Abstract
Low-carbohydrate ketogenic diet (LCKD), originally used as a treatment for childhood epilepsy is currently gaining acceptance as a nutritional therapy for obesity and type 2 diabetes. In addition, this diet has a positive effect on body weight, blood glucose level, glycosylated hemoglobin, plasma lipid profile, and neurological disorders. This review focuses on the therapeutic effectiveness, negative effects, and the rationale of using LCKD for the treatment of type 2 diabetes. It is shown that LCKD contributes to the reduction in the intake of insulin and oral antidiabetic drugs in patients with type 2 diabetes. Furthermore, the data presented in this review reveal the efficacy and cost-effectiveness of LCKD in the management of type 2 diabetes.
Keywords: Low-carbohydrate ketogenic diet, Type 2 diabetes, Obesity, HbA1c
Significance of the Study
This review unravels the therapeutic effects of low-carbohydrate ketogenic diet in diabetes.
Introducing low carbohydrate diet therapy in diabetic management may lead to better quality of life and in reducing the health care costs of diabetes.
Efficacy of carbohydrate restriction can lead to significant reduction in the units of insulin required, cessation, or reduction of the dose of antidiabetic agents.
Introduction
According to Centers for Disease Control and Prevention (CDC), the number of people with diabetes has almost tripled in the last couple of decades [1]. Approximately about 425 million people worldwide have diabetes now, which is expected to reach to 629 million by 2045 [2]. A similar trend in diabetes has been noticed in Kuwait and other Arab countries [3]. In addition to the diagnosed cases, several millions live with undiagnosed diabetes and many are at the risk of developing diabetes [3]. The direct costs of diagnosed diabetes (about 30.3 million people) and the reduced productivity were $327 billion in the United States alone in 2017 [1]. In addition, the costs for managing the complications of diabetes add to this economic burden [4].
Complications of Diabetes
A majority of the type 2 diabetic patients develop hypertension. The exact mechanism of this link is not understood; however, it is suggested that to minimize the level of insulin, our body attempts to hold more fluid in the blood vessels, which ultimately leads to hypertension and weakening of the cardiovascular system. Depending on the duration and the severity of hyperglycemia, diabetic patients develop microvascular complications such as retinopathy, nephropathy, and neuropathy. Diabetic retinopathy occurs due to the damage of the retinal blood vessels and nerves. It is the most common cause of vision problems and blindness among adults [5] and it is estimated that more than 60% of diabetic patients develop retinopathy [5]. Nephropathy is another complication that results from prolonged diabetes. The signs of this disease are microalbuminuria and increase in glomerular filtration rate [6]. Diabetic neuropathy results from the nerve damage that occurs in diabetic patients [7]. Large nerve fiber neuropathy causes loss of vibration sensation, loss of proprioception, loss of reflexes, and reduced nerve conduction velocity, and small nerve fibers injury results in burning pain, reduced pain, and temperature sensation, leading to foot ulceration [7]. Patients with diabetic neuropathy may lose bladder control and the ability to walk, and male patients suffer from erectile dysfunction or impotence. Autonomic neuropathy usually causes heart rate abnormalities, abnormal sweating, and gastroparesis [8].
Diabetic patients show abnormalities in lipid profile. During diabetes, there is an increase in the level of both low-density lipoproteins (LDLs) and very low-density lipoproteins (VLDLs) and a decrease in level of high-density lipoproteins (HDLs). Alterations in lipid profile and high blood pressure increase the risk of heart attack and stroke [9, 10]. Abnormal lipid profile and hyperglycemia causes glycation, oxidation, tyrozylation, and methylation that result in chronic inflammation.
The major factor that leads to the development of diabetic complications is the glycation of proteins. Glycated hemoglobin in the form of HbA1c is an important indicator of glycemic control, and dietary carbohydrate restriction induces substantial improvement of HbA1c [11, 12]. Glycemic control together with reasonable weight control remains the primary requirement for effective diabetic management [11, 13]. In this regard, dietary carbohydrate restriction using low-carbohydrate ketogenic diet (LCKD) is found to be very effective in reducing body weight and type 2 diabetes. Therefore, the ultimate purpose of this study is to review the current knowledge on the benefits and possible risks of using LCKDs in the treatment of type 2 diabetes.
Low-Carbohydrate Ketogenic Diets
William Banting in the fourth edition of his paper entitled “Letter on Corpulence, Addressed to the Public,” published in May 1869, reported that a low-carbohydrate diet is very effective in reducing body weight [14]. He apparently lost 22.7 kg in mass and 33 cm in girth in 6 months after following this diet therapy. This is the first report on the use of low-carbohydrate diet for the treatment of obesity. Since 1955, scientists were experimenting on the concept that fat can be eaten ad libitum and still induce weight loss in obese subjects. Prior to its use for obesity, LCKD have been used in the treatment of diabetes and pediatric epilepsy [15]. Recent studies indicate that LCKD is also beneficial in the treatment of attention deficit/hyperactivity disorders [16], infantile spasms, bipolar illness [17, 18], Alzheimer's disease, and brain tumor [19].
A high-fat diet changes the body's metabolism to a new direction [20, 21]. In the liver, fatty acids undergo partial oxidation to form ketone bodies. As ketone bodies are formed in excess than the body's ability to metabolize, they are accumulated in the body resulting in the condition called ketosis. The high-fat diet that causes ketosis is called a ketogenic diet [20, 21]. Although there are several studies on the effects of LCKD in obesity, diabetes, and epilepsy, the amount of low-carbohydrate intake in each study varies. Therefore, in order to compare the effectiveness of ketogenic diets in various studies, Feinman et al. [22] have recently suggested definitions of low-carbohydrate diets in different forms, which are given in Table 1.
Table 1.
Amount of carbohydrate (g/day) in different types of diet [22]
| Type of diet | Amount of carbohydrate, g/day |
|---|---|
| VLCKD | 20–50 |
| Low carbohydrate | <130 |
| Moderate carbohydrate | 130–230 |
| High carbohydrate | >230 |
| VLCKD, very low-carbohydrate ketogenic diet. | |
In addition to the Atkins diet [23, 24], there are other similar diets that are in common practice. They are the Kwasniewski's, Zone [25], and the South Beach Diet [26]. The carbohydrate content of these diets is given in Table 2. Based on the influence of ketogenic diet in the various therapeutic uses reported from our laboratory, the subjects were recommended a low-carbohydrate, high protein and fat diet containing 20 g of carbohydrates, polyunsaturated and monounsaturated fats (5 tablespoons olive oil), and micronutrients (vitamins and minerals) in the form of 1 capsule/day. An additional 20 g of carbohydrates was given 12 weeks later [27]. The subjects were fully restricted from consuming any kind of alcoholic beverages and certain type of foods as shown in Table 3.
Table 2.
| Type of diet | Content, % of nutrients |
|---|---|
| Atkins diet* | 3–16% − carbohydrates |
| 28–34% − protein | |
| 55–65% − fats | |
| Kwasniewski's diet | 9.2% − carbohydrates |
| 14% − protein | |
| 76.8% − fats | |
| Zone diet | 40% − carbohydrates |
| 30% − protein | |
| 30% − fats | |
Percentage range depends on individual status.
Table 3.
Recommended and restricted food in ketogenic diet (adapted from Dashti et al. [27])
| Recommended food in ketogenic diet | |
| Proteins | Fish: tuna, sardine, prawns, shrimps, lobster |
| Meat: kababs, sausages, minced | |
| Poultry: chicken, eggs | |
| Cheese: full-fat cheese |
|
| Vegetables/fruits | Spinach, watercress, eggplant, parsley, mulberry, coriander, mint, artichoke, okra, cabbage, mushroom, avocado, leek, carrot, radish, celery, cauliflower, green pepper, lettuce, cucumber, tomato, 10–15 olives/day, lemon, strawberry-6/day, avocado, berries-10/day |
| Oil | Olive oil (5 tablespoons) added to the salad, flax seed oil |
| Restricted food in ketogenic diet | |
| Carbohydrates | Flour, potato, macaroni, spaghetti, noodles, bread, rice, sugar, sweets, honey, cakes |
| Fruits/drinks | All fruit juices, all soft drinks |
Is Ketosis Safe?
One of the major questions faced by the scientific community is whether ketosis is safe. In this regard, some of the interesting natural facts concerning ketogenic diets and ketosis are as follows: mother's milk that has a high fat content is the best natural food formula for human development and growth. All of us approach ketosis during development and during the diurnal cycle. During starvation, the body's metabolism is switched to ketosis, which acts as a regulator of the body's calorie intake by suppressing hunger. Birds use fat which is the primary metabolic fuel for migration, and most importantly, early man's prehistoric diets consisted of LCKDs [28].
