Abstract
Inflammation, due to infectious pathogens or other non-infectious stimuli, during pregnancy is associated with elevated risk for neurodevelopmental disorders such as schizophrenia and autism in the offspring. Although historically identified through retrospective epidemiologic studies, the relationship between maternal immune activation and offspring neurodevelopmental disease risk is now well established because of clinical studies which utilized prospective birth cohorts, serologically confirmed infection records, and subsequent long-term offspring follow-up. These efforts have been corroborated by preclinical research which demonstrates anatomical, biochemical, and behavioural alterations that resemble the clinical features of psychiatric illnesses. Intervention studies further demonstrate causal roles of inflammatory mediators, such as cytokines, in these long-lasting changes in behaviour and brain. This review summarizes a selection of maternal immune activation literature that explores the relationship between these inflammatory mediators and the neuropsychiatric-like effects later observed in the offspring. This literature is presented alongside emerging information regarding SARS-CoV-2 infection in pregnancy, with discussion of how these data may inform future research regarding the effects of the present coronavirus pandemic on emerging birth cohorts.
Keywords: Schizophrenia, Autism, Inflammation, Behaviour
1. Introduction
Psychiatric illnesses such as schizophrenia and autism are complex neurological conditions. Both are common developmental disorders that affect approximately 1% of the population and cause significant societal burden (Knapp et al., 2004; Lai et al., 2014). The symptoms that accompany them are diverse and often debilitating, including hallucinations, delusions, emotional flattening, avolition, social impairments, and cognitive dysfunction (Tandon et al., 2013). Both schizophrenia and autism are associated with prenatal risk factors, including maternal immune activation. The ongoing Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) pandemic has resulted in millions of infections worldwide with a novel viral pathogen. Infected individuals include pregnant women, and the impact this will have on psychiatric disease risk for the offspring is not yet known. Consideration of the known literature on maternal immune activation (MIA) and psychiatric illness risk in the context of the novel virus may help inform future public health initiatives. There is need for continued research of the factors that influence psychiatric illnesses risk to improve disease diagnosis, treatment and prognosis, and exposure to SARS-CoV-2 in utero, or its associated maternal immune response, may contribute to increased risk for the development of psychiatric illnesses for future generations.
The aetiology of schizophrenia and autism are believed to involve a complex interplay of genetic and environmental factors (Michel et al., 2012). Schizophrenia is polygenic, and genome-wide association studies (GWAS) have characterized associations among many genes (Ripke et al., 2014). Notably, schizophrenia gene associations were enriched in immune tissues, including major histocompatibility complex (MHC) and factors in acquired immunity such as B-lymphocyte lineages (Dhindsa and Goldstein, 2016; Ripke et al., 2014; Sekar et al., 2016). The evidence for genetic involvement in autism points to risk associated with rare, large effect variants; however, evidence for autism-specific risk factors has not been substantiated (Myers et al., 2020). While the role for genetics in autism continue to emerge, genetic links to the immune system are increasingly well established for schizophrenia.
Prior to the identification of a genetic link to the immune system, the role of inflammation as an environmental risk factor for schizophrenia was realized through epidemiological studies. Specifically, inflammation due to infection in pregnant women was linked to increased risk of psychiatric illness in their offspring as they reached adulthood (Brown, 2006, 2012; Brown et al., 2004; Brown and Meyer, 2018). Early epidemiological studies produced conflicting results, but later studies focussed on serologically confirmed influenza cases using serum antibodies or other markers of inflammation and have provided additional evidence for maternal inflammation during development as a risk factor for schizophrenia (Brown et al., 2004; Ellman and Susser, 2009; Scola and Duong, 2017). Similar associations have been described for autism risk (Atladóttir et al., 2010; Jiang et al., 2016; Lee et al., 2015; Ornoy et al., 2015; Patterson, 2011; Ravaccia and Ghafourian, 2020; Zerbo et al., 2015). Whether a similar increase in neurodevelopmental diseases will occur in the years following SARS-CoV-2 emergence remains to be seen. This short review seeks to summarize pre-existing knowledge regarding maternal inflammation due to infectious disease and the elevated risk for neuropsychiatric outcomes in the offspring, with discussion of the potential impact for the future of the field in the years following SARS-CoV-2 emergence.
2. Clinical evidence for adverse outcomes in MIA-exposed offspring
Large prospective birth cohorts with maternal health data and serum samples, as well as long-term offspring follow-up, have been a valuable resource in revealing links between disease outbreaks and psychiatric illness. Serologically confirmed exposure to influenza B, but not influenza A, in pregnancy was associated with significantly lower cognitive scores in offspring at age 7 who would later develop schizophrenia (Ellman et al., 2009). This association was not found in control offspring also exposed to Influenza B, suggesting the influence of other factors in offspring cognitive performance and eventual diagnoses (Ellman et al., 2009).
The impact of maternal inflammation on offspring brain development has been further demonstrated by associating offspring cognitive performance and behaviour phenotypes to inflammatory markers in maternal sera. Foetal exposure to elevated levels of interleukin-8 (IL-8) was associated with significant anatomical deviations among offspring diagnosed with schizophrenia as adults, including abnormalities in brain regions implicated in psychiatric illnesses; however, control participants did not show any relationship between neuroanatomy and IL-8 levels in maternal sera (Ellman et al., 2010). Levels of maternal IL-6 and C-reactive protein correlated with altered functional connectivity in the salience network in neonates, as well as behaviour and cognitive performance in the offspring as neonates and toddlers (Spann et al., 2018). Maternal inflammation also affects offspring externalizing and internalizing symptoms associated with depressive, impulsive, and withdrawn phenotypes, but the timing of exposure and the sex of the offspring influence these outcomes (Mac Giollabhui et al., 2019). These data reveal that maternal inflammation influences offspring neurodevelopmental trajectory, affecting cognition, emotion, behaviour, and risk for psychiatric and mood disorders. It should be considered that maternal inflammation induced by SARS-CoV-2 infection may also influence offspring neurodevelopment. While prospective clinical studies have uncovered vital associations between inflammation and offspring outcomes in human cohorts, they have limited ability to probe underlying mechanisms (Brown and Meyer, 2018). Preclinical models have enabled fundamental research to investigate these physiological and molecular mechanisms of inflammation and developmental alterations.
3. Preclinical evidence for offspring neurodevelopmental abnormalities in MIA models
Basic science has supported the findings from prospective clinical studies and further demonstrated a role of inflammation in the emergence of neurodevelopmental psychiatric disease using animal models. Maternal immune activation (MIA) models are the most frequently seen neurodevelopmental models and involve induction of an immune challenge to rodents during pregnancy. Various inflammatory agents have been examined in pregnant rats or mice, with experimental endpoints ranging from acute maternal or embryonic samples to longer-term assessment of adult offspring. The agents most commonly administered are not infectious pathogens, but immune stimulants such as lipopolysaccharide (LPS) or polyinosinic:polycytidylic acid (polyI:C, a synthetic double stranded RNA molecule) which mimic bacterial and viral stimulation of the innate immune system, respectively (Boksa, 2010; Brown and Meyer, 2018; Chlodzinska et al., 2011; Soumiya et al., 2011; Winship et al., 2019). These models helped establish that physiological challenges in utero result in psychiatric illness-like abnormalities in experimental settings.
