Abstract
The ubiquitin-proteasome system modifies different cellular and protein functions. Its dysregulation may lead to disrupted proteostasis associated with multiple pathologies and aging. Pharmacological regulation of proteasome functions is already an important part of the treatment of several diseases. 1,4-dihydropyridine (1,4-DHP) derivatives possess different pharmacological activities, including antiaging and neuroprotective. The aim of this study was to investigate the effects of several 1,4-DHP derivatives on mRNA expression levels of proteasomal genes Psma3, Psmb5, and Psmc6 in several organs of rats. Rats were treated with metcarbatone, etcarbatone, glutapyrone, styrylcarbatone, AV-153-Na, or AV-153-Ca per os for three days. mRNA expression levels were determined with real-time polymerase chain reaction (PCR). For AV-153-Na and AV-153-Ca, we also determined the expression of the Psma6 gene. In the kidney, metcarbatone, etcarbatone, styrylcarbatone, and AV-153-Na increased the expression of all analysed genes. Glutapyrone increased the expression of Psmb5 and Psmc6 but did not affect the expression of Psma3. In the blood, glutapyrone increased Psmb5 expression. In the liver, AV-153-Na increased the expression of Psma6 and Psmc6 but lowered the expression of Psmb5, while AV-153-Ca only increased Psma6 expression. The ability of 1,4-DHP derivatives to increase the expression of proteasome subunit genes might hold a therapeutic potential in conditions associated with impaired proteasomal functions, but further research is needed.
Key words: AV-153-Ca, AV-153-Na, etcarbatone, gene expression, glutapyrone, impaired proteasomal functions, metcarbatone, pharmacological activities, proteasome subunits, styrylcarbatone, ubiquitin-proteasome system
Abstract
Ubikvitin-proteasomski sustav utječe na različite funkcije bjelančevina i stanica. Poremećaji u njegovoj regulaciji mogu dovesti do poremećaja u proteostazi koji su povezani s nastankom različitih bolesti i sa starenjem, ali se sustav može regulirati u liječenju pojedinih bolesti lijekovima poput 1,4 dihidropiridinskih (1,4 DHP) derivata, koji štite živčani sustav i usporavaju starenje. Cilj ovoga istraživanja bio je utvrditi djelovanje nekoliko 1,4 DHP derivata na ekspresiju glasničke RNA (mRNA) u proteasomskim genima Psma3, Psmb5 i Psmc6 u više štakorskih organa. Štakori su tri dana dobivali oralne doze metkarbatona, etkarbatona, glutapirona, stirilkarbatona, AV-153-Na ili AV-153-Ca, a genska se ekspresija u mRNA utvrdila polimeraznom lančanom reakcijom u stvarnom vremenu (engl. real-time polymerase chain reaction, krat. PCR). Osim toga, utvrdili smo djelovanje derivata AV‑153‑Na i AV‑153‑Ca na ekspresiju gena Psma6. U bubrezima su metkarbaton, etkarbaton, stirilkarbatoni AV‑153‑Na povećali ekspresiju svih analiziranih gena. Glutapiron je povećao ekspresiju Psmb5 i Psmc6, ali ne i Psma3. U krvi je glutapiron povećao gensku ekspresiju Psmb5. U jetrima je AV‑153‑Na povećao ekspresiju Psma6 i Psmc6, istodobno smanjivši ekspresiju Psmb5. AV‑153‑Ca utjecao je samo na Psma6, povećavši mu ekspresiju. Sposobnost 1,4-DHP derivata da povećaju gensku ekspresiju proteasomskih podjediničnih proteina obećava u smislu mogućnosti liječenja bolesti povezanih s poremećenom proteasomskom funkcijom, no nužna su daljnja istraživanja.
Ključne riječi: AV-153-Ca, AV-153-Na, etkarbaton, glutapiron, metkarbaton, stirilkarbaton, poremećena proteasomska funkcija, proteasomske podjedinice, ubikvitin-proteasomski sustav
Proteostasis, or cellular protein homeostasis, relies on the regulation of protein synthesis, folding, conformational maintenance, and degradation (1). Deviations from optimal proteostasis can result in serious pathologies and accelerate the aging of an organism. Proteostasis is maintained by several control systems, all of them of equal importance (1). Protein degradation is partly regulated by the ubiquitin-proteasome system (UPS), which ensures rapid and specific turnover of proteins. UPS modifies cellular and protein functions, including cell cycle, cell signalling, DNA repair, chromatin modifications, and protein trafficking (2). These are mediated by ATPase (ubiquitination enzymes encoded by the Psmc genes in rodents) and the 20S core particle. The latter has a cylinder-like structure consisting of 28 proteins arranged in four heptameric rings. The outer ring is formed by alpha subunits encoded by the Psma genes in rodents (PSMA in humans). The inner, beta subunits are encoded by the Psmb genes (PSMB in humans) (2). Proteasome gene expression is triggered by the nuclear respiratory factor 1 (Nrf1). This transcription factor is, in turn, regulated by the mammalian target of rapamycin complex 1 (mTORC1) (3) and a feedback mechanism compensating proteasome dysfunction (4). Nrf1-dependent transcription of proteasomal genes is also increased by pharmacological inhibition of proteasomes (2). Pharmacological inhibition of proteasome function is important for the treatment of several diseases such as cancer (5), whereas stimulators of proteasome activity are being researched (6) as potential remedies against neurodegenerative diseases and as antiaging agents. It was shown that proteasome activity in long-living mammal species is higher than in short-living animals (7). Several natural substances were found to stimulate proteasome activity (8), synthetic molecules are also intensively studied (9).
In our opinion, testing of the impact of 1,4-dihydropyridine (1,4-DHP) derivatives, a vast group of compounds with different pharmacological activities, on the UPS could be a prospective research branch. A big group of these compounds has been synthesised in the Latvian Institute of Organic Synthesis over the last few years. Some of them manifest interesting effects besides antioxidant activity (9), as they can modify cell proliferation (10), bind DNA and proteins, or stimulate DNA repair by activating DNA repair enzymes (11, 12). These novel 1,4‑DHP derivatives have a weak Ca2+ channel blocker activity and are water-soluble unlike “classical” Ca2+ channel blockers, which are hydrophobic. Yet they can modify the expression of several genes and proteins (13, 14), including the proteasome gene Psma6 (15). The present work aimed to expand our previous research by studying the effects of several 1,4‑DHP derivatives on mRNA expression levels of proteasomal genes Psma3, Psmb5, and Psmc6 in several organs of rats to see if they have pharmacological potential as UPS modulators.
