Abstract
Mesenchymal stromal cell (MSC)-derived exosomes play a promising role in regenerative medicine. Their trophic and immunomodulatory potential has made them a promising candidate for cardiac regeneration and repair. Numerous studies have demonstrated that MSC-derived exosomes can replicate the anti-inflammatory, anti-apoptotic, and pro-angiogenic and anti-fibrotic effects of their parent cells and are considered a substitute for cell-based therapies. In addition, their lower tumorigenic risk, superior immune tolerance, and superior stability compared with their parent stem cells make them an attractive option in regenerative medicine. The therapeutic effects of MSC-derived exosomes have consequently been evaluated for application in cardiac regeneration and repair. In this review, we summarize the potential mechanisms and therapeutic effects of MSC-derived exosomes in cardiac regeneration and repair and provide evidence to support their clinical application.
Key words: mesenchymal stromal cell, exosome, myocardial infarction, cardiac regeneration and repair
Graphical abstract
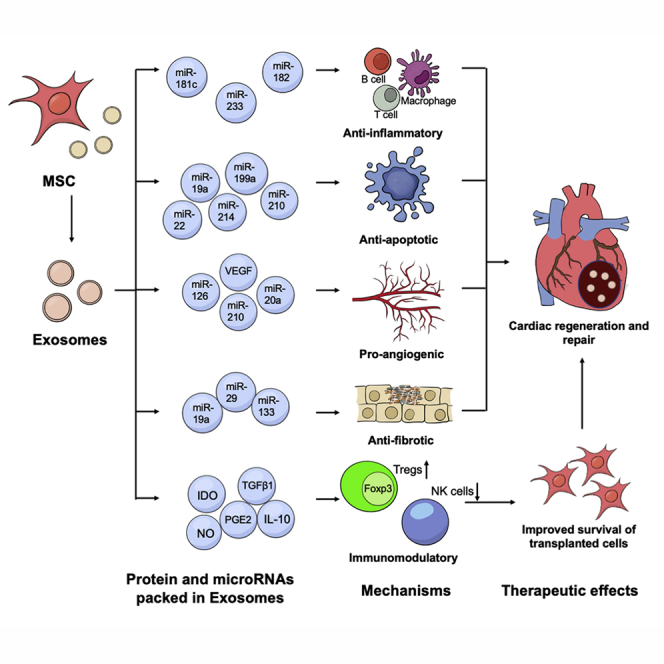
In this article Liao, Tse and colleagues summarize the mechanisms that underlie the therapeutic efficacy of MSC-derived exosomes in cardiac regeneration and repair and their potential role in enhancing the therapeutic efficacy of cell-based therapy. Moreover, they discuss the potential approaches to improve the production and therapeutic effects of MSC-derived exosomes.
Main text
Background
Cardiovascular diseases, in particular coronary heart disease (CHD), remain the leading cause of morbidity and mortality worldwide (Virani et al., 2020). Among all CHDs, acute myocardial infarction (MI) is a major cause of death. Consequent complications such as heart failure contribute to a significant medical, social, and financial burden. A broad spectrum of therapeutic reperfusion strategies is available, such as thrombolytic therapy or primary percutaneous coronary intervention, as well as anti-remodeling medications such as angiotensin-converting enzyme drugs and β-blockers. Nonetheless there is no effective pharmacological intervention that prevents cardiomyocyte death due to myocardial ischemia/reperfusion (I/R) injury (Heusch and Gersh, 2017). This I/R injury may also contribute to cardiac fibrosis, myocardial remodeling, cardiac arrhythmia, and, eventually, heart failure (Frank et al., 2012). At the end stage, the only available therapy for heart failure is heart transplantation or permanent left ventricular (LV) support. There is therefore tremendous interest in and need for novel therapies for post-MI LV remodeling and dysfunction.
In the last few years great advances have been made in cell-based therapies. The ultimate aim of such therapies is to generate sufficient functional cardiomyocytes to compensate for those lost following MI, either with exogenous cells or by activating endogenous regeneration and repair mechanisms (Menasche, 2018). Mesenchymal stem cells (MSCs) are multipotent stem cells that reside in various organs and can be derived from multiple tissues or cells including bone marrow (BM) stroma, adipose tissue, muscle, skin, fallopian tissue, umbilical cord blood, menstrual blood, and even induced pluripotent stem cells (iPSCs) (Caplan, 1991; Jackson et al., 2010; Lian et al., 2010). Their strong paracrine effects make MSCs a promising candidate for endogenous regeneration and repair pathways. There is evidence that intravenous, intracoronary, or intramyocardial administration of MSCs can improve LV function in an MI model (Bagno et al., 2018; Hu et al., 2016; Liao et al., 2019). Moreover, our previous study showed that pre-transplantation systemic intravenous administration of MSCs improved retention and therapeutic efficacy of intramyocardially transplanted exogenous cells (Sun et al., 2021).
Accumulating evidence shows that MSC-derived exosomes can ameliorate cardiac function and improve cardiac regeneration and repair (de Abreu et al., 2020). In this review, we discuss the therapeutic effects of MSC-derived exosomes in cardiac regeneration and repair following MI and describe the potential mechanisms underlying the benefits of MSC-exosome-based therapies. Additionally, we describe potential approaches to improve the efficacy and production of MSC-derived exosomes.
The advantages of MSC-derived exosomes over their parent MSCs
Exosomes are extracellular vesicles with a diameter of 30–150 nm. Previous studies have shown that they are secreted by endosomes, stored in the multivesicular endosomes (MVEs), and released through exocytosis (Doyle and Wang, 2019). The destiny of exosomes is either release into the extracellular space through fusion of MVEs with the cell plasma membrane or degradation by lysosomes along with MVEs (Doyle and Wang, 2019). The exosome transfers messages of the target cells through three possible pathways (Figure 1). First, the membrane receptors as well as protein or microRNA (miRNA) composition can be transferred to recipient cells by membrane fusion. Second, there are several protein ligands such as heat-shock proteins in the exosome membrane that can activate recipient cells through receptors in recipient cells. Third, the composition of exosomes such as protein and miRNAs can be transferred to recipient cells by endocytosis (Loyer et al., 2014). Exosomes have been considered one of the main mediators that regulate cell-cell communication. Barile et al. (2012) observed that exosomes secreted by MSCs were absorbed by cardiomyocytes. This ultrastructural evidence indicated that there is indeed an MSC-cardiomyocyte communication modulated by exosomes and that MSCs exerted their cardioprotective effect partly through releasing exosomes. Administration of exosomes derived from MSCs has been shown recently to recapitulate the cardioprotective effect of MSCs (de Abreu et al., 2020). There are several advantages of using exosomes rather than cell-based therapies. First, the risk of tumorigenicity in MSC-based therapies has been observed in previous studies (Barkholt et al., 2013), whereas no tumor formation has been observed in exosome-based therapies. Second, inflammatory cytokine interferon-γ (IFN-γ) can increase expression of HLA and immune rejection of transplanted MSCs (Sun et al., 2015), whereas no such effects have been reported following transplantation of MSC-derived exosomes. Third, MSC-derived exosomes are more stable to store compared with MSCs (Lener et al., 2015). Fourth, the production cost of MSC-derived exosomes is lower than that of their parent cells. Finally, as the particle size of exosomes is much smaller than that of their parent cells, exosomes could travel across capillaries without plugging. Therefore, MSC-derived exosomes are more efficacious than their parent cells after intravenous administration. In summary, mounting evidence demonstrates that exosomes not only recapitulate the beneficial effects of their parent cells but also overcome some of the limitations of parent cells (Table 1).
Figure 1.
Pathways for exosomes to target toward and fuse with the receiving cells
First, membrane fusion: activated receptors as well as the composition of exosomes can be transferred to the target cells by membrane fusion. Second, binding receptors: exosomes can activate target cells through binding their protein ligands to the receptors in recipient cells. Third, endocytosis: when exosomes are taken up by the target cells, the protein or miRNA composition that they carry can be released into target cells. MSC, mesenchymal stromal cell.
Table 1.
The advantages of MSC-derived exosomes over their parent cells
| MSC | MSC-derived exosomes | |
|---|---|---|
| Risk of tumorigenicity | low | no |
| Immune rejection | low | no |
| Stability | low | high |
| Production cost | high | low |
| Therapeutic efficacy after systemic delivery | low | high |
MSC, mesenchymal stromal cell.