Behaviour tasks in rodent models can reveal the specific nature of the abnormalities present in MIA offspring. Behaviour tests are a means of demonstrating face validity and relating functional deficits to underlying anatomic or biochemical abnormalities which cannot be achieved in patient populations. In rodents, the positive symptoms are frequently assessed using locomotor activity in a novel environment, or following a pharmacological challenge (amphetamine, apomorphine, or MK-801). Negative symptoms can be assessed through altered social interaction tasks, or sucrose-preference tests (Jones et al., 2011, Moore et al., 2006, Winship et al., 2019). Many rodent tests have been validated to assess cognition, and those with relevance to schizophrenia have been extensively reviewed (Young et al., 2009). Briefly, cognitive flexibility (set-shifting and reversal learning) and object-in-place memory are typically disturbed in schizophrenia models (Ballendine et al., 2015; Howland et al., 2012; Zhang et al., 2012). In a pair of recent publications, my colleagues and I conducted a battery of behaviour tasks to assess the three main symptom domains of schizophrenia in polyI:C-exposed rat offspring (Lins et al, 2018, 2019). We found significantly increased locomotor activity following MK-801 administration compared to controls in males (Lins et al., 2018), but not females (Lins et al., 2019). While altered locomotor activity is a well-established consequence of MIA in rodents, other studies report hypo-locomotion following systemic MK-801, proposed to indicate NMDA receptor hypofunction (Van den Eynde, 2014). Further, MIA offspring of both sexes displayed reduced social interaction in a three-chambered sociability task (Lins et al, 2018, 2019). Cognitive deficits in polyI:C-exposed offspring were found in multiple tasks, including reduced oddity preference, impaired visual, but not tactile, recognition memory; impaired late-phase reversal learning; and facilitated set-shifting performance (Lins et al., 2018, 2019). While we did not observe altered sensorimotor gating in MIA offspring, other studies report polyI:C-exposure in utero impairs prepulse inhibition (Ballendine et al., 2015; Basta-Kaim et al., 2015; Howland et al., 2012; Wolff and Bilkey, 2008, 2010).
Behaviour studies also reveal autism-like behaviours in offspring following MIA (al-Haddad et al., 2019; Estes and McAllister, 2015; Hsiao and Patterson, 2011; Wang et al., 2017). While schizophrenia-focussed studies test offspring in adulthood, autism-focussed research assess behaviours in young animals and report reduced sociability and play, altered ultrasonic vocalizations, and repetitive behaviours such as increased marble-burying (al-Haddad et al., 2019; Choi et al., 2016; Shin Yim et al., 2017).
Overall, behaviour abnormalities in MIA offspring have been widely reported (Ballendine et al., 2015; Missault et al., 2014; Piontkewitz et al., 2011; Van den Eynde et al., 2014; Zuckerman and Weiner, 2005) with inconsistencies occasionally seen (Mueller et al., 2021). These differences may be driven by several factors, including variations in the response to immune stimulant polyI:C, or additional stressors present during gestation (Bronson and Bale, 2014; Giovanoli et al., 2013, 2016; Monte et al., 2017; Moriyama et al., 2013; Mueller et al., 2019). A recent paper described factors that underlie differential outcomes in MIA with the goal of improving reproducibility, while illustrating that factors that contribute to neurodevelopment are diverse (Kentner et al., 2019).
Like the prospective birth cohorts that followed human pregnancies with long-term follow-up of the offspring, preclinical research has sought to link inflammatory molecules in maternal sera to offspring outcomes, and to modulate developmental outcomes through interventions. IL-6 has been repeatedly implicated in MIA studies (Smith et al., 2007; Wu et al., 2017). In a mouse model, maternal treatment with IL-6 alone was sufficient to produce impaired PPI and latent inhibition in the offspring. Further, neurodevelopmental abnormalities were absent in IL-6 knockout mice, or mice administered anti-IL-6 antibody to block IL-6 receptors (Smith et al., 2007). IL-6 signalling in the placenta is also required for the development of abnormal phenotypes, as shown in a study that assessed autism-like behaviours of reduced sociability and increased marble burying. Inhibition of IL-6 signalling through knockout of placental IL-6 receptors prevented behaviour deficits, prevented increased IL-6 gene expression in the foetal brain, and prevented cerebellar neurodevelopmental abnormalities (Wu et al., 2017). In similar fashion, polyI:C-exposed mouse offspring displayed autism-like behaviours of altered ultrasonic vocalization, increased marble burying, and reduced sociability alongside elevated foetal brain IL-17Ra mRNA, and abnormal gene expression patterns, resulting in irregular cortical lamination. These aberrations were prevented by modulation of IL-17a signalling through anti-IL-17a antibody administration, or by knockout of retinoic acid receptor-related orphan nuclear receptor gamma t-dependent effector T lymphocytes (Choi et al., 2016). Other inflammatory molecules such as TNFα were associated with cerebellar abnormalities such as a loss in Purkinje cells, and altered synapse formation in MIA-exposed offspring (Pendyala et al., 2017). Another approach to ameliorate the effects of MIA used a chemokine receptor antagonist G31P to block CXCL1, the rat chemokine receptor analogous to the human IL-8 receptor. With this intervention, crossmodal memory deficits in polyI:C-exposed offspring were restored to control levels, although deficits in PPI, associative memory, and cognitive flexibility were not affected (Ballendine et al., 2015). These data reveal that stimulation of the maternal immune response in pregnancy has potential for profound effects on offspring development, cognition, and behaviour; however, modulation of the immune response through some targets can ameliorate deficits and rescue the trajectory of neurodevelopment.
4. Considerations for the future: from historical epidemiology to a present-day pandemic
The SARS-CoV-2 pandemic has impacted healthcare and economies on a global scale (Chakraborty and Maity, 2020). Vaccine distribution, initiated in December 2020, ushers in a potential turning point with a promise of ‘return to normalcy’ (Oliver et al., 2020), but historical understanding of viral epidemics suggest that the full impact of the SARS-CoV-2 pandemic may not be fully appreciated for several decades. As outlined above, a long-term consequence of infectious disease outbreaks is increased psychiatric diagnoses as the offspring of infection-exposed pregnancies reach adulthood (Scola and Duong, 2017; Watson et al., 2021; Zimmer et al., 2021).