Materials and methods
Animals
This study was approved by the Animal Ethics Committee of the Food and Veterinary Service (Riga, Latvia) and was carried out according to the guidelines of the 1986 European Convention for the Protection of Vertebrate Animals Used for Experimental and other Scientific Purposes (16). Male Wistar rats (215.0±5.6 g) were purchased from the Laboratory of Experimental Animals, Riga Stradins University, Riga, Latvia. Animals were kept at 22±0.5 °C with a 12 h light/dark cycle and fed standard laboratory diet.
Chemicals
All drugs used in the study – metcarbatone, etcarbatone, glutapyrone, styrylcarbatone (J-9-125), and AV-153 Na and Ca salts (Figure 1) were synthesised at the Latvian Institute of Organic Synthesis. Other chemicals were purchased from Sigma-Aldrich Chemie (Taufkirchen, Germany).
Figure 1.
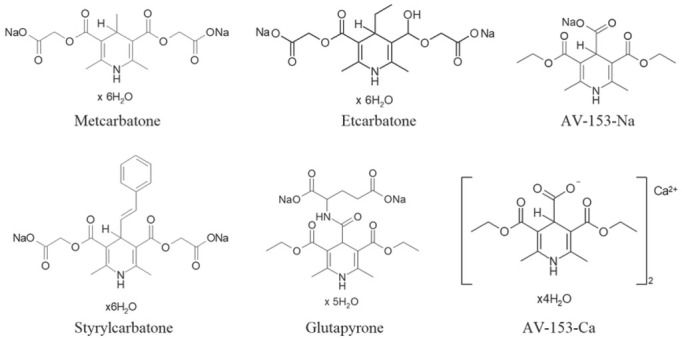
Formulas of 1,4-dihydropyridine derivatives used in the study
Experimental design
Rats were divided into control and treatment groups. The latter received 0.05 mg/kg or 0.5 mg/kg of metcarbatone, etcarbatone, glutapyrone, styrylcarbatone, AV-153-Na, or AV-153-Ca per os by gavage for three days. The rats were then euthanised and their organ samples (kidneys, blood, and liver) taken and frozen in liquid nitrogen until analysis. There were two sets of rats. Kidneys and blood were taken from the control group (11 animals) and groups treated with metcarbatone, etcarbatone, glutapyrone, and styrylcarbatone (3–4 animals per group). Kidneys and liver were taken from the control group (10 animals) and groups treated with AV-153-Na and AV-153-Ca (3–5 animals per group).
RNA extraction and cDNA synthesis
Total RNA was isolated from the kidneys, blood, and liver with a TRI reagent (Sigma Aldrich, Taufkirchen, Germany). RNA was purified from DNA with a DNA-free kit (Ambion, Austin, TX, USA) and its quantity and purity determined with a NanoPhotometer ® NP 80 spectrophotometer (ImplenGMBH, Munich, Germany).
The quality of RNA was analysed with gel electrophoresis. cDNA was synthesised from the obtained RNA (5 μg from kidneys and liver, and 2 μg from blood) with random hexamer primers (RevertAid™ First Strand cDNA Synthesis Kit, Fermentas, Vilnius, Lithuania).
Real-time reverse-transcription polymerase chain reaction tests
mRNA expression of Psma3, Psmb5 and Psmc6, and a reference gene (RNA-polymerase II) in the kidney, blood, and liver was determined using the SYBR® Green PCR Master Mix (Applied Biosystems, Foster City, CA, USA) according to the instructions provided by the manufacturer. For the AV-153-Na and AV-153-Ca treatment groups, we also determined the expression of the Psma6 gene.
Primers were designed using Primer‑BLAST software ( 1 7 ) . Primer sequences were: Psma3 – 5’‑CACCATCCTCTGGTGTCCATT‑3’ (forward) and 5’‑CGCAGATATCCTCAATTACCCAAC‑3’ (reverse) ( fragment size 1 2 8 b p ) ; P s m b 5 – 5’‑AGGTGCCTACATTGCTTCCC‑3’ (forward) and 5’‑GAGATGCGTTCCTTGTTGCG‑3’ (reverse) (fragment size 159 bp); Psmc6 – 5’‑TACATTGGGGAAAGCGCTCG‑3’ (forward) and 5’‑TCAGAAAACCGACGACCACC‑3’ (reverse) (fragment size 116 bp); and Psma6 – 5’‑GTGTGCGCTACGGGGTGTA‑3’ (forward) and 5’‑AGTCACGGTGCTGGAATCCA‑3’ (reverse) (fragment size 247 bp). The choice of the reference RNA‑polymerase II gene was described earlier (18). Primer sequences for this gene were 5’‑GCCAGAGTCTCCCATGTGTT‑3’ (forward) and 5’‑GTCGGTGGGACTCTGTTTGT‑3’ (reverse) (amplified fragment size 135 bp).
Oligonucleotides were supplied by Metabion International AG (Martinsried, Germany). qPCR reactions were performed using a StepOne™ Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). Cycling conditions were as follows: one cycle at 95 ºC for 10 min, 40 cycles at 95 ºC for 15 sec, and one cycle at 60 ºC for 1 min (Applied Biosystems StepOne software, version 2.1). The specificity of amplification products was verified by dissociation curve: one cycle at 95 ºC for 15 sec, one at 60 ºC for 1 min, and one at 95 ºC for 15 sec. The cycle threshold (Ct) values are presented in Tables 1 and 2.
Table 1.
