Abstract
Objective
Tracheal intubation and mechanical ventilation provide essential support for patients with respiratory failure, but the course of mechanical ventilation may be complicated by adverse ventilator-associated events (VAE). VAE may be infection-associated or not. We sought to understand how the frequency of subglottic suction, an indicator of the quantity of sputum produced by ventilated patients, relates to onset of all VAE and infection-associated VAE.
Design
We performed a case-crossover study including 87 patients with VAE and evaluated 848 days in the pre-VAE period at risk for VAE.
Setting & Participants
Critically ill subjects were recruited from the medical intensive care unit of an academic medical center.
Methods
We used the number of as-needed subglottic suctioning events performed per calendar day to quantify sputum production and compared the immediate pre-VAE period to the preceding period. We used CDC surveillance definitions for VAE and to categorize whether events were infection-associated or not.
Results
Sputum quantity measured by subglottic suction frequency is greater in the period immediately prior to VAE, compared to the preceding period. However, it does not discriminate well between infection-associated VAE and VAE without associated infection.
Conclusions
Subglottic suction frequency may serve as a valuable marker of sputum quantity, and is associated with risk for VAE. However, our results require validation in a broader population of mechanically ventilated patients and intensive care settings.
Keywords: ventilator-associated complications, ventilator-associated pneumonia, subglottic suction
Introduction
Tracheal intubation and mechanical ventilation are life-saving interventions to address respiratory failure, but adverse events occur during the course of mechanical ventilation that can result in prolonged critical illness and increased risk of death. Traditional surveillance focused on infectious causes of adverse events, particularly ventilator-associated lower respiratory tract infection (VA-LRTI), including ventilator-associated pneumonia and tracheobronchitis. But other adverse events, such as congestive heart failure or septic shock, can also result in the need for increased ventilator support.
In 2013, the Centers for Disease Control and Prevention (CDC)’s National Healthcare Safety Network (NHSN) implemented a new surveillance strategy for adverse ventilator-associated events (VAE), which advanced a broader, objective definition of ventilator-associated complications (VAC).1–3 There are three tiers of VAC: (1) VAC without signs of infection; infection-associated VAC (IVAC), which are accompanied by leukocytosis/leukopenia or fever/hypothermia, as well as altered antibiotic treatment; and (3) possible ventilator-associated pneumonia (PVAP), which are IVAC with sputum studies suggestive of VA-LRTI. The NHSN definitions of VAE are well validated to predict ventilator-associated morbidity and mortality.4–6 However, current VAE surveillance definitions capture a different population than clinically defined VA-LRTI, which are typically diagnosed by physical examination findings such as sputum production, laboratory test results, radiographic findings, and microbiologic test results.7–18
Sputum production is a physical examination finding that is often used in the clinical diagnosis of VA-LRTI, but its relationship to VAE defined by CDC NHSN criteria is unknown. It is challenging to quantify sputum production during mechanical ventilation, but an indicator of sputum production is the frequency with which subglottic suction is performed. Subglottic suction is a procedure performed on an as-needed basis by nurses, respiratory therapists, and physicians to clear lower airway secretions via the endotracheal tube and reduce the risk of VA-LRTI.19–21 We sought to evaluate the relationship between the quantity of sputum produced, measured by the frequency of subglottic suctioning, and VAE. Specifically, we sought to define how subglottic suction frequency in the immediate pre-VAE period relates to subglottic suction frequency in the preceding period, and to define whether sputum quantity measured by subglottic suction frequency discriminates between VAC and IVAC/VAP. Here we report the results of a case-crossover study designed to address these questions.
Materials & Methods
Study Design
We performed a case-crossover study to define the relationship between the quantity of sputum production (primary exposure), measured as the frequency with which nurses and respiratory therapists chose to perform as-needed subglottic suctioning, and the occurrence of VAE (primary outcome). We also analyzed whether VAE were categorized as VAC without suspected infection or as IVAC/PVAP. For the primary analysis, the day of VAE served as the case period. The days prior to VAE during which subjects could have been diagnosed with VAE (i.e., after at least two days of stable or decreasing positive end-expiratory pressure (PEEP) and fraction of inspired oxygen (FiO2) requirements) served as the control period. As sensitivity analyses, we treated the day before VAE as the case period, or two days before VAE as case period, with the antecedent days during which VAE might have been diagnosed as the control period.
