Abstract
Psoriasis is a systemic immune-mediated inflammatory disease characterized by uncontrolled keratinocyte proliferation and poor differentiation. Cinnamaldehyde (CIN) has been shown to inhibit the proliferation and inflammatory response of primary and immortalized immune cells. However, to the best of our knowledge, the role of CIN in the progression of psoriasis remains unclear. Therefore, the present study aimed to investigate the biological role of CIN in psoriasis. To mimic abnormal proliferation and differentiation in keratinocytes in vitro, normal human epidermal keratinocytes (NHEKs) were stimulated with M5 (IL-1α, IL-17A, IL-22, oncostatin M and TNF-α). The viability and proliferation of NHEKs were analyzed using Cell Counting Kit-8 and 5-Ethynyl-2′-deoxyuridine assays, respectively. Western blotting was used to analyze the expression levels of keratin 1, filaggrin and loricrin in NHEKs. The results of the present study revealed that CIN significantly inhibited the proliferation and cell cycle progression, and promoted the differentiation of M5-stimulated NHEKs. CIN also markedly attenuated the extent of oxidative stress-induced damage in M5-stimulated NHEKs. Moreover, CIN ameliorated M5-induced inflammatory injury in NHEKs, as evidenced by the decreased levels of multiple inflammatory factors. Furthermore, CIN notably downregulated the expression levels of phosphorylated (p)-inhibitor of NF-κB, p-p65 and p-JNK in M5-stimulated NHEKs. In conclusion, the present data suggested that CIN may protect NHEKs against M5-induced hyperproliferation and inflammatory injury via inhibition of NF-κB and JNK signaling pathways. These results provide a novel insight on the role of CIN in psoriasis.
Keywords: psoriasis, cinnamaldehyde, hyperproliferation, inflammation, keratinocytes
Introduction
Psoriasis is one of the most common types of inflammatory-related skin diseases, which is characterized by uncontrolled proliferation and the poor differentiation of keratinocytes (1–3). Psoriasis is most commonly caused by genetic, immune and infection factors (4). For example, a diet with poor nutrition or low intake of omega-3 fatty acids can stimulate the development of psoriasis (5). However, to the best of our knowledge, the underlying mechanisms of the occurrence and development of psoriasis have not yet been fully elucidated, and the current treatment methods available for patients with psoriasis remain unsatisfactory. Therefore, the development of more effective strategies for the treatment of psoriasis is the current focus of research.
In the affected skin areas of psoriasis, the expression levels of chemokines [such as IL-8 and C-X-C motif chemokine ligand 1 (CXCL1)], proinflammatory cytokines (such as IL-1β) and antimicrobial peptides [such as S100 calcium binding protein A7 (S100A7), β-defensin 2 and cathelicidin antimicrobial peptide (LL-37)] are upregulated, leading to the hyperproliferation of epithelial cells (6,7). Keratinocytes are the predominant type of epithelial cells of the epidermis (8). Emerging evidence has shown that treatment with M5 can induce psoriasis-like changes in cultured keratinocytes, including increased cell proliferation and inflammation, and poor differentiation (9). Thus, in the present study, in order to mimic abnormal proliferation and differentiation in keratinocytes in vitro, keratinocytes were stimulated with M5.
It has been reported that the NF-κB and JNK signaling pathways play crucial roles in the immune system and inflammatory response (10,11). For instance, salidroside, which is an extract of Sedum rosea, a perennial herb (12), can inhibit inflammation and keratinocyte proliferation by downregulating NF-κB and STAT3-related signaling pathways, thus improving the symptoms of psoriasis (12).
Cinnamaldehyde (CIN) is a Traditional Chinese medicine that is derived from the dried bark of the Cinnamomum lauraceae (Cinnamomum cassia Presl) plant (13). CIN was previously reported to exert potent fungicidal, antitumor, immunological and anti-inflammatory activities (14). In addition, CIN has been found to downregulate the expression levels of the proinflammatory cytokines, TNF-α, IL-1β and IL-6, in lipopolysaccharide (LPS)-stimulated macrophages (15). CIN has also been discovered to inhibit bleomycin-induced idiopathic pulmonary fibrosis in mice by inhibiting the production of inflammatory cytokines and reactive oxygen species (ROS) (16). However, to the best of our knowledge, the biological effect of CIN on psoriasis, particularly regarding its effect on keratinocyte proliferation, inflammation and differentiation, remains unclear. Therefore, the present study aimed to investigate the biological role of CIN in keratinocytes exposed to M5 to mimic abnormal proliferation and differentiation, and whether it can act as a therapeutic agent against psoriasis via the NF-κB and JNK signaling pathways.
Materials and methods
Cell lines and culture
Normal human epidermal keratinocytes (NHEKs) were obtained from ScienCell Research Laboratories, Inc. NHEKs were cultured in DMEM (Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10% FBS (Thermo Fisher Scientific, Inc.), 100 U/ml penicillin (Thermo Fisher Scientific, Inc.) and 100 mg/ml streptomycin (Thermo Fisher Scientific, Inc.), and maintained in an incubator at 37°C in the presence of 5% CO2. NHEKs were treated with 10 ng/ml M5 [IL-1α, IL-17A, IL-22, oncostatin M (OSM) and TNF-α; PeproTech China] for 24 h at 37°C to mimic abnormal proliferation and differentiation of keratinocytes (9).
