Abstract
Setting:
Laparoscopic sleeve gastrectomy (LSG) is gaining popularity as a bariatric option. Gastric leak (GL) is the most dreaded septic complication of LSG. Early detection and treatment of this complication may improve outcomes.
Objectives:
This study investigates biomarkers that might be useful to predict GL before its clinical presentation in patients who underwent LSG.
Patients and Methods:
This study, prospective observational, was carried out in 151 patients, who underwent LSG for morbid obesity between February 2014 and October 2019. Blood samples were collected before the operation and on post-operative days one, three and five to dose serum C-reactive protein (CRP), pro-calcitonin (PCT), fibrinogen, white blood cells (WBCs) count and neutrophil-to-lymphocyte ratio (NLR).
Results:
GL occurred in 6 patients (3.97%). According to the receiver operating characteristics curve, NLR detected leak with remarkably higher sensitivity (100%) and specificity (100%) than CRP, fibrinogen, WBC on all the days and higher than PCT in post-operative days 3 and 5. Moreover, the area under the curve (AUC) of NLR (AUC = 1) was higher than the AUC of CRP, fibrinogen, WBC on all the days and higher than PCT in post-operative days 3 and 5, suggesting important statistical significance.
Conclusions:
Because NLR and PCT detected GL with remarkably higher sensitivity and specificity than CRP, fibrinogen and WBC, these two markers seem to be more accurate for the early detection of this complication.
Keywords: C-reactive protein, gastric leak, laparoscopic sleeve gastrectomy, neutrophil-to-lymphocyte ratio, procalcitonin
INTRODUCTION
Laparoscopic sleeve gastrectomy (LSG) gained a place among the surgical bariatric procedures for the treatment of severe obesity. It was first described by Gagner[1] in superobese patients as an effective way to reach an adequate weight to undergo a more complex procedure. In recent times, LSG taken on importance as a separate procedure to lose weight.[2,3,4] Its major advantages are relative technical simplicity, lack of anastomosis, better reproducibility, shorter operative time, no risk of internal hernia, absence of dumping syndrome, absence of malabsorption and preserved access to the upper gastrointestinal tract.[5,6,7,8] However, post-operative complications can be severe and sometimes fatal. The most frequent complications are gastric leak (GL) with a rate between 0%[9] and 7%[10] that can lead to abdominal sepsis or chronic gastric fistula,[11,12,13,14,15] haemorrhage (1.0%–1.2%) and strictures (0.6%–1.0%).[6,7,8,16,17,18,19,20] Some of these complications (GL and abscess) are potentially severe and could also appear after patient's discharge. Early detection and treatment of post-operative complications may improve outcomes. In the last decade, several studies have demonstrated that some serum inflammatory markers, especially C-reactive protein (CRP) and pro-calcitonin (PCT), can effectively detect and/or predict post-operative infectious complications (PICs) after abdominal surgery,[21,22] mainly after colorectal surgery.[23,24,25,26] However, few studies investigated the relationships between acute-phase parameters and the appearance of a GL after bariatric surgery.[27,28,29]
The aim of this study, prospective observational, is to analyse the evolution of serum levels of acute-phase proteins (CRP, PCT and fibrinogen), white blood cells (WBCs) count and neutrophil-to-lymphocyte ratio (NLR) in the first post-operative days after LSG, to evaluate if they could be useful to predict GL before its clinical presentation.
PATIENTS AND METHODS
We performed a prospective, single-centre, observational study in a group of 151 consecutive obese patients, who underwent LSG in our centre between February 2014 and October 2019.
In this study, we included patients who met the following criteria: body mass index (BMI) >40 kg/m2 or 35 kg/m2 with at least one obesity-related comorbidity.
Contraindications to LSG included severe gastro-oesophageal reflux disease and Barrett's oesophagus.[30] Before surgery, each patient was evaluated and followed for at least 6 months by a multidisciplinary team, including the psychologist, the endocrinologist, the gastroenterologist and the nutritionist. Patients were fully informed about the entire procedure by surgeons and anaesthetists. Informed consent was obtained from all individual participants included in the study.
Operative technique
Our operative technique has been previously described.[5]
All patients were double checked with a methylene blue test and upper gastrointestinal transit, (X-ray, computed tomography scan or by endoscopy), on the 2nd post-operative day and if no leakage was detected, a liquid diet was started. The patients were discharged on the 5th post-operative day after eating mashed food.
The database, used to collect information, included the following details: gender, age at the time of surgery, comorbidities, duration of the surgical procedure, eventual conversion from laparoscopic to open procedure, intra and early post-operative complications and duration of hospitalisation. Complications' severity was graded according to the Dindo-Clavien classification.[31,32]
An intention-to-treat analysis was performed. Patients who required conversion to open procedure were included in the analysis. Blood samples were collected the day before the operation and on post-operative days one, three and five to dese serum CRP, pro-calcitonin, fibrinogen and WBCs.
Stapling line leak was defined as a leak of contrast material shown by upper gastrointestinal transit (radiography and/or computed tomography scan).
Comparison between GL and non-GL groups after LSG was performed.
Statistical analysis
The characteristics of the study sample were analysed using descriptive statistics. The discrete and nominal variables were expressed using frequencies and percentages; continuous variables were expressed as means and standard deviations. The Mann–Whitney U-test was performed for testing differences between groups, according to the presence or absence of GL.
Receiver operating characteristic (ROC) curve analysis was performed, and the respective areas under the curve (AUC) with standard errors and 95% confidence intervals (CIs) were calculated to evaluate the predictive value of procalcitonin (PCT) level, CRP level, fibrinogen level, WBC count and NLR for the diagnosis of GL as post-operative complication. If the CI did not include the 0.5 value, the parameter had an ability to distinguish between the two groups (presence or absence of GL). The sensitivity and specificity of these parameters were calculated. Positive predictive value (PPV) and negative predictive value (NPV) were calculated for the specific sensitivity and specificity, considering the disease prevalence. The maximal Youden index (sensitivity + specificity − 1) was calculated to establish cutoff values. A parameter for which the AUC was equal to 0.05 was no predictive of GL; on the contrary, a parameter for which the AUC was closet to 1 was the most predictive of GL.
Statistical analyses were carried out using the Stata Statistical Software: Release 15 (Stata Corp LP, College Station, TX, USA). All the tests were two tailed, and P < 0.05 was considered statistically significant.
RESULTS
Among the 151 patients included in the study, 96 (63.58%) were females and 55 (36.42%) were males, with a mean age of 45 ± 16 years.
In six (3.97%) patients, GL was observed in post-operative follow-up, while none was detected during the operation. GL rate was also strongly linked to the learning curve. In fact, we diagnosed four leaks in the first 50 cases (8%) and two among the cases 51–151 (1.9%). The leakage was diagnosed on post-operative days five (in 1 patient), six (in 2 patients), eight (in 2 patients) and ten (in 1 patient). Therefore, 83.3% of the GLs occurred between post-operative days six and ten. Three patients (50%) had been discharged before diagnosis of leak.
