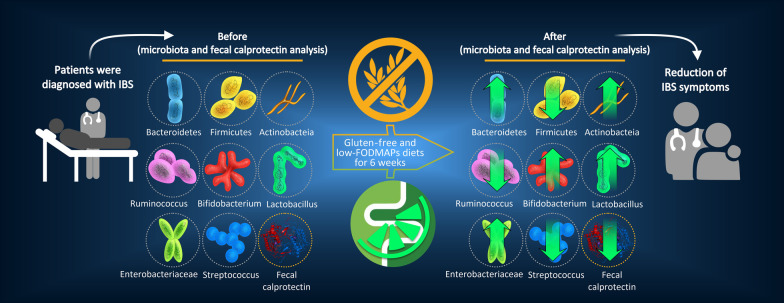
Abstract
Background and objective
Recently, dietary restriction of fermentable carbohydrates (a low-FODMAP diet) in combination with a gluten-free diet (GFD) has been proposed to reduce the symptoms in irritable bowel syndrome (IBS) patients. Different studies reported that IBS has been associated with dysbiosis in the gut microbiota. Additionally, a few studies have reported inflammation in the gastrointestinal (GI) system of adults with IBS. In this study, we aimed to investigate the effects of low FODMAP-gluten free diet (LF-GFD) on clinical symptoms, intestinal microbiota diversity, and fecal calprotectin (FC) level in Iranian patients with IBS.
Design
In this clinical trial study, 42 patients with IBS (Rome IV criteria) underwent LF-GFD intervention for 6 weeks. Symptoms were assessed using the IBS symptom severity scoring (IBS-SSS), and fecal samples were collected at baseline and after intervention and analyzed by quantitative 16 S rRNA PCR assay. The diversity of gut microbiota compared before and after 6 weeks of dietary intervention. FC was also analyzed by the ELISA method.
Results
Thirty patients (mean age 37.8 ± 10.7 years) completed the 6-week diet. The IBS-SSS was significantly (P = 0.001) reduced after LF-GFD intervention compared to the baseline. Significant microbial differences before and after intervention were noticed in fecal samples. A significant increase was found in Bacteroidetes, and the Firmicutes to Bacteroidetes (F/B) ratio was significantly (P = 0.001) decreased after the dietary intervention. The value of FC was significantly decreased after 6 weeks of dietary intervention (P = 0.001).
Conclusions
Our study suggests that patients with IBS under an LF-GFD had a significant improvement in IBS symptoms severity, with reduced FC level following normalization of their gut microbiota composition. Further rigorous trials are needed to establish a long-term efficacy and safety of this dietary intervention for personalized nutrition in IBS.
Clinical Trial Registry Number: IRCT20100524004010N26.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12876-021-01868-5.
Keywords: Irritable bowel syndrome, Gluten-free diet, Low-FODMAP, Gut microbiota, IBS-SSS, Iran
Introduction
Irritable bowel syndrome (IBS) is one of the most commonly diagnosed functional gastrointestinal disorders accounting for around 10% of the world’s population [1]. This functional bowel disorder can be classified into different categories: diarrhea-predominant (IBS-D), constipation-predominant (IBS-C), mixed (IBS-M), and unclassified (IBS-U) [2].
The pathogenesis of IBS is complex and not yet clearly defined, but numerous pathophysiological mechanisms have been proposed including brain-gut dysfunction, intestinal dysmotility, visceral hypersensitivity, inflammation, psychosocial stressors, altered levels of gastrointestinal neuropeptides and hormones, and particularly imbalance in the composition of gut microbiota, called “microbiome dysbiosis” [3, 4]. It is now well evident that microbiome dysbiosis-related disorders and IBS have been reported to have similar clinical features, and more recently, IBS has been found to be in association with the bacterial community shift in the large intestine [5]. Furthermore, restoration and modulation of gut microbiota through consumption of probiotics, prebiotics, and symbiotic, which result in improvement of the IBS-related symptoms highlights the important role of gut microbiota dysbiosis in the pathogenesis of IBS [6, 7].
Normal gut microbiome inhibits the overgrowth and colonization of pathogenic organisms by competition for nutrients and attachment sites in the gut epithelium, production of antibacterial substances, as well as enhancement of the host immune responses [8, 9]. Several previous studies have described that alterations in the gut microbiota such as increased number of Enterobacteriaceae and Bacteroidetes or decreased number of beneficial organisms like Bifidobacterium and Lactobacillaceae, can trigger a gut immune response, impair gastrointestinal functions and enhance disease susceptibility [6, 10].
A majority of IBS patients suffer from abdominal discomfort or pain and bloating after ingestion of certain food items that may contain IBS-associated triggers [11, 12]. In addition, a number of studies suggest that environmental factors such as diet, lifestyle, and antibiotics and medications exert a significant impact on the gut microbiome [13]. Recently, dietary components including wheat, gluten, and fermentable oligo-di-mono-saccharides and polyols (FODMAP) have been suggested to play an essential role in the induction of IBS symptoms [14]. Accordingly, there is some evidence supporting a clinically relevant positive effect for low-FODMAP and gluten-free diets in patients with IBS [15].
The significant effect acknowledged to date is alterations in gut microbiota by varying gluten intake, such as altering total microbiota abundance and changing the relative amount of Bifidobacteria [12, 16]. However, the roles of alterations of gut microbiota due to the reduction in FODMAP intake in ongoing efficacy have yet to be explored. Herein, we aimed to investigate the impact of a 6-week LF-GFD on gut microbiota alterations in Iranian patients with IBS. Additionally, in order to determine the inflammation status of patients, we examined the level of fecal calprotectin (FC) before and after dietary intervention.
Materials and methods
Study population
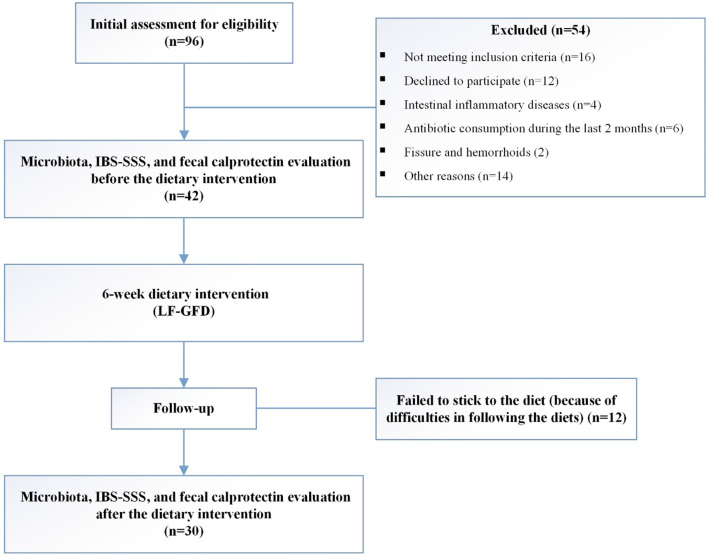
Ninety-six consecutive IBS patients were screened for eligibility; 54 patients did not meet the initial assessment criteria, and 12 patients discontinued the dietary intervention during the follow-up phase and 30 patients completed the study. A flowchart of the recruitment process and study design is shown in Fig. 1. Forty-two eligible patients with IBS, aged 18–59 years (mean age 37.8 ± 10.7 years), and had a physician diagnosis of IBS according to the Rome IV diagnostic criteria, and did not have any other gastrointestinal disorders were recruited in this study [17]. Patients with the following criteria were excluded: patients with a history of celiac disease (CD), inflammatory bowel disease (IBD), liver diseases, gastrointestinal surgery, cancer, use of non-steroidal anti-inflammatory drugs (NSAIDs), excessive alcohol consumption, systemic use of immunosuppressive agents, and poorly controlled psychiatric disease. In addition, colonoscopy and biopsy specimens were performed in order to include patients with normal mucosa and excluding those with fissures, hemorrhoids, and microscopic colitis. Moreover, those who used broad-spectrum antibiotics, probiotics, or any other drugs that affect the bowel function within the last 4 weeks prior to the study were also excluded. Active participation in another form of dietary therapy at the time of enrollment (i.e., low carbohydrate, high protein) was not considered in this study.