The potential role of MSC-derived exosomes in cardiac regeneration and repair
Use of exosomes for cardiac repair is still at an early stage, and clinical studies of MSC-derived exosomes in the treatment of cardiovascular disease are ongoing. Table 2 summarizes the findings of pre-clinical studies (Arslan et al., 2013; Bian et al., 2014; Feng et al., 2014; He et al., 2018; Ju et al., 2018; Liu et al., 2020; Luther et al., 2018; Lv et al., 2019; Ma et al., 2017, 2018; Mao et al., 2019; Ni et al., 2019; Shao et al., 2017; Wang et al., 2017, 2018a; Xiao et al., 2018; Xu et al., 2020; Yu et al., 2013; Zhao et al., 2019) on the use of MSC-derived exosomes in cardiac regeneration and repair. Application of exosomes derived from BM-MSCs (Bian et al., 2014; Feng et al., 2014; He et al., 2018; Liu et al., 2020; Luther et al., 2018; Lv et al., 2019; Ma et al., 2018; Shao et al., 2017; Wang et al., 2017, 2018a; Xiao et al., 2018; Xu et al., 2020; Yu et al., 2013; Zhao et al., 2019), embryonic stem cell (ESC)-MSCs (Arslan et al., 2013), umbilical cord (UC)-MSCs (Ma et al., 2017; Ni et al., 2019; Xu et al., 2020), adipose (AD)-MSCs (Xu et al., 2020), and cardiac MSCs (Ju et al., 2018) resulted in significantly improved cardiac function in a pre-clinical model of MI. Among all these exosomes, BM-MSC-derived exosomes and extracellular vesicle size from 30 to 180 nm were most widely used for cardiac repair and regeneration. Intramyocardial and intravenous transplantation were the two most commonly used modes of delivery. For studies in mice, 20–50 μg of MSC-derived exosomes is used for intravenous administration while 1–600 μg is used for intramyocardial administration. For studies in rats, 40–400 μg of MSC-derived exosomes is used for intravenous administration while 20–80 μg is used for intramyocardial administration. In most of these studies, exosomes were administered immediately after induction of MI. Other studies demonstrated that when administration occurred 30 min, 48 h, and even 1 week after MI, cardiac function was still improved. Moreover, to define the therapeutic role of MSC-derived exosomes, the following issues needed to be clarified: (1) the source of their parent cells; (2) methods of isolation and particle size distribution; and (3) the components of exosomes. Here, we illustrate the mechanisms that underlie the therapeutic efficacy of MSC-derived exosomes in cardiac regeneration and repair and their potential role in enhancing the therapeutic efficacy of cell-based therapy.
Table 2.
Pre-clinical studies of MSC-derived exosomes for cardiac regeneration and repair
| No. | Authors | Year | Model | Cell source | Cell type | Size of EV | Administration | Time | Dose | Effect |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Arslan et al. | 2013 | myocardial I/R injury (mouse) | human | ESC-MSC | – | intracoronary | 5 min prior to reperfusion to 3 h after | 0.4 μg/mL | reduced MI size, decreased LV dilation, increased cardiac function, decreased ATP loss |
| 2 | Yu et al. | 2013 | myocardial I/R injury (rat) | rat | BM-MSC | average: 100 nm | intramyocardial | immediately | derived from 4 × 106 MSC | reduced CM apoptosis, improved cardiac function |
| 3 | Bian et al. | 2014 | MI (rat) | human | BM-MSC | 47–180 nm | intramyocardial | 30 min after MI | 80 μg | improved cardiac function, decreased MI size, promoted angiogenesis |
| 4 | Feng et al. | 2014 | MI (mouse) | mouse | BM-MSC | average: 57.4 nm | intramyocardial | immediately | 1 μg | decreased MI size, decreased apoptosis |
| 5 | Ma et al. | 2017 | MI (rat) | human | UC-MSC | average: 96 nm | intravenous | immediately | 400 μg | improved cardiac function, decreased apoptosis, increased angiogenesis |
| 6 | Shao et al. | 2017 | MI (rat) | rat | BM-MSC | – | intramyocardial | immediately | 20 μg | improved cardiac function, reduced MI size, decreased inflammation |
| 7 | Wang et al. | 2017 | MI (mouse) | mouse | BM-MSC | – | intravenous | immediately | – | improved angiogenesis and cardiac function |
| 8 | He et al. | 2018 | MI (mouse) | mouse | BM-MSC | – | intravenous | 48 h after MI | 20 μg | improved cardiac function, decreased apoptosis, increased angiogenesis |
| 9 | Ju et al. | 2018 | MI (mouse) | mouse | C-MSC | average: 120 nm | intramyocardial | immediately | 50 μg | improved cardiac function, increased angiogenesis |
| 10 | Luther et al. | 2018 | MI (mouse) | mouse | BM-MSC | – | pericardial sac | immediately | 12.5 μg | decreased apoptosis |
| 11 | Wang et al. | 2018 | MI (mouse) | mouse | BM-MSC | 30–150 nm | intravenous | immediately | 50 μg | improved cardiac function, increased angiogenesis, decreased MI size, decreased inflammation |
| 12 | Xiao et al. | 2018 | MI (mouse) | human | BM-MSC | – | intramyocardial | 30 min after MI | 5 μg | improved cardiac function, decreased apoptosis |
| 13 | Xu et al. | 2018 | MI (rat) | human | BM-MSC, AD-MSC, UC-MSC | BM-MSC: 40–100 nm; AD-MSC: 30–100 nm; UC-MSC: 10–90 nm | intramyocardial | 30 min after MI | derived from 1.5 × 106 MSC | improved cardiac function, decreased MI size, decreased apoptosis and inflammation |
| 14 | Ma et al. | 2018 | MI (mouse) | mouse | BM-MSC | <150 nm | intramyocardial | 1 week after MI | 600 μg | improved cardiac function, increased angiogenesis |
| 15 | Mao et al. | 2019 | MI (rat) | human | MSC | 30–150 nm | intravenous | immediately | 40 μg | decreased MI size, decreased apoptosis and inflammation |
| 16 | Ni et al. | 2019 | MI (rat) | human | UC-MSC | 40–90 nm | intramyocardial | immediately | 50 μg | improved cardiac function, decreased MI size, reduced apoptosis, increased angiogenesis |
| 17 | Lv et al. | 2019 | MI (rat) | rat | BM-MSC | average: 90 nm | intramyocardial | 30 min after MI | 80 μg | improved cardiac function, reduced MI size, decreased apoptosis and inflammation, increased angiogenesis |
| 18 | Zhao et al. | 2019 | myocardial I/R injury (mouse) | mouse | BM-MSC | 50–150 nm | intramyocardial | immediately | 50 μg | decreased MI size and inflammation |
| 19 | Liu et al. | 2020 | MI (rat) | human | BM-MSC | average: 50 nm | intramyocardial | immediately | 30 μg | improved cardiac function, decreased apoptosis |
MSC, mesenchymal stromal cell; EV, extracellular vesicle; MI, myocardial infarction; I/R, ischemia/reperfusion; ESC, embryonic stem cell; BM, bone marrow; UC, umbilical cord; AD, adipose; LV, left ventricle; ATP, adenosine triphosphate; CM, cardiomyocyte.
Anti-inflammatory effects
Previous studies showed that MSCs have multiple anti-inflammatory effects that include regulating the polarization of macrophages, inhibiting the activation of effector T cells, and suppressing the secretion of B cells (Corcione et al., 2006; Di Nicola et al., 2002; Le Blanc and Mougiakakos, 2012). Recent studies show that MSC-derived exosomes replicate these anti-inflammatory effects of their parent cells. Both intramyocardial (Lv et al., 2019; Shao et al., 2017; Xu et al., 2020; Zhao et al., 2019) and intravenous (Mao et al., 2019; Wang et al., 2018a) transplantation of MSC-derived exosomes can decrease pro-inflammatory immune cell infiltration in the infarcted heart. Sun et al. (2018) demonstrated that MSC-derived exosomes switched pro-inflammatory macrophages to anti-inflammatory macrophages. Collino et al. (2015) showed that miRNAs play a key role in exosome-based therapies. This anti-inflammatory effect may also be mediated by exosomal miRNAs, evidenced by the promotion of switching of M1 macrophages to M2 macrophages in the peri-infarcted area by miR-182-enriched MSC-derived exosomes (Zhao et al., 2019). Moreover, miR-233-enriched MSC-derived exosomes, which were taken up by macrophages, downregulated SEMA3A and STAT3 and reduced the inflammatory response in macrophages (Wang et al., 2015). In addition, MSC-derived exosomes regulated effector T cells and B cells, inhibited T cell proliferation, and induced activated effective T cell apoptosis (Mokarizadeh et al., 2012). miR-181c has been regarded as a key miRNA in this exosome-mediated T cell regulation; miR-181c-enriched MSC-derived exosomes were shown to blunt the Toll-like receptor 4 (TLR4) signaling pathway-mediated release of pro-inflammatory factors (Li et al., 2016). A recent study also showed that MSC-derived exosomes fully reproduced the immunosuppressive effects of MSCs on B cell proliferation and immunoglobulin production by mediating the secretion of immunoglobulins (Budoni et al., 2013). Although the precise mechanism of these anti-inflammatory effects remains unclear, current clinical trials show that intravenous administration of MSC-derived exosomes is a safe and effective treatment for immune disease (Kordelas et al., 2014).