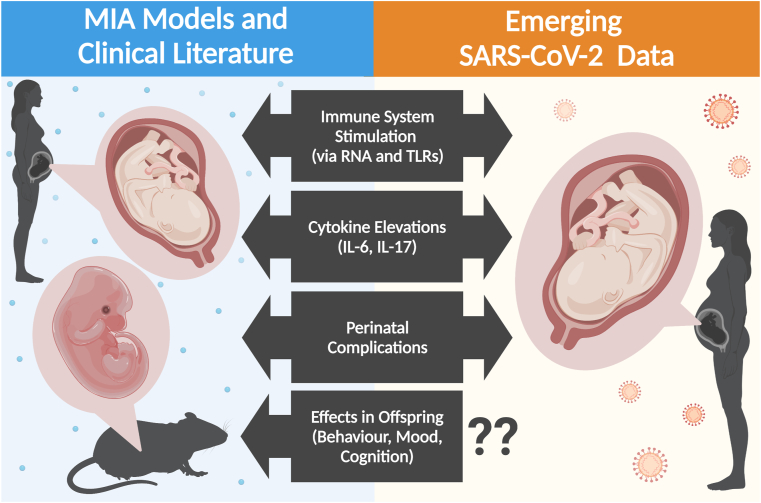
SARS-CoV-2 is an RNA virus and the immune response it initiates bears some similarities to that of polyI:C, the synthetic RNA virus mimetic commonly used in preclinical MIA research. Detailed characterization of the current knowledge of the SARS-CoV-2 immune response is beyond the scope of this short review, but notably includes serum elevations of cytokines implicated in psychiatric illness-like outcomes by preclinical MIA research, such as IL-6 and IL-17 (Azkur et al., 2020). Much like viral mimetic polyI:C, SARS-CoV-2 is recognized by toll-like receptors (TLR) and initiates NF-κB/MAPK signalling pathways, resulting in cytokine release (Alexopoulou et al., 2001; Azkur et al., 2020; Reisinger et al., 2015). Notably, polyI:C is largely recognized by TLR3, while SARS-CoV-2 recognition involves TLR7, which result in distinct offspring behaviour abnormalities (Meyer, 2019; Missig et al., 2020; Moreno-Eutimio et al., 2020). MIA with TLR3 activation often results in heightened anxiety-like behaviour in offspring, but MIA with TLR7 activation produced the opposite effect (Missig et al., 2020). While the behaviour domains altered in offspring following TLR3- and TLR4-mediated MIA are similar, including ultrasonic vocalizations, sociability, and repetitive behaviours, they are qualitatively different (Missig et al., 2020). While preclinical research should not be presumed to translate directly to clinical events, consideration of the similarities and differences between immune stimuli and the maternal immune response in well-characterized models and SARS-CoV-2 may inform expectations about long-term effects in the years following the pandemic (Fig. 1).
Fig. 1.
Summary of preclinical and clinical events associated with maternal immune activation and neurodevelopmental effects in the offspring. Factors which may be present in, or induced by, SARS-CoV-2 infection, similar to those reported in prior MIA literature. The effects of SARS-CoV-2 infection during pregnancy and birth is a rapidly evolving area and data continues to emerge. Created with BioRender.com.
It is increasingly recognized that the degree of risk conferred by MIA is not uniform (Meyer, 2019, Mueller et al., 2021). A recent review from Meyer (2019) proposed factors that may contribute to differential offspring susceptibility, highlighting heterogeneity of inflammatory stimuli as an important element. Regarding MIA due to infectious pathogens, the most severe risk for adverse neurodevelopmental outcomes appears to follow infections where vertical transmission occurs (rubella virus; herpes simplex virus; cytomegalovirus; Zika virus; Toxoplasma gondii parasite) (Meyer, 2019). In addition to a greater latent psychiatric illness risk (schizophrenia, autism, bipolar disorder, major depressive disorder) than non-teratogenic agents, these pathogens can result in congenital consequences not limited to microcephaly, cerebral palsy, and blindness/deafness (Coyne and Lazear, 2016, Meyer, 2019). The potential for vertical transmission in SARS-CoV-2 infected pregnancies is a concern, as well as teratogenicity and other perinatal complications, due to their associations with offspring risk of neurodevelopmental disorders and adverse events (al-Haddad et al., 2019, Alzamora et al., 2020, Brown, 2006, Brown, 2012, Chen et al., 2020, Chen et al., 2020, Deverman and Patterson, 2009, Hayakawa et al., 2020, Meyer, 2019, Ornoy et al., 2015, Zimmer et al., 2021). Early data suggested that vertical transmission of SARS-CoV-2 does not occur or is incredibly rare, does not appear to be teratogenic, nor to increase risk of stillbirth, preterm labour, or miscarriage (Alzamora et al., 2020; H. Chen et al., 2020; Y. Chen et al., 2020; Dashraath et al., 2020; Hayakawa et al., 2020; Marim et al., 2020; Zimmer et al., 2021). More recent data challenge this with reports of associations between SARS-CoV-2 in pregnancy and perinatal complications such as preterm birth, low birth weight, intrauterine growth restriction, and placental abnormalities including inflammation and malperfusion (Cavalcante et al., 2021). Continued study of these emerging data regarding SARS-CoV-2 in pregnancy is of critical importance now and into the future.
Increased risk of neurodevelopmental disease is also associated with pathogens that typically do not cross the placenta (influenza, upper respiratory infections) (al-Haddad et al., 2019). This suggests that the maternal immune response influences neurodevelopmental trajectory, altering offspring susceptibility to psychiatric illness (al-Haddad et al., 2019). As suggested by the name, SARS-CoV-2 is largely defined by respiratory symptoms, lending face similarities to infections known to be associated with psychiatric illness risk (Bordallo et al., 2020; Shuid et al., 2021). MIA via infection likely intersects with maternal stress and in the context of MIA due to SARS-CoV-2, these interacting factors are especially pertinent. Elevated stress has been documented in pregnant women during the pandemic, including clinically relevant anxiety and depression (Barišić, 2020, Kotlar et al., 2021, Meyer, 2019, Preis et al., 2020).
In addition to stress, other maternal and perinatal factors are believed to contribute to offspring risk. This was recently reviewed (Meyer, 2019), and such factors should be considered regarding SARS-CoV-2 as they may assist in identifying at-risk populations in the coming years. The presence of gestational diabetes, micronutrient deficiencies, and microbiome alterations may all interact with pathogen-induced maternal inflammation to confer increased risk, as well as postnatal events such as trauma, stress, or Δ-9-tetrahydrocannabinol exposure (Brown, 2011; Debost et al., 2017; Giovanoli et al., 2013, 2016; Hsiao et al., 2013; Li et al., 2018; Mattei and Pietrobelli, 2019; Money et al., 2018; Morais et al., 2018; Moretti et al., 2015; Zamberletti et al., 2015). Alongside interactions that may exacerbate offspring susceptibility, resilience to the neurodevelopmental consequences of MIA may also be conferred by several factors, including robust maternal nutrition and microbiome (Hsiao et al., 2013; Luan et al., 2018). In addition, a recent mouse study found transcriptional differences in cortical and subcortical brain tissue, and distinct plasma cytokine profiles among offspring that were susceptible or resilient (see Mueller et al., 2021 for further reading).
While the impact of the present pandemic is undoubtedly unmatched, recent history has seen the emergence of similar viruses (Watson et al., 2021; Zimmer et al., 2021). Within the past 20 years, two coronaviruses have emerged and spread to pandemic proportions, namely severe acute respiratory syndrome coronavirus (SARS, now also known as SARS-CoV-1) in 2003 and Middle East respiratory syndrome coronavirus (MERS, or MERS-CoV) in 2012 (Singh, 2016; Zimmer et al., 2021). Although their reach and impact were not seen at the scale of SARS-CoV-2, these events may provide valuable insight into the anticipated effects of the current pandemic. The MERS-CoV outbreak was too recent for data on schizophrenia emergence, and offspring born during the SARS-CoV-1 pandemic are only now reaching the age associated with diagnosis (Watson et al., 2021; Zimmer et al., 2021). Reports of neurodevelopmental outcomes in these birth cohorts are still forthcoming. Given the age of onset for autism is earlier than schizophrenia, it is unclear why reports are not yet available, but the characterization of SARS-CoV-1 and MERS-CoV birth cohorts regarding autism incidence and presentation should be prioritized due to potential to inform the response to SARS-CoV-2.
