Abstract
Alzheimer’s disease (AD) is the most common cause of dementia, which affects more than 5 million individuals in the United States. Unfortunately, no effective therapies are currently available to prevent development of AD or to halt progression of the disease. It has been proposed that monoacylglycerol lipase (MAGL), the key enzyme degrading the endocannabinoid 2-arachidonoylglycerol (2-AG) in the brain, is a therapeutic target for AD based on the studies using the APP transgenic models of AD. While inhibition of 2-AG metabolism mitigates β-amyloid (Aβ) neuropathology, it is still not clear whether inactivation of MAGL alleviates tauopathies as accumulation and deposition of intracellular hyperphosphorylated tau protein are the neuropathological hallmark of AD. Here we show that JZL184, a potent MAGL inhibitor, significantly reduced proinflammatory cytokines, astrogliosis, phosphorylated GSK3β and tau, cleaved caspase-3, and phosphorylated NF-kB while it elevated PPARγ in P301S/PS19 mice, a tau mouse model of AD. Importantly, Tau transgenic mice treated with JZL184 displayed improvements in spatial learning and memory retention. In addition, inactivation of MAGL ameliorates deteriorations in expression of synaptic proteins in P301S/PS19 mice. Our results provide further evidence that MAGL is a promising therapeutic target for AD.
Keywords: 2-arachidonoylglycerol, monoacylglycerol lipase, Alzheimer’s disease, neuroinflammation, tauopathies, cognition
Introduction
Alzheimer’s disease (AD), the most common form of dementia, is a degenerative brain disease characterized by neuroinflammation, accumulation of β-amyloid (Aβ) plaques and neurofibrillary tangles, neurodegeneration, synaptic dysfunction, and cognitive decline. Based on ‘the 2020 Alzheimer’s Disease Facts and Figures’ reported by Alzheimer’s Association, more than 5 million Americans age 65 and older are living with Alzheimer’s dementia in 2020 and AD is the sixth-leading cause of death in the US. However, no effective pharmacotherapies are currently available for prevention and treatment of AD. This is largely due to the fact that etiology of AD is multifactorial and development of AD is linked to multiple mechanisms or signaling pathways. This means that the ideal AD therapy should be able to modify the disease through multiple mechanisms.
Endocannabinoids are endogenous bioactive lipid mediators involved in a variety of physiological and pathological processes. The endocannabinoid 2-arachidonoylglycerol (2-AG) is the most abundant endogenous cannabinoid and a full agonist for both CB1R and CB2R [1]. Several lines of evidence indicate that 2-AG displays anti-inflammatory and neuroprotective properties in responses to harmful insults [2–6]. 2-AG is primarily synthesized from diacylglycerol (DAG) by diacylglycerol lipases α/β and metabolized by the enzymes, including monoacylglycerol lipase (MAGL) and α/β hydrolase domain-containing protein 6 and 12 (ABHD6/12). 2-AG is also converted by cyclooxygenase-2 (COX-2) to a new class of prostaglandins, prostaglandin glycerol esters (PG-Gs), when COX-2 is excessively activated during inflammation [6, 7]. Although these enzymes are capable of metabolizing of 2-AG, it has been estimated that 85% of 2-AG in the brain is degraded by MAGL [8, 9], suggesting that MAGL plays a dominant role in control of endogenous 2-AG levels in the brain. Recent studies provided further evidence that 2-AG in neurons and astrocytes is primarily hydrolyzed by MAGL, while it in microglial cells is predominantly degraded by ABHD12 [10, 11]. The immediate metabolites of 2-AG are arachidonic acid (AA), a precursor of prostaglandins catalyzed by COX-1/2 and leukotrienes via arachidonate 5-lipoxygenase (LOX). Prostaglandins (i.e., PGE2) and leukotrienes are important inflammatory mediators or proinflammatory [12, 13], while 2-AG is anti-inflammatory and neuroprotective [2–6, 14]. Apparently, inactivation of MAGL would result in augmentation of anti-inflammatory and neuroprotective 2-AG signaling, while lowering proinflammatory and neurotoxic eicosanoid levels [7, 9, 10, 15], suggesting that inhibition of 2-AG metabolism in the brain will lead to activate neuroprotective signaling pathways and concurrently turn off signaling pathways that are detrimental to brain function [16, 17]. Indeed, previous studies from our group and others