Study Setting
We evaluated 90 consecutive VAE that occurred across 87 unique subjects, over 3.5 years in the medical intensive care unit (MICU) of the Hospital of the University of Pennsylvania (HUP), a tertiary academic medical center. All patients who depend on mechanical ventilation at HUP were monitored for VAE, and all VAE were reported to NHSN. VAC and IVAC were captured by review of HUP NHSN reporting records. The study was approved by the University of Pennsylvania Institutional Review Board (protocol #824613) with a waiver of informed consent.
Study Population
Subjects were eligible for inclusion if they had been admitted to the HUP MICU, required mechanical ventilation for a period greater than two calendar days (i.e., at risk for VAE), and experienced a VAE event within the enrollment period. We chose a case-crossover design and mixed-effects modeling approach to account for the heterogeneity of lung disease and other medical comorbidities in this critically ill population.
Clinical Data Collection
VAE dates and VAC versus IVAC categorization were obtained from NSHN reporting records. Subject demographics, medical diagnoses, laboratory, and microbiology data were obtained from the Penn Data Store, a repository of electronic health record data. Subglottic suction events were extracted from daily flowsheets that report all activity related to mechanical ventilation.
Definition of Exposures
The primary exposure was the quantity of sputum production, defined as number of as-needed subglottic suction events performed per calendar day. Calendar days were used because VAE (outcome of interest) are adjudicated per calendar day.
Definition of Outcomes
The primary outcome was VAE, and we compared the frequency of subglottic suction on the day of VAE and in the preceding 48 hours to the pre-VAE period. To evaluate whether quantity of sputum discriminates between VAC and IVAC, we similarly compared subglottic suction frequency across VAE categories.
Precision and Sample Size
We determined the necessary sample size by specifying the desired precision with which to detect the effect of sputum quantity to discriminate the pre-VAE and VAE risk periods.22 We anticipated that with 85 subjects we could detect an absolute risk difference attributable to sputum quantity of 15% with posterior certainty 4% - 26%. With the same subject number, we anticipated detecting a 25% absolute risk of IVAC versus VAC attributable to sputum quantity, with 95% posterior certainty 4 – 45%.
Statistical Methods and Models
We organized data with R statistical software version 3.6.1 and generated plots using the “ggplot2” package.23 We evaluated potential confounders by comparing clinical variables of interest across exposure groups (high versus low frequency of subglottic suction at onset of mechanical ventilation) using Wilcoxon rank-sum testing (continuous variables) and Fisher’s exact test (categorical variables). For the primary analysis, we performed Bayesian multilevel logistic regression with random, subject-level slopes and intercepts to address the possibility of confounding by subject-level factors. Like conditional logistic regression, this approach accounts for subject-level strata in the case-crossover design.24–27 To permit partial pooling of parameter estimates and rigorous evaluation of model fit, Bayesian models were fit using Stan Hamiltonian Monte Carlo (HMC) version 2.19 via the “rethinking”, “rstan”, and “brms” packages.28–30 Models were fit with 4 chains of 2000 iterations, then confirmed with HMC diagnostics (no divergent iterations, no iterations saturating maximum tree depth, Rhat statistic < 1.1 for all parameters, and E-BFMI > 0.2), and by examining the posterior distributions.31 We characterized posterior credibility intervals and type S error.32
Availability of Data
Data, model code, and code used to produce the figures are available at https://github.com/bjklab/Subglottic-suction-frequency-and-adverse-events-during-mechanical-ventilation.
Results
Sputum quantity at onset of mechanical ventilation does not predict time to VAE.
We evaluated 90 consecutive VAE, which occurred across 87 unique subjects. For subjects with more than one VAE, we considered only the first VAE, so 87 VAE (47 VAC without associated infection; 40 IVAC, including 12 PVAP) were included in the final analysis. The median number of subglottic suction events at onset of mechanical ventilation (first full calendar day on ventilator) was 7. We first compared clinical characteristics across 39 subjects with high (greater than median) sputum quantity and 48 subjects with low (less than or equal to median) sputum quantity at the onset of mechanical ventilation (Table 1). We noted no significant differences in subject demographics, medical history, or features of critical illness at onset of mechanical ventilation. We did note a disparity in bacterial growth from respiratory culture in the 7-days prior to mechanical ventilation onset: only 1 (2.1%) of the subjects with low sputum quantity had recent bacterial growth from respiratory culture, versus 5 (13%) of subjects with high sputum quantity. But this difference was not statistically significant. We next evaluated the sputum quantity at onset of mechanical ventilation as a predictor of time to VAE. A Weibull parametric survival model showed no significant differences in time to VAE attributable to sputum quantity (Supplemental Figure 1).
Table 1. Comparison of clinical characteristics among subjects high or low sputum production at onset of mechanical ventilation.