Several experts have called for priority investigation of MIA in light of SARS-CoV-2 emergence (Holmes et al., 2020; López-Díaz et al., 2021; McAlonan et al., 2020). Rapid translation of existing knowledge into clinical practice and the development of pathogen-specific knowledge will benefit public health for future generations (McAlonan et al., 2020). The scale of the pandemic presents the opportunity to improve upon past studies, and the collection of prospective data now (maternal sera, perinatal complication data, offspring follow-up, and neuroimaging) will form a vital resource for future research (Ricardo Martins-Filho et al., 2020; Zimmer et al., 2021).
Special consideration must be given to sex effects when considering the risk for offspring of exposed pregnancies, and this should continue be prioritized in MIA research moving forward. While long-term data following the offspring of SARS-CoV-2 pregnancies have yet to be realized, prior work in the field of MIA have demonstrated that sex as a biological variable influences outcomes, and sex-based bias is present in the field with a preferential inclusion of male animals in preclinical studies (Coiro and Pollak, 2019; Goldstein et al., 2021; Mac Giollabhui et al., 2019; Will et al., 2017). The relative lack of evidence of how MIA manifests in female offspring may present a challenge in the years post-pandemic. This illustrates how continued emphasis reducing sex bias in biological studies is paramount to improving translational capacity (Clayton, 2018; Wainer et al., 2020).
5. Conclusion
Associations between inflammation in pregnancy and offspring psychiatric illness risk has been demonstrated through GWAS, epidemiological, clinical, and preclinical research. Maternal inflammation impacts offspring neurodevelopment, and the elevated immune response associated with infectious disease is associated with increased risk of neurodevelopmental disorders. The long-term neuropsychiatric sequelae of SARS-CoV-2 infection during pregnancy on offspring is unknown, yet the data reviewed here suggest that elevated risk for offspring neurodevelopmental disorders is a distinct possibility. As a result, the SARS-CoV-2 pandemic will shape the field of psychoneuroimmunology for years to come.
Declaration of competing interest
None.
Acknowledgements
The author thanks John Howland and Sarah Hellewell for their suggestions and insight during the preparation of this manuscript.
Biography

Brittney Lins. Dr. Brittney Lins is a Canadian early career researcher trained in neuroscience and interested in cognition, behaviour, inflammation, and pharmacology. She is currently undertaking postdoctoral research at Curtin University (2019-present) within the Curtin Health Innovation Research Institute in Perth, Australia.
Dr. Lins completed her Ph.D. at the University of Saskatchewan, Canada (conferred 2020), as well as prior M.Sc. (2014) and B.Sc.Hons. (2013) degrees. Her graduate research was conducted under the supervision of Prof. John Howland, where she investigated the relationships between maternal inflammatory mediators during pregnancy and the development of cognitive and behavioural abnormalities in the offspring. Behaviours of interest included both spontaneous and rewarded cognitive tests, as well as sociability, locomotion, and prepulse inhibition. Previously, she applied acute glutamatergic and dopaminergic interventions to both disrupt and rescue cognitive performance in operant conditioning tasks.
In her current postdoctoral role, Dr. Lins is mentored by Professors Lindy Fitzgerald and John Mamo at Curtin University, where she is applying her knowledge of inflammation, cognition, and behaviour to investigate damage mechanisms in models of traumatic brain injury and neurodegeneration. Various imaging techniques complement her prior expertise to study the influence of glial cells on degeneration and functional losses.
References
- al-Haddad B.J.S., Oler E., Armistead B., Elsayed N.A., Weinberger D.R., Bernier R., Burd I., Kapur R., Jacobsson B., Wang C., Mysorekar I., Rajagopal L., Adams Waldorf K.M. The fetal origins of mental illness. Am. J. Obstet. Gynecol. 2019;221:549–562. doi: 10.1016/j.ajog.2019.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexopoulou L., Holt A.C., Medzhitov R., Flavell R.A. Recognition of double-stranded RNA and activation of NF-κB by Toll-like receptor 3. Nature. 2001;413:732–738. doi: 10.1038/35099560. [DOI] [PubMed] [Google Scholar]
- Alzamora M.C., Paredes T., Caceres D., Webb C.M., Webb C.M., Valdez L.M., Valdez L.M., La Rosa M., La Rosa M. Severe COVID-19 during pregnancy and possible vertical transmission. Am. J. Perinatol. 2020;37:861–865. doi: 10.1055/s-0040-1710050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atladóttir H.Ó., Thorsen P., Østergaard L., Schendel D.E., Lemcke S., Abdallah M., Parner E.T. Maternal infection requiring hospitalization during pregnancy and autism spectrum disorders. J. Autism Dev. Disord. 2010;40:1423–1430. doi: 10.1007/s10803-010-1006-y. [DOI] [PubMed] [Google Scholar]
- Azkur A.K., Akdis M., Azkur D., Sokolowska M., van de Veen W., Brüggen M.C., O'Mahony L., Gao Y., Nadeau K., Akdis C.A. Immune response to SARS-CoV-2 and mechanisms of immunopathological changes in COVID-19. Allergy Eur. J. Allergy Clin. Immunol. 2020;75:1564–1581. doi: 10.1111/all.14364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballendine S.A., Greba Q., Dawicki W., Zhang X., Gordon J.R., Howland J.G. Behavioral alterations in rat offspring following maternal immune activation and ELR-CXC chemokine receptor antagonism during pregnancy: implications for neurodevelopmental psychiatric disorders. Prog. Neuro-Psychopharmacol. Biol. Psychiatry. 2015;57:155–165. doi: 10.1016/j.pnpbp.2014.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barišić A. Conceived in the covid-19 crisis: impact of maternal stress and anxiety on fetal neurobehavioral development. J. Psychosom. Obstet. Gynaecol. 2020;41(3):246. doi: 10.1080/0167482X.2020.1755838. [DOI] [PubMed] [Google Scholar]
- Basta-Kaim A., Fijał K., Ślusarczyk J., Trojan E., Głombik K., Budziszewska B., Leśkiewicz M., Regulska M., Kubera M., Lasoń W., Wędzony K. Prenatal administration of lipopolysaccharide induces sex-dependent changes in glutamic acid decarboxylase and parvalbumin in the adult rat brain. Neuroscience. 2015;287:78–92. doi: 10.1016/J.NEUROSCIENCE.2014.12.013. [DOI] [PubMed] [Google Scholar]
- Boksa P. Effects of prenatal infection on brain development and behavior: a review of findings from animal models. Brain Behav. Immun. 2010;24:881–897. doi: 10.1016/j.bbi.2010.03.005. [DOI] [PubMed] [Google Scholar]