demonstrated that inactivation of MAGL reduces Aβ formation and accumulation, neuroinflammation and neurodegeneration and improves synaptic and cognitive functions in APP transgenic mice, animal models of AD [15, 18–20]. However, it is still not clear whether inhibition of 2-AG metabolism mitigates tauopathies as intracellular neurofibrillary tangles that represent hyperphosphorylated tau proteins are an important neuropathological hallmark of AD [21, 22]. Here we show that inactivation of MAGL attenuates neuroinflammation and tau phosphorylation and prevents deterioration in synaptic proteins and cognitive decline in P301S/PS19 mice, a tau mouse model of AD, supporting generalizability of MAGL as a promising therapeutic target for AD.
RESULTS
JZL184 mitigates neuroinflammation in P301S/PS19 mice
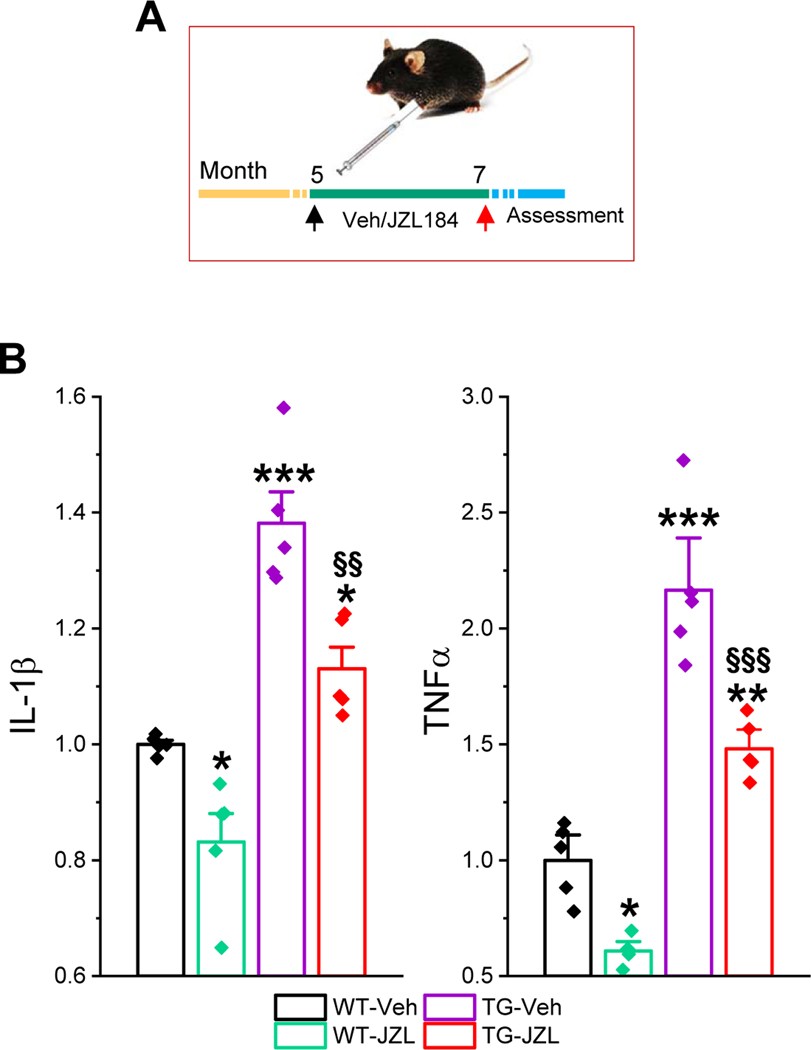
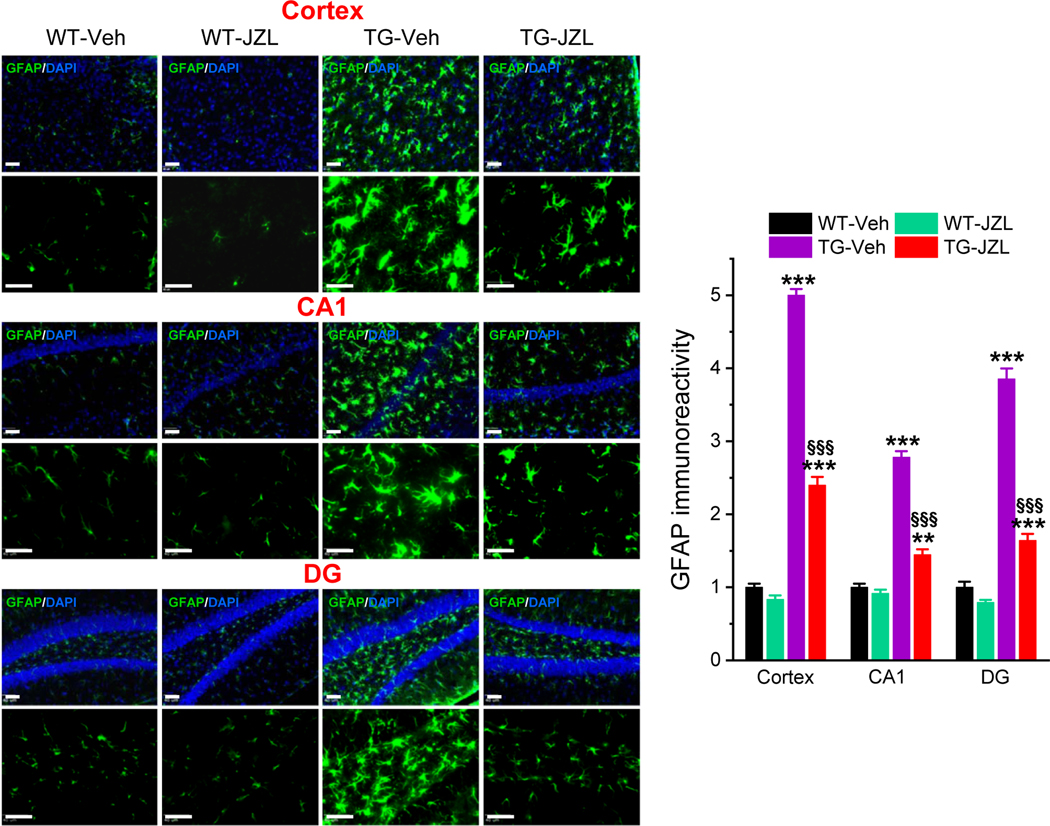
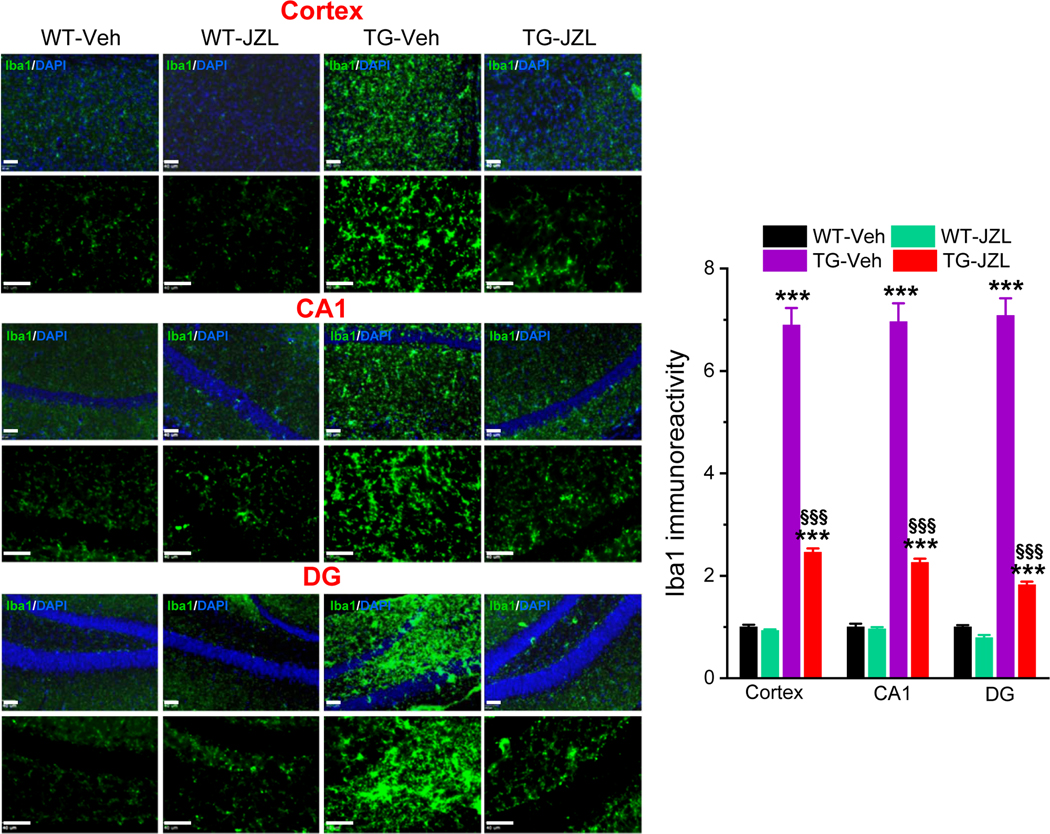
Neuroinflammation is one of the major pathogenic mechanisms that contribute to neuropathology and cognitive decline in AD. Previous studies demonstrate that pharmacological inhibition MAGL attenuates neuroinflammation evident by decreases in proinflammatory cytokines and astroglial reactivity in APP transgenic mice [15, 18–20]. To determine whether inhibition of MAGL produces anti-inflammatory effects in tau P301S/PS19 transgenic (TG) mice [23], we treated mice at 5 months of age with JZL184 (10 mg/kg, three times/week for 8 weeks, Figure 1A), as described previously [15]. We first assessed hippocampal cytokines IL-1β and TNFα in 7-month old TG mice and age-matched wild type (WT) control mice treated with vehicle or JZL184 using ELISA analysis. As shown in Figure 1B, the levels of IL-1β and TNFα were significantly elevated in PS19 TG mice when compared with WT mice. Treatment with JZl184 significantly decreased hippocampal IL-1β and TNFα in both WT and TG mice. These results indicate that inhibition of 2-AG metabolism with JZL184 reduces neuroinflammation in tau TG mice. To further confirm anti-inflammatory effects of MAGL inactivation in tau TG mice, GFAP immunoreactivity, a marker for inflammatory responses in astrocytes, was assessed in the brain of P301S TG mice treated with vehicle or JZL184 three times a week for 8 weeks. As shown in Figure 2, GFAP immunoreactivity was robustly reduced in the cortex and hippocampus of 7-month old P301S TG mice treated with JZL184. Next, we detected Iba1 immunoreactivity, a marker for inflammatory responses in microglia, as microglial cells are the innate immune system of the brain and play an important role in neuroinflammatory processes. As shown in Figure 3, JZL184 also greatly suppressed reactive microglial cells in the cortex and hippocampus of the PS301S TG mice. These results are consistent with previous observations where inhibition of MAGL mitigates neuroinflammation in APP TG mice [15, 18, 19].
Figure 1. Inactivation of MAGL reduces proinflammatory cytokines in P301S/PS19 transgenic (TG) mice.
A) Schematic illustration of the experimental protocol. P301S tau TG mice and their age-matched wild-type (WT) controls received vehicle or JZL184 (10 mg/kg, i.p.) three times per week starting at 5 months of age for 8 weeks. All the assessments were made at 7 months of age. B) Changes in hippocampal IL-1β and TNFα in mice treated with JZL184. Cytokines were detected using ELISA analysis. Data are means ±SEM. *P<0.05, ***P<0.001 compared with WT-vehicle; §§P<0.01, §§§P<0.001 compared with TG-vehicle (ANOVA with Bonferroni test, n=5/group).
Figure 2. Inhibition of 2-AG metabolism attenuates reactivity of astrocytes in P301S TG mice.
Immunoreactivity of astrocytic marker GFAP (green) in the cortex and hippocampus is reduced in tau TG mice that received JZL184. Scale bars: 40 μm. Data are means ±SEM. **P<0.01, ***P<0.001 compared with WT-vehicle control; §§§P<0.001 compared with TG-vehicle (ANOVA with Bonferroni post-hoc test, n=4 mice per group).
Figure 3. Inhibition of 2-AG metabolism attenuates reactivity of microglial cells in P301S TG mice.
Immunoreactivity of microglial marker Iba-1 (green) in the cortex and hippocampus is reduced in tau TG mice that received JZL184. Scale bars: 40 μm. Data are means ±SEM. **P<0.01, ***P<0.001 compared with WT-vehicle control; §§§P<0.001 compared with TG-vehicle (ANOVA with Bonferroni post-hoc test, n=4 mice per group).