What Are Ketone Bodies?
Ketone bodies are synthesized in the mitochondria of liver cells by incomplete oxidation of free fatty acids. Ketone bodies are used as an energy source in the body including the brain. During normal dietary conditions, small amount of ketone bodies that are too small to cause any metabolic effects are produced in the body [29]. As mentioned earlier, accumulation of excess of ketone bodies in the body is ketosis. There are 3 types of ketone bodies. They are acetoacetate (AcAc), β-hydroxybutyrate (BHB), and acetone. In the formation of ketone bodies, two molecules of acetyl CoA are condensed to form a molecule of acetoacetyl CoA. Acetoacetyl CoA combines with another molecule of acetyl CoA in the presence of the hepatic enzyme, HMG-CoA synthase, to form 3-hydroxy-3-methylglutaryl CoA (HMG-CoA). HMG-CoA is cleaved into acetyl CoA and AcAc by another enzyme, HMG-CoA lyase. AcAc is reduced to BHB by the action of BHB dehydrogenase or undergoes spontaneous decarboxylation to acetone which is excreted in the breath and urine [29].
Beneficial Effect of Ketogenic Diet in Type 2 Diabetes Patients
In this review, we report on the beneficial and side effects of ketogenic diet based on the studies that were conducted during the last 20 years in our laboratory and the studies of other investigators that substantiate our view. The first paper from our laboratory was published in 2003, where we studied the effects of ketogenic diet in eliminating or preventing the risk factors of heart disease in obese patients [30]. In further studies we addressed the long-term effects of ketogenic diet in reducing body weight in obese subjects [31] and in obese subjects with hypercholesteremia [27] or type 2 diabetes [32, 33]. Also, we focused on the effect of ketogenic diet in animal models of cardiac tolerance to global ischemia [34] and diabetes [35, 36]. Although we studied the diverse effects of ketogenic diets in humans and animal models, based on the focus of this review, only those studies that are directly involved with the effects of diabetes in human subjects and animal models [32, 33, 35, 36] are discussed here.
In a study on the effect of ketogenic diet in obese diabetic subjects [32], it has been convincingly shown that long-term administration LCKD has significant beneficial effects in obese diabetic subjects. In this study, 64 healthy obese subjects were divided into two groups. Group I consisting of 31 subjects with a BMI >30, having a blood glucose level >6.1 mmol/L, and 33 subjects with a normal blood glucose level were included in this study. The body weight, blood glucose level, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, urea, and Cr were determined before and at 8, 16, 24, 48, and 56 weeks after the administration of the ketogenic diet. All 64 subjects followed a ketogenic diet consisting of <20 g of carbohydrates and 80–100 g of proteins. Twelve weeks later an additional 20 g of carbohydrates was added to the diet. The diet was supplemented with micronutrients and 5 tablespoons of olive oil. The participants continued with their routine exercise of at least a daily walk of 45 min. Thus, this study consists of the routine exercise pattern together with this diet.
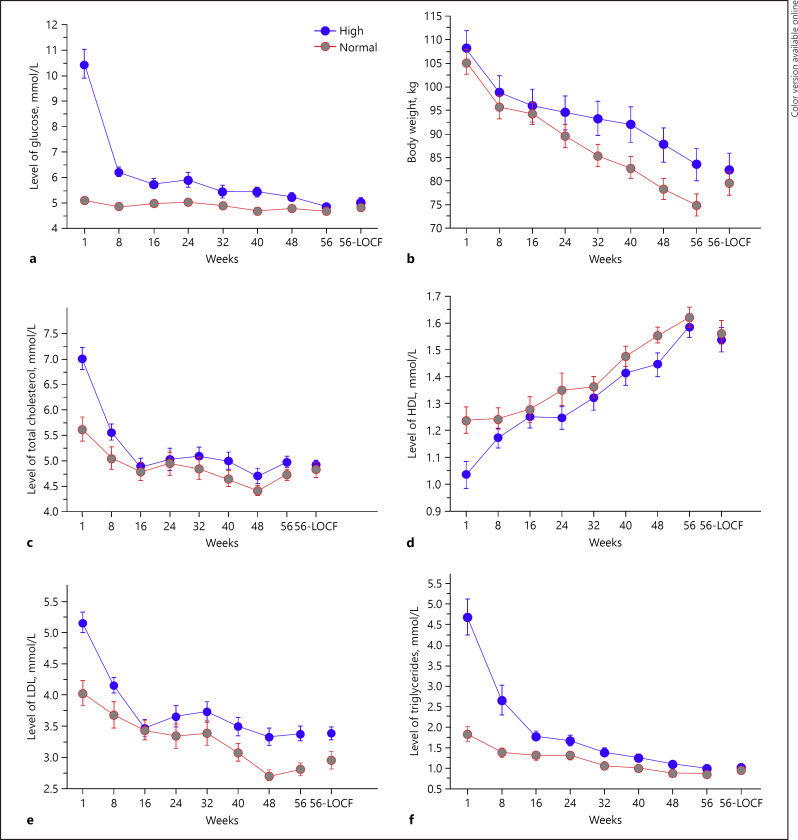
The results of this study showed that the blood glucose, body weight, total cholesterol, LDL, and triglycerides of both the groups decreased significantly from the start until the 56th week (Fig. 1; Table 4). Following the administration of LCKD, in patients with high glucose level (Group 1), there was a 24.4 ± 6.7% and 50.9 ± 12.5% reduction in body weight and glucose level, respectively, after 56 weeks. Patients belonging to the normal glucose level also showed a reduction in the body weight and glucose level, the percentage reduction in body weight and glucose level after 56 weeks was 27.2 ± 6.0 and 7.4 ± 11.9, respectively. Changes in glucose level in both the groups were statistically significant. HDL cholesterol increased significantly (Fig. 1; Table 4). HDL is a measure of good cholesterol and LDL is a measure of bad cholesterol whereas the total cholesterol is the sum of HDL, LDL, and the VLDL. As mentioned above, 5 tablespoons of olive oil was also included in the diet. Since olive oil is rich in monounsaturated fatty acids and antioxidant substances, the beneficial effects of olive oil may be attributed to these properties. Since the participants were involved in exercise prior to entering the diet program and continued the routine exercise, the observed beneficial effects were not related to exercise.
Fig. 1.
Changes in the level of blood glucose (a), body weight (b), total cholesterol (c), level of HDL (d), LDL (e), and triglycerides (f) after the administration of ketogenic diet for 56 weeks [27]. HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Table 4.
Statistical significance between week 1 and week 56 observation of various parameters studied in diabetic subjects (n = 31 [27])
| Week 1 | Week 56 | p value | |
|---|---|---|---|
| Weight, kg | 108.1+21.2 | 83.5+18.0 | <0.0001 |
| Glucose, mmol/L | 10.5+3.0 | 4.9+0.6 | <0.0001 |
| Total cholesterol, mmol/L | 6.8+1.1 | 4.9+0.5 | <0.0001 |
| Triglycerides, mmol/L | 4.7+2.5 | 1.0+0.2 | <0.0001 |
| LDL, mmol/L | 5.2+0.9 | 3.4+0.6 | <0.0001 |
| HDL, mmol/L | 1.0+0.3 | 1.6+0.2 | <0.0001 |
| Urea, µmol/L | 5.8+0.9 | 5.0+1.1 | <0.0111 |
LDL, low-density lipoprotein; HDL, high-density lipoprotein.
In another study [33] concerning the effects of ketogenic diet in comparison with low-calorie diet in type 2 diabetes it has been shown that although both LCD and LCKD had beneficial effects on type 2 diabetes, the ketogenic diet has more advantages over the conventional LCD in obese diabetic subjects. Three hundred and sixty-three overweight and obese subjects (86 men and 277 women) were enrolled in this 24-week trial. Among the men, 28 were diabetic and 58 were nondiabetic; among the women, 74 were diabetic and 203 were nondiabetic. Altogether, 102 participants had type 2 diabetes and 261 were nondiabetic. The inclusion criteria of diabetic subjects in this study were as follows. The age of the patient should be at least 18 years, the BMI should be >25 kg/m2 and the fasting serum glucose level should be >125 mg/dL (>6.9 mmol/L). Patients with renal insufficiency, liver disease, or unstable cardiovascular disease were excluded from the study. The participants were asked to select either a low-calorie diet or LCKD. Initially, participants in the LCKD group were given about 20 g/day of carbohydrates.
