Abstract
We herein report on a series of four patients presented to our tertiary care centre with features of multisystem inflammatory syndrome in children and cardiac involvement. Two of our patients had recent exposure to a COVID-19-positive patient, one had recent documented infection, and another had no known positive contact. All the patients were tested positive for severe acute respiratory syndrome coronavirus 2 immunoglobulin G antibody at the time of presentation. All of them fulfilled the diagnostic criteria according to the World Health Organization Centers for Disease Control or the British guidelines for MIS-C (fever for ≥3 days, multisystem involvement (at least two), elevated markers of inflammation, and no other alternative diagnosis).
Cardiac involvement was variable ranging from isolated ectasia of the coronary arteries to full-blown pancarditis such as severe biventricular dysfunction, multi-valvar involvement, and pericardial effusion.
All our patients received intravenous immunoglobulin (2 g/kg), methylprednisolone, and aspirin and some required inotropic support and ICU admission.
Remarkably, all our patients showed significant improvement in their cardiac disease within a few days as evident on serial echocardiographic evaluation. However, we stress the need for long-term follow-up as one of our patients demonstrated mild LV myocardial scarring as evident by gadolinium late enhancement on a cardiac MRI.
Keywords: Multisystem inflammatory syndrome in children (MIS-C), cardiac manifestations, COVID-19, coronary involvement, myocardial dysfunction
Case 1
An 8-year-old boy previously healthy was presented with 7 days history of high-grade fever, abdominal pain, and erythematous rash.
Upon presentation to the pediatric emergency department, the patient was noted to be lethargic and febrile. His vital signs showed a heart rate of 130 beats per minute and a systolic blood pressure of 84/46 mmHg. Physical exam was remarkable for bilateral non-purulent bulbar (limbus-sparing) conjunctivitis, cracked and dry lips, strawberry tongue, a faint macular rash, hepatosplenomegaly, scrotal, and lower limb oedema. Cardiac evaluation revealed a grade II–III systolic murmur over the apical area. He had exposure to COVID-19 4 weeks earlier. Upon presentation, SARS-CoV-2 reverse transcriptase polymerase chain reaction (CFX96 Platform) was negative, however, serology for SARS-CoV-2 immunoglobulin G was positive. Other laboratory tests were remarkable for anaemia, thrombocytopenia, acute liver, and kidney injury, hypoalbuminemia, elevated inflammatory markers as well as Troponin T and Pro-BNP (Table 1).
Table 1.
Laboratory results upon presentation
| Reference | Case 1 | Case 2 | Case 3 | Case 4 | |
|---|---|---|---|---|---|
| Pro-BNP | 5–125 pg/ml | 13,369 | 1698 | 21,436 | 3872 |
| Troponin T | 0–0.03 ng/L | 0.083 | 0.045 | 0.026 | 0.255 |
| WBC | 4.5–13.5 K/Ul | 10,297 | 7800 | 26,200 | 12,400 |
| Neutrophils | 29–75% | 75% | 91% | 94% | 87% |
| Lymphocytes | 16–75% | 18% | 6% | 4% | 9% |
| Haematocrit | 35–45% | 29 | 31 | 32 | 40 |
| Platelets | 150–450 K/Ul | 95,600 | 206,000 | 316,000 | 110,000 |
| CRP | <0.03 mg/dl | 140 | 87 | 308 | 265 |
| ESR | 0–20 mm/hour | – | 44 | 64 | 31 |
| Ferritin | 7–140 ng/ml | 748 | 248 | 324 | 1050 |
| Fibrinogen | 1.7–4 g/L | 1.6 | 4.27 | 7.64 | 7.33 |
| D-dimer | <0.5 µg/ml | 2319 | 1842 | 1534 | 705 |
| LDH | 135–223 U/L | 232 | 391 | 460 | 356 |
| Albumin | 36–53 g/L | 23 | 32 | 35 | 38 |
| ALT | 0–41 U/L | 46 | 20 | 16 | 30 |
| AST | 0–40 U/L | 35 | 43 | 23 | 63 |
| Interleukin-6 | Below 7 pg/ml | 26 | – | 341 | 61 |
| Creatinine | 0.6–1.2 mg/dl | 0.9 (baseline 0.4) | 0.3 | 0.5 | 1.1 |
Cardiac ultrasound revealed
Mildly dilated left ventricle with left ventricular internal diameter in diastole = 4.8 cm (z score: 1.7 on the 95th percentile), decreased left ventricular systolic function with estimated ejection fraction around 54% by M-mode and 50% by Simpson’s biplane method, depressed four-chamber global longitudinal strain of −14%, moderate-to-severe mitral valve regurgitation with mitral valve anterior leaflet tip prolapse, normal left atrial volume = 23 ml/m2, trace aortic valve regurgitation, mild pulmonary insufficiency, moderate tricuspid regurgitation, bilateral small pleural effusions, tiny pericardial effusion, and normal proximal coronary arteries.
