Abstract
Honeysuckle has been used in the treatment of influenza virus infection for thousands of years in China. However, its main active components and the functional mechanisms remain to be elucidated. Here, four honeysuckle extracts, including acids extract, flavonoids extract, total extract and acids-flavonoids mixture, were prepared to clarify the main active antiviral components. The cytopathic effect reduction assay showed that all the four extracts inhibited the replication of influenza viruses H1N1, H3N2 and the oseltamivir-resistant mutant strain H1N1-H275Y. The acids-flavonoids mixture had the strongest inhibitory effects in vitro with EC50 values of 3.8, 4.1, and > 20 μg/mL against H1N1, H3N2 and H1N1-H275Y, respectively, showing competitive antiviral activity with oseltamivir and ribavirin. Honeysuckle acids extract also showed the most significant antiviral activity in vivo. Oral administration of the acids extract at a dosage of 600 mg/kg/d effectively alleviated viral pneumonia, maintained body weight and improved the survival rate to 30% of the mice infected with a lethal dose of H1N1. The results of time-of-drug addition experiment and neuraminidase (NA) inhibition assay showed that honeysuckle extracts had a broad-spectrum inhibitory effect against influenza virus NAs. The flavonoid extract showed the strongest inhibitory effect on the NA of influenza virus H7N9 with an IC50 of 24.7 μg/mL. These results suggested that these extracts might exert their antiviral activity by suppressing the release of influenza viruses. Briefly, our findings demonstrate that acids and flavonoids extracts of honeysuckle are the major antiviral active components, and the acids extract has the potential to be developed into an antiviral agent against influenza virus, especially for oseltamivir-resistant viruses.
Keywords: Honeysuckle extract, Antiviral activity, Influenza A virus (IAV), Oseltamivir-resistant
Introduction
Influenza A virus is a considerable threat to human health and life, and can cause seasonal and widespread epidemics or pandemics with high levels of morbidity and mortality (Taubenberger and Kash 2010; Zhang et al. 2012). The circulating strains of seasonal Influenza viruses A/FM/1/47 (H1N1) and A/Beijing/32/92 (H3N2) are the primary causes of pandemic infections. In addition, the highly pathogenic avian A/Vietnam/1203/2004 (H5N1) virus and the recently reported A/Anhui/1/2013 (H7N9) virus have shown potential to cause severe influenza pandemics, with estimated mortality rates of 60% and 30%, respectively (Chen et al. 2004; Gao et al. 2013; Zhang et al. 2013). Vaccination may be helpful in controlling influenza infection. However, vaccine shortcomings, such as time lag, limited efficacy, side effects and storage difficulty, have restricted their use (Levine and Sztein 2004; Cinatl et al. 2007; Hampson 2008; Rohrs et al. 2014). Antiviral drugs are the most effective option for treating influenza infection.
Currently, there are three classes of anti-influenza virus drugs with different antiviral mechanisms, including M2 ion channel blockers (amantadine and rimantadine), viral RNA synthesis inhibitors (ribavirin and T-705) and neuraminidase (NA) inhibitors (oseltamivir and zanamivir) (Hay et al. 1985; De Clercq 2006; Furuta et al. 2013). However, because of the frequent emergence of drug-resistant variants, M2 ion channel blockers are no longer used in clinic (Hayden 2006; Ison 2011; Lee and Yen 2012). In addition, ribavirin causes toxic side effects and has a limited therapeutic effect. More importantly, NA inhibitor-resistant influenza viruses have emerged (Thorlund et al. 2011; Leang et al. 2013), limiting the future utility of NA inhibitors.
Traditional Chinese medicines have been regarded as sources of therapeutic agents with a long history (Koehn and Carter 2005; Cai et al. 2015), and have many advantages, such as multiple target effects, abundant availability and few toxic side effects. Honeysuckle (HS, Lonicera japonica) is the dry bud or early flower of Lonicera japonica, a well-known Chinese herbal medicine that has been used for the effective treatment of influenza virus infection for thousands of years (Zhou et al. 2015). To clarify the main antiviral components of honeysuckle and the underlying mechanisms of their action, we assessed the inhibitory activity of the total extract, organic acids extract, flavonoids extract and acids-flavonoids mixture of honeysuckle against IAVs (H1N1 and H3N2) in vitro and in vivo. We also investigated their inhibitory effects of these honeysuckle compounds on the oseltamivir-resistant strain H1N1-H275Y. Moreover, we studied the inhibitory effect of these extracts at different stages of the influenza virus life cycle and sought to determine their potential targets. Our findings demonstrate the potential utility of honeysuckle acids extract as an alternative antiviral agent for attenuating influenza virus, especially the oseltamivir-resistant mutant strain.
Materials and Methods
Cells and Virus
Madin-Darby canine kidney cells (MDCK) and human cervical cancer cells (HeLa) were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA). MDCK and HeLa cells were cultured in Dulbecco’s Modified Eagle’s medium (DMEM) and RPMI-1640 medium supplemented with 10% (V/V) fetal bovine serum (FBS) respectively, and were maintained in an atmosphere containing 5% CO2 at 37 °C.
Influenza viruses A/FM/1/47 (H1N1), A/Beijing/32/92 (H3N2), H1N1 oseltamivir-resistant mutant strain possessing a histidine-to-tyrosine substitution at position 275 (H275Y) of NA were conserved at the Department of Microbiology, School of Life Science and Technology, China Pharmaceutical University. Influenza A viruses were propagated in 10-day-old embryonated chicken eggs. All these viruses were stored at − 80 °C (Jung et al. 2010).
Reagents
Oseltamivir carboxylate (OC) were purchased from MedChemExpress (Shanghai, China). Ribavirin (RBV) was obtained from Sichuan Baili Pharmaceutical Co., Ltd. (Chengdu, China). Both of them were dissolved in double-distilled water to a 10 mg/mL concentration for use as stock solutions. 2′-(4-methyl-umbelliferyl)-α-D-N-acetylneuraminic acid sodium salt hydrate (MUNANA), 2-(N-morpholino) ethanesulfonic acid (MES) and 3-[4,5-dimethyl-thiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma-Aldrich Co., St. (Louis, MO, USA). DMEM, RPMI-1640 and FBS were purchased from Gibco Co. (NY, USA).
