Abstract
Early identification and treatment significantly improve clinical outcomes of psychotic disorders. Recent studies identified protein components of the complement and coagulation systems as key pathways implicated in psychosis. These specific protein alterations are integral to the inflammatory response and can begin years before the onset of clinical symptoms of psychotic disorder. Critically, they have recently been shown to predict the transition from clinical high risk to first-episode psychosis, enabling stratification of individuals who are most likely to transition to psychotic disorder from those who are not. This reinforces the concept that the psychosis spectrum is likely a central nervous system manifestation of systemic changes and highlights the need to investigate plasma proteins as diagnostic or prognostic biomarkers and pathophysiological mediators. In this review, we integrate evidence of alterations in proteins belonging to the complement and coagulation protein systems, including the coagulation, anticoagulation, and fibrinolytic pathways and their dysregulation in psychosis, into a consolidated mechanism that could be integral to the progression and manifestation of psychosis. We consolidate the findings of altered blood proteins relevant for progression to psychotic disorders, using data from longitudinal studies of the general population in addition to clinical high-risk (CHR) individuals transitioning to psychotic disorder. These are compared to markers identified from first-episode psychosis and schizophrenia as well as other psychosis spectrum disorders. We propose the novel hypothesis that altered complement and coagulation plasma levels enhance their pathways’ activating capacities, while low levels observed in key regulatory components contribute to excessive activation observed in patients. This hypothesis will require future testing through a range of experimental paradigms, and if upheld, complement and coagulation pathways or specific proteins could be useful diagnostic or prognostic tools and targets for early intervention and preventive strategies.
Subject terms: Neuroscience, Biological techniques
Introduction
Psychotic disorders such as schizophrenia are among the most severe mental disorders, with large individual and societal costs [1]. Early identification and intervention are associated with improved symptomatic and functional outcomes [2, 3]. The liability to psychosis likely exists on a spectrum within the general population [4, 5].
Operationalized criteria for the clinical high-risk (CHR) state [6] serve to identify vulnerable groups at enhanced risk of psychotic disorders [3]. Identification of risk factors for development of psychosis in high-risk individuals is a key aspect of active research [7, 8]. Prognostication of risk is difficult based on clinical symptoms alone, with only ~20–35% of CHR individuals developing psychosis at 3 years. There is thus an urgent need to identify clinically translatable early biomarkers of psychosis risk [9]. Recently, baseline plasma proteomic biomarkers, predominantly components of the complement and coagulation pathways, have been found to accurately discriminate between CHR individuals who do and do not go on to develop a first psychotic episode [10]. This suggests that dysregulation of these systems could play a crucial role in early detection and may provide insights into the early pathophysiology of psychosis.
Here, we propose that the complement and coagulation systems have a central role in the development of psychosis phenotypes. We review existing evidence for immune and coagulation dysfunction in the blood associated with the psychosis phenotypic spectrum. We then focus on the physiological roles of complement and coagulation before examining how dysfunction of these pathways may give rise to pathology. We integrate novel findings into a refinement of the prevailing “two-hit” hypothesis, where early genetic and/or environmental developmental disruptions to the developing central nervous system (CNS) (“first-hit”) increase the vulnerability of the individual to subsequent, late environmental disruptions (“second-hit”), leading to the development of CNS manifestation [11–13]. This proposed theory integrates complement and coagulation dysregulation that can lead to immune activation, contributing to the development of psychotic disorder. Finally, we consider the potential implications in relation to treatment of psychotic disorders and propose directions for future research.
Evidence of complement and coagulation dysfunction in psychosis
The pathophysiology of psychosis is complex, with multiple and heterogeneous biological and environmental factors identified across the developmental lifespan [14]. Inflammation and immune activation are implicated in psychotic disorders such as schizophrenia and bipolar disorder and other major mental disorders. A strong genetic association was established by genome-wide association studies between the major histocompatibility complex (MHC) and schizophrenia [15], including the MHC genetic locus encoding some complement components. This association was partly explained by allelic structural variation of the complement component 4 (C4) gene [16]. In the same study, C4 RNA expression was found to be increased in postmortem brain samples from patients with schizophrenia compared to controls and on immunohistochemical analysis was noted to localize to neurons and synapses [16]. Overexpression of C4A in mice revealed reduced cortical synapse density, increased microglial engulfment of synapses, as well as altered mouse behavior [17]. Transcriptomic studies identified cross-tissue gene signatures in both brain and blood associated with schizophrenia, including pathways mediating immune functions, for instance, differentially expressed complement receptors and regulators [18]. Blood plasma biomarker studies reported elevated complement activation in first-episode psychosis (FEP) [19], with inconsistent results regarding total C3 and unaltered C4 plasma levels [20, 21], although C4B variant specific as well as C4B and C4-S variant deficiency have been observed [22]. Several studies analyzing complement pathway-specific activity have indicated increased complement activation in schizophrenia patients, although inconsistent findings have also been reported [20, 23]. Other links between complement and coagulation pathway dysregulation and schizophrenia include enhanced coagulation activation [24, 25], altered blood protein levels, and abnormal protein phosphorylation [26] and changes in the fibrinolytic [27] and anticoagulant [28] pathways.
The physiological role of complement and coagulation pathways
The complement and coagulation pathways (KEGG: hsa04610) are central to host defense against pathogen infection and injury. The complement system is a key component of the innate immune defense. It is composed of plasma and cell-bound proteins, which are activated via three distinct pathways: the classical pathway (CP, antibody–antigen complex), lectin pathway (LP, carbohydrates), and alternative pathway (AP, contact activation), which converge at the level of C3 activation leading to formation of a membrane-attack complex [29, 30]. Innate immune defense mechanisms can trigger coagulation to limit the invasiveness of the pathogens as well as respond to injury [31], through fibrin formation either by intrinsic (contact activation) or extrinsic (tissue factor (TF)) pathway activation [32]. This is subsequently regulated by plasminogen activation-mediated fibrinolysis [33]. Increased evidence of molecular crosstalk between coagulation and inflammation [34] suggests that coagulation activation can increase inflammation, which in turn amplifies coagulation [35].
Complement and coagulation protein synthesis
In response to infection or injury, immune cells express inflammatory mediators such as cytokines, causing hepatocytes to secrete acute phase proteins, including the complement and coagulation proteins [36, 37]. The acute phase response to cytokine signaling induces profound changes in the plasma proteome [38]. There is evidence of increased levels of IL-6 in schizophrenia patients [39], and other proinflammatory cytokines were observed to be increased in CHR individuals who later developed psychosis compared to those who did not [40], linking inflammatory cytokines to complement and coagulation protein expression. While the liver is the primary site of synthesis for acute phase proteins, extrahepatic synthesis of complement and coagulation components has been reported at serum‐restricted sites [41].
Complement and coagulation pathway activation
Complement and coagulation proteins commonly circulate in plasma as zymogens or procofactors that must be converted into active enzymes or cofactors via limited proteolysis through pathway-specific activation mechanisms [42]. Complement activation relies on recognition of localized molecular patterns, which triggers proteolysis, while regulatory components support clearance of inflammatory mediators after elimination of the trigger [43]. Once triggered, a powerful cascade-like activation of proteins result in: opsonization (C3b, iC3b (AP); C4b (CP, LP)) of pathogens or cells marked for phagocytic clearance; generation of signaling molecules promoting chemotaxis of leukocytes (C5a); and direct cell lysis and killing (C5b-9) [30, 44]. In addition to pathogen killing, complement activation also promotes clearance of immune complexes, or apoptotic cells, and acts as an interface between innate and adaptive immunity [45, 46]. The coagulation TF pathway requires exposure of TF with plasma [47], while the intrinsic (FXII-mediated) pathway is triggered by contact with anionic surfaces, both converging at thrombin-mediated fibrin clot formation [48]. Coagulation is linked to innate immunity as it also contributes to host–pathogen defenses [32] by limiting pathogen dissemination and supporting pathogen killing [31]. Dysregulation of the complement and coagulation systems is caused by continuous activation of either proteolytic pathway (e.g., through unresolved, prolonged infection) or inadequate regulation (e.g., as a result of inherited or acquired deficiencies). This may lead directly to pathology [44] or contribute to infection-related complications such as thrombosis [31]. Pathway-specific dysregulation leads to different phenotypes seen in several diseases, which further vary with genetic or nongenetic risk factors predisposing to disease [49, 50].
Molecular crosstalk of complement and coagulation proteins
Expression level differences in complement and coagulation proteins set their pathways’ activating capacities, thus influencing susceptibility to diseases involving complement and coagulation activation, as well as fibrinolytic and anticoagulant pathway activity. In addition, the molecular crosstalk of these evolutionarily linked systems may contribute to concurrent activation and amplification, which may further aggravate pathology. Relevant molecular protein crosstalk of complement and coagulation proteins identified in Table 1(B) and their functional effect on complement and coagulation activity is summarized in Table 2.
Table 1.
(A) Topmost implicated plasma proteins of the coagulation and complement pathways implicated in longitudinal studies observing conversion to psychotic disorder, or psychotic experiences, and clinical high-risk (CHR) transition to psychotic disorder, as well as case-control comparison for schizophrenia and psychosis spectrum disorders. (B) Overview of topmost implicated plasma proteins of the coagulation and complement pathways identified in two or more studies.