Cell Counting Kit-8 (CCK-8) assay
CIN (MedChemExpress) was dissolved with DMSO. NHEKs were treated with CIN (0, 5, 10, 20, 40 or 80 µM) for 24 h at 37°C. A CCK-8 assay was used to evaluate the viability of NHEKs. Briefly, NHEKs (5×103 cells/well) were plated into a 96-well plate. Then, 10 µl CCK-8 reagent (Dojindo Molecular Technologies, Inc.) was added to each well and further incubated for 2 h at 37°C. The absorbance of each well was measured at a wavelength of 450 nm using a spectrophotometer (Bio-Rad Laboratories, Inc.) (17).
5-Ethynyl-2′-deoxyuridine (EdU) staining
An EdU DNA Proliferation in vitro Detection kit (Guangzhou RiboBio Co., Ltd.) was used to evaluate the proliferation of NHEKs, according to the manufacturer's protocol. Briefly, NHEKs were seeded into 24-well plates (2.5×105 cells/well) and incubated with 50 µM EdU at room temperature for 2 h. Following the incubation, cells were stained with Apollo staining solution for 30 min at 37°C in the dark. EdU-positive cells were observed using a fluorescence microscope (Olympus Corporation) (18).
Cell cycle distribution analysis
Flow cytometry was used to evaluate the cell cycle distribution of NHEKs. Briefly, cells were fixed in 70% ethanol overnight at 4°C, then incubated with 10 µl propidium iodide/RNase staining buffer (BD Biosciences) for 30 min at 37°C in the dark. The cell cycle distribution of NHEKs was analyzed using a FACScan™ flow cytometer (BD Biosciences). FlowJo software (version 10.6.2; FlowJo LLC) was used to analyze the data. Flow cytometry was performed according to methods outlined in previous studies (19,20).
Western blotting
RIPA lysis buffer (Beyotime Institute of Biotechnology) was used to extract the protein from cells. Total protein concentration was determined using a BCA protein assay kit (cat. no. AS1086; Aspen Biotechnology Co., Ltd.) and 30 µg protein per lane was separated via SDS-PAGE on 10% gel. The separated proteins were subsequently transferred onto PVDF membranes (EMD Millipore) and blocked with 5% skimmed milk powder diluted in TBS with 0.1% Tween-20 at room temperature for 1 h. The membranes were then incubated with the following primary antibodies at 4°C overnight: Anti-cyclin E1 (1:1,000; cat. no. ab33911), anti-CDK2 (1:1,000; cat. no. ab32147), anti-CDK inhibitor 1B (p27Kip1; 1:1,000; cat. no. ab32034), anti-keratin 1 (1:1,000; cat. no. ab185628), anti-filaggrin (1:1,000; cat. no. sc-66192), anti-loricrin (1:1,000; cat. no. ab137533), anti-keratin 5 (1:1,000; cat. no. ab64081), anti-keratin 10 (1:1,000; cat. no. ab237775), anti-inhibitor of NF-κB (IκBα; 1:1,000; cat. no. ab32518), anti-phosphorylated (p)-IκBα (1:1,000; cat. no. ab92700), anti-p65 (1:1,000; cat. no. ab140751), anti-p-p65 (1:1,000; cat. no. ab239882), anti-JNK (1:1,000; cat. no. ab199380), anti-p-JNK (1:1,000; cat. no. ab131499) and anti-β-actin (1:1,000; cat. no. ab8226). β-actin was used as an internal control. Following the primary antibody incubation, the membranes were incubated with an anti-rabbit secondary antibody (1:5,000; cat. no. ab96899) at room temperature for 1 h. Protein bands were visualized with an ECL kit (cat. no. AS1059-3; Aspen Biotechnology Co., Ltd.) and densitometric analysis was performed using AlphaEaseFC software (version 4.0; ProteinSimple). Anti-filaggrin was purchased from Santa Cruz Biotechnology, Inc., while all other antibodies were obtained from Abcam. All procedures were carried out according to previous studies (19,21).
ROS analysis
The production of ROS was detected using a ROS assay kit (Beyotime Institute of Biotechnology). Briefly, NHEKs (1×105 cells/well) were plated into 6-well plates for 24 h. Then, the cells were treated with CIN (5 or 10 µM) for 24 h at 37°C. Following which, NHEKs were stained with 2′-7′-Dichlorofluorescin diacetate for 20 min at 37°C. Subsequently, the cells were collected and gently washed. The fluorescence intensity was subsequently detected using a FACScan™ flow cytometer (BD Biosciences). FlowJo software (version 10.6.2; FlowJo LLC) was used to analyze the data. All procedures were carried out according to a previous study (22).
ELISA
Malondialdehyde (MDA) assay kit (cat. no. A003-1) and Reduced glutathione (GSH) assay kit (cat. no. A006-2-1) were purchased from Nanjing Jiancheng Bioengineering Institute. These specific ELISA kits were used to determine the concentrations of MDA and GSH in NHEKs, according to the manufacturer's instructions (23).
Reverse transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from cells using TRIpure Total RNA Extraction Reagent (ELK Biotechnology Co., Ltd.) according to the manufacturer's protocol. Total RNA was reverse transcribed into cDNA using an EntiLink™ 1st Strand cDNA Synthesis kit (ELK Biotechnology Co., Ltd.), according to the manufacturer's protocol. qPCR was subsequently performed on StepOne™ Real-Time PCR instrument (Thermo Fisher Scientific, Inc.) using an EnTurbo™ SYBR Green PCR SuperMix kit (ELK Biotechnology Co., Ltd.). The following qPCR thermocycling conditions were used: 3 min at 95°C, followed by 40 cycles of 10 sec at 95°C, 30 sec at 58°C, and 30 sec at 72°C. The primers used for qPCR are listed in Table I. β-actin was used as the internal control. mRNA levels were quantified using the 2−ΔΔCq method (24). All procedures were carried out according to a previous study (23).