Inhibition of 2-AG metabolism alleviates tauopathies in P301S/PS19 TG mice
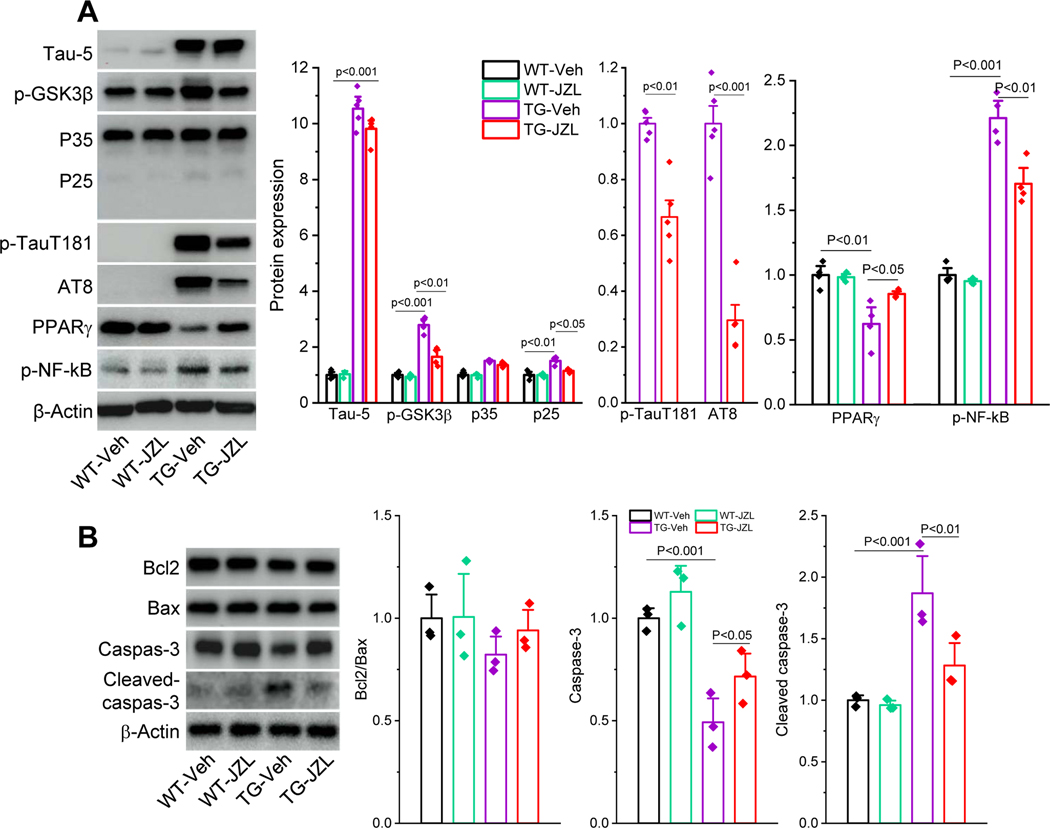
Accumulation of intracellular neurofibrillary tangles resulting from hyperphosphorylated tau is the neuropathological hallmark of AD [21, 22]. However, no studies have been conducted to assess whether disruption of 2-AG metabolism reduces phosphorylated tau (p-tau). To this end, we assessed total tau (Tau-5) and p-tau, including p-tau Thr181 (p-tauT181), and p-tau Ser202/Thr205 (AT8) in the hippocampus of PS19 TG mice treated with JZL184. As shown in Figure 4A, p-tauT181 and AT8 were robustly elevated in TG mice, indicating increased p-tau proteins in P301S TG mice. Importantly, while JZL184 did not induce changes in Tau-5, which is the transgene and overexpressed in this line of tau TG mice, it significantly reduced p-tauT181 and AT8 in TG mice, indicating that inactivation of MAGL decreases p-tau. Since tau phosphorylation is largely regulated by glycogen synthase kinase-3β (GSK3β) and cyclin-dependent kinase 5 (Cdk5) [24–27], we detected phosphorylated GSK3β (p-GSK3β) and P35/25 that interact with Cdk5. As shown in Figure 4A, p-GSK3β and P25 were significantly elevated in TG mice that received the vehicle, but the increase was reduced in TG mice treated JZL184, indicating JZL184-reduced p-tau is likely through suppression of p-GSK3β and P25.
Figure 4. Inactivation of MAGL alleviates tauopathies in P301S TG mice.
A) Immunoblot analysis of tau-5 (total tau), p-GSK3β, P35/25, p-tauT181, p-tau Ser202/Thr205 (AT8), PPARγ and p-NF-kB in the hippocampus in P301S TG mice that received JZL184 for 8 weeks. The values of Tau-5, p-GSK3β, P35/25, PPARγ and p-NF-kB are normalized to WT-vehicle, while p-tauT181 and AT8 are normalized to TG-vehicle as these phosphorylated tau proteins are not detectable in WT mice. Data are means ±SEM (ANOVA with Fisher PLSD test, n=4~5/group). B) Immunoblot analysis of Bcl2, Bax, caspase-3 and cleaved caspase-3 in hippocampal tissues from PS19 TG mice that received JZL184 for 8 weeks. Data are means ±SEM (ANOVA with Fisher PLSD test, n=3/group)
We also assessed PPARγ, an important nuclear receptors displaying anti-inflammatory and neuroprotective effects, and phosphorylated NF-kB (p-NF-kB), an important transcription factor regulating expression of genes involved in inflammation and neurodegeneration [28–31]. We observed that hippocampal expression of PPARγ was significantly reduced, while p-NF-kB was increased in 7-months old P301S TG mice. However, JZL184 prevented a decrease in expression of PPARγ and an increase in p-NF-kB (Figure 4A).
It is generally accepted that apoptosis is one of the important mechanisms contributing to loss of neurons in AD [32–34]. To determine whether apoptosis occurs in 7-month old P301S tau TG mice and whether MAGL inactivation mitigates tau-induced apoptosis, we detected expression of Bcl2, Bax, and caspase-3, which are involved in apoptotic cascades [35, 36], in hippocampal tissues from WT and TG mice treated with vehicle or JZL184. As shown in Figure 4B, expression of caspase-3 was significantly decreased, while cleaved caspase-3 was increased in the hippocampus in TG mice treated with vehicle. However, these changes were prevented by inactivation of MAGL with JZL184. We noticed that expression of Bcl2 was slightly reduced while Bax was elevated in vehicle-treated TG mice. Although these changes were reversed in animals treated with JZL184, there were no statistically significant differences in expression of Bcl2, Bax, or in the ratio of Bcl2/Bax between WT and TG mice treated with vehicle or JZL184 (Figure 4B). These results suggest that apoptosis occurs in tau TG mice and that MAGL inactivation reduces apoptosis primarily through a caspase-3-dependent signaling pathway in tau TG mice. This is consistent with the observations where caspase-3/cleaved caspase-3, but not Bcl2/Bax pathways, contribute to neuropathology in P301S tau TG mice [37].
Inactivation of MAGL improves cognitive function in P301S/PS19 TG mice
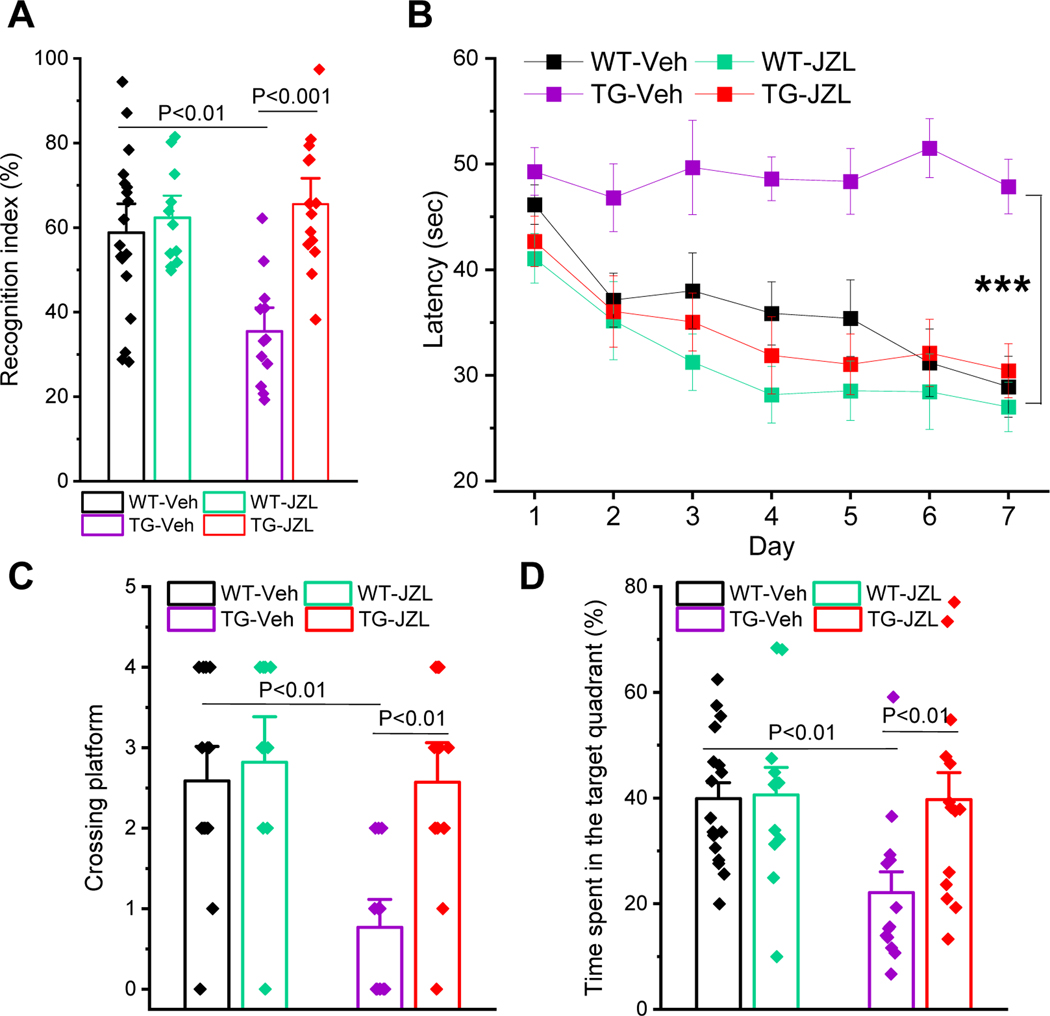
Inactivation of MAGL has been shown to improve spatial learning and memory in APP transgenic mice [15, 20, 38]. However, it is still not clear whether inactivation of MAGL prevents cognitive decline in tau transgenic models of AD. To determine whether inhibition of 2-AG metabolism improves cognitive function, we assessed spatial learning and memory retention using the novel object recognition (NOR) and the Morris water maze tests, as described previously [15, 20, 38–41], in P301S tau TG mice treated with JZL184 for 8 weeks. As shown in Figure 5A, memory retention was significantly impaired in 7-month-old P301S TG mice treated with vehicle. However, the impairment was prevented in TG mice treated with JZL184. The cognitive improvement by inactivation of MAGL in P301S TG mice is further confirmed by the Morris water maze test. As shown in Figure 5B~D, JZL184 significantly improves spatial learning and memory retention in P301S TG mice, similar to APP transgenic mice [15, 20, 38]. These results suggest that inactivation of MAGL is capable of preventing cognitive decline in both APP and tau transgenic models of AD.
