Evans A.K., Getselter D., Gil S., Lowry C.A., Vale W.W., Chen A. A triple urocortin knockout mouse model reveals an essential role for urocortins in stress recovery. Proc Natl Acad Sci U S A. 2010;107:19020–19025. doi: 10.1073/pnas.1013761107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Saisho Y., Butler A.E., Meier J.J., Monchamp T., Allen-Auerbach M., Pizza R.A., Butler P.C. Pancreas volumes in humans from birth to age one hundred taking into account sex, obesity, and presence of type-2 diabetes. Clin Anat. 2007;20:933–942. doi: 10.1002/ca.20543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Syed A.B., Mahal R.S., Schumm L.P., Dachman A.H. Pancreas size and volume on computed tomography in normal adults. Pancreas. 2012;41:589–595. doi: 10.1097/MPA.0b013e318237457f. [DOI] [PubMed] [Google Scholar]
- 27.Taylor T.V., Rimmer S., Holt S., Jeacock J., Lucas S. Sex differences in gallstone pancreatitis. Ann Surg. 1991;214:667–670. doi: 10.1097/00000658-199112000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Farnbacher M.J., Radespiel-Tröger M., König M.D., Wehler M., Hahn E.G., Schneider H.T. Pancreatic endoprostheses in chronic pancreatitis: Criteria to predict stent occlusion. Gastrointest Endosc. 2006;63:60–66. doi: 10.1016/j.gie.2005.08.046. [DOI] [PubMed] [Google Scholar]
- 29.Elliott I.A., Epelboym I., Winner M., Allendorf J.D., Haigh P.I. Population-level incidence and predictors of surgically induced diabetes and exocrine insufficiency after partial pancreatic resection. Perm J. 2017;21:16–095. doi: 10.7812/TPP/16-095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Makni A., Rebai W., Daghfouss A., Ayadi S., Fterich F., Chebbi F., Ksantini R., Jouini M., Kacem M., Ben Safta Z. Risk factors associated with pancreatic fistula after distal pancreatectomy. Tunis Med. 2012;90:148–153. [PubMed] [Google Scholar]
- 31.Orion K.C., Najafian A., Ehlert B.A., Malas M.B., Black J.H., 3rd, Abularrage C.J. Gender predicts rupture of pancreaticoduodenal artery aneurysms. Ann Vasc Surg. 2016;36:1–6. doi: 10.1016/j.avsg.2016.03.007. [DOI] [PubMed] [Google Scholar]
- 32.Coulier B. The hypoechoic ventral embryologic cephalic pancreas: a large clinical prospective study. J Belge Radiol. 1996;79:120–124. [PubMed] [Google Scholar]
- 33.Ikeda M., Sato T., Morozumi A., Fujino M.A., Yoda Y., Ochiai M., Kobayashi K. Morphologic changes in the pancreas detected by screening ultrasonography in a mass survey, with special reference to main duct dilatation, cyst formation, and calcification. Pancreas. 1994;9:508–512. doi: 10.1097/00006676-199407000-00014. [DOI] [PubMed] [Google Scholar]
- 34.Andriulli A., Masoero G., Amato A., Felder M., Benitti V., Dobrilla G., De La Pierre M., Verme G. Serum immunoreactive cationic trypsinogen response to secretin in normal subjects. Am J Gastroenterol. 1983;78:579–583. [PubMed] [Google Scholar]
- 35.Tiscornia O.M., Mingorance F.N.L., Repetti J.A., Bank S., Tiscornia-Wasserman P.G. Exocrine pancreas function - sex, age, estrogenic and cholinergic tone - preliminary clinical value of citrates “magic powder”. Acta Gastroenterol Latinoamericana. 2019;49:324–330. [Google Scholar]
- 36.Tiscornia O.M., Cresta M.A., Celener D., de Lehmann E.S., Tumilasci O., Scacchi P., Dreiling D.A. Estrogen effects on basal bile-pancreatic secretion and the exocrine-endocrine pancreatic gland in the rat. Mt Sinai J Med. 1986;53:462–469. [PubMed] [Google Scholar]
- 37.Tsuchitani M., Sato J., Kokoshima H. A comparison of the anatomical structure of the pancreas in experimental animals. J Toxicol Pathol. 2016;29:147–154. doi: 10.1293/tox.2016-0016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tangoku A., Doi R., Chowdhury P., Blevins G.T., Pasley J.N., Rayford P.L. Exogenous administration of estradiol and cholecystokinin alters exocrine pancreatic function in rats. Int J Pancreatol. 1993;13:81–86. doi: 10.1007/BF02786075. [DOI] [PubMed] [Google Scholar]
- 39.Beaudoin A.R., Grondin G., St-jean P., Vachereau A., Cabana C., Grossman A. Steroids and the secretory function of the exocrine pancreas. Endocrinology. 1986;119:2106–2117. doi: 10.1210/endo-119-5-2106. [DOI] [PubMed] [Google Scholar]
- 40.Grossman A., Boctor A.M., Band P., Lane B. Role of steroids in secretion-modulating effect of triamcinolone and estradiol on protein synthesis and secretion from the rat exocrine pancreas. J Steroid Biochem. 1983;19:1069–1081. doi: 10.1016/0022-4731(83)90399-0. [DOI] [PubMed] [Google Scholar]
- 41.Beaudoin A.R., Grondin G., St Jean P., Pettengill O., Longnecker D.S., Grossman A. Marked differences in immunocytological localization of [3H]estradiol-binding protein in rat pancreatic acinar tumor cells compared with normal acinar cells. Endocrinology. 1991;128:1617–1622. doi: 10.1210/endo-128-3-1617. [DOI] [PubMed] [Google Scholar]
