Abstract
Severe coronavirus disease-2019 (COVID-19) is frequently associated with microvascular thrombosis, especially in the lung, or macrovascular thrombosis, mainly venous thromboembolism, which significantly contributes to the disease mortality burden. COVID-19 patients also exhibit distinctive laboratory abnormalities that are compatible with a prothrombotic state. The key event underlying COVID-19-associated thrombotic complications is an excessive host inflammatory response to severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2) infection generating multiple inflammatory mediators, mainly cytokines and complement activation products. The latter, along with the virus itself, the increased levels of angiotensin II and hypoxia, drive the major cellular changes promoting thrombosis, which include: (1) aberrant expression of tissue factor by activated alveolar epithelial cells, monocytes-macrophages and neutrophils, and production of other prothrombotic factors by activated endothelial cells (ECs) and platelets; (2) reduced expression of physiological anticoagulants by dysfunctional ECs, and (3) suppression of fibrinolysis by the endothelial overproduction of plasminogen activator inhibitor-1 and, likely, by heightened thrombin-mediated activation of thrombin-activatable fibrinolysis inhibitor. Moreover, upon activation or death, neutrophils and other cells release nuclear materials that are endowed with potent prothrombotic properties. The ensuing thrombosis significantly contributes to lung injury and, in most severe COVID-19 patients, to multiple organ dysfunction. Insights into the pathogenesis of COVID-19-associated thrombosis may have implications for the development of new diagnostic and therapeutic tools.
Keywords: SARS-COV-2, Thrombosis, COVID, Infection, Prothrombotic state
Introduction
Coronavirus disease-2019 (COVID-19) is a viral illness caused by severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2). Since its emergence in late 2019, the disease has rapidly achieved pandemic proportions causing remarkably high mortality worldwide. Although most people infected with SARS-CoV-2 are totally asymptomatic or have a mild illness, some patients (about 5%) usually present with progressive respiratory failure (acute respiratory distress syndrome, ARDS), and even multiple organ dysfunction.1,2 Accumulating clinical and pathological evidence indicates that severe SARS-CoV-2 infection is frequently associated with a prothrombotic state which can manifest as microvascular or macrovascular thrombosis, and that these complications significantly contribute to the mortality burden of COVID-19 patients. Microvascular thrombosis occurs mainly in the lung, as documented by several autopsy reports.3–6 Indeed, in addition to diffuse alveolar damage, platelet-fibrin thrombi are frequently seen in the small pulmonary vasculature in almost all the examined lungs. Importantly, alveolar-capillary microthrombi were 9 times as prevalent in patients with Covid-19 as in patients who died from ARDS secondary to influenza A (H1N1) infection.7 Pulmonary microvascular thrombosis also appears more pronounced in severe SARS-CoV-2 infection than in other human coronavirus infections targeting the lower respiratory tract, namely SARS-CoV and Middle East respiratory syndrome coronavirus (MERS-CoV).8 In COVID-19 patients with more severe disease, thrombosis of the microcirculation may also be seen in other organs (heart, kidney, brain, and liver).4–6
Among macrovascular thrombotic events reported in COVID-19, venous thromboembolism (VTE), which includes deep vein thrombosis (DVT) and pulmonary embolism (PE) is the most frequent, with a cumulative incidence of 16,7 to 49% in critically ill patients admitted to the intensive care unit (ICU), and with PE being the most common complication.9–13 Notably, VTE may occur despite standard thromboprophylaxis. Moreover, COVID-19 ARDS patients develop more thrombotic complications, mainly PE, than non-COVID-19 ARDS patients, and patients suffering from a thrombotic complication had more than a 5-fold increase in all-cause mortality.10,12 Because the frequency of PE far exceeds that of DVT in most reports on COVID-19 patients, it has been proposed that the occlusion of pulmonary vessels in these patients results from pulmonary thrombosis rather than embolism.13,14 In hospitalized, non-severely ill patients receiving standard thromboprophylaxis, the incidence of VTE is obviously much lower, ranging from 0 to about 6%.9,14–16 Arterial thrombosis has also been reported in patients with COVID-19, including myocardial infarction,11,17 ischemic stroke11,18 and peripheral thrombosis,19,20 with rates < 3%.10,11,15 Patients with COVID-19 may also experience bleeding complications. A multicentre study of 400 hospitalized patients with COVID-19 reported an overall bleeding rate of 4.8% and a severe bleeding rate of 2.3%.15
Based on the extensive clinical evidence summarized above, thrombotic events emerge as critical issues in severe COVID-19 and can be listed among life-threatening complications of the disease. This implies that patients suffering from severe COVID-19 have haemostatic abnormalities that predispose to thrombosis, commonly referred to as hypercoagulability or prothrombotic state.
In this review, we will 1) shortly summarize the distinctive laboratory haemostatic abnormalities in patients with COVID-19, 2) discuss the possible pathogenetic mechanisms of COVID-19-associated thrombosis, and 3) describe the new diagnostic and therapeutic tools that are being developed.
Laboratory Haemostatic Abnormalities
Routine assays
The most frequent finding in patients with COVID-19-associated coagulopathy is an increased plasma D-dimer concentration, which is found in almost 50% of patients and has attracted particular attention because of its prognostic significance. Markedly higher D-dimer levels (usually more than three-fold the upper limit of normal) were consistently observed in severely affected patients (requiring critical care support) and in nonsurvivors. Significantly, exceedingly high D-dimer levels on hospital admission or a progressive elevation during the hospitalization are associated with an increased need for mechanical ventilation and an increased risk of death.21–24 Therefore, COVID-19 patients who have markedly raised D-dimer on admission should be carefully checked even in the absence of other laboratory abnormalities or severe symptoms because the presence of high D-dimer is strongly suggestive of clotting activation and increased thrombin generation.
Thrombocytopenia is uncommon in COVID-19 patients, and, when present, it is usually mild. Even in patients with the most severe illness, the platelet count is generally greater than 100×109/L,12,21, 23,25–27 and less than 5% of patients displays a lower count.23,25 In very advanced cases, a progressive decrease in platelet count may occur, indicating platelet consumption.21,22,26 However, despite the relatively small changes in platelet count in patients with COVID-19, two studies suggest that thrombocytopenia may have prognostic significance. Lippi et al., in a meta-analysis of nine studies, showed that the platelet count was significantly lower in patients with more severe COVID-19 and correlated with mortality.28 Moreover, in the most extensive so-far analysis of thrombocytopenia in patients with COVID-19 (1476 patients, 238 of whom were nonsurvivors), Yang et al. found a significant association between thrombocytopenia and in-hospital mortality.29 The latter study is also important because it confirms the relatively low frequency of thrombocytopenia which, with 125×109/L as the lower limit of the normal range, occurred in about 20% of the patients.
Prothrombin time (PT) is normal in non-critically ill COVID-19 patients, whereas it is only mildly prolonged in severe cases.22,23 When performed on admission, PT is more prolonged, albeit modestly, in patients who need critical care support and in nonsurvivors.23
Fibrinogen is markedly increased in Covid-19 patients,12,22,30,31 which likely reflects the inflammatory acute phase response. However, a sudden drop in fibrinogen level below 1.0 g/L has been reported in terminal COVID-19 patients shortly before they died.22
The coagulation abnormalities of patients with Covid-19 in relation to the disease severity are often heterogeneous and somewhat difficult to interpret, particularly with respect to platelet count and PT. A meta-analysis of 34 studies (with a pooled population of 6,492 COVID-19 patients) showed that elevated D-dimer levels, low platelet count, and prolonged PT occur more often in severe COVID-19 patients and in nonsurvivors.32 Therefore, monitoring these biomarkers in COVID-19 patients might aid in the early detection of severe disease. As a matter of fact, the guidance for the management of coagulopathy in COVID-19 released by the International Society on Thrombosis and Haemostasis (ISTH) recommends testing and monitoring routine haemostatic markers, including, in decreasing order of importance, D-dimer, prothrombin time, platelet count and fibrinogen.33 The alterations of routine haemostatic tests, when combined, are reminiscent of the pattern seen in DIC. However, there are significant differences from classical DIC associated with sepsis. First, thrombocytopenia is the most common haemostatic abnormality in the latter condition and is usually much more pronounced (≤ 50×109/L);34,35 of note, severe thrombocytopenia is strongly associated with mortality either when present at admission or when appearing during the hospital stay.35 In addition, patients with sepsis-associated DIC have much lower levels of several coagulation factors, including those involved in the PT assay, namely prothrombin, factor (F)V, FVII, FX, and fibrinogen, because of consumption, and a marked reduction in plasma levels of physiological anticoagulant proteins antithrombin (AT) and protein C (PC).36,37 These changes are almost absent in COVID-19 patients (see below). It is worth mentioning that one previous study22 reported that 71% of non-survivors COVID-19 patients fulfilled the ISTH criteria for DIC, compared with only 0.6% among survivors, suggesting that DIC is common in severe SARS-CoV-2 infection. However, several other investigators did not find evidence of DIC in their patients,12,15,30,31,38,39 leading to the conclusion that DIC is rare in COVID-19.
Other assays
Global coagulation assays on whole blood, including thromboelastography (TEG) and thromboelastometry (ROTEM), have consistently demonstrated a hypercoagulable profile in patients with COVID-19,30,31,38,40 except for one study reporting ROTEM parameters in the normal range.41 Thrombin generation assay (TGA), a global test performed in plasma with or without thrombomodulin, has been used by some investigators to evaluate the COVID-19-associated coagulopathy with results that are difficult to interpret because the assay is sensitive to anticoagulants and the majority of COVID-19 hospitalized patients receive either standard thromboprophylaxis with low molecular weight heparin (LMWH) or, in more severe cases, higher doses of LMWH or standard heparin. In general, TGA parameters were reported to be normal despite the use of anticoagulants,41,42 a finding interpreted as evidence of hypercoagulability.42 Using plasma samples from COVID-19 patients treated to neutralize anticoagulants prior to the assay, White et al.43 found normal thrombin generation in both critical and noncritical patients, whereas Hardy et al.44 observed increased thrombin potential over the first week after ICU admission.
Thrombin-antithrombin complex (TAT) and prothrombin fragment 1+2 (F1+2) have also been measured in COVID-19 patients as in vivo thrombin formation markers. TAT plasma concentration was elevated in some studies,41,45,46 with significantly higher values in ICU compared with non-ICU patients,45 but normal43 or moderately increased47 in others. F1+2 was normal41,43 or marginally increased.48 In the latter study, in a cohort of 114 critically ill COVID-19 patients, F1+2 levels were significantly higher in those with thrombosis and were more discriminant than D-dimer in identifying thrombotic manifestations. FVIII and von Willebrand Factor (vWF) are consistently and considerably elevated in COVID-19 patients, particularly in severe cases,12,31,41,45 likely reflecting endothelial cell activation (see next section). Taken together, these results indicate that most hospitalized COVID-19 patients, especially those with severe disease, have haemostatic abnormalities that are compatible with a prothrombotic state.
Pathophysiology
The urgent need to understand how SARS-CoV-2 induces a prothrombotic state and to develop effective therapeutic approaches for COVID-19-associated thrombosis has prompted an unprecedented number of studies. Despite this enormous effort, the mechanisms underpinning haemostatic abnormalities and thrombosis in COVID-19 patients still remain to be fully elucidated. Being COVID-19 a severe infection, it can be reasonably postulated that they may overlap, at least in part, with those of sepsis-induced DIC. Like classical bacterial sepsis, severe SARS-CoV-2 infection is characterized by an excessive inflammatory response of the host which is thought to drive, together with a virus-induced lowering of antiviral defences (low interferon response, lymphocytopenia), the progressive worsening of lung function and, in most severe cases with systemic inflammatory response, multiorgan failure ultimately leading to the death of COVID-19 patients. Several recent reviews have been published on this topic.49–51 Briefly, SARS-CoV-2, through its surface spike (S) protein, primarily infects alveolar epithelial cells, especially type 2 cells, which express the highest levels of angiotensin-converting enzyme 2 (ACE2), the best characterized entry receptor for the virus.52 This leads to cell activation and/or death by apoptosis and pyroptosis and to the release of damage-associated molecular patterns (DAMPs).