- Bordallo B., Bellas M., Cortez A.F., Vieira M., Pinheiro M. Severe COVID-19: what have we learned with the immunopathogenesis? Advances in Rheumatology. 2020;60:50. doi: 10.1186/s42358-020-00151-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bronson S.L., Bale T.L. Prenatal stress-induced increases in placental inflammation and offspring hyperactivity are male-specific and ameliorated by maternal antiinflammatory treatment. Endocrinology. 2014;155(7):2635–2646. doi: 10.1210/en.2014-1040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown A.S. The environment and susceptibility to schizophrenia. Prog. Neurobiol. 2011;93(1):23–58. doi: 10.1016/j.pneurobio.2010.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown A.S. Epidemiologic studies of exposure to prenatal infection and risk of schizophrenia and autism. Dev. Neurobiol. 2012;72:1272–1276. doi: 10.1002/dneu.22024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown A.S. Prenatal Infection as a risk factor for schizophrenia. Schizophr. Bull. 2006;32:200–202. doi: 10.1093/schbul/sbj052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown A.S., Begg M.D., Gravenstein S., Schaefer C.A., Wyatt R.J., Bresnahan M., Babulas V.P., Susser E.S. Serologic evidence of prenatal influenza in the etiology of schizophrenia. Arch. Gen. Psychiatr. 2004;61:774. doi: 10.1001/archpsyc.61.8.774. [DOI] [PubMed] [Google Scholar]
- Brown A.S., Meyer U. Maternal immune activation and neuropsychiatric illness: a translational research perspective. Am. J. Psychiatr. 2018;175:1073–1083. doi: 10.1176/appi.ajp.2018.17121311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavalcante M.B., Cavalcante C.T., de M.B., Sarno M., Barini R., Kwak-Kim J. Maternal immune responses and obstetrical outcomes of pregnant women with COVID-19 and possible health risks of offspring. J. Reprod. Immunol. 2021;143 doi: 10.1016/j.jri.2020.103250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chakraborty I., Maity P. COVID-19 outbreak: migration, effects on society, global environment and prevention. Sci. Total Environ. 2020;728:138882. doi: 10.1016/j.scitotenv.2020.138882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H., Guo J., Wang C., Luo F., Yu X., Zhang W., Li J., Zhao D., Xu D., Gong Q., Liao J., Yang H., Hou W., Zhang Y. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: a retrospective review of medical records. Lancet. 2020;395:809–815. doi: 10.1016/S0140-6736(20)30360-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y., Peng H., Wang L., Zhao Y., Zeng L., Gao H., Liu Y. Infants born to mothers with a new coronavirus (COVID-19) Front. Pediatr. 2020;8 doi: 10.3389/fped.2020.00104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chlodzinska N., Gajerska M., Bartkowska K., Turlejski K., Djavadian R.L. Lipopolysaccharide injected to pregnant mice affects behavior of their offspring in adulthood. Acta Neurobiol. Exp. 2011;71:519–527. doi: 10.55782/ane-2011-1868. [DOI] [PubMed] [Google Scholar]
- Choi G.B., Yim Y.S., Wong H., Kim Sangdoo, Kim H., Kim Sangwon V., Hoeffer C.A., Littman D.R., Huh J.R. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science. 2016;351:933–939. doi: 10.1126/science.aad0314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clayton J.A. Applying the new SABV (sex as a biological variable) policy to research and clinical care. Physiol. Behav. 2018;187:2–5. doi: 10.1016/j.physbeh.2017.08.012. [DOI] [PubMed] [Google Scholar]
- Coiro P., Pollak D.D. Sex and gender bias in the experimental neurosciences: the case of the maternal immune activation model. Transl. Psychiatry. 2019;9:90. doi: 10.1038/s41398-019-0423-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coyne C.B., Lazear H.M. Zika virus - reigniting the TORCH. Nat. Rev. Microbiol. 2016;14(11):707–715. doi: 10.1038/nrmicro.2016.125. [DOI] [PubMed] [Google Scholar]
- Dashraath P., Wong J.L.J., Lim M.X.K., Lim L.M., Li S., Biswas A., Choolani M., Mattar C., Su L.L. Coronavirus disease 2019 (COVID-19) pandemic and pregnancy. Am. J. Obstet. Gynecol. 2020;222:521–531. doi: 10.1016/j.ajog.2020.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Debost J.P., Larsen J.T., Munk-Olsen T., Mortensen P.B., Meyer U., Petersen L. Joint effects of exposure to prenatal infection and peripubertal psychological trauma in schizophrenia. Schizophr. Bull. 2017;43(1):171–179. doi: 10.1093/schbul/sbw083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deverman B.E., Patterson P.H. Cytokines and CNS development. Neuron. 2009;64:61–78. doi: 10.1016/j.neuron.2009.09.002. [DOI] [PubMed] [Google Scholar]
- Dhindsa R.S., Goldstein D.B. Schizophrenia: from genetics to physiology at last. Nature. 2016;530:162–163. doi: 10.1038/nature16874. [DOI] [PubMed] [Google Scholar]
- Ellman L.M., Deicken R.F., Vinogradov S., Kremen W.S., Poole J.H., Kern D.M., Tsai W.Y., Schaefer C.A., Brown A.S. Structural brain alterations in schizophrenia following fetal exposure to the inflammatory cytokine interleukin-8. Schizophr. Res. 2010;121:46–54. doi: 10.1016/j.schres.2010.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellman L.M., Susser E.S. The promise of epidemiologic studies: neuroimmune mechanisms in the etiologies of brain disorders. Neuron. 2009;64:25–27. doi: 10.1016/j.neuron.2009.09.024. [DOI] [PubMed] [Google Scholar]
- Ellman L.M., Yolken R.H., Buka S.L., Torrey E.F., Cannon T.D. Cognitive functioning prior to the onset of psychosis: the role of fetal exposure to serologically determined influenza infection. Biol. Psychiatr. 2009;65:1040–1047. doi: 10.1016/j.biopsych.2008.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Estes M.L., McAllister A.K. Immune mediators in the brain and peripheral tissues in autism spectrum disorder. Nat. Rev. Neurosci. 2015;16:469–486. doi: 10.1038/nrn3978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giovanoli S., Engler H., Engler A., Richetto J., Voget M., Willi R., Winter C., Riva M.A., Mortensen P.B., Feldon J., Schedlowski M., Meyer U. Stress in puberty unmasks latent neuropathological consequences of prenatal immune activation in mice. Science. 2013;339(6123):1095–1099. doi: 10.1126/science.1228261. [DOI] [PubMed] [Google Scholar]
