Abstract
Mesenchymal stem cells (MSCs), a kind of multipotent stem cells with self-renewal ability and multi-differentiation ability, have become the “practical stem cells” for the treatment of diseases. MSCs have immunomodulatory properties and can be used to treat autoimmune diseases, such as systemic lupus erythematosus (SLE) and Crohn’s disease. MSCs also can be used in cancer and aging. At present, many clinical experiments are using MSCs. MSCs can reduce the occurrence of inflammation and apoptosis of tissue cells, and promote the proliferation of endogenous tissue and organ cells, so as to achieve the effect of repairing tissue and organs. MSCs presumably also play an important role in Corona Virus Disease 2019 (COVID-19) infection.
Keywords: Mesenchymal stem cells, Autoimmune diseases, Aging, Inflammation, Cancer, COVID-19
Introduction
When mesenchymal stem cells (MSCs) were isolated and cultured at an early stage, they were found to be fibroblast-like in shape and were called fibroblast colony-forming units or bone marrow stromal fibroblasts [1]. A large number of studies have shown that MSCs secrete a variety of nutrient factors or growth factors while promoting the survival of a variety of cells through paracrine effects [2]. However, MSCs lack specific surface markers, are multipotent adult stem cells, exist in a variety of tissues, and are targeted to tumors [3, 4]. Although there are many reports about the application of MSCs in the clinic, so far, many clinical studies have not enough evidence to prove that they are effective treatment methods. Therefore, it is particularly important to explore the progress of the clinical application of MSCs in depth.
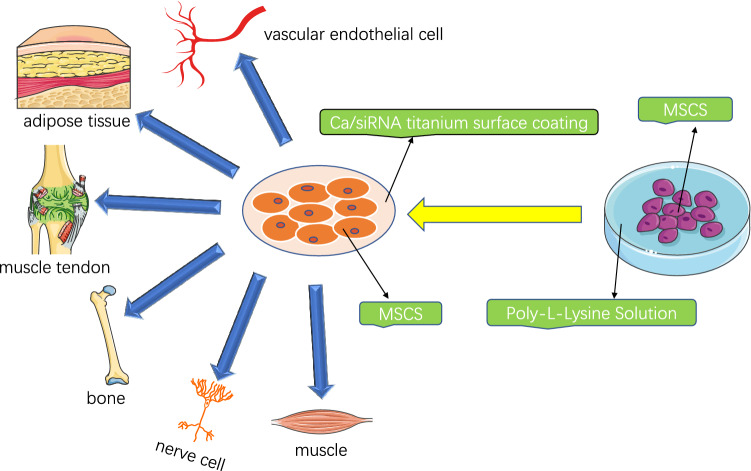
MSCs can differentiate into a variety of tissue cells derived from mesoderm and neuroectoderm, such as osteoblasts, chondrocytes, adipocytes, tenocytes, smooth muscle cells, bone marrow stromal cells, fibroblasts and a variety of vascular endothelial cells, and even neurons and glial cells of the nervous system [5–7]. Moreover, proteins secreted by MSCs have anti-microbial, anti-fibrotic, and regenerative effects, and have effects on cell proliferation, differentiation, immune regulation, angiogenesis, wound healing, and tissue regeneration [8–10]. Therefore, MSCs have become the “practical stem cells” for the treatment of multiple systemic diseases [11]. Moreover, MSCs have a unique function of secreting cytokines. The active proteins secreted by MSCs have played an important role in tissue healing. For example, they can construct collateral circulation, alleviate ischemic symptoms, inhibit inflammation, regulate immunity, maintain the local microenvironment homeostasis of ulcer wounds, and, most importantly, activate and recruit autologous stem cells for repair [12–14]. To make MSCs better applied in the clinic, the culture and differentiation are particularly important. Studies have found that Poly-L-Lysine can provide a good microenvironment for the culture of MSCs while enhancing the functionality and stemness of MSCs [15]. Moreover, the Ca/small interfering RNA (siRNA) titanium surface coating can effectively control the differentiation of MSCs, which is also a new method to control MSC differentiation and tissue regeneration [16] (Fig. 1).
Fig. 1.
MSC can differentiate into many kinds of tissue cells from mesoderm and neuroectoderm. Poly-l-lysine can provide a good microenvironment for the culture of MSCs, and enhance the function and dryness of MSCs. Ca/siRNA titanium coating can effectively control the differentiation of mesenchymal stem cells
Applications of MSCs in diabetes mellitus
Insufficient insulin secretion due to dysfunction of islet beta cells is one of the common features of type 1 and type 2 diabetes, and many patients need lifelong insulin therapy. Studies have shown that a single injection of autologous bone marrow MSCs can protect and maintain the residual function of pancreatic beta cells, but nonetheless, MSCs cannot replace hypoglycemic drugs [17] (Table 2). Therefore, more research is required. Recently, researchers have found the existence of stem cells in the islets of adult mice, which provides theoretical and technical support for the cultivation of functional islet organoids. It is promising to treat diabetes by transplanting functional islet organoids in the future [18]. Researchers have also conducted relevant studies on damaging islets. Studies have shown that the dedifferentiation of islet beta cells is an important mechanism of impaired islet function and may also be an adaptive expression of beta cells in the diabetic state. It is exciting that MSCs can reverse the dedifferentiation of human islets in type 2 diabetes and repair the morbid islets. This study provides a fresh and strong evidence for the treatment of diabetes with MSCs. Specific practice is to reverse the dedifferentiation of β cells by co-culture of MSCs with type 2 diabetic islets. The inflammatory rescue signal released by type 2 diabetic islets with high inflammation level stimulates MSCs to exert their repair function, reduces the level of islet inflammation, reverses the dedifferentiation of β cells, and thus improves islet function. Molecular mechanism studies have shown that hyperinflammatory type 2 diabetic islets secrete interleukin-1beta (IL-1β) and tumor necrosis factor-alpha (TNF-α), and stimulate MSC to secrete interleukin-1 receptor antagonist (IL-1Ra), and IL-1Ra inhibits islet inflammation, thereby reversing the dedifferentiation of β cells in type 2 diabetic islets and repairing islet function. Through this dialog response mechanism, MSCs can adjust the level of inflammation in the islets to a physiological range according to the need of the damaged islets and achieve a precise repair effect [19, 20] (Table 2). In the treatment of diabetes-induced ulcers, the main ways of stem cell transplantation are intravascular injection and local injection. However, in both approaches, stem cells are difficult to migrate to diabetic wounds, so there are problems such as low transplantation efficiency and short-cell action time. Recently, some researchers have used polymer gel to transport MSCs to ulcer wounds, because the polymer gel has a certain anti-reactive oxygen radical effect and can reduce the apoptosis of stem cells when co-transplanted, so it has become a new choice for scientists. At the same time, the study showed that the polymer gel could significantly reduce the inflammatory response mediated by the TNF-α factor, stimulate the formation of human skin fibroblasts and the proliferation of microvascular endothelial cells, thereby improving the efficiency of tissue repair and significantly shortening the healing time of diabetic ulcers. The efficiency of MSCs in the treatment of diabetic foot ulcers also depends on the use of polymer gels [21].
Table 2.
Important on-going clinical research on mesenchymal stem cells
| Source | Clinical research | Research status | References |
|---|---|---|---|
| BM-MSCs | Diabetes mellitus | A single injection of autologous BM-MSCs can protect and maintain the residual function of pancreatic beta cells, but nonetheless, MSCs cannot replace hypoglycemic drugs. Further research is needed | [17] |
| Stem cells in the islets of adult mice | Diabetes mellitus | It is promising to treat diabetes by transplanting functional islet organoids in the future | [18] |
| MSCs | Diabetes mellitus | Reversing the dedifferentiation of β cells in type 2 diabetic islets and repairing islet function | [19, 20] |
| MSCs | Autoimmune diseases | Apoptosis and metabolic inactivation of MSC have immunomodulatory potential, and regulatory T cells and monocytes play an important role | [26] |
| Remestemcel-L | COVID-19 ARDS | Remestemcel-L has an immunomodulatory effect and has potential applications in the treatment of COVID-19 ARDS | [34] |
| MSCs | Vaccines | Most pathogens have no effective antibodies, which suggest the limitations of the five existing vaccine platforms. The successful establishment of the stem cell vaccine, the sixth vaccine platform, can point out a new direction for future vaccine research and development | [47] |
| MSCs | Cancer | To use MSCs as the carriers of anti-tumor drugs to achieve effective tumor treatment | [88] |
| Gene-directed enzyme prodrug therapy or suicide gene therapy | [92, 93] | ||
| MSCs | Ischemic diseases | The mechanism of MSCs in the treatment of myocardial infarction is not that MSCs migrate to the injured myocardium differentiate into cardiomyocytes but secrete cytokines | [107, 108] |
| MSCs | Anti-aging | YAP plays a key role in maintaining the youthful state of human adult stem cells and may play a role of “de-senescence” | [123] |
| MSCs | OA | A new approach for gene therapy of OA targeting DGCR8 | [129] |
| Exosomes (or similar particles) derived from MSCs may suppress OA development | [130] | ||
| MSC-derived exosomes |
Cancer therapy Gene therapy Drug delivery |
MSC-derived exosomes have been suggested as possible cell-free substitutes for intact MSCs | [137] |
| MSCs | POF | The relevant mechanisms need to be further elucidated | [138–142] |
| BM-MSCs | Organ transplantation | Immune tolerance can be induced by the infusion of autologous BM-MSCs without taking immunosuppressive agents | [143] |
Applications of MSCs in autoimmune diseases
Since MSCs were reported to have immunosuppressive functions in 1998, their application prospects in the treatment of autoimmune diseases have been attracting great attention [22]. MSCs were first demonstrated to modulate immunosuppression in vitro and in vivo in 2002 [23]. MSCs have a strong impact on both innate and adaptive immune responses. The prevailing view is that MSCs are strong inhibitors of the immune system [24]. They inhibit the activation and proliferation of T and B lymphocytes and may also affect the maturation of dendritic cells [25]. MSCs affect the function of most immune effector cells through direct contact with immune cells and local microenvironment factors. It has been found that the immunomodulatory effects of MSCs are mainly mediated by cytokines secreted by MSCs. However, recently, apoptosis and metabolic inactivation of MSC have been shown to have immunomodulatory potential, and regulatory T cells and monocytes play an important role [26] (Table 2). Recent studies have focused on the regulation of T-cell-mediated immune response, because interferon gamma (IFN-γ) and TNF-α produced by T cells can endow mesenchymal stem cells with immunosuppressive function, which is enhanced by cytokines (such as IL-17) stimulation [27]. Recently, the immunosuppressive effects of MSCs have been studied in different directions. Studies have shown that MSCs under hypoxic conditions, through insulin-like growth factor 2 (IGF-2), confer metabolic bias on oxidative phosphorylation of macrophages during maturation, induce the formation of anti-inflammatory macrophages and promote Treg differentiation, thereby effectively inhibiting autoimmune diseases. Analysis of macrophages revealed that IGF-2 did not affect the transition of mature macrophages to macrophages with anti-inflammatory function, but acted on the process of differentiation and maturation of monocytes to macrophages, making mature macrophages under the regulation of IGF-2 have sustained immunosuppressive capacity. This finding provides new strategies and effective molecules for the treatment of autoimmune diseases [28]. There are also studies, showing that placental MSCs can significantly improve the survival of patients with graft-versus-host disease (GVHD) [29]. Moreover, because MSCs have multiple immunomodulatory effects [30], some researchers have applied them to the treatment of systemic lupus erythematosus. Current clinical data show that MSC transplantation can significantly improve the symptoms and alleviate the condition of patients with lupus erythematosus, so MSCs have important value in the treatment of systemic lupus erythematosus [31]. In summary, MSCs have great value and significance in the treatment of autoimmune diseases, and it is believed that in the near future, through further research, they will make a great contribution to the treatment of autoimmune diseases in humans.