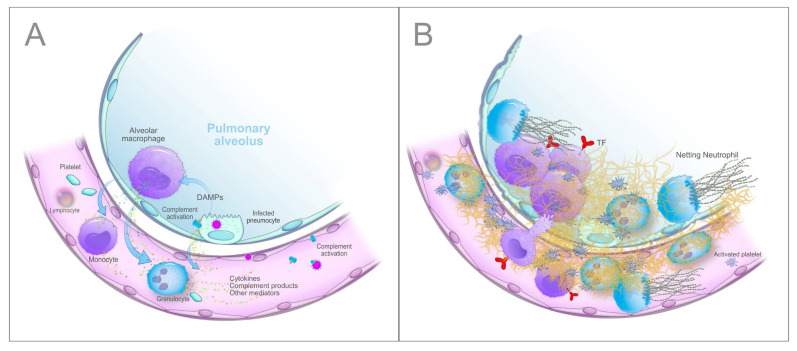
Given the close proximity to pneumocytes, alveolar macrophages are the first immune cells that recognize DAMPs and probably also the virus and/or its unique constituents (PAMPs, pathogen-associated molecular patterns) through specific receptors (PRRs, pattern recognition receptors, primarily the TLRs, Toll-like receptors), and respond with the synthesis and release of large amounts of proinflammatory mediators, mainly cytokines and chemokines. Cytokines secreted by alveolar macrophages, i.e., interleukin (IL)-6, IL-10 and tumour necrosis factor-alpha (TNF-α), have been detected in the lung by immunohistochemistry53,54 and increased circulating levels of numerous cytokines, including IL-6, IL-8, IL-1β, and TNF-α, that correlate with disease severity have been reported in COVID-19 patients.23,25,27,49 Moreover, SARS-Cov-2 activates the complement (C) alternative and lectin pathways through at least 2 surface proteins, i.e., the S protein, which, being heavily glycosylated with L-fucose and mannose, binds mannose-binding lectin (MBL), and the nucleocapsid protein (N protein), which binds MBL-associated serine protease 2 (MASP-2),55,56 thus generating other potent proinflammatory mediators including the anaphylatoxins C3a and C5a, C3b and C5b-9 complex (membrane attack complex, MAC). The involvement of the complement system in COVID-19 is strongly supported by the extensive deposition of C5b-9, C4d, and MASP-2 within the lung microvasculature.57 Strikingly, the SARS-CoV-2 S glycoprotein colocalized with C5b-9 and C4d in a subset of cases. In addition, patients with COVID-19 have elevated serum C5a and sC5b-9 levels.58,59 All the generated inflammatory mediators then cause the recruitment into the alveolar and interstitial spaces of monocytes-macrophages and neutrophils, which are activated and, in their turn, release additional cytokines (the so-called “cytokine storm”), proteases and other mediators, such as reactive oxygen species (ROS), which work in concert to trigger a self-amplifying inflammatory cascade eventually resulting in a further injury of alveolar epithelial cells and hypoxic respiratory failure. In this setting, the vascular endothelium also plays a pivotal role. ACE2 is widely expressed on endothelial cells, and the presence of SARS-CoV-2 elements within these cells has been well documented.60 Direct viral infection, combined with the milieu of proinflammatory mediators, will cause endothelial dysfunction/injury that strongly contributes to inflammation and tissue damage. Finally, platelets are activated by cytokines and complement products, as well as by the interaction with injured vessels and activated leukocytes, and release the content of their granules, including numerous proinflammatory mediators that further amplify the inflammatory reaction.61,62 As expected, COVID-19 patients exhibit a marked elevation of acute-phase reactants (e.g., C-reactive protein, fibrinogen and ferritin). A schematic summary of the host inflammatory response to SARS-CoV-2 infection is reported in Figure 1.
Figure 1.
A. Schematic overview of the main steps of the hyperinflammatory response to SARS-CoV-2 infection in the lung. B. Tissue damage and fibrin deposition in the lung induced by inflammatory cells recruited by cytokines and complement activation products. Details in the text.
In the light of these findings, a plausible hypothesis is that the plethora of inflammatory mediators generated during the persistent inflammatory status, along with the virus-derived components, drives thrombus formation in severe COVID-19 patients mainly through 1) the upregulation of the cellular procoagulant pathways, 2) the downregulation of physiological anticoagulant pathways and 3) the impairment of fibrinolysis. Virtually all cells participating in the inflammatory process, including lung epithelial cells, resident macrophages, monocytes, neutrophils, platelets, and endothelial cells, may variably contribute to these mechanisms.
Upregulation of Cellular Procoagulant Pathways: the Role of Tissue Factor
Currently, the aberrant in vivo expression of tissue factor (TF) is thought to play a pivotal role in almost all forms of thrombosis, including the sepsis-associated widespread microthrombosis.36,37,63 TF appears to be the major trigger of the coagulation cascade also in several viral infections as inferred from in vitro findings and animal studies.64,65
Lung epithelial cells
Lung epithelial cells are the first target of SARS-CoV-2. In vitro studies suggest that the basal expression of TF in these cells may be upregulated by infection with some viruses64 or exposure to a mixture of cytokines.66 Moreover, mice infected with influenza A virus H1N1 show both an increased TF expression in lung epithelial cells and signs of clotting activation.64 Concerning COVID-19, a preprint study suggests that lung epithelial cells infected in vitro with SARS-CoV-2 undergo transcriptional changes in several coagulation genes that make them prothrombotic.67 Specifically, TF is upregulated while two major coagulation inhibitors, TFPI (tissue factor pathway inhibitor) and protein S, are either unmodified or downregulated. Similar transcriptional changes were found ex vivo in cells isolated from bronchoalveolar lavage fluid (BALF) of COVID-19 patients, although the cell type(s) undergoing these changes were not identified. If confirmed, these findings could contribute to explain the initial fibrin deposition in the alveoli.
Monocytes and macrophages
Monocytes and macrophages have long been recognized as major players in the activation of blood coagulation, through the synthesis of TF in response to several inflammatory mediators. These cells, like many others, upon activation or during apoptosis, release small-membrane vesicles bearing TF, called microparticles (TF+ MPs) or microvesicles, that can be transferred to the surface of other cells via specific receptors (for instance, P-selectin glycoprotein ligand-1, PSGL-1 on leukocyte-derived MPs and P-selectin on activated platelets or endothelial cells), making the recipient cell capable of triggering and propagating coagulation.68 The best example illustrating the role of monocytes-macrophages in clotting activation is sepsis-associated DIC, in which the aberrant expression of TF by these cells has been amply documented both in experimental and human studies.36,37 TF can also be induced in these cells after viral infection.64 As mentioned above, monocytes and macrophages play a key role in the overwhelming inflammatory response observed in COVID-19 patients,53,69 being responsible, once activated, for the unrestrained release of cytokines, ROS and proteases, which sustain the inflammatory process and aggravate lung injury. Therefore, although direct proof is still lacking, it can be argued that, in patients with severe COVID-19, alveolar macrophages and recruited monocytes-macrophages synthesize and express active TF on their surface, in response to several inflammatory mediators, mainly cytokines and complement activation products (Figure 1B). Among cytokines, IL-6 appears of particular importance because a) it stimulates TF synthesis by human monocytes in vitro,70 and seems to be critical for in vivo activation of blood coagulation in animal models;71 b) it is present in the lung of patients with COVID-19, as shown by immunohistochemistry;53 c) its serum level is significantly elevated in nonsurvivors and progressively increases with illness deterioration.21,72 In addition, IL-6 is one of the key mediators of the cytokine storm and can suppress normal T cell activation, which may partly explain the occurrence of lymphopenia.49,72 Concerning the complement-derived mediators, C5a is known to induce TF expression in human monocytes,73,74 and its plasma level is significantly increased in patients with COVID-19.58,59 The possible involvement of monocyte TF in SARS-CoV-2 infection is supported by a recent study of Hottz et al.75 These investigators demonstrated that patients with severe COVID-19, but not those with mild disease or asymptomatic, exhibit platelet activation, as assessed by increased P-selectin and CD63 surface expression, increased plasma levels of thromboxane B2 (TXB2, a metabolite from platelet TXA2 synthesis), and a greater number of platelet-monocyte aggregates. Interestingly, exposure to plasma from severe COVID-19 patients increased the activation of control platelets in vitro. More relevant in the context of this discussion, platelet-monocyte interaction resulted in strong TF expression by the monocytes. Both platelet activation and monocyte TF expression were associated with fibrinogen and D-dimers levels, and were increased in patients requiring invasive mechanical ventilation or patients who died in hospital. Notably, platelets from severe COVID-19 patients were able to induce TF expression in monocytes from healthy volunteers, a phenomenon that was inhibited by platelet P-selectin neutralization or integrin αIIb/β3 blockade (with the aggregation inhibitor abciximab). The plasma factor(s) responsible for platelet activation and platelet-dependent monocyte TF expression were not identified, but they could well be cytokines and/or complement activation products. The concept that activated platelets form heterotypic aggregates with monocytes and neutrophils in the setting of infection and inflammation is rather well established76 and has been confirmed in COVID-19 patients also by other investigators.62 These aggregates might play a role in mechanical occlusion of the microvasculature in addition to altering the functional cell responses, eventually leading to TF expression. A definitive understanding of the in vivo expression of monocyte-macrophage TF and the role thereof in blood clotting activation and thrombosis associated with COVID-19 remains to be settled. In ARDS secondary to conditions other than COVID-19, studies performed with BALF and pulmonary tissues have consistently reported that activation of coagulation with fibrin formation is mainly due to TF upregulation in alveolar and recruited macrophages as well as in lung epithelial cells, likely as a response to inflammatory mediators.66,77,78 Therefore, studies at the tissue level, particularly in the lung, which is the main target organ of SARS-CoV-2, are warranted.
Neutrophils
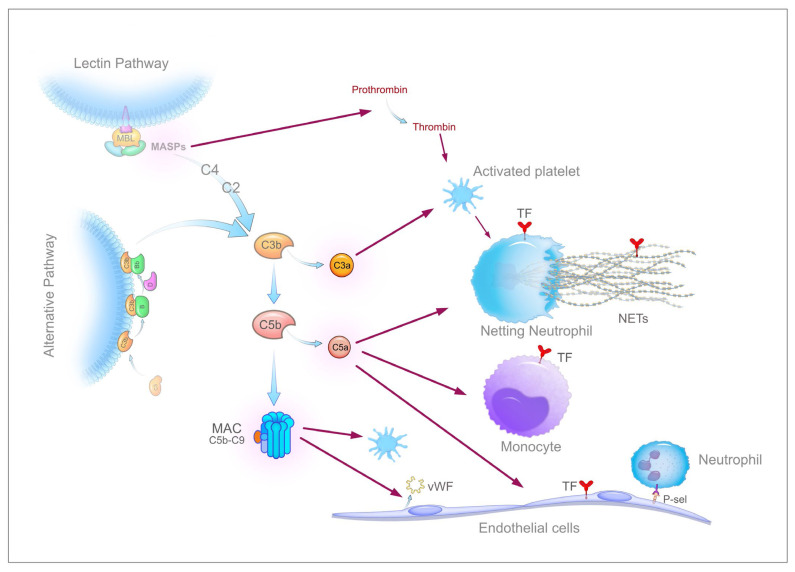
Increased neutrophil counts have been associated with disease severity and poor prognosis in COVID-1927,79 and the neutrophil-to-lymphocyte ratio is an independent risk factor for severe disease.80 In addition, neutrophil infiltration of pulmonary capillaries has been described in autopsy specimens.4,57 Neutrophils reportedly express TF in response to inflammatory stimuli.81,82 Another remarkable feature of activated neutrophils is the capacity to release neutrophil extracellular traps (NETs), a process named NETosis.83 NETs are networks of chromatin filaments made up of histones and DNA strands, decorated with proteins and lysosomal enzymes (myeloperoxidase, elastase and cathepsin G, among others) and are released by neutrophils upon exposure to a variety of stimuli such as microorganisms and their products, inflammatory mediators, including cytokines and complement activation products, and ROS. Noteworthily, activated platelets are potent inducers of NET formation as a consequence of their interaction with neutrophils. Extracellular traps can be actively extruded also by other activated innate immune cells, such as mast cells, eosinophils, and mononuclear phagocytes, and individual components (histones and DNA, mainly as nucleosomes) can be passively released by dying cells. NET formation involves the unwinding of nuclear DNA fibres and the nuclear membrane breakdown before the final active discharge in the extracellular milieu. This process is mediated by NF-kB (nuclear factor kappa-light-chain-enhancer of activated B cells) signalling, peptidyl arginine deiminase 4 (PAD4), and neutrophil elastase (NE), which cooperate to modify histones and enable DNA decondensation, and by ROS via NADPH oxidase. Originally characterized as a major innate immunity mechanism to trap and eventually neutralize invading microorganisms,83 NETs, when not properly regulated, have the potential to propagate inflammation, to cause cell and tissue injury, and to trigger thrombus formation, and thus they play a major role in numerous diseases,84 including viral infections.85 NETs are now emerging as important players in COVID-19 as well, as clearly suggested by several lines of evidence. First, numerous investigators demonstrated that patients with COVID-19 exhibit elevated serum or plasma levels of known markers of NETs, namely myeloperoxidase (MPO)-DNA complex, citrullinated histone H3 (cit-H3), and cell-free DNA.46,86–89 Cell-free DNA and/or MPO-DNA levels were found to be consistently correlated with disease severity, being higher in hospitalized patients undergoing mechanical ventilation than in those breathing room air,86,87,89 and in nonsurvivors as compared to survivors.87 Plasma NETs also correlate with respiratory failure severity and the Sequential Organ Failure Assessment (SOFA) score.87 In patients who recover from COVID-19, plasma NETs decrease to levels similar to those of healthy subjects, suggesting the normalization of NET formation.87 In a case-control study of hospitalized COVID-19 patients, the serum levels of NET markers and calprotectin, a marker of neutrophil activation, were associated with a higher risk of thrombotic events despite prophylactic anticoagulation.90 Interestingly, plasma or serum from patients with COVID-19 was able to stimulate NET release when added to neutrophils isolated from healthy donors,86,87 suggesting the presence of factors capable of triggering NETosis, such as cytokines and complement activation products. Second, neutrophils isolated from COVID-19 patients with severe disease presented signs of in vivo activation, for example, a significant decrease in granularity87 or a significant increase of “low-density granulocytes,” a specific form of neutrophils characterized by a high propensity to spontaneously form NETs.89 As a matter of fact, when cultured in vitro in the absence of any stimulus, these cells released markedly higher levels of NETs compared to normal neutrophils.86–88 Third, examination of autopsy lung specimens from COVID-19 patients by immunofluorescence revealed the presence of microvascular thrombi containing, in addition to platelets and fibrin, numerous neutrophils releasing NETs.87,88,91,92 Colocalization of cit-H3-positive neutrophils with platelets suggests that the interaction between these cells contributes to the release of NETs. Such interaction is further supported by the significantly higher levels of circulating platelet-neutrophil aggregates in COVID-19 patients compared with healthy adults.62,87 In a detailed immunohistochemical study of post-mortem lung specimens from 4 COVID-19 patients,92 NETs were consistently located in the airway compartment, often associated with fibrin that occluded some alveoli or bronchioles, in the interstitial compartment, especially in areas infiltrated by neutrophils and macrophages, and in the vascular compartment, mainly in arterioles containing neutrophil-rich microthrombi. Notably, NETs were not detected in lung specimens of 4 patients who died from a