- 42.Beaudoin A.R., St-Jean P., Proulx J., Grondin G. Influence of steroids on the exocrine pancreas: Presence of laminated bodies in the acinar lumen following castration and adrenalectomy. Pancreas. 1989;4:219–227. doi: 10.1097/00006676-198904000-00011. [DOI] [PubMed] [Google Scholar]
- 43.Logsdon C.D., Moessner J., Williams J.A., Goldfine I.D. Glucocorticoids increase amylase mRNA levels, secretory organelles, and secretion in pancreatic acinar AR42J cells. J Cell Biol. 1985;100:1200–1208. doi: 10.1083/jcb.100.4.1200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hilgendorf I., Gellersen O., Emmrich J., Mikkat U., Rohwedel J., Krammer H.J., Müller P.K., Kruse C. Estradiol has a direct impact on the exocrine pancreas as demonstrated by enzyme and vigilin expression. Pancreatology. 2001;1:24–29. doi: 10.1159/000055788. [DOI] [PubMed] [Google Scholar]
- 45.Tangoku A., Doi R., Chowdhury P., Blevins G.T., Jr., Pasley J.N., Chang L.W., Rayford P.L. Humoral factors that induce alterations of the pancreas in rats with obstructive jaundice. Pancreas. 1993;8:103–108. doi: 10.1097/00006676-199301000-00018. [DOI] [PubMed] [Google Scholar]
- 46.Pang Y., Kartsonaki C., Turnbull I., Guo Y., Yang L., Bian Z., Chen Y., Millwood I.Y., Bragg F., Gong W., Xu Q., Kang Q., Chen J., Li L., Holmes M.V., Chen Z. Metabolic and lifestyle risk factors for acute pancreatitis in Chinese adults: a prospective cohort study of 0.5 million people. PLoS Medicine. 2018;15 doi: 10.1371/journal.pmed.1002618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Spanier B., Bruno M.J., Dijkgraaf M.G. Incidence and mortality of acute and chronic pancreatitis in the Netherlands: a nationwide record-linked cohort study for the years 1995–2005. World J Gastroenterol. 2013;19:3018–3026. doi: 10.3748/wjg.v19.i20.3018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Setiawan V.W., Pandol S.J., Porcel J., Wilkens L.R., Le Marchand L., Pike M.C., Monroe K.R. Prospective study of alcohol drinking, smoking, and pancreatitis: the multiethnic cohort. Pancreas. 2016;45:819–825. doi: 10.1097/MPA.0000000000000657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zarnescu N.O., Costea R., Zarnescu Vasiliu E.C., Neagu S. Clinico-biochemical factors to early predict biliary etiology of acute pancreatitis: age, female gender, and ALT. J Med Life. 2015;8:523–526. [PMC free article] [PubMed] [Google Scholar]
- 50.Zhao Z.H., Hu L.H., Ren H.B., Zhao A.J., Qian Y.Y., Sun X.T., Su S., Zhu S.G., Yu J., Zou W.B., Guo X.R., Wang L., Li Z.S., Liao Z. Incidence and risk factors for post-ERCP pancreatitis in chronic pancreatitis. Gastrointest Endosc. 2017;86:519–524.e1. doi: 10.1016/j.gie.2016.12.020. [DOI] [PubMed] [Google Scholar]
- 51.Bang S., Suh J.H., Park B.K., Park S.W., Song S.Y., Chung J.B. The relationship of anatomic variation of pancreatic ductal system and pancreaticobiliary diseases. Yonsei Med J. 2006;47:243–248. doi: 10.3349/ymj.2006.47.2.243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Morse B., Centeno B., Vignesh S. Autoimmune pancreatitis: updated concepts of a challenging diagnosis. Am J Med. 2014;127:1010.e1–1010.e9. doi: 10.1016/j.amjmed.2014.04.033. [DOI] [PubMed] [Google Scholar]
- 53.Kanno A., Nishimori I., Masamune A., Kikuta K., Hirota M., Kuriyama S., Tsuji I., Shimosegawa T., Research Committee on Intractable Diseases of Pancreas Nationwide epidemiological survey of autoimmune pancreatitis in Japan. Pancreas. 2012;41:835–839. doi: 10.1097/MPA.0b013e3182480c99. [DOI] [PubMed] [Google Scholar]
- 54.Lu C.W., Liu L.C., Hsieh Y.C., Yang L.H., Chen R.J., Hsieh C.H. Increased admission serum estradiol level is correlated with high mortality in patients with severe acute pancreatitis. J Gastroenterol. 2013;48:374–381. doi: 10.1007/s00535-012-0636-6. [DOI] [PubMed] [Google Scholar]
- 55.Feng J.-Y., Liu K.-T., Abraham E., Chen C.-Y., Tsai P.-Y., Chen Y.-C., Lee Y.-C., Yang K.-Y. Serum estradiol levels predict survival and acute kidney injury in patients with septic shock-a prospective study. PLoS One. 2014;9 doi: 10.1371/journal.pone.0097967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Sánchez-Hurtado L.A., Lugo-Cob R., Tejeda-Huezo B.C., Esquivel-Chávez A., Cano-Oviedo A.A., Zamora-Varela S., Gomez-Flores S.S., Arvizo-Tachiquin P., Baltazar-Torres J.A. Serum estradiol level at intensive care unit admission and mortality in critically ill patients. Indian J Crit Care Med. 2018;22:96–99. doi: 10.4103/ijccm.IJCCM_395_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Liane B.J., Magee C. Guerilla warfare on the pancreas? A case of acute pancreatitis from a supplement known to contain anabolic-androgenic steroids. Mil Med. 2016;181:e1395–e1397. doi: 10.7205/MILMED-D-15-00575. [DOI] [PubMed] [Google Scholar]