- Giovanoli S., Engler H., Engler A., Richetto J., Feldon J., Riva M.A., Schedlowski M., Meyer U. Preventive effects of minocycline in a neurodevelopmental two-hit model with relevance to schizophrenia. Transl. Psychiatry. 2016;6(4) doi: 10.1038/tp.2016.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein J.M., Cohen J.E., Mareckova K., Holsen L., Whitfield-Gabrieli S., Gilman S.E., Buka S.L., Hornig M. Impact of prenatal maternal cytokine exposure on sex differences in brain circuitry regulating stress in offspring 45 years later. Proc. Natl. Acad. Sci. Unit. States Am. 2021;118 doi: 10.1073/pnas.2014464118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayakawa S., Komine-Aizawa S., Mor G.G. Covid-19 pandemic and pregnancy. J. Obstet. Gynaecol. Res. 2020;46:1958–1966. doi: 10.1111/jog.14384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmes E.A., O'Connor R.C., Perry V.H., Tracey I., Wessely S., Arseneault L., Ballard C., Christensen H., Cohen Silver R., Everall I., Ford T., John A., Kabir T., King K., Madan I., Michie S., Przybylski A.K., Shafran R., Sweeney A., Worthman C.M., Yardley L., Cowan K., Cope C., Hotopf M., Bullmore E. Multidisciplinary research priorities for the COVID-19 pandemic: a call for action for mental health science. The Lancet Psychiatry. 2020;7:547–560. doi: 10.1016/S2215-0366(20)30168-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howland J.G., Cazakoff B.N., Zhang Y. Altered object-in-place recognition memory, prepulse inhibition, and locomotor activity in the offspring of rats exposed to a viral mimetic during pregnancy. Neuroscience. 2012;201:184–198. doi: 10.1016/j.neuroscience.2011.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsiao E.Y., Patterson P.H. Activation of the maternal immune system induces endocrine changes in the placenta via IL-6. Brain Behav. Immun. 2011;25:604–615. doi: 10.1016/j.bbi.2010.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsiao E.Y., McBride S.W., Hsien S., Sharon G., Hyde E.R., McCue T., Codelli J.A., Chow J., Reisman S.E., Petrosino J.F., Patterson P.H., Mazmanian S.K. Microbiota modulate behavioral and physiological abnormalities associated with neurodevelopmental disorders. Cell. 2013;155(7):1451–1463. doi: 10.1016/j.cell.2013.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang H., Xu L., Shao L., Xia R., Yu Z., Ling Z., Yang F., Deng M., Ruan B. Maternal infection during pregnancy and risk of autism spectrum disorders: a systematic review and meta-analysis. Brain Behav. Immun. 2016;58:165–172. doi: 10.1016/j.bbi.2016.06.005. [DOI] [PubMed] [Google Scholar]
- Jones C.A., Watson D.J.G., Fone K.C.F. Animal models of schizophrenia. Br. J. Pharmacol. 2011 doi: 10.1111/j.1476-5381.2011.01386.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kentner A.C., Bilbo S.D., Brown A.S., Hsiao E.Y., McAllister A.K., Meyer U., Pearce B.D., Pletnikov Mikhail V., Yolken R.H., Bauman M.D. Maternal immune activation: reporting guidelines to improve the rigor, reproducibility, and transparency of the model. Neuropsychopharmacology. 2019;44:245–258. doi: 10.1038/s41386-018-0185-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knapp M., Mangalore R., Simon J. The global costs of schizophrenia. Schizophr. Bull. 2004;30:279–293. doi: 10.1093/oxfordjournals.schbul.a007078. [DOI] [PubMed] [Google Scholar]
- Kotlar B., Gerson E., Petrillo S., Langer A., Tiemeier H. The impact of the COVID-19 pandemic on maternal and perinatal health: a scoping review. Reprod. Health. 2021 doi: 10.1186/s12978-021-01070-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai M.C., Lombardo M.V., Baron-Cohen S. The Lancet. Elsevier B.V.; 2014. Autism; pp. 896–910. [DOI] [PubMed] [Google Scholar]
- Lee B.K., Magnusson C., Gardner R.M., Blomström Å., Newschaffer C.J., Burstyn I., Karlsson H., Dalman C. Maternal hospitalization with infection during pregnancy and risk of autism spectrum disorders. Brain Behav. Immun. 2015;44:100–105. doi: 10.1016/j.bbi.2014.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y., Dugyala S.R., Ptacek T.S., Gilmore J.H., Frohlich F. Maternal immune activation alters adult behavior, gut microbiome and juvenile brain oscillations in ferrets. eNeuro. 2018;5(5) doi: 10.1523/ENEURO.0313-18.2018. ENEURO.0313-18.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lins B.R., Hurtubise J.L., Roebuck A.J., Marks W.N., Zabder N.K., Scott G.A., Greba Q., Dawicki W., Zhang X., Rudulier C.D., Gordon J.R., Howland J.G. Prospective analysis of the effects of maternal immune activation on rat cytokines during pregnancy and behavior of the male offspring relevant to schizophrenia. eNeuro. 2018;5 doi: 10.1523/ENEURO.0249-18.2018. ENEURO.0249-18.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lins B.R., Marks W.N., Zabder N.K., Greba Q., Howland J.G. Maternal immune activation during pregnancy alters the behavior profile of female offspring of sprague dawley rats. eneuro. 2019;6 doi: 10.1523/ENEURO.0437-18.2019. ENEURO.0437-18.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- López-Díaz Á., Ayesa-Arriola R., Crespo-Facorro B., Ruiz-Veguilla M. COVID-19 infection during pregnancy and risk of neurodevelopmental disorders in offspring: time for collaborative research. Biol. Psychiatr. 2021;89:e29–e30. doi: 10.1016/j.biopsych.2020.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luan W., Hammond L.A., Vuillermot S., Meyer U., Eyles D.W. Maternal vitamin D prevents abnormal dopaminergic development and function in a mouse model of prenatal immune activation. Sci. Rep. 2018;8(1):9741. doi: 10.1038/s41598-018-28090-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mac Giollabhui N., Breen E.C., Murphy S.K., Maxwell S.D., Cohn B.A., Krigbaum N.Y., Cirillo P.M., Perez C., Alloy L.B., Drabick D.A.G., Ellman L.M. Maternal inflammation during pregnancy and offspring psychiatric symptoms in childhood: timing and sex matter. J. Psychiatr. Res. 2019;111:96–103. doi: 10.1016/J.JPSYCHIRES.2019.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marim F., Karadogan D., Eyuboglu T.S., Emiralioglu N., Gurkan C.G., Toreyin Z.N., Akyil F.T., Yuksel A., Arikan H., Serifoglu I., Gursoy T.R., Sandal A., Akgun M. Lessons learned so far from the pandemic: a review on pregnants and neonates with COVID-19. Eurasian J. Med. 2020;52:202–210. doi: 10.5152/eurasianjmed.2020.20118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattei D., Pietrobelli A. Micronutrients and brain development. Curr Nutr Rep. 2019;8(2):99–107. doi: 10.1007/s13668-019-0268-z. [DOI] [PubMed] [Google Scholar]
- McAlonan G.M., Murphy D.G.M., Edwards D.A. Multidisciplinary: research priorities for the COVID-19 pandemic. The Lancet Psychiatry. 2020;7:e35. doi: 10.1016/j.jmii.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer U. Neurodevelopmental resilience and susceptibility to maternal immune activation. Trends Neurosci. 2019;42(11):793–806. doi: 10.1016/j.tins.2019.08.001. [DOI] [PubMed] [Google Scholar]
- Michel M., Schmidt M.J., Mirnics K. Immune system gene dysregulation in autism and schizophrenia. Dev. Neurobiol. 2012;72:1277–1287. doi: 10.1002/dneu.22044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Missault S., Van Den Eynde K., Vanden Berghe W., Fransen E., Weeren A., Timmermans J.P., Kumar-Singh S., Dedeurwaerdere S. The risk for behavioural deficits is determined by the maternal immune response to prenatal immune challenge in a neurodevelopmental model. Brain Behav. Immun. 2014;42:138–146. doi: 10.1016/j.bbi.2014.06.013. [DOI] [PubMed] [Google Scholar]
