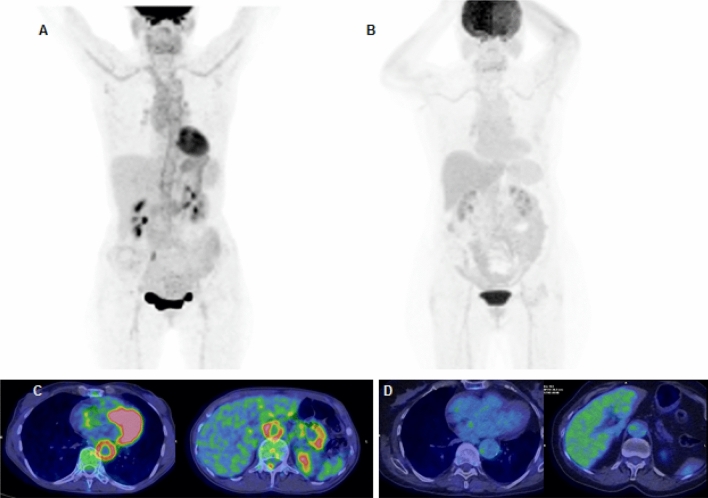
Abstract
Aim
The diagnosis, severity and extent of a sterile inflammation or a septic infection could be challenging since there is not one single test able to achieve an accurate diagnosis. The clinical use of 18F-fluorodeoxyglucose ([18F]FDG) positron emission tomography/computed tomography (PET/CT) imaging in the assessment of inflammation and infection is increasing worldwide. The purpose of this paper is to achieve an Italian consensus document on [18F]FDG PET/CT or PET/MRI in inflammatory and infectious diseases, such as osteomyelitis (OM), prosthetic joint infections (PJI), infective endocarditis (IE), prosthetic valve endocarditis (PVE), cardiac implantable electronic device infections (CIEDI), systemic and cardiac sarcoidosis (SS/CS), diabetic foot (DF), fungal infections (FI), tuberculosis (TBC), fever and inflammation of unknown origin (FUO/IUO), pediatric infections (PI), inflammatory bowel diseases (IBD), spine infections (SI), vascular graft infections (VGI), large vessel vasculitis (LVV), retroperitoneal fibrosis (RF) and COVID-19 infections.
Methods
In September 2020, the inflammatory and infectious diseases focus group (IIFG) of the Italian Association of Nuclear Medicine (AIMN) proposed to realize a procedural paper about the clinical applications of [18F]FDG PET/CT or PET/MRI in inflammatory and infectious diseases. The project was carried out thanks to the collaboration of 13 Italian nuclear medicine centers, with a consolidate experience in this field. With the endorsement of AIMN, IIFG contacted each center, and the pediatric diseases focus group (PDFC). IIFG provided for each team involved, a draft with essential information regarding the execution of [18F]FDG PET/CT or PET/MRI scan (i.e., indications, patient preparation, standard or specific acquisition modalities, interpretation criteria, reporting methods, pitfalls and artifacts), by limiting the literature research to the last 20 years. Moreover, some clinical cases were required from each center, to underline the teaching points. Time for the collection of each report was from October to December 2020.
Results
Overall, we summarized 291 scientific papers and guidelines published between 1998 and 2021. Papers were divided in several sub-topics and summarized in the following paragraphs: clinical indications, image interpretation criteria, future perspectivess and new trends (for each single disease), while patient preparation, image acquisition, possible pitfalls and reporting modalities were described afterwards. Moreover, a specific section was dedicated to pediatric and PET/MRI indications. A collection of images was described for each indication.
Conclusions
Currently, [18F]FDG PET/CT in oncology is globally accepted and standardized in main diagnostic algorithms for neoplasms. In recent years, the ever-closer collaboration among different European associations has tried to overcome the absence of a standardization also in the field of inflammation and infections. The collaboration of several nuclear medicine centers with a long experience in this field, as well as among different AIMN focus groups represents a further attempt in this direction. We hope that this document will be the basis for a “common nuclear physicians’ language” throughout all the country.
Supplementary Information
The online version contains supplementary material available at 10.1007/s40336-021-00445-w.
Keywords: PET, FDG, Imaging, Infection, Inflammation
Introduction
Since the initial clinical use of [18F]FDG, nuclear medicine physicians and clinicians, have understood the potentiality of this radiopharmaceutical for imaging sterile inflammatory processes and also septic inflammation (infection). Indeed, foci of sterile or septic inflammation were, initially, considered as pitfalls of PET/CT scans and a source of false positivity in oncological patients. With increasing experience and expansion of the application’s fields of PET/CT, it became clear that [18F]FDG could have been successfully used to study patients with inflammatory diseases. For sure, the first clinical application, in this regard, was fever of unknown origin (FUO) since [18F]FDG PET/CT was able to change the clinical management of patients in a high percentage of cases.
In the last 2 decades, more clinical applications have been investigated with excellent results in some pathologies, such as large vessel vasculitis (LVV), systemic and cardiac sarcoidosis (SS/CS), spine infections (SI), but with less promising results in other pathologies, such as prosthetic joint infections (PJI).
Clinical studies continue to be performed and published and, in particular, studies with appropriate reference standard, or multicenter studies, or head-to-head comparisons between different diagnostic techniques, will continue to improve our knowledge and correct use of [18F]FDG PET in inflammatory diseases.
The Italian Association of Nuclear Medicine (AIMN) created, almost 20 years ago, a study group to investigate the role of nuclear medicine imaging in sterile and septic inflammatory processes.
Regarding [18F]FDG PET for the imaging of sterile and septic inflammatory processes, there many different imaging protocols and interpretation criteria have been published, thus making it difficult, in some cases, to reliably use [1].
Therefore, in the last 2 years, the main purposes of the AIMN study group focussed on infection and inflammation, have been to (1) carefully study all protocols proposed in the literature, and (2) summarize and analyse them, with the final goal of providing practical indications for nuclear medicine physicians about how to use [18F]FDG PET appropriately and how to correctly interpret images in different inflammatory diseases.
Materials and methods
To accomplish the goal of this extensive literature search, we analysed all published papers in the last 20 years dealing with [18F]FDG PET/CT or PET/magnetic resonance imaging (MRI) in inflammation or infection. Amongst all papers retrieved, we selected the most relevant, based on our experience and on the methodology used in the manuscripts. Papers were divided in several sub-topics, each one characterized by the following paragraphs: clinical indications, image interpretation criteria, future perspectives and new trends (for each single disease), while patient preparation, image acquisition, possible pitfalls and reporting modalities were described afterwards.
Overall, we summarized 291 scientific papers and guidelines published between 1998 and 2021.
When a national or European guideline or a consensus paper was available, we mainly referred to it [1–21].
Results
From all 291 retrieved studies, clinical indications and the image interpretation criteria for SIs, diabetic foot infections (DFI), osteomyelitis (OM), LVV, vascular graft infections (VGI), infectious endocarditis (IE), cardiovascular implantable electronic device (CIEDs) infections, left ventricular assist device associated (LVADs) infections, CS, retroperitoneal fibrosis (RF), FUO, SS, tuberculosis (TBC), invasive fungal infections (IFI), HIV-infection, SARS-CoV-2 infection and inflammatory bowel diseases (IBD), were carefully reported.
Then, we summarized the results of recent applications of PET/MRI and in pediatric patients in this setting of disease.
Finally, a practical guide for patient preparation, image acquisition protocol, possible pitfalls and how to prepare the medical report, has also been reported.
Role of 18F-FDG PET/CT in spine infections
SI can involve vertebral bone (spondylitis), intervertebral disc (discitis) or both vertebral bodies and corresponding intervertebral disc with frequent extension to the contiguous soft tissues (spondylodiscitis) [2]. SI has been classified as primary or secondary according to the modality of infection. Primary SI has a hematogenous origin while secondary SI is due to direct contamination, in most cases after surgical procedures. SI incidence has progressively increased in the last years. Men, aged between 50 and 70 years, are more affected than women [2, 3]. The most frequent predisposing factors are endocarditis, diabetes mellitus, septic arthritis, urinary tract infections, immunodeficiency, long-term steroid use, malignancy and spinal surgery [2, 3]. The most common etiological agents of SI are pyogenic bacteria, such as Staphylococcus aureus, followed by Streptococci; less frequently, it can be caused by non-pyogenic agents such as Mycobacterium Tuberculosis, fungi and parasites [2]. The most frequent region involved is the lumbar spine, followed by dorsal and cervical regions [3]. The diagnosis of SI is based on clinical signs (focal back pain, fever and/or neurological deficit), laboratory tests (elevated inflammation indexes, positive blood culture) and imaging findings [2, 3]. The mean time for the diagnosis of SI is still too long and the rate of complications increases with a delayed diagnosis. An early diagnosis is required to prevent irreversible complications. Diagnostic imaging is necessary to confirm the clinical suspicious of SI. MRI acquired with STIR or fat-saturated T2-weighted sequences and T1-weighted SE fat-suppressed pre- and post i.v. injection of contrast media, is considered the most accurate technique for the diagnosis of primary SI [2, 4]. However, some studies show similar diagnostic accuracy of MRI and [18F]FDG PET/CT in primary SI underlining the importance of the complementarity of both diagnostic procedures [5, 6]. MRI can be replaced by [18F]FDG PET/CT in patients with contraindications to MRI and in case of MRI inconclusive report [2, 3]. [18F]FDG PET/CT should be performed in patients with suspected secondary SI, since the presence of post-surgical reactive tissue does not affect the outcome of the diagnostic procedure. Finally, [18F]FDG PET/CT should be performed for the evaluation of treatment response [2, 3, 7, 8] having preferably acquired a basal scan before the start of treatment. Moreover, it is important to know that MRI showed highest sensitivity in diagnosing epidural and spinal abscesses while [18F]FDG PET/CT was more sensitive in diagnosing paravertebral and psoas abscesses [5]. In primary and mostly in secondary SI, the nuclear medicine technique seems to show superior diagnostic value in the early course of disease (< 15 days) [5]. Last but not least, [18F]FDG PET/CT allows to study the total-body of patient, identifying other potential foci of infection [9]. The specificity of [18F]FDG PET/CT remains the crucial issue since it is not able to differentiate infection from marked degenerative disease or malignancy, for this reason, we must always evaluate the entire medical history of the patient.
Clinical indications
Diagnosis of suspected SI in patients with elevated C-reactive protein (CRP) and/or erythrocyte sedimentation rate (ESR);
Diagnosis of suspected primary SI;
Diagnosis of secondary SI (at least 4 months after surgery) [5];
Suspected recurrence of disease in patients with previous SI;
Assessment of the extent of the SI and identification of complications (i.e., paravertebral, psoas abscess);
Evaluation of antibiotic therapy response in primary and secondary SI.
Interpretation criteria
Qualitative analysis: [18F]FDG PET/CT is considered negative for SI if [18F]FDG uptake of the suspected site is smooth and homogeneous with similar intensity of adjacent vertebrae and surrounding soft tissue [3]. [18F]FDG PET/CT is considered positive for SI if [18F]FDG uptake of the suspected site is higher than the adjacent vertebrae and surrounding soft tissue [3].
A useful score for the qualitative interpretation of PET images has been proposed by Hungenbach et al.:
Score 0: Normal findings and physiological [18F]FDG distribution (no infection);
Score 1: slightly increased uptake in the inter- or paravertebral region (no infection);
Score 2: clearly increased uptake of a linear or disciform pattern in the intervertebral space (discitis);
Score 3: clearly increased uptake of a linear or disciform pattern in the intervertebral space and involvement of ground or cover plate or both plates of the adjacent vertebrae (spondylodiscitis);
Score 4: clearly increased uptake of a linear or disciform pattern in the intervertebral space and involvement of ground or cover plate or both plates of the adjacent vertebrae associated with surrounding soft-tissue abscess (spondylodiscitis) [10].
Quantitative analysis: SUVmax is the most validated semi-quantitative parameter. No cut-off has been established to differentiate between positive and negative findings; SUVmax > 3 seems to be compatible with SI [11]. Semi-quantitative analysis is also useful to evaluate the response to therapy; in particular, a follow-up SUVmax reduction compared to basal value is considered suggestive of a good response to treatment. Literature reports a cut-off of ΔSUVmax ((SUVmax before treatment − SUVmax after treatment)/SUVmax before treatment) between 25 and 43% suggestive of metabolic response to therapy [7, 12].
In summary, SI needs to have a fast and early diagnosis to prevent irreversible complications. MRI is considered the most accurate technique for the diagnosis of primary SI and shows high sensitivity in diagnosing epidural and spinal abscesses. [18F]FDG PET/CT can be performed in patients with contraindications to MRI and in case of the suspect of secondary SI. [18F]FDG PET/CT is highly sensitive for the diagnosis of paravertebral and psoas abscesses and for the evaluation of treatment response, mostly in secondary SI. It is recommended to perform a basal PET/CT scan before or immediately at the beginning of antibiotic treatment.
Future perspectives and new trends
[18F]FDG PET/MRI is a multimodality imaging that can match the high sensitivity of [18F]FDG PET/CT in detecting metabolic alterations with the high specificity of MRI for spine lesions evaluation; therefore, it represents the ideal tool for a one-stop shop benefit. Even if only a limited number of studies of PET/MRI in patients with SI are available now [13].
Some experimental studies are now in progress to find new radiopharmaceuticals, as 68 Ga-DOTA-Biotin, to improve the specificity of [18F]FDG in SI diagnosis [14].
Role of [18F]FDG PET/CT in diabetic foot infections
It has been estimated that up to 15% of patients with longstanding diabetes will develop foot complications such as ulcers and infections and more than 50% of wounds are infected at their presentation. The development of an infection represents a dramatic event for both patients and society, being associated to considerable morbidity, increased risk of major complications that often require foot amputation, and high mortality rate. Therefore, DFIs represent one of the most relevant health issues worldwide [15, 16]. Diabetic neuropathy, micro/macro angiopathy and immune system alterations, play a central role in the development of foot ulcers that may remain unrecognized by the patient and may be difficult to heal because of the presence of peripheral artery disease. Foot ulcers become the entryway of several pathogens thus predisposing to the development of a soft tissue infection (STI). If not promptly identified, the infection could reach the bone, thus causing OM that is one of the most disabling complications of uncontrolled diabetes. Charcot osteoarthropathy, with or without a superimposed infection, is another foot complication of diabetes that further complicates the diagnosis. Indeed, the most important diagnostic challenge is to differentiate between these three conditions since they require very different therapeutic approaches [17]. In case of OM, it is also important to correctly evaluate the extent of the infection and of affected bones.
Although the gold standard for the diagnosis of an infection is still represented by the isolation of causative micro-organisms, through bone biopsy or cultures from ulcers, these approaches are often invasive and associated to risk of sample contaminations [18, 19]. Therefore, imaging plays a crucial role in the diagnosis of infective process and for the evaluation of its extent, thus providing important information to plan the most appropriate therapy. Plain X-ray film usually represents the first step imaging modality since it provides an anatomical overview of the foot and detects any pre-existing condition that should be known for the correct interpretation of second level imaging modalities. Despite the specificity for detecting an infection is low, MRI is the radiologic modality of choice for the study of DFI since it provides high quality images with high definition between soft tissues and bone, being able to achieve the diagnosis of infection with a good accuracy [17]. From a nuclear medicine point of view, the gold standard imaging modality for diagnosis of infective complications of DF, still remains radiolabeled white blood cell (WBC) scintigraphy [20–23], especially if performed following EANM recommendations [20, 24, 25], as also demonstrated in previous meta-analysis [26] and by a large retrospective study comparing radiolabeled WBC scintigraphy, [18F]FDG PET/CT and MRI [23]. However, also [18F]FDG PET/CT is gaining an interesting role in imaging infections and inflammation particularly in lesions of the forefoot [27]. Nevertheless, data in literature show discordant results, mainly due to the lack of univocal interpretation criteria for PET imaging. From the systematic review and meta-analysis published in 2013 by Treglia and colleagues, per-patient-based analysis showed a pooled sensitivity of 74% and a specificity of 91% [28]. However, this meta-analysis included only four papers [29–32]. In another more recent systematic review and meta-analysis comparing radiolabeled WBC scintigraphy, [18F]FDG PET/CT and MRI, these three imaging modalities showed comparable sensitivities for the detection of DF osteomyelitis (DFO) whereas, both radiolabeled WBC scintigraphy and [18F]FDG PET/CT showed highest specificity [21]. In a recently published retrospective study, in 251 patients, aiming to compare different imaging modalities in detecting OM, STI and Charcot, [18F]FDG PET/CT showed significantly higher specificity compared with MRI (95.7% vs 83.6% respectively) in detecting STI, especially in mid/hindfoot and a good sensitivity, specificity and accuracy in detecting pedal OM, in particular in the forefoot [23].
Clinical indications
Diagnosis of foot complications (in particular for the detection of forefoot infections and the evaluation of its extent from soft tissues to bone);
Therapeutic monitoring and follow-up (although not completely addressed);
Interpretation criteria
Qualitative analysis: Definite and commonly accepted image interpretation criteria still do not exist for [18F]FDG PET/CT in DF. Image interpretation relies on personal experience and on the evaluation of co-registered CT scan. Qualitative assessment of the uptake, in terms of pattern (focal or diffused), intensity (for example, comparing the uptake with contra-lateral side) and the description of number of foci and their location, should always be the first step. In a large retrospective study, OM was defined in presence of focal or diffused uptake higher than contralateral side clearly involving the bone; STI was defined when focal or diffused uptake was detected only on soft tissue and without bone involvement (Fig. 1) [23]. Nevertheless, while this interpretation criteria is easily adaptable to the forefoot, differential diagnosis between bone infection, STI and Charcot neuro-osteoarthropathy in the mid-hindfoot is much more complicated and sometimes impossible by [18F]FDG.
Fig. 1.
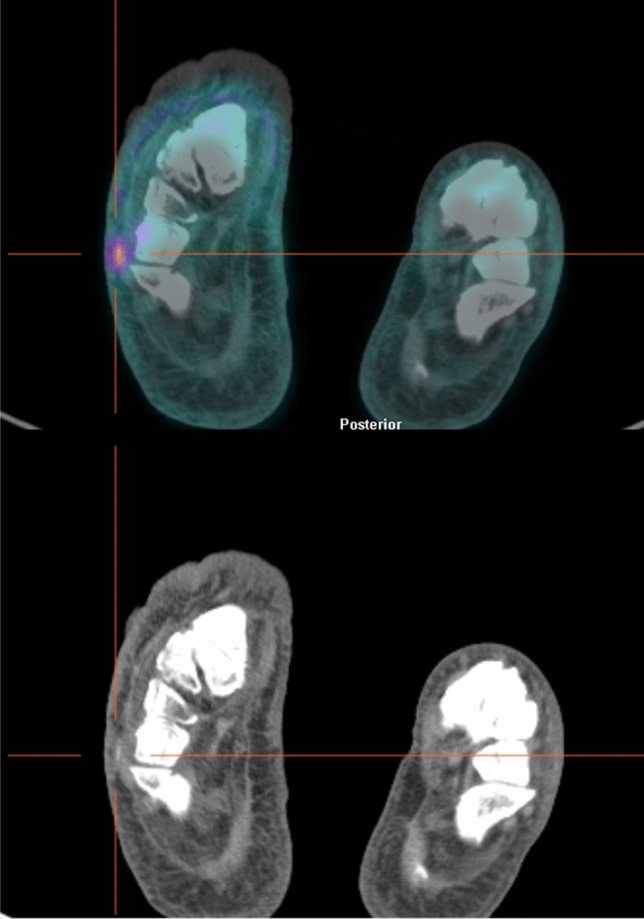
Axial views of [18F]FDG PET/CT images (top) and low-dose CT scan (bottom) of a diabetic patient with suspected osteomyelitis of right foot. The scan identified a focal uptake on a cutaneous/subcutaneous ulcer of the soft tissues of plantar surface without bone involvement, thus ruling out the diagnosis of osteomyelitis
In some recent published papers, the interpretation criteria to discriminate between OM and Charcot have been described, concluding that qualitative assessment of [18F]FDG pattern could allow such differentiation [23, 33]. However, these criteria are not universally adopted among the centers, thus explaining the [18F]FDG PET/CT heterogeneity in reported accuracy. Moreover, many concerns still exist on interpretation criteria of [18F]FDG PET/CT performed during antibiotic therapy, especially when the scan has to be compared with a basal study for the evaluation of treatment efficacy.
Semi-quantitative analysis: SUVmax is also possible, but despite several authors explored its possible role, no well-defined thresholds able to differentiate between infection and sterile inflammation, have yet been identified. Moreover, its role for follow-up and therapy assessment studies is still unclear.
In summary, [18F]FDG PET/CT represents a useful and non-invasive imaging modality for the assessment of foot complications in diabetic patients but the low specificity of [18F]FDG in differentiating between infection and sterile inflammation still represents the major drawback of this modality. The lack of standardized interpretation criteria is another important limitation; therefore, the accuracy of this modality in discriminating between OM, STI and Charcot still relies on personal experience.
Future perspectives and new trends
Aiming at developing a more specific radiopharmaceutical for PET imaging WBC have also been labelled with [18F]FDG showing promising results [34]. The short half-life of 18F, indeed, represents a major limiting factor since delayed and late images cannot be performed. PET/MRI is also demonstrating a great role in musculoskeletal infections and, in future, it will gain an important application in defining the different complications of DF. Hopefully, this modality will solve the most challenging clinical scenarios in DFI [35].
Role of [18F]FDG PET/CT in osteomyelitis and prosthetic joint infections
OM is an inflammatory process of the bone caused by an infecting microorganism with associated bone destruction. Hematogenous OM is mainly seen in pre-pubertal children and in elderly patients and it is due to bone seeding of bacteria present in the bloodstream, often deriving from other sites of occult infections (oro-pharingeal, gastro-intestinal, urinary, etc.). Secondary OMs are more common and often occur after bone-exposed fractures and after joint replacement. Joint prosthetic replacements are increasing in the world and it is estimated that, by 2030, 2 million of total hip or total knee arthroplasties will be annually performed in United States. PJI occurs in 1% to 2% of primary and up to 4% of revision arthroplasties.
The infection typically spreads in the soft tissues without respecting its boundaries, while in the bone, it spreads in the periosteal area or inside the medullary cavity, invading the vascular channels and, raising the intraosseous pressure, reducing the blood flow until causing the typical septic bone necrosis.
The clinical diagnosis of infection is based on clinical and laboratory criteria such as hyperpyrexia, neutrophilic granulocytosis, ESR and CRP which, although highly sensitive (increased in 97% of cases) are not specific and can also be detected in rheumatic diseases and neoplasms and vice versa, can be normal in the presence of a chronic or only moderately active infection. The differential diagnosis between aseptic inflammation and infection by means of nuclear medicine examination, is essential, particularly in the presence of metal hardware or deep sites that limit the study with radiological techniques [36, 37]. [18F]FDG PET/CT is increasingly being used in the diagnosis of PJI, especially for hip arthroplasty. Differing from leukocyte scintigraphy, [18F]FDG accumulation in infections does not depend on leukocyte migration but it is mainly related to the glycolytic activity of the cells involved in the inflammatory response [27]. Compared with radiolabeled WBC scintigraphy, [18F]FDG PET/CT offers advantages such as time efficiency, increased resolution, and the use of low-dose CT [38, 39]. Nevertheless, it has a lower diagnostic accuracy respect to WBC scintigraphy as previously well demonstrated [36, 37, 40].
The detection of peri-prosthetic inflammation using [18F]FDG PET/CT had a sensitivity of 90–100% for septic cases and of 45–80% in cases of increased abrasion and aseptic foreign-body reactions. However, reliable differentiation between abrasion-induced and bacterial caused inflammation is not possible using [18F]FDG PET/CT. In patients with knee arthroplasty, only uptake at the bone/prosthesis interface was considered as being consistent with infection.
In post-traumatic osteomyelitis and fracture-related infections the sensitivity of [18F]FDG PET/CT ranges between 86 and 94% and specificity between 76 and 100%. Recent fractures and the presence of metallic hardware may decrease the accuracy of [18F]FDG PET/CT [1, 41].
Clinical indications
[18F]FDG PET/CT has high sensitivity but a low specificity compared with radiolabeled WBC scintigraphy and it is mainly indicated in chronic infections characterized by a prevalent population of macrophages and monocytes while the component of neutrophilic granulocytes is minimal. Therefore, the most frequent clinical indications for [18F]FDG PET/CT are in the diagnosis and in therapy monitoring of:
Chronic pelvic OM (including after pressure ulceration);
Chronic OM (e.g. of long bone);
Chronic destructive septic arthritis;
Prosthetic joint infections, infected fracture non-unions, infected fracture fixation implants (healed fracture); however, until now, no well-established clinical indications and interpretation criteria are available.
Interpretation criteria
Qualitative analysis: different patterns (focal, linear, and diffused), in intensity of [18F]FDG uptake and, in comparison to areas of physiological distribution have been used to assess positivity in bone infections. For example, the distribution of the activity uptake along the bone-prosthesis interface of the shaft, the decreasing of activity in a dual time modality of acquisition, and so on [42]. The major limitation of [18F]FDG is the inability of the radiopharmaceutical to discriminate between malignancy, infection, and inflammation [1]. Moreover, there is still not a universal agreement on the validity of these criteria. Repeated PET studies after antibiotics treatment can be useful to confirm the diagnosis and assess the response to this treatment. The femoral component is considered the most reliable indicator for peri-prosthetic hip infection, since the accumulation around the head, neck, and distal tip of the prosthesis can remain for up to 2 years after implant [43].
In summary, [18F]FDG PET/CT allows reliable prediction of peri-prosthetic septic inflammatory tissue reactions. Because of the high negative predictive value of this method, a negative PET result in the setting of a diagnostically unclear situation eliminates the need for revision surgery. In contrast, a positive PET result gives no clear differentiation regarding the cause of inflammation. At the present time, the role of [18F]FDG imaging in the evaluation of arthroplasty infection and of post-fracture infections remains to be determined [44, 45].
Future perspectives and new trends
Several other radiopharmaceuticals showed potential relevance in OM, such as [18F]-Fluoroetiltirosine (FET), [18F]-Choline, [18F]-Fluoride and others. In some selected cases, PET/MRI could also be helpful by combining the sensitivity of [18F]FDG PET/CT with the specificity of MRI.
Role of [18F]FDG PET/CT in large vessel vasculitis
Primary systemic vasculitides are classified by the diameter of the vessels that are predominantly involved [46–49]. The increasing availability and improvement of imaging techniques are making a deep impact in the evaluation and follow-up of patients with vasculitis, particularly for those with LVV [50]. LVV are vasculitides affecting large arteries more often than other vasculitides. Large arteries are aorta and its major branches. LVV include giant cell arteritis (GCA) and Takayasu arteritis (TAK). The histopathologic features of TAK and GCA are indistinguishable; both are granulomatous vasculitides involving the aorta and its major branches. TAK and GCA occur predominantly in females and share many clinical and radiographic features. The age at onset is generally used to distinguish between the two diseases. TAK is a disease predominantly of younger individuals (under 40 years) while GCA is a disease of older individuals (over 50 years). GCA has a predilection for the branches of the carotid and vertebral arteries, and often involves the temporal artery [49–51]. While cranial manifestations of GCA are well recognized, it has been increasingly appreciated that GCA is a systemic disease that extends beyond the superficial temporal arteries and can cause manifestations including large-artery stenosis or aortic involvement (aortitis, aneurysm formation and dissection) [52]. Several prospective imaging studies in patients with newly diagnosed GCA have shown that a majority of patients with GCA have inflammation of the aorta and its branches [50]. The usefulness of [18F]FDG PET/CT in diagnosing LVV has been borne out by several studies. In a recent systematic review and meta-analysis, [18F]FDG PET/CT had a sensitivity of 90% and a specificity of 98% for GCA, while it had a sensitivity of 87% and a specificity of 73% for TAK [53]. [18F]FDG PET/CT is particularly valuable in securing the diagnosis of LVV in patients with less typical manifestations, such as FUO, or when other diagnostic tests are inconclusive, e.g. when temporal artery biopsy is negative in GCA. The role of [18F]FDG PET/CT is less well established in predicting the course of the disease and its complications [50].
