Abstract
Reliable data on causes of death (COD) in preterm infants are needed to assess perinatal care and current clinical guidelines. In this retrospective observational analysis of all deceased preterm infants born < 37 weeks’ gestational age (n = 278) at a Swedish tertiary neonatal intensive care unit, we compared preliminary COD from Medical Death Certificates with autopsy defined COD (2002–2018), and assessed changes in COD between two periods (period 1:2002–2009 vs. period 2:2011–2018; 2010 excluded due to centralized care and seasonal variation in COD). Autopsy was performed in 73% of all cases and was more than twice as high compared to national infant autopsy rates (33%). Autopsy revised or confirmed a suspected preliminary COD in 34.9% of the cases (23.6% and 11.3%, respectively). Necrotizing enterocolitis (NEC) as COD increased between Period 1 and 2 (5% vs. 26%). The autopsy rate did not change between the two study periods (75% vs. 71%). We conclude that autopsy determined the final COD in a third of cases, while the incidence of NEC as COD increased markedly during the study period. Since there is a high risk to determine COD incorrectly based on clinical findings in preterm infants, autopsy remains a valuable method to obtain reliable COD.
Subject terms: Health care, Medical research, Signs and symptoms
Introduction
The autopsy has been fundamental to understand anatomy and pathophysiology in medicine and neonatology. Despite proactive obstetric and neonatal care, prematurity remains the most common worldwide cause of death (COD) among children under the age of 5 years1,2. As autopsy is “the gold standard” for accurately determining COD, autopsy contributes to the continued development of neonatal care and possibly improved survival in preterm infants3,4.
When reporting causes of death, it is customary to use the International Classification of Diseases (ICD) by the WHO5. To assign prematurity as the COD is not recommended by the WHO, unless no other COD can be confirmed. Despite these recommendations, there has been a continued use of prematurity as COD instead of defining the specific COD6–8. In mortality reports, ICD-codes are grouped into categories to provide coherent descriptions, although, classification systems often differ between reports and/or countries. Based on Wigglesworth’s classification of perinatal mortality9, the International Collaborative Effort (ICE) offered a classification method for infant mortality in 1989 to enable international comparisons10. Still, reports continue to use different classifications11–14, and some, including the ICE, use the classification of prematurity related deaths despite the recommendations from the WHO8. New classification systems have been proposed, yet no consensus has been reached15,16. In Sweden, death is certified in two stages: the Notification of Death and the Medical Death Certificate (MDC)6. Writing a MDC to define COD utilizes the WHO guidelines. A chronology of events leading to death is listed in the MDC, including the duration of each condition. The direct COD is defined as the final condition causing death5,6.
Unfortunately, there is a global trend for decreasing autopsy rates in infants17, which has also been seen in Sweden. A report from 2014 showed that infant autopsy rates had decreased from 71 to 35% for boys and from 64 to 31% for girls since 19877. The former chief editor of JAMA George Lundberg warned in 1988 that autopsy reduction will negatively affect provided care, since lack of quality control of care will result in reduced knowledge of beneficial or negative effects of treatments18. Accurately defined COD reports are also needed for reliable and meaningful statistics. Records of preterm deaths are frequently incorrect, with flaws in the definition of live births, underreporting of births and deaths, and inaccurately stated CODs19. Some have advocated for postmortem MRI as a supplement to autopsies20,21. Although MRI brings some diagnostic value, it has been shown to miss more than half of the findings an autopsy would have provided, and neglects histological and microbiological features20,21.
The aim of this study was to determine the incidence of autopsy in preterm infants treated at our NICU between 2002 and 2018, and define how many of the preliminary CODs were revised by these autopsies. We also wanted to determine if there have been changes in the CODs over time. A better understanding of the significance of accurately determining the COD by autopsy in preterm infants may lead to the development of standardized protocols for defining COD.
Results
The mortality for all premature infants born in Uppsala was 3.8% (n = 105/2763) during Period 1 and 5.0% (n = 160/3200) during Period 2 (p = 0.059). A total of 278 infants died during 2002–2018. No differences in birth weight, gestational age, degree of immaturity or age at death was found between the two periods (Table 1). There was a higher percentage of boys who died compared to girls in Period 2 (63.1% vs. 36.9%; p = 0.006; Table 1).
Table 1.
Characteristics of deceased infants.