Figure 5. Inhibition of MAGL improves cognitive function in P301S TG mice.
A) The novel object recognition test in 7-month-old Pv TG mice that received JZL184 for 8 weeks. Data are means ±SEM (ANOVA with Bonferroni post-hoc test, n=11~18/group). B) The Morris water maze test was performed in 7-month-old P301S TG mice that received JZL184 for 8 weeks. Data are means ±SEM (***P<0.001, ANOVA with repeated measures, n=11~17/group). C) Number of platform crossing during the probe test. The probe trial was conducted 24 hrs after 7 days of acquisition training. Data are means ±SEM (ANOVA with Bonferroni post-hoc test, n=11~17/group). D) The percentage of time spent in search of the target quadrant during the probe trial. Data are means ±SEM (ANOVA with Bonferroni post-hoc test, n=11~17/group).
Inhibition of 2-AG metabolism prevents deterioration in expression of synaptic proteins in P301S/PS19 TG mice
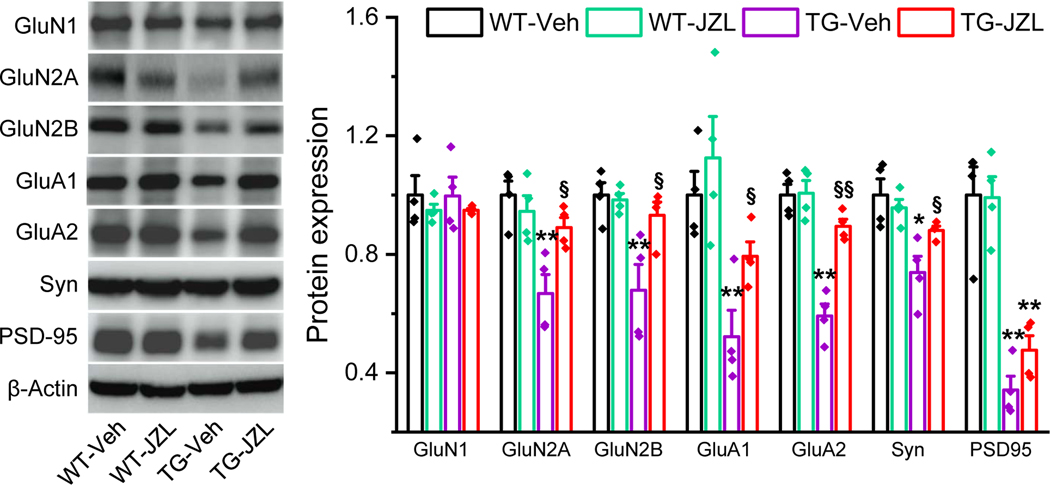
It has been well recognized that cognitive deficits in AD is primarily due to loss of the synaptic integrity [40, 42]. Proper and efficient synaptic transmission and plasticity in the brain are largely dependent on expression and function of excitatory glutamate receptors. Our previous studies showed that inactivation of MAGL in APP TG mice prevents downregulation of glutamate receptor subunits in the brain [15, 38]. To determine whether JZL184 prevents deterioration in expression of glutamate receptors in tau TG mice, we detected expression of AMPA and NMDA glutamate receptor subunits, including GluA1, GluA2, GluN1, GluN2A and GluN2B, in P301S tau TG mice treated with vehicle or JZL184. As shown in Figure 6, expression of GluA1, GluA2, GluN2A, and GluN2B in the hippocampus was significantly downregulated in P301S TG mice treated with vehicle. However, expression of these glutamate receptor subunits in mice treated with JZL184 was returned to the levels in WT mice, suggesting that inhibition of 2-AG metabolism is also capable of preventing deterioration in expression of glutamate receptor subunits. To further assess whether inactivation of MAGL prevents changes in expression of synaptic proteins in P301S TG mice, we also detected synaptophysin (Syn) and PSD-95, pre- and post-synaptic markers. We observed that hippocampal expression of Syn and PSD-95 was significantly reduced in 7-months old P301S TG mice. However, treatment with JZL184 attenuated downregulation of Syn. Although expression of PSD-95 in JZL184-treated animals was slightly increased, but no statistical significance. Our results provide evidence that restraint of 2-AG metabolism by inactivation of MAGL averts downregulation of synaptic proteins in tau TG mice.
Figure 6. Inactivation of MAGL prevents deterioration in expression of synaptic proteins in P301S TG mice.
Immunoblot analysis of hippocampal expression of AMPA (GluA1 and GluA2), and NMDA (GluN1, GluN2A, and GluN2B) receptor subunits, PSD-95, synaptophysin (Syn), and PPARγ in 7- month-old P301S TG mice treated with vehicle or JZL184. Data are means ±SEM. *P<0.05; **P<0.01 compared with the WT-vehicle, §P<0.05, §§P<0.01 compared with the TG-vehicle (ANOVA with Fisher’s PLSD test, n=4 mice per group).
DISCUSSION
It has been proposed that MAGL inhibition, which augments 2-AG signaling and reduces eicosanoid levels [8, 9], is a promising therapy for AD [15]. However, MAGL proposed as a therapeutic target is largely based on the studies using the APP transgenic models of AD [15, 18–20, 38]. Since intracellular neurofibrillary tangles resulting from accumulation of hyperphosphorylated tau proteins is one of the important neuropathological hallmarks of AD, it is still not clear whether inhibition of 2-AG metabolism alleviates tauopathies. To ascertain generalizability of the results from APP TG mice, we used P301S/PS19 tau TG mice, a tau mouse model of AD [23]. We demonstrate in the present study that JZL184, a highly selective and potent MAGL inhibitor [9, 43, 44], significantly reduces neuroinflammation, phosphorylated tau, and cleaved caspase-3, and prevents deteriorations in expression of synaptic proteins, and improves cognitive function in P301S TG mice. Our results provide further evidence that inactivation of MAGL is capable of alleviating both Aβ neuropathology and tauopathies, supporting the notion that MAGL is a promising therapeutic target for AD.
It is generally accepted that the etiology of AD is multifactorial and complex. Apparently, AD involves multiple etiopathogenic mechanisms or signaling pathways. This means that the ideal AD therapy should be able to modify the disease through multiple mechanisms. Several lines of evidence suggest that 2-AG is an important endogenous signaling molecule maintaining brain homeostasis by modulating synaptic transmission and plasticity, resolving neuroinflammation, and protecting neurons from harmful insults [2–6, 14, 17, 45–48]. Intriguingly, the immediate metabolite of 2-AG is arachidonic acid (AA), a precursor of prostaglandins and leukotrienes, which are proinflammatory and neurotoxic [12, 13]. Apparently, inactivation of MAGL results in augmentation of anti-inflammatory and neuroprotective 2-AG signaling, while lowering proinflammatory and neurotoxic eicosanoid levels [6, 7, 16]. This means that inactivation of MAGL will turn on anti-inflammatory and neuroprotective signaling pathways and concurrently off signaling pathways that are detrimental to homeostatic maintenance of brain function [16, 17]. Indeed, previous studies provide evidence showing that pharmacological or genetic inactivation of MAGL enhances long-term potentiation (LTP), learning and memory [15, 49, 50]. Importantly, inactivation of MAGL produces profound anti-inflammatory and neuroprotective effects and improves synaptic and cognitive functions in animal models of AD and MPTP model of Parkinson’s disease, and close head injury [9, 15, 18, 19, 41]. Thus, MAGL has been proposed as a therapeutic target for neurodegenerative diseases [15, 38, 41, 51–54].