Clinical indications
Diagnosis of suspected LVV (evaluation of disease extent and disease activity) in patients with elevated CRP and/or ESR;
Assessment of therapy efficacy (Fig. 2);
Exclusion of other causes of systemic symptoms;
As a guide for biopsy (potential use);
Surrogate endpoints in clinical trials [54].
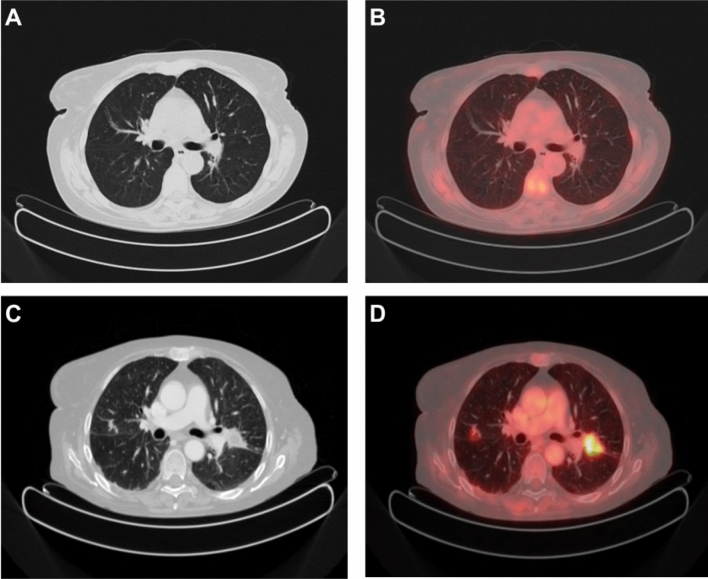
Fig. 2.
>50-year-old female with Giant Cell Arteritis (GCA). MIP (A, B) and axial PET/CT views before and after steroid treatment. Baseline (A, C): pathologic tracer uptake at ascending aorta, descending aorta, thoracic aorta, aortic arc, anonymous artery, subclavian arteries, common carotid arteries and abdominal aorta. Vascular tracer uptake greater than the hepatic one (grade 3 according to Meller reference scale). After treatment (B, D): no pathological uptake 3 months after steroid and immunosuppressive therapy (ongoing)
Interpretation criteria
Qualitative analysis: visual [18F]FDG vessel uptake is the grading system currently used. Meller et al. in 2003 proposed a four-point grading scale, comparing large vessels to hepatic [18F]FDG uptake (“0” = no uptake; “1” = uptake inferior to the liver; “2” = uptake similar to the liver; “3” = uptake superior to the liver). This approach is characterized by high specificity (about 100%, but with variable sensitivity levels between 56 and 77%) and high inter- and intra-observer reproducibility (90% and 93% respectively) [55]. A vascular uptake degree equal to the liver (Grade 2) can be considered “suspect” for LVV, while a lower grade (Grade 1) is likely to exclude its presence. A recently published study [56] shows that up to 45% of control patients without LVV can express, during visual assessment, a vascular uptake of Grade 1, 14% of Grade 2, and 4% of Grade 3. Type (i.e., linear, segmental, or focal) and intensity of the vessel wall [18F]FDG uptake, also must be taken into consideration: in LVV the radiopharmaceutical distribution type is “spread” (or initially segmental), while a “focal” type can easily be attributable to atherosclerotic plaques.
Semi-quantitative analysis: an important overlap of the SUVmax value between normal controls and LVV patients and the absence of reference recognized values of normality significantly limit its use in diagnosing vasculitides [57]. Total vascular score (TVS) takes into consideration seven vascular districts (carotid arteries, subclavian, axillary, iliac, femoral, thoracic aorta, abdominal aorta) assigning to each one an uptake score (ranging from “0” to “3”) based on the intensity degree (maximum TVS of 21). A TVS equal and/or superior to 6 ± 0.2 is considered highly specific for the presence of vasculitic disease [55, 58]. This approach is still debated and not yet universally accepted. Ratio of SUVmax vascular and SUVmax of a reference organ (liver, lung, vascular pool) reflects the limits already indicated above, and at the moment, its use is suggested only for clinical research or in doubtful case [59].
Future perspectives and new trends
The [18F]FDG PET/MRI is a multimodality imaging that combines the high sensitivity of [18F]FDG PET/CT in detecting vascular/perivascular inflammation with the high specificity of MRI. [18F]FDG PET/MRI is feasible in LVV with visual and quantitative results highly comparable to PET/CT for precisely determining disease extent and disease activity in LVV [60].
Role of [18F]FDG PET/CT in vascular graft infections
Despite the incidence of VGI being low, ranging from 1.5% to 6%, it represents an extremely severe complication of vascular surgery being associated with bad prognosis and high mortality rate [61]. Clinical presentation may vary according to time elapsed from surgery, type and site of the device, causative pathogen and to patient-related risk factors.
A prompt and accurate diagnosis is mandatory to correctly manage this kind of infection and to avoid the risk of major complications [24, 62]. Nevertheless, the diagnosis can be really challenging.
A combination of microbiology, laboratory tests, and imaging is crucial [63, 64]; however, an univocal diagnostic strategy and a well-defined flow-chart do not exist yet, thus resulting in different approaches among various centers.
Several imaging modalities are currently used for diagnosis and follow-up of VGI [65]. From a radiologic point of view, CT and computed tomography angiography (CTA) represent the gold standard imaging modalities for their ability to provide an anatomical overview of vascular tree and to detect suggestive signs for infection (peri-graft gas or fluid collection, fistulae, adjacent soft tissue abscesses). However, the reported sensitivity and specificity is 67% and 63%, respectively [66] that may be even lower in low-grade infections [64]. MRI could be potentially used but at the moment its role in diagnosing VGI has not been extensively studied.
On the other hand, nuclear medicine offers the possibility to explore the underlying physiopathology of the infective process thus providing functional information. One of the main strong points of NM is the possibility to achieve an accurate differentiation between infection and sterile inflammation by performing radiolabeled white blood cells (WBC) scintigraphy and following well-standardized protocols for images acquisitions and display and well-defined interpretation criteria [20, 67]. 18F-fluorodeoxyglucose ([18F]FDG) positron emission tomography/computed tomography (PET/CT) has also gained an important role in the field of infective and inflammatory diseases. Nevertheless, well-standardized acquisition protocols and interpretation criteria still do not exist in the specific field of VGI [27]. From several meta-analysis and systematic reviews emerges a high sensitivity but a low specificity for this modality [65, 68–70], mainly related to the high rate of false positives results that may be observed in presence of physiological post-surgical inflammation due to foreign body reaction induced by synthetic materials [71, 72]. In one of the most recently published systematic reviews and meta-analysis, [69] the reported pooled sensitivity is 96%, ranging from 81% [73] and 100% [72, 74–76], whereas the reported pool specificity is 74%, ranging between 29% [77] and 92% [78]. This extreme variability in literature is mainly related, not only to the low specificity of [18F]FDG itself in discriminating between infection and sterile inflammation, but also to the lack of univocal interpretation criteria for [18F]FDG PET/CT, thus underlining the need of a standardization.
Clinical indications
Interpretation criteria
Several interpretation criteria have been proposed but at the moment, none have been universally accepted. The presence of focal [18F]FDG uptake, with dotted configuration, is a good diagnostic marker showing high specificity and positive predictive value [71]. Conversely, mild and homogeneous uptake is usually observed in non-infected graft, due to foreign body reaction and chronic sterile inflammation [82], but it could also be consistent with low-grade infection. The exact location of the focal uptake, its distribution and intensity should be recorded as well as [18F]FDG uptake in regional lymph nodes. Also the evaluation of graft border at co-registered CT scan is an important aspect, being the combination of irregular borders and focal [18F]FDG uptake, highly predictive of VGI [71].
Qualitative analysis: as always, the evaluation of [18F]FDG intensity derives from a qualitative assessment and some visual grading scales have been proposed [75, 83–85]. In grade 0, [18F]FDG uptake is similar to the background; in grade I, [18F]FDG uptake is similar to inactive muscles and fat (low [18F]FDG uptake); in grade II, the uptake is higher than those observed in inactive muscles and fat (moderate [18F]FDG uptake); in grade III, it is lower than the physiological urinary uptake by the bladder (strong [18F]FDG uptake) and in grade IV, it is comparable with the physiological urinary uptake by the bladder (very strong [18F]FDG uptake) [85]. Focal uptake, with an intensity grade > II is suspicious of VGI [84].
Semi-quantitative analysis: several authors explored the possible role of SUVmax or target-to-background ratio (T/B), normalizing the uptake for background activity in the liver or blood pool (usually in the cava vein) [86]. However, no definitive cut-off able to discriminate between an infection and sterile inflammation has been identified yet.
In conclusion, the clinical use of [18F]FDG PET/CT imaging in the assessment of suspected VGI has increased worldwide. At the moment, the evaluation of uptake pattern (focal vs diffused) seems to be a good diagnostic marker able to distinguish between VGI and sterile inflammation; however, several pitfalls must be considered when interpreting the scan.
Future perspectives and new trends
Interesting and preliminary results can be obtained with [18F]FDG-labelled autologous leukocytes. Such an approach can improve the sensitivity of PET imaging in this scenario, identifying the site and extent of the infection. However, this technique is not free from pitfalls and at the moment, there are only limited studies in this field [87, 88]. Also, in the era of artificial intelligence application, textural analysis has shown preliminary potentialities in the characterization of the [18F]FDG uptake heterogeneity as a feasible and promising tool to diagnose VGI that may improve the clinical decision-making process [89].
Role of [18F]FDG-PET/CT in infectious endocarditis, cardiovascular implantable electronic device infections, left ventricular assist device associated, infections and cardiac sarcoidosis
In this document, we provide a standard for [18F]FDG PET/(diagnostic) CT imaging procedures in IE, CIED, including LVAD and CS, which is based on the most recent EANM, ESC and EHRA guideline/Procedural recommendations. This standard should be applied in clinical practice and integrated in clinical trials for optimal procedural standardization. The needs of proper hybrid equipment, dedicated imaging acquisition protocols, specific expertise for imaging reading and imaging interpretations in this field are discussed, emphasizing the need of a specific reference framework within a Cardiovascular Multidisciplinary Team Approach. Cardiovascular infections are burdened by high morbidity and mortality. The spectra of micro-organisms causing cardiovascular infections are very broad and may involve various components of the native structure of the heart as well as implanted devices. Since most matrix-embedded bacteria resist to medications and host defences, the surgery represents the only chance to definitely eradicate the infection, but a timely diagnosis is of paramount importance. However, cardiovascular infection diagnosis is challenging, and requires a multimodal approach and a multidisciplinary team. Indeed, the introduction of a team of specialists has radically improved the management of IE, CIEDs infections, including Left Ventricular Assist Device Associated (LVADs) Infections and several classifications, algorithms and flow-charts are currently applied in clinical practice [90].
Nuclear medicine has gained a great relevance in the diagnosis and follow-up of these conditions as demonstrated by the introduction, in 2015, of [18F]FDG PET/CT and radiolabeled WBC SPECT/CT, in the diagnostic algorithm of IE, CIEDs and LVADs infections [90]. In general, whole body [18F]FDG PET/CT, provides information not only about the presence of local infection and its extent, but also on the identification of extracardiac manifestations and portal of entry (POE), thus contributing to a better prognostic assessment and to the selection of the most suitable medical strategy [91]. [18F]FDG PET/CT diagnostic performances can be improved when it is used together with CTA since it is able to identify higher number of lesions, to provide additional meaningful information about cardiac valves and to better distinguish infection limited to the pocket or leads from a more severe extented infection in patients with suspected CIED infection [92]. Using this approach, it is possible to reduce the number of doubtful scans and to provide a more accurate classification of patients compared to the Duke criteria [93–95].
According to literature, sensitivity and specificity of [18F]FDG PET/CT in prosthetic valve endocarditis (PVE) are 73–100% and 71–100%, respectively. [18F]FDG PET/CT improved the sensitivity of the modified Duke criteria from 52–70% to 91–97% [96] by reducing the number of possible prosthetic valve endocarditis (PVE) cases [94, 97–102]. In native valve endocarditis (NVE) diagnosis, [18F]FDG PET/CT has a relatively limited role due to its low sensitivity which is hampered by both IE pathophysiology and technical factors [103]. Nonetheless, patients with NVE may benefit from [18F]FDG PET/CT since it may successfully identify extracardiac manifestations—mainly embolic stroke or septic embolization to bone, spleen or kidneys—that are reported in a significant proportion of patients with both NVE and PVE.
In patients with suspected CIED infection, [18F]FDG PET/CT shows very high diagnostic accuracy in detecting pocket/generator infection (sensitivity = 93%, specificity = 98%), in which mild inflammatory changes after device implantation usually do not extend beyond 6 weeks. In cases of lead-related IE, [18F]FDG PET/CT is highly specific (88%) with low sensitivity (65%), because small vegetation(s)—often under the spatial resolution of the system—are characterized by low-metabolic activity [104–106].
In LVAD infections, [18F]FDG PET/CT allows precise anatomic location and accurate extent of a suspected infection [107] being highly accurate, in particular, for the assessment of overall device—infections (pooled sensitivity and specificity of 95% and 91%, respectively). Similar performance has been reported for pump/pocket infections (pooled sensitivity and specificity of 97% and 93%, respectively), while when only assessing the driveline, [18F]FDG PET/CT pooled sensitivity and specificity increased (97% and 99%, respectively).
The role of [18F]FDG PET/CT for the diagnosis of extracardiac sarcoidosis is supported by a large amount of literature (see also the section on systemic sarcoidosis) [40]. The assessment of CS has been included in international guidelines [40, 108] and should be recommended in all the cases when a patient is scanned with [18F]FDG PET/CT for systemic sarcoidosis. There is no distinct pattern of [18F]FDG uptake that is pathognomonic for CS, though focal or focal on diffused uptake is suggestive of the disorder. [18F]FDG PET/CT sensitivity and specificity for CS have been reported at 89 and 78%, respectively. Using semi-quantitative analysis, it is possible to reaching a sensitivity of 97.3% and a specificity of 83.6% for the diagnosis of CS [109].
Recently, procedural recommendations on PET/CT imaging in inflammatory-, infective-, infiltrative, and innervation (4Is)-related cardiovascular disease have been published, suggesting a classification of [18F]FDG findings to facilitate imaging interpretation (Tables 1 and 2) [110] .
Table 1.
Classification for [18F]FDG findings suggested by the EANM Procedural recommendations for PET/CT imaging of IE and CIEDs infections
| Typical findings | Atypical findings |
|---|---|
|
Presence of focal, heterogenous, valvular/peri-valvular [18F]FDG uptake persisting on NAC images and corresponding to an area of suspected infection on echocardiography or CTA High [18F]FDG signal in the absence of prior use of surgical adhesives Presence of focal [18F]FDG uptake in organs with low background uptake consistent with septic embolism, mycotic aneurysms, or the portal of entry (POE) |
Diffused, homogeneous, valvular [18F]FDG uptake that is absent on NAC images Low [18F]FDG signal |
Table 2.
Classification for [18F]FDG findings suggested by the EANM Procedural recommendations for PET/CT imaging of CS
Modified from [110]
| [18F]FDG PET/CT finding | Perfusion finding (SPECT or PET) | CMR findings | Final interpretation |
|---|---|---|---|
| No [18F]FDG uptake | Normal | No LGE | No CS (< 10%) |
| Diffused [18F]FDG uptake (homogeneous) | Normal | No LGE | Uptake most likely due to suboptimal patient preparation |
| Isolated lateral wall [18F]FDG uptake | Normal | No LGE | Uptake possibly related to a normal variant |
| No [18F]FDG uptake | Small perfusion defect | One focal area of LGE (alternative diagnosis are most likely) | Possible CS (50–90%) |
| Focal area of [18F]FDG uptake | Normal | ||
| No [18F]FDG uptake | Multiple non-contiguous areas of perfusion defect | Typical LGE | Probable CS (50–90%) |
| Focal or focal on diffused[18F]FDG uptake | Resting perfusion defect | ||
| Focal area of [18F]FDG uptake + extracardiac findings | Normal | Typical LGE | Active cardiac sarcoidosis (> 90%) |
| Focal on diffused[18F]FDG uptake | Perfusion defect | Typical LGE | Active inflammation with scar in the same location with either diffused inflammation or suboptimal preparation |
| Focal area of [18F]FDG uptake in an area of normal perfusion | Perfusion defect | Typical LGE | Presence of both inactive scar and inflammation in different segments of the myocardium or inactive scar and false-positive physiological [18F]FDG uptake |
LGE late gadolinium enhancement
Clinical indications
Suspected PVE, and/or septic embolisms, spread of infection, and POE in both PVE and NVE;
Suspected CIED infection and/or, extent of the disease burden in a proven CIED infection, and/or positive blood culture in a patient with CIED;
Suspected infection of LVAD, and/or evaluation of the extent of infection of LVAD, and/or positive blood culture in a patient with LVAD;
Suspicion of cardiac sarcoidosis (Fig. 3);
Monitoring of treatment in patients with established cardiac sarcoidosis.
Fig. 3.
A male patient with a recent history of ischaemic stroke, with a suspicion for cardiac sarcoidosis. A MR showed a late enhancement in the septum, while B at PET images, a high and diffused FDG uptake was found in the anterior and lateral wall of the left ventricle. Moreover, in the lateral wall a focal FDG uptake was found. The pattern of focal-on-diffused uptake was compatible with active sarcoidosis
Interpretation criteria for IE
Qualitative analysis:
Location of the [18F]FDG uptake at the valve should be described as intravalvular (in the leaflets), valvular (following the supporting structure of the valve) or perivalvular (next to the valve). A perivalvular signal is in favour of infection, even if infection cannot be excluded in case of intravalvular or valvular [18F]FDG signal. Extra-cardiac site(s) of [18F]FDG uptake should be described (spleen, liver, lungs, kidneys, intervertebral disks).
Pattern of the [18F]FDG uptake at the valve should be described as focal or diffused and homogeneous or heterogeneous. Focal and heterogeneous uptake is consistent with IE.
Intensity of the [18F]FDG uptake at the valve: the chance of infection increases with the intensity of the [18F]FDG signal at the native valve/prosthesis.
Semi-quantitative analysis: Several indices have been evaluated to quantify the intensity of [18F]FDG signal in IE. As for other domains, SUVmax is the easiest parameter to measure [18F]FDG uptake in the valvular region.
Interpretation criteria for CIEDs and LVADs infections
Qualitative analysis: relies on the evaluation of:
Presence/absence of the [18F]FDG uptake.
Persistency on NAC images: the presence and location of the signal and its persistency on NAC PET images should be described according to the signal intensity and its location. A focal or linear [18F]FDG signal located on or alongside a lead on CT and persisting on NAC images is in favour of infection. Late PET acquisitions might prove particularly useful in case of persistent high blood signal on PET images acquired at 1 h p.i.
-
Location:
In CIED infections the presence of [18F]FDG uptake should be described as pertinent:- To generator/pocket (superficial or deep).
- To the leads specifying if it involves the intravascular or the intra-cardiac portion of the lead(s) or both. [18F]FDG uptake along pacing leads appearing as multiple hot spots, in particular in the same location as mobile elements on echocardiography and in association with septic pulmonary emboli, is highly suggestive of pacing lead infection [111]. The contrast between [18F]FDG signal along the pacing lead and residual blood signal is usually improved with delayed PET acquisition (3 h p.i) [112]. A focal [18F]FDG signal is often present at the point of entry of the lead into the subclavian vein that resembles a focal inflammation.
- To cardiac valve.
- To the pericardium.
-
To other sites.In LVADs infections, the presence of [18F]FDG uptake should be assessed separately and described as pertinent to the five components of the device or to other sites:
- Driveline exit site.
- Driveline within the subcutaneous tissues.
- Pump.
- Inflow cannula.
- Outflow cannula.
- To other sites.
- Pattern of the [18F]FDG uptake: focal or linear.
- Intensity of [18F]FDG uptake: moderate [18F]FDG uptake in relation to post-operative residual inflammation can be found up to 2 months after CIED implantation but is usually of lower intensity than in the case of infection.
Semi-quantitative analysis: semi-quantitative ratio of maximum activity concentration of the pocket device over mean count rate of lung parenchyma [111] or normalization of SUVmax around the CIEDs to the mean hepatic or blood pool activity might help in differentiating mild postoperative residual inflammation up to 2 months after device implantation versus infection [113].
Interpretation criteria for CS
Qualitative analysis: in addition to the parameters previously described for the other clinical indications (location of [18F]FDG uptake at the left or right cameras, pattern of the uptake, extent and intensity especially in comparison with prior assessments), the classification of [18F]FDG findings proposed by recent EANM guidelines, and summarized in Table 2, could be helpful [110].
Semi-quantitative analysis: SUVmax on [18F]FDG PET/CT seems to be the only independent predictor among clinical and imaging variables for diagnosing CS and for therapy efficacy assessment [114, 115].
Future perspectives
Although still limited in clinical practice PET/MR imaging could have a great potential in cardiovascular infectious and inflammatory diseases, in particular in CS and myocarditis [116]. Given the possible interaction between ferromagnetic material and magnetic field, IE and device infection are not among the major indications for PET/MR. Particular caution is warranted in case of MR-compatible, implanted material such as stents or sternal wires, as these can lead to incorrect attenuation maps and thus to incorrect attenuation-corrected PET data [117]. Several new tailored radiopharmaceuticals are being developed for imaging myocarditis and pericarditis, where the role of [18F]FDG PET/CT is limited, but at the moment are still in preclinical phases.
Role of 18F-FDG PET/CT in retroperitoneal fibrosis
Retroperitoneal fibrosis is a rare disease characterized by the presence of a retroperitoneal tissue, consisting of chronic inflammation and marked fibrosis, which often entraps the ureters or other abdominal organs [118]. The idiopathic form of RF accounts for more than two thirds of cases, with the rest being secondary to other factors (neoplasms, infections, trauma, radiotherapy, surgery, and use of certain drugs).
The gold standard for the diagnosis of RF is still histopathology with its typical inflammatory infiltrate (mononuclear cells, chronic inflammation, fibroblast proliferation and excessive extracellular matrix) [119]. CE-CT allows a better delineation of periaortitis and its extension to adjacent structures [120]. The majority of available studies suggests that [18F]FDG PET/CT could be useful in evaluating the disease extent and activity at the time of diagnosis [48]. An over-expression of glucose transporter (GLUT-1, GLUT-3) and an increased hexokinase type II (HK-II) activity stimulated by cytokines or mutagens lead to an incremental glucose consumption, and a consequent 18F]FDG uptake, by inflammatory cells [121]. Idiopathic RF could be included under the umbrella term of chronic periaortitis, along with inflammatory abdominal aortic aneurysms and perianeurysmal retroperitoneal fibrosis [119, 121]. The availability of imaging techniques has provided non-invasive and reliable methods of diagnosis and follow-up in patients with chronic periaortitis [50]. In idiopathic RF, the aorta is not dilated and the surrounding fibro-inflammatory tissue generally entraps abdominal organs such as ureters; in inflammatory abdominal aortic aneurysms, the tissue develops around a dilated aorta, but does not cause obstructions; finally, in perianeurysmal RF, the fibro-inflammatory tissue involves an inflammatory aneurysm and generally entraps the adjacent organs [121]. [18F]FDG PET/CT may be useful in diagnosing RPF. Vascular uptake at [18F]FDG PET/CT was observed in the abdominal aorta and/or iliac artery and thoracic aorta and/or its branches in 100 and 43% of patients with RF, respectively, but none in unaffected controls [122]. With regard to the use of PET in following up patients with RF, PET findings normalized in six out of seven patients after immunosuppressive therapy, whereas CT showed a residual mass in all patients, probably representing inactive residual tissue [123]. These data suggest that PET may be more specific than CT in defining disease activity in treated RF.
Clinical indications
Diagnosis of suspected RF (evaluation of disease extent and disease activity) in patients with elevated CRP and/or ESR [118];
Assessment of exacerbation and progression of disease, highlighting new foci of inflammation not documented at morphological imaging [124];
Evaluation of disease activity during/after treatment in patients with normal inflammatory markers and stable residual mass on repeated MDCT [125];
Evaluation of correct time to proceed to ureteral stent removal [126];
Discrimination between active and residual fibrotic tissue [27].
Interpretation criteria
Qualitative analysis: the evaluation of the metabolic activity of RF with [18F]FDG PET/CT can be done by visual analysis. It is usually performed in a four-point graded scale, based on the tissue-to-liver [18F]FDG uptake ratio (0: no uptake, 1: uptake less than that of the liver, 2: uptake equal to that of the liver, and 3: uptake greater than that of the liver); a visual score above 1 is usually considered a positive criterion for active RPF [27, 119].
Semi-quantitative analysis: SUVmax values and T/B could also be used. In the recent past standardization of [18F]FDG PET/CT studies using semi-quantitative analysis to achieve interchangeability in multicentric trials was recommended [127].
Future perspectives and new trends
The [18F]FDG PET/MRI is a multimodality imaging that combines the high sensitivity of [18F]FDG PET/CT in detecting vascular/perivascular inflammation with the high specificity. PET/MRI is a feasible tool in RF combining metabolic imaging ([18F]FDG uptake) of inflammatory cells with high signal intensities at DWI, T2-W and T1-W images (most probably due to edema-related structural tissue changes and hypercellularity in active fase of disease [128].
Role of [18F]FDG PET/CT in fever of unknown origin
FUO is the acronymous of fever of unknown origin and refers to a prolonged febrile illness without an established etiology despite intensive evaluation and diagnostic testing [129]. Its definition has changed several times since 1961 to the present day. Petersdorf and Beeson defined FUO as an illness of more than 3 weeks’ duration with fever higher than 38.3 °C (101 °F) on several occasions and diagnosis uncertain after 1 week of study in the hospital [130]. Actually, current definition of FUO is as follows:
Temperature ≥ 38.3 °C (101 °F) on at least two occasions;
Duration of illness ≥ 3 weeks or multiple febrile episodes in ≥ 3 weeks;
Not immunocompromised (defined as neutropenia for at least 1 week in the 3 months before the start of the fever; known HIV-infection; known hypogammaglobulinemia or use of 10-mg prednisone or equivalent for at least 2 weeks in the 3 months before the start of fever);
Uncertain diagnosis despite thorough history-taking, physical examination, and the following investigations: ESR or CRP, haemoglobin, platelet count, leukocyte count and differentiation, electrolytes, creatinine, total serum protein, protein electrophoresis, alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase, lactate dehydrogenase, creatine kinase, ferritin, antinuclear antibodies, rheumatoid factor, microscopic urinalysis, three blood cultures, urine culture, chest x-ray, abdominal ultrasonography, and tuberculin skin test or interferon gamma release assay [131].
Causes of FUO include more than 200 categories [132], but nowadays, the four major causes are represented by infections, malignancies (so-called “pyrogenic cancers” such as lymphoma, leukaemia, colon cancers, pancreatic cancer, sarcoma and brain tumours), noninfectious inflammatory diseases (NIID; which includes autoimmune, autoinflammatory and granulomatous disease, and vasculitides), and miscellaneous causes [129, 132–134]. Approximately, 30% of patients with FUO may eventually be diagnosed with a rheumatologic disease [134]. Similar to FUO, the etiology of inflammation of unknown origin (IUO) may vary from a self-limited disease to a malignancy. IUO is defined as an illness of at least 3 weeks’ duration with signs of inflammation but temperatures not > 38.3 °C on more than three occasions; CRP > 30 mg/L and/or ESR > age/2 in men or (age + 10)/2 in women on more than three occasions; and uncertain diagnosis despite appropriate investigations after at least three days in hospital or three outpatient visits [132]. [18F]FDG PET/CT plays an important role in the diagnosis of patient with FUO and IUO, because [18F]FDG accumulates both in inflammatory cells (granulocytes, lymphocytes, monocytes/macrophages) and neoplastic cells with increased rate of glycolysis. A search for potential diagnostic clues (PDCs) in a complete and repeated history-taking, physical examination, and the essential investigations are important for diagnosing FUO. PDCs are defined as all localizing signs, symptoms, and abnormalities potentially indicating a certain diagnosis. When PDCs are absent [18F]FDG PET/CT should be performed to guide additional diagnostic tests [135, 136].