| (A) | |||||||
|---|---|---|---|---|---|---|---|
| Reference | No. of samples | Sample | Disease | Age range | Method | Complement | Coagulation |
| Transition studies—psychosis | |||||||
| English et al. [59]* | 37 PD, 38 no PD and 40 PE, 66 no PE | Age 12, blood. Comparison of age 18 PD vs non-PD | Psychotic disorder | 12 | LC–MS/MS | C1R (↑), C1S (↓), CFD (↑), C6 (↑), C7 (↑), C4BP (↓), CFH (↑), CFI (↑), CLU (↑), VTN (↑), IGHM (↓) | FXII (↑), FXI (↑), FIX (↑), FII (↑), FV (↑), FXIII (↑), PLG (↑), SERPINF2 (↑), A2M (↓) |
| Föcking et al. [60]* | 64 PE, 67 no PE | Age 12, blood. Comparison of age 18 PE vs non-PE | Psychotic experiences | 12 | LC–MS/MS | C1RL (↑), C5 (↑), C8 (↑), C4BP (↓), CFH (↑), VTN (↑), IGHM (↓), IGG (↓) | PLG (↑), A2M (↓) |
| Madrid-Gambin et al. [61] | 48 PE, 67 no PE | Comparison of blood at age 12 against PE at age 18 | Psychotic experiences | 12 | Targeted proteomics (DIA) | VTN (↑) | F11 (↑), HC2 (↑), PLG (↑), SERPINF2 (↑) |
| Perkins et al. [40] | 32 CHR psychosis, 35 HC, 40 CHR no psychosis | Transition vs nontransition | Clinical high risk for psychosis | 12–35 | Multianalyte profiling, immunoassay | VTN (↑) | FVII (↑), vWF (↑), A2M (↑) |
| Mongan et al. [10]* | 49 transition to psychosis and 84 no transition, 61 PE, 61 HC | Blood, Transition vs nontransition | Schizophrenia (SZ) | 18–27 and 12 | LC–MS/MS | C1QA (↑), C1QB (↑), C1R (↑), C1S (↑), C1RL (↑), C2 (↑), C3 (↑), C4A (↑), C4B (↑), C5 (↓), C6 (↑), C7 (↑), C8A (↑), C8B (↓), C9 (↓), CFB (↑), CFHR1 (↑), CFHR2 (↑), CFHR5 (↑), CLUS (↑), CFI (↑), CFH (↑), C4BPA (↓), FCN3 (↓), VTN (↓), IGHM (↓) | A2M (↓), F2 (↑), F9 (↑), F10 (↑), F11 (↑), F12 (↑), F13A (↓), F13B (↑), PLG (↑), SERPING1 (↓), SERPINA1 (↓), SERPINA5 (↓), SERPINA10 (↓), PROZ (↓), HC2 (↓), PROC (↓), PROS (↓), SERPINC1 (↑), SERPIND1 (↑) |
| Case–control studies—psychosis | |||||||
| Chan et al. [58] | 127 first onset SZ, 204 HC | Control vs FEP | SZ | 18–49 | Multianalyte profiling, immunoassay | FVII (↑), vWF (↑), A2M (↑) | |
| Herberth et al. [143] | 17 SZ, 17 HC | Control vs FEP | SZ | 22–39 | Multianalyte profiling, immunoassay | A2M (↓) | |
| Li et al. [56]* | 10 SZ, 10 HC and 47 SZ, 53 HC | Blood, case-control | SZ | 24–58.8 | LC–MS/MS | C4BPB (↓), C8B (↑), IGHM (↑) | F7 (↓), PROS (↓), SERPINA5 (↑) |
| Jaros et al. [26]* | 20 SZ, 20 HC | Blood, case-control | SZ | 22–41.4 | Immobilized metal ion affinity chromatography (IMAC) combined with LC–MS/MS | C4BPA (↑), C6 (↑), CFB (↑), FCN3 (↑) | |
| Levin et al. [25]* | 22 SZ, 33 HC | Blood, case-control | SZ | 18–44 | LC–MS/MS | IGHM(↓) | F13B (↓) |
| Cooper et al. [144] | 60 SZ, 77 HC, 892 blood spot samples | Blood, neonatal blood spots | SZ | 23.7–43.7 | Multiple reaction monitoring mass spectrometry | C4A (↑), C4BPA (↓), C9 (↑), CLUS (↑) | |
| Walss-Bass et al. [145] | 60 SZ, 20 HC | Blood | SZ | 41.1–43.7 | X-aptamer technology | C4A (↑) | |
| Ramsey et al. [146] | 133 SZ, 133 HC | Blood, female vs male | SZ | 16.5–49.6 | Multianalyte profiling, immunoassay | C3 (↑) | F7 (↓), SERPINA1 (↑) |
| Moriyana et al. [147]* | 6 SZ, 6 HC | Umbilical arterial serum | SZ | 22.8–38.3 | LC–MS/MS | C1QB (↑), C1QC (↑), C1R (↑), C1S (↑), C2 (↑), C3 (↑), C4A (↑), C4B (↑), C5 (↑), C7 (↓), C9 (↑), CFB (↑), CFI (↑), C6 (↑), CLU (↑), VTN (↑), IGHM (↑) | F2 (↑), F10 (↑), F12 (↑), F13B (↑), KLKB1 (↑), SERPINA5 (↑), SERPINC1 (↑), SERPIND1 (↑), HC2 (↑), SERPINF2 (↑), SERPING1 (↑), |
| Jiang et al. [148]* | 20 SZ, 10 HC plus 40 SZ, 40 HC | Leukocyte profiling | First-episode SZ | 17.3–33.3 | Proteomic signatures | C1QBP (↑), C1QC (↑), C1R (↑), C4B (↑), C4BPA (↑), C6 (↑), C8B (↓), CD59 (↑), CFB (↑), CFD (↑), CFI (↑), CFH (↑), CR1 (↑) | |
| Gupta et al. [149] | 2 SZ, 2 HC | Cerebrospinal fluid | SZ | 23–28 (SZ) and 53–60 (HC) | Proteomics, iTRAQ | A2M (↓) | |
| Velasquez et al. [150] | 12 SZ, 8 HC | Case-control, brain samples of mitochondria (MIT), crude nuclear fraction (NUC), and cytoplasm (CYT) | SZ | Not stated | Quantitative proteomics, using iTRAQ labeling and SRM | C3 (↑) | |
| Case–control studies—psychosis spectrum | |||||||
| Domenici et al. [55] | 245 MDD, 229 SZ, 254 HC | Blood, case-control | Major depressive Disorder (MDD) and SZ | 27.1–67.5 | Multianalyte profiling, immunoassay | C3 (↑) | A2M (↑), F7 (↑), SERPINA1 (↑) |
| Yang et al. [151] | 24 MDD, 12 HC and 98 MDD, 49 HC | Blood, case-control | Major depressive Disorder (MDD), suicide attempters and nonattempters | 16–46.5 | 2-DE-MALDI-TOF/TOF MS and iTRAQ-LC-MS/MS, western blots and ELISAQ | CFB(↑) | F7 (↑), F10 (↓), SERPINA1 (↑) |
| Turck et al. [152] | 39 MDD, 24 responders, 15 nonresponders | Antidepressant treatment | MDD | 27.4–64.6 | C7 (↓), CFHR1 (↑), CFHR2 (↑), CFHR5 (↑) | F5 (↑), F10 (↓), FGA (↑), FGB (↑), SERPING1 (↓) | |
| Stelzhammer et al. [153] | 40 MDD, 63 HC | Depression, case-control, drugnaive | MDD | 26.4–53.8 | LC–MS/MS | C4B (↑) | |
| Gui et al. [154] | 20 MDD, 20 HC | Blood | MDD | 18–60 | iTRAQ-based quantitative proteomics (and metabolomics) | CFH (↓) | |
| de Jesus et al. [155] | 14 BPD, 12 HC, 23 SZ, other PD:4 | Serum | Bipolar Disorder (BPD), SZ, other Psychotic Disorders | 23–55 | 2D-DiGE | C4A (↑) | |
| Haenisch et al. [156] | 17 BPD, 46 HC | Blood, case-control | BPD | 21–47 | Multianalyte profiling, immunoassay | C3 (↑) | |
| (B) | |||||||
|---|---|---|---|---|---|---|---|
| Pathway | Nested population-based studies (ALSPAC) | Clinical high risk (CHR) – transition (T) vs non-transition (NT) studies | FEP and schizophrenia case-control studies | Other psychosis spectrum case-control studies | |||
| Complement (↑) | CFH, VTN | CFH, VTN C1Q, C1R, CFI, C5, C7, C8 | C4-B, CFB, C6 C1Q, C1R, C3, CFI, C9, CLU | C3 | |||
| Complement (↓) | C4BP, IGM | C4BP, IGM | |||||
| Coagulation (↑) | FXI, PLG SERPINF2 | FXI, PLG FXII, FIX, FII | FXI, PLG FXII, FIX, FII, SERPINF2 | FVII, SERPINA1 | |||
| Coagulation (↓) | A2M | FXI, PLG FXII, FIX, FII | FX | ||||
In (A), topmost proteins are selected by significance (p < 0.05) of expression level fold changes. Studies that conducted comprehensive pathway analyses are labeled with asterisk (*). Upregulation (↑) and downregulation (↓) are indicated for each protein—shown with corresponding gene name.
In (B), overview of protein markers is depicted for longitudinal conversion to psychotic disorder and clinical high risk (CHR)—transition (T) vs nontransition (NT) studies, first-episode psychosis (FEP) and schizophrenia case–control studies, and other psychosis spectrum disorder case–control studies. Longitudinal studies of a general population observing conversion to psychotic experiences and psychotic disorder in the ALSPAC cohort are shown in a separate column. Fold change direction shown for upregulation (↑) and downregulation (↓) is indicated for each protein- shown with corresponding gene name. Only studies that showed consistent fold change direction were included. Proteins marked in bold were found to be altered in >2 studies.
A2M alpha-2-macroglobulin, ADAMTS13 a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 also known as von Willebrand factor-cleaving protease (VWFCP), C1Q complement component 1q, C1R complement component 1r, C1RL complement component 1r Like, C1S complement component 1s, C4BP complement 4 binding protein, C5 complement C5, C6 complement C6, C7 complement C7, C8 complement C8, C8A complement C8A, CFB complement factor B, CFD complement factor D, CFH complement factor H, CFI complement factor I, CLU clusterin, FIC3 ficolin 3, FII prothrombin, FIX factor IX, FVII factor VII, FXI factor XI, FXII factor XII, FXIII factor XIII, IGG immunglobulin G, IGHM immunoglobulin heavy constant Mu, IL10 interleukin 10, IL13 interleukin 13, IL15 interleukin 15, IL8 interleukin 8, PLG plasminogen, PROS vitamin K-dependent protein S, PROZ vitamin K-dependent protein Z, SERPINA7 serpin peptidase inhibitor, clade A member 7, SERPIND1 serpin family D member 1, SERPINF2 serpin family F member 2, SERPING1 plasma protease C1 inhibitor, VTN vitronectin, vWF van Willebrand factor.
Table 2.
Overview of molecular crosstalk identified in the topmost implicated plasma proteins of the coagulation and complement pathways.
| Coagulation | Complement | Effect on Complement | Effect on Coagulation | Reference |
|---|---|---|---|---|
| FXII (↑) | C1Q, C1R (↑) | FXII activates C1 complex |
C1Q inhibits activation of FXII in vitro Platelet activation |
[157–159] |
| FII, FIX, FXI (↑) | C3 (↑) | FIIa, FIXa, and FXIa cleave C3→C3a and C5→C5a |
C3a and C5a increase platelet activation C5a increases tissue factor activity C5a increases expression of PAI-1 on mast cells |
[160–163] |
| FX (↓) | C3, C5 (↑) | FXa cleaves C3 into C3a and C5 into C5a | [160] | |
| PLG (↑) | C5 (↑) | PLN cleaves C5 into C5a | [141] | |
| PLG (↑) | C4BP (↓) |
C4BP binds PLG and increases activation of plasminogen (PLG) to plasmin (PLN) C4BP binds PROS resulting decreased cofactor function of protein S for activated protein C |
[164, 165] | |
| FXI, FXII (↑) | SERPING1 | SERPING1 inhibits FXIa and FXIIa | [166] | |
| SERPING1 | C1R (↑) | SERPING1 inhibits C1R | [167] | |
| A2M (↓) | Thrombin, FXa, PLG (↑) | A2M, C3, and C4 are structurally similar and evolutionarily related | A2M is a protease inhibitor of thrombin, FXa, plasmin | [168–170] |
| Platelets | C7, C8, C9 | C5b6789 (C5b-9) forms the lytic membrane-attack complex |
C5b6789 (C5b-9) affects: Platelet activation; Increased binding of coagulation factors Va and Xa; Increased release of factor V from platelet alpha-granules; Induces endothelial cells to secrete von Willebrand factor; C7 binding interaction with PLG enhances tPA-mediated PLG activation |
[164, 171–173] |
| vWF, TM | CFH (↑) |
CFH and VWF binding interaction enhances CFH cofactor activity and VWF-mediated platelet aggregation CFH and TM binding interaction enhances CFH cofactor activity |
[174–176] | |
| CLU (↑) | CLU binds to C5b-7 and inhibits generation of C5b-9 | [177] | ||
| Thrombin–antithrombin | VTN (↑) | VNT binds to C5b-7 and inhibits C9 polymerization | VTN binds to the thrombin–antithrombin complex (TAT) | [178–180] |
| Protein S | C4BP | C4BP interaction with protein S has no effect on the inhibition of complement activation | C4BP can bind anticoagulant protein S, resulting in a decreased cofactor function of protein S for activated protein C | [181] |
Fold change direction shown for upregulation (↑) and downregulation (↓) is indicated for each protein—shown with corresponding gene name. Proteins identified are shown in bold.
A number of studies (Table 1(B)) found significantly altered levels in crosstalk proteins, which potentially impact the activation, amplification, and regulation of both proteolytic systems. Key crosstalk interactions appear to affect generation of complement opsonins and chemotactic effector molecules and activation of coagulation pathways.
Plasma protein changes in psychosis spectrum disorders
A growing body of literature supports the significant contribution of (neuro)inflammation in the pathogenesis of psychotic disorders [51–54]. Several studies demonstrate changes in plasma protein levels, years before the onset of psychotic experiences or psychotic disorder [10, 40, 55–60]. These highlight the importance of longitudinal studies to identify potential pathophysiological alterations, such as those observed in relation to complement and coagulation pathways [10, 59, 60]. Table 1(A) summarizes the findings for complement and coagulation markers from studies describing altered blood proteins relevant for progression to psychotic disorders, as seen in longitudinal studies of the general population with conversion to psychotic experiences or psychotic disorder as well as CHR individuals transitioning to psychotic disorder. These are compared to markers identified for first-episode psychosis (FEP) and schizophrenia as well as other psychosis spectrum disorders, as shown in Table 1(A).
This breadth of data implicates dysregulation of the complement and coagulation pathways (see Supplementary Table 1 for protein function) suggesting a plausible mechanism, observed years before transition to psychotic experiences [60] or psychotic disorder [59], which was validated in CHR individuals who transitioned to psychotic disorder versus those who did not [10, 40, 58]. There is some observed overlap in protein markers between transition studies and case–control studies assessing FEP or schizophrenia, which further solidifies the relevance of these markers to psychosis. Case–control studies do appear to highlight differences in blood biomarkers (Table 1), however, it is unclear whether these are due to differential study design or reflect pathophysiological changes that are different prior to onset of psychosis that become quiescent in the later stages of CNS manifestation.