Application of MSCs in COVID-19
The Corona Virus Disease 2019 (COVID-19) has swept the world, and more and more people are infected with the virus. Therefore, the treatment of patients has become the direction of many scientists. COVID-19 is characterized by pneumonia, lymphopenia, lymphocyte depletion, and cytokine storm [32]. At present, MSC therapy is one of the key research objects [33, 34] (Table 2). Remestemcel-L is an allogeneic MSC-derived from MSCs isolated from unrelated donor bone marrow and prepared by culture and expansion. Remestemcel-L, administered by intravenous infusion, has an immunomodulatory effect by down-regulating the production of proinflammatory cytokines, increasing the production of anti-inflammatory cytokines, and recruiting naturally occurring anti-inflammatory cells into relevant tissues to combat the inflammatory process associated with a variety of diseases. In this outbreak, diseases with potentially fatal inflammation caused by cytokine storms are the leading causes of death in patients with COVID-19. A similar course of cytokine storm can be observed in acute GVHD [35–37]. Therefore, remestemcel-L has potential applications in the treatment of COVID-19 acute respiratory distress syndrome (ARDS). And the researchers conducted a randomized, placebo-controlled study of 60 patients with chronic obstructive pulmonary disease (COPD). The analysis showed that remestemcel-L significantly improved respiratory function in patients with similarly elevated inflammatory biomarkers, which were also observed in COVID-19 ARDS patients (Table 1). These results provide a theoretical basis for evaluating remestemcel-L in the treatment of COVID-19 ARDS patients. However, a phase III clinical trial of remestemcel-L for the treatment of COVID-19 ARDS failed to meet the goal of 30 days mortality reduction. The trial included 300 patients and the goal was to reduce mortality by 43% over 30 days. On the other hand, some researchers have also confirmed that MSCs have a significant effect on the treatment of patients infected with COVID-19 [38]. Studies have shown that plasma cytokine levels in severe COVID-19 patients are significantly increased, suggesting that cytokine storm plays an important role in the lethal process of COVID-19 [39–41]. Therefore, some researchers have proposed a new idea that the use of mature MSC transplantation technology can rapidly and significantly improve the prognosis of severe and critical patients, effectively avoid the cytokine storm, without obvious side effects. Moreover, the safety and reliability of this method have been tested by clinical practice, which provides new hope for reducing the mortality rate of severe and critical patients with new coronary artery disease [42]. In addition, viral-induced acute respiratory distress syndrome is also one of the important causes of death. Recent studies have found that MSCs have great potential in the treatment of acute respiratory distress syndrome through immune regulation, proliferation, differentiation and repair, and anti-microbial effects [34, 43]. A prospective, double-blind, multicenter, randomized trial was conducted, and the results showed that a single dose of MSCs was safe for moderate-to-severe ARDS, but its effect needed to be evaluated in larger clinical trials, and the survival rate of MSCs still needed to be improved [44, 45].
Table 1.
Clinical trials of mesenchymal stem cell therapy
| Types of mesenchymal stem cells | Disease | Sample | Operation | Consequence | References |
|---|---|---|---|---|---|
| Placental mesenchymal stem cells | Graft-versus-host disease |
Eighty-four patients who received allogeneic hematopoietic stem cell transplantation |
Forty-four of them were also injected with placental mesenchymal stem cells | The 1-year survival rate was 67 percent, significantly higher than the average for other treatment regimens in the hospital | Baygan et al. [29] |
| Placental mesenchymal stem cells | knee arthritis | Twenty patients with knee arthritis | Randomly divided into two groups, one group was injected with placental mesenchymal stem cells as the experimental group, and the other group was injected with normal saline as the control group | Patients in the treatment group showed significant improvement at 8 weeks, and improvements were also observed at 24 weeks after the injection | Khalifeh Soltani et al. [56] |
| Fat mesenchymal stem cells | Osteoarthritis | Eighteen patients with knee osteoarthritis | Fat mesenchymal stem cells were injected into the knee, including three dose-increasing queues: low dose group, medium dose group and high dose group | After 6 months, WOMAC score decreased and KSS knee score and KSS functional score significantly improved | Jo et al. [57] |
| Allogeneic mesenchymal stem cells | Chronic obstructive pulmonary disease | Sixty patients with chronic obstructive pulmonary disease | Remestemcel-l was administered intravenously in a randomized, placebo-controlled study | Significantly improved respiratory function | Taghavi-Farahabadi et al. [34] |
| Autologous mesenchymal stem cells | An anal fistula caused by Crohn's disease | Twelve patients with Crohn's disease cause anal fistula | The patients' own MSCS were cultured and amplified in vitro, and then the bioabsorbable materials were used as carriers to fill the fistula of the patients regularly | The anal fistula of ten patients was completely closed, and the anal fistula of two patients improved | Dietz et al. [59] |
| Placental mesenchymal stem cells | Hemorrhagic cystitis |
Eighty-four patients who received allogeneic hematopoietic stem cell transplantation |
Forty-four of them were also injected with placental mesenchymal stem cells | One-year survival was as high as 90% | Baygan et al. [29] |
However, the above treatment of the disease cannot solve the problem at the root cause. The most fundamental is how to successfully prevent COVID-19 infection. Therefore, the global pandemic of the current epidemic makes it particularly urgent to develop vaccines [46]. Some researchers have tried to use new stem cell vaccines. In this study, the researchers used the characteristics of heterologous transfusion of MSCs and immune regulation function to over-express the structural protein of COVID-19, so that MSCs become a micro-factory with continuous production of viral proteins, to achieve the effect of mimicking viral infection. After subcutaneous and intramuscular injection in mice, the antibody against new coronavirus can be detected successfully by enzyme-linked immunosorbent assay (ELISA) analysis in serum after only 20 days. At the same time, multiple modifications of various COVID-19 structural proteins can be carried out on MSCs to achieve a cocktail effect and greatly improve the success rate of stem cell vaccines. Moreover, this has confirmed the efficiency and safety of stem cell vaccines from a new perspective, provided a new perspective for the development of vaccines, and has important significance for the development of vaccines for many pathogens without vaccines. Moreover, currently, most pathogens have no effective antibodies, which suggest the limitations of the five existing vaccine platforms. The successful establishment of the stem cell vaccine, the sixth vaccine platform, can point out a new direction for future vaccine research and development [47] (Table 2). With the extensive development of research, the immune regulatory properties, anti-inflammatory properties, and the characteristics of repairing damaged tissues of MSCs are expected to play an important role in this antiviral campaign.
Applications of MSCs in inflammation
MSCs have immune regulatory functions and strong paracrine effects in the inflammatory environment, which can be used to treat inflammatory diseases, and their role remains to be further developed [48, 49]. A large number of studies have shown that MSCs can regulate inflammation and promote tissue regeneration in a variety of ways, so as to alleviate or even treat a variety of inflammatory diseases, such as inflammatory bowel disease [50] and arthritis [51]. However, the effects of MSCs are not immutable but strictly regulated by the type and level of inflammatory cytokines. In inflammatory diseases, inflammation changes frequently. The dynamic changes in the function and composition of immune cells and immune molecules in the inflammatory microenvironment can affect the application of MSCs in inflammation [52]. Some inflammatory factors have been found to endow MSCs with immunosuppressive function [53]. Moreover, the diversity of inflammatory factors and the dynamic changes of their levels determine the plasticity of the immune regulatory capacity of MSCs [54]. For example, IL-17 regulates the immunosuppressive function of MSC and enhances the immunosuppressive effect of MSC by down-regulating mRNA degradation factors, forming inflammatory factors [55]. Recently, scientists have found that an anti-inflammatory factor (CD200) is highly expressed in placental MSCs, inhibiting the production of proinflammatory factors but also increasing the formation of anti-inflammatory response factors and immune regulatory factors, thus playing a role in the treatment of diseases [43]. In addition, studies have shown that placental mesenchymal stem cells are safe and effective in the treatment of knee arthritis. Researchers randomly divided 20 patients with knee arthritis into two groups: one group was injected with placental MSCs as the experimental group, and the other group was injected with saline as the control group. The results showed that patients in the experimental group had a significant improvement at 8 weeks, and the improvement was also observed at 24 weeks after injection [56]. Placental MSCs can also significantly improve the survival rate of patients with hemorrhagic cystitis. Among 84 patients who received allogeneic hematopoietic stem cell transplantation, 44 of them received placental MSCs at the same time. The results showed that the 1-year survival rate of hemorrhagic cystitis patients treated with placental MSCs was as high as 90% [29] (Table 1).
MSCs not only play a significant role in knee arthritis but also play an important role in osteoarthritis (OA), Crohn’s disease, and other places. For example, researchers have injected autologous fat MSC into the local joint cavity to treat OA (Phase I-II clinical trial). The results show that this can reduce the local inflammatory reaction of joints, reduce pain, improve knee function, and promote the regeneration of articular cartilage. After 6 months, the Western Ontario and McMaster Universities (WOMAC) score decreased and the Keen Society Score (KSS) knee score and KSS function score improved significantly [57]. Researchers have also found that autologous MSCs can be used to treat anal fistulas caused by Crohn’s disease with the help of bio-resorptive materials, while most patients with Crohn’s disease who receive allogeneic bone marrow mesenchymal stem cells (BM-MSCs) for anal fistulas have not seen recurrence 4 years later [58]. The researchers screened 12 patients with anal fistula caused by Crohn's disease. After the patient's own MSCs were cultured and expanded in vitro, they were filled into the patient's anal fistula and fistula regularly with bioresorbable materials as carriers for treatment. Six months later, anal fistulas were completely closed in 10 patients, improved in 2 patients, and no serious complications related to MSC therapy occurred. Therefore, MSCs are safe and effective in curing anal fistula caused by refractory Crohn’s disease [59] (Table 1).