Kidney and blood real-time PCR cycle threshold (Ct) values
| Kidneys | Blood | |||||||
|---|---|---|---|---|---|---|---|---|
| RNSpolII | Psma3 | Psmb5 | Psmc6 | RNSpolII | Psma3 | Psmb5 | Psmc6 | |
| Control | 20,6336 | 21,2849 | 20,2966 | 18,5916 | 19,3183 | 24,7317 | 23,9346 | 23,4343 |
| 19,9632 | 21,0402 | 20,1468 | 18,5301 | 19,1140 | 24,9544 | 23,9330 | 23,6485 | |
| 20,5162 | 20,6843 | 19,9793 | 18,4140 | 19,5568 | 23,5918 | 23,6477 | 22,6171 | |
| 19,7661 | 20,8542 | 20,0899 | 18,4168 | 19,5473 | 25,3724 | 23,5371 | 23,2719 | |
| 20,4493 | 21,1992 | 20,3281 | 18,7329 | 19,2752 | 24,2432 | 23,6730 | 22,2758 | |
| 20,0575 | 20,7519 | 19,8123 | 18,2283 | 18,0247 | 24,1311 | 23,7417 | 21,8996 | |
| 19,8994 | 20,8551 | 20,1670 | 18,5733 | 19,6721 | 24,1230 | 22,6278 | 22,9333 | |
| 19,9792 | 21,2709 | 20,5719 | 18,8297 | 18,5737 | 23,8446 | 23,0603 | 22,4729 | |
| 20,3273 | 21,4559 | 20,4872 | 18,9060 | 18,9473 | 24,8475 | 24,5772 | 23,0626 | |
| 20,7163 | 21,1733 | 20,4132 | 18,8730 | 19,7215 | 24,5549 | 23,4031 | 23,6338 | |
| 20,1879 | 21,1945 | 20,4594 | 18,7461 | 18,7025 | 24,7850 | 24,1693 | ||
| 18,7703 | 24,3940 | 24,0506 | ||||||
| Metcarbatone 0.05 mg/kg | 21,1421 | 21,1700 | 20,2690 | 18,7102 | 19,1992 | 24,9793 | 24,9819 | 23,4209 |
| 20,6063 | 20,9449 | 20,0149 | 18,6103 | 18,2122 | 25,0053 | 24,6967 | 22,8411 | |
| 20,9352 | 21,4263 | 20,5656 | 18,9078 | 19,6951 | 24,3028 | 23,8090 | 22,9598 | |
| 20,5415 | 20,9494 | 20,0938 | 18,4946 | 18,7135 | 24,5634 | 24,1763 | 22,6806 | |
| Metcarbatone 0.5 mg/kg | 20,8324 | 20,9915 | 20,1140 | 18,8390 | 18,9062 | 25,2567 | 23,8337 | 23,6736 |
| 21,1187 | 21,0831 | 20,1682 | 18,7917 | 19,2715 | 25,2724 | 24,5450 | 23,6550 | |
| 21,0912 | 21,1248 | 20,3896 | 18,8515 | 18,5728 | 24,6956 | 23,9355 | 22,8742 | |
| 20,6640 | 21,1151 | 20,0875 | 18,7441 | 18,6701 | 25,2925 | 24,5882 | 23,2121 | |
| Etcarbatone 0.05 mg/kg | 20,6521 | 21,3114 | 20,2236 | 18,6247 | 18,8542 | 24,8812 | 24,1604 | 22,9606 |
| 21,2283 | 21,3560 | 20,3069 | 18,6993 | 19,1319 | 24,8758 | 24,7965 | 22,9433 | |
| 21,3387 | 21,4437 | 20,1647 | 18,7293 | 18,1798 | 24,6700 | 24,4486 | 22,5183 | |
| Etcarbatone 0.5 mg/kg | 20,6957 | 21,2241 | 20,1316 | 18,4727 | 19,2888 | 24,9479 | 24,4943 | 23,1595 |
| 21,4283 | 21,6113 | 20,5876 | 18,8926 | 19,2577 | 24,8016 | 24,0260 | 23,0911 | |
| 21,0995 | 21,5415 | 20,3180 | 18,8051 | 18,9519 | 24,5479 | 24,2079 | 22,4058 | |
| 20,9716 | 21,4134 | 20,4994 | 18,8108 | 19,0836 | 24,6532 | 24,6596 | 22,5140 | |
| Styrylcarbatone 0.05 mg/kg | 21,4476 | 21,4452 | 20,5627 | 18,6984 | 19,5796 | 23,9558 | 23,0535 | 22,6217 |
| 20,9010 | 21,0415 | 20,1628 | 18,3986 | 19,0160 | 24,8889 | 24,1739 | 23,2842 | |
| 21,3434 | 21,2509 | 20,2865 | 18,6662 | 18,1215 | 23,5157 | 23,3238 | 21,4245 | |
| 21,6191 | 22,0462 | 21,5846 | 18,9777 | 19,5722 | 25,4267 | 24,9225 | 23,4448 | |
| Styrylcarbatone 0.5 mg/kg | 21,5660 | 21,5113 | 21,2236 | 18,9375 | 19,1745 | 23,8688 | 23,2048 | 22,2596 |
| 21,2131 | 21,5601 | 20,7720 | 18,7713 | 19,5076 | 24,7026 | 23,6100 | 23,0168 | |
| 21,0897 | 21,4685 | 20,4881 | 18,7617 | 18,9420 | 24,6156 | 23,8653 | 23,2664 | |
| 21,4455 | 21,6779 | 20,9215 | 18,9282 | |||||
| Glutapyrone 0.05 mg/kg | 22,0678 | 22,8544 | 21,6248 | 19,8827 | 18,9078 | 23,7505 | 22,4539 | 22,0790 |
| 21,0281 | 22,1696 | 20,8820 | 19,3440 | 19,1537 | 23,5877 | 22,7654 | 22,3149 | |
| 21,1787 | 21,9149 | 20,8096 | 19,0287 | 20,1436 | 25,5499 | 23,8626 | 23,8251 | |
| 21,3287 | 21,5062 | 20,4549 | 18,7512 | 19,6614 | 24,1258 | 23,0498 | 23,1506 | |
| Glutapyrone 0.5 mg/kg | 21,0105 | 21,8076 | 20,5749 | 18,8806 | 19,3639 | 24,6586 | 23,1130 | 22,9958 |
| 20,6910 | 21,6908 | 20,2656 | 18,6775 | 19,6876 | 25,6195 | 23,7417 | 24,0393 | |
| 21,1142 | 21,4909 | 20,2270 | 18,7062 | 19,4588 | 24,3839 | 23,1421 | 22,8727 | |
| 21,3715 | 21,4707 | 20,0467 | 18,8292 | 19,4118 | 24,7691 | 23,3703 | 23,2199 | |
Table 2.