He was admitted to the paediatric ICU and started on vasoactive medications, and treated with intravenous immunoglobulin at a dose of 2g/kg, methylprednisolone (1 mg/kg/dose IV every 12 hours), and low-dose aspirin.
His cardiac left ventricular function normalised (left ventricular ejection fraction: 60–65%) and was weaned off vasoactive infusions by 4th day of admission. He developed on the 5th DOA, a brief episode of sinus bradycardia reaching 40 BPM with premature atrial contractions, which was resolved spontaneously. He was transferred out of the PICU on the 8th DOA, and discharged home at the 9th DOA. His last echocardiogram was done 80 days post–presentation, which demonstrated normal biventricular systolic function (left ventricular ejection fraction: 60–65%; four-chamber left ventricular longitudinal strain: −21%), normal left ventricular dimensions (left ventricular internal diastolic diameter: 4.5 cm), mild mitral, and tricuspid regurgitation. Troponin T levels gradually returned to normal prior to discharge. Pro-BNP decreased significantly to 600 pg/ml on day number 4 and returned to normal on his first post-discharge clinic visit.
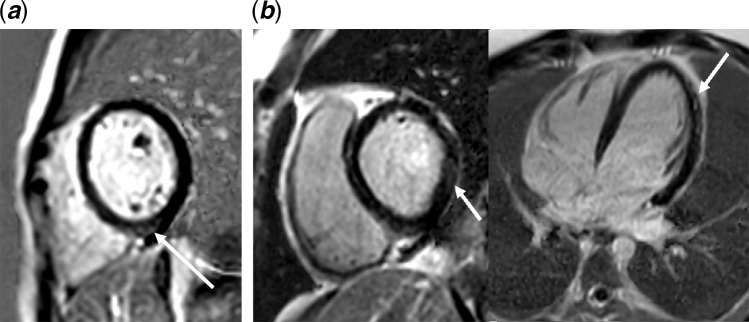
Cardiac MRI performed 3.5 months later showed minimal areas of delayed enhancement in the mid inferior septum and mid inferior wall representing mild squeal following myocarditis (Fig 1). Twenty-hour Holter monitor did not reveal any ectopic beats.
Figure 1.
Shows the results of gadolinium late enhancement by MRI. (a) Patient #1: Short-axis PSIR showing minimal areas of delayed enhancement in the mid myocardial, mid inferior wall minimal, and mid myocardial delayed enhancement was noted in the mid inferior wall (arrow). (b) Patient #4: Short-axis and four-chamber PSIR showing moderate patchy delayed enhancement in the subepicardial and mid myocardial region in the inferior and inferolateral wall and inferior septum of the mid and basal left ventricle (arrows). PSIR = phase-sensitive inversion recovery.
Case 2
A 5-year-old girl previously healthy was presented to the pediatric ED with 4 days history of high-grade fever associated with vomiting and erythematous rash.
Upon presentation, her vital signs were stable, her physical exam was positive for tachycardia, conjunctivitis, cracked lips, and erythematous rash.
The patient had exposure to COVID-19 4 weeks earlier, In the PED, she had a negative SARS-CoV-2 PCR and a positive serology for SARS-CoV-2 (SARS-CoV-2 IgG) antibodies.
Her laboratory tests were remarkable for anaemia, neutrophilia, low albumin, positive troponin, elevated Pro-BNP, and inflammatory markers (Table 1).
Cardiac ultrasound revealed traces of pericardial effusion, mild mitral regurgitation, mildly reduced left ventricular function left ventricular ejection fraction: 53 % by M-mode, and ectasia of the right coronary artery: measuring 2.8 mm, (z score: +2.2) proximally and 2.5 mm, (z score: +1.4) distally. And normal left main coronary artery measuring 1.7 mm (z score: −1.8).