It is noteworthy that only a small number of studies use comprehensive analyses such as discovery proteomics to assess the entire complement and coagulation pathways as indicated in Table 1(A). While some studies provide limited data, the study summary (Table 1(B)) clearly shows a robust identification of complement and coagulation biomarkers across all longitudinal studies (complement: high CFH and VTN and low C4BP and IGM; coagulation: high FXI, PLG, and low A2M). This consistency may be attributed to the fact that three out of five studies were conducted in the ALSPAC cohort, a longitudinal study of the general population. In this study, mostly well children were observed, some of whom went on to have psychotic experiences [59–61], and distinct criteria for conversion to psychotic experiences or psychotic disorder were compared. Interestingly, protease inhibitor A2M was consistently downregulated across all conversion and transition studies. A2M was also identified as a marker in schizophrenia case-control studies, albeit not consistently down- or upregulated. We hypothesize that this may reflect differences in clinical stage of illness or possibly the influences of different study design. Table 1(B) further highlights overlapping complement and coagulation proteins, identified for both, longitudinal general population studies with conversion to psychosis and CHR-transition studies, as well as case-control studies (complement: C1Q and C1R; coagulation: FXI and PLG), but also highlights some clear differences that may be a result of distinct study designs or pathophysiological mechanisms during the development of psychosis versus endpoint FEP. In addition, studies involving other psychosis spectrum disorders identified distinct complement and coagulation markers (Table 1(A)), although a lack of comprehensive proteomics discovery studies in these phenotypes discourage speculation as to the relevance of specific markers at this stage.
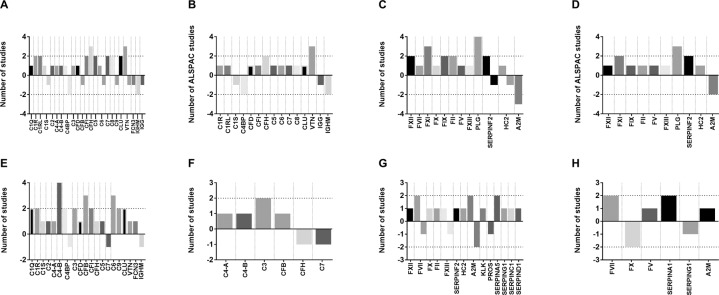
The topmost complement and coagulation proteins consistently identified across all studies are shown in Fig. 1. Overall, there is a distinctive pattern seen with robust and repeated findings of altered complement and coagulation protein levels in conversion or transition to psychosis and case-control studies. In the longitudinal studies, we observe an upregulation of complement and coagulation components and downregulation of key regulatory molecules, which is not reflected in the cross-sectional case-control studies.
Fig. 1. Overview of topmost implicated plasma proteins of the complement and coagulation pathways identified in two or more studies.
Number of studies showing altered complement proteins in A longitudinal conversion and CHR-transition studies, B longitudinal general population studies of the ALSPAC cohort, C case-control psychosis studies, and D case-control psychosis spectrum studies. Number of studies showing altered coagulation proteins in E longitudinal conversion and CHR-transition studies, F longitudinal general population studies of the ALSPAC cohort, G case-control psychosis studies, and H case–control psychosis spectrum studies. Positive or negative depiction indicates direction of fold change found in each study, e.g., a mention of −2 means that two studies found this protein to be downregulated, threshold indicated as two studies.
We propose a novel mechanistic hypothesis whereby complement and coagulation protein upregulation results in enhanced activating capacity of the complement and coagulation pathways. Both systems, once triggered, are able to generate effector molecules (such as chemotactic complement anaphylatoxins C3a and C5a) with functions in the inflammatory response.
Therefore, both systems are less likely to reestablish homeostasis post challenge, leading to enhanced complement and coagulation activity and initiation of pathological mechanisms, which advance progression to psychosis. The discrepancies between cross-sectional and longitudinal studies may indicate temporal changes in complement and coagulation protein alterations according to illness stage or simply the lack of comprehensive discovery case-control studies.
To unravel the pathophysiological mechanisms, it is necessary to view the complement and coagulation pathways as integrated and interlinked defense systems that are upregulated in response to cytokine stimuli of the acute phase response to infection or injury. Given the CNS manifestations of psychotic disorder, it is also important to consider their joint effect on the blood-brain barrier (BBB) and in the brain. Here, we briefly review their physiological roles in the blood and the brain.
Complement and coagulation proteins affect BBB integrity
BBB pathology is recognized as a central factor in the development of many neurological disorders [62]. BBB dysfunction in psychosis might be relevant to many aspects of disrupted neuronal and synaptic function. While it is not clear whether BBB changes are the cause or consequence of neuropathology [63], it is possible that these drive each other, contributing to disease progression. BBB integrity and function [64] disruption, due to systemic inflammation [65] or infection [66], can occur through cytokines affecting BBB integrity [65], as well as CNS functions [67]. For instance, complement C5a-receptor interactions modulate cytokine generation [68] and receptor signaling increases BBB permeability in neuroinflammatory disease [69]. Coagulation proteases thrombin and activated protein C can affect brain pathophysiology by interfering with synapse homeostasis [70] and BBB function [71]. While the plasminogen activation system is key to fibrinolysis [72], in the brain it performs nonfibrinolytic functions [73]. For instance, tissue plasminogen activator (tPA) induces the conversion of plasminogen to plasmin, and promotes BBB permeability via both plasmin‐independent and plasmin‐dependent pathways [73, 74]. Low plasmin activity has been observed in schizophrenia patients [27], as well as low tPA-inhibitor, PAI-1 [75].
While BBB dysfunction is often secondary to the primary insult, it has been suggested as a primary cause in multiple sclerosis, epilepsy, and Alzheimer’s disease [76]. BBB components can actively promote neuroinflammation [76], and BBB disruption may facilitate plasma proteins to enter the brain thereby modulating neuroinflammatory responses [77] and upsetting the fine balance of complement and coagulation components expressed in the brain.
Complement and coagulation in the brain
We provide evidence for the role of systemic alterations of complement and coagulation proteins in the blood. While these are primarily expressed in the liver, all complement components can be locally produced in the brain [78]. The local role of the complement and coagulation systems in the brain in terms of psychosis pathophysiology is not completely established.
Local complement synthesis, modulated by proinflammatory cytokines, was confirmed in the context of Alzheimer’s disease in human astrocytes and microglia, and in this context serves mainly to aid clearance of apoptotic cells or debris [79] with subsequently identified CNS roles in synaptic function [80]. During fetal development, complement proteins contribute to diverse neurodevelopmental processes, while dysregulation can alter the correct balance manifesting in neurodevelopmental disorders [43]. Complement classical pathway (CP) components have been described to aid synaptic elimination [81] and microglial-mediated synaptic pruning [82]. Excessive synaptic pruning on the other hand has not only been associated with developmental and brain diseases but also with normal postnatal CNS development [83]. Therefore, altered complement expression and dysregulation in the brain could result in enhanced synapse pruning, neuronal injury, and neurotoxicity, as seen in neuroinflammatory and neurodegenerative diseases [84].
Coagulation components, in particular the plasminogen activation system, influence numerous aspects of brain function [74, 85], neuroinflammatory processes [86], and neurodegeneration [87], with evidence that fibrin increases axonal damage, microglial activation, and clinical severity in neuroinflammatory disease [88]. The importance of the fibrinolytic system and plasminogen activation in fibrin removal is well characterized, and there is evidence that the plasminogen activation system contributes to brain function [72, 74, 89]. In addition, fibrinogen leakage upon BBB disruption leads to activation of innate immunity in the CNS [90] and fibrin(ogen) deposits in the CNS has a pathogenic role in neurodegenerative and other diseases [91]. Fibrinogen can drive neuroinflammatory responses through CD11b/CD18 (complement receptor 3) [83, 92] and induces microglial-mediated synaptic elimination, while deposition of different coagulation factors may trigger exacerbation of inflammation [93]. For instance, coagulation factors TF, thrombin, or fibrinogen are described as potential drivers of inflammation in disease models [94–98].
These integral roles of complement and coagulation components in neurodevelopment and CNS function, alongside the observed complement and coagulation changes, link systemic changes in blood with changes in synaptic plasticity, vulnerability to neuroinflammation, and neurodegeneration in the brain. However, it is not clear whether synaptic changes observed in psychosis [99, 100] occur as a result of systemic alterations, synergistic changes in the blood and brain or whether they represent potentially independent pathophysiological mechanisms.
Role of complement and coagulation during development
Novel findings indicate that progression to future psychotic experiences and disorder are associated with the presence of an early inflammatory phenotype driven by complement and coagulation pathway-related mechanisms [10, 59, 60]. This enhanced inflammatory-complement and coagulant tone could be triggered by prenatal infection [101, 102], a long-known risk factor for schizophrenia. While normal pregnancy is characterized by an enhanced innate and suppressed adaptive immune response [103], increased plasma levels of complement [104, 105] and acute phase proteins [106] could lead to enhanced complement activity. Complement activation and dysregulation contributes to multiple adverse pregnancy outcomes, and highlights the importance of complement regulation at the fetal-maternal interface [107] and a possible association with maternal immune activation [108–110].
Longitudinal studies, from in utero to early childhood, allow examination of associations between the prenatal environment, brain development, and later behavioral alterations [109]. These could clarify whether the observed complement and coagulation changes are present at birth or a result of maternal immune activation [108–110] and could extend into adolescence. While studies have identified altered cytokine expression in association with risk for psychosis [111], these studies have not yet addressed the involvement of the complement and coagulation pathways in these phenotypes at early developmental stages.
A notable potential postnatal influencing factor is that of childhood adversity. Individuals with a history of childhood adversity have higher levels of circulating markers of acute and chronic inflammation [112, 113] and there is evidence of increased hypothalamic-pituitary-adrenal axis activation and blunted response to stress in FEP [114]. Exposure to stress is a trigger of inflammation associated with neuroinflammation and neurodegenerative disease [115]. Recent evidence indicates that stress can activate the inflammatory response in the brain as well as peripherally [116, 117]. We have observed dysregulated expression of a number of complement proteins in mice exposed to chronic social stress, which were altered in the same direction as seen in subjects progressing to psychotic experiences [60]. This suggests that complement level alterations may also reflect exposure to stress.
Complement is integral to the early immune response against microbial infection [45], which is a recognized risk factor for psychotic disorders. For instance, maternal infection with influenza early in gestation or toxoplasmosis are associated with an increased risk of schizophrenia for the offspring [101, 118], as well as postnatal infections later in life contributing to risk [52]. While an enhanced inflammatory-complement phenotype should support resolution of infection, if unable to reestablish homeostasis post challenge, this could then contribute to chronic inflammation [119].
Complement and coagulation alterations potentially represent a key biological vulnerability, or predisposition, in the progression to psychosis
Several factors likely contribute to the development and eventual CNS manifestation of psychotic disorder and the broader psychosis spectrum. The two-hit hypothesis for schizophrenia suggests that a genetic and prenatal environmental “first-hit” affects brain development, and establishes increased vulnerability to a “second-hit” that may occur later in life [11–13].
We put forward a novel extension of the two-hit hypothesis; we propose that altered complement and coagulation protein levels set their pathways activating capacity, thus influencing susceptibility to immune activation (Fig. 2). This “at-the-ready” state can lead to increased activation when triggered and- as a result of low regulatory components- leads to amplification of these systems. Importantly, lack of key regulatory proteins in these pathways cause dysregulation and pan-pathway crosstalk further amplifies the generation of effector molecules. While under normal circumstances, activation and regulation of these systems are finely balanced and work to reestablish homeostasis in response to an inflammatory insult [42], we propose that the observed alterations may represent a biological vulnerability, or predisposition, in the progression to psychosis. We hypothesize that psychosis is a multiple-hit pathology where several triggering events in a biologically vulnerable individual might lead to psychosis. The observed alterations in complement and coagulation components may represent another “hit” adding to many other genetic or environmental risk factors. We define this distinct vulnerability or “hit” as “dysregulated complement and coagulation activation, and dysfunction of the fibrinolytic and anticoagulant pathways.” A loss of regulatory components could lead to enhanced and persistent complement and coagulation dysfunction. Further research is required to determine whether the enhanced activating capacity or the lack of regulation is a key pathophysiological factor in the development of psychotic disorder and if loss of regulatory components is a result of inherited or acquired deficiency. Overall, complement deficiencies are rare primary immunodeficiency disorders, poorly characterized clinically as they have been difficult to detect, and are probably underdiagnosed in clinical practice [120]. Deficiencies of complement components can be inherited or acquired, partial or complete, and increase susceptibility to infections. Infectious diseases are well known to provoke psychiatric symptoms such as viral infection with herpes simplex virus [121, 122], measles [123] and neuropsychiatric associations with human immunodeficiency virus [124], respiratory virus infections including influenza [125], and most recently SARS-CoV-2 [126–128].