Fig. 1.
Schematic of recruitment process and study design
Study design and procedures
This study was an uncontrolled, open-label clinical trial study, which was conducted in the Research Institute for Gastroenterology and Liver Diseases (RIGLD) at Taleghani Hospital in Tehran, Iran. All the participants were recruited between March 2018 and August 2019. This was a dietary trial and the dietitian was not blinded. Patients were referred to the trained research dietitian at the RIGLD and were counseled on their allocated diets. A standardized questionnaire including demographic information, usual diet, medication history, and underlying health condition and clinical symptoms was recorded for all patients. All IBS patients fulfilling inclusion criteria were counseled by the dietitian and given an LF-GFD for a 6-week run-in period. Fecal samples were collected from all patients before and after the completion of the study period for microbiota analysis. In order to assess the effect of the dietary interventions and the severity of IBS symptoms, the IBS symptom severity score (IBS-SSS) was applied [18]. The overall IBS-SSS ranges from 0 to 500, and all patients were divided into 3 severity groups by use of the accepted cut-off values: < 175, mild IBS; 175–300, moderate IBS; > 300, severe IBS. A higher score implicates more severe symptoms. Patients also classified into the following categories: (1) Defecation, (2) Changes in frequency, and (3) Changes in form of stool.
The study protocol was approved by the Ethical Review Committee of RIGLD at Shahid Beheshti University of Medical Sciences (Project No. IR.SBMU.RIGLD.REC.1396.154). The study was performed according to the revised Declaration of Helsinki 2013 [19] and informed consents were obtained from all subjects and/or their legal guardians prior to sample collection. This study was registered in the Iranian Registry of Clinical Trials on 20/10/2018 and the clinical trial registration number is IRCT20100524004010N26.
Collection of fecal samples
Fresh stool samples were collected from every subject enrolled in this study at baseline and after 6 weeks following dietary therapy. All samples were homogenized through agitation using a vortex and divided into three aliquots within 3 h of defecation. The aliquots were immediately frozen and stored at − 80 °C in screw-capped cryovial tubes until used for DNA extraction.
DNA extraction from fecal samples
Total DNA content was extracted from stool samples using the QIAamp DNA Stool Mini Kit (Qiagen Retsch GmbH, Hannover, Germany) according to the manufacturer’s instructions with some modifications. DNA concentration was quantified by NanoDrop ND-2000 Spectrophotometer (NanoDrop products, Wilmington, DE, USA). The concentration and purity of extracted DNA were assessed by Nanodrop (DeNovix Inc., USA). Extracted DNA samples were stored at − 20 °C until further analysis.
Diet
The main aim of the nutritional assessment was the evaluation of participants’ nutritional status. After investigation of the recordings of the dietary recall for 3 days (1 weekend and 2 workdays), a personalized LF-GFD adjusted to match energy, macronutrients, and micronutrients daily requirements was designed for each participant. To be precise, if a food is gluten-free, it does not necessarily mean that it will be low in FODMAP contents. In other words, some gluten-containing foods are low FODMAP foods. However, in this study, we planned to apply an intervention that was not only low in FODMAP contents but also was free of gluten. In Additional file 1: Table S1, some examples of high FODMAP gluten-free foods and some examples of gluten-containing low FODMAP foods are described, all of which were eliminated in the LF-GFD. All of the diets were prepared by a trained nutritionist who was responsible for doing nutrition counseling (an average of 1 h), prescribing the diets, and being in contact with patients through telephone and/or email. Compliance with the diet was determined by the evaluation of the 3 days dietary recall by the same nutritionist. At the beginning of the study, an in-depth GFD review, high FODMAP foods, and low FODMAP alternatives were explained to all the participants, in addition to the education of how to modify the FODMAP content towards the LFD, as Iranian dishes have a high level of FODMAP content. An example of one day LF-GFD which should have been followed during the study and a regular diet before start of the study is shown in Table 1.
Table 1.
Examples of the two different prescribed diets before and during the study for a typical day
| Meal | LF-GFD | Regular diet |
|---|---|---|
| Breakfast |
1 cup of tea 90 g of gluten-free muffin |
1 glass of milk 3 slices of bread 3 teaspoons of cherry jam |
| Morning snack | 1 banana | 1–3 slices of cookies |
| Lunch |
20 tablespoons of rice 90 g of seafood Vegetable salad (1 cucumber + 1 tomato + 1 cup of lettuce) |
20 tablespoon of rice ½ cup of onion ½ cup of beans 60 g of red meat 3 slices of bread |
| Afternoon snack |
1 cup of tea 30 g of gluten-free biscuits |
1 cup of Fruit of the season 100 g of ice cream |
| Dinner |
2 slices of whole grain gluten-free bread 60 g of chicken 100 g of carrots |
90 g of chicken or regular pasta 100 g of tomato 3 slices of bread 1 cup of regular yogurt |
| During the day | 90 g of grapes | 2 cup of Fruit of the season |
Dietary data represent the typical diet for a patient with an approximate energy expenditure of 1600 kcal/day
FODMAP fermentable, oligosaccharides, disaccharides, monosaccharides, and polyols, GFD gluten-free diet, LFD low FODMAP diet, LF-GFD low FODMAP gluten-free diet
Microbiota analysis by quantitative real-time PCR (qPCR)
In the current study, a qPCR assay was performed for the enumeration of eight bacterial phyla, families, and genera including Firmicutes, Bacteroidetes, Actinobacteria, Enterobacteriaceae, Bifidobacterium, Lactobacillus, Ruminococcus, and Streptococcus. The qPCR was carried out by SYBR Green chemistry using universal and group-specific primers based on the bacterial 16 S rRNA sequences presented in Additional file 2: Table S2. Each PCR reaction was performed in a final volume of 25 µL, comprising of 12.5 µL of SYBR green PCR master mix (Ampliqon, Odense, Denmark), 1 µL of 10 pmol of forward and reverse primers, and 100 ng of the DNA template. The reaction parameters for amplification were 95 °C for 10 min and 40 cycles at 95 °C for 20 s, 30 s of annealing at optimal temperature for each primer pair as indicated in Additional file 2: Table S2, and 72 °C for 20 s. All PCR amplifications were carried out in triplicate by using a Rotor-Gene® Q (Qiagen, Germany) real-time PCR system. The accuracy of amplification was determined by melting curve analysis with increasing temperature from 60 to 95 °C (at the regular increment of 0.5 °C for 5 s) to confirm the specificity of amplification. The relative abundance of each taxon before and after the dietary therapy was calculated according to the ratio of 16 S rRNA copy number of specific bacteria to total 16 S rRNA copy number of universal bacteria using the previously described method [20]. Accordingly, the average Ct value obtained from each primer pair was transformed into a percentage using the following formula:
The “Eff. Univ” refers to the calculated efficiency of the universal primers (2 = 100 % and 1 = 0 %) and “Eff. Spec” indicates the efficiency of the taxon-specific primers. “Ct univ” and “Ct spec” represent the threshold cycles registered by the thermocycler. “X” represents the percentage (%) of 16 S taxon-specific copy numbers existing in a sample.
Fecal calprotectin measurement
Before and after the dietary therapy, first-morning stool samples were collected from patients and stored at − 80 °C after initial storage. FC was measured by using BÜHLMANN fCAL® ELISA kit (Bühlmann Laboratories, Schönenbuch, Switzerland) according to the manufacturer’s instructions. An FC level < 50 µg/g stool was considered to be within the normal range.
Statistical analysis
Spearman’s correlation analysis was used for non-parametric values and student t-test and Mann-Whitney test were used for the analysis of parametric data. The PCA (principle component analysis) plot was drawn by using the FactoMineR and Factoextra packages from the open-source statistical program R version 3.6.1 (R Core Team, Vienna, Austria). The relative abundance of microbiota was graphed using GraphPad Prism software version 8.3.0 (GraphPad Software, San Diego, CA, USA). Differences were considered to be statistically significant when *P < 0.05, **P < 0.01, and ***P < 0.001.