Applications of MSCs in bone formation
MSCs are of great significance in bone-related diseases, bone tissue engineering and regenerative medicine [60, 61]. Scientists have found that many factors can promote the osteogenic differentiation of MSCs. For example, nanomembranes composed of collagen (Col) and sodium alginate (AA) show relatively high stiffness, and MSCs coated with Col/AA nanomembranes have higher osteogenic differentiation efficiency compared with other nanomembranes-coated MSCs. It was found that Col/AA nanofilm coating utilized extracellular signal-related kinase and p38 mitogen-activated protein kinase (p38MAPK) signals to activate transcriptional co-activators of PDZ-binding motifs to stimulate osteogenesis [62]. Moreover, the transcriptional co-activator of the PDZ-binding motif (TAZ) plays an important role in the osteogenesis of BM-MSCs and strongly activates the expression of osteogenic differentiation markers. Studies have shown that insulin receptor substrate 1 (IRS-1) gene modification increases TAZ expression through phosphatidylinositol 3 kinase-protein kinase B (PI3K-PKA) signaling and promotes the osteogenic differentiation pathway of rat BM-MSCs [63]. For BM-MSCs, it is a great challenge to maintain the nature of their regenerative applications in culture. However, researchers have found that a culture environment mimicking the bone marrow niche can be established to regulate the activity of BM-MSCs. In this study, researchers extracted soluble factors from human bone marrow and used them for tissue regeneration culture of bone marrow stromal cells. It has been found that Lipocalin-2 and prolactin are key factors in the bone marrow and are involved in regulating the activity of BM-MSC. Treatment of cells with Lipocalin-2 and prolactin delayed cell senescence in BM-MSCs and triggered cell osteogenesis and cartilage formation. Furthermore, further studies showed that BM-MSCs pretreated with Lipocalin-2 and prolactin could also enhance the repair of skull defects in mice. These all demonstrate that appropriate methods can be used to elicit BM-MSC properties in vitro to improve cell-based tissue regeneration in vivo [64].
Recently, researchers have made many discoveries about adipose-derived MSCs. For example, the reduction of endogenous Smad4 and Jak1 can inhibit the osteogenic differentiation of human adipose stem cells, while the expression of Smad4 and Jak1 is inhibited by MiR-125a-3p. It is suggested that MiR-125a-3p can negatively regulate osteoblast differentiation of human adipose-derived MSCs by targeting Smad4 and Jak1 [65]. To enhance the osteogenic potential of human adipose-derived mesenchymal stem cells (hADMSCs), novel multifunctional nano-scaffold chitosan (CTs) functionalized super-magnetic halloysite nanotubes (M-HNTs) were synthesized and decorated with calcium phosphate 2-D nanosheets (CaP) (called M-HNTs-CTs-CaP). It was found that M-HNTs-CTs-CaP had a strong osteogenic ability of hADMSCs and could be used as a highly applicable next-generation nano-scaffold for bone tissue engineering applications [66]. Speaking of nano-scaffolds, researchers have also fabricated ideal zein/poly L-lactic acid (PLLA) nanofibers by coaxial electrospinning and loaded them with bone morphogenetic protein 2 (BMP-2) and dexamethasone (DEX) for dual-controlled release for bone tissue engineering applications. In vitro osteogenesis studies showed that drug-loaded nanofiber scaffolds induced osteogenic differentiation. In addition, the dual-controlled release of BMP-2 and DEX enhanced the osteogenic differentiation of MSCs caused by synergistic effects. Therefore, zein/PLLA nanofiber scaffolds loaded with BMP-2 and DEX have great potential in bone tissue engineering applications [67].
Applications of MSCs in cancer
MSCs are one of the important components of the tumor microenvironment [68, 69]. Therefore, much research on cancer is about MSCs. Mesenchymal cells contribute to regulating the microenvironment of stem cells and malignant cells. The primary changes in the hematopoietic microenvironment can lead to secondary tumor diseases. MSC dysfunction may induce leukemia [70]. Some studies have found that acute myeloid leukemia injures the osteogenic differentiation process of MSCs and down-regulates the regulatory molecules required for the hematopoietic process. It reveals that cancer cells regulate specific stromal cells and impair normal tissue function to promote the carcinogenesis process [71–73]. Other studies have found that cancer cells can use MSCs to assist themselves in metastasis, because MSCs are attracted to the vicinity of the tumor by cancer cells during the process of cancer cell metastasis and promote tumor development and metastasis [74–80]. For example, MSCs can promote breast cancer metastasis, but their ability depends on the response to transforming growth factor beta (TGFβ) signals. Among the tumor-associated inflammatory factors, TGFβ is considered to be a key determinant of malignancy [81]. Researchers found that TGFβ could significantly inhibit C-X-C motif chemokine ligand 12 (CXCL12) secreted by MSCs, and over-expression of CXCL12 in MSCs could eliminate the tumor-promoting metastasis ability of MSCs [82]. This study revealed that MSCs originally bound to tumor cells evolved into MSCs that promoted tumor metastasis under the regulation of TGFβ. Moreover, by analyzing the sample information of breast cancer patients, we found that a high level of C-X-C chemokine receptor type 7 (CXCR7) expression and a low level of CXCL12 expression are typical characteristics of breast cancer, and are closely related to the survival rate of breast cancer patients. This study not only reveals a new mechanism of the interaction between TGFβ and MSCs in determining tumor cell metastasis but also provides a new marker molecule for the molecular diagnosis of breast cancer patients. Some cytokines have regulatory effects on breast cancer stem cells. For example, IL8 activates the self-renewal and regeneration ability of cancer stem cells through the IL8-CXCR1 pathway. BM-MSCs can secrete more cytokines such as IL8 and IL6 under the induction of IL6, and migrate from bone marrow to the site of breast carcinoma in situ. And to promote the self-renewal of cancer stem cells and the growth of in situ cancer, leading to further deterioration of cancer [83].
In terms of treatment, tumor-targeted drug delivery has the potential to improve therapeutic efficacy and reduce the non-specific toxicity of anti-cancer drugs [84–87]. BM-MSCs have been shown to migrate specifically to inflammatory sites, including tumors, and are expected to serve as tumor-specific carriers to deliver antineoplastic drugs. Therefore, some scientists use MSCs as the carriers of anti-tumor drugs to achieve effective tumor treatment [88] (Table 2). Interferon alpha (IFNα) has been used clinically to treat various types of tumors; however, because of its short half-life, significant therapeutic effects require high doses, which often lead to serious side effects. Therefore, researchers have successfully constructed MSCs that can continuously secrete IFNα, which can exert long-lasting anti-tumor effects and eliminate side effects associated with high clinical doses of recombinant IFNα. Moreover, MSC-IFNα can effectively inhibit the proliferation of B16 melanoma cells and promote melanoma cell apoptosis, and its anti-tumor mechanism is related to natural killer cells and CD8 + T cells. This study shows that MSCs are an ideal tumor-targeting therapy and drug sustained release carrier, which provides an innovative strategy for the clinical treatment of tumors [89]. In addition, interferon gamma-induced protein 10 kDa (IP-10) is a potent chemoattractant that enhances anti-tumor activity and mediates tumor regression through multiple mechanisms. In the mouse model of melanoma lung metastasis, hADMSCs expressing IP-10 can inhibit the growth of tumor cells and significantly prolong survival time. This suggests that hADMSCs expressing IP-10 can reduce the growth and metastasis of melanoma by targeting metastatic tumor sites. This also may be a novel and effective strategy for the treatment of melanoma [90].
And in colorectal cancer (CRC), radiotherapy is well known for remain one of the cornerstones for improving the prognosis of patients with CRC. Radiotherapy of CRC not only helps destroy cancer cells but also remodels the tumor microenvironment by enhancing the tumor specificity of BM-MSCs from the peripheral circulation. Some researchers found that BM-MSC showed anti-tumor effects by secreting certain cytokines (TNF-a, IFN-γ) when irradiated with low doses of ultraviolet and X-ray. The above cytokines inhibit the proliferation and induce apoptosis of CRC cells. All these indicate that BM-MSC can potentially promote the role of radiotherapy in CRC [91].
Gene-directed enzyme prodrug therapy (GDEPT) or suicide gene therapy is another novel treatment for cancer. MSC is used as a gene carrier to transfer suicide gene into tumor cells, which is based on the characteristics of MSCs secreting exosomes and tending to tumor tissue. In the presence of the prodrug, MSCs chemotactic into the tumor site activate the prodrug to its cytotoxic metabolites. The cytotoxic metabolites kill the adjacent tumor cells to achieve the anti-tumor effect. At the same time, the targeted administration reduces the toxic and side effects of anti-tumor drugs [92, 93] (Table 2).
Angiogenesis is a common and important physiological or pathological behavior during the growth of solid tumors [94, 95]. MSCs can secrete a variety of proangiogenic factors. Some animal experiments have proved that MSCs promote the growth of breast cancer cells [96], ovarian cancer cells [97], melanoma cells [98], and gastric cancer cells [99]. MSCs promote the growth of tumors. It is speculated that MSCs secrete growth factors to promote the growth of tumor cells, promote the angiogenesis of tumor tissue, and participate in the construction of tumor stem cell microenvironment [100, 101]. Therefore, whether MSC can be used in the treatment of tumors is full of great controversy in theory. It needs rigorous scientific research with large clinical samples.
Application of MSCs in angiogenesis
The formation of blood vessels is fundamental to development, and the deregulation of blood vessels leads to severe diseases [102–104]. MSCs participate in the links of angiogenesis, including the proliferation of endothelial cells in the early stage and the maturation of blood vessels in the late stage. MSCs improve blood supply by releasing angiopoietin and vascular endothelial growth factors to increase angiogenesis in damaged areas [105]. MSCs transplantation has made great progress in the treatment of ischemic heart disease [106]. At present, the mechanism of MSCs in the treatment of myocardial infarction is not that MSCs migrate to the injured myocardium differentiate into cardiomyocytes but secrete cytokines [107, 108] (Table 2). The main reason why ischemic diseases are difficult to recover completely is that the stimulation intensity of angiogenesis is not enough, and the lack of appropriate angiogenesis leads to insufficient blood supply. The emergence of MSC has brought hope to the treatment of diseases with insufficient blood supply such as atherosclerosis [109] and stroke [110].
