Abstract
Herbs and dietary supplement-induced liver injury (HILI) is the leading cause of drug-induced liver injury in China. Among different hepatotoxic herbs, the pyrrolizidine alkaloid (PA)-producing herb Gynura japonica contributes significantly to HILI by inducing hepatic sinusoidal obstruction syndrome (HSOS), a liver disorder characterized by hepatomegaly, hyperbilirubinemia, and ascites. In China, G. japonica has been used as one of the plant species for Tu-San-Qi and is often misused with non-PA-producing Tu-San-Qi (Sedum aizoon) or even San-Qi (Panax notoginseng) for self-medication. It has been reported that over 50% of HSOS cases are caused by the intake of PA-producing G. japonica. In this review, we provide comprehensive information to distinguish these Tu-San-Qi-related herbal plant species in terms of plant/medicinal part morphologies, medicinal indications, and chemical profiles. Approximately 2156 Tu-San-Qi-associated HSOS cases reported in China from 1980 to 2019 are systematically reviewed in terms of their clinical manifestation, diagnostic workups, therapeutic interventions, and outcomes. In addition, based on the application of our developed mechanism-based biomarker of PA exposure, our clinical findings on the definitive diagnosis of 58 PA-producing Tu-San-Qi-induced HSOS patients are also elaborated. Therefore, this review article provides the first comprehensive report on 2214 PA-producing Tu-San-Qi (G. japonica)-induced HSOS cases in China, and the information presented will improve public awareness of the significant incidence of PA-producing Tu-San-Qi (G. japonica)-induced HSOS and facilitate future prevention and better clinical management of this severe HILI.
Keywords: herb-induced liver injury, hepatic sinusoidal obstruction syndrome, pyrrolizidine alkaloids, Tu-San-Qi, Gynura japonica
Introduction
Drug-induced liver injury (DILI) is a common adverse drug reaction and one of the major causes of liver failure [1–3]. The incidence of DILI in China has been estimated to be 23.8 per 100,000 inhabitants, which is higher than that in Western countries [4]. Herbs and dietary supplement-induced liver injury (HILI), particularly due to the use of medicinal herbs, has been found to be the leading cause (~26.8%) of DILI in China [4]. The diagnosis of DILI, especially HILI caused by the intake of herbal medicines and herbal dietary supplements, is challenging due to the lack of characteristic clinical features and specific tests for this condition.
Hepatic sinusoidal obstruction syndrome (HSOS, formerly called hepatic venoocclusive disease) is a hepatic vascular disease resulting from injury to hepatic sinusoidal endothelial cells that leads to obstruction of the small hepatic veins [5]. Severe HSOS often causes multiorgan multi failure, resulting in a high mortality rate greater than 80% [6]. The clinical manifestations of HSOS, including hepatomegaly, jaundice, ascites, and weight gain, are nonspecific and highly similar to the symptoms of other liver diseases, such as acute hepatitis, decompensated cirrhosis, and Budd–Chiari syndrome (BCS) [7].
Exposure to pyrrolizidine alkaloids (PAs) has been determined to be one of the two major causes of HSOS [7–9]. PAs are common phytotoxins widely distributed across the plant kingdom in up to 13 distantly related angiosperm families, covering ~3% of flowering plants [10–12]. Humans are exposed to PAs by direct consumption of PA-producing plants used as herbal medicine, herbal teas, and dietary supplements [8, 13] or by the intake of PA-contaminated foodstuffs such as milk, teas, and honey [14–16]. The earliest case of PA-induced HSOS was reported in 1920 and resulted from the ingestion of PA-contaminated wheat [17]. Since then, based on our literature research, over 15,000 PA poisoning cases have been documented in many countries and regions, including Afghanistan, Britain, China, Germany, Hong Kong (China), India, Jamaica, South Africa, Switzerland, and the United States [18]. The major cause of HSOS in China is the ingestion of a PA-producing medicinal herb, Gynura japonica (Thunb.) Juel (G. japonica), accounting for 50%–89% of reported HSOS cases [19].
It has been reported that G. japonica contains predominantly four hepatotoxic PAs, namely, seneciphylline, senecionine, seneciphylline N-oxide, and senecionine N-oxide [9, 20, 21]. PA-producing G. japonica has long been frequently misused with the non-PA-producing traditional Chinese medicine (TCM) herbs Tu-San-Qi (Sedum aizoon L. (S. aizoon)) and San-Qi (Panax notoginseng (Burkill) F.H. Chen (P. notoginseng)). Among them, P. notoginseng is a highly valuable Chinese materia medica with a hemostatic effect and is mainly used for the treatment of trauma and ischemic cardiovascular diseases. It is documented in the Chinese pharmacopoeia and is one of the most common herbs prescribed by TCM practitioners. On the other hand, G. japonica and S. aizoon are often used as folk medicines in China and usually via self-medication. These three medicinal herbs are difficult to distinguish by self-medicating individuals or even by TCM practitioners because they have the same or similar herbal names, similar appearances, and similar medicinal indications. In addition, in most cases, the provided herbs are in the form of a powder, capsule, decoction, or liquor, rather than the original plant and/or medicinal part of the raw materials, further complicating the situations for the users. Therefore, PA-induced HSOS due to the intake of PA-producing Tu-San-Qi (G. japonica) has been frequently reported in China [9, 20, 22].