Sample Preparations
Honeysuckle was provided by Anhui Qimen Huangshan Yaogu Science and Technology Co., Ltd. The four honeysuckle extracts were prepared at the Department of Pharmaceutics, China Pharmaceutical University.
The Honeysuckle Flavonoids Extract
100 g honeysuckle was added with 1200 mL 70% ethanol and then refluxed at 85 °C for 2 h. After filtration, 900 mL 70% ethanol was added to the residue and refluxed for 2 h. After the second filtration, the filtrates from the two refluxed-steps were combined and evaporated under reduced pressure. The obtained extractum (150 mL) was mixed with petroleum ether (3–4 times the volume of obtained extractum), and the petroleum ether layer was discarded. The resultant concentrated solution was mixed with ethyl acetate (3–4 times the volume of the concentrated solution) and the ethyl acetate layer was collected. The ethyl acetate extraction step repeated twice. The ethyl acetate layers were combined and evaporated to dryness under reduced pressure.
The Honeysuckle Acids Extract
The water layer from the initial flavonoids extraction step was adjusted to pH 2 with 1 mol/L dilute hydrochloric acid. The acids extract was obtained through precipitation with ethyl acetate (4 times volume) twice. Then the extract was mixed and evaporated to dryness under reduced pressure.
The Honeysuckle Acids-Flavonoids Combination
The acids-flavonoids mixture was prepared by mixing 0.85 g acids extract and 0.75 g flavonoids extract.
The Total Extract Liquid of Honeysuckle
Crushed honeysuckle was mixed with 10 times volume of 60% ethanol and refluxed in water bath at 40 °C for 1.5 h. After filtration, the filtrate was collected. The same procedure was repeated twice with the remaining residue. The overall filtrates were mixed and kept at 4 °C for 24 h. The filtrates were evaporated under reduced pressure, and then dissolved in distilled water and lyophilized to get purified total extract.
After conversion, 1 g flavonoid fraction is equivalent to 33.3 g honeysuckle; 1 g organic acid fraction is equivalent to 28.6 g honeysuckle; 1 g flavonoid-acid mixture is equivalent to 30.8 g honeysuckle; 1 g total extract is equivalent to 3.3 g honeysuckle.
Each honeysuckle extract was dissolved in dimethyl sulfoxide (DMSO) or in 0.5% CMC-Na solution to a 100 mg/mL concentration as stock solution for in vitro or in vivo studies, respectively.
Cytotoxic Activity of Honeysuckle Extracts on MDCK or HeLa Cells
MDCK or HeLa cells (5 × 103 cells/well) were cultured in 96-well plates for 24 h. Then, the medium was replaced with the maintenance medium (DMEM/RPMI-1640 with 1% FBS) containing honeysuckle extracts at final concentrations of 0, 25, 50, 100, 200 and 400 μg/mL. After incubation for 48 h, the cell viability was measured by MTT assays as described previously (Tian et al. 2011). In brief, 10 μL of MTT solution (5 mg/mL) was added to each well. After incubation at 37 °C for 4 h, the MTT solution was replaced with DMSO (150 μL/well). The plate was shaken gently for 10 min to dissolve the precipitate of formazan. Absorbance at 490 nm was determined with a microplate reader. The cell viability was calculated using the following equation: cell viability (%) = (mean optical density of test—mean optical density of background)/(mean optical density of cell controls—mean optical density of background) × 100. The half maximal cytotoxic concentration (CC50) was calculated by regression analysis in GraphPad Prism 5.
TCID50 Determination of IAV
MDCK or HeLa cells (5 × 103 cells/well) were grown in 96-well plates for 24 h. The virus stock was serial two fold diluted by serum-free medium. Then, 100 μL of the serial virus dilution was added to each well at 37 °C for 2 h. The cells were incubated in the maintenance medium at 37 °C for 48 h. The cell viability was measured by MTT assay. The TCID50 (50% tissue culture infectious dose) was determined using the Reed-Müench method.
Cytopathic Effect (CPE) Reduction Assay
MDCK or HeLa cells (5 × 103 cells/well) were cultured in 96-well plates for 24 h and incubated with 100 TCID50 of influenza H1N1, H3N2 and H1N1-H275Y viruses at 37 °C for 2 h. Then, the cells were incubated with the maintenance medium in the presence of various concentrations of honeysuckle extracts (1, 2.5, 5, 10, and 20 μg/mL). 2 μg/mL oseltamivir and ribavirin were used as positive controls and 0.5% CMC-Na solution was used as placebo. After 48 h incubation, the CPE was observed with microscopy, and the viability of cells was measured by MTT assay. The inhibition rate of the test drugs was calculated using the following equation (Ding et al. 2014): inhibition rate (%) = (mean optical density of test—mean optical density of virus controls) / (mean optical density of cell controls—mean optical density of virus controls) × 100. The cell control group was MDCK cells treated with DMSO, and the virus control group was MDCK cells only infected with virus (the placebo group). The EC50 values were determined using regression analysis, and the selectivity index (SI) was defined as the ratio of CC50 to EC50.
Inhibitory Efficacy of Honeysuckle Extracts on Different Stages of Viral Life Cycle
To investigate the inhibitory effect of honeysuckle at different stages of the influenza virus life cycle, a time-of-drug-addition experiment was carried out as previously described (Yu et al. 2016). In the first protocol (pre-infection), MDCK cells were pre-incubated with several concentrations of honeysuckle extracts (1, 2.5, 5, 10, and 20 μg/mL) for 12 h before virus infection. In the second protocol (co-infection), virus and honeysuckle extracts were added to MDCK cells simultaneously and incubated for 2 h. In both protocols, the cells were incubated with the maintenance medium for 48 h after virus adsorption. In the third protocol (post-infection), after infected with the influenza virus for 2 h, the maintenance medium containing honeysuckle extracts was added to MDCK cells. Cell viability was determined by MTT assay.