The patient received intravenous immunoglobulins (2 g/kg), methylprednisolone (1 mg/kg/dose IV every 12 hours), and aspirin. She was defervesced on the 2nd DOA and discharged home on the 4th DOA.
Her latest echocardiogram performed 1 month later showed normal cardiac structure and function with resolution of the right coronary ectasia and no evidence of coronary aneurysmal dilatation.
Case 3
A 4-year-old boy known to have thalassaemia trait was presented to the pediatric ED with high-grade fever, conjunctivitis, vomiting, diarrhoea, and skin rash of 6 days duration.
On initial assessment, he was tachycardic with heart rate: 160–180 BPM and hypotensive BP: 85/53 mmHg. The remainder of the physical exam was significant for bilateral non-exudative and limbic-sparing conjunctivitis, cracked dry lips, and diffuse macular rash.
Laboratory tests were remarkable for leukocytosis with neutrophil predominance, microcytic anaemia, low albumin, elevated Pro-BNP, and inflammatory markers (Table 1).
Cardiac ultrasound revealed mild mitral regurgitation, mildly reduced left ventricular systolic function (ejection fraction: 50%), with prominent left anterior descending artery measuring 2.8 mm (z score 1.8), while right coronary artery was “normal” measuring 2.4 mm (z score 0.0).
The patient had no known exposure to COVID-19. He was tested negative for SARS-CoV-2 by PCR, however, he had positive serology SARS-CoV-2 (SARS-CoV-2 IgG) antibody upon presentation.
The patient was admitted to the PICU and started on vasoactive medications. He also received intravenous immunoglobulin (2 g/kg), methylprednisolone (1 mg/kg/dose IV every 12 hours), and aspirin.
He defervesced and was weaned off inotropic support by the 2nd DOA. He was transferred to regular floor on the 3rd DOA and discharged home after 6 days of hospitalisation.
His latest echocardiogram 14 days later showed normal heart structure and function. His coronary artery sizes regressed right coronary artery to 1.9 from 2.4 mm and left anterior descending from 2.8 to 1.7 mm.
Case 4
A 17-year-old boy previously healthy was presented with 5 days history of fever associated with throat pain, left cervical pain, and swelling.
In the pediatric ED, he was found to be lethargic, tachycardia (heart rate: 130 BPM) and hypotensive BP: 87/56 mmHg. Physical exam was remarkable for bilateral conjunctivitis, cracked dry lips, and palpable large tender left cervical lymph node (measuring 2.2 * 2.6 cm by CT scan). He had normal cardiac examination with regular rhythm and no murmurs.
His laboratory investigations were remarkable for thrombocytopenia, neutrophilia, elevated inflammatory markers, troponin, Pro-BNP, and D-dimer (Table 1).
Cardiac ultrasound showed traces of mitral regurgitation, significant biventricular systolic, and diastolic dysfunction (left ventricular ejection fraction by M-mode around 30%) as well as decreased left ventricular longitudinal strain measured at (−8%) from the four-chamber view.
The patient tested positive for COVID-19 1 month earlier. His SARS-CoV-2 PCR and SARS-CoV-2 (SARS-CoV-2 IgG) antibody were both positive upon presentation.
He was admitted to the CCU and started on vasoactive medications and Ivabradine for sinus tachycardia, and he received IVIG (2 g/kg), methylprednisolone, and aspirin.
He was weaned off inotropic support and switched to carvedilol and perindopril by the 2nd DOA, he defervesced on the 1st DOA, and was transferred to regular floor by the 3rd DOA and discharged home after 8 days of hospitalisation. The cardiac Troponin T level gradually decreased over 7 days, (from 0.255 ng/L on admission to 0.04 ng/L). Pro-BNP continued to increase from 3870 ng/L on admission and peaked at 21,480 ng/L on the 3rd DOA. The levels decreased significantly over 2 days to 12,600 ng/L and returned to normal levels on the first follow-up clinic visit.
His latest echocardiogram 52 days later showed improved albeit lower normal left ventricular systolic function (left ventricular ejection fraction = 56% by M-mode and Simpson’s Bi-plane), low normal left ventricular longitudinal strain (−18%), normal right ventricular systolic function, and normal proximal coronaries. The patient was weaned of carvedilol and remained on perindopril for borderline systemic arterial hypertension and borderline LVEF.