Table I.
Primer sequences.
| Gene | Primer sequences (5′à3′) |
|---|---|
| IL-1β | F: ACGATGCACCTGTACGATCACT |
| R: GAGAACACCACTTGTTGCTCCA | |
| IL-8 | F: ACTGAGAGTGATTGAGAGTGGAC |
| R: AACCCTCTGCACCCAGTTTTC | |
| CXCL1 | F: AACCGAAGTCATAGCCACACTC |
| R: CTTCTCCTAAGCGATGCTCAAA | |
| S100A7 | F: GCACAAATTACCTCGCCGAT |
| R: GACATTTTATTGTTCCTGGGGTC | |
| β-defensin2 | F: ATGTCATCCAGTCTTTTGCCC |
| R: TGCGTATCTTTGGACACCATAG | |
| LL-37 | F: CGTGCTATAGATGGCATCAACC |
| R: GCCCGTCCTTCTTGAAGTCA | |
| β-actin | F: GTCCACCGCAAATGCTTCTA |
| R: TGCTGTCACCTTCACCGTTC |
F, forward; R, reverse; CXCL1, C-X-C motif chemokine ligand 1; S100A7, S100 calcium binding protein A7; LL-37, cathelicidin antimicrobial peptide.
Statistical analysis
Statistical analyses were performed using GraphPad Prism software (version 7.0; GraphPad Software, Inc.). The CCK-8 assay was repeated five times, while all other experiments were repeated three times. All data are presented as the mean ± SD. Statistical differences between at least three groups were determined using a one-way ANOVA followed by a Tukey's post hoc test. P<0.05 was considered to indicate a statistically significant difference.
Results
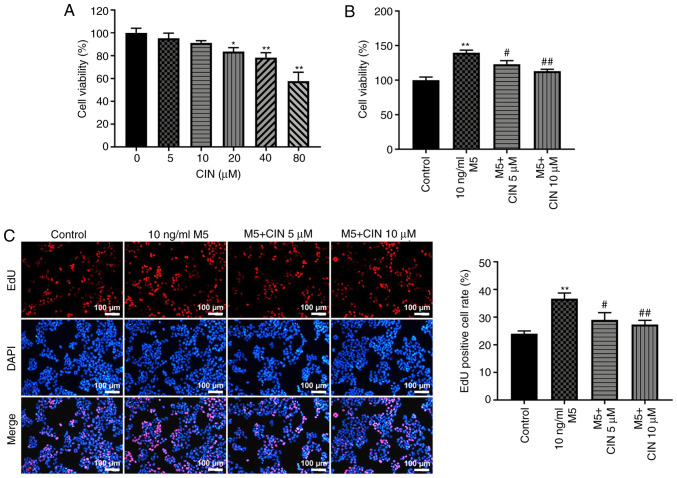
CIN treatment inhibits the proliferation of M5-stimulated NHEKs
To determine the cytotoxic effect of CIN on NHEKs, a CCK-8 assay was used. As shown in Fig. 1A, treatment with 5 or 10 µM CIN exerted a small effect on the viability of NHEKs. However, treatment with 20, 40 or 80 µM CIN significantly reduced the viability of NHEKs. Therefore, 5 and 10 µM CIN were selected as the optimal drug doses for use in subsequent experiments. The effects of CIN on the viability and proliferation of M5-treated NHEKs were subsequently investigated. The results of the CCK-8 and EdU staining assays indicated that M5 significantly increased the viability and proliferation of NHEKs, respectively, compared with the control group; however, these effects were reversed by CIN treatment (Fig. 1B and C). These data suggested that CIN may inhibit the viability and proliferation of M5-stimulated NHEKs.
Figure 1.
CIN inhibits the proliferation of M5-stimulated NHEKs. (A) CIN was dissolved with DMSO. NHEKs were treated with CIN (0, 5, 10, 20, 40 or 80 µM) for 24 h. Cell viability was detected by CCK-8 assay. (B) NHEKs were treated with CIN (5 or 10 µM) for 24 h, and then treated with 10 ng/ml M5 for 24 h. Cell viability was detected by CCK-8 assay. (C) EdU staining assay was used to detect cell proliferation. (Scale bar, 100 µm). n=3. *P<0.05, **P<0.01 vs. control group; #P<0.05, ##P<0.01 vs. 10 ng/ml M5 group. CIN, cinnamaldehyde; NHEKs, normal human epidermal keratinocytes; CCK-8, Cell Counting Kit-8; Edu, 5-Ethynyl-2′-deoxyuridine.
