Abstract
Background
Ongoing measures to improve pediatric continuous kidney replacement therapy (CKRT) have lowered mortality rates, shifting the focus to survivor functional status. While septic acute kidney injury generates new morbidity in pediatric critically ill patients, acquired morbidities and functional status of CKRT population are unknown. We predicted that CKRT survivors are at risk for new morbidity and would have worse functional status at PICU discharge compared to baseline, and aimed to describe associated factors.
Methods
Retrospective cohort study over 24 months of CKRT patients surviving to PICU discharge in a quaternary children’s hospital. Functional outcome was determined by Functional Status Scale (FSS).
Results
FSS scores were higher at PICU and hospital discharge compared to baseline. Of 45 CKRT survivors, 31 (69%) had worse FSS score at PICU discharge and 51% had new morbidity (≥3 increase in FSS); majority qualified as moderate to severe disability (FSS ≥10). Four patients (9%) had new tracheostomy, 3 (7%) were ventilator dependent, and 10 (22%) were dialysis dependent. Most (23/45, 51%) required outpatient rehabilitation. Cumulative days on sedation, controlled for illness severity, were independently associated with worse FSS at PICU discharge (aOR 25.18 (3.73, 169.92)). In adjusted analyses, duration of sedation was associated with new morbidity, while neurologic comorbidity, percent fluid overload at CKRT start, and nonrenal comorbidity were associated with moderate to severe disability at PICU discharge when controlled for baseline FSS.
Conclusions
CKRT survivors, with new morbidity and worse functional outcomes at PICU discharge, are a newly described vulnerable population requiring targeted follow-up. Deliberate decrease of sedation exposure in patients with decreased clearance due to organ dysfunction needs to be studied as a modifiable risk factor.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00467-021-05177-7.
Keywords: CKRT, ICU, Functional status, Children, AKI
Introduction
Continuous kidney replacement therapy (CKRT) is routinely used in the pediatric intensive care unit (PICU) to support critically ill children with acute kidney injury (AKI) [1], fluid overload, and metabolic derangements. Traditionally, outcome of pediatric CKRT has been reported as crude mortality, ranging from 58% in the early 2000s to 38% recently, with multiple organ dysfunction, greater percent fluid overload, underlying primary disease process, and young age reported as risk factors [2, 3]. Decreasing mortality rates and better appreciation of lasting impact of PICU stay among survivors have led to reporting outcomes based on morbidity and quality of life in the pediatric literature [4, 5]. Considering the trend reported from more recent studies, it is reasonable to surmise that ongoing measures to improve the practice and use of CKRT will likely continue to lower mortality rates. With the decrease in mortality, survivor quality of life, new morbidity, and functional status will become the focus for improvement in patient outcomes. Although septic AKI has been shown to generate new morbidity in pediatric critically ill patients [6], to date, the acquired morbidities and functional status of the pediatric CKRT population remain unknown.
We predicted that survivors of CKRT are at risk for new morbidity and would have worse functional status at PICU discharge compared to their preadmission (baseline) state. We aimed to describe factors associated with new morbidity, moderate to severe disability, and new technology dependence.
Methods
This was a retrospective cohort study of patients requiring CKRT and surviving to discharge from the PICUs at Texas Children’s Hospital in Houston, TX, from January 2014 through December 2015. Texas Children’s Hospital is a quaternary center with a 32-bed PICU and a 36-bed cardiac intensive care unit and has 1200 annual CKRT days on average. Patients who received modified CKRT with ECMO and patients over the age of 21 years were excluded from this study. The data were extracted from the electronic medical record by a single reviewer and included demographic information, CKRT indication and duration, comorbidities (adjudicated by two investigators from the medical record based on a priori agreement between authors, Supplemental Table1), organ function at admission and discharge, surgical procedures performed, and medications administered. Information about the patient’s ability to complete the activities of daily living was collated from the physical and occupational therapists’ documentation. The number of days on sedation and neuromuscular blockade was calculated from the medication administration records. Typical sedation regimen during the study period in our institution was with opioid (fentanyl) and benzodiazepine (midazolam) infusions with dexmedetomidine infusion added as needed. Data on nutrition was obtained from the institutional quality database for CKRT, where nutrition status is assessed based on z-scores for height and weight and classified as no, mild, moderate, or severe malnutrition by a critical care nephrology dietitian (MWV). The indication for CKRT was extracted from the nephrology notes when available, and from the intensivists’ notes otherwise. Estimated glomerular filtration (eGFR) rate was calculated using the modified Schwartz equation [7]. The Institutional Review Board of Baylor College of Medicine approved this study and waived requirement of informed consent.