Results
Baseline characteristics
Out of 96 IBS patients, 30 patients completed the study period (a six-week of LF-GFD), of whom 15 (50%) patients were male, mean age 35.7 years (range 21–57 years), and 15 patients (50%) were female, mean age 39.8 years (range 18–59 years). Most of the IBS patients were sub-classified into IBS-D according to the Rome IV criteria (Table 2).
Table 2.
Baseline characteristics of study participants at enrollment
| Baseline characteristics | Number (%) |
|---|---|
| Age (years), mean (range) | 37.8 (18–59) |
| Gender (M/F) | 15/15 |
| Smoking, yes (%) | 9 (30 %) |
| IBS-SSS, mean (range) | 326 (250–475) |
| IBS subtypes | |
| IBS-D | 16 (54) |
| IBS-C | 10 (33) |
| IBS-M | 3 (10) |
| IBS-U | 1 (3) |
| Ethnicity | |
|
Persian Turk Kurd Lur Others |
10 11 4 3 2 |
Effects of the GFD + low-FODMAP diet on the microbiota diversity
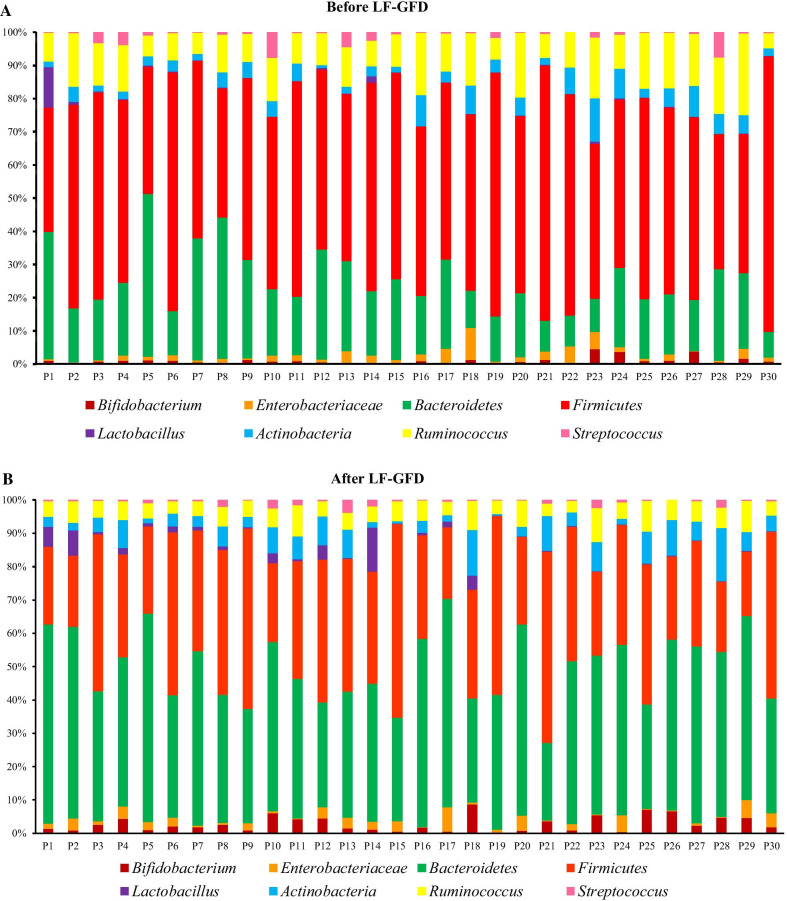
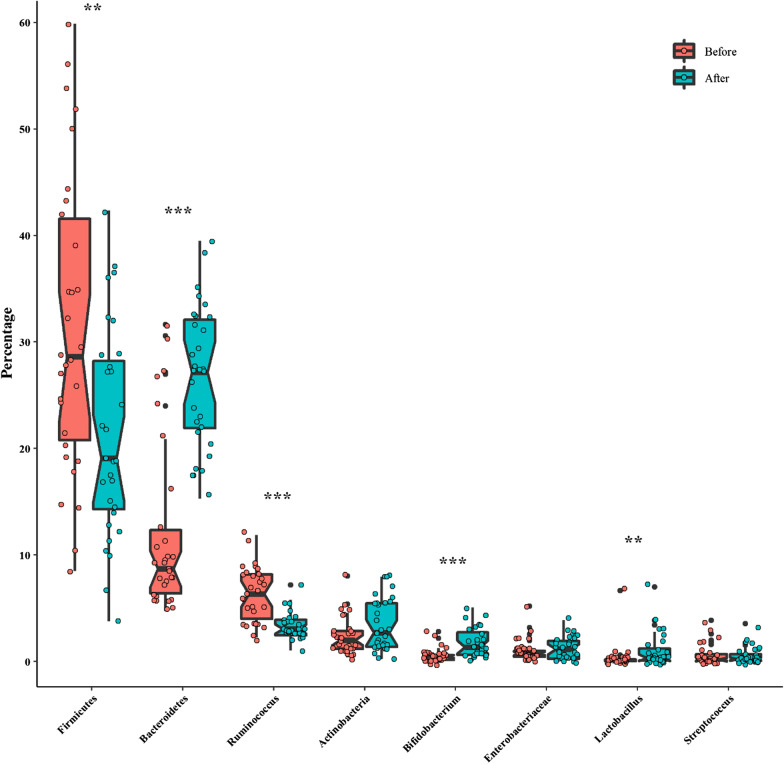
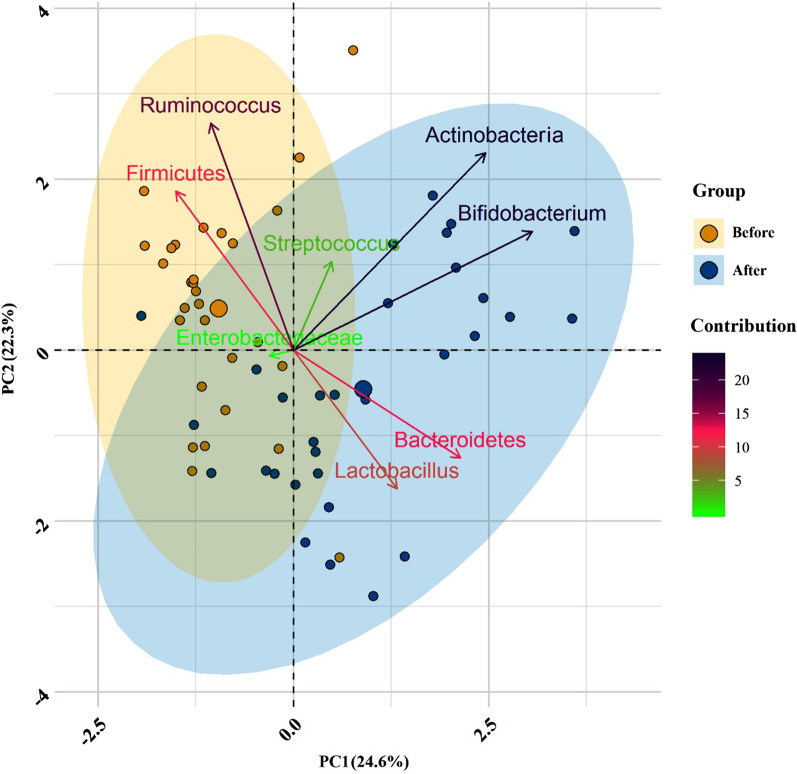
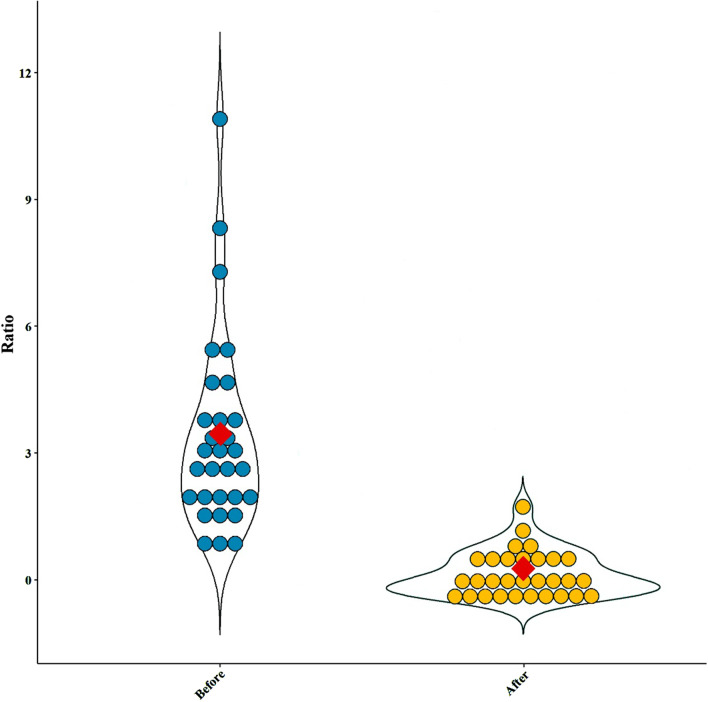
Our intestinal microbiota analysis was based on qPCR amplification to calculate the ratio of 16 S rRNA copy number of each bacterial taxa before and after the dietary intervention. The relative abundance and diversity of intestinal microbiota among IBS patients before and after the LF-GFD intervention is illustrated in Fig. 2a and b. The predominant phylum in IBS patients before the LF-GFD was Firmicutes (31.59 %), which significantly reduced after the dietary intervention (22.17%; P = 0.003). Bacteroidetes was significantly the most abundant phylum after the dietary intervention, (from 11.69% (baseline) to 26.65%; P = 0.001). The relative abundance of phylum Actinobacteria was also increased after the dietary intervention, but this difference was not statistically significant (P = 0.12). No significant (P = 0.63) alterations were also observed in the relative abundance of Enterobacteriaceae before and after the intervention. However, the percentage of Ruminococcaceae significantly decreased from 6.4 to 3.45% (P = 0.001). At the genus level, the relative abundance of Bifidobacterium and Lactobacillus significantly (P = 0.001 and P = 0.006, respectively) increased after the intervention, whereas the percentage of Streptococcus remained almost similar to its baseline abundance with no significant changes (P = 0.72). The mean percentage and distribution of the selected bacterial taxa before and after LF-GFD in IBS patients are illustrated in Fig. 3 and Additional file 3: Figure S1. In addition, principal component analysis (PCA) also revealed that taxonomic profiles were notably different in the microbial communities before and after the dietary intervention among IBS patients as schematically depicted in Fig. 4. The ratio of Firmicutes to Bacteroidetes (F/B) was significantly decreased (P = 0.001) and shifted from 2.6:1 to 0.8:1 before and after the dietary intervention, respectively. The F/B ratio before and after LF-GFD in IBS patients is shown in Fig. 5.
Fig. 2.
The relative abundance and diversity of intestinal microbiota in IBS patients before and after LF-GFD are illustrated in (a) and (b), respectively. Each color corresponds to a type of microbiota included in this study