Anti-apoptotic effects
Previous studies showed that intramyocardial (Feng et al., 2014; Liu et al., 2020; Lv et al., 2019; Ni et al., 2019; Shao et al., 2017; Wang et al., 2017; Xiao et al., 2018; Xu et al., 2020; Yu et al., 2013), intravenous (He et al., 2018; Ma et al., 2017; Mao et al., 2019; Wang et al., 2018a), intracoronary (Arslan et al., 2013) or intrapericardial sac (Luther et al., 2018) transplantation of MSC-derived exosomes could ameliorate apoptosis of endogenous cardiomyocytes. The infiltrated inflammatory cells released numerous reactive oxygen species that led to programmed cell death in the peri-infarcted area. MSC-derived exosomes may reduce oxidative stress and improve the anti-apoptotic ability of cardiomyocytes in the peri-infarcted area. A recent study showed that administration of MSC-derived exosomes in an I/R mouse model increased the ATP level, reduced oxidative stress, and improved cardiomyocyte survival (Arslan et al., 2013). Previous studies showed that miRNAs including miR-19a (He et al., 2018), miR-22 (Feng et al., 2014), miR-199a (Zhu et al., 2019), and miR-214 (Wang et al., 2018b) play key roles in these exosome-mediated anti-apoptotic effects. Exosomes derived from GATA4-overexpressed MSCs exerted anti-apoptotic and cardioprotective effects by delivering anti-apoptotic miRNAs, including miR-19a (He et al., 2018). These anti-apoptotic effects were abolished by inhibition of miR-19a (He et al., 2018). Researchers attributed the anti-apoptotic effects to downregulation of phosphatase and tensin homolog (PTEN) (Chen et al., 2013). Nonetheless, the clinical safety of miR-19a-enriched exosomes is still under debate. Although there are limited data on the arrhythmogenic effect of miR-19a-enriched exosomes, overexpression of miR-19a may induce arrhythmias, since miR-19a aberrantly inhibits connexin-43 expression (Danielson et al., 2013). Feng et al. (2014) showed that after co-culture with cardiomyocytes, MSCs transferred miR-22 into cardiomyocytes through exosome release. Intramyocardial administration of miR-22-enriched MSC-derived exosomes was shown to decrease cardiomyocyte apoptosis in the ischemic area and reduce infarct size (Feng et al., 2014). In a kidney ischemia/perfusion model, miR-199a-enriched MSC-derived exosomes decreased apoptosis in renal cells by modulating Sema3A and activating Akt and ERK pathways (Zhu et al., 2019). miR-199a is one of the key miRNAs that blocked negative regulators of cardiomyocyte proliferation in a neonatal rat model (Eulalio et al., 2012). Indeed, overexpression of miR-199a exerted cardioprotective effects and promoted cardiac regeneration and repair after MI (Eulalio et al., 2012). Exosomes derived from miR-214-enriched MSCs facilitated cardiac stem cell survival via reduction of reactive oxygen species production. On the contrary, depletion of miR-214 by miR-214 inhibitor or miR-214 mimics impaired the anti-apoptotic effects of these miR-214-enriched exosomes (Wang et al., 2018b). miR-214 exerted its anti-apoptotic effects mainly through regulation of sodium/calcium exchanger 1, cyclophilin D, Bcl-2 like protein 11, and PTEN (Aurora et al., 2012; Lv et al., 2014; Wang et al., 2018b). Nonetheless, miR-214 may induce dilated cardiomyocytes in the long term (Lv et al., 2014). This side effect may limit the clinical application of miR-214-enriched exosomes. Although no report is available on miR-210-enriched exosomes, they may be also involved in the exosome-mediated anti-apoptosis effect. miR-210 promoted endogenous cardiac regeneration through upregulation of β-catenin and Bcl-2 and downregulation of adenomatous polyposis coli, p16, and caspase-3 (Arif et al., 2017).
Pro-angiogenic effects
Previous research showed that intravenous (He et al., 2018; Ma et al., 2017; Wang et al., 2017, 2018a) and intramyocardial (Bian et al., 2014; Ju et al., 2018; Lv et al., 2019; Ma et al., 2018; Ni et al., 2019) administration can improve angiogenesis during cardiac regeneration and repair. Our study showed that iPSC-MSCs ameliorated limb ischemia through both vascular differentiation and paracrine secretion, such as vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (Lian et al., 2010). Recently, Hu et al. (2015) reported that intramuscular administration of exosomes derived from iPSC-MSCs partly replicated the pro-angiogenic effects of their parent cells. In their in vitro study, iPSC-MSC-derived exosomes promoted human umbilical vein endothelial cell migration, proliferation, tube formation, and angiogenesis-related gene and molecule expression (Hu et al., 2015). miRNAs and proteins packaged in the MSC-derived exosomes may mainly account for these angiogenic effects. Endothelial cells treated with MSC-derived exosomes demonstrated an increased ability for proliferation, migration, and tube formation. Intravenous administration of these exosomes significantly increased cardiac function and angiogenesis in a rat model of MI (Ma et al., 2017). Researchers concluded that the pro-angiogenic effects were due to increased expression of platelet-derived growth factor D (PDGF-D). As well as PDGF-D, several miRNAs are involved in MSC-derived exosome-mediated pro-angiogenic effects. Evidence suggests that miR-126- and VEGF-enriched MSC-derived exosomes promote pro-angiogenic mRNA expression in ischemic tissues, while the pro-angiogenic effects were weakened in exosomes derived from miR-126 knockdown MSCs in the treated group (Du et al., 2017). Wang et al. (2017) showed that MSC-derived exosomes promoted angiogenesis in the mouse model of MI through delivery of miR-210. Exosome-derived MSCs with miR-210 silence demonstrated a significantly decreased pro-angiogenic effect both in vitro and in vivo (Wang et al., 2017). Interestingly, preconditioning MSCs with ischemic stroke serum significantly increased miR-20a expression (Kim et al., 2016). Nonetheless, the effects of miR-20a on neovascularization are still under debate. Inhibition of miR-20a has been suggested to exert pro-angiogenic or anti-angiogenic effects (Doebele et al., 2010; Pin et al., 2012; Suárez et al., 2008).