Therapeutic Efficacy Study in Mice
The 18–22 g six-week-old male ICR mice were obtained from Nantong University (Nantong, Jiangsu, China) and the experiment was performed as previously described (Ding et al. 2014). Ninety-eight mice were divided into seven groups: normal control group (uninfected), placebo group (infected mice with saline), four honeysuckle extracts-treated groups (600 mg/kg/d), and the ribavirin-treated group (positive control, 100 mg/kg/d). Mice were inoculated intranasally with 8 × LD50 (50% mice lethal dose) mouse-adapted A/FM/1/47 (H1N1) virus suspended in 50 μL normal saline. After infection for 2 h, honeysuckle extracts, ribavirin or saline (placebo) were administered once daily for 5 days by oral gavage. Survival and weight were observed daily for 14 days. On day 5 and day 6 after infection, four mice per group were randomly selected and euthanized for lung pathological examinations. The lung index was calculated using the equation: Lung index = lung weight/body weight × 100. The lung injury score was evaluated according to the degree of congestion and edema of the lungs, the degree of bronchial necrosis, the degree of alveolar and bronchial epithelium injury, and the degree of inflammatory infiltration by the professional.
qRT-PCR Analysis
The viral load in the lung tissues was determined by quantitative real time RT-PCR (qRT-PCR). RNA was extracted from the lungs by homogenizing with Trizol (Invitrogen, USA) reagent, and subsequently reverse transcribes to cDNA. qRT-PCR was performed on an ABI 7500 Real-Time PCR System (ABI, USA). The GAPDH was used as endogenous control. The primer for H1N1 (FM1) detection: Forward: 5′’-GAGAAAGAAGTCCTTGTGC-3′ and Reverse: 5′-TCTATCATTCCAGTCCATCCC-3′.
Hematoxylin and Eosin Staining
The lungs of mice from each group were fixed in 4% paraformaldehyde and embedded in paraffin. Then, sections (5 μm) were stained with hematoxylin and eosin (HE). Histological examinations were performed using an Olympus microscope.
Neuraminidase (NA) Inhibition Assay
An NA inhibition assay was performed as previously described (Kim et al. 2013). Allantoic fluid containing the influenza viruses H1N1, H1N1-H275Y or H3N2 and the cell lysate of CHO expressing viral NAs, including A/Vietnam/1203/2004 (H5N1), A/Anhui/1/2013 (H7N9) and H7N9-R294K were used for the NA inhibition assay. First, the allantoic fluid or cell lysate was incubated with serial honeysuckle extracts (15.625, 31.25, 62.5, 125, 250 μg/mL) at 37 °C in a 96-well black OptiPlate for 5 min. Then, 20 μmol/L fluorescent substrate MUNANA was added and the plates were incubated at 37 °C for 30 min. Fluorescence was measured using an AFLx800 fluorescence microplate reader (Bio-Tek) at excitation and emission wavelengths of 360 and 450 nm respectively.
Statistical Analysis
All assays were performed in triplicate. Values in the texts and figures are presented as the mean ± SD. Data were analyzed statistically by an un-paired, two-tailed Student’s t-test. The significance was defined as P < 0.05 (*P < 0.05, **P < 0.01, ***P < 0.001).
Results
Antiviral Activity of Honeysuckle Extracts against H1N1 and H3N2 In Vitro
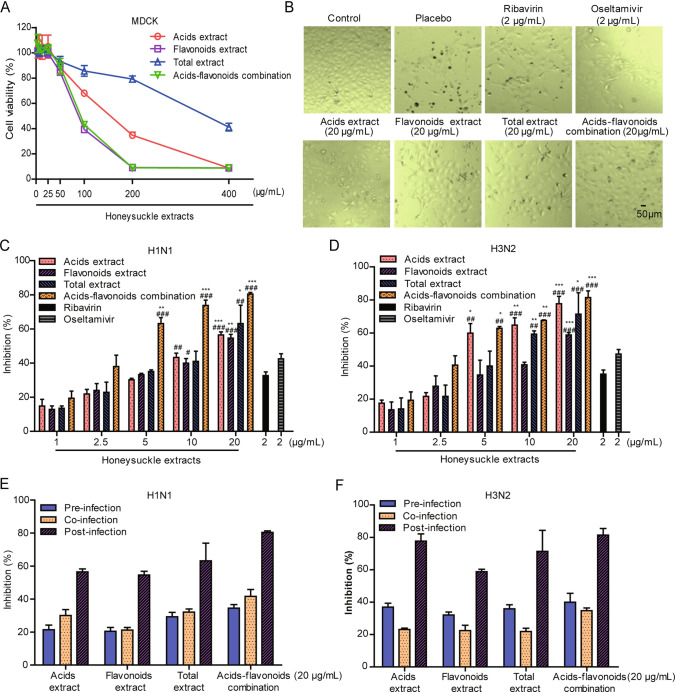
The cytotoxicity of the honeysuckle extracts in MDCK and Hela cells was examined. As shown in Fig. 1A, upon treatments with the four honeysuckle extracts at 25 μg/mL, cell viability remained at least 95% at 48 h post-infection, indicating that the treatments induced limited cytotoxicity at concentrations less of 25 μg/mL. In addition, the CC50 and EC50 values of the four honeysuckle extracts on the influenza viruses were determined (Table 1). The total extract induced the lowest cytotoxicity, with a CC50 value of 350.0 μg/mL in MDCK cells. The acids-flavonoids mixture showed the most effective antiviral activity against H1N1 and H3N2 with EC50 values of 3.8 μg/mL and 4.1 μg/mL, respectively.
Fig. 1.
Inhibitory effects of honeysuckle extracts on influenza viruses H1N1 and H3N2 in MDCK cells. A Cellular toxicity of honeysuckle extracts in MDCK cells. B Microscopic images for H1N1 virus-infected, 20 μg/mL honeysuckle extracts-treated, 2 μg/mL OC-treated, and 2 μg/mL ribavirin-treated MDCK cells at 48 h post-infection. C Inhibitory rates of honeysuckle extracts (1–20 μg/mL) against influenza virus A/FM/1/47 (H1N1), and D H3N2 in MDCK cells. 2 μg/mL oseltamivir carboxylate and ribavirin were used as positive controls. Inhibitory rates of honeysuckle extracts (20 μg/mL) against H1N1 (E) and H3N2 (F) influenza viruses with different infection protocols in MDCK cells. The results were presented as the mean ± S.D. of three independent experiments, *P < 0.05; **P < 0.01; ***P < 0.001, compared with the oseltamivir-treated group, #P < 0.05; ##P < 0.01; ###P < 0.001, compared with the ribavirin-treated group.
Table 1.