Liver real-time PCR cycle threshold (Ct) values
| Kidneys | Liver | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| RNSpolII | Psma3 | Psmb5 | Psmc6 | Psma6 | RNSpolII | Psma3 | Psmb5 | Psmc6 | Psma6 | |
| Control | 20,5970 | 22,5839 | 19,2551 | 19,1623 | 19,9986 | 19,8560 | 23,0557 | 21,1670 | 18,6385 | 19,8489 |
| 20,4384 | 22,5746 | 19,0794 | 18,8645 | 19,5620 | 19,5794 | 22,9181 | 21,1984 | 18,6208 | 19,6455 | |
| 20,4358 | 22,5837 | 19,1140 | 18,8865 | 19,7310 | 19,9334 | 23,3011 | 21,1796 | 19,6168 | 20,4226 | |
| 20,5461 | 22,7381 | 19,0685 | 19,1585 | 19,7124 | 19,7203 | 23,1264 | 21,1636 | 19,4941 | 20,2836 | |
| 20,3772 | 22,3517 | 19,0689 | 19,2026 | 20,2427 | 19,1888 | 22,9224 | 21,0717 | 19,1761 | 19,8373 | |
| 20,3640 | 22,2591 | 18,9613 | 18,9177 | 19,7695 | 19,7114 | 23,1850 | 21,1812 | 19,1708 | 20,0905 | |
| 20,4210 | 22,1444 | 19,0667 | 18,8171 | 19,3981 | 19,8140 | 23,5071 | 21,3909 | 19,3937 | 20,2497 | |
| 20,5429 | 22,3808 | 19,2618 | 20,5558 | 19,7631 | 23,3798 | 21,3486 | 19,0927 | 20,2443 | ||
| 20,4095 | 22,3303 | 18,9823 | 20,3727 | 19,6435 | 23,5475 | 21,2771 | 19,3322 | 20,4155 | ||
| 19,9648 | 22,0525 | 18,7116 | 19,2846 | |||||||
| AV-153-Na 0.05 mg/kg | 21,1756 | 22,9314 | 19,3199 | 19,5803 | 20,0066 | 19,3193 | 22,6985 | 21,0440 | 18,3758 | 18,9400 |
| 22,0513 | 23,4752 | 19,6423 | 19,9317 | 20,6172 | 18,8415 | 22,8245 | 20,9705 | 18,3795 | 19,0355 | |
| 21,4856 | 23,2240 | 19,4647 | 19,6623 | 19,8643 | 19,5196 | 22,7070 | 21,0585 | 18,3857 | 19,0187 | |
| 21,6234 | 23,2381 | 19,5993 | 19,5928 | 19,6469 | 18,8631 | 22,7883 | 20,9608 | 18,3031 | 18,9503 | |
| AV-153-Na 0.5 mg/kg | 21,9367 | 23,2775 | 19,4898 | 19,8077 | 20,0014 | 19,6059 | 22,7776 | 20,9764 | 18,4171 | 19,0567 |
| 21,6564 | 23,2385 | 19,5634 | 19,4900 | 19,8338 | 19,6035 | 22,8215 | 21,0093 | 18,4078 | 19,0701 | |
| 21,2077 | 23,2004 | 19,4074 | 19,5859 | 19,9741 | 19,5527 | 22,8589 | 20,9877 | 18,3254 | 19,1177 | |
| AV-153-Ca 0.05 mg/kg | 19,7019 | 22,0370 | 19,1800 | 18,7166 | 19,3175 | 19,3393 | 22,9415 | 20,8086 | 19,1408 | 19,8715 |
| 19,8564 | 22,2813 | 19,3695 | 18,7706 | 19,2143 | 19,6280 | 23,3113 | 21,0154 | 19,1477 | 20,3142 | |
| 20,1111 | 21,8349 | 19,0583 | 18,6593 | 19,3757 | 19,3697 | 22,8512 | 20,9235 | 18,9642 | 20,2187 | |
| 19,6506 | 23,1935 | 21,0658 | 19,3068 | 20,0824 | ||||||
| AV-153-Ca 0.5 mg/kg | 20,3086 | 22,0432 | 19,5138 | 18,8374 | 19,3591 | 19,4528 | 22,9892 | 20,7577 | 19,0212 | 19,3202 |
| 20,5201 | 22,7760 | 18,8733 | 19,2125 | 20,2666 | 19,5880 | 23,2170 | 21,2606 | 19,0527 | 19,6300 | |
| 19,5801 | 21,8729 | 18,8382 | 18,7051 | 19,3437 | 18,8662 | 23,0295 | 21,3512 | 18,8270 | 19,3601 | |
| 20,1326 | 22,0602 | 18,9902 | 18,8147 | 19,6876 | 19,5368 | 23,2560 | 21,5740 | 19,0952 | 19,4394 | |
| 20,1226 | 21,8402 | 18,8235 | 18,8101 | 19,4492 | 19,5666 | 23,0126 | 21,1141 | 18,7163 | 19,2355 | |
Statistics
Reference gene stability was analysed with BestKeeper provided in RefFinder (https://www.heartcure.com). Among studied organs, standard deviation (SD) values ranged from 0.22 to 0.54, and the coefficient of variance (CV) ranged from 1.13 to 2.63. These values are consistent with those reported for stable housekeeping genes (19). Gene expression data were expressed using the 2-ΔΔCt method – mean fold difference with standard error of the mean (SEM) (20). The P values were calculated from delta values using one-way ANOVA followed by Dunnett’s test for multiple comparisons between the groups. In all tests, the P value of <0.05 was considered statistically significant. All analyses were run on the GraphPad Prism 6 version 6.01 software (GraphPad Software, San Diego, CA, USA).
Results
Metcarbatone
In the kidney, metcarbatone at both doses significantly increased the expression of the Psma3, Psmb5, and Psmc6 genes (Table 3). The higher dose produced a more pronounced effect for Psma3 and Psmb5. The increase ranged from 1.43-fold with Psmc6 (0.05 mg/kg: P=0.010; 0.5 mg/kg: P=0.015) to 1.69-fold with Psmb5 (0.5 mg/kg: P=0.001). In the blood, metcarbatone significantly decreased the Psmc6 gene expression by 0.60 (0.5 mg/kg: P=0.032).
Table 3.