Anti-fibrotic effects
Previous research showed that intravenous (Wang et al., 2018a) and intramyocardial (Bian et al., 2014; Lv et al., 2019; Ni et al., 2019; Shao et al., 2017) transplantation of MSC-derived exosomes could ameliorate cardiac fibrosis during cardiac regeneration and repair. Cardiac regeneration and repair and deposition of extracellular collagen are two parallel processes during the reparative phase. The balance between scar formation and cardiac regeneration is crucial for cardiac repair. Scar formation provides mechanical strength and circumvents ventricular rupture, and is beneficial for cardiac repair in the short term. Nonetheless it is inversely correlated with cardiac regeneration in the long term. Therefore, anti-fibrotic therapies are optimized to target the reactive fibrosis that occurs during ventricular remodeling. These are supported by a previous study wherein systemic inhibition of fibrosis formation signals during the inflammatory phase following MI exacerbated cardiac impairment, while inhibition of systemic fibrosis ameliorated cardiac remodeling and dysfunction in the post-MI reparative phase (Ikeuchi et al., 2004). The miRNA in the MSC-derived exosomes ameliorated collagen deposition during cardiac remodeling. Apart from anti-apoptotic effects, miR-19a regulated several extracellular matrix proteins, including thrombospondin-1 and connective tissue growth factor (CTGF) (Wen et al., 2012). Whether miR-19a-enriched exosomes regulated post-MI fibrosis requires further exploration. A previous study showed that miR-29 was expressed in BM-MSCs (Jin et al., 2016). miR-29 is a well-defined miRNA that regulates cardiac fibrosis. Lower miR-29 expression leads to more extracellular matrix deposit, culminating in cardiac fibrosis (van Rooij et al., 2008). Although no direct evidence supports the anti-fibrotic effect of miR-29, a pharmacological study showed that MI rats treated with carvedilol had reduced myocardial fibrosis that was miR-29 dependent (Zhu et al., 2013). Interestingly, swim training ameliorated the downregulated expression of miR-29 and decreased the MI-induced fibrotic gene expression (Melo et al., 2014). These studies highlight the pivotal role of miR-29 in cardiac fibrosis development following MI. A recent study also showed that miR-133-overexpressed MSCs reduced infarct size following MI (Chen et al., 2017). The anti-fibrotic effects of miR-133 may be due to regulation of the collagen α1 chain and CTGF (Castoldi et al., 2012; Duisters et al., 2009).
Taken together, exosomes carrying different contents may elicit different therapeutic responses. Exosomes expressing abundant miR-182, miR-233, and miR-181c elicit beneficial effects mainly through anti-inflammatory responses; exosomes expressing abundant miR-19a, miR-22, miR-199a, miR-214, and miR-210 modulate the survival of cardiomyocytes in the ischemic region; exosomes expressing abundant miR-126, miR-210, and miR-20a improve cardiac regeneration and repair by promoting angiogenesis; and exosomes expressing abundant miR-19a, miR-29, and miR-133 inhibit fibrosis during cardiac regeneration and repair. Post-MI regeneration and repair can be divided into two phases: a pro-inflammatory phase, which features pro-inflammatory cytokine release, inflammatory cell infiltration, and phagocytosis of necrotic cell debris; and a reparative phase, characterized by resolution of inflammation and formation of collagen-based scar (Cahill et al., 2017). Therefore, anti-inflammatory and anti-apoptotic exosomes may benefit cardiac regeneration during the pro-inflammatory phase, whereas pro-angiogenetic and anti-fibrotic exosomes promote vascularization and cardiac regeneration during the reparative phase. The miRNAs and downstream pathways relevant to the anti-inflammatory, anti-apoptotic, pro-angiogenic, and anti-fibrotic effects are summarized in Table 3.
Table 3.
The miRNAs and underlying pathways for MSC-derived exosome-mediated cardiac regeneration
| Function | Route of administration | miRNA | Pathway | Target cell | Reference |
|---|---|---|---|---|---|
| Anti-inflammatory | intramyocardial, intravenous | miR-182 | TLR4 signal | macrophage | Zhao et al., 2019 |
| miR-233 | SEMA3A; STAT3 | macrophage | Wang et al., 2015 | ||
| miR-181c | TLR4 signal | T cell | Mokarizadeh et al., 2012 | ||
| Anti-apoptotic | intramyocardial, intravenous, intracoronary, intrapericardial | miR-19a | PTEN | cardiomyocyte | He et al., 2018 |
| miR-22 | MeCP2 | Feng et al., 2014 | |||
| miR-199a | Sema3A | Zhu et al., 2019 | |||
| miR-214 | sodium/calcium exchanger 1; cyclophilin D; Bcl-2 like protein 11; PTEN | Wang et al., 2018b | |||
| miR-210 | β-catenin; Bcl-2; adenomatous polyposis coli; p16; caspase-3 | Arif et al., 2017 | |||
| Pro-angiogenic | intramyocardial, intravenous | miR-126 | Spred-1 | endothelial cell | Du et al., 2017 |
| miR-210 | Efna3 | Wang et al., 2017 | |||
| miR-20a | p38 MAP kinase | Pin et al., 2012 | |||
| Anti-fibrotic | intramyocardial, intravenous | miR-19a | thrombopondin-1; CTGF | fibroblast | Wen et al., 2012 |
| miR-29 | fibrotic gene | Melo et al., 2014 | |||
| miR-133 | collagen α1 chain; CTGF | Chen et al., 2017 |
MSC, mesenchymal stromal cell; TLR4, Toll-like receptor 4; PTEN, phosphatase and tensin homolog; CTGF, connective tissue growth factor.
Potential role in enhancing therapeutic efficacy of cell-based therapy
Recently, our group has investigated the immunomodulatory effect of human iPSC (hiPSC)-MSCs on improving the survival and therapeutic efficacy of cells following intramyocardial transplantation into a mouse model of MI (Sun et al., 2021). Our results showed that pre-transplantation systemic administration of hiPSC-MSCs increased systemic regulatory T cell (Treg) activation, decreased the number of splenic natural killer (NK) cells and inflammation, enhanced survival of transplanted hiPSC-MSCs and hiPSC-cardiomyocytes following intramyocardial transplantation, and improved the therapeutic efficacy at 4 weeks post MI. Previous studies showed that MSC-derived exosomes could upregulate CD4+ T cell differentiation into Tregs and downregulate the activation of NK cells (Kordelas et al., 2014; Zhang et al., 2014). The immunomodulatory effects of MSCs were orchestrated by immunosuppressive factors including indoleamine 2,3-dioxygenase, transforming growth factor β1, prostaglandin E2, nitric oxide, and interleukin-10 (English et al., 2009; Maggini et al., 2010; Nauta et al., 2006; Ren et al., 2012; van Koppen et al., 2012). Recently, researchers proposed that these immunosuppressive factors were packed in the extracellular vesicles, especially exosomes (Lener et al., 2015). Indeed, subcutaneous administration of MSC-derived exosomes improved the survival of transplanted immortal human keratinocytes in a rat skin wound model (Zhang et al., 2015). As a result, if hiPSC-MSC-derived exosomes can recapitulate the immunomodulatory effect of their parent cells, pre-transplantation systemic administration of hiPSC-MSC-derived exosomes is a promising strategy by which to improve the cell retention of cell-based therapy because of their advantages of low tumorigenicity and superior immune tolerance and stability. Isolation of immunomodulatory exosomes from MSCs may provide a promising means by which to combine cell-based therapies and exosome-based therapies for cardiac regeneration and repair.
Approaches to improve the production and therapeutic effects of MSC-derived exosomes
Although MSC-derived exosomes represent a promising candidate for cardiac regeneration and repair, their low-level production from routine culture conditions limits their therapeutic efficacy. Different culture conditions may result in altered production of MSC-derived exosomes and even different components in the released exosomes. For example, culture of MSCs with stroke serum, obtained from the blood of mice with middle cerebral artery occlusion, demonstrated remarkably increased miR-20a expression in their released exosomes when compared with MSCs cultured with fetal bovine serum or normal serum (Kim et al., 2016). It is evident that culture conditions will considerably affect the constituents of its relevant exosomes as well as their production. It is of paramount importance to define the appropriate culture conditions to improve the production of exosomes without compromising their therapeutic effect. Apart from culture medium, cell type also influences the efficacy of exosome-based therapies, since exosomes from different microenvironments may have a different focus on a certain cell type. For example, extracellular vesicles derived from the nervous system are preferentially absorbed by cells in the nervous system (Pegtel et al., 2014). Whether co-culturing of MSCs with cardiomyocytes can improve the efficacy of MSC-derived exosomes remains to be revealed.
As mentioned above, the destiny of exosomes is either degradation by lysosomes or release into the extracellular space. Therefore, increased production of MSC-derived exosomes can be realized by decreasing degradation and increasing exosome secretion. For example, inhibition of P2X7 receptors, soluble NSF attachment protein receptors, tumor suppressor-activated pathway 6, rhomboid domain-containing protein 1, or phospholipase D2 compromises exosome release. Overexpressing these mediators may enhance exosome extracellular secretion and prevent exosome degradation (Phan et al., 2018). Regulating the key mediators in the exosome degradation or secretion pathways may promote effective MSC-derived exosome secretion, culminating in increased exosome production.