The six GLs were treated by endoscopic placement of a double pigtail drainage. In two cases (33.3%), GL was complicated by intra-abdominal abscess. In one case, it was radiologically drained, while the other one required laparoscopic surgical toilette and drainage. In terms of age, gender, BMI, comorbidities, intraoperative blood loss and operative time, both groups (GL and no GL) were similar (P = ns), whereas duration of hospitalisation was significantly longer in the GL group (P < 0.001). The conversion from laparoscopic to open procedure was observed but in the absence of GL. These parameters are given in Table 1.
Table 1.
Baseline clinical characteristics of the patients (n=151)
| Gastric leak (n=6) | No gastric leak (n=145) | P | |
|---|---|---|---|
| Age (years)* | 46±12 | 44±17 | 0.188 |
| Gender, n (%) | |||
| Female | 5 (83.33) | 91 (62.76) | 0.870 |
| Male | 1 (16.67) | 54 (37.24) | |
| BMI* | 42.48±4.78 | 40.40±0.57 | 0.538 |
| Comorbidities, n (%) | |||
| Type 2 DM | 2 (33.33) | 36 (32.43) | 0.637 |
| Hypertyension | 2 (33.33) | 38 (34.23) | 0.666 |
| Dislypidemia | 3 (50.00) | 57 (51.35) | 0.636 |
| Obstructive sleep apnea | 3 (50.00) | 58 (52.25) | 0.619 |
| Intraoperative blood loss (mL)* | 34.11±3.24 | 33.61±7.16 | 0.768 |
| Conversion, n (%) | / | 3 (1.99) | / |
| Operative time (min)* | 60.22±14.12 | 58.31±12.57 | 0.478 |
| Hospital stay (days)* | 19.61±10.80 | 4.52±1.25 | <0.001 |
*Mean±SD. SD: Standard deviation, BMI: body mass index, DM: Diabetes mellitus
Thirty-day mortality rate was 0.7% (1 patient in the GL group), due to a massive pulmonary embolism because of severe sepsis.
Pre-operative and post-operative day 1 CRP values were similar in both groups (P = ns). On post-operative days 3 and 5, CRP was significantly higher in patients with GL (P = 0.020 and P < 0.001, respectively).
PCT levels were significantly higher in post-operative day 1 (P < 0.001).
Serum levels of fibrinogen were increased only in post-operative day 5 in patients with GL (P < 0.001) and were decreased in patients without leakage. Serum CRP, PCT and fibrinogen levels are reported in Table 2.
Table 2.
Mean values of serum c-reactive protein, pro-calcitonin and fibrinogen between the two groups (absence or presence of gastric leak)
| Time | Total (n=151) | Gastric leak | P* | |
|---|---|---|---|---|
| Absent (n=145) | Present (n=6) | |||
| CRP | ||||
| Preoperative, mean±SD | 1.14±0.67 | 1.14±0.69 | 1.17±0.31 | 0.443 |
| 1 day after, mean±SD | 4.14±1.43 | 4.11±1.39 | 4.72±2.10 | 0.679 |
| 3 days after, mean±SD | 5.37±2.22 | 5.24±2.08 | 7.85±3.39 | 0.020 |
| 5 days after, mean±SD | 4.54±3.55 | 3.95±1.57 | 15.28±9.61 | <0.001 |
| PCT | ||||
| Preoperative, mean±SD | 0.04±0.02 | 0.04±0.02 | 0.03±0.01 | 0.335 |
| 1 day after, mean±SD | 0.20±0.64 | 0.07±0.03 | 2.61±1.44 | <0.001 |
| 3 days after, mean±SD | 0.44±1.79 | 0.09±0.17 | 6.92±4.53 | <0.001 |
| 5 days after, mean±SD | 0.49±2.23 | 0.07±0.13 | 8.36±6.03 | <0.001 |
| Fibrinogen | ||||
| Preoperative, mean±SD | 344.27±54.10 | 343.37±55.06 | 361.00±29.39 | 0.380 |
| 1 day after, mean±SD | 399.57±54.32 | 399.48±55.12 | 401.33±40.09 | 0.757 |
| 3 days after, mean±SD | 416.30±90.86 | 413.11±90.81 | 475.17±75.55 | 0.104 |
| 5 days after, mean±SD | 388.66±97.56 | 374.87±72.11 | 643.67±155.51 | <0.001 |
*Wilcoxon rank-sum (Mann–Whitney) test. CRP: C-Reactive protein; PCT: Pro-calcitonin, SD: Standard deviation
Pre-operative values of WBC and NLR were similar in GL and no-GL group. In POD 1, WBC count increased in both groups, being slightly higher in the GL group, although not reaching statistical significance. WBC count was significantly higher in the third and fifth post-operative days in the GL group. NLR was significantly higher in POD 1, 3 and 5 in patients with GL (P < 0.001). Serum WBC and NLR levels are reported in Table 3.
Table 3.
Mean values of white blood cells count and neutrophil-to-lymphocyte ratio between the two groups (absence or presence of gastric leak)
| Time | Total (n=151) | Gastric leak | P* | |
|---|---|---|---|---|
| Absent (n=145) | Present (n=6) | |||
| WBC | ||||
| Preoperative, mean±SD | 8.37±1.39 | 8.36±1.39 | 8.51±1.51 | 0.795 |
| 1 day after, mean±SD | 10.41±1.39 | 10.36±1.30 | 11.44±2.48 | 0.161 |
| 3 days after, mean±SD | 9.53±1.41 | 9.39±1.11 | 12.10±3.22 | 0.014 |
| 5 days after, mean±SD | 8.81±1.33 | 8.66±0.94 | 11.40±3.63 | 0.031 |
| NLR | ||||
| Preoperative, mean±SD | 1.83±0.20 | 1.84±0.20 | 1.74±0.09 | 0.276 |
| 1 day after, mean±SD | 2.06±0.45 | 1.98±0.29 | 3.44±0.64 | <0.001 |
| 3 days after, mean±SD | 2.03±0.56 | 1.91±0.20 | 4.14±0.90 | <0.001 |
| 5 days after, mean±SD | 2.01±0.85 | 1.83±0.15 | 5.37±1.47 | <0.001 |
*Wilcoxon rank-sum (Mann–Whitney) test. WBC count: White blood cells count; NLR: Neutrophil-to-lymphocyte ratio, SD: Standard deviation
Using ROC analysis, the mean ± standard error of the area under the curve (AUC) (AUC ± SE) for CRP levels on post-operative day 1 was 0.550 ± 0.135, 95% CI = 0.285–0.815, and a cutoff level at 7.24 for predicting GL achieved 33.33% sensitivity and 98.20% specificity with PPV of 50.03% and NPV of 96.46% [Table 4 and Figure 1a].
