Abstract
Purpose:
Biomarkers of response and resistance to FLT3 tyrosine kinase inhibitors (TKIs) are still emerging and optimal clinical combinations remain unclear. The purpose of this study is to identify co-occurring mutations that influence clinical response to the novel FLT3 inhibitor pexidartinib (PLX3397).
Experimental Design:
We performed targeted sequencing of pre-treatment blasts from 29 patients with FLT3 internal tandem duplication (ITD) mutations treated on the phase I/II trial of pexidartinib in relapsed/refractory FLT3-ITD+ AML. We sequenced 37 samples from 29 patients with available material, including 8 responders and 21 non-responders treated at or above the recommended phase 2 dose of 3000mg.
Results:
Consistent with other studies, we identified mutations in NRAS, TP53, IDH2 and a variety of epigenetic and transcriptional regulators only in non-responders. Among the most frequently mutated genes in non-responders was CCND3. A total of 3 individual mutations in CCND3 (Q276*, S264R, and T283A) were identified in 2/21 non-responders (one patient had both Q276* and S264R). No CCND3 mutations were found in pexidartinib responders. Expression of the Q276* and T283A mutations in FLT3-ITD MV4;11 cells conferred resistance to apoptosis, decreased cell cycle arrest, and increased proliferation in the presence of pexidartinib and other FLT3 inhibitors. Inhibition of CDK4/6 activity in CCND3 mutant MV4;11 cells restored pexidartinib-induced cell cycle arrest but not apoptosis.
Conclusions:
Mutations in CCND3, a gene not commonly mutated in AML, are a novel cause of clinical primary resistance to FLT3 inhibitors in AML and may have sensitivity to CDK4/6 inhibition.
Translational Relevance
Systematic genomic interrogation of patients treated on clinical trials of novel cancer therapeutics is necessary to uncover response biomarkers that can direct treatment to patients most likely to benefit and reveal potential targets for combination therapy. In a phase I/II trial of the FLT3 inhibitor pexidartinib in acute myeloid leukemia (AML), we observed that recurrent mutations in NRAS, IDH2, TP53, and other epigenetic and transcriptional regulators are associated with poor response to pexidartinib. In addition, recurrent oncogenic mutations in the gene encoding cyclin D3 (CCND3) are associated with primary resistance to pexidartinib. CCND3 mutations cause decreased cell cycle arrest and reduced apoptosis with FLT3 inhibitor treatment, representing a novel mechanism of resistance to FLT3 inhibitors. CDK4/6 inhibition with palbociclib can restore cell cycle arrest in response to FLT3 inhibition. Combined CKD4/6 and FLT3 inhibition may be a promising potential therapeutic strategy in FLT3-mutant AML.
Introduction
Genomic studies have revealed the mutational landscape of de novo acute myeloid leukemia (AML) and shed light on the impact of somatic mutations on prognosis(1,2). Constitutively activating mutations in FMS-Like Tyrosine Kinase 3 (FLT3), most commonly internal tandem duplication (ITD) mutations in the juxtamembrane domain or point mutations in the tyrosine kinase domain (TKD), are the most frequently observed mutations in AML(2,3). FLT3-ITD mutations are particularly associated with poor prognosis(4,5). A randomized study of the Type II FLT3 tyrosine kinase inhibitor (TKI) quizartinib compared with chemotherapy in relapsed/refractory (R/R) FLT3-ITD+ AML (QuANTUM-R) demonstrated superior survival for patients treated with quizartinib, underscoring the clinical efficacy of FLT3 TKIs(6). However, duration of response to quizartinib and other Type II FLT3 inhibitors(7–11) (which bind only the inactive kinase conformation) is limited by development of acquired resistance-conferring FLT3 kinase domain (KD) mutations. KD mutations, particularly at the D835 activation loop residue, are a commonly reported mechanism of clinical resistance to Type II FLT3 inhibitors such as quizartinib and sorafenib(7–11), which bind only the inactive kinase conformation. Type I FLT3 inhibitors such as crenolanib and gilteritinib, which bind the active kinase conformation, can circumvent resistance caused by FLT3 D835 mutations(12,13) and have demonstrated clinical activity in R/R FLT3-mutated AML(14,15). A randomized phase 3 study of gilteritinib (ADMIRAL) compared to salvage chemotherapy (NCT02421939) in AML showed a significant overall survival benefit for gilteritinib compared to chemotherapy (9.3 months vs 5.6 months)(16), leading to the Food and Drug Administration (FDA) approval of gilteritinib in FLT3-mutated R/R AML.
Systematic translational studies of patients treated with targeted therapy are essential to understand predictors of clinical response and elucidate clinically relevant resistance mechanisms. Sequencing of patients treated with crenolanib or gilteritinib therapy have revealed that alternative, non-FLT3-dependent resistance mechanisms are responsible for the majority of clinical resistance to these Type I FLT3 inhibitors. Ras pathway mutations appear to be a particularly common mechanism of resistance to both gilteritinib and crenolanib. In a recent report from our group, treatment-emergent Ras/MAPK pathway mutations were acquired in 15 of 41 patients (36.6%) treated with gilteritinib, including activating mutations in NRAS (13/41 patients; 31.7%), KRAS (3/41 patients; 7.3%), PTPN11 (3/41 patients; 7.3%), CBL (2/41 patients; 4.9%), and BRAF (1/41 patients; 2.4%)(17). Two other patients in this study developed new BCR-ABL1 fusions, in keeping with other reports(17,18). In a similar report of crenolanib resistance, mutations in RAS pathway genes including NRAS, KRAS, PTPN11 and CBL were noted in poor responders and/or were acquired or expanded on crenolanib treatment(19). In addition, mutations in a variety of genes involved in epigenetics (IDH1/2, TET2), splicing (U2AF1), cohesin (STAG2), transcription (ASXL1, CEBPA) and TP53 pathway (TP53, PPM1D) were associated with poor response to crenolanib(19). In contrast, a minority of patients developed resistance due to FLT3 KD mutations, though the FLT3 F691L “gatekeeper” mutation did emerge in 2 of 26 and 5 of 41 patients resistant to crenolanib and gilteritinib, respectively(17,19). The role of these non-FLT3 dependent “off-target” mechanisms in Type II FLT3 inhibitor resistance has not been well-characterized, with previous studies instead suggesting that “on-target” FLT3 KD mutations are the major mechanism of resistance to this class of FLT3 inhibitors(11,20).