COVID-19-unrelated cardiac cause. In COVID-19 patients, neutrophil-enriched thrombi were restricted to the lung in one study 92 but present also in renal and cardiac microvessels in another.91 The concentration of NETs was about 10 times higher in tracheal aspirate fluid than in plasma samples from COVID-19 patients and airway fluid from healthy controls.87,88 Finally, infection of neutrophils from healthy individuals with SARS-CoV-2 induced the release of NETs, which were cytotoxic for lung epithelial cells in vitro.88 Altogether, these studies strongly suggest a key role of NETs in the pathophysiology of lung injury and microthrombosis (Figure 1B); but how do NET-releasing granulocytes promote thrombus formation? NETs are endowed with tremendous thrombogenic potential. They per se can promote thrombosis by providing a three-dimensional scaffold for recruitment of platelets and other cells and by adsorbing several prothrombotic proteins such as vWF, fibronectin, fibrinogen, and even cell-derived TF.93 NETs colocalize with fibrin and interact closely with fibrin strands in the thrombus, thus potentially influencing thrombus organization and stability. Mechanistically, NET’s constituents are primarily responsible for thrombus formation because they display a variety of prothrombotic activities.36 Histones, the most abundant proteins in NETs, induce platelet activation and expression of procoagulant properties, phosphatidylserine exposure on erythrocyte membrane, TF expression on monocytes and endothelial cells, prothrombin autoactivation, impairment of the protein C pathway and interference with the anticoagulant activity of heparin-like substances.36,94 Most of these effects are attributable to histone H4 and, to a lesser extent, H3. In addition, histones were shown to promote prothrombin activation by FXa in the absence of FVa and surface phospholipids, both in vitro and in mice, thus providing a new mechanism to disseminate intravascular coagulation.95 Double-stranded DNA serves as a suitable negatively charged surface that initiates the intrinsic pathway of coagulation by favouring the auto-activation of FXII and potentiating FXI activation by thrombin. Among proteins and enzymes hosted in NETs, elastase cleaves the major physiological anticoagulants TFPI, AT, and TM, myeloperoxidase oxidizes and inactivates TM, and cathepsin G augments platelet activation on the NET surface. Finally, NETs can harbour neutrophil or blood-derived TF. The relative role of each of these mechanisms in triggering COVID-19-associated thrombosis is difficult to establish. Skendros et al.46 unravelled the combined importance of TF and complement. They found a significant increase of plasma MPO/DNA complexes in COVID-19 patients, which positively correlated with TAT level, suggesting a possible role in the activation of blood coagulation. More importantly, using confocal immunofluorescence microscopy, they observed spontaneous formation of NETs bearing high amounts of TF in neutrophils collected from patients with severe COVID-19. Patients’ neutrophils also exhibited a 5-fold increase in TF mRNA levels as compared to normal cells. In addition, when neutrophils isolated from healthy individuals were treated with COVID-19 platelet-rich plasma (PRP), they expressed increased levels of TF mRNA and generated TF-bearing NETs, two phenomena that could be prevented by a specific C5a receptor antagonist. Furthermore, targeting C3 with the specific inhibitor compstatin also decreased neutrophil TF at both the mRNA and protein levels. These findings, coupled with the high plasma levels of sC5b-9 and C5a in COVID-19 patients,58,59 clearly suggest a pivotal role of complement and NETs in COVID-19-associated thrombosis. Furthermore, the complement system may exert additional prothrombotic effects56,96 (Figure 2). MASP-1 and MASP-2, two serine proteases that initiate the lectin complement pathway, can activate prothrombin to thrombin, and MASP-1 can also directly convert fibrinogen to fibrin and activate FXII. C3a and MAC induce platelet activation and increase platelet prothrombinase activity, thus contributing to clotting activation. C5a and MAC also activate ECs leading to increased P-selectin expression, release of vWF, and expression of cell surface prothrombinase activity. C4b-binding protein (C4BP) binds to and inhibits protein S.
Figure 2.
Overview of the thrombogenic potential derived from the activation of the complement system. Details in the text.
Like NETs and/or their constituents, another nuclear product, namely high-mobility group box 1 (HMGB1), can be actively secreted by a variety of stimulated immune cells or passively released by necrotic cells. HMGB1 displays strong proinflammatory properties in the extracellular milieu and is thought to play an essential role in numerous acute and chronic systemic inflammatory diseases, including sepsis.36,97 In systemic inflammation, HMGB-1 levels rise into the circulation and in other biological fluids and exhibit a direct correlation with disease severity. In this context, two observations are of particular interest and suggest a possible involvement of this protein in microvascular thrombosis of different aetiologies: 1) the direct correlation between serum HMGB1 and severity of pneumonia caused by various agents, including viruses; 2) the capacity of HMGB1 to stimulate TF expression in monocytes and ECs and to reduce the activity of thrombin-TM complex with a consequent reduction in protein C activation.36,97 In COVID-19 patients, the levels of serum HMGB1 are elevated and correlate with the severity of the disease and with a poor clinical outcome.98,99 Moreover, in vitro, exogenous HMGB1 induces the expression of SARS-CoV-2 entry receptor ACE2 in alveolar epithelial cells in a RAGE (receptor for advanced glycosylation end products)-dependent manner. Significantly, genetic or pharmacological inhibition of the HMGB1-RAGE pathway blocks ACE2 expression.99 This mechanism adds to the known ability of HMGB1 to stimulate the production of inflammatory cytokines by immune cells (e.g., monocytes-macrophages) via TLRs.97 Therefore, HMGB1 might play an important role in COVID-19, although more studies are required.
Platelets
Activated platelets closely cooperate with monocytes-macrophages and neutrophils to promote thrombosis during SARS-CoV-2 infection. Platelets, however, may participate in the activation of coagulation and thrombus formation by additional mechanisms.36,63 First, in quiescent platelets, variable levels of TF mRNA and protein have been detected, increasing upon stimulation with different agonists. Moreover, activated platelets provide a suitable phospholipid surface (anionic phospholipids, mainly phosphatidylserine) that accelerates the coagulation reactions many folds and renders clotting enzymes less susceptible to fluid phase protease inhibitors. Finally, stimulated platelets release from dense granules soluble polyphosphates that are composed of 60–100 linear-linked phosphate subunits and represent a suitable surface for the activation of the contact system, thereby inducing thrombin generation independently of TF and bradykinin formation. The role of the contact system in COVID-19-associated thrombosis has not been addressed so far. However, the potential benefits of targeting the contact system activation are discussed in recent reviews.100,101
Endothelial cells
As alluded to above, there is compelling evidence that endothelial cells play a pivotal role in COVID-19, as they are the target of both the virus and the multiple inflammatory mediators generated by the hyperinflammatory response and, as a consequence, undergo activation/injury. In vitro infection of endothelial cells with some viruses has been shown to induce TF expression.64,65 Likewise, cultured endothelial cells have long been known to respond to cytokines, including TNFα, IL-1β, and IL-6, as well as to complement activation products with the expression of TF.36,37 To our knowledge, data on the direct and/or indirect effects of SARS-CoV-2 on endothelial TF are unavailable. DiNicolantonio and McCarty102 hypothesized that SARS-CoV-2, after incorporation into endosomes within endothelial cells, can activate endosomal NADPH oxidase and that the resulting local production of superoxide/hydrogen peroxide leads to activation of NF-kB and increased TF expression. Though unproven, this hypothesis is interesting based on the finding that Covid-19 patients display overactivation of NADPH oxidase 2 (Nox2), which is more intense in severe than non-severe patients.103 Moreover, patients with thrombotic complications have higher Nox2 activation than those without, suggesting the possible involvement of Nox2 in thrombus formation. Despite the lack of evidence for the in vivo TF expression during SARS-CoV-2 infection, activated endothelial cells may contribute to thrombus formation by other important mechanisms. They adopt a proinflammatory phenotype that supports the recruitment of monocytes-macrophages and neutrophils (both expressing a great procoagulant potential) through the exposure of adhesion molecules (P-selectin and intercellular adhesion molecule 1, ICAM-1, among others), thus favoring local thrombus formation. In this context, P-selectin is of particular importance because it is per se a potent stimulus for induction of TF in monocytes104 and, as mentioned above, it binds TF+ MPs of leukocyte origin. MPs are then internalized, and the TF moiety is recycled to the cell surface, thus inducing a substantial increase in the cell procoagulant potential.68,105 Endothelial ICAM-1, on the other hand, is overexpressed in COVID-19 post-mortem lung samples compared to H1N1 and control lung samples.54 In addition, activated endothelial cells are known to secrete von Willebrand factor (vWF) in its highly platelet-agglutinating form (i.e., ultra-large vWF multimers) from Weibel-Palade bodies, eventually resulting in platelet activation and platelet-mediated clotting stimulation (Figure 2). As a matter of fact, increased plasma levels of vWF along with a decrease in vWF-cleaving protease ADAMTS-13 (a disintegrin and metalloprotease with thrombospondin type 1 motif, member 13, that reduces the thrombogenicity of released vWF by cleaving ultralarge multimers) have been reported by several investigators.12,31,41,106–108 The extent to which changes in ADAMTS-13/vWF ratio contribute to thrombosis and illness severity in COVID-19 patients remains to be fully elucidated. Bazzan et al.,107 in a cohort of 88 COVID-19 patients, found lower ADAMTS-13 (and higher vWF) levels in nonsurvivors compared with survivors, and ADAMTS-13 levels < 30% were significantly associated with higher mortality. Cugno et al.,59 in a cohort of 148 patients with COVID-19 of different severity, reported that plasma levels of sC5b-9 and vWF were strongly correlated with each other and disease severity. Interestingly, sC5b-9 and vWF declined simultaneously during remission, suggesting that, in addition to proinflammatory cytokines and direct SARS-CoV-2 infection, complement activation plays a key role in mediating COVID-19-associated endotheliopathy. Several studies have reported the increase of other markers of endothelial activation/damage, including soluble TM (sTM),45,59,109 soluble E-selectin,59 soluble P-selectin (sP-selectin, a marker of endothelial and platelet activation),45,110 and TFPI.43 In one of these studies,45, the levels of sP-selectin, like those of vWF, were significantly higher in ICU than in non-ICU COVID-19 patients, suggesting a more severe endothelial activation-damage, although a contribution of activated platelets cannot be excluded. sTM, instead, was not different in ICU versus non-ICU patients. However, its plasma concentration, like vWF antigen, correlated with clinical outcome and was a predictive marker of mortality in COVID-19. In another study,110 ICU COVID-19 patients had significantly and persistently elevated plasma levels of hyaluronic acid and syndecan-1, besides sP-selectin, suggesting degradation of the endothelial glycocalyx with profound consequences on vascular function, such as, for example, a decrease in nitric oxide production that will promote platelet recruitment to vascular endothelium. Moreover, the plasma levels of heparan sulfate and heparanase were significantly higher in COVID-19 patients than in healthy controls, and heparanase activity was associated with disease severity.111 Of note, LMWH prophylaxis in these patients reduced heparanase activity, which translates into a beneficial effect because heparanase can activate macrophages and induce the secretion of cytokines. Finally, circulating endothelial cells, which are considered reliable markers of endothelial injury, were significantly higher in COVID-19 patients than in healthy subjects at earlier phase of the disease, further supporting the role of endothelial damage in COVID-19.112
During SARS-CoV-2 infection, at least two additional mechanisms may contribute to the prothrombotic state. First, COVID-19 patients reportedly have increased plasma levels of angiotensin II (ATII) due to the downregulation of cell surface ACE2 expression that follows the interaction with SARS-CoV-2 S-protein.52,113 ATII contributes to the clinical and pathological manifestations of COVID-19 and, more importantly, it is endowed with proinflammatory and prothrombotic properties, including the capacity to induce TF expression in both monocytes and endothelial cells.113,114
Second, a rather common finding in patients with COVID-19 is severe hypoxia. Although not formally tested in the context of SARS-CoV-2 infection, hypoxia, through the release of HIFs (hypoxia-inducible transcription factors), is likely to induce a prothrombotic state by stimulating TF synthesis in monocytes-macrophages and endothelial cells, and by downregulating natural anticoagulants protein S and TFPI.115, 116
Changes in Physiological Anticoagulant Mechanisms
Downregulation of physiological anticoagulant pathways, characterized by a marked reduction in plasma levels of major anticoagulant proteins (antithrombin, protein C and protein S) and by a decreased expression of endothelial anticoagulant properties (heparan sulphate, TM and endothelial protein C receptor), is a common feature of sepsis-associated haemostatic alterations and contributes to widespread thrombosis of the microcirculation seen in this condition.36,37 In striking contrast, plasma concentration of antithrombin, proteins C and protein S are not significantly altered in patients with COVID-19, even in those with severe disease.12,42,43,45 Some investigators reported a slight decrease of these parameters, particularly in non-surviving patients, but the plasma levels rarely dropped below 80% of normal.22,31,41 The anticoagulant activities of endothelial cells have not been formally investigated in SARS-CoV-2 infection, neither in cell cultures nor at the tissue level. Considering that endothelial cells are major targets of the virus and of the numerous proinflammatory mediators present in severe COVID-19 patients, impairment of endothelial anticoagulant properties can be safely hypothesized in the context of endothelial dysfunction-injury. The reported increase of plasma levels of sTM, heparan sulphate and TFPI in COVID-19 patients (see above) would support this view, although more data are needed to reach definitive conclusions.