Cytotoxicity and antiviral activity of honeysuckle extracts.
| Compounds | CC50a | H1N1 | H3N2 | |||
|---|---|---|---|---|---|---|
| MDCK | HeLa | EC50b | SIc | EC50 | SI | |
| Acids extract | 139.8 ± 9.3 | 143.3 ± 6.0 | 15.6 ± 1.5 | 8.9 | 5.6 ± 0.6 | 24.9 |
| Flavonoids extract | 82.4 ± 0.8 | 76.9 ± 1.4 | 17.2 ± 3.8 | 4.8 | 14.1 ± 2.5 | 5.9 |
| Total extract | 350.0 ± 14.5 | 178.1 ± 2.7 | 14.3 ± 7.0 | 25.2 | 7.4 ± 1.0 | 48.6 |
| Acids-flavonoids | 88.3 ± 3.9 | 116.5 ± 2.9 | 3.8 ± 0.3 | 23.5 | 4.1 ± 0.5 | 21.8 |
| Oseltamivir | > 500 | 2.6 ± 0.1 | > 192 | 3.1 ± 0.1 | > 161 | |
| Ribavirin | 13.7 | 3.5 ± 0.1 | 3.9 | NDd | ND | |
The results were the mean ± S.D. of three independent experiments.
aConcentration of drugs required to reduce the viability of normal MDCK cells by 50% (μg/mL).
bConcentration of drugs required to improve the viability of influenza virus-infected cells by 50% (μg/mL).
cSelectivity index, CC50/EC50.
dNot determined.
To investigate whether these honeysuckle extracts can inhibit the replication of the H1N1 and H3N2 influenza viruses, a CPE reduction assay was performed in MDCK cells. The CPE reduction was confirmed by direct microscopic observation. Each honeysuckle treatment cell group had a considerably lower CPE than the placebo group (H1N1 infected cells treated with DMSO) (Fig. 1B). All four honeysuckle treatment protected MDCK cells from influenza virus infection in a dose-dependent manner while inducing low cytotoxicity (Fig. 1C, 1D). In addition, acids-flavonoids mixture exhibited stronger suppressive effects than the three extracts in MDCK cells, especially at a final concentration of 20 μg/mL, with inhibition rates of 80.4% and 81.4% against H1N1 and H3N2, respectively. At a concentration of 2.5 μg/mL, the acids-flavonoids mixture had an anti-influenza-virus activity effect against H1N1 and H3N2 similar to that of oseltamivir and ribavirin.
To clarify the possible anti-influenza mechanism of the four honeysuckle extracts treatments, their inhibitory effects on influenza virus replication were examined using three different infection protocols. As shown in Fig. 1E–1F, when 20 μg/mL honeysuckle extracts were added 2 h post-infection, the inhibition rates against H1N1 and H3N2 viruses were much higher than those observed after the ‘pre-infection’ or ‘co-infection’ treatment of infected cells, indicating that honeysuckle treatments mainly inhibited the replication and release of influenza virus but not viral adsorption or penetration.
Taken together, these results indicate that all four honeysuckle extracts treatments effectively inhibited the replication of H1N1 and H3N2 influenza viruses in MDCK cells, and the honeysuckle acids-flavonoids mixture had the strongest inhibitory effects in vitro.
Honeysuckle Extracts Effectively Inhibited the Replication of Oseltamivir-Resistant Mutant Influenza Virus H1N1-H275Y In Vitro
In addition to wild type influenza viruses, we also investigated the inhibitory effects of the honeysuckle extracts treatments on the oseltamivir-resistant mutant strain H1N1-H275Y in vitro. As shown in Fig. 2, four honeysuckle extracts obviously inhibited the replication of the influenza virus H1N1-H275Y in a dose-dependent manner in MDCK cells. The inhibition rate of the acids-flavonoids mixture against H1N1-H275Y virus was 47.8% at a concentration of 20 μg/mL, while the inhibition rates of oseltamivir and ribavirin were only 3.5% and 14.8%, respectively, showing that acids-flavonoids mixture can effectively inhibit the replication of the oseltamivir-resistant virus in vitro.
Fig. 2.
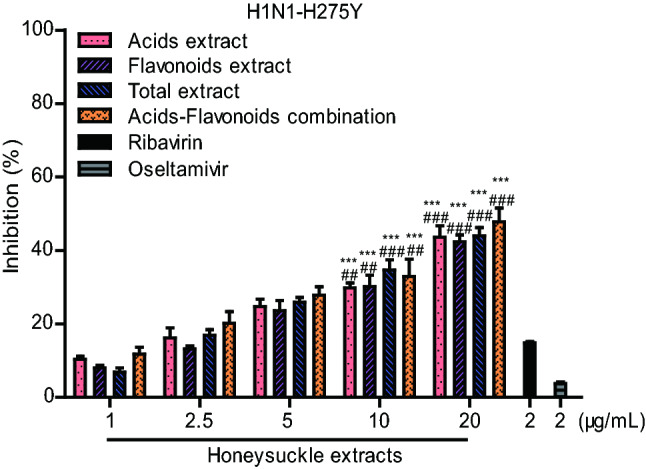
Inhibitory effects of honeysuckle extracts on oseltamivir-resistant mutant influenza virus H1N1-H275Y in MDCK cells. 2 μg/mL oseltamivir carboxylate and ribavirin were used as positive controls. The results were presented as the mean ± S.D. of three independent experiments, *P < 0.05; **P < 0.01; ***P < 0.001, compared with the oseltamivir-treated group, #P < 0.05; ##P < 0.01; ###P < 0.001, compared with the ribavirin-treated group.