Fig. 3.
Box plot for the distribution of the selected bacterial taxa by the median abundance that constitutes the fecal microbiota in IBS patients before and after LF-GFD
Fig. 4.
Bacterial community clustering using principal component analysis (PCA) before and after LF-GFD in IBS patients. Percentage values in parentheses next to PC1 and PC2 represent the percentage of variance explained by each component. Arrows show the contribution of each type of microbiota on the PC1 and PC2. Each data point denotes an individual patient, colored based on their group
Fig. 5.
Violin plots showing the Firmicutes to Bacteroidetes (F/B) ratio before and after LF-GFD in IBS patients. This ratio was significantly (P = 0.001) decreased after the dietary intervention compared with baseline. Mean values of the F/B ratio are marked as the red rhombus
Effects of the GFD + low-FODMAP diet on IBS symptom severity
Figure 6 represents IBS symptom severity in each patient before and after the dietary intervention. Based on the IBS-SSS, the severity of IBS symptoms was classified as mild (n = 1), moderate (n = 12) and severe (n = 17) at the baseline and before the dietary intervention. After the end of the dietary intervention, the number of patients in each classification changed to mild (n = 8), moderate (n = 20), and severe (n = 2), accordingly (Table 3. Totally, IBS-SSS decreased in 22/30 (73.3%) patients after the dietary intervention compared to the baseline, and this clinical improvement was statistically significant (P = 0.001). Approximately, 53 of patients after the end of the dietary intervention experienced a 30–60% reduction in IBS-SSS, while only 3.3% of patients of experienced more than 60% reduction (Table 4).
Fig. 6.
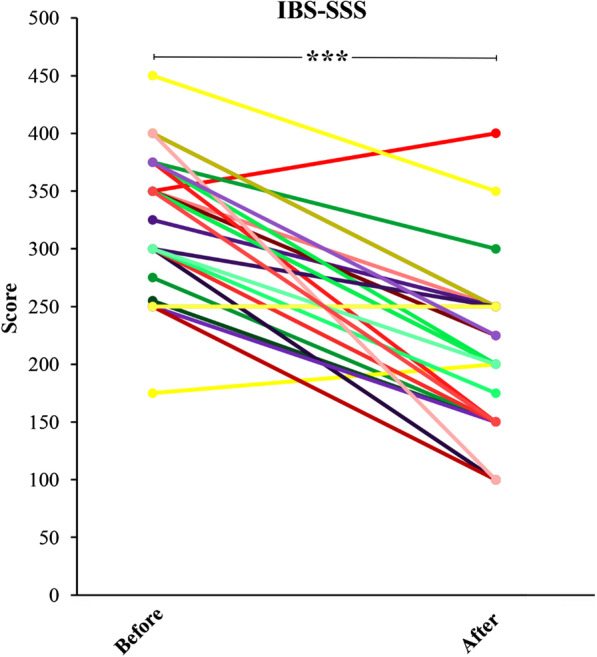
IBS symptom severity score (IBS-SSS) in IBS patients before and after the dietary intervention. IBS-SSS was reduced in patients after the end of the dietary intervention compared with baseline (P = 0.001)
Table 3.
Severity scores for mild, moderate and severe IBS patients before and after the dietary intervention
| IBS-SSS | Before | IBS-SSS | After | ||||
|---|---|---|---|---|---|---|---|
| Mean (± SD) | Median | Range | Mean (± SD) | Median | Range | ||
| Mild (n = 1) | 175 (-) | 175 | – | Mild (n = 8) | 146.8 (± 19.5) | 150 | 100–175 |
| Moderate (n = 12) | 277.5 (± 23.4) | 287.5 | 250–300 | Moderate (n = 20) | 231.25 (± 28.6) | 237.5 | 200–300 |
| Severe (n = 17) | 369.1 (± 29.1) | 350 | 300–450 | Severe (n = 2) | 375 (± 25) | 375 | 350–400 |
Table 4.
Frequency distribution of IBS-SSS reduction
| IBS-SSS reduction category | After dietary intervention n (%) | P value |
|---|---|---|
| Decreased/Increased/unchanged | 22 (73.3)/2 (6.6)/6 (20) | > 0.001 |
| < 30% reduction | 5 (16.6) | |
| 30–60% reduction | 16 (53.3) | |
| > 60% reduction | 1 (3.3) |
Fecal calprotectin
The mean FC values in the IBS patients at the baseline and after a 6-week LF-GFD was shown in Fig. 7. The value of FC was significantly decreased after 6 weeks of diets, from 83.4 at baseline to 37.3 (P < 0.001). In addition, we did not observe any significant correlation between FC level and microbiota diversity before and after the diets.
Fig. 7.