| Both periods | Period 1 2002–2009 | Period 2 2011–2018 | p value | |
|---|---|---|---|---|
| Population size | 265 | 105 | 160 | – |
| Gestational age, weeks—median (range) | 25 (21–36) | 26 (21–36) | 25 (21–36) | 0.255 |
| Birth weight, g—median (range) | 742 (312–3970) | 800 (312–3655) | 710 (322–3970) | 0.256 |
| Gestational age of those deceased—number (% of total in period) | ||||
| 32–36 weeks | 37 (14.0) | 16 (15.2) | 21 (13.1) | 0.627 |
| 28–31 weeks | 51 (19.2) | 19 (18.1) | 32 (20.0) | 0.700 |
| < 28 weeks | 177 (66.8) | 70 (66.7) | 107 (66.9) | 0.972 |
| Distribution of sex—number (% of total in period) | ||||
| Girls | 109 (41.1) | 50 (47.6) | 59 (36.9) | 0.265 |
| Boys | 156 (58.9) | 55 (52.4) | 101 (63.1) | 0.373 |
| Postnatal age at death in days—median (range) | 7(0–308) | 5 (0–239) | 8 (0–308) | 0.361 |
| Timing of death—number (% of total in period) | ||||
| 0–24 h | 38 (14.3) | 15 (14.3) | 23 (14.4) | 0.234 |
| 1–6 days | 90 (34.0) | 41 (39.0) | 49 (30.6) | 0.230 |
| 7–28 days | 80 (30.2) | 29 (27.6) | 51 (31.9) | 0.635 |
| > 28 days, up to one year | 57 (21.5) | 20 (19.0) | 37 (23.1) | 0.429 |
Autopsy data are summarized in Table 2. The autopsy rate over the whole study period was 73.0% (n = 203/278), and did not change between the study periods (75% vs. 71%). Boys were autopsied in 70.2% of cases and girls in 76.9%. The main reason for an autopsy not being performed was parental refusal (n = 61/75; 81.3%). In 14.7% cases, there was no declared reason for autopsy not being performed. The frequency of autopsies was lower in those who died after 7 days of life (n = 96/141; 64.9%) as compared to those who died earlier (n = 107/137; 78.1%). Postmortem examinations changed the preliminary COD or confirmed the suspected COD in 35.0% (n = 71/203) of all autopsied cases (23.7% of the preliminary CODs were revised completely, and 11.3% of the suspected CODs were confirmed). In 5% (n = 10/203) of the autopsied cases it was not possible to define if autopsy altered the COD, due to lack of data on the preliminary COD. In Supplement 1, the preliminary COD and autopsy confirmed COD is listed for each individual where autopsy completely revised the COD. The CODs where autopsy enabled confirmation of a suspected COD are also listed in Supplement 1.
Table 2.
Number of performed autopsies, diagnosis after autopsy of revised and confirmed suspected causes of death (CODs), and reasons for not performing autopsy (2002–2018).
| Number | Percent | |
|---|---|---|
| Number of performed autopsies | 203/278 | 73.0 |
| Revised CODs after autopsy | 48/203 | 23.6 |
| Confirmed suspected CODs after autopsy | 23/203 | 11.3 |
| Not changed CODs after autopsy | 112/203 | 55.2 |
| Clinical CODs more precise than autopsy CODs* | 10/203 | 4.9 |
| Not available | 10/203 | 4.9 |
| Revised and confirmed suspected CODs after autopsy | 71/203 | 34.9 |
| Congenital anomaly | 6/71 | 8.5 |
| Asphyxia | 1/71 | 1.4 |
| Respiratory | 13/71 | 18.3 |
| IVH | 4/71 | 5.6 |
| Infection/sepsis | 21/71 | 29.6 |
| NEC | 14/71 | 19.7 |
| Other | 12/71 | 16.9 |
| Reasons for not performing autopsy | 75/278 | 27.0 |
| Parental refusal | 61/75 | 81.3 |
| Lack of medical indication | 2/75 | 2.7 |
| Other | 1/75 | 1.3 |
| Unknown | 11/75 | 14.7 |
*If the autopsy defined the COD as “prematurity”, the clinical diagnosis was used as a definitive COD. In 3 of these 10 cases, the clinical COD was also prematurity.
Cause of death: comparison between Period 1 and Period 2
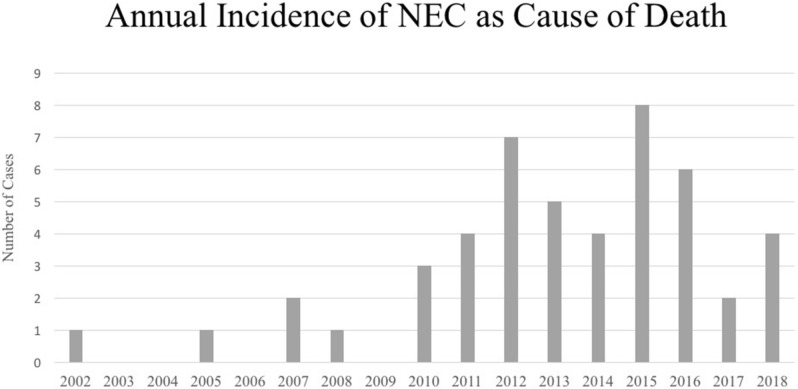
NEC as the COD increased from 5.0% (n = 5/101) to 25.2% (n = 40/159) (p < 0.001) from Period 1 to Period 2, whereas IVH as the COD decreased (p = 0.042) (Table 3). The annual incidence of NEC as a COD is shown in Fig. 1. Period 2 had fewer cases of RDS (p = 0.039) as COD than Period 1. The isolated spontaneous intestinal perforations (n = 4) were either consequences of anomalies or perforation without NEC. The type of infections as the COD during Period 1 and Period 2 was viral (n = 2), fungal (n = 2), bacterial pneumonia (n = 3) and systemic bacterial (n = 34).
Table 3.