The anti-aging effect of MSCs
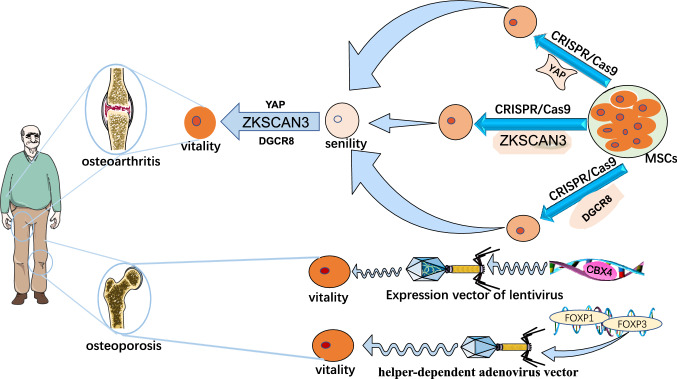
Aging is a major problem facing mankind, and the pursuit of immortality is an eternal topic [111–114]. After more and more studies, mesenchymal stem cells have been found to play a great role in combating aging. Cell senescence and stem cell depletion, as important markers of aging, are important factors driving the occurrence and development of senile diseases [115–119]. The study found that many symptoms of senile patients with the debilitating syndrome were improved without adverse reactions after the reinfusion of young MSCs. The safety and efficacy of MSCs have been demonstrated, and it is also possible to keep the elderly old [120, 121]. As a result, more and more scientists are committed to applying MSCs to anti-aging. Researchers found that zinc-finger proteins with Kruppel associated box (KRAB) and SCAN domain 3 (ZKSCAN3) showed down-regulated expression in human premature aging MSCs, replicative senescent MSCs, and primary MSCs isolated from the elderly individuals, suggesting a potential link between ZKSCAN3 and senescence regulation. Furthermore, human embryonic stem cells and MSCs with the targeted knockout of ZKSCAN3 were successfully obtained for the first time using Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)/CRISPR-associated protein-9 nuclease (Cas9)-mediated gene editing technology and stem cell directed induced differentiation technology. ZKSCAN3 was found to delay human stem cell senescence in an autophagy-independent manner. Thus, revealing a new function and mechanism of ZKSCAN3 as an epigenetic regulator to regulate human stem cell homeostasis and inhibit its senescence. This discovery expands people's understanding of the new functions of the zinc-finger protein family, deepens the understanding of the epigenetic mechanism of aging, and provides clues and ideas for delaying stem cell aging and preventing aging-related diseases [122]. At the same time, studies have also found that Yes-associated protein (YAP), the main effector of Hippo signaling, is a single protein factor that can maintain the young state of human MSCs. Researchers have also used CRISPR/Cas9-mediated gene editing to obtain YAP-specific knockout human embryonic stem cells and human MSCs [123] (Table 2). The researchers found that human MSCs lacking YAP exhibited a severe accelerated aging phenotype. Overexpression of YAP in YAP-deficient human MSCs can effectively reverse the accelerated senescence phenotype of cells, suggesting that YAP plays a key role in maintaining the youthful state of human adult stem cells and may play a role of "de-senescence" (Fig. 2). Meanwhile, human induced pluripotent stem cell (iPSC)-derived MSCs (iMSCs) has proven to be a useful clinically relevant source of MSCs, and iMSCs can obtain gene markers for revitalization regardless of donor age and cell source. These genes are expressed in pluripotent stem cells but not in parental MSCs. Moreover, iMSCs can obtain a secreted proteome similar to that of primary MSCs, which highlights their ability to conduct through paracrine signaling pathways. These suggest that human induced pluripotent stem cell-derived MSCs from older organisms may be able to “rejuvenate” [124].
Fig. 2.
MSCs targeting ZKSCAN3, YAP, and DGCR8 genes were obtained by CRISPR/Cas9-mediated gene editing technology. Then, gene therapy was used to introduce these “stem cell anti-aging factors” to treat osteoarthritis, and studies have proved its feasibility. To treat osteoporosis, researchers have used a third-generation adenovirus vector (helper-dependent adenovirus vector). The two single nucleotides in exon three of the FOXO3 gene in human embryonic stem cells were replaced by gene editing technology, thus inhibiting the phosphorylation and degradation of FOXO3 protein in cells, promoting the aggregation of FOXO3 in the nucleus, and activating the expression of downstream target genes. It can effectively delay cell senescence, resist external stress, and enhance cardiovascular homeostasis
Moreover, the aging of MSCs is considered to be one of the important triggers for the pathogenesis of OA [125]. YAP cooperates with the TEA domain transcription factor (TEAD) to activate the expression of Forkhead box D1 (FOXD1), a geo-protective protein [126]. The key role of the YAP-FOXD1 “young pathway” in human stem cell de-aging and OA treatment was established. It confirms the feasibility of treating OA by introducing the “rejuvenation” factor of stem cells into the gene and provides a new solution for the intervention of aging-related diseases such as OA, with potential clinical transformation value [127]. Researchers have also found that chromobox protein homologue 4 (CBX4), a component of polycomb inhibitory complex 1 (PRC1), plays an important role in maintaining cell identity and organ development through gene silencing. First, CBX4 protein showed down-regulated expression in human premature aging MSCs, replicative aging MSCs, and primary MSCs isolated from elderly individuals, so the researchers cloned the gene coding sequence of CBX4 into the expression vector of lentivirus and introduced it into the aging human MSCs and found that these stem cells became more “young” and restored the rapid growth characteristics (Fig. 2). Researchers pointed out that this may be because CBX4 maintains nucleolar rDNA heterochromatin homeostasis by interacting with nucleolus and heterochromatin proteins, ensures proper transcription of rRNA, prevents protein over translation, and thus maintains the young state of cells [128]. This study reveals the key role of CBX4 in counteracting cellular senescence by maintaining nucleolar homeostasis and provides a potential therapeutic target for senescence-related diseases. Researchers have also reported that the DiGeorge syndrome critical region gene 8 (DGCR8), a key factor of the miRNA synthesis pathway, inhibits the senescence of human MSCs by stabilizing heterochromatin, providing a new potential intervention target for delaying organ senescence and preventing aging-related diseases. In this study, DGCR8-deficient human MSCs were also obtained by CRISPR/Cas9-mediated gene editing and stem cell directed differentiation. Further studies revealed that DGCR8 could form a complex with nuclear membrane protein Lamin B1 and heterochromatin proteins KRAB-association protein 1 (KAP1) and heterochromatin protein 1 (HP1) to participate in maintaining the stability of heterochromatin structure, thus maintaining the young state of human mesenchymal stem cells. At the same time, gene therapy based on DGCR8 over-expression can effectively inhibit the aging of mouse joint tissue, promote the regeneration of articular cartilage, and alleviate the pathological phenotype of injurious OA and senescent OA. This reveals a new approach for gene therapy of OA targeting DGCR8 [129] (Table 2). This is another innovative discovery in the field of OA gene therapy following stem cell “young factor” CBX4 and “young pathway” YAP-FOXD1. These findings all prove the feasibility of introducing the “stem cell senescence factor” into gene therapy for OA and have potential clinical application value (Fig. 2). Recent studies have also indicated that exosomes (or similar particles) derived from MSCs may suppress OA development. This provides a new idea for the clinical treatment of OA [130] (Table 2).
Finally, studies have consistently shown that Forkhead box O (FOXO) transcription factors are important determinants of aging and longevity. And mammals have four FOXO genes: FOXO1, FOXO3, FOXO4, and FOXO6. Studies have shown that in BMSCs, the expression level of Forkhead box P1 (FOXP1) decreases with age, in contrast to the expression level of p16(INK4a), a marker of aging. Conditional depletion of FOXP1 in bone marrow MSCs leads to premature aging features, including increased bone marrow obesity, decreased bone mass, and impaired MSC. This suggests that FOXP1 may be a promising molecular target for delaying the senescence of MSCs and treating human osteoporosis [131] (Fig. 2). Forkhead box O3 (FOXO3) is closely related to delaying cell senescence, resisting external stress, and enhancing cardiovascular homeostasis [132]. Activation of FOXO3 can resist the malignant transformation of cells by inducing expression of tumor suppressor genes. The researchers eventually replaced two single nucleotides in exon 3 of the FOXO3 gene in human embryonic stem cells by gene editing using a third-generation adenovirus vector, helper-dependent adenovirus vector. Thus, it can inhibit the phosphorylation and degradation of FOXO3 protein in cells, promote the aggregation of FOXO3 in the nucleus, and then activate the expression of downstream target genes [133]. By comparing with wild-type cells, FOXO3-activated vascular cells exhibited delayed senescence and increased resistance to oxidative damage. This study provides insights into the mechanism of FOXO3-mediated vascular protection and suggests that FOXO3 activation may provide a method to generate more effective and safe biomaterials for cell replacement therapy (Fig. 2).
Perspectives of MSCs in the treatment of clinical diseases
MSCs are more and more widely used in clinical practice. In addition to the above-mentioned aspects, there are also the following aspects. For example, in future wound repair and plastic reconstruction, MSCs exist in normal skin and play a key role in wound healing. Exogenous implanted MSCs can promote the regenerative healing of injured skin. Exosomes, an effective component of MSCs, have attracted more and more attention in the field of wound healing and skin regeneration as a new and potential therapeutic method, and have put forward new prospects and expectations for future wound repair and plastic reconstruction [134–136]. Among them, exosomal microRNAs derived from MSCs can be used in cancer therapy, gene therapy, drug delivery, regenerative medicine, and some other biomedical applications. There are some limitations in the application of MSC, including the controversial use in the presence of tumor. MSC-derived exosomes have been suggested as possible cell-free substitutes for intact MSCs. If this view is proved, it will provide unlimited possibilities for the treatment of diseases in the future [137] (Table 2). In recent years, with the emergence of regenerative medicine, more and more studies on MSCs in the treatment of premature ovarian failure (POF), and more and more studies have shown that MSCs have great clinical potential in the treatment of POF, but the relevant mechanisms need to be further elucidated [138–142] (Table 2). In terms of organ transplantation, studies have found that immune tolerance can be induced by the infusion of autologous BM-MSCs without taking immunosuppressive agents. This provides evidence for MSCs to solve the problem of transplant rejection in humans. It is believed that MSCs will be widely used in clinical organ transplantation through further research [143] (Table 2).
The controversial clinical treatment of MSCs
The definition of MSCs is not clear, and the lack of uniform molecular markers has led to the use of different molecular markers of MSCs in different laboratories. Moreover, MSCs contain too many cell types and have been associated with unproven medical applications, so it has been suggested to rename them as “multipotent mesenchymal stromal cells” [144, 145]. It has also been suggested to classify cells based on their surface proteins and to name them “medicinal signaling cells” because of their ability to transform into other cell types in vitro and to come to the site of injury [146–148]. Some MSCs are promising tools for tissue engineering and regenerative medicine; however, the cellular source of bone regeneration remains controversial [149]. Meanwhile, CRC radiotherapy for colorectal cancer can not only help kill cancer cells [150], but also rebuild the tumor microenvironment by enhancing the tumor specificity of BM-MSC [151–154]. However, the role played by in situ mesenchymal stem cells and recruited stem cells under radiotherapy conditions has not been fully elucidated. The influence of BM-MSC cells on tumor progression under non-radiotherapy conditions is still controversial: on one hand, BM-MSC can regulate immune cell function, leading to reduced immune sensitivity, leading to increased risk of tumor recurrence; on the other hand, it can also secrete a variety of cytokines, which have anti-cancer effects [155–157].