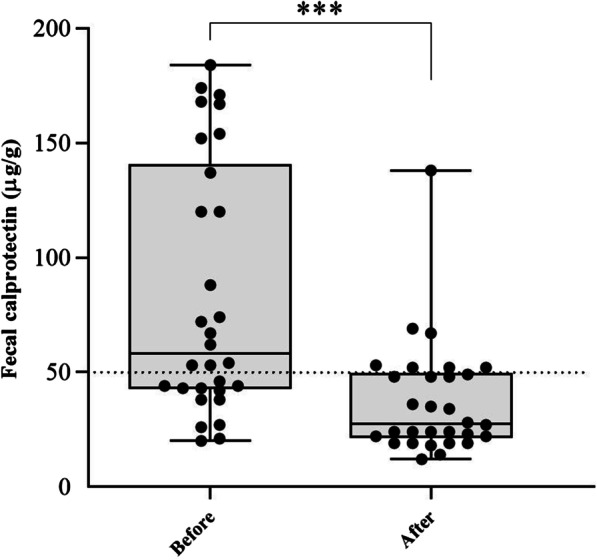
Values of fecal calprotectin level in IBS patients before and after a six-week of LF-GFD. ***P < 0.001
Discussion
It has been hypothesized that a subset of patients with IBS may have an intolerance of dietary triggers such as gluten and highly FODMAP-containing foods, which can alter the gut microbiota and the metabolome of patients with IBS leading to worsening of their symptoms [21, 22]. This has led researchers to recommend LF-GFD as the most widely adopted diets to improve IBS symptoms [23, 24]. Despite the growing popularity of the GFD and low-FODMAP diet in patients with IBS, the beneficial impact of such dietary interventions on the restoration of intestinal microbiota dysbiosis has been lacking in this population.
In this 6-week, controlled, dietary intervention study, a clinically significant improvement in IBS-SSS was observed after the dietary intervention compared to the baseline. Several other clinical trials have also shown that a low-FODMAP diet associates with an improvement in IBS symptom scores and effectively increased the quality of life in patients with IBS [15, 22, 25–27]. It is now well documented that IBS is a condition in which several pathophysiological mechanisms are involved in its development and symptom severity. Among them, a distinct fecal microbiota composition and microbiome dysbiosis has been proposed as one of the key factors associated with the disease symptom severity [28–30].
There is increasing evidence that dietary interventions using low-FODMAP diets could improve functional gastrointestinal symptoms in IBS patients particularly through interactions with the gut microbiota [11, 15]. Furthermore, another study suggested that reduction of FODMAPs intake in a GFD consistently and significantly improved the gastrointestinal symptoms in IBS patients who were finally classified as non-celiac gluten sensitivity (NCGS) [31]. In our study, we found that the relative abundance of phylum Firmicutes was higher prior to the start of dietary interventions compared to the phylum Bacteroidetes in patients with IBS. However, there was a clear tendency to increased Bacteroidetes after the dietary interventions, and subsequently, the F/B ratio was significantly decreased. In a recent study by Dieterich et al., clinical and neurological symptoms of NCGS patients who consumed a low-FODMAP diet and especially the GFD significantly improved [32]. In addition, they reported a significant increase in the numbers of Bacteroidetes following a 2-week GFD compared to the low-FODMAP diet (P < 0.01). Furthermore, data obtained from a randomized clinical trial in childhood IBS demonstrated that individuals who respond to a low-FODMAP diet have a greater capacity for saccharolytic metabolism mainly due to higher proportions of Bacteroidaceae, Erysipilotrichaceae, and Clostridiales species than non-responders [33]. Rajilić-Stojanović et al. also reported an approximately twofold increase in the F/B ratio as the major bacterial phyla in 62 IBS patients (Rome II criteria) compared with 46 healthy subjects [34]. This finding has been observed in several other studies, in which the abundance of Firmicutes was enriched together with a reduced abundance of Bacteroidetes in the IBS subjects compared to healthy individuals [35, 36]. In contrast, other studies reported an increase in the content and abundance of Bacteroidetes members in the IBS patients compared to non-IBS subjects [37, 38]. Bacteroidetes are known as complex carbohydrate digesters which are specialized in degrading specific types of dietary fibers in order to maximize energy intake from these kinds of carbohydrates [39, 40].
Several studies have reported a significant depletion in Actinobacteria in the gut of patients with IBS [34, 36, 41]. On the other hand, other studies reported an increase in the relative abundance of Actinobacteria among IBS patients compared to healthy controls [35, 42, 43]. In our study the relative abundance of Actinobacteria was increased, although not statistically significant, after the LF-GFD intervention. In line with our results, McIntosh et al. also reported that a 3-week low-FODMAP diet increased Actinobacteria richness and diversity in patients with IBS [22]. In contrast, a recent study had demonstrated that gut bacteria such as Actinobacteria, Bifidobacterium, and Faecalibacterium prausnitzii were significantly decreased in IBS patients throughout a 9-week low-FODMAP diet along with a reduction in total SCFAs, n-butyric acid, and serum levels of proinflammatory cytokines (IL-6 and IL-8) as compared to baseline [44]. However, the overall inconsistency and differences in the abovementioned results contribute to the difference among study populations, IBS subtypes, duration of dietary intervention, level of dietary adherence, washout period in crossover studies, and variations in taste or other contents outside of FODMAP carbohydrates that may affect the dysbiotic gut microbiota and disease outcomes.
A number of studies have shown that the low-FOMAPs diet decreases the abundance of Bifidobacterium in the gut of IBS patients [12, 16, 22, 25, 27, 44]. Collectively, previous studies have also demonstrated that GFD induced a decrease in Bifidobacterium spp. in the intestinal microbiome of healthy human subjects, celiac disease (CD), and NCGS patients, raising potential concerns after the consumption of a GFD [32, 45, 46]. However, Collado et al. reported increased prevalence of certain Bifidobacterium species including B. adolescentis, B. lactis, and B. dentium after a GFD in CD subjects [47]. In another study from Brazil, the fecal counts of Bifidobacteria were significantly higher in GFD treated-CD (T-CD) patients compared to the healthy subjects [46, 48]. Interestingly, we observed that the relative abundances of lactate-producing bacteria Bifidobacterium and Lactobacillus were increased after the LF-GFD intervention in IBS patients. To our knowledge, no previous studies have evaluated the effects of the LF-GFD intervention simultaneously on IBS patients. Our findings propose that at least in patients with IBS implementing LF-GFD may lead to an increase in the relative abundances of Bifidobacterium and Lactobacillus, although contrary to the other studies which applied these dietary interventions separately.
The family Enterobacteriaceae (phylum Proteobacteria), contains several pathogenic genera such as Escherichia, Shigella, Salmonella, and Campylobacter. Generally, these aerobes were found to be slightly enriched in IBS patients and significantly correlated with IBS symptoms. Moreover, an increase in some of these pathogenic microbiota may contribute to the low rate of mucosal inflammation through overexpression of proinflammatory cytokines IL-6 and IL-8 as seen in IBS patients [29, 34, 49, 50]. In contrast, Tana et al. found no difference in Enterobacteriaceae count between IBS patients and healthy controls. In another study by De Palma et al., Enterobacteriaceae were increased in a group of healthy adult subjects who were on a one-month GFD [51]. In our study, we found no difference in the relative abundance of Enterobacteriaceae before and after the dietary intervention.
The genus Streptococcus is among the dominant bacterial groups present in the upper gastrointestinal tract [52]. Some reports have also showed high fecal amounts of Streptococcus spp., pathogenic bacteria which causes increased expression levels of IL-6 [53], in IBS patients [34], and particularly in IBS-D subtypes [54, 55]. We found no difference in the relative abundance of Streptococcus after the dietary intervention compared to the baseline. Our results are in agreement with another study that found no difference in the relative abundance of Streptococcus species for the low FODMAP diet compared with the sham diet [25]. Furthermore, Bennet et al. also demonstrated no difference in abundance of Streptococcus between responders and non-responders patients with IBS after a 4-week low-FODMAP diet [27].
In this study, over half of the patients had abnormal FC values before dietary intervention. Moreover, a major part of the patients were diagnosed to suffer from IBS-D subtype. It has been reported that alterations in some groups of gut microbiota may induce pro-inflammatory cytokines like IL-6 and IL-8, and thus such IBS patients probably suffer from a chronic low-grade inflammation [56]. In addition, a subgroup of IBS patients known as “IBS-D” may experience a higher level of inflammation [57, 58]. Therefore, alterations in certain groups of gut microbiota and the presence of IBS-D subtype can justify abnormal level of FC among such patients in our study.