The functional outcomes were assigned based on information from the chart at baseline (premorbid), ICU admission, ICU discharge, and hospital discharge. The Functional Status Scale (FSS) was used to describe the patient’s function at each time point [8]. The primary outcome was change in FSS on ICU discharge. The FSS tool has six domains: mental status, sensory, communication, motor function, feeding, and respiratory. Each domain ranges from normal (score of 1) to very severe dysfunction (score of 5) and adds up to an overall score of normal (score of 6) to very severe dysfunction (score of 30) and can describe the functional outcomes in a reliable, rapid, and well-defined way for a large range of ages [8]. We evaluated additional secondary outcomes: new morbidity, moderate to severe disability, new technology dependence, and FSS at hospital discharge. New morbidity was defined as an FSS score increase from baseline greater than or equal to three points. Moderate to severe disability was defined as an FSS score equal to or greater than 10 [9, 10]. FSS was adjudicated by three investigators in pairs at each time point (MS, NTP, AAA). Interrater observer agreement was >95%.
Statistical analysis
Continuous variables were summarized as median (IQR) and tested by rank sum test. Categorical variables were summarized as counts (percentages) and compared using chi-square or Fisher’s exact as appropriate. FSS was treated as a continuous and ordinal variable. Change in FSS from baseline to ICU discharge was analyzed as ordinal change as well as by two binary definitions of ≥10 increase versus <10 increase and worsened functional status (FSS ICU discharge > FSS baseline) versus intact survival (FSS ICU discharge ≤ FSS baseline). New morbidity was a categorical variable.
Generalized linear models (GLM) were used to analyze multivariable associations with outcomes (ordinal logistic model used for FSS ordinal change and binomial logistic model used for binary FSS outcomes). Regression models were fit in two stages: (1) using backwards stepwise selection based on Bayesian information criterion (BIC) minimization to determine most associated risk factors that occurred before or on admission, then (2) adding all during admission variables and using backwards stepwise selection based on BIC minimization to determine the most associated risk factors at all time points. Due to sparse data, only factors with univariate associations p < 0.2 were investigated for inclusion in the multivariable model. Continuous risk factors were binned into binary categories based on median value and tested for model inclusion separately with lowest univariate BIC indicating better fit. FSS at baseline was excluded due to tautological association with the binary outcome of worse FSS at ICU discharge. Conversely, FSS at baseline was forced in the model to ensure control for FSS at baseline in the model of binary outcome of FSS ≥10 at ICU discharge. Associations were reported as odds ratios (95% confidence intervals) and predicted probabilities were computed at mean levels of covariates in each model. All analyses were completed with Stata SE 15.
Results
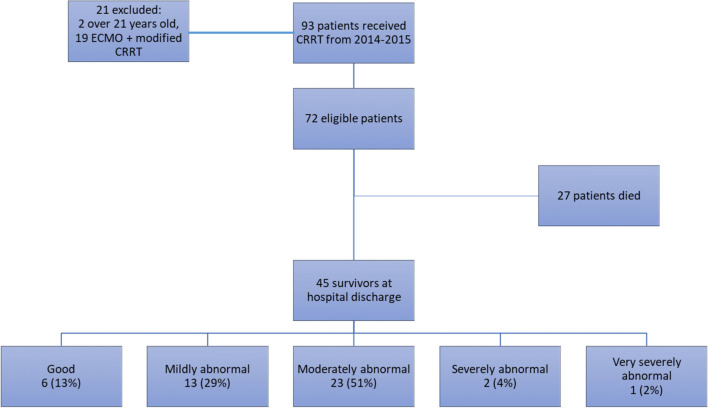
Of the 72 patients, 45 survived to hospital discharge; 42% were female (Fig. 1, Supplemental Table 2). The reasons for ICU admission were sepsis/septic shock in 27% (12/45), liver failure in 20% (9/45), multiple organ failure in 18% (8/45), metabolic coma due to suspected inborn error in 7% (3/45), thrombocytopenia-associated microangiopathy in 7% (3/45), shock (hemorrhagic, cardiac) in 7% (3/45), solid organ transplant in 7% (3/45), cardiac arrest in 4% (2/45), and oncological in 4% (2/45). PELOD score on ICU admission was 12 (IQR 10, 21) (Table 1); number of comorbidities was 2 (IQR 1, 4), 20% had no comorbidity, and 31% had four to seven comorbidities (Supplementary Table1). One patient (2%) had a tracheostomy and six patients (13%) had gastrostomy tubes on admission. Thirty-three patients (73%) had normal nutritional status on admission, 11 (24%) had mild malnutrition, and one (2%) had moderate malnutrition. Forty (89%) patients required invasive mechanical ventilation; length of ventilation was 8 days (IQR 3, 32). The highest oxygenation index (OI) was 8.5 (IQR 3, 19.6). The median sedation duration was 8 days (IQR 2, 25) and neuromuscular blockade duration was 1 day (IQR 0, 5). Thirty-four patients (76%) had surgery in the operating room under general anesthesia during their ICU stay. Four patients (9%) required cardiopulmonary resuscitation and 21 (47%) had new abnormal head imaging during the ICU admission (Supplemental Table 2 and 3).
Fig. 1.
CONSORT diagram of cohort
Table 1.