- 58.Lee J., Goldberg I.J. Hypertriglyceridemia-induced pancreatitis created by oral estrogen and in vitro fertilization ovulation induction. J Clin Lipidol. 2008;2:63–66. doi: 10.1016/j.jacl.2007.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Trenque-Tessereau M.G., Picot C., Herment N., Trenque T.C. Combined estradiol/gestodene and acute pancreatitis. Ann Pharmacother. 2005;39:1953–1954. doi: 10.1345/aph.1G292. [DOI] [PubMed] [Google Scholar]
- 60.Perego E., Scaini A., Romano F., Franciosi C., Uggeri F. Estrogen-induced severe acute pancreatitis in a male. J Pancreas. 2004;5:353–356. [PubMed] [Google Scholar]
- 61.Davidoff F., Tishler S., Rosoff C. Marked hyperlipidemia and pancreatitis associated with oral contraceptive therapy. N Engl J Med. 1973;289:552–555. doi: 10.1056/NEJM197309132891103. [DOI] [PubMed] [Google Scholar]
- 62.Heinig J., Simon P., Weiss F.U., Zimmer K.P., Domschke W., Lerch M.M. Treatment of menstruation-associated recurrence of hereditary pancreatitis with pharmacologic ovarian suppression. Am J Med. 2002;113:164. doi: 10.1016/s0002-9343(02)01147-6. [DOI] [PubMed] [Google Scholar]
- 63.Navina S., Acharya C., DeLany J.P., Orlichenko L.S., Baty C.J., Shiva S.S., Durgampudi C., Karlsson J.M., Lee K., Bae K.T., Furlan A., Behari J., Liu S., McHale T., Nichols L., Papachristou G.I., Yadav D., Singh V.P. Lipotoxicity causes multisystem organ failure and exacerbates acute pancreatitis in obesity. Sci Transl Med. 2011;3:107ra110. doi: 10.1126/scitranslmed.3002573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Durgampudi C., Noel P., Patel K., Cline R., Trivedi R.N., DeLany J.P., Yadav D., Papachristou G.I., Lee K., Acharya C., Jaligama D., Navina S., Murad F., Singh V.P. Acute lipotoxicity regulates severity of biliary acute pancreatitis without affecting its initiation. Am J Pathol. 2014;184:1773–1784. doi: 10.1016/j.ajpath.2014.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Camhi S.M., Bray G.A., Bouchard C., Greenway F.L., Johnson W.D., Newton R.L., Ravussin E., Ryan D.H., Smith S.R., Katzmarzyk P.T. The relationship of waist circumference and BMI to visceral, subcutaneous, and total body fat: sex and race differences. Obesity (Silver Spring) 2011;19:402–408. doi: 10.1038/oby.2010.248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Guleken Z., Ozbeyli D., Acikel-Elmas M., Oktay S., Alev B., Sirvanci S., Ogunc A.V., Cakir O.K. The effect of estrogen receptor agonists on pancreaticobiliary duct ligation induced experimental acute pancreatitis. J Physiol Pharmacol. 2017;68:847–858. [PubMed] [Google Scholar]
- 67.Yang S.J., Chen H.M., Hsieh C.H., Hsieh C.-H., Hsu J.-T., Yeh C.-N., Yeh T.-S., Hwang T.-L., Jan Y.-Y., Chen M.-F. Akt pathway is required for oestrogen-mediated attenuation of lung injury in a rodent model of cerulein-induced acute pancreatitis. Injury. 2011;42:638–642. doi: 10.1016/j.injury.2010.07.242. [DOI] [PubMed] [Google Scholar]
- 68.Rao K.N., Eagon P.K., Okamura K., Van Thiel D.H., Gavaler J.S., Kelly R.H., Lombardi B. Acute hemorrhagic pancreatic necrosis in mice. Induction in male mice treated with estradiol. Am J Pathol. 1982;109:8–14. [PMC free article] [PubMed] [Google Scholar]
- 69.Catalano R.D., Kyriakou T., Chen J., Easton A., Hillhouse E.W. Regulation of corticotropin-releasing hormone type 2 receptors by multiple promoters and alternative splicing: identification of multiple splice variants. Mol Endocrinol. 2003;17:395–410. doi: 10.1210/me.2002-0302. [DOI] [PubMed] [Google Scholar]
- 70.Hirota M., Shimosegawa T., Masamune A., Kikuta K., Kume K., Hamada S., Kihara Y., Satoh A., Kimura K., Tsuji I., Kuriyama S., Research Committee of Intractable Pancreatic Diseases The sixth nationwide epidemiological survey of chronic pancreatitis in Japan. Pancreatology. 2012;12:79–84. doi: 10.1016/j.pan.2012.02.005. [DOI] [PubMed] [Google Scholar]
- 71.Yen S., Hsieh C.C., Macmahon B. Consumption of alcohol and tobacco and other risk factors for pancreatitis. Am J Epidemiol. 1982;116:407–414. doi: 10.1093/oxfordjournals.aje.a113425. [DOI] [PubMed] [Google Scholar]
- 72.Ryu J.K., Lee J.K., Kim Y.T., Lee D.K., Seo D.W., Lee K.T., Kim H.G., Kim J.S., Lee H.S., Kim T.N., Rho M.H., Moon J.H., Liee J., Choi H.S., Lee W.J., Yoo B.M., Yoon Y.B. Korean Multicenter Study Group on Chronic Pancreatitis. Clinical features of chronic pancreatitis in Korea: a multicenter nationwide study. Digestion. 2005;72:207–211. doi: 10.1159/000089414. [DOI] [PubMed] [Google Scholar]
- 73.Shah R., Haydek C., Mulki R., Qayed E. Incidence and predictors of 30-day readmissions in patients hospitalized with chronic pancreatitis: a nationwide analysis. Pancreatology. 2018;18:386–393. doi: 10.1016/j.pan.2018.04.006. [DOI] [PubMed] [Google Scholar]
- 74.Rothenbacher D., Löw M., Hardt P.D., Klör H.U., Ziegler H., Brenner H. Prevalence and determinants of exocrine pancreatic insufficiency among older adults: results of a population-based study. Scand J Gastroenterol. 2005;40:697–704. doi: 10.1080/00365520510023116. [DOI] [PubMed] [Google Scholar]