Genetic modification of their parent cells is another efficient means to improve the therapeutic effect of exosomes. The therapeutic efficacy of MSC-derived exosomes can be improved by overexpression or knockdown of certain RNAs or proteins. Several DNA or RNA sequences have been regarded as cardioprotective or pro-angiogenic. For example, modifying MSCs by transfecting them with a recombinant adenovirus carrying the Akt gene sequence could increase the expression of PDGF-D in its related exosomes (Ma et al., 2017). These genetically modified MSCs and their related exosomes have an increased ability to promote neovascularization, cardiac regeneration, and cardiac repair. Both DNA sequences and RNA sequences could be regarded as modified target and non-coding RNAs that were increased in the exosomes derived from genetically modified MSCs, and have been considered to play a pivotal role in cardiac regeneration and repair following MI.
Although there is no consensus on the optimum delivery route for MSC-derived exosomes, intramyocardial transplantation, especially transendocardial transplantation, may be the most efficacious (Collantes et al., 2017; Kanelidis et al., 2017). Nonetheless, previous studies showed that more than 90% of cells were lost within the first few days following transplantation because a large percentage of cells spilled out from the myocardium when they were delivered via direct intramyocardial injection. The same problem exists for administration of exosome-based therapy. An alternative approach is to use cardiac tissue engineering to produce a scaffold and then seed exosomes onto the patch to optimize retention and engraftment.
Limitations of MSC-derived exosomes
Although MSC-derived exosomes are a promising substitute for MSCs in regenerative medicine, there are several considerations that should be addressed prior to their clinical application. First, exosomes are currently isolated according to their vesicle size. If different vesicle sizes reflect different components in exosomes, more accurate isolation methods need to be adopted, since different-size-defined exosomes may have variable therapeutic efficacy for cardiac regeneration and repair. Second, exosomes are stored in MVEs before release into intercellular substance. Extracting MSC-derived exosomes from MVEs before they are released may help to isolate purified exosomes, as MSC-derived exosomes isolated from culture media and blood may be contaminated by exosomes from other cell types. Third, a well-defined method for single-exosome analysis is currently unavailable. Investigating cargoes from a single exosome rather than the whole exosome population would provide a better understanding of the exact mechanisms underlying exosome-based therapies. Finally, it is pertinent to define the appropriate microenvironment in which to generate therapeutic exosomes for cardiac regeneration and repair. Recent evidence demonstrates that MSC-derived exosomes may contain different compounds and exert different therapeutic effects in different microenvironments. For example, MSCs may secrete immunomodulatory exosomes in the pro-inflammatory phase after MI and then pro-angiogenic exosomes in the reparative phase of cardiac regeneration and repair. Taken together, difficulty in detecting exosomes in vivo, dynamic secretion and uptake at the tissue level, and lack of an efficient purification route are all challenges for current exosome studies.
Perspective and conclusion
Mounting evidence suggests that exosomes derived from MSCs can be a potential therapy to promote cardiac regeneration and repair after MI. The therapeutic role of MSC-derived exosomes is extremely complex. Demonstrating the components in exosomes and their exact interaction with other cells in the infarcted heart may help researchers to understand their therapeutic effects and optimize the effects of exosome-based therapies. Exosome research is still in its infancy although a pre-clinical large animal study is currently ongoing. Studying the therapeutic efficacy of MSC-exosome-based therapy in this porcine model or other pre-clinical large animal model could provide further evidence to support clinical translation of MSC-exosome-based therapy. Moreover, our previous study showed that hiPSC-MSCs demonstrated superior therapeutic efficacy for cardiac repair and regeneration in comparison with BM-MSCs due to their increased survival after transplantation (Lian et al., 2010). Therefore, it is of great interest to investigate whether hiPSC-MSC-derived exosomes have therapeutic benefits for cardiac repair and regeneration that are superior to those of BM-MSC-derived exosomes. Our previous study also showed that pre-transplantation systemic intravenous administration of hiPSC-MSCs induced immunomodulation and facilitated the survival of intramyocardially transplanted cells to improve cardiac function in MI. Pre-transplantation systemic administration of MSC-derived exosomes may be a promising strategy to improve cell retention of cell-based therapy. In conclusion, studying exosomes provides insight into the exact mechanism of cardiac regeneration and repair, helps optimize cell-based therapies, and promotes the development of precision medicine.
Author contributions
S.-J.S.: Literature search, data collection, data analysis, paper writing. R.W.: Data interpretation, figures. F.L.: Literature search. S.-Y.L.: Study design, data analysis, data interpretation, paper revising. H.-F.T.: Study design, paper revising.
Contributor Information
Song-Yan Liao, Email: lsy923@hku.hk.
Hung-Fat Tse, Email: hftse@hku.hk.
References
- Arif M., Pandey R., Alam P., Jiang S., Arghya P., Ahmed R.P. MicroRNA-210-mediated proliferation, survival, and angiogenesis promote cardiac repair post myocardial infarction in rodents. J. Mol. Med. 2017;95:1369–1385. doi: 10.1007/s00109-017-1591-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arslan F., Lai R.C., Smeets M.B., Akeroyd L., Choo A., Aguor E.N.E., Timmers L., van Rijen H.V., Doevendans P.A., Pasterkamp G. Mesenchymal stem cell-derived exosomes increase ATP levels, decrease oxidative stress and activate PI3K/Akt pathway to enhance myocardial viability and prevent adverse remodeling after myocardial ischemia/reperfusion injury. Stem Cell Res. 2013;10:301–312. doi: 10.1016/j.scr.2013.01.002. [DOI] [PubMed] [Google Scholar]
- Aurora A.B., Mahmoud A.I., Luo X., Johnson B.A., van Rooij E., Matsuzaki S., Humphries K.M., Hill J.A., Bassel-Duby R., Sadek H.A. MicroRNA-214 protects the mouse heart from ischemic injury by controlling Ca2+ overload and cell death. J. Clin. Invest. 2012;122:1222. doi: 10.1172/JCI59327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bagno L., Hatzistergos K.E., Balkan W., Hare J.M. Mesenchymal stem cell-based therapy for cardiovascular disease: progress and challenges. Mol. Ther. 2018;26:1610–1623. doi: 10.1016/j.ymthe.2018.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barile L., Gherghiceanu M., Popescu L.M., Moccetti T., Vassalli G. Ultrastructural evidence of exosome secretion by progenitor cells in adult mouse myocardium and adult human cardiospheres. J. Biomed. Biotechnol. 2012;2012:354605. doi: 10.1155/2012/354605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barkholt L., Flory E., Jekerle V., Lucas-Samuel S., Ahnert P., Bisset L., Buscher D., Fibbe W., Foussat A., Kwa M. Risk of tumorigenicity in mesenchymal stromal cell-based therapies—bridging scientific observations and regulatory viewpoints. Cytotherapy. 2013;15:753–759. doi: 10.1016/j.jcyt.2013.03.005. [DOI] [PubMed] [Google Scholar]