Conclusion
MSCs have good application prospects, and they have no or low immunogenicity, no MHC restriction, can induce host immune tolerance, reduce transplant rejection, and can be applied in tissue engineering, tissue and organ transplantation, gene therapy, immunotherapy, and so on in the future. And MSCs presumably also play a great role in COVID-19. It is believed that after further research by scientists. MSCs can play a significant role in the complex diseases facing human beings at present.
Abbreviations
- AA
Alginate
- ARDS
Acute respiratory distress syndrome
- BM-MSCs
Bone marrow mesenchymal stem cells
- BMP-2
Bone morphogenetic protein 2
- CBX4
Chromobox protein homologue 4
- CRISPR
Clustered Regularly Interspaced Short Palindromic Repeats
- Cas9
CRISPR-associated protein-9 nuclease
- Col
Collagen
- COPD
Chronic obstructive pulmonary disease
- COVID-19
Corona Virus Disease 2019
- CRC
Colorectal cancer
- CXCR7
C-X-C chemokine receptor type 7
- CXCL12
C-X-C motif chemokine ligand 12
- DGCR8
DiGeorge syndrome critical region gene 8
- DEX
Dexamethasone
- ELISA
Enzyme-linked immunosorbent assay
- FOXD1
Forkhead box D1
- FOXO3
Forkhead box O3
- FOXP1
Forkhead box P1
- GVHD
Graft-versus-host disease
- hADMSCs
Human adipose-derived mesenchymal stem cells
- HP1
Heterochromatin protein 1
- IFNα
Interferon alpha
- IGF-2
Insulin-like growth factor 2
- IL-1β
Interleukin-1beta
- IFN-γ
Interferon gamma
- iMSCs
Induced Mesenchymal Stem Cells
- iPSCs
Induced pluripotent stem cells
- IP-10
Interferon gamma-induced protein 10 kDa
- IRS-1
Insulin receptor substrate 1
- KAP1
KRAB-association protein 1
- KSS
Keen Society Score
- KRAB
Kruppel associated box
- MSCs
Mesenchymal stem cells
- OA
Osteoarthritis
- POF
Premature ovarian failure
- PLLA
Poly l-lactic acid
- p38MAPK
P38 mitogen-activated protein kinase
- PI3K-PKA
Phosphatidylinositol 3 kinase- protein kinase B
- siRNA
Small interfering RNA
- SLE
Systemic lupus erythematosus
- TGFβ
Transforming growth factor beta
- WOMAC
Western Ontario and McMaster Universities
- YAP
Yes-associated protein
Author contributions
XZ and ZL conceived the work. GG wrote and drafted the manuscript. CF, WL, RL, CW, XC, YY, YZ, YS, YK, ZL, and XZ discussed and editing the manuscript. All authors read and approved the final version of the manuscript.
Funding
This work was supported partly by National Natural Science Foundation of China (81541153 and 81972366); Guangdong Key Laboratory funds of Systems Biology and Synthetic Biology for Urogenital Tumors (2017B030301015); Southern Marine Science and Engineering Guangdong Laboratory Zhanjiang (ZJW-2019-007); The Public Service Platform of South China Sea for R&D Marine Biomedicine Resources (GDMUK201808); Doctoral Research Initiation Fund of Guangdong Medical University (B2012001).
Declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
Not required.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guanwen Gao and Chenyang Fan have contributed equally to this work.
Contributor Information
Zesong Li, lzssc@email.szu.edu.cn.
Xiao Zhu, Email: bioxzhu@yahoo.com.
References
- 1.Beyer Nardi N, da SilvaMeirelles L. Mesenchymal stem cells: isolation, in vitro expansion and characterization. Handb Exp Pharmacol. 2006;1:249–282. doi: 10.1007/3-540-31265-X_11. [DOI] [PubMed] [Google Scholar]
- 2.Kupcova Skalnikova H. Proteomic techniques for characterisation of mesenchymal stem cell secretome. Biochimie. 2013;95:2196–2211. doi: 10.1016/j.biochi.2013.07.015. [DOI] [PubMed] [Google Scholar]
- 3.Lei T, et al. Calreticulin as a special marker to distinguish dental pulp stem cells from gingival mesenchymal stem cells. Int J Biol Macromol. 2021;178:229–239. doi: 10.1016/j.ijbiomac.2021.02.126. [DOI] [PubMed] [Google Scholar]
- 4.Kulanthaivel S, Agarwal T, Sharan Rathnam VS, Pal K, Banerjee I. Cobalt doped nano-hydroxyapatite incorporated gum tragacanth-alginate beads as angiogenic-osteogenic cell encapsulation system for mesenchymal stem cell based bone tissue engineering. Int J Biol Macromol. 2021;179:101–115. doi: 10.1016/j.ijbiomac.2021.02.136. [DOI] [PubMed] [Google Scholar]
- 5.Cortes-Hernandez LE, Eslami SZ, Alix-Panabieres C. Circulating tumor cell as the functional aspect of liquid biopsy to understand the metastatic cascade in solid cancer. Mol Asp Med. 2020;72:100816. doi: 10.1016/j.mam.2019.07.008. [DOI] [PubMed] [Google Scholar]
- 6.Glassberg MK, Csete I, Simonet E, Elliot SJ. Stem cell therapy for COPD: hope and exploitation. Chest. 2021 doi: 10.1016/j.chest.2021.04.020. [DOI] [PubMed] [Google Scholar]
- 7.Pan G, Liu Y, Shang L, Zhou F, Yang S. EMT-associated microRNAs and their roles in cancer stemness and drug resistance. Cancer Commun (Lond) 2021;41:199–217. doi: 10.1002/cac2.12138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Andrzejewska A, Lukomska B, Janowski M. Concise review: mesenchymal stem cells: from roots to boost. Stem Cells. 2019;37:855–864. doi: 10.1002/stem.3016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bulati M, et al. The immunomodulatory properties of the human amnion-derived mesenchymal stromal/stem cells are induced by inf-gamma produced by activated lymphomonocytes and are mediated by cell-to-cell contact and soluble factors. Front Immunol. 2020;11:54. doi: 10.3389/fimmu.2020.00054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lin B, et al. Tumor-infiltrating lymphocytes: warriors fight against tumors powerfully. Biomed Pharmacother. 2020;132:110873. doi: 10.1016/j.biopha.2020.110873. [DOI] [PubMed] [Google Scholar]
- 11.Mirzaei H, et al. Therapeutic application of multipotent stem cells. J Cell Physiol. 2018;233:2815–2823. doi: 10.1002/jcp.25990. [DOI] [PubMed] [Google Scholar]
- 12.Amann B, Ludemann C, Ratei R, Schmidt-Lucke JA. Autologous bone-marrow stem-cell transplantation for induction of arteriogenesis for limb salvage in critical limb ischaemia. Zent Chir. 2009;134:298–304. doi: 10.1055/s-0029-1224532. [DOI] [PubMed] [Google Scholar]
- 13.Luque-Campos N, et al. The macrophage response is driven by mesenchymal stem cell-mediated metabolic reprogramming. Front Immunol. 2021;12:624746. doi: 10.3389/fimmu.2021.624746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Taniguchi N, et al. True significance of N-acetylglucosaminyltransferases GnT-III, V and alpha1,6 fucosyltransferase in epithelial-mesenchymal transition and cancer. Mol Asp Med. 2021;79:100905. doi: 10.1016/j.mam.2020.100905. [DOI] [PubMed] [Google Scholar]
- 15.Heo JS, et al. Poly-L-lysine prevents senescence and augments growth in culturing mesenchymal stem cells ex vivo. Biomed Res Int. 2016;2016:8196078. doi: 10.1155/2016/8196078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Song W, Yang C, Le Svend D, Zhang QY, Kjems J. Calcium-MicroRNA complex-functionalized nanotubular implant surface for highly efficient transfection and enhanced osteogenesis of mesenchymal stem cells. ACS Appl Mater Interfaces. 2018;10:7756–7764. doi: 10.1021/acsami.7b18289. [DOI] [PubMed] [Google Scholar]
- 17.Carlsson PO, Schwarcz E, Korsgren O, Le Blanc K. Preserved beta-cell function in type 1 diabetes by mesenchymal stromal cells. Diabetes. 2015;64:587–592. doi: 10.2337/db14-0656. [DOI] [PubMed] [Google Scholar]
- 18.Wang D, et al. Long-term expansion of pancreatic islet organoids from resident procr(+) progenitors. Cell. 2020;180(1198–1211):e1119. doi: 10.1016/j.cell.2020.02.048. [DOI] [PubMed] [Google Scholar]
- 19.Wang L, et al. Mesenchymal stem cells ameliorate beta cell dysfunction of human type 2 diabetic islets by reversing beta cell dedifferentiation. EBioMedicine. 2020;51:102615. doi: 10.1016/j.ebiom.2019.102615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Xu F, et al. HOXD13 suppresses prostate cancer metastasis and BMP4-induced epithelial-mesenchymal transition by inhibiting SMAD1. Int J Cancer. 2021;148:3060–3070. doi: 10.1002/ijc.33494. [DOI] [PubMed] [Google Scholar]
- 21.Moon KC, et al. Potential of allogeneic adipose-derived stem cell-hydrogel complex for treating diabetic foot ulcers. Diabetes. 2019;68:837–846. doi: 10.2337/db18-0699. [DOI] [PubMed] [Google Scholar]
- 22.Wei X, et al. Mesenchymal stem cells: a new trend for cell therapy. Acta Pharmacol Sin. 2013;34:747–754. doi: 10.1038/aps.2013.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bartholomew A, et al. Mesenchymal stem cells suppress lymphocyte proliferation in vitro and prolong skin graft survival in vivo. Exp Hematol. 2002;30:42–48. doi: 10.1016/s0301-472x(01)00769-x. [DOI] [PubMed] [Google Scholar]
- 24.Poggi A, Zocchi MR. Immunomodulatory properties of mesenchymal stromal cells: still unresolved "Yin and Yang". Curr Stem Cell Res Ther. 2019;14:344–350. doi: 10.2174/1574888X14666181205115452. [DOI] [PubMed] [Google Scholar]
- 25.Herrero C, Perez-Simon JA. Immunomodulatory effect of mesenchymal stem cells. Braz J Med Biol Res. 2010;43:425–430. doi: 10.1590/s0100-879x2010007500033. [DOI] [PubMed] [Google Scholar]
- 26.Song N, Scholtemeijer M, Shah K. Mesenchymal ntial. Trends Pharmacol Sci. 2020;41:653–664. doi: 10.1016/j.tips.2020.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Obermajer N, et al. Conversion of Th17 into IL-17A(neg) regulatory T cells: a novel mechanism in prolonged allograft survival promoted by mesenchymal stem cell-supported minimized immunosuppressive therapy. J Immunol. 2014;193:4988–4999. doi: 10.4049/jimmunol.1401776. [DOI] [PubMed] [Google Scholar]
- 28.Du L, et al. IGF-2 preprograms maturing macrophages to acquire oxidative phosphorylation-dependent anti-inflammatory properties. Cell Metab. 2019;29:1363–1375 e1368. doi: 10.1016/j.cmet.2019.01.006. [DOI] [PubMed] [Google Scholar]
- 29.Baygan A, et al. Safety and side effects of using placenta-derived decidual stromal cells for graft-versus-host disease and hemorrhagic cystitis. Front Immunol. 2017;8:795. doi: 10.3389/fimmu.2017.00795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pistoia V, Raffaghello L. Mesenchymal stromal cells and autoimmunity. Int Immunol. 2017;29:49–58. doi: 10.1093/intimm/dxx008. [DOI] [PubMed] [Google Scholar]