- 75.Sankaran S.J., Xiao A.Y., Wu L.M., Windsor J.A., Forsmark C.E., Petrov M.S. Frequency of progression from acute to chronic pancreatitis and risk factors: a meta-analysis. Gastroenterology. 2015;149:1490–1500.e1. doi: 10.1053/j.gastro.2015.07.066. [DOI] [PubMed] [Google Scholar]
- 76.Fujii M., Ohno Y., Yamada M., Kamada Y., Miyoshi E. Impact of fatty pancreas and lifestyle on the development of subclinical chronic pancreatitis in healthy people undergoing a medical checkup. Environ Health Prev Med. 2019;24:10. doi: 10.1186/s12199-019-0763-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Buscher H.C., Wilder-Smith O.H., van Goor H. Chronic pancreatitis patients show hyperalgesia of central origin: a pilot study. Eur J Pain. 2006;10:363–370. doi: 10.1016/j.ejpain.2005.06.006. [DOI] [PubMed] [Google Scholar]
- 78.Nakamura S., Yamada T., Hashimoto T., Takahashi S., Sogawa M., Ohara H., Nakazawa T., Sano H., Kuno A., Joh T., Nomura T., Arakawa T., Itoh M. Estradiol alleviates acinar cell apoptosis and chronic pancreatitis in male Wistar Bonn/Kobori rats. Pancreas. 2003;26:e59–e66. doi: 10.1097/00006676-200304000-00024. [DOI] [PubMed] [Google Scholar]
- 79.Diakopoulos K.N., Lesina M., Wörmann S., Song L., Aichler M., Schild L., Artati A., Römisch-Margl W., Wartmann T., Fischer R., Kabiri, Zischka H., Halangk, Demir I.E., Pilsak C., Walch A., Mantzoros C.S., Steiner J.M., Erkan M., Schmid R.M., Witt H., Adamski J., Algül H. Impaired autophagy induces chronic atrophic pancreatitis in mice via sex- and nutrition-dependent processes. Gastroenterology. 2015;148:626–638.e17. doi: 10.1053/j.gastro.2014.12.003. [DOI] [PubMed] [Google Scholar]
- 80.Levy N., Paruthiyil S., Zhao X., Vivar O.I., Saunier E.F., Griffin C., Tagliaferri M., Cohen I., Speed T.P., Leitman D.C. Unliganded estrogen receptor-beta regulation of genes is inhibited by tamoxifen. Mol Cell Endocrinol. 2010;315:201–207. doi: 10.1016/j.mce.2009.08.030. [DOI] [PubMed] [Google Scholar]
- 81.Gullo L., Migliori M., Falconi M., Pederzoli P., Bettini R., Casadei R., Delle Fave G., Corleto V.D., Ceccarelli C., Santini D., Tomassetti P. Nonfunctioning pancreatic endocrine tumors: a multicenter clinical study. Am J Gastroenterol. 2003;98:2435–2439. doi: 10.1111/j.1572-0241.2003.07704.x. [DOI] [PubMed] [Google Scholar]
- 82.Estrozi B., Bacchi C.E. Neuroendocrine tumors involving the gastroenteropancreatic tract: a clinicopathological evaluation of 773 cases. Clinics (Sao Paulo) 2011;66:1671–1675. doi: 10.1590/S1807-59322011001000002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kann P.H., Ivan D., Pfützner A., Forst T., Langer P., Schaefer S. Preoperative diagnosis of insulinoma: low body mass index, young age, and female gender are associated with negative imaging by endoscopic ultrasound. Eur J Endocrinol. 2007;157:209–213. doi: 10.1530/EJE-07-0117. [DOI] [PubMed] [Google Scholar]
- 84.Sun L., Wang Y., Jiang F., Qian W., Shao C., Jin Z. Prevalence of pancreatic cystic lesions detected by magnetic resonance imaging in the Chinese population. J Gastroenterol Hepatol. 2019;34:1656–1662. doi: 10.1111/jgh.14658. [DOI] [PubMed] [Google Scholar]
- 85.Calvani J., Lopez P., Sarnacki S., Molina T.J., Gibaut L., Fabre M., Scharfmann R., Capito C., Galmiche L. Solid pseudopapillary neoplasms of the pancreas do not express major pancreatic markers in pediatric patients. Hum Pathol. 2019;83:29–35. doi: 10.1016/j.humpath.2018.08.010. [DOI] [PubMed] [Google Scholar]
- 86.Kurokawa S., Hirabayashi K., Hadano A., Yamada M., Tajiri T., Nakamura N. Do solid pseudopapillary neoplasms shrink after menopause?: Review of the literature. Pancreas. 2015;44:998–999. doi: 10.1097/MPA.0000000000000358. [DOI] [PubMed] [Google Scholar]
- 87.Regi P., Salvia R., Cena C., Girelli R., Frigerio I., Bassi C. Cystic "feminine" pancreatic neoplasms in men. Do any clinical alterations correlate with these uncommon entities? Int J Surg. 2013;11:157–160. doi: 10.1016/j.ijsu.2012.12.008. [DOI] [PubMed] [Google Scholar]
- 88.Shi S., Zhou Y., Hu C. Clinical manifestations and multi-slice computed tomography characteristics of solid pseudopapillary neoplasms of the pancreas between males and females. BMC Med Imaging. 2019;19:87. doi: 10.1186/s12880-019-0390-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Tognarini I., Tonelli F., Nesi G., Martineti V., Galli G., Gozzini A., Colli E., Zonefrati R., Paglierani M., Marini F., Sorace S., Cavalli T., Cavalli L., Tanini A., Brandi M.L. In vitro effects of oestrogens, antioestrogens and SERMs on pancreatic solid pseudopapillary neoplasm-derived primary cell culture. Cell Oncol. 2010;32:331–343. doi: 10.3233/CLO-2010-0522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Suzuki Y., Sugiyama M., Abe N., Fujioka Y., Atomi Y. Immunohistochemical similarities between pancreatic mucinous cystic tumor and ovarian mucinous cystic tumor. Pancreas. 2008;36:e40–e46. doi: 10.1097/mpa.0b013e3181584643. [DOI] [PubMed] [Google Scholar]