Written instructions concerning the importance and how to complete the food records were given to all participants before the beginning of the study. All the paricipants realized the seriousness of this and were very cooperative in completing the take-home food record as per the directions. All the participants submitted the completed food records at the beginning of the study and at weeks 4, 8, 12, 16, 20, and 24. Similar to the previous study mentioned above [33], all the parameters such as blood glucose level, body weight, total cholesterol, LDL, HDL, and triglycerides were determined before and at 4, 8, 12, 16, 20, and 24 weeks after the administration of the LCD or LCKD. In addition, changes in hemoglobin and glycosylated hemoglobin at the time points mentioned above were measured. Hypoglycemic episodes and symptomatic side effects in participants in both the diet groups were assessed by direct interview of the participant on a biweekly basis and necessary medication adjustment were done.
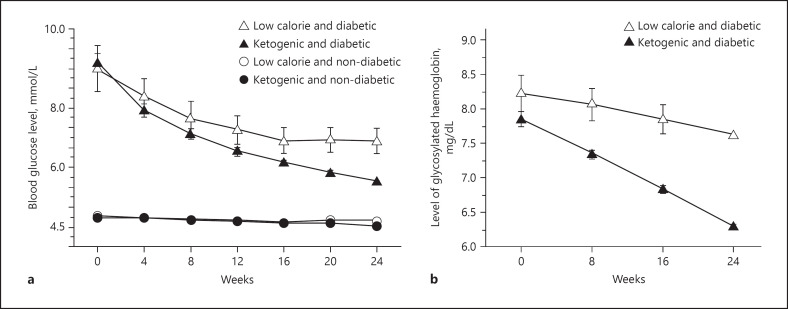
The results of this study showed that although the blood glucose level decreased in both the groups, the effectiveness of LCKD was more beneficial than the LCD group (Fig. 2). Furthermore, with regard to the blood glucose levels, the effectiveness of the LCKD was much greater (p < 0.0001) in the diabetic LCKD group than in the LCD group as compared to their initial and final measurements. A similar pattern was observed in the HbA1c levels (Fig. 2). As compared with the initial (week 1) and final (week 24), the effectiveness of the LCKD was much greater in normalizing the HbA1c level in the diabetic LCKD group than in the LCD group. There was a significant difference (p < 0.0001) in the body weight of both diabetic and nondiabetic participants in both the low calorie and ketogenic diet program. Similar to that observed in the above-mentioned parameters, there was a decrease in lipid levels in both diabetic and nondiabetic participants of the LCKD and LCD groups. The LCKD group, however, showed a significant decrease (p < 0.0001) in triglyceride, total cholesterol, and LDL levels and a significant increase in the HDL level (p < 0.0001) in the LCKD group as compared to the LCD group. In general, the LCD and LCKD had beneficial effects on all the parameters examined. Interestingly, these changes were pronounced in subjects who were on the LCKD as compared with those on the LCD.
Fig. 2.
Changes in blood glucose (a) and glycosylated hemoglobin (b) level in diabetic and nondiabetic subjects after the administration of a low-calorie diet or a LCKD for 24 weeks. Black circles, ketogenic and nondiabetic; black triangles, ketogenic and diabetic; white circles, low calorie and nondiabetic; white triangles, low calorie and diabetic[33]. LCKD, low-carbohydrate ketogenic diet.
In addition to human subjects, we have carried out several studies on the effect of ketogenic diet on animal models. In one of the studies in experimental rats, it has been shown that LCKD enhances cardiac tolerance to global ischemia [34]. In other studies in diabetic rat models, it is found that LCKD has protective and therapeutic effects [35, 36]. Since the focus of this review is on the role of LCKD in diabetes, studies concerning its effect on cardiac protection in animal models will not be discussed here. Concerning the studies on the beneficial effect of LCKD in diabetes, two sets of experiments were conducted. In one set of the experiments, the animals were pre-fed with LCKD for a period of 8 weeks and then diabetes was induced to study its protective effect. These studies are referred to as pre-fed or protective experiments. In the second set of experiments, diabetes was induced using streptozotocin (STZ) in rats and afterward they were fed with LCKD to understand its therapeutic role (post-fed or therapeutic experiments). Sixty-three animals were used in each pre-fed or post-fed experiment. The animals were divided into normal, high-carbohydrate diet, and LCKD groups based on the type of diet given [35, 36].
In both pre-fed and post-fed experiment, each group was further divided into three subgroups containing 7 rats in each subgroup, such as control, sham, and diabetes rats. In the pre-fed experiments, during the 8th week, the diabetic rats were injected with STZ dissolved in saline to induce diabetes. The control rats were kept without injection whereas rats of the sham group were injected with only saline. In the pre-fed or protective groups, the blood glucose and the body weight were measured on the day of the STZ injection. Thereafter, weekly from the 8th week until the end of the experiment at the 12th week, blood glucose and the body weight were measured [36]. In the post-fed (therapeutic) experiments, blood glucose and body weight were monitored on the day of the STZ injection and thereafter from week 1 until the end of the experiment at the 12th week [35].
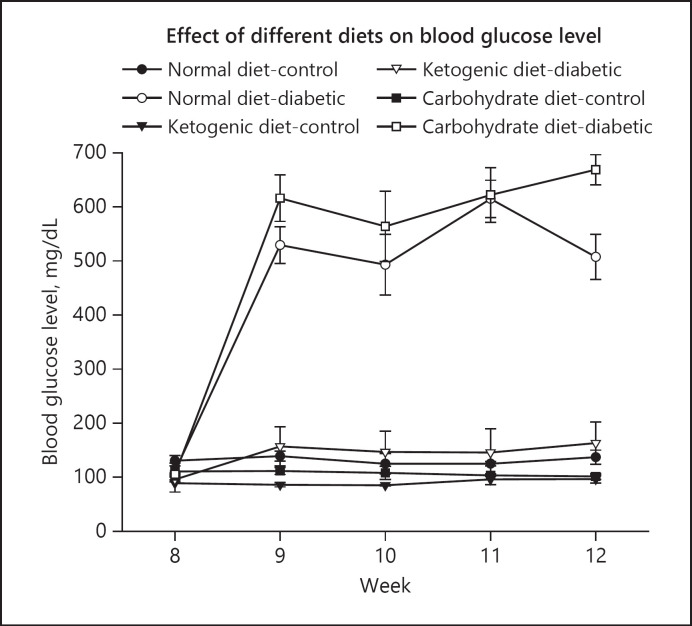
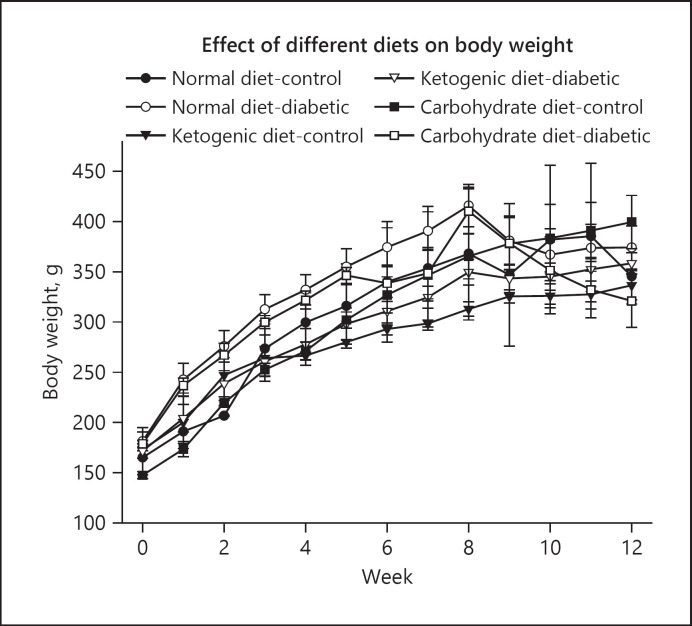
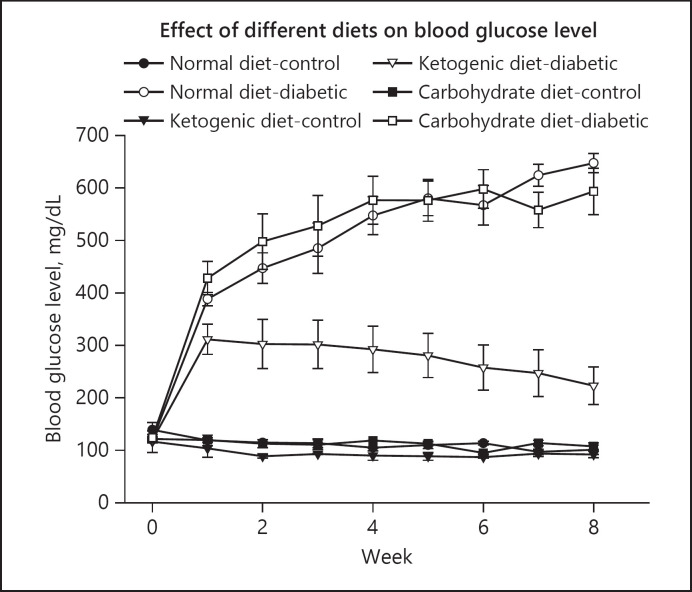
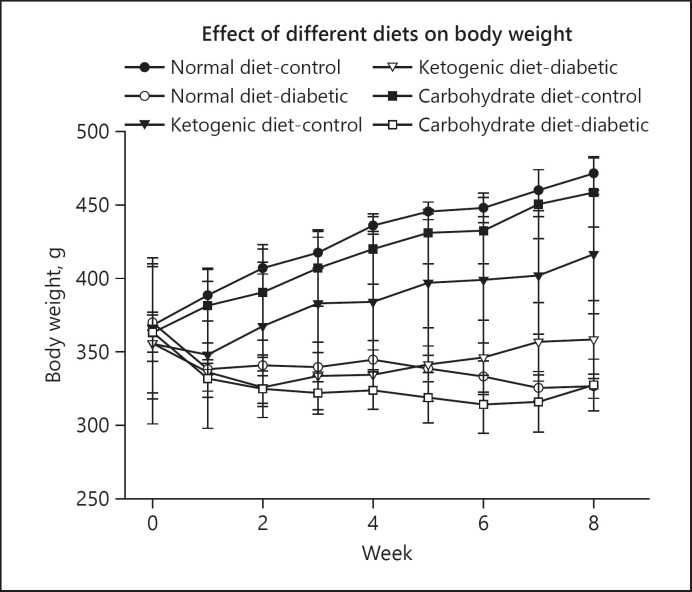
The results of this study showed that in the pre-fed (protective) experiments [36], after the administration of STZ the blood glucose levels in normal diet-diabetic and high carbohydrate-diabetic groups were increased from 105 mg/dL upto 507.3 mg/dL and 668.6 mg/dL, respectively, at the end of the study. However, in the LCKD group, the blood glucose levels remained within the normal range of 100 mg/dL throughout the duration of the experiment (Fig. 3). The body weight increased gradually in all groups (normal diet-control and high carbohydrate-control) during the first 8 weeks. The increase in normal-diet and high-carbohydrate diet groups were significantly higher (Fig. 4) as compared to LCKD groups. After 8 weeks following the administration of STZ, there was a drop in body weight in both normal-diet and high-carbohydrate diet groups, while the body weight continued to increase constantly in the LCKD group [36].
Fig. 3.
Effect of normal diet, high-carbohydrate diet, and LCKD on blood glucose level (mg/dL) in control and diabetic rats in the pre-fed experiments. The values are mean ± SEM (n = 42; [36]). LCKD, low-carbohydate ketogenic diet.