Cause of death in relation to year of birth.
| Both periods | Period 1 2002–2009 | Period 2 2011–2018 | p value | |
|---|---|---|---|---|
| Total deaths (n) | 265 | 105 | 160 | – |
| Total deaths with determined cause of death (n) | 260 | 101 | 159 | – |
| Cause of death—number of cases (% of total) | n | n (%) | n (%) | |
|---|---|---|---|---|
| Congenital anomalies | 18 | 10 (9.9) | 8 (5.0) | 0.132 |
| Neural anomaly | 3 | 2 (2.0) | 1 (0.6) | 0.562 |
| Cardiac anomaly | 3 | 2 (2.0) | 1 (0.6) | 0.562 |
| Other anomaly | 9 | 3 (3.0) | 6 (3.8) | 1.000 |
| Chromosomal abnormalities | 3 | 3 (3.0) | 0 (0.0) | 0.058 |
| Asphyxia | 17 | 10 (9.6) | 7 (4.4) | 0.080 |
| Intrauterine | 8 | 4 (4.0) | 4 (2.5) | 0.715 |
| Perinatal | 8 | 6 (5.9) | 2 (1.3) | 0.059 |
| Postnatal | 1 | 0 (0.0) | 1 (0.6) | 1.000 |
| Respiratory | 75 | 31 (30.7) | 44 (27.3) | 0.674 |
| RDS | 10 | 7 (6.9) | 3 (1.9) | 0.039 |
| BPD | 12 | 5 (5.0) | 7 (4.4) | 0.837 |
| Pulmonary hypoplasia | 20 | 8 (7.9) | 12 (7.5) | 0.912 |
| PPHN | 10 | 5 (5.0) | 5 (3.1) | 0.461 |
| Pneumothorax | 5 | 1 (1.0) | 4 (2.5) | 0.651 |
| Haemothorax | 4 | 3 (3.0) | 1 (0.6) | 0.302 |
| Miscellaneous | 14 | 2 (2.0) | 12 (7.5) | 0.087 |
| IVH | 22 | 13 (12.9) | 9 (5.7) | 0.042 |
| Infection/sepsis | 41 | 19 (18.8) | 22 (13.8) | 0.283 |
| Early | 11 | 3 (3.0) | 8 (5.0) | 0.537 |
| Late | 30 | 16 (15.8) | 14 (8.8) | 0.083 |
| NEC | 45 | 5 (5.0) | 40 (25.2) | < 0.001 |
| NEC with Sepsis | 16 | 2 (2.0) | 14 (8.8) | 0.053 |
| NEC without sepsis | 29 | 3 (3.0) | 26 (16.4) | 0.001 |
| Other | 42 | 13 (12.9) | 29 (18.2) | 0.252 |
| Shock/anemia/bleeding | 8 | 1 (1.0) | 7 (4.4) | 0.156 |
| Volvulus/malrotation | 4 | 1 (1.0) | 3 (1.9) | 1.000 |
| Isolated spontaneous intestinal perforation | 4 | 0 (0.0) | 4 (2.5) | 0.160 |
| Metabolic/electrolyte/endocrine disorders | 4 | 2 (2.0) | 2 (1.3) | 0.643 |
| Tumor | 3 | 0 (0.0) | 3 (1.9) | 0.285 |
| Twin-twin transfusion syndrome | 2 | 2 (1.0) | 1 (0.6) | 0.562 |
| CNS-related | 3 | 1 (1.0) | 2 (1.3) | 1.000 |
| Other | 8 | 4 (4.0) | 4 (2.5) | 0.715 |
| Prematurity | 3 | 2 (2.0) | 1 (0.6) | 0.562 |
| Unknown | 2 | 0 (0.0) | 2 (1.3) | 0.523 |
Figure 1.
Annual incidence of NEC as cause of death from 2002 to 2018.
Cause versus timing of death
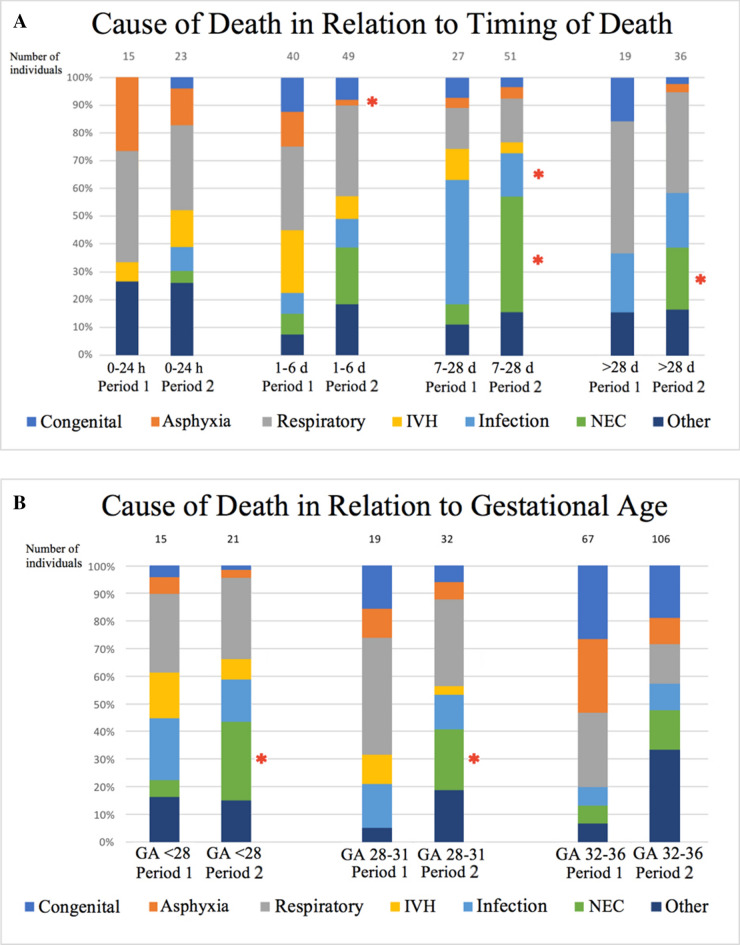
As shown in Fig. 2a, fewer patients died due to perinatal asphyxia between day 1–6 (p = 0.050) in Period 2 than in Period 1. Cases with NEC as the COD increased for infants who died on days 7–28 and after 28 days (p = 0.001 and p = 0.025, respectively), while infection as the COD decreased (p = 0.006) between Period 1 and 2 for those infants who died on days 7–28. Median days of life for patients with NEC as cause of death was 4 (IQR = 8.5) in Period 1 and 11 (IQR = 11.5) in Period 2 (p = 0.023).