- 91.Tanaka S., Kawamura T., Nakamura N., Teramoto K., Arii S. Mucinous cystadenocarcinoma of the pancreas developing during hormone replacement therapy. Dig Dis Sci. 2007;52:1326–1328. doi: 10.1007/s10620-006-9482-9. [DOI] [PubMed] [Google Scholar]
- 92.Nishihara K., Tsuneyoshi M., Ohshima A., Yamaguchi K. Papillary cystic tumor of the pancreas. Is it a hormone-dependent neoplasm? Pathol Res Pract. 1993;189:521–526. doi: 10.1016/S0344-0338(11)80359-6. [DOI] [PubMed] [Google Scholar]
- 93.Izumo A., Yamaguchi K., Eguchi T., Nishiayama K., Yamamoto H., Yonemasu H., Yao T., Tanaka M., Tsuneyoshi M. Mucinous cystic tumor of the pancreas: Immunohistochemical assessment of 'ovarian-type stroma'. Oncol Rep. 2003;10:515–525. [PubMed] [Google Scholar]
- 94.Yeh M.M., Tang L.H., Wang S., Robert M.E., Zheng W., Jain D. Inhibin expression in ovarian-type stroma in mucinous cystic neoplasms of the pancreas. Appl Immunohistochem Mol Morphol. 2004;12:148–152. doi: 10.1097/00129039-200406000-00009. [DOI] [PubMed] [Google Scholar]
- 95.Ingkakul T., Warshaw A.L., Fernández-Del Castillo C. Epidemiology of intraductal papillary mucinous neoplasms of the pancreas: sex differences between 3 geographic regions. Pancreas. 2011;40:779–780. doi: 10.1097/MPA.0b013e31821f27fb. [DOI] [PubMed] [Google Scholar]
- 96.Jonnalagadda S., Janec E.M. Sex-based differences in pancreatic and biliary disease. Practical Gastroenterol. 2006;30:49–67. [Google Scholar]
- 97.Tikhomirov V., Tikhomirova S., Sieber S., Schiffman M.K. A pancreatic intraductal papillary mucinous tumor causing recurrent acute pancreatitis at the onset of menstrual periods. J Clin Gastroenterol. 2000;31:172–174. doi: 10.1097/00004836-200009000-00019. [DOI] [PubMed] [Google Scholar]
- 98.Georgiadou D., Sergentanis T.N., Sakellariou S., Filkippakis G.M., Zagouri F., Psaltopoulou T., Lazaris A.C., Patsouris E., Gounaris A., Zografos G.C. Immunohistochemical expression of sex steroid hormone receptors, cell cycle regulators and angiogenesis factors in intraductal papillary mucinous neoplasms: an explorative study. Anticancer Res. 2015;35:1049–1056. [PubMed] [Google Scholar]
- 99.Mack T.M., Paganini-Hill A. Epidemiology of pancreas cancer in Los Angeles. Cancer. 1981;47:1474–1483. doi: 10.1002/1097-0142(19810315)47:6+<1474::aid-cncr2820471406>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 100.Eberle C.A., Bracci P.M., Holly E.A. Anthropometric factors and pancreatic cancer in a population-based case-control study in the San Francisco Bay area. Cancer Causes Control. 2005;16:1235–1244. doi: 10.1007/s10552-005-0354-y. [DOI] [PubMed] [Google Scholar]
- 101.Etxeberria J., Goicoa T., López-Abente G., Riebler A., Ugarte M.D. Spatial gender-age-period-cohort analysis of pancreatic cancer mortality in Spain (1990–2013) PLoS One. 2017;12 doi: 10.1371/journal.pone.0169751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Ma J., Siegel R., Jemal A. Pancreatic cancer death rates by race among US men and women, 1970–2009. J Natl Cancer Inst. 2013;105:1694–1700. doi: 10.1093/jnci/djt292. [DOI] [PubMed] [Google Scholar]
- 103.Gordon-Dseagu V.L., Devesa S.S., Goggins M., Stolzenberg-Solomon R. Pancreatic cancer incidence trends: evidence from the Surveillance, Epidemiology and End Results (SEER) population-based data. Int J Epidemiol. 2018;47:427–439. doi: 10.1093/ije/dyx232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Carriaga M.T., Henson D.E. Liver, gallbladder, extrahepatic bile ducts, and pancreas. Cancer. 1995;75:171–190. doi: 10.1002/1097-0142(19950101)75:1+<171::aid-cncr2820751306>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 105.Baylor S.M., Berg J.W. Cross-classification and survival characteristics of 5,000 cases of cancer of the pancreas. J Surg Oncol. 1973;5:335–358. doi: 10.1002/jso.2930050410. [DOI] [PubMed] [Google Scholar]
- 106.Seoane-Mato D., Nuñez O., Fernández-de-Larrea N., Pérez-Gómez B., Pollán M., López-Abente G., Aragonés N. Long-term trends in pancreatic cancer mortality in Spain (1952–2012) BMC Cancer. 2018;18:625. doi: 10.1186/s12885-018-4494-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Koyanagi Y.N., Matsuo K., Ito H., Tamakoshi A., Sugawara Y., Hidaka A., Wada K., Oze I., Kitamura Y., Liu R., Mizoue T., Sawada N., Nagata C., Wakai K., MNakayama T., Sadakane A., Tanaka K., Inoue M., Tsugane S., Sasazuki S. Body-mass index and pancreatic cancer incidence: a pooled analysis of nine population-based cohort studies with more than 340,000 Japanese subjects. J Epidemiol. 2018;28:245–252. doi: 10.2188/jea.JE20160193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Arslan A.A., Helzlsouer K.J., Kooperberg C., Shu X.O., Steplowski E., Bueno-de-Mesquita H.B., Fuchs C.S., Gross M.D., Jacobs E.J., Lacroix A.Z., Petersen G.M., Stolzenberg-Solomon R.Z., Zheng W., Albanes D., Amundadottir L., Bamlet W.R., Barricarte A., Bingham S.A., Boeing H., Boutron-Ruault M.