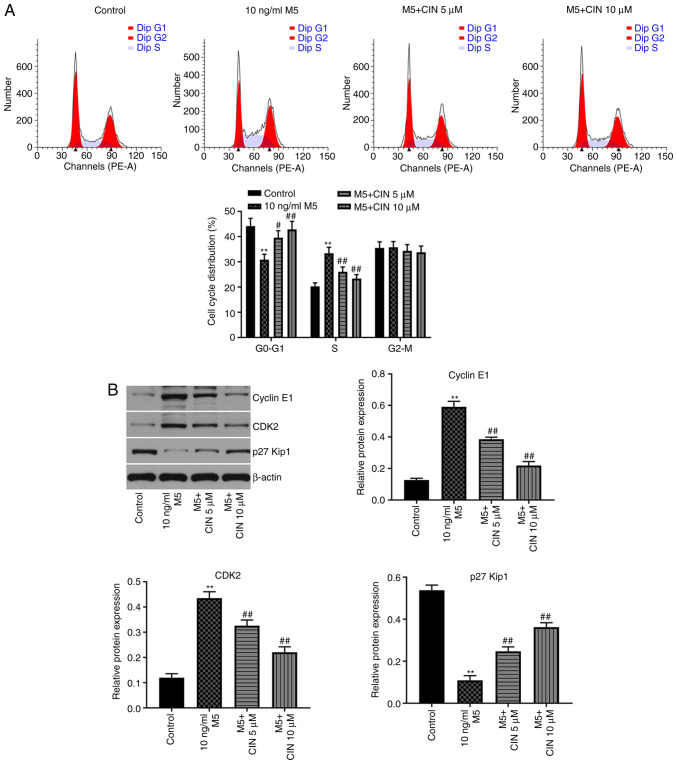
CIN induces cell cycle arrest in M5-stimulated NHEKs
Next, to evaluate the effect of CIN on the cell cycle distribution of M5-stimulated NHEKs, flow cytometry was used. As shown in Fig. 2A, the percentage of NHEKs in the G0/G1 phase was significantly decreased following stimulation with M5, while the number of cells in the S phase increased; however, these changes were reversed by CIN treatment. In addition, the results of the western blotting analysis revealed that M5 notably upregulated the levels of cyclin E1 and CDK2, and downregulated the expression of p27Kip1, compared with the control group (Fig. 2B). CIN significantly downregulated the expression levels of cyclin E1 and CDK2, and upregulated the expression levels of p27Kip1 in M5-stimulated NHEKs compared with the M5 treatment group (Fig. 2B). These results suggested that CIN may induce cell cycle arrest in M5-stimulated NHEKs.
Figure 2.
CIN induces cell cycle arrest in M5-stimulated NHEKs. NHEKs were treated with CIN (5 or 10 µM) for 24 h, and then treated with 10 ng/ml M5 for 24 h. (A) Flow cytometry was used to measure the cell cycle distribution in NHEKs. (B) Western blotting was used to detect the expression levels of cyclin E1, CDK2 and p27Kip1 in NHEKs. The relative expression levels of cyclin E1, CDK2 and p27Kip1 in NHEKs were normalized to β-actin. n=3. **P<0.01 vs. control group; #P<0.05, ##P<0.01 vs. 10 ng/ml M5 group. CIN, cinnamaldehyde; NHEKs, normal human epidermal keratinocytes; p27Kip1, CDK inhibitor 1B.
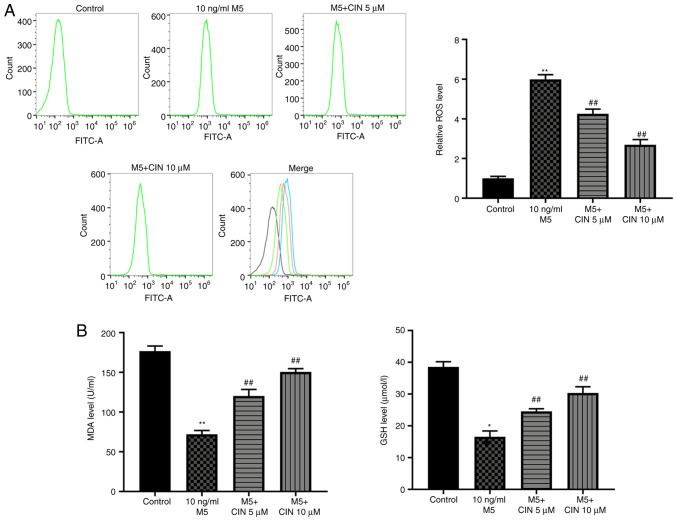
CIN attenuates M5-induced oxidative stress damage in NHEKs
To determine whether CIN could protect NHEKs against M5-induced oxidative stress, the production of intracellular ROS and the levels of MDA and GSH were detected. As shown in Fig. 3A and B, M5 stimulation significantly increased ROS production, and decreased the levels of MDA and GSH in NHEKs compared with the control group, suggesting that M5 may induce oxidative stress in NHEKs. However, treatment with CIN significantly reversed M5-induced oxidative stress in NHEKs (Fig. 3A and B). Taken together, these data indicated that CIN may attenuate M5-induced oxidative stress damage in NHEKs.
Figure 3.
CIN attenuates oxidative stress damage in M5-stimulated NHEKs. NHEKs were treated with CIN (5 or 10 µM) for 24 h, and then treated with 10 ng/ml M5 for 24 h. (A) Flow cytometry was used to measure the production of intracellular ROS in NHEKs. (B) ELISA was used to detect the levels of MDA and GSH in the supernatants of NHEKs. n=3. *P<0.05, **P<0.01 vs. control group; ##P<0.01 vs. 10 ng/ml M5 group. CIN, cinnamaldehyde; NHEKs, normal human epidermal keratinocytes; ROS, reactive oxygen species; MDA, malondialdehyde; GSH, glutathione.
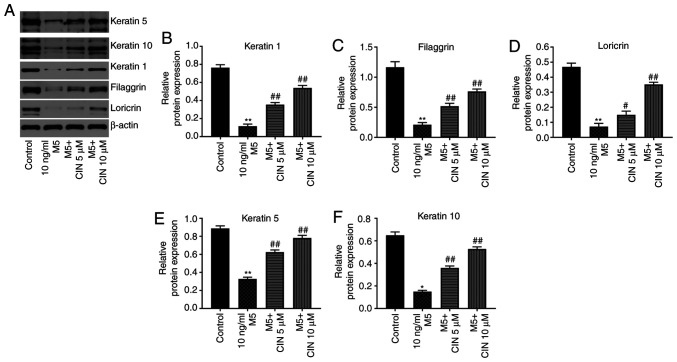
CIN promotes the differentiation of M5-stimulated NHEKs
Western blotting was performed to determine whether CIN could affect the differentiation of M5-treated NHEKs. Compared with the control group, M5 stimulation significantly downregulated the expression levels of keratin 1, filaggrin, loricrin, keratin 5 and keratin 10 in NHEKs; however, these changes were significantly reversed by CIN treatment (Fig. 4A-F). These data suggested that CIN may promote the differentiation of NHEKs following M5 exposure.