Categorical variables are described as number (%); continuous variables are described as median (IQR). Categorical variables are compared with chi-square test or Fisher’s exact test; continuous variables are compared with Wilcoxon rank-sum test.
| Characteristic | Whole Cohort | 7 or Fewer Suction Episodes (N = 48) | More than 7 Suction Episodes (N = 39) | p-value |
|---|---|---|---|---|
| Demographics | ||||
| Age (years) | 58 (48, 66) | 60 (52, 66) | 53 (44, 66) | 0.2 |
| Gender | 0.57 | |||
| Male | 45 (52%) | 23 (48%) | 22 (56%) | |
| Female | 42 (48%) | 25 (52%) | 17 (44%) | |
| Race | 0.35 | |||
| Asian | 3 (3.4%) | 3 (6.2%) | 0 (0%) | |
| Black | 23 (26%) | 12 (25%) | 11 (28%) | |
| Unknown | 6 (6.9%) | 2 (4.2%) | 4 (10%) | |
| White | 55 (63%) | 31 (65%) | 24 (62%) | |
| Comorbidities | ||||
| Chronic Obstructive Pulmonary Disease (COPD) | 1 (1.1%) | 0 (0%) | 1 (2.6%) | 0.45 |
| Asthma | 13 (15%) | 10 (21%) | 3 (7.7%) | 0.16 |
| Interstitial Lung Disease (ILD) | 0 (0%) | 0 (0%) | 0 (0%) | |
| Lymphoma or Leukemia | 3 (3.4%) | 1 (2.1%) | 2 (5.1%) | 0.58 |
| Diabetes mellitus | 11 (13%) | 4 (8.3%) | 7 (18%) | 0.21 |
| Congestive Heart Failure | 26 (30%) | 14 (29%) | 12 (31%) | >0.99 |
| Cirrhosis | 12 (14%) | 8 (17%) | 4 (10%) | 0.58 |
| Features of Critical Illness | ||||
| Subglottic Suction Events at MV Onset | 7.0 (4.0, 9.5) | 4.0 (2.0, 6.0) | 10.0 (8.0, 11.5) | <0.001 |
| Duration of MV (days) | 15 (9, 29) | 15 (9, 27) | 16 (10, 31) | 0.36 |
| Ventilator Day of VAE | 7 (4, 11) | 7 (4, 11) | 8 (5, 11) | 0.28 |
| IVAC | 40 (46%) | 19 (40%) | 21 (54%) | 0.27 |
| PVAP | 12 (14%) | 5 (10%) | 7 (18%) | 0.48 |
| Fever Week Before MV Onset | 15 (17%) | 9 (19%) | 6 (15%) | 0.9 |
| Respiratory Culture Order Week Before MV Onset | 9 (10%) | 4 (8.3%) | 5 (13%) | 0.51 |
| Respiratory Culture Growth Week Before MV Onset | 6 (6.9%) | 1 (2.1%) | 5 (13%) | 0.085 |
| WBC at MV Onset (1e3 cells/mm3) | 13 (9, 19) | 13 (9, 18) | 12 (9, 19) | 0.79 |
| Serum Cr at MV Onset (mg/dL) | 1.22 (0.83, 2.08) | 1.13 (0.80, 2.01) | 1.33 (0.85, 2.14) | 0.57 |
| AST at MV Onset (units/L) | 41 (22, 74) | 42 (21, 110) | 41 (23, 53) | 0.44 |
| ALT at MV Onset (units/L) | 27 (16, 50) | 30 (18, 54) | 21 (15, 36) | 0.3 |
| Maximum FiO2 at MV Onset (%) | 100 (60, 100) | 100 (60, 100) | 100 (55, 100) | 0.76 |
| Maximum PEEP at MV Onset (cm H2O) | 7.50 (5.00, 10.00) | 7.25 (5.00, 10.00) | 7.50 (5.00, 10.00) | 0.67 |
| Norepinephrine at MV Onset | 43 (49%) | 25 (52%) | 18 (46%) | 0.74 |
| Epinephrine at MV Onset | 9 (10%) | 5 (10%) | 4 (10%) | >0.99 |
| Vasopression at MV Onset | 23 (26%) | 16 (33%) | 7 (18%) | 0.17 |
| Dopamine at MV Onset | 4 (4.6%) | 2 (4.2%) | 2 (5.1%) | >0.99 |
| Vancomycin (IV) in Week Before MV Onset | 27 (31%) | 16 (33%) | 11 (28%) | 0.78 |
| Metronidazole in Week Before MV Onset | 4 (4.6%) | 2 (4.2%) | 2 (5.1%) | >0.99 |
| Linezolid in Week Before MV Onset | 2 (2.3%) | 2 (4.2%) | 0 (0%) | 0.5 |
| Cefazolin in Week Before MV Onset | 1 (1.1%) | 0 (0%) | 1 (2.6%) | 0.45 |
| Piperacillin-tazobactam in Week Before MV Onset | 13 (15%) | 7 (15%) | 6 (15%) | >0.99 |
| Cefepime in Week Before MV Onset | 12 (14%) | 5 (10%) | 7 (18%) | 0.48 |
| Meropenem in Week Before MV Onset | 6 (6.9%) | 3 (6.2%) | 3 (7.7%) | >0.99 |
Sputum quantity increases in the immediate pre-VAE period.