Baseline characteristics of the whole cohort, patients with intact survival (ICU discharge FSS same as admission or better), and worse functional status (FSS on ICU discharge higher than baseline FSS)
| Baseline characteristics | All surviving CKRT patients n = 45 |
Intact survival n = 14 |
Higher FSS at ICU discharge n = 31 |
p-value |
|---|---|---|---|---|
| Female | 19 (42%) | 6 (32%) | 18 (69%) | 0.954 |
| Age, years | 4 (0.8–11) | 4.5 (0.8–12) | 4 (0.8–10) | 0.720 |
| Total number of comorbidities | 2 (1, 4) | 2.5 (1, 5) | 2 (1, 4) | 0.412 |
| Genetic comorbidity | 7 (16%) | 5 (71%) | 2 (29%) | 0.023` |
| Tracheostomy on admission | 1 (2%) | 1 (100%) | 0 | 0.331` |
| G-tube dependent on admission | 6 (13%) | 4 (67%) | 2 (33%) | 0.065` |
| Baseline FSS | 6 (6, 9) | 8.5 (6, 17) | 6 (6, 6) | 0.007 |
| PELOD on ICU admission | 12 (10, 31) | 10.5 (0, 31) | 22 (10, 31) | 0.247 |
| Cardiopulmonary Resuscitation | 4 (9%) | 1 (25%) | 3 (75%) | 1.000` |
| New seizure | 4 (9%) | 2 (50%) | 2 (50%) | 0.578` |
| Mechanical ventilation | 40 (89%) | 12 (30%) | 28 (70%) | 0.639` |
| Length of invasive mechanical ventilation, days | 8 (3, 32) | 5 (1, 8) | 11 (5, 34) | 0.056 |
| ≥ 10 days | 3 (14%) | 19 (86%) | 0.013 | |
| Highest OI | 8.5 (3, 19.6) | 5 (2.4, 23.4) | 9.3 (3, 19.6) | 0.515 |
| Days on sedation | 8 (2, 25) | 3.5 (1, 8) | 10 (0, 34) | 0.127 |
| ≥ 9 days | 3 (14%) | 18 (86%) | 0.023 | |
| Days on neuromuscular blockade | 1 (0, 5) | 0 (0, 3) | 2 (0, 7) | 0.097 |
| ≥ 1 day | 5 (19%) | 22 (81%) | 0.025 | |
| Days on parenteral nutrition | 8 (0, 17) | 5 (0, 10) | 8 (3, 25) | 0.192 |
| Days on CKRT | 9 (3, 18) | 5 (2, 8) | 12 (6, 27) | 0.020 |
| Repeat CKRT | 3 (7%) | 0 | 3 (100%) | 0.541` |
| PELOD at CKRT start | 31 (12, 32) | 26.5 (10, 40) | 32 (12, 32) | 0.630 |
| % fluid overload at CKRT start | 34.5 (11, 70) | 26.5 (2.6, 49) | 42.5 (18, 74) | 0.124 |
| Hemodialysis | 26 (58%) | 8 (31%) | 18 (69%) | 0.003 |
CKRT, continuous kidney replacement therapy; FSS, Functional Status Scale; GI, Gastrointestinal; ICU, intensive care unit; OI, oxygenation index; OR, operating room
The indications for CKRT were AKI in 64% (29/45); AKI and fluid overload in 9% (4/45); hyperammonemia, AKI, and fluid overload in 7% (3/45); metabolic acidosis in 4% (2/45); hyperammonemia and AKI in 2% (1/45); hyperammonemia in 4% (2/45); fluid overload in 2% (1/45); acidosis and fluid overload in 2% (1/45); acidosis, fluid overload, and hyperkalemia in 2% (1/45); and hyperkalemia in 2% (1/45). PELOD score at CKRT start was 31 (IQR 12, 32). CKRT duration was 9 days (IQR 3, 18). Three patients (7%) required repeat runs of CKRT, and 26 (58%) were transitioned to intermittent hemodialysis after CKRT cessation. Ten of the 45 survivors (22%) were dialysis dependent with an estimated glomerular filtration rate (eGFR) < 15 ml/m2/min at ICU discharge. Thirty-one patients (69%) had outpatient follow-up in renal clinic after hospital discharge.
Four patients (9%) developed new chronic respiratory failure requiring a new tracheostomy; three of these were also ventilator dependent at ICU discharge. Thirteen (29%) were newly dependent on a gastrostomy tube for nutrition, 11 (24%) were dependent on a nasogastric tube, and 2 (4%) were dependent on parenteral nutrition. Seventeen additional patients (38%) had dysfunctional swallowing requiring a change in the thickness of feeds.
The majority of the patients required rehabilitation after their ICU admission. Only eight (18%) were discharged home without any therapy. Most of the survivors (23/45, 51%) required outpatient rehabilitation, while 14 (31%) were transferred to inpatient rehabilitation at hospital discharge.