Table 4.
Results of receiver operating characteristic analysis showing the predictive value of c-reactive protein, procalcitonin, fibrinogen, white blood cell and neutrophil-to-lymphocyte ratio for absence/presence of gastric leak after laparoscopic sleeve gastrectomy
| AUC | 95% CI | SE | Cutoff value | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | |
|---|---|---|---|---|---|---|---|---|
| CRP | ||||||||
| Preoperative | 0.631 | 0.468-0.795 | 0.083 | 0.7 | 100 | 41.44 | 8.45 | 100 |
| 1 day after | 0.550 | 0.285-0.815 | 0.135 | 7.24 | 33.33 | 98.20 | 50.03 | 96.46 |
| 3 days after | 0.984 | 0.951-1 | 0.016 | 7.33 | 100 | 90.09 | 35.30 | 100 |
| 5 days after | 0.966 | 0.901-1 | 0.033 | 5.59 | 83.33 | 100 | 100 | 99.11 |
| PCT | ||||||||
| Preoperative | 0.615 | 0.453-0.777 | 0.083 | 0.04 | 100 | 34.23 | 7.60 | 100 |
| 1 day after | 1 | 1-1 | 0 | 0.55 | 100 | 100 | 100 | 100 |
| 3 days after | 0.996 | 0.985-1 | 0.005 | 0.14 | 83.33 | 99.10 | 83.35 | 99.10 |
| 5 days after | 0.967 | 0.905-1 | 0.031 | 1.64 | 83.33 | 100 | 100 | 99.11 |
| Fibrinogen | ||||||||
| Preoperative | 0.664 | 0.503-0.824 | 0.082 | 328 | 100 | 38.74 | 8.11 | 100 |
| 1 day after | 0.553 | 0.291-0.814 | 0.133 | 402 | 33.33 | 99.10 | 66.69 | 96.49 |
| 3 days after | 0.985 | 0.956-1 | 0.015 | 388 | 100 | 90.99 | 37.51 | 100 |
| 5 days after | 0.957 | 0.876-1 | 0.041 | 529 | 83.33 | 100 | 100 | 99.11 |
| WBC | ||||||||
| Preoperative | 0.665 | 0.486-0.844 | 0.091 | 6.5 | 100 | 34.23 | 7.60 | 100 |
| 1 day after | 0.652 | 0.325-0.978 | 0.166 | 11.84 | 66.67 | 81.98 | 16.67 | 97.85 |
| 3 days after | 0.984 | 0.951-1 | 0.016 | 10.18 | 100 | 89.19 | 33.34 | 100 |
| 5 days after | 0.949 | 0.856-1 | 0.048 | 6.57 | 83.33 | 100 | 100 | 99.11 |
| NLR | ||||||||
| Preoperative | 0.724 | 0.567-0.881 | 0.080 | 1.74 | 83.33 | 73.87 | 14.71 | 98.79 |
| 1 day after | 1 | 1-1 | 0 | 2.74 | 100 | 100 | 100 | 100 |
| 3 days after | 1 | 1-1 | 0 | 3.22 | 100 | 100 | 100 | 100 |
| 5 days after | 1 | 1-1 | 0 | 3.64 | 100 | 100 | 100 | 100 |
AUC: Area under the curve, CI: Confidence interval, SE: Standard error. PPV: Positive predictive value. NPV: Negative predictive value, WBC: White blood cells; NLR: Neutrophil-to-lymphocyte ratio, PCT: Procalcitonin, CRP: C-reactive protein
Figure 1.
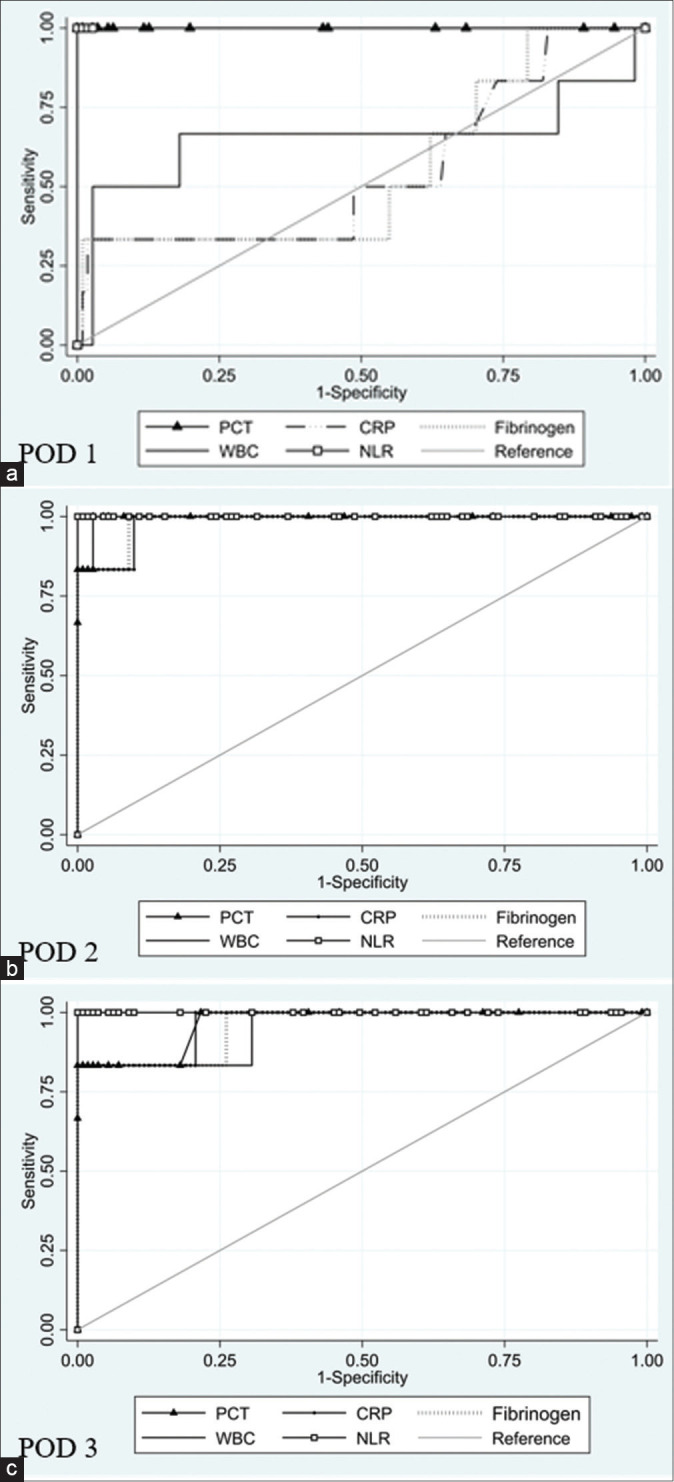
ROC curve on POD 1 (a), 3 (b) and 5 (c). ROC: Receiver operative carachteristics. PCT: Procalcitonin. CRP: C reactive protein. WBC: White blood cells. NLR: Neuthrophil-to-lymphocyte ratio. POD: Post-operative day
The AUC ± SE of PCT levels on post-operative day 1 was 1 ± 0, 95% CI = 1–1, and a PCT cutoff level at 0.55 with 100% sensitivity and 100% specificity with PPV of 100% and NPV 100% [Table 4 and Figure 1a].