- 31.Wang D, et al. The regulation of the Treg/Th17 balance by mesenchymal stem cells in human systemic lupus erythematosus. Cell Mol Immunol. 2017;14:423–431. doi: 10.1038/cmi.2015.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol. 2020;20:269–270. doi: 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Atluri S, Manchikanti L, Hirsch JA. Expanded umbilical cord mesenchymal stem cells (UC-MSCs) as a therapeutic strategy in managing critically Ill COVID-19 patients: the case for compassionate use. Pain Phys. 2020;23:E71–E83. [PubMed] [Google Scholar]
- 34.Taghavi-Farahabadi M, Mahmoudi M, Soudi S, Hashemi SM. Hypothesis for the management and treatment of the COVID-19-induced acute respiratory distress syndrome and lung injury using mesenchymal stem cell-derived exosomes. Med Hypotheses. 2020;144:109865. doi: 10.1016/j.mehy.2020.109865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kurtzberg J, et al. Study 275: updated expanded access program for remestemcel-L in steroid-refractory acute graft-versus-host disease in children. Biol Blood Marrow Transpl. 2020;26:855–864. doi: 10.1016/j.bbmt.2020.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kurtzberg J, et al. A phase 3, single-arm, prospective study of remestemcel-l, ex vivo culture-expanded adult human mesenchymal stromal cells for the treatment of pediatric patients who failed to respond to steroid treatment for acute graft-versus-host disease. Biol Blood Marrow Transpl. 2020;26:845–854. doi: 10.1016/j.bbmt.2020.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kebriaei P, et al. A phase 3 randomized study of remestemcel-l versus placebo added to second-line therapy in patients with steroid-refractory acute graft-versus-host disease. Biol Blood Marrow Transpl. 2020;26:835–844. doi: 10.1016/j.bbmt.2019.08.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zumla A, et al. Reducing mortality and morbidity in patients with severe COVID-19 disease by advancing ongoing trials of Mesenchymal Stromal (stem) Cell (MSC) therapy—achieving global consensus and visibility for cellular host-directed therapies. Int J Infect Dis. 2020;96:431–439. doi: 10.1016/j.ijid.2020.05.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Li J, et al. Clinical features of familial clustering in patients infected with 2019 novel coronavirus in Wuhan, China. Virus Res. 2020;286:198043. doi: 10.1016/j.virusres.2020.198043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Huang C, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wu JC, et al. Features of maintenance hemodialysis patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. Clin J Am Soc Nephrol. 2020 doi: 10.2215/CJN.04160320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Leng Z, et al. Transplantation of ACE2(-) mesenchymal stem cells improves the outcome of patients with COVID-19 pneumonia. Aging Dis. 2020;11:216–228. doi: 10.14336/AD.2020.0228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kong T, et al. Immunomodulatory effect of CD200-positive human placenta-derived stem cells in the early phase of stroke. Exp Mol Med. 2018;50:e425. doi: 10.1038/emm.2017.233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wilson JG, et al. Mesenchymal stem (stromal) cells for treatment of ARDS: a phase 1 clinical trial. Lancet Respir Med. 2015;3:24–32. doi: 10.1016/S2213-2600(14)70291-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Matthay MA, et al. Treatment with allogeneic mesenchymal stromal cells for moderate to severe acute respiratory distress syndrome (START study): a randomised phase 2a safety trial. Lancet Respir Med. 2019;7:154–162. doi: 10.1016/S2213-2600(18)30418-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Haider II, Tiwana F, Tahir SM. Impact of the COVID-19 pandemic on adult mental health. Pak J Med Sci. 2020;36:S90–S94. doi: 10.12669/pjms.36.COVID19-S4.2756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liu J, Jiao H, Yin X. Engineered human mesenchymal stem cells as new vaccine platform for COVID-19. bioRxiv, 2020.2006.2020.163030, 2020. Doi:10.1101/2020.06.20.163030 (2020).
- 48.Mannerstrom B, et al. Epigenetic alterations in mesenchymal stem cells by osteosarcoma-derived extracellular vesicles. Epigenetics. 2019;14:352–364. doi: 10.1080/15592294.2019.1585177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zhu X, Fan WG, Li DP, Kung H, Lin MC. Heme oxygenase-1 system and gastrointestinal inflammation: a short review. World J Gastroenterol. 2011;17:4283–4288. doi: 10.3748/wjg.v17.i38.4283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Panes J, et al. Expanded allogeneic adipose-derived mesenchymal stem cells (Cx601) for complex perianal fistulas in Crohn’s disease: a phase 3 randomised, double-blind controlled trial. Lancet. 2016;388:1281–1290. doi: 10.1016/S0140-6736(16)31203-X. [DOI] [PubMed] [Google Scholar]
- 51.Augello A, Tasso R, Negrini SM, Cancedda R, Pennesi G. Cell therapy using allogeneic bone marrow mesenchymal stem cells prevents tissue damage in collagen-induced arthritis. Arthritis Rheum. 2007;56:1175–1186. doi: 10.1002/art.22511. [DOI] [PubMed] [Google Scholar]
- 52.Shi Y, et al. Immunoregulatory mechanisms of mesenchymal stem and stromal cells in inflammatory diseases. Nat Rev Nephrol. 2018;14:493–507. doi: 10.1038/s41581-018-0023-5. [DOI] [PubMed] [Google Scholar]
- 53.Ren G, et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell. 2008;2:141–150. doi: 10.1016/j.stem.2007.11.014. [DOI] [PubMed] [Google Scholar]
- 54.Wang Y, Chen X, Cao W, Shi Y. Plasticity of mesenchymal stem cells in immunomodulation: pathological and therapeutic implications. Nat Immunol. 2014;15:1009–1016. doi: 10.1038/ni.3002. [DOI] [PubMed] [Google Scholar]
- 55.Wang Y, et al. TNF-alpha-induced LRG1 promotes angiogenesis and mesenchymal stem cell migration in the subchondral bone during osteoarthritis. Cell Death Dis. 2017;8:e2715. doi: 10.1038/cddis.2017.129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Khalifeh Soltani S, et al. Safety and efficacy of allogenic placental mesenchymal stem cells for treating knee osteoarthritis: a pilot study. Cytotherapy. 2019;21:54–63. doi: 10.1016/j.jcyt.2018.11.003. [DOI] [PubMed] [Google Scholar]
- 57.Jo CH, et al. Intra-articular injection of mesenchymal stem cells for the treatment of osteoarthritis of the knee: a proof-of-concept clinical trial. Stem Cells. 2014;32:1254–1266. doi: 10.1002/stem.1634. [DOI] [PubMed] [Google Scholar]
- 58.Barnhoorn MC, et al. Long-term evaluation of allogeneic bone marrow-derived mesenchymal stromal cell therapy for crohn's disease perianal fistulas. J Crohns Colitis. 2020;14:64–70. doi: 10.1093/ecco-jcc/jjz116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Dietz AB, et al. Autologous mesenchymal stem cells, applied in a bioabsorbable matrix, for treatment of perianal fistulas in patients with crohn's disease. Gastroenterology. 2017;153:59–62 e52. doi: 10.1053/j.gastro.2017.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Chen YH, et al. YY1 and HDAC9c transcriptionally regulate p38-mediated mesenchymal stem cell differentiation into osteoblasts. Am J Cancer Res. 2018;8:514–525. [PMC free article] [PubMed] [Google Scholar]
- 61.Yao D, et al. Bone metabolism regulation: Implications for the treatment of bone diseases. Biomed Pharmacother. 2020;129:110494. doi: 10.1016/j.biopha.2020.110494. [DOI] [PubMed] [Google Scholar]
- 62.Hwang JH, et al. Artificial cellular nano-environment composed of collagen-based nanofilm promotes osteogenic differentiation of mesenchymal stem cells. Acta Biomater. 2019;86:247–256. doi: 10.1016/j.actbio.2018.12.044. [DOI] [PubMed] [Google Scholar]
- 63.Wang N, Xue P, Li Z, Li Y. IRS-1 increases TAZ expression and promotes osteogenic differentiation in rat bone marrow mesenchymal stem cells. Biol Open. 2018 doi: 10.1242/bio.036194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tsai TL, Li WJ. Identification of bone marrow-derived soluble factors regulating human mesenchymal stem cells for bone regeneration. Stem Cell Rep. 2017;8:387–400. doi: 10.1016/j.stemcr.2017.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gu Z, Long J, Li Y, Wang X, Wang H. MiR-125a-3p negatively regulates osteoblastic differentiation of human adipose derived mesenchymal stem cells by targeting Smad4 and Jak1. Am J Transl Res. 2019;11:2603–2615. [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 66.Lee YJ, Lee SC, Jee SC, Sung JS, Kadam AA. Surface functionalization of halloysite nanotubes with supermagnetic iron oxide, chitosan and 2-D calcium-phosphate nanoflakes for synergistic osteoconduction enhancement of human adipose tissue-derived mesenchymal stem cells. Colloids Surf B Biointerfaces. 2019;173:18–26. doi: 10.1016/j.colsurfb.2018.09.045. [DOI] [PubMed] [Google Scholar]
- 67.Li R, Ma Y, Zhang Y, Zhang M, Sun D. Potential of rhBMP-2 and dexamethasone-loaded Zein/PLLA scaffolds for enhanced in vitro osteogenesis of mesenchymal stem cells. Colloids Surf B Biointerfaces. 2018;169:384–394. doi: 10.1016/j.colsurfb.2018.05.039. [DOI] [PubMed] [Google Scholar]
- 68.Tang X, Huang Y, Lei J, Luo H, Zhu X. The single-cell sequencing: new developments and medical applications. Cell Biosci. 2019;9:53. doi: 10.1186/s13578-019-0314-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Liu J, Li D, Luo H, Zhu X. Circular RNAs: the star molecules in cancer. Mol Asp Med. 2019;70:141–152. doi: 10.1016/j.mam.2019.10.006. [DOI] [PubMed] [Google Scholar]
- 70.Raaijmakers MH, et al. Bone progenitor dysfunction induces myelodysplasia and secondary leukaemia. Nature. 2010;464:852–857. doi: 10.1038/nature08851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Baryawno N, et al. A Cellular Taxonomy of the Bone Marrow Stroma in Homeostasis and Leukemia. Cell. 2019;177:1915–1932 e1916. doi: 10.1016/j.cell.2019.04.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Guo B, Li D, Du L, Zhu X. piRNAs: biogenesis and their potential roles in cancer. Cancer Metastasis Rev. 2020;39:567–575. doi: 10.1007/s10555-020-09863-0. [DOI] [PubMed] [Google Scholar]