Pexidartinib (PLX3397) is a Type II, selective small molecule kinase inhibitor of CSF1R, KIT and FLT3-ITD with unique activity against the FLT3 F691L mutation. Based on its activity against CSF1R, pexidartinib was recently approved for the treatment of advanced tenosynovial giant cell tumor, a neoplasm whose growth is related to CSF1R. In preclinical studies, pexidartinib demonstrated equipotent activity in cells expressing FLT3-ITD/F691L mutations compared to cells expressing native FLT3-ITD, though it remains vulnerable to FLT3 D835 mutations(11). A phase 1/2 open-label, sequential dose escalation study of continuous oral administration of pexidartinib demonstrated clinical activity in R/R FLT3-ITD+ AML(21). In an effort to understand the impact of coexisting mutations on response pexidartinib therapy, we performed capture-based sequencing of 585 malignancy-associated genes in pre-treatment blasts isolated from patients enrolled on a phase I/II trial of pexidartinib in relapsed/refractory AML (NCT01349049) with the goal of identifying mutations associated with resistance to pexidartinib.
Materials and Methods
Patient Samples.
These studies were conducted according to the Declaration of Helsinki guidelines. Samples were obtained following written informed consent from patients enrolled on the Phase I/II clinical trial of pexidartinib in relapsed or refractory AML (NCT01349049). Collection and analysis of samples was approved by the UCSF institutional review board (IRB) and the individual IRB of all institutions participating in the above-referenced pexidartinib clinical trial. Details of the clinical trial and results are reported elsewhere(21). We sequenced a total of 29 unique patients: 8 responders, 21 non-responders (Table S1). Response to treatment was evaluated using a modified version of the International Working Group (IWG) response criteria as described previously(21). Any patient not achieving a protocol-defined response was considered a non-responder. Patient blasts and T-cells were isolated from pre-treatment blood or bone marrow samples by fluorescence-activated cell sorting (FACS) for cell surface markers (see Figure S1). All responders with available material (defined as having either a pre-treatment FACS-purified blast sample or pre-treatment mononuclear cells containing leukemia in addition to a FACS-purified T cell somatic control) were sequenced using our custom capture panel (n=8). Samples from non-responder patients with available material and who were treated at or above the recommended phase 2 dose (RP2D) of 3000mg were sequenced (n=21). Samples from additional time points were sequenced when available. All patients had a history of detectable FLT3-ITD mutations prior to study enrollment and were considered relapsed or refractory after at least one line of AML chemotherapy. Targeted sequencing of CCND3 was done after PCR amplification from pre-treatment genomic DNA samples in an additional 21 patients treated at 3000mg of pexidartinib and above using primers CCND3 E5F ATGTGTTGGGAGCTGTCCTT and CCND3 E5R GGTAGGACCAGATCCCTTGG.
Genomic Sequencing and Analysis.
Targeted next generation sequencing of 585 genes (Table S2) with a known role in malignant transformation was conducted using the Memorial Sloan Kettering integrated mutation profiling of actionable cancer targets panel (MSK IMPACT) as previously described(22). To call FLT3-ITD, PINDEL v2.3 was used and results were restricted to duplication events present in the canonical ITD sites in more than 10 reads with at least one read from each strand. Mutational calls were made by alignment to common unmatched normal samples and then filtered using matched sorted T cells for single nucleotide polymorphisms and events with similar variant allele frequencies.
Cell Lines.
MV4;11 cells were obtained from the laboratory of Scott Kogan and authenticated by Promega STR analysis. All cell lines were documented to be mycoplasma-free by PCR testing 2–4 times per year. Cells were thawed and used within approximately 3 months for the indicated experiments. CCND3 targeted shRNA sequence GCTTCTCCTGTGTGATTGACA was cloned into plasmid pMK1222 (kind gift of J. Weissman lab). Plasmid containing the full length CCND3 cDNA cloned into pDONR221 backbone was obtained from the Harvard PlasmID Repository (Boston, MA). Mutations were introduced by Quikchange site directed mutagenesis (Agilent Technologies; Santa Clara, CA) per manufacturer’s protocol and cloned into MSCV puro plasmid using Gateway recombination (ThermoFisher; Waltham, MA). Stable MV4;11 cell lines were generated by lentiviral infection of CCND3 targeted shRNA followed by retroviral infection with empty vector or the appropriate mutated CCND3 plasmid as previously described(20).
Cell Proliferation, Apoptosis and Cell Cycle Analysis.
Pexidartinib was obtained under material transfer agreement from Plexxikon Inc. AC220 (quizartinib), palbociclib, gilteritinib, midostaurin, crenolanib, CT7001 (CDK7 inhibitor), and BAY1251152 (CDK9 inhibitor) were purchased from Selleckchem (Houston, TX) and sorafenib was purchased from LC Labs (Woburn, MA). For proliferation assays, exponentially growing cells were seeded at a concentration of 2 × 105/cells per mL in the presence of inhibitor at the indicated concentration in RPMI + 10% FCS on day 0 and counted every 2–3 days for the indicated time period. Cells were diluted as necessary to maintain a maximal cell density of < 2 ×106 cells/mL to allow for exponential grown and the appropriate dilution factor was factored in to obtain total cell numbers. Fold proliferation was calculated by normalizing cell numbers on each day to starting cell number on day 0.
For assessment of apoptosis, exponentially growing cells were plated in the presence of inhibitor at the indicated concentration in RPMI + 10% FCS for 48 hours. Cells were fixed with 4% paraformaldehyde (Electron Microscopy Sciences; Hatfield, PA) and permeabilized with 100% methanol (Electron Microscopy Sciences; Hatfield, PA) followed by staining with Alexa Fluor 647-conjugated anti-active caspase-3 antibody (BD Pharmingen; San Jose, CA). Cells were run on a LSRFortessa (BD Pharmingen; San Jose, CA) cell analyzer and data was analyzed using FlowJo (Tree Star Inc; Ashland, OR). Percentage of live cells was determined by negative staining for activated caspase-3.
For cell cycle analysis, exponentially growing cells were plated in the presence of inhibitor at the indicated concentration in RPMI + 10% FCS for 24 hours. Cells were then incubated for 2 hours in EdU (5-ethynyl-2´-deoxyuridine) and stained using the Click-iT Plus EdU Alexa Fluor 647 Flow Cytometry Kit (ThermoFisher; Santa Clara, CA) and DAPI (4’,6-Diamidino-2-Phenylindole, Dihydrochloride) per manufacturer’s protocol. Cells were run on a LSRFortessa (BD Pharmingen; San Jose, CA) cell analyzer and analyzed for DNA content distribution using FlowJo (Tree Star Inc; Ashland, OR).