Figure 4.
CIN promotes the differentiation of M5-stimulated NHEKs. NHEKs were treated with CIN (5 or 10 µM) for 24 h, and then treated with 10 ng/ml M5 for 24 h. (A-F) Western blotting was used to detect the expression levels of keratin 5, keratin 10, keratin 1, filaggrin and loricrin in NHEKs. The relative expression levels of keratin 1, filaggrin, loricrin, keratin 5 and keratin 10 in NHEKs were normalized to β-actin. n=3. *P<0.05, **P<0.01 vs. control group; #P<0.05, ##P<0.01 vs. 10 ng/ml M5 group. CIN, cinnamaldehyde; NHEKs, normal human epidermal keratinocytes.
CIN attenuates inflammatory injury in M5-stimulated NHEKs
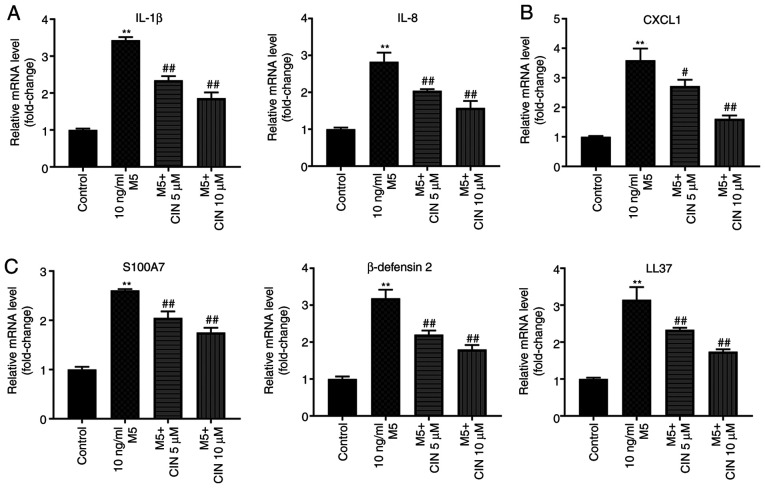
It was previously reported that M5 promoted the production of inflammatory mediators, such as cytokines (IL-1β), chemokines (IL-8 and CXCL1) and antimicrobial peptide pairs (S100A7, β-defensin 2 and LL-37) in keratinocytes (6). To validate whether CIN could attenuate the inflammatory response in NHEKs following M5 exposure, RT-qPCR was performed. As shown in Fig. 5A-C, M5 stimulation significantly upregulated the expression levels of IL-1β, IL-8, CXCL1, S100A7, β-defensin 2 and LL-37 in NHEKs compared with the control group; however, these M5-induced changes were significantly decreased following CIN treatment. These findings suggested that CIN may attenuate inflammatory injury in M5-stimulated NHEKs.
Figure 5.
CIN attenuates inflammatory injury in M5-stimulated NHEKs. NHEKs were treated with CIN (5 or 10 µM) for 24 h, and then treated with 10 ng/ml M5 for 24 h. Reverse transcription-quantitative PCR was performed to measure the expression levels of (A) IL-1β, IL-8, (B) CXCL1, (C) S100A7, β-defensin2 and LL-37 in NHEKs. n=3. **P<0.01 vs. control group; #P<0.05, ##P<0.01 vs. 10 ng/ml M5 group. CIN, cinnamaldehyde; NHEKs, normal human epidermal keratinocytes; CXCL1, C-X-C motif chemokine ligand 1; S100A7, S100 calcium binding protein A7; LL-37, cathelicidin antimicrobial peptide.
CIN inhibits the proliferation and associated inflammation of M5-treated NHEKs by downregulating the NF-κB and JNK signaling pathways
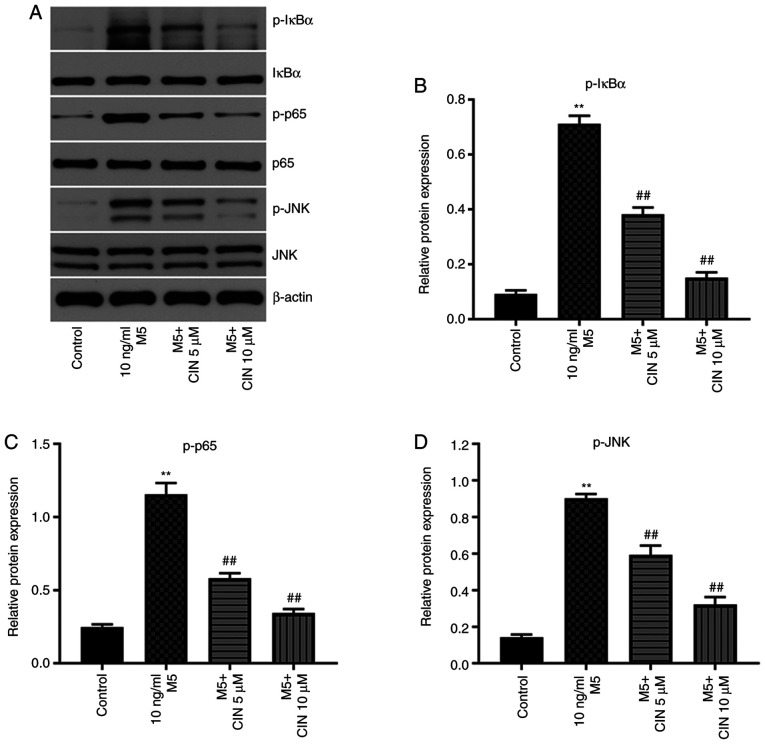
Previous studies have shown that the NF-κB and JNK signaling pathways play important roles in psoriasis (25,26). Thus, western blotting was used to analyze the expression levels of p-IκBα, p-p65 and p-JNK in NHEKs. M5 stimulation significantly upregulated the expression levels of p-IκBα, p-p65 and p-JNK in NHEKs compared with the control group; however, these M5-induced changes were significantly reversed by CIN treatment (Fig. 6A-D). These results indicated that CIN may inhibit the proliferation and associated inflammation of M5-treated NHEKs by downregulating the NF-κB and JNK signaling pathways.