Therapeutic Efficacy of Honeysuckle Extracts against H1N1 In Vivo
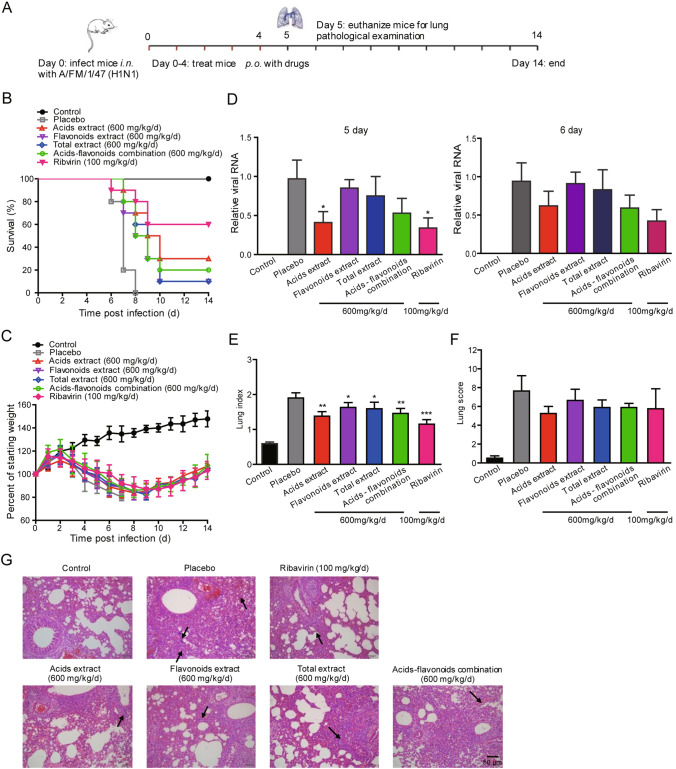
The inhibitory efficacy of four honeysuckle extracts on influenza A virus in vivo was evaluated in an H1N1-infected mouse model. The experimental protocol is shown in Fig. 3A. On ‘day 0’, the mice were infected intranasally with a lethal dose of H1N1 virus. At 2 h post infection, drugs were delivered orally to mice once daily for 5 days (days 0–4).
Fig. 3.
Therapeutic efficacy of honeysuckle extracts against the influenza virus in mice. A Experimental protocol of testing honeysuckle extracts in H1N1-infected mice. Fourteen uninfected ICR mice were as control group. ICR mice were intranasally infected with a mouse-adapted influenza virus H1N1 (8 × LD50). At 2 h post-infection, fourteen mice per group were treated with saline (placebo group), four honeysuckle extracts (600 mg/kg/d), or ribavirin (100 mg/kg/d) twice daily for 5 successive days (days 0–4) by oral gavage, respectively. On day 5 and day 6 post-infection, four mice per group were euthanized for lung pathological examinations. Survival rate (B) and body weight (C) were monitored for 14 days (n = 10). Viral load (D), Lung index (E) and Lung score (F) of mice infected with influenza virus H1N1 (n = 4). G H&E staining of sectioned lungs (n = 4). The histopathological changes are indicated by arrows. Data shown as the mean ± S.D. *P < 0.05; **P < 0.01; ***P < 0.001, compared with the placebo group.
As shown in Fig. 3B, acids extract-treated mice were obviously protected from death caused by influenza virus, whereas all the mice administered the placebo died within 8 days. Treatment of the mice with 600 mg/kg/d of honeysuckle acids extract, flavonoids extract, total extract or acids-flavonoids mixture increased survival to 30%, 10%, 10% and 20%, respectively. In addition, administration of the four honeysuckle extracts effectively protected the infected mice from body weight loss (Fig. 3C). Despite a similar trend in body weight loss during the first 9 days post-infection, the mice administered the honeysuckle extracts treatments regained weight starting on day 10, whereas infected mice treated with placebo showed significant body weight loss until their death.
On ‘day 5’ and ‘day 6’, we used qPCR to detect the viral load in the lungs of the treated mice. Acids extract and ribavirin treatments significantly inhibited viral RNA expression on day 5 but only slightly suppressed viral RNA expression on day 6 (Fig. 3D). Four mice per group were randomly selected and euthanized for use in lung pathological examinations on day 5. The four honeysuckle extracts treatments effectively suppressed the influenza-induced increases in lung indexes to a different degree (Fig. 3E). At a dosage of 600 mg/kg/d, the lung index of the honeysuckle acids extract-treated mice was 1.4, a statistically significant decrease compared to that of the placebo group (lung index: 1.9). The four honeysuckle extracts treatments administered at a dosage of 600 mg/kg/d protected the lungs of mice from damage caused by influenza virus infection. On day 5, features of extensive lung damage, such as alveolar wall congestion, infiltration of inflammatory cells and bronchial epithelium necrosis, were observed in the placebo group (lung score: 7.63) (Fig. 3F, 3G). In the honeysuckle acids extract-treated mice, the lung damage was notably attenuated (lung score: 5.25).
These data indicated that the honeysuckle acids extract significantly reduced body weight loss, inhibited lung damage and prevented the death of influenza-infected mice. The anti-influenza virus effects of the honeysuckle acids extract was slightly weaker than that of the acids-flavonoids mixture in vitro, but in vivo, and the pharmaco dynamic study demonstrated the potent therapeutic efficacy of the honeysuckle acids extract against infection with influenza virus H1N1.