-C., Buring J.E., CHanock S.J., Clipp S., Gaziano J.M., Giovannuicci E.L., Hankinson S.E., Hartge P., Hoover R.N., Hunter D.J., Hutchinson A., Jacobs K.B., Kraft P., Lynch S.M., Manjer J., Manson J.E., McTiernan A., McWilliams R.R., Mendelsohn J.B., Michaud D.S., Palli D., Rohan T.E., Slimani N., Thomas G., Tjønneland A., Tobias G.S., Trichopoulos D., Virtamo J., Wolpin B.M., Yu K., Zeleniuch-Jacquotte A., Patel A.V. Pancreatic Cancer Cohort Consortium (PanScan). Anthropometric measures, body mass index, and pancreatic cancer: a pooled analysis from the Pancreatic Cancer Cohort Consortium (PanScan) Arch Intern Med. 2010;170:791–802. doi: 10.1001/archinternmed.2010.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Navarro Silvera S.A., Miller A.B., Rohan T.E. Hormonal and reproductive factors and pancreatic cancer risk: a prospective cohort study. Pancreas. 2005;30:369–374. doi: 10.1097/01.mpa.0000160301.59319.ba. [DOI] [PubMed] [Google Scholar]
- 110.Hohla F., Hopfinger G., Romeder F., Hohla F., Hopfinger G., Romeder F., Rinnerthaler G., Bezan A., Stättner S., Hauser-Kronberger C., Ulmer H., Greil R. Female gender may predict response to FOLFIRINOX in patients with unresectable pancreatic cancer: a single institution retrospective review. Int J Oncol. 2014;44:319–326. doi: 10.3892/ijo.2013.2176. [DOI] [PubMed] [Google Scholar]
- 111.Pendharkar S.A., Mathew J., Petrov M.S. Age- and sex-specific prevalence of diabetes associated with diseases of the exocrine pancreas: a population-based study. Dig Liver Dis. 2017;49:540–544. doi: 10.1016/j.dld.2016.12.010. [DOI] [PubMed] [Google Scholar]
- 112.Robles-Diaz G., Diaz-Sanchez V., Mendez J.P., Altamirano A., Wolpert E. Low serum testosterone/dihydrotestosterone ratio in patients with pancreatic carcinoma. Pancreas. 1987;2:684–687. doi: 10.1097/00006676-198711000-00010. [DOI] [PubMed] [Google Scholar]
- 113.Fernández-del Castillo C., Robles-Díaz G., Díaz-Sánchez V., Altamirano A. Pancreatic cancer and androgen metabolism: high androstenedione and low testosterone serum levels. Pancreas. 1990;5:515–518. doi: 10.1097/00006676-199009000-00003. [DOI] [PubMed] [Google Scholar]
- 114.Fyssas I., Syrigos K.N., Konstandoulakis M.M., Papadopoulos S., Milingos N., Anapliotou M., Waxman J., Golematis B.C. Sex hormone levels in the serum of patients with pancreatic adenocarcinoma. Horm Metab Res. 1997;29:115–118. doi: 10.1055/s-2007-979002. [DOI] [PubMed] [Google Scholar]
- 115.Skipworth R.J., Moses A.G., Sangster K., Sturgeon C.M., Voss A.C., Fallon M.T., Anderson R.A., Ross J.A., Fearon K.C.H. Interaction of gonadal status with systemic inflammation and opioid use in determining nutritional status and prognosis in advanced pancreatic cancer. Supportive Care Cancer. 2011;19:391–401. doi: 10.1007/s00520-010-0832-y. [DOI] [PubMed] [Google Scholar]
- 116.Hollande E., Fanjul M., Houti N., Faye J.C., Courriere P. Expression of estrogen receptors during growth of human pancreatic adenocarcinoma cells (Capan-1)-relationship with differentiation. In Vitro Cell Dev Biol Anim. 1998;34:593–599. doi: 10.1007/s11626-998-0120-z. [DOI] [PubMed] [Google Scholar]
- 117.Konduri S., Schwarz M.A., Cafasso D., Schwarz R.E. Androgen receptor blockade in experimental combination therapy of pancreatic cancer. J Surg Res. 2007;142:378–386. doi: 10.1016/j.jss.2006.09.034. [DOI] [PubMed] [Google Scholar]
- 118.Schumacher G., Kataoka M., Roth J.A., Mukhopadhyay T. Potent antitumor activity of 2-methoxyestradiol in human pancreatic cancer cell lines. Clin Cancer Res. 1999;5:493–499. [PubMed] [Google Scholar]
- 119.Schumacher G., Hoffmann J., Cramer T., Spinelli A., Jacob D., Bahra M., Pratschke J., Pfitzmann, Schmidt S., Lage H. Antineoplastic activity of 2-methoxyestradiol in human pancreatic and gastric cancer cells with different multidrug-resistant phenotypes. J Gastroenterol Hepatol. 2007;22:1469–1473. doi: 10.1111/j.1440-1746.2007.05033.x. [DOI] [PubMed] [Google Scholar]
- 120.Qanungo S., Basu A., Das M., Haldar S. 2-Methoxyestradiol induces mitochondria dependent apoptotic signaling in pancreatic cancer cells. Oncogene. 2002;21:4149–4157. doi: 10.1038/sj.onc.1205508. [DOI] [PubMed] [Google Scholar]
- 121.Yao L., Zou X., Jin C., Fan J., Guo Z., Lin Q., Li J., Fu D. The roles of ID-1 in human pancreatic ductal adenocarcinoma and the therapeutic effects of 2-methoxyestradiol. Carcinogenesis. 2018;39:728–737. doi: 10.1093/carcin/bgy035. [DOI] [PubMed] [Google Scholar]