The neuroprotective effects of MAGL inhibition observed in the present study likely benefit from multiple signaling pathways based on the anti-inflammatory and neuroprotective properties of its substrate 2-AG and the proinflammatory and neurotoxic properties of the 2-AG metabolites (Figure 7). It has been demonstrated earlier that inactivation of MAGL suppresses LPS-induced inflammation and reduces MPTP-induced neuronal loss [9]. These neuroprotective effects by disruption of MAGL are not mediated via 2-AG signaling that binds to CB1R or CB2R, but through prostaglandin signaling derived from AA, the 2-AG immediate metabolite catalyzed by MAGL. Consistent with these observations, MAGL inhibition-produced suppression of gliosis, cytokine production, and Aβ formation in APP TG mice is also independent of CB1R or CB2R [15, 18, 38]. In contrast, 2-AG has been shown to protect neurons from traumatic brain injury via a CB1R-dpendeent mechanism [5, 55]. Especially, 2-AG or inhibition of MAGL attenuates Aβ, proinflammatory and excitotoxic insults-induced neuroinflammation, neurodegeneration, and apoptosis via a CB1R-mediated suppression of caspase-3 cleavage, MAPK, ERK1/2, NF-kB, and COX-2 in vitro [2, 4]. Thus, MAGL inactivation-produced beneficial effects are closely associated with augmentation of 2-AG signaling and concurrent reduction of its metabolites through CB1R or CB2R dependent and independent mechanisms.
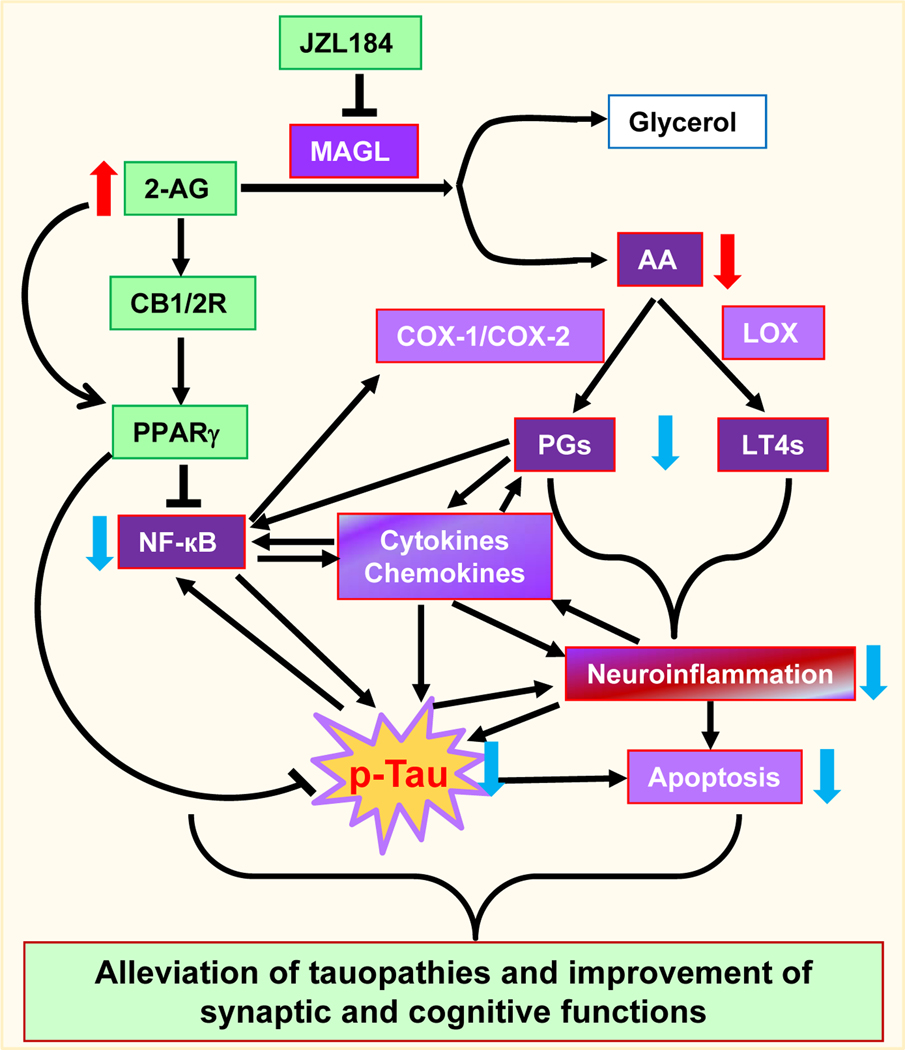
Figure 7. Signaling pathways mediating the beneficial effects produced by inhibition of 2-AG metabolism.
Inactivation of monoacylglycerol lipase (MAGL) by JZL184 augments 2-AG signaling and decreases 2-AG immediate metabolite arachidonic acid (AA) and AA-derived prostaglandins (PGs) through cyclooxygenase 1 and 2 (COX-1/2) and leukotrienes (LT4s) through 5-lipoxygenase (LOX). 2-AG possesses anti-inflammatory and neuroprotective properties, while prostaglandins and leukotrienes are proinflammatory and neurotoxic. Enhanced 2-AG by inactivation of MAGL stimulates expression and activity PPARγ through CB1/2-dependent and independent pathways, resulting in inhibition of NF-κB. Reduced NF-kB transcriptional activity and the amount of prostaglandins and leukotrienes decrease proinflammatory cytokines and chemokines and neuroinflammation as well as apoptosis, which in turn suppress tau phosphorylation and tauopathies. These events may benefit to maintaining brain homeostasis and the integrity of synapses, and thus improving synaptic and cognitive functions.
Peroxisome proliferator-activated receptor-γ (PPARγ), a member of nuclear receptors, displays anti-inflammatory and neuroprotective effects through interacting with NF-kB, resulting in reduction of expression of genes involved in inflammation and neurodegeneration [28–31]. Our previous studies show that 2-AG- or MAGL-inhibition-induced suppression of COX-2 and p-NF-kB is mediated via CB1R-dpendent promotion of PPARγ expression in vitro [3], suggesting that interactions occur between PPARγ and NF-kB in 2-AG-produced anti-inflammatory and neuroprotective effects against harmful insults [6]. Our latest studies provide further evidence that 2-AG is an endogenous PPARγ, which mediates 2-AG- or MAGL inactivation-produced resolution of neuroinflammation and protection of neurons by inhibition of NF-kB signaling in APP TG mice [20]. In the present study, we observed that MAGL inactivation elevates PPARγ and decreases p-NF-kB in tau TG mice, suggesting that PPARγ signaling plays an important role in MAGL inactivation produced mitigation of neuroinflammation, apoptosis, and tauopathies (Figure 7).
Senile plaques and neurofibrillary tangles are the two neuropathological hallmarks of AD. Senile plaques are extracellular accumulation and deposition of Aβ, while neurofibrillary tangles are intracellular deposition of hyperphosphorylated tau proteins. The behavioral symptoms of AD are largely associated with the accumulation of plaques and tangles [21]. Several lines of evidence show that tau stability is largely associated with molecular chaperons, including heat shock protein 90 (Hsp90) and heat shock protein 70 (Hsp70), two major molecular chaperones that are involved in tau misfolding, self-aggregation, and clearance [35, 36, 56, 57]. It has been shown that inhibition of Hsp90 or Hsp70 reduces tauopathies [58, 59]. Although we did not assess Hsp90 and Hsp70 in the present study, it is possible that Hsp90 or Hsp70 might be involved in the MAGL inactivation-alleviated tauopathies, which warrants further research.
While both Aβ and p-tau contribute to neuropathology, synaptic and cognitive declines in AD, accumulated evidence suggests that tau proteins are essential or required for Aβ-induced neurotoxicity, neurodegeneration and synaptic dysfunction [22, 60, 61]. This means that hyperphosphorylated tau plays an important role in driving cognitive decline in AD. In addition, clinical trials of Aβ-targeting therapies are largely failed. This suggests that efficacious AD therapies may need to alleviate both Aβ and tau pathologies. The results from the previous studies where inactivation of MAGL alleviates neuropathology and improves synaptic and cognitive function in APP TG mice [15, 18–20, 38] together with the findings from the present study in tau TG mice suggest that inhibition of 2-AG metabolism is capable of curbing both Aβ and tau neuropathologies and preventing cognitive decline and that MAGL is a promising therapeutic target.