The diagnosis of patients with FUO or IUO represents a challenging medical problem. Timely identification and precise localization of the causes of FUO (and IUO) is critical for decision on further diagnostic procedures and the initiation of appropriate treatment [137]. In this setting, [18F]FDG PET/CT can be useful in identifying origins of FUO (or IUO) with a whole-body imaging characterized by high resolution, high sensitivity in low-grade infections and in early stage of inflammations, improved anatomical resolution (with CT component) and a relative low radiation exposure (about 15 mSv rather than 20–25 mSv of a diagnostic CT scan) [129, 135, 138, 139]. Takeuchi et al. found that after a series of unsuccessful investigations for fever workup, patients with negative PET/CT results have approximately 6 times higher than average chance of spontaneous remission than those with positive PET/CT results. These findings suggest that a negative PET/CT result can be a good predictor of favourable prognosis in patients with undiagnosed classic FUO after a series of unsuccessful investigations [140]. In previous years, before PET imaging expanded, other radiopharmaceutcals were routinely used in FUO such as: 67 Ga-citrate, radiolabeled white blood cells (with 111In-Oxine or 99mTc-HMPAO), anti-granulocytes antibodies, each with their own advantages and disadvantages. Of these, 99mTc-HMPAO-WBC scintigraphy remains a valid alternative to [18F]FDG, and, it has been suggested, in particular, in these patients with a high pre-test probability of infection (clinically and laboratory evaluated) [141]. By contrast, [18F]FDG is certainly more indicated in all cases with a low pre-test probability of infection [141].
Most existing meta-analyses, focussing on [18F]FDG PET/CT sensitivity in FUO (or IUO), are limited by (a) different definitions of FUO, (b) observational data involving small samples, (c) immunocompromised patients enrolled, (d) retrospective studies, (e) outdated case definitions, (f) poor generalizability, (g) differences in [18F]FDG PET/CT timing, and also (h) the absence of a reference standard for the investigation of FUO (or IUO) to enable estimates of diagnostic accuracy [129, 139]. Two meta-analyses reported a sensitivity of 85% (95% CI 81–88%; 15 studies) and of 98% (95% CI 94–99%; 9 studies), respectively, while Bharucha et al. in 2007 reported an achieved diagnosis in 56% of patients, consistent with the previous concept of “overall helpfulness” (54%) [139]. Besson et al. in 2016 claimed that sensitivity, specificity and derived parameters (positive predictive value, negative predictive value, likelihood ratios) are potentially unsuitable in FUO (or IUO) [142]. Pijl et al. recently evaluated the diagnostic utility of [18F]FDG PET/CT in intensive care patients with bloodstream infection. In this setting, [18F]FDG PET/CT is very important because patients are in critical clinical conditions, poorly collaborating and cannot wait for a WBC scan. [18F]FDG allowed to identify an infection focus with a a sensitivity of 90.9% and a specificity of 87.5%, leading to a change in clinical management in about 47% of cases [143].
Clinical indications
Evaluation of unknown sites of inflammation (particularly in patients with low probability of infection);
Evaluation of unknown sites of neoplastic disease as causes of systemic symptoms;
Guide biopsy;
Assessment of therapeutic efficacy;
Assessment of prognostic value [140].
Once the cause of FUO (or IUO) is correctly identified (vasculitis, endocarditis, etc.), the role and timing of [18F]FDG PET/CT imaging in the management of patient will be that corresponding to the specific pathology.
Interpretation criteria
Qualitative analysis is based on the identification of all sites of pathological radiopharmaceutical uptake towards which the diagnostic path should be addressed. False-negative results were reported in diseases such as systemic lupus erythematosus, cytomegalovirus infection, toxoplasmosis, urinary infection, septicaemia, pyelonephritis and Crohn’s disease [144] and in sites of para-physiological radiopharmaceutical uptake.
Future perspectives and new trends
Since FUO is very often an atypical presentation of a specific disease, the possibility of using new acquisition modalities, new tracers or PET/MRI overlaps with what is described in the specific paragraphs of the other pathologies.
Role of [18F]FDG PET/CT in sarcoidosis and tuberculosis
Sarcoidosis and tuberculosis are granulomatous diseases with heterogeneous clinical manifestations. The typical lesion of sarcoidosis is granuloma with non-caseous necrosis, while caseous necrosis granuloma is characteristic of TBC. Lung is the most commonly involved organ; however, in patients with sarcoidosis and tuberculosis any organ or tissue can be involved. Tuberculosis is due to mycobacterium tuberculosis while the exact etiology of sarcoidosis is yet to be determined. Sarcoidosis might be the result of an exaggerated immune reaction after exposure to unidentified antigens in subjects who are genetically susceptible [145]. The clinical course of sarcoidosis is variable. The overall prognosis is generally good. In about half of patients, disease spontaneously resolves within 2–5 years. In some cases, it progresses to pulmonary fibrosis for poor drug response without a known reason (about 10–15%). Usually, an efficient immune response eliminates tuberculosis bacilli. In some patients, a latent infection may run asymptomatic with the restriction of non-replicating mycobacteria bacilli inside granulomas. Only in a small proportion of subjects, the tuberculosis infection clinically appears with active inflammation and symptoms [146]. Severity of these diseases ranges from a completely asymptomatic clinical picture to a massive pulmonary involvement leading to important respiratory failure. The most common symptoms are a persistent cough and fatigue. All these symptoms are quite non-specific and can delay diagnosis. Tuberculosis remains a global emergency in developing countries. India is the most affected region, followed by China and Indonesia [147]. The pulmonary radiological signs of sarcoidosis and tuberculosis are quite heterogeneous. A typical sign of sarcoidosis is the presence of diffused micro-nodular pulmonary infiltration with lymphatic distribution often associated with intra and interlobular septal thickening [148]. Another radiological manifestation is the presence of bilateral peri-bronco-vascular and peri-hilar consolidations. Lung and lymph nodes (hilar and mediastinal) are usually both involved. In pulmonary tuberculosis, we find radiological signs as parenchymal consolidations in any lobe (also multilobar consolidations), ground glass opacities, pleural thickening and pleural effusion, micro-nodular infiltration, parenchymal scar (calcified or not) and hilar and mediastinal lymphadenopathy [149–151]. Fibrosis, bronchiectasis, parenchymal cavities, volume loss are outcomes of a possible evolution. Extra-thoracic involvement in sarcoidosis is present in more than 30% of cases and it can be the major or the only disease manifestations. Skin involvement is the most common extra-thoracic manifestation, and it can manifest as skin nodules, erythema nodosum, rashes and plaques [145, 148]. Other common manifestations are enlarged peripheral lymph nodes, arthritis and uveitis. Liver, spleen and retroperitoneal lymph nodes are frequent involved. Other gastro-intestinal localizations are rare. Sarcoidosis can involve cardiac tissue (a potentially life-threatening localization) with clinical manifestations that include atrioventricular block, ventricular arrhythmias, congestive heart failure, sudden death and consequences of impairment in sympathetic nerve activity [152, 153]. The left ventricle is the most commonly involved chamber. Extra-pulmonary tuberculosis occurs in about 20% of patients. Musculoskeletal involvement is quite frequent with prevalent involvement of the spine (in approximately 50% of cases) [152]. The most common form is the spondylodiscitis (Pott’s disease) usually localized in the lower dorsal or upper lumbar spine [149]. Multifocal bone involvement is not rare. Neurologic involvement in sarcoidosis e tuberculosis is not common. The most frequently affected sites are the cranial nerves, meninges and brain parenchyma. Symptoms related to neurologic involvement are peripheral pain, dysesthesia and various autonomic disturbances. The main consequences of chronic granulomatous diseases are fibrosis and the related damage to the involved organs. The goals in the management of these patients are to prevent or limit organ damage, relieve symptoms and improve the quality of life [151–154]. [18F]FDG PET/CT is not routinely included in the workup for granulomatous diseases. Chest radiography is the first diagnostic modality. The Scadding criteria based on chest radiography that classify sarcoidosis in five stages correlates poorly with symptom severity, extra-pulmonary disease, pulmonary function tests and prognosis [145]. The high-resolution CT is the elective method to study pulmonary parenchyma morphology. Anyhow, radiological techniques do not correlate well with the state of inflammatory activity [155]. Biochemical markers (ACE, sIL-2R, neopterin) are useful but negative serologic findings do not exclude the presence of active disease [145, 156–158]. Symptoms like coughing and dyspnea are not specific. [18F]FDG PET/CT is a sensitive technique to assess the inflammatory activity in sarcoidosis and tuberculosis by detecting and quantifying the level of inflammatory foci in pulmonary parenchyma and elsewhere in the body. Sarcoidosis and tubercular active lesions show an increased [18F]FDG uptake [157, 159–163]. [18F]FDG uptake reflects the high glycolytic activity of activated macrophages and lymphocytes, prominent in granulomatous inflammatory processes [164, 165]. End-stage fibrosis may show anatomical abnormalities but the disease itself has become inactive [156]. [18F]FDG PET/CT scan could play an important role in tuberculosis patients with negative sputum or in sarcoidosis patients with low level of serological inflammatory markers (ACE, sIL-2R, neopterin). Several studies suggest [18F]FDG PET/CT to be superior in evaluating disease activity compared to serological inflammatory markers, bronchoalveolar lavage and to conventional radiological techniques [145, 156]. [18F]FDG PET/CT is an effective procedure to define the extent of disease accurately and it is able to detect more sarcoidosis or tuberculosis lesions than any other routine radiological technique. A whole-body evaluation in a single examination is not possible with other imaging modalities. Pulmonary and extra-pulmonary involvements (skin, bone, bone marrow, SNC, myocardium …) are simultaneously evaluated, with time and cost-saving implications [149, 157, 166, 167]. Bone biopsy is not necessary in patients with proven disease and typical bone involvement based on [18F]FDG PET/CT or MRI [168]. [18F]FDG PET/CT may be superior to CT or MRI for detecting bone and bone marrow sarcoidosis [169]. An accurate assessment of the extent of the disease allows a better prognostic evaluation in these patients. [18F]FDG PET/CT is an excellent tool to demonstrate hidden or atypical lesions [157, 170] and to identify the most accessible site for biopsy (cutaneous nodules, superficial lymph nodes, conjunctival lesions, lacrimal or parotid gland lesions) for a rapid diagnosis [156, 161]. [18F]FDG PET/CT is probably the most important tool for monitoring the treatment response and for planning clinical management [158, 160, 163, 166, 167, 170, 171]. During treatment, sometimes granulomatous lesions do not decrease in size also for a long time. Radiological techniques may be unable to establish whether the disease is in remission or not. [18F]FDG uptake in granulomas decreases after proper treatment [172] and it could be predictive for the response to treatment. [18F]FDG PET/CT is useful in therapeutic decision-making in patient refractory to first line therapy [173] and it allows to identify residual active disease versus scarring after therapy [154, 159, 174]. Non-responder patients should be treated with further therapy [154, 158, 161]. Patients with residual ongoing inflammatory sarcoidosis might benefit from a change or increase of the anti-inflammatory therapy. Standard treatment duration for tuberculosis is about 6 months. Early treatment response assessment may allow a rapid identification of patients who respond poorly to treatment [146]. After 1 month of anti-tuberculosis treatment, a decrease of approximately one-third in SUVmax of pulmonary and extra-pulmonary lesions is indicative of a good response [149, 175]. In patients with radiographic lesions suggestive of old healed tuberculosis and with suspected recurrence of the disease, [18F]FDG PET/CT can evaluate the metabolic status of lesions to identify patient at high risk of disease recurrence [146, 174, 176]. Atypical manifestations and imaging findings can make diagnosis challenging. [18F]FDG PET/CT can direct the diagnostic hypothesis in patients with non-specific symptoms [154].
Clinical indications
Evaluation of disease activity;
Differential diagnosis between reversible granuloma from irreversible fibrosis;
Diagnosis of occult disease;
Evaluation of disease extent in initial studies;
Evaluation of prognostic assessment;
Evaluation of treatment response;
Evaluation of early treatment response;
Identification of most suitable site for biopsy;
Performing diagnostic hypothesis in patients with non-specific symptoms
Interpretation criteria
[18F]FDG uptake of granulomatous lesions correlates with disease activity [151, 157]. The immune status of the host or the presence of comorbidity can affect the SUVmax of lesions.
Qualitative analysis:
Right para-tracheal and bilateral hilar lymph nodes involvement: increased [18F]FDG uptake defines the so-called lambda sign (λ), an imaging finding suggestive of active sarcoidosis, similar to the lambda sign of 67Ga-citrate scintigraphy, an investigation no longer carried out [177, 178]. [18F]FDG PET/CT is superior to 67Ga-citrate scintigraphy in the evaluation of active sarcoidosis [179]. The presence of lambda sign (λ) whether or not combined with lung parenchymal active disease, supports the likelihood of sarcoidosis [156].
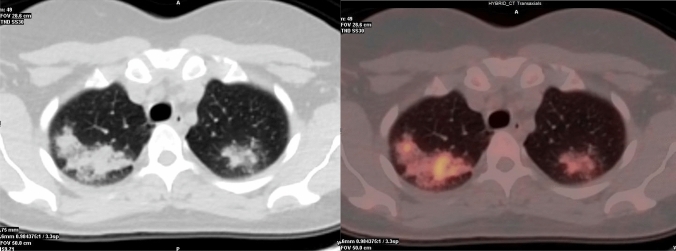
Pulmonary involvement with cavitation: cavitation is the radiological hallmark of post-primary tuberculosis PTB and it is radiographically evident in 20–45% of patients. Cavitation may progress to endobronchial spread and results in a typical ‘tree in bud’ pattern in addition to cavitation. These radiological markers, indicative of active tuberculosis [149] usually show increased [18F]FDG uptake. Radiological features indicative of inactive tuberculosis are thin-walled smooth cavities, fibrosis, and parenchymal, nodal, or pleural calcifications [149]. In systemic tuberculosis, any site of active disease shows elevated [18F]FDG uptake (Fig. 4).
Pulmonary miliary involvement: in both sarcoidosis and tuberculosis, it is possible to detect a pulmonary miliary pattern that consist of innumerable micro-nodules (1–3 mm) randomly distributed throughout both lungs [149]. In these patients, it is possible to see a mild diffused inhomogeneous pulmonary uptake. In active disease, micro-nodules may coalesce to form focal or diffused consolidation with more intense [18F]FDG uptake.
Pleural effusion involvement: in case of pleural effusion, it is possible to see a smooth thickening of visceral and parietal pleura with diffusedly intense [18F]FDG uptake similar to pleural mesothelioma [149, 150].
Skin involvement: moderate/intense radiopharmaceutical uptake is detectable on active cutaneous lesions with a focal or diffused distribution pattern related to the type of lesion (skin nodules, erythema nodosum, rashes, plaques …).
Lacrimal and salivary glands involvement: another typical imaging finding suggestive of active sarcoidosis is the so-called “panda sign”, an uptake pattern characterized by increased and symmetric radiopharmaceutical accumulation in the grossly enlarged lacrimal and parotid glands. The radiopharmaceutical uptake of the submandibular glands is only minimally increased [178].
Myocardial involvement: [18F]FDG PET/CT is a sensitive test to evaluate the myocardial involvement [180]. The elective approach should consider performing a perfusion and an inflammation evaluation at the same time. Dual perfusion-inflammation PET/CT scan seems to be the elective way but this approach can only be possible in a medical center equipped with cyclotron. The combined interpretation of the two procedures (perfusion and inflammation [18F]FDG PET/CT scan) allows evaluating the presence of active myocardial localizations and their response to treatment. In cardiac sarcoidosis involvement, perfusion is normal or decreased and in the same involved area, [18F]FDG uptake is increased. Fibrosis usually demonstrates reduced perfusion and [18F]FDG uptake [154]. However, besides [18F]FDG PET/CT for cardiac involvement, a myocardial perfusion scan should be recommended [156].
Neurological involvement: granulomatous lesions may appear hypo- or hypermetabolic depending on the neurologic site or brain area involved.
Musculoskeletal involvement: usually demonstrates avid [18F]FDG activity [181] and high radiopharmaceutical uptake. Some radiological signs associated with tuberculous spondylodiscitis are: frequent localization in the posterior portion of the vertebra, bone destruction due to the appearance of areas of osteolysis, narrowing of the disc, swelling and padding of the paravertebral soft tissues, deformation of the vertebral with somatic collapse. In severe cases, deformation of the spine can occur (kyphotic or gibbus deformity). The signs of repair are represented by osteosclerosis.
Fig. 4.
Initial evaluation of a > 30-year-old woman from Argentina with FUO and recent finding of pulmonary bilateral consolidations. 18F-FDG PET/CT shows moderate/high radiotracer uptake in solid/sub-solid consolidations localized in the upper areas of both lungs. Mild 18F-FDG uptake coexists in enlarged hilar and mediastinal lymph nodes. Histopathologic evaluation establishes the diagnosis of sarcoidosis
Quantitative analysis: viable Mycobacterium tuberculosis may persist in a quiescent state for years. The detection of mild increased [18F]FDG uptake (SUVmax 1.5 or little more) in radiological features suggestive of old healed tuberculosis lesions (nodules or scars) do not necessarily represent active disease, but might reflect a state of equilibrium between the host’s immune response and the replicating bacilli [151]. Latent infection may develop to active disease. Patients with radiological features suggestive of old healed tuberculosis but with increased [18F]FDG uptake may be at higher risk of relapse of disease [149, 176]. A SUVmax above 2.0 may reflect ongoing disease even if the patient has no clinical symptoms [151]. In patients with active sarcoidosis or tuberculosis, [18F]FDG PET/CT scan shows increased radiopharmaceutical uptake in any site of disease with high level of SUVmax. Usually, tuberculous spondylodiscitis have higher SUVmax compared with pyogenic spondylodiscitis [151, 182].
Future perspectives and new trends
Future perspectives may concern the use of alternative PET/TC radiopharmaceuticals (gallium-68 citrate, choline derivatives, 18F-Fluoro-l-thymidine, 18F-Fluoromisonidazole) or a combined use of 2 radiopharmaceuticals at the same time. However, further studies are required [151, 183]. The greater availability of hybrid tomographs PET/MRI could improve the study of extra-pulmonary involvement (cardiac, hepatic, spinal, musculoskeletal, splenic, neurologic involvement). Although [18F]FDG PET/CT is not included in the standard workup for sarcoidosis and tuberculosis, it appears to be a promising and valid technique for the management of patients affected by these diseases. [18F]FDG PET/CT is useful in the initial diagnosis to assess disease extent, to valuate inflammatory activity and to assess early or final treatment response. Moreover, [18F]FDG PET/CT is able to reveal extra-pulmonary disease lesions, the most suitable site for biopsy and it can be helpful to direct the diagnosis in patients with non-typical symptoms. It is legitimate to think that a proper interpretation of both CT and PET findings (both radiological and metabolic features) is necessary to improve the accuracy of the examination.
Role of [18F]FDG PET/CT in management of invasive fungal infections and HIV-infection
IFI are common in immunosuppressed patients, particularly those with human immunodeficiency virus (HIV) infection, recipients of solid organ or hematologic stem cell transplants and oncologic patients [184]. IFI are not caused by a single organism but by a number of different species of fungi that have been generally divided into yeasts and moulds. Candida spp is the most common type of yeast whilst Aspergillus spp is the most common type of mould. Cryptococcus and Pneumocystis are more likely to be found in HIV-positive patients [185]. Immunocompromised patients are prone to develop disseminated multisite disease, with the lungs being the most common site of IFI; the dissemination to other organ systems, including the central nervous system, heart, kidney, and liver, usually occurs by a hematogenous dissemination [186]. Early diagnosis or exclusion of infection is of utmost importance for the optimal management of these patients and, considering that the sites of [18F]FDG accumulation in infectious tissue are migratory inflammatory cells, micro-organisms and granulation tissue, [18F]FDG PET/CT may be useful to identify lesions associated with early IFI, reveal the extent of the disease, and monitor the treatment response to antifungal therapy [187, 188]. Immunosuppression due to HIV-infection results in a variety of opportunistic infections or tumours. On HIV-infection, resting lymphocytes are activated and switch to glycolysis, increasing their glucose uptake by around 20-fold over 24 h. This increased cellular glucose utilization by activated lymphocytes in affected nodes can result in increased [18F]FDG uptake, quantifiable by [18F]FDG PET/CT. According to recent studies, [18F]FDG PET/CT proved useful in detecting both malignancies and infections in HIV-positive patients [187, 189–191]. Furthermore, [18F]FDG PET/CT may prove useful for evaluating the effectiveness of highly active anti-retroviral therapy (HAART) in suppressing HIV replication, but also in evaluating common side effects of treatment such as lipodystrophy and immune reconstitution inflammatory syndrome (IRIS) [189].
Clinical indications
Although no appropriateness criteria have been developed to date for [18F]FDG PET/CT in management of immunocompromised and HIV-positive patients with suspected or known IFI, some recent evidence and common expert opinions concluded that major indications are as follows [187–193]:
To detect IFI in immunocompromised patients with persistent fever when conventional CT is inconclusive;
To identify clinically occult and disseminated IFI in immunocompromised and HIV-positive patients (Fig. 5); to monitor response to IFI treatment after adequate duration of empirical therapy, and CT suggests persistent lesions, to diagnose HIV-related opportunistic infections, associated neoplasms, and Castleman’s disease, to monitor response to HAART and evaluate common side effects of treatment in HIV-positive patients (Fig. 6).
Fig. 5.
> 50-year-old woman with non-Hodgkin lymphoma, stage III, and secondary immunosuppression due to CHT and myelofibrosis. CT transaxial (A) and 18F-FDG PET/CT fused transaxial (B) imaging show mild pericardial and pleural effusion. The patient had fever without response to antibiotic therapy and pancytopenia. CT transaxial scan after contrast media injection (C) and 18F-FDG PET/CT fused transaxial (D) imaging after 6 months of corticosteroid therapy show persistent pericardial and pleural effusion and multiple pulmonary nodular lesions, later diagnosed as aspergillomas, which show intense 18F-FDG uptake. Typical Aspergillus lesion in left pulmonary hilar region, SUVmax 6.0
Fig. 6.
> 30-year-old man with HIV with fever and severe weight loss. CT transaxial (A) and 18F-FDG PET-TC fused transaxial (B) imaging show diffused and intense splenic 18F-FDG uptake (SUVmax 4.2), greater than liver. Further finding is a focal intense 18F-FDG uptake (SUVmax 9.0) localized at superior pole of the spleen, without consensual alterations at CT scan. These findings, along with mild diffused lymph nodal uptake (SUVmax 4.1), are suggestive for lymphomatous disease, but the patient was eventually diagnosed with disseminated histoplasmosis. CT transaxial (C) and 18F-FDG PET-TC fused transaxial (D) imaging after 1 month of follow-up show mild peritoneal effusion, persistent inhomogeneous and intense 18F-FDG splenic uptake, greater than liver, without focal hypermetabolic areas
Interpretation criteria
Qualitative analysis for IFI: PET images should be visually analysed by looking for increased [18F]FDG uptake, taking into consideration the pattern (focal, linear, diffused), homogeneity, intensity and relationship to areas of physiologic distribution, then PET information is compared with morphologic information obtained by previous instrumental exams, such as CT, MRI or US [188, 192]. In contrast to its use in oncology, quantitative analysis with SUV has not been validated in IFI, therefore, SUV in this field should be used with caution in clinical practice and may be used as a purely descriptive means of reporting, but the measurement should not be used to render a specific diagnosis [27, 194]. Most research articles on the subject have defined interpretation criteria for the purposes of the study, although no definitive consensus has been agreed on [195, 196]. IFI have been known to accumulate [18F]FDG and the uptake in these focal lesions was found to correspond to disease activity, making [18F]FDG PET/CT a useful tool for IFI follow-up [197].
Qualitative and semi-quantitative analysis for HIV: in HIV-positive patients it is recommended to perform qualitative visual and semi-quantitative analysis evaluating SUVmax and SUVmean, symmetry, pattern and intensity of [18F]FDG uptake in bone marrow, liver, spleen and principal nodal region. When possible, a comparison with previous [18F]FDG PET/CT exams should be performed [189, 198]. Splenic [18F]FDG uptake greater than hepatic [18F]FDG uptake is commonly observed in HIV-positive patients in the earlier stages of the disease and may indicate a lymphomatous involvement of the spleen [189]. In HIV-positive patients with subclinical neurologic dysfunction [18F]FDG PET/CT may show relative hypermetabolism in the basal ganglia, especially in the striatum, followed by late cortical and subcortical hypometabolism or globally reduced cortical 18F-FDG uptake [189]. Immune reconstitution inflammatory syndrome (IRIS) must be considered when evaluating patients undergoing HAART, showing worsening manifestations of underlying opportunistic infections (OI) or neoplasia or with abrupt and frequently atypical presentation of an occult OI or neoplasia. These patients showed increased [18F]FDG uptake in bone marrow, spleen and lymph nodes prior to HAART initiation and a persistent abnormally elevated bone marrow and spleen metabolism 4–8 weeks after HAART initiation, that reflects the inflammatory response restoration induced by therapy [189, 193, 199, 200]. Furthermore, diffused [18F]FDG uptake in subcutaneous fat could be related to HIV-associated lipodystrophy syndrome (HALS) in long-term HAART patients and could be mistaken for infectious disease [191, 201].
Remarks
In IFI, [18F]FDG PET/CT may be useful to identify lesions associated with early infection, reveal the extent of the disease, and monitor the treatment response to antifungal therapy. In HIV-infection [18F]FDG PET/CT proved useful in detecting both malignancies and infections in HIV-positive patients. Furthermore, [18F]FDG PET/CT may prove useful for evaluating the effectiveness of highly active anti-retroviral therapy (HAART) in suppressing HIV replication, but also in evaluating common side effects of treatment such as lipodystrophy and immune reconstitution inflammatory syndrome (IRIS).
Role of [18F]FDG PET/CT in SARS-CoV-2 infection
Since its beginning in December 2019 in Wuhan, Hubei province of China, the coronavirus COVID-19 (SARS-CoV-2) pandemic of zoonotic origin is affecting world public health and testing the health systems of the various countries. Coronaviruses are non-segmented, enveloped positive-sense ribonucleic acid viruses from the Coronaviridae family. This terrible global health crisis of our time has involved more than 185 countries worldwide with millions of cases and thousands of related deaths. In concordance with current evidence, the infection is mainly based on person-to-person transmission, especially via respiratory droplets, oro-faecal transmission, but also via contact with organic fluids contaminating surfaces. The clinical scenario is very different from case to case and may be mild with symptoms as fever, cough, fatigue, ageusia, shortness of breath, myalgia, anosmia and in some patients, gastrointestinal infection symptoms or much more severe; in these cases, it could lead the patient to the need for hospitalization in intensive care in consequence of complications (especially in the elderly and patients with comorbidities) such as acute respiratory distress syndrome (ARDS), cytokines storm and thromboembolism. The reverse-transcriptase polymerase chain reaction (RT-PCR) is a nuclear acid test and represents the gold standard for the diagnosis of COVID-19 infection, but false-negative results are possible and responsible of a missed diagnosis causing the spread of the epidemic. As a consequence, clinical diagnosis is an alternative and it is mainly based on exposure history, symptoms, serological tests and imaging; chest high-resolution CT represents the main imaging diagnostic tool and is the routine-preferred method for screening, diagnosis, course severity assessment, and therapy monitoring. The role of [18F]FDG PET/CT in infectious/inflammatory disease is continuously growing in the field of infectious and inflammatory diseases.