Immunoblotting.
Exponentially growing MV4;11 cells stably expressing CCND3 isoforms were plated in RPMI medium 1640 + 10% FCS supplemented with inhibitor at the indicated concentration. After the indicated incubation time, the cells were washed in phosphate buffered saline and lysed and processed as previously described(23). Immunoblotting was performed using indicated antibodies from Cell Signaling Technologies (Beverly, MA) anti-cyclin D3 (2936S), anti-CDK4 (12790S), anti-CDK6 (3136S), anti-phospho-RB (9307S), anti-RB (9313S), anti-phospho-STAT5 (9359S), anti-STAT5 (94205S), anti-phospho-ERK (9101S), anti-ERK(9107S), anti-phospho-AKT (4060S), anti-AKT (9272S), anti-cyclin D2 (3741S), anti-actin (4967S), and anti-GAPDH (sc-25778; Santa Cruz Biotechnology; Dallas, TX). Data shown are representative of multiple experiments.
Results
Mutations Identified in Pexidartinib-Treated FLT3-ITD AML Patients
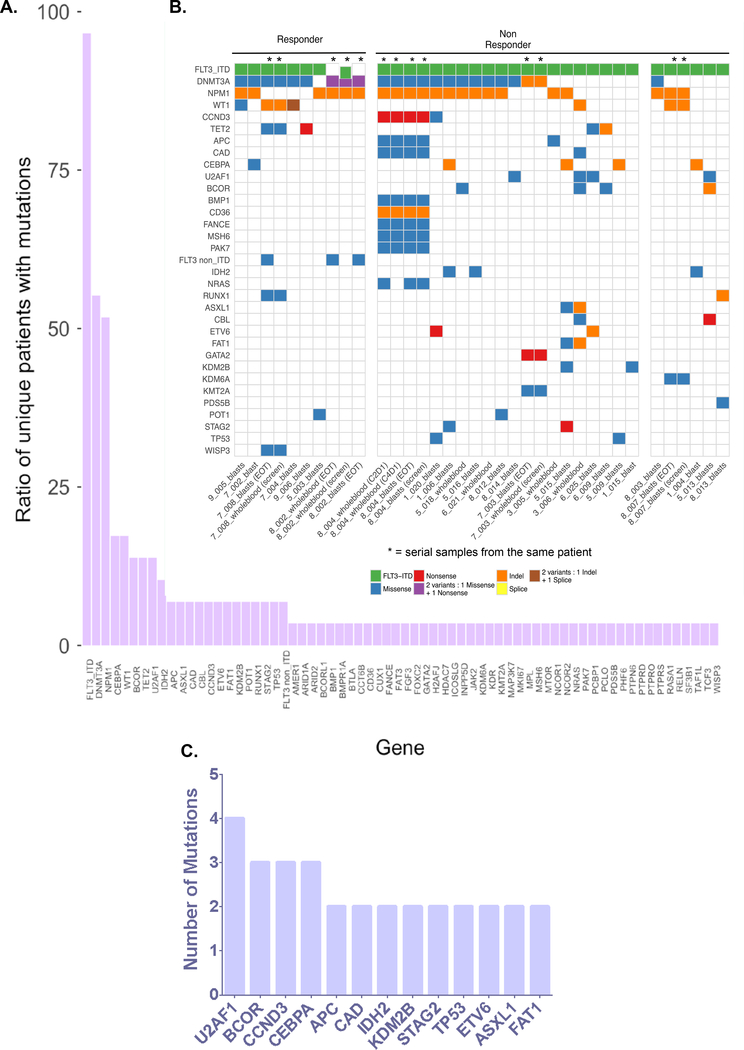
We identified 225 mutations in 68 different genes in 37 samples from 29 unique patients (Figure 1 and Table S3). 24 patients had only 1 evaluable sample (from the pre-treatment time point), 3 patients had 2 evaluable samples (from 2 time points), 1 patient had 3 evaluable samples (from 2 time points) and 1 patient had 4 evaluable samples (from 4 time points). All patients had a FLT3-ITD mutation identified by next generation sequencing (Table S4) in the pre-treatment sample. One patient (8_002) lost the FLT3-ITD mutation at the relapse/end-of-treatment timepoint, as has been previously described at relapse on other FLT3 inhibitors(17). An average of 5 mutations were observed per sample (range 3 to 14).
Figure 1. Mutations in FLT3-ITD+ Relapsed/Refractory AML Patients.
(A) Ratio of patients with mutations per gene. (B) Distribution of somatic mutations in individual patients organized by pexidartinib response status. Screen = pre-treatment (screening) time point; EOT = end of treatment time point; C2D1 = cycle 2 day 1; C4D1 = cycle 4 day 1; whole blood = sample from unsorted peripheral blood; blasts = sample from sorted blasts. (C) Mutations identified recurrently in pexidartinib non-responders.
The range of mutations identified in this patient cohort was similar to that described in large sequencing studies of newly diagnosed AML patients(1,2). The frequencies of AML classifications were similar to another published cohort of FLT3-ITD AML patients treated with chemotherapy23, but also included mutations in genes not commonly identified in AML (Figure 1A and 1B). Classification of mutations revealed clustering of genes in functional classes previously linked to AML (Figures 1B and S2). Genomic classification based on patterns of co-mutation using a previously defined algorithm(1) revealed enrichment for patients with co-occurring NPM1 mutations (known to co-occur with FLT3-ITD)(1) as well as a high frequency of mutations in chromatin-modification and spliceosome genes (Figure S2) similar to that observed in chemotherapy-treated FLT3-ITD patients(24).
The pattern of mutations associated with no response to pexidartinib (Figure 1B–C) was consistent with mutation patterns reported in prior studies of crenolanib and gilteritinib resistance(17,19). Similar to crenolanib(19) and gilteritinib(17), mutations in MAPK pathway genes NRAS (1/21, 4.8%) and CBL (2/21; 9.5%) were observed in pexidartinib non-responders. Mutations in TP53 (2/21; 9.5%) and IDH2 (3/21; 14.3%), which have been shown to confer resistance to crenolanib(19), were also exclusively observed in pexidartinib non-responders. Similar to the pattern in crenolanib poor-responders(19), mutations in cohesin complex members, splicing factors, epigenetic regulators and/or transcription factors such as U2F1, STAG2, ASXL1, BCOR and CEBPA were amongst the most common recurrently mutated genes in pexidartinib non-responders (Figure 1C). In contrast to crenolanib, where TET2 frameshift/nonsense mutations were associated with poor response(19), TET2 mutations did not segregate with differential pexidartinib response (Figure 1B).