MATERIALS AND METHODS
Animals
P301S tau transgenic mice (B6;C3-Tg(Prnp-MAPT*P301S)PS19Vle/J, stock number: 008169) were obtained from the Jackson Lab [23]. Both male and female P301S mice and age-matched non-transgenic wild-type (WT) littermates at 7 months of age were used in the present study. 4-Nitrophenyl-4-[bis (1,3-benzodioxol-5-yl)(hydroxy)methyl]piperidine-1-carboxylate (JZL184, provided by the NIH Mental Health Institute Chemical Synthesis and Drug Supply Program), a potent and selective inhibitor of MAGL, was dissolved in the vehicle containing Tween-80 (10%), DMSO (10%), and saline (80%). Animals were treated with either vehicle or JZL184 (10 mg/kg, i.p.) three times per week starting at 5 months of age for 8 weeks, as described previously [15, 20]. All the observations and assessments in mice were made at 7 months of age.
All the experiments were performed in compliance with the US Department of Health and Human Services Guide for the Care and Use of Laboratory Animals. The care and use of the animals reported in this study were approved by the Institutional Animal Care and Use Committee of University of Texas Health San Antonio. The experiments, whenever possible, were performed in a blinded fashion.
ELISA.
Levels of IL-1β and TNFα in hippocampal tissues of P301S TG mice that received vehicle control or JZL184 were detected using a colorimetric IL1β and TNFα ELISA kits (IL-1β: Cat# RAB0275, Sigma-Aldrich, and TNFα: Cat# MBS825075, MyBioSource) according to the instructions provided by the manufacturers.
Immunoblot
Western blot assay was conducted to determine expression of GluA1, GluA2, GluN1, GluN2A, and GluN2B, tau-5, AT8, p-tauT181, p-GSK3β, p35/25, caspase-3, cleaved caspase-3, Bcl2, Bax, PPARγ, PSD95, synaptophysin (Syn) in the hippocampus from animals that received vehicle or JZL184. Hippocampal tissue was extracted and immediately homogenized in RIPA lysis buffer and protease inhibitors, and incubated on ice for 30 min, then centrifuged for 10 min at 10,000 rpm at 4°C. Supernatants were fractionated on 4–15% SDS-PAGE gels and transferred onto PVDF membranes (Bio-Rad). The membrane was incubated with anti-GluA1 (1:1,000, Abcam, Cat# AB31232), GluA2 (1:1,000, Abcam, Cat# AB133477), GluN1 (1:1,000, Abcam, Cat# AB109182), GluN2A (1:1,000, LSBio, Cat# LS-B7707), GluN2B (1:1,000, Abcam, Cat# AB65783), tau-5 (1:1,000, ThermoFisher, Cat# AHB0042), AT8 (1:200, ThermoFisher, Cat# MN1020), p-tauT181 (1:1,000, Cell Signaling Technology, Cat# 12885), p-GSK3β (1:1,000, Cell Signaling Technology, Cat# 9323), p35/25 (1:1,000, Cell Signaling Technology, Cat# 2680), caspase-3 (1:1,000, Cell Signaling Technology, Cat# 9665), cleaved caspase-3 (1:1,000, Cell Signaling Technology, Cat# 9661), Bcl2 (1:1,000, Cell Signaling Technology, Cat# 2876), Bax (1:1,000, Cell Signaling Technology, Cat# 2772), PPARγ (1:1,000, Abcam, Cat# AB27649), Syn (1:500, Abcam, Cat# AB8049), and PSD95 (1:1,000, Abcam, Cat# AB2723) at 4°C overnight. The blots were washed and incubated with a secondary antibody (goat anti-rabbit 1:2,000, Cell Signaling Technology, Cat# 7074) at room temperature for 1 hr. Proteins were visualized by enhanced chemiluminescence (Amersham Biosciences, UK). The densities of specific bands were quantified by densitometry using FUJIFILM Multi Gauge software (version 3.0). Band densities were quantified and converted to the total amount of protein loaded in each well as determined by mouse anti β-actin (1:2,000, Santa Cruz Biotechnology, Cat# SC-47778) as described previously [15, 20, 39, 40].
Immunohistochemistry
Immunohistochemical analyses were performed to determine astrocytic and microglial markers (GFAP and Iba1) in coronal brain sections, as described previously [15, 20, 41]. Animals were anesthetized with ketamine/Xylazine (200/10 mg/kg) and subsequently transcardially perfused with PBS followed by 4% paraformaldehyde in phosphate buffer. The brains were quickly removed from the skulls and fixed in 4% paraformaldehyde overnight, and then transferred into the PBS containing 30% sucrose until sinking to the bottom of the small glass jars. Cryostat sectioning was made on a freezing Vibratome at 40 μm and series sections (every 10th section from each animals) were collected in 0.1M phosphate buffer. Free floating sections were immunostained using specific antibodies for GFAP (1:500, Sigma-Aldrich, Cat# G3893) an Iba1 (1:500, Sigma-Aldrich, Cat# MABN92) followed by incubation with the corresponding fluorescent-labeled secondary antibody. 4′−6-Diamidino-2-phenylindole (DAPI), a fluorescent stain that binds strongly to DNA, was used it to detect cell nuclei in the sections. The sections were then mounted on slides for immunofluorescence detection using a Zeiss deconvolution microscope. The immunoreactivity (in arbitrary densitometric units) of GFAP and Iba1 in the cortex and hippocampus in each image was analyzed and quantified using SlideBook 6.0, as described previously [15, 20, 38, 41].
Novel object recognition
The novel object recognition (NOR) test was performed as described previously [40, 62]. Briefly, animals were first allowed to acclimate to the testing environment (habituation). The test included two stages: training and testing. In the first stage of the test, the animal was confronted with two identical objects, placed in an open field, and in the second stage, the animal was exposed to two dissimilar objects placed in the same open field: one familiar object, used in the first phase, and the other novel object. Exploration of an object was defined as time spent with the head oriented towards and within two cm of the object. The time spent exploring each of the objects in stage two was detected by a video-camera using an EthoVision video tracking system (Noldus). The recognition index (RI) was calculated based on the following equation: RI =TN/TN+TF), where TN is the exploration time devoted to the novel object and TF is the exploration time for the familiar object, as described previously [63, 64].
Morris water Maze
The classic Morris water maze (MWM) test was used to determine spatial learning and memory, as described previously [15, 20, 38–41]. Animals were initially randomized grouped to receive different treatments. The test was performed 24 hrs after last cessation of JZL184 injections. A circular water tank (diameter 120 cm) was filled with water and the water was made opaque with non-toxic white paint. A round platform (diameter 15 cm) was hidden 1 cm beneath the surface of the water at the center of a given quadrant of the water tank. PS19 TG mice and their age-matched littermates received learning acquisition training for 7 days (7 sessions), and each session consisted of 4 trials. For each trial, the mouse was released from the wall of the tank and allowed to search, find, and stand on the platform for 10 seconds within the 60-second trial period. For each training session, the starting quadrant and sequence of the four quadrants from where the mouse was released into the water tank were randomly chosen so that it was different among the separate sessions for each animal and was different for individual animals. The mice in the water pool were recorded by a video-camera using an EthoVision video tracking system (Noldus). A probe test to assess memory retention was conducted 24 hours after the completion of the training acquisition. During the probe test, the platform was removed from the pool, and the task performances were recorded for 60 seconds.
Data analysis
Data are presented as mean ± S.E.M. Unless stated otherwise, one- or two-way-analysis of variance (ANOVA) followed by post-hoc tests were used for statistical comparison when appropriate. Differences were considered significant when P< 0.05.
Acknowledgement
The authors also thank NIH Mental Health Institute Chemical Synthesis and Drug Supply Program for providing JZL184.
Funding
This work was supported by National Institutes of Health grants R01NS076815, R01MH113535, and R01AG058621 (to C.C.) and by startup funds from UT Health San Antonio, Joe R. & Teresa Lozano Long School of Medicine (to C.C.).
Footnotes
Ethics Approval
All the animal experiments were performed in compliance with the US Department of Health and Human Services Guide for the Care and Use of Laboratory Animals. The care and use of the animals reported in this study were approved by the Institutional Animal Care and Use Committee of University of Texas Health San Antonio.
Data availability
Data supporting the findings of this manuscript are available from the corresponding authors upon request.
Code availability
Not applicable
Consent to participate
Not applicable
Consent for publication
Not applicable
Conflict of interest
The authors declare no conflict of interest.