- Bian S., Zhang L., Duan L., Wang X., Min Y., Yu H. Extracellular vesicles derived from human bone marrow mesenchymal stem cells promote angiogenesis in a rat myocardial infarction model. J. Mol. Med. 2014;92:387–397. doi: 10.1007/s00109-013-1110-5. [DOI] [PubMed] [Google Scholar]
- Budoni M., Fierabracci A., Luciano R., Petrini S., Di Ciommo V., Muraca M. The immunosuppressive effect of mesenchymal stromal cells on B lymphocytes is mediated by membrane vesicles. Cell Transplant. 2013;22:369–379. doi: 10.3727/096368911X582769. [DOI] [PubMed] [Google Scholar]
- Cahill T.J., Choudhury R.P., Riley P.R. Heart regeneration and repair after myocardial infarction: translational opportunities for novel therapeutics. Nat. Rev. Drug Discov. 2017;16:699–717. doi: 10.1038/nrd.2017.106. [DOI] [PubMed] [Google Scholar]
- Caplan A.I. Mesenchymal stem cells. J. Orthop. Res. 1991;9:641–650. doi: 10.1002/jor.1100090504. [DOI] [PubMed] [Google Scholar]
- Castoldi G., Di Gioia C.R.T., Bombardi C., Catalucci D., Corradi B., Gualazzi M.G., Leopizzi M., Mancini M., Zerbini G., Condorelli G. MiR-133a regulates collagen 1A1: potential role of miR-133a in myocardial fibrosis in angiotensin II-dependent hypertension. J. Cell Physiol. 2012;227:850–856. doi: 10.1002/jcp.22939. [DOI] [PubMed] [Google Scholar]
- Chen Y., Zhao Y., Chen W., Xie L., Yang J., Chen Y., Wei L., Shen Z. MicroRNA-133 overexpression promotes the therapeutic efficacy of mesenchymal stem cells on acute myocardial infarction. Stem Cell Res. Ther. 2017;8:268. doi: 10.1186/s13287-017-0722-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y.J., Huang T.Z.-P., Seok T.H., Ding T.J., Kataoka T.M., Zhang T.Z., Hu T.X., Wang T.G., Lin T.Z., Wang T.S. mir-17-92 cluster is required for and sufficient to induce cardiomyocyte proliferation in postnatal and adult hearts. Circ. Res. 2013;112:1557–1566. doi: 10.1161/CIRCRESAHA.112.300658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collantes M., Pelacho B., Garcia-Velloso M., Gavira J., Abizanda G., Palacios I., Rodriguez-Borlado L., Alvarez V., Prieto E., Ecay M. Non-invasive in vivo imaging of cardiac stem/progenitor cell biodistribution and retention after intracoronary and intramyocardial delivery in a swine model of chronic ischemia reperfusion injury. J. Transl. Med. 2017;15:56. doi: 10.1186/s12967-017-1157-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collino F., Bruno S., Incarnato D., Dettori D., Neri F., Provero P., Pomatto M., Oliviero S., Tetta C., Quesenberry P.J. AKI recovery induced by mesenchymal stromal cell-derived extracellular vesicles carrying microRNAs. J. Am. Soc. Nephrol. 2015;26:2349–2360. doi: 10.1681/ASN.2014070710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corcione A., Benvenuto F., Ferretti E., Giunti D., Cappiello V., Cazzanti F., Risso M., Gualandi F., Mancardi G.L., Pistoia V. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006;107:367–372. doi: 10.1182/blood-2005-07-2657. [DOI] [PubMed] [Google Scholar]
- Danielson L., Park D., Rotllan N., Chamorro-Jorganes A., Guijarro M., Fernandez-Hernando C., Fishman G.I., Phoon C., Hernando E. Cardiovascular dysregulation of miR-17-92 causes a lethal hypertrophic cardiomyopathy and arrhythmogenesis. FASEB J. 2013;27:1460–1467. doi: 10.1096/fj.12-221994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Abreu R.C., Fernandes H., da Costa Martins P.A., Sahoo S., Emanueli C., Ferreira L. Native and bioengineered extracellular vesicles for cardiovascular therapeutics. Nat. Rev. Cardiol. 2020;17:685–697. doi: 10.1038/s41569-020-0389-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Nicola M., Carlo-Stella C., Magni M., Milanesi M., Longoni P.D., Matteucci P., Grisanti S., Gianni A.M. Human bone marrow stromal cells suppress T-lymphocyte proliferation induced by cellular or nonspecific mitogenic stimuli. Blood. 2002;99:3838–3843. doi: 10.1182/blood.v99.10.3838. [DOI] [PubMed] [Google Scholar]
- Doebele C., Bonauer A., Fischer A., Scholz A., Reiss Y., Urbich C., Hofmann W.-K., Zeiher A.M., Dimmeler S. Members of the microRNA-17-92 cluster exhibit a cell-intrinsic antiangiogenic function in endothelial cells. Blood. 2010;115:4944. doi: 10.1182/blood-2010-01-264812. [DOI] [PubMed] [Google Scholar]
- Doyle L.M., Wang M.Z. Overview of extracellular vesicles, their origin, composition, purpose, and methods for exosome isolation and analysis. Cells. 2019;8:727. doi: 10.3390/cells8070727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du W., Zhang K., Zhang S., Wang R., Nie Y., Tao H., Han Z., Liang L., Wang D., Liu J. Enhanced proangiogenic potential of mesenchymal stem cell-derived exosomes stimulated by a nitric oxide releasing polymer. Biomaterials. 2017;133:70–81. doi: 10.1016/j.biomaterials.2017.04.030. [DOI] [PubMed] [Google Scholar]
- Duisters F.R., Tijsen J.A., Schroen J.B., Leenders E.J., Lentink W.V., Van Der Made G.I., Herias M.V., Van Leeuwen E.R., Schellings E.M., Barenbrug E.P. miR-133 and miR-30 regulate connective tissue growth factor: implications for a role of microRNAs in myocardial matrix remodeling. Circ. Res. 2009;104:170–178. doi: 10.1161/CIRCRESAHA.108.182535. [DOI] [PubMed] [Google Scholar]
- English K., Ryan J.M., Tobin L., Murphy M.J., Barry F.P., Mahon B.P. Cell contact, prostaglandin E2 and transforming growth factor beta 1 play non-redundant roles in human mesenchymal stem cell induction of CD4+ CD25 High forkhead box P3+ regulatory T cells. Clin. Exp. Immunol. 2009;156:149–160. doi: 10.1111/j.1365-2249.2009.03874.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eulalio A., Mano M., Dal Ferro M., Zentilin L., Sinagra G., Zacchigna S., Giacca M. Functional screening identifies miRNAs inducing cardiac regeneration. Nature. 2012;492:376. doi: 10.1038/nature11739. [DOI] [PubMed] [Google Scholar]
- Feng Y., Huang W., Wani M., Yu X., Ashraf M. Ischemic preconditioning potentiates the protective effect of stem cells through secretion of exosomes by targeting Mecp2 via miR-22. PLoS One. 2014;9:e88685. doi: 10.1371/journal.pone.0088685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frank A., Bonney M., Bonney S., Weitzel L., Koeppen M., Eckle T. Myocardial ischemia reperfusion injury: from basic science to clinical bedside. Semin. Cardiothorac. Vasc. Anesth. 2012;16:123–132. doi: 10.1177/1089253211436350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He J.-G., Li H.-R., Han J.-X., Li B.-B., Yan D., Li H.-Y., Wang P., Luo Y. GATA-4-expressing mouse bone marrow mesenchymal stem cells improve cardiac function after myocardial infarction via secreted exosomes. Sci. Rep. 2018;8:9047. doi: 10.1038/s41598-018-27435-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heusch G., Gersh B.J. The pathophysiology of acute myocardial infarction and strategies of protection beyond reperfusion: a continual challenge. Eur. Heart J. 2017;38:774–784. doi: 10.1093/eurheartj/ehw224. [DOI] [PubMed] [Google Scholar]
- Hu G.-W., Li Q., Niu X., Hu B., Liu J., Zhou S.-M., Guo S.-C., Lang H.-L., Zhang C.-Q., Wang Y. Exosomes secreted by human-induced pluripotent stem cell-derived mesenchymal stem cells attenuate limb ischemia by promoting angiogenesis in mice. Stem Cell Res. Ther. 2015;6:10. doi: 10.1186/scrt546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu X., Xu Y., Zhong Z., Wu Y., Zhao J., Wang Y., Cheng H., Kong M., Zhang F., Chen Q. A large-scale investigation of hypoxia-preconditioned allogeneic mesenchymal stem cells for myocardial repair in nonhuman primates: paracrine activity without remuscularization. Circ. Res. 2016;118:970–983. doi: 10.1161/CIRCRESAHA.115.307516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikeuchi M., Tsutsui H., Shiomi T., Matsusaka H., Matsushima S., Wen J., Kubota T., Takeshita A. Inhibition of TGF-beta signaling exacerbates early cardiac dysfunction but prevents late remodeling after infarction. Cardiovasc. Res. 2004;64:526–535. doi: 10.1016/j.cardiores.2004.07.017. [DOI] [PubMed] [Google Scholar]