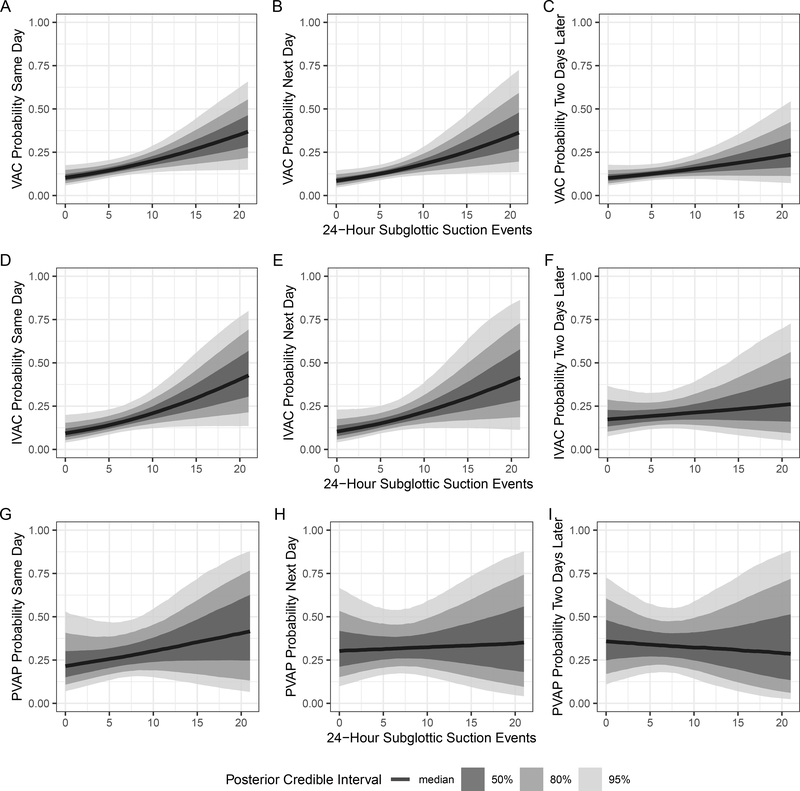
Among the 87 included subjects, we evaluated 848 ventilator-days at risk for VAE (i.e., not including the first two days of mechanical ventilation). Though sputum quantity at onset of mechanical ventilation was not associated with time to VAE, we did observe an association between increased subglottic suction events and increased risk of both VAC and IVAC. Subglottic suction events across the pre-VAE period and across VAE types are summarized in Supplemental Table 1. Figure 1 shows the binomial model predicted probability of VAC or IVAC based on the number of observed subglottic suction events across all ventilator-days at risk for VAE. With subglottic suctioning at 10 versus 5 events over a 24-hour period, the absolute probability of VAC increases by 3.07% (type S error < 0.024) the same day, 2.97% (type S error < 0.018) the next day, and 1.71% (95% CI includes 0) the day after. With subglottic suctioning at 10 versus 5 events over a 24-hour period, the absolute IVAC probability by 3.57% (type S error < 0.035) the same day, 3.43% (type S error < 0.018) increased risk of IVAC the next day, and 1.25% (95% CI includes 0) the day after. We also evaluated the impact of 15 versus 5 subglottic suction events over a 24-hour period and found VAC probability increases by 6.75% (type S error < 0.024) the same day, 6.67% (type S error < 0.018) the next day, and 3.67% (95% CI includes 0) two days later; with the same increase, IVAC probability increases by 8.1% (type S error < 0.035) the same day, 7.67% (95% CI includes 0) the next day, and 2.57% (95% CI includes 0) two days later. Given the small number of observed PVAP events, posterior credibility intervals were wide. But a positive relationship was observed between subglottic suction events and same-day PVAP risk, which increases by 2.82% (95% CI includes 0) as subglottic suction events increase from 5 to 10, and by 5.86% (95% CI includes 0) as events increase from 5 to 15.