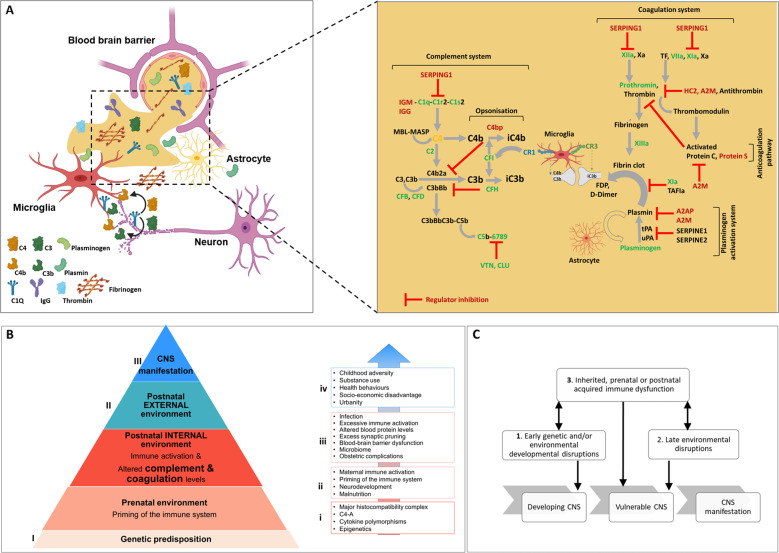
Fig. 2. Mechanism by which infection and inflammation (inherited and acquired risk) influence the progression to psychosis.
A Complement and coagulation proteins and pathways show upregulated components in green (proteomics studies) or blue (transcriptomic studies). Downregulated components are shown in red. Regulatory interactions are highlighted with an inhibition arc colored in red ├. Genetic variant is shown in orange. A blood-brain barrier (BBB) cross-section with complement and coagulation proteins at the neurovascular interface and extravasation into the central nervous system (CNS) following disintegration of the BBB. Activation of the coagulation and plasminogen activation and complement system promoting inflammation and microglia-mediated cellular damage. B The pyramid describes the cumulative risk factors in the progression toward psychosis spectrum disorders illustrating the integrated impact of genetic predisposition and prenatal environment (priming of the immune system), postnatal internal environment (immune activation, altered complement, and coagulation levels), postnatal external environment, leading to central nervous system (CNS) manifestation. Notably, each risk factor is not considered in isolation but as a significant contributing factor and each of the “hits” may have an additive effect on the progression to psychotic disorder. Points of risk assessment and intervention: (I) Risk assessment and stratification, e.g., prenatal environment and genetic risk association, (II) stratification, e.g., clinical high risk (CHR), proposed predicative blood biomarkers and intervention, e.g., cognitive behavioral therapy (CBT), Omega-3 supplementation and proposed anti-inflammatory (complement) and anticoagulant therapeutics. (III) Intervention, e.g., cognitive behavioral therapy (CBT) and antipsychotic drugs. The box and arrow breaks down the individual components as bullet points associated with (i) genetic predisposition, (ii) prenatal environment, (iii) postnatal internal environment, and (iv) postnatal external environment. C The two-hit hypothesis of schizophrenia states that early genetic and/or environmental developmental disruptions (“first-hit”) to the developing central nervous system (CNS) increase the vulnerability of the individual to subsequent, late environmental disruptions (“second-hit”), leading to the development of CNS manifestation. The multiple-hit theory introduces another hit of inherited, prenatal, or postnatal acquired immune dysfunction that sets complement and coagulation pathway activating capacity and regulatory ability integral to development of psychotic disorder (adapted from [12]). A2M alpha-2-macroglobulin, APC activated protein C, AT antithrombin, C1Q complement component 1q, C1r complement C1r subcomponent, C1S complement component 1s, C2 complement C2, C3 complement C3, C3b is the larger of two elements formed by the cleavage of complement component 3, opsonins, C3bBb complement C3 convertase, C4 complement C4, C4b is the larger of two elements formed by the cleavage of complement component 4, opsonin, C4b2a complement C3 convertase, C5b6789 membrane-attack complex, CFB complement factor B, CFD complement factor D, CFH complement factor H, CLU clusterin, D-dimer a fibrin degradation product, FDP fibrin degradation products, FII prothrombin; HC2 heparin cofactor II or SERPIND1, iC3b inactive component 3b, opsonin, IGG immunoglobulin G, IGM immunglobulin M, MASP mannose-associated serine protease, MBL mannose-binding lectin, PAI-1 plasminogen activator inhibitor-1, PROS vitamin K-dependent protein S, SERPING1 plasma protease C1 inhibitor, TF tissue factor, tPA tissue plasminogen activator, VIIa plasma factor VIIa, VTN vitronectin, Xa factor Xa, XIa factor XIa, XIIa factor XIIa, XIIIa factor XIIIa, an enzyme of the blood coagulation system that crosslinks fibrin.
Susceptibility to infections, when the delicate balance between complement and coagulation homeostasis is altered, may contribute to enhanced vulnerability to psychosis as a result of multiple risk factors that influence the development of psychosis, and may have an additive effect (“hit”) on the progression to psychotic disorder. For instance, genetic [16] or early environmental [107, 109] psychosis risk factors are kept in check, unless complement or coagulation pathways are triggered, through, e.g., environmental risk factors [129], or infection [118, 130, 131].
We hypothesize that altered complement and coagulation component levels set their activating capacity. Once triggered (e.g., infection) these cascades generate effector molecules (e.g., opsonins, anaphylatoxins, active serine proteases, fibrin clot). We propose that dysregulation (lack or loss of regulatory components) is the key underlying mechanism driving the amplification of these pathways leading to immune activation and inability to reestablish homeostasis. Peripheral dysregulated complement and coagulation activation leads to cellular and possibly vascular damage, which over time, contributes to BBB leakiness, potential loss of transport regulation, and neuroinflammation [69] and eventual loss of BBB integrity. Plasma extravasation into the brain and potentially altered local expression of complement and coagulation proteins upset the balance of physiological mechanisms (e.g., synaptic plasticity). In addition to extravasation of activated complement and coagulation components, their expression in the brain could be also altered, albeit not necessarily similarly, thus setting activating and regulatory capacity in the brain. Increased complement opsonization could affect complement pathway activating capacity and opsonin-mediated synaptic pruning. The extravasation of coagulation components into the CNS can drive neuroinflammatory responses through deposition of different coagulation factors. Fibrinogen can induce further microglial-mediated synaptic elimination and coagulation serine proteases are further able to generate complement effector molecules (Fig. 2A).
While normal activity of neuroinflammation acts mainly to restore brain homeostasis, prenatal genetic and environmental “first-hit” vulnerability may contribute to the development of CNS manifestation. The pyramid (Fig. 2B) describes the cumulative risk factors in the progression toward psychosis spectrum disorders illustrating the integrated impact of genetic predisposition and prenatal environment (priming of the immune system), postnatal internal environment (immune activation, altered complement, and coagulation levels), and postnatal external environment, leading to CNS manifestation.
Notably, each risk factor is not considered in isolation but as a significant contributing factor and each of the “hits” may have an additive effect on the progression to psychotic disorder. Increased immune activation may contribute to these or even present as a distinct “hit” of inherited, prenatal or postnatal acquired immune dysfunction that sets complement and coagulation pathway activating capacity and regulatory ability and is integral to development of psychotic disorder (Fig. 2C).
It remains a question of debate and further research whether complement and coagulation dysfunction can be seen as a distinct “hit” or is better described as a phase of the immune activation (“second-hit”).
These risk factors in the progression to psychosis may be counterbalanced by a number of measures. Therapeutic interventions at this stage may contribute to reduced neuroinflammation and maintenance of the BBB to prevent progression to psychotic disorder. There is some evidence that therapies targeting inflammatory and immune pathways show efficacy in treatment trials for psychosis. These include nonsteroidal anti-inflammatory drugs as adjuncts to antipsychotics in patients with schizophrenia [132] or other drugs such as minocycline [133], statins, and omega-3 fatty acids [134].
Complement therapeutics are emerging for other disease phenotypes characterized by complement deficiency and dysregulation [135, 136], any association with psychotic symptoms have not been reported yet. Interestingly, anticoagulant therapy has been associated with remission of psychotic symptoms [137] and therefore, anticoagulants could be considered in future research as adjunctive treatments, for instance, target-specific anticoagulants [138]. The efficacy of fibrin-targeting therapeutics needs further investigation [139] considering fibrinogen extravasation into the CNS as a mediator of neurodegeneration [90].
Future directions
Outstanding questions remain. For example, it is unknown whether the altered blood proteome observed in psychosis arises from dysfunctional hepatic or nonhepatic cytokine signaling, acute phase response dysfunction, genetic risk factors, or consumption-mediated deficiency of key regulatory complement and coagulation components. Further investigation is needed to determine whether blood plasma protein expression is correlated with the brain, noting that all key components significantly altered in blood proteome studies are also expressed in the brain and contribute to key CNS functions [78, 80, 81, 140].
To understand the mechanisms of immune activation, we need to investigate the underlying inherited, prenatal or postnatal acquired immune dysfunction mechanisms and their key triggers that cause complement and coagulation dysfunction in psychosis and whether the pathophysiological roles of the complement and/or coagulation systems are distinct “hits” in the progression to psychosis. Therefore, we require further knowledge of the (i) mechanisms that lead to differential protein synthesis (congenital deficiency) or consumption-associated acquired deficiency, (ii) distinct triggers of these proteolytic cascades, (iii) genetic or environmental factors that contribute to dysregulation and altered activating capacity, and (iv) functional consequences of molecular crosstalk.
To confirm whether complement or coagulation deficiency leads to neuropsychiatric disorders, we require the development of mouse models. In line with our findings suggesting that dysregulation of the complement protein pathway reflects past exposure to stress [60], a chronic social stress or an environmental “two-hit” animal model in complement deficient mice could determine whether complement deficiency influences pathophysiology.
Second, it is unclear whether complement and coagulation pathways are dysregulated in the prenatal environment or become established during postnatal development among those who later develop psychotic disorder. Therefore, studies of longitudinal cohorts analyzing the complement and coagulation activation fragments, which are generated when triggering these pathways, are needed and will give an in-depth view of pathway-specific dysregulation in individuals with phenotypes across the psychosis spectrum.
Third, the role of C4 variants [16] (“first-hit”) on the functional effect of C4BP regulating complement C4b-A and -B variants that derives from C4A or C4B needs further investigation. The effect of low plasma concentration of C4BP [59, 60] in conjunction with low protein S on the anticoagulant pathway [28] needs further clarification in these patients. The role of C4BP in the activation of plasminogen [141], due to the association of the plasminogen activation system with schizophrenia [27], merits biochemical investigation of the synergistic impact of this crosstalk interaction on systemic complement and coagulation activity. While C4 and plasminogen/plasmin have been found in the brain, and C4BP was immunohistochemically detected on apoptotic cells in Alzheimer’s brain [142], it remains unclear whether molecular crosstalk or any of the complement and coagulation expression changes that we have observed translate to the psychosis brain.
Lastly, while ours and several other studies have covaried for environmental and other factors, such as BMI, gender, or infection, residual confounding is always possible in observational studies.
Conclusions
There is now compelling evidence for a key role of complement and coagulation alterations in psychosis spectrum disorders and we propose that these changes lead to increased risk for psychosis. Intriguingly, the evidence may be most robust for dysregulation in early psychosis phenotypes, such as in advance of psychotic experiences among apparently well children, or prior to the onset of psychotic disorder among the high-risk population. The observed pattern of protein changes in these pathways has been consistent and points to the presence of chronic immune activation as a risk factor for psychosis. Critically, while the conceptualization of psychosis as an inflammatory condition and immune activation is not new, our data provide a novel understanding and refocus on the consolidated functions of the complement and coagulation pathways contributing to the progression to psychotic disorder. These pathways may provide objective biomarkers of psychosis, and enable identification of therapeutic targets for early and more effective intervention strategies and treatment.