Changes in Fibrinolysis
Suppression of fibrinolysis is widely recognized as a significant player in the pathophysiology of sepsis-associated DIC, as it contributes to widespread microvascular thrombosis by preventing fibrin removal. It has been attributed mainly to upregulation of plasminogen activator inhibitor-1 (PAI-1) and, more recently, to activation of thrombin activatable fibrinolysis inhibitor (TAFI) by enhanced thrombin formation, a hallmark of sepsis.35–37 Likewise, impaired fibrinolysis, mainly due to increased PAI-1 levels, seems to play a critical role in viral diseases.64 Unfortunately, studies evaluating fibrinolysis in COVID-19 are scarce and little information is available so far, both at tissue/cellular and systemic level.
In the lung, the main target of SARS-CoV-2, where microvascular thrombosis and alveolar fibrin deposition are most prominent and substantially contribute to ARDS, the role of fibrinolysis has not been investigated. In ARDS, secondary to other causes, fibrin formation is not adequately counterbalanced by fibrin removal because fibrinolysis is impaired. Measurements in BALF from patients with ARDS or severe pneumonia requiring mechanical ventilation have shown a marked increase in PAI-1, the most powerful fibrinolysis inhibitor, which was associated with mortality in ARDS patients.77,78 Whether a similar mechanism might operate in COVID-19 patients remains a matter of speculation.117,118 It should be considered that NETs and their components (histones, DNA, and particularly histone/DNA complexes), abundantly released during COVID-19, modify the fibrin structure by increasing the fiber thickness and stabilize the clot by binding to large fibrin degradation products, thereby exerting an inhibitory effect on fibrinolysis.119 Clearly, specific investigations with BALF and pulmonary tissue from COVID-19 patients are required to shed light on this important issue.
At systemic level, a decreased fibrinolytic capacity, commonly referred to as fibrinolysis shutdown, has been proposed by several investigators based on global assays performed with whole blood, such as TEG and ROTEM.42,120–123 However, the interpretation of these findings is difficult, considering that the ability of standard viscoelastic tests to detect hypofibrinolysis is rather limited. Using a variant of the ROTEM assay, in which blood is challenged with exogenous t-PA, two studies reported a marked resistance to fibrinolysis in severe COVID-19 patients,42,120 which was more pronounced in ICU COVID-19 patients with thrombosis than in ICU patients without thrombosis.42 A similar fibrinolytic resistance was shown by Blasi et al.41 in a small group of COVID-19 patients, using a global assay which measures the t-PA induced lysis time of plasma clots (plasma clot lysis time, CLT), but no difference was seen between patients admitted to ICU and ward. Because all patients received LMWH, which shortens CLT at clinically relevant concentrations, the fibrinolytic defect might have been underestimated in these patients.41 In a longitudinal study of 21 COVID-19 patients with daily measurements during the ICU stay, reduced fibrinolysis, assessed by another global plasma assay named global fibrinolytic capacity test, was frequently observed.44
Data on the main proteins of the fibrinolytic system and on markers of fibrinolysis activation, which might help in better understanding the fibrinolytic state of COVID-19 patients, are rather limited. In general, the studies on plasma fibrinolytic proteins reported increased levels of PAI-1 and, when measured, also of t-PA41,42,45,59,124,125 with some exceptions.39,126 Some investigators found that t-PA and/or PAI-1 were significantly higher in ICU than in non-ICU COVID-19 patients,42,124,125 whereas others found no difference.41,45,59 White et al.43 reported significantly increased levels of t-PA, but not of PAI-1, in critical COVID-19 patients. The plasma levels of t-PA/PAI-1 complexes were increased in a series of 147 COVID-19 patients as compared to controls, and patients with thrombotic disease had higher values than those without; moreover, t-PA/PAI-1 complexes were correlated with disease severity and, like D-dimer, were an independent risk factor for death.127 In the study of Zuo et al.,124 involving 118 COVID-19 patients, both t-PA and PAI-1 were associated with poor clinical outcomes. Interestingly, plasma samples from COVID-19 patients with high t-PA, when tested with a spontaneous clot lysis assay (i.e., in the absence of exogenous t-PA), exhibited significantly enhanced fibrinolysis compared to samples with low t-PA and samples from healthy controls. According to the authors, there is a subset of COVID-19 patients with exceedingly high levels of t-PA, which is prone to fibrinolysis, suggesting a possible mechanism that explains the enhanced bleeding risk, at least in part, observed in some patients with COVID-19. The role of TAFI activation as a mechanism that impairs fibrinolysis and facilitates fibrin deposition has not been sufficiently addressed so far, despite the knowledge that COVID-19 is associated with a hypercoagulability state and a consequent increase in thrombin generation. In this respect, only Nougier et al.,42 in their study on 78 COVID-19 patients, reported not only raised t-PA and PAI-1 but also increased plasma levels of TAFIa/i (a combination of activated TAFI and its inactivated form). The latter, like t-PA and PAI-1, was significantly higher in ICU than in non-ICU patients. Plasma levels of α2-antiplasmin were reported to be in the normal range both in ICU and non-ICU patients,42,45 whereas plasminogen level was normal at admission and slightly, albeit significantly, reduced in ICU patients, suggesting a mild consumption with the progression of the disease.128
Concerning the evaluation of fibrinolysis activation, plasma levels of plasmin-α2-antiplasmin complex (PAP), also called plasmin-α2-plasmin inhibitor complex (PIC), which reflect ongoing in vivo plasmin generation, were found to be significantly increased in COVID-19 patients.41,109,127 One study reported that PAP level did not differ between ICU and general ward patients,41 while another127 showed that it was higher in patients with thrombosis and correlated with disease severity. Ranucci et al.,126 in a study of 20 COVID-19 patients, observed increased levels of PAI-2, normally absent from plasma (except in pregnant women), especially in nonsurvivors, but normal t-PA (PAI-1 was not measured); at follow-up, PAI-2 increased further in nonsurvivors reaching a level 6-fold higher than that of survivors, with a significant difference between the two groups. PAP levels were not significantly modified; however, the baseline PAI-2/PAP ratio was significantly higher in nonsurvivors than in survivors. Finally, in another study of 20 critical COVID-19 patients, Tang et al.129 evaluated the trajectory of fibrinolytic changes and found that PAP levels decreased over time and became significantly lower in nonsurvivors during the late stage in ICU, which coincided with a significant increase of t-PA-PAI-1 complex. The findings were interpreted as a PAI-1-mediated hypofibrinolysis status that might suggest the use of thrombolytic therapy with t-PA.
The mechanisms behind the changes in t-PA and PAI-1 have not been specifically investigated in COVID-19 patients. However, considering the pivotal role of the endothelium in the pathophysiology of SARS-CoV-2 infection, increased t-PA and PAI-1 plasma levels most probably reflect endothelial activation-injury induced by the virus itself and by the numerous inflammatory mediators released in response to the infection. Among the latter, IL-6 seems, once again, one of the most important as suggested by Kang et al.,125 who showed, albeit in few patients, that treatment with tocilizumab (a monoclonal antibody targeting the IL-6 receptor) decreased PAI-1 and improved the clinical features. Moreover, angiotensin II might also contribute to fibrinolytic changes because it is increased in COVID-19 patients130 and is known to induce the expression of PAI-1 in endothelial cells.131 In ARDS secondary to conditions other than COVID-19, lung macrophages have been reported to be the main cellular source of PAI-1 and, in some studies, also of PAI-2,77,132 although the contribution of damaged alveolar epithelial cells and activated platelets cannot be overlooked.77,133 In a preprint study,67 lung epithelial cells infected in vitro with SARS-CoV-2 showed an upregulation of several fibrinolytic genes, including t-PA, u-PA, the u-PA receptor, and PAI-2. Therefore, while it can be safely concluded that SARS-CoV-2 infection is associated with multifaceted changes of the fibrinolytic system, we need to understand how these changes impact the fibrinolytic process in the lung parenchyma of COVID-19 patient.
Conclusion and Perspectives
Over the last months, considerable progress has been made in the understanding of the complex events involved in the pathological derangement of the haemostatic process that leads to macrovascular and/or microvascular thrombosis associated with COVID-19. The extent to which the current knowledge will prove useful for developing new diagnostic and therapeutic tools remains to be established. Although many laboratory tests are frequently altered in COVID-19 patients (e.g., global assays, markers of coagulation activation, and markers of endothelial activation), the diagnosis and monitoring of haemostatic abnormalities are still based on routine assays, such as D-dimer level, prothrombin time, fibrinogen level, and platelet count. Interestingly, measuring D-dimer in combination with IL-6 greatly improves the specificity and sensitivity for early prediction of the severity of COVID-19.134 A recent study in septic patients reported that D-dimer corrected for thrombin and plasmin generation provides a better picture of the in vivo coagulation-fibrinolysis balance and displays a much higher prognostic value than standard D-dimer.135 Whether the “corrected” D-dimer may represent a useful prognostic marker in patients with COVID-19 deserves investigation. Some new emerging parameters that could be of clinical utility await further investigation. For instance, elevated plasma levels of complement activation products (C5a and sC5b-9) and markers of NETs (cell-free DNA, MPO-DNA complex, and cit-H3) paralleling the disease severity have been consistently reported in COVID-19 patients.58,59,86,87,89 Moreover, circulating HMGB-1 is augmented in COVID-19 patients and may have prognostic value.98,99 These proteins, therefore, might be new sensitive biomarkers of disease progression and useful predictors of outcome in SARS-CoV-2 infection.
Regarding the supportive treatment for haemostatic abnormalities associated with SARS-CoV-2 infection, different strategies can be envisaged in light of insights into the pathogenetic mechanisms underlying macro and microthrombosis in COVID-19 patients. Considering the high rate of thromboembolic complications among hospitalised patients, prophylactic administration of heparin, particularly LMWH, is recommended for all hospitalised COVID-19 patients in the absence of contraindications. The use of more intense intermediate to therapeutic LMWH or heparin dose, especially in ICU patients, is still a matter of debate.136 The central role of cytokines, particularly IL-6, in the pathophysiology of SARS-CoV-2 infection and the associated thrombotic complications provides a rationale for using tocilizumab and similar drugs in COVID-19 patients.137 Several studies have indeed reported beneficial effects of the drug in these patients.138 Clinical trials on the efficacy and safety of tocilizumab in COVID-19 are underway. Besides cytokines, also the complement system and the inflammatory mediators derived from its activation appear to play a major pathophysiological role in COVID-19 hyperinflammatory response and ensuing thrombotic complications. Therefore, targeting complement at different levels, especially at C5 or C3 level, may represent a therapeutic option.139,140 Consistent with this notion, blocking C5 with eculizumab (a monoclonal antibody that binds to C5 with high affinity and inhibits its cleavage into C5a and C5b, thus preventing the generation of the terminal complement complex C5b-9) or C3 with AMY101 (a small peptide that binds to C3 and impairs the cleavage of the protein by C3 convertases, thus preventing propagation and amplification of complement activation) has shown promising results in terms of reduction of inflammatory responses and improvement of clinical conditions, eventually leading to successful disease outcomes. As expected, AMY101 exhibited a broader anti-inflammatory effect.141 These and other complement inhibiting drugs are being studied in clinical trials.139 Finally, considering the emerging role of NETs and/or their constituents in dysregulation of haemostasis and consequent micro or macrothrombosis associated with SARS-CoV-2 infection, targeting these compounds at different levels could be a novel therapeutic strategy.142 Inhibitors of the enzymes PAD4, NADPH-oxidase, and elastase, all of which are involved in NETs formation, are possible candidates for active regulation of NETs. Dismantling of NETs by deoxyribonuclease (DNase) 1 could be another potential treatment of COVID-19. In small series of patients with severe disease, administration of nebulized Dornase alfa, a recombinant human DNase 1, resulted in a significant improvement of pulmonary function and a reduction of inflammatory markers.143,144 Therefore, appropriately designed clinical trials are required. Since histones, the most toxic NET components, along with HMGB-1, seem to be critical mediators of organ dysfunction and death in COVID-19 patients, an attractive therapeutic approach could be the development of effective histone and HMGB-1 antagonists.
Footnotes
Competing interests: The authors declare no conflict of Interest.