No Newly Mutated Off-Target Genes Identified At Relapse on Pexidartinib
Two responders (7_008 and 8_002) had sequencing of both pre-treatment and relapse samples allowing the identification of mutations associated with acquired pexidartinib resistance. In each case of acquired resistance, new FLT3 TKD mutations known to confer resistance to pexidartinib were observed with high variant allele frequency in the relapse sample (patient 7_008: N676K, VAF=0.41 and patient 8_002: D835Y, VAF=0.42; Figure S3), supporting the central role of on-target mutations in acquired FLT3 inhibitor resistance. These results are consistent with a prior report of these samples from our group using a different deep sequencing platform(11). Significantly, no newly mutated genes were detectable in relapse samples from either patient, though patient 7_008 did acquire second distinct mutations in TET2 and WT1 at the time of resistance (Figure S3) demonstrating clonal evolution in the dominant clone at relapse In the 3 remaining patients sequenced at multiple time points (non-responders), mutations and mutation frequencies were largely stable over time (Figure S3), suggesting the absence of significant changes in clonal dynamics with inhibitor treatment in these patients.
Recurrent CCND3 Mutations Are Associated with Primary Resistance to Pexidartinib
We next turned our attention to potential mediators of primary resistance. Among pexidartinib non-responders, in addition to genes known to be commonly mutated in AML, we observed recurrent mutations in CCND3 (VAF range: 0.06 to 0.39), APC (VAF range: 0.28 to 0.49), CAD (VAF range: 0.43 to 0.53), and KDM2B (VAF range: 0.43 to 0.48) (Figure 1C). Mutations in these genes are not commonly implicated in AML or myeloid malignancy pathogenesis, though it is also likely that most prior targeted genomic sequencing studies in AML have not included these genes. Another recurrently mutated gene FAT1 (VAF range: 0.46 to 0.42) is also infrequently mutated in AML, but mutations have been previously described in AML patients with FLT3-ITD mutations(24). While mutations in the tumor suppressor gene APC are well-described in colorectal cancer (CRC), they have not been observed in AML(25) and the APC mutations observed in this cohort did not occur at mutational hotspots associated with CRC. Dysregulation of the histone demethylase KDM2B has been linked to a variety of cancers, including hematologic malignancies(26). However, KDM2B mutations have not been observed in AML nor proven to be oncogenic in other malignancies. Similarly, CAD gene amplification has been associated with deficiencies in DNA mismatch repair in cancer cell lines(27), but mutations in CAD are not known to be pathogenic in any cancer. Though the mutations in APC, CAD, KDM2B and FAT1 observed in our cohort are not known oncogenic mutations (Figure S4), we did observe known oncogenic mutations in CCND3, the gene encoding cyclin D3. Mutations in CCND3 were found in 2 non-responders, including one patient (1_020) with 2 distinct CCND3 mutations (T283A and S264R; Table 1). Among mutations found exclusively in non-responders, mutations in CCND3 occurred at the second highest frequency of any gene (Figure 1C). Mutations in CCND3 have rarely been reported in AML(28,29) and have not been identified in larger AML genomic sequencing efforts(1,2,24).
Table 1.
CCND3 Mutations Identified in Pexidartinib Non-Responders
| SAMPLE ID | CHR | POSITION | GENE | cDNA CHANGE | PROTEIN CHANGE | DEPTH | VAF | EFFECT |
|---|---|---|---|---|---|---|---|---|
| 1_020_blasts | 6 | 41903765 | CCND3 | c.792C>A | p.S264R | 753 | 0.065450644 | Non synonymous codon |
| 6 | 41903710 | CCND3 | c.847A>G | p.T283A | 835 | 0.141187926 | Non synonymous codon | |
| 8_004_blasts (screen) | 6 | 41903731 | CCND3 | c.826C>T | p.Q276* | 1174 | 0.338524035 | Stop gained |
| 8_004_wholeblood (C2D1) | 6 | 41903731 | CCND3 | c.826C>T | p.Q276* | 1179 | 0.357380688 | Stop gained |
| 8_004_wholeblood (C4D1) | 6 | 41903731 | CCND3 | c.826C>T | p.Q276* | 1302 | 0.394010113 | Stop gained |
| 8_004_blasts (EOT) | 6 | 41903731 | CCND3 | c.826C>T | p.Q276* | 955 | 0.391138637 | Stop gained |
While rare in AML, recurrent missense, nonsense and frameshift mutations in CCND3 have been frequently identified in sporadic and HIV-associated Burkitt lymphoma and to a lesser degree in diffuse large B cell lymphoma (DLBCL)(30). Missense mutations in the conserved threonine 283 (T283) residue are known to affect D-type cyclin phosphorylation and stability(31) and, along with nonsense/frameshift mutations in the C terminus of cyclin D3, result in accumulation of mutant isoforms associated with increased proliferative advantage in Burkitt lymphoma cells(30). Importantly, both the Q276* and T283A mutations identified in pexidartinib-resistant patients are recurrently observed mutations in Burkitt lymphoma and DLBCL(30).
Functional Validation of Cyclin D3 Mutations as a Mechanism of Resistance to Pexidartinib
Mutation or overexpression of cyclin D3 have not been previously associated with FLT3 inhibitor resistance. However, D cyclins have been shown to function downstream of FLT3-ITD mediated signaling. Treatment of FLT3-ITD driven cells with pharmacologic FLT3 inhibitors results in downregulation of cyclin D2 and D3, retinoblastoma protein (RB) dephosphorylation and cell cycle arrest(32). Inhibition of CDK4/6 activity in these cells can impair proliferation and induce cell cycle arrest(32). In light of these findings, we hypothesized that CCND3 mutations resulting in cyclin D3 stabilization and overexpression may abrogate downstream cell cycle arrest induced by FLT3 inhibitor treatment.
To determine whether cyclin D3 expression is necessary for the proliferation of FLT3-ITD driven cells, we first performing shRNA knockdown of cyclin D3 in the FLT3-ITD mutant MV4;11 cell line and measured proliferation (Figure 2A and 2B). Knockdown of cyclin D3 mildly impaired proliferation of MV4;11 cells over time, suggesting cyclin D3 is not an essential mediator of cell cycle progression in these cells. Overexpression of wild type or mutant cyclin D3 in cells with knockdown of native cyclin D3 restored proliferation to levels commensurate with parental MV4;11 cells (Figure 2B).