In this review article, PA-producing G. japonica is first compared with and distinguished from the two non-PA-producing herbal plants, S. aizoon and P. notoginseng, in terms of their plant morphologies, distributions, medicinal effects, and chemical profiles. A total of 2156 Tu-San-Qi-induced HSOS cases reported from 1980 to 2019 in China are then reviewed by addressing their clinical features, diagnostic workups, therapeutic interventions, and outcomes. Furthermore, by using our developed mechanism-based biomarker, the definitive diagnosis of 58 PA-producing Tu-San-Qi (G. japonica)-induced HSOS patients is also elaborated.
Differentiation of Tu-San-Qi-related herbs
Among the three Tu-San-Qi-related plant species, P. notoginseng, S. aizoon, and G. japonica belong to the Araliaceae, Crassulaceae, and Compositae families, respectively [23–27]. It is worth noting that G. japonica (Thunb.) Juel is the currently adopted Latin name by the World Flora Online, a globally recognized database for the list of authentic names for all known plant species [28] and is also used in the present article. Other names, such as G. segetum (Lour.) Merr., G. pinnatifida (Lour.) DC., and G. truncata Kerr, which have been used in many previous publications, are considered ‘synonyms’ representing the same plant species. The main features that distinguish the three Tu-San-Qi plant species and their medicinal parts (root/rhizome) are summarized in Table 1. In terms of the overall morphologies of the plants, P. notoginseng is an erect herb and is the shortest among the three (Fig. 1a), S. aizoon is a succulent herb with a height between that of the other two plants (Fig. 1b), and G. japonica is the tallest plant (Fig. 1c). Compared to the whole plants, the medicinal parts (root/rhizome) of the three plants are relatively difficult to differentiate in terms of their main distinguishing features: (i) the spindle-shaped main root of P. notoginseng is thick and fleshy and often has branches with braided protrusions, and its cross-section is gray-green (Fig. 1d); (ii) the rhizome of S. aizoon is nearly lignified with a hard texture and a dark brown cross-section (Fig. 1e); and (iii) the outer skin of the root of G. japonica is an earthy brown, and the cross-section is grayish-white (Fig. 1f). In terms of the geographical distributions, P. notoginseng is mainly distributed in Southwest and Southeast China, S. aizoon is distributed in most regions of the country except South China, and G. japonica is mainly distributed in Southwest, East, and Southeast China (Table 1). All three plant species have been documented to have similar clinical efficacies in promoting blood circulation and detumescence and relieving pain. They have all been recorded in ancient Chinese materia medica and are widely used in TCM practice. In addition, the aerial part of PA-producing G. japonica is also consumed as a vegetable and herbal tea in some areas of China [29]. Modern pharmacological studies have identified that G. japonica contains hepatotoxic PA [9, 20, 21], while P. notoginseng and S. aizoon are non-PA-producing herbs.
Table 1.
The main features of three commonly used Tu-San-Qi-related herbs.
| Items | Comparison of three kinds of 三七 San-Qi | ||
|---|---|---|---|
| Chinese name | 三七 | 景天三七 | 菊三七 |
| (Prescribed, nontoxic) | (Self-medication, nontoxic) | (Self-medication, toxic) | |
| Chinese Pinyin name | San-Qi | Jing-Tian-San-Qi | Ju-San-Qi |
| Synonym | Tian-Qi, Dian-San-Qi, Jin-Bu-Huan | Tu-San-Qi, Fei-Cai, Ba-Xian-Cao | Tu-San-Qi, Jian-Zhong-Xiao, Ru-Xiang-Cao, Nai-Cao |
| Origin | Roots of Panax notoginseng (Burkill) F.H. Chen | Roots or whole herbs of Sedum aizoon L. | Roots or whole herbs of Gynura japonica (Thunb.) Juel |
| Plant morphologies | Herbs, perennial, 20–60 cm tall | Herbs, perennial, fleshy, hairless, 20–80 cm tall | Herbs, perennial, high and robust, 60–100 cm or taller |
| Growing environment | Forests; ASL 1200–1800 m | Scrub, ravine edges in valleys, grasslands on rocky slopes, and so on; ASL1000–3100 m | Valleys, grassy slopes, forests, forest margins; ASL 1200–3000 m |
| Distribution in China and the world | SE Yunnan; cultivated in Fujian, SW Guangxi, Jiangxi, Zhejiang [N Vietnam] | Anhui, Gansu, Hebei, Heilongjiang, Henan, Hubei, Jiangsu, Jiangxi, Jilin, Liaoning, Nei Mongol, Ningxia, Qinghai, Shaanxi, Shandong, Shanxi, Sichuan, Zhejiang [Korea, Japan, Mongolia, Russia] | Anhui, Fujian, Guangxi, Guizhou, Hebei, Henan, Hubei, Hunan, Jiangsu, Jiangxi, Shaanxi, Sichuan, Taiwan, Yunnan, Zhejiang [Japan, Nepal, Thailand] |
| Macroscopic features of the medicinal part |
The roots are conical, spindle-shaped, or irregularly shaped, 1–6 cm long and 1–4 cm in diameter. The surface is grayish yellow to brownish black, with waxy luster, stem marks on the top, knob-like protrusions around, intermittent longitudinal wrinkles on the sides, and root fractures. The weight and texture are solid. After breaking, the section is grayish green, yellowish green, or grayish white; It smells a bit scent, tastes sweet after bitter |
The rhizome is short and slightly blocky; the surface is grayish brown, with several roots and varying thickness; the texture is hard and the section is dark brown or white; It smells a bit scent, tastes harsh |
The root is a fist-shaped round block with a length of 3–6 cm and a diameter of about 3 cm. The surface is grayish brown or brownish yellow. There are many tumor-like protrusions on the whole. There are often stems or buds on the top of the protrusions. Broken. It is solid, not easy to break, the section is not flat, it is white when fresh, and the dry is yellowish; It has no smell, tastes slightly bitter |
| Function and indication in folk medicine |
1. Stop bleeding and dissipate blood stasis 2. eliminate swelling and relieve pain. Treat various hemorrhagic diseases |
1. dissipate blood stasis and stop bleeding, detoxify. Mainly treat vomiting blood, hemoptysis, defecation hemorrhage, traumatic bleeding, dysmenorrhea, traumatic injury, insect bite and so on |
1. Stop bleeding and dissipate blood stasis 2. Eliminate swelling and relieve pain 3. Dissipate heat and detoxify. Mainly treat vomiting blood, hemoptysis, defecation hemorrhage, traumatic bleeding, dysmenorrhea, traumatic injury, insect bite, etc. |
| Bioactive constituents | Ginsenosides, amino acids, volatile oil | Monosaccharide, disaccharide, flavonoids |
Pyrrolizidine alkaloids, 3-epi-diosgenin-3-β-D-glucopyranoside |
| Usage and dosage |
Oral: 3–9 g (decoction), 1–3 g (powder) External: moderate amount |
Oral: 15–30 g (decoction), 30–60 g (Fresh herbs in squeezing juice) External: moderate amount |
Oral: 3–15 g (roots in decoction), 1.5–3 g (roots in powder), 10–30 g (whole herbs or leaves) External: moderate amount |
| Earliest literature |
Compendium of Materia Medica (本草纲目) A.D. 1596 |
Jiuhuang Materia Medica (救荒本草) A.D. 1406 |
Diannan Materia Medica (滇南本草) A.D. 1436 |
Fig. 1. Photographs of the three commonly used Tu-San-Qi-related herbs.