Inhibitory Effects of Honeysuckle Extracts against the Neuraminidase Activity of Influenza Viruses
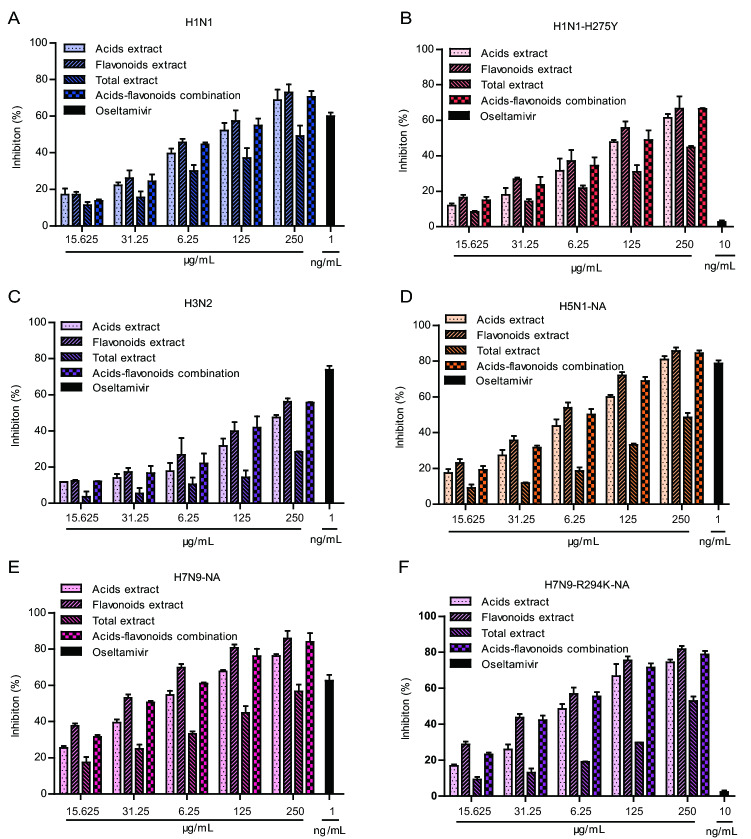
The results of the time-of-drug addition experiment indicated that honeysuckle extracts primarily inhibited the replication and release of the influenza virus after entrance into host cells (Fig. 1E, 1F). Influenza virus neuraminidase can hydrolyze sialic acid residues in progeny virus to promote the release of progeny virus particles from host cells, thus playing an important role in the spread of the influenza virus. Therefore, we speculated that honeysuckle extracts may inhibit the neuraminidase activity of influenza viruses. The inhibitory effects of the honeysuckle extracts treatments on the neuraminidase activity of H1N1, H1N1-H275Y, H3N2 influenza viruses, CHO-expressed H5N1-NA, CHO-expressed H7N9-NA and CHO-expressed H7N9-R294K-NA were examined using MUNANA, a specific fluorescent substrate. Figure 4 shows that the four honeysuckle extracts treatments significantly inhibited the neuraminidase activity of multi-influenza viruses in a dose-dependent manner. Among the four honeysuckle extracts treatments, the flavonoids extract showed the best anti-neuraminidase activity. Moreover, the four honeysuckle extracts treatments had a greater inhibitory effect against recombinant NA from the H7N9 influenza virus than against NA from the other influenza viruses. As shown in Table 2, the IC50 value of the honeysuckle flavonoids extract against NA of H7N9 influenza virus was 24.7 μg/mL. These results demonstrated that the honeysuckle extracts treatments had broad-spectrum inhibitory activity against NA in multiple influenza viruses, including the oseltamivir-resistant mutant strains.
Fig. 4.
Inhibition of neuraminidase activity by four honeysuckle extracts. Neuraminidase (NA) inhibition of four honeysuckle extracts (15.625–250 μg/mL) against the influenza viruses: (A) H1N1, (B) oseltamivir-resistant H1N1-H275Y, (C) H3N2, (D) CHO-expressed H5N1-NA, (E) CHO-expressed H7N9-NA and F CHO-expressed H7N9-R294K-NA. Oseltamivir carboxylate (1 nmol/L or 10 nmol/L) was used as a positive control. The results were shown as the mean ± S.D. of three independent experiments.
Table 2.
IC50 of honeysuckle extracts against neuraminidase activity of influenza A virus.
| NA | IC50a ± SD (μg/mL) | |||
|---|---|---|---|---|
| Acids extract | Flavonoids extract | Total extract | Acids-flavonoids | |
| H1N1 | 112.3 ± 17.7 | 90.9 ± 8.6 | 305.1 ± 23.7 | 100.1 ± 11.4 |
| H3N2 | 332.6 ± 34.5 | 196.0 ± 23.4 | 947.3 ± 39.5 | 203.8 ± 9.9 |
| H5N1 | 78.8 ± 5.5 | 54.7 ± 3.4 | 283.9 ± 29.0 | 63.8 ± 0.6 |
| H7N9 | 55.9 ± 5.1 | 24.7 ± 2.3 | 170.0 ± 5.2 | 35.2 ± 3.1 |
| H1N1-H275Y | 150.4 ± 13.6 | 108.4 ± 17.0 | 350.1 ± 36.9 | 125.7 ± 14.7 |
| H7N9-R294K | 80.1 ± 6.3 | 44.1 ± 5.4 | 291.2 ± 26.2 | 53.1 ± 6.0 |
The results were the mean ± S.D. of three independent experiments.
aConcentration of drugs required to reduce the activity of neuraminidase by 50%.
Discussion
Honeysuckle has been used for the effective treatment of influenza virus infection for thousands of years in China (Zhou et al. 2015). It efficiently inhibits influenza virus replication in vitro and in vivo. However, the effective components and functional mechanisms had not been previously identified. Here, we performed a systematic evaluation of the activity of the four honeysuckle extracts treatments against the influenza viruses and found that the honeysuckle acids-flavonoids mixture had the strongest inhibitory effect in vitro, as indicated by the higher selectivity index (SI > 23). To verify whether honeysuckle extracts have broad spectrum activity against influenza virus, we evaluated the effects of four honeysuckle extracts treatments against different influenza virus strains, such as A/FM/1/47 (H1N1) and A/Beijing/32/92 (H3N2). As expected, acids-flavonoids mixture dramatically inhibited both strains.
Influenza virus is an important pathogens of human acute respiratory infection and poses a long-term danger to human health. Additional drug-resistant strains are gradually emerging in addition to oseltamivir resistance of the H1N1 influenza virus, which has persisted since 2007 (Collins et al. 2008). The common neuraminidase mutation at amino acid 274 or 275 (H274Y or H275Y) in the influenza virus does not affect its adaptability or replication, which has led to the outbreaks of drug-resistant influenza and epidemics (Hurt 2014; Tarbet et al. 2014). Importantly, the honeysuckle acids-flavonoids mixture at a concentration of 20 μg/mL displayed a sufficient inhibitory effect on the oseltamivir-resistant H1N1-H275Y strain, with an inhibition rate of 47.8%, while the inhibition rate of OC against H1N1-H275Y was only 3.5% (Cheng et al. 2016; Jin et al. 2018).
Furthermore, the oral administration of the honeysuckle acids extract treatment effectively protected mice infected with H1N1 influenza virus from death. At a dosage of 600 mg/kg/d, they reduced body weight loss, alleviated influenza-induced acute lung injury, improved lung parameters and conferred a 30% survival rate on the mice. These data are not consistent with the results we observed in vitro showing that acids-flavonoids mixture had the best antiviral activity. We believe the potential reason is that the crude extracts have different metabolism and absorption rates in vitro and in vivo. We will carry out experiment on drug metabolism in the future.