Supplementary information
Acknowledgements
This research was funded by the Irish Health Research Board, ILP POR-2019-005 and HRB ILP POR-2017-039, and the Wellcome Trust, 220438Z/20/Z. DM was funded under the Irish Clinical Academic Training (ICAT) Programme which is supported by the Wellcome Trust and the Health Research Board (Grant Number 203930/B/16/Z), the Health Service Executive National Doctors Training and Planning, and the Health and Social Care, Research and Development Division, Northern Ireland. The funders had no role in the preparation, review, or approval of the manuscript or the decision to submit the manuscript for publication. The authors are extremely grateful to all the families who took part in this study, the midwives for their help in recruiting them, and the whole-ALSPAC team, which includes interviewers, computer and laboratory technicians, clerical workers, research scientists, volunteers, managers, receptionists, and nurses. The UK Medical Research Council and Wellcome (Grant ref: 102215/2/13/2) and the University of Bristol provide core support for ALSPAC. The European Network of National Schizophrenia Networks Studying Gene-Environment Interactions (EU-GEI) Project is funded by grant agreement HEALTH-F2-2010-241909 (Project EU-GEI) from the European Community’s Seventh Framework Programme. The authors also thank the Mass Spectrometry Core Facility at UCD Conway Institute, UCD, for support in the development of our proteomic workflows.
Competing interests
The authors declare conflict of interest as follows: DM, GC, and DRC have a patent pending (UK Patent Application No. 1919155.0, “Biomarkers to predict psychosis”). MH and MF declare no conflict of interest. The same samples used for datasets in this work have been used in other publications [10, 59, 60].
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Meike Heurich, Melanie Föcking.
Contributor Information
Meike Heurich, Email: heurichm@cardiff.ac.uk.
David R. Cotter, Email: drcotter@rcsi.ie
Supplementary information
The online version contains supplementary material available at 10.1038/s41380-021-01197-9.
References
- 1.Gustavsson A, Svensson M, Jacobi F, Allgulander C, Alonso J, Beghi E, et al. Cost of disorders of the brain in Europe 2010. Eur Neuropsychopharmacol. 2011;21:718–79. doi: 10.1016/j.euroneuro.2011.08.008. [DOI] [PubMed] [Google Scholar]
- 2.McGorry PD, Killackey E, Yung A. Early intervention in psychosis: concepts, evidence and future directions. World Psychiatry. 2008;7:148–56. doi: 10.1002/j.2051-5545.2008.tb00182.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Singh SP. Early intervention in psychosis. Br J Psychiatry. 2010;196:343–5. doi: 10.1192/bjp.bp.109.075804. [DOI] [PubMed] [Google Scholar]
- 4.Kelleher I, Keeley H, Corcoran P, Lynch F, Fitzpatrick C, Devlin N, et al. Clinicopathological significance of psychotic experiences in non-psychotic young people: evidence from four population-based studies. Br J Psychiatry. 2012;201:26–32. doi: 10.1192/bjp.bp.111.101543. [DOI] [PubMed] [Google Scholar]
- 5.Healy C, Brannigan R, Dooley N, Coughlan H, Clarke M, Kelleher I, et al. Childhood and adolescent psychotic experiences and risk of mental disorder: a systematic review and meta-analysis. Psychol Med. 2019;49:1589–99. doi: 10.1017/S0033291719000485. [DOI] [PubMed] [Google Scholar]
- 6.Yung AR, Yuen HP, McGorry PD, Phillips LJ, Kelly D, Dell’Olio M, et al. Mapping the onset of psychosis: the comprehensive assessment of at-risk mental states. Aust N Z J Psychiatry. 2005;39:964–71. doi: 10.1080/j.1440-1614.2005.01714.x. [DOI] [PubMed] [Google Scholar]
- 7.Schmidt A, Cappucciati M, Radua J, Rutigliano G, Rocchetti M, Dell’Osso L, et al. Improving prognostic accuracy in subjects at clinical high risk for psychosis: systematic review of predictive models and meta-analytical sequential testing simulation. Schizophr Bull. 2017;43:375–88. doi: 10.1093/schbul/sbw098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Addington J, Stowkowy J, Liu L, Cadenhead KS, Cannon TD, Cornblatt BA, et al. Clinical and functional characteristics of youth at clinical high-risk for psychosis who do not transition to psychosis. Psychol Med. 2019;49:1670–7. doi: 10.1017/S0033291718002258. [DOI] [PubMed] [Google Scholar]
- 9.Fusar-Poli P, Bonoldi I, Yung AR, Borgwardt S, Kempton MJ, Valmaggia L, et al. Predicting psychosis: meta-analysis of transition outcomes in individuals at high clinical risk. Arch Gen Psychiatry. 2012;69:220–9. doi: 10.1001/archgenpsychiatry.2011.1472. [DOI] [PubMed] [Google Scholar]
- 10.Mongan D, Focking M, Healy C, Susai SR, Heurich M, Wynne K, et al. Development of proteomic prediction models for transition to psychotic disorder in the clinical high-risk state and psychotic experiences in adolescence. JAMA Psychiatry. 2021;78:77–90. doi: 10.1001/jamapsychiatry.2020.2459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bayer TA, Falkai P, Maier W. Genetic and non-genetic vulnerability factors in schizophrenia: the basis of the “two hit hypothesis”. J Psychiatr Res. 1999;33:543–8. doi: 10.1016/s0022-3956(99)00039-4. [DOI] [PubMed] [Google Scholar]
- 12.Maynard TM, Sikich L, Lieberman JA, LaMantia AS. Neural development, cell-cell signaling, and the “two-hit” hypothesis of schizophrenia. Schizophr Bull. 2001;27:457–76. doi: 10.1093/oxfordjournals.schbul.a006887. [DOI] [PubMed] [Google Scholar]
- 13.Feigenson KA, Kusnecov AW, Silverstein SM. Inflammation and the two-hit hypothesis of schizophrenia. Neurosci Biobehav Rev. 2014;38:72–93. doi: 10.1016/j.neubiorev.2013.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zwicker A, Denovan-Wright EM, Uher R. Gene-environment interplay in the etiology of psychosis. Psychol Med. 2018;48:1925–36. doi: 10.1017/S003329171700383X. [DOI] [PubMed] [Google Scholar]
- 15.Consortium SWGotPG. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511:421–7. doi: 10.1038/nature13595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sekar A, Bialas AR, de Rivera H, Davis A, Hammond TR, Kamitaki N, et al. Schizophrenia risk from complex variation of complement component 4. Nature. 2016;530:177–83. doi: 10.1038/nature16549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yilmaz M, Yalcin E, Presumey J, Aw E, Ma M, Whelan CW, et al. Overexpression of schizophrenia susceptibility factor human complement C4A promotes excessive synaptic loss and behavioral changes in mice. Nat Neurosci. 2021;24:214–24. doi: 10.1038/s41593-020-00763-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hess JL, Tylee DS, Barve R, de Jong S, Ophoff RA, Kumarasinghe N, et al. Transcriptome-wide mega-analyses reveal joint dysregulation of immunologic genes and transcription regulators in brain and blood in schizophrenia. Schizophrenia Res. 2016;176:114–24. doi: 10.1016/j.schres.2016.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kopczynska M, Zelek W, Touchard S, Gaughran F, Di Forti M, Mondelli V, et al. Complement system biomarkers in first episode psychosis. Schizophrenia Res. 2019;204:16–22. [DOI] [PMC free article] [PubMed]
- 20.Boyajyan A, Khoyetsyan A, Chavushyan A. Alternative complement pathway in schizophrenia. Neurochem Res. 2010;35:894–8. doi: 10.1007/s11064-010-0126-2. [DOI] [PubMed] [Google Scholar]
- 21.Laskaris L, Zalesky A, Weickert CS, Di Biase MA, Chana G, Baune BT, et al. Investigation of peripheral complement factors across stages of psychosis. Schizophrenia Res. 2019;204:30–7. doi: 10.1016/j.schres.2018.11.035. [DOI] [PubMed] [Google Scholar]
- 22.Woo JJ, Pouget JG, Zai CC, Kennedy JL. The complement system in schizophrenia: where are we now and what’s next? Mol Psychiatry. 2020;25:114–30. doi: 10.1038/s41380-019-0479-0. [DOI] [PubMed] [Google Scholar]
- 23.Mayilyan KR, Arnold JN, Presanis JS, Soghoyan AF, Sim RB. Increased complement classical and mannan-binding lectin pathway activities in schizophrenia. Neurosci Lett. 2006;404:336–41. doi: 10.1016/j.neulet.2006.06.051. [DOI] [PubMed] [Google Scholar]
- 24.Yang Y, Wan C, Li H, Zhu H, La Y, Xi Z, et al. Altered levels of acute phase proteins in the plasma of patients with schizophrenia. Anal Chem. 2006;78:3571–6. doi: 10.1021/ac051916x. [DOI] [PubMed] [Google Scholar]
- 25.Levin Y, Wang L, Schwarz E, Koethe D, Leweke FM, Bahn S. Global proteomic profiling reveals altered proteomic signature in schizophrenia serum. Mol Psychiatry. 2010;15:1088–100. doi: 10.1038/mp.2009.54. [DOI] [PubMed] [Google Scholar]
- 26.Jaros JA, Martins-de-Souza D, Rahmoune H, Rothermundt M, Leweke FM, Guest PC, et al. Protein phosphorylation patterns in serum from schizophrenia patients and healthy controls. J Proteom. 2012;76:43–55. doi: 10.1016/j.jprot.2012.05.027. [DOI] [PubMed] [Google Scholar]
- 27.Hoirisch-Clapauch S, Nardi AE. Markers of low activity of tissue plasminogen activator/plasmin are prevalent in schizophrenia patients. Schizophrenia Res. 2014;159:118–23. doi: 10.1016/j.schres.2014.08.011. [DOI] [PubMed] [Google Scholar]
- 28.Hoirisch-Clapauch S, Amaral OB, Mezzasalma MA, Panizzutti R, Nardi AE. Dysfunction in the coagulation system and schizophrenia. Transl Psychiatry. 2016;6:e704. doi: 10.1038/tp.2015.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Walport MJ. Complement. N Engl J Med. 2001;344:1058–66. doi: 10.1056/NEJM200104053441406. [DOI] [PubMed] [Google Scholar]
- 30.Merle NS, Noe R, Halbwachs-Mecarelli L, Fremeaux-Bacchi V, Roumenina LT. Complement system part II: role in immunity. Front Immunol. 2015;6:257. doi: 10.3389/fimmu.2015.00257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Antoniak S. The coagulation system in host defense. Res Pr Thromb Haemost. 2018;2:549–57. doi: 10.1002/rth2.12109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Smith SA, Travers RJ, Morrissey JH. How it all starts: initiation of the clotting cascade. Crit Rev Biochem Mol Biol. 2015;50:326–36. doi: 10.3109/10409238.2015.1050550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Castellino FJ, Ploplis VA. Structure and function of the plasminogen/plasmin system. Thrombosis Haemost. 2005;93:647–54. doi: 10.1160/TH04-12-0842. [DOI] [PubMed] [Google Scholar]
- 34.Conway EM. Complement-coagulation connections. Blood Coagul Fibrinolysis. 2018;29:243–51. doi: 10.1097/MBC.0000000000000720. [DOI] [PubMed] [Google Scholar]
- 35.Esmon CT, Xu J, Lupu F. Innate immunity and coagulation. J Thromb Haemost. 2011;9:182–8. doi: 10.1111/j.1538-7836.2011.04323.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ceciliani F, Giordano A, Spagnolo V. The systemic reaction during inflammation: the acute-phase proteins. Protein Pept Lett. 2002;9:211–23. doi: 10.2174/0929866023408779. [DOI] [PubMed] [Google Scholar]
- 37.Bode JG, Albrecht U, Haussinger D, Heinrich PC, Schaper F. Hepatic acute phase proteins–regulation by IL-6- and IL-1-type cytokines involving STAT3 and its crosstalk with NF-kappaB-dependent signaling. Eur J Cell Biol. 2012;91:496–505. doi: 10.1016/j.ejcb.2011.09.008. [DOI] [PubMed] [Google Scholar]
- 38.Cray C, Zaias J, Altman NH. Acute phase response in animals: a review. Comp Med. 2009;59:517–26. [PMC free article] [PubMed] [Google Scholar]
- 39.Muller N, Riedel M, Ackenheil M, Schwarz MJ. Cellular and humoral immune system in schizophrenia: a conceptual re-evaluation. World J Biol Psychiatry. 2000;1:173–9. doi: 10.3109/15622970009150588. [DOI] [PubMed] [Google Scholar]