References
- 1.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, Liu L, Shan H, Lei CL, Hui DSC, Du B, Li LJ, Zeng G, Yuen KY, Chen RC, Tang CL, Wang T, Chen PY, Xiang J, Li SY, Wang JL, Liang ZJ, Peng YX, Wei L, Liu Y, Hu YH, Peng P, Wang JM, Liu JY, Chen Z, Li G, Zheng ZJ, Qiu SQ, Luo J, Ye CJ, Zhu SY, Zhong NS China Medical Treatment Expert Group for Covid-19. Clinical Characteristics of Coronavirus Disease 2019 in China. N Engl J Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020;323:1239–1242. doi: 10.1001/jama.2020.2648. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 3.Carsana L, Sonzogni A, Nasr A, Rossi RS, Pellegrinelli A, Zerbi P, Rech R, Colombo R, Antinori S, Corbellino M, Galli M, Catena E, Tosoni A, Gianatti A, Nebuloni M. Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: a two-centre descriptive study. Lancet Infect Dis. 2020;20:1135–1140. doi: 10.1016/S1473-3099(20)30434-5. doi: 10.1016/S1473-3099(20)30434-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fox SE, Akmatbekov A, Harbert JL, Li G, Quincy Brown J, Vander Heide RS. Pulmonary and cardiac pathology in African American patients with COVID-19: an autopsy series from New Orleans. Lancet Respir Med. 2020;8:681–686. doi: 10.1016/S2213-2600(20)30243-5. doi: 10.1016/S2213-2600(20)30243-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Dolhnikoff M, Duarte-Neto AN, de Almeida Monteiro RA, da Silva LFF, de Oliveira EP, Saldiva PHN, Mauad T, Negri EM. Pathological evidence of pulmonary thrombotic phenomena in severe COVID-19. J Thromb Haemost. 2020;18:1517–1519. doi: 10.1111/jth.14844. doi: 10.1111/jth.14844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Falasca L, Nardacci R, Colombo D, Lalle E, Di Caro A, Nicastri E, Antinori A, Petrosillo N, Marchioni L, Biava G, D’Offizi G, Palmieri F, Goletti D, Zumla A, Ippolito G, Piacentini M, Del Nonno F. Postmortem Findings in Italian Patients With COVID-19: A Descriptive Full Autopsy Study of Cases with and without Comorbidities. J Infect Dis. 2020;222:1807–1815. doi: 10.1093/infdis/jiaa578. doi: 10.1093/infdis/jiaa578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, Vanstapel A, Werlein C, Stark H, Tzankov A, Li WW, Li VW, Mentzer SJ, Jonigk D. Pulmonary Vascular Endothelialitis, Thrombosis, and Angiogenesis in Covid-19. N Engl J Med. 2020;383:120–128. doi: 10.1056/NEJMoa2015432. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McFadyen JD, Stevens H, Peter K. The Emerging Threat of (Micro)Thrombosis in COVID-19 and Its Therapeutic Implications. Circ Res. 2020;127:571–587. doi: 10.1161/CIRCRESAHA.120.317447. doi: 10.1161/CIRCRESAHA.120.317447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Middeldorp S, Coppens M, van Haaps TF, Foppen M, Vlaar AP, Müller MCA, Bouman CCS, Beenen LFM, Kootte RS, Heijmans J, Smits LP, Bonta PI, van Es N. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J Thromb Haemost. 2020;18:1995–2002. doi: 10.1111/jth.14888. doi: 10.1111/jth.14888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Klok FA, Kruip MJHA, van der Meer NJM, Arbous MS, Gommers DAMPJ, Kant KM, Kaptein FHJ, van Paassen J, Stals MAM, Huisman MV, Endeman H. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: An updated analysis. Thromb Res. 2020;191:148–150. doi: 10.1016/j.thromres.2020.04.041. doi: 10.1016/j.thromres.2020.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lodigiani C, Iapichino G, Carenzo L, Cecconi M, Ferrazzi P, Sebastian T, Kucher N, Studt JD, Sacco C, Alexia B, Sandri MT, Barco S. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb Res. 2020;191:9–14. doi: 10.1016/j.thromres.2020.04.024. doi: 10.1016/j.thromres.2020.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Helms J, Tacquard C, Severac F, Leonard-Lorant I, Ohana M, Delabranche X, Merdji H, Clere-Jehl R, Schenck M, Fagot Gandet F, Fafi-Kremer S, Castelain V, Schneider F, Grunebaum L, Anglés-Cano E, Sattler L, Mertes PM, Meziani F CRICS TRIGGERSEP Group (Clinical Research in Intensive Care and Sepsis Trial Group for Global Evaluation and Research in Sepsis) High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Med. 2020;46:1089–1098. doi: 10.1007/s00134-020-06062-x. doi: 10.1007/s00134-020-06062-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Poissy J, Goutay Julien, Caplan Morgan, Parmentier Erika, Duburcq Thibault, Lassalle Fanny, Jeanpierre Emmanuelle, Rauch Antoine, Labreuche Julien, Susen Sophie and Lille ICU Haemostasis COVID-19 Group. Pulmonary Embolism in Patients With COVID-19. Circulation. 2020;142:184–186. doi: 10.1161/CIRCULATIONAHA.120.047430. doi: 10.1161/CIRCULATIONAHA.120.047430. [DOI] [PubMed] [Google Scholar]
- 14.Cattaneo M, Bertinato EM, Birocchi S, Brizio C, Malavolta D, Manzoni M, Muscarella G, Orlandi M. Pulmonary embolism or pulmonary thrombosis in COVID-19? is the recommendation to use high-dose heparin for thromboprophylaxis justified? Thromb Haemost. 2020;120:1230–1232. doi: 10.1055/s-0040-1712097. doi: 10.1055/s-0040-1712097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Al-Samkari H, Karp Leaf RS, Dzik WH, Carlson JCT, Fogerty AE, Waheed A, Goodarzi K, Bendapudi PK, Bornikova L, Gupta S, Leaf DE, Kuter DJ, Rosovsky RP. COVID-19 and coagulation: bleeding and thrombotic manifestations of SARS-CoV-2 infection. Blood. 2020;136:489–500. doi: 10.1182/blood.2020006520. doi: 10.1182/blood.2020006520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mei F, Fan J, Yuan J, Liang Z, Wang K, Sun J, Guan W, Huang M, Li Y, Zhang WW. Comparison of venous thromboembolism risks between COVID-19 pneumonia and community-acquired pneumonia patients. Arterioscler Thromb Vasc Biol. 2020;40:2332–2337. doi: 10.1161/ATVBAHA.120.314779. doi: 10.1161/ATVBAHA.120.314779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zheng YY, Ma YT, Zhang JY, Xie X. COVID-19 and the cardiovascular system. Nat Rev Cardiol. 2020;17:259–260. doi: 10.1038/s41569-020-0360-5. doi: 10.1038/s41569-020-0360-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Oxley TJ, Mocco J, Majidi S, Kellner CP, Shoirah H, Singh IP, De Leacy RA, Shigematsu T, Ladner TR, Yaeger KA, Skliut M, Weinberger J, Dangayach NS, Bederson JB, Tuhrim S, Fifi JT. Large-Vessel Stroke as a Presenting Feature of Covid-19 in the Young. N Engl J Med. 2020;382:e60. doi: 10.1056/NEJMc2009787. doi: 10.1056/NEJMc2009787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mestres G, Puigmacià R, Blanco C, Yugueros X, Esturrica M, Riambau V. Risk of peripheral arterial thrombosis in COVID-19. J Vasc Surg. 2020;72:756–757. doi: 10.1016/j.jvs.2020.04.477. doi: 10.1016/j.jvs.2020.04.477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Warrior K, Chung PA, Ahmed N, Soult MC, Simpson KP. Acute Limb Ischemia Due to Arterial Thrombosis Associated with Coronavirus Disease 2019. Crit Care Explor. 2020;2:e0140. doi: 10.1097/CCE.0000000000000140. doi: 10.1097/CCE.0000000000000140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844–847. doi: 10.1111/jth.14768. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, Zhang L, Fan G, Xu J, Gu X, Cheng Z, Yu T, Xia J, Wei Y, Wu W, Xie X, Yin W, Li H, Liu M, Xiao Y, Gao H, Guo L, Xie J, Wang G, Jiang R, Gao Z, Jin Q, Wang J, Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lippi G, Favaloro EJ. D-dimer is Associated with Severity of Coronavirus Disease 2019: A Pooled Analysis. Thromb Haemost. 2020;120:876–878. doi: 10.1055/s-0040-1709650. doi: 10.1055/s-0040-1709650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen G, Wu D, Guo W, Cao Y, Huang D, Wang H, Wang T, Zhang X, Chen H, Yu H, Zhang X, Zhang M, Wu S, Song J, Chen T, Han M, Li S, Luo X, Zhao J, Ning Q. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest. 2020;130:2620–2629. doi: 10.1172/JCI137244. doi: 10.1172/JCI137244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen T, Wu D, Chen H, Yan W, Yang D, Chen G, Ma K, Xu D, Yu H, Wang H, Wang T, Guo W, Chen J, Ding C, Zhang X, Huang J, Han M, Li S, Luo X, Zhao J, Ning Q. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. 2020;368:m1091. doi: 10.1136/bmj.m1091. doi: 10.1136/bmj.m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lippi G, Plebani M, Henry BM. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin Chim Acta. 2020;506:145–148. doi: 10.1016/j.cca.2020.03.022. doi: 10.1016/j.cca.2020.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yang X, Yang Q, Wang Y, Wu Y, Xu J, Yu Y, Shang Y. Thrombocytopenia and its association with mortality in patients with COVID-19. J Thromb Haemost. 2020;18:1469–1472. doi: 10.1111/jth.14848. doi: 10.1111/jth.14848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ranucci M, Ballotta A, Di Dedda U, Bayshnikova E, Dei Poli M, Resta M, Falco M, Albano G, Menicanti L. The procoagulant pattern of patients with COVID-19 acute respiratory distress syndrome. J Thromb Haemost. 2020;18:1747–1751. doi: 10.1111/jth.14854. doi: 10.1111/jth.14854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Panigada M, Bottino N, Tagliabue P, Grasselli G, Novembrino C, Chantarangkul V, Pesenti A, Peyvandi F, Tripodi A. Hypercoagulability of COVID-19 patients in intensive care unit. A report of thromboelastography findings and other parameters of hemostasis. J Thromb Haemost. 2020;18:1738–1742. doi: 10.1111/jth.14850. doi: 10.1111/jth.14850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhu J, Pang J, Ji P, Zhong Z, Li H, Li B, Zhang J, Lu J. Coagulation dysfunction is associated with severity of COVID-19: A meta-analysis. J Med Virol. 2021;93:962–972. doi: 10.1002/jmv.26336. doi: 10.1002/jmv.26336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Thachil J, Tang N, Gando S, Falanga A, Cattaneo M, Levi M, Clark C, Iba T. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J Thromb Haemost. 2020;18:1023–1026. doi: 10.1111/jth.14810. doi: 10.1111/jth.14810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Levi M. Platelets in Critical Illness. Semin Thromb Hemost. 2016;42:252–257. doi: 10.1055/s-0035-1570080. doi: 10.1055/s-0035-1570080. [DOI] [PubMed] [Google Scholar]
- 35.Semeraro F, Colucci M, Caironi P, Masson S, Ammollo CT, Teli R, Semeraro N, Magnoli M, Salati G, Isetta M, Panigada M, Tonetti T, Tognoni G, Latini R, Pesenti A, Gattinoni L. Platelet Drop and Fibrinolytic Shutdown in Patients with Sepsis. Crit Care Med. 2018;46:e221–e228. doi: 10.1097/CCM.0000000000002919. doi: 10.1097/CCM.0000000000002919. [DOI] [PubMed] [Google Scholar]
- 36.Semeraro N, Ammollo CT, Semeraro F, Colucci M. Coagulopathy of Acute Sepsis. Semin Thromb Hemost. 2015;41:650–658. doi: 10.1055/s-0035-1556730. doi: 10.1055/s-0035-1556730. [DOI] [PubMed] [Google Scholar]
- 37.Levi M, van der Poll T. Coagulation and sepsis. Thromb Res. 2017;149:38–44. doi: 10.1016/j.thromres.2016.11.007. doi: 10.1016/j.thromres.2016.11.007. [DOI] [PubMed] [Google Scholar]