References
- 1.Sugiura T, Kishimoto S, Oka S and Gokoh M (2006) Biochemistry, pharmacology and physiology of 2-arachidonoylglycerol, an endogenous cannabinoid receptor ligand. Prog Lipid Res 45:405–46. doi: 10.1016/j.plipres.2006.03.003 [DOI] [PubMed] [Google Scholar]
- 2.Chen X, Zhang J and Chen C (2011) Endocannabinoid 2-arachidonoylglycerol protects neurons against beta-amyloid insults. Neuroscience 178:159–68. doi: 10.1016/j.neuroscience.2011.01.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Du H, Chen X, Zhang J and Chen C (2011) Inhibition of COX-2 expression by endocannabinoid 2-arachidonoylglycerol is mediated via PPAR-gamma. Br J Pharmacol 163:1533–49. doi: 10.1111/j.1476-5381.2011.01444.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhang J and Chen C (2008) Endocannabinoid 2-arachidonoylglycerol protects neurons by limiting COX-2 elevation. J Biol Chem 283:22601–11. doi: 10.1074/jbc.M800524200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Panikashvili D, Simeonidou C, Ben-Shabat S, Hanus L, Breuer A, Mechoulam R and Shohami E (2001) An endogenous cannabinoid (2-AG) is neuroprotective after brain injury. Nature 413:527–31. doi: 10.1038/35097089 [DOI] [PubMed] [Google Scholar]
- 6.Xu JY and Chen C (2015) Endocannabinoids in Synaptic Plasticity and Neuroprotection. Neuroscientist 21:152–168. doi: 10.1177/1073858414524632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hermanson DJ, Gamble-George JC, Marnett LJ and Patel S (2014) Substrate-selective COX-2 inhibition as a novel strategy for therapeutic endocannabinoid augmentation. Trends Pharmacol Sci 35:358–67. doi: 10.1016/j.tips.2014.04.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Blankman JL, Simon GM and Cravatt BF (2007) A comprehensive profile of brain enzymes that hydrolyze the endocannabinoid 2-arachidonoylglycerol. Chem Biol 14:1347–56. doi: 10.1016/j.chembiol.2007.11.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nomura DK, Morrison BE, Blankman JL, Long JZ, Kinsey SG, Marcondes MC, Ward AM, Hahn YK, Lichtman AH, Conti B and Cravatt BF (2011) Endocannabinoid hydrolysis generates brain prostaglandins that promote neuroinflammation. Science 334:809–13. doi: 10.1126/science.1209200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Viader A, Blankman JL, Zhong P, Liu X, Schlosburg JE, Joslyn CM, Liu QS, Tomarchio AJ, Lichtman AH, Selley DE, Sim-Selley LJ and Cravatt BF (2015) Metabolic Interplay between Astrocytes and Neurons Regulates Endocannabinoid Action. Cell Rep 12:798–808. doi: 10.1016/j.celrep.2015.06.075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Viader A, Ogasawara D, Joslyn CM, Sanchez-Alavez M, Mori S, Nguyen W, Conti B and Cravatt BF (2016) A chemical proteomic atlas of brain serine hydrolases identifies cell type-specific pathways regulating neuroinflammation. Elife 5:e12345. doi: 10.7554/eLife.12345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hein AM and O’Banion MK (2009) Neuroinflammation and memory: the role of prostaglandins. Mol Neurobiol 40:15–32. doi: 10.1007/s12035-009-8066-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Salmon JA and Higgs GA (1987) Prostaglandins and leukotrienes as inflammatory mediators. Br Med Bull 43:285–96. [DOI] [PubMed] [Google Scholar]
- 14.Shohami E, Cohen-Yeshurun A, Magid L, Algali M and Mechoulam R (2011) Endocannabinoids and traumatic brain injury. Br J Pharmacol 163:1402–10. doi: 10.1111/j.1476-5381.2011.01343.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chen R, Zhang J, Wu Y, Wang D, Feng G, Tang YP, Teng Z and Chen C (2012) Monoacylglycerol lipase is a therapeutic target for Alzheimer’s disease. Cell Rep 2:1329–39. doi: 10.1016/j.celrep.2012.09.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chen C (2016) Endocannabinoid metabolism in neurodegenerative diseases. Neuroimmunol Neuroinflamm 3:268–270. doi: 10.20517/2347-8659.2016.46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen C (2015) Homeostatic regulation of brain functions by endocannabinoid signaling. Neural Regen Res 10:691–2. doi: 10.4103/1673-5374.156947 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Piro JR, Benjamin DI, Duerr JM, Pi Y, Gonzales C, Wood KM, Schwartz JW, Nomura DK and Samad TA (2012) A dysregulated endocannabinoid-eicosanoid network supports pathogenesis in a mouse model of Alzheimer’s disease. Cell Rep 1:617–23. doi: 10.1016/j.celrep.2012.05.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pihlaja R, Takkinen J, Eskola O, Vasara J, Lopez-Picon FR, Haaparanta-Solin M and Rinne JO (2015) Monoacylglycerol lipase inhibitor JZL184 reduces neuroinflammatory response in APdE9 mice and in adult mouse glial cells. J Neuroinflammation 12:81. doi: 10.1186/s12974-015-0305-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang J, Hu M, Teng Z, Tang YP and Chen C (2014) Synaptic and cognitive improvements by inhibition of 2-AG metabolism are through upregulation of microRNA-188–3p in a mouse model of Alzheimer’s disease. J Neurosci 34:14919–33. doi: 10.1523/jneurosci.1165-14.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bloom GS (2014) Amyloid-β and tau: the trigger and bullet in Alzheimer disease pathogenesis. JAMA Neurol 71:505–8. doi: 10.1001/jamaneurol.2013.5847 [DOI] [PubMed] [Google Scholar]
- 22.Rapoport M, Dawson HN, Binder LI, Vitek MP and Ferreira A (2002) Tau is essential to beta -amyloid-induced neurotoxicity. Proc Natl Acad Sci U S A 99:6364–9. doi: 10.1073/pnas.092136199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yoshiyama Y, Higuchi M, Zhang B, Huang SM, Iwata N, Saido TC, Maeda J, Suhara T, Trojanowski JQ and Lee VM (2007) Synapse loss and microglial activation precede tangles in a P301S tauopathy mouse model. Neuron 53:337–51. doi: 10.1016/j.neuron.2007.01.010 [DOI] [PubMed] [Google Scholar]
- 24.Takashima A (2006) GSK-3 is essential in the pathogenesis of Alzheimer’s disease. J Alzheimers Dis 9:309–17. doi: 10.3233/jad-2006-9s335 [DOI] [PubMed] [Google Scholar]
- 25.Turab Naqvi AA, Hasan GM and Hassan MI (2020) Targeting Tau Hyperphosphorylation via Kinase Inhibition: Strategy to Address Alzheimer’s Disease. Curr Top Med Chem 20:1059–1073. doi: 10.2174/1568026620666200106125910 [DOI] [PubMed] [Google Scholar]
- 26.Patrick GN, Zukerberg L, Nikolic M, de la Monte S, Dikkes P and Tsai LH (1999) Conversion of p35 to p25 deregulates Cdk5 activity and promotes neurodegeneration. Nature 402:615–22. doi: 10.1038/45159 [DOI] [PubMed] [Google Scholar]
- 27.Kimura T, Ishiguro K and Hisanaga S (2014) Physiological and pathological phosphorylation of tau by Cdk5. Front Mol Neurosci 7:65. doi: 10.3389/fnmol.2014.00065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bensinger SJ and Tontonoz P (2008) Integration of metabolism and inflammation by lipid-activated nuclear receptors. Nature 454:470–7. doi: 10.1038/nature07202 [DOI] [PubMed] [Google Scholar]
- 29.Villapol S (2018) Roles of Peroxisome Proliferator-Activated Receptor Gamma on Brain and Peripheral Inflammation. Cell Mol Neurobiol 38:121–132. doi: 10.1007/s10571-017-0554-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ju Hwang C, Choi DY, Park MH and Hong JT (2019) NF-κB as a Key Mediator of Brain Inflammation in Alzheimer’s Disease. CNS Neurol Disord Drug Targets 18:3–10. doi: 10.2174/1871527316666170807130011 [DOI] [PubMed] [Google Scholar]
- 31.Shabab T, Khanabdali R, Moghadamtousi SZ, Kadir HA and Mohan G (2017) Neuroinflammation pathways: a general review. Int J Neurosci 127:624–633. doi: 10.1080/00207454.2016.1212854 [DOI] [PubMed] [Google Scholar]
- 32.Bamberger ME and Landreth GE (2002) Inflammation, apoptosis, and Alzheimer’s disease. Neuroscientist 8:276–83. doi: 10.1177/1073858402008003013 [DOI] [PubMed] [Google Scholar]
- 33.Behl C (2000) Apoptosis and Alzheimer’s disease. J Neural Transm (Vienna) 107:1325–44. doi: 10.1007/s007020070021 [DOI] [PubMed] [Google Scholar]
- 34.Shimohama S (2000) Apoptosis in Alzheimer’s disease--an update. Apoptosis 5:9–16. doi: 10.1023/a:1009625323388 [DOI] [PubMed] [Google Scholar]
- 35.Alani B, Salehi R, Sadeghi P, Khodagholi F, Digaleh H, Jabbarzadeh-Tabrizi S, Zare M and Korbekandi H (2015) Silencing of Hsp70 intensifies 6-OHDA-induced apoptosis and Hsp90 upregulation in PC12 cells. J Mol Neurosci 55:174–183. doi: 10.1007/s12031-014-0298-3 [DOI] [PubMed] [Google Scholar]