Herbal plants and medicinal parts of Panax notoginseng (a, d), Sedum aizoon (b, e) and Gynura japonica (c, f).
The reported Tu-San-Qi-induced HSOS cases
We performed a systematic search of the literature on Tu-San-Qi-induced HSOS from the first clinical case documented in 1980 [30] until the end of 2019 in China. All the articles were searched via PubMed and three Chinese-language databases, including the China National Knowledge Infrastructure (CNKI), VIP, and Wanfang databases. The terms ‘sanqi’, ‘pyrrolizidine alkaloids’, ‘sinusoidal obstruction syndrome’, and ‘hepatic venoocclusive disease’ were searched in the PubMed database. In the three Chinese-language databases, the terms ‘san qi’ (三七), ‘bi luo wan’ (吡咯烷), ‘gan dou zu se’ (肝窦阻塞) and ‘gan xiao jing mai bi se’ (肝小静脉闭塞) were searched. The latest search was performed on December 31, 2019. Case reports are defined as manuscripts that document available clinical information for individual patients; otherwise, if the information is documented for more than one patient, the manuscript is defined as a case series. From 1980 to 2019, there were (i) 91 Tu-San-Qi-induced HSOS case reports involving 124 patients; (ii) 87 Tu-San-Qi-induced HSOS case series involving 1645 patients; and (iii) 24 Tu-San-Qi-related HSOS case series (i.e., induced by mixed etiologies including Tu-San-Qi) involving 387 patients. In total, from 1980 to 2019, 2156 Tu-San-Qi-related HSOS cases have been reported in China.
Case reports
Among the 91 Tu-San-Qi-induced HSOS case reports, 124 patients were from 74 institutions located in 20 provinces or regions in China (Table 2). These patients were admitted between 1973 and 2019. The specific clinical features, diagnostic workup, therapy, and outcome information were provided for individual patients in the original reports. The demographics and clinical features of these patients are summarized in Table 3.
Table 2.
Distribution of Tu-San-Qi-induced HSOS case reports and case series reported across China.
| Provinces or regions | Patient numbers | ||
|---|---|---|---|
| Case report | Case series | Total | |
| Jiangsu | 17 | 754 | 771 |
| Zhejiang | 30 | 300 | 330 |
| Henan | 3 | 244 | 247 |
| Hubei | 12 | 127 | 139 |
| Anhui | 5 | 90 | 95 |
| Beijing | 10 | 33 | 43 |
| Guangdong | 4 | 22 | 26 |
| Jiangxi | 1 | 21 | 22 |
| Shanghai | 8 | 11 | 19 |
| Shandong | 11 | 8 | 19 |
| Liaoning | 3 | 15 | 18 |
| Hunan | 4 | 6 | 10 |
| Shaanxi | 2 | 8 | 10 |
| Jilin | 7 | 0 | 7 |
| Hebei | 0 | 6 | 6 |
| Tianjin | 1 | 0 | 1 |
| Fujian | 1 | 0 | 1 |
| Chongqing | 1 | 0 | 1 |
| Guizhou | 1 | 0 | 1 |
| Yunnan | 1 | 0 | 1 |
| Gansu | 1 | 0 | 1 |
Table 3.