- 73.Li K, Luo H, Luo H, Zhu X. Clinical and prognostic pan-cancer analysis of m6A RNA methylation regulators in four types of endocrine system tumors. Aging (Albany NY) 2020;12:23931–23944. doi: 10.18632/aging.104064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Berger L, Shamai Y, Skorecki KL, Tzukerman M. Tumor specific recruitment and reprogramming of mesenchymal stem cells in tumorigenesis. Stem Cells. 2016;34:1011–1026. doi: 10.1002/stem.2269. [DOI] [PubMed] [Google Scholar]
- 75.Zou Z, Tao T, Li H, Zhu X. mTOR signaling pathway and mTOR inhibitors in cancer: progress and challenges. Cell Biosci. 2020;10:31. doi: 10.1186/s13578-020-00396-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zou Z, et al. The pan-cancer analysis of the two types of uterine cancer uncovered clinical and prognostic associations with m6A RNA methylation regulators. Mol Omics. 2021 doi: 10.1039/d0mo00113a. [DOI] [PubMed] [Google Scholar]
- 77.Zhu X, Li S, Xu B, Luo H. Cancer evolution: a means by which tumors evade treatment. Biomed Pharmacother. 2020;133:111016. doi: 10.1016/j.biopha.2020.111016. [DOI] [PubMed] [Google Scholar]
- 78.Liang B, Ding H, Huang L, Luo H, Zhu X. GWAS in cancer: progress and challenges. Mol Genet Genomics. 2020;295:537–561. doi: 10.1007/s00438-020-01647-z. [DOI] [PubMed] [Google Scholar]
- 79.Zhu X, et al. The rs391957 variant cis-regulating oncogene GRP78 expression contributes to the risk of hepatocellular carcinoma. Carcinogenesis. 2013;34:1273–1280. doi: 10.1093/carcin/bgt061. [DOI] [PubMed] [Google Scholar]
- 80.Xie S, Wu Z, Qi Y, Wu B, Zhu X. The metastasizing mechanisms of lung cancer: recent advances and therapeutic challenges. Biomed Pharmacother. 2021;138:111450. doi: 10.1016/j.biopha.2021.111450. [DOI] [PubMed] [Google Scholar]
- 81.de Araujo Farias V, Carrillo-Galvez AB, Martin F, Anderson P. TGF-beta and mesenchymal stromal cells in regenerative medicine, autoimmunity and cancer. Cytokine Growth Factor Rev. 2018;43:25–37. doi: 10.1016/j.cytogfr.2018.06.002. [DOI] [PubMed] [Google Scholar]
- 82.Yu PF, et al. Downregulation of CXCL12 in mesenchymal stromal cells by TGFbeta promotes breast cancer metastasis. Oncogene. 2017;36:840–849. doi: 10.1038/onc.2016.252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Jia D, et al. An autocrine inflammatory forward-feedback loop after chemotherapy withdrawal facilitates the repopulation of drug-resistant breast cancer cells. Cell Death Dis. 2017;8:e2932. doi: 10.1038/cddis.2017.319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lu G, Luo H, Zhu X. Targeting the GRP78 pathway for cancer therapy. Front Med (Lausanne) 2020;7:351. doi: 10.3389/fmed.2020.00351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Liang X, Li D, Leng S, Zhu X. RNA-based pharmacotherapy for tumors: from bench to clinic and back. Biomed Pharmacother. 2020;125:109997. doi: 10.1016/j.biopha.2020.109997. [DOI] [PubMed] [Google Scholar]
- 86.Li S, Zhang Z, Lai WF, Cui L, Zhu X. How to overcome the side effects of tumor immunotherapy. Biomed Pharmacother. 2020;130:110639. doi: 10.1016/j.biopha.2020.110639. [DOI] [PubMed] [Google Scholar]
- 87.Wu Z, Li S, Zhu X. The mechanism of stimulating and mobilizing the immune system enhancing the anti-tumor immunity. Front Immunol. 2021;12:682435. doi: 10.3389/fimmu.2021.682435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Mirzaei H, et al. Application of mesenchymal stem cells in melanoma: a potential therapeutic strategy for delivery of targeted agents. Curr Med Chem. 2016;23:455–463. doi: 10.2174/0929867323666151217122033. [DOI] [PubMed] [Google Scholar]
- 89.Xu C, et al. Interferon-alpha-secreting mesenchymal stem cells exert potent antitumor effect in vivo. Oncogene. 2014;33:5047–5052. doi: 10.1038/onc.2013.458. [DOI] [PubMed] [Google Scholar]
- 90.Mirzaei H, et al. The therapeutic potential of human adipose-derived mesenchymal stem cells producing CXCL10 in a mouse melanoma lung metastasis model. Cancer Lett. 2018;419:30–39. doi: 10.1016/j.canlet.2018.01.029. [DOI] [PubMed] [Google Scholar]
- 91.Feng H, et al. Bone marrow-derived mesenchymal stromal cells promote colorectal cancer cell death under low-dose irradiation. Br J Cancer. 2018;118:353–365. doi: 10.1038/bjc.2017.415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Mohammadi M, Jaafari MR, Mirzaei HR, Mirzaei H. Mesenchymal stem cell: a new horizon in cancer gene therapy. Cancer Gene Ther. 2016;23:285–286. doi: 10.1038/cgt.2016.35. [DOI] [PubMed] [Google Scholar]
- 93.Moradian Tehrani R, et al. Mesenchymal stem cells: a new platform for targeting suicide genes in cancer. J Cell Physiol. 2018;233:3831–3845. doi: 10.1002/jcp.26094. [DOI] [PubMed] [Google Scholar]
- 94.Yang Y, et al. Programmed death ligand-1 regulates angiogenesis and metastasis by participating in the c-JUN/VEGFR2 signaling axis in ovarian cancer. Cancer Commun (Lond) 2021;41:511–527. doi: 10.1002/cac2.12157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Shi Y, et al. Bevacizumab biosimilar LY01008 compared with bevacizumab (Avastin) as first-line treatment for Chinese patients with unresectable, metastatic, or recurrent non-squamous non-small-cell lung cancer: A multicenter, randomized, double-blinded, phase III trial. Cancer Commun (Lond) 2021 doi: 10.1002/cac2.12179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Karnoub AE, et al. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature. 2007;449:557–563. doi: 10.1038/nature06188. [DOI] [PubMed] [Google Scholar]
- 97.Coffelt SB, et al. The pro-inflammatory peptide LL-37 promotes ovarian tumor progression through recruitment of multipotent mesenchymal stromal cells. Proc Natl Acad Sci USA. 2009;106:3806–3811. doi: 10.1073/pnas.0900244106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Djouad F, et al. Immunosuppressive effect of mesenchymal stem cells favors tumor growth in allogeneic animals. Blood. 2003;102:3837–3844. doi: 10.1182/blood-2003-04-1193. [DOI] [PubMed] [Google Scholar]
- 99.Quante M, et al. Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell. 2011;19:257–272. doi: 10.1016/j.ccr.2011.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zhu W, et al. Mesenchymal stem cells derived from bone marrow favor tumor cell growth in vivo. Exp Mol Pathol. 2006;80:267–274. doi: 10.1016/j.yexmp.2005.07.004. [DOI] [PubMed] [Google Scholar]
- 101.Yu JM, Jun ES, Bae YC, Jung JS. Mesenchymal stem cells derived from human adipose tissues favor tumor cell growth in vivo. Stem Cells Dev. 2008;17:463–473. doi: 10.1089/scd.2007.0181. [DOI] [PubMed] [Google Scholar]
- 102.Carmeliet P. Angiogenesis in health and disease. Nat Med. 2003;9:653–660. doi: 10.1038/nm0603-653. [DOI] [PubMed] [Google Scholar]
- 103.Carmeliet P, Jain RK. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011;473:298–307. doi: 10.1038/nature10144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Xu P, et al. Cancer neoantigen: boosting immunotherapy. Biomed Pharmacother. 2020;131:110640. doi: 10.1016/j.biopha.2020.110640. [DOI] [PubMed] [Google Scholar]
- 105.Watt SM, et al. The angiogenic properties of mesenchymal stem/stromal cells and their therapeutic potential. Br Med Bull. 2013;108:25–53. doi: 10.1093/bmb/ldt031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Kanelidis AJ, Premer C, Lopez J, Balkan W, Hare JM. Route of delivery modulates the efficacy of mesenchymal stem cell therapy for myocardial infarction: a meta-analysis of preclinical studies and clinical trials. Circ Res. 2017;120:1139–1150. doi: 10.1161/CIRCRESAHA.116.309819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Nazari-Shafti TZ, et al. Human mesenchymal stromal cells and derived extracellular vesicles: Translational strategies to increase their proangiogenic potential for the treatment of cardiovascular disease. Stem Cells Transl Med. 2020;9:1558–1569. doi: 10.1002/sctm.19-0432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Sun L, et al. Down-regulated exosomal MicroRNA-221—3p derived from senescent mesenchymal stem cells impairs heart repair. Front Cell Dev Biol. 2020;8:263. doi: 10.3389/fcell.2020.00263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Du W, et al. VCAM-1+ placenta chorionic villi-derived mesenchymal stem cells display potent pro-angiogenic activity. Stem Cell Res Ther. 2016;7:49. doi: 10.1186/s13287-016-0297-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Xue P, Wang M, Yan G. Mesenchymal stem cell transplantation as an effective treatment strategy for ischemic stroke in Asia: a meta-analysis of controlled trials. Ther Clin Risk Manag. 2018;14:909–928. doi: 10.2147/TCRM.S161326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Hajishengallis G, Li X, Chavakis T. Immunometabolic control of hematopoiesis. Mol Aspects Med. 2021;77:100923. doi: 10.1016/j.mam.2020.100923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Safiri S, et al. Burden of tracheal, bronchus, and lung cancer and its attributable risk factors in 204 countries and territories, 1990 to 2019. J Thorac Oncol. 2021;16:945–959. doi: 10.1016/j.jtho.2021.03.030. [DOI] [PubMed] [Google Scholar]
- 113.Wu Q, Yu X, Li J, Sun S, Tu Y. Metabolic regulation in the immune response to cancer. Cancer Commun (Lond) 2021 doi: 10.1002/cac2.12182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Lajoie AC, Lafontaine AL, Kaminska M. The spectrum of sleep disorders in parkinson disease: a review. Chest. 2021;159:818–827. doi: 10.1016/j.chest.2020.09.099. [DOI] [PubMed] [Google Scholar]