Figure 2.
(a) Distribution in percent of causes of death at various time intervals (0–24 h; 1–6 days; 7–28 days; > 28 days) during Period 1 and Period 2. *p < 0.05; significant change between Period 1 and Period 2 within each time interval. Congenital, congenital anomalies; Respiratory, respiratory causes; IVH, intraventricular hemorrhage; Infection, infection with septicemia; NEC, necrotizing enterocolitis. See Table 4 for further categorization of causes of death. (b) Distribution in percent of causes of death at various degrees of immaturity (gestational age < 28 weeks, 28–31 weeks and 32–36 weeks) during Period 1 and Period 2. *p < 0.05; significant change between Period 1 and Period 2 within each degree of immaturity. GA; gestational age; Congenital, congenital anomalies; Respiratory, respiratory causes; IVH, intraventricular hemorrhage; Infection, infection with septicemia; NEC, necrotizing enterocolitis. See Table 4 for further categorization of causes of death.
Cause of death versus degree of immaturity
As presented in Fig. 2b, the incidence of NEC as COD in Period 2 was higher than in Period 1 for very preterm (p = 0.028) and extremely preterm infants (p < 0.001). There were no other significant differences in COD in the gestational age groups between Periods 1 and 2 (Fig. 2b).
Discussion
In this study involving 278 preterms, we found an autopsy rate of 73% and a comparable distribution between sexes. The autopsy rate in this cohort was more than twice as high compared to national rates of infant autopsies in Sweden7. This might be explained by our local tradition of offering postmortem investigations at a regular basis to all parents in order to increase autopsy rates in preterm infants at UUCH, and thereby improving our knowledge of the COD. Infants who died at a higher postnatal age were autopsied less frequently than those who died at a lower postnatal age. Although unclear from this study, this difference in autopsy rates depending on postnatal age may be a consequence of there being more time to find a definitive diagnosis during a longer stay at the NICU, which is partly substantiated by de Séveaux et al. who found only minor additional findings with higher postnatal age at death21. Parents refusing to consent to autopsy was the main reason for autopsies not taking place, which is in agreement with previous findings21,22. Reduced parental consent may depend on the information provided by the health care regarding the procedure and potential benefit of an autopsy22,23. In many cases, the responsibility for obtaining consent for autopsy lies with the medical staff who may not fully understand the importance of the autopsy when discussing the procedure with the parents22,23. For the parents, the correct COD for their lost infant may be of importance for future pregnancy decisions, since genetic anomalies or syndromes can be difficult to diagnose solely on clinical examination of very preterm infants21.
Our main finding is that post-mortem examinations contributed considerably to determining the COD as evidenced by changing or confirming the suspected preliminary COD in a third of cases. This is consistent with results from previous studies, where a percentage of 25–36% CODs were found to be altered by autopsy in centers with generally low autopsy rates21,22,24. A higher autopsy rate might be expected to decrease the number of times the preliminary COD is revised, given that in studies with lower autopsy rates, the autopsy is performed when the COD is unclear to the clinician17,21,25. However, as shown in this study, even with relatively high autopsy rates the risk of changing the preliminary COD remains high. Thus, our data are consistent with the notion that improving autopsy rates will yield a higher quality of COD and therefore be more useful in understanding COD trends in the NICU.
The decrease of IVH as a COD between the two studied periods, is consistent with national findings in Sweden26 and may be attributed to more refined ventilatory strategies27 and more consistent treatment with antenatal corticosteroids28. Being a referral hospital for our region, the reduced rate of RDS as a COD could be due to the changed policy during Period 2 of referring all mothers at risk of delivering prematurely before 28 weeks GA to our hospital, an observation that needs further evaluation. We found that the incidence of congenital anomalies as a COD in our cohort was lower than in other comparable studies in high income countries29. One possible explanation could be consistent maternity care, improved prenatal screening for congenital anomalies and Swedish abortion practices. Another reason could be the targeting of a more precise COD in our study rather than applying a general diagnosis like congenital anomalies as the primary COD.