Demographic and clinical features of 124 individual patients from 91 case reports.
| Variables | Number of patients | Percentage of patients | Values (mean ± SD) |
|---|---|---|---|
| Gender (n = 124) | |||
| Male | 68 | 54.8% | NA |
| Female | 56 | 45.2% | NA |
| Age (years) (n = 123) | NA | NA | 54.16 ± 21.52 |
| Tu-San-Qi formulation (n = 124) | |||
| Unclear | 39 | 31.4% | NA |
| Decoction | 35 | 28.2% | NA |
| Medicinal liquor | 26 | 21.0% | NA |
| Tea | 8 | 6.5% | NA |
| Powder | 13 | 10.5% | NA |
| Fresh product | 3 | 2.4% | NA |
| Tu-San-Qi ingestion | |||
| Duration (month) | 105 | NA | 3.17 ± 9.17 |
| Dosage regimen (g/day) | 41 | NA | 66.24 ± 180.97 |
| Total dosage (g) | 53 | NA | 2701.30 ± 6425.59 |
| Clinical presentation (n = 122) | |||
| Ascites | 122 | 100% | NA |
| Hepatomegaly | 79 | 64.8% | NA |
| Jaundice | 70 | 57.4% | NA |
| Pleural effusion | 50 | 41.0% | NA |
| Lower limbs edema | 50 | 41.0% | NA |
| Splenomegaly | 41 | 33.6% | NA |
| Upper gastrointestinal bleeding | 7 | 5.7% | NA |
| Laboratory testa (n = 117) | |||
| ALT (U/L) | 102 | 87.2% | 236.59 ± 385.08 |
| AST (U/L) | 99 | 84.6% | 222.15 ± 329.27 |
| Total bilirubin (μmol/L) | 99 | 84.6% | 64.72 ± 91.08 |
| Albumin (g/L) | 87 | 74.4% | 32.25 ± 6.31 |
| GGT (U/L) | 66 | 56.4% | 131.96 ± 138.93 |
| PT (s) | 51 | 43.6% | 17.93 ± 7.51 |
| ALP (U/L) | 50 | 42.7% | 180.50 ± 112.19 |
| Imaging examination (n = 120) | |||
| Ultrasound | 99 | 82.5% | NA |
| Computed tomography | 91 | 75.8% | NA |
| Angiography | 30 | 25.0% | NA |
| Magnetic resonance imaging | 25 | 20.8% | NA |
| Upper gastrointestinal endoscopy (n = 120) | 26 | 21.7% | NA |
| Esophageal and gastric varices (n = 26) | 9 | 34.6% | NA |
| Liver biopsy (n = 120) | 45 | 37.5% | NA |
| Therapeutic treatments (n = 108) | |||
| Diuretics | 90 | 83.3% | NA |
| Anticoagulation | 27 | 25.0% | NA |
| Albumin | 25 | 23.1% | NA |
| Paracentesis | 22 | 20.4% | NA |
| Liver transplantation | 4 | 3.7% | NA |
| Outcome (n = 107) | |||
| Survival | 88 | 82.2% | NA |
| Death | 19 | 17.8% | NA |
n number of patients with available information, s second, NA Not applicable.
aALT normal reference range: <40 U/L; AST normal reference range: <35 U/L; ALP normal reference range: 35–100 U/L; GGT normal reference range: <45 U/L; Total bilirubin normal reference range: 5–28 µmol/L; Serum albumin levels normal reference range: 40–55 g/L; PT normal reference range: 10–15 s.
The three most common clinical features among these patients were typical HSOS symptoms, such as ascites, hepatomegaly, and jaundice, while only seven patients presented upper gastrointestinal bleeding. Herbal decoction was the most common form of Tu-San-Qi ingested. The average duration between the intake of Tu-San-Qi and the onset of clinical symptoms was 3.17 ± 9.17 months (range: 2 days–3 years). The average total intake dose of Tu-San-Qi during the entire period was 2701.30 ± 6425.59 g of crude herb (range: 10–18,000 g). Most of the patients showed elevated serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma glutamyl transpeptidase (GGT), and total bilirubin levels. Some patients showed mildly decreased serum albumin levels and a prolonged prothrombin time (PT). Most patients underwent ultrasound and computed tomography (CT) for the diagnosis of HSOS. In addition, 26 patients underwent upper gastrointestinal endoscopy, 9 of whom (34.6%) had positive gastroesophageal varices, while 45 patients underwent liver biopsy for further confirmative diagnosis. Conventional drug therapy, including diuretics, paracentesis, and albumin infusion, was prescribed for the treatment of ascites for most patients. Anticoagulation therapy was prescribed for 27 patients. Liver transplantation was performed for four patients. Based on the available survival time and status information provided for 107 patients, after treatment, 88 patients survived, while 19 patients died.
Case series reports
Among the 87 Tu-San-Qi-induced HSOS case series, 1645 patients were from 55 institutions in 14 provinces or regions in China (Table 2). These patients were admitted between 1996 and 2018. Both case reports and case series reports revealed that there was a high prevalence of Tu-San-Qi poisoning in East China, especially in the areas near Shanghai, including Jiangsu and Zhejiang provinces. This is consistent with the geographic distribution of PA-producing Tu-San-Qi (G. Japonica), indicating a high risk of PA exposure in those areas.
The main clinical manifestations of these patients were ascites, hepatomegaly, jaundice, pleural effusion, lower limb edema, splenomegaly, upper gastrointestinal bleeding, gastroesophageal varices, and hepatic encephalopathy. The patients used Tu-San-Qi to self-treat trauma, arthritis, arthralgia, back pain, leg pain, hemoptysis, cerebral hemorrhage, and irregular menstruation. The forms of Tu-San-Qi ingested included decoction, liquor, powder, tea, and steamed root/rhizome. The duration between the use of Tu-San-Qi and the onset of clinical symptoms was reported in 49 case series and ranged from 1 day to 20 years. The total intake dose of Tu-San-Qi during the entire period was clearly reported in 16 case series and ranged from 100 to 127 kg. The demographic information is shown in Fig. 2. Diagnostic workup was clearly reported in 77 case series. Liver biopsy was performed for 363 patients from 42 case series. Therapeutic treatments were reported for 1003 (60.97%) patients from 52 case series. Diuretics and albumin were two of the most commonly used drugs for treating the patients. Other drugs, including hepatoprotective drugs, anticoagulants, and those that improve circulation and paracentesis, were also used. For patients who did not respond to anticoagulant and nonanticoagulant therapy, transjugular intrahepatic portosystemic shunts (TIPSs) were implanted. Liver transplantation was reported for 2 (0.12%) patients from 2 case series. Outcomes were reported for 646 (39.27%) patients from 48 case series, including recovery for 331 (20.12%) patients from 31 case series, chronicity for 126 (7.66%) patients from 23 case series, and death for 189 (11.49%) patients from 30 case series (Fig. 2). On the other hand, for the 387 patients from the 24 Tu-San-Qi-related HSOS case series, because their HSOS was induced by mixed etiologies rather than Tu-San-Qi alone, their detailed clinical data are not interpreted in the present article.