Fig. 4.
Effect of normal diet, high-carbohydrate diet, and LCKD on body weight in control and diabetic rats in the pre-fed (protective) experiment. The values are mean ± SEM (n = 42; [36]). LCKD, low-carbohydrate ketogenic diet.
In the post-fed group, after the administration of STZ, the blood glucose level significantly increased in all diabetic groups compared to control [35]. The increase in the blood glucose level of the LCKD group was significantly lower (p < 0.005 and p < 0.01) than the other diabetic groups (Fig. 5). Also, starting from week 6 the significant difference in the blood glucose level between control and diabetic groups of LCKD disappeared which may indicate that blood glucose level is getting close to normal <200 mg/dL toward the end of the experiment (Fig. 5). With STZ administration there was a significant drop (Fig. 6) in the body weight in both normal-diet and high-carbohydrate diet groups, while there was no significant change in the body weight between control and diabetic groups of the LCKD group [35].
Fig. 5.
Effect of different diets: normal diet, high-carbohydrate diet, and LCKD on blood glucose level (mg/dL) in control and diabetic rats in the post-fed experiments. The values are mean ± SEM (n = 42; [35]). LCKD, low-carbohydrate ketogenic diet.
Fig. 6.
Effect of different diets: normal diet, high-carbohydrate diet, and LCKD on body weight in control and diabetic rats. The values are mean ± SEM (n = 42; [35]). LCKD, low-carbohydrate ketogenic diet.
Selected studies of other investigators [37, 38, 39, 40, 41, 42, 43, 44, 45] on the effect of LCKD in diabetes that support our findings are given in Table 5. As mentioned in our studies and studies of other investigators, antidiabetic medications were decreased or eliminated in most of the participants of the LCKD dietary program [33, 37, 38, 39, 40, 41, 42, 43, 44, 45]. It is advised that, until we fully understand the mechanism of LCKDs' function, medical monitoring for hypoglycemia, electrolyte abnormalities, and dehydration is vital in patients those who are LCKD and taking antidiabetic or diuretic medications. Furthermore, the message from most of the above studies is that in addition to its therapeutic value, LCKD is safe to use for a longer period in obese diabetic subjects without any adverse effect on glucose metabolism or insulin resistance [11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25].
Table 5.
A summary of selected studies showing the beneficial effects of LCKD in type 2 diabetes
| Reference | Design and methods | Results |
|---|---|---|
| Samaha et al. [39] | Comparison of LCD with LFD for 6 months in obese subjects with 39% prevalence of diabetes | LCD almost 3 times more effective in reducing body weight. Fasting blood glucose reduced by 26 and 5% in LCD group and LFD group, respectively. LCD showed a favorable effect on the lipid profile |
| Yancy et al. [40] | Effect of increased level of ketones in very low-calorie ketogenic diet on hepatic glucose output in diabetic patients | Strong inverse correlation between circulating ketones and hepatic glucose output suggesting that higher levels of ketones have favorable effects on glycemic control in diabetics |
| Westman et al. [41] | Comparison of LCKD without calorie restriction with LGID, reduced-calorie diet for 24 h in obese and type 2 diabetic patients | The LCKD group showed greater improvements in hemoglobin A1c, body weight, and HDL cholesterol as compared to the LGID group. Diabetes medications were reduced or eliminated in 95.2% of the participants in the LCKD group versus 62% in the LGID group |
| Nielsen and Joensson [42] | Comparison of a 20% carbohydrate diet with a 55–60% carbohydrate diet on body weight, glycemic control and cardiovascular outcome in obese diabetes patients | There was improvement in HbA1c and body weight in patients who have used low-carbohydrate diet. These patients have suffered very few cardiovascular events while the controls have suffered several cardiovascular events |
| Saslow et al. [43] | Aimed at understanding the effect of MCCRD and LCKD in obese and type 2 diabetic participants | Participants in the LCKD group had greater reductions in HbA1c levels and body weight as compared to MCCRD group. However, reduction in body weight was not significantly different between the two groups |
| Walton et al. [44] | Efficacy of LCKD containing <30 g of carbohydrates daily in obese diabetic females for 90 days | Significant reduction in body weight, HbA1c level, and blood pressure while eliciting favorable changes in blood lipids. LCKD functionally reversed the diabetes |
LCD, low-carbohydrate diet; LFD, low-fat diet; LCKD, low-carbohydrate ketogenic diet; LGID, low-glycemic index; HDL, high-density lipoprotein; MCCRD, moderate-carbohydrate calorie-restricted diet; HbA1c, glycated hemoglobin.
Mechanism of Ketogenic Diet Action in Obesity and Diabetes
There is a tendency for type II diabetes and obesity to occur together [46]. Although the exact molecular mechanism that links obesity to diabetes needs to be elucidated, one of the causes of metabolic syndrome may be the ectopic accumulation of lipids [47]. Various pieces of existing evidence suggest the effectiveness of LCKD in weight loss and in a preferential loss of fat mass [48, 49]. A recent study showed that a high-protein diet also effectively reduces hepatic fat [50]. On the other hand excessive intake of carbohydrate is harmful to individuals with insulin resistance. Therefore, diet choices have a major impact on diabetes and obesity. Several possible mechanisms on the role of LCKD in reducing body weight have been suggested. Each gram of glycogen is stored in 3 g of water; therefore, the initial weight loss could be due to glycogen depletion and water excretion [51]. There is a decrease in metabolic efficiency resulting in greater loss of energy in the form of heat [52] and in the form of ketones in urine, sweat, and feces. Ketones also have a diuretic [53] and appetite suppression effect [54]. High fat content in LCKD delays the digestion providing a sense of fullness [55]. More importantly, utilization of fat as body fuel promotes fat loss and therefore weight loss [40].