Change in FSS score at ICU and hospital discharge
Baseline FSS score was normal (6 (IQR 6, 9)); the majority of the cohort (31/45 (69%)) had good baseline function (baseline FSS score of 6–7). Overall functional status was worse at ICU discharge compared to baseline (median 10 (IQR 8, 13)) (p < 0.0001 compared to baseline FSS) with the majority (31/45, 69%) having a worse FSS score at ICU discharge compared to baseline. Eight patients (18%) had no change from baseline, and six (13%) had an improved FSS score at ICU discharge. Four of the six patients with improvement in FSS score had a genetic comorbidity.
FSS score was 8 (IQR 6, 11) at hospital discharge (p = 0.02 compared to baseline FSS). Fifteen patients (33%) were discharged with good function, 16 patients (34%) were discharged with mildly abnormal function, 13 patients (29%) had moderately abnormal function, and one survivor (2%) had very severely abnormal function at discharge.
In univariate analyses, significant risk factors for developing worse FSS at ICU discharge compared to baseline included having a non-genetic morbidity (OR 46.51 (CI 2.49, 869.56)) and cumulative sedation duration of 9 days or more (OR 12.68 (95% CI 1.45, 110.50)). Interestingly, PELOD at ICU admission was not associated with worse FSS at ICU discharge or hospital discharge. PELOD score was forced into multivariable models to control for severity of illness. Despite no independent association with FSS at ICU discharge, inclusion of PELOD strengthened the association for independent effect of cumulative duration of sedation and did not change the point estimate associations between non-genetic morbidity and surgery (Table 2). However, PELOD was independently associated with FSS on hospital discharge, along with days of sedation and non-pulmonary comorbidity (Table 3)
Table 2.
Multivariable ordinal regression of factors associated with change in FSS at ICU discharge (compared to FSS at baseline)
| Risk factors for change in FSS score at ICU discharge compared to baseline | ||
|---|---|---|
| Variable | Adjusted odds ratio (95% CI) | p-value |
| PELOD on ICU admission | 0.99 (0.93, 1.04) | 0.641 |
| Genetic comorbidity | 0.05 (<0.01, 0.63) | 0.021 |
| Days on sedation (≥ 9) | 25.18 (3.73, 169.92) | 0.001 |
| Surgery in OR | 0.11 (0.01, 0.87) | 0.036 |
FSS, Functional Status Scale; OR, operating room
Table 3.
Multivariable generalized linear models (GLM) for higher FSS at hospital discharge compared to baseline
| Risk factors for higher FSS at hospital discharge (FSS hospital discharge > FSS baseline) | ||
|---|---|---|
| Variable | Adjusted odds ratio (95% CI) | p-value |
| Pulmonary comorbidity | 0.02 (<0.01, 0.39) | 0.009 |
| Days on sedation (≥ 9) | 49.14 (4.60, 524.42) | 0.001 |
| PELOD on ICU admission | 1.06 (1.00, 1.12) | 0.049 |
FSS, Functional Status Scale; ICU, intensive care unit; PELOD, Pediatric Logistic Organ Dysfunction score
New morbidity and moderate to severe disability
Twenty-three of the 45 patients (51%) qualified as having a new morbidity based on three or more points increase in their FSS score. In multivariate analysis, duration of sedation (OR 24.50 (95% CI 3.01, 199.34)) was independently associated with new morbidity at ICU discharge, while genetic comorbidity and having surgery in the OR were associated with lower odds of new morbidity (Table 4).
Table 4.
Multivariable logistic regression of factors associated with new morbidity at ICU discharge and multivariable generalized linear model (GLM) of factors associated with moderate to severe disability (FSS ≥ 10 at ICU discharge). Baseline FSS was forced into the models to control for preexisting functional impairment
| Risk factors for new morbidity | ||
|---|---|---|
| Variable | Adjusted odds ratio (95% CI) | p-value |
| Genetic comorbidity | 0.02 (<0.01, 0.40) | 0.010 |
| Days on sedation (≥ 9) | 24.50 (3.01, 199.34) | 0.003 |
| Surgery in OR | 0.05 (<0.01, 0.56) | 0.015 |
| Percent fluid overload | 1.03 (1.00, 1.06) | 0.067 |
| Risk factors for moderate to severe disability at ICU discharge | ||
| Adjusted odds ratio (95% CI) | p-value | |
| Baseline FSS > 6 | 0.63 (0.04, 10.02) | 0.744 |
| Neurologic comorbidity | 20.02 (1.01, 383.75) | 0.047 |
| Renal comorbidity | 0.03 (<0.01, 0.59) | 0.021 |
| Days on sedation | 1.14 (0.99, 1.32) | 0.067 |
| Percent fluid overload | 1.04 (1.0, 1.08) | 0.040 |
FSS, Functional Status Scale; ICU, intensive care unit; OR, operating room
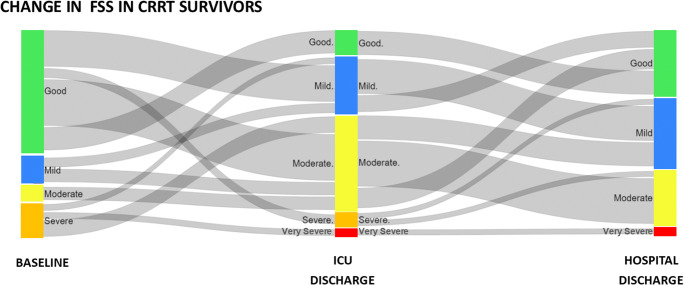
The majority of the cohort had FSS ≥ 10 on ICU discharge (26/45, 58%); therefore, they were classified as moderate or severe disability (Fig. 2, Supplemental Table 4). Patients with moderate to severe disability at ICU discharge were more likely to have neurologic or GI/liver comorbidities and less likely to have renal comorbidities. They had longer length of ventilation, longer duration of CKRT, and longer exposure to sedation and neuromuscular blockade. Their peak OI and percent fluid overload were higher (Supplemental Table 4). On multivariable analysis, when controlled for baseline FSS, the presence of a neurologic comorbidity (OR 20.02 (95% CI 1.01, 383.75)) and percent fluid overload at CKRT start (OR 1.04 (1.0, 1.08)) were independently associated with moderate to severe disability, while patients with renal comorbidity at baseline were less likely to have moderate to severe disability at ICU discharge (OR 0.03 (95% CI <0.01, 0.59)) (Table 4).