- 122.Banerjee S.N., Sengupta K., Banerjee S., Saxena N.K., Banerjee S.K. 2-Methoxyestradiol exhibits a biphasic effect on VEGF-A in tumor cells and upregulation is mediated through ER-alpha: a possible signaling pathway associated with the impact of 2-ME2 on proliferative cells. Neoplasia. 2003;5:417–426. doi: 10.1016/s1476-5586(03)80044-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Goncharov A.I., Maslakova A.A., Polikarpova A.V., Bulanova E.A., Guseva A.A., Morozov I.A., Rubtsov P.M., Smirnova O.V., Schelkunova T.A. progesterone inhibits proliferation and modulates expression of proliferation-related genes in classical progesterone receptor-negative human BxPC3 pancreatic adenocarcinoma cells. J Steroid Biochem Mol Biol. 2017;165:293–304. doi: 10.1016/j.jsbmb.2016.07.007. [DOI] [PubMed] [Google Scholar]
- 124.Yazdani S., Kasajima A., Onodera Y., Mcnamara K.M., Ise K., Nakamura Y., Tachibana T., Motoi F., Unno M., Sasano H. Progesterone arrested cell cycle progression through progesterone receptor isoform A in pancreatic neuroendocrine neoplasm. J Steroid Biochem Mol Biol. 2018;178:243–253. doi: 10.1016/j.jsbmb.2018.01.003. [DOI] [PubMed] [Google Scholar]
- 125.Boorman G.A., Banas D.A., Eustis S.L., Haseman J.K., Proliferative exocrine pancreatic lesions in rats The effect of sample size on the incidence of lesions. Toxicol Pathol. 1987;15:451–456. doi: 10.1177/019262338701500410. [DOI] [PubMed] [Google Scholar]
- 126.Hayashi Y., Katayama H. Promoting effect of testosterone propionate on experimental exocrine pancreatic tumors by 4-hydroxyaminoquinoline 1-oxide in rats. Toxicol Lett. 1981;9:349–354. doi: 10.1016/0378-4274(81)90009-6. [DOI] [PubMed] [Google Scholar]
- 127.Sumi C., Longnecker D.S., Roebuck B.D., Brinck-Johnsen T. Inhibitory effects of estrogen and castration on the early stage of pancreatic carcinogenesis in Fischer rats treated with azaserine. Cancer Res. 1989;49:2332–2336. [PubMed] [Google Scholar]
- 128.Lhoste E.F., Roebuck B.D., Stern J.E., Longnecker D.S. Effect of orchiectomy and testosterone on the early stages of azaserine-induced pancreatic carcinogenesis in the rat. Pancreas. 1987;2:38–43. doi: 10.1097/00006676-198701000-00006. [DOI] [PubMed] [Google Scholar]
- 129.Longnecker D.S., Sumi C. Effects of sex steroid hormones on pancreatic cancer in the rat. Int J Pancreatol. 1990;7:159–165. doi: 10.1007/BF02924233. [DOI] [PubMed] [Google Scholar]
- 130.Kokkinakis D.M. Sex differences in the dietary modulation of pancreatic cancer in Syrian hamsters treated continuously with N-nitroso(2-hydroxypropyl)(2-oxopropyl)amine. Carcinogenesis. 1990;11:1909–1913. doi: 10.1093/carcin/11.11.1909. [DOI] [PubMed] [Google Scholar]
- 131.Moore P.K., Benson D., Kehler M., Moore E.E., Fragoso M., Silliman C.C., Barnett CC JR The plasma fraction of stored erythrocytes augments pancreatic cancer metastasis in male versus female mice. J Surg Res. 2010;164:23–27. doi: 10.1016/j.jss.2010.05.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Pitchumoni C.S., Yegneswaran B. Acute pancreatitis in pregnancy. World J Gastroenterol. 2009;15(45):5641–5646. doi: 10.3748/wjg.15.5641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Jones B.A., Girling J.C. Biliary and pancreatic disease in pregnancy. Fetal Maternal Med Rev. 2010;21:263–282. [Google Scholar]
- 134.Papadakis E.P., Sarigianni M., Mikhailidis D.P., Mamopoulos A., Karagiannis V. Acute pancreatitis in pregnancy: an overview. Eur J Obstet Gynecol Reprod Biol. 2011;159:261–266. doi: 10.1016/j.ejogrb.2011.07.037. [DOI] [PubMed] [Google Scholar]
- 135.Wiseman J.E., Yamamoto M., Nguyen T.D., Bonadio J., Imagawa D.K. Cystic pancreatic neoplasm in pregnancy: a case report and review of the literature. Arch Surg. 2008;143:84–86. doi: 10.1001/archsurg.2007.4. [DOI] [PubMed] [Google Scholar]
- 136.Naganuma S., Honda K., Noriki S., Kimura S., Murakami M., Konerei K., Katayama K., Yamaguchi A., Itoh H. Ruptured mucinous cystic neoplasm with an associated invasive carcinoma of pancreatic head in a pregnant woman: report of a case and review of literature. Pathol Int. 2011;61:28–33. doi: 10.1111/j.1440-1827.2010.02609.x. [DOI] [PubMed] [Google Scholar]
- 137.Iacopi S., Lombardo C., Menonna F., Mazzeo S., Caramella D., Amorese G., Vistoli F., Boggi U. Management of pregnancy-associated pancreatic cystic tumors: review of the literature and results of a Pancreas Club Inc. Survey. Pancreatology. 2018;18:905–912. doi: 10.1016/j.pan.2018.09.010. [DOI] [PubMed] [Google Scholar]
- 138.Lo A.C., Soliman A.S., El-Ghawalby N., Abdel-Wahab M., Fathy O., Khaled H.M., Omar S., Hamilton S.R., Greenson J.K., Abbruzzese J.L. Lifestyle, occupational, and reproductive factors in relation to pancreatic cancer risk. Pancreas. 2007;35:120–129. doi: 10.1097/mpa.0b013e318053e7d3. [DOI] [PubMed] [Google Scholar]
- 139.Wahi M.M., Shah N., Schrock C.E., Rosemurgy A.S., 2nd, Goldin S.B. Reproductive factors and risk of pancreatic cancer in women: a review of the literature. Ann Epidemiol. 2009;19:103–111. doi: 10.1016/j.annepidem.2008.11.003. [DOI] [PubMed] [Google Scholar]
- 140.Kreiger N., Lacroix J., Sloan M. Hormonal factors and pancreatic cancer in women. Ann Epidemiol. 2001;11:563–567. doi: 10.1016/s1047-2797(01)00219-8. [DOI] [PubMed] [Google Scholar]