The effect of metcarbatone on Psma3, Psmb5 and Psmc6 gene expression in the kidney and blood
| Metcarbatone (mg/kg) | Kidney fold difference (SEM range) | Blood fold difference (SEM range) | |
|---|---|---|---|
| Psma3 | Control | 1.00 (0.93–1.07) | 1.00 (0.87–1.14) |
| 0.05 | 1.44 (1.34–1.54)* | 0.76 (0.56–1.04) | |
| 0.50 | 1.61 (1.50–1.74)** | 0.53 (0.48–0.58) | |
| Psmb5 | Control | 1.00 (0.93–1.08) | 1.00 (0.84–1.18) |
| 0.05 | 1.51 (1.40–1.63)** | 0.55 (0.39–0.77) | |
| 0.50 | 1.69 (1.60–1.79)** | 0.58 (0.51–0.67) | |
| Psmc6 | Control | 1.00 (0.94–1.07) | 1.00 (0.90–1.12) |
| 0.05 | 1.43 (1.34–1.54)* | 0.83 (0.68–1.01) | |
| 0.50 | 1.43 (1.34–1.53)* | 0.60 (0.55–0.64)* |
*P<0.05 and **P<0.01 compared to control
Etcarbatone
The Psma3, Psmb5, and Psmc6 gene expression significantly increased in the kidney (Table 4), from 1.36-fold at the higher dose for Psma3 (P=0.046) to 1.82-fold at the lower dose for Psmb5 (P=0.002). No significant differences were detected in the blood.
Table 4.
The effect of etcarbatone on Psma3, Psmb5 and Psmc6 gene expression in the kidney and blood
| Etcarbatone (mg/kg) | Kidney fold difference (SEM range) | Blood fold difference (SEM range) | |
|---|---|---|---|
| Psma3 | Control | 1.00 (0.93–1.07) | 1.00 (0.87–1.14) |
| 0.05 | 1.46 (1.29–1.65)* | 0.61 (0.52–0.70) | |
| 0.50 | 1.36 (1.29–1.43)* | 0.85 (0.84–0.87) | |
| Psmb5 | Control | 1.00 (0.93–1.08) | 1.00 (0.84–1.18) |
| 0.05 | 1.82 (1.56–2.12)** | 0.45 (0.37–0.55) | |
| 0.50 | 1.61 (1.52–1.71)** | 0.66 (0.59–0.74) | |
| Psmc6 | Control | 1.00 (0.94–1.07) | 1.00 (0.90–1.12) |
| 0.05 | 1.72 (1.52–1.95)** | 0.79 (0.71–0.88) | |
| 0.50 | 1.62 (1.53–1.72)** | 1.07 (0.99–1.17) |
*P<0.05 and **P<0.01 compared to control
Styrylcarbatone
Styrylcarbatone significantly increased the expression of the Psma3, Psmb5 and Psmc6 genes (Table 5). It was the most pronounced for Psmc6 – up to 2.05-fold at the lower dose (P<0.0001). No significant differences were detected in the blood.
Table 5.
The effect of styrylcarbatone on Psma3, Psmb5 and Psmc6 gene expression in the kidney and blood
| Styrylcarbatone (mg/kg) | Kidney fold difference (SEM range) | Blood fold difference (SEM range) | |
|---|---|---|---|
| Psma3 | Control | 1.00 (0.93–1.07) | 1.00 (0.87–1.14) |
| 0.05 | 1.65 (1.53–1.79)** | 0.99 (0.78–1.26) | |
| 0.50 | 1.53 (1.43–1.64)** | 1.13 (0.93–1.37) | |
| Psmb5 | Control | 1.00 (0.93–1.08) | 1.00 (0.84–1.18) |
| 0.05 | 1.63 (1.39–1.90)** | 0.87 (0.64–1.18) | |
| 0.50 | 1.41 (1.36–1.47)* | 1.18 (0.97–1.44) | |
| Psmc6 | Control | 1.00 (0.94–1.07) | 1.00 (0.90–1.12) |
| 0.05 | 2.05 (1.98–2.13)*** | 1.09 (0.90–1.32) | |
| 0.50 | 1.83 (1.75–1.91)*** | 1.08 (0.84–1.39) |
*P<0.05, **P<0.01, and ***P<0.0001 compared to control
Glutapyrone
In the kidney, the higher dose of glutapyrone significantly increased Psmb5 expression 1.73-fold (P=0.003; Table 6) and Psmc6 expression up to 1.59-fold (P=0.004). In the blood, the lower dose of glutapyrone increased Psmb5 expression 2.04-fold (P=0.036).
Table 6.
The effect of glutapyrone on Psma3, Psmb5 and Psmc6 gene expression in the kidney and blood
| Glutapyrone (mg/kg) | Kidney fold difference (SEM range) | Blood fold difference (SEM range) | |
|---|---|---|---|
| Psma3 | Control | 1.00 (0.93–1.07) | 1.00 (0.87–1.14) |
| 0.05 | 1.10 (0.95–1.26) | 1.49 (1.27–1.74) | |
| 0.50 | 1.21 (1.05–1.39) | 0.99 (0.86–1.14) | |
| Psmb5 | Control | 1.00 (0.93–1.08) | 1.00 (0.84–1.18) |
| 0.05 | 1.40 (1.26–1.55) | 2.04 (1.94–2.14)* | |
| 0.50 | 1.73 (1.49–2.01)** | 1.66 (1.56–1.77) | |
| Psmc6 | Control | 1.00 (0.94–1.07) | 1.00 (0.90–1.12) |
| 0.05 | 1.46 (1.28–1.66)* | 1.30 (1.19–1.42) | |
| 0.50 | 1.59 (1.46–1.73)** | 0.96 (0.84–1.11) |
*P<0.05 and **P<0.01 compared to control
AV-153-Na
AV-153-Na significantly increased the expression of Psma3, Psmb5, Psmc6, and Psma6 at both doses (Table 7) in the kidney. The higher dose resulted in the highest (2.17-fold) increase in Psma6 (P=0.0007), while the lower dose increased it 1.91-fold (P=0.0002). Psmc6 gene expression increased 1.61-fold at the higher dose (P=0.017), but the lower dose decreased the Psmb5 gene expression by 0.79 (P=0.029).
Table 7.