- 38.Spiezia L, Boscolo A, Poletto F, Cerruti L, Tiberio I, Campello E, Navalesi P, Simioni P. COVID-19-Related Severe Hypercoagulability in Patients Admitted to Intensive Care Unit for Acute Respiratory Failure. Thromb Haemost. 2020;120:998–1000. doi: 10.1055/s-0040-1710018. doi: 10.1055/s-0040-1710018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Paparella D, Colucci M, Squiccimarro E, Raimondo P, De Palma F, Ranieri P, Mariggiò MA, Grasso S. Clotting abnormalities in critically ill COVID-19 patients are inconsistent with overt disseminated intravascular coagulation. Thromb Res. 2020;196:272–275. doi: 10.1016/j.thromres.2020.09.015. doi: 10.1016/j.thromres.2020.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pavoni V, Gianesello L, Pazzi M, Stera C, Meconi T, Covani Frigieri F. Evaluation of coagulation function by rotation thromboelastometry in critically ill patients with severe COVID-19 pneumonia. J Thromb Thrombolysis. 2020;50:281–286. doi: 10.1007/s11239-020-02130-7. https://doi.org/10.1007/s11239-020-02130-7 https://doi.org/10.1007/s11239-020-02130-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Blasi A, von Meijenfeldt FA, Adelmeijer J, Calvo A, Ibañez C, Perdomo J, Reverter JC, Lisman T. In vitro hypercoagulability and ongoing in vivo activation of coagulation and fibrinolysis in COVID-19 patients on anticoagulation. J Thromb Haemost. 2020;18:2646–2653. doi: 10.1111/jth.15043. doi: 10.1111/jth.15043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Nougier C, Benoit R, Simon M, Desmurs-Clavel H, Marcotte G, Argaud L, David JS, Bonnet A, Negrier C, Dargaud Y. Hypofibrinolytic state and high thrombin generation may play a major role in SARS-COV2 associated thrombosis. J Thromb Haemost. 2020;18:2215–2219. doi: 10.1111/jth.15016. doi: 10.1111/jth.15016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.White D, MacDonald S, Edwards T, Bridgeman C, Hayman M, Sharp M, Cox-Morton S, Duff E, Mahajan S, Moore C, Kirk M, Williams R, Besser M, Thomas W. Evaluation of COVID-19 coagulopathy; laboratory characterization using thrombin generation and nonconventional haemostasis assays. Int J Lab Hematol. 2021;43:123–130. doi: 10.1111/ijlh.13329. doi: 10.1111/ijlh.13329. [DOI] [PubMed] [Google Scholar]
- 44.Hardy M, Michaux I, Lessire S, Douxfils J, Dogné JM, Bareille M, Horlait G, Bulpa P, Chapelle C, Laporte S, Testa S, Jacqmin H, Lecompte T, Dive A, Mullier F. Prothrombotic disturbances of hemostasis of patients with severe COVID-19: A prospective longitudinal observational study. Thromb Res. 2021;197:20–23. doi: 10.1016/j.thromres.2020.10.025. doi: 10.1016/j.thromres.2020.10.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Goshua G, Pine AB, Meizlish ML, Chang CH, Zhang H, Bahel P, Baluha A, Bar N, Bona RD, Burns AJ, Dela Cruz CS, Dumont A, Halene S, Hwa J, Koff J, Menninger H, Neparidze N, Price C, Siner JM, Tormey C, Rinder HM, Chun HJ, Lee AI. Endotheliopathy in COVID-19-associated coagulopathy: evidence from a single-centre, cross-sectional study. Lancet Haematol. 2020;7:e575–e582. doi: 10.1016/S2352-3026(20)30216-7. doi: 10.1016/S2352-3026(20)30216-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Skendros P, Mitsios A, Chrysanthopoulou A, Mastellos DC, Metallidis S, Rafailidis P, Ntinopoulou M, Sertaridou E, Tsironidou V, Tsigalou C, Tektonidou M, Konstantinidis T, Papagoras C, Mitroulis I, Germanidis G, Lambris JD, Ritis K. Complement and tissue factor-enriched neutrophil extracellular traps are key drivers in COVID-19 immunothrombosis. J Clin Invest. 2020;130:6151–6157. doi: 10.1172/JCI141374. doi: 10.1172/JCI141374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Umemura Y, Yamakawa K, Kiguchi T, Nishida T, Kawada M, Fujimi S. Hematological Phenotype of COVID-19-Induced Coagulopathy: Far from Typical Sepsis-Induced Coagulopathy. J Clin Med. 2020;9:2875. doi: 10.3390/jcm9092875. doi: 10.3390/jcm9092875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Al-Samkari H, Song F, Van Cott EM, Kuter DJ, Rosovsky R. Evaluation of the prothrombin fragment 1.2 in patients with coronavirus disease 2019 (COVID-19) Am J Hematol. 2020;95:1479–1485. doi: 10.1002/ajh.25962. doi: 10.1002/ajh.25962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sarzi-Puttini P, Giorgi V, Sirotti S, Marotto D, Ardizzone S, Rizzardini G, Antinori S, Galli M. COVID-19, cytokines and immunosuppression: what can we learn from severe acute respiratory syndrome? Clin Exp Rheumatol. 2020;38:337–342. Epub 2020 Mar 22. [PubMed] [Google Scholar]
- 50.Wang J, Jiang M, Chen X, Montaner LJ. Cytokine storm and leukocyte changes in mild versus severe SARS-CoV-2 infection: Review of 3939 COVID-19 patients in China and emerging pathogenesis and therapy concepts. J Leukoc Biol. 2020;108:17–41. doi: 10.1002/JLB.3COVR0520-272R. doi: 10.1002/JLB.3COVR0520-272R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tay MZ, Poh CM, Rénia L, MacAry PA, Ng LFP. The trinity of COVID-19: immunity, inflammation and intervention. Nat Rev Immunol. 2020;20:363–374. doi: 10.1038/s41577-020-0311-8. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gheblawi M, Wang K, Viveiros A, Nguyen Q, Zhong JC, Turner AJ, Raizada MK, Grant MB, Oudit GY. Angiotensin-Converting Enzyme 2: SARS-CoV-2 Receptor and Regulator of the Renin-Angiotensin System: Celebrating the 20th Anniversary of the Discovery of ACE2. Circ Res. 2020;126:1456–1474. doi: 10.1161/CIRCRESAHA.120.317015. doi: 10.1161/CIRCRESAHA.120.317015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang C, Xie J, Zhao L, Fei X, Zhang H, Tan Y, Nie X, Zhou L, Liu Z, Ren Y, Yuan L, Zhang Y, Zhang J, Liang L, Chen X, Liu X, Wang P, Han X, Weng X, Chen Y, Yu T, Zhang X, Cai J, Chen R, Shi ZL, Bian XW. Alveolar macrophage dysfunction and cytokine storm in the pathogenesis of two severe COVID-19 patients. EBioMedicine. 2020;57:102833. doi: 10.1016/j.ebiom.2020.102833. doi: 10.1016/j.ebiom.2020.102833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Nagashima S, Mendes MC, Camargo Martins AP, Borges NH, Godoy TM, Miggiolaro AFRDS, da Silva Dezidério F, Machado-Souza C, de Noronha L. Endothelial Dysfunction and Thrombosis in Patients With COVID-19-Brief Report. Arterioscler Thromb Vasc Biol. 2020;40:2404–2407. doi: 10.1161/ATVBAHA.120.314860. doi: 10.1161/ATVBAHA.120.314860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yu J, Yuan X, Chen H, Chaturvedi S, Braunstein EM, Brodsky RA. Direct activation of the alternative complement pathway by SARS-CoV-2 spike proteins is blocked by factor D inhibition. Blood. 2020;136:2080–2089. doi: 10.1182/blood.2020008248. doi: 10.1182/blood.2020008248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Song WC, FitzGerald GA. COVID-19, microangiopathy, hemostatic activation, and complement. J Clin Invest. 2020;130:3950–3953. doi: 10.1172/JCI140183. doi: 10.1172/JCI140183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Magro C, Mulvey JJ, Berlin D, Nuovo G, Salvatore S, Harp J, Baxter-Stoltzfus A, Laurence J. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID-19 infection: A report of five cases. Transl Res. 2020;220:1–13. doi: 10.1016/j.trsl.2020.04.007. doi: 10.1016/j.trsl.2020.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cugno M, Meroni PL, Gualtierotti R, Griffini S, Grovetti E, Torri A, Panigada M, Aliberti S, Blasi F, Tedesco F, Peyvandi F. Complement activation in patients with COVID-19: A novel therapeutic target. J Allergy Clin Immunol. 2020;146:215–217. doi: 10.1016/j.jaci.2020.05.006. doi: 10.1016/j.jaci.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Cugno M, Meroni PL, Gualtierotti R, Griffini S, Grovetti E, Torri A, Lonati P, Grossi C, Borghi MO, Novembrino C, Boscolo M, Uceda Renteria SC, Valenti L, Lamorte G, Manunta M, Prati D, Pesenti A, Blasi F, Costantino G, Gori A, Bandera A, Tedesco F, Peyvandi F. Complement activation and endothelial perturbation parallel COVID-19 severity and activity. J Autoimmun. 2021;116:102560. doi: 10.1016/j.jaut.2020.102560. doi: 10.1016/j.jaut.2020.102560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Varga Z, Flammer AJ, Steiger P, Haberecker M, Andermatt R, Zinkernagel AS, Mehra MR, Schuepbach RA, Ruschitzka F, Moch H. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395:1417–1418. doi: 10.1016/S0140-6736(20)30937-5. doi: 10.1016/S0140-6736(20)30937-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Koupenova M. Potential role of platelets in COVID-19: Implications for thrombosis. Res Pract Thromb Haemost. 2020 Jun 21;4(5):737–740. doi: 10.1002/rth2.12397. doi: 10.1002/rth2.12397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Manne BK, Denorme F, Middleton EA, Portier I, Rowley JW, Stubben C, Petrey AC, Tolley ND, Guo L, Cody M, Weyrich AS, Yost CC, Rondina MT, Campbell RA. Platelet gene expression and function in patients with COVID-19. Blood. 2020;136:1317–1329. doi: 10.1182/blood.2020007214. doi: 10.1182/blood.2020007214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Grover SP, Mackman N. Tissue factor: an essential mediator of hemostasis and trigger of thrombosis. Arterioscler Thromb Vasc Biol. 2018;38:709–725. doi: 10.1161/ATVBAHA.117.309846. doi: 10.1161/ATVBAHA.117.309846. [DOI] [PubMed] [Google Scholar]
- 64.Mackman N, Antoniak S, Wolberg AS, Kasthuri R, Key NS. Coagulation Abnormalities and Thrombosis in Patients Infected With SARS-CoV-2 and Other Pandemic Viruses. Arterioscler Thromb Vasc Biol. 2020;40:2033–2044. doi: 10.1161/ATVBAHA.120.314514. doi: 10.1161/ATVBAHA.120.314514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Subramaniam S, Scharrer I. Procoagulant activity during viral infections. Front Biosci (Landmark Ed) 2018;23:1060–1081. doi: 10.2741/4633. doi: 10.2741/4633. [DOI] [PubMed] [Google Scholar]
- 66.Bastarache JA, Wang L, Geiser T, Wang Z, Albertine KH, Matthay MA, Ware LB. The alveolar epithelium can initiate the extrinsic coagulation cascade through expression of tissue factor. Thorax. 2007;62:608–616. doi: 10.1136/thx.2006.063305. doi: 10.1136/thx.2006.063305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.FitzGerald ES, Jamieson AM. Unique transcriptional changes in coagulation cascade genes in SARS-CoV-2-infected lung epithelial cells: A potential factor in COVID-19 coagulopathies. bioRxiv preprint. [DOI]
- 68.Osterud B, Bjorklid E. Tissue factor in blood cells and endothelial cells. Front Biosci (Elite Ed) 2012;4:289–299. doi: 10.2741/e376. [DOI] [PubMed] [Google Scholar]
- 69.Merad M, Martin JC. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol. 2020;20:355–362. doi: 10.1038/s41577-020-0331-4. doi: 10.1038/s41577-020-0331-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Neumann FJ, Ott I, Marx N, Luther T, Kenngott S, Gawaz M, Kotzsch M, Schömig A. Effect of human recombinant interleukin-6 and interleukin-8 on monocyte procoagulant activity. Arterioscler Thromb Vasc Biol. 1997;17:3399–3405. doi: 10.1161/01.atv.17.12.3399. doi: 10.1161/01.atv.17.12.3399. [DOI] [PubMed] [Google Scholar]
- 71.van der Poll T, de Jonge E, Levi M. Regulatory role of cytokines in disseminated intravascular coagulation. Semin Thromb Hemost. 2001;27:639–651. doi: 10.1055/s-2001-18868. doi: 10.1055/s-2001-18868. [DOI] [PubMed] [Google Scholar]
- 72.Chen X, Zhao B, Qu Y, Chen Y, Xiong J, Feng Y, Men D, Huang Q, Liu Y, Yang B, Ding J, Li F. Detectable Serum Severe Acute Respiratory Syndrome Coronavirus 2 Viral Load (RNAemia) Is Closely Correlated with Drastically Elevated Interleukin 6 Level in Critically Ill Patients with Coronavirus Disease 2019. Clin Infect Dis. 2020;71:1937–1942. doi: 10.1093/cid/ciaa449. doi: 10.1093/cid/ciaa449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Muhlfelder TW, Niemetz J, Kreutzer D, Beebe D, Ward PA, Rosenfeld SI. C5 chemotactic fragment induces leukocyte production of tissue factor activity: a link between complement and coagulation. J Clin Invest. 1979;63:147–150. doi: 10.1172/JCI109269. doi: 10.1172/JCI109269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Landsem A, Fure H, Christiansen D, Nielsen EW, Østerud B, Mollnes TE, Brekke OL. The key roles of complement and tissue factor in Escherichia coli induced coagulation in human whole blood. Clin Exp Immunol. 2015;182:81–89. doi: 10.1111/cei.12663. https://doi.org/10.1111/cei.12663 https://doi.org/10.1111/cei.12663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Hottz ED, Azevedo-Quintanilha IG, Palhinha L, Teixeira L, Barreto EA, Pão CRR, Righy C, Franco S, Souza TML, Kurtz P, Bozza FA, Bozza PT. Platelet activation and platelet-monocyte aggregate formation trigger tissue factor expression in patients with severe COVID-19. Blood. 2020;136:1330–1341. doi: 10.1182/blood.2020007252. doi: 10.1182/blood.2020007252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Freedman JE, Loscalzo J. Platelet-monocyte aggregates: bridging thrombosis and inflammation. Circulation. 2002;105:2130–2132. doi: 10.1161/01.cir.0000017140.26466.f5. doi: 10.1161/01.cir.0000017140.26466.f5. [DOI] [PubMed] [Google Scholar]