The occurrence of NEC as the COD was significantly higher during Period 2 as compared to Period 1. The increase of NEC was especially evident in extremely preterm infants and deaths during day 7–28 and after 28 days of life. This is likely due to NEC being a disease occurring after 1–2 of weeks of life30 and that the risk of NEC increases with decreasing gestational age31. Another potential factor in the overall increase of NEC as a COD could be that improvements in clinical care over time enable the infants to survive long enough to develop NEC, as has been suggested previously32. The median age at death for patients with NEC as COD increased significantly between Period 1 and Period 2. An increased incidence of NEC in Sweden has also been reported for these two time periods (2004–2007 vs. 2014–2016; 6% vs. 10%) with a simultaneously increased survival in the lowest gestational ages (70% vs. 77%)33. A lower reported incidence of surgically treated NEC from our unit to the Swedish Neonatal Quality Register in infants born between 22 and 27 weeks GA (22–24 GA, 5% vs. 7.5%; 25–27 GA, 1.5% vs. 3%)26, with similar to lower mortality rates, suggests that other factors than local case clustering of NEC from viral or bacterial outbreaks (so called epidemic NEC) contribute to NEC incidence in our study34,35. The increase in NEC as a COD cannot be only ascribed to more autopsies allowing more detailed diagnosis, since the autopsy rate was similar during both periods. Nor could any difference in breast-feeding strategies (same during both periods) or probiotic strategies (not introduced) explain this redistribution of CODs with higher NEC during the second period.
The retrospective observational study design seems relevant for this type of study, and seems to define the prevalence of different CODs and the value of autopsy in determining the definitive COD. However, given the observational design it is difficult to draw too strong conclusions regarding the causes for the observed changes in COD over time, and particularly for NEC, why further research is needed to explain these differences in COD. Structured national and international surveying of neonatal care is increasing worldwide36, and might have impact on understanding COD and changes in COD over time. Some other studies on infant mortality have systematically assigned prematurity as the major COD13, ignoring the recommendations of the WHO8. We found 10 cases in this cohort where prematurity was registered as the COD, however 7 of them could later be sorted into other categories using data from medical records and/or autopsies. Only in 3 out of 278 cases did prematurity remain as the final COD. The low rate of prematurity as COD demonstrates the feasibility of following the WHO recommendation regarding not assigning prematurity as the primary COD. Some of the complications of prematurity are difficult to diagnose in preterm infants, since symptoms may not be specific or may appear after a delay. In our study, nearly 75% of all diagnoses were autopsy-verified, substantiating the COD. All autopsies were performed by the same pathologist, which results in a high level of qualitative continuity in performing autopsies22. In addition, the neonatologist involved in the patient’s care usually attended the postmortem examination, thereby providing the pathologist with more detailed pre-mortem clinical findings, which facilitated the examination and supported the definitive COD.
This study highlights the importance of post-mortem examinations, which continue to yield valuable information and contribute to determine the definitive COD in preterm infants. Besides observing an increase in NEC and a decrease in IVH between the two periods, we found that the results from the autopsy revised or confirmed the suspected preliminary COD in a third of cases during the entire study period. Interestingly, 73% of all deceased preterm infants during this period were autopsied and not only the ones with uncertain diagnoses. Our findings suggest that it would be beneficial to reverse the negative trend in autopsy rates in preterm infants, in order to obtain reliable COD data.
Methods
Study setting and ethical consideration
This study is a retrospective observational study of all preterm deaths from 2002 to 2018 and a 2-period sub-analysis (2002–2009 vs. 2011–2018) in a tertiary NICU at Uppsala University Children’s Hospital (UUCH). In order to exclude overlap of early and late deaths, and seasonal variations in COD, 2010 was excluded from the 2-period analysis. The study has been approved by the Regional Ethical Review Board of Uppsala, D:nr 2019-05033, and by the Uppsala Biobank, D:nr BbA-827-2019-094. With these endorsements infants were legally approved to be included in this study. Parental informed consent to perform an autopsy was obtained. All methods in this study were performed in accordance with relevant operating guidelines and regulations. Data were de-identified before analysis, according to operative ethical regulations.
Inclusion of participants
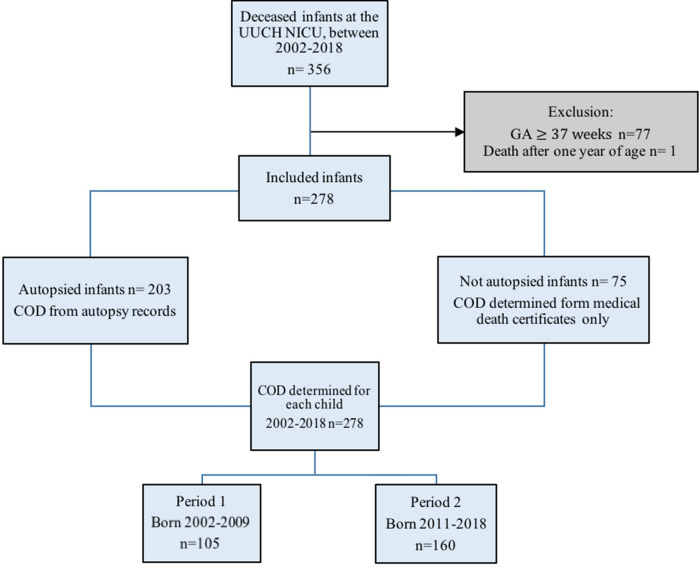
Figure 3 shows a flowchart of the selection of participants. All live born infants admitted to our NICU who subsequently died during the period of 2002 to 2018 were extracted from the Swedish Neonatal Quality Registry (SNQ) (n = 356). To study solely infant mortality, patients who died after 1 year of age were excluded (n = 1). Term infants (≥ 37 weeks gestation) were excluded (n = 77). Thus, there were a total of 278 infants that met inclusion criteria and had none of the exclusion criteria.