- 36.Alani B, Salehi R, Sadeghi P, Zare M, Khodagholi F, Arefian E, Hakemi MG and Digaleh H (2014) Silencing of Hsp90 chaperone expression protects against 6-hydroxydopamine toxicity in PC12 cells. J Mol Neurosci 52:392–402. doi: 10.1007/s12031-013-0163-9 [DOI] [PubMed] [Google Scholar]
- 37.Zhang YH, Wang DW, Xu SF, Zhang S, Fan YG, Yang YY, Guo SQ, Wang S, Guo T, Wang ZY and Guo C (2018) α-Lipoic acid improves abnormal behavior by mitigation of oxidative stress, inflammation, ferroptosis, and tauopathy in P301S Tau transgenic mice. Redox Biol 14:535–548. doi: 10.1016/j.redox.2017.11.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhang J and Chen C (2018) Alleviation of Neuropathology by Inhibition of Monoacylglycerol Lipase in APP Transgenic Mice Lacking CB2 Receptors. Mol Neurobiol 55:4802–4810. doi: 10.1007/s12035-017-0689-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Chen R, Zhang J, Fan N, Teng ZQ, Wu Y, Yang H, Tang YP, Sun H, Song Y and Chen C (2013) Delta9-THC-caused synaptic and memory impairments are mediated through COX-2 signaling. Cell 155:1154–65. doi: 10.1016/j.cell.2013.10.042 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Song Y, Hu M, Zhang J, Teng ZQ and Chen C (2019) A novel mechanism of synaptic and cognitive impairments mediated via microRNA-30b in Alzheimer’s disease. EBioMedicine 39:409–421. doi: 10.1016/j.ebiom.2018.11.059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhang J, Teng Z, Song Y, Hu M and Chen C (2015) Inhibition of monoacylglycerol lipase prevents chronic traumatic encephalopathy-like neuropathology in a mouse model of repetitive mild closed head injury. J Cereb Blood Flow Metab 35:443–53. doi: 10.1038/jcbfm.2014.216 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Selkoe DJ (2002) Alzheimer’s disease is a synaptic failure. Science 298:789–91. doi: 10.1126/science.1074069 [DOI] [PubMed] [Google Scholar]
- 43.Long JZ, Li W, Booker L, Burston JJ, Kinsey SG, Schlosburg JE, Pavon FJ, Serrano AM, Selley DE, Parsons LH, Lichtman AH and Cravatt BF (2009) Selective blockade of 2-arachidonoylglycerol hydrolysis produces cannabinoid behavioral effects. Nat Chem Biol 5:37–44. doi: 10.1038/nchembio.129 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Long JZ, Nomura DK and Cravatt BF (2009) Characterization of monoacylglycerol lipase inhibition reveals differences in central and peripheral endocannabinoid metabolism. Chem Biol 16:744–53. doi: 10.1016/j.chembiol.2009.05.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Song Y, Zhang J and Chen C (2015) Fine-tuning of synaptic upscaling at excitatory synapses by endocannabinoid signaling is mediated via the CB1 receptor. Sci Rep 5:16257. doi: 10.1038/srep16257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Katona I and Freund TF (2008) Endocannabinoid signaling as a synaptic circuit breaker in neurological disease. Nat Med 14:923–30. doi: 10.1038/nm.f.1869 [DOI] [PubMed] [Google Scholar]
- 47.Heifets BD and Castillo PE (2009) Endocannabinoid signaling and long-term synaptic plasticity. Annu Rev Physiol 71:283–306. doi: 10.1146/annurev.physiol.010908.163149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Crimins JL, Rocher AB, Peters A, Shultz P, Lewis J and Luebke JI (2011) Homeostatic responses by surviving cortical pyramidal cells in neurodegenerative tauopathy. Acta Neuropathol 122:551–64. doi: 10.1007/s00401-011-0877-0 [DOI] [PubMed] [Google Scholar]
- 49.Pan B, Wang W, Zhong P, Blankman JL, Cravatt BF and Liu QS (2011) Alterations of endocannabinoid signaling, synaptic plasticity, learning, and memory in monoacylglycerol lipase knock-out mice. J Neurosci 31:13420–30. doi: 10.1523/jneurosci.2075-11.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang Z, Wang W, Zhong P, Liu SJ, Long JZ, Zhao L, Gao HQ, Cravatt BF and Liu QS (2015) Blockade of 2-arachidonoylglycerol hydrolysis produces antidepressant-like effects and enhances adult hippocampal neurogenesis and synaptic plasticity. Hippocampus 25:16–26. doi: 10.1002/hipo.22344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fowler CJ (2012) Monoacylglycerol lipase - a target for drug development? Br J Pharmacol 166:1568–85. doi: 10.1111/j.1476-5381.2012.01950.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gil-Ordonez A, Martin-Fontecha M, Ortega-Gutierrez S and Lopez-Rodriguez ML (2018) Monoacylglycerol lipase (MAGL) as a promising therapeutic target. Biochem Pharmacol 157:18–32. doi: 10.1016/j.bcp.2018.07.036 [DOI] [PubMed] [Google Scholar]
- 53.Grabner GF, Zimmermann R, Schicho R and Taschler U (2017) Monoglyceride lipase as a drug target: At the crossroads of arachidonic acid metabolism and endocannabinoid signaling. Pharmacol Ther 175:35–46. doi: 10.1016/j.pharmthera.2017.02.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mulvihill MM and Nomura DK (2013) Therapeutic potential of monoacylglycerol lipase inhibitors. Life Sci 92:492–7. doi: 10.1016/j.lfs.2012.10.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Panikashvili D, Mechoulam R, Beni SM, Alexandrovich A and Shohami E (2005) CB1 cannabinoid receptors are involved in neuroprotection via NF-kappa B inhibition. J Cereb Blood Flow Metab 25:477–84. doi: 10.1038/sj.jcbfm.9600047 [DOI] [PubMed] [Google Scholar]
- 56.Dou F, Netzer WJ, Tanemura K, Li F, Hartl FU, Takashima A, Gouras GK, Greengard P and Xu H (2003) Chaperones increase association of tau protein with microtubules. Proc Natl Acad Sci U S A 100:721–6. doi: 10.1073/pnas.242720499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Woo JA, Liu T, Zhao X, Trotter C, Yrigoin K, Cazzaro S, Narvaez E, Khan H, Witas R, Bukhari A, Makati K, Wang X, Dickey C and Kang DE (2017) Enhanced tau pathology via RanBP9 and Hsp90/Hsc70 chaperone complexes. Hum Mol Genet 26:3973–3988. doi: 10.1093/hmg/ddx284 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Abisambra J, Jinwal UK, Miyata Y, Rogers J, Blair L, Li X, Seguin SP, Wang L, Jin Y, Bacon J, Brady S, Cockman M, Guidi C, Zhang J, Koren J, Young ZT, Atkins CA, Zhang B, Lawson LY, Weeber EJ, Brodsky JL, Gestwicki JE and Dickey CA (2013) Allosteric heat shock protein 70 inhibitors rapidly rescue synaptic plasticity deficits by reducing aberrant tau. Biol Psychiatry 74:367–74. doi: 10.1016/j.biopsych.2013.02.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Luo W, Dou F, Rodina A, Chip S, Kim J, Zhao Q, Moulick K, Aguirre J, Wu N, Greengard P and Chiosis G (2007) Roles of heat-shock protein 90 in maintaining and facilitating the neurodegenerative phenotype in tauopathies. Proc Natl Acad Sci U S A 104:9511–6. doi: 10.1073/pnas.0701055104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Ittner LM, Ke YD, Delerue F, Bi M, Gladbach A, van Eersel J, Wölfing H, Chieng BC, Christie MJ, Napier IA, Eckert A, Staufenbiel M, Hardeman E and Götz J (2010) Dendritic function of tau mediates amyloid-beta toxicity in Alzheimer’s disease mouse models. Cell 142:387–97. doi: 10.1016/j.cell.2010.06.036 [DOI] [PubMed] [Google Scholar]
- 61.Shipton OA, Leitz JR, Dworzak J, Acton CE, Tunbridge EM, Denk F, Dawson HN, Vitek MP, Wade-Martins R, Paulsen O and Vargas-Caballero M (2011) Tau protein is required for amyloid {beta}-induced impairment of hippocampal long-term potentiation. J Neurosci 31:1688–92. doi: 10.1523/jneurosci.2610-10.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Leger M, Quiedeville A, Bouet V, Haelewyn B, Boulouard M, Schumann-Bard P and Freret T (2013) Object recognition test in mice. Nat Protoc 8:2531–7. doi: 10.1038/nprot.2013.155 [DOI] [PubMed] [Google Scholar]
- 63.Antunes M and Biala G (2012) The novel object recognition memory: neurobiology, test procedure, and its modifications. Cogn Process 13:93–110. doi: 10.1007/s10339-011-0430-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Botton PH, Costa MS, Ardais AP, Mioranzza S, Souza DO, da Rocha JB and Porciúncula LO (2010) Caffeine prevents disruption of memory consolidation in the inhibitory avoidance and novel object recognition tasks by scopolamine in adult mice. Behav Brain Res 214:254–9. doi: 10.1016/j.bbr.2010.05.034 [DOI] [PubMed] [Google Scholar]