Figure 6.
CIN inhibits the proliferation and inflammation of M5-treated NHEKs via downregulation of NF-κB and JNK signaling pathways. NHEKs were treated with CIN (5 or 10 µM) for 24 h, and then treated with 10 ng/ml M5 for 24 h. (A-D) Western blotting was performed to measure the expression levels of p-IκBα, p-p65 and p-JNK in NHEKs. The relative expression levels of p-IκBα, p-p65 and p-JNK in NHEKs were normalized to IκBα, p65 and JNK. n=3. **P<0.01 vs. control group; ##P<0.01 vs. 10 ng/ml M5 group. CIN, cinnamaldehyde; NHEKs, normal human epidermal keratinocytes; p-, phosphorylated; IκBα, inhibitor of NF-κB.
Discussion
Psoriasis is a common, chronic, relapsing inflammatory skin disease (27–29). Numerous previous studies have indicated that certain Traditional Chinese medicines may exert promising therapeutic effects in psoriasis (30). For instance, Xu et al (12) reported that salidroside inhibited the inflammatory response and hyperproliferation of keratinocytes. In addition, Liu et al (31) found that cimifugin could inhibit oxidative stress and the inflammation of TNF-α-stimulated keratinocytes. He et al (32) demonstrated that triptolide inhibited the proliferation and cell cycle distribution of IL-22-stimulated HaCaT cells. In addition, Wu et al (33) reported that diosgenin inhibited the proliferation of HaCaT cells following LPS/IL-22 exposure via inducing cell cycle arrest. However, to the best of our knowledge, the role of CIN in the progression of psoriasis remains unclear. The results of the present study found that CIN could inhibit the proliferation and associated inflammatory response of M5-stimualted NHEKs, in addition to inducing cell cycle arrest, which was consistent with the findings of previous studies (30–33).
Accumulating evidence has shown that the NF-κB and JNK signaling pathways play an important role in the pathogenesis of psoriasis (25,26). For example. Yang et al (25) demonstrated that Datura metel attenuated imiquimod-induced psoriasis-like dermatitis via inhibition of the NF-κB signaling pathway. Liu et al (31) showed that cimifugin inhibited oxidative stress and inflammation in TNF-α-treated keratinocytes by inactivating the NF-κB and JNK signaling pathways. Consistent with these previous findings, the present study confirmed that CIN could inhibit the phosphorylation of IκBα and p65 in M5-stimulated NHEKs, indicating that CIN may inhibit the NF-κB signaling pathway in M5-stimulated NHEKs. In addition, JNK has been suggested to represent a potential target for the treatment of psoriasis (34). In addition, Hammouda et al (34) reported that JNK promoted the occurrence of psoriasis. Consistent with these findings, the current results revealed that M5 markedly upregulated the expression levels of p-JNK in NHEKs. However, treatment with CIN significantly downregulated the expression levels of p-JNK in M5-stimulated NHEKs. Taken together with the aforementioned findings, these data indicated that CIN may inhibit the proliferation and associated inflammatory response of NHEKs following M5 exposure via inhibition of the NF-κB and JNK signaling pathways.
It has been reported that keratinocyte proliferation is inhibited and differentiation is promoted during the treatment of psoriasis (35). In the present study, CIN markedly upregulated the expression levels of keratin 1, filaggrin, loricrin, keratin 5 and keratin 10 in NHEKs. Thus, these findings indicated that CIN promoted keratinocyte differentiation.
However, there are some limitations of the current study. Additional in-depth and detailed studies that further research the underlying mechanisms by which CIN regulates the NF-κB and JNK signaling pathways are required. In addition, in order to further verify the relationship between the NF-κB and JNK signaling pathways and psoriasis, NF-κB and JNK signaling inhibitors will be used in future studies to obtain more complete information. Thus, further experiments are needed in the future.
In conclusion, the findings of the present study suggested that CIN may inhibit the proliferation and associated inflammation of M5-stimulated NHEKs via inhibition of the NF-κB and JNK signaling pathways. Therefore, CIN may represent a potential agent for the treatment of psoriasis.
Acknowledgements
Not applicable.
Funding Statement
The present study was supported by Chinese National Natural Science Foundation (grant no. 81703105).