Chu et al. [202] found that the SARS-CoV-2 infection significantly upregulates inflammatory mediators. This fact has suggested the insight that [18F]FDG PET/CT may be a potential imaging tool for COVID-19. The study of Deng et al. who have also argued for the possible [18F]FDG PET/CT utility, as a sensitive tool to detect and monitor inflammatory diseases, such as viral pneumonia, its progression, and treatment outcomes moves in this direction [203, 204].
Clinical indications
The role of [18F]FDG PET/CT should be reflected in changes in uptake patterns and locations during virus exposure, which may be useful for monitoring the effects of treatment. Despite this insight, no high-quality evidence or guideline are currently available to consider [18F]FDG PET/CT as an integral and structured part of the COVID-19-related pneumonia diagnostic flow-chart. Joob and Wiwanitkit who are of this opinion, have highlighted that [18F]FDG PET/CT is still not recommended in infectious pneumonia, and especially have warned of the risk of disease spreading in PET departments [205]. Other authors are of the same opinion; in particular Treglia [206] highlights that an increased metabolic activity in pulmonary or lymph nodal lesions in patients with COVID-19 infection is not surprising and it is not specific since other infective/inflammatory diseases can show the same aspect. As a consequence, Treglia states that [18F]FDG PET/CT should not be recommended for the evaluation of patients with known or suspected COVID-19 infection remaining the chest CT the imaging gold standard. Moreover, so far, there are no data suggesting an added value of [18F]FDG PET/CT compared to chest CT in the management or outcome of patients with COVID-19 infection and the hypothesis that [18F]FDG PET/CT could be useful in this setting is excessive and not demonstrated.
Interpretation criteria
CT patterns:
Peripheral ground-glass opacities and/or consolidative opacities in more than two pulmonary lobes (especially involving both lungs); during the disease progression, the lesions increase and spread, the inflammatory exudation, consolidation, and density increase, and other signs like pulmonary vascular shadow thickening, air bronchi sign, paving stone sign, interlobular septal thickening also called “crazy paving” and pleural effusion appear. A scoring system evaluation as the CO-RADS categorical CT assessment scheme for patients suspected of having COVID-19 pneumonia is suggested [207]; it consists of six scores: 0 non interpretable (scan technically insufficient for assigning a score), 1 very low (normal or noninfectious), 2 low (typical for other infections but not COVID-19), 3 equivocal/unsure (features compatible with COVID-19 but also other diseases), 4 high (suspected COVID-19), 5 very high (typical for COVID-19), 6 proven (RT-PCR positive for SARS-CoV-2). Despite the CT component of hybrid imaging sessions used for attenuation correction show a lower diagnostic quality not equivalent to that of dedicated chest CT (in particular a lack of deep inspiration can contribute to higher density in the posterior lung fields), it could be sufficient to diagnose or suspect COVID-19 infection.
PET patterns:
COVID-19 pulmonary infections are characterized by 18F-FDG uptake (Fig. 7) and reflects a significant inflammatory burden, similarly to that elicited by the Middle East respiratory syndrome or the H1N1 pandemic influenza virus [208–211]. After the infection, the cascade of reactions activates inflammatory cells such as monocytes, neutrophils, effector T cells and determine a local chemokines release. Activated neutrophils are heavily dependent on anaerobic glycolysis, requiring increased glucose supply; this results in high [18F]FDG uptake confirmed by high SUVmax values; granulocytes and macrophages also facilitate glucose transport under chronic conditions [212]. COVID-19 infections could be accompanied by lymphadenopathy [213]; Qin et al. [214], who moves in this direction, reported that lung lesions of patients with COVID-19 pneumonia are characterized by a high [18F]FDG uptake as well as lymph nodes. So far, most of the reports available, are case reports, case series or, rarely, retrospective studies reporting about incidentally detected COVID-19 infections as the papers by Wakfie-Corieh et al. [215], Pallardy et al. [216], Halsey et al. [217] and Setti et al. [218]. Otherwise, Dietz et al. [219] prospectively enrolled patients admitted with COVID-19 and performed [18F]FDG PET/CT from day 6 to day 14 of the onset of symptoms with the aim of assessing the inflammatory status at the presumed peak of the inflammatory phase in non-critically ill patients. They concluded that there was no correlation between PET lung inflammatory status and chest CT evolution or short-term clinical outcome; consequently, the inflammatory process at the presumed peak of the inflammatory phase in COVID-19 patients was obvious in [18F]FDG PET/CT scans.
Fig. 7.
Left panel: axial PET, CT and fused PET/CT (1a, 1b, 1c), sagittal PET, CT and fused PET/CT (2a, 2b, 2c) and coronal PET, CT and fused PET/CT (3a, 3b, 3c) images of a 18F-FDG PET/CT scan performed in a 59 year old male patient for oral carcinoma follow-up. Images revealed intense tracer uptake on diffused interstitial alterations with ground-glass appearance on both lungs; subsequent RT-PCR revealed COVID-19 positivity. Right panel: axial PET, CT and fused PET/CT (1a, 1b, 1c), sagittal PET, CT and fused PET/CT (2a, 2b, 2c) and coronal PET, CT and fused PET/CT (3a, 3b, 3c) images of a 18F-FDG PET/CT scan performed in the same patient for restaging of oral carcinoma purpose and after completion of COVID-19 specific therapy; images demonstrated complete disappearance of pathological uptake and resolution of interstitial alterations
Future perspectives and new trends
Large prospective studies contemporary to the evolution of the pandemic are desirable to investigate and evaluate the effective role of PET in this field. [18F]FDG PET/CT cannot be used in an emergency setting because it is more complex than chest CT and because of a possible risk of disease spreading due to the long procedure time; but, nevertheless it is not recommended for the diagnosis of this infectious disease, incidental findings are frequent during a pandemic scenario and the [18F]FDG PET/CT pattern needs to be well known and well described by nuclear medicine physicians. Further studies are desirable to confirm or controvert the insight of a possible role of [18F]FDG PET/CT in the diagnosis, follow-up and therapy monitoring of SARS-CoV-2 infection.
Role of 18F-FDG PET/CT in inflammatory bowel disease
IBD is a group of chronic relapsing inflammatory disorders of the gastrointestinal (GI) tract, represented by two subtypes, Crohn’s Disease (CD) and Ulcerative Colitis (UC), with typically different bowel location and inflammation pattern [220]. IBD has shown growing incidence in the last decade with a prevalence > 0.3% and a predominant age of onset between 15 and 30 years [221, 222]. A complex interplay of luminal microflora, external environment and disturbances in the immune response are concomitant etiological factors in genetically predisposed hosts [223]. IBD presents an important impact on quality of life with GI symptoms (including diarrhoea, weight loss, abdominal pain and vomiting) and extraintestinal manifestations (rheumatologic, dermatologic and ocular) [224]. Diagnosis of IBD is currently based on clinical, instrumental and histological criteria and treatment depends on disease severity [221, 225]. Although endoscopy and histological findings are considered the diagnostic gold standard, they are not devoid of limitations [226]. Other less invasive imaging techniques, such as ultrasound (US), CT and MRI, are performed to complete the diagnostic workup, with a more accurate overview of the disease [221, 227–229].
In the nuclear medicine field, in addition to WBC SPECT/CT, [18F]FDG PET/CT is a non-invasive, whole-body and hybrid technique, emerging as a reliable tool in the investigation of IBD, especially in active phases of disease, even if specific protocols have not been defined yet [220, 223, 225, 230, 231].
Clinical indications
Diagnosis in patients with suspected IBD in equivocal cases [141, 232];
- Initial assessment of the disease (anatomic location, extent, grade of activity):
- Extra-intestinal disease assessment (i.e. sacro-ileitis and lymph nodes involvement).
- Evaluation of complications (especially differential diagnosis between fibrotic and inflammatory strictures with consequent possibility to guide treatment strategy).
- Early evaluation of therapy efficacy.
- Follow-up and monitoring disease evolution
[18F]FDG PET/CT can be used when other conventional studies, particularly endoscopy, are non-diagnostic or infeasible [223, 225], as in severe and acute phase of disease, because of the high risk of complications (i.e. toxic megacolon or perforation), or in presence of strictures [227, 228].
Interpretation criteria
Qualitative analysis: [18F]FDG PET/CT is considered negative for IBD when a diffused and mild glucose uptake is detectable in bowel [226]. [18F]FDG PET/CT is considered positive for IBD when a segmental and significant increased glucose uptake in the intestinal tract is observed [226]. In addition, the uptake distribution pattern can be useful to discriminate between CD or UC: CD can involve any segment of the GI tract, more frequently the distal small bowel, showing discontinuous patches of inflammation; UC primarily involves the rectum with a proximally extension in a continuous way [233].
Semi-quantitative analysis: SUVmax is the more validated semi-quantitative parameter for the evaluation of glucose uptake in IBD. To date, no defined cut-off has been identified to differentiate positive and negative findings [228, 234]. However, a comparison between SUVmax of the bowel segments involved and liver is encouraged. The liver activity can be used as reference, because hepatic glucose uptake is relatively stable and reproducible. A bowel SUVmax lower or equal to liver can be considered normal, while a bowel SUVmax major than liver is suggestive for IBD. No standardized scales of evaluation have been defined yet [224, 226, 227].
Future perspectives and new trends
[18F]FDG PET/CT could be co-registered to MRI, rather than CT, significantly improving diagnostic morphological information about bowel wall and decreasing the radiation dose [228, 234, 235]. In different clinical protocols, [18F]FDG-WBC PET/CT demonstrated a higher specificity than [18F]FDG PET/CT, showing a lower radiopharmaceutical uptake in healthy GI and urinary tract, but it needs further studies [222, 225]. In preclinical studies, new and more specific radiopharmaceuticals are under investigation, particularly, 18F-Deoxy-Arabino Furanosyl Cytosine (18F-D-FAC) probe with preferential uptake in activated CD8 + lymphocytes and 18F-DPA-714, a radioligand of a translocator protein overexpressed in activated macrophages [222, 225].
Role of 18F-FDG PET/MRI in infection and inflammation
Imaging of inflammatory and infective disease include radiography, conventional scintigraphy with different radiopharmaceutical agents, CT with or without contrast enhancement (c.e.), MR and PET. Each of these has some limitations and therefore cannot be considered the only option for diagnosis or follow-up of inflammatory and infection disease. Hybrid imaging can overcome these limitations by combining morphological and functional information. In particular, PET/MR scanner offers superior soft tissue contrast to CT even without the use of contrast agents, and it is very appealing for application in the paediatric population. Furthermore, the association of MR that is able to assess blood vessels density, perfusion properties and cellular membrane integrity with PET that can evaluate the metabolic properties of the cells, represent an important tool for the infection and inflammation disease [236, 237], as reported above in the majority of future perspectives paragraphs.
Clinical indications
From the available data in literature emerged that [18F]FDG PET/MR can be indicated in some clinical indications in infection and inflammation, such as:
Interpretation criteria
The imaging interpretation should consider the PET, MR and PET/MR parts for each clinical indication. Therefore, a strong collaboration between nuclear medicine physician and radiologist is mandatory for the correct interpretation of the images, and the preparation of the final report.
In case of limbic encephalitis [238], typical patterns at MR images are shown, such as a T2 hyperintensity of the amygdala, of the hippocampus and/or insula and volume alterations of the mesial temporal structures. For PET images, the presence of any hypo- or hypermetabolism of the limbic system and any extra-limbic metabolic changes indicative/potentially associated with limbic encephalitis should be noted.
In case of spondylodiscitis [13], MR images often demonstrated the presence of hyperintense signal alterations on T2-weighted images or turbo inversion recovery magnitude (TIRM) sequences in the disk space appearing in both adjacent vertebrae as bone marrow and paravertebral soft-tissue edema, while the lesion showed a decreased signal intensity on non-contrast T1-weighted images. For PET part, the focal FDG uptake in spinal disks greater than that in the surrounding tissue (on the basis of a visual qualitative analysis) is considered to be suggestive.
In case of CS [239, 240], at MR images, the presence of T2 hyperintensity and/or a late gadolinium enhancement are usually suggestive of a cardiac sarcoidosis. In PET images, [18F]FDG uptake should be classified into four patterns (none, diffused, focal, or focal on diffused) for each subject. Focal and focal-on-diffused patterns of [18F]FDG uptake should be considered to be positive findings indicative of cardiac involvement. The presence or absence of right ventricle free-wall [18F]FDG activity should also be evaluated. Finally, the 17-segment model recommended by the American Heart Association should be used for the evaluation of extension of disease.
In case of LVV [60, 241], MR scan is interpreted by considering the thickening (> 3 mm) and contrast enhancement of the vessel wall on axial T1-weighted VIBE sequences. The presence of signal elevation in the vessel wall and visual or quantitatively measurable (> 3 mm) wall thickening on coronal T2-weighted STIR sequences is indicative for an active LVV. For PET images, a visual score is usually used, indicating the intensity of arterial [18F]FDG uptake in relation to liver uptake [i.e. 0 = no uptake; I = low-grade uptake (uptake lower than liver uptake); II = intermediate-grade uptake (similar to liver uptake); III = high-grade uptake (higher than liver uptake)].
In case of RF [128], MR images showed a hyper-intense signal on T2-weighted, DWI and contrast-enhanced images. While for the interpretation of PET images, a specific score could be used in relation to the uptake in normal liver, as follows: 0 = no pathological uptake; 1 = pathological uptake but less than liver uptake; 2 = pathological uptake similar to liver uptake; 3 = pathological uptake more than liver uptake.
In case of CD, [18F]FDG PET/MRI allows precise localization of the inflamed intestinal loop and permites the differential diagnosis between fibrotic stenosis and inflammatory stenosis. Co-acquisition also enables more accurate spatial and temporal matching of MR anatomy with PET data, reducing miss-registration artifacts due to motion and peristalsis [242, 243]. Attention should be paid to understand non-specific activity in transit in bowel lumen. A second bed image at 2 h p.i. could help in evaluating bowel wall activity versus lumen activity.
Future perspectives and new trends
PET/MRI has great potential to expand in the field of inflammation infection, not only with [18F]FDG but also with new radiopharmaceuticals targeting bacteria or white blood cell antigens to provide, in vivo, histological characterization of the underlying pathological process and to guide personalized therapy. Indeed, MRI, being a multi-parametric imaging modality with radiation emission, is suitable to study several inflammatory and infective processes particularly on central nervous system, spine, pelvis, soft tissues, heart and vessels.
Role of 18F-FDG PET/CT in pediatric inflammation diseases
In the pediatric population, [18F]FDG PET/CT imaging may play a role in several inflammatory or infectious diseases, including fever of FUO, IBD, musculoskeletal inflammation and infection (e.g. OM), fungal infection, chronic granulomatous disease and vasculitis (e.g. Henoch-Schonlein purpura, Kawasaki disease, infantile polyarteritis nodosa, and Takayasu disease), [245]. The advantage of using [18F]FDG PET/CT imaging for assessing inflammatory or infectious diseases in the pediatric population relies on the high spatial resolution and lesion-to-background ratio [246]. Other important aspects which favour [18F]FDG PET/CT imaging is the short exam duration, which facilitates the compliance of the young patients, and the use of optimized protocols which can result in a low radiation burden [245]. A limitation of [18F]FDG PET/CT imaging is the suboptimal specificity in distinguishing inflammatory/infectious processes from tumour [190].
Clinical indications
According to the available literature, the main clinical indications in the pediatric population are:
Assessment of FUO: one challenge in clinical medicine is the difficulty in diagnosing fever of unknown origin. This entity has varied definitions, ranging from fever lasting longer than 1 week that remains unexplained even after an extensive work-up in children, to any fever lasting 3 weeks. Most often, the etiology is infectious, but rheumatologic or neoplastic issues may also give rise to this clinical scenario [247]. However, in children with FUO, literature about the value of [18F]FDG PET/CT is scarce and the studies included relatively low numbers of patients. In a recent study on 110 children, Pijl et al found that [18F]FDG PET/CT is a valuable diagnostic tool in the evaluation of children with FUO, since it may detect a true underlying cause in almost half (62%) of all cases where none was found otherwise [248]. Endocarditis, systemic juvenile idiopathic arthritis, and inflammatory bowel disorder and cholangitis were the most common causes. CRP and leukocyte count seem to be positively and negatively associated, respectively, with finding a true positive focus of fever on [18F]FDG PET/CT, which may contribute to a priori assessment on the outcome of [18F]FDG PET/CT. Furthermore, pathological findings help to direct the therapeutic work-up in these patients [249];
Evaluation of vasculitis: vasculitis can affect arteries and veins of any size. In children it is found in conditions such as Henoch-Schonlein purpura, Kawasaki disease, infantile polyarteritis nodosa, and Takayasu disease. Diagnosis typically relies on biopsy, conventional angiography, ultrasound, or MR angiography. However, PET has been shown capable of revealing vasculitis in adults [250] and incipient evidence exists on the value of PET/CT for assessing active vascular inflammation in Takayasu arteritis, the most common granulomatous inflammation of large arteries in children. In Takayasu arteritis, lack of overt clinical symptoms and the absence of currently available reliable serologic biomarkers conceivably contribute a delay in diagnosis. [18F]FDG PET/CT in conjunction with MRI offers a promising approach in assessing inflammatory activity within the vasculature prior to the development of vessel damage (wall thickening, dilatation/aneurysm, stenosis, occlusion) and there is accumulating evidence supporting the role and the utility of PET/MRI as the best hybrid system in characterizing disease [251];
Assessment of peripheral OM: patients with acute OM can present with a variety of symptoms that are influenced by the age of the child and causative pathogen. Moreover, multifocal involvement is more frequently present in particular in newborns. [18F]FDG PET/CT have a major advantage of assessing the whole body in one imaging session to confirm or exclude multifocal involvement [252]. Gram-positive bacteria, especially Staphylococcus aureus and Streptococcus pyrogens, are by far the most common pathogens and account for 80%–90% of the cases. Kingelle kingae, a Gram-negative bacterium, is a causative organism for osteoarticular infections and is mainly present in younger children [253]. [18F]FDG PET/CT is the radionuclide modality of choice in the evaluation of spinal OM, in occult bacterial infections, and when hematogenous spread is suspected [5];
IBD: in children, CD and UC account for the vast majority of inflammatory bowel disease. Imaging is used not only to diagnose but also to assess disease extent, detect and characterize complications, and differentiate active from quiescent diseases. Studies of [18F]FDG PET/CT in children with inflammatory bowel disease have demonstrated high sensitivity, specificity, and accuracy of this non-invasive technique [254, 255]. Furthermore, [18F]FDG PET/MRI has several advantages over [18F]FDG PET/CT in children with inflammatory bowel disease, including providing better anatomical and soft tissue detail, the benefit of functional magnetic resonance diffusion weighted imaging, as well as apparent diffusion coefficient maps, and a lower patient radiation exposure [252].
Although, still requiring further evidence, [18F]FDG PET/CT imaging appears to be indicated in other diseases, such as:
Fungal infection, mainly for defining the best location for biopsy and to detect all infectious sites in the body, to evaluate response to therapy and to help in therapy decision-making on whether to stop, continue, or modify treatment [256].
Detection of infective foci related to assistive devices or valvular prosthesis and diagnosis or exclusion of myocarditis in patients treated for a variety of congenital heart diseases [257].
Juvenile autoimmune diabetes or T1D is an autoimmune disease characterized by the disruption of pancreatic β-cells that are responsible for insulin secretion [258].
Interpretation criteria
Correct interpretation requires accurate knowledge of physiological [18F]FDG distribution. Furthermore, children may present physiologic [18F]FDG uptake in the thymus and in the brown fat (e.g. in case of low temperature in the waiting room).
Any focal radiopharmaceutical uptake in the bone should be considered suspicious although bone marrow uptake can be noticed in patients with inflammatory disease or infection. Knowledge of any tumour disease in patient history is mandatory for a correct interpretation. Indeed, a direct association has been shown between bone marrow glucose uptake and host systemic inflammation in cancer patients [259]. Furthermore, a diffusedly increased bone marrow FDG uptake can be present in patients due to increased hematopoiesis following administration of granulocyte-colony stimulating factors [260].
Likewise, evaluation of any uptake in the spleen should be approached cautiously with knowledge of full medical history (including previous medication: i.e. recent cytokine administration). Diffused spleen uptake may reflect activation of B-cells, which may be associated to many inflammatory diseases [261]. Interpretation may be done visually and/or assisted by quantitative analysis recording SUV.
In conclusion, the role of [18F]FDG PET/CT still needs to be fully investigated in pediatric patients undergoing the scan to assess inflammatory or infectious diseases. [18F]FDG PET/CT may be used with optimized protocol to reduce radiation doses in children. With the increase of the studies in literature, the use of [18F]FDG PET/CT will also increase in the clinical practice in the next years.
Future perspectives and new trends
Beyond [18F]FDG PET/CT, also PET/MRI may play a future role in the assessment of inflammatory and infectious diseases in children. At the moment, there are few studies documenting the value of this imaging method in children and young adults. Furthermore PET/MRI should be compared in head-to-head studies to assess the diagnostic gain compared to PET/CT.
Patient preparation
The list below includes several general considerations for all patients:
[18F]FDG-PET/CT is contraindicated in case of pregnancy.
Recommendations regarding breastfeeding are reported in the ICRP [27, 262].
Patients are recommended to fast at least 6 h before the administration of [18F]FDG [27].
Patients should drink 0.5–1 L of water (or receive non-glucose containing fluid intravenously) in the 2 h before [18F]FDG PET/CT to increase renal excretion of radiopharmaceutical agent [143].
In diabetic patients treated with insulin, [18F]FDG should be administrated at least 4 h after the last rapid-acting insulin dose [27] or 6 h after the last short-acting dose and no intermediate or long-acting insulin on the day [18F]FDG PET/CT is performed. Use of metformin may increase FDG uptake of the gastrointestinal tract but does not necessarily have to be stopped before scan [143].
Blood glucose levels < 11 mmol/L (< 180 mg/dL) should be indicated, although, high blood glucose level in unstable or poorly controlled diabetes is not an absolute contraindication to the exam [263].
When intensive care unit (ICU) patients are scheduled for [18F]FDG PET/CT, doctors should ensure that all sources of glucose or carbohydrates are timely stopped before scan (glucose infusions, parenteral nutrition, etc.) [143].
The discontinuance of corticosteroids is recommended, after clinical evaluation, particularly for LVV [3, 27].
Avoid strenuous physical activities for 24 h before [18F]FDG administration [98].
Relax in an adequately temperature-controlled room [20–22 °C (68–71.6 °F)] to minimize physiologic uptake in muscles and brown fat at the moment of and after administration of [18F]FDG [98]. In some cases, [18F]FDG uptake in brown fat could be reduced by beta-blocking drugs (e.g. orally administered 20 mg propranolol 1 h before [18F]FDG injection) [98].
In case of SI, DF and OM, discontinuance of antibiotic therapy is not necessary, although it is important to know its last administration time.
For the evaluation of post-surgical SI, it is recommended to perform [18F]FDG-PET/CT after at least 4 months after surgery (to avoid false-positive findings) [3].
Considerations for specific conditions:
In oncologic patients, in case of suspected SI, [18F]FDG-PET/CT should be performed at least 1 month after the end of chemotherapy [3].
For patients with suspected IE, CIED, LVAD and CS a high-fat–enriched diet lacking carbohydrates for 12–24 h prior to the exam associated with a prolonged fasting period (12–18 h) is necessary. For the same purpose, the intravenous heparin use (50 IU/kg) about 15 min before [18F]FDG administration may also be used [110].
In case of SARS-COV-2 every step of the patient flow at all levels including arrival, waiting area, during the injection and scan and the return home should be managed in concordance with the operational guidelines for the management of diagnostic activities and patients in the nuclear medicine departments recommended by the EANM [264] and by the American College of Nuclear Medicine [265].
In patients with IBD the administration of high quantity of fluids could be useful to obtain a proper distension of bowel segments. A previous medical therapy with rifaximine for 2 days before PET scan could allow an adequate intestinal cleanse, reducing unspecific [18F]FDG uptake [225, 266].
For PET/MRI the patient preparation is similar to those undergoing [18F]FDG PET/CT, in accordance with the European guidelines [267, 268]. Careful attention should be paid to the presence of metal objects or incompatible devices in the patient, due to the strong magnetic field (3 T). Table 3 reports the contraindications to MR examination. In case of doubts about the compatibility of the devices with PET/MR, the following website http://www.mrisafety.com can be consulted.
In pediatric patients all the above rules, according to pathology, should be followed. Limiting the use of mobile devices for playing is important [269]. [18F]FDG activity should be calculated according to the EANM pediatric dosage card [270].
Table 3.
Contraindications to PET/MR scan
| Potential contraindications to an MR examination [290] |
|---|
| Aneurysm clip(s) |
| Any metallic fragment or foreign body |
| Coronary and peripheral artery stents |
| Aortic stent graft |
| Prosthetic heart valves and annuloplasty rings |
| Cardiac occluder devices |
| Vena cava filters and embolisation coils |
| Haemodynamic monitoring and temporary pacing devices, eg, Swan–Ganz catheter |
| Haemodynamic support devices |
| Cardiac pacemaker |
| Implanted cardioverter-defibrillator (ICD) |
| Retained transvenous pacemaker and defibrillator leads |
| Electronic implant or device, eg, insulin pump or other infusion pump |
| Permanent contraceptive devices, diaphragm, or pessary |
| Cochlear, otologic, or other ear implant |
| Neurostimulation system |
| Shunt (spinal or intraventricular) |
| Vascular access port and/or catheter |
| Tissue expander (eg, breast) |
| Joint replacement (eg, hip, knee, etc.) |
| Any type of prosthesis (eg, eye, penile, etc.) |
| Tattoo or permanent makeup |
| Known claustrophobia |
| Body piercing jewellery |
| Hearing aid |
| Renal insufficiency |
| Known/possible pregnancy or breastfeeding |
Image acquisition
Standardized imaging protocols exist only for IE, CIED, LVAD, CS and VGI, where acquisition, reconstruction and post-processing pipeline have been recently summarized in joint procedural recommendations for cardiovascular imaging [110]. For all the other infective and inflammatory disorders, the same acquisition, reconstruction and post-processing steps described in the EANM procedural guidelines for tumour imaging and for inflammations and infections are recommended [27, 268].
The administered activity of [18F]FDG is approximately 2.5–5 MBq/kg according to available device [27]. Whole-body PET/CT acquisitions should start approximately 50–60 min after injection, and they are performed from base of skull to mid-thigh by placing patient in supine position with raised arms. The study may be completed by a late and focussed acquisition of the suspected field of interest, and it could be extended to feet in selected patients (i.e., in patients with DF, FUO or systemic infections to detect occult foci of infection).
For imaging IE, CIED, LVAD, CS and VGI, the administration of iodinated contrast may be useful to obtain a full diagnostic CT scan.
A dual time acquisition has been proposed for several clinical indications:
For imaging SI;
For differentiating infections from malignancies or infected from non-infected fractures [271];
In HIV-positive patients;
For increasing T/B and contrast resolution in LVV and IE.
However, the performance, overall reliability and usefulness of dual time point imaging has mixed results, limiting its routine clinical use for evaluating incidental foci of uptake on [18F]FDG PET/CT [272].
If the scan is performed for therapy evaluation of long-term follow-up, it is important to apply the same interval from injection to image acquisition of the basal study, to make SUVmax measurements more comparable to each other.
Imaging acquisition protocols of PET/MRI for each clinical indication are reported in (Table 4).
Table 4.