Figure 1. Increased sputum quantity is associated with increased risk for VAE.
Panels depict the results of random effects binomial regression models relating VAE to the frequency of subglottic suctioning. Panels A, B, and C show the association between increased sputum quantity and absolute probability of VAC on the same day, VAC on the next day, and VAC two days later, respectively. Panels D, E, and F show the between IVAC and subglottic suctioning, and panels G, H, and I the association between PVAP and subglottic suctioning, in the same order. Best estimates of the association are represented with a dark line, surrounded by bands of posterior certainty. The left panels show the primary analysis (case period defined as the day of VAE), and the center and right panels show sensitivity analyses with the case period defined as one- or two-days prior to VAE.
Sputum quantity alone does not discriminate IVAC from VAC.
Greater sputum quantity was associated with increased probability of VAC, IVAC, and PVAP. To determine whether sputum quantity could be used to discriminate IVAC from VAC, or PVAP from VAC/IVAC (not including PVAP), we evaluated a model comparing subglottic suction events in the period before IVAC versus VAC, and a model comparing subglottic suction events in the period before PVAP versus VAC/IVAC. No level of subglottic suction events reliably discriminated IVAC from VAC. Greater than 15 subglottic suction events was associated with greater probability of IVAC, but this threshold did not have high posterior certainty (Figure 2). Subglottic suction events did not discriminate PVAP from VAC/IVAC. In an exploratory secondary analysis, we evaluated a multivariable model including subglottic suctioning and other clinical variables that may contribute to respiratory failure to determine features not included in the IVAC definition (i.e., excluding temperature and white blood cell count) that discriminate IVAC from VAC. In our cohort of medical intensive care patients, we found that serum liver enzymes, alanine aminotransferase (ALT) and aspartate aminotransferase (AST), best discriminated IVAC and VAC, with elevated enzymes favoring VAC. The odds ratio of IVAC per standard deviation increase in ALT was 0.51 (95% CI 0.26 – 0.92), per standard deviation increase in AST was 0.56 (95% CI 0.26 – 0.996), and per standard deviation increase in the sum of AST and ALT was 0.54 (95% CI 0.27 – 0.98). Number of pressors and number of antibiotics favored VAC and IVAC, respectively. But unlike AST and ALT, these alone did not discriminate well (Supplemental Figure 2).
Figure 2. Subglottic suction events do not distinguish IVAC from VAC.
The results of random effects binomial regression comparing the subglottic suctioning in the pre-IVAC period to the period before non-infectious VAC (panels A, B, and C), or the pre-PVAP period to the period before VAC and IVAC (excluding PVAP) (panels D, E, and F) are shown. Best estimates of the association are represented with a dark line, surrounded by bands of posterior certainty. Very frequent subglottic suctioning is associated with IVAC, but no number of observed suction events discriminates IVAC from VAC with high certainty.
The clinical decision to send sputum for bacterial culture depends on fever more than sputum quantity.
The observed association between sputum quantity and VAE risk prompted us to examine the relationship between sputum quantity and the behavior of clinicians. In a multivariable model utilizing all mechanical ventilation days (in contrast to the analyses above, which utilized only the pre-VAE period), we examined the relationship between subglottic suction events, fever, and the decision to order a sputum culture. Whereas fever was associated with the probability of a sputum culture order (1.9 (95% CI 1.5 – 2.5)), sputum quantity was not (Supplemental Figure 3).
Discussion
Using a case-crossover design, we investigated the relationship between sputum production during mechanical ventilation, measured by subglottic suction frequency, and adverse ventilator-associated events (VAE) with the goal of understanding whether sputum quantity reliably reflects underlying lung disease that will result in increased oxygen (FiO2 and PEEP) requirement. We quantified sputum production as the number of subglottic suction events per calendar day, and we categorized VAE using the CDC’s NHSN definitions of VAC and IVAC. In this cohort of medical intensive care patients with VAE, sputum quantity measured by subglottic suction frequency was associated with both IVAC and VAC. Though the association with IVAC was stronger, sputum quantity did not discriminate well between infection-associated and non-infectious VAE. Secondary analyses revealed other variables, prominently AST and ALT, that better discriminated IVAC from VAC in this cohort. The fact that IVAC was less likely in subjects with evidence of livery injury suggests pulmonary edema may have driven VAE in these cases. We found no association between sputum quantity at onset of mechanical ventilation and time to VAE, supporting VAE as a sign of new lung pathology that develops during mechanical ventilation, rather than progression of pathology already present at time of intubation.