- Jackson W.M., Nesti L.J., Tuan R.S. Potential therapeutic applications of muscle-derived mesenchymal stem and progenitor cells. Expert Opin. Biol. Ther. 2010;10:505–517. doi: 10.1517/14712591003610606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin M., Wu Y., Wang J., Ye W., Wang L., Yin P., Liu W., Pan C., Hua X. MicroRNA-29 facilitates transplantation of bone marrow-derived mesenchymal stem cells to alleviate pelvic floor dysfunction by repressing elastin. Stem Cell Res. Ther. 2016;7:167. doi: 10.1186/s13287-016-0428-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ju C., Shen Y., Ma G., Liu Y., Cai J., Kim I.-M., Weintraub N.L., Liu N., Tang Y. Transplantation of cardiac mesenchymal stem cell-derived exosomes promotes repair in ischemic myocardium. J. Cardiovasc. Trans. Res. 2018;11:420–428. doi: 10.1007/s12265-018-9822-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanelidis J.A., Premer M.C., Lopez M.J., Balkan M.W., Hare M.J. Route of delivery modulates the efficacy of mesenchymal stem cell therapy for myocardial infarction: a meta-analysis of preclinical studies and clinical trials. Circ. Res. 2017;120:1139–1150. doi: 10.1161/CIRCRESAHA.116.309819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim E.H., Kim D.H., Kim H.R., Kim S.Y., Kim H.H., Bang O.Y. Stroke serum priming modulates characteristics of mesenchymal stromal cells by controlling the expression miRNA-20a. Cell Transplant. 2016;25:1489–1499. doi: 10.3727/096368916X690430. [DOI] [PubMed] [Google Scholar]
- Kordelas L., Rebmann V., Ludwig A.K., Radtke S., Ruesing J., Doeppner T.R., Epple M., Horn P.A., Beelen D.W., Giebel B. MSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease. Leukemia. 2014;28:970–973. doi: 10.1038/leu.2014.41. [DOI] [PubMed] [Google Scholar]
- Le Blanc K., Mougiakakos D. Multipotent mesenchymal stromal cells and the innate immune system. Nat. Rev. Immunol. 2012;12:383–396. doi: 10.1038/nri3209. [DOI] [PubMed] [Google Scholar]
- Lener T., Gimona M., Aigner L., Borger V., Buzas E., Camussi G., Chaput N., Chatterjee D., Court F.A., Del Portillo H.A. Applying extracellular vesicles based therapeutics in clinical trials—an ISEV position paper. J. Extracell. Vesicles. 2015;4:30087. doi: 10.3402/jev.v4.30087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Liu L., Yang J., Yu Y., Chai J., Wang L., Ma L., Yin H. Exosome derived from human umbilical cord mesenchymal stem cell mediates MiR-181c attenuating burn-induced excessive inflammation. EBioMedicine. 2016;8:72–82. doi: 10.1016/j.ebiom.2016.04.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lian Q., Zhang Y., Zhang J., Zhang H.K., Wu X., Zhang Y., Lam F.F., Kang S., Xia J.C., Lai W.H. Functional mesenchymal stem cells derived from human induced pluripotent stem cells attenuate limb ischemia in mice. Circulation. 2010;121:1113–1123. doi: 10.1161/CIRCULATIONAHA.109.898312. [DOI] [PubMed] [Google Scholar]
- Liao S., Zhang Y., Ting S., Zhen Z., Luo F., Zhu Z., Jiang Y., Sun S., Lai W.-H., Lian Q. Potent immunomodulation and angiogenic effects of mesenchymal stem cells versus cardiomyocytes derived from pluripotent stem cells for treatment of heart failure. Stem Cell Res. Ther. 2019;10:78. doi: 10.1186/s13287-019-1183-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X., Li X., Zhu W., Zhang Y., Hong Y., Liang X., Fan B., Zhao H., He H., Zhang F. Exosomes from mesenchymal stem cells overexpressing MIF enhance myocardial repair. J. Cell Physiol. 2020;235:8010–8022. doi: 10.1002/jcp.29456. [DOI] [PubMed] [Google Scholar]
- Loyer X., Vion A.-C., Tedgui A., Boulanger C.M. Microvesicles as cell-cell messengers in cardiovascular diseases. Circ. Res. 2014;114:345–353. doi: 10.1161/CIRCRESAHA.113.300858. [DOI] [PubMed] [Google Scholar]
- Luther K.M., Haar L., McGuinness M., Wang Y., Lynch T.L., Phan A., Song Y., Shen Z., Gardner G., Kuffel G. Exosomal miR-21a-5p mediates cardioprotection by mesenchymal stem cells. J. Mol. Cell Cardiol. 2018;119:125–137. doi: 10.1016/j.yjmcc.2018.04.012. [DOI] [PubMed] [Google Scholar]
- Lv G., Shao S., Dong H., Bian X., Yang X., Dong S. MicroRNA-214 protects cardiac myocytes against H2O2-induced injury. J. Cell Biochem. 2014;115:93–101. doi: 10.1002/jcb.24636. [DOI] [PubMed] [Google Scholar]
- Lv K., Li Q., Zhang L., Wang Y., Zhong Z., Zhao J., Lin X., Wang J., Zhu K., Xiao C. Incorporation of small extracellular vesicles in sodium alginate hydrogel as a novel therapeutic strategy for myocardial infarction. Theranostics. 2019;9:7403–7416. doi: 10.7150/thno.32637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma J., Zhao Y., Sun L., Sun X., Zhao X., Sun X., Qian H., Xu W., Zhu W. Exosomes derived from Akt-modified human umbilical cord mesenchymal stem cells improve cardiac regeneration and promote angiogenesis via activating platelet-derived growth factor D. Stem Cells Transl. Med. 2017;6:51–59. doi: 10.5966/sctm.2016-0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma T., Chen Y., Chen Y., Meng Q., Sun J., Shao L., Yu Y., Huang H., Hu Y., Yang Z. MicroRNA-132, delivered by mesenchymal stem cell-derived exosomes, promote angiogenesis in myocardial infarction. Stem Cells Int. 2018;2018:3290372. doi: 10.1155/2018/3290372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maggini J., Mirkin G., Bognanni I., Holmberg J., Piazzón I.M., Nepomnaschy I., Costa H., Cañones C., Raiden S., Vermeulen M. Mouse bone marrow-derived mesenchymal stromal cells turn activated macrophages into a regulatory-like profile (macrophages and MSC) PLoS One. 2010;5:e9252. doi: 10.1371/journal.pone.0009252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mao Q., Liang X.-L., Zhang C.-L., Pang Y.-H., Lu Y.-X. LncRNA KLF3-AS1 in human mesenchymal stem cell-derived exosomes ameliorates pyroptosis of cardiomyocytes and myocardial infarction through miR-138-5p/Sirt1 axis. Stem Cell Res. Ther. 2019;10:393. doi: 10.1186/s13287-019-1522-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melo S.F.S., Fernandes T., Baraúna V.G., Matos K.C., Santos A.A.S., Tucci P.J.F., Oliveira E.M. Expression of MicroRNA-29 and collagen in cardiac muscle after swimming training in myocardial-infarcted rats. Cell Physiol. Biochem. 2014;33:657–669. doi: 10.1159/000358642. [DOI] [PubMed] [Google Scholar]
- Menasche P. Cell therapy trials for heart regeneration—lessons learned and future directions. Nat. Rev. Cardiol. 2018;15:659–671. doi: 10.1038/s41569-018-0013-0. [DOI] [PubMed] [Google Scholar]
- Mokarizadeh A., Delirezh N., Morshedi A., Mosayebi G., Farshid A.-A., Mardani K. Microvesicles derived from mesenchymal stem cells: potent organelles for induction of tolerogenic signaling. Immunol. Lett. 2012;147:47–54. doi: 10.1016/j.imlet.2012.06.001. [DOI] [PubMed] [Google Scholar]
- Nauta A., Kruisselbrink A., Lurvink E., Willemze R., Fibbe W. Mesenchymal stem cells inhibit generation and function of both CD34 super(+)-derived and monocyte-derived dendritic cells. J. Immunol. 2006;177:2080–2087. doi: 10.4049/jimmunol.177.4.2080. [DOI] [PubMed] [Google Scholar]