Cardiac MRI performed for 50 days post-presentation showed moderate patchy delayed enhancement in the subepicardial and mid myocardial region in the inferior and inferolateral wall and inferior septum of the mid and basal left ventricle suggestive of prior myocarditis (Fig 1). Twenty-hour Holter monitor did not reveal any ectopic beats. The patient was advised to refrain from sports activity and is scheduled for repeat CMRI in 6 months.
Discussion
Initially with the emergence of the novel coronavirus, the paediatric population was considered a low risk for developing severe COVID-19 infection and complications.1 Later in the course of the pandemic, around April, 2020, a serious entity linked to severe acute respiratory syndrome coronavirus 2 was described in the paediatric age group for the first time. Manifestations overlapping with many features of Kawazaki disease, toxic shock syndrome, and macrophage-activating syndrome. This entity was eventually referred to as multisystem inflammatory syndrome in children.2 Of interest were the high prevalence and the variable degree of cardiac involvement.
Although the exact prevalence of the cardiovascular manifestations is not yet well defined, it has been reported to be 66% by a multicentre case series involving 1116 hospitalised patients at 66 US hospitals.3 Even higher prevalence rate was reported by another large case series involving 186 patients by Feldstein et al.4
Based on large case series, the risk of developing MIS-C appears to vary by race and ethnicity affecting mostly Blacks and Hispanics.4,5 To date, few case reports have been published regarding MIS-C affecting patients in the Middle East area.6
According to a study by Kaushik et al, the main cardiovascular manifestations of MIS-C were left ventricular dysfunction and low ejection fraction, which were present in 63% of cases. Severely decreased left ventricular ejection fraction <30% was only noted in 4 patients out of 33.7 Those results were concordant with a larger study, and almost 80% of the patients had cardiac involvement that was evidenced by either an increase in the cardiac biomarkers (troponin and BNP) or the decrease in the left ventricular ejection fraction; albeit severe decrease in left ventricular ejection fraction <30% was only noted in 5 patients out of 149.8
All of our patients had ventricular dysfunction, three out of the four patients had mild decrease in their left ventricular systolic function with left ventricular ejection fraction ranging between 45 and 50%, which is the most common described degree of ventricular depression as discussed above and only one patient had severe ventricular dysfunction with an left ventricular ejection fraction of 20%. ACE inhibitors were used as an afterload-reducing agent in the case of decreased LV systolic function. In our fourth case, carvedilol was added for the treatment of severe LV systolic dysfunction and was weaned off gradually.
Myocardial strain is currently used as a more sensitive marker for myocardial function since it can detect subtle ventricular dysfunction even in the absence of depressed LVEF,9 strain abnormalities was seen in the two patients who was presented with left ventricular systolic dysfunction, and improved in parallel to the improvement in left ventricular ejection fraction.
We observe that the degree of the initial ventricular dysfunction didn’t correlate with the short-term outcomes. All our defervesced patients in hospital and follow-up echocardiography showed significant improvement with rapid recovery of the left ventricular function specially in our fourth case whose left ventricular ejection fraction increased to 50% by the fourth day of admission. Similarly, two case series reported by Kaushik et al7 and Chiotos et al8 highlighted the fast recovery of the left ventricular systolic function specifically prior to the hospital discharge in the first study and by the fifth day of admission in the second.
Although the exact mechanism of myocardial injury in MIS-C and its prognosis has not been yet fully elucidated, its similarity with the clinical course of Kawasaki-related myocarditis might point for a common pathophysiology.
Interestingly, this fast recovery of the left ventricular systolic function was also seen in patients with Kawasaki-related myocarditis as noted in a small series evaluating the progression of the left ventricular contractility in 25 patients diagnosed with acute Kawasaki at the time of diagnosis and after treatment, 57% of those with initially depressed systolic function normalised within 24 hours, 85% within 48 hours, and all but 1 patient by 6 weeks.10
To date, data on long-term consequences and myocardial sequela following MIS-C is still missing. A recent small case series reported cardiac MRI findings on three children and adolescents diagnosed with MIS-C during the acute phase, which showed diffuse myocardial oedema with no evidence of replacement fibrosis or focal necrosis;11 similarly, a larger study conducted on patients who have suffered from Kawasaki disease, also refuted the occurrence of myocardial fibrosis.12 Alarming was the presence of late enhancing lesions almost 3 months after the presentation in two of our patients. Of interest, those were the patients with low left ventricular ejection fraction and decreased left ventricular longitudinal strain.