Fig. 2.
Sankey diagram of change in Functional Status Scale (FSS) at baseline, on admission, at ICU discharge, and at hospital discharge. Colored bars represent the categories of FSS while the horizontal ribbons represent number of patients moving from one category to the next at the different time points
Discussion
We demonstrated that the majority of pediatric CKRT survivors had higher FSS scores on ICU discharge compared to baseline and over half developed new morbidity. Almost 60% of survivors were classified as having moderate to severe disability at ICU discharge. Majority of the patients required inpatient and outpatient rehabilitation. While FSS modestly improved at hospital discharge, it was still higher than baseline. Independent predictors of worse FSS at ICU discharge were duration of sedation, while genetic comorbidity and surgery during the admission were associated with lack of change in FSS. Duration of sedation was also associated with new morbidity, while neurological comorbidity and fluid overload were independent predictors for moderate to severe disability. Non-pulmonary comorbidity, longer sedation, and higher PELOD score on ICU admission were associated with higher FSS scores at hospital discharge.
In 2018, Pinto et al. described the long-term morbidity for children under 18 years admitted to the PICU with any diagnosis 3 years post discharge [9]. At discharge, 5.2% of patients were classified as having new morbidity. The concerning finding from this study is that, contrary to the general assumption that survivor functionality would improve over time, at 6-month and at 3-year follow-up, new disability rates increased to 6.5% and 10.4%, respectively, demonstrating a sustained increase in morbidity even 3 years after discharge. Considering over half of our population developed new morbidity at discharge, our findings are sobering. This fraction is staggeringly high compared to recently reported post-discharge morbidity in children with acute respiratory failure, where 20% of the cohort developed functional decline at PICU discharge [11].
Nonrenal outcomes have been explored only in septic AKI in children. Fitzgerald et al. described mortality and new morbidity in a pediatric population with severe sepsis and AKI based on Pediatric Overall Performance Category (POPC) score at admission compared to at discharge or 90 days after study start date [6]. In this study, 37% of survivors with severe sepsis and severe AKI developed new disability, and 24% had moderate disability at discharge. Of note, only 60% patients in that study required kidney replacement therapy, and the morbidity for that subset was not delineated. Our population had a higher rate of morbidity at discharge than children with severe AKI and sepsis. Recently, Starr and colleagues demonstrated higher FSS scores at discharge in pediatric septic shock patients with AKI, and lower quality of life at 3 months after discharge with poor physical performance in AKI survivors [12]. Intriguingly, neurocognitive problems have been described to persist in 2-year follow-up in patients who develop AKI in the setting of severe malaria [13, 14]. Collectively, these findings highlight the connection between kidney injury and global poor outcomes in critical illness.
Interestingly, in our cohort, there was a significant protective effect from having pre-existing kidney disease against developing moderate to severe disability at discharge. We did not compare the severity of illness for the eleven survivors with pre-existing renal disease to the remainder of the cohort, but good global outcomes have been demonstrated in children receiving CKRT primarily for a renal indication in other cohorts [15, 16]. Although not examined separately, there were more patients who received intermittent HD after CKRT in the worse FSS group compared to intact survival; whether this reflects the adverse impact of delayed kidney recovery on functional outcomes remains speculative and should be further investigated. Having a genetic diagnosis also suggested to be protective against developing new morbidity, and four of the six patients who had an improvement in their FSS score had a genetic comorbidity. We hypothesize that this finding might be due to the presence of toxic metabolites affecting patients’ central nervous system prior to treatment with CKRT and benefit from purification therapy directed at the underlying disorder, as inborn error of metabolism is an indication for CKRT in pediatrics [17]. Alternatively, FSS tool might lack sensitivity to capture potentially subtle changes that happen due to ceiling effect or parental preference to limit invasive interventions in this population. We categorized inborn errors of metabolism within the comorbidity category of genetic diseases, and not as a category on their own. While these patients frequently require extracorporeal therapy only for blood purification and do not always develop AKI, there were only three patients with inborn errors of metabolism in our genetic comorbidity cohort, significantly limiting our ability to generate specific conclusions within our data set. Either way, this observation likely is not generalizable to other PICU survivor cohorts.