The AUC ± SE of fibrinogen levels on post-operative day 1 was 0.553 ± 0.133, 95% CI = 0.291–0.814, and a fibrinogen cutoff level at 402 with 33.33% sensitivity and 99.10% specificity with PPV of 66.69% and NPV of 96.49% [Table 4 and Figure 1a].
The AUC ± SE of WBC count on post-operative day 1 was 0.652 ± 0.166, 95% CI = 0.325–0.978, and a cutoff level at 11.84 cell/mm3 for predicting GL achieved 66.67% sensitivity and 81.98% specificity with PPV of 16.67% and NPV of 97.85% [Table 4 and Figure 1a], whereas the AUC ± SE of the NLR was 1 ± 0, 95% CI = 1-1, and a cutoff level at 2.74 achieved 100% sensitivity and 100% specificity with PPV of 100% and NPV of 100% [Table 4 and Figure 1a].
The AUC ± SE of CRP levels on post-operative day 3 was 0.984 ± 0.016, 95% CI = 0.951-1, and a cutoff level at 7.33 achieved 100% sensitivity and 90.09% specificity with PPV of 35.30% and NPV of 100% [Table 4 and Figure 1b].
The AUC ± SE of PCT levels on post-operative day 3 was 0.996 ± 0.005, 95% CI = 0.985–1, and a cutoff level at 0.14 with 83.33% sensitivity and 99.10% specificity with PPV of 83.35% and NPV of 99.10% [Table 4 and Figure 1b].
The AUC ± SE of fibrinogen levels on post-operative day 3 was 0.985 ± 0.015, 95% CI = 0.956–1, and a cutoff level at 388 with 100% sensitivity and 90.99% specificity with PPV of 37.51% and NPV of 100% [Table 4 and Figure 1b].
The AUC ± SE of WBC count on post-operative day 3 was 0.984 ± 0.016, 95% CI = 0.951–1, and a cutoff level at 10.18 cell/mm3 for predicting GL achieved 100% sensitivity and 89.19% specificity with PPV of 33.34% and NPV of 100% [Table 4 and Figure 1b], whereas the AUC ± SE of the NLR was 1 ± 0, 95% CI = 1–1, and a cutoff level at 3.22 achieved 100% sensitivity and 100% specificity with PPV of 100% and NPV of 100% [Table 4 and Figure 1b].
The AUC ± SE of CRP levels on post-operative day 5 was 0.966 ± 0.033, 95% CI = 0.901–1, and a cutoff level at 5.59 achieved 83.33% sensitivity and 100% specificity with PPV of 100% and NPV of 99.11% [Table 4 and Figure 1c].
The AUC ± SE of PCT levels on post-operative day 5 was 0.967 ± 0.031, 95% CI = 0.905–1, and a cutoff level at 1.64 with 83.33% sensitivity and 100% specificity with PPV of 100% and NPV of 99.11% [Table 4 and Figure 1c].
The AUC ± SE of fibrinogen levels on post-operative day 5 was 0.957 ± 0.041, 95% CI = 0.876–1, and a cutoff level at 529 with 83.33% sensitivity and 100% specificity with PPV of 100% and NPV of 99.11% [Table 4 and Figure 1c].
The AUC ± SE of WBC count on post-operative day 5 was 0.949 ± 0.048, 95% CI = 0.856–1, and a cutoff level at 6.57 cell/mm3 with 83.33% sensitivity and 100% specificity with PPV of 100% and NPV of 99.11% [Table 4 and Figure 1c], whereas the AUC ± SE of the NLR was 1 ± 0, 95% CI = 1–1, and a cutoff level at 3.64 for predicting GL achieved 100% sensitivity and 100% specificity with PPV of 100% and NPV of 100% [Table 4 and Figure 1c].
Three patients (1.99%) required conversion from laparoscopic to open procedure, two out of the first 50 cases (4%) of the series, and one among cases 51–151 (0.99%). The following conditions required conversion to laparotomy: Technical difficulties with fundus dissection due to peri-oesophagitis; prepiloric lesion during greater curvature dissection; haemorrhage for lesion of short gastric vessels. In none of these cases GL was observed.
DISCUSSION
After LSG, the leakage of the suture line lead to increased mortality, morbidity and hospital stay.[33,34] Its rate after LSG is 0%–7%.[9,10] In our study, this rate was 3.97% (6/151).
The diagnosis of GL is still based on clinical signs (pain, fever and tachycardia), laboratory findings (leucocytosis), methylene blue test (appearance of methylene blue dye through drain), and upper gastrointestinal transit (radiography and CT scan). This conventional attitude may cause important delays in diagnosis,[27,35] even because the initial clinical manifestations are very subtle.[27,36] Aurora et al.,[19] in their systematic analysis of 4,888 patients, affirmed that 50% of GLs occur after post-operative day 10. In our study, 83.3% of the GLs occurred between post-operative days six and ten. Therefore, an effective indicator could aid in the early diagnosis of the leakage before that clinical signs appear. Several biochemical tests are used to identify inflammatory activity in post-operative patients, including dosage of PCT, CRP and fibrinogen.
Recent studies in surgical patients have also shown that after orthopaedic, cardiac and thoracic surgery, PCT was better for detecting post-operative infections than CRP.[37,38,39]
After LSG, Kassir et al. showed that a mean post-operative PCT was 0.062 ng/mL in uncomplicated patients versus 0.108 ng/mL in those with complications (P = 0.0006) and concluded that PCT measured during the post-operative period were correlated with the occurrence of post-operative complications.[29] Muñoz et al. concluded that PCT levels in the first day and especially in the second day post-operative can predict septic complications after LSG.[28] In our study, serum PCT values were increased in the GL group on post-operative day 1, 3 and 5. The AUC of PCT levels on post-operative day 1 was 1, with 100% sensitivity and 100% specificity.