The effect of AV-153-Na on Psma3, Psmb5, Psmc6 and Psma6 gene expression in the kidney and liver
| AV-153-Na (mg/kg) | Kidneys fold difference (SEM range) | Liver fold difference (SEM range) | |
|---|---|---|---|
| Psma3 | Control | 1.00 (0.97–1.03) | 1.00 (0.95–1.05) |
| 0.05 | 1.28 (1.21–1.35)* | 0.94 (0.82–1.08) | |
| 0.50 | 1.28 (1.12–1.46)* | 1.23 (1.19–1.26) | |
| Psmb5 | Control | 1.00 (0.99–1.02) | 1.00 (0.94–1.04) |
| 0.05 | 1.64 (1.51–1.78)*** | 0.79 (0.71–0.87)* | |
| 0.50 | 1.68 (1.47–1.91)*** | 1.09 (1.08–1.11) | |
| Psmc6 | Control | 1.00 (0.97–1.03) | 1.00 (0.92–1.09) |
| 0.05 | 1.39 (1.29–1.51)** | 1.19 (1.07–1.33) | |
| 0.50 | 1.47 (1.30–1.66)** | 1.61 (1.59–1.62)* | |
| Psma6 | Control | 1.00 (0.92–1.08) | 1.00 (0.94–1.06) |
| 0.05 | 2.00 (1.78–2.25)*** | 1.49 (1.32–1.68)** | |
| 0.50 | 2.17 (1.87–2.52)*** | 1.91 (1.86–1.95)*** |
*P<0.05, **P<0.01, and ***P<0.001 compared to control
AV-153-Ca
In the kidney, AV-153-Ca significantly affected only the Psmb5 gene expression, which decreased by 0.62 at the lower dose (P=0.0043; Table 8). In the blood, Psma6 gene expression increased 1.35-fold at the lower dose. Other tested genes were not affected significantly.
Table 8.
The effect of AV-153-Ca on Psma3, Psmb5, Psmc6 and Psma6 gene expression in the kidney and liver
| AV-153-Ca (mg/kg) | Kidney fold difference (SEM range) | Liver fold difference (SEM range) | |
|---|---|---|---|
| Psma3 | Control | 1.00 (0.97–1.03) | 1.00 (0.95–1.05) |
| 0.05 | 0.89 (0.76–1.03) | 0.96 (0.94–0.99) | |
| 0.50 | 1.00 (0.92–1.09) | 0.89 (0.81–0.97) | |
| Psmb5 | Control | 1.00 (0.99–1.02) | 1.00 (0.94–1.04) |
| 0.05 | 0.62 (0.55–0.71)** | 1.05 (1.03–1.08) | |
| 0.50 | 0.85 (0.75–0.95) | 0.82 (0.71–0.95) | |
| Psmc6 | Control | 1.00 (0.97–1.03) | 1.00 (0.92–1.09) |
| 0.05 | 0.85 (0.77–0.93) | 0.89 (0.86–0.93) | |
| 0.50 | 0.90 (0.84–0.96) | 0.96 (0.88–1.05) | |
| Psma6 | Control | 1.00 (0.92–1.08) | 1.00 (0.94–1.06) |
| 0.05 | 1.03 (0.96–1.11) | 0.87 (0.82–0.93) | |
| 0.50 | 0.98 (0.89–1.07) | 1.35 (1.22–1.48)* |
*P<0.05 and **P<0.01 compared to control
Discussion
Most of the tested 1,4-DHP derivatives increased gene expression levels in the kidney but were mainly without a significant effect in the blood and liver. The general sensitivity of the kidney cells to 1,4‑DHP could simply be explained by accumulation of the compounds in the kidney before excretion, but there are no data to support it.
Comparing the effects of 1,4‑DHP derivatives on different proteasome subunit genes, we noticed that subunit mRNA expression did not follow a uniform pattern. Other authors have also reported divergent effects of drugs on different proteasome subunit gene expression. For example, cocaine mainly upregulated the PSMB1 and PSMA5 subunits and downregulated the PSMA6 subunit but did not affect the PSMB2 and PSMB5 subunits (21).
In the kidney, metcarbatone, etcarbatone, styrylcarbatone, and AV-153-Na increased the expression of all analysed genes, but glutapyrone and AV-153-Ca showed varying effects. Glutapyrone did not affect the expression of Psma3, which codes for the outer ring subunit of the 20S proteasome, but did increase the expression of Psmc6, which encodes for the subunit of the 19S regulatory complex. At the higher dose glutapyrone also increased the expression of Psmb5, which codes for the inner ring subunit of the 20S proteasome. Interestingly, while AV-153-Na increased Psmb5 expression in the kidney, AV-153-Ca decreased it. Furthermore, AV-153-Ca was the only compound that decreased the expression of any of the tested genes in the kidney.
In the blood, glutapyrone increased only Psmb5 expression. An earlier study (15) reported that glutapyrone increased the mRNA expression of Psma6, another subunit of the outer ring of the 20S proteasome, both in the kidney and blood. Metcarbatone at the higher dose decreased Psmc6 expression in blood.
In the liver, AV-153-Na upregulated the expression of Psma6 and also of Psmc6 at the higher dose but downregulated Psmb5 at the lower dose. The higher dose of AV-153-Ca increased only Psma6 expression.
1,4‑DHP derivatives as prospective drugs have already shown antioxidant activities and a wide range of antiaging, antibacterial, anticancer, and neuroprotective actions (10). Glutapyrone, a representative of the novel group of 1,4‑DHP derivatives with weak Ca2+ channel blocker activity has very low toxicity and multiple pharmacological properties, including concomitant effects on multiple neurotransmitter systems and antioxidant activities (22). Carbatone, another compound of this group, administered orally, showed fast absorption in the gastrointestinal tract and 62 % bioavailability. It quickly spreads across tissues and is excreted mostly through urine and faeces. This group of 1,4-DHP derivatives seems to have a very low cytotoxicity at the tested doses (unpublished data).
It also seems that the protective antioxidant activities of 1,4-DHP derivatives are achieved by targeting the mitochondria. They might be working through direct scavenging of reactive oxygen species and decomposition of hydrogen peroxide. Furthermore, they stimulate cell growth and differentiation (23). An in vitro study in human osteoblast-like cells treated with 1,4-DHP derivatives (23) demonstrated de novo glutathione synthesis, indicating the involvement of the NRF2 signalling pathway in the action of these compounds. It also suggested that the bioactivity of 1,4-DHP derivatives is associated with 4-hydroxynonenal and related second messengers of free radicals, but precise bioactivity mechanisms remain to be elucidated. The increase in proteasomal gene expression by 1,4-DHP derivatives may have similar beneficial mechanisms as those reported for antioxidants, which elevate transcription levels of 26S proteasome subunits responsible for removal of damaged proteins and attenuating the progression of human diseases related to oxidative stress (24).