- 77.Glas GJ, Van Der Sluijs KF, Schultz MJ, Hofstra JJ, Van Der Poll T, Levi M. Bronchoalveolar hemostasis in lung injury and acute respiratory distress syndrome. J Thromb Haemost. 2013;11:17–25. doi: 10.1111/jth.12047. doi: 10.1111/jth.12047. [DOI] [PubMed] [Google Scholar]
- 78.Günther A, Mosavi P, Heinemann S, Ruppert C, Muth H, Markart P, Grimminger F, Walmrath D, Temmesfeld-Wollbrück B, Seeger W. Alveolar fibrin formation caused by enhanced procoagulant and depressed fibrinolytic capacities in severe pneumonia. Comparison with the acute respiratory distress syndrome. Am J Respir Crit Care Med. 2000;161:454–462. doi: 10.1164/ajrccm.161.2.9712038. doi: 10.1164/ajrccm.161.2.9712038. [DOI] [PubMed] [Google Scholar]
- 79.Liu X, Zhang R, He G. Hematological findings in coronavirus disease 2019: indications of progression of disease. Ann Hematol. 2020;99:1421–1428. doi: 10.1007/s00277-020-04103-5. doi: 10.1007/s00277-020-04103-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Liu J, Liu Y, Xiang P, Pu L, Xiong H, Li C, Zhang M, Tan J, Xu Y, Song R, Song M, Wang L, Zhang W, Han B, Yang L, Wang X, Zhou G, Zhang T, Li B, Wang Y, Chen Z, Wang X. Neutrophil-to-lymphocyte ratio predicts critical illness patients with 2019 coronavirus disease in the early stage. J Transl Med. 2020;18:206. doi: 10.1186/s12967-020-02374-0. doi: 10.1186/s12967-020-02374-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Maugeri N, Brambilla M, Camera M, Carbone A, Tremoli E, Donati MB, de Gaetano G, Cerletti C. Human polymorphonuclear leukocytes produce and express functional tissue factor upon stimulation. J Thromb Haemost. 2006;4:1323–30. doi: 10.1111/j.1538-7836.2006.01968.x. doi: 10.1111/j.1538-7836.2006.01968.x. [DOI] [PubMed] [Google Scholar]
- 82.Ritis K, Doumas M, Mastellos D, Micheli A, Giaglis S, Magotti P, Rafail S, Kartalis G, Sideras P, Lambris JD. A novel C5a receptor-tissue factor cross-talk in neutrophils links innate immunity to coagulation pathways. J Immunol. 2006;177:4794–4802. doi: 10.4049/jimmunol.177.7.4794. doi: 10.4049/jimmunol.177.7.4794. [DOI] [PubMed] [Google Scholar]
- 83.Brinkmann V, Zychlinsky A. Neutrophil extracellular traps: is immunity the second function of chromatin? J Cell Biol. 2012;198:773–783. doi: 10.1083/jcb.201203170. doi: 10.1083/jcb.201203170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Papayannopoulos V. Neutrophil extracellular traps in immunity and disease. Nat Rev Immunol. 2018;18:134–147. doi: 10.1038/nri.2017.105. [DOI] [PubMed] [Google Scholar]
- 85.Schönrich G, Raftery MJ. Neutrophil Extracellular Traps Go Viral. Front Immunol. 2016;7:366. doi: 10.3389/fimmu.2016.00366. doi: 10.3389/fimmu.2016.00366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Zuo Y, Yalavarthi S, Shi H, Gockman K, Zuo M, Madison JA, Blair C, Weber A, Barnes BJ, Egeblad M, Woods RJ, Kanthi Y, Knight JS. Neutrophil extracellular traps in COVID-19. JCI Insight. 2020;5:e138999. doi: 10.1172/jci.insight.138999. doi: 10.1172/jci.insight.138999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Middleton EA, He XY, Denorme F, Campbell RA, Ng D, Salvatore SP, Mostyka M, Baxter-Stoltzfus A, Borczuk AC, Loda M, Cody MJ, Manne BK, Portier I, Harris ES, Petrey AC, Beswick EJ, Caulin AF, Iovino A, Abegglen LM, Weyrich AS, Rondina MT, Egeblad M, Schiffman JD, Yost CC. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood. 2020;136:1169–1179. doi: 10.1182/blood.2020007008. doi: 10.1182/blood.2020007008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Veras FP, Pontelli MC, Silva CM, Toller-Kawahisa JE, de Lima M, Nascimento DC, Schneider AH, Caetité D, Tavares LA, Paiva IM, Rosales R, Colón D, Martins R, Castro IA, Almeida GM, Lopes MIF, Benatti MN, Bonjorno LP, Giannini MC, Luppino-Assad R, Almeida SL, Vilar F, Santana R, Bollela VR, Auxiliadora-Martins M, Borges M, Miranda CH, Pazin-Filho A, da Silva LLP, Cunha LD, Zamboni DS, Dal-Pizzol F, Leiria LO, Siyuan L, Batah S, Fabro A, Mauad T, Dolhnikoff M, Duarte-Neto A, Saldiva P, Cunha TM, Alves-Filho JC, Arruda E, Louzada-Junior P, Oliveira RD, Cunha FQ. SARS-CoV-2-triggered neutrophil extracellular traps mediate COVID-19 pathology. J Exp Med. 2020;217:e20201129. doi: 10.1084/jem.20201129. doi: 10.1084/jem.20201129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Leppkes M, Knopf J, Naschberger E, Lindemann A, Singh J, Herrmann I, Stürzl M, Staats L, Mahajan A, Schauer C, Kremer AN, Völkl S, Amann K, Evert K, Falkeis C, Wehrfritz A, Rieker RJ, Hartmann A, Kremer AE, Neurath MF, Muñoz LE, Schett G, Herrmann M. Vascular occlusion by neutrophil extracellular traps in COVID-19. EBioMedicine. 2020;58:102925. doi: 10.1016/j.ebiom.2020.102925. doi: 10.1016/j.ebiom.2020.102925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zuo Y, Zuo M, Yalavarthi S, Gockman K, Madison JA, Shi H, Woodard W, Lezak SP, Lugogo NL, Knight JS, Kanthi Y. Neutrophil extracellular traps and thrombosis in COVID-19. J Thromb Thrombolysis. 2021;51:446–453. doi: 10.1007/s11239-020-02324-z. doi: 10.1007/s11239-020-02324-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Nicolai L, Leunig A, Brambs S, Kaiser R, Weinberger T, Weigand M, Muenchhoff M, Hellmuth JC, Ledderose S, Schulz H, Scherer C, Rudelius M, Zoller M, Höchter D, Keppler O, Teupser D, Zwißler B, von Bergwelt-Baildon M, Kääb S, Massberg S, Pekayvaz K, Stark K. Immunothrombotic Dysregulation in COVID-19 Pneumonia Is Associated with Respiratory Failure and Coagulopathy. Circulation. 2020;142:1176–1189. doi: 10.1161/CIRCULATIONAHA.120.048488. doi: 10.1161/CIRCULATIONAHA.120.048488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Radermecker C, Detrembleur N, Guiot J, Cavalier E, Henket M, d’Emal C, Vanwinge C, Cataldo D, Oury C, Delvenne P, Marichal T. Neutrophil extracellular traps infiltrate the lung airway, interstitial, and vascular compartments in severe COVID-19. J Exp Med. 2020;217:e20201012. doi: 10.1084/jem.20201012. doi: https://doi.org/10.1084/jem.20201012 https://doi.org/10.1084/jem.20201012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Fuchs TA, Brill A, Duerschmied D, Schatzberg D, Monestier M, Myers DD, Jr, Wrobleski SK, Wakefield TW, Hartwig JH, Wagner DD. Extracellular DNA traps promote thrombosis. Proc Natl Acad Sci U S A. 2010;107:15880–15885. doi: 10.1073/pnas.1005743107. doi: 10.1073/pnas.1005743107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Ammollo CT, Semeraro N, Carratù MR, Colucci M, Semeraro F. Histones differentially modulate the anticoagulant and profibrinolytic activities of heparin, heparin derivatives, and dabigatran. J Pharmacol Exp Ther. 2016;356:305–313. doi: 10.1124/jpet.115.229823. doi: 10.1124/jpet.115.229823. [DOI] [PubMed] [Google Scholar]
- 95.Abrams ST, Su D, Sahraoui Y, Lin Z, Cheng Z, Nesbitt K, Alhamdi Y, Harrasser M, Du M, Foley JH, Lillicrap D, Wang G, Toh CH. Assembly of alternative prothrombinase by extracellular histones initiates and disseminates intravascular coagulation. Blood. 2021;137:103–114. doi: 10.1182/blood.2019002973. doi: 10.1182/blood.2019002973. [DOI] [PubMed] [Google Scholar]
- 96.Fletcher-Sandersjöö A, Bellander BM. Is COVID-19 associated thrombosis caused by overactivation of the complement cascade? A literature review. Thromb Res. 2020;194:36–41. doi: 10.1016/j.thromres.2020.06.027. doi: 10.1016/j.thromres.2020.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Colavita L, Ciprandi G, Salpietro A, Cuppari C. HMGB1: A pleiotropic activity. Pediatr Allergy Immunol. 2020;31(Suppl 26):63–65. doi: 10.1111/pai.13358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Chen L, Long X, Xu Q, Tan J, Wang G, Cao Y, Wei J, Luo H, Zhu H, Huang L, Meng F, Huang L, Wang N, Zhou X, Zhao L, Chen X, Mao Z, Chen C, Li Z, Sun Z, Zhao J, Wang D, Huang G, Wang W, Zhou J. Elevated serum levels of S100A8/A9 and HMGB1 at hospital admission are correlated with inferior clinical outcomes in COVID-19 patients. Cell Mol Immunol. 2020;17:992–994. doi: 10.1038/s41423-020-0492-x. doi: 10.1038/s41423-020-0492-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Chen R, Huang Y, Quan J, Liu J, Wang H, Billiar TR, Lotze MT, Zeh HJ, Kang R, Tang D. HMGB1 as a potential biomarker and therapeutic target for severe COVID-19. Heliyon. 2020;6:e05672. doi: 10.1016/j.heliyon.2020.e05672. doi: 10.1016/j.heliyon.2020.e05672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Meini S, Zanichelli A, Sbrojavacca R, Iuri F, Roberts AT, Suffritti C, Tascini C. Understanding the Pathophysiology of COVID-19: Could the Contact System Be the Key? Front Immunol. 2020;11:2014. doi: 10.3389/fimmu.2020.02014. doi: 10.3389/fimmu.2020.02014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.van de Veerdonk FL, Netea MG, van Deuren M, van der Meer JW, de Mast Q, Bruggemann RJ, van der Hoeven H. Kallikrein-kinin blockade in patients with COVID-19 to prevent acute respiratory distress syndrome. Elife. 2020;9:e57555. doi: 10.7554/eLife.57555. doi: 10.7554/eLife.57555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.DiNicolantonio JJ, McCarty M. Thrombotic complications of COVID-19 may reflect an upregulation of endothelial tissue factor expression that is contingent on activation of endosomal NADPH oxidase. Open Heart. 2020;7:e001337. doi: 10.1136/openhrt-2020-001337. doi: 10.1136/openhrt-2020-001337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Violi F, Oliva A, Cangemi R, Ceccarelli G, Pignatelli P, Carnevale R, Cammisotto V, Lichtner M, Alessandri F, De Angelis M, Miele MC, D’Ettorre G, Ruberto F, Venditti M, Pugliese F, Mastroianni CM. Nox2 activation in Covid-19. Redox Biol. 2020;36:101655. doi: 10.1016/j.redox.2020.101655. doi: 10.1016/j.redox.2020.101655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Celi A, Pellegrini G, Lorenzet R, De Blasi A, Ready N, Furie BC, Furie B. P-selectin induces the expression of tissue factor on monocytes. Proc Natl Acad Sci U S A. 1994;91:8767–8771. doi: 10.1073/pnas.91.19.8767. doi: 10.1073/pnas.91.19.8767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Collier ME, Mah PM, Xiao Y, Maraveyas A, Ettelaie C. Microparticle associated tissue factor is recycled by endothelial cells resulting in enhanced surface tissue factor activity. Thromb Haemost. 2013;110:966–976. doi: 10.1160/TH13-01-0055. [DOI] [PubMed] [Google Scholar]
- 106.Morici N, Bottiroli M, Fumagalli R, Marini C, Cattaneo M. Role of von Willebrand Factor and ADAMTS-13 in the pathogenesis of thrombi in SARS-CoV-2 Infection: Time to Rethink. Thromb Haemost. 2020;120:1339–1342. doi: 10.1055/s-0040-1713400. doi: 10.1055/s-0040-1713400. [DOI] [PubMed] [Google Scholar]