Our data suggest that there is a correlation between consuming LF-GFD and decrease of FC which is in line with a study performed by Shulman et al. [59] but are in contrast with a few previous studies which suggested that FC concentration was not increased in IBS patients compared with control subjects [60, 61]. Therefore, it is noteworthy to administrate the diet or a combination of diets of choice for gut microbiota-associated disorders such as IBS in order to normalize the dysbiotic communities of microbiota. However, these controversial findings in the above-mentioned studies may be due to differences in the study design and population.
Conclusions
In summary, our study suggests that patients with IBS who consumed LF-GFD had a significant improvement in IBS symptoms and normalization of their gut microbiota. In addition, our findings indicate inflammation in IBS patients. In which consuming LF-GFD can downregulate intestinal inflammation, and consequently decrease IBS-SSS. To the best of our knowledge, there has been no comprehensive analysis of the effects of LF-GFD on the gut microbiota in a group of unselected Iranian patients with IBS. We also suggest evaluating the effects of this combined dietary intervention on the metabolic output of gut microbiota and its integration with supplementary probiotics to avoid side effects on health due to the unfavorable alteration of the intestinal microbiota in IBS individuals. Future studies are still required to validate the robustness of our findings, and to establish a long-term efficacy and safety of this dietary intervention for personalized nutrition in IBS.
Supplementary Information
Additional file 1 Examples of high FODMAP gluten free foods and some examples of gluten-containing low FODMAP foods.
Additional file 2 The taxon-specific primers used in this study.
Additional file 3 Pie charts representing the mean percentage of the bacterial taxa that constitute the fecal microbiota in IBS patients before and after LFGFD.
Acknowledgements
The authors wish to thank the laboratory staff specially Mrs. Masoumeh Azimirad of the Foodborne and Waterborne Diseases Research Center, Research Institute for Gastroenterology and Liver Diseases, Shahid Beheshti University of Medical Sciences, Tehran, Iran to their sincere assistance.
Authors’ contributions
SS collected the samples and KN performed the real-time PCR analysis; MRN and AY designed and supervised the study; KN, AY, and MO participated in data analysis; KN, AY, and HH wrote the manuscript; MRN, AS, HD, CC, PI, and MRZ critically revised the manuscript. All authors read and approved the final manuscript.
Funding
This study was supported by a Grant (No. RIGLD 961) from Foodborne and Waterborne Diseases Research Center, Research Institute for Gastroenterology and Liver Diseases, Shahid Beheshti Statement of authors University of Medical Sciences, Tehran, Iran.
Availability of data and materials
The datasets supporting the conclusions of this article are included within the article and its additional file.
Declarations
Ethics approval and consent to participate
The study protocol was approved by the Ethical Review Committee of RIGLD at Shahid Beheshti University of Medical Sciences (Project No. IR.SBMU.RIGLD.REC.1396.154). The study was performed according to the revised Declaration of Helsinki 2013 [19] and informed consents were obtained from all subjects and/or their legal guardians prior to sample collection.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no conflicts of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Mohammad Rostami-Nejad, Email: m.rostamii@gmail.com.
Abbas Yadegar, Email: a.yadegar@sbmu.ac.ir, Email: babak_y1983@yahoo.com.
References
- 1.Oka P, Parr H, Barberio B, Black CJ, Savarino EV, Ford AC. Global prevalence of irritable bowel syndrome according to Rome III or IV criteria: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2020;5(10):908–917. doi: 10.1016/S2468-1253(20)30217-X. [DOI] [PubMed] [Google Scholar]
- 2.Schmulson MJ, Drossman DA. What is new in Rome IV. J Neurogastroenterol Motil. 2017;23(2):151–163. doi: 10.5056/jnm16214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chey WD, Kurlander J, Eswaran S. Irritable bowel syndrome: a clinical reviewirritable bowel syndromeirritable bowel syndrome. JAMA. 2015;313(9):949–958. doi: 10.1001/jama.2015.0954. [DOI] [PubMed] [Google Scholar]
- 4.Javanmard A, Ashtari S, Sabet B, Davoodi SH, Rostami-Nejad M, Akbari ME, Niaz A, Mortazavian AM. Probiotics and their role in gastrointestinal cancers prevention and treatment; an overview. Gastroenterol Hepatol Bed Bench. 2018;11(4):284. [PMC free article] [PubMed] [Google Scholar]
- 5.Holtmann G, Shah A, Morrison MJDD. Pathophysiology of functional gastrointestinal disorders: a holistic overview. Dig Dis. 2017;35(S1):5–13. doi: 10.1159/000485409. [DOI] [PubMed] [Google Scholar]
- 6.Shukla R, Ghoshal U, Dhole TN, Ghoshal UC. Fecal microbiota in patients with irritable bowel syndrome compared with healthy controls using real-time polymerase chain reaction: an evidence of dysbiosis. Dig Dis Sci. 2015;60(10):2953–2962. doi: 10.1007/s10620-015-3607-y. [DOI] [PubMed] [Google Scholar]
- 7.Rostami-Nejad M, Ishaq S, Al Dulaimi D, Zali MR, Rostami K. The role of infectious mediators and gut microbiome in the pathogenesis of celiac disease. Arch Iran Med. 2015;18(4):244–249. [PubMed] [Google Scholar]
- 8.Lee BJ, Bak YT. Irritable bowel syndrome, gut microbiota and probiotics. J Neurogastroenterol Motil. 2011;17(3):252–266. doi: 10.5056/jnm.2011.17.3.252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Azimirad M, Rostami-Nejad M, Rostami K, Naji T, Zali MR. The susceptibility of celiac disease intestinal microbiota to Clostridium difficile infection. Am J Gastroenterol. 2015;110(12):1740–1741. doi: 10.1038/ajg.2015.360. [DOI] [PubMed] [Google Scholar]
- 10.Ng QX, Soh AYS, Loke W, Lim DY, Yeo W-S. The role of inflammation in irritable bowel syndrome (IBS) J Inflamm Res. 2018;11:345. doi: 10.2147/JIR.S174982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bohn L, Storsrud S, Liljebo T, Collin L, Lindfors P, Tornblom H, Simren M. Diet low in FODMAPs reduces symptoms of irritable bowel syndrome as well as traditional dietary advice: a randomized controlled trial. Gastroenterology. 2015;149(6):1399–1407.e1392. doi: 10.1053/j.gastro.2015.07.054. [DOI] [PubMed] [Google Scholar]
- 12.Staudacher HM, Lomer MCE, Anderson JL, Barrett JS, Muir JG, Irving PM, Whelan K. Fermentable carbohydrate restriction reduces luminal bifidobacteria and gastrointestinal symptoms in patients with irritable bowel syndrome. J Nutr. 2012;142(8):1510–1518. doi: 10.3945/jn.112.159285. [DOI] [PubMed] [Google Scholar]
- 13.Chong PP, Chin VK, Looi CY, Wong WF, Madhavan P, Yong VC. The microbiome and irritable bowel syndrome: a review on the pathophysiology, current research and future therapy. Front Microbiol. 2019;10:1136–1136. doi: 10.3389/fmicb.2019.01136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.De Giorgio R, Volta U, Gibson PR. Sensitivity to wheat, gluten and FODMAPs in IBS: facts or fiction? Gut. 2016;65(1):169–178. doi: 10.1136/gutjnl-2015-309757. [DOI] [PubMed] [Google Scholar]