The downregulation of UPS genes in the liver seems to correspond with age. In old mice, this downregulation was reported to lead to the accumulation of IκBα in the cytoplasm, which prevented the activation of the NF-κB protein, which is important for hepatocyte survival and liver health (25). Older age also seems to be associated with lower mRNA levels of both proteasome beta subunits, which are directly involved in the proteolytic function of the proteasome and antioxidant activity (26), but some healthy centenarians were reported to have proteasome subunit mRNA levels close to young donors. Furthermore, one study showed that a stable transfection of either PSMB1 or PSMB5 enhanced proteasome function and resistance to oxidative stress (27). This is why our findings of increased proteasome subunit gene expression by 1,4‑DHP derivatives seem promising in terms of pharmacotherapy.
Downregulated proteasomal gene expression is also associated with several pathologies. For instance, in patients with schizophrenia, dentate granule neurons showed decreased expression of several proteasome subunit and other genes involved in protein processing by proteasomes and ubiquitin, resulting in a deficient ubiquitin-proteasome function that can lead to reduced neuron responsiveness (28). In patients with Parkinson’s disease, both catalytic and regulatory subunits of the UPS, including the PSMA3 gene analysed in this study, showed decreased gene expression in substantia nigra (29). This might lead to decreased levels of the 26S proteasome complex, insufficient degradation of short-lived proteins such as cyclins, and accumulation of ubiquitinated proteins, which can eventually result in dopaminergic neuronal damage. In this kind of pathologies, the potential of 1,4-DHP derivatives to increase proteasomal gene expression might lead to restored proteasome function and therapeutic effect. However, additional research is needed to determine whether 1,4‑DHP derivatives increase proteasomal protein expression in the same way as they increase gene expression.
To sum up, our research has confirmed the ability of several 1,4-DHP derivatives to increase the expression of proteasome subunit genes. This might be a promising property for the development of drugs for conditions associated with impaired proteasomal functions and low mRNA levels of proteasome subunits.
Acknowledgements
This work was supported from the Grant of the Latvian Council of Science 278/2012 and the National Research Program “Biomedicine 2014”.
Footnotes
Conflicts of interest
The authors declare that they have no conflict of interest.
References
- 1.Klaips CL, Jayaraj GG, Hartl FU. Pathways of cellular proteostasis in aging and disease. J Cell Biol. 2018;217:51–63. doi: 10.1083/jcb.201709072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Albornoz N, Bustamante H, Soza A, Burgos P. Cellular responses to proteasome inhibition: molecular mechanisms and beyond. Int J Mol Sci. 2019;20(14):3379. doi: 10.3390/ijms20143379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Motosugi R, Murata S. Dynamic regulation of proteasome expression. Front Mol Biosci. 2019;6:30. doi: 10.3389/fmolb.2019.00030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Marshall RS, Vierstra RD. Dynamic regulation of the 26S proteasome: from synthesis to degradation. Front Mol Biosci. 2019;6:40. doi: 10.3389/fmolb.2019.00040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sherman DJ, Li J. Proteasome inhibitors: harnessing proteostasis to combat disease. Molecules. 2020;25(3):671. doi: 10.3390/molecules25030671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Coleman RA, Trader DJ. Methods to discover and evaluate proteasome small molecule stimulators. Molecules. 2019;24(12):2341. doi: 10.3390/molecules24122341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dammann P. Slow aging in mammals - Lessons from African mole-rats and bats. Semin Cell Dev Biol. 2017;70:154–63. doi: 10.1016/j.semcdb.2017.07.006. [DOI] [PubMed] [Google Scholar]
- 8.Cuanalo-Contreras K, Moreno-Gonzalez I. Natural products as modulators of the proteostasis machinery: implications in neurodegenerative diseases. Int J Mol Sci. 2019;20(19):4666. doi: 10.3390/ijms20194666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang Y, Loh C, Chen J, Mainolfi N. Targeted protein degradation mechanisms. Drug Discov Today Technol. 2019;31:53–60. doi: 10.1016/j.ddtec.2019.01.001. [DOI] [PubMed] [Google Scholar]
- 10.Velena A, Zarkovic N, Gall Troselj K, Bisenieks E, Krauze A, Poikans J, Duburs G. 1,4‑Dihydropyridine derivatives: dihydronicotinamide analogues-model compounds targeting oxidative stress. Oxid Med Cell Longev. 2016;2016:1892412. doi: 10.1155/2016/1892412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bruvere I, Bisenieks E, Poikans J, Uldrikis J, Plotniece A, Pajuste K, Rucins M, Vigante B, Kalme Z, Gosteva M, Domracheva I, Velena A, Vukovic T, Milkovic L, Duburs G, Zarkovic N. Dihydropyridine derivatives as cell growth modulators in vitro. O x i d Med Cell Longev. 2017;2017:4069839. doi: 10.1155/2017/4069839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Leonova E, Rostoka E, Sauvaigo S, Baumane L, Selga T, Sjakste N. Study of interaction of antimutagenic 1,4-dihydropyridine AV-153-Na with DNA-damaging molecules and its impact on DNA repair activity. PeerJ. 2018;6:e4609. doi: 10.7717/peerj.4609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Leonova E, Ošiņa K,, Duburs G, Bisenieks E, Germini D, Vassetzky Y, Sjakste N. Metal ions modify DNA-protecting and mutagen-scavenging capacities of the AV-153 1,4-dihydropyridine. Mutat Res. 2019;845:403077. doi: 10.1016/j.mrgentox.2019.06.007. [DOI] [PubMed] [Google Scholar]
- 14.Ošiņa K, Rostoka E, Isajevs S, Sokolovska J, Sjakste T, Sjakste N. Effects of an antimutagenic 1,4-dihydropyridine AV-153 on expression of nitric oxide synthases and DNA repair-related enzymes and genes in kidneys of rats with a streptozotocin model of diabetes mellitus. Basic Clin Pharmacol Toxicol. 2016;119:458–63. doi: 10.1111/bcpt.12617. [DOI] [PubMed] [Google Scholar]
- 15.Ošiņa K, Leonova E, Isajevs S, Baumane L, Rostoka E, Sjakste T, Bisenieks E, Duburs G, Vīgante B, Sjakste N. Modifications of expression of genes and proteins involved in DNA repair and nitric oxide metabolism by carbatonides [disodium-2,6-dimethyl-1,4- dihydropyridine-3,5-bis(carbonyloxyacetate) derivatives] in intact and diabetic rats. Arh Hig Rada Toksikol. 2017;68:212–27. doi: 10.1515/aiht-2017-68-2945. [DOI] [PubMed] [Google Scholar]
- 16.Ošiņa K, Rostoka E, Sokolovska J, Paramonova N, Bisenieks E, Duburs G, Sjakste N, Sjakste T. 1,4-dihydropyridine derivatives without Ca2+-antagonist activity up-regulate Psma6 mRNA expression in kidneys of intact and diabetic rats. Cell Biochem Funct. 2016;34:3–6. doi: 10.1002/cbf.3160. [DOI] [PubMed] [Google Scholar]
- 17.European Convention for the Protection of Vertebrate Animals Used for Experimental and other Scientific Purposes [displayed 31 May. 2021. https://www.coe.int/en/web/conventions/full-list/-/conventions/treaty/123 Council of Europe. Available at.