- Ni J., Liu X., Yin Y., Zhang P., Xu Y.-W., Liu Z. Exosomes derived from TIMP2-modified human umbilical cord mesenchymal stem cells enhance the repair effect in rat model with myocardial infarction possibly by the Akt/Sfrp2 pathway. Oxid. Med. Cell Longev. 2019;2019:1958941. doi: 10.1155/2019/1958941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pegtel D.M., Peferoen L., Amor S. Extracellular vesicles as modulators of cell-to-cell communication in the healthy and diseased brain. Philos. Trans. R. Soc. B Biol. Sci. 2014;369:20130516. doi: 10.1098/rstb.2013.0516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phan J., Kumar P., Hao D., Gao K., Farmer D., Wang A. Engineering mesenchymal stem cells to improve their exosome efficacy and yield for cell-free therapy. J. Extracell. Vesicles. 2018;7:1522236. doi: 10.1080/20013078.2018.1522236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pin A.-L., Houle F.o., Guillonneau M.v., Paquet E.R., Simard M.J., Huot J. miR-20a represses endothelial cell migration by targeting MKK3 and inhibiting p38 MAP kinase activation in response to VEGF. Angiogenesis. 2012;15:593–608. doi: 10.1007/s10456-012-9283-z. [DOI] [PubMed] [Google Scholar]
- Ren G., Chen X., Dong F., Li W., Ren X., Zhang Y., Shi Y. Concise review: mesenchymal stem cells and translational medicine: emerging issues. Stem Cells Transl. Med. 2012;1:51–58. doi: 10.5966/sctm.2011-0019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shao L., Zhang Y., Lan B., Wang J., Zhang Z., Zhang L., Xiao P., Meng Q., Geng Y.-j., Yu X.-y. MiRNA-sequence indicates that mesenchymal stem cells and exosomes have similar mechanism to enhance cardiac repair. Biomed. Res. Int. 2017;2017:4150705. doi: 10.1155/2017/4150705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suárez Y., Fernández-Hernando C., Yu J., Gerber S.A., Harrison K.D., Pober J.S., Iruela-Arispe M.L., Merkenschlager M., Sessa W.C. Dicer-dependent endothelial microRNAs are necessary for postnatal angiogenesis. Proc. Natl. Acad. Sci. U S A. 2008;105:14082. doi: 10.1073/pnas.0804597105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun S.-J., Lai W.-H., Jiang Y., Zhen Z., Wei R., Lian Q., Liao S.-Y., Tse H.-F. Immunomodulation by systemic administration of human induced pluripotent stem cell-derived mesenchymal stromal cells to enhance the therapeutic efficacy of cell-based therapy for treatment of myocardial infarction. Theranostics. 2021;11:1641–1654. doi: 10.7150/thno.46119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun X., Shan A., Wei Z., Xu B. Intravenous mesenchymal stem cell-derived exosomes ameliorate myocardial inflammation in the dilated cardiomyopathy. Biochem. Biophys. Res. Commun. 2018;503:2611–2618. doi: 10.1016/j.bbrc.2018.08.012. [DOI] [PubMed] [Google Scholar]
- Sun Y.Q., Zhang Y., Li X., Deng M.X., Gao W.X., Yao Y., Chiu S.M., Liang X., Gao F., Chan C.W. Insensitivity of human iPS cells-derived mesenchymal stem cells to interferon-gamma-induced HLA expression potentiates repair efficiency of hind limb ischemia in immune humanized NOD scid gamma mice. Stem Cells. 2015;33:3452–3467. doi: 10.1002/stem.2094. [DOI] [PubMed] [Google Scholar]
- van Koppen A., Joles J.A., van Balkom B.W.M., Lim S.K., de Kleijn D., Giles R.H., Verhaar M.C. Human embryonic mesenchymal stem cell-derived conditioned medium rescues kidney function in rats with established chronic kidney disease. PLoS One. 2012;7:e38746. doi: 10.1371/journal.pone.0038746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Rooij E., Sutherland L.B., Thatcher J.E., Dimaio J.M., Naseem R.H., Marshall W.S., Hill J.A., Olson E.N. Dysregulation of microRNAs after myocardial infarction reveals a role of miR-29 in cardiac fibrosis. Proc. Natl. Acad. Sci. U S A. 2008;105:13027–13032. doi: 10.1073/pnas.0805038105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Virani S.S., Alonso A., Benjamin E.J., Bittencourt M.S., Callaway C.W., Carson A.P., Chamberlain A.M., Chang A.R., Cheng S., Delling F.N. Heart disease and stroke statistics—2020 update: a report from the American Heart Association. Circulation. 2020;141:e139–e151. doi: 10.1161/CIR.0000000000000757. [DOI] [PubMed] [Google Scholar]
- Wang N., Chen C., Yang D., Liao Q., Luo H., Wang X., Zhou F., Yang X., Yang J., Zeng C. Mesenchymal stem cells-derived extracellular vesicles, via miR-210, improve infarcted cardiac function by promotion of angiogenesis. Biochim. Biophys. Acta Mol. Basis Dis. 2017;1863:2085–2092. doi: 10.1016/j.bbadis.2017.02.023. [DOI] [PubMed] [Google Scholar]
- Wang X., Chen Y., Zhao Z., Meng Q., Yu Y., Sun J., Yang Z., Chen Y., Li J., Ma T. Engineered exosomes with ischemic myocardium-targeting peptide for targeted therapy in myocardial infarction. J. Am. Heart Assoc. 2018;7:e008737. doi: 10.1161/JAHA.118.008737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X., Gu H., Qin D., Yang L., Huang W., Essandoh K., Wang Y., Caldwell C.C., Peng T., Zingarelli B. Exosomal miR-223 contributes to mesenchymal stem cell-elicited cardioprotection in polymicrobial sepsis. Sci. Rep. 2015;5:13721. doi: 10.1038/srep13721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Zhao R., Liu D., Deng W., Xu G., Liu W., Rong J., Long X., Ge J., Shi B. Exosomes derived from miR-214-enriched bone marrow-derived mesenchymal stem cells regulate oxidative damage in cardiac stem cells by targeting CaMKII. Oxid. Med. Cell Longev. 2018;2018:4971261. doi: 10.1155/2018/4971261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen Z., Zheng S., Zhou C., Yuan W., Wang J., Wang T. Bone marrow mesenchymal stem cells for post-myocardial infarction cardiac repair: microRNAs as novel regulators. J. Cell Mol. Med. 2012;16:657–671. doi: 10.1111/j.1582-4934.2011.01471.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao C., Wang K., Xu Y., Hu H., Zhang N., Wang Y., Zhong Z., Zhao J., Li Q., Zhu D. Transplanted mesenchymal stem cells reduce autophagic flux in infarcted hearts via the exosomal transfer of miR-125b. Circ. Res. 2018;123:564–578. doi: 10.1161/CIRCRESAHA.118.312758. [DOI] [PubMed] [Google Scholar]
- Xu H., Wang Z., Liu L., Zhang B., Li B. Exosomes derived from adipose tissue, bone marrow, and umbilical cord blood for cardioprotection after myocardial infarction. J. Cell Biochem. 2020;121:2089–2102. doi: 10.1002/jcb.27399. [DOI] [PubMed] [Google Scholar]
- Yu B., Gong M., Wang Y., Millard R., Pasha Z., Yang Y., Ashraf M., Xu M. Cardiomyocyte protection by GATA-4 gene engineered mesenchymal stem cells is partially mediated by translocation of miR-221 in microvesicles. PLoS One. 2013;8:e73304. doi: 10.1371/journal.pone.0073304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang B., Wang M., Gong A., Zhang X., Wu X., Zhu Y., Shi H., Wu L., Zhu W., Qian H. HucMSC-exosome mediated-Wnt4 signaling is required for cutaneous wound healing. Stem Cells. 2015;33:2158–2168. doi: 10.1002/stem.1771. [DOI] [PubMed] [Google Scholar]
- Zhang B., Yin Y., Lai R.C., Tan S.S., Choo A.B., Lim S.K. Mesenchymal stem cells secrete immunologically active exosomes. Stem Cells Dev. 2014;23:1233–1244. doi: 10.1089/scd.2013.0479. [DOI] [PubMed] [Google Scholar]
- Zhao J., Li X., Hu J., Chen F., Qiao S., Sun X., Gao L., Xie J., Xu B. Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc. Res. 2019;115:1205–1216. doi: 10.1093/cvr/cvz040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu G., Pei L., Lin F., Yin H., Li X., He W., Liu N., Gou X. Exosomes from human-bone-marrow-derived mesenchymal stem cells protect against renal ischemia/reperfusion injury via transferring miR-199a-3p. J. Cell Physiol. 2019;234:23736–23749. doi: 10.1002/jcp.28941. [DOI] [PubMed] [Google Scholar]
- Zhu J.-N., Chen R., Fu Y.-H., Lin Q.-X., Huang S., Guo L.-L., Zhang M.-Z., Deng C.-Y., Zou X., Zhong S.-L. Smad3 inactivation and MiR-29b upregulation mediate the effect of carvedilol on attenuating the acute myocardium infarction-induced myocardial fibrosis in rat. PLoS One. 2013;8:e75557. doi: 10.1371/journal.pone.0075557. [DOI] [PMC free article] [PubMed] [Google Scholar]