The number of days a patient received sedation significantly correlated with the development of higher FSS and new morbidity on univariate and multivariable analyses controlling for severity of illness. Sedation days positively correlated with length of ventilation, days on parenteral nutrition, days on CKRT, and days on neuromuscular blockade, which are all therapies associated with a higher severity of illness. Associations between opioid use and cognitive impairments after pediatric critical illness have been described, but interestingly, relationship between kidney impairment during PICU stay and outcomes has not been explored [4, 5]. Similarly, association between severity of illness and cognitive impairment has not been widely reported, but this might be due to inability to truly capture chronic disease burden and actual severity of illness with current scoring systems. In pediatric acute respiratory failure, exposure to certain classes of sedatives and opioids, such as methadone and clonidine, has been associated with functional decline and impaired quality of life post discharge [11]. Kidney dysfunction and treatment with extracorporeal therapies fundamentally impact pharmacokinetics of many therapeutics, changing the volume of distribution and clearance [18–20]. It is plausible to propose that children who have abnormal kidney function and therefore require CKRT would be at higher risk of accumulation of sedative agents and their active metabolites. We were not able to assess cumulative dose and number of sedative agents used in our patients. It would be interesting to see if our results can be replicated in other PICUs with alternative sedation protocols. Our findings add yet another dimension to post intensive care syndrome in pediatrics, raising the question of stratification of patients not only according to primary diagnostic categories but also according to pattern of organ failure during PICU stay to explore outcome associations and devise interventional approaches in post-PICU follow-up.
Our study had limitations. Retrospective approach prevents establishing casuality between clinical characteristics and worse functional outcome. We were limited in covariates that could be included in our statistical models due to sample size. We chose to focus on functional status of survivors at ICU discharge, as we wanted to capture the patients after the ICU stay while still in the hospital to explore this potential window of opportunity for intervention. We did not have long-term follow-up data on our patients and cannot evaluate their trajectory post hospital discharge. Further studies could leverage planned medical follow-up as opportunities to assess FSS for longitudinal changes. As a single-center study, our findings might have limited external validity, although our cohort is similar to other CKRT populations from resource-replete settings [3, 21, 22]. While days on sedation were associated with all outcomes, the estimates were often imprecise (indicated by wide CI) due to our sample size; thus, actual magnitude of effects should be interpreted with caution. We were not able to precisely measure exposure to sedation as cumulative dosing data was not available to the investigators. In addition, as would be expected, there is high correlation between days on sedation and days on CKRT, days on neuromuscular blockade, and length of ventilation. Due to this known correlation between risk factors (and to limited sample size thus limited number of predictors we can allow in the model), we used the two-stage model fitting technique to first determine which predictors were more correlated and then only included those with low p-values and BIC in the final model—this resulted in restricting to only the most associated predictors included in the model and removing multiple predictors that measure similar risks (such as length of ventilation and days on sedation). With the growing recognition of ICU-related morbidity, including deconditioning and prolonged immobilization, on adverse outcomes, it would be crucial to explore whether different approaches to sedation management have differential impact on functional status of patients. Although we only documented new abnormal head imaging findings relying on radiology reports, we were not able to ascertain the exact indication for imaging and we did not have data on longitudinal imaging results. We did not assess evolution of extra-renal organ failure between ICU admission and CKRT start. Pattern of evolution of organ dysfunction, prior to CKRT start as well as during and after, might provide additional prognostic implications.
Similar to survivors of the neonatal intensive care units who are followed in a multidisciplinary clinic focusing on their neurodevelopment, some institutions have follow-up clinics for PICU survivors with congenital heart disease or traumatic brain injury [23]. As we continue to discover the impact critical illness has on pediatric survivors, it may become apparent that these follow-up clinics are necessary in other discrete populations, such as the CKRT population, whose longitudinal follow-up has largely had a nephrocentric focus until now. Risk factors for adverse outcomes, if confirmed in multicentral studies, constitute both potential interventional targets during hospitalization and also stratification tools for facilitating cohorting for optimal follow-up. Patients with highest risk might benefit from targeted follow-up in multidisciplinary clinics that incorporate developmental pediatrics with nephrology and other subspecialty follow-up [24].
Conclusions
CKRT survivors develop worse functional outcomes and new morbidity at PICU discharge and have higher FSS scores at hospital discharge. This is a newly described vulnerable population requiring global developmental follow-up. Deliberate decrease of sedation exposure in patients with decreased clearance due to organ dysfunction needs to be studied as a modifiable risk factor. Given the high proportion of survivors qualifying for moderate to severe disability, inpatient modifiable risk factors as well as outpatient follow-up need further study to allocate proper resources and improve outcomes.