Figure 2. Cyclin D3 is Dispensable To Proliferation of MV4;11 Cells but Cyclin D3 Mutations Confer Resistance to FLT3 TKI Treatment.
(A) Western blot analysis of cyclin D3 expression in MV4;11 cells after knockdown and overexpression of empty vector or indicated cyclin D3 isoforms. (B) Fold proliferation of indicated cyclin D3 shRNA and cyclin D3 isoform-expressing MV4;11 cells over a sixteen day period. EV=empty vector, WT=wild type. Error bars represent standard deviation of 3 technical replicates. Curves shown are representative of multiple experiments. (C) Fold proliferation of cyclin D3 knockdown and overexpressing MV4;11 cells over the indicated time period in the presence of 4nM AC220 (quizartinib) or 1000nM PLX3397 (pexidartinib) (p-values derived from one-way ANOVA comparison of Q276* and T283A expressing cells to EV). Error bars represent standard deviation of 3 technical replicates. Curves shown are representative of multiple experiments. (D) Western blot analysis of indicated cyclin D3 knockdown and overexpressing of MV4;11 cells 2, 6 and 24 hours after exposure to 1000 nM of PLX3397. Western Blot analysis using anti-cyclin D3, anti-CDK4, anti-CDK6, anti-phospho-Rb, anti-Rb, anti-actin, anti-phospho-ERK, anti-phospho-AKT, anti-phospho-STAT5, anti-ERK, anti-AKT and anti-STAT5 antibodies performed on whole cell lysates. Data shown representative of multiple experiments. EV=empty vector, WT=wild type.
In contrast, cells overexpressing cyclin D3 Q276* and T283A mutations exhibited increased proliferative capacity and decreased cell cycle arrest in the setting of treatment with pexidartinib and quizartinib (Figure 2C, 3A and S5). Similar increased proliferation was observed in the presence of other clinically relevant FLT3 inhibitors gilteritinib, crenolanib and midostaurin (Figure S5). Cells overexpressing wild type (WT) cyclin D3 also exhibited decreased cell cycle arrest in the setting of FLT3 TKI treatment, which did not translate to significantly increased proliferation with drug exposure compared to empty vector (EV) control (Figure 2C). Interestingly, overexpression of both WT and mutant cyclin D3 isoforms conferred protection from apoptosis induced by a variety of FLT3 inhibitors (Figure 3 and Figure S5). Consistent with the function of cyclin D3 downstream of FLT3-ITD signaling, MV4;11 cells expressing stabilizing cyclin D3 Q276* and T283A mutations treated with pexidartinib exhibited sustained cyclin D3 expression over time compared to cells expressing empty vector (EV) or WT cyclin D3, despite equivalent modulation of downstream FLT3 signaling as measured by STAT5, ERK and AKT phosphorylation (Figure 2D). CDK4 expression levels were upregulated in cyclin D3 Q276* and T283A expressing cells while CDK6 expression levels were decreased, suggesting a preferential role for active cyclin D3/CDK4 complexes in mediating proliferation in these cells. Overexpression of cyclin D3, particularly the Q276* and T283A mutant isoforms, resulted in increased RB phosphorylation that was sustained in the setting of pexidartinib treatment. In aggregate, these data suggest that, while not critical for FLT3-ITD mediated cell cycle progression, downstream overexpression and stabilization of cyclin D3 can impair FLT3 TKI-mediated cell cycle arrest and apoptosis.
Figure 3. CDK4/6 Inhibition Restores Cell Cycle Arrest and Inhibition of Proliferation in Response to FLT3 TKI Treatment.
(A) Cell cycle analysis of cyclin D3 knockdown and overexpressing MV4;11 cells after 24 hours of treatment with 4nM AC220 or 1000nM PLX3397 with and without 500nM palbociclib. (B) Fold proliferation of cyclin D3 overexpressing MV4;11 cells over 10 days in the presence of 1000nM PLX3397 and 500nM palbociclib (p-values derived from one-way ANOVA comparison of Q276* and T283A expressing cells to EV). Error bars represent standard deviation of 3 technical replicates. Curves shown are representative of multiple experiments. (C) Western blot analysis of indicated cyclin D3 expressing of MV4;11 cells after 20 hour exposure to 1000nM PLX3397 and 500nM palbociclib. Western Blot analysis using anti-cyclin D3, anti-CDK4, anti-CDK6, anti-phospho-Rb, anti-Rb, anti-GAPDH, anti-phospho-STAT5, and anti-STAT5 antibodies performed on whole cell lysates. Data shown represents a single experiment. (D) Average normalized percentage of live cells as assessed by caspase-3 activation at 48h following treatment with 1000 nM PLX3397 with and without palbociclib for MV4;11 cells after knockdown and overexpression of indicated cyclin D3 isoforms (error bars represent s.d. replicate experiments; n = 9; p-values derived from paired t-test).
CDK4/6 Inhibition is Insufficient to Overcome Resistance to Apoptosis Conferred by Cyclin D3 Mutations
The small molecule CDK4-CDK6 inhibitor palbociclib has demonstrated activity in combination with the aromatase inhibitor letrazole in the treatment of advanced estrogen receptor positive breast cancer(33). The proposed mechanism of action of CDK4/6 inhibition is prevention of RB phosphorylation, resulting in subsequent inactivation of RB-mediated transcriptional corepressor and chromatin modifier activity on genes required for cell cycle progression. The anti-proliferative effect of CDK4/6 inhibitors may be potentiated by signal transduction inhibitors, which cause downstream cyclin D downregulation. This therapeutic potential has led to the development of small molecule inhibitors targeted against both FLT3 and CDK4/6(34,35).