Several studies have found that CRP is a useful predictor of septic complications following colorectal and oesophagogastric resection.[40,41,42,43,44] After LSG, early rise of serum CRP before clinical and radiological presentation of leakage was shown in the literature, which might lead to early diagnosis and reduction of morbidity and mortality.[27,29] Increased serum CRP levels after operation decrease versus normal levels at the third post-operative day. A CRP level that remains elevated (>125 mg/L) at the post-operative day 3 should be an indication for CT scan to look for sepsis.[45,46] After LSG, Albanopoulos et al. demonstrated that on day 3, the sensitivity and specificity of CRP reached 100% and on day 5, CRP achieved 83.2% sensitivity and 100% specificity.[27] Kassir et al.[29] reported that mean CRP was 61.3 mg/L in patient without complications and 161.3 mg/L in case of complications (P = 0.0233), and affirmed that it do not provide any predictive benefit, because the average was elevated in few spuriously high numbers, 'but when the test was applied on an individual basis it is no longer predictive of a complication'.
In our study, on post-operative day 1 the CRP level achieved 33.33% sensitivity, and 98.20% specificity, whereas a PCT level achieved 100% sensitivity and 100% specificity. Moreover, the AUC of the PCT level was 1, remarkably higher than the AUC of the CRP level (0.5503). On post-operative day 3 and 5, the AUC, sensitivity and specificity of PCT and CRP were similar.
Fibrinogen is a 350-KDa glycoprotein, synthesised mainly by the liver.[47] After converted to insoluble fibrin by activated thrombin, fibrinogen plays an important role in regulating blood clotting. Moreover, fibrinogen is a pro-inflammatory protein produced in response to elevated serum cytokines (IL-6 and IL-1β) and CRP levels[48,49,50] and usually acts as an acute-phase protein, and its level increases in response to wound healing, infection and inflammation.[51]
In the current study, we observed a slight increase of fibrinogen levels in most cases, and patients with GL showed significantly higher levels only at fifth post-operative day and on post-operative day 1, 3 and 5 sensitivity, specificity and AUC were remarkably lower than the sensitivity, specificity and AUC of PCT and CRP.
Traditionally, the WBCs count has been used as a monitor for post-operative infectious complications; however, according to some recent studies, the value of WBC count in this scenario might be overestimated.[24,27,30,41,52] In our study, WBC count increased in both groups on post-operative day 1, being slightly higher in the GL group, but not reaching statistical significance. In the first post-operative day, the AUC was only 0.652, with sensitivity of 66.67% and specificity of 81.98%. Therefore, measurements of WBC count contribute a little to the early detection of GL complication.
Muñoz et al.[28] in a prospective study evaluated the efficacy of various acute-phase parameters determined 24 and 48 h after LSG for predicting septic complications in the post-operative course within an Enhanced Recovery After Surgery Program. They found that the areas under the curve (AUC) of CRP and PCT levels was 1.00, remarkably higher than the AUC of the WBC (0.858) or fibrinogen level (0.779). These results are in line with ours.
A novel inflammatory marker derived from the WBC count, the NLR, is becoming increasingly used with promising results in this context.[52,53,54,55,56,57,58] NLR is a ratio of absolute count of neutrophils to the absolute count of lymphocytes. Neutrophil play an important role in the acute inflammatory response to tissue injury.[59] Lymphocytopenia after a major surgery is related to cortisol production and neuroendocrine stress.[60] Thus, NLR, as a ratio of neutrophil to lymphocyte count, represents the two inversely related immune pathways: One representing unrestrained inflammation and the other latent immune pathway.
Medina Fernández et al.[52] affirm that this parameter shows a parallel evaluation to CRP in patients with post-operative infectious complications (PICs), and values of NLR persistently >7–8 should be considered suspicious for PICs, since post-operative values in uncomplicated patients remain around 5.
In our study at the first, third and fifth post-operative days, values of NLR were significantly increased in the GL group. On post-operative day 1, 3 and 5 sensitivity (100%), specificity (100%) and AUC (1) were higher than the sensitivity, specificity and AUC of CRP, WBC and fibrinogen. The AUC, sensitivity and specificity were also higher than AUC, sensitivity and specificity of PCT in third and fifth post-operative days, while in the first post-operative day the values were comparable. Therefore, the high sensitivity (100%) and specificity (100%) and AUC (1) of the NLR and PCT recorded in the first post-operative day may facilitate the early detection of GL complication. Finally, it must be considered that CRP, fibrinogen and WBC had in P.O. days 1 and 3 a high NPV but a low PPV Table 4, while PCT and NLR had in P.O. days 1 and 3 a high value of both NPV and PPV. The value for NLR was even higher (100%) [Table 4].
However, our study, being a preliminary study, has, obviously, some limitations: The small sample size and the collection of data in a single centre only are the most important limitations which restricts the statistical power of our analysis and since it may influence the evaluation of the calibration of the biomarker of infection. Moreover, a higher lead time bias between the onset of GL and the calculation of levels of serum biomarker may also contribute to the performance of predictive benefit. Therefore, although these findings are interesting, they need verification in a larger sample. More studies are needed to evaluate NLR as an accurate and early predictor of GL. Moreover, the next studies need to be designed in a more detailed and robust way.
However, as for as we are aware, this is the first prospective study that evaluates the NLR, together with PCT, CRP, fibrinogen and WBC, in patients who underwent LSG.
CONCLUSIONS
Because of the severity of the complications (intra-abdominal abscess, generalised peritonitis) associated with a GL after LSG, it is imperative to identify the problem and act as early as possible. Clinical signs (pain, fever, tachycardia), laboratory findings (leucocytosis), methylene blue test and upper gastrointestinal transit (radiography, CT scan), may cause important delays in diagnosis. Elevated levels of serum PCT, CRP, fibrinogen, WBC and NLR at post-operative days, could be a help for the early detection of GL after LSG. Because NLR and PCT detected GL with remarkably higher sensitivity and specificity than CRP, fibrinogen and WBC, these two markers seem to be more accurate for the early detection of this complication.
In the light of this study, the protocol for our patients is as follows:
Patients without clinical symptoms (no pain, no fever, no tachycardia), with normal WBC count (4.8–10.8 migl/mmc), normal CRP (<0.5 mg/L), normal fibrinogen (200–400 mg/dL), normal PCT (<0.5 mg/dL) and normal NLR (2.11–1.55) are discharged on post-operative day 3 without undergo upper gastrointestinal transit test
Patients with high values of NLR and/or PCT on post-operative day 1 and/or 3, undergo upper gastrointestinal transit test, also without clinical signs. If a GL is reported, we manage it with endoscopic placement of a double pigtail drainage. If no leak is reported, if these inflammatory markers are still high, another radiographic test is performed in post-operative day 5 or 6. If this control is still negative, the patient is discharged and followed with outpatient checks. Otherwise, if markers return to normal, the patient is discharged without controls.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
Acknowledgements
All authors disclose the absence of any financial and personal relationships with other people or organisations that could inappropriately influence (bias) their work.