Supplementary Information
(DOCX 38 kb)
Acknowledgements
The authors would like to acknowledge Mr. Uluc Arikan for his assistance in the creation of the figures.
This study was conducted at Baylor College of Medicine-affiliated Texas Children’s Hospital in Houston, TX.
Availability of data and material
Data will be available through BCM IRB application.
Code availability
Not applicable
Funding
Internal funding from Baylor College of Medicine supported the study.
Declarations
Ethics approval
Baylor College of Medicine Institutional Review Board approved the study.
Consent to participate
Consent was waived by the IRB.
Consent for publication
Not applicable
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Le KN, Moffett BS, Ocampo EC, Zaki J, Mossad EB. Impact of dexmedetomidine on early extubation in pediatric cardiac surgical patients. Intensive Care Med. 2011;37:686–690. doi: 10.1007/s00134-011-2140-5. [DOI] [PubMed] [Google Scholar]
- 2.Symons JM, Chua AN, Somers MJ, Baum MA, Bunchman TE, Benfield MR, Brophy PD, Blowey D, Fortenberry JD, Chand D, Flores FX, Hackbarth R, Alexander SR, Mahan J, McBryde KD, Goldstein SL. Demographic characteristics of pediatric continuous renal replacement therapy: a report of the prospective pediatric continuous renal replacement therapy registry. Clin J Am Soc Nephrol. 2007;2:732–738. doi: 10.2215/CJN.03200906. [DOI] [PubMed] [Google Scholar]
- 3.Cortina G, McRae R, Hoq M, Donath S, Chiletti R, Arvandi M, Gothe RM, Joannidis M, Butt W. Mortality of critically ill children requiring continuous renal replacement therapy: effect of fluid overload, underlying disease, and timing of initiation. Pediatr Crit Care Med. 2019;20:314–322. doi: 10.1097/PCC.0000000000001806. [DOI] [PubMed] [Google Scholar]
- 4.Heneghan JA, Pollack MM. Morbidity: changing the outcome paradigm for pediatric critical care. Pediatr Clin N Am. 2017;64:1147–1165. doi: 10.1016/j.pcl.2017.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Manning JC, Pinto NP, Rennick JE, Colville G, Curley MAQ. Conceptualizing post intensive care syndrome in children-the PICS-p framework. Pediatr Crit Care Med. 2018;19:298–300. doi: 10.1097/PCC.0000000000001476. [DOI] [PubMed] [Google Scholar]
- 6.Fitzgerald JC, Basu RK, Akcan-Arikan A, Izquierdo LM, Pineres Olave BE, Hassinger AB, Szczepanska M, Deep A, Williams D, Sapru A, Roy JA, Nadkarni VM, Thomas NJ, Weiss SL, Furth S, Sepsis PRevalence, OUtcomes, and Therapies Study Investigators and Pediatric Acute Lung Injury and Sepsis Investigators Network Acute kidney injury in pediatric severe sepsis: an independent risk factor for death and new disability. Crit Care Med. 2016;44:2241–2250. doi: 10.1097/CCM.0000000000002007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schwartz GJ, Munoz A, Schneider MF, Mak RH, Kaskel F, Warady BA, Furth SL. New equations to estimate GFR in children with CKD. J Am Soc Nephrol. 2009;20:629–637. doi: 10.1681/ASN.2008030287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pollack MM, Holubkov R, Glass P, Dean JM, Meert KL, Zimmerman J, Anand KJ, Carcillo J, Newth CJ, Harrison R, Willson DF, Nicholson C, Kennedy E, Shriver National Institute of Child Health, Human Development Collaborative Pediatric Critical Care Research Network Functional Status Scale: new pediatric outcome measure. Pediatrics. 2009;124:e18–e28. doi: 10.1542/peds.2008-1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pinto NP, Rhinesmith EW, Kim TY, Ladner PH, Pollack MM. Long-term function after pediatric critical illness: results from the survivor outcomes study. Pediatr Crit Care Med. 2017;18:e122–e130. doi: 10.1097/PCC.0000000000001070. [DOI] [PubMed] [Google Scholar]
- 10.Pollack MM, Holubkov R, Funai T, Berger JT, Clark AE, Meert K, Berg RA, Carcillo J, Wessel DL, Moler F, Dalton H, Newth CJ, Shanley T, Harrison RE, Doctor A, Jenkins TL, Tamburro R, Dean JM, Kennedy E, Shriver National Institute of Child Health, Human Development Collaborative Pediatric Critical Care Research Network Simultaneous prediction of new morbidity, mortality, and survival without new morbidity from pediatric intensive care: a new paradigm for outcomes assessment. Crit Care Med. 2015;43:1699–1709. doi: 10.1097/CCM.0000000000001081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Watson RS, Asaro LA, Hutchins L, Bysani GK, Killien