The level of HbA1c is considered as a gold standard index for the diagnosis and management of diabetes and indicates the level of oxidative stress [56]. Various studies from our laboratory and other international laboratories have convincingly demonstrated that administration of LCKD decreases the level of HbA1c in diabetic patients [32, 33, 35, 36, 57, 58]. Furthermore, as HbA1c is an indicator of oxidative stress, it is suggested that ketogenic diet induces a reduction in the generation of reactive oxygen species and improves the oxidative status. Gumbiner and his colleagues [38] showed that the improvement in the blood glucose levels is due to the direct effect of the ketone bodies on the hepatic glucose output. It has also been shown that in addition to normalizing the blood glucose level, LCKD was effective in reversing diabetic associated complications [59].
Concerning the mechanism of ketogenic diet action, it is worth mentioning that a mitochondrial link is suggested with regard to the antiseizure potential of ketogenic diet [60]. In the rat hippocampus, it has been found that the anticonvulsive effects of ketogenic diet was associated with an increase in mitochondrial biogenesis [61]. In support of this view, studies from our laboratory [34] have also shown an increase in mitochondrial biogenesis in rat cardiac muscle following the administration of a LCKD (Fig. 7). Other investigators have also shown that ketogenic diets stimulate mitochondrial biogenesis, improve mitochondrial function, decrease oxidative stress [61, 62], and contribute to reducing the glycolytic rate due to increases in lipid oxidation and mitochondrial respiration [62]. These studies, therefore, suggest that ketogenic diets have the potential to be used as a possible treatment for mitochondrial disorders [63]. Mitochondrial disease generally occurs in tissues with high energy demands such as the brain, muscle, heart, and endocrine system [64, 65, 66]. Although there appears to be an association with the mechanism of ketogenic diet action and mitochondrial biogenesis in oxidative stress, further studies are necessary to understand the underlying molecular mechanisms of ketogenic diet function.
Fig. 7.
Micrographs showing the ultrastructure of the mitochondria of rat cardiac muscle of an adult rat that was fed normal rat chow (a) and one that was fed with LCKD (b). There is an increase in the number of mitochondria in rats fed with low-carbohydrate diet. Scale bar, 1 µm [34]. LCKD, low-carbohydrate ketogenic diet.
Now it is well established that the use of ketogenic diets in weight loss therapy is effective. More than 90% of the diabetic patients are obese and here is a direct link between type 2 diabetes and obesity [67]. It is shown that a modest 5% weight loss can significantly improve the HbA1c levels in obese diabetic subjects [68]. Although some investigators suggest that there is not any metabolic advantages in low-carbohydrate diets and that weight loss results from the increased satiety effect and reduced caloric intake [69], several other investigators provide evidences that contradict the above-mentioned view [70]. Further evidences suggest ketone bodies are directly involved in the modulation of ghrelin and leptin level that influence appetite [71]. In general, based on the current evidence the weight loss effect of LCKD could be due to the reduction in appetite due to higher satiety effect of proteins [69, 72], effects on appetite control of hormones such as ghrelin and leptin [71], and the direct appetite-suppressant action of the ketone bodies [73]. Further evidence suggests that the weight loss effect of LCKD could also be due to the reduction in lipogenesis and increased lipolysis [72], the increased metabolic requirements of gluconeogenesis and the thermic effect of proteins [42, 70], and reduced resting respiratory quotient due to greater metabolic efficiency in consuming fats [71].
The main concern regarding the use of ketogenic diet with a high protein and fat content is that this diet will causes adverse effects by altering their lipid profile drastically [9, 10]. On the other hand, several recent studies have demonstrated that a low-carbohydrate diet produces significant benefits in the lipid profile [9, 10]. Following the administration of a ketogenic diet, there was a reduction in total cholesterol, increase in HDL, and decrease in the level of blood triglycerides [27, 74, 75, 76]. Saturated fatty acids that are involved in increased risk for cardiovascular diseases and insulin resistance [10] are found to be more associated with dietary carbohydrate [77, 78]. There is a direct link between higher level of insulin and the activation of HMG-CoA reductase a key enzyme in cholesterol biosynthesis. Thus, a reduction in dietary carbohydrate together with an appropriate cholesterol intake will lead to an inhibition of cholesterol biosynthesis. Results from our laboratory and other recent studies have shown that LCKD decreased the level of triglycerol and LDL cholesterol and increased the level of HDL cholesterol [27]. Similar results were obtained when obese subjects with high cholesterol level and obese subjects with diabetes were treated with LCKD for a longer period. In summary, several studies on the effect of ketogenic diets on cardiovascular disease suggest that it is safe to use ketogenic diet in diabetic subjects as well as in subjects with high cholesterol level [30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 79]. In addition to the mechanisms of ketogenic action mentioned above, ketogenic diets are involved in various other pathological conditions [16, 17, 18, 19, 20] such as neurodegenerative diseases, brain trauma, polycystic ovary syndrome, cancer, and osteoporosis, the mechanisms of which are not mentioned in this review.
Side Effects of Ketogenic Diet
During the shift from carbohydrate to fat-based energy utilization (keto-adaptation) there is a feeling of fatigue, lethargy, and headache [80]. In diabetic patients, due to the efficacy of LCKD, hypoglycemia may occur [81, 82] and could result in a significant reduction in the units of insulin required to be administered or cessation or reduction in the doses of oral drugs administered for type 2 diabetes [40, 83, 84]. Therefore, patients those who are on insulin or antidiabetic drugs should be monitored carefully.
Negative events of ketogenic diet include dehydration, dyselectrolytemia, and hypovitaminosis [85]. In addition, short-term LCKD impairs bone mass density and mechanical properties of bone [86]. Therefore, to minimize the above-mentioned side effects, daily supplements of electrolytes, multivitamins, potassium citrate and calcium, vitamin D, and minerals should be given during the period of ketogenic diet administration. Another side effect of LCKD is the formation of kidney stones and increased production and the decreased excretion of uric acid. This is due to limited fluid intake and the suppression of thirst by ketone bodies. Similarly, ketone bodies are also involved in the suppression of food intake. Hyperuricemia leads to urate stone formation. It should be noted that studies from our laboratory have observed a decrease in serum level of urea. To rectify this issue, inclusion of 5% carbohydrate composition in the diet is recommended [87].
Another concern regarding the ketogenic diet is that the subjects who are on this diet have a reduction in the intake of healthy foods such as fruits and vegetables that contain polyphenols and antioxidants that fight against the free radicals. Type 2 diabetes is associated with oxidative stress and limiting the supply of polyphenols and antioxidants may increase the imbalance of antioxidant-oxidation system in our body [88]. In order to overcome this situation, it is suggested to supplement the ketogenic diet with extracts of polyphenols and antioxidants, especially in patients with type 2 diabetes.
Constipation is also a noted as a side effect of LCKD, which could be due to the decreased fiber content and dehydration due to the suppression of thirst by ketones [89]. This situation can be prevented by increasing the fiber content in the diet, increasing fluid intake and using laxatives [90]. In general, studies of various investigators have convincingly shown that these supplementation and modification can eliminate the adverse effects LCKD.
Conclusion
The information presented in this review shows efficacy of ketogenic diet in glycemic control in hyperlipidemic diabetic patients with type 2 diabetes. In addition to reduction in body weight and improving lipid profile, there was a significant improvement in HbA1c and reduction in the intake of insulin and oral antidiabetic drugs in patients with type 2 diabetes. Low carbohydrate shifts the body to an alternate metabolic pathway that stabilizes insulin resistance, normalizes blood glucose, glycosylated hemoglobin and hepatic, renal and plasma lipid profile in type 2 diabetic patients. Due to the significant effect of LCKD in lowering blood glucose level and contributing to the reduction of insulin and antidiabetic medication, the diabetic patients on LCKD diet should be routinely monitored to understand the optimal adjustments for insulin, antidiabetic, and diuretic medications in order to avoid hypoglycemia and dehydration complications.
Conflict of Interest Statement
The authors have no conflicts of interest to declare.
Funding Sources
Some of the studies mentioned in this review are funded by Kuwait University.
Author Contributions
All the authors have equally contributed to preparation and editing of the manuscript.
Acknowledgements
The authors would like to thank Ms. Alaa Al-Khalifa, who was involved in the animal studies mentioned in this review and Mrs. Elizabeth Mathew, chief technician, Department of Medical Laboratory Sciences, Faculty of Allied Health Sciences, Kuwait University, for her expert technical advice and assistance.