Given the central role of CDK4/6 activity in facilitating cyclin D-mediated cell cycle progression, we tested the activity of palbociclib in combination with FLT3 TKI treatment in cyclin D3 overexpressing MV4;11 cells. Palbociclib restored cell cycle arrest and inhibition of proliferation in cyclin D3 overexpressing cell lines in the presence of pexidartinib (Figure 3A and 3B). Similar inhibition of proliferation was observed when palbociclib was combined with other clinically-relevant FLT3 TKIs (Figure S5). Correspondingly, significant abrogation of RB phosphorylation was demonstrated only with palbociclib treatment in cyclin D3 overexpressing cell lines (Figure 3C), while FLT3 signaling as measured by STAT5 phosphorylation was suppressed in all pexidartinib-treated conditions. As before, downregulation of cyclin D3 Q276* and T283A isoforms was not observed with pexidartinib treatment, alone or in combination with palbociclib, despite clear downregulation of native cyclin D3 (MIG EV and CCND3 WT; Figure 3C) and cyclin D2 (all cell lines; Figure 3C). Similarly, total CDK4 (but not CDK6) levels, were clearly decreased with pexidartinib treatment alone or in combination with palbociclib, in the setting of native but not mutant cyclin D3 (Figure 3C). The addition of palbociclib to pexidartinib was insufficient to restore apoptosis in cyclin D3-overexpressing cell lines (Figure 3D), suggesting resistance to apoptosis conferred by cyclin D3 overexpression is independent of CDK4/6 kinase activity. Neither addition of a CDK7 or CDK9 inhibitor, expected to have broader activity to inhibit expression of anti-apoptotic proteins, could overcome resistance to pexidartinib induced by cyclin D3 overexpression (Figure S6).
Discussion
In this study, we have identified mutations in CCND3 as a novel mechanism of primary clinical resistance to FLT3 inhibitors in AML. CCND3 mutations have been rarely identified in AML to date. However, existing evidence suggests mutations in CCND3 and its related family members, CCND1 and CCND2, may be enriched in certain phenotypic or genetic contexts. A recent study of MLL-rearranged pediatric and adult AML patients identified CCND3 mutations in 5 pediatric MLL-rearranged AML patients (8.9%) and 2 adult MLL-rearranged AML patients (3.3%)(29). In this study, a low rate of recurrent mutations of CCND1 (n = 3, 2.9%) and CCND2 (n = 8, 7.6%) were found in pediatric t(8;21) AML patients but not in MLL-rearranged patients. These findings are consistent with another recent report of frequent mutations of CCND1 and CCND2 in patients with core-binding factor (CBF) AML but not in other AML subtypes(36). Of note, in our study, neither of the 2 patients with identified CCND3 mutations had co-existing MLL-rearrangements. Targeted sequencing of CCND3 in all patients with available material treated at the recommended phase II pexidartinib dose of 3000mg and above (an additional 21 patients) revealed no other patients with CCND3 mutations (data not shown).
Though CCND3 mutations appear to be a rare event in AML overall, the occurrence of these mutations in R/R FLT3-ITD AML patients with FLT3 inhibitor resistance is indicative that even rare mutations can have important consequences that impact response to therapy. Additionally, the recent discoveries of CCND1 and CCND2 mutations in CBF AML(36) and CCND3 mutations in MLL-rearranged AML(29) suggest that the cyclin D axis may have pathogenic significance and serve as a potential therapeutic target in certain AML subtypes. In this way, rare but recurrent pathway mutations can collectively implicate particular biological nodes for therapeutic targeting. Our current model places cyclin D3 downstream of FLT3-ITD signaling, mediating cellular proliferation though activation of CDK4/6, subsequent RB phosphorylation and cell cycle entry. Consistent with our data, a role for CDK4/6 inhibition has been suggested in in AML(29). While our data suggest that proliferative effects induced by cyclin D3 stabilization and overexpression can be mitigated by inhibition CDK4/6 kinase activity, the anti-apoptotic effect of cyclin D3 overexpression remains unaffected. It is known that molecular effects of cyclin D extend beyond its role in CDK activation(31) and overexpression of cyclin D has been previously implicated in resistance to chemotherapy-mediated apoptosis(37), suggesting the role of cyclin D3 mutations in mediating FLT3 inhibitor resistance is complex and multi-functional.
In addition to CCND3 mutations, we also identified mutations in genes previously associated with resistance to Type I FLT3 inhibitors in pexidartinib non-responders, suggesting a common spectrum of “off-target” mutations associated with FLT3 TKI resistance. These commonly mutated genes included members of the cohesin complex, splicing factors, epigenetic regulators and/or transcription factors. However, unlike with crenolanib, TET2 frameshift/nonsense mutations were not predictive of poor response to pexidartinib. As with crenolanib and gilteritinib, mutations in NRAS, CBL, IDH2 and TP53 were associated with poor response to pexidartinib. However, mutations in other genes involved in the Ras pathway (such as PTPN11) or other signaling mutations were not identified in our study. The lack of other Ras pathway mutations may be reflective of the small numbers in our study and limited number of samples with serial banked samples to assess for mutations enriched on treatment or with relapse.
It is likely that as more broadly targeted sequencing studies are done in AML patients treated with both targeted and chemotherapy, additional pathogenic abnormalities found at overall low frequency in AML will appear with higher prevalence in certain molecular subtypes and may impact response to both conventional and targeted therapy. Understanding the functional interplay between mutational events, genetic backdrop and treatment response is a complex, but critically important problem. Expanded genomic profiling of relapsed/refractory AML and development of experimental models incorporating complex mutation profiles within the context that they appear in patients is essential for optimization of future therapeutic strategies and to inform therapeutic decision-making.
Supplementary Material
Acknowledgements:
C. Smith is an ASH Faculty Scholar and a Damon Runyon-Richard Lumsden Foundation Clinical Investigator supported (in part) by the Damon Runyon Cancer Research Foundation (CI-99-18). This work was supported in part by grants from the NCI (1RO1 CA176091) (N. Shah), (K08 CA187577) (C. Smith) and by grant CA-1U54OD020355-01 to R. Levine. This work was also supported by a Research Scholar Grant from the American Cancer Society (132032-RSG-18-063-01-TBG) (C. Smith). N. Shah wishes to acknowledge the Edward S. Ageno Foundation for continued support. R. Levine is supported by National Cancer Institute award R35 CA197594-01A1. A. Viny. is supported by a National Cancer Institute career development grant K08 CA215317, the William Raveis Charitable Fund Fellowship of the Damon Runyon Cancer Research Foundation (DRG 117-15), and an EvansMDS Young Investigator grant from the Edward P. Evans Foundation. This work was supported in part by MSKCC Cancer Center Core Grant P30 CA008748.