Imaging acquisition protocols for PET/MR
| Clinical indication, ref | PET protocol | MR protocol |
|---|---|---|
| Limbic encephalitis, 1 |
Brain: one bed position for 20 min (static) Whole-body: 4 min/bed |
Head and neck radiofrequency coil Brain sequences: T1-MPRAGE FLAIR SWI STIR TSE T2-TSE c.e. MPRAGE Whole-body sequences: T1-VIBE T2-HASTE DWI c.e. T1-VIBE Contrast enhancement: gadobutrol (0.2 mmol/kg b.w.) |
| Spondylodiscitis, 2 | Whole-body: 5-min/bed |
Integrated spine coil Spine: T1-weighted TSE sagittal T2-weighted TSE transverse T1-weighted TSE transverse Whole-body sequences: T1-VIBE T2-HASTE Contrast enhancement: gadobutrol (0.1 mmol/kg b.w.) |
| Cardiac sarcoidosis, 3,4 | Thoracic: one bed of 25 min centred over the heart (static) |
Thoracic sequences: T2-weighted coronal T2- HASTE Breath-hold cine imaging with TrueFISP and TIRM Late gadolinium-enhanced (15 min following administration of c.e.) PSIR Contrast enhancement: gadobutrol (0.1 mmol/kg b.w.) |
| Large vessels vasculitis and retroperitoneal fibrosis, 5–7 | Whole-body: 4 min/bed |
Whole-body sequences: T1-VIBE T2-HASTE STIR DWI (only for retroperitoneal fibrosis) |
MPRAGE magnetization-prepared rapid acquisition with gradient-echo, FLAIR fluid-attenuated inversion recovery, SWI susceptibility weighted-imaging, STIR short-tau inversion recovery, TSE turbo spin-echo, VIBE volume interpolated breath-hold examination, HASTE half Fourire acquisition single shot turbo spin echo, DWI diffusion weighted imaging, TrueFISP fast imaging with steady state procession, TIRM turbo inversion recovery magnitude, PSIR phase-sensitive version recovery
Possible pitfalls
Due to the low specificity of [18F]FDG in differentiating an infection from an acute/chronic sterile inflammation and the lack of standardized interpretation criteria, several pitfalls should be considered.
In SI, false-positive results are possible in case of inflammatory or degenerative disc disease, bone tumours and metastases, recent vertebral fractures and post-operative inflammation [273].
In spondylodiscitis, the presence of erosive osteochondrosis can show focally elevated [18F]FDG uptake as well as hyperintense T2-weighted and hypointense T1-weighted MR signals; however, these MR signal patterns can also be found in inflammatory processes and degenerative diseases (i.e. degenerative disk space narrowing) [274].
In DF, the presence of concomitant orthopedic comorbidities, for example pre-existing fractures, degenerative arthrosis, gout or other form of arthritis, could show intense uptake of [18F]FDG, thus representing a possible source of misdiagnosis. Moreover, another important diagnostic challenge is represented by Charcot osteoarthropathy. This complication is characterized by intense and diffused uptake of [18F]FDG, mainly at mid-hindfoot, that does not allow to determine if there is or not a superimposed infection (Fig. 3). Indeed, in these cases, a radiolabeled WBC scintigraphy in combination with bone marrow scan is more appropriate [17, 20, 22].
Same applies for OM in general, and after surgery in particular, non-specific [18F]FDG uptake can be found in healing tissues and may last for months after the intervention. Bone fractures may also show [18F]FDG positivity for 1–2 years, as well as degenerative changes. A cause of false-positive [18F]FDG uptake in patients with metallic implants might be explained by high glucose metabolism in the joint capsule and around the prosthesis neck, caused by inflammation due to granulomatous foreign body reaction against polyethylene debris particles [1].
For LVV, diagnostic accuracy of PET/CT declines by nearly 50% after the start of steroid or immunosuppressive treatments. SUVmax reduction is notable 3 months after treatment, while no more reduction between 3 and 6 months after the end of the therapy is appreciable [51, 275]. Minimal persistent inflammation of the vessel wall after treatment, vascular remodelling, and resistance to treatment (similar to what happens for antibiotics) are possible hypothesis. False positive results mainly due to increased [18F]FDG uptake in macrophage-rich areas in atherosclerotic plaques and low spatial resolution of PET and PET/CT scanners actually employed, can be possible limitations [276].
In case of VGI, [18F]FDG uptake may be observed also in non-infected graft for many years after surgery and it may depend on type of graft material (Dacron prosthesis usually show higher uptake than Goretex), type of surgery (open surgery or endovascular approach) and patient variability [277]. Therefore, when interpreting a [18F]FDG PET/CT scan is very important to carefully review patient history and comorbidities. Indeed, other diseases may interfere with a correct interpretation, for example, venous thrombosis, vasculitis or RF could cause misdiagnosis. Old peri-prosthetic blood collections infiltrated by macrophages can also give false-positive results due to the high glicometabolic activity of these cells. Moreover, [18F]FDG-avid processes that are close to the graft and show an overlap on the graft itself, for example, extra-bone spondylodiscitis or psoas abscesses or in general infective/inflammatory processes of surrounding soft tissues, could be wrongly interpreted as positive findings. Moreover, despite the focal pattern being considered, at the moment, the best criterion to diagnose an infection, in an entirely infected device this criterion cannot be applied. Finally, efforts should be directed to limit artifact movements.
False-negative results are possible in case of infection with low-virulence bacteria, previous antimicrobial therapy, epidural abscesses or extensive vertebral arthrodesis [273].
Possible pitfalls in case of IE, CIED, LVAD, CS can be summarized as follows:
Surgical adhesives and post-operative inflammation can result in false-positive findings soon after valve surgery.
Post-operative inflammation may also result in falsely positive scan depending on the level of risk for infection [263]; in non-complicated valve surgery, scans < 3 weeks surgery can be considered.
Incomplete myocardial suppression of [18F]FDG (scarce fasting time before PET).
Lipomatous hypertrophy of the interatrial septum.
[18F]FDG-avid processes close to the graft but not involving the device.
Primary cardiac tumours and cardiac metastasis.
Libman-Sacks endocarditis.
Post-surgical sterile inflammation (i.e., foreign body reaction).
Artifacts caused by device.
False increase of [18F]FDG uptake in RPF may be related to beam-hardening artifact, which should not be overlooked since many patients have vascular calcifications [278].
All pitfalls mentioned can occur, since a reason for FUO can be any of the other inflammatory/infective disorders mentioned in this paper.
Sarcoidosis and pulmonary tuberculosis lesions are quite morphologically heterogeneous. They may appear as nodular, micro-nodular, mass or sub-solid opacity and it can be very difficult to differentiate from lung carcinoma [151, 161, 279]. SUV measurements from both active granulomatous diseases and malignant lesions can be similarly high with significant overlap. [18F]FDG PET/CT does not reliably distinguish between granulomatous active and malignant lesions [150]. The suspicion of lymphoma should be considered in presence of intrathoracic or abdominal metabolically active lymphadenopathies.
In search of IFI, the most common challenge in evaluating a [18F]FDG PET/CT scan, is the presence of potential causes of false-negative results, such as: lesion size, low metabolic rate, hyperglycemia, lesions masked by adjacent high physiologic uptake, concomitant drug use interfering with uptake, such as ongoing steroid therapy in systemic disorders. Also, the presence of potential causes of false-positive results, should be considered, such as: injection artifacts and external contamination, reconstruction artifacts from attenuation correction, normal physiologic uptake, pathologic uptake not related to IFI, such as neoplasia or other infection [27]. Main limitation in evaluating IFI is the absence of a validated SUVmax cut-off or other parameters to differentiate malignant lesions from fungal lesions [188]. In HIV-positive patients special care must be taken in evaluating [18F]FDG PET/CT because persistent generalized lymphadenopathy and benign hypermetabolic foci are common, especially in the context of high viremia, and can lead to false-positive interpretations of malignancy [198].
Potential pitfalls for SARS-CoV-2 patients are drug-induced interstitial pneumonia (everolimus, nivolumab), or interstitial pneumonias of different etiologies, responsible for similar PET/CT pattern [280].
Interestingly, some studies [281, 282] have reported that the lung inflammatory burden in COVID-19 infection is characterized by fluoro-choline uptake. It has been speculated that the systemic hyper-inflammation (also defined as macrophage activation syndrome), or cytokine storm, requires an increase in choline consumption to synthesize phosphatidylcholine and stimulate phagocytosis, organelle biogenesis, secretory functions, and endocytosis; as a consequence, 18F-Fluorocholine that is a synthetic analogue of the naturally choline, could be useful in the detection and quantification of the macrophage activity in pulmonary interstitial infiltrates of Covid-19 pneumonia. Also, mild 68Ga-PSMA-11 mild uptake has been identified in correspondence with peripheral ground-glass opacities of both lungs in a prostate cancer patient, as reported by Stasiak et al. [283].
In patients with IBD, false-positive cases can occur in normal bowel (for high turnover of intestinal mucosa, increased peristaltic activity of muscular layer and collapse of bowel loops) and in diabetic patients assuming hypoglycemic oral therapy, in diverticulitis, in infectious colitis or in case of malignancies [194, 225, 226, 284].
False-negative cases can occur in disease with a low-grade activity and in case of recent administration of high dose of corticosteroid [224, 226, 285].
In pediatric patients, false-positive [18F]FDG uptake has been described in brain or the genitourinary system as well as false-negative [18F]FDG PET/CT in urinary tract infection has been described [286]. Gastrointestinal and reactive lymph nodes may also cause false-positive findings. Special attention should be paid to [18F]FDG-avid pitfalls, including non-ossifying fibroma, fractures and muscular distortion [287–289].
Finally, when [18F]FDG PET/CT is performed on patients with kidney and/or liver failure, scan potentially results in reduced background clearance, reduced metabolism of FDG, and poor image quality [143].
Most common [18F]FDG PET/CT pitfalls imaging in inflammation and infections are shown in Supplementary Table 1.
PET reporting
As a general rule, [18F]FDG PET/CT should be reported according to the specific interpretation criteria adopted for each clinical indication.
Images should be described in terms of both anatomical and functional/metabolic point of view and should be correlated with patient’s clinical signs as far as possible.
The following common scheme of reporting [18F]FDG PET/CT should be applied to all the clinical indications:
Personal data (patients’ name, date of birth, medical record number, date of [18F]FDG PET/CT execution).
Anamnestic information (clinical history, comorbidities, previous surgery, diagnostic tests and ongoing treatments).
Technical data (radiopharmaceutical, dose, type of camera, the interval between [18F]FDG injection and the images acquisition).
- Description of findings:
- Presence/absence of lesion/s;
- Pattern of uptake (focal vs diffused, homogeneous vs non-homogeneous, continuous vs discontinuous for IBD);
- Location;
- Extent (in case of involvement of contiguous soft tissues or regional lymph-nodes);
- Evaluation of CT component: in particular in VGI where the graft borders and other radiological signs of infection, such as dislocation of the graft or presence of gas, should also be reported;
- Intensity of uptake by SUVmax evaluation: this could be reported, not for diagnostic purposes but to provide a reference parameter for further PET studies that will be performed for therapy evaluation or long-term follow-up. To this purpose, it is important to clearly mention the precise timing between [18F]FDG injection and images acquisition and to indicate the type of tomograph, since both these aspects could have a great influence on SUVmax calculation, thus making this parameter not comparable. In some clinical indications, the report should include specific scores (i.e. for SI, LVV and retroperitoneal fibrosis), as reported in the specific paragraphs on imaging interpretation.
Possible differential diagnosis.
Comparison with previous [18F]FDG PET/CT or PET/MRI if performed.
Conclusions: in this section should be indicated the most probable diagnosis based on the specific interpretation of the findings. It would also eventually recommend follow-up or integration with additional imaging or laboratory tests for the confirmation or exclusion of diagnosis.
Discussion
Several considerations can be drawn from this paper. First of all, we noted that the clinical applications of FDG in the field of inflammatory and infective diseases is rapidly expanding. In some diseases, [18F]FDG shows clear superiority over other nuclear medicine modalities (i.e., WBC scan or anti-granulocyte antibody scan) such as for SI, LVV, EI, RF, sarcoidosis and FI. The main reasons are: (1) the histopathological features of the diseases and (2) the availability of well-established image interpretation criteria. For other pathologies, such as IBD, VGI, OM, DFI and FUO, [18F]FDG shows some limitations. Finally, in some cases such as PJI, early post-operative VGI, high pre-test probability of infection in patients with FUO, post-operative evaluation of CD, brain abscesses and fracture-related infections with metal fixators, [18F]FDG has a limited role, while WBC should be preferred due to its higher diagnostic accuracy.
In some other pathologies, such us SARS-Cov-2 infections, HIV infections and tuberculosis, the role of [18F]FDG is still unclear, and it may be employed only in specific settings.
Moreover, several rheumatic diseases such as rheumatoid arthritis, Sjogren syndrome, Bechet diseases, and others, have not been mentioned in the present article due to the paucity of available scientific evidence.
The most important aspect is that, for many of the diseases, there is either not well-established image interpretation criteria, nor relevant scientific publications comparing [18F]FDG PET/CT with other radiological or nuclear medicine imaging modalities. Moreover, the standard of references, such as histological confirmation, is often poor.
Conclusions
In conclusion, the use of [18F]FDG is rapidly expanding in the field of inflammation/infection imaging. However, for most diseases there still lacks the publication of large case–control, multicentric studies with appropriate gold standard, to correctly define the clinical role of [18F]FDG and standardize image interpretation criteria.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
Authors wish to thank Dr. Rossella Ruta, Dr. Alessia Branca and Dr. Coruzzi Chiara for their contribution in medline search.
Declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The research was conducted in accordance with the Declaration of Helsinki.
Research involving human participants and/or animals
The present manuscript involves human participants.
Informed consent
In each center, all patients gave their consent for the potential publication of their images.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Massimiliano Casali, Chiara Lauri, Corinna Altini, Francesco Bertagna, Gianluca Cassarino, Angelina Cistaro, AnnaPaola Erba, Cristina Ferrari, CiroGabriele Mainolfi, Andrea Palucci, and Napoleone Prandini equally contributed as first author.
Domenico Albano, Luca Burroni, Alberto Cuocolo, Laura Evangelista, Elena Lazzeri, Natale Quartuccio, Brunella Rossi, Giuseppe Rubini, Martina Sollini, Annibale Versari, and Alberto Signore equally contributed as last author.
References
- 1.Glaudemans AW, Israel O, Slart RH. Pitfalls and limitations of radionuclide and hybrid imaging in infection and inflammation. Semin Nucl Med. 2015;45(6):500–512. doi: 10.1053/j.semnuclmed.2015.02.005. [DOI] [PubMed] [Google Scholar]
- 2.Raghavan M, Lazzeri E, Palestro CJ. Imaging of spondylodiscitis. Semin Nucl Med. 2018;48(2):131–147. doi: 10.1053/j.semnuclmed.2017.11.001. [DOI] [PubMed] [Google Scholar]
- 3.Lazzeri E, Bozzao A, Cataldo MA, Petrosillo N, Manfrè L, Trampuz A, et al. Joint EANM/ESNR and ESCMID-endorsed consensus document for the diagnosis of spine infection (spondylodiscitis) in adults. Eur J Nucl Med Mol Imaging. 2019;46(12):2464–2487. doi: 10.1007/s00259-019-04393-6. [DOI] [PubMed] [Google Scholar]
- 4.Kawakyu-O'Connor D, Bordia R, Nicola R. Magnetic resonance imaging of spinal emergencies. Magn Reson Imaging Clin N Am. 2016;24(2):325–344. doi: 10.1016/j.mric.2015.11.004. [DOI] [PubMed] [Google Scholar]
- 5.Smids C, Kouijzer IJ, Vos FJ, Sprong T, Hosman AJ, de Rooy JW, et al. A comparison of the diagnostic value of MRI and (18)F-FDG-PET/CT in suspected spondylodiscitis. Infection. 2017;45(1):41–49. doi: 10.1007/s15010-016-0914-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fuster D, Tomás X, Mayoral M, Soriano A, Manchón F, Cardenal C, et al. Prospective comparison of whole-body (18)F-FDG PET/CT and MRI of the spine in the diagnosis of haematogenous spondylodiscitis. Eur J Nucl Med Mol Imaging. 2015;42(2):264–271. doi: 10.1007/s00259-014-2898-0. [DOI] [PubMed] [Google Scholar]
- 7.Nanni C, Boriani L, Salvadori C, Zamparini E, Rorato G, Ambrosini V, et al. FDG PET/CT is useful for the interim evaluation of response to therapy in patients affected by haematogenous spondylodiscitis. Eur J Nucl Med Mol Imaging. 2012;39(10):1538–1544. doi: 10.1007/s00259-012-2179-8. [DOI] [PubMed] [Google Scholar]
- 8.Riccio SA, Chu AK, Rabin HR, Kloiber R. Fluorodeoxyglucose positron emission tomography/computed tomography interpretation criteria for assessment of antibiotic treatment response in pyogenic spine infection. Can Assoc Radiol J. 2015;66(2):145–152. doi: 10.1016/j.carj.2014.08.004. [DOI] [PubMed] [Google Scholar]
- 9.Kouijzer IJ, Vos FJ, Janssen MJ, van Dijk AP, Oyen WJ, Bleeker-Rovers CP. The value of 18F-FDG PET/CT in diagnosing infectious endocarditis. Eur J Nucl Med Mol Imaging. 2013;40(7):1102–1107. doi: 10.1007/s00259-013-2376-0. [DOI] [PubMed] [Google Scholar]
- 10.Hungenbach S, Delank KS, Dietlein M, Eysel P, Drzezga A, Schmidt MC. 18F-fluorodeoxyglucose uptake pattern in patients with suspected spondylodiscitis. Nucl Med Commun. 2013;34(11):1068–1074. doi: 10.1097/MNM.0b013e328365abec. [DOI] [PubMed] [Google Scholar]
- 11.Altini C, Lavelli V, Niccoli-Asabella A, Sardaro A, Branca A, Santo G, et al. Comparison of the diagnostic value of mri and whole body (18)F-FDG PET/CT in diagnosis of spondylodiscitis. J Clin Med. 2020;9(5):1581. doi: 10.3390/jcm9051581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Righi E, Carnelutti A, Muser D, Di Gregorio F, Cadeo B, Melchioretto G, et al. Incremental value of FDG-PET/CT to monitor treatment response in infectious spondylodiscitis. Skelet Radiol. 2020;49(6):903–912. doi: 10.1007/s00256-019-03328-4. [DOI] [PubMed] [Google Scholar]
- 13.Fahnert J, Purz S, Jarvers JS, Heyde CE, Barthel H, Stumpp P, et al. Use of simultaneous 18F-FDG PET/MRI for the detection of spondylodiskitis. J Nucl Med. 2016;57(9):1396–1401. doi: 10.2967/jnumed.115.171561. [DOI] [PubMed] [Google Scholar]
- 14.Asti M, Iori M, Erba PA, Atti G, Farioli D, Guidotti C, Versari A, et al. Radiosynthesis of 68Ga-labelled DOTA-biocytin (68Ga-r-BHD) and assessment of its pharmaceutical quality for clinical use. Nucl Med Commun. 2012;33:1179–1187. doi: 10.1097/MNM.0b013e3283573e05. [DOI] [PubMed] [Google Scholar]
- 15.Ramsey SD, Newton K, Blough D, McCulloch DK, Sandhu N, Reiber GE, et al. Incidence, outcomes, and cost of foot ulcers in patients with diabetes. Diabetes Care. 1999;22(3):382–387. doi: 10.2337/diacare.22.3.382. [DOI] [PubMed] [Google Scholar]
- 16.Reiber GE, Lipsky BA, Gibbons GW. The burden of diabetic foot ulcers. Am J Surg. 1998;176(2A Suppl):5s–10s. doi: 10.1016/S0002-9610(98)00181-0. [DOI] [PubMed] [Google Scholar]
- 17.Lauri C, Leone A, Cavallini M, Signore A, Giurato L, Uccioli L. Diabetic foot infections: the diagnostic challenges. J Clin Med. 2020;9(6):1779. doi: 10.3390/jcm9061779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Álvaro-Afonso FJ, Lázaro-Martínez JL, Aragón-Sánchez J, García-Morales E, García-Álvarez Y, Molines-Barroso RJ. Inter-observer reproducibility of diagnosis of diabetic foot osteomyelitis based on a combination of probe-to-bone test and simple radiography. Diabetes Res Clin Pract. 2014;105(1):e3–5. doi: 10.1016/j.diabres.2014.04.024. [DOI] [PubMed] [Google Scholar]
- 19.Malone M, Bowling FL, Gannass A, Jude EB, Boulton AJ. Deep wound cultures correlate well with bone biopsy culture in diabetic foot osteomyelitis. Diabetes Metab Res Rev. 2013;29(7):546–550. doi: 10.1002/dmrr.2425. [DOI] [PubMed] [Google Scholar]
- 20.Signore A, Jamar F, Israel O, Buscombe J, Martin-Comin J, Lazzeri E. Clinical indications, image acquisition and data interpretation for white blood cells and anti-granulocyte monoclonal antibody scintigraphy: an EANM procedural guideline. Eur J Nucl Med Mol Imaging. 2018;45(10):1816–1831. doi: 10.1007/s00259-018-4052-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lauri C, Tamminga M, Glaudemans A, Juárez Orozco LE, Erba PA, Jutte PC, et al. Detection of osteomyelitis in the diabetic foot by imaging techniques: a systematic review and meta-analysis comparing MRI, white blood cell scintigraphy, and FDG-PET. Diabetes Care. 2017;40(8):1111–1120. doi: 10.2337/dc17-0532. [DOI] [PubMed] [Google Scholar]
- 22.Lauri C, Glaudemans A, Signore A. Leukocyte imaging of the diabetic foot. Curr Pharm Des. 2018;24(12):1270–1276. doi: 10.2174/1381612824666180227094116. [DOI] [PubMed] [Google Scholar]
- 23.Lauri C, Glaudemans A, Campagna G, Keidar Z, Kurash MM, Georga S, et al. Comparison of white blood cell scintigraphy, FDG PET/CT and MRI in suspected diabetic foot infection: results of a large retrospective multicenter study. J Clin Med. 2020;9(6):1645. doi: 10.3390/jcm9061645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chakfé N, Diener H, Lejay A, Assadian O, Berard X, Caillon J, et al. Editor’s choice—European Society for Vascular Surgery (ESVS) 2020 clinical practice guidelines on the management of vascular graft and endograft infections. Eur J Vasc Endovasc Surg. 2020;59(3):339–384. doi: 10.1016/j.ejvs.2019.10.016. [DOI] [PubMed] [Google Scholar]
- 25.Roca M, de Vries EF, Jamar F, Israel O, Signore A. Guidelines for the labelling of leucocytes with (111)In-oxine. Inflammation/infection taskgroup of the European Association of Nuclear Medicine. Eur J Nucl Med Mol Imaging. 2010;37(4):835–841. doi: 10.1007/s00259-010-1393-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Capriotti G, Chianelli M, Signore A. Nuclear medicine imaging of diabetic foot infection: results of meta-analysis. Nucl Med Commun. 2006;27(10):757–764. doi: 10.1097/01.mnm.0000230065.85705.b3. [DOI] [PubMed] [Google Scholar]
- 27.Jamar F, Buscombe J, Chiti A, Christian PE, Delbeke D, Donohoe KJ, et al. EANM/SNMMI guideline for 18F-FDG use in inflammation and infection. J Nucl Med. 2013;54(4):647–658. doi: 10.2967/jnumed.112.112524. [DOI] [PubMed] [Google Scholar]
- 28.Treglia G, Sadeghi R, Annunziata S, Zakavi SR, Caldarella C, Muoio B, et al. Diagnostic performance of fluorine-18-fluorodeoxyglucose positron emission tomography for the diagnosis of osteomyelitis related to diabetic foot: a systematic review and a meta-analysis. Foot (Edinb) 2013;23(4):140–148. doi: 10.1016/j.foot.2013.07.002. [DOI] [PubMed] [Google Scholar]
- 29.Familiari D, Glaudemans AW, Vitale V, Prosperi D, Bagni O, Lenza A, et al. Can sequential 18F-FDG PET/CT replace WBC imaging in the diabetic foot? J Nucl Med. 2011;52(7):1012–1019. doi: 10.2967/jnumed.110.082222. [DOI] [PubMed] [Google Scholar]
- 30.Nawaz A, Torigian DA, Siegelman ES, Basu S, Chryssikos T, Alavi A. Diagnostic performance of FDG-PET, MRI, and plain film radiography (PFR) for the diagnosis of osteomyelitis in the diabetic foot. Mol Imaging Biol. 2010;12(3):335–342. doi: 10.1007/s11307-009-0268-2. [DOI] [PubMed] [Google Scholar]