- 141.Luo A.J., Feng R.H., Wang X.W., Wang F.Z. Older age at first birth is a risk factor for pancreatic cancer: a meta-analysis. Hepatobiliary Pancreat Dis Int. 2016;15:125–130. doi: 10.1016/s1499-3872(16)60063-2. [DOI] [PubMed] [Google Scholar]
- 142.Zhu B., Zou L., Han J., Chen W., Shen N., Zhong R., Li J., Chen X., Liu C., Shi Y., Miao X. Parity and pancreatic cancer risk: evidence from a meta-analysis of twenty epidemiologic studies. Sci Rep. 2014;4:5313. doi: 10.1038/srep05313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol. 2020;20:269–270. doi: 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Jin J.M., Bai P., He W., Wu F., Liu X.-F., Han D.-M., Liu S., Yang J.-K. Gender differences in patients with COVID-19: focus on severity and mortality. Front Public Health. 2020;8:152. doi: 10.3389/fpubh.2020.00152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Gebhard C., Regitz-Zagrosek V., Neuhauser H.K., Morgan R., Klein S.L. Impact of sex and gender on COVID-19 outcomes in Europe. Biol Sex Differ. 2020;11:29. doi: 10.1186/s13293-020-00304-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Kragholm K., Andersen M.P., Gerds T.A., Butt J.H., Østergaard L., Polcwiartek C., Phelps M., Gislason G.H., Torp-Pedersen C., Køber L., Schou M., Fosbøl E.L. Association between male sex and outcomes of Coronavirus Disease 2019 (Covid-19) - a Danish nationwide, register-based study. Clin Infect Dis. 2020 Jul 8 doi: 10.1093/cid/ciaa924. [E-pub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Redd W.D., Zhou J.C., Hathorn K.E., McCarty T.R., Bazarbashi A.N., Thompson C.C., Shen L., Chan W.W. Prevalence and characteristics of gastrointestinal symptoms in patients with severe acute respiratory syndrome coronavirus 2 infection in the United States: a multicenter cohort study. Gastroenterology. 2020;159:765–767.e2. doi: 10.1053/j.gastro.2020.04.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Sierpinski R., Pinkas J., Jankowski M., Zgliczynski W.S., Wierzba W., Gujski M., Szumowski L. Sex differences in the frequency of gastrointestinal symptoms and olfactory or taste disorders in 1942 nonhospitalized patients with coronavirus disease 2019 (COVID-19) Pol Arch Intern Med. 2020;130:501–505. doi: 10.20452/pamw.15414. [DOI] [PubMed] [Google Scholar]
- 150.Wang F., Wang H., Fan J., Zhang Y., Wang H., Zhao Q. Pancreatic injury patterns in patients with COVID-19 pneumonia. Gastroenterology. 2020;159:367–370. doi: 10.1053/j.gastro.2020.03.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Aloysius M.M., Thatti A., Gupta A., Sharma N., Bansal P., Goyal H. COVID-19 presenting as acute pancreatitis. Pancreatology. 2020;20:1026–1027. doi: 10.1016/j.pan.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Hadi A., Werge M.P., Kristiansen K.T., Pedersen U.G., Karsetensen J.G., Novovic S., Gluud L.L. Coronavirus Disease-19 (COVID-19) associated with severe acute pancreatitis: Case report on three family members. Pancreatology. 2020;20:665–667. doi: 10.1016/j.pan.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Dirweesh A., Li Y., Trikudanathan G., Mallery J.S., Freeman M.L., Amateau S.K. Clinical outcomes of acute pancreatitis in patients with coronavirus disease 2019. Gastroenterology. 2020;159:1972–1974. doi: 10.1053/j.gastro.2020.07.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Samanta J., Gupta R., Singh M.P., Patnaik I., Kumar A., Kochhar R. Coronavirus disease 2019 and the pancreas. Pancreatology. 2020;20:1567–1575. doi: 10.1016/j.pan.2020.10.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Inamdar S., Benias P.C., Liu Y. Prevalence, risk factors, and outcomes of hospitalized patients with coronavirus disease 2019 presenting as acute pancreatitis. Gastroenterology. 2020;159:2226–2228.e2. doi: 10.1053/j.gastro.2020.08.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Mukherjee R., Smith A., Sutton R. Covid-19-related pancreatic injury. Br J Surg. 2020;107 doi: 10.1002/bjs.11645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Shah S.C., Khalili H., Gower-Rousseau C., Olen O., Benchimol E.I., Lynge E., Nielsen K.R., Brassard P., Vutcovici M., Bitton A., Bernstein C.N., Leddin D., Tamim H., Stefansson T., Loftus E.V., Jr., Moum B., Tang W., Ng S.C., Gearry R., Sincic B., Bell S., Sands B.E., Lakatos P.L., Végh Z., Ott C., Kaplan G.G., Burisch J., Colombel J.-F. Sex-based differences in incidence of inflammatory bowel diseases—pooled analysis of population-based studies from Western countries. Gastroenterology. 2018;155:1079–1089.e3. doi: 10.1053/j.gastro.2018.06.043. [DOI] [PubMed] [Google Scholar]
- 158.Soldin O.P., Guo T., Weiderpass E., Tractenberg R.E., Hilakivi-Clarke L., Soldin S.J. Steroid hormone levels in pregnancy and 1 year postpartum using isotope dilution tandem mass spectrometry. Fertil Steril. 2005;84:701–710. doi: 10.1016/j.fertnstert.2005.02.045. [DOI] [PMC free article] [PubMed] [Google Scholar]