REFERENCES
- 1.Ren CJ, Patterson E, Gagner M. Early results of laparoscopic biliopancreatic diversion with duodenal switch: A case series of 40 consecutive patients. Obes Surg. 2000;10:514–23. doi: 10.1381/096089200321593715. [DOI] [PubMed] [Google Scholar]
- 2.Baltasar A, Serra C, Pérez N, Bou R, Bengochea M, Ferri L. Laparoscopic sleeve gastrectomy: A multi-purpose bariatric operation. Obes Surg. 2005;15:1124–8. doi: 10.1381/0960892055002248. [DOI] [PubMed] [Google Scholar]
- 3.Cottam D, Qureshi FG, Mattar SG, Sharma S, Holover S, Bonanomi G, et al. Laparoscopic sleeve gastrectomy as an initial weight-loss procedure for high-risk patients with morbid obesity. Surg Endosc. 2006;20:859–63. doi: 10.1007/s00464-005-0134-5. [DOI] [PubMed] [Google Scholar]
- 4.Carubbi F, Ruscitti P, Pantano I, Alvaro S, Benedetto PD, Liakouli V, et al. Jejunoileal bypass as the main procedure in the onset of immune-related conditions: The model of BADAS. Expert Rev Clin Immunol. 2013;9:441–52. doi: 10.1586/eci.13.26. [DOI] [PubMed] [Google Scholar]
- 5.Giuliani A, Romano L, Papale E, Puccica I, Di Furia M, Salvatorelli A, et al. Complications of postlaparoscopic sleeve gastric resection: review of surgical technique. Minerva Chir. 2019;74:213–7. doi: 10.23736/S0026-4733.19.07883-0. [DOI] [PubMed] [Google Scholar]
- 6.Deitel M, Gagner M, Erickson AL, Crosby RD. Third international summit: Current status of sleeve gastrectomy. Surg Obes Relat Dis. 2011;7:749–59. doi: 10.1016/j.soard.2011.07.017. [DOI] [PubMed] [Google Scholar]
- 7.Sarela AI, Dexter SP, O'Kane M, Menon A, McMahon MJ. Long-term follow-up after laparoscopic sleeve gastrectomy: 8-9-year results. Surg Obes Relat Dis. 2012;8:679–84. doi: 10.1016/j.soard.2011.06.020. [DOI] [PubMed] [Google Scholar]
- 8.Kehagias I, Spyropoulos C, Karamanakos S, Kalfarentzos F. Efficacy of sleeve gastrectomy as sole procedure in patients with clinically severe obesity (BMI≤50 kg/m (2)) Surg Obes Relat Dis. 2013;9:363–9. doi: 10.1016/j.soard.2011.12.011. [DOI] [PubMed] [Google Scholar]
- 9.Bellanger DE, Greenway FL. Laparoscopic sleeve gastrectomy, 529 cases without a leak: Short-term results and technical considerations. Obes Surg. 2011;21:146–50. doi: 10.1007/s11695-010-0320-y. [DOI] [PubMed] [Google Scholar]
- 10.Stroh C, Birk D, Flade-Kuthe R, Frenken M, Herbig B, Höhne S, et al. A nationwide survey on bariatric surgery in Germany-results 2005-2007. Obes Surg. 2009;19:105–12. doi: 10.1007/s11695-008-9736-z. [DOI] [PubMed] [Google Scholar]
- 11.Burgos AM, Braghetto I, Csendes A, Maluenda F, Korn O, Yarmuch J, et al. Gastric leak after laparoscopic-sleeve gastrectomy for obesity. Obes Surg. 2009;19:1672–7. doi: 10.1007/s11695-009-9884-9. [DOI] [PubMed] [Google Scholar]
- 12.Casella G, Soricelli E, Rizzello M, Trentino P, Fiocca F, Fantini A, et al. Nonsurgical treatment of staple line leaks after laparoscopic sleeve gastrectomy. Obes Surg. 2009;19:821–6. doi: 10.1007/s11695-009-9840-8. [DOI] [PubMed] [Google Scholar]
- 13.Tan JT, Kariyawasam S, Wijeratne T, Chandraratna HS. Diagnosis and management of gastric leaks after laparoscopic sleeve gastrectomy for morbid obesity. Obes Surg. 2010;20:403–9. doi: 10.1007/s11695-009-0020-7. [DOI] [PubMed] [Google Scholar]
- 14.Giuliani A, Romano L, Marchese M, Necozione S, Cianca G, Schietroma M, et al. Gastric leak after laparoscopic sleeve gastrectomy: Management with endoscopic double pigtail drainage. A systematic review. Surg Obes Relat Dis. 2019;15:1414–9. doi: 10.1016/j.soard.2019.03.019. [DOI] [PubMed] [Google Scholar]
- 15.Romano L, Giuliani A, Cianca G, Di Sibio A, Carlei F, Amicucci G, et al. A case of intrasplenic displacement of an endoscopic double-pigtail stent as a treatment for laparoscopic sleeve gastrectomy leak [published correction appears in Int J Surg Case Rep 2019;56:49]? Int J Surg Case Rep. 2018;53:367–9. doi: 10.1016/j.ijscr.2018.11.008. doi:10.1016/j.ijscr.2018.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jen S, Simillis C, Efthimiou E. A very challenging leak from a sleeve gastrectomy. Surg Obes Relat Dis. 2013;9:e56–9. doi: 10.1016/j.soard.2012.06.002. [DOI] [PubMed] [Google Scholar]
- 17.Nedelcu M, Skalli M, Delhom E, Fabre JM, Nocca D. New CT scan classification of leak after sleeve gastrectomy. Obes Surg. 2013;23:1341–3. doi: 10.1007/s11695-013-1002-3. [DOI] [PubMed] [Google Scholar]
- 18.Nedelcu AM, Skalli M, Deneve E, Fabre JM, Nocca D. Surgical management of chronic fistula after sleeve gastrectomy. Surg Obes Relat Dis. 2013;9:879–84. doi: 10.1016/j.soard.2013.02.010. [DOI] [PubMed] [Google Scholar]
- 19.Aurora AR, Khaitan L, Saber AA. Sleeve gastrectomy and the risk of leak: A systematic analysis of 4,888 patients. Surg Endosc. 2012;26:1509–15. doi: 10.1007/s00464-011-2085-3. [DOI] [PubMed] [Google Scholar]
- 20.Khoursheed M, Al-Bader I, Mouzannar A, Ashraf A, Bahzad Y, Al-Haddad A, et al. Postoperative Bleeding and Leakage After Sleeve Gastrectomy: a Single-Center Experience. Obes Surg. 2016;26:3007. doi: 10.1007/s11695-016-2317-7. [DOI] [PubMed] [Google Scholar]
- 21.Gans SL, Atema JJ, van Dieren S, Groot Koerkamp B, Boermeester MA. Diagnostic value of C-reactive protein to rule out infectious complications after major abdominal surgery: A systematic review and meta-analysis. Int J Colorectal Dis. 2015;30:861–73. doi: 10.1007/s00384-015-2205-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Adamina M, Steffen T, Tarantino I, Beutner U, Schmied BM, Warschkow R. Meta-analysis of the predictive value of C-reactive protein for infectious complications in abdominal surgery. Br J Surg. 2015;102:590–8. doi: 10.1002/bjs.9756. [DOI] [PubMed] [Google Scholar]