References
- 1.Centers for Disease Control and Prevention. National diabetes fact sheet: national estimates and general information on diabetes and prediabetes in the United States, 2011. s.l: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. 2011.
- 2.International Diabetes Federation . IDF diabetes atlas. 8th ed. Brussels, Belgium: International Diabetes Federation; 2017. Available online: http://www.diabetesatlas.org. [PubMed] [Google Scholar]
- 3.Abuyassin B, Laher I. Diabetes epidemic sweeping the Arab world. World J Diabetes. 2016;7((8)):165–74. doi: 10.4239/wjd.v7.i8.165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.American Diabetes Association Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018 May;41((5)):917–28. doi: 10.2337/dci18-0007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fong DS, Aiello LP, Ferris FL, 3rd, Klein R. Diabetic retinopathy. Diabetes Care. 2004;27((10)):2540–53. doi: 10.2337/diacare.27.10.2540. [DOI] [PubMed] [Google Scholar]
- 6.Gross JL, de Azevedo MJ, Silveiro SP, Canani LH, Caramori ML, Zelmanovitz T. Diabetic nephropathy: diagnosis, prevention, and treatment. Diabetes Care. 2005;28((1)):164–76. doi: 10.2337/diacare.28.1.164. [DOI] [PubMed] [Google Scholar]
- 7.American Diabetes Association Standards of medical care in diabetes 2007 [position statement] Diabetes Care. 2007;30:S4–S41. doi: 10.2337/dc07-S004. [DOI] [PubMed] [Google Scholar]
- 8.Avogaro A, Giorda C, Maggini M, Mannucci E, Raschetti R, Lombardo F, et al. Incidence of coronary heart disease in type 2 diabetic men and women: impact of microvascular complications, treatment, and geographic location. Diabetes Care. 2007;30((5)):1241–7. doi: 10.2337/dc06-2558. [DOI] [PubMed] [Google Scholar]
- 9.Shai I, Schwarzfuchs D, Henkin Y, Shahar DR, Witkow S, Greenberg I, et al. Weight loss with a low-carbohydrate, mediterranean, or low-fat diet. N Engl J Med. 2008;359((3)):229–41. doi: 10.1056/NEJMoa0708681. [DOI] [PubMed] [Google Scholar]
- 10.Volek JS, Phinney SD, Forsythe CE, Quann EE, Wood RJ, Puglisi MJ, et al. Carbohydrate restriction has a more favorable impact on the metabolic syndrome than a low fat diet. Lipids. 2009;44((4)):297–309. doi: 10.1007/s11745-008-3274-2. [DOI] [PubMed] [Google Scholar]
- 11.Individualizing Your Patient's A1C Target (2018). Available from: http://guidelines.diabetes.ca/reduce-complications/a1ctarget.
- 12.Higgins V, Piercy J, Roughley A, Milligan G, Leith A, Siddall J, et al. Trends in medication use in patients with type 2 diabetes mellitus: a long-term view of real-world treatment between 2000 and 2015. Diabetes Metab Syndr Obes. 2016;9:371–80. doi: 10.2147/DMSO.S120101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.American Diabetes Association Nutrition recommendations and interventions for diabetes: 2013. Diabetes Care. 2013;36((Suppl 1)):S12–32. doi: 10.2337/dc07-S048. [DOI] [PubMed] [Google Scholar]
- 14.Banting W. Open letter on being overweight. 4th ed. 1869. Available from: https://onlinelibrary.wiley.com/doi/pdf/10.1002/j.15508528.1993.tb00605. [Google Scholar]
- 15.Paoli A. Ketogenic diet for obesity: friend or foe? Int J Environ Res Public Health. 2014;11((2)):2092–107. doi: 10.3390/ijerph110202092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Murphy P, Likhodii SS, Hatamian M, McIntyre Burnham W. Effect of the ketogenic diet on the activity level of Wistar rats. Pediatr Res. 2005;57((3)):353–7. doi: 10.1203/01.PDR.0000150804.18038.79. [DOI] [PubMed] [Google Scholar]
- 17.Kossoff EH, Pyzik PL, McGrogan JR, Vining EP, Freeman JM. Efficacy of the ketogenic diet for infantile spasms. Pediatrics. 2002;109((5)):780–3. doi: 10.1542/peds.109.5.780. [DOI] [PubMed] [Google Scholar]
- 18.Murphy P, Likhodii S, Nylen K, Burnham WM. The antidepressant properties of the ketogenic diet. Biol Psychiatry. 2004;56((12)):981–3. doi: 10.1016/j.biopsych.2004.09.019. [DOI] [PubMed] [Google Scholar]
- 19.Seyfried TN, Mukherjee P. Targeting energy metabolism in brain cancer: review and hypothesis. Nutr Metab. 2005;2:30. doi: 10.1186/1743-7075-2-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chester B, Babu JR, Greene MW, Geetha T. The effects of popular diets on type 2 diabetes management. Diabetes Metab Res Rev. 2019;35((8)):e3188. doi: 10.1002/dmrr.3188. [DOI] [PubMed] [Google Scholar]
- 21.Krebs HA. The regulation of the release of ketone bodies by the liver. Adv Enzyme Regul. 1966;4:339–54. doi: 10.1016/0065-2571(66)90027-6. [DOI] [PubMed] [Google Scholar]
- 22.Feinman RD, Pogozelski WK, Astrup A, Bernstein RK, Fine EJ, Westman EC, et al. Dietary carbohydrate restriction as the first approach in diabetes management: critical review and evidence base. Nutrition. 2015;31((1)):1–13. doi: 10.1016/j.nut.2014.06.011. [DOI] [PubMed] [Google Scholar]
- 23.Atkins R. Dr Atkins' new diet revolution. New York, NY: Avon Books; 1998. [Google Scholar]
- 24.The Atkins trial kit handbook: a simple guide to doing Atkins. Ronkonkoma, NY: Atkins Nutritionals; 2001. [Google Scholar]
- 25.Sears B. The zone: a dietary road map. New York, NY: HarperCollins; 1995. [Google Scholar]
- 26.Agatston A. The south beach diet: the delicious, doctor-designed, foolproof plan for fast and healthy weight loss. New York, NY: St. Martin's Press; 2005. [Google Scholar]
- 27.Dashti HM, Al-Zaid NS, Mathew TC, Al-Mousawi M, Talib H, Asfar SK, et al. Long term effects of ketogenic diet in obese subjects with high cholesterol level. Mol Cell Biochem. 2006;286((1–2)):1–9. doi: 10.1007/s11010-005-9001-x. [DOI] [PubMed] [Google Scholar]
- 28.Cordain L, Eaton SB, Miller JB, Mann N, Hill K. The paradoxical nature of hunter-gatherer diets: meat-based, yet non-atherogenic. Eur J Clin Nutr. 2002;56((Suppl 1)):S42–52. doi: 10.1038/sj.ejcn.1601353. [DOI] [PubMed] [Google Scholar]
- 29.Mitchell GA, Kassovska-Bratinova S, Boukaftane Y, Robert MF, Wang SP, Ashmarina L, et al. Medical aspects of ketone body metabolism. Clin Invest Med. 1995;18((3)):193–216. [PubMed] [Google Scholar]
- 30.Dashti HM, Bo-Abbas YY, Asfar SK, Mathew TC, Hussein T, Behbahani A, et al. Ketogenic diet modifies the risk factors of heart disease in obese patients. Nutrition. 2003;19((10)):901–2. doi: 10.1016/s0899-9007(03)00161-8. [DOI] [PubMed] [Google Scholar]
- 31.Dashti HM, Mathew TC, Hussein T, Asfar SK, Behbehani AI, Al-Sayer HM, et al. Long term effects of ketogenic diet in obese subjects. Exp Clin Cardiol. 2004;9((3)):200–5. [PMC free article] [PubMed] [Google Scholar]
- 32.Dashti HM, Mathew TC, Khadada M, Al-Mousawi M, Talib H, Asfar SK, et al. Beneficial effects of ketogenic diet in obese diabetic subjects. Mol Cell Biochem. 2007;302((1–2)):249–56. doi: 10.1007/s11010-007-9448-z. [DOI] [PubMed] [Google Scholar]
- 33.Hussain TA, Mathew TC, Dashti AA, Asfar S, Al-Zaid N, Dashti HM. Effect of low-calorie versus low-carbohydrate ketogenic diet in type 2 diabetes. Nutrition. 2012;28((10)):1016–21. doi: 10.1016/j.nut.2012.01.016. [DOI] [PubMed] [Google Scholar]