Fig. 2. Patient information from the care series reports.
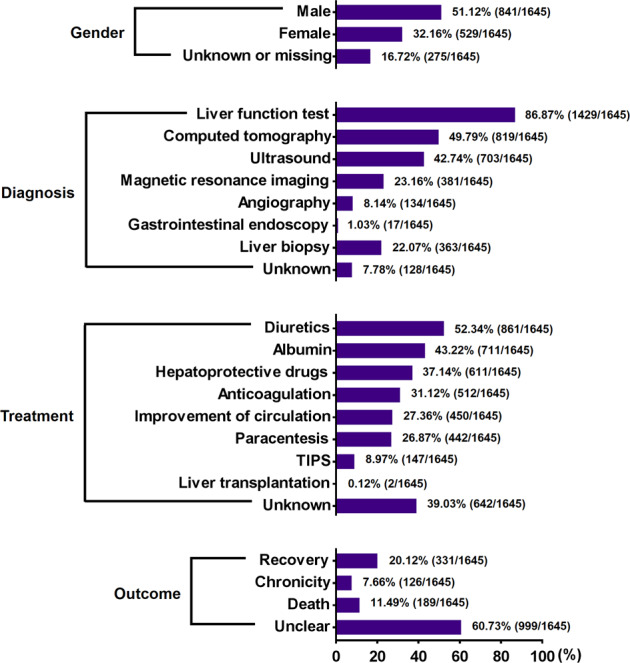
Demographics, diagnostic workups, therapeutic interventions, and outcomes of 1645 individual patients from 87 case series reported across China.
The current diagnosis and treatment method
As reported, the clinical features in Tu-San-Qi-induced HSOS patients vary from asymptomatic to severe conditions. The symptoms usually appear from a few weeks to several years after the intake of Tu-San-Qi. The typical manifestations of these patients, including abdominal distention, abdominal pain, ascites, malaise, hepatomegaly, jaundice, and edema, are nonspecific and similar to the symptoms and signs seen in patients suffering from other liver diseases, such as viral hepatitis, decompensated cirrhosis, and BCS [7]. The liver function test mostly shows an elevation in serum total bilirubin, ALT, AST, ALP, and GGT levels. Traditional imaging examinations, including ultrasound, CT, and magnetic resonance imaging (MRI) showing hepatomegaly, splenomegaly, and ascites, usually give no specific information for the diagnosis of PA-induced HSOS. Angiography can be used to differentiate between PA-induced HSOS and BCS. Liver biopsy can be applied to confirm the diagnosis, although it is not a routine practice in the clinic and cannot be performed on patients with severe ascites and/or poor coagulative function [31]. Recently, an expert consensus on the diagnostic criteria for PA-induced HSOS, which was referred to as the “Nanjing criteria”, was announced by the Hepatobiliary Diseases Committee of the Chinese Society of Gastroenterology [32]. The “Nanjing criteria” include (1) abdominal distention and/or pain in the hepatic region, as well as hepatomegaly and ascites; (2) elevation in serum total bilirubin or abnormal laboratory liver tests; (3) evidence on enhanced CT or MRI; or (4) pathological evidence that excludes other types of liver injury, plus a history of ingesting PA-producing herbs. However, because in most cases, patients can only provide an ambiguous history of the drug/herb used and imaging examinations largely rely on personal experience and expertise, the definitive diagnosis of Tu-San-Qi-induced HSOS remains a challenge for physicians and gastroenterologists.
To date, there is no uniform approach to the treatment of PA-producing Tu-San-Qi-induced HSOS. The first and most important step in the management of this condition is to avoid further injury by withdrawing Tu-San-Qi intake and providing supportive and symptomatic care. Diuretics, hepatoprotective drugs, albumin infusion, and nutrition support, as well as drugs that can improve the microcirculation, such as prostaglandin E1, salvia, Dai-Kenchu-To (also called ‘DaJianZhong-Tang’), and glucocorticoids, are commonly used and have been shown to aid in patient recovery [33–35]. Anticoagulant therapy has been applied for patients with ascites and jaundice during the acute/subacute stage [32]. TIPSs, which are widely used in the treatment of portal hypertension-related complications in patients with cirrhosis, appear to result in a favorable profile for Tu-San-Qi-induced HSOS patients who do not respond to supportive symptomatic/anticoagulant treatment [36–38]. Furthermore, the recently developed anticoagulant-TIPS ladder therapeutic strategy has been reported to significantly improve the prognosis of patients with PA-induced HSOS [32, 36]. In light of the high mortality of severe HSOS, for late stage/severe cases, liver transplantation is the only way to save the patient’s life, but this is not guaranteed [39]. In addition, defibrotide, a polydisperse mixture of oligonucleotides with local antithrombotic, anti-ischemic, and anti-inflammatory activities, has also been claimed to be useful for treating hematopoietic stem cell transplantation-induced HSOS, which is the major cause of HSOS in Western countries [39–42]. However, it has not been authorized for clinical use in China, and studies on its clinical use in PA-induced HSOS patients are still lacking.