The relationship between sputum quantity and VAE is of interest because sputum quantity is often clinically interpreted as a sign of VA-LRTI, and the relationship between VAE and VA-LRTI remains unclear.7–10 The finding that sputum quantity, measured by subglottic suction frequency, is higher at the time of VAE and immediately before VAE, relative to the earlier pre-VAE period, validates sputum quantity as a physical examination sign associated with risk for worsening respiratory failure. However, sputum quantity did not discriminate well between IVAC and VAC. Greater sputum quantity was associated with VAC without fever or leukocytosis, as well as with IVAC.
There are several possible explanations for the discordance between sputum quantity and the NHSN criteria used to discriminate infection-associated adverse events. One possible explanation is that the number of subglottic suction events does not consistently represent the quantity of sputum produced. Misclassification of sputum production could result in poor discrimination between IVAC and VAC. Another possible explanation is that NHSN IVAC criteria, which are known to exclude a large proportion of clinically diagnosed VA-LRTI,11 exclude true VA-LRTI events that would drive a stronger association with sputum quantity. A third possible explanation is that sputum production, though commonly interpreted as a clinical sign of infection, is nonspecific, or that sputum quality (e.g., purulence) is more specific than sputum quantity. Indeed, we observed that clinician orders for respiratory bacterial culture were not associated with sputum quantity, whereas culture orders were associated with fever. This likely reflects the clinical suspicion for non-infectious etiologies (e.g., pulmonary edema) that can also drive increased sputum quantity. Further complicating efforts to use subglottic suction frequency to discriminate IVAC and VAC is the potential protective effect against infection exerted by suctioning.19–21 Subglottic suctioning may help to prevent some VAE that it would otherwise herald.
Several limitations of our study must be noted. The case-crossover design was chosen because VAE are rare outcomes, and the exposure of interest – sputum quantity – is dynamic and most likely to impact VAE risk acutely. However, our exclusive focus on VAE cases leaves open important questions about the generalizability of our findings across the broader population of patients who depend upon mechanical ventilation. Likewise, our cohort was enrolled from the medical intensive care unit of a single, academic medical center, and medical history on subjects was limited to ICD-10 codes associated with their intensive care unit admission. Sputum quantity may differ significantly in surgical, neurological, or pediatric intensive care patients, and practices for management of tracheal secretions may differ across institutions. Our findings must be validated with studies of sputum quantity across a broader population of patients dependent on mechanical ventilation.
Nevertheless, our study demonstrates a significant, positive association between subglottic suction events and VAE risk, supporting the validity of subglottic suction frequency ascertained from electronic medical records as a means to quantify sputum production. The relationships between the respiratory tract microbiome, lung immune activation, and sputum production during mechanical ventilation warrant further study, as does the impact of sputum production on clinical behavior. The frequency of subglottic suction events appears to serve as a valuable marker of sputum quantity for these future analyses.
Conclusions
The frequency of subglottic suction events, which are performed on an as-needed basis for patients dependent on mechanical ventilation, may serve as a valuable marker of sputum quantity, which is associated with risk for risk for adverse ventilator-associated events (VAE). However, the relationship between subglottic suction events, sputum quantity, and VAE must be validated in a broader population of mechanically ventilated patients and intensive care settings.
Supplementary Material
Supplemental Figure 1. No association is observed between subglottic suction episodes at onset of mechanical ventilation and time to VAE.
Results of a parametric Weibull survival model, relating survival without VAE (days) to the number of subglottic suction events over the first full calendar day of mechanical ventilation, are shown, suggesting no association between admission sputum quantity and subsequent VAE. The best estimates of the association is represented with a dark line, surrounded by bands of posterior certainty.
Supplemental Figure 2. Exploratory analyses of clinical variables that discriminate IVAC and VAC suggest a role for AST, ALT, number of pressors, and number of antibiotics.
The association between the sum of AST and ALT and probability of IVAC versus VAC are shown, stratified by number of pressors and number of antibiotics. Best estimates of the associations are represented with a dark line, surrounded by bands of posterior certainty.
Supplemental Figure 3. The clinical decision to perform sputum culture is associated with fever but not subglottic suction events.
Results of a random effects binomial regression relating subglottic suction events and fever to the clinical decision to send sputum for bacterial culture are shown. Best estimates of the association are represented with a dark line, surrounded by bands of posterior certainty.
Supplemental Table 1. Subglottic suction frequency across days relative to VAE and across VAE type. The median and interquartile range (IQR) of subglottic suction events per calendar day are given for the day of VAE, the day prior, two days prior, and the antecedent period. Summaries stratified by VAE type are also provided.