The time-of-drug addition experiment showed that the honeysuckle extract inhibits influenza virus release primarily because of their effective inhibition of NA. In particular, the honeysuckle extracts treatments had inhibitory activity against NA from H1N1, H3N2, H5N1, H7N9 and the oseltamivir-resistant mutant strain. Honeysuckle extracts may interact with the surface proteins of influenza virus, such as NA, which is important for virus release from host cells (Sakai et al. 2017). However, further in-depth study is still needed. It remains to be revealed whether honeysuckle extracts have inhibitory effects through other mechanisms, such as the inhibition of RNA polymerase activity, inhibition of influenza virus vRNP outside the nucleus, and/or activation of host cell immune function.
In conclusion, our study found that among the four honeysuckle extracts, including acids extract, flavonoids extract, total extract and acids-flavonoids mixture, the acids extract components of honeysuckle are the key antiviral component. They suppresses IAVs infection mainly by inhibiting viral NAs activity. With its broad-spectrum antiviral activity against IAVs, honeysuckle acids extract shows great potential for the clinical management of influenza virus infections.
Acknowledgements
This work was supported by the Fundamental Research Funds for the Central Universities (2632017PT03) and the National Natural Science Foundation (No. 81700756).
Author Contributions
MG, CZ and JD designed the experiments. XK and QG contributed tested compounds. ML, YW, and JJ carried out the experiments. MG, ML and YW analyzed the data. MG and ML wrote the paper. CZ and MG checked and finalized the manuscript. All authors read and approved the final manuscript.
Compliance with Ethical Standards
Conflict of interest
The authors declare that they have no conflict of interest.
Animal and Human Rights Statement
The related expression was observed in the animal experiments and was approved by the Science and Technology Department of Jiangsu Province (SYXK 2016-0011) and the Animal Ethics and Experimentation Committee of China Pharmaceutical University. All institutional and national guidelines for the care and use of laboratory animals were followed.
Footnotes
Mengwei Li and Yuxu Wang contributed equally to this work.
Contributor Information
Changlin Zhou, Email: cl_zhou@cpu.edu.cn.
Min Guo, Email: 1020152471@cpu.edu.cn.
References
- Cai WT, Li YT, Chen SR, Wang ML, Zhang AD, Zhou HB, Chen HC, Jin ML. 14-Deoxy-11,12-dehydroandrographolide exerts anti-influenza A virus activity and inhibits replication of H5N1 virus by restraining nuclear export of viral ribonucleoprotein complexes. Antiviral Res. 2015;118:82–92. doi: 10.1016/j.antiviral.2015.03.008. [DOI] [PubMed] [Google Scholar]
- Chen H, Deng G, Li Z, Tian G, Li Y, Jiao P, Zhang L, Liu Z, Webster RG, Yu K. The evolution of H5N1 influenza viruses in ducks in southern China. Proc Natl Acad Sci U S A. 2004;101:10452–10457. doi: 10.1073/pnas.0403212101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng W, Ren J, Huang Q, Long H, Jin H, Zhang L, Liu H, van Ofwegen L, Lin W. Pregnane steroids from a gorgonian coral Subergorgia suberosa with anti-flu virus effects. Steroids. 2016;108:99–104. doi: 10.1016/j.steroids.2016.02.003. [DOI] [PubMed] [Google Scholar]
- Cinatl J, Jr, Michaelis M, Doerr HW. The threat of avian influenza A (H5N1). Part IV: development of vaccines. Med Microbiol Immunol. 2007;196:213–225. doi: 10.1007/s00430-007-0052-3. [DOI] [PubMed] [Google Scholar]
- Collins PJ, Haire LF, Lin YP, Liu J, Russell RJ, Walker PA, Skehel JJ, Martin SR, Hay AJ, Gamblin SJ. Crystal structures of oseltamivir-resistant influenza virus neuraminidase mutants. Nature. 2008;453:1258–1261. doi: 10.1038/nature06956. [DOI] [PubMed] [Google Scholar]
- De Clercq E. Antiviral agents active against influenza A viruses. Nat Rev Drug Discov. 2006;5:1015–1025. doi: 10.1038/nrd2175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding Y, Dou J, Teng Z, Yu J, Wang T, Lu N, Wang H, Zhou C. Antiviral activity of baicalin against influenza A (H1N1/H3N2) virus in cell culture and in mice and its inhibition of neuraminidase. Arch Virol. 2014;159:3269–3278. doi: 10.1007/s00705-014-2192-2. [DOI] [PubMed] [Google Scholar]
- Furuta Y, Gowen BB, Takahashi K, Shiraki K, Smee DF, Barnard DL. Favipiravir (T-705), a novel viral RNA polymerase inhibitor. Antiviral Res. 2013;100:446–454. doi: 10.1016/j.antiviral.2013.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao R, Cao B, Hu Y, Feng Z, Wang D, Hu W, Chen J, Jie Z, Qiu H, Xu K, Xu X, Lu H, Zhu W, Gao Z, Xiang N, Shen Y, He Z, Gu Y, Zhang Z, Yang Y, Zhao X, Zhou L, Li X, Zou S, Zhang Y, Li X, Yang L, Guo J, Dong J, Li Q, Dong L, Zhu Y, Bai T, Wang S, Hao P, Yang W, Zhang Y, Han J, Yu H, Li D, Gao GF, Wu G, Wang Y, Yuan Z, Shu Y. Human infection with a novel avian-origin influenza A (H7N9) virus. N Engl J Med. 2013;368:1888–1897. doi: 10.1056/NEJMoa1304459. [DOI] [PubMed] [Google Scholar]
- Hampson AW. Vaccines for pandemic influenza. The history of our current vaccines, their limitations and the requirements to deal with a pandemic threat. Ann Acad Med Singap. 2008;37:510–517. [PubMed] [Google Scholar]
- Hay AJ, Wolstenholme AJ, Skehel JJ, Smith MH. The molecular basis of the specific anti-influenza action of amantadine. EMBO J. 1985;4:3021–3024. doi: 10.1002/j.1460-2075.1985.tb04038.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayden FG. Antiviral resistance in influenza viruses—Implications for management and pandemic response. N Engl J Med. 2006;354:785–788. doi: 10.1056/NEJMp068030. [DOI] [PubMed] [Google Scholar]