- 31.Kagna O, Srour S, Melamed E, Militianu D, Keidar Z. FDG PET/CT imaging in the diagnosis of osteomyelitis in the diabetic foot. Eur J Nucl Med Mol Imaging. 2012;39(10):1545–1550. doi: 10.1007/s00259-012-2183-z. [DOI] [PubMed] [Google Scholar]
- 32.Schwegler B, Stumpe KD, Weishaupt D, Strobel K, Spinas GA, von Schulthess GK, et al. Unsuspected osteomyelitis is frequent in persistent diabetic foot ulcer and better diagnosed by MRI than by 18F-FDG PET or 99mTc-MOAB. J Intern Med. 2008;263(1):99–106. doi: 10.1111/j.1365-2796.2007.01877.x. [DOI] [PubMed] [Google Scholar]
- 33.Diez AIG, Fuster D, Morata L, Torres F, Garcia R, Poggio D, et al. Comparison of the diagnostic accuracy of diffusion-weighted and dynamic contrast-enhanced MRI with (18)F-FDG PET/CT to differentiate osteomyelitis from Charcot neuro-osteoarthropathy in diabetic foot. Eur J Radiol. 2020;132:109299. doi: 10.1016/j.ejrad.2020.109299. [DOI] [PubMed] [Google Scholar]
- 34.Rastogi A, Bhattacharya A, Prakash M, Sharma S, Mittal BR, Khandelwal N, et al. Utility of PET/CT with fluorine-18-fluorodeoxyglucose-labeled autologous leukocytes for diagnosing diabetic foot osteomyelitis in patients with Charcot’s neuroarthropathy. Nucl Med Commun. 2016;37(12):1253–1259. doi: 10.1097/MNM.0000000000000603. [DOI] [PubMed] [Google Scholar]
- 35.Glaudemans AW, Prandini N, Girolamo MDI, Argento G, Lauri C, Lazzeri E, et al. Hybrid imaging of musculoskeletal infections. Q J Nucl Med Mol Imaging. 2018;62(1):3–13. doi: 10.23736/S1824-4785.17.03045-X. [DOI] [PubMed] [Google Scholar]
- 36.Glaudemans A, Jutte PC, Cataldo MA, Cassar-Pullicino V, Gheysens O, Borens O, et al. Consensus document for the diagnosis of peripheral bone infection in adults: a joint paper by the EANM, EBJIS, and ESR (with ESCMID endorsement) Eur J Nucl Med Mol Imaging. 2019;46(4):957–970. doi: 10.1007/s00259-019-4262-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Signore A, Sconfienza LM, Borens O, Glaudemans A, Cassar-Pullicino V, Trampuz A, et al. Consensus document for the diagnosis of prosthetic joint infections: a joint paper by the EANM, EBJIS, and ESR (with ESCMID endorsement) Eur J Nucl Med Mol Imaging. 2019;46(4):971–988. doi: 10.1007/s00259-019-4263-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zoccali C, Teori G, Salducca N. The role of FDG-PET in distinguishing between septic and aseptic loosening in hip prosthesis: a review of literature. Int Orthop. 2009;33(1):1–5. doi: 10.1007/s00264-008-0575-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Manthey N, Reinhard P, Moog F, Knesewitsch P, Hahn K, Tatsch K. The use of [18 F]fluorodeoxyglucose positron emission tomography to differentiate between synovitis, loosening and infection of hip and knee prostheses. Nucl Med Commun. 2002;23(7):645–653. doi: 10.1097/00006231-200207000-00009. [DOI] [PubMed] [Google Scholar]
- 40.Slart R, Glaudemans A, Lancellotti P, Hyafil F, Blankstein R, Schwartz RG, et al. A joint procedural position statement on imaging in cardiac sarcoidosis: from the Cardiovascular and Inflammation & Infection Committees of the European Association of Nuclear Medicine, the European Association of Cardiovascular Imaging, and the American Society of Nuclear Cardiology. J Nucl Cardiol. 2018;25(1):298–319. doi: 10.1007/s12350-017-1043-4. [DOI] [PubMed] [Google Scholar]
- 41.Govaert GA, IJpma FF, McNally M, McNally E, Reininga IH, Glaudemans AW. Accuracy of diagnostic imaging modalities for peripheral post-traumatic osteomyelitis—a systematic review of the recent literature. Eur J Nucl Med Mol Imaging. 2017;44(8):1393–1407. doi: 10.1007/s00259-017-3683-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Basu S, Kwee TC, Saboury B, Garino JP, Nelson CL, Zhuang H, et al. FDG PET for diagnosing infection in hip and knee prostheses: prospective study in 221 prostheses and subgroup comparison with combined (111)In-labeled leukocyte/(99m)Tc-sulfur colloid bone marrow imaging in 88 prostheses. Clin Nucl Med. 2014;39(7):609–615. doi: 10.1097/RLU.0000000000000464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zhuang H, Chacko TK, Hickeson M, Stevenson K, Feng Q, Ponzo F, et al. Persistent non-specific FDG uptake on PET imaging following hip arthroplasty. Eur J Nucl Med Mol Imaging. 2002;29(10):1328–1333. doi: 10.1007/s00259-002-0886-2. [DOI] [PubMed] [Google Scholar]
- 44.Love C, Palestro CJ. Nuclear medicine imaging of bone infections. Clin Radiol. 2016;71(7):632–646. doi: 10.1016/j.crad.2016.01.003. [DOI] [PubMed] [Google Scholar]
- 45.Romanò CL, Petrosillo N, Argento G, Sconfienza LM, Treglia G, Alavi A, et al. The role of imaging techniques to define a peri-prosthetic hip and knee joint infection: multidisciplinary consensus statements. J Clin Med. 2020;9(8):2548. doi: 10.3390/jcm9082548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Glaudemans AW, de Vries EF, Galli F, Dierckx RA, Slart RH, Signore A. The use of (18)F-FDG-PET/CT for diagnosis and treatment monitoring of inflammatory and infectious diseases. Clin Dev Immunol. 2013;2013:623036. doi: 10.1155/2013/623036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Versari A, Pipitone N, Casali M, Jamar F, Pazzola G. Use of imaging techniques in large vessel vasculitis and related conditions. Q J Nucl Med Mol Imaging. 2018;62(1):34–39. doi: 10.23736/S1824-4785.17.03044-8. [DOI] [PubMed] [Google Scholar]
- 48.Meller J, Sahlmann CO, Gürocak O, Liersch T, Meller B. FDG-PET in patients with fever of unknown origin: the importance of diagnosing large vessel vasculitis. Q J Nucl Med Mol Imaging. 2009;53(1):51–63. [PubMed] [Google Scholar]
- 49.Jennette JC, Falk RJ, Bacon PA, Basu N, Cid MC, Ferrario F, et al. 2012 revised international chapel hill consensus conference nomenclature of vasculitides. Arthritis Rheum. 2013;65(1):1–11. doi: 10.1002/art.37715. [DOI] [PubMed] [Google Scholar]
- 50.Muratore F, Pipitone N, Salvarani C, Schmidt WA. Imaging of vasculitis: state of the art. Best Pract Res Clin Rheumatol. 2016;30(4):688–706. doi: 10.1016/j.berh.2016.09.010. [DOI] [PubMed] [Google Scholar]
- 51.Kermani TA, Crowson CS, Muratore F, Schmidt J, Matteson EL, Warrington KJ. Extra-cranial giant cell arteritis and Takayasu arteritis: how similar are they? Semin Arthritis Rheum. 2015;44(6):724–728. doi: 10.1016/j.semarthrit.2015.01.005. [DOI] [PubMed] [Google Scholar]
- 52.Muratore F, Kermani TA, Crowson CS, Green AB, Salvarani C, Matteson EL, et al. Large-vessel giant cell arteritis: a cohort study. Rheumatology (Oxford) 2015;54(3):463–470. doi: 10.1093/rheumatology/keu329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Soussan M, Nicolas P, Schramm C, Katsahian S, Pop G, Fain O, et al. Management of large-vessel vasculitis with FDG-PET: a systematic literature review and meta-analysis. Medicine (Baltimore) 2015;94(14):e622. doi: 10.1097/MD.0000000000000622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Pelletier-Galarneau M, Ruddy TD. PET/CT for diagnosis and management of large-vessel vasculitis. Curr Cardiol Rep. 2019;21(5):34. doi: 10.1007/s11886-019-1122-z. [DOI] [PubMed] [Google Scholar]
- 55.Blockmans D, de Ceuninck L, Vanderschueren S, Knockaert D, Mortelmans L, Bobbaers H. Repetitive 18F-fluorodeoxyglucose positron emission tomography in giant cell arteritis: a prospective study of 35 patients. Arthritis Rheum. 2006;55(1):131–137. doi: 10.1002/art.21699. [DOI] [PubMed] [Google Scholar]
- 56.Prieto-González S, Espígol-Frigolé G, García-Martínez A, Alba MA, Tavera-Bahillo I, Hernández-Rodríguez J, et al. The expanding role of imaging in systemic vasculitis. Rheum Dis Clin N Am. 2016;42(4):733–751. doi: 10.1016/j.rdc.2016.07.009. [DOI] [PubMed] [Google Scholar]
- 57.Lehmann P, Buchtala S, Achajew N, Haerle P, Ehrenstein B, Lighvani H, et al. 18F-FDG PET as a diagnostic procedure in large vessel vasculitis-a controlled, blinded re-examination of routine PET scans. Clin Rheumatol. 2011;30(1):37–42. doi: 10.1007/s10067-010-1598-9. [DOI] [PubMed] [Google Scholar]
- 58.Blockmans D, De Ceuninck L, Vanderschueren S, Knockaert D, Mortelmans L, Bobbaers H. Repetitive 18-fluorodeoxyglucose positron emission tomography in isolated polymyalgia rheumatica: a prospective study in 35 patients. Rheumatology (Oxford) 2007;46(4):672–677. doi: 10.1093/rheumatology/kel376. [DOI] [PubMed] [Google Scholar]
- 59.Slart R. FDG-PET/CT(A) imaging in large vessel vasculitis and polymyalgia rheumatica: joint procedural recommendation of the EANM, SNMMI, and the PET Interest Group (PIG), and endorsed by the ASNC. Eur J Nucl Med Mol Imaging. 2018;45(7):1250–1269. doi: 10.1007/s00259-018-3973-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Einspieler I, Thürmel K, Pyka T, Eiber M, Wolfram S, Moog P, et al. Imaging large vessel vasculitis with fully integrated PET/MRI: a pilot study. Eur J Nucl Med Mol Imaging. 2015;42(7):1012–1024. doi: 10.1007/s00259-015-3007-8. [DOI] [PubMed] [Google Scholar]
- 61.Wilson WR, Bower TC, Creager MA, Amin-Hanjani S, O’Gara PT, Lockhart PB, et al. Vascular graft infections, mycotic aneurysms, and endovascular infections: a scientific statement from the American Heart Association. Circulation. 2016;134(20):e412–e460. doi: 10.1161/CIR.0000000000000457. [DOI] [PubMed] [Google Scholar]
- 62.Kilic A, Arnaoutakis DJ, Reifsnyder T, Black JH, 3rd, Abularrage CJ, Perler BA, et al. Management of infected vascular grafts. Vasc Med. 2016;21(1):53–60. doi: 10.1177/1358863X15612574. [DOI] [PubMed] [Google Scholar]
- 63.FitzGerald SF, Kelly C, Humphreys H. Diagnosis and treatment of prosthetic aortic graft infections: confusion and inconsistency in the absence of evidence or consensus. J Antimicrob Chemother. 2005;56(6):996–999. doi: 10.1093/jac/dki382. [DOI] [PubMed] [Google Scholar]
- 64.Lyons OT, Baguneid M, Barwick TD, Bell RE, Foster N, Homer-Vanniasinkam S, et al. Diagnosis of aortic graft infection: a case definition by the Management of Aortic Graft Infection Collaboration (MAGIC) Eur J Vasc Endovasc Surg. 2016;52(6):758–763. doi: 10.1016/j.ejvs.2016.09.007. [DOI] [PubMed] [Google Scholar]
- 65.Lauri C, Iezzi R, Rossi M, Tinelli G, Sica S, Signore A, et al. Imaging modalities for the diagnosis of vascular graft infections: a consensus paper amongst different specialists. J Clin Med. 2020;9(5):1510. doi: 10.3390/jcm9051510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Reinders Folmer EI, Von Meijenfeldt GCI, Van der Laan MJ, Glaudemans A, Slart R, Saleem BR, et al. Diagnostic imaging in vascular graft infection: a systematic review and meta-analysis. Eur J Vasc Endovasc Surg. 2018;56(5):719–729. doi: 10.1016/j.ejvs.2018.07.010. [DOI] [PubMed] [Google Scholar]
- 67.Signore A, Lauri C, Galli F. Radiolabeled probes targeting infection and inflammation for personalized medicine. Curr Pharm Des. 2014;20(14):2338–2345. doi: 10.2174/13816128113196660666. [DOI] [PubMed] [Google Scholar]
- 68.Khaja MS, Sildiroglu O, Hagspiel K, Rehm PK, Cherry KJ, Turba UC. Prosthetic vascular graft infection imaging. Clin Imaging. 2013;37(2):239–244. doi: 10.1016/j.clinimag.2012.07.008. [DOI] [PubMed] [Google Scholar]
- 69.Kim SJ, Lee SW, Jeong SY, Pak K, Kim K. A systematic review and meta-analysis of (18)F-fluorodeoxyglucose positron emission tomography or positron emission tomography/computed tomography for detection of infected prosthetic vascular grafts. J Vasc Surg. 2019;70(1):307–313. doi: 10.1016/j.jvs.2019.01.051. [DOI] [PubMed] [Google Scholar]
- 70.Rojoa D, Kontopodis N, Antoniou SA, Ioannou CV, Antoniou GA. 18F-FDG PET in the diagnosis of vascular prosthetic graft infection: a diagnostic test accuracy meta-analysis. Eur J Vasc Endovasc Surg. 2019;57(2):292–301. doi: 10.1016/j.ejvs.2018.08.040. [DOI] [PubMed] [Google Scholar]
- 71.Spacek M, Belohlavek O, Votrubova J, Sebesta P, Stadler P. Diagnostics of “non-acute” vascular prosthesis infection using 18F-FDG PET/CT: our experience with 96 prostheses. Eur J Nucl Med Mol Imaging. 2009;36(5):850–858. doi: 10.1007/s00259-008-1002-z. [DOI] [PubMed] [Google Scholar]
- 72.Tokuda Y, Oshima H, Araki Y, Narita Y, Mutsuga M, Kato K, et al. Detection of thoracic aortic prosthetic graft infection with 18F-fluorodeoxyglucose positron emission tomography/computed tomography. Eur J Cardiothorac Surg. 2013;43(6):1183–1187. doi: 10.1093/ejcts/ezs693. [DOI] [PubMed] [Google Scholar]
- 73.Berger P, Vaartjes I, Scholtens A, Moll FL, De Borst GJ, De Keizer B, et al. Differential FDG-PET uptake patterns in uninfected and infected central prosthetic vascular grafts. Eur J Vasc Endovasc Surg. 2015;50(3):376–383. doi: 10.1016/j.ejvs.2015.06.007. [DOI] [PubMed] [Google Scholar]
- 74.Chang CY, Chang CP, Shih CC, Yang BH, Cheng CY, Chang CW, et al. Added value of dual-time-point 18F-FDG PET/CT with delayed imaging for detecting aortic graft infection: an observational study. Medicine (Baltimore) 2015;94(27):e1124. doi: 10.1097/MD.0000000000001124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Sah BR, Husmann L, Mayer D, Scherrer A, Rancic Z, Puippe G, et al. Diagnostic performance of 18F-FDG-PET/CT in vascular graft infections. Eur J Vasc Endovasc Surg. 2015;49(4):455–464. doi: 10.1016/j.ejvs.2014.12.024. [DOI] [PubMed] [Google Scholar]
- 76.Wassélius J, Malmstedt J, Kalin B, Larsson S, Sundin A, Hedin U, et al. High 18F-FDG Uptake in synthetic aortic vascular grafts on PET/CT in symptomatic and asymptomatic patients. J Nucl Med. 2008;49(10):1601–1605. doi: 10.2967/jnumed.108.053462. [DOI] [PubMed] [Google Scholar]
- 77.Guenther SP, Cyran CC, Rominger A, Saam T, Kazmierzcak PM, Bagaev E, et al. The relevance of 18F-fluorodeoxyglucose positron emission tomography/computed tomography imaging in diagnosing prosthetic graft infections post cardiac and proximal thoracic aortic surgery. Interact Cardiovasc Thorac Surg. 2015;21(4):450–458. doi: 10.1093/icvts/ivv178. [DOI] [PubMed] [Google Scholar]
- 78.Keidar Z, Engel A, Hoffman A, Israel O, Nitecki S. Prosthetic vascular graft infection: the role of 18F-FDG PET/CT. J Nucl Med. 2007;48(8):1230–1236. doi: 10.2967/jnumed.107.040253. [DOI] [PubMed] [Google Scholar]
- 79.Husmann L, Sah BR, Scherrer A, Burger IA, Stolzmann P, Weber R, et al. 18F-FDG PET/CT for therapy control in vascular graft infections: a first feasibility study. J Nucl Med. 2015;56(7):1024–1029. doi: 10.2967/jnumed.115.156265. [DOI] [PubMed] [Google Scholar]
- 80.Husmann L, Ledergerber B, Anagnostopoulos A, Stolzmann P, Sah BR, Burger IA, et al. The role of FDG PET/CT in therapy control of aortic graft infection. Eur J Nucl Med Mol Imaging. 2018;45(11):1987–1997. doi: 10.1007/s00259-018-4069-1. [DOI] [PubMed] [Google Scholar]
- 81.Dong W, Li Y, Zhu J, Xia J, He L, Yun M, et al. Detection of aortic prosthetic graft infection with (18)F-FDG PET/CT imaging, concordance with consensus MAGIC graft infection criteria. J Nucl Cardiol. 2020;28(3):1005–1016. doi: 10.1007/s12350-020-02227-9. [DOI] [PubMed] [Google Scholar]
- 82.Keidar Z, Pirmisashvili N, Leiderman M, Nitecki S, Israel O. 18F-FDG uptake in noninfected prosthetic vascular grafts: incidence, patterns, and changes over time. J Nucl Med. 2014;55(3):392–395. doi: 10.2967/jnumed.113.128173. [DOI] [PubMed] [Google Scholar]
- 83.Saleem BR, Berger P, Vaartjes I, de Keizer B, Vonken EJ, Slart RH, et al. Modest utility of quantitative measures in (18)F-fluorodeoxyglucose positron emission tomography scanning for the diagnosis of aortic prosthetic graft infection. J Vasc Surg. 2015;61(4):965–971. doi: 10.1016/j.jvs.2014.11.005. [DOI] [PubMed] [Google Scholar]
- 84.Bruggink JL, Glaudemans AW, Saleem BR, Meerwaldt R, Alkefaji H, Prins TR, et al. Accuracy of FDG-PET-CT in the diagnostic work-up of vascular prosthetic graft infection. Eur J Vasc Endovasc Surg. 2010;40(3):348–354. doi: 10.1016/j.ejvs.2010.05.016. [DOI] [PubMed] [Google Scholar]
- 85.Fukuchi K, Ishida Y, Higashi M, Tsunekawa T, Ogino H, Minatoya K, et al. Detection of aortic graft infection by fluorodeoxyglucose positron emission tomography: comparison with computed tomographic findings. J Vasc Surg. 2005;42(5):919–925. doi: 10.1016/j.jvs.2005.07.038. [DOI] [PubMed] [Google Scholar]
- 86.Zogala D, Rucka D, Ptacnik V, Cerny V, Trnka J, Varejka P, et al. How to recognize stent graft infection after endovascular aortic repair: the utility of 18F-FDG PET/CT in an infrequent but serious clinical setting. Ann Nucl Med. 2019;33(8):594–605. doi: 10.1007/s12149-019-01370-9. [DOI] [PubMed] [Google Scholar]
- 87.Yilmaz S, Aliyev A, Ekmekcioglu O, Ozhan M, Uslu L, Vatankulu B, et al. Comparison of FDG and FDG-labeled leukocytes PET/CT in diagnosis of infection. Nuklearmedizin. 2015;54(6):262–271. doi: 10.3413/Nukmed-0724-15-02. [DOI] [PubMed] [Google Scholar]
- 88.Sood A, Bhattacharya A, Aggarwal P, Basher RK, Mittal BR. 18F-FDG-labeled autologous leukocyte PET-CT in a patient with aortic valve-tube graft infection after Bentall procedure. Clin Nucl Med. 2019;44(3):e161–e162. doi: 10.1097/RLU.0000000000002447. [DOI] [PubMed] [Google Scholar]
- 89.Saleem BR, Beukinga RJ, Boellaard R, Glaudemans AW, Reijnen MM, Zeebregts CJ, et al. Textural features of (18)F-fluorodeoxyglucose positron emission tomography scanning in diagnosing aortic prosthetic graft infection. Eur J Nucl Med Mol Imaging. 2017;44(5):886–894. doi: 10.1007/s00259-016-3599-7. [DOI] [PubMed] [Google Scholar]
- 90.Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta JP, Del Zotti F, et al. 2015 ESC guidelines for the management of infective endocarditis: the task force for the management of infective endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM) Eur Heart J. 2015;36(44):3075–3128. doi: 10.1093/eurheartj/ehv319. [DOI] [PubMed] [Google Scholar]
- 91.Bernhardt AM, Pamirsad MA, Brand C, Reichart D, Tienken M, Barten MJ, et al. The value of fluorine-18 deoxyglucose positron emission tomography scans in patients with ventricular assist device specific infections†. Eur J Cardiothorac Surg. 2017;51(6):1072–1077. doi: 10.1093/ejcts/ezx016. [DOI] [PubMed] [Google Scholar]
- 92.Ahmed FZ, James J, Cunnington C, Motwani M, Fullwood C, Hooper J, et al. Early diagnosis of cardiac implantable electronic device generator pocket infection using 18F-FDG-PET/CT. Eur Heart J Cardiovasc Imaging. 2015;16(5):521–530. doi: 10.1093/ehjci/jeu295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Pizzi MN, Roque A, Fernández-Hidalgo N, Cuéllar-Calabria H, Ferreira-González I, Gonzàlez-Alujas MT, et al. Improving the diagnosis of infective endocarditis in prosthetic valves and intracardiac devices with 18F-fluordeoxyglucose positron emission tomography/computed tomography angiography: initial results at an infective endocarditis referral center. Circulation. 2015;132(12):1113–1126. doi: 10.1161/CIRCULATIONAHA.115.015316. [DOI] [PubMed] [Google Scholar]
- 94.Pizzi MN, Dos-Subirà L, Roque A, Fernández-Hidalgo N, Cuéllar-Calabria H, Domènech AP, et al. (18)F-FDG-PET/CT angiography in the diagnosis of infective endocarditis and cardiac device infection in adult patients with congenital heart disease and prosthetic material. Int J Cardiol. 2017;248:396–402. doi: 10.1016/j.ijcard.2017.08.008. [DOI] [PubMed] [Google Scholar]
- 95.Chen W, Kim J, Molchanova-Cook OP, Dilsizian V. The potential of FDG PET/CT for early diagnosis of cardiac device and prosthetic valve infection before morphologic damages ensue. Curr Cardiol Rep. 2014;16(3):459. doi: 10.1007/s11886-013-0459-y. [DOI] [PubMed] [Google Scholar]
- 96.Gomes A, Glaudemans A, Touw DJ, van Melle JP, Willems TP, Maass AH, et al. Diagnostic value of imaging in infective endocarditis: a systematic review. Lancet Infect Dis. 2017;17(1):e1–e14. doi: 10.1016/S1473-3099(16)30141-4. [DOI] [PubMed] [Google Scholar]
- 97.Saby L, Laas O, Habib G, Cammilleri S, Mancini J, Tessonnier L, et al. Positron emission tomography/computed tomography for diagnosis of prosthetic valve endocarditis: increased valvular 18F-fluorodeoxyglucose uptake as a novel major criterion. J Am Coll Cardiol. 2013;61(23):2374–2382. doi: 10.1016/j.jacc.2013.01.092. [DOI] [PubMed] [Google Scholar]
- 98.Rouzet F, Chequer R, Benali K, Lepage L, Ghodbane W, Duval X, et al. Respective performance of 18F-FDG PET and radiolabeled leukocyte scintigraphy for the diagnosis of prosthetic valve endocarditis. J Nucl Med. 2014;55(12):1980–1985. doi: 10.2967/jnumed.114.141895. [DOI] [PubMed] [Google Scholar]
- 99.Ricciardi A, Sordillo P, Ceccarelli L, Maffongelli G, Calisti G, Di Pietro B, et al. 18-fluoro-2-deoxyglucose positron emission tomography-computed tomography: an additional tool in the diagnosis of prosthetic valve endocarditis. Int J Infect Dis. 2014;28:219–224. doi: 10.1016/j.ijid.2014.04.028. [DOI] [PubMed] [Google Scholar]
- 100.Bartoletti M, Tumietto F, Fasulo G, Giannella M, Cristini F, Bonfiglioli R, et al. Combined computed tomography and fluorodeoxyglucose positron emission tomography in the diagnosis of prosthetic valve endocarditis: a case series. BMC Res Notes. 2014;7:32. doi: 10.1186/1756-0500-7-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Fagman E, van Essen M, Lindqvist JF, Snygg-Martin U, Bech-Hanssen O, Svensson G. 18F-FDG PET/CT in the diagnosis of prosthetic valve endocarditis. Int J Cardiovasc Imaging. 2016;32(4):679–686. doi: 10.1007/s10554-015-0814-8. [DOI] [PubMed] [Google Scholar]
- 102.Salomäki SP, Saraste A, Kemppainen J, Bax JJ, Knuuti J, Nuutila P, et al. (18)F-FDG positron emission tomography/computed tomography in infective endocarditis. J Nucl Cardiol. 2017;24(1):195–206. doi: 10.1007/s12350-015-0325-y. [DOI] [PubMed] [Google Scholar]
- 103.Pelletier-Galarneau M, Abikhzer G, Harel F, Dilsizian V. Detection of native and prosthetic valve endocarditis: incremental attributes of functional FDG PET/CT over morphologic imaging. Curr Cardiol Rep. 2020;22(9):93. doi: 10.1007/s11886-020-01334-w. [DOI] [PubMed] [Google Scholar]
- 104.Juneau D, Golfam M, Hazra S, Zuckier LS, Garas S, Redpath C, et al. Positron emission tomography and single-photon emission computed tomography imaging in the diagnosis of cardiac implantable electronic device infection: a systematic review and meta-analysis. Circ Cardiovasc Imaging. 2017;10(4):e005772. doi: 10.1161/CIRCIMAGING.116.005772. [DOI] [PubMed] [Google Scholar]
- 105.Erba PA, Pizzi MN, Roque A, Salaun E, Lancellotti P, Tornos P, et al. Multimodality imaging in infective endocarditis: an imaging team within the endocarditis team. Circulation. 2019;140(21):1753–1765. doi: 10.1161/CIRCULATIONAHA.119.040228. [DOI] [PubMed] [Google Scholar]
- 106.Ploux S, Riviere A, Amraoui S, Whinnett Z, Barandon L, Lafitte S, et al. Positron emission tomography in patients with suspected pacing system infections may play a critical role in difficult cases. Heart Rhythm. 2011;8(9):1478–1481. doi: 10.1016/j.hrthm.2011.03.062. [DOI] [PubMed] [Google Scholar]
- 107.Dell’Aquila AM, Mastrobuoni S, Alles S, Wenning C, Henryk W, Schneider SR, et al. Contributory role of fluorine 18-fluorodeoxyglucose positron emission tomography/computed tomography in the diagnosis and clinical management of infections in patients supported with a continuous-flow left ventricular assist device. Ann Thorac Surg. 2016;101(1):87–94. doi: 10.1016/j.athoracsur.2015.06.066. [DOI] [PubMed] [Google Scholar]
- 108.Birnie DH, Sauer WH, Bogun F, Cooper JM, Culver DA, Duvernoy CS, et al. HRS expert consensus statement on the diagnosis and management of arrhythmias associated with cardiac sarcoidosis. Heart Rhythm. 2014;11(7):1305–1323. doi: 10.1016/j.hrthm.2014.03.043. [DOI] [PubMed] [Google Scholar]
- 109.Youssef G, Beanlands RS, Birnie DH, Nery PB. Cardiac sarcoidosis: applications of imaging in diagnosis and directing treatment. Heart. 2011;97(24):2078–2087. doi: 10.1136/hrt.2011.226076. [DOI] [PubMed] [Google Scholar]
- 110.Slart RHJA, Glaudemans AWJM, Gheysens O, Lubberink M, Kero T, Dweck MR, et al. Procedural recommendations of cardiac PET/CT imaging: standardization in inflammatory-, infective-, infiltrative-, and innervation (4Is)-related cardiovascular diseases: a joint collaboration of the EACVI and the EANM. Eur J Nucl Med Mol Imaging. 2020;48:1016–1039. doi: 10.1007/s00259-020-05066-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Sarrazin JF, Philippon F, Tessier M, Guimond J, Molin F, Champagne J, et al. Usefulness of fluorine-18 positron emission tomography/computed tomography for identification of cardiovascular implantable electronic device infections. J Am Coll Cardiol. 2012;59(18):1616–1625. doi: 10.1016/j.jacc.2011.11.059. [DOI] [PubMed] [Google Scholar]