ACKNOWLEDGEMENTS
BJK is supported by the National Institute for Allergy and Infectious Diseases (K23 AI121485 and L30 AI120149), as well as Centers for Disease Control and Prevention (CDC) contract awards (BAA 200-2016-91964 and 200-2018-02919). BJK and DP are supported by the CDC Healthcare-Associated Infection Prevention Epicenters Program (U54CK000485). All authors report no conflicts of interest relevant to this article. There are no relevant disclosures.
DATA AVAILABILITY
Deidentified data, as well as analysis scripts and model code are available at github.com/bjklab.
REFERENCES
- 1.Klompas M Complications of mechanical ventilation–the CDC’s new surveillance paradigm. The New England journal of medicine. 2013;368(16):1472–1475. [DOI] [PubMed] [Google Scholar]
- 2.Magill SS, Klompas M, Balk R, et al. Developing a new, national approach to surveillance for ventilator-associated events*. Critical care medicine. 2013;41(11):2467–2475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Magill SS, Rhodes B, Klompas M. Improving ventilator-associated event surveillance in the national healthcare safety network and addressing knowledge gaps: Update and review. Current opinion in infectious diseases. 2014;27(4):394–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kobayashi H, Uchino S, Takinami M, Uezono S. The impact of Ventilator-Associated events in critically ill subjects with prolonged mechanical ventilation. Respiratory care. 2017;62(11):1379–1386. [DOI] [PubMed] [Google Scholar]
- 5.Meagher AD, Lind M, Senekjian L, et al. Ventilator-associated events, not ventilator-associated pneumonia, is associated with higher mortality in trauma patients. The journal of trauma and acute care surgery. 2019;87(2):307–314. [DOI] [PubMed] [Google Scholar]
- 6.Klompas M, Li L, Menchaca JT, Gruber S, Centers for Disease Control and Prevention Epicenters Program. Ultra-Short-Course antibiotics for patients with suspected Ventilator-Associated pneumonia but minimal and stable ventilator settings. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America. 2017;64(7):870–876. [DOI] [PubMed] [Google Scholar]
- 7.Fan Y, Gao F, Wu Y, Zhang J, Zhu M, Xiong L. Does ventilator-associated event surveillance detect ventilator-associated pneumonia in intensive care units? A systematic review and meta-analysis. Critical care / the Society of Critical Care Medicine. 2016;20(1):338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Klompas M Discordance between novel and traditional surveillance definitions for ventilator-associated pneumonia: Insights and opportunities to improve patient care. Infection control and hospital epidemiology: the official journal of the Society of Hospital Epidemiologists of America. 2014;35(9):1196–1198. [DOI] [PubMed] [Google Scholar]
- 9.Ramirez-Estrada S, Peña-Lopez Y, Kalwaje Eshwara V, Rello J. Ventilator-associated events versus ventilator-associated respiratory infections-moving into a new paradigm or merging both concepts, instead? Annals of translational medicine. 2018;6(21):425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stevens JP, Silva G, Gillis J, et al. Automated surveillance for ventilator-associated events. Chest. 2014;146(6):1612–1618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Klompas M Ventilator-Associated events: What they are and what they are not. Respiratory care. 2019;64(8):953–961. [DOI] [PubMed] [Google Scholar]
- 12.Cocoros NM, Klompas M. Ventilator-Associated events and their prevention. Infectious disease clinics of North America. 2016;30(4):887–908. [DOI] [PubMed] [Google Scholar]
- 13.Lewis SC, Li L, Murphy MV, Klompas M, CDC Prevention Epicenters. Risk factors for ventilator-associated events: A case-control multivariable analysis. Critical care medicine. 2014;42(8):1839–1848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Klompas M Potential strategies to prevent ventilator-associated events. American journal of respiratory and critical care medicine. 2015;192(12):1420–1430. [DOI] [PubMed] [Google Scholar]
- 15.Klompas M, Anderson D, Trick W, et al. The preventability of ventilator-associated events. The CDC prevention epicenters wake up and breathe collaborative. American journal of respiratory and critical care medicine. 2015;191(3):292–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Rello J, Ramírez-Estrada S, Romero A, et al. Factors associated with ventilator-associated events: An international multicenter prospective cohort study. European journal of clinical microbiology & infectious diseases: official publication of the European Society of Clinical Microbiology. 2019;38(9):1693–1699. [DOI] [PubMed] [Google Scholar]
- 17.Liu J, Zhang S, Chen J, et al. Risk factors for ventilator-associated events: A prospective cohort study. American journal of infection control. 2019;47(7):744–749. [DOI] [PubMed] [Google Scholar]
- 18.Vaewpanich J, Akcan-Arikan A, Coss-Bu JA, Kennedy CE, Starke JR, Thammasitboon S. Fluid overload and kidney injury score as a predictor for Ventilator-Associated events. Frontiers in pediatrics. 2019;7:204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Damas P, Frippiat F, Ancion A, et al. Prevention of ventilator-associated pneumonia and ventilator-associated conditions: A randomized controlled trial with subglottic secretion suctioning. Critical care medicine. 2015;43(1):22–30. [DOI] [PubMed] [Google Scholar]
- 20.Caroff DA, Li L, Muscedere J, Klompas M. Subglottic secretion drainage and objective outcomes: A systematic review and Meta-Analysis. Critical care medicine. 2016;44(4):830–840. [DOI] [PubMed] [Google Scholar]
- 21.Mao Z, Gao L, Wang G, et al. Subglottic secretion suction for preventing ventilator-associated pneumonia: An updated meta-analysis and trial sequential analysis. Critical care / the Society of Critical Care Medicine. 2016;20(1):353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rothman KJ, Greenland S. Planning study size based on precision rather than power. Epidemiology. 2018;29(5):599–603. [DOI] [PubMed] [Google Scholar]
- 23.R Core Team. R: A language and environment for statistical computing. Published online 2018.