- 15.Halmos EP, Power VA, Shepherd SJ, Gibson PR, Muir JG. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology. 2014;146(1):67–75.e65. doi: 10.1053/j.gastro.2013.09.046. [DOI] [PubMed] [Google Scholar]
- 16.Halmos EP, Christophersen CT, Bird AR, Shepherd SJ, Gibson PR, Muir JG. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut. 2015;64(1):93–100. doi: 10.1136/gutjnl-2014-307264. [DOI] [PubMed] [Google Scholar]
- 17.Lacy BE, Patel NK. Rome criteria and a diagnostic approach to irritable bowel syndrome. J Clin Med. 2017;6(11):99. doi: 10.3390/jcm6110099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Windgassen S, Moss-Morris R, Goldsmith K, Chalder T. Key mechanisms of cognitive behavioural therapy in irritable bowel syndrome: the importance of gastrointestinal related cognitions, behaviours and general anxiety. J Psychosom Res. 2019;118:73–82. doi: 10.1016/j.jpsychores.2018.11.013. [DOI] [PubMed] [Google Scholar]
- 19.Association WM. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310(20):2191–2194. doi: 10.1001/jama.2013.281053. [DOI] [PubMed] [Google Scholar]
- 20.Bacchetti De Gregoris T, Aldred N, Clare AS, Burgess JG. Improvement of phylum- and class-specific primers for real-time PCR quantification of bacterial taxa. J Microbiol Methods. 2011;86(3):351–356. doi: 10.1016/j.mimet.2011.06.010. [DOI] [PubMed] [Google Scholar]
- 21.Skodje GI, Sarna VK, Minelle IH, Rolfsen KL, Muir JG, Gibson PR, Veierød MB, Henriksen C, Lundin KEA. Fructan, rather than gluten, induces symptoms in patients with self-reported non-celiac gluten sensitivity. Gastroenterology. 2018;154(3):529–539.e522. doi: 10.1053/j.gastro.2017.10.040. [DOI] [PubMed] [Google Scholar]
- 22.McIntosh K, Reed DE, Schneider T, Dang F, Keshteli AH, De Palma G, Madsen K, Bercik P, Vanner S. FODMAPs alter symptoms and the metabolome of patients with IBS: a randomised controlled trial. Gut. 2017;66(7):1241–1251. doi: 10.1136/gutjnl-2015-311339. [DOI] [PubMed] [Google Scholar]
- 23.Vazquez-Roque MI, Camilleri M, Smyrk T, Murray JA, Marietta E, O'Neill J, Carlson P, Lamsam J, Janzow D, Eckert D, et al. A controlled trial of gluten-free diet in patients with irritable bowel syndrome-diarrhea: effects on bowel frequency and intestinal function. Gastroenterology. 2013;144(5):903–911.e903. doi: 10.1053/j.gastro.2013.01.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wilson B, Rossi M, Kanno T, Hough R, Probert C, Irving P, James Mason A, Lomer M, Whelan K. PWE-126 low fodmap diet effect on IBS gastrointestinal microbiome and metabolites and prediction of response. Gut. 2018;67(Suppl 1):A181–A182. [Google Scholar]
- 25.Staudacher HM, Lomer MCE, Farquharson FM, Louis P, Fava F, Franciosi E, Scholz M, Tuohy KM, Lindsay JO, Irving PM, et al. A diet low in FODMAPs reduces symptoms in patients with irritable bowel syndrome and a probiotic restores bifidobacterium species: a randomized controlled trial. Gastroenterology. 2017;153(4):936–947. doi: 10.1053/j.gastro.2017.06.010. [DOI] [PubMed] [Google Scholar]
- 26.Rao SS, Yu S, Fedewa A. Systematic review: dietary fibre and FODMAP-restricted diet in the management of constipation and irritable bowel syndrome. Aliment Pharmacol Ther. 2015;41(12):1256–1270. doi: 10.1111/apt.13167. [DOI] [PubMed] [Google Scholar]
- 27.Bennet SMP, Bohn L, Storsrud S, Liljebo T, Collin L, Lindfors P, Tornblom H, Ohman L, Simren M. Multivariate modelling of faecal bacterial profiles of patients with IBS predicts responsiveness to a diet low in FODMAPs. Gut. 2018;67(5):872–881. doi: 10.1136/gutjnl-2016-313128. [DOI] [PubMed] [Google Scholar]
- 28.Tap J, Derrien M, Tornblom H, Brazeilles R, Cools-Portier S, Dore J, Storsrud S, Le Neve B, Ohman L, Simren M. Identification of an intestinal microbiota signature associated with severity of irritable bowel syndrome. Gastroenterology. 2017;152(1):111–123.e118. doi: 10.1053/j.gastro.2016.09.049. [DOI] [PubMed] [Google Scholar]
- 29.Pittayanon R, Lau JT, Yuan Y, Leontiadis GI, Tse F, Surette M, Moayyedi P. Gut microbiota in patients with irritable bowel syndrome: a systematic review. Gastroenterology. 2019;157(1):97–108. doi: 10.1053/j.gastro.2019.03.049. [DOI] [PubMed] [Google Scholar]
- 30.Chong PP, Chin VK, Looi CY, Wong WF, Madhavan P, Yong VC. The microbiome and irritable bowel syndrome: a review on the pathophysiology, current research and future therapy. Front Microbiol. 2019;10:1136. doi: 10.3389/fmicb.2019.01136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Biesiekierski JR, Peters SL, Newnham ED, Rosella O, Muir JG, Gibson PR. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology. 2013;145(2):320–328.e323. doi: 10.1053/j.gastro.2013.04.051. [DOI] [PubMed] [Google Scholar]
- 32.Dieterich W, Schuppan D, Schink M, Schwappacher R, Wirtz S, Agaimy A, Neurath MF, Zopf Y. Influence of low FODMAP and gluten-free diets on disease activity and intestinal microbiota in patients with non-celiac gluten sensitivity. Clin Nutr (Edinb, Scotl) 2019;38(2):697–707. doi: 10.1016/j.clnu.2018.03.017. [DOI] [PubMed] [Google Scholar]
- 33.Chumpitazi BP, Cope JL, Hollister EB, Tsai CM, McMeans AR, Luna RA, Versalovic J, Shulman RJ. Randomised clinical trial: gut microbiome biomarkers are associated with clinical response to a low FODMAP diet in children with the irritable bowel syndrome. Aliment Pharmacol Ther. 2015;42(4):418–427. doi: 10.1111/apt.13286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rajilic-Stojanovic M, Biagi E, Heilig HG, Kajander K, Kekkonen RA, Tims S, de Vos WM. Global and deep molecular analysis of microbiota signatures in fecal samples from patients with irritable bowel syndrome. Gastroenterology. 2011;141(5):1792–1801. doi: 10.1053/j.gastro.2011.07.043. [DOI] [PubMed] [Google Scholar]
- 35.Jeffery IB, O'Toole PW, Ohman L, Claesson MJ, Deane J, Quigley EM, Simren M. An irritable bowel syndrome subtype defined by species-specific alterations in faecal microbiota. Gut. 2012;61(7):997–1006. doi: 10.1136/gutjnl-2011-301501. [DOI] [PubMed] [Google Scholar]
- 36.Krogius-Kurikka L, Lyra A, Malinen E, Aarnikunnas J, Tuimala J, Paulin L, Makivuokko H, Kajander K, Palva A. Microbial community analysis reveals high level phylogenetic alterations in the overall gastrointestinal microbiota of diarrhoea-predominant irritable bowel syndrome sufferers. BMC Gastroenterol. 2009;9:95. doi: 10.1186/1471-230X-9-95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Malinen E, Krogius-Kurikka L, Lyra A, Nikkila J, Jaaskelainen A, Rinttila T, Vilpponen-Salmela T, von Wright AJ, Palva A. Association of symptoms with gastrointestinal microbiota in irritable bowel syndrome. World J Gastroenterol. 2010;16(36):4532–4540. doi: 10.3748/wjg.v16.i36.4532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ponnusamy K, Choi JN, Kim J, Lee SY, Lee CH. Microbial community and metabolomic comparison of irritable bowel syndrome faeces. J Med Microbiol. 2011;60(Pt 6):817–827. doi: 10.1099/jmm.0.028126-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.De Filippo C, Cavalieri D, Di Paola M, Ramazzotti M, Poullet JB, Massart S, Collini S, Pieraccini G, Lionetti P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci. 2010;107(33):14691–14696. doi: 10.1073/pnas.1005963107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kolodziejczyk AA, Zheng D, Elinav E. Diet–microbiota interactions and personalized nutrition. Nat Rev Microbiol. 2019;17(12):742–753. doi: 10.1038/s41579-019-0256-8. [DOI] [PubMed] [Google Scholar]