Footnotes
Conflict of Interest Statement: N.P.S. has received research funding for the conduct of clinical trials from ARIAD Pharmaceuticals and Ambit Biosciences and research funding from Daiichi-Sankyo and Plexxikon, Inc. C.C.S. has received research funding from Plexxikon, Inc. and Astellas Pharma. R.L.L. is on the supervisory board of QIAGEN and is a scientific advisor to Loxo (until Feb 2019), Imago, C4 Therapeutics, and Isoplexis. He receives research support from and consulted for Celgene and Roche and has consulted for Lilly, Jubilant, Janssen, Astellas, Morphosys, and Novartis. He has received honoraria from Roche, Lilly, and Amgen for invited lectures and from Celgene and Gilead for grant reviews.
References
- 1.Papaemmanuil E, Gerstung M, Bullinger L, Gaidzik VI, Paschka P, Roberts ND, et al. Genomic Classification and Prognosis in Acute Myeloid Leukemia. N Engl J Med 2016;374(23):2209–21 doi 10.1056/NEJMoa1516192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cancer Genome Atlas Research N, Ley TJ, Miller C, Ding L, Raphael BJ, Mungall AJ, et al. Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N Engl J Med 2013;368(22):2059–74 doi 10.1056/NEJMoa1301689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Papaemmanuil E, Dohner H, Campbell PJ. Genomic Classification in Acute Myeloid Leukemia. N Engl J Med 2016;375(9):900–1 doi 10.1056/NEJMc1608739. [DOI] [PubMed] [Google Scholar]
- 4.Kottaridis PD, Gale RE, Frew ME, Harrison G, Langabeer SE, Belton AA, et al. The presence of a FLT3 internal tandem duplication in patients with acute myeloid leukemia (AML) adds important prognostic information to cytogenetic risk group and response to the first cycle of chemotherapy: analysis of 854 patients from the United Kingdom Medical Research Council AML 10 and 12 trials. Blood 2001;98(6):1752–9. [DOI] [PubMed] [Google Scholar]
- 5.Frohling S, Schlenk RF, Breitruck J, Benner A, Kreitmeier S, Tobis K, et al. Prognostic significance of activating FLT3 mutations in younger adults (16 to 60 years) with acute myeloid leukemia and normal cytogenetics: a study of the AML Study Group Ulm. Blood 2002;100(13):4372–80 doi 10.1182/blood-2002-05-1440. [DOI] [PubMed] [Google Scholar]
- 6.Cortes JE, Khaled S, Martinelli G, Perl AE, Ganguly S, Russell N, et al. Quizartinib versus salvage chemotherapy in relapsed or refractory FLT3-ITD acute myeloid leukaemia (QuANTUM-R): a multicentre, randomised, controlled, open-label, phase 3 trial. The Lancet Oncology 2019;20(7):984–97 doi 10.1016/S1470-2045(19)30150-0. [DOI] [PubMed] [Google Scholar]
- 7.Cortes J, Perl AE, Dohner H, Kantarjian H, Martinelli G, Kovacsovics T, et al. Quizartinib, an FLT3 inhibitor, as monotherapy in patients with relapsed or refractory acute myeloid leukaemia: an open-label, multicentre, single-arm, phase 2 trial. Lancet Oncol 2018. doi 10.1016/S1470-2045(18)30240-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cortes JE, Tallman MS, Schiller GJ, Trone D, Gammon G, Goldberg SL, et al. Phase 2b study of two dosing regimens of quizartinib monotherapy in FLT3-ITD mutated, relapsed or refractory AML. Blood 2018. doi 10.1182/blood-2018-01-821629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Baker SD, Zimmerman EI, Wang YD, Orwick S, Zatechka DS, Buaboonnam J, et al. Emergence of polyclonal FLT3 tyrosine kinase domain mutations during sequential therapy with sorafenib and sunitinib in FLT3-ITD-positive acute myeloid leukemia. Clin Cancer Res 2013. doi 10.1158/1078-0432.CCR-13-1323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Man CH, Fung TK, Ho C, Han HH, Chow HC, Ma AC, et al. Sorafenib treatment of FLT3-ITD(+) acute myeloid leukemia: favorable initial outcome and mechanisms of subsequent nonresponsiveness associated with the emergence of a D835 mutation. Blood 2012;119(22):5133–43 doi 10.1182/blood-2011-06-363960. [DOI] [PubMed] [Google Scholar]
- 11.Smith CC, Zhang C, Lin KC, Lasater EA, Zhang Y, Massi E, et al. Characterizing and Overriding the Structural Mechanism of the Quizartinib-Resistant FLT3 “Gatekeeper” F691L Mutation with PLX3397. Cancer Discov 2015;5(6):668–79 doi 10.1158/2159-8290.CD-15-0060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Smith CC, Lasater EA, Lin KC, Wang Q, McCreery MQ, Stewart WK, et al. Crenolanib is a selective type I pan-FLT3 inhibitor. Proc Natl Acad Sci U S A 2014. doi 10.1073/pnas.1320661111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lee LY, Hernandez D, Rajkhowa T, Smith SC, Raman JR, Nguyen B, et al. Preclinical studies of gilteritinib, a next-generation FLT3 inhibitor. Blood 2017;129(2):257–60 doi 10.1182/blood-2016-10-745133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Perl AE, Altman JK, Cortes J, Smith C, Litzow M, Baer MR, et al. Selective inhibition of FLT3 by gilteritinib in relapsed or refractory acute myeloid leukaemia: a multicentre, first-in-human, open-label, phase 1–2 study. Lancet Oncol 2017;18(8):1061–75 doi 10.1016/S1470-2045(17)30416-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cortes JE, Kantarjian HM, Kadia TM, Borthakur G, Konopleva M, Garcia-Manero G, et al. Crenolanib besylate, a type I pan-FLT3 inhibitor, to demonstrate clinical activity in multiply relapsed FLT3-ITD and D835 AML. Journal of Clinical Oncology 2016;34(15_suppl):7008- doi 10.1200/JCO.2016.34.15_suppl.7008. [DOI] [Google Scholar]
- 16.Perl AE, Martinelli G, Cortes JE, Neubauer A, Berman E, Paolini S, et al. Gilteritinib or Chemotherapy for Relapsed or Refractory FLT3-Mutated AML. N Engl J Med 2019;381(18):1728–40 doi 10.1056/NEJMoa1902688. [DOI] [PubMed] [Google Scholar]