Figure 3.
Inclusion of participants, data extraction and period division.
For the comparison between preliminary COD and definitive COD according to autopsy, there were 203 infants with an autopsy (Fig. 3). Medical Death Certificate (MDCs) were examined to determine preliminary cause of death. If multiple MDCs were issued, the MDC issued prior to autopsy was used to avoid the preliminary COD being influenced by autopsy findings. If the MDC was issued after autopsy findings, the discharge note was examined to determine the presumed COD prior to autopsy. MDC and the discharge note were written by the attending physician well acquainted with the patient, and confirmed by the head physician in charge of the ward. An autopsy included a macroscopic review of tissues with a neonatologist present, immune-histological analysis linked to symptomology, bacterial cultures and PCR-analysis if suspicion for viral infection from clinical symptoms or from macroscopic findings was present. The present neonatologist provided clinical data, contributing to the definitive cause of death as determined by autopsy. All infant autopsies were performed by the same pathologist and none was restricted in scope. Parental informed consent was required for autopsy. All parents are offered autopsy by the attending physician on a regular basis, to define the definitive COD and/or other diseases relevant for the course of care and for future pregnancies. For the individuals who were not autopsied, the reason for an autopsy not being performed was defined.
Data extraction and study variables
Perinatal and postnatal data were collected from the SNQ. Perinatal data included time of birth, GA, sex, and birth weight. GA was documented as complete weeks and days, determined by ultrasound. Postnatal data included whether autopsy was performed or not, days of life, and if recorded, why an autopsy was not performed. If the autopsy was not performed due to parental reluctance no retrospective inquiry was performed, since this was considered ethically inappropriate. Missing data in SNQ were complemented by retrieving Medical Death Certificates (MDC) and charts from Cosmic (electronic journal database after 2008) or from Regional archives (before 2008). Autopsy records were collected from the pathology chart system Sympathy. Premature birth rates for the Uppsala Region were obtained from SNQ. Cause of death was determined according to ICD-10 codes8. Definitive COD was determined from autopsy records for those who underwent autopsy (n = 203). For the rest (n = 75), MDCs and discharge notes were used for definitive COD. Causes of death were categorized by groups based on ICE10 and further refined in alignment with categories used in recent publications11–13. The final categories are presented in Table 4. The categories were established prior to data collection, to avoid the categorization being influenced by the data.
Table 4.
Categories for causes of death.
| Congenital anomalies | Intraventricular hemorrhage |
| Neural anomaly | Infection/sepsis |
| Cardiac anomaly | Infection/sepsis, early |
| Other anomaly | Infection/sepsis, late |
| Chromosomal abnormalities | Necrotizing enterocolitis (NEC) |
| Asphyxia | NEC without sepsis |
| Intrauterine | NEC with sepsis |
| Perinatal | Other |
| Postnatal | Shock/anemia/bleeding |
| Respiratory causes | Volvulus/malrotation |
| Bronchopulmonary dysplasia | Isolated spontaneous intestinal perforation |
| Respiratory distress syndrome | Metabolic/electrolyte/endocrine disorders |
| Pulmonary hypoplasia | Tumor |
| Persistent pulmonary hypertension | Twin-to-twin transfusion syndrome |
| Pneumothorax | CNS-related |
| Haemothorax | Other |
| Miscellaneous | Prematurity |
For autopsied infants, MDCs were examined to compare preliminary and definitive COD. If the MDC was issued after autopsy findings, the discharge note was examined to determine the presumed COD prior to autopsy. The preliminary COD and the definitive COD were compared to see if the definitive COD was altered due to information arising from the autopsy. The outcome after autopsy could be one of three: no change of COD; revised COD; or confirmed suspected but not yet established COD. Suspected COD could for instance be a suspected infection, but without positive cultures or identification of the location of the infection. If COD from the autopsy was “Prematurity” (n = 10), but the clinicians found a more precise COD, the more precise COD was used as the definitive COD, in line with the recommendations from the WHO8.
Statistical analyses
Descriptive statistics were calculated in Excel, Version 15.27 (161010). The rest of the statistical analysis was performed in SPSS version 26. A p value < 0.05 was considered significant. Comparisons between groups for continuous data were done using the Mann–Whitney U-test. For trend difference analysis in COD from 2002 to 2009 (n = 105) and 2011–2018 (n = 160), both autopsied and not autopsied infants were included. A chi-square test was used to compare the frequency of each diagnosis between the two periods, and complemented with a Fisher’s exact test if less than 5 occurrences were observed. The comparison between preliminary COD and definitive COD according to autopsy was performed on autopsied individuals from 2002 to 2018 (n = 203).
Supplementary Information
Acknowledgements
The authors are indebted to our secretary Maria Wibeck, for her skilled and tireless help in collecting data from SNQ.