- 112.Leccisotti L, Perna F, Lago M, Leo M, Stefanelli A, Calcagni ML, et al. Cardiovascular implantable electronic device infection: delayed vs standard FDG PET-CT imaging. J Nucl Cardiol. 2014;21(3):622–632. doi: 10.1007/s12350-014-9896-2. [DOI] [PubMed] [Google Scholar]
- 113.Memmott MJ, James J, Armstrong IS, Tout D, Ahmed F. The performance of quantitation methods in the evaluation of cardiac implantable electronic device (CIED) infection: a technical review. J Nucl Cardiol. 2016;23(6):1457–1466. doi: 10.1007/s12350-015-0106-7. [DOI] [PubMed] [Google Scholar]
- 114.Flores RJ, Flaherty KR, Jin Z, Bokhari S. The prognostic value of quantitating and localizing F-18 FDG uptake in cardiac sarcoidosis. J Nucl Cardiol. 2020;27(6):2003–2010. doi: 10.1007/s12350-018-01504-y. [DOI] [PubMed] [Google Scholar]
- 115.Ohira H, Tsujino I, Ishimaru S, Oyama N, Takei T, Tsukamoto E, et al. Myocardial imaging with 18F-fluoro-2-deoxyglucose positron emission tomography and magnetic resonance imaging in sarcoidosis. Eur J Nucl Med Mol Imaging. 2008;35(5):933–941. doi: 10.1007/s00259-007-0650-8. [DOI] [PubMed] [Google Scholar]
- 116.Vita T, Okada DR, Veillet-Chowdhury M, Bravo PE, Mullins E, Hulten E, et al. Complementary value of cardiac magnetic resonance imaging and positron emission tomography/computed tomography in the assessment of cardiac sarcoidosis. Circ Cardiovasc Imaging. 2018;11(1):e007030. doi: 10.1161/CIRCIMAGING.117.007030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Nensa F, Kloth J, Tezgah E, Poeppel TD, Heusch P, Goebel J, et al. Feasibility of FDG-PET in myocarditis: comparison to CMR using integrated PET/MRI. J Nucl Cardiol. 2018;25(3):785–794. doi: 10.1007/s12350-016-0616-y. [DOI] [PubMed] [Google Scholar]
- 118.Tanaka T, Masumori N. Current approach to diagnosis and management of retroperitoneal fibrosis. Int J Urol. 2020;27(5):387–394. doi: 10.1111/iju.14218. [DOI] [PubMed] [Google Scholar]
- 119.Milojevic ITG, Milojevic B, Sobic-Saranovic DP, Artiko VM. Impact of hybrid molecular imaging in retroperitoneal fibrosis: a systematic review. Rheumatol Int. 2018;38(2):179–187. doi: 10.1007/s00296-017-3798-y. [DOI] [PubMed] [Google Scholar]
- 120.Guignard R, Simukoniene M, Garibotto V, Ratib O. 18F-FDG PET/CT and contrast-enhanced CT in a one-stop diagnostic procedure: a better strategy for management of patients suffering from retroperitoneal fibrosis? Clin Nucl Med. 2012;37(5):453–459. doi: 10.1097/RLU.0b013e3182478bd3. [DOI] [PubMed] [Google Scholar]
- 121.Vaglio A, Salvarani C, Buzio C. Retroperitoneal fibrosis. Lancet. 2006;367(9506):241–251. doi: 10.1016/S0140-6736(06)68035-5. [DOI] [PubMed] [Google Scholar]
- 122.Salvarani C, Pipitone N, Versari A, Vaglio A, Serafini D, Bajocchi G, et al. Positron emission tomography (PET): evaluation of chronic periaortitis. Arthritis Rheum. 2005;53(2):298–303. doi: 10.1002/art.21074. [DOI] [PubMed] [Google Scholar]
- 123.Vaglio A, Greco P, Versari A, Filice A, Cobelli R, Manenti L, et al. Post-treatment residual tissue in idiopathic retroperitoneal fibrosis: active residual disease or silent “scar” ? A study using 18F-fluorodeoxyglucose positron emission tomography. Clin Exp Rheumatol. 2005;23(2):231–234. [PubMed] [Google Scholar]
- 124.Nakajo M, Jinnouchi S, Tanabe H, Tateno R, Nakajo M. 18F-fluorodeoxyglucose positron emission tomography features of idiopathic retroperitoneal fibrosis. J Comput Assist Tomogr. 2007;31(4):539–543. doi: 10.1097/01.rct.0000284388.45579.05. [DOI] [PubMed] [Google Scholar]
- 125.Piccoli GB, Consiglio V, Arena V, Pelosi E, Anastasios D, Ragni F, et al. Positron emission tomography as a tool for the ‘tailored’ management of retroperitoneal fibrosis: a nephro-urological experience. Nephrol Dial Transplant. 2010;25(8):2603–2610. doi: 10.1093/ndt/gfq051. [DOI] [PubMed] [Google Scholar]
- 126.Treglia G, Mattoli MV, Bertagna F, Giubbini R, Giordano A. Emerging role of fluorine-18-fluorodeoxyglucose positron emission tomography in patients with retroperitoneal fibrosis: a systematic review. Rheumatol Int. 2013;33(3):549–555. doi: 10.1007/s00296-012-2576-0. [DOI] [PubMed] [Google Scholar]
- 127.Boellaard R, Oyen WJ, Hoekstra CJ, Hoekstra OS, Visser EP, Willemsen AT, et al. The Netherlands protocol for standardisation and quantification of FDG whole body PET studies in multi-centre trials. Eur J Nucl Med Mol Imaging. 2008;35(12):2320–2333. doi: 10.1007/s00259-008-0874-2. [DOI] [PubMed] [Google Scholar]
- 128.Ruhlmann V, Poeppel TD, Brandt AS, Grüneisen J, Ruhlmann M, Theysohn JM, et al. (18)F-FDG PET/MRI evaluation of retroperitoneal fibrosis: a simultaneous multiparametric approach for diagnosing active disease. Eur J Nucl Med Mol Imaging. 2016;43(9):1646–1652. doi: 10.1007/s00259-016-3351-3. [DOI] [PubMed] [Google Scholar]
- 129.Kouijzer IJE, Mulders-Manders CM, Bleeker-Rovers CP, Oyen WJG. Fever of unknown origin: the value of FDG-PET/CT. Semin Nucl Med. 2018;48(2):100–107. doi: 10.1053/j.semnuclmed.2017.11.004. [DOI] [PubMed] [Google Scholar]
- 130.Petersdorf RG, Beeson PB. Fever of unexplained origin: report on 100 cases. Medicine (Baltimore) 1961;40:1–30. doi: 10.1097/00005792-196102000-00001. [DOI] [PubMed] [Google Scholar]
- 131.Vanderschueren S, Del Biondo E, Ruttens D, Van Boxelaer I, Wauters E, Knockaert DD. Inflammation of unknown origin versus fever of unknown origin: two of a kind. Eur J Intern Med. 2009;20(4):415–418. doi: 10.1016/j.ejim.2009.01.002. [DOI] [PubMed] [Google Scholar]
- 132.Kan Y, Wang W, Liu J, Yang J, Wang Z. Contribution of 18F-FDG PET/CT in a case-mix of fever of unknown origin and inflammation of unknown origin: a meta-analysis. Acta Radiol. 2019;60(6):716–725. doi: 10.1177/0284185118799512. [DOI] [PubMed] [Google Scholar]
- 133.Loizidou A, Aoun M, Klastersky J. Fever of unknown origin in cancer patients. Crit Rev Oncol Hematol. 2016;101:125–130. doi: 10.1016/j.critrevonc.2016.02.015. [DOI] [PubMed] [Google Scholar]
- 134.Mulders-Manders CM, Simon A, Bleeker-Rovers CP. Rheumatologic diseases as the cause of fever of unknown origin. Best Pract Res Clin Rheumatol. 2016;30(5):789–801. doi: 10.1016/j.berh.2016.10.005. [DOI] [PubMed] [Google Scholar]
- 135.Takeuchi M, Dahabreh IJ, Nihashi T, Iwata M, Varghese GM, Terasawa T. Nuclear imaging for classic fever of unknown origin: meta-analysis. J Nucl Med. 2016;57(12):1913–1919. doi: 10.2967/jnumed.116.174391. [DOI] [PubMed] [Google Scholar]
- 136.Cascini GL, De Palma D, Matteucci F, Biggi A, Rambaldi PF, Signore A, et al. Fever of unknown origin, infection of subcutaneous devices, brain abscesses and endocarditis. Nucl Med Commun. 2006;27(3):213–222. doi: 10.1097/00006231-200603000-00003. [DOI] [PubMed] [Google Scholar]
- 137.Dong MJ, Zhao K, Liu ZF, Wang GL, Yang SY, Zhou GJ. A meta-analysis of the value of fluorodeoxyglucose-PET/PET-CT in the evaluation of fever of unknown origin. Eur J Radiol. 2011;80(3):834–844. doi: 10.1016/j.ejrad.2010.11.018. [DOI] [PubMed] [Google Scholar]
- 138.Kaya A, Ergul N, Kaya SY, Kilic F, Yilmaz MH, Besirli K, et al. The management and the diagnosis of fever of unknown origin. Expert Rev Anti Infect Ther. 2013;11(8):805–815. doi: 10.1586/14787210.2013.814436. [DOI] [PubMed] [Google Scholar]
- 139.Bharucha T, Rutherford A, Skeoch S, Alavi A, Brown M, Galloway J. Diagnostic yield of FDG-PET/CT in fever of unknown origin: a systematic review, meta-analysis, and Delphi exercise. Clin Radiol. 2017;72(9):764–771. doi: 10.1016/j.crad.2017.04.014. [DOI] [PubMed] [Google Scholar]
- 140.Takeuchi M, Nihashi T, Gafter-Gvili A, García-Gómez FJ, Andres E, Blockmans D, et al. Association of 18F-FDG PET or PET/CT results with spontaneous remission in classic fever of unknown origin: a systematic review and meta-analysis. Medicine (Baltimore) 2018;97(43):e12909. doi: 10.1097/MD.0000000000012909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Viscido A, Aratari A, Maccioni F, Signore A, Caprilli R. Inflammatory bowel diseases: clinical update of practical guidelines. Nucl Med Commun. 2005;26(7):649–655. doi: 10.1097/01.mnm.0000169205.21377.6a. [DOI] [PubMed] [Google Scholar]
- 142.Besson FL, Chaumet-Riffaud P, Playe M, Noel N, Lambotte O, Goujard C, et al. Contribution of (18)F-FDG PET in the diagnostic assessment of fever of unknown origin (FUO): a stratification-based meta-analysis. Eur J Nucl Med Mol Imaging. 2016;43(10):1887–1895. doi: 10.1007/s00259-016-3377-6. [DOI] [PubMed] [Google Scholar]
- 143.Pijl JP, Londema M, Kwee TC, Nijsten MWN, Slart R, Dierckx R, et al. FDG-PET/CT in intensive care patients with bloodstream infection. Crit Care. 2021;25(1):133. doi: 10.1186/s13054-021-03557-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Sioka C, Assimakopoulos A, Fotopoulos A. The diagnostic role of (18)F fluorodeoxyglucose positron emission tomography in patients with fever of unknown origin. Eur J Clin Invest. 2015;45(6):601–608. doi: 10.1111/eci.12439. [DOI] [PubMed] [Google Scholar]
- 145.Sobic-Saranovic D, Artiko V, Obradovic V. FDG PET imaging in sarcoidosis. Semin Nucl Med. 2013;43(6):404–411. doi: 10.1053/j.semnuclmed.2013.06.007. [DOI] [PubMed] [Google Scholar]
- 146.Yu WY, Lu PX, Assadi M, Huang XL, Skrahin A, Rosenthal A, et al. Updates on (18)F-FDG-PET/CT as a clinical tool for tuberculosis evaluation and therapeutic monitoring. Quant Imaging Med Surg. 2019;9(6):1132–1146. doi: 10.21037/qims.2019.05.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Yu WY, Zhang QQ, Xiao Y, Tan WG, Li XD, Lu PX. Correlation between 18F-FDG PET CT SUV and symptomatic or asymptomatic pulmonary tuberculosis. J Xray Sci Technol. 2019;27(5):899–906. doi: 10.3233/XST-190543. [DOI] [PubMed] [Google Scholar]
- 148.Valeyre D, Prasse A, Nunes H, Uzunhan Y, Brillet PY, Müller-Quernheim J. Sarcoidosis. Lancet. 2014;383(9923):1155–1167. doi: 10.1016/S0140-6736(13)60680-7. [DOI] [PubMed] [Google Scholar]
- 149.Skoura E, Zumla A, Bomanji J. Imaging in tuberculosis. Int J Infect Dis. 2015;32:87–93. doi: 10.1016/j.ijid.2014.12.007. [DOI] [PubMed] [Google Scholar]
- 150.Soussan M, Brillet PY, Mekinian A, Khafagy A, Nicolas P, Vessieres A, et al. Patterns of pulmonary tuberculosis on FDG-PET/CT. Eur J Radiol. 2012;81(10):2872–2876. doi: 10.1016/j.ejrad.2011.09.002. [DOI] [PubMed] [Google Scholar]
- 151.Ankrah AO, Glaudemans A, Maes A, Van de Wiele C, Dierckx R, Vorster M, et al. Tuberculosis. Semin Nucl Med. 2018;48(2):108–130. doi: 10.1053/j.semnuclmed.2017.10.005. [DOI] [PubMed] [Google Scholar]
- 152.Ungprasert P, Ryu JH, Matteson EL. Clinical manifestations, diagnosis, and treatment of sarcoidosis. Mayo Clin Proc Innov Qual Outcomes. 2019;3(3):358–375. doi: 10.1016/j.mayocpiqo.2019.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Nery PB, Beanlands RS, Nair GM, Green M, Yang J, McArdle BA, et al. Atrioventricular block as the initial manifestation of cardiac sarcoidosis in middle-aged adults. J Cardiovasc Electrophysiol. 2014;25(8):875–881. doi: 10.1111/jce.12401. [DOI] [PubMed] [Google Scholar]
- 154.Akaike G, Itani M, Shah H, Ahuja J, Gunes BY, Assaker R, et al. PET/CT in the diagnosis and workup of sarcoidosis: focus on atypical manifestations. Radiographics. 2018;38(5):1536–1549. doi: 10.1148/rg.2018180053. [DOI] [PubMed] [Google Scholar]
- 155.Malaisamy S, Dalal B, Bimenyuy C, Soubani AO. The clinical and radiologic features of nodular pulmonary sarcoidosis. Lung. 2009;187(1):9–15. doi: 10.1007/s00408-008-9118-2. [DOI] [PubMed] [Google Scholar]
- 156.Keijsers RGM, Grutters JC. In which patients with sarcoidosis is FDG PET/CT indicated? J Clin Med. 2020;9(3):890. doi: 10.3390/jcm9030890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Mostard RL, Vöö S, van Kroonenburgh MJ, Verschakelen JA, Wijnen PA, Nelemans PJ, et al. Inflammatory activity assessment by F18 FDG-PET/CT in persistent symptomatic sarcoidosis. Respir Med. 2011;105(12):1917–1924. doi: 10.1016/j.rmed.2011.08.012. [DOI] [PubMed] [Google Scholar]
- 158.Sobic-Saranovic D, Grozdic I, Videnovic-Ivanov J, Vucinic-Mihailovic V, Artiko V, Saranovic D, et al. The utility of 18F-FDG PET/CT for diagnosis and adjustment of therapy in patients with active chronic sarcoidosis. J Nucl Med. 2012;53(10):1543–1549. doi: 10.2967/jnumed.112.104380. [DOI] [PubMed] [Google Scholar]
- 159.Mostard RL, Verschakelen JA, van Kroonenburgh MJ, Nelemans PJ, Wijnen PA, Vöö S, et al. Severity of pulmonary involvement and (18)F-FDG PET activity in sarcoidosis. Respir Med. 2013;107(3):439–447. doi: 10.1016/j.rmed.2012.11.011. [DOI] [PubMed] [Google Scholar]
- 160.Kosterink JG. Positron emission tomography in the diagnosis and treatment management of tuberculosis. Curr Pharm Des. 2011;17(27):2875–2880. doi: 10.2174/138161211797470183. [DOI] [PubMed] [Google Scholar]
- 161.Sánchez-Montalvá A, Barios M, Salvador F, Villar A, Tórtola T, Molina-Morant D, et al. Usefulness of FDG PET/CT in the management of tuberculosis. PLoS ONE. 2019;14(8):e0221516. doi: 10.1371/journal.pone.0221516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kim IJ, Lee JS, Kim SJ, Kim YK, Jeong YJ, Jun S, et al. Double-phase 18F-FDG PET-CT for determination of pulmonary tuberculoma activity. Eur J Nucl Med Mol Imaging. 2008;35(4):808–814. doi: 10.1007/s00259-007-0585-0. [DOI] [PubMed] [Google Scholar]
- 163.Mostard RL, van Kroonenburgh MJ, Drent M. The role of the PET scan in the management of sarcoidosis. Curr Opin Pulm Med. 2013;19(5):538–544. doi: 10.1097/MCP.0b013e328363ed0d. [DOI] [PubMed] [Google Scholar]
- 164.Fu Y, Maianu L, Melbert BR, Garvey WT. Facilitative glucose transporter gene expression in human lymphocytes, monocytes, and macrophages: a role for GLUT isoforms 1, 3, and 5 in the immune response and foam cell formation. Blood Cells Mol Dis. 2004;32(1):182–190. doi: 10.1016/j.bcmd.2003.09.002. [DOI] [PubMed] [Google Scholar]
- 165.Mochizuki T, Tsukamoto E, Kuge Y, Kanegae K, Zhao S, Hikosaka K, et al. FDG uptake and glucose transporter subtype expressions in experimental tumor and inflammation models. J Nucl Med. 2001;42(10):1551–1555. [PubMed] [Google Scholar]
- 166.Lefebvre N, Argemi X, Meyer N, Mootien J, Douiri N, Sferrazza-Mandala S, et al. Clinical usefulness of (18)F-FDG PET/CT for initial staging and assessment of treatment efficacy in patients with lymph node tuberculosis. Nucl Med Biol. 2017;50:17–24. doi: 10.1016/j.nucmedbio.2017.04.003. [DOI] [PubMed] [Google Scholar]
- 167.Demura Y, Tsuchida T, Uesaka D, Umeda Y, Morikawa M, Ameshima S, et al. Usefulness of 18F-fluorodeoxyglucose positron emission tomography for diagnosing disease activity and monitoring therapeutic response in patients with pulmonary mycobacteriosis. Eur J Nucl Med Mol Imaging. 2009;36(4):632–639. doi: 10.1007/s00259-008-1009-5. [DOI] [PubMed] [Google Scholar]
- 168.Shariatmaghani S, Salari R, Sahebari M, Tabrizi PS, Salari M. Musculoskeletal manifestations of sarcoidosis: a review article. Curr Rheumatol Rev. 2019;15(2):83–89. doi: 10.2174/1573397114666180425111901. [DOI] [PubMed] [Google Scholar]
- 169.Mostard RL, Prompers L, Weijers RE, van Kroonenburgh MJ, Wijnen PA, Geusens PP, et al. F-18 FDG PET/CT for detecting bone and bone marrow involvement in sarcoidosis patients. Clin Nucl Med. 2012;37(1):21–25. doi: 10.1097/RLU.0b013e3182335f9b. [DOI] [PubMed] [Google Scholar]
- 170.Sathekge M, Maes A, Kgomo M, Stoltz A, Pottel H, Van de Wiele C. Impact of FDG PET on the management of TBC treatment. A pilot study. Nuklearmedizin. 2010;49(1):35–40. doi: 10.3413/nukmed-0270. [DOI] [PubMed] [Google Scholar]
- 171.Ambrosini V, Zompatori M, Fasano L, Nanni C, Nava S, Rubello D, et al. (18)F-FDG PET/CT for the assessment of disease extension and activity in patients with sarcoidosis: results of a preliminary prospective study. Clin Nucl Med. 2013;38(4):e171–e177. doi: 10.1097/RLU.0b013e31827a27df. [DOI] [PubMed] [Google Scholar]
- 172.Aide N, Allouache D, Ollivier Y, de Raucourt S, Switsers O, Bardet S. Early 2′-deoxy-2′-[18F]fluoro-D-glucose PET metabolic response after corticosteroid therapy to differentiate cancer from sarcoidosis and sarcoid-like lesions. Mol Imaging Biol. 2009;11(4):224–228. doi: 10.1007/s11307-008-0191-y. [DOI] [PubMed] [Google Scholar]
- 173.Vorselaars AD, Crommelin HA, Deneer VH, Meek B, Claessen AM, Keijsers RG, et al. Effectiveness of infliximab in refractory FDG PET-positive sarcoidosis. Eur Respir J. 2015;46(1):175–185. doi: 10.1183/09031936.00227014. [DOI] [PubMed] [Google Scholar]
- 174.Malherbe ST, Shenai S, Ronacher K, Loxton AG, Dolganov G, Kriel M, et al. Persisting positron emission tomography lesion activity and Mycobacterium tuberculosis mRNA after tuberculosis cure. Nat Med. 2016;22(10):1094–1100. doi: 10.1038/nm.4177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Martinez V, Castilla-Lievre MA, Guillet-Caruba C, Grenier G, Fior R, Desarnaud S, et al. (18)F-FDG PET/CT in tuberculosis: an early non-invasive marker of therapeutic response. Int J Tuberc Lung Dis. 2012;16(9):1180–1185. doi: 10.5588/ijtld.12.0010. [DOI] [PubMed] [Google Scholar]
- 176.Jeong YJ, Paeng JC, Nam HY, Lee JS, Lee SM, Yoo CG, et al. (18)F-FDG positron-emission tomography/computed tomography findings of radiographic lesions suggesting old healed tuberculosis. J Korean Med Sci. 2014;29(3):386–391. doi: 10.3346/jkms.2014.29.3.386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Thomas J, Shagos GS, Firuz I. Lambda sign in sarcoidosis using PET/CT. Clin Case Rep. 2019;7(1):236–237. doi: 10.1002/ccr3.1937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Oksüz MO, Werner MK, Aschoff P, Pfannenberg C. 18F-FDG PET/CT for the diagnosis of sarcoidosis in a patient with bilateral inflammatory involvement of the parotid and lacrimal glands (panda sign) and bilateral hilar and mediastinal lymphadenopathy (lambda sign) Eur J Nucl Med Mol Imaging. 2011;38(3):603. doi: 10.1007/s00259-010-1679-7. [DOI] [PubMed] [Google Scholar]
- 179.Keijsers RG, Grutters JC, Thomeer M, Du Bois RM, Van Buul MM, Lavalaye J, et al. Imaging the inflammatory activity of sarcoidosis: sensitivity and inter observer agreement of (67)Ga imaging and (18)F-FDG PET. Q J Nucl Med Mol Imaging. 2011;55(1):66–71. [PubMed] [Google Scholar]
- 180.Bois JP, Muser D, Chareonthaitawee P. PET/CT evaluation of cardiac sarcoidosis. PET Clin. 2019;14(2):223–232. doi: 10.1016/j.cpet.2018.12.004. [DOI] [PubMed] [Google Scholar]
- 181.Arepally GM. Heparin-induced thrombocytopenia. Blood. 2017;129(21):2864–2872. doi: 10.1182/blood-2016-11-709873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Rivas-Garcia A, Sarria-Estrada S, Torrents-Odin C, Casas-Gomila L, Franquet E. Imaging findings of Pott’s disease. Eur Spine J. 2013;22(Suppl 4):567–578. doi: 10.1007/s00586-012-2333-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Hara T, Kosaka N, Suzuki T, Kudo K, Niino H. Uptake rates of 18F-fluorodeoxyglucose and 11C-choline in lung cancer and pulmonary tuberculosis: a positron emission tomography study. Chest. 2003;124(3):893–901. doi: 10.1378/chest.124.3.893. [DOI] [PubMed] [Google Scholar]
- 184.Tang JL, Kung HC, Lei WC, Yao M, Wu UI, Hsu SC, et al. High incidences of invasive fungal infections in acute myeloid leukemia patients receiving induction chemotherapy without systemic antifungal prophylaxis: a prospective observational study in Taiwan. PLoS ONE. 2015;10(6):e0128410. doi: 10.1371/journal.pone.0128410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Sanguinetti M, Posteraro B, Beigelman-Aubry C, Lamoth F, Dunet V, Slavin M, et al. Diagnosis and treatment of invasive fungal infections: looking ahead. J Antimicrob Chemother. 2019;74(Suppl 2):ii27–ii37. doi: 10.1093/jac/dkz041. [DOI] [PubMed] [Google Scholar]
- 186.Patterson TF, Thompson GR, 3rd, Denning DW, Fishman JA, Hadley S, Herbrecht R, et al. Practice guidelines for the diagnosis and management of aspergillosis: 2016 update by the infectious diseases society of America. Clin Infect Dis. 2016;63(4):e1–e60. doi: 10.1093/cid/ciw326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Sharma P, Mukherjee A, Karunanithi S, Bal C, Kumar R. Potential role of 18F-FDG PET/CT in patients with fungal infections. AJR Am J Roentgenol. 2014;203(1):180–189. doi: 10.2214/AJR.13.11712. [DOI] [PubMed] [Google Scholar]
- 188.Douglas AP, Thursky KA, Worth LJ, Drummond E, Hogg A, Hicks RJ, et al. FDG PET/CT imaging in detecting and guiding management of invasive fungal infections: a retrospective comparison to conventional CT imaging. Eur J Nucl Med Mol Imaging. 2019;46(1):166–173. doi: 10.1007/s00259-018-4062-8. [DOI] [PubMed] [Google Scholar]
- 189.Davison JM, Subramaniam RM, Surasi DS, Cooley T, Mercier G, Peller PJ. FDG PET/CT in patients with HIV. AJR Am J Roentgenol. 2011;197(2):284–294. doi: 10.2214/AJR.10.6332. [DOI] [PubMed] [Google Scholar]
- 190.Kung BT, Seraj SM, Zadeh MZ, Rojulpote C, Kothekar E, Ayubcha C, et al. An update on the role of (18)F-FDG-PET/CT in major infectious and inflammatory diseases. Am J Nucl Med Mol Imaging. 2019;9(6):255–273. [PMC free article] [PubMed] [Google Scholar]
- 191.Zhuang H, Codreanu I. Growing applications of FDG PET-CT imaging in non-oncologic conditions. J Biomed Res. 2015;29(3):189–202. doi: 10.7555/JBR.29.20140081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Li Y, Wang Q, Wang X, Li X, Wu H, Wang Q, et al. Expert Consensus on clinical application of FDG PET/CT in infection and inflammation. Ann Nucl Med. 2020;34(5):369–376. doi: 10.1007/s12149-020-01449-8. [DOI] [PubMed] [Google Scholar]
- 193.Hammoud DA, Boulougoura A, Papadakis GZ, Wang J, Dodd LE, Rupert A, et al. Increased metabolic activity on 18F-fluorodeoxyglucose positron emission tomography-computed tomography in human immunodeficiency virus-associated immune reconstitution inflammatory syndrome. Clin Infect Dis. 2019;68(2):229–238. doi: 10.1093/cid/ciy454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Rahman WT, Wale DJ, Viglianti BL, Townsend DM, Manganaro MS, Gross MD, et al. The impact of infection and inflammation in oncologic (18)F-FDG PET/CT imaging. Biomed Pharmacother. 2019;117:109168. doi: 10.1016/j.biopha.2019.109168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Leroy-Freschini B, Treglia G, Argemi X, Bund C, Kessler R, Herbrecht R, et al. 18F-FDG PET/CT for invasive fungal infection in immunocompromised patients. QJM. 2018;111(9):613–622. doi: 10.1093/qjmed/hcy128. [DOI] [PubMed] [Google Scholar]
- 196.Hot A, Maunoury C, Poiree S, Lanternier F, Viard JP, Loulergue P, et al. Diagnostic contribution of positron emission tomography with [18F]fluorodeoxyglucose for invasive fungal infections. Clin Microbiol Infect. 2011;17(3):409–417. doi: 10.1111/j.1469-0691.2010.03301.x. [DOI] [PubMed] [Google Scholar]
- 197.Ankrah AO, Klein HC, Span LFR, de Vries EFJ, Dierckx R, Sathekge MM, et al. The role of PET in monitoring therapy in fungal infections. Curr Pharm Des. 2018;24(7):795–805. doi: 10.2174/1381612824666171213101648. [DOI] [PubMed] [Google Scholar]
- 198.Mhlanga JC, Durand D, Tsai HL, Durand CM, Leal JP, Wang H, et al. Differentiation of HIV-associated lymphoma from HIV-associated reactive adenopathy using quantitative FDG PET and symmetry. Eur J Nucl Med Mol Imaging. 2014;41(4):596–604. doi: 10.1007/s00259-013-2671-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Chang CC, Sheikh V, Sereti I, French MA. Immune reconstitution disorders in patients with HIV infection: from pathogenesis to prevention and treatment. Curr HIV/AIDS Rep. 2014;11(3):223–232. doi: 10.1007/s11904-014-0213-0. [DOI] [PubMed] [Google Scholar]
- 200.Boulougoura A, Sereti I. HIV infection and immune activation: the role of coinfections. Curr Opin HIV AIDS. 2016;11(2):191–200. doi: 10.1097/COH.0000000000000241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Caton MT, Jr, Miskin N, Hyun H. 18F-FDG uptake in subcutaneous fat preceding clinical diagnosis of human immunodeficiency virus-associated lipodystrophy. Clin Nucl Med. 2018;43(12):e475–e476. doi: 10.1097/RLU.0000000000002298. [DOI] [PubMed] [Google Scholar]
- 202.Chu H, Chan JF, Wang Y, Yuen TT, Chai Y, Hou Y, et al. Comparative replication and immune activation profiles of SARS-CoV-2 and SARS-CoV in human lungs: an ex vivo study with implications for the pathogenesis of COVID-19. Clin Infect Dis. 2020;71(6):1400–1409. doi: 10.1093/cid/ciaa410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Deng Y, Lei L, Chen Y, Zhang W. The potential added value of FDG PET/CT for COVID-19 pneumonia. Eur J Nucl Med Mol Imaging. 2020;47(7):1634–1635. doi: 10.1007/s00259-020-04767-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Guedj E, Cammilleri S, Verger A. Predictive medicine: towards a multi-parametric imaging for a personal risk stratification. Eur J Nucl Med Mol Imaging. 2017;44(2):196–198. doi: 10.1007/s00259-016-3522-2. [DOI] [PubMed] [Google Scholar]