- 23.Singh PP, Zeng IS, Srinivasa S, Lemanu DP, Connolly AB, Hill AG. Systematic review and meta-analysis of use of serum C-reactive protein levels to predict anastomotic leak after colorectal surgery. Br J Surg. 2014;101:339–46. doi: 10.1002/bjs.9354. [DOI] [PubMed] [Google Scholar]
- 24.Silvestre J, Rebanda J, Lourenço C, Póvoa P. Diagnostic accuracy of C-reactive protein and procalcitonin in the early detection of infection after elective colorectal surgery-A pilot study. BMC Infect Dis. 2014;14:444. doi: 10.1186/1471-2334-14-444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lagoutte N, Facy O, Ravoire A, Chalumeau C, Jonval L, Rat P, et al. C-reactive protein and procalcitonin for the early detection of anastomotic leakage after elective colorectal surgery: Pilot study in 100 patients. J Visc Surg. 2012;149:e345–9. doi: 10.1016/j.jviscsurg.2012.09.003. [DOI] [PubMed] [Google Scholar]
- 26.Cousin F, Ortega-Deballon P, Bourredjem A, Doussot A, Giaccaglia V, Fournel I. Diagnostic accuracy of procalcitonin and c-reactive protein for the early diagnosis of intra-abdominal infection after elective colorectal surgery: A meta-analysis. Ann Surg. 2016;264:252–6. doi: 10.1097/SLA.0000000000001545. [DOI] [PubMed] [Google Scholar]
- 27.Albanopoulos K, Alevizos L, Natoudi M, Dardamanis D, Menenakos E, Stamou K, et al. C-reactive protein, white blood cells, and neutrophils as early predictors of postoperative complications in patients undergoing laparoscopic sleeve gastrectomy. Surg Endosc. 2013;27:864–71. doi: 10.1007/s00464-012-2526-7. [DOI] [PubMed] [Google Scholar]
- 28.Muñoz JL, Ruiz-Tovar J, Miranda E, Berrio DL, Moya P, Gutiérrez M, et al. C-Reactive protein and procalcitonin as early markers of septic complications after laparoscopic sleeve gastrectomy in morbidly obese patients within an enhanced recovery after surgery program. J Am Coll Surg. 2016;222:831–7. doi: 10.1016/j.jamcollsurg.2016.01.059. [DOI] [PubMed] [Google Scholar]
- 29.Kassir R, Blanc P, Bruna Tibalbo LM, Breton C, Lointier P. C-Reactive protein and procalcitonin for the early detection of postoperative complications after sleeve gastrectomy: Preliminary study in 97 patients. Surg Endosc. 2015;29:1439–44. doi: 10.1007/s00464-014-3821-2. [DOI] [PubMed] [Google Scholar]
- 30.Schietroma M, Pessia B, Colozzi S, Romano L, Vicentini V, Giuliani A, et al. HLA-DR (cellular and soluble) and inflammatory serum markers in patients after trauma: can they predict outcome?? Chirurgia. 2019;32:221–7. doi: 10.23736/S0394-9508.18.04890-8. [Google Scholar]
- 31.Dindo D, Demartines N, Clavien PA. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13. doi: 10.1097/01.sla.0000133083.54934.ae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250:187–96. doi: 10.1097/SLA.0b013e3181b13ca2. [DOI] [PubMed] [Google Scholar]
- 33.Sakran N, Goitein D, Raziel A, Keidar A, Beglaibter N, Grinbaum R, et al. Gastric leaks after sleeve gastrectomy: A multicenter experience with 2,834 patients. Surg Endosc. 2013;27:240–5. doi: 10.1007/s00464-012-2426-x. [DOI] [PubMed] [Google Scholar]
- 34.Rosenthal RJ, International Sleeve Gastrectomy Expert Panel. Diaz AA, Arvidsson D, Baker RS, Basso N, et al. International sleeve gastrectomy expert panel consensus statement: Best practice guidelines based on experience of 12,000 cases. Surg Obes Relat Dis. 2012;8:8–19. doi: 10.1016/j.soard.2011.10.019. [DOI] [PubMed] [Google Scholar]
- 35.Di Furia M, Romano L, Salvatorelli A, Brandolin D, Lomanto D, Cianca G, et al. Indocyanine green fluorescent angiography during laparoscopic sleeve gastrectomy: Preliminary results. Obes Surg. 2019;29:3786–90. doi: 10.1007/s11695-019-04085-y. [DOI] [PubMed] [Google Scholar]
- 36.Schietroma M, Pessia B, Mattei A, Romano L, Giuliani A, Carlei F. Temperature-neutrophils-multiple organ failure grading for complicated intra-abdominal infections. Surg Infect (Larchmt) 2019;21:69–74. doi: 10.1089/sur.2019.092. [DOI] [PubMed] [Google Scholar]
- 37.Hunziker S, Hügle T, Schuchardt K, Groeschl I, Schuetz P, Mueller B, et al. The value of serum procalcitonin level for differentiation of infectious from noninfectious causes of fever after orthopaedic surgery. J Bone Joint Surg Am. 2010;92:138–48. doi: 10.2106/JBJS.H.01600. [DOI] [PubMed] [Google Scholar]
- 38.Falcoz PE, Laluc F, Toubin MM, Puyraveau M, Clement F, Mercier M, et al. Usefulness of procalcitonin in the early detection of infection after thoracic surgery. Eur J Cardiothorac Surg. 2005;27:1074–8. doi: 10.1016/j.ejcts.2005.02.022. [DOI] [PubMed] [Google Scholar]
- 39.Jebali MA, Hausfater P, Abbes Z, Aouni Z, Riou B, Ferjani M. Assessment of the accuracy of procalcitonin to diagnose postoperative infection after cardiac surgery. Anesthesiology. 2007;107:232–8. doi: 10.1097/01.anes.0000271871.07395.ad. [DOI] [PubMed] [Google Scholar]
- 40.Matthiessen P, Henriksson M, Hallböök O, Grunditz E, Norén B, Arbman G. Increase of serum C-reactive protein is an early indicator of subsequent symptomatic anastomotic leakage after anterior resection. Colorectal Dis. 2008;10:75–80. doi: 10.1111/j.1463-1318.2007.01300.x. [DOI] [PubMed] [Google Scholar]