- 24.Maclure M The case-crossover design: A method for studying transient effects on the risk of acute events. American journal of epidemiology. 1991;133(2):144–153. [DOI] [PubMed] [Google Scholar]
- 25.Maclure M, Mittleman MA. Should we use a case-crossover design? Annual review of public health. 2000;21:193–221. [DOI] [PubMed] [Google Scholar]
- 26.Hosmer DW Jr., Lemeshow S, Sturdivant RX. Applied Logistic Regression. John Wiley & Sons; 2013:313–376. [Google Scholar]
- 27.Rice K Equivalence between conditional and Random-Effects likelihoods for Pair-Matched Case-Control studies. Journal of the American Statistical Association. 2008;103(481):385–396. [Google Scholar]
- 28.Bürkner P-C. Brms: An R package for bayesian multilevel models using stan. Journal of Statistical Software, Articles. 2017;80(1):1–28. [Google Scholar]
- 29.Carpenter B, Gelman A, Hoffman M, et al. Stan: A probabilistic programming language. Journal of statistical software. 2017;76(1):1–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.McElreath R Statistical Rethinking: A Bayesian Course with Examples in R and Stan. 1 edition. CRC Press/Taylor & Francis Group; 2016. [Google Scholar]
- 31.Gabry J, Simpson D, Vehtari A, Betancourt M, Gelman A. Visualization in bayesian workflow. Journal of the Royal Statistical Society: Series A (Statistics in Society). 2019;182(2):389–402. [Google Scholar]
- 32.Gelman A, Tuerlinckx F. Type S error rates for classical and bayesian single and multiple comparison procedures. Computational statistics. 2000;15(3):373–390. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Figure 1. No association is observed between subglottic suction episodes at onset of mechanical ventilation and time to VAE.
Results of a parametric Weibull survival model, relating survival without VAE (days) to the number of subglottic suction events over the first full calendar day of mechanical ventilation, are shown, suggesting no association between admission sputum quantity and subsequent VAE. The best estimates of the association is represented with a dark line, surrounded by bands of posterior certainty.
Supplemental Figure 2. Exploratory analyses of clinical variables that discriminate IVAC and VAC suggest a role for AST, ALT, number of pressors, and number of antibiotics.
The association between the sum of AST and ALT and probability of IVAC versus VAC are shown, stratified by number of pressors and number of antibiotics. Best estimates of the associations are represented with a dark line, surrounded by bands of posterior certainty.
Supplemental Figure 3. The clinical decision to perform sputum culture is associated with fever but not subglottic suction events.
Results of a random effects binomial regression relating subglottic suction events and fever to the clinical decision to send sputum for bacterial culture are shown. Best estimates of the association are represented with a dark line, surrounded by bands of posterior certainty.
Supplemental Table 1. Subglottic suction frequency across days relative to VAE and across VAE type. The median and interquartile range (IQR) of subglottic suction events per calendar day are given for the day of VAE, the day prior, two days prior, and the antecedent period. Summaries stratified by VAE type are also provided.
Data Availability Statement
Data, model code, and code used to produce the figures are available at https://github.com/bjklab/Subglottic-suction-frequency-and-adverse-events-during-mechanical-ventilation.
Deidentified data, as well as analysis scripts and model code are available at github.com/bjklab.