Development of a mechanism-based biomarker for assisting the diagnosis of PA-producing Tu-San-Qi-induced HSOS
The major problem in the diagnosis of the Tu-San-Qi-induced HSOS patients described above is that most were based on the exclusion of other etiologies of liver disease and/or retrospective identification of the history of Tu-San-Qi intake. However, routine clinical liver tests are nonspecific, and people who had ingested Tu-San-Qi often did not consider it as a medicine and would not take the initiative to report their history of Tu-San-Qi intake because Tu-San-Qi is usually consumed for self-medication; for example, it is cultivated by patients themselves or given by other relatives/friends rather than prescribed by TCM practitioners. Further complicating matters, among all the above reported Tu-San-Qi-induced HSOS cases, none of the causative Tu-San-Qi samples were collected and authentically identified, and thus, G. japonica and even the non-PA-producing S. aizoon or P. notoginseng were suspected to be responsible for the condition. Because of public unawareness and the lack of a definitive diagnostic method for Tu-San-Qi-induced HSOS, the incidence reported thus far might be highly underestimated, and the currently documented cases may represent just the tip of the iceberg. A definitive diagnostic method for PA-producing Tu-San-Qi-induced HSOS is thus urgently and crucially needed to appropriately address this safety issue.
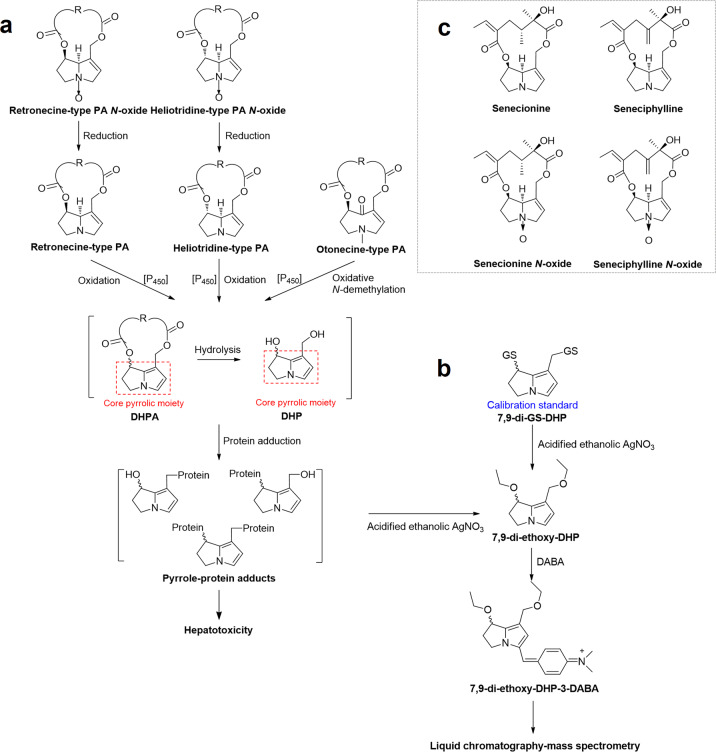
Metabolic activation of PAs and the formation of pyrrole-protein adducts
Based on the structures of their necine bases, PAs can be classified into retronecine-type, heliotridine-type, otonecine-type, and platynecine-type. The first three types of PA possess a 1,2-unsaturated double bond in the necine base and are hepatotoxic (Fig. 3a) [10, 11]. Retronecine-type and heliotridine-type PAs commonly coexist with their corresponding pyrrolizidine alkaloid N-oxides (PA N-oxides) with variable compositions in different plants [43–45]. Catalyzed by hepatic cytochrome P450 monooxygenases (CYPs), especially CYP3A4, toxic PAs are metabolically activated to generate dehydropyrrolizidine alkaloids (DHPAs). All DHPAs contain an identical core pyrrolic moiety regardless of the structures of their parent PAs. DHPA is chemically and biologically reactive and can rapidly hydrolyze to the less reactive (±)-6,7-dihydro-7-hydroxy-1-hydroxymethyl-5H-pyrrolizine (DHP). Both DHPA and DHP can covalently bind with cellular proteins to form pyrrole-protein adducts, causing damage to some functional proteins and resulting in hepatotoxicity, such as HSOS (Fig. 3a) [8, 10, 46–50]. Moreover, we recently confirmed that PA N-oxides also induce HSOS in humans [21]. Using rodent models, we determined that PA N-oxides induce hepatotoxicity via their metabolic reduction to the corresponding PA mediated by intestinal microbiota as well as intestinal and hepatic CYPs, in particular CYP1A2 and CYP2D6. The resultant PAs then undergo CYP-mediated metabolic bioactivation in the liver to generate DHPAs, which interact with cellular proteins, leading to hepatotoxicity [21, 49]. Furthermore, we also demonstrated that the hepatotoxic potency of PA N-oxides was significantly lower than that of the corresponding PAs primarily due to the significantly lower oral absorption of PA N-oxides and the uncompleted biotransformation of PA N-oxides to PAs [21, 51].
Fig. 3. The mechanism of PA-induced hepatotoxicity.
Metabolic activation of PAs and N-oxides to induce hepatotoxicity (a), precolumn derivatization scheme and analysis of pyrrole-protein adducts (b), and chemical structures of senecionine, seneciphylline, senecionine N-oxide and seneciphylline N-oxide identified in Gynura japonica (c). DHPA dehydropyrrolizidine alkaloids, DHP (±)-6,7-dihydro-7-hydroxy-1-hydroxymethyl-5H-pyrrolizine, DABA 4-dimethylaminobenzaldehyde.