- 17.McMahon CM, Ferng T, Canaani J, Wang ES, Morrissette JJ, Eastburn DJ, et al. Clonal selection with Ras pathway activation mediates secondary clinical resistance to selective FLT3 inhibition in acute myeloid leukemia. Cancer discovery 2019:CD-18-1453 doi 10.1158/2159-8290.Cd-18-1453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kasi PM, Litzow MR, Patnaik MM, Hashmi SK, Gangat N. Clonal evolution of AML on novel FMS-like tyrosine kinase-3 (FLT3) inhibitor therapy with evolving actionable targets. Leuk Res Rep 2016;5:7–10 doi 10.1016/j.lrr.2016.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang H, Savage S, Schultz AR, Bottomly D, White L, Segerdell E, et al. Clinical resistance to crenolanib in acute myeloid leukemia due to diverse molecular mechanisms. Nature communications 2019;10(1):244 doi 10.1038/s41467-018-08263-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Smith CC, Wang Q, Chin CS, Salerno S, Damon LE, Levis MJ, et al. Validation of ITD mutations in FLT3 as a therapeutic target in human acute myeloid leukaemia. Nature 2012;485(7397):260–3 doi 10.1038/nature11016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Smith CC, Levis MJ, Frankfurt O, Pagel JM, Roboz GJ, Stone RM, et al. A phase 1/2 study of the oral FLT3 inhibitor pexidartinib in relapsed/refractory FLT3-ITD-mutant acute myeloid leukemia. Blood Adv 2020;4(8):1711–21 doi 10.1182/bloodadvances.2020001449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cheng DT, Mitchell TN, Zehir A, Shah RH, Benayed R, Syed A, et al. Memorial Sloan Kettering-Integrated Mutation Profiling of Actionable Cancer Targets (MSK-IMPACT): A Hybridization Capture-Based Next-Generation Sequencing Clinical Assay for Solid Tumor Molecular Oncology. The Journal of molecular diagnostics : JMD 2015;17(3):251–64 doi 10.1016/j.jmoldx.2014.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Smith CC, Lasater EA, Zhu X, Lin KC, Stewart WK, Damon LE, et al. Activity of ponatinib against clinically-relevant AC220-resistant kinase domain mutants of FLT3-ITD. Blood 2013;121(16):3165–71 doi 10.1182/blood-2012-07-442871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Garg M, Nagata Y, Kanojia D, Mayakonda A, Yoshida K, Haridas Keloth S, et al. Profiling of somatic mutations in acute myeloid leukemia with FLT3-ITD at diagnosis and relapse. Blood 2015;126(22):2491–501 doi 10.1182/blood-2015-05-646240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Rowan AJ, Lamlum H, Ilyas M, Wheeler J, Straub J, Papadopoulou A, et al. APC mutations in sporadic colorectal tumors: A mutational “hotspot” and interdependence of the “two hits”. Proc Natl Acad Sci U S A 2000;97(7):3352–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Andricovich J, Kai Y, Peng W, Foudi A, Tzatsos A. Histone demethylase KDM2B regulates lineage commitment in normal and malignant hematopoiesis. J Clin Invest 2016;126(3):905–20 doi 10.1172/JCI84014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen S, Bigner SH, Modrich P. High rate of CAD gene amplification in human cells deficient in MLH1 or MSH6. Proc Natl Acad Sci U S A 2001;98(24):13802–7 doi 10.1073/pnas.241508098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yan XJ, Xu J, Gu ZH, Pan CM, Lu G, Shen Y, et al. Exome sequencing identifies somatic mutations of DNA methyltransferase gene DNMT3A in acute monocytic leukemia. Nat Genet 2011;43(4):309–15 doi 10.1038/ng.788. [DOI] [PubMed] [Google Scholar]
- 29.Matsuo H, Yoshida K, Fukumura K, Nakatani K, Noguchi Y, Takasaki S, et al. Recurrent CCND3 mutations in MLL-rearranged acute myeloid leukemia. Blood Adv 2018;2(21):2879–89 doi 10.1182/bloodadvances.2018019398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schmitz R, Young RM, Ceribelli M, Jhavar S, Xiao W, Zhang M, et al. Burkitt lymphoma pathogenesis and therapeutic targets from structural and functional genomics. Nature 2012;490(7418):116–20 doi 10.1038/nature11378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Musgrove EA, Caldon CE, Barraclough J, Stone A, Sutherland RL. Cyclin D as a therapeutic target in cancer. Nat Rev Cancer 2011;11(8):558–72 doi 10.1038/nrc3090. [DOI] [PubMed] [Google Scholar]
- 32.Wang L, Wang J, Blaser BW, Duchemin AM, Kusewitt DF, Liu T, et al. Pharmacologic inhibition of CDK4/6: mechanistic evidence for selective activity or acquired resistance in acute myeloid leukemia. Blood 2007;110(6):2075–83 doi 10.1182/blood-2007-02-071266. [DOI] [PubMed] [Google Scholar]
- 33.Finn RS, Martin M, Rugo HS, Jones S, Im S-A, Gelmon K, et al. Palbociclib and Letrozole in Advanced Breast Cancer. New England Journal of Medicine 2016;375(20):1925–36 doi doi: 10.1056/NEJMoa1607303. [DOI] [PubMed] [Google Scholar]
- 34.Li C, Liu L, Liang L, Xia Z, Li Z, Wang X, et al. AMG 925 is a dual FLT3/CDK4 inhibitor with the potential to overcome FLT3 inhibitor resistance in acute myeloid leukemia. Mol Cancer Ther 2015;14(2):375–83 doi 10.1158/1535-7163.MCT-14-0388. [DOI] [PubMed] [Google Scholar]
- 35.Li Z, Wang X, Eksterowicz J, Gribble MW Jr., Alba GQ, Ayres M, et al. Discovery of AMG 925, a FLT3 and CDK4 dual kinase inhibitor with preferential affinity for the activated state of FLT3. J Med Chem 2014;57(8):3430–49 doi 10.1021/jm500118j. [DOI] [PubMed] [Google Scholar]
- 36.Eisfeld AK, Kohlschmidt J, Schwind S, Nicolet D, Blachly JS, Orwick S, et al. Mutations in the CCND1 and CCND2 genes are frequent events in adult patients with t(8;21)(q22;q22) acute myeloid leukemia. Leukemia 2016. doi 10.1038/leu.2016.332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Biliran H Jr., Wang Y, Banerjee S, Xu H, Heng H, Thakur A, et al. Overexpression of cyclin D1 promotes tumor cell growth and confers resistance to cisplatin-mediated apoptosis in an elastase-myc transgene-expressing pancreatic tumor cell line. Clin Cancer Res 2005;11(16):6075–86 doi 10.1158/1078-0432.CCR-04-2419. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.