- 107.Bazzan M, Montaruli B, Sciascia S, Cosseddu D, Norbiato C, Roccatello D. Low ADAMTS 13 plasma levels are predictors of mortality in COVID-19 patients. Intern Emerg Med. 2020;15:861–863. doi: 10.1007/s11739-020-02394-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Huisman A, Beun R, Sikma M, Westerink J, Kusadasi N. Involvement of ADAMTS13 and von Willebrand factor in thromboembolic events in patients infected with SARS-CoV-2. Int J Lab Hematol. 2020;42:e211–e212. doi: 10.1111/ijlh.13244. doi: 10.1111/ijlh.13244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Bouck EG, Denorme F, Holle LA, Middelton EA, Blair A, de Laat B, Schiffman JD, Yost CC, Rondina MT, Wolberg AS, Campbell RA. COVID-19 and Sepsis Are Associated with Different Abnormalities in Plasma Procoagulant and Fibrinolytic Activity. Arterioscler Thromb Vasc Biol. 2021;41:401–414. doi: 10.1161/ATVBAHA.120.315338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Fraser DD, Patterson EK, Slessarev M, Gill SE, Martin C, Daley M, Miller MR, Patel MA, Dos Santos CC, Bosma KJ, O’Gorman DB, Cepinskas G. Endothelial Injury and Glycocalyx Degradation in Critically Ill Coronavirus Disease 2019 Patients: Implications for Microvascular Platelet Aggregation. Crit Care Explor. 2020;2:e0194. doi: 10.1097/CCE.0000000000000194. doi: 10.1097/CCE.0000000000000194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Buijsers B, Yanginlar C, de Nooijer A, Grondman I, Maciej-Hulme ML, Jonkman I, Janssen NAF, Rother N, de Graaf M, Pickkers P, Kox M, Joosten LAB, Nijenhuis T, Netea MG, Hilbrands L, van de Veerdonk FL, Duivenvoorden R, de Mast Q, van der Vlag J. Increased Plasma Heparanase Activity in COVID-19 Patients. Front Immunol. 2020;11:575047. doi: 10.3389/fimmu.2020.575047. doi: 10.3389/fimmu.2020.575047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Nizzoli ME, Merati G, Tenore A, Picone C, Consensi E, Perotti L, Ferretti VV, Sambo M, Di Sabatino A, Iotti GA, Arcaini L, Bruno R, Belliato M. Circulating endothelial cells in COVID-19. Am J Hematol. 2020;95:E187–E188. doi: 10.1002/ajh.25881. doi: 10.1002/ajh.25881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Miesbach W. Pathological Role of Angiotensin II in Severe COVID-19. TH Open. 2020;4:e138–e144. doi: 10.1055/s-0040-1713678. doi: 10.1055/s-0040-1713678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Celi A, Cianchetti S, Dell’Omo G, Pedrinelli R. Angiotensin II, tissue factor and the thrombotic paradox of hypertension. Expert Rev Cardiovasc Ther. 2010;8:1723–9. doi: 10.1586/erc.10.161. doi: 10.1586/erc.10.161. [DOI] [PubMed] [Google Scholar]
- 115.Matsuura Y, Yamashita A, Iwakiri T, Sugita C, Okuyama N, Kitamura K, Asada Y. Vascular wall hypoxia promotes arterial thrombus formation via augmentation of vascular thrombogenicity. Thromb Haemost. 2015;114:158–72. doi: 10.1160/TH14-09-0794. doi: 10.1160/TH14-09-0794. [DOI] [PubMed] [Google Scholar]
- 116.Gupta N, Zhao YY, Evans CE. The stimulation of thrombosis by hypoxia. Thromb Res. 2019;181:77–83. doi: 10.1016/j.thromres.2019.07.013. doi: 10.1016/j.thromres.2019.07.013. [DOI] [PubMed] [Google Scholar]
- 117.Whyte CS, Morrow GB, Mitchell JL, Chowdary P, Mutch NJ. Fibrinolytic abnormalities in acute respiratory distress syndrome (ARDS) and versatility of thrombolytic drugs to treat COVID-19. J Thromb Haemost. 2020;18:1548–1555. doi: 10.1111/jth.14872. doi: 10.1111/jth.14872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Medcalf RL, Keragala CB, Myles PS. Fibrinolysis and COVID-19: A plasmin paradox. J Thromb Haemost. 2020;18:2118–2122. doi: 10.1111/jth.14960. doi: 10.1111/jth.14960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Varjú I, Longstaff C, Szabó L, Farkas ÁZ, Varga-Szabó VJ, Tanka-Salamon A, Machovich R, Kolev K. DNA, histones and neutrophil extracellular traps exert anti-fibrinolytic effects in a plasma environment. Thromb Haemost. 2015;113:1289–98. doi: 10.1160/TH14-08-0669. doi: 10.1160/TH14-08-0669. [DOI] [PubMed] [Google Scholar]
- 120.Weiss E, Roux O, Moyer JD, Paugam-Burtz C, Boudaoud L, Ajzenberg N, Faille D, de Raucourt E. Fibrinolysis Resistance: A Potential Mechanism Underlying COVID-19 Coagulopathy. Thromb Haemost. 2020;120:1343–1345. doi: 10.1055/s-0040-1713637. doi: 10.1055/s-0040-1713637. [DOI] [PubMed] [Google Scholar]
- 121.Ibañez C, Perdomo J, Calvo A, Ferrando C, Reverter JC, Tassies D, Blasi A. High D dimers and low global fibrinolysis coexist in COVID19 patients: what is going on in there? J Thromb Thrombolysis. 2020;15:1–5. doi: 10.1007/s11239-020-02226-0. doi: 10.1007/s11239-020-02226-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Wright FL, Vogler TO, Moore EE, Moore HB, Wohlauer MV, Urban S, Nydam TL, Moore PK, McIntyre RC., Jr Fibrinolysis Shutdown Correlation with Thromboembolic Events in Severe COVID-19 Infection. J Am Coll Surg. 2020;231:193–203.e1. doi: 10.1016/j.jamcollsurg.2020.05.007. doi: 10.1016/j.jamcollsurg.2020.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Creel-Bulos C, Auld SC, Caridi-Scheible M, Barker N, Friend S, Gaddh M, Kempton CL, Maier C, Nahab F, Sniecinski R. Fibrinolysis Shutdown and Thrombosis in a COVID-19 ICU. Shock. 2021;55:316–320. doi: 10.1097/SHK.0000000000001635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Zuo Y, Warnock M, Harbaugh A, Yalavarthi S, Gockman K, Zuo M, Madison JA, Knight JS, Kanthi Y, Lawrence DA. Plasma tissue plasminogen activator and plasminogen activator inhibitor-1 in hospitalized COVID-19 patients. Sci Rep. 2021;11:1580. doi: 10.1038/s41598-020-80010-z. doi: 10.1038/s41598-020-80010-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Kang S, Tanaka T, Inoue H, Ono C, Hashimoto S, Kioi Y, Matsumoto H, Matsuura H, Matsubara T, Shimizu K, Ogura H, Matsuura Y, Kishimoto T. IL-6 trans-signaling induces plasminogen activator inhibitor-1 from vascular endothelial cells in cytokine release syndrome. Proc Natl Acad Sci U S A. 2020;117:22351–22356. doi: 10.1073/pnas.2010229117. doi: 10.1073/pnas.2010229117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ranucci M, Sitzia C, Baryshnikova E, Di Dedda U, Cardani R, Martelli F, Corsi Romanelli M. Covid-19-Associated Coagulopathy: Biomarkers of Thrombin Generation and Fibrinolysis Leading the Outcome. J Clin Med. 2020;9:3487. doi: 10.3390/jcm9113487. doi: 10.3390/jcm9113487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Jin X, Duan Y, Bao T, Gu J, Chen Y, Li Y, Mao S, Chen Y, Xie W. The values of coagulation function in COVID-19 patients. PLoS ONE. 2020;15:e0241329. doi: 10.1371/journal.pone.0241329. https://doi.org/10.1371/journal.pone.0241329 https://doi.org/10.1371/journal.pone.0241329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Henry BM, Benoit SW, Hoehn J, Lippi G, Favaloro EJ, Benoit JL. Circulating Plasminogen Concentration at Admission in Patients with Coronavirus Disease 2019 (COVID-19) Semin Thromb Hemost. 2020;46:859–862. doi: 10.1055/s-0040-1715454. DOI https://doi.org/10.1055/s-0040-1715454 https://doi.org/10.1055/s-0040-1715454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Tang N, Bai H, Xiong D, Sun Z. Specific coagulation markers may provide more therapeutic targets in COVID-19 patients receiving prophylactic anticoagulant. J Thromb Haemost. 2020;18:2428–2430. doi: 10.1111/jth.14988. doi: 10.1111/jth.14988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Wu Z, Hu R, Zhang C, Ren W, Yu A, Zhou X. Elevation of plasma angiotensin II level is a potential pathogenesis for the critically ill COVID-19 patients. Crit Care. 2020;24:290. doi: 10.1186/s13054-020-03015-0. doi: 10.1186/s13054-020-03015-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Vaughan DE, Lazos SA, Tong K. Angiotensin II regulates the expression of plasminogen activator inhibitor-1 in cultured endothelial cells. A potential link between the renin-angiotensin system and thrombosis. J Clin Invest. 1995;95:995–1001. doi: 10.1172/JCI117809. doi: 10.1172/JCI117809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Chapman HA, Yang XL, Sailor LZ, Sugarbaker DJ. Developmental expression of plasminogen activator inhibitor type 1 by human alveolar macrophages. Possible role in lung injury. J Immunol. 1990;145:3398–3405. [PubMed] [Google Scholar]
- 133.Idell S, James KK, Levin EG, Schwartz BS, Manchanda N, Maunder RJ, Martin TR, McLarty J, Fair DS. Local abnormalities in coagulation and fibrinolytic pathways predispose to alveolar fibrin deposition in the adult respiratory distress syndrome. J Clin Invest. 1989;84:695–705. doi: 10.1172/JCI114217. doi: 10.1172/JCI114217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Gao Y, Li T, Han M, Li X, Wu D, Xu Y, Zhu Y, Liu Y, Wang X, Wang L. Diagnostic utility of clinical laboratory data determinations for patients with the severe COVID-19. J Med Virol. 2020;92:791–796. doi: 10.1002/jmv.25770. doi: 10.1002/jmv.25770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Semeraro F, Ammollo CT, Caironi P, Masson S, Latini R, Panigada M, Pesenti A, Semeraro N, Gattinoni L, Colucci M. D-dimer corrected for thrombin and plasmin generation is a strong predictor of mortality in patients with sepsis. Blood Transfus. 2020;18:304–311. doi: 10.2450/2019.0175-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Spyropoulos AC, Levy JH, Ageno W, Connors JM, Hunt BJ, Iba T, Levi M, Samama CM, Thachil J, Giannis D, Douketis JD Subcommittee on Perioperative, Critical Care Thrombosis, Haemostasis of the Scientific, Standardization Committee of the International Society on Thrombosis and Haemostasis. Scientific and Standardization Committee communication: Clinical guidance on the diagnosis, prevention, and treatment of venous thromboembolism in hospitalized patients with COVID-19. J Thromb Haemost. 2020;18:1859–1865. doi: 10.1111/jth.14929. doi: 10.1111/jth.14929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Zhang C, Wu Z, Li JW, Zhao H, Wang GQ. Cytokine release syndrome in severe COVID-19: interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality. Int J Antimicrob Agents. 2020;55:105954. doi: 10.1016/j.ijantimicag.2020.105954. doi: 10.1016/j.ijantimicag.2020.105954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Khiali S, Khani E, Entezari-Maleki T. A Comprehensive Review of Tocilizumab in COVID-19 Acute Respiratory Distress Syndrome. J Clin Pharmacol. 2020;60:1131–1146. doi: 10.1002/jcph.1693. doi: 10.1002/jcph.1693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Polycarpou A, Howard M, Farrar CA, Greenlaw R, Fanelli G, Wallis R, Klavinskis LS, Sacks S. Rationale for targeting complement in COVID-19. EMBO Mol Med. 2020;12:e12642. doi: 10.15252/emmm.202012642. doi: 10.15252/emmm.202012642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Risitano AM, Mastellos DC, Huber-Lang M, Yancopoulou D, Garlanda C, Ciceri F, Lambris JD. Complement as a target in COVID-19? Nat Rev Immunol. 2020;20:343–344. doi: 10.1038/s41577-020-0320-7. doi: 10.1038/s41577-020-0320-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Mastellos DC, Pires da Silva BGP, Fonseca BAL, Fonseca NP, Auxiliadora-Martins M, Mastaglio S, Ruggeri A, Sironi M, Radermacher P, Chrysanthopoulou A, Skendros P, Ritis K, Manfra I, Iacobelli S, Huber-Lang M, Nilsson B, Yancopoulou D, Connolly ES, Garlanda C, Ciceri F, Risitano AM, Calado RT, Lambris JD. Complement C3 vs C5 inhibition in severe COVID-19: Early clinical findings reveal differential biological efficacy. Clin Immunol. 2020;220:108598. doi: 10.1016/j.clim.2020.108598. doi: 10.1016/j.clim.2020.108598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Barnes BJ, Adrover JM, Baxter-Stoltzfus A, Borczuk A, Cools-Lartigue J, Crawford JM, Daßler-Plenker J, Guerci P, Huynh C, Knight JS, Loda M, Looney MR, McAllister F, Rayes R, Renaud S, Rousseau S, Salvatore S, Schwartz RE, Spicer JD, Yost CC, Weber A, Zuo Y, Egeblad M. Targeting potential drivers of COVID-19: Neutrophil extracellular traps. J Exp Med. 2020;217:e20200652. doi: 10.1084/jem.20200652. doi: 10.1084/jem.20200652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Okur HK, Yalcin K, Tastan C, Demir S, Yurtsever B, Karakus GS, Kancagi DD, Abanuz S, Seyis U, Zengin R, Hemsinlioglu C, Kara M, Yildiz ME, Deliceo E, Birgen N, Pelit NB, Cuhadaroglu C, Kocagoz AS, Ovali E. Preliminary report of in vitro and in vivo effectiveness of dornase alfa on SARS-CoV-2 infection. New Microbes New Infect. 2020;37:100756. doi: 10.1016/j.nmni.2020.100756. doi: 10.1016/j.nmni.2020.100756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Weber AG, Chau AS, Egeblad M, Barnes BJ, Janowitz T. Nebulized in-line endotracheal dornase alfa and albuterol administered to mechanically ventilated COVID-19 patients: a case series. Mol Med. 2020;26:91. doi: 10.1186/s10020-020-00215-w. doi: 10.1186/s10020-020-00215-w. [DOI] [PMC free article] [PubMed] [Google Scholar]