- 34.Al-Zaid NS, Dashti HM, Mathew TC, Juggi JS. Low carbohydrate ketogenic diet enhances cardiac tolerance to global ischaemia. Acta Cardiol. 2007;62((4)):381–9. doi: 10.2143/AC.62.4.2022282. [DOI] [PubMed] [Google Scholar]
- 35.Al-Khalifa A, Mathew TC, Al-Zaid NS, Mathew E, Dashti HM. Therapeutic role of low-carbohydrate ketogenic diet in diabetes. Nutrition. 2009;25((11–12)):1177–85. doi: 10.1016/j.nut.2009.04.004. [DOI] [PubMed] [Google Scholar]
- 36.Al-Khalifa A, Mathew TC, Al-Zaid NS, Mathew E, Dashti H. Low carbohydrate ketogenic diet prevents the induction of diabetes using streptozotocin in rats. Exp Toxicol Pathol. 2011;63((7–8)):663–9. doi: 10.1016/j.etp.2010.05.008. [DOI] [PubMed] [Google Scholar]
- 37.Phinney SD, Bistrian BR, Wolfe RR, Blackburn GL. The human metabolic response to chronic ketosis without caloric restriction: physical and biochemical adaptation. Metab Clin Exp. 1983;32((8)):757–68. doi: 10.1016/0026-0495(83)90105-1. [DOI] [PubMed] [Google Scholar]
- 38.Gumbiner B, Wendel JA, McDermott MP. Effects of diet composition and ketosis on glycemia during very-low-energy-diet therapy in obese patients with non-insulin-dependent diabetes mellitus. Am J Clin Nutr. 1996;63((1)):110–5. doi: 10.1093/ajcn/63.1.110. [DOI] [PubMed] [Google Scholar]
- 39.Samaha FF, Iqbal N, Seshadri P, Chicano KL, Daily DA, McGrory J, et al. A low-carbohydrate as compared with a low-fat diet in severe obesity. N Engl J Med. 2003;348((21)):2074–81. doi: 10.1056/NEJMoa022637. [DOI] [PubMed] [Google Scholar]
- 40.Yancy WS, Jr, Foy M, Chalecki AM, Vernon MC, Westman EC. A low-carbohydrate, ketogenic diet to treat type 2 diabetes. Nutr Metab. 2005;2:34. doi: 10.1186/1743-7075-2-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Westman EC, Yancy WS, Mavropoulos JC, Marquart M, McDuffie JR. The effect of a low-carbohydrate, ketogenic diet versus a low-glycemic index diet on glycemic control in type 2 diabetes mellitus. Nutr Metab. 2008;5((1)):36. doi: 10.1186/1743-7075-5-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Nielsen J, Joensson E. Low‐carbohydrate diet in type 2 diabetes: stable improvement of bodyweight and glycemic control during 44 months follow‐up. Nutr Metab. 2008;5((1)):14. doi: 10.1186/1743-7075-5-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Saslow LR, Daubenmier JJ, Moskowitz JT, Kim S, Murphy EJ, Phinney SD, et al. Twelve-month outcomes of a randomized trial of a moderate-carbohydrate versus very low-carbohydrate diet in overweight adults with type 2 diabetes mellitus or prediabetes. Nutr Diabetes. 2017;7((12)):304–29. doi: 10.1038/s41387-017-0006-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Walton CM, Perry K, Hart RH, Berry SL, Bikman BT. Recent evidence suggests that ketogenic diets may playan impactful role in type II diabetes and obesity. Improvement in glycemic and lipid profiles in type 2 diabetics with a 90-day ketogenic diet. J Diabetes Res. 2019:1–6. doi: 10.1155/2019/8681959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rallis S. Optimizing glycemic control in type 2 diabeticpatients through the use of a low-carbohydrate, high-fat, ketogenic diet: a review of 2 patientsin primary care diabetes, metabolic syndrome and obesity. Targets Ther. 2019;12:299–303. doi: 10.2147/DMSO.S195994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hossain P, Kawar B, El Nahas M. Obesity and diabetes in the developing world: a growing challenge. N Engl J Med. 2007;356((3)):213–5. doi: 10.1056/NEJMp068177. [DOI] [PubMed] [Google Scholar]
- 47.Molavi B, Rasouli N, Kern PA. The prevention and treatment of metabolic syndrome and high-risk obesity. Curr Opin Cardiol. 2006;21((5)):479–85. doi: 10.1097/01.hco.0000240586.76344.f5. [DOI] [PubMed] [Google Scholar]
- 48.Volek JS, Sharman MJ, Love DM, Avery NG, Gómez AL, Scheett TP, et al. Body composition and hormonal responses to a carbohydrate-restricted diet. Metab Clin Exp. 2002;51((7)):864–70. doi: 10.1053/meta.2002.32037. [DOI] [PubMed] [Google Scholar]
- 49.Xu C, Markova M, Seebeck N, Loft A, Hornemann S, Gantert T, et al. High-protein diet more effectively reduces hepatic fat than low-protein diet despite lower autophagy and FGF21 levels. Liver Int. 2020:1–16. doi: 10.1111/liv.14596. [DOI] [PubMed] [Google Scholar]
- 50.Goss AM, Gower B, Soleymani T, Stewart M, Pendergrass M, Lockhart M, et al. Effects of weight loss during a very low carbohydrate diet on specific adipose tissue depots and insulin sensitivity in older adults with obesity: a randomized clinical trial. Nutr Metab. 2020;17((1)):64. doi: 10.1186/s12986-020-00481-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Crowe TC. Safety of low-carbohydrate diets. Obes Rev. 2005;6((3)):235–45. doi: 10.1111/j.1467-789X.2005.00196.x. [DOI] [PubMed] [Google Scholar]
- 52.Kasper H, Thiel H, Ehl M. Response of body weight to a low carbohydrate, high fat diet in normal and obese subjects. Am J Clin Nutr. 1973;26((2)):197–204. doi: 10.1093/ajcn/26.2.197. [DOI] [PubMed] [Google Scholar]
- 53.Olsson KE, Saltin B. Variation in total body water with muscle glycogen changes in man. Acta Physiol Scand. 1970;80((1)):11–8. doi: 10.1111/j.1748-1716.1970.tb04764.x. [DOI] [PubMed] [Google Scholar]
- 54.Meckling KA, Gauthier M, Grubb R, Sanford J. Effects of a hypocaloric, low-carbohydrate diet on weight loss, blood lipids, blood pressure, glucose tolerance, and body composition in free-living overweight women. Can J Physiol Pharmacol. 2002;80((11)):1095–105. doi: 10.1139/y02-140. [DOI] [PubMed] [Google Scholar]
- 55.Mei J, Lindqvist A, Krabisch L, Rehfeld JF, Erlanson-Albertsson C. Appetite suppression through delayed fat digestion. Physiol Behav. 2006;89((4)):563–8. doi: 10.1016/j.physbeh.2006.07.020. [DOI] [PubMed] [Google Scholar]
- 56.Kennedy L, Mehl TD, Riley WJ, Merimee TJ. Non-enzymatically glycosylated serum protein in diabetes mellitus: an index of short-term glycaemia. Diabetologia. 1981;21((2)):94–8. doi: 10.1007/BF00251273. [DOI] [PubMed] [Google Scholar]
- 57.Neilsen JV, Joensson E, Nilsson AK. Lasting improvement of hyperglycaemia and bodyweight: low-carbohydrate diet in type 2 diabetes. A brief report. Ups J Med Sci. 2005;110:179–83. [PubMed] [Google Scholar]
- 58.Willi SM, Martin K, Datko FM, Brant BP. Treatment of type 2 diabetes in childhood using a very-low-calorie diet. Diabetes Care. 2004;27((2)):348–53. doi: 10.2337/diacare.27.2.348. [DOI] [PubMed] [Google Scholar]
- 59.Poplawski MM, Mastaitis JW, Isoda F, Grosjean F, Zheng F, Mobbs CV. Reversal of diabetic nephropathy by a ketogenic diet. PLoS One. 2011;6((4)):e18604. doi: 10.1371/journal.pone.0018604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bough K. Energy metabolism as part of the anticonvulsant mechanism of the ketogenic diet. Epilepsia. 2008;49((Suppl 8)):91–3. doi: 10.1111/j.1528-1167.2008.01846.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Bough KJ, Wetherington J, Hassel B, Pare JF, Gawryluk JW, Greene JG, et al. Mitochondrial biogenesis in the anticonvulsant mechanism of the ketogenic diet. Ann Neurol. 2006;60((2)):223–35. doi: 10.1002/ana.20899. [DOI] [PubMed] [Google Scholar]