- 41.Rigsbee L, Agans R, Shankar V, Kenche H, Khamis HJ, Michail S, Paliy O. Quantitative profiling of gut microbiota of children with diarrhea-predominant irritable bowel syndrome. Am J Gastroenterol. 2012;107(11):1740–1751. doi: 10.1038/ajg.2012.287. [DOI] [PubMed] [Google Scholar]
- 42.Rangel I, Sundin J, Fuentes S, Repsilber D, de Vos WM, Brummer RJ. The relationship between faecal-associated and mucosal-associated microbiota in irritable bowel syndrome patients and healthy subjects. Aliment Pharmacol Ther. 2015;42(10):1211–1221. doi: 10.1111/apt.13399. [DOI] [PubMed] [Google Scholar]
- 43.Casen C, Vebo HC, Sekelja M, Hegge FT, Karlsson MK, Ciemniejewska E, Dzankovic S, Froyland C, Nestestog R, Engstrand L, et al. Deviations in human gut microbiota: a novel diagnostic test for determining dysbiosis in patients with IBS or IBD. Aliment Pharmacol Ther. 2015;42(1):71–83. doi: 10.1111/apt.13236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hustoft TN, Hausken T, Ystad SO, Valeur J, Brokstad K, Hatlebakk JG, Lied GA. Effects of varying dietary content of fermentable short-chain carbohydrates on symptoms, fecal microenvironment, and cytokine profiles in patients with irritable bowel syndrome. Neurogastroenterol Motil Off J Eur Gastrointest Motil Soc. 2017;29(4):1–9. doi: 10.1111/nmo.12969. [DOI] [PubMed] [Google Scholar]
- 45.Hansen LBS, Roager HM, Sondertoft NB, Gobel RJ, Kristensen M, Valles-Colomer M, Vieira-Silva S, Ibrugger S, Lind MV, Maerkedahl RB, et al. A low-gluten diet induces changes in the intestinal microbiome of healthy Danish adults. Nat Commun. 2018;9(1):4630. doi: 10.1038/s41467-018-07019-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chander AM, Yadav H, Jain S, Bhadada SK, Dhawan DK. Cross-talk between gluten, intestinal microbiota and intestinal mucosa in celiac disease: recent advances and basis of autoimmunity. Front Microbiol. 2018;9:2597. doi: 10.3389/fmicb.2018.02597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Collado MC, Donat E, Ribes-Koninckx C, Calabuig M, Sanz Y. Imbalances in faecal and duodenal Bifidobacterium species composition in active and non-active coeliac disease. BMC Microbiol. 2008;8(1):232. doi: 10.1186/1471-2180-8-232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Golfetto L, de Senna FD, Hermes J, Beserra BT, Franca Fda S, Martinello F. Lower bifidobacteria counts in adult patients with celiac disease on a gluten-free diet. Arq Gastroenterol. 2014;51(2):139–143. doi: 10.1590/S0004-28032014000200013. [DOI] [PubMed] [Google Scholar]
- 49.Ghoshal UC, Shukla R, Ghoshal U, Gwee K-A, Ng SC, Quigley EMM. The gut microbiota and irritable bowel syndrome: friend or foe? Int J Inflam. 2012;2012:151085–151085. doi: 10.1155/2012/151085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Maccaferri S, Candela M, Turroni S, Centanni M, Severgnini M, Consolandi C, Cavina P, Brigidi P. IBS-associated phylogenetic unbalances of the intestinal microbiota are not reverted by probiotic supplementation. Gut Microbes. 2012;3(5):406–413. doi: 10.4161/gmic.21009. [DOI] [PubMed] [Google Scholar]
- 51.De Palma G, Nadal I, Collado MC, Sanz Y. Effects of a gluten-free diet on gut microbiota and immune function in healthy adult human subjects. Br J Nutr. 2009;102(8):1154–1160. doi: 10.1017/S0007114509371767. [DOI] [PubMed] [Google Scholar]
- 52.Booijink CC, El-Aidy S, Rajilic-Stojanovic M, Heilig HG, Troost FJ, Smidt H, Kleerebezem M, De Vos WM, Zoetendal EG. High temporal and inter-individual variation detected in the human ileal microbiota. Environ Microbiol. 2010;12(12):3213–3227. doi: 10.1111/j.1462-2920.2010.02294.x. [DOI] [PubMed] [Google Scholar]
- 53.Jalanka-Tuovinen J, Salojarvi J, Salonen A, Immonen O, Garsed K, Kelly FM, Zaitoun A, Palva A, Spiller RC, de Vos WM. Faecal microbiota composition and host-microbe cross-talk following gastroenteritis and in postinfectious irritable bowel syndrome. Gut. 2014;63(11):1737–1745. doi: 10.1136/gutjnl-2013-305994. [DOI] [PubMed] [Google Scholar]
- 54.Kassinen A, Krogius-Kurikka L, Makivuokko H, Rinttila T, Paulin L, Corander J, Malinen E, Apajalahti J, Palva A. The fecal microbiota of irritable bowel syndrome patients differs significantly from that of healthy subjects. Gastroenterology. 2007;133(1):24–33. doi: 10.1053/j.gastro.2007.04.005. [DOI] [PubMed] [Google Scholar]
- 55.Salonen A, de Vos WM, Palva A. Gastrointestinal microbiota in irritable bowel syndrome: present state and perspectives. Microbiology (Reading, England) 2010;156(Pt 11):3205–3215. doi: 10.1099/mic.0.043257-0. [DOI] [PubMed] [Google Scholar]
- 56.Bashashati M, Moradi M, Sarosiek I. Interleukin-6 in irritable bowel syndrome: a systematic review and meta-analysis of IL-6 (-G174C) and circulating IL-6 levels. Cytokine. 2017;99:132–138. doi: 10.1016/j.cyto.2017.08.017. [DOI] [PubMed] [Google Scholar]
- 57.Melchior C, Simrén M. Positive effect of bimodal release ondansetron in irritable bowel syndrome with diarrhea: relevance of low-grade inflammation? In: LWW. 2020. [DOI] [PubMed]
- 58.Zhang Y, Wu XX, Li S, Wu JF, Han S, Lin ZJ, Ding SZ, Gong WJ. Peroxiredoxin 1 as an inflammatory marker in diarrhea-predominant and postinfectious irritable bowel syndrome. Neurogastroenterol Motil. 2020;32(2):e13741. doi: 10.1111/nmo.13741. [DOI] [PubMed] [Google Scholar]
- 59.Shulman RJ, Eakin MN, Czyzewski DI, Jarrett M, Ou C-N. Increased gastrointestinal permeability and gut inflammation in children with functional abdominal pain and irritable bowel syndrome. J Pediatr. 2008;153(5):646–650. doi: 10.1016/j.jpeds.2008.04.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Dunlop SP, Hebden J, Campbell E, Naesdal J, Olbe L, Perkins AC, Spiller RC. Abnormal intestinal permeability in subgroups of diarrhea-predominant irritable bowel syndromes. Am J Gastroenterol. 2006;101(6):1288–1294. doi: 10.1111/j.1572-0241.2006.00672.x. [DOI] [PubMed] [Google Scholar]
- 61.Marshall J, Thabane M, Garg A, Clark W, Meddings J, Collins S, Investigators W. Intestinal permeability in patients with irritable bowel syndrome after a waterborne outbreak of acute gastroenteritis in Walkerton, Ontario. Aliment Pharmacol Ther. 2004;20(11–12):1317–1322. doi: 10.1111/j.1365-2036.2004.02284.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1 Examples of high FODMAP gluten free foods and some examples of gluten-containing low FODMAP foods.
Additional file 2 The taxon-specific primers used in this study.
Additional file 3 Pie charts representing the mean percentage of the bacterial taxa that constitute the fecal microbiota in IBS patients before and after LFGFD.
Data Availability Statement
The datasets supporting the conclusions of this article are included within the article and its additional file.