- 40.Perkins DO, Jeffries CD, Addington J, Bearden CE, Cadenhead KS, Cannon TD, et al. Towards a psychosis risk blood diagnostic for persons experiencing high-risk symptoms: preliminary results from the NAPLS project. Schizophr Bull. 2015;41:419–28. doi: 10.1093/schbul/sbu099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lubbers R, van Essen MF, van Kooten C, Trouw LA. Production of complement components by cells of the immune system. Clin Exp Immunol. 2017;188:183–94. doi: 10.1111/cei.12952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ricklin D, Hajishengallis G, Yang K, Lambris JD. Complement: a key system for immune surveillance and homeostasis. Nat Immunol. 2010;11:785–97. doi: 10.1038/ni.1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Degn SE, Thiel S. Humoral pattern recognition and the complement system. Scand J Immunol. 2013;78:181–93. doi: 10.1111/sji.12070. [DOI] [PubMed] [Google Scholar]
- 44.Ricklin D, Lambris JD. Complement in immune and inflammatory disorders: pathophysiological mechanisms. J Immunol. 2013;190:3831–8. doi: 10.4049/jimmunol.1203487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Dunkelberger JR, Song W-C. Complement and its role in innate and adaptive immune responses. Cell Res. 2010;20:34–50. doi: 10.1038/cr.2009.139. [DOI] [PubMed] [Google Scholar]
- 46.Noris M, Remuzzi G. Overview of complement activation and regulation. Semin Nephrol. 2013;33:479–92. doi: 10.1016/j.semnephrol.2013.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cimmino G, Ciccarelli G, Golino P. Role of tissue factor in the coagulation network. Semin Thrombosis Hemost. 2015;41:708–17. doi: 10.1055/s-0035-1564045. [DOI] [PubMed] [Google Scholar]
- 48.Palta S, Saroa R, Palta A. Overview of the coagulation system. Indian J Anaesth. 2014;58:515–23. doi: 10.4103/0019-5049.144643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wong EKS, Kavanagh D. Diseases of complement dysregulation—an overview. Semin Immunopathol. 2018;40:49–64. doi: 10.1007/s00281-017-0663-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.de Cordoba SR, Tortajada A, Harris CL, Morgan BP. Complement dysregulation and disease: from genes and proteins to diagnostics and drugs. Immunobiology. 2012;217:1034–46. doi: 10.1016/j.imbio.2012.07.021. [DOI] [PubMed] [Google Scholar]
- 51.Meyer U. Developmental neuroinflammation and schizophrenia. Prog Neuro-Psychopharmacol Biol Psychiatry. 2013;42:20–34. doi: 10.1016/j.pnpbp.2011.11.003. [DOI] [PubMed] [Google Scholar]
- 52.Khandaker GM, Cousins L, Deakin J, Lennox BR, Yolken R, Jones PB. Inflammation and immunity in schizophrenia: implications for pathophysiology and treatment. Lancet Psychiatry. 2015;2:258–70. doi: 10.1016/S2215-0366(14)00122-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tomasik J, Rahmoune H, Guest PC, Bahn S. Neuroimmune biomarkers in schizophrenia. Schizophrenia Res. 2016;176:3–13. doi: 10.1016/j.schres.2014.07.025. [DOI] [PubMed] [Google Scholar]
- 54.Khandaker GM, Dantzer R. Is there a role for immune-to-brain communication in schizophrenia? Psychopharmacology. 2016;233:1559–73. doi: 10.1007/s00213-015-3975-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Domenici E, Wille DR, Tozzi F, Prokopenko I, Miller S, McKeown A, et al. Plasma protein biomarkers for depression and schizophrenia by multi analyte profiling of case-control collections. PLoS ONE. 2010;5:e9166. doi: 10.1371/journal.pone.0009166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Li Y, Zhou K, Zhang Z, Sun L, Yang J, Zhang M, et al. Label-free quantitative proteomic analysis reveals dysfunction of complement pathway in peripheral blood of schizophrenia patients: evidence for the immune hypothesis of schizophrenia. Mol Biosyst. 2012;8:2664–71. doi: 10.1039/c2mb25158b. [DOI] [PubMed] [Google Scholar]
- 57.Schwarz E, Guest PC, Rahmoune H, Harris LW, Wang L, Leweke FM, et al. Identification of a biological signature for schizophrenia in serum. Mol Psychiatry. 2012;17:494–502. doi: 10.1038/mp.2011.42. [DOI] [PubMed] [Google Scholar]
- 58.Chan MK, Krebs MO, Cox D, Guest PC, Yolken RH, Rahmoune H, et al. Development of a blood-based molecular biomarker test for identification of schizophrenia before disease onset. Transl Psychiatry. 2015;5:e601. doi: 10.1038/tp.2015.91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.English JA, Lopez LM, O’Gorman A, Focking M, Hryniewiecka M, Scaife C, et al. Blood-based protein changes in childhood are associated with increased risk for later psychotic disorder: evidence from a nested case-control study of the ALSPAC Longitudinal Birth Cohort. Schizophr Bull. 2018;44:297–306. doi: 10.1093/schbul/sbx075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Focking M, Sabherwal S, Cates HM, Scaife C, Dicker P, Hryniewiecka M, et al. Complement pathway changes at age 12 are associated with psychotic experiences at age 18 in a longitudinal population-based study: evidence for a role of stress. Mol Psychiatry. 2021;26:524–33. [DOI] [PMC free article] [PubMed]
- 61.Madrid-Gambin F, Focking M, Sabherwal S, Heurich M, English JA, O’Gorman A, et al. Integrated lipidomics and proteomics point to early blood-based changes in childhood preceding later development of psychotic experiences: evidence from the Avon Longitudinal Study of Parents and Children. Biol Psychiatry. 2019;86:25–34. doi: 10.1016/j.biopsych.2019.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sweeney MD, Sagare AP, Zlokovic BV. Blood-brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat Rev Neurol. 2018;14:133–50. doi: 10.1038/nrneurol.2017.188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Pollak TA, Drndarski S, Stone JM, David AS, McGuire P, Abbott NJ. The blood-brain barrier in psychosis. Lancet Psychiatry. 2018;5:79–92. doi: 10.1016/S2215-0366(17)30293-6. [DOI] [PubMed] [Google Scholar]
- 64.Abbott NJ, Patabendige AA, Dolman DE, Yusof SR, Begley DJ. Structure and function of the blood-brain barrier. Neurobiol Dis. 2010;37:13–25. doi: 10.1016/j.nbd.2009.07.030. [DOI] [PubMed] [Google Scholar]
- 65.Varatharaj A, Galea I. The blood-brain barrier in systemic inflammation. Brain Behav Immun. 2017;60:1–12. doi: 10.1016/j.bbi.2016.03.010. [DOI] [PubMed] [Google Scholar]
- 66.Chaudhuri JD. Blood brain barrier and infection. Med Sci Monit. 2000;6:1213–22. [PubMed] [Google Scholar]
- 67.Banks WA. Blood-brain barrier transport of cytokines: a mechanism for neuropathology. Curr Pharm Des. 2005;11:973–84. doi: 10.2174/1381612053381684. [DOI] [PubMed] [Google Scholar]
- 68.Yan C, Gao H. New insights for C5a and C5a receptors in sepsis. Front Immunol. 2012;3:368. doi: 10.3389/fimmu.2012.00368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Jacob A, Alexander JJ. Complement and blood–brain barrier integrity. Mol Immunol. 2014;61:149–52. doi: 10.1016/j.molimm.2014.06.039. [DOI] [PubMed] [Google Scholar]
- 70.De Luca C, Virtuoso A, Maggio N, Papa M. Neuro-coagulopathy: blood coagulation factors in central nervous system diseases. Int J Mol Sci. 2017;18:10. doi: 10.3390/ijms18102128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Festoff BW, Sajja RK, van Dreden P, Cucullo L. HMGB1 and thrombin mediate the blood-brain barrier dysfunction acting as biomarkers of neuroinflammation and progression to neurodegeneration in Alzheimer’s disease. J Neuroinflammation. 2016;13:194. [DOI] [PMC free article] [PubMed]
- 72.Draxler DF, Medcalf RL. The fibrinolytic system-more than fibrinolysis? Transfus Med Rev. 2015;29:102–9. doi: 10.1016/j.tmrv.2014.09.006. [DOI] [PubMed] [Google Scholar]
- 73.Niego B, Medcalf RL. Plasmin-dependent modulation of the blood-brain barrier: a major consideration during tPA-induced thrombolysis? J Cereb Blood Flow Metab. 2014;34:1283–96. doi: 10.1038/jcbfm.2014.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Medcalf RL. Fibrinolysis: from blood to the brain. J Thromb Haemost. 2017;15:2089–98. doi: 10.1111/jth.13849. [DOI] [PubMed] [Google Scholar]
- 75.Elmi S, Sahu G, Malavade K, Jacob T. Role of tissue plasminogen activator and plasminogen activator inhibitor as potential biomarkers in psychosis. Asian J psychiatry. 2019;43:105–10. doi: 10.1016/j.ajp.2019.05.021. [DOI] [PubMed] [Google Scholar]
- 76.Daneman R, Prat A. The blood-brain barrier. Cold Spring Harb Perspect Biol. 2015;7:a020412. doi: 10.1101/cshperspect.a020412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Najjar S, Pahlajani S, De Sanctis V, Stern JNH, Najjar A, Chong D. Neurovascular unit dysfunction and blood-brain barrier hyperpermeability contribute to schizophrenia neurobiology: a theoretical integration of clinical and experimental evidence. Front Psychiatry. 2017;8:83. doi: 10.3389/fpsyt.2017.00083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Veerhuis R, Nielsen HM, Tenner AJ. Complement in the brain. Mol Immunol. 2011;48:1592–603. doi: 10.1016/j.molimm.2011.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Veerhuis R, Janssen I, De Groot CJ, Van Muiswinkel FL, Hack CE, Eikelenboom P. Cytokines associated with amyloid plaques in Alzheimer’s disease brain stimulate human glial and neuronal cell cultures to secrete early complement proteins, but not C1-inhibitor. Exp Neurol. 1999;160:289–99. doi: 10.1006/exnr.1999.7199. [DOI] [PubMed] [Google Scholar]
- 80.Tenner AJ, Stevens B, Woodruff TM. New tricks for an ancient system: physiological and pathological roles of complement in the CNS. Mol Immunol. 2018;102:3–13. doi: 10.1016/j.molimm.2018.06.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Stevens B, Allen NJ, Vazquez LE, Howell GR, Christopherson KS, Nouri N, et al. The classical complement cascade mediates CNS synapse elimination. Cell. 2007;131:1164–78. doi: 10.1016/j.cell.2007.10.036. [DOI] [PubMed] [Google Scholar]
- 82.Hong S, Beja-Glasser VF, Nfonoyim BM, Frouin A, Li S, Ramakrishnan S, et al. Complement and microglia mediate early synapse loss in Alzheimer mouse models. Science. 2016;352:712–6. doi: 10.1126/science.aad8373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Schafer DP, Lehrman EK, Kautzman AG, Koyama R, Mardinly AR, Yamasaki R, et al. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron. 2012;74:691–705. doi: 10.1016/j.neuron.2012.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Bonifati DM, Kishore U. Role of complement in neurodegeneration and neuroinflammation. Mol Immunol. 2007;44:999–1010. doi: 10.1016/j.molimm.2006.03.007. [DOI] [PubMed] [Google Scholar]
- 85.Medcalf RL, Lawrence DA. Editorial: the role of the plasminogen activating system in neurobiology. Front Cell Neurosci. 2016;10:222. doi: 10.3389/fncel.2016.00222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Mehra A, Ali C, Parcq J, Vivien D, Docagne F. The plasminogen activation system in neuroinflammation. Biochim Biophys Acta. 2016;1862:395–402. doi: 10.1016/j.bbadis.2015.10.011. [DOI] [PubMed] [Google Scholar]
- 87.Akassoglou K, Strickland S. Nervous system pathology: the fibrin perspective. Biol Chem. 2002;383:37–45. doi: 10.1515/BC.2002.004. [DOI] [PubMed] [Google Scholar]