Concerning coronary involvement, none of our patients had coronary aneurysm, however, coronary aneurysm has been reported in approximately 6–24% of patients, nevertheless the most described coronary abnormality is diffuse mild coronary artery dilation as seen in our second and third cases.13
Conclusion
We report on a small case series of MIS-C patients stressing the cardiovascular presentation and short-term follow-up. We found the same cardiovascular manifestations in our Middle Eastern patients, as reported from Europe and the Americans, indicating a universal cardiac presentation of this syndrome. Moreover, we stress the need for long-term follow-up as one of our patients demonstrated mild LV myocardial scarring as evident by gadolinium late enhancement on a cardiac MRI, which was performed 3 months after his “recovery” and apparently normalisation of his global LV systolic function by echocardiography.
Acknowledgements
We thank Miss M.A. and Miss R.T. for their help in the preparation of the manuscript.
Financial support
This research received no specific grant from any funding agency, commercial, or not-for-profit sectors.
Conflicts of interest
None.
Ethical standards
Patient data has been anonymised and the Institutional Review Board approval was waivered as this represent a descriptive small case series.
References
- 1. Rabinowicz S, Leshem E, Pessach IM. COVID-19 in the pediatric population-review and current evidence. Curr Infect Dis Rep 2020; 22: 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. World Health Organization. Multisystem inflammatory syndrome in children and adolescents temporally related to COVID-19, 2020. Retrieved May 15, 2020, from https://www.who.int/news-room/commentaries/detail/multisystem-inflammatory-syndrome-in-children-and-adolescents-with-covid-19
- 3. Narayanan SN, Ahamed MZ, Safia M, et al. Cardiovascular involvement in Kawasaki disease. Indian Pediatr 2005; 42: 918–922. [PubMed] [Google Scholar]
- 4. Feldstein LR, Rose EB, Horwitz SM, et al. Multisystem inflammatory syndrome in U.S. children and adolescents. N Engl J Med 2020; 383: 334–346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Davies P, Evans C, Kanthimathinathan HK, et al. Intensive care admissions of children with paediatric inflammatory multisystem syndrome temporally associated with SARS-CoV-2 (PIMS-TS) in the UK: a multicenter observational study. Lancet Child Adolesc Health 2020; 4: 669–677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Almoosa ZA, Al Ameer HH, AlKadhem SM, et al. Multisystem inflammatory syndrome in children, the real disease of COVID-19 in pediatrics – a multicenter case series from Al-Ahsa, Saudi Arabia. Cureus 2020; 12: e11064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Kaushik S, Aydin SI, Derespina KR, et al. Multisystem inflammatory syndrome in children associated with severe acute respiratory syndrome coronavirus 2 infection (MIS-C): a multi-institutional study from New York City. J Pediatr 2020; 224: 24–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Chiotos K, Bassiri H, Behrens EM, et al. Multisystem inflammatory syndrome in children during the coronavirus 2019 pandemic: a case series. J Pediatric Infect Dis Soc 2020; 9: 393–398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Tops LF, Delgado V, Marsan NA, Bax JJ. Myocardial strain to detect subtle left ventricular systolic dysfunction. Eur J Heart Fail 2017; 19: 307–313. [DOI] [PubMed] [Google Scholar]
- 10. Moran AM, Newburger JW, Sanders SP, et al. Abnormal myocardial mechanics in Kawasaki disease: rapid response to gamma-globulin. Am Heart J 2000; 139 (Pt 1): 217–223. [DOI] [PubMed] [Google Scholar]
- 11. Blondiaux E, Parisot P, Redheuil A, et al. Cardiac MRI in children with multisystem inflammatory syndrome associated with COVID-19. Radiology 2020; 297: E283–E288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Bratis K, Hachmann P, Child N, et al. Cardiac magnetic resonance feature tracking in Kawasaki disease convalescence. Ann Pediatr Cardiol 2017; 10: 18–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Sperotto F, et al. Cardiac manifestations in SARS-CoV-2-associated multisystem inflammatory syndrome in children: a comprehensive review and proposed clinical approach. Eur J Pediatr 2021; 180: 307–322. [DOI] [PMC free article] [PubMed] [Google Scholar]