- Hurt AC. The epidemiology and spread of drug resistant human influenza viruses. Curr Opin Virol. 2014;8:22–29. doi: 10.1016/j.coviro.2014.04.009. [DOI] [PubMed] [Google Scholar]
- Ison MG. Antivirals and resistance: influenza virus. Curr Opin Virol. 2011;1:563–573. doi: 10.1016/j.coviro.2011.09.002. [DOI] [PubMed] [Google Scholar]
- Jin J, Chen Y, Wang D, Ma L, Guo M, Zhou C, Dou J. The inhibitory effect of sodium baicalin on oseltamivir-resistant influenza a virus via reduction of neuraminidase activity. Arch Pharm Res. 2018;41:664–676. doi: 10.1007/s12272-018-1022-6. [DOI] [PubMed] [Google Scholar]
- Jung EJ, Lee KH, Seong BL. Reverse genetic platform for inactivated and live-attenuated influenza vaccine. Exp Mol Med. 2010;42:116–121. doi: 10.3858/emm.2010.42.2.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JH, Resende R, Wennekes T, Chen HM, Bance N, Buchini S, Watts AG, Pilling P, Streltsov VA, Petric M, Liggins R, Barrett S, McKimm-Breschkin JL, Niikura M, Withers SG. Mechanism-based covalent neuraminidase inhibitors with broad-spectrum influenza antiviral activity. Science. 2013;340:71–75. doi: 10.1126/science.1232552. [DOI] [PubMed] [Google Scholar]
- Koehn FE, Carter GT. The evolving role of natural products in drug discovery. Nat Rev Drug Discov. 2005;4:206–220. doi: 10.1038/nrd1657. [DOI] [PubMed] [Google Scholar]
- Leang SK, Deng YM, Shaw R, Caldwell N, Iannello P, Komadina N, Buchy P, Chittaganpitch M, Dwyer DE, Fagan P, Gourinat AC, Hammill F, Horwood PF, Huang QS, Ip PK, Jennings L, Kesson A, Kok T, Kool JL, Levy A, Lin C, Lindsay K, Osman O, Papadakis G, Rahnamal F, Rawlinson W, Redden C, Ridgway J, Sam IC, Svobodova S, Tandoc A, Wickramasinghe G, Williamson J, Wilson N, Yusof MA, Kelso A, Barr IG, Hurt AC. Influenza antiviral resistance in the Asia-Pacific region during 2011. Antiviral Res. 2013;97:206–210. doi: 10.1016/j.antiviral.2012.12.016. [DOI] [PubMed] [Google Scholar]
- Lee SM, Yen HL. Targeting the host or the virus: current and novel concepts for antiviral approaches against influenza virus infection. Antiviral Res. 2012;96:391–404. doi: 10.1016/j.antiviral.2012.09.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine MM, Sztein MB. Vaccine development strategies for improving immunization: the role of modern immunology. Nat Immunol. 2004;5:460–464. doi: 10.1038/ni0504-460. [DOI] [PubMed] [Google Scholar]
- Rohrs S, Kalthoff D, Beer M. A model for early onset of protection against lethal challenge with highly pathogenic H5N1 influenza virus. Vaccine. 2014;32:2631–2636. doi: 10.1016/j.vaccine.2014.03.019. [DOI] [PubMed] [Google Scholar]
- Sakai T, Nishimura SI, Naito T, Saito M. Influenza A virus hemagglutinin and neuraminidase act as novel motile machinery. Sci Rep. 2017;7:45043. doi: 10.1038/srep45043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tarbet EB, Hamilton S, Vollmer AH, Luttick A, Ng WC, Pryor M, Hurst BL, Crawford S, Smee DF, Tucker SP. A zanamivir dimer with prophylactic and enhanced therapeutic activity against influenza viruses. J Antimicrob Chemother. 2014;69:2164–2174. doi: 10.1093/jac/dku127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taubenberger JK, Kash JC. Influenza virus evolution, host adaptation, and pandemic formation. Cell Host Microb. 2010;7:440–451. doi: 10.1016/j.chom.2010.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorlund K, Awad T, Boivin G, Thabane L. Systematic review of influenza resistance to the neuraminidase inhibitors. BMC Infect Dis. 2011;11:134. doi: 10.1186/1471-2334-11-134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian L, Wang ZY, Wu H, Wang S, Wang Y, Wang YY, Xu JW, Wang LY, Qi FC, Fang ML, Yu DH, Fang XX. Evaluation of the anti-neuraminidase activity of the traditional Chinese medicines and determination of the anti-influenza A virus effects of the neuraminidase inhibitory TCMs in vitro and in vivo. J Ethnopharmacol. 2011;137:534–542. doi: 10.1016/j.jep.2011.06.002. [DOI] [PubMed] [Google Scholar]
- Yu J, Wang D, Jin J, Xu J, Li M, Wang H, Dou J, Zhou C. Antiviral activity of SA-2 against influenza A virus in vitro/vivo and its inhibition of RNA polymerase. Antiviral Res. 2016;127:68–78. doi: 10.1016/j.antiviral.2016.01.011. [DOI] [PubMed] [Google Scholar]
- Zhang J, Liu T, Tong X, Li G, Yan J, Ye X. Identification of novel virus inhibitors by influenza A virus specific reporter cell based screening. Antiviral Res. 2012;93:48–54. doi: 10.1016/j.antiviral.2011.10.014. [DOI] [PubMed] [Google Scholar]
- Zhang Q, Shi J, Deng G, Guo J, Zeng X, He X, Kong H, Gu C, Li X, Liu J, Wang G, Chen Y, Liu L, Liang L, Li Y, Fan J, Wang J, Li W, Guan L, Li Q, Yang H, Chen P, Jiang L, Guan Y, Xin X, Jiang Y, Tian G, Wang X, Qiao C, Li C, Bu Z, Chen H. H7N9 influenza viruses are transmissible in ferrets by respiratory droplet. Science. 2013;341:410–414. doi: 10.1126/science.1240532. [DOI] [PubMed] [Google Scholar]
- Zhou Z, Li XH, Liu JX, Dong L, Chen Q, Liu JL, Kong HH, Zhang QY, Qi X, Hou DX, Zhang L, Zhang GQ, Liu YC, Zhang YJ, Li J, Wang J, Chen X, Wang H, Zhang JF, Chen HL, Zen K, Zhang CY. Honeysuckle-encoded atypical microRNA2911 directly targets influenza A viruses. Cell Res. 2015;25:39–49. doi: 10.1038/cr.2014.130. [DOI] [PMC free article] [PubMed] [Google Scholar]