Funding
The present study was supported by Chinese National Natural Science Foundation (grant no. 81703105).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Authors' contributions
ZD made major contributions to the conception, design and manuscript drafting of this study. JL, HQ and LW were responsible for data acquisition, analysis and interpretation, as well as manuscript revision. ML made substantial contributions to the conception and design of this study, and revised the manuscript critically for important intellectual content. All authors agreed to be accountable for all aspects of the work. All authors have read and approved the final manuscript. All authors confirm the authenticity of all the raw data.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Schleicher SM. Psoriasis: Pathogenesis, assessment, and therapeutic update. Clin Podiatr Med Surg. 2016;33:355–366. doi: 10.1016/j.cpm.2016.02.004. [DOI] [PubMed] [Google Scholar]
- 2.Nakajima K, Sano S. Mouse models of psoriasis and their relevance. J Dermatol. 2018;45:252–263. doi: 10.1111/1346-8138.14112. [DOI] [PubMed] [Google Scholar]
- 3.Lee J, Song K, Hiebert P, Werner S, Kim TG, Kim YS. Tussilagonone ameliorates psoriatic features in keratinocytes and imiquimod-induced psoriasis-like lesions in mice via NRF2 activation. J Invest Dermatol. 2020;140:1223–1232.e4. doi: 10.1016/j.jid.2019.12.008. [DOI] [PubMed] [Google Scholar]
- 4.Girolomoni G, Strohal R, Puig L, Bachelez H, Barker J, Boehncke WH, Prinz JC. The role of IL-23 and the IL-23/T(H) 17 immune axis in the pathogenesis and treatment of psoriasis. J Eur Acad Dermatol Venereol. 2017;31:1616–1626. doi: 10.1111/jdv.14433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Madden SK, Flanagan KL, Jones G. How lifestyle factors and their associated pathogenetic mechanisms impact psoriasis. Clin Nutr. 2020;39:1026–1040. doi: 10.1016/j.clnu.2019.05.006. [DOI] [PubMed] [Google Scholar]
- 6.Rabeony H, Petit-Paris I, Garnier J, Barrault C, Pedretti N, Guilloteau K, Jegou JF, Guillet G, Huguier V, Lecron JC, et al. Inhibition of keratinocyte differentiation by the synergistic effect of IL-17A, IL-22, IL-1α, TNFα and oncostatin M. PLoS One. 2014;9:e101937. doi: 10.1371/journal.pone.0101937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Niyonsaba F, Ushio H, Nakano N, Ng W, Sayama K, Hashimoto K, Nagaoka I, Okumura K, Ogawa H. Antimicrobial peptides human beta-defensins stimulate epidermal keratinocyte migration, proliferation and production of proinflammatory cytokines and chemokines. J Invest Dermatol. 2007;127:594–604. doi: 10.1038/sj.jid.5700599. [DOI] [PubMed] [Google Scholar]
- 8.Zhang D, Wang Y, Xia Y, Huo J, Zhang Y, Yang P, Zhang Y, Wang X. Repression of miR-142-3p alleviates psoriasis-like inflammation by repressing proliferation and promoting apoptosis of keratinocytes via targeting Sema3A. Mol Cell Probes. 2020;52:101573. doi: 10.1016/j.mcp.2020.101573. [DOI] [PubMed] [Google Scholar]
- 9.Nguyen LTH, Ahn SH, Nguyen UT, Yang IJ. Dang-Gui-Liu-Huang Tang a traditional herbal formula, ameliorates imiquimod-induced psoriasis-like skin inflammation in mice by inhibiting IL-22 production. Phytomedicine. 2018;47:48–57. doi: 10.1016/j.phymed.2018.04.051. [DOI] [PubMed] [Google Scholar]
- 10.Hoesel B, Schmid JA. The complexity of NF-κB signaling in inflammation and cancer. Mol Cancer. 2013;12:86. doi: 10.1186/1476-4598-12-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Matulewicz N, Karczewska-Kupczewska M. Insulin resistance and chronic inflammation. Postepy Hig Med Dosw (Online) 2016;70:1245–1258. [PubMed] [Google Scholar]
- 12.Xu F, Xu J, Xiong X, Deng Y. Salidroside inhibits MAPK, NF-κB, and STAT3 pathways in psoriasis-associated oxidative stress via SIRT1 activation. Redox Rep. 2019;24:70–74. doi: 10.1080/13510002.2019.1658377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sun L, Liu LN, Li JC, Lv YZ, Zong SB, Zhou J, Wang ZZ, Kou JP, Xiao W. The essential oil from the twigs of Cinnamomum cassia Presl inhibits oxytocin-induced uterine contraction in vitro and in vivo. J Ethnopharmacol. 2017;206:107–114. doi: 10.1016/j.jep.2017.05.023. [DOI] [PubMed] [Google Scholar]
- 14.Klibanov AM, Giannousis PP. Geometric specificity of alcohol dehydrogenases and its potential for separation of trans and cis isomers of unsaturated aldehydes. Proc Natl Acad Sci USA. 1982;79:3462–3465. doi: 10.1073/pnas.79.11.3462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kim ME, Na JY, Lee JS. Anti-inflammatory effects of trans-cinnamaldehyde on lipopolysaccharide-stimulated macrophage activation via MAPKs pathway regulation. Immunopharmacol Immunotoxicol. 2018;40:219–224. doi: 10.1080/08923973.2018.1424902. [DOI] [PubMed] [Google Scholar]
- 16.Yan L, Song F, Li H, Li Y, Li J, He QY, Zhang D, Wang F, Zhang M, Zhao H, et al. Submicron emulsion of cinnamaldehyde ameliorates bleomycin-induced idiopathic pulmonary fibrosis via inhibition of inflammation, oxidative stress and epithelial-mesenchymal transition. Biomed Pharmacother. 2018;102:765–771. doi: 10.1016/j.biopha.2018.03.145. [DOI] [PubMed] [Google Scholar]