- 41.Ortega-Deballon P, Radais F, Facy O, d'Athis P, Masson D, Charles PE, et al. C-reactive protein is an early predictor of septic complications after elective colorectal surgery. World J Surg. 2010;34:808–14. doi: 10.1007/s00268-009-0367-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schietroma M, Carlei F, Cecilia EM, Piccione F, Sista F, De Vita F, et al. A prospective randomized study of systemic inflammation and immune response after laparoscopic nissen fundoplication performed with standard and low-pressure pneumoperitoneum. Surg Laparosc Endosc Percutan Tech. 2013;23:189–96. doi: 10.1097/SLE.0b013e3182827e51. [DOI] [PubMed] [Google Scholar]
- 43.Woeste G, Müller C, Bechstein WO, Wullstein C. Increased serum levels of C-reactive protein precede anastomotic leakage in colorectal surgery. World J Surg. 2010;34:140–6. doi: 10.1007/s00268-009-0304-z. [DOI] [PubMed] [Google Scholar]
- 44.Dutta S, Fullarton GM, Forshaw MJ, Horgan PG, McMillan DC. Persistent elevation of C-reactive protein following esophagogastric cancer resection as a predictor of postoperative surgical site infectious complications. World J Surg. 2011;35:1017–25. doi: 10.1007/s00268-011-1002-1. [DOI] [PubMed] [Google Scholar]
- 45.Schuetz P, Albrich W, Mueller B. Procalcitonin for diagnosis of infection and guide to antibiotic decisions: Past, present and future. BMC Med. 2011;9:107. doi: 10.1186/1741-7015-9-107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Csendes A, Burgos AM, Roizblatt D, Garay C, Bezama P. Inflammatory response measured by body temperature, C-reactive protein and white blood cell count 1, 3, and 5 days after laparotomic or laparoscopic gastric bypass surgery. Obes Surg. 2009;19:890–3. doi: 10.1007/s11695-008-9702-9. [DOI] [PubMed] [Google Scholar]
- 47.Kim I, Kim HG, Kim H, Kim HH, Park SK, Uhm CS, et al. Hepatic expression, synthesis and secretion of a novel fibrinogen/angiopoietin-related protein that prevents endothelial-cell apoptosis. Biochem J. 2000;346(Pt 3):603–10. [PMC free article] [PubMed] [Google Scholar]
- 48.Tennent GA, Brennan SO, Stangou AJ, O'Grady J, Hawkins PN, Pepys MB. Human plasma fibrinogen is synthesized in the liver. Blood. 2007;109:1971–4. doi: 10.1182/blood-2006-08-040956. [DOI] [PubMed] [Google Scholar]
- 49.Yamaguchi T, Yamamoto Y, Yokota S, Nakagawa M, Ito M, Ogura T. Involvement of interleukin-6 in the elevation of plasma fibrinogen levels in lung cancer patients. Jpn J Clin Oncol. 1998;28:740–4. doi: 10.1093/jjco/28.12.740. [DOI] [PubMed] [Google Scholar]
- 50.Miki C, Konishi N, Ojima E, Hatada T, Inoue Y, Kusunoki M. C-reactive protein as a prognostic variable that reflects uncontrolled up-regulation of the IL-1-IL-6 network system in colorectal carcinoma. Dig Dis Sci. 2004;49:970–6. doi: 10.1023/b:ddas.0000034556.48527.6e. [DOI] [PubMed] [Google Scholar]
- 51.Weisel JW. Fibrinogen and fibrin. Adv Protein Chem. 2005;70:247–99. doi: 10.1016/S0065-3233(05)70008-5. [DOI] [PubMed] [Google Scholar]
- 52.Medina Fernández FJ, Muñoz-Casares FC, Arjona-Sánchez A, Casado-Adam A, Gómez-Luque I, Garcilazo Arismendi DJ, et al. Postoperative time course and utility of inflammatory markers in patients with ovarian peritoneal carcinomatosis treated with neoadjuvant chemotherapy, cytoreductive surgery, and HIPEC. Ann Surg Oncol. 2015;22:1332–40. doi: 10.1245/s10434-014-4096-5. [DOI] [PubMed] [Google Scholar]
- 53.Forget P, Dinant V, De Kock M. Is the Neutrophil-to-Lymphocyte Ratio more correlated than C-reactive protein with postoperative complications after major abdominal surgery? PeerJ. 2015;3:e713. doi: 10.7717/peerj.713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kahramanca S, Kaya O, Özgehan G, Irem B, Dural I, Küçükpınar T, et al. Are neutrophil-lymphocyte ratio and platelet-lymphocyte ratio as effective as Fournier's gangrene severity index for predicting the number of debridements in Fourner's gangrene? Ulus Travma Acil Cerrahi Derg. 2014;20:107–12. doi: 10.5505/tjtes.2014.62829. [DOI] [PubMed] [Google Scholar]
- 55.Turak O, Özcan F, Işleyen A, Başar FN, Gül M, Yilmaz S, et al. Usefulness of neutrophil-to-lymphocyte ratio to predict in-hospital outcomes in infective endocarditis. Can J Cardiol. 2013;29:1672–8. doi: 10.1016/j.cjca.2013.05.005. [DOI] [PubMed] [Google Scholar]
- 56.Terradas R, Grau S, Blanch J, Riu M, Saballs P, Castells X, et al. Eosinophil count and neutrophil-lymphocyte count ratio as prognostic markers in patients with bacteremia: A retrospective cohort study. PLoS One. 2012;7:e42860. doi: 10.1371/journal.pone.0042860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.de Jager CP, Wever PC, Gemen EF, Kusters R, van Gageldonk-Lafeber AB, van der Poll T, et al. The neutrophil-lymphocyte count ratio in patients with community-acquired pneumonia. PLoS One. 2012;7:e46561. doi: 10.1371/journal.pone.0046561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.de Jager CP, van Wijk PT, Mathoera RB, de Jongh-Leuvenink J, van der Poll T, Wever PC. Lymphocytopenia and neutrophil-lymphocyte count ratio predict bacteremia better than conventional infection markers in an emergency care unit. Crit Care. 2010;14:R192. doi: 10.1186/cc9309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Segel GB, Halterman MW, Lichtman MA. The paradox of the neutrophil's role in tissue injury. J Leukoc Biol. 2011;89:359–72. doi: 10.1189/jlb.0910538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gennari R, Dominioni L, Imperatori A, Bianchi V, Maroni P, Dionigi R. Alterations in lymphocyte subsets as prognosticators of postoperative infections. Eur J Surg. 1995;161:493–9. [PubMed] [Google Scholar]