Development of a mechanism-based biomarker of PA-exposure
The abovementioned pyrrole-protein adducts have been implicated to play a critical role in PA-induced hepatotoxicity [10]. In 2011, our group developed a specific and sensitive method for the qualitative determination of pyrrole-protein adducts in the blood of a PA-producing Tu-San-Qi-induced HSOS patient in Zhongshan Hospital, Shanghai, China [9]. This novel analytical method involves (i) reaction of blood pyrrole-protein adducts with AgNO3 to form 7,9-di-ethoxy-DHP; (ii) treatment of 7,9-di-ethoxy-DHP with 4-dimethylaminobenzaldehyde (DABA) to produce the analyte 7,9-di-ethoxy-DHP-3-DABA; and (iii) analysis of the analyte by liquid chromatography–mass spectrometry (Fig. 3b). This novel method has been successfully applied in the diagnosis of one HSOS patient, who claimed taking Tu-San-Qi as self-medication with HSOS confirmed by liver biopsy [11]. The herb consumed by this patient was authenticated as G. japonica, and four hepatotoxic PAs/PA N-oxides, namely, senecionine, seneciphylline, senecionine N-oxide, and seneciphylline N-oxide (Fig. 3c), were identified in the herb. Pyrrole-protein adducts were unequivocally detected in the blood of this Tu-San-Qi-exposed patient. Moreover, an animal study showed a good correlation of liver injury with the ingestion of G. japonica [21]. Therefore, for the first time, we provided firm evidence of PA-induced HSOS caused by the intake of PA-producing Tu-San-Qi (G. japonica) in humans and demonstrated pyrrole-protein adducts as a mechanism-based biomarker of PA exposure for assisting in the definitive diagnosis of PA-producing Tu-San-Qi-induced HSOS in the clinic.
Afterwards, our group further improved this method by using 7,9-di-GS-DHP (Fig. 3b) to construct a calibration curve for the quantitative analysis of pyrrole-protein adducts in biological specimens [20]. With this method, we were able to perform the simultaneous qualitative and quantitative determination of the biomarker pyrrole-protein adducts in blood samples. We then further tested the blood samples of 86 suspected DILI patients with unknown etiology (excluding virtual infection, alcohol, orthodox drugs, etc.) and 18 suspected PA-producing Tu-San-Qi-induced HSOS patients who claimed to have ingested Tu-San-Qi [20, 21, 52, 53]. Pyrrole-protein adducts were detected in all 18 suspected Tu-San-Qi-induced HSOS patients and 39 (~45%) of the suspected DILI patients but not in any of the healthy volunteers, indicating an unrecognized high prevalence of PA-induced liver injury in China. Among all the diagnosed PA-exposure patients, 15 patients provided samples of their consumed herbs. All herbs were identified as PA-producing Tu-San-Qi (G. japonica). Furthermore, the remaining confirmed PA-induced DILI patients were also considered to have Tu-San-Qi-induced HSOS based on their claims of using Tu-San-Qi as self-medication for tonic or improvement of blood circulation, although the herbal samples were unavailable. Therefore, together with the abovementioned 2156 Tu-San-Qi-related HSOS cases reported in China, through the end of 2019, a total of 2214 PA-producing Tu-San-Qi (G. japonica)-induced HSOS patients were diagnosed in China.
Currently, there is no gold standard for the diagnosis of DILI. In the clinic, a diagnosis is commonly made retrospectively or after the exclusion of other causes. Therefore, the rates of misdiagnosis for DILI are very high, with reports of up to 47.1% and 28.5% in Europe and America, respectively [54–56]. The discovery of a drug-specific biomarker would undoubtedly help assist in making accurate and confirmative diagnosis of DILI. However, it is challenging to find a specific biomarker, such as a protein or a metabolite, at a commonly high level among thousands of other metabolites and proteins. This situation would be even more challenging if applied to HILI because of the much more complex compositions of herbal medicines. Until now, no clinically applicable biomarkers have been successfully validated and approved in herbal medicine research due to their inadequate sensitivity and specificity [57–59]. Our developed biomarker, the group of pyrrole-protein adducts, aided in providing the first and direct evidence of PA-induced HSOS resulting from the intake of PAs, especially PA-producing Tu-San-Qi in the present cases. This biomarker is mechanism-based, specific, and only present in biological specimens after PA exposure and achieves a new step in overcoming the difficulties encountered with HILI. Our developed biomarker has been well recognized and used to aid in the diagnosis of both PA-producing herb-induced liver toxicity, such as the abovementioned PA-producing Tu-San-Qi-induced HSOS [52, 60], and PA-contaminated food-induced liver injury, such as PA-contaminated grain-induced liver disease in Ethiopia [61]. Moreover, based on its successful application in assisting in the definitive diagnosis of PA-induced HSOS, this biomarker has been included as the diagnostic index for the confirmation of PA intake in the “Nanjing criteria” [32].