Abbreviations
- BPD
Bronchopulmonary dysplasia
- COD
Cause of death
- IVH
Intraventricular hemorrhage
- MDC
Medical death certificate
- NEC
Necrotizing enterocolitis
- RDS
Respiratory distress syndrome
- SNQ
Swedish neonatal quality registry
- UUCH
Uppsala University Children’s Hospital
Author contributions
A.H. performed the collection of data, statistical analysis, wrote and edited the manuscript. L.M. contributed through data analysis, writing and revising of the manuscript. K.E. performed all autopsies included in this study and assisted in data collection. L.D.N. partook in data analysis as well as writing and revising the manuscript. R.S. was the one to set up the study and further partook in data collection, data analysis, writing and revising of the manuscript. The manuscript has been read and approved for submission by all authors.
Funding
Open access funding provided by Uppsala University. HKH Kronprinsessan Lovisas Stiftelse (Grant Number 2018-00459).
Data availability
All data generated or analyzed during this study are included in this published article (and its Supplementary Information files).
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-93358-7.
References
- 1.Chawanpaiboon S, Vogel JP, Moller A-B, Lumbiganon P, Petzold M, Hogan D, et al. Global, regional, and national estimates of levels of preterm birth in 2014: a systematic review and modelling analysis. Lancet Glob. Health. 2019;7(1):e37–46. doi: 10.1016/S2214-109X(18)30451-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.GBD 2016 Causes of Death Collaborators Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: a systematic analysis for the global burden of disease study 2016. Lancet. 2017;390(10100):1151–1210. doi: 10.1016/S0140-6736(17)32152-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Blencowe H, Cousens S, Oestergaard MZ, Chou D, Moller A-B, Narwal R, et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: a systematic analysis and implications. Lancet. 2012;379(9832):2162–2172. doi: 10.1016/S0140-6736(12)60820-4. [DOI] [PubMed] [Google Scholar]
- 4.Petrou S, Henderson J, Bracewell M, Hockley C, Wolke D, Marlow N. Pushing the boundaries of viability: the economic impact of extreme preterm birth. Early Hum. Dev. 2006;82(2):77–84. doi: 10.1016/j.earlhumdev.2006.01.002. [DOI] [PubMed] [Google Scholar]
- 5.World Health Organization . ICD-10: International statistical classification of diseases and related health problems. World Health Organization; 2011. [Google Scholar]
- 6.Brooke HL, Talbäck M, Hörnblad J, Johansson LA, Ludvigsson JF, Druid H, et al. The Swedish cause of death register. Eur. J. Epidemiol. 2017;32(9):765–773. doi: 10.1007/s10654-017-0316-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.National Board of Health and Welfare. Causes of Death 2014 [Internet]. 2015 https://www.socialstyrelsen.se/globalassets/sharepoint-dokument/artikelkatalog/statistik/2015-8-1.pdf. Accessed 3 Oct 2019.
- 8.World Health Organization. The WHO application of ICD-10 to deaths during the perinatal period: ICD-PM [Internet]. World Health Organization; 2016 https://apps.who.int/iris/bitstream/handle/10665/249515/9789241549752-eng.pdf?sequence=1. Accessed 15Oct 2019.
- 9.Wigglesworth JS. Monitoring perinatal mortality. A pathophysiological approach. Lancet. 1980;2(8196):684–686. doi: 10.1016/S0140-6736(80)92717-8. [DOI] [PubMed] [Google Scholar]
- 10.Alberman E, Bergsjø P, Cole S, Evans S, Hartford R, Hoffman H, et al. International Collaborative Effort (ICE) on birthweight; plurality; and perinatal and infant mortality. I: Methods of data collection and analysis. Acta Obstet. Gynecol. Scand. 1989;68(1):5–10. doi: 10.3109/00016348909087681. [DOI] [PubMed] [Google Scholar]
- 11.Berrington JE, Hearn RI, Bythell M, Wright C, Embleton ND. Deaths in preterm infants: changing pathology over 2 decades. J. Pediatr. 2012;160(1):49–53.e1. doi: 10.1016/j.jpeds.2011.06.046. [DOI] [PubMed] [Google Scholar]
- 12.Park JH, Chang YS, Sung S, Ahn SY, Park WS. Trends in overall mortality, and timing and cause of death among extremely preterm infants near the limit of viability. Gay N, editor. PLoS ONE. 2017;12(1):e0170220. doi: 10.1371/journal.pone.0170220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Patel RM, Kandefer S, Walsh MC, Bell EF, Carlo WA, Laptook AR, et al. Causes and timing of death in extremely premature infants from 2000 through 2011. N. Engl. J. Med. 2015;372(4):331–340. doi: 10.1056/NEJMoa1403489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Stoll BJ, Hansen NI, Bell EF, Walsh MC, Carlo WA, Shankaran S, et al. Trends in care practices, morbidity, and mortality of extremely preterm neonates, 1993–2012. JAMA. 2015;314(10):1039–1051. doi: 10.1001/jama.2015.10244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dias BAS, Santos ETD, Andrade MAC. Classification systems for avoidability of infant deaths: different methods, different repercussions? Cad Saude Publica. 2017;33(5):e00125916. doi: 10.1590/0102-311x00125916. [DOI] [PubMed] [Google Scholar]