- 88.Davalos D, Baeten KM, Whitney MA, Mullins ES, Friedman B, Olson ES, et al. Early detection of thrombin activity in neuroinflammatory disease. Ann Neurol. 2014;75:303–8. doi: 10.1002/ana.24078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Bardehle S, Rafalski VA, Akassoglou K. Breaking boundaries-coagulation and fibrinolysis at the neurovascular interface. Front Cell Neurosci. 2015;9:354. doi: 10.3389/fncel.2015.00354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Petersen MA, Ryu JK, Akassoglou K. Fibrinogen in neurological diseases: mechanisms, imaging and therapeutics. Nat Rev Neurosci. 2018;19:283–301. doi: 10.1038/nrn.2018.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Vilar R, Fish RJ, Casini A, Neerman-Arbez M. Fibrin(ogen) in human disease: both friend and foe. Haematologica. 2020;105:284–96. doi: 10.3324/haematol.2019.236901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Erdei A, Lukácsi S, Mácsik-Valent B, Nagy-Baló Z, Kurucz I, Bajtay Z. Non-identical twins: different faces of CR3 and CR4 in myeloid and lymphoid cells of mice and men. Semin Cell Dev Biol. 2019;85:110–21. doi: 10.1016/j.semcdb.2017.11.025. [DOI] [PubMed] [Google Scholar]
- 93.Göbel K, Eichler S, Wiendl H, Chavakis T, Kleinschnitz C, Meuth SG. The coagulation factors fibrinogen, thrombin, and factor XII in inflammatory disorders—a systematic review. Front Immunol. 2018;9:1731. doi: 10.3389/fimmu.2018.01731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Strukova SM. Thrombin as a regulator of inflammation and reparative processes in tissues. Biochem Biokhimiia. 2001;66:8–18. doi: 10.1023/a:1002869310180. [DOI] [PubMed] [Google Scholar]
- 95.Adams RA, Passino M, Sachs BD, Nuriel T, Akassoglou K. Fibrin mechanisms and functions in nervous system pathology. Mol interventions. 2004;4:163–76. doi: 10.1124/mi.4.3.6. [DOI] [PubMed] [Google Scholar]
- 96.Akassoglou K, Adams RA, Bauer J, Mercado P, Tseveleki V, Lassmann H, et al. Fibrin depletion decreases inflammation and delays the onset of demyelination in a tumor necrosis factor transgenic mouse model for multiple sclerosis. Proc Natl Acad Sci USA. 2004;101:6698–703. doi: 10.1073/pnas.0303859101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Schoenmakers SH, Reitsma PH, Spek CA. Blood coagulation factors as inflammatory mediators. Blood Cells Mol Dis. 2005;34:30–7. doi: 10.1016/j.bcmd.2004.09.001. [DOI] [PubMed] [Google Scholar]
- 98.Davalos D, Akassoglou K. Fibrinogen as a key regulator of inflammation in disease. Semin Immunopathol. 2012;34:43–62. doi: 10.1007/s00281-011-0290-8. [DOI] [PubMed] [Google Scholar]
- 99.Ong WY, Garey LJ. Ultrastructural features of biopsied temporopolar cortex (area 38) in a case of schizophrenia. Schizophrenia Res. 1993;10:15–27. doi: 10.1016/0920-9964(93)90072-q. [DOI] [PubMed] [Google Scholar]
- 100.Pennington K, Beasley CL, Dicker P, Fagan A, English J, Pariante CM, et al. Prominent synaptic and metabolic abnormalities revealed by proteomic analysis of the dorsolateral prefrontal cortex in schizophrenia and bipolar disorder. Mol Psychiatry. 2008;13:1102–17. doi: 10.1038/sj.mp.4002098. [DOI] [PubMed] [Google Scholar]
- 101.Brown AS, Derkits EJ. Prenatal infection and schizophrenia: a review of epidemiologic and translational studies. Am J Psychiatry. 2010;167:261–80. doi: 10.1176/appi.ajp.2009.09030361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Brown AS, Patterson PH. Maternal infection and schizophrenia: implications for prevention. Schizophrenia Bull. 2011;37:284–90. doi: 10.1093/schbul/sbq146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Sacks G, Sargent I, Redman C. An innate view of human pregnancy. Immunol Today. 1999;20:114–8. doi: 10.1016/s0167-5699(98)01393-0. [DOI] [PubMed] [Google Scholar]
- 104.Baines M, Millar K, Mills P. Studies of complement levels in normal human pregnancy. Obstet Gynecol. 1974;43:806–10. [PubMed] [Google Scholar]
- 105.Hopkinson N, Powell R. Classical complement activation induced by pregnancy: implications for management of connective tissue diseases. J Clin Pathol. 1992;45:66–7. doi: 10.1136/jcp.45.1.66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Comeglio P, Fedi S, Liotta AA, Cellai AP, Chiarantini E, Prisco D, et al. Blood clotting activation during normal pregnancy. Thrombosis Res. 1996;84:199–202. doi: 10.1016/0049-3848(96)00176-4. [DOI] [PubMed] [Google Scholar]
- 107.Girardi G. Complement activation, a threat to pregnancy. Semin Immunopathol. 2018;40:103–11. doi: 10.1007/s00281-017-0645-x. [DOI] [PubMed] [Google Scholar]
- 108.Cannon M, Clarke MC, Cotter DR. Priming the brain for psychosis: maternal inflammation during fetal development and the risk of later psychiatric disorder. Am J Psychiatry. 2014;171:901–5. doi: 10.1176/appi.ajp.2014.14060749. [DOI] [PubMed] [Google Scholar]
- 109.Boulanger-Bertolus J, Pancaro C, Mashour GA. Increasing role of maternal immune activation in neurodevelopmental disorders. Front Behav Neurosci. 2018;12:230. doi: 10.3389/fnbeh.2018.00230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Minakova E, Warner BB. Maternal immune activation, central nervous system development and behavioral phenotypes. Birth Defects Res. 2018;110:1539–50. doi: 10.1002/bdr2.1416. [DOI] [PubMed] [Google Scholar]
- 111.Momtazmanesh S, Zare-Shahabadi A, Rezaei N. Cytokine alterations in schizophrenia: an updated review. Front Psychiatry. 2019;10:892. doi: 10.3389/fpsyt.2019.00892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Baumeister D, Akhtar R, Ciufolini S, Pariante CM, Mondelli V. Childhood trauma and adulthood inflammation: a meta-analysis of peripheral C-reactive protein, interleukin-6 and tumour necrosis factor-alpha. Mol Psychiatry. 2016;21:642–9. doi: 10.1038/mp.2015.67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Rasmussen LJH, Moffitt TE, Arseneault L, Danese A, Eugen-Olsen J, Fisher HL, et al. Association of adverse experiences and exposure to violence in childhood and adolescence with inflammatory burden in young people. JAMA Pediatr. 2020;174:38–47. [DOI] [PMC free article] [PubMed]
- 114.Borges S, Gayer-Anderson C, Mondelli V. A systematic review of the activity of the hypothalamic-pituitary-adrenal axis in first episode psychosis. Psychoneuroendocrinology. 2013;38:603–11. doi: 10.1016/j.psyneuen.2012.12.025. [DOI] [PubMed] [Google Scholar]
- 115.Liu Y-Z, Wang Y-X, Jiang C-L. Inflammation: the common pathway of stress-related diseases. Front Hum Neurosci. 2017;11:316. doi: 10.3389/fnhum.2017.00316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Rohleder N. Stimulation of systemic low-grade inflammation by psychosocial stress. Psychosom Med. 2014;76:181–9. doi: 10.1097/PSY.0000000000000049. [DOI] [PubMed] [Google Scholar]
- 117.Calcia MA, Bonsall DR, Bloomfield PS, Selvaraj S, Barichello T, Howes OD. Stress and neuroinflammation: a systematic review of the effects of stress on microglia and the implications for mental illness. Psychopharmacology. 2016;233:1637–50. doi: 10.1007/s00213-016-4218-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Yolken RH, Dickerson FB, Fuller Torrey E. Toxoplasma and schizophrenia. Parasite Immunol. 2009;31:706–15. doi: 10.1111/j.1365-3024.2009.01131.x. [DOI] [PubMed] [Google Scholar]
- 119.Lachmann PJ. The amplification loop of the complement pathways. Adv Immunol. 2009;104:115–49. doi: 10.1016/S0065-2776(08)04004-2. [DOI] [PubMed] [Google Scholar]
- 120.Sjöholm AG, Jönsson G, Braconier JH, Sturfelt G, Truedsson L. Complement deficiency and disease: an update. Mol Immunol. 2006;43:78–85. doi: 10.1016/j.molimm.2005.06.025. [DOI] [PubMed] [Google Scholar]
- 121.Chiveri L, Sciacco M, Prelle A. Schizophreniform disorder with cerebrospinal fluid PCR positivity for herpes simplex virus type 1. Eur Neurol. 2003;50:182–3. doi: 10.1159/000073062. [DOI] [PubMed] [Google Scholar]
- 122.Oommen KJ, Johnson PC, Ray CG. Herpes simplex type 2 virus encephalitis presenting as psychosis. Am J Med. 1982;73:445–8. doi: 10.1016/0002-9343(82)90751-3. [DOI] [PubMed] [Google Scholar]
- 123.Hiroshi H, Seiji K, Toshihiro K, Nobuo K. [An adult case suspected of recurrent measles encephalitis with psychiatric symptoms] Seishin Shinkeigaku Zasshi. 2003;105:1239–46. [PubMed] [Google Scholar]
- 124.Munjal S, Ferrando SJ, Freyberg Z. Neuropsychiatric aspects of infectious diseases: an update. Crit Care Clin. 2017;33:681–712. doi: 10.1016/j.ccc.2017.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Bohmwald K, Gálvez NMS, Ríos M, Kalergis AM. Neurologic alterations due to respiratory virus infections. Front Cell Neurosci. 2018;12:386. doi: 10.3389/fncel.2018.00386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Rogers JP, Chesney E, Oliver D, Pollak TA, McGuire P, Fusar-Poli P, et al. Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. Lancet Psychiatry. 2020;7:611–27. doi: 10.1016/S2215-0366(20)30203-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Ferrando SJ, Klepacz L, Lynch S, Tavakkoli M, Dornbush R, Baharani R, et al. COVID-19 psychosis: a potential new neuropsychiatric condition triggered by novel coronavirus infection and the inflammatory response? Psychosomatics. 2020;S0033–3182:30151. doi: 10.1016/j.psym.2020.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Jasti M, Nalleballe K, Dandu V, Onteddu S. A review of pathophysiology and neuropsychiatric manifestations of COVID-19. J Neurol. 2021;268:2007–12. [DOI] [PMC free article] [PubMed]
- 129.van Os J, Kenis G, Rutten BP. The environment and schizophrenia. Nature. 2010;468:203–12. doi: 10.1038/nature09563. [DOI] [PubMed] [Google Scholar]
- 130.Dickerson F, Jones-Brando L, Ford G, Genovese G, Stallings C, Origoni A, et al. Schizophrenia is associated with an aberrant immune response to Epstein-Barr virus. Schizophrenia Bull. 2019;45:1112–9. doi: 10.1093/schbul/sby164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Berger M, Burkhardt E, Yung A, Nelson B, Francey S, Lin A, et al. M22. IGG antibodies to toxoplasma gondii are associated with increased long-term risk for psychosis in individuals at ultra-high risk for psychosis. Schizophrenia Bull. 2020;46:S141–2. [Google Scholar]
- 132.Sommer IE, van Westrhenen R, Begemann MJH, de Witte LD, Leucht S, Kahn RS. Efficacy of anti-inflammatory agents to improve symptoms in patients with schizophrenia: an update. Schizophrenia Bull. 2013;40:181–91. doi: 10.1093/schbul/sbt139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Xiang YQ, Zheng W, Wang SB, Yang XH, Cai DB, Ng CH, et al. Adjunctive minocycline for schizophrenia: a meta-analysis of randomized controlled trials. Eur Neuropsychopharmacol. 2017;27:8–18. doi: 10.1016/j.euroneuro.2016.11.012. [DOI] [PubMed] [Google Scholar]
- 134.Amminger GP, Schafer MR, Papageorgiou K, Klier CM, Cotton SM, Harrigan SM, et al. Long-chain omega-3 fatty acids for indicated prevention of psychotic disorders: a randomized, placebo-controlled trial. Arch Gen Psychiatry. 2010;67:146–54. doi: 10.1001/archgenpsychiatry.2009.192. [DOI] [PubMed] [Google Scholar]
- 135.Ricklin D, Mastellos DC, Reis ES, Lambris JD. The renaissance of complement therapeutics. Nat Rev Nephrol. 2018;14:26–47. doi: 10.1038/nrneph.2017.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Carpanini SM, Torvell M, Morgan BP. Therapeutic inhibition of the complement system in diseases of the central nervous system. Front Immunol. 2019;10:362. [DOI] [PMC free article] [PubMed]
- 137.Hoirisch-Clapauch S, Nardi AE. Psychiatric remission with warfarin: Should psychosis be addressed as plasminogen activator imbalance? Med Hypotheses. 2013;80:137–41. doi: 10.1016/j.mehy.2012.11.011. [DOI] [PubMed] [Google Scholar]