- 62.Danial NN, Hartman AL, Stafstrom CE, Thio LL. How does the ketogenic diet work? Four potential mechanisms. J Child Neurol. 2013;28((8)):1027–33. doi: 10.1177/0883073813487598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Santra S, Gilkerson RW, Davidson M, Schon EA. Ketogenic treatment reduces deleted mitochondrial DNAs in cultured human cells. Ann Neurol. 2004;56((5)):662–9. doi: 10.1002/ana.20240. [DOI] [PubMed] [Google Scholar]
- 64.Stafford P, Abdelwahab MG, Kim DY, Preul MC, Rho JM, Scheck AC. The ketogenic diet reverses gene expression patterns and reduces reactive oxygen species levels when used as an adjuvant therapy for glioma. Nutr Metab. 2010;7:74. doi: 10.1186/1743-7075-7-74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kang HC, Lee YM, Kim HD. Mitochondrial disease and epilepsy. Brain Dev. 2013;35((8)):757–61. doi: 10.1016/j.braindev.2013.01.006. [DOI] [PubMed] [Google Scholar]
- 66.Wallace DC, Fan W, Procaccio V. Mitochondrial energetics and therapeutics. Annu Rev Pathol. 2010;5:297–348. doi: 10.1146/annurev.pathol.4.110807.092314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Ibrahim M, Tuomilehto J, Aschner P, Beseler L, Cahn A, Eckel RH, et al. Global status of diabetes prevention and prospects for action: a consensus statement. Diabetes Metab Res Rev. 2018;34((6)):e3021. doi: 10.1002/dmrr.3021. [DOI] [PubMed] [Google Scholar]
- 68.American Diabetes Association 4. Lifestyle management: standards of medical care in diabetes-2018. Diabetes Care. 2018 Jan;41((Suppl 1)):S38–S72. doi: 10.2337/dc18-S004. [DOI] [PubMed] [Google Scholar]
- 69.Westerterp-Plantenga MS, Nieuwenhuizen A, Tome D, Soenen S, Westerterp KR. Dietary protein, weight loss, and weight maintenance. Annu Rev Nutr. 2009;29:21–41. doi: 10.1146/annurev-nutr-080508-141056. [DOI] [PubMed] [Google Scholar]
- 70.Feinman RD, Fine EJ. Nonequilibrium thermodynamics and energy efficiency in weight loss diets. Theor Biol Med Model. 2007;4:27–1. doi: 10.1186/1742-4682-4-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Paoli A, Grimaldi K, Bianco A, Lodi A, Cenci L, Parmagnani A. Medium term effects of a ketogenic diet and a mediterranean diet on resting energy expenditure and respiratory ratio. BMC Proc. 2012;6((Suppl 3)):P37. [Google Scholar]
- 72.Veldhorst M, Smeets A, Soenen S, Hochstenbach-Waelen A, Hursel R, Diepvens K, et al. Protein-induced satiety: effects and mechanisms of different proteins. Physiol Behav. 2008;94:300–7. doi: 10.1016/j.physbeh.2008.01.003. [DOI] [PubMed] [Google Scholar]
- 73.Johnstone AM, Horgan GW, Murison SD, Bremner DM, Lobley GE. Effects of a high-protein ketogenic diet on hunger, appetite, and weight loss in obese men feeding ad libitum. Am J Clin Nutr. 2008;87:44–55. doi: 10.1093/ajcn/87.1.44. [DOI] [PubMed] [Google Scholar]
- 74.Veldhorst MA, Westerterp-Plantenga MS, Westerterp KR. Gluconeogenesis and energy expenditure after a high-protein, carbohydrate-free diet. Am J Clin Nutr. 2009;90:519–26. doi: 10.3945/ajcn.2009.27834. [DOI] [PubMed] [Google Scholar]
- 75.Paoli A, Rubini A, Volek JS, Grimaldi KA. Beyond weight loss: a review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur J Clin Nutr. 2013;67:789–96. doi: 10.1038/ejcn.2013.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Bueno NB, de Melo IS, de Oliveira SL, da Rocha Ataide T. Very-low-carbohydrate ketogenic diet v. Low-fat diet for long-term weight loss: a meta-analysis of randomised controlled trials. Br J Nutr. 2013;110:1178–87. doi: 10.1017/S0007114513000548. [DOI] [PubMed] [Google Scholar]
- 77.Bisschop PH, de Metz J, Ackermans MT, Endert E, Pijl H, Kuipers F, et al. Dietary fat content alters insulin-mediated glucose metabolism in healthy men. Am J Clin Nutr. 2001;73:554–9. doi: 10.1093/ajcn/73.3.554. [DOI] [PubMed] [Google Scholar]
- 78.Bisschop PH, Pereira Arias AM, Ackermans MT, Endert E, Pijl H, Kuipers F, et al. The effects of carbohydrate variation in isocaloric diets on glycogenolysis and gluconeogenesis in healthy men. J Clin Endocrinol Metab. 2000;85:1963–7. doi: 10.1210/jcem.85.5.6573. [DOI] [PubMed] [Google Scholar]
- 79.Westman EC, Mavropoulos J, Yancy WS, Volek JS. A review of low-carbohydrate ketogenic diets. Curr Atheroscler Rep. 2003;5:476–83. doi: 10.1007/s11883-003-0038-6. [DOI] [PubMed] [Google Scholar]
- 80.Sumithran P, Proietto J. Ketogenic diets for weight loss: a review of their principles, safety and efficacy. Obes Res Clin Pract. 2008;2:1–3. doi: 10.1016/j.orcp.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 81.Daly ME, Paisey R, Millward BA, Eccles C, Williams K, Hammersley S, et al. Short-term effects of severe dietary carbohydrate restriction advice in type 2 diabetes- a randomized controlled trial. Diabet Med. 2006;23:15–20. doi: 10.1111/j.1464-5491.2005.01760.x. [DOI] [PubMed] [Google Scholar]
- 82.Daly ME, Piper J, Paisey R, Darby T, George L, Ball C, et al. Efficacy of carbohydrate restriction in obese type 2 diabetes patients. Diabet Med. 2006;23((2)):26. [Google Scholar]
- 83.Boden G, Sargrad K, Homko C, Mozzoli M, Stein TP. Effect of a low-carbohydrate diet on appetite, blood glucose levels, and insulin resistance in obese patients with type 2 diabetes. Ann Intern Med. 2005;142:403–11. doi: 10.7326/0003-4819-142-6-200503150-00006. [DOI] [PubMed] [Google Scholar]
- 84.Nielsen J, Jonsson E. Low-carbohydrate diet in type 2 diabetes. Stable improvement of bodyweight and glycaemic control during 22 months follow-up. Nutr Metab. 2006;3:22–7. doi: 10.1186/1743-7075-3-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Ballaban-Gil K, Callahan C, O'Dell C, Pappo M, Moshe S, Shinnar S. Complications of the ketogenic diet. Epilepsia. 1998;39:744–8. doi: 10.1111/j.1528-1157.1998.tb01160.x. [DOI] [PubMed] [Google Scholar]
- 86.Bielohuby M, Matsuura M, Herbach N, Kienzle E, Slawik M, Hoeflich A, et al. Short-term exposure to low-carbohydrate, high-fat diets induces low bone mineral density and reduces bone formation in rats. J Bone Miner Res. 2010;25:275–84. doi: 10.1359/jbmr.090813. [DOI] [PubMed] [Google Scholar]
- 87.Hartman AL, Vining EP. Clinical aspects of the ketogenic diet. Epilepsia. 2007;48:31–42. doi: 10.1111/j.1528-1167.2007.00914.x. [DOI] [PubMed] [Google Scholar]
- 88.Milder JB, Liang LP, Patel M. Acute oxidative stress and systemic Nrf2 activation by the ketogenic diet. Neurobiol Dis. 2010;40:238–44. doi: 10.1016/j.nbd.2010.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Volek JS, Gomez AL, Kraemer WJ. Fasting and postprandial lipoprotein responses to a low-carbohydrate diet supplemented with n-3 fatty acids. J Am Coll Nutr. 2000;19:383–91. doi: 10.1080/07315724.2000.10718935. [DOI] [PubMed] [Google Scholar]
- 90.Vamecq J, Vallee L, Lesage F, Gressens P, Stables JP. Antiepileptic popular ketogenic diet: emerging twists in an ancient story. Prog Neurobiol. 2005;75:1–28. doi: 10.1016/j.pneurobio.2004.11.003. [DOI] [PubMed] [Google Scholar]