- 115.Zhou S, et al. Age-related intrinsic changes in human bone-marrow-derived mesenchymal stem cells and their differentiation to osteoblasts. Aging Cell. 2008;7:335–343. doi: 10.1111/j.1474-9726.2008.00377.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Wang Y, et al. Interplay between orphan nuclear receptors and androgen receptor-dependent or-independent growth signalings in prostate cancer. Mol Asp Med. 2021;78:100921. doi: 10.1016/j.mam.2020.100921. [DOI] [PubMed] [Google Scholar]
- 117.Ding H, et al. Transferrin receptor 1 ablation in satellite cells impedes skeletal muscle regeneration through activation of ferroptosis. J Cachexia Sarcopenia Muscle. 2021 doi: 10.1002/jcsm.12700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Libby AE, Jones B, Lopez-Santiago I, Rowland E, Levi M. Nuclear receptors in the kidney during health and disease. Mol Asp Med. 2021;78:100935. doi: 10.1016/j.mam.2020.100935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Torti SV, Torti FM. Iron: the cancer connection. Mol Asp Med. 2020;75:100860. doi: 10.1016/j.mam.2020.100860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Golpanian S, et al. Allogeneic human mesenchymal stem cell infusions for aging frailty. J Gerontol A Biol Sci Med Sci. 2017;72:1505–1512. doi: 10.1093/gerona/glx056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Golpanian S, et al. Rationale and design of the allogeneiC human mesenchymal stem cells (hMSC) in patients with aging fRAilTy via intravenoUS delivery (CRATUS) study: A phase I/II, randomized, blinded and placebo controlled trial to evaluate the safety and potential efficacy of allogeneic human mesenchymal stem cell infusion in patients with aging frailty. Oncotarget. 2016;7:11899–11912. doi: 10.18632/oncotarget.7727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Hu H, et al. ZKSCAN3 counteracts cellular senescence by stabilizing heterochromatin. Nucl Acids Res. 2020;48:6001–6018. doi: 10.1093/nar/gkaa425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Zhang M, Eshraghian EA, Jammal OA, Zhang Z, Zhu X. CRISPR technology: the engine that drives cancer therapy. Biomed Pharmacother. 2020;133:111007. doi: 10.1016/j.biopha.2020.111007. [DOI] [PubMed] [Google Scholar]
- 124.Spitzhorn LS, et al. Human iPSC-derived MSCs (iMSCs) from aged individuals acquire a rejuvenation signature. Stem Cell Res Ther. 2019;10:100. doi: 10.1186/s13287-019-1209-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Alsalameh S, Amin R, Gemba T, Lotz M. Identification of mesenchymal progenitor cells in normal and osteoarthritic human articular cartilage. Arthritis Rheum. 2004;50:1522–1532. doi: 10.1002/art.20269. [DOI] [PubMed] [Google Scholar]
- 126.Tang J, Xu Z, Huang L, Luo H, Zhu X. Transcriptional regulation in model organisms: recent progress and clinical implications. Open Biol. 2019;9:190183. doi: 10.1098/rsob.190183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Fu L, et al. Up-regulation of FOXD1 by YAP alleviates senescence and osteoarthritis. PLoS Biol. 2019;17:e3000201. doi: 10.1371/journal.pbio.3000201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Ren X, et al. Maintenance of nucleolar homeostasis by CBX4 alleviates senescence and osteoarthritis. Cell Rep. 2019;26:3643–36563647. doi: 10.1016/j.celrep.2019.02.088. [DOI] [PubMed] [Google Scholar]
- 129.Deng L, et al. Stabilizing heterochromatin by DGCR8 alleviates senescence and osteoarthritis. Nat Commun. 2019;10:3329. doi: 10.1038/s41467-019-10831-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Mianehsaz E, et al. Mesenchymal stem cell-derived exosomes: a new therapeutic approach to osteoarthritis? Stem Cell Res Ther. 2019;10:340. doi: 10.1186/s13287-019-1445-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Li H, et al. FOXP1 controls mesenchymal stem cell commitment and senescence during skeletal aging. J Clin Investig. 2017;127:1241–1253. doi: 10.1172/JCI89511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Martins R, Lithgow GJ, Link W. Long live FOXO: unraveling the role of FOXO proteins in aging and longevity. Aging Cell. 2016;15:196–207. doi: 10.1111/acel.12427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Yan P, et al. FOXO3-engineered human esc-derived vascular cells promote vascular protection and regeneration. Cell Stem Cell. 2019;24(447–461):e448. doi: 10.1016/j.stem.2018.12.002. [DOI] [PubMed] [Google Scholar]
- 134.Hu P, et al. Mesenchymal stromal cells-exosomes: a promising cell-free therapeutic tool for wound healing and cutaneous regeneration. Burns Trauma. 2019;7:38. doi: 10.1186/s41038-019-0178-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Hu P, et al. Corrigendum to: 'Mesenchymal stromal cells-exosomes: a promising cell-free therapeutic tool for wound healing and cutaneous regeneration'. Burns Trauma. 2020;8:tkaa007. doi: 10.1093/burnst/tkaa007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Tang Z, Li D, Hou S, Zhu X. The cancer exosomes: clinical implications, applications and challenges. Int J Cancer. 2020;146:2946–2959. doi: 10.1002/ijc.32762. [DOI] [PubMed] [Google Scholar]
- 137.Asgarpour K, et al. Exosomal microRNAs derived from mesenchymal stem cells: cell-to-cell messages. Cell Commun Signal. 2020;18:149. doi: 10.1186/s12964-020-00650-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Yoon SY, et al. Recovery of ovarian function by human embryonic stem cell-derived mesenchymal stem cells in cisplatin-induced premature ovarian failure in mice. Stem Cell Res Ther. 2020;11:255. doi: 10.1186/s13287-020-01769-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Chen L, et al. Effect of stem cell transplantation of premature ovarian failure in animal models and patients: a meta-analysis and case report. Exp Ther Med. 2018;15:4105–4118. doi: 10.3892/etm.2018.5970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Li J, et al. Human chorionic plate-derived mesenchymal stem cells transplantation restores ovarian function in a chemotherapy-induced mouse model of premature ovarian failure. Stem Cell Res Ther. 2018;9:81. doi: 10.1186/s13287-018-0819-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Ding L, et al. Transplantation of UC-MSCs on collagen scaffold activates follicles in dormant ovaries of POF patients with long history of infertility. Sci China Life Sci. 2018;61:1554–1565. doi: 10.1007/s11427-017-9272-2. [DOI] [PubMed] [Google Scholar]
- 142.Ling L, et al. Effects of low-intensity pulsed ultrasound (LIPUS)-pretreated human amnion-derived mesenchymal stem cell (hAD-MSC) transplantation on primary ovarian insufficiency in rats. Stem Cell Res Ther. 2017;8:283. doi: 10.1186/s13287-017-0739-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Casiraghi F, et al. Kidney transplant tolerance associated with remote autologous mesenchymal stromal cell administration. Stem Cells Transl Med. 2020;9:427–432. doi: 10.1002/sctm.19-0185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Jariyal H, Gupta C, Srivastava A. Hyaluronic acid induction on breast cancer stem cells unfolds subtype specific variations in stemness and epithelial-to-mesenchymal transition. Int J Biol Macromol. 2020;160:1078–1089. doi: 10.1016/j.ijbiomac.2020.05.236. [DOI] [PubMed] [Google Scholar]
- 145.Oh Y, Ahn CB, Marasinghe M, Je JY. Insertion of gallic acid onto chitosan promotes the differentiation of osteoblasts from murine bone marrow-derived mesenchymal stem cells. Int J Biol Macromol. 2021;183:1410–1418. doi: 10.1016/j.ijbiomac.2021.05.122. [DOI] [PubMed] [Google Scholar]
- 146.Sipp D, Robey PG, Turner L. Clear up this stem-cell mess. Nature. 2018;561:455–457. doi: 10.1038/d41586-018-06756-9. [DOI] [PubMed] [Google Scholar]
- 147.Galipeau J, Weiss DJ, Dominici M. Response to nature commentary “clear up this stem-cell mess”. Cytotherapy. 2019;21:1–2. doi: 10.1016/j.jcyt.2018.11.007. [DOI] [PubMed] [Google Scholar]
- 148.Jiang B, et al. Concise review: mesenchymal stem cells derived from human pluripotent cells, an unlimited and quality-controllable source for therapeutic applications. Stem Cells. 2019;37:572–581. doi: 10.1002/stem.2964. [DOI] [PubMed] [Google Scholar]
- 149.Zhang Y, et al. An in vitro comparative study of multisource derived human mesenchymal stem cells for bone tissue engineering. Stem Cells Dev. 2018;27:1634–1645. doi: 10.1089/scd.2018.0119. [DOI] [PubMed] [Google Scholar]
- 150.Liu Z, et al. Imaging genomics for accurate diagnosis and treatment of tumors: a cutting edge overview. Biomed Pharmacother. 2021 doi: 10.1016/j.biopha.2020.111173. [DOI] [PubMed] [Google Scholar]
- 151.Tang Z, Xu Z, Zhu X, Zhang J. New insights into molecules and pathways of cancer metabolism and therapeutic implications. Cancer Commun (Lond) 2021;41:16–36. doi: 10.1002/cac2.12112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Tan S, Li D, Zhu X. Cancer immunotherapy: pros, cons and beyond. Biomed Pharmacother. 2020;124:109821. doi: 10.1016/j.biopha.2020.109821. [DOI] [PubMed] [Google Scholar]
- 153.Li K, Luo H, Huang L, Luo H, Zhu X. Microsatellite instability: a review of what the oncologist should know. Cancer Cell Int. 2020;20:16. doi: 10.1186/s12935-019-1091-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Weiskirchen R, Weiskirchen S, Tacke F. Organ and tissue fibrosis: Molecular signals, cellular mechanisms and translational implications. Mol Asp Med. 2019;65:2–15. doi: 10.1016/j.mam.2018.06.003. [DOI] [PubMed] [Google Scholar]
- 155.Pan G, Liu Y, Shang L, Zhou F, Yang S. EMT-associated microRNAs and their roles in cancer stemness and drug resistance. Cancer Commun (Lond) 2021 doi: 10.1002/cac2.12138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Jonkers J, Pai P, Sukumar S. Multiple roles of HOX proteins in metastasis: let me count the ways. Cancer Metastasis Rev. 2020;39:661–679. doi: 10.1007/s10555-020-09908-4. [DOI] [PubMed] [Google Scholar]
- 157.Angthong C, Kunkanjanawan H. Ankle synovium-derived mesenchymal stem cells for the treatment of osteochondral lesion of the talus: a novel cell harvesting technique and clinical applications. Eur Rev Med Pharmacol Sci. 2020;24:8273–8280. doi: 10.26355/eurrev_202008_22623. [DOI] [PubMed] [Google Scholar]