- 17.Ye F, Zhang J, Zhang Q, Zhang J, Chen C. Preliminary study on the mechanism of long noncoding RNA SENCR regulating the proliferation and migration of vascular smooth muscle cells. J Cell Physiol. 2020;235:9635–9643. doi: 10.1002/jcp.29775. [DOI] [PubMed] [Google Scholar]
- 18.Zhu J, Liu B, Wang Z, Wang D, Ni H, Zhang L, Wang Y. Exosomes from nicotine-stimulated macrophages accelerate atherosclerosis through miR-21-3p/PTEN-mediated VSMC migration and proliferation. Theranostics. 2019;9:6901–6919. doi: 10.7150/thno.37357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Farahzadi R, Fathi E, Vietor I. Mesenchymal stem cells could be considered as a candidate for further studies in cell-based therapy of alzheimer's disease via targeting the signaling pathways. ACS Chem Neurosci. 2020;11:1424–1435. doi: 10.1021/acschemneuro.0c00052. [DOI] [PubMed] [Google Scholar]
- 20.Fathi E, Vietor I. Mesenchymal stem cells promote caspase expression in Molt-4 leukemia cells via GSK-3α/Β and ERK1/2 signaling pathways as a therapeutic strategy. Curr Gene Ther. 2021;21:81–88. doi: 10.2174/1566523220666201005111126. [DOI] [PubMed] [Google Scholar]
- 21.Fathi E, Farahzadi R, Javanmardi S, Vietor I. L-carnitine extends the telomere length of the cardiac differentiated CD117+−expressing stem cells. Tissue Cell. 2020;67:101429. doi: 10.1016/j.tice.2020.101429. [DOI] [PubMed] [Google Scholar]
- 22.Wang J, Li XM, Bai Z, Chi BX, Wei Y, Chen X. Curcumol induces cell cycle arrest in colon cancer cells via reactive oxygen species and Akt/GSK3β/cyclin D1 pathway. J Ethnopharmacol. 2018;210:1–9. doi: 10.1016/j.jep.2017.06.037. [DOI] [PubMed] [Google Scholar]
- 23.Fathi E, Valipour B, Sanaat Z, Nozad Charoudeh H, Farahzadi R. Interleukin-6, −8, and TGF-β secreted from mesenchymal stem cells show functional role in reduction of telomerase activity of leukemia cell Via Wnt5a/β-catenin and p53 pathways. Adv Pharm Bull. 2020;10:307–314. doi: 10.34172/apb.2020.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 25.Yang BY, Cheng YG, Liu Y, Liu Y, Tan JY, Guan W, Guo S, Kuang HX. Datura Metel L. Ameliorates imiquimod-induced psoriasis-like dermatitis and inhibits inflammatory cytokines production through TLR7/8-MyD88-NF-κB-NLRP3 Inflammasome Pathway. Molecules. 2019;24:2157. doi: 10.3390/molecules24112157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hotamisligil GS, Davis RJ. Cell signaling and stress responses. Cold Spring Harb Perspect Biol. 2016;8:a006072. doi: 10.1101/cshperspect.a006072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Deng Y, Chang C, Lu Q. The inflammatory response in psoriasis: A comprehensive review. Clin Rev Allergy Immunol. 2016;50:377–389. doi: 10.1007/s12016-016-8535-x. [DOI] [PubMed] [Google Scholar]
- 28.Gooderham MJ, Papp KA, Lynde CW. Shifting the focus - the primary role of IL-23 in psoriasis and other inflammatory disorders. J Eur Acad Dermatol Venereol. 2018;32:1111–1119. doi: 10.1111/jdv.14868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Oliveira Mde F, Rocha Bde O, Duarte GV. Psoriasis: Classical and emerging comorbidities. An Bras Dermatol. 2015;90:9–20. doi: 10.1590/abd1806-4841.20153038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lin W, Yu Q, Qin Y, Dai L, Xiao J, Jiao L, Liu S, Ye S, Zhang J, Chen M. To explore the clinical efficacy of Traditional Chinese Medicine bath in the treatment of psoriasis vulgaris with blood-heat syndrome and its effect on related cytokines based on different temperature and different concentration. Medicine. 2020;99:e20172. doi: 10.1097/MD.0000000000020172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu A, Zhao W, Zhang B, Tu Y, Wang Q, Li J. Cimifugin ameliorates imiquimod-induced psoriasis by inhibiting oxidative stress and inflammation via NF-κB/MAPK pathway. Biosci Rep. 2020;40:BSR20200471. doi: 10.1042/BSR20200471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.He Q, Zhang B, Hu F, Long J, Shi Q, Pi X, Chen H, Li J. Triptolide inhibits the proliferation of HaCaT cells induced by IL22 via upregulating miR-181b-5p. Drug Des Devel Ther. 2020;14:2927–2935. doi: 10.2147/DDDT.S254466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wu S, Zhao M, Sun Y, Xie M, Le K, Xu M, Huang C. The potential of Diosgenin in treating psoriasis: Studies from HaCaT keratinocytes and imiquimod-induced murine model. Life Sci. 2020;241:117115. doi: 10.1016/j.lfs.2019.117115. [DOI] [PubMed] [Google Scholar]
- 34.Hammouda MB, Ford AE, Liu Y, Zhang JY. The JNK signaling pathway in inflammatory Skin disorders and cancer. Cells. 2020;9:857. doi: 10.3390/cells9040857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wang C, Zong J, Li Y, Wang X, Du W, Li L. miR-744-3p regulates keratinocyte proliferation and differentiation via targeting KLLN in psoriasis. Exp Dermatol. 2019;28:283–291. doi: 10.1111/exd.13888. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.