- 18.Ye J, Coulouris G, Zaretskaya I, Cutcutache I, Rozen S, Madden TL. Primer-BLAST: A tool to design target-specific primers for polymerase chain reaction. BMC Bioinformatics. 2012;13:134. doi: 10.1186/1471-2105-13-134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sokolovska J, Isajevs S, Sugoka O, Sharipova J, Lauberte L, Svirina D, Rostoka E, Sjakste T, Kalvinsh I, Sjakste N. Correction of glycaemia and GLUT1 level by mildronate in rat streptozotocin diabetes mellitus model. Cell Biochem Funct. 2011;29:55–63. doi: 10.1002/cbf.1719. [DOI] [PubMed] [Google Scholar]
- 20.Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP. Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: BestKeeper-Excel-based tool using pair-wise correlations. Biotechnol L e t t. 2 0 0 4 ; 2 6 : 5 0 9 – 1 5 . d o i : 10.1023/b:bile.0000019559.84305.47. [DOI] [PubMed]
- 21.Taylor SC, Nadeau K, Abbasi M, Lachance C, Nguyen M, Fenrich J. The ultimate qPCR experiment: producing publication quality, reproducible data the first time. Trends Biotechnol. 2 0 1 9 ; 3 7 : 7 6 1 – 7 4 . doi: 1 0 . 1 0 1 6 / j . tibtech.2018.12.002. [DOI] [PubMed]
- 22.Caputi FF, Carboni L, Mazza D, Candeletti S, Romualdi P. Cocaine and ethanol target 26S proteasome activity and gene expression in neuroblastoma cells. Drug Alcohol Depend. 2016;161:265–75. doi: 10.1016/j.drugalcdep.2016.02.012. [DOI] [PubMed] [Google Scholar]
- 23.Misane I, Klusa V, Dambrova M, Germane S, Duburs G, Bisenieks E, Rimondini R, Ogren SO. “Atypical” neuromodulatory profile of glutapyrone, a representative of a novel ‘class’ of amino acid-containing dipeptide-mimicking 1,4-dihydropyridine (DHP) compounds: in vitro and in vivo studies. Eur Neuropharmacol. 1998;8:329–47. doi: 10.1016/s0924-977x(97)00097-7. [DOI] [PubMed] [Google Scholar]
- 24.Milkovic L, Vukovic T, Zarkovic N, Tatzber F, Bisenieks E, Kalme Z, Bruvere I, Ogle Z, Poikans J, Velena A, Duburs G. Antioxidative 1,4-dihydropyridine derivatives modulate oxidative stress and growth of human osteoblast-like cells in vitro. Antioxidants (Basel) 2018;7(9):123. doi: 10.3390/antiox7090123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kwak MK, Wakabayashi N, Greenlaw JL, Yamamoto M, Kensler TW. Antioxidants enhance mammalian proteasome expression through the Keap1-Nrf2 signaling pathway. Mol Cell Biol. 2003;23:8786–94. doi: 10.1128/MCB.23.23.8786-8794.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Huber N, Sakai N, Eismann T, Shin T, Kuboki S, Blanchard J, Schuster R, Edwards MJ, Wong HR, Lentsch AB. Age-related decrease in proteasome expression contributes to defective nuclear factor-kappaB activation during hepatic ischemia/reperfusion. Hepatology. 2009;49:1718–28. doi: 10.1002/hep.22840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chondrogianni N, Petropoulos I, Franceschi C, Friguet B, Gonos ES. Fibroblast cultures from healthy centenarians have an active proteasome. Exp Gerontol. 2000;35:721–8. doi: 10.1016/s0531-5565(00)00137-6. [DOI] [PubMed] [Google Scholar]
- 28.Chondrogianni N, Stratford FL, Trougakos IP, Friguet B, Rivett AJ, Gonos ES. Central role of the proteasome in senescence and survival of human fibroblasts: induction of a senescence-like phenotype upon its inhibition and resistance to stress upon its activation. J Biol Chem. 2003;278:28026. doi: 10.1074/jbc.M301048200. –. [DOI] [PubMed] [Google Scholar]
- 29.Altar CA, Jurata LW, Charles V, Lemire A, Liu P, Bukhman Y, Young TA, Bullard J, Yokoe H, Webster MJ, Knable MB, Brockman JA. Deficient hippocampal neuron expression of proteasome, ubiquitin, and mitochondrial genes in multiple schizophrenia cohorts. Biol Psychiatry. 2005;58:85–96. doi: 10.1016/j.biopsych.2005.03.031. [DOI] [PubMed] [Google Scholar]
- 30.Grünblatt E, Mandel S, Jacob-Hirsch J, Zeligson S, Amariglo N, Rechavi G, Li J, Ravid R, Roggendorf W, Riederer P, Youdim MBH. Gene expression profiling of parkinsonian substantia nigra pars compacta; alterations in ubiquitin-proteasome, heat shock protein, iron and oxidative stress regulated proteins, cell adhesion/cellular matrix and vesicle trafficking genes. J Neural Transm. 2004;111:1543–73. doi: 10.1007/s00702-004-0212-1. [DOI] [PubMed] [Google Scholar]