- Missig G., Robbins J.O., Mokler E.L. Sex-dependent neurobiological features of prenatal immune activation via TLR7. Mol. Psychiatr. 2020;25:2330–2341. doi: 10.1038/s41380-018-0346-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Money K.M., Barke T.L., Serezani A., Gannon M., Garbett K.A., Aronoff D.M., Mirnics K. Gestational diabetes exacerbates maternal immune activation effects in the developing brain. Mol. Psychiatr. 2018;23(9):1920–1928. doi: 10.1038/mp.2017.191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monte A.S., Mello B.S.F., Borella V.C.M., da Silva Araujo T., da Silva F.E.R., Sousa F.C.F., de Oliveira A.C.P., Gama C.S., Seeman M.V., Vasconcelos S.M.M., Lucena D.F., Macêdo D. Two-hit model of schizophrenia induced by neonatal immune activation and peripubertal stress in rats: study of sex differences and brain oxidative alterations. Behav. Brain Res. 2017;331:30–37. doi: 10.1016/j.bbr.2017.04.057. [DOI] [PubMed] [Google Scholar]
- Moore H., Jentsch J.D., Ghajarnia M., Geyer M., Grace A. A neurobehavioral systems analysis of adult rats exposed to methylazoxymethanol acetate on E17: implications for the neuropathology of schizophrenia. Biol Psychiatr. 2006 doi: 10.1016/j.biopsych.2006.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morais L.H., Felice D., Golubeva A.V., Moloney G., Dinan T.G., Cryan J.F. Strain differences in the susceptibility to the gut-brain axis and neurobehavioural alterations induced by maternal immune activation in mice. Behav. Pharmacol. 2018;29(2 and 3-Spec Issue):181–198. doi: 10.1097/FBP.0000000000000374. [DOI] [PubMed] [Google Scholar]
- Moreno-Eutimio M.A., López-Macías C., Pastelin-Palacios R. Bioinformatic analysis and identification of single-stranded RNA sequences recognized by TLR7/8 in the SARS-CoV-2, SARS-CoV, and MERS-CoV genomes. Microb. Infect. 2020;22(4–5):226–229. doi: 10.1016/j.micinf.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moriyama C., Galic M.A., Mychasiuk R., Pittman Q.J., Perrot T.S., Currie R.W., Esser M.J. Prenatal transport stress, postnatal maternal behavior, and offspring sex differentially affect seizure susceptibility in young rats. Epilepsy Behav. 2013;29(1):19–27. doi: 10.1016/j.yebeh.2013.06.017. [DOI] [PubMed] [Google Scholar]
- Moretti S., Franchi S., Castelli M., Amodeo G., Somaini L., Panerai A., Sacerdote P. Exposure of adolescent mice to delta-9-tetrahydrocannabinol induces long-lasting modulation of pro- and anti-inflammatory cytokines in hypothalamus and Hippocampus similar to that observed for peripheral macrophages. J. Neuroimmune Pharmacol. 2015;10(2):371–379. doi: 10.1007/s11481-015-9592-2. 2015 Jun. [DOI] [PubMed] [Google Scholar]
- Mueller F.S., Scarborough J., Schalbetter S.M., Richetto J., Kim E., Couch A., Yee Y., Lerch J.P., Vernon A.C., Weber-Stadlbauer U., Meyer U. Behavioral, neuroanatomical, and molecular correlates of resilience and susceptibility to maternal immune activation. Mol. Psychiatr. 2021;26:396–410. doi: 10.1038/s41380-020-00952-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mueller F.S., Richetto J., Hayes L.N., Zambon A., Pollak D.D., Sawa A., Meyer U., Weber-Stadlbauer U. Influence of poly(I:C) variability on thermoregulation, immune responses and pregnancy outcomes in mouse models of maternal immune activation. Brain Behav. Immun. 2019;80:406–418. doi: 10.1016/j.bbi.2019.04.019. [DOI] [PubMed] [Google Scholar]
- Myers S.M., Challman T.D., Bernier R., Bourgeron T., Chung W.K., Constantino J.N., Eichler E.E., Jacquemont S., Miller D.T., Mitchell K.J., Zoghbi H.Y., Martin C.L., Ledbetter D.H. Insufficient evidence for “autism-specific” genes. Am. J. Hum. Genet. 2020;106:587–595. doi: 10.1016/j.ajhg.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliver S.E., Gargano J.W., Marin M., Wallace M., Curran K.G., Chamberland M., McClung N., Campos-Outcalt D., Morgan R.L., Mbaeyi S., Romero J.R., Talbot H.K., Lee G.M., Bell B.P., Dooling K. The advisory committee on immunization practices' interim recommendation for use of pfizer-BioNTech COVID-19 vaccine — United States, december 2020. MMWR Morb. Mortal. Wkly. Rep. 2020;69:1922–1924. doi: 10.15585/mmwr.mm6950e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ornoy A., Weinstein-Fudim L., Ergaz Z. Prenatal factors associated with autism spectrum disorder (ASD) Reprod. Toxicol. 2015;92:595–601. doi: 10.1016/j.reprotox.2015.05.007. [DOI] [PubMed] [Google Scholar]
- Patterson P.H. Maternal infection and immune involvement in autism. Trends Mol. Med. 2011;17:389–394. doi: 10.1016/j.molmed.2011.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pendyala G., Chou S., Jung Y., Coiro P., Spartz E., Padmashri R., Li M., Dunaevsky A. Maternal immune activation causes behavioral impairments and altered cerebellar cytokine and synaptic protein expression. Neuropsychopharmacology. 2017;42:1435–1446. doi: 10.1038/npp.2017.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piontkewitz Y., Arad M., Weiner I. Abnormal trajectories of neurodevelopment and behavior following in utero insult in the rat. Biol. Psychiatr. 2011;70:842–851. doi: 10.1016/j.biopsych.2011.06.007. [DOI] [PubMed] [Google Scholar]
- Preis H., Mahaffey B., Heiselman C., Lobel M. Vulnerability and resilience to pandemic-related stress among U.S. women pregnant at the start of the COVID-19 pandemic. Soc. Sci. Med. 2020 doi: 10.1016/j.socscimed.2020.113348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ravaccia D., Ghafourian T. Critical role of the maternal immune system in the pathogenesis of autism spectrum disorder. Biomedicines. 2020;8:1–21. doi: 10.3390/biomedicines8120557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reisinger S., Khan D., Kong E., Berger A., Pollak A., Pollak D.D. The Poly(I:C)-induced maternal immune activation model in preclinical neuropsychiatric drug discovery. Pharmacol. Ther. 2015;149:213–226. doi: 10.1016/j.pharmthera.2015.01.001. [DOI] [PubMed] [Google Scholar]
- Ricardo Martins-Filho P., Moura Tanajura D., Santos H.P., Jr., Santana Santos V. Correspondence COVID-19 during pregnancy: potential risk for neurodevelopmental disorders in neonates? Eur. J. Obstet. Gynecol. 2020;250:255–256. doi: 10.1101/2020.04.07.20053744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ripke S., Neale B.M., Corvin A. Biological insights from 108 schizophrenia-associated genetic loci. Nature. 2014;511:421–427. doi: 10.1038/nature13595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scola G., Duong A. Prenatal maternal immune activation and brain development with relevance to psychiatric disorders. Neuroscience. 2017;346:403–408. doi: 10.1016/J.NEUROSCIENCE.2017.01.033. [DOI] [PubMed] [Google Scholar]