- 16.Nakamura AM, Dove MS, Minnal A, Damesyn M, Curtis MP. Infant mortality: development of a proposed update to the Dollfus classification of infant deaths. Public Health Rep. 2015;130(6):632–642. doi: 10.1177/003335491513000613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bassat Q, Castillo P, Alonso PL, Ordi J, Menéndez C. Resuscitating the dying autopsy. PLoS Med. 2016;13(1):e1001927. doi: 10.1371/journal.pmed.1001927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lundberg GD. Now is the time to emphasize the autopsy in quality assurance. JAMA. 1988;260(23):3488–3488. doi: 10.1001/jama.1988.03410230106040. [DOI] [PubMed] [Google Scholar]
- 19.Paulson J, Ramsini W. Unregistered deaths among extremely low birthweight infants—Ohio, 2006. JAMA. 2008;299(3):282–283. doi: 10.1001/jama.299.3.282. [DOI] [Google Scholar]
- 20.Cohen MC, Paley MN, Griffiths PD, Whitby EH. Less invasive autopsy: benefits and limitations of the use of magnetic resonance imaging in the perinatal postmortem. Pediatr. Dev. Pathol. 2008;11(1):1–9. doi: 10.2350/07-01-0213.1. [DOI] [PubMed] [Google Scholar]
- 21.de Sévaux JLH, Nikkels PGJ, Lequin MH, Groenendaal F. The value of autopsy in neonates in the 21st century. Neonatology. 2019;115(1):89–93. doi: 10.1159/000493003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Elder DE. Autopsy after death due to extreme prematurity. Arch. Child. Fetal Neonatal Ed. 2005;90(3):F270–f272. doi: 10.1136/adc.2004.062380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mjörnheim B, Rosendahl A, Eriksson L. Attitudes of nurses and physicians about clinical autopsy in neonatal and adult hospital care: a survey in Sweden. Nurs. Res. 2015;2015(64):264–271. doi: 10.1097/NNR.0000000000000105. [DOI] [PubMed] [Google Scholar]
- 24.Killeen OG, Burke C, Devaney D, Clarke TA. The value of the perinatal and neonatal autopsy. Ir. Med. J. 2004;97(8):241–244. [PubMed] [Google Scholar]
- 25.Seske LM, Muglia LJ, Hall ES, Bove KE, Greenberg JM. Infant mortality, cause of death, and vital records reporting in Ohio, United States. Matern. Child Health J. 2017;21(4):727–733. doi: 10.1007/s10995-016-2159-x. [DOI] [PubMed] [Google Scholar]
- 26.SNQ Report Group. Årsrapport 2018, Yearly Report 2018 [Internet]. 2019 Aug https://www.medscinet.com/PNQ/uploads/website/arsrapporter/Neonatalregistrets%20%C3%85rsrapport%202018._3.0.pdf. Accessed 11 Oct 2019.
- 27.Klingenberg C, Wheeler KI, McCallion N, Morley CJ, Davis PG. Volume-targeted versus pressure-limited ventilation in neonates. Cochrane Database Syst Rev. 2017 doi: 10.1002/14651858.CD003666.pub4/full. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Roberts D, Brown J, Medley N, Dalziel SR. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst. Rev. 2017;21(3):CD004454. doi: 10.1002/14651858.CD004454.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lehtonen L, Gimeno A, Parra-Llorca A, Vento M. Early neonatal death: a challenge worldwide. Semin. Fetal Neonatal Med. 2017;22(3):153–160. doi: 10.1016/j.siny.2017.02.006. [DOI] [PubMed] [Google Scholar]
- 30.Rasiah V, Yajamanyam PK, Ewer AK. Necrotizing enterocolitis: current perspectives. Res. Rep. Neonatol. 2014 doi: 10.2147/RRN.S36576. [DOI] [Google Scholar]
- 31.Samuels N, van de Graaf RA, de Jonge RCJ, Reiss IKM, Vermeulen MJ. Risk factors for necrotizing enterocolitis in neonates: a systematic review of prognostic studies. BMC Pediatr. 2017;17(1):105. doi: 10.1186/s12887-017-0847-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Travers CP, Clark RH, Spitzer AR, Das A, Garite TJ, Carlo WA. Exposure to any antenatal corticosteroids and outcomes in preterm infants by gestational age: prospective cohort study. BMJ. 2017;356:3j1039. doi: 10.1136/bmj.j1039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Norman M, Hallberg B, Abrahamsson T, Björklund LJ, Domellöf M, Farooqi A, et al. Association between year of birth and 1-year survival among extremely preterm infants in Sweden during 2004–2007 and 2014–2016. JAMA. 2019;321(12):1188–1199. doi: 10.1001/jama.2019.2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bagci S, Eis-Hübinger AM, Franz AR, Bierbaum G, Heep A, Schildgen O, et al. Detection of astrovirus in premature infants with necrotizing enterocolitis. Pediatr. Infect. J. 2008;27(4):347–350. doi: 10.1097/INF.0b013e318162a17a. [DOI] [PubMed] [Google Scholar]
- 35.Meinzen-Derr J, Morrow AL, Hornung RW, Donovan EF, Dietrich KN, Succop PA. Epidemiology of necrotizing enterocolitis temporal clustering in two neonatology practices. J. Pediatr. 2009;154(5):656–661. doi: 10.1016/j.jpeds.2008.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kiechl-Kohlendorfer U, Simma B, Urlesberger B, Maurer-Fellbaum U, Wald M, Wald M, et al. Low mortality and short-term morbidity in very preterm infants in Austria 2011–2016. Acta Paediatr. 2019;108(8):1419–1426. doi: 10.1111/apa.14767. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analyzed during this study are included in this published article (and its Supplementary Information files).