EY, Angus DC, Wypij D, Curley MAQ. Risk factors for functional decline and impaired quality of life after pediatric respiratory failure. Am J Respir Crit Care Med. 2019;200:900–909. doi: 10.1164/rccm.201810-1881OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Starr MC, Banks R, Reeder RW, Fitzgerald JC, Pollack MM, Meert KL, PS MQ, Mourani PM, Chima RS, Sorenson S, Varni JW, Hingorani S, Zimmerman JJ, Life After Pediatric Sepsis Evaluation (LAPSE) Investigators Severe acute kidney injury is associated with increased risk of death and new morbidity after pediatric septic shock. Pediatr Crit Care Med. 2020;21:e686–e695. doi: 10.1097/PCC.0000000000002418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hickson MR, Conroy AL, Bangirana P, Opoka RO, Idro R, Ssenkusu JM, John CC. Acute kidney injury in Ugandan children with severe malaria is associated with long-term behavioral problems. PLoS One. 2019;14:e0226405. doi: 10.1371/journal.pone.0226405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Conroy AL, Opoka RO, Bangirana P, Idro R, Ssenkusu JM, Datta D, Hodges JS, Morgan C, John CC. Acute kidney injury is associated with impaired cognition and chronic kidney disease in a prospective cohort of children with severe malaria. BMC Med. 2019;17:98. doi: 10.1186/s12916-019-1332-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Symons JM, Brophy PD, Gregory MJ, McAfee N, Somers MJ, Bunchman TE, Goldstein SL. Continuous renal replacement therapy in children up to 10 kg. Am J Kidney Dis. 2003;41:984–989. doi: 10.1016/s0272-6386(03)00195-1. [DOI] [PubMed] [Google Scholar]
- 16.Riley AA, Watson M, Smith C, Guffey D, Minard CG, Currier H, Akcan Arikan A. Pediatric continuous renal replacement therapy: have practice changes changed outcomes? A large single-center ten-year retrospective evaluation. BMC Nephrol. 2018;19:268. doi: 10.1186/s12882-018-1068-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yetimakman AF, Kesici S, Tanyildiz M, Bayrakci B. Continuous renal replacement therapy for treatment of severe attacks of inborn errors of metabolism. J Pediatr Intensive Care. 2019;8:164–169. doi: 10.1055/s-0039-1683991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Moffett BS, Morris J, Munoz F, Arikan AA. Population pharmacokinetic analysis of vancomycin in pediatric continuous renal replacement therapy. Eur J Clin Pharmacol. 2019;75:1089–1097. doi: 10.1007/s00228-019-02664-7. [DOI] [PubMed] [Google Scholar]
- 19.Nehus EJ, Mizuno T, Cox S, Goldstein SL, Vinks AA. Pharmacokinetics of meropenem in children receiving continuous renal replacement therapy: validation of clinical trial simulations. J Clin Pharmacol. 2016;56:291–297. doi: 10.1002/jcph.601. [DOI] [PubMed] [Google Scholar]
- 20.Gist KM, Mizuno T, Goldstein SL, Vinks A. Retrospective evaluation of milrinone pharmacokinetics in children with kidney injury. Ther Drug Monit. 2015;37:792–796. doi: 10.1097/FTD.0000000000000214. [DOI] [PubMed] [Google Scholar]
- 21.de Galasso L, Emma F, Picca S, Di Nardo M, Rossetti E, Guzzo I. Continuous renal replacement therapy in children: fluid overload does not always predict mortality. Pediatr Nephrol. 2016;31:651–659. doi: 10.1007/s00467-015-3248-6. [DOI] [PubMed] [Google Scholar]
- 22.de Galasso L, Picca S, Guzzo I. Dialysis modalities for the management of pediatric acute kidney injury. Pediatr Nephrol. 2020;35:753–765. doi: 10.1007/s00467-019-04213-x. [DOI] [PubMed] [Google Scholar]
- 23.Goldsworthy M, Franich-Ray C, Kinney S, Shekerdemian L, Beca J, Gunn J. Relationship between social-emotional and neurodevelopment of 2-year-old children with congenital heart disease. Congenit Heart Dis. 2016;11:378–385. doi: 10.1111/chd.12320. [DOI] [PubMed] [Google Scholar]
- 24.Kashani K, Rosner MH, Haase M, Lewington AJP, O’Donoghue DJ, Wilson FP, Nadim MK, Silver SA, Zarbock A, Ostermann M, Mehta RL, Kane-Gill SL, Ding X, Pickkers P, Bihorac A, Siew ED, Barreto EF, Macedo E, Kellum JA, Palevsky PM, Tolwani AJ, Ronco C, Juncos LA, Rewa OG, Bagshaw SM, Mottes TA, Koyner JL, Liu KD, Forni LG, Heung M, Wu VC. Quality improvement goals for acute kidney injury. Clin J Am Soc Nephrol. 2019;14:941–953. doi: 10.2215/CJN.01250119. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX 38 kb)
Data Availability Statement
Data will be available through BCM IRB application.