- Sekar A., Bialas A.R., de Rivera H., Davis A., Hammond T.R., Kamitaki N., Tooley K., Presumey J., Baum M., Van Doren V., Genovese G., Rose S.A., Handsaker R.E., Schizophrenia Working Group of the Psychiatric Genomics Consortium. Daly M.J., Carroll M.C., Stevens B., McCarroll S.A. Schizophrenia risk from complex variation of complement component 4. Nature. 2016;530:177–183. doi: 10.1038/nature16549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin Yim Y., Park A., Berrios J., Lafourcade M., Pascual L.M., Soares N., Yeon Kim J., Kim S., Kim H., Waisman A., Littman D.R., Wickersham I.R., Harnett M.T., Huh J.R., Choi G.B. Reversing behavioural abnormalities in mice exposed to maternal inflammation. Nature. 2017;549:482–487. doi: 10.1038/nature23909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shuid A.N., Jayusman P.A., Shuid N., Ismail J., Kamal Nor N., Mohamed I.N. Association between viral infections and risk of autistic disorder: an overview. Int. J. Environ. Res. Publ. Health. 2021;18:2817. doi: 10.3390/ijerph18062817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh S.K. Middle East respiratory syndrome virus pathogenesis. Semin. Respir. Crit. Care Med. 2016;37:572–577. doi: 10.1055/s-0036-1584796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith S.E.P., Li J., Garbett K., Mirnics K., Patterson P.H. Maternal immune activation alters fetal brain development through interleukin-6. J. Neurosci. 2007;27:10695–10702. doi: 10.1523/JNEUROSCI.2178-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soumiya H., Fukumitsu H., Furukawa S. Prenatal immune challenge compromises the normal course of neurogenesis during development of the mouse cerebral cortex. J. Neurosci. Res. 2011;89:1575–1585. doi: 10.1002/jnr.22704. [DOI] [PubMed] [Google Scholar]
- Spann M.N., Monk C., Scheinost D., Peterson B.S. Maternal immune activation during the third trimester is associated with neonatal functional connectivity of the salience network and fetal to toddler behavior. J. Neurosci. 2018;38:2877–2886. doi: 10.1523/JNEUROSCI.2272-17.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tandon R., Gaebel W., Barch D.M., Bustillo J., Gur R.E., Heckers S., Malaspina D., Owen M.J., Schultz S., Tsuang M., Van Os J., Carpenter W. Definition and description of schizophrenia in the DSM-5. Schizophr. Res. 2013;150:3–10. doi: 10.1016/j.schres.2013.05.028. [DOI] [PubMed] [Google Scholar]
- Van den Eynde K., Missault S., Fransen E., Raeymaekers L., Willems R., Drinkenburg W., Timmermans J.-P., Kumar-Singh S., Dedeurwaerdere S. Hypolocomotive behaviour associated with increased microglia in a prenatal immune activation model with relevance to schizophrenia. Behav. Brain Res. 2014;258:179–186. doi: 10.1016/j.bbr.2013.10.005. [DOI] [PubMed] [Google Scholar]
- Wainer Z., Carcel C., Hickey M., Schiebinger L., Schmiede A., McKenzie B., Jenkins C., Webster J., Woodward M., Hehir A., Solomon B., de Costa C., Lukaszyk C., Colville D.J., Dempsey E., Wright G.M., Mishra G.D., Fisher J.R.W., Kulkarni J., Mitchell J.A., Hutchison K., Thompson K., Jorm L., Chappell L., van der Meulen M., Henry A., DiGiacomo M., Huxley R., Ivers R., Peters S., Rogers W.A., Wang X., Norton R. Sex and gender in health research: updating policy to reflect evidence. Med. J. Aust. 2020;212:57–62. doi: 10.5694/mja2.50426. e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C., Geng H., Liu W., Zhang G. Prenatal, perinatal, and postnatal factors associated with autism: a meta-analysis. Medicine (Baltim.) 2017;96 doi: 10.1097/MD.0000000000006696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watson C.J., Thomas R.H., Solomon T., Michael B.D., Nicholson T.R., Pollak T.A. COVID-19 and psychosis risk: real or delusional concern? Neurosci. Lett. 2021;741:135491. doi: 10.1016/j.neulet.2020.135491. [DOI] [PubMed] [Google Scholar]
- Will T.R., Proaño S.B., Thomas A.M., Kunz L.M., Thompson K.C., Ginnari L.A., Jones C.H., Lucas S.-C., Reavis E.M., Dorris D.M., Meitzen J. Problems and progress regarding sex bias and omission in neuroscience research. eneuro. 2017;4 doi: 10.1523/ENEURO.0278-17.2017. ENEURO.0278-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winship I.R., Dursun S.M., Baker G.B., Balista P.A., Kandratavicius L., Maia-de-Oliveira J.P., Hallak J., Howland J.G. An overview of animal models related to schizophrenia. Can. J. Psychiatr. 2019;64:5–17. doi: 10.1177/0706743718773728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolff A.R., Bilkey D.K. The maternal immune activation (MIA) model of schizophrenia produces pre-pulse inhibition (PPI) deficits in both juvenile and adult rats but these effects are not associated with maternal weight loss. Behav. Brain Res. 2010;213:323–327. doi: 10.1016/j.bbr.2010.05.008. [DOI] [PubMed] [Google Scholar]
- Wolff A.R., Bilkey D.K. Immune activation during mid-gestation disrupts sensorimotor gating in rat offspring. Behav. Brain Res. 2008;190:156–159. doi: 10.1016/J.BBR.2008.02.021. [DOI] [PubMed] [Google Scholar]
- Wu W.L., Hsiao E.Y., Yan Z., Mazmanian S.K., Patterson P.H. The placental interleukin-6 signaling controls fetal brain development and behavior. Brain Behav. Immun. 2017;62:11–23. doi: 10.1016/j.bbi.2016.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young J.W., Powell S.B., Risbrough V., Marston H.M., Geyer M.A. Using the MATRICS to guide development of a preclinical cognitive test battery for research in schizophrenia. Pharmacol. Ther. 2009;122:150–202. doi: 10.1016/j.pharmthera.2009.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamberletti E., Gabaglio M., Prini P., Rubino T., Parolaro D. Cortical neuroinflammation contributes to long-term cognitive dysfunctions following adolescent delta-9-tetrahydrocannabinol treatment in female rats. Eur. Neuropsychopharmacol. 2015;25(12):2404–2415. doi: 10.1016/j.euroneuro.2015.09.021. [DOI] [PubMed] [Google Scholar]
- Zerbo O., Qian Y., Yoshida C., Grether J.K., Van de Water J., Croen L.A. Maternal infection during pregnancy and autism spectrum disorders. J. Autism Dev. Disord. 2015;45:4015–4025. doi: 10.1007/s10803-013-2016-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y., Cazakoff B.N., Thai C.A., Howland J.G. Prenatal exposure to a viral mimetic alters behavioural flexibility in male, but not female, rats. Neuropharmacology. 2012;62:1299–1307. doi: 10.1016/j.neuropharm.2011.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmer A., Youngblood A., Adnane A., Miller B.J., Goldsmith D.R. Prenatal exposure to viral infection and neuropsychiatric disorders in offspring: a review of the literature and recommendations for the COVID-19 pandemic. Brain Behav. Immun. 2021;91:756–770. doi: 10.1016/j.bbi.2020.10.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuckerman L., Weiner I. Maternal immune activation leads to behavioral and pharmacological changes in the adult offspring. J. Psychiatr. Res. 2005;39:311–323. doi: 10.1016/j.jpsychires.2004.08.008. [DOI] [PubMed] [Google Scholar]