- 138.Bauer KA. Pros and cons of new oral anticoagulants. Hematol Am Soc Hematol Educ Program. 2013;2013:464–70. doi: 10.1182/asheducation-2013.1.464. [DOI] [PubMed] [Google Scholar]
- 139.Ryu JK, Rafalski VA, Meyer-Franke A, Adams RA, Poda SB, Rios Coronado PE, et al. Fibrin-targeting immunotherapy protects against neuroinflammation and neurodegeneration. Nat Immunol. 2018;19:1212–3. doi: 10.1038/s41590-018-0232-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Lee JD, Coulthard LG, Woodruff TM. Complement dysregulation in the central nervous system during development and disease. Semin Immunol. 2019;45:101340. doi: 10.1016/j.smim.2019.101340. [DOI] [PubMed] [Google Scholar]
- 141.Agarwal V, Talens S, Grandits AM, Blom AM. A novel interaction between complement inhibitor C4b-binding protein and plasminogen that enhances plasminogen activation. J Biol Chem. 2015;290:18333–42. doi: 10.1074/jbc.M114.619494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Trouw LA, Nielsen HM, Minthon L, Londos E, Landberg G, Veerhuis R, et al. C4b-binding protein in Alzheimer’s disease: binding to Abeta1-42 and to dead cells. Mol Immunol. 2008;45:3649–60. doi: 10.1016/j.molimm.2008.04.025. [DOI] [PubMed] [Google Scholar]
- 143.Herberth M, Rahmoune H, Schwarz E, Koethe D, Harris LW, Kranaster L, et al. Identification of a molecular profile associated with immune status in first-onset schizophrenia patients. Clin Schizophr Relat Psychoses. 2014;7:207–15. [PubMed] [Google Scholar]
- 144.Cooper JD, Ozcan S, Gardner RM, Rustogi N, Wicks S, van Rees GF, et al. Schizophrenia-risk and urban birth are associated with proteomic changes in neonatal dried blood spots. Transl Psychiatry. 2017;7:1290. doi: 10.1038/s41398-017-0027-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Walss-Bass C, Lokesh GLR, Dyukova E, Gorenstein DG, Roberts DL, Velligan D, et al. X-aptamer technology identifies C4A and ApoB in blood as potential markers for schizophrenia. Mol Neuropsychiatry. 2019;5:52–9. doi: 10.1159/000492331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Ramsey JM, Schwarz E, Guest PC, van Beveren NJ, Leweke FM, Rothermundt M, et al. Distinct molecular phenotypes in male and female schizophrenia patients. PLoS ONE. 2013;8:e78729. doi: 10.1371/journal.pone.0078729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Moriyama Y, Kotani T, Ushida T, Imai K, Nakano T, Tsuda H, et al. Altered proteomic profile in umbilical arterial serum from mothers with schizophrenia. Schizophrenia Res. 2018;197:615–7. doi: 10.1016/j.schres.2018.02.024. [DOI] [PubMed] [Google Scholar]
- 148.Jiang J, Peng C, Sun L, Li J, Qing Y, Hu X, et al. Leukocyte proteomic profiling in first-episode schizophrenia patients: does oxidative stress play central roles in the pathophysiology network of schizophrenia? Antioxid Redox Signal. 2019;31:579–88. doi: 10.1089/ars.2019.7805. [DOI] [PubMed] [Google Scholar]
- 149.Gupta AK, Pokhriyal R, Khan MI, Kumar DR, Gupta R, Chadda RK, et al. Cerebrospinal fluid proteomics for identification of alpha2-macroglobulin as a potential biomarker to monitor pharmacological therapeutic efficacy in dopamine dictated disease states of Parkinson’s disease and schizophrenia. Neuropsychiatr Dis Treat. 2019;15:2853–67. doi: 10.2147/NDT.S214217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Velasquez E, Martins-de-Souza D, Velasquez I, Carneiro GRA, Schmitt A, Falkai P, et al. Quantitative subcellular proteomics of the orbitofrontal cortex of schizophrenia patients. J Proteome Res. 2019;18:4240–53. doi: 10.1021/acs.jproteome.9b00398. [DOI] [PubMed] [Google Scholar]
- 151.Yang Y, Chen J, Liu C, Fang L, Liu Z, Guo J, et al. The extrinsic coagulation pathway: a biomarker for suicidal behavior in major depressive disorder. Sci Rep. 2016;6:32882. doi: 10.1038/srep32882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Turck CW, Guest PC, Maccarrone G, Ising M, Kloiber S, Lucae S, et al. Proteomic differences in blood plasma associated with antidepressant treatment response. Front Mol Neurosci. 2017;10:272. doi: 10.3389/fnmol.2017.00272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Stelzhammer V, Haenisch F, Chan MK, Cooper JD, Steiner J, Steeb H, et al. Proteomic changes in serum of first onset, antidepressant drug-naive major depression patients. Int J Neuropsychopharmacol. 2014;17:1599–608. doi: 10.1017/S1461145714000819. [DOI] [PubMed] [Google Scholar]
- 154.Gui SW, Liu YY, Zhong XG, Liu X, Zheng P, Pu JC, et al. Plasma disturbance of phospholipid metabolism in major depressive disorder by integration of proteomics and metabolomics. Neuropsychiatr Dis Treat. 2018;14:1451–61. doi: 10.2147/NDT.S164134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.de Jesus JR, Galazzi RM, de Lima TB, Banzato CEM, de Almeida Lima ESLF, de Rosalmeida Dantas C, et al. Simplifying the human serum proteome for discriminating patients with bipolar disorder of other psychiatry conditions. Clin Biochem. 2017;50:1118–25. doi: 10.1016/j.clinbiochem.2017.06.009. [DOI] [PubMed] [Google Scholar]
- 156.Haenisch F, Alsaif M, Guest PC, Rahmoune H, Dickerson F, Yolken R, et al. Multiplex immunoassay analysis of plasma shows prominent upregulation of growth factor activity pathways linked to GSK3beta signaling in bipolar patients. J Affect Disord. 2014;156:139–43. doi: 10.1016/j.jad.2013.12.008. [DOI] [PubMed] [Google Scholar]
- 157.Ghebrehiwet B, Silverberg M, Kaplan AP. Activation of the classical pathway of complement by Hageman factor fragment. J Exp Med. 1981;153:665–76. doi: 10.1084/jem.153.3.665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Peerschke E, Reid K, Ghebrehiwet B. Platelet activation by C1q results in the induction of alpha IIb/beta 3 integrins (GPIIb-IIIa) and the expression of P-selectin and procoagulant activity. J Exp Med. 1993;178:579–87. doi: 10.1084/jem.178.2.579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Rehmus E, Greene B, Everson B, Ratnoff O. Inhibition of the activation of Hageman factor (factor XII) by complement subcomponent C1q. J Clin Investig. 1987;80:516–21. doi: 10.1172/JCI113100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Amara U, Flierl MA, Rittirsch D, Klos A, Chen H, Acker B, et al. Molecular intercommunication between the complement and coagulation systems. J Immunol. 2010;185:5628. doi: 10.4049/jimmunol.0903678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Polley M, Nachman R. Human platelet activation by C3a and C3a des-arg. J Exp Med. 1983;158:603–15. doi: 10.1084/jem.158.2.603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Ikeda K, Nagasawa K, Horiuchi T, Tsuru T, Nishizaka H, Niho Y. C5a induces tissue factor activity on endothelial cells. Thrombosis Haemost. 1997;77:394–8. [PubMed] [Google Scholar]
- 163.Wojta J, Kaun C, Zorn G, Ghannadan M, Hauswirth AW, Sperr WR, et al. C5a stimulates production of plasminogen activator inhibitor-1 in human mast cells and basophils. Blood. 2002;100:517–23. doi: 10.1182/blood.v100.2.517. [DOI] [PubMed] [Google Scholar]
- 164.Reinartz J, Hänsch GM, Kramer MD. Complement component C7 is a plasminogen-binding protein. J Immunol. 1995;154:844–50. [PubMed] [Google Scholar]
- 165.van de Poel RH, Meijers JC, Bouma BN. The interaction between anticoagulant protein S and complement regulatory C4b-binding protein (C4BP) Trends Cardiovasc Med. 2000;10:71–6. doi: 10.1016/s1050-1738(00)00051-7. [DOI] [PubMed] [Google Scholar]
- 166.Cicardi M, Zingale L, Zanichelli A, Pappalardo E, Cicardi B. C1 inhibitor: molecular and clinical aspects. Springer Semin Immunopathol. 2005;27:286–298. [DOI] [PubMed]
- 167.Bergamaschini L, Miedico A, Cicardi M, Coppola R, Faioni EN, Agostoni A. Consumption of C4b-binding protein (C4BP) during in vivo activation of the classical complement pathway. Clin Exp Immunol. 1999;116:220–4. doi: 10.1046/j.1365-2249.1999.00874.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Rehman AA, Ahsan H, Khan FH. α-2-Macroglobulin: a physiological guardian. J Cell Physiol. 2013;228:1665–75. doi: 10.1002/jcp.24266. [DOI] [PubMed] [Google Scholar]
- 169.Downing MR, Bloom JW, Mann KG. Comparison of the inhibition of thrombin by three plasma protease inhibitors. Biochemistry. 1978;17:2649–53. doi: 10.1021/bi00606a030. [DOI] [PubMed] [Google Scholar]
- 170.Sottrup-Jensen L, Stepanik TM, Kristensen T, Lønblad PB, Jones CM, Wierzbicki DM, et al. Common evolutionary origin of alpha 2-macroglobulin and complement components C3 and C4. Proc Natl Acad Sci USA. 1985;82:9–13. doi: 10.1073/pnas.82.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Polley MJ, Nachman RL. Human complement in thrombin-mediated platelet function: uptake of the C5b-9 complex. J Exp Med. 1979;150:633–45. doi: 10.1084/jem.150.3.633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Sims PJ, Faioni E, Wiedmer T, Shattil S. Complement proteins C5b-9 cause release of membrane vesicles from the platelet surface that are enriched in the membrane receptor for coagulation factor Va and express prothrombinase activity. J Biol Chem. 1988;263:18205–12. [PubMed] [Google Scholar]
- 173.Wiedmer T, Esmon C, Sims P. On the mechanism by which complement proteins C5b-9 increase platelet prothrombinase activity. J Biol Chem. 1986;261:14587–92. [PubMed] [Google Scholar]
- 174.Heurich M, Preston RJ, O’Donnell VB, Morgan BP, Collins PW. Thrombomodulin enhances complement regulation through strong affinity interactions with factor H and C3b-Factor H complex. Thrombosis Res. 2016;145:84–92. doi: 10.1016/j.thromres.2016.07.017. [DOI] [PubMed] [Google Scholar]
- 175.Delvaeye M, Noris M, De Vriese A, Esmon CT, Esmon NL, Ferrell G, et al. Thrombomodulin mutations in atypical hemolytic-uremic syndrome. N Engl J Med. 2009;361:345–57. doi: 10.1056/NEJMoa0810739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Rayes J, Roumenina LT, Dimitrov JD, Repessé Y, Ing M, Christophe O, et al. The interaction between factor H and VWF increases factor H cofactor activity and regulates VWF prothrombotic status. Blood. 2014;123:121–5. doi: 10.1182/blood-2013-04-495853. [DOI] [PubMed] [Google Scholar]
- 177.Tschopp J, Chonn A, Hertig S, French L. Clusterin, the human apolipoprotein and complement inhibitor, binds to complement C7, C8 beta, and the b domain of C9. J Immunol. 1993;151:2159–65. [PubMed] [Google Scholar]
- 178.de Boer HC, de Groot PG, Bouma BN, Preissner K. Ternary vitronectin-thrombin-antithrombin III complexes in human plasma. Detection and mode of association. J Biol Chem. 1993;268:1279–83. [PubMed] [Google Scholar]
- 179.Declerck P, De Mol M, Alessi M, Baudner S, Paques E, Preissner K, et al. Purification and characterization of a plasminogen activator inhibitor 1 binding protein from human plasma. Identification as a multimeric form of S protein (vitronectin) J Biol Chem. 1988;263:15454–61. [PubMed] [Google Scholar]
- 180.Podack ER, Kolb WP, Müller-Eberhard HJ. The SC5b-7 complex: formation, isolation, properties, and subunit composition. J Immunol. 1977;119:2024–9. [PubMed] [Google Scholar]
- 181.Dahlback B. Inhibition of protein Ca cofactor function of human and bovine protein S by C4b-binding protein. J Biol Chem. 1986;261:12022–7. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.