- 205.Joob B, Wiwanitkit V. 18F-FDG PET/CT and COVID-19. Eur J Nucl Med Mol Imaging. 2020;47(6):1348. doi: 10.1007/s00259-020-04762-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Treglia G (2020) The role of (18)F-FDG PET for COVID-19 infection: myth versus reality. Clin Transl Imaging: 1–2. [DOI] [PMC free article] [PubMed]
- 207.Prokop M, van Everdingen W, van Rees Vellinga T, van Ufford HQ, Stöger L, Beenen L, et al. CO-RADS: a categorical CT assessment scheme for patients suspected of having COVID-19-definition and evaluation. Radiology. 2020;296(2):E97–E104. doi: 10.1148/radiol.2020201473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Chefer S, Thomasson D, Seidel J, Reba RC, Bohannon JK, Lackemeyer MG, et al. Modeling [(18)F]-FDG lymphoid tissue kinetics to characterize nonhuman primate immune response to middle east respiratory syndrome-coronavirus aerosol challenge. EJNMMI Res. 2015;5(1):65. doi: 10.1186/s13550-015-0143-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Jonsson CB, Camp JV, Wu A, Zheng H, Kraenzle JL, Biller AE, et al. Molecular imaging reveals a progressive pulmonary inflammation in lower airways in ferrets infected with 2009 H1N1 pandemic influenza virus. PLoS ONE. 2012;7(7):e40094. doi: 10.1371/journal.pone.0040094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Albano D, Bertagna F, Bertoli M, Bosio G, Lucchini S, Motta F, et al. Incidental findings suggestive of COVID-19 in asymptomatic patients undergoing nuclear medicine procedures in a high-prevalence region. J Nucl Med. 2020;61(5):632–636. doi: 10.2967/jnumed.120.246256. [DOI] [PubMed] [Google Scholar]
- 211.Albano D, Camoni L, Rinaldi R, Bertagna F, Giubbini R. 18F-FDG PET/CT metabolic behavior of COVID-19 pneumonia: a series of 4 patients with RT-PCR confirmation. Clin Nucl Med. 2020;45(8):e378–e380. doi: 10.1097/RLU.0000000000003150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Jones HA, Marino PS, Shakur BH, Morrell NW. In vivo assessment of lung inflammatory cell activity in patients with COPD and asthma. Eur Respir J. 2003;21(4):567–573. doi: 10.1183/09031936.03.00048502. [DOI] [PubMed] [Google Scholar]
- 213.Chung M, Bernheim A, Mei X, Zhang N, Huang M, Zeng X, et al. CT imaging features of 2019 novel coronavirus (2019-nCoV) Radiology. 2020;295(1):202–207. doi: 10.1148/radiol.2020200230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Qin C, Liu F, Yen TC, Lan X. (18)F-FDG PET/CT findings of COVID-19: a series of four highly suspected cases. Eur J Nucl Med Mol Imaging. 2020;47(5):1281–1286. doi: 10.1007/s00259-020-04734-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Wakfie-Corieh CG, Blanes García AM, Ferrando-Castagnetto F, Valhondo-Rama R, Ortega Candil A, Rodríguez Rey C, et al. Assessment of extra-parenchymal lung involvement in asymptomatic cancer patients with COVID-19 pneumonia detected on (18)F-FDG PET-CT studies. Eur J Nucl Med Mol Imaging. 2020;48(3):768–776. doi: 10.1007/s00259-020-05019-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Pallardy A, Rousseau C, Labbe C, Liberge R, Bodet-Milin C, Kraeber-Bodere F, et al. Incidental findings suggestive of COVID-19 in asymptomatic cancer patients undergoing 18F-FDG PET/CT in a low prevalence region. Eur J Nucl Med Mol Imaging. 2021;48(1):287–292. doi: 10.1007/s00259-020-05014-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Halsey R, Priftakis D, Mackenzie S, Wan S, Davis LM, Lilburn D, et al. COVID-19 in the act: incidental 18F-FDG PET/CT findings in asymptomatic patients and those with symptoms not primarily correlated with COVID-19 during the United Kingdom coronavirus lockdown. Eur J Nucl Med Mol Imaging. 2021;48(1):269–281. doi: 10.1007/s00259-020-04972-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Setti L, Kirienko M, Dalto SC, Bonacina M, Bombardieri E. FDG-PET/CT findings highly suspicious for COVID-19 in an Italian case series of asymptomatic patients. Eur J Nucl Med Mol Imaging. 2020;47(7):1649–1656. doi: 10.1007/s00259-020-04819-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Dietz M, Chironi G, Claessens YE, Farhad RL, Rouquette I, Serrano B, et al. COVID-19 pneumonia: relationship between inflammation assessed by whole-body FDG PET/CT and short-term clinical outcome. Eur J Nucl Med Mol Imaging. 2021;48(1):260–268. doi: 10.1007/s00259-020-04968-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Seoane-Viaño I, Gómez-Lado N, Lázare-Iglesias H, Rey-Bretal D, Lamela-Gómez I, Otero-Espinar FJ, et al. Evaluation of the therapeutic activity of melatonin and resveratrol in inflammatory bowel disease: a longitudinal PET/CT study in an animal model. Int J Pharm. 2019;15:118713. doi: 10.1016/j.ijpharm.2019.118713. [DOI] [PubMed] [Google Scholar]
- 221.Rubin DT, Ananthakrishnan AN, Siegel CA, Sauer BG, Long MD. ACG clinical guideline: ulcerative colitis in adults. Am J Gastroenterol. 2019;114:384–413. doi: 10.14309/ajg.0000000000000152. [DOI] [PubMed] [Google Scholar]
- 222.Kaaru E, Bianchi A, Wunder A, Rasche V, Stiller D. Molecular imaging in preclinical models of IBD with nuclear imaging techniques: state-of-the-art and perspectivess. Inflamm Bowel Dis. 2016;22:2491–2498. doi: 10.1097/MIB.0000000000000904. [DOI] [PubMed] [Google Scholar]
- 223.Dmochowska N, Wardill HA-O, Hughes PA. Advances in imaging specific mediators of inflammatory bowel disease. Int J Mol Sci. 2018 doi: 10.3390/ijms19092471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Bettenworth D, Reuter S, Hermann S, Weckesser M, Kerstiens L, Stratis A, Nowacki TM, et al. Translational 18F-FDG PET/CT imaging to monitor lesion activity in intestinal inflammation. J Nucl Med. 2013;54:748–755. doi: 10.2967/jnumed.112.112623. [DOI] [PubMed] [Google Scholar]
- 225.Caobelli F, Evangelista L, Quartuccio N, Familiari D, Altini C, Castello A, et al. Role of molecular imaging in the management of patients affected by inflammatory bowel disease: state-of-the-art. World J Radiol. 2016;8:829–845. doi: 10.4329/wjr.v8.i10.829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Perlman SB, Hall BS, Reichelderfer M. PET/CT imaging of inflammatory bowel disease. Semin Nucl Med. 2013;43:420–426. doi: 10.1053/j.semnuclmed.2013.06.006. [DOI] [PubMed] [Google Scholar]
- 227.Treglia G, Quartuccio N, Sadeghi R, Farchione A, Caldarella C, Bertagna F, Fania P, et al. Diagnostic performance of fluorine-18-fluorodeoxyglucose positron emission tomography in patients with chronic inflammatory bowel disease: a systematic review and a meta-analysis. J Crohns Colitis. 2013;7:345–354. doi: 10.1016/j.crohns.2012.08.005. [DOI] [PubMed] [Google Scholar]
- 228.Pellino G, Nicolai E, Catalano OA, Campione S, D'Armiento FP, Salvatore M, et al. PET/MR versus PET/CT imaging: impact on the clinical management of small-bowel Crohn’s disease. J Crohns Colitis. 2016;10:277–285. doi: 10.1093/ecco-jcc/jjv207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Glaudemans AW, Maccioni F, Mansi L, Dierckx RA, Signore A. Imaging of cell trafficking in Crohn’s disease. J Cell Physiol. 2010;223(3):562–571. doi: 10.1002/jcp.22069. [DOI] [PubMed] [Google Scholar]
- 230.Holtmann MH, Uenzen M, Helisch A, Dahmen A, Mudter J, Goetz M, et al. 18F-Fluorodeoxyglucose positron-emission tomography (PET) can be used to assess inflammation non-invasively in Crohn’s disease. Dig Dis Sci. 2012;57(10):2658–2668. doi: 10.1007/s10620-012-2190-8. [DOI] [PubMed] [Google Scholar]
- 231.Neurath MF, Vehling D, Schunk K, Holtmann M, Brockmann H, Helisch A, et al. Noninvasive assessment of Crohn’s disease activity: a comparison of 18F-fluorodeoxyglucose positron emission tomography, hydromagnetic resonance imaging, and granulocyte scintigraphy with labeled antibodies. Am J Gastroenterol. 2002;97(8):1978–1985. doi: 10.1111/j.1572-0241.2002.05836.x. [DOI] [PubMed] [Google Scholar]
- 232.Dorbala S, Ando Y, Bokhari S, Dispenzieri A, Falk RH, Ferrari VA, et al. ASNC/AHA/ASE/EANM/HFSA/ISA/SCMR/SNMMI expert consensus recommendations for multimodality imaging in cardiac amyloidosis: part 2 of 2-diagnostic criteria and appropriate utilization. J Cardiac Fail. 2019;25(11):854–865. doi: 10.1016/j.cardfail.2019.08.002. [DOI] [PubMed] [Google Scholar]
- 233.Chandler MB, Zeddun SM, Borum ML. The role of positron emission tomography in the evaluation of inflammatory bowel disease. Ann N Y Acad Sci. 2011;1228:59–63. doi: 10.1111/j.1749-6632.2011.06032.x. [DOI] [PubMed] [Google Scholar]
- 234.Domachevsky LA-O, Leibovitzh H, Avni-Biron I, Lichtenstein L, Goldberg N, Nidam M, et al. Correlation of 18F-FDG PET/MRE metrics with inflammatory biomarkers in patients with Crohn’s disease: a pilot study. Contrast Media Mol Imaging. 2017;2017:7167292. doi: 10.1155/2017/7167292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Catalano OA, Gee MS, Nicolai E, Selvaggi F, Pellino G, Cuocolo A, et al. Evaluation of quantitative PET/MR enterography biomarkers for discrimination of inflammatory strictures from fibrotic strictures in Crohn disease. Radiology. 2016;278:792–800. doi: 10.1148/radiol.2015150566. [DOI] [PubMed] [Google Scholar]
- 236.Bailey DL, Pichler BJ, Gückel B, Barthel H, Beer AJ, Botnar R, et al. Combined PET/MRI: from status quo to status go. Summary report of the fifth international workshop on PET/MR imaging; February 15–19, 2016; Tübingen, Germany. Mol Imaging Biol. 2016;18(5):637–650. doi: 10.1007/s11307-016-0993-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Glaudemans AW, Quintero AM, Signore A. PET/MRI in infectious and inflammatory diseases: will it be a useful improvement? Eur J Nucl Med Mol Imaging. 2012;39(5):745–749. doi: 10.1007/s00259-012-2060-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Deuschl CA-O, Rüber T, Ernst L, Fendler WP, Kirchner J, Mönninghoff CA-O, et al. 18F-FDG-PET/MRI in the diagnostic work-up of limbic encephalitis. PLoS ONE. 2020;15:e0227906. doi: 10.1371/journal.pone.0227906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Hanneman K, Kadoch M, Guo HH, Jamali M, Quon A, Iagaru A, Herfkens R, et al. Initial experience with simultaneous 18F-FDG PET/MRI in the evaluation of cardiac sarcoidosis and myocarditis. Clin Nucl Med. 2017;42:e328–e334. doi: 10.1097/RLU.0000000000001669. [DOI] [PubMed] [Google Scholar]
- 240.Wisenberg G, Thiessen JD, Pavlovsky W, Butler J, Wilk B, Prato FS. Same day comparison of PET/CT and PET/MR in patients with cardiac sarcoidosis. J Nucl Cardiol. 2020;27:2118–2129. doi: 10.1007/s12350-018-01578-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Laurent C, Ricard L, Fain O, Buvat I, Adedjouma A, Soussan M, et al. PET/MRI in large-vessel vasculitis: clinical value for diagnosis and assessment of disease activity. Sci Rep. 2019;9:12388. doi: 10.1038/s41598-019-48709-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Catalano O, Maccioni F, Lauri C, Auletta S, Dierckx R, Signore A. Hybrid imaging in Crohn’s disease: from SPECT/CT to PET/MR and new image interpretation criteria. Q J Nucl Med Mol Imaging. 2018;62(1):40–55. doi: 10.23736/S1824-4785.17.03053-9. [DOI] [PubMed] [Google Scholar]
- 243.Catalano OA, Wu V, Mahmood U, Signore A, Vangel M, Soricelli A, et al. Diagnostic performance of PET/MR in the evaluation of active inflammation in Crohn disease. Am J Nucl Med Mol Imaging. 2018;8(1):62–69. [PMC free article] [PubMed] [Google Scholar]
- 244.Maccioni F, Patak MA, Signore A, Laghi A. New frontiers of MRI in Crohn’s disease: motility imaging, diffusion-weighted imaging, perfusion MRI, MR spectroscopy, molecular imaging, and hybrid imaging (PET/MRI) Abdom Imaging. 2012;37(6):974–982. doi: 10.1007/s00261-012-9890-6. [DOI] [PubMed] [Google Scholar]
- 245.Servaes S. Imaging infection and inflammation in children with (18)F-FDG PET and (18)F-FDG PET/CT. J Nucl Med Technol. 2011;39:179–182. doi: 10.2967/jnmt.111.088385. [DOI] [PubMed] [Google Scholar]
- 246.Parisi MT, Otjen JP, Stanescu AL, Shulkin BL. Radionuclide imaging of infection and inflammation in children: a review. Semin Nucl Med. 2018;48:148–165. doi: 10.1053/j.semnuclmed.2017.11.002. [DOI] [PubMed] [Google Scholar]
- 247.Mojtahedi A, Penna D, Cistaro A. Fever of unknown origin. In: Cistaro A, editor. Atlas of PET/CT in pediatric patients. Milano: Springer Milan; 2014. pp. 245–247. [Google Scholar]
- 248.Pijl JA-O, Kwee TC, Legger GE, Peters HJH, Armbrust W, Schölvinck EH, et al. Role of FDG-PET/CT in children with fever of unknown origin. Eur J Nucl Med Mol Imaging. 2020;47:1596–1604. doi: 10.1007/s00259-020-04707-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 249.Pijl JP, Kwee TC, Legger GE, Peters HJH, Armbrust W, Schölvinck EH, et al. Role of FDG-PET/CT in children with fever of unknown origin. Eur J Nucl Med Mol Imaging. 2020;47(6):1596–1604. doi: 10.1007/s00259-020-04707-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Walter MA, Melzer RA, Schindler C, Müller-Brand J, Tyndall A, Nitzsche EU. The value of [18F]FDG-PET in the diagnosis of large-vessel vasculitis and the assessment of activity and extent of disease. Eur J Nucl Med Mol Imaging. 2005;32(6):674–681. doi: 10.1007/s00259-004-1757-9. [DOI] [PubMed] [Google Scholar]
- 251.Einspieler I, Henninger M, Mergen V, Wendorff H, Haller B, Beyer LP, et al. 18F-FDG PET/MRI compared with clinical and serological markers for monitoring disease activity in patients with aortitis and chronic periaortitis. Clin Exp Rheumatol. 2020;38:99–106. [PubMed] [Google Scholar]
- 252.Signore A, Glaudemans A, Gheysens O, Lauri C, Catalano OA. Nuclear medicine imaging in pediatric infection or chronic inflammatory diseases. Semin Nucl Med. 2017;47(3):286–303. doi: 10.1053/j.semnuclmed.2016.12.005. [DOI] [PubMed] [Google Scholar]
- 253.Yagupsky P, Porsch E, St Geme JW., 3rd Kingella kingae: an emerging pathogen in young children. Pediatrics. 2011;127:557–565. doi: 10.1542/peds.2010-1867. [DOI] [PubMed] [Google Scholar]
- 254.Malham M, Hess S, Nielsen RG, Husby S, Høilund-Carlsen PF. PET/CT in the diagnosis of inflammatory bowel disease in pediatric patients: a review. Am J Nucl Med Mol Imaging. 2014;4:225–230. [PMC free article] [PubMed] [Google Scholar]
- 255.Skehan SJ, Issenman R, Mernagh J, Nahmias C, Jacobson K. 18F-fluorodeoxyglucose positron tomography in diagnosis of paediatric inflammatory bowel disease. Lancet. 1999;354:836–837. doi: 10.1016/S0140-6736(99)02824-X. [DOI] [PubMed] [Google Scholar]
- 256.Ankrah AO, Sathekge MM, Dierckx RA, Glaudemans AW. Imaging fungal infections in children. Clin Transl Imaging. 2016;4:57–72. doi: 10.1007/s40336-015-0159-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Milanesi O, Stellin G, Zucchetta P. Nuclear medicine in pediatric cardiology. Semin Nucl Med. 2017;47(2):158–169. doi: 10.1053/j.semnuclmed.2016.10.008. [DOI] [PubMed] [Google Scholar]
- 258.Signore A, Anzola KL, Auletta S, Varani M, Petitti A, Pacilio M, et al. Current status of molecular imaging in inflammatory and autoimmune disorders. Curr Pharm Des. 2018;24:743–753. doi: 10.2174/1381612824666180130115153. [DOI] [PubMed] [Google Scholar]
- 259.Dolan RD, McLees NG, Irfan A, McSorley ST, Horgan PG, Colville D, et al. The relationship between tumor glucose metabolism and host systemic inflammatory responses in patients with cancer: a systematic review. J Nucl Med. 2019;60(4):467–471. doi: 10.2967/jnumed.118.216697. [DOI] [PubMed] [Google Scholar]
- 260.Cistaro A, Cassalia L, Ferrara C, Quartuccio N, Evangelista L, Bianchi M, et al. Italian multicenter study on accuracy of (18)F-FDG PET/CT in assessing bone marrow involvement in pediatric hodgkin lymphoma. Clin Lymphoma Myeloma Leuk. 2018;18(6):e267–e273. doi: 10.1016/j.clml.2018.04.002. [DOI] [PubMed] [Google Scholar]
- 261.Liu Y. Clinical significance of diffusedly increased splenic uptake on FDG-PET. Nucl Med Commun. 2009;30(10):763–769. doi: 10.1097/MNM.0b013e32832fa254. [DOI] [PubMed] [Google Scholar]
- 262.Stewart FA, Akleyev AV, Hauer-Jensen M, Hendry JH, Kleiman NJ, Macvittie TJ, et al. ICRP publication 118: ICRP statement on tissue reactions and early and late effects of radiation in normal tissues and organs–threshold doses for tissue reactions in a radiation protection context. Ann ICRP. 2012;41(1–2):1–322. doi: 10.1016/j.icrp.2012.02.001. [DOI] [PubMed] [Google Scholar]
- 263.Erba PA, Lancellotti P, Vilacosta I, Gaemperli O, Rouzet F, Hacker M, et al. Recommendations on nuclear and multimodality imaging in IE and CIED infections. Eur J Nucl Med Mol Imaging. 2018;45(10):1795–1815. doi: 10.1007/s00259-018-4025-0. [DOI] [PubMed] [Google Scholar]
- 264.Paez D, Gnanasegaran G, Fanti S, Bomanji J, Hacker M, Sathekge M, et al. COVID-19 pandemic: guidance for nuclear medicine departments. Eur J Nucl Med Mol Imaging. 2020;47(7):1615–1619. doi: 10.1007/s00259-020-04825-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Tulchinsky M, Osmany S. The American college of nuclear medicine guidance on operating procedures for a nuclear medicine facility during COVID-19 pandemic. Clin Nucl Med. 2020;46(7):571–574. doi: 10.1097/RLU.0000000000003146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Franquet E, Palmer MR, Gifford AE, Selen DJ, Chen Y-CS, Sedora-Roman N, Joyce RM, et al. Rifaximin suppresses background intestinal 18F-FDG uptake on PET/CT scans. Nucl Med Commun. 2014;35:1026–1031. doi: 10.1097/MNM.0000000000000170. [DOI] [PubMed] [Google Scholar]
- 267.Nobili FA-O, Arbizu J, Bouwman FA-O, Drzezga A, Agosta F, Nestor P, et al. European Association of Nuclear Medicine and European Academy of Neurology recommendations for the use of brain (18) F-fluorodeoxyglucose positron emission tomography in neurodegenerative cognitive impairment and dementia: Delphi consensus. Eur J Neurol. 2018;25(10):1201–1217. doi: 10.1111/ene.13728. [DOI] [PubMed] [Google Scholar]
- 268.Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W, et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42(2):328–354. doi: 10.1007/s00259-014-2961-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Bai X, Wang X, Zhuang H. Relationship between the elevated muscle FDG uptake in the distal upper extremities on PET/CT scan and prescan utilization of mobile devices in young patients. Clin Nucl Med. 2018;43(3):168–173. doi: 10.1097/RLU.0000000000001967. [DOI] [PubMed] [Google Scholar]
- 270.Lassmann M, Biassoni L, Monsieurs M, Franzius C. The new EANM paediatric dosage card: additional notes with respect to F-18. Eur J Nucl Med Mol Imaging. 2008;35(9):1666–1668. doi: 10.1007/s00259-008-0799-9. [DOI] [PubMed] [Google Scholar]
- 271.Brown TL, Spencer HJ, Beenken KE, Alpe TL, Bartel TB, Bellamy W, et al. Evaluation of dynamic [18F]-FDG-PET imaging for the detection of acute post-surgical bone infection. PLoS ONE. 2012;7(7):e41863. doi: 10.1371/journal.pone.0041863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Gunes BY, Onsel C, Sonmezoglu K, Ozaras R, Halac M, Tabak F, et al. Diagnostic value of F-18 FDG PET/CT in patients with spondylodiscitis: is dual time point imaging time worthy? Diagn Microbiol Infect Dis. 2016;85(3):381–385. doi: 10.1016/j.diagmicrobio.2016.03.021. [DOI] [PubMed] [Google Scholar]
- 273.Treglia G, Pascale M, Lazzeri E, van der Bruggen W, Delgado Bolton RC, Glaudemans A. Diagnostic performance of (18)F-FDG PET/CT in patients with spinal infection: a systematic review and a bivariate meta-analysis. Eur J Nucl Med Mol Imaging. 2020;47(5):1287–1301. doi: 10.1007/s00259-019-04571-6. [DOI] [PubMed] [Google Scholar]
- 274.D’Aprile P, Nasuto M, Tarantino A, Cornacchia S, Guglielmi G, Jinkins JR. Magnetic resonance imaging in degenerative disease of the lumbar spine: fat saturation technique and contrast medium. Acta Biomed. 2018;89:208–219. doi: 10.23750/abm.v89i1-S.7024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Gotthardt M, Bleeker-Rovers CP, Boerman OC, Oyen WJ. Imaging of inflammation by PET, conventional scintigraphy, and other imaging techniques. J Nucl Med. 2010;51(12):1937–1949. doi: 10.2967/jnumed.110.076232. [DOI] [PubMed] [Google Scholar]
- 276.Puppo C, Massollo M, Paparo F, Camellino D, Piccardo A, Naseri MSZ, et al. Giant cell arteritis: a systematic review of the qualitative and semiquantitative methods to assess vasculitis with 18F-fluorodeoxyglucose positron emission tomography. Biomed Res Int. 2014;2014:574248. doi: 10.1155/2014/574248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Osborne MT, Hulten EA, Murthy VL, Skali H, Taqueti VR, Dorbala S, et al. Patient preparation for cardiac fluorine-18 fluorodeoxyglucose positron emission tomography imaging of inflammation. J Nucl Cardiol. 2017;24(1):86–99. doi: 10.1007/s12350-016-0502-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Moroni G, Castellani M, Balzani A, Dore R, Bonelli N, Longhi S, et al. The value of (18)F-FDG PET/CT in the assessment of active idiopathic retroperitoneal fibrosis. Eur J Nucl Med Mol Imaging. 2012;39(10):1635–1642. doi: 10.1007/s00259-012-2144-6. [DOI] [PubMed] [Google Scholar]
- 279.Deppen SA, Blume JD, Kensinger CD, Morgan AM, Aldrich MC, Massion PP, et al. Accuracy of FDG-PET to diagnose lung cancer in areas with infectious lung disease: a meta-analysis. JAMA. 2014;312(12):1227–1236. doi: 10.1001/jama.2014.11488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Artigas C, Lemort M, Mestrez F, Gil T, Flamen P. COVID-19 pneumonia mimicking immunotherapy-induced pneumonitis on 18F-FDG PET/CT in a patient under treatment with nivolumab. Clin Nucl Med. 2020;45(8):e381–e382. doi: 10.1097/RLU.0000000000003152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.García Vicente AM, Castrejón ÁS. Incidental COVID-19 pneumonia on 18F-fluorocholine PET/CT. Clin Nucl Med. 2020;45:376–377. doi: 10.1097/RLU.0000000000003189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 282.Savelli G, Bonacina M, Rizzo A, Zaniboni A. Activated macrophages are the main inflammatory cell in COVID-19 interstitial pneumonia infiltrates. Is it possible to show their metabolic activity and thus the grade of inflammatory burden with (18)F-fluorocholine PET/CT? Med Hypotheses. 2020;144:109885. doi: 10.1016/j.mehy.2020.109885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Stasiak CA-O, Cardoso FR, de Almeida SA-O, Rosado-de-Castro PA-O. Incidental finding of COVID-19 infection after [(68)Ga]Ga-PSMA-11 PET/CT imaging in a patient with prostate cancer. Eur J Nucl Med Mol Imaging. 2021;48:653–654. doi: 10.1007/s00259-020-04932-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Keidar Z. FDG PET/CT imaging in diabetic patients—a special emphasis on imaging of infection. Curr Pharm Des. 2018;24:806–813. doi: 10.2174/1381612824666171129202647. [DOI] [PubMed] [Google Scholar]
- 285.Brodersen JB, Hess S. FDG-PET/CT in inflammatory bowel disease: is there a future? PET Clin. 2020;15:153–162. doi: 10.1016/j.cpet.2019.11.006. [DOI] [PubMed] [Google Scholar]
- 286.Sturm E, Rings EH, Schölvinck EH, Gouw AS, Porte RJ, Pruim J. Fluordeoxyglucose positron emission tomography contributes to management of pediatric liver transplantation candidates with fever of unknown origin. Liver Transpl. 2006;12(11):1698–1704. doi: 10.1002/lt.20922. [DOI] [PubMed] [Google Scholar]
- 287.Shon IH, Fogelman I. F-18 FDG positron emission tomography and benign fractures. Clin Nucl Med. 2003;28(3):171–175. doi: 10.1097/01.RLU.0000053508.98025.01. [DOI] [PubMed] [Google Scholar]
- 288.Pagano M, Berta M, Postini AM, Bianchi M, Del Prever AB, Defilippi C, et al. Nonossifying fibroma: a possible pitfall in F18-FD-PET/CT imaging of Hodgkin’s disease. Radiol Case Rep. 2011;6(2):271. doi: 10.2484/rcr.v6i2.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Lindholm H, Johansson O, Jonsson C, Jacobsson H. The distribution of FDG at PET examinations constitutes a relative mechanism: significant effects at activity quantification in patients with a high muscular uptake. Eur J Nucl Med Mol Imaging. 2012;39(11):1685–1690. doi: 10.1007/s00259-012-2202-0. [DOI] [PubMed] [Google Scholar]
- 290.Shellock FG, Crues JV. MR procedures: biologic effects, safety, and patient care. Radiology. 2004;232:635–652. doi: 10.1148/radiol.2323030830. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.