The clinical features of 15 Tu-San-Qi-induced HSOS patients and the PA/PA N-oxide contents of their ingested Tu-San-Qi samples
The aforementioned 15 HSOS patients were admitted to Zhongshan Hospital from April 2009 to June 2013 and claimed to have ingested Tu-San-Qi [20]. Most of the patients showed elevated serum ALT (8/15, 53.3%) and AST (13/15, 86.7%) levels (Table 4, Part A). The median value of total bilirubin was 38.1 μmol/L with a range of 11.7–198 μmol/L. Imaging examinations showed diffuse, patchy hepatic enhancement, periportal edema, and ascites but excluded other vascular diseases. Hepatic pathology demonstrated the expansion and congestion of the sinus, endothelial swelling, wall thickening, and incomplete lumen occlusion of the small hepatic vessels. Mortality was 40% (6/15) (Table 4, Part A). The patients ingested Tu-San-Qi in different formulations. All patients provided herb samples (whole plants or medicinal parts) used to make the formulations, and all the provided herbs were authenticated as G. japonica. As shown in Table 4, Part A, the duration between the intake of Tu-San-Qi and the onset of clinical symptoms ranged from 4 to 730 days. The total dose of the ingested herbs for the entire period was estimated to range from 350 to 1650 g. The total content of PAs/PA N-oxides was determined in all herbs and found to vary substantially, with a range of 0.27–13.6 mg/g herb (Table 4, Part B). Based on the available information, the lowest PA content that induced HSOS in this cohort is estimated to be 7.58 mg PA/day (calculated based on P9 in Table 4). Among all herb samples tested, seneciphylline N-oxide and senecionine N-oxide, rather than the corresponding PAs, were identified as the predominant forms. It is worth noting that PA N-oxides were found as the sole or predominant form of PAs in 14/15 samples, while only 1 herb sample (GJ1) contained a substantially lower portion (12%) of PA N-oxides. Although the liver toxicity induced by PA N-oxide has been demonstrated to be significantly lower than that of the corresponding PAs [21], our findings suggested that PA N-oxides, rather than the PAs themselves, caused liver damage in most (14/15) of these PA-producing Tu-San-Qi-induced HSOS patients.
Table 4.
Clinic features of hepatic sinusoidal obstruction syndrome (HSOS) patients associated with Tu-San-Qi exposure (Part A) and contents of pyrrolizidine alkaloids (PA) and pyrrolizidine alkaloids N-oxides (PA N-oxide) in herbal samples ingested by patients (Part B).
| Part A | ||||||||
|---|---|---|---|---|---|---|---|---|
| Patient code | Liver function testa | Formulation | Duration (days) | Dosage | Blood level of pyrrole-protein adducts (nM)b | Outcome (at ~100 days after diagnosis)c | ||
| ALT (U/L) | AST (U/L) | Total bilirubin (µmol/L) | ||||||
| P1 | 37.0 | 68.0 | 26.4 | Tea | 4 | Unclear | 6.07 | Recovery |
| P2 | 61.0 | 58.0 | 129.6 | Decoction | 15 | 2000 mL | 12.90 | Death |
| P3 | 19.0 | 34.0 | 198.0 | Medicinal liquor | 730 | Unclear | 11.80 | Death |
| P4 | 20.0 | 56.0 | 38.1 | Capsule | 31 | Unclear | 15.90 | Death |
| P5 | 15.0 | 46.0 | 26.4 | Unclear | 72 | Unclear | 4.70 | Recovery |
| P6 | 1027.0 | 930.0 | 12.8 | Boiled egg with herb | 22 (take every other day) | 150 g/day | 74.40 | Liver transplant |
| P7 | 37.0 | 40.0 | 34.0 | Medicinal liquor | 7 | 15–30 mL | 11.50 | Recovery |
| P8 | 177.0 | 123.0 | 28.9 | Medicinal liquor | 121 | 100 mL/day | 0.33 | Recovery |
| P9 | 128.8 | 80.3 | 38.9 | Powder | 204 | ~2 g/day | 10.70 | Death |
| P10 | 37.0 | 71.0 | 40.8 | Decoction | 11 | 50 mL/day | 20.02 | Recovery |
| P11 | 549.0 | 395.0 | 11.7 | Medicinal liquor | 45 | 100–150 mL/day | 32.11 | Chronicity |
| P12 | 394.0 | 537.0 | 43.8 | Decoction | 10 | 60 mL/day | 0.65 | Death |
| P13 | 11.0 | 12.0 | 41.2 | Powder | 100 | Unclear | 0.14 | Recovery |
| P14 | 369.0 | 576.0 | 47.5 | Medicinal liquor | 7 | Unclear | 8.50 | Death |
| P15 | 344.0 | 237.0 | 17.6 | Unclear | 10 | Unclear | 0.14 | Chronicity |
| Part B | |||||
|---|---|---|---|---|---|
| Herb code | PA content (mg/g herb) | PA N-oxide content (mg/g herb) | Total PA + PA N-oxide contents (mg/g herb) (% of PA N-oxide)b | ||
| Senecionine | Seneciphylline | Senecionine N-oxide | Seneciphylline N-oxide | ||
| GJ1 | 0.109 ± 0.002 | 0.132 ± 0.003 | 0.029 ± 0.000 | 0.004 ± 0.000 | 0.274 ± 0.005 (12.0%) |
| GJ2 | ND | ND | 0.484 ± 0.064 | 0.345 ± 0.041 | 0.829 ± 0.105 (100.0%) |
| GJ3 | 0.076 ± 0.005 | 0.145 ± 0.003 | 4.198 ± 0.447 | 3.467 ± 0.212 | 7.886 ± 0.656 (97.2%) |
| GJ4 | 0.056 ± 0.007 | 0.239 ± 0.027 | 0.810 ± 0.102 | 1.179 ± 0.142 | 2.283 ± 0.268 (87.1%) |
| GJ5 | 0.029 ± 0.003 | 0.055 ± 0.004 | 4.597 ± 0.581 | 2.772 ± 0.462 | 7.452 ± 1.049 (98.9%) |
| GJ6 | 0.011 ± 0.002 | 0.032 ± 0.009 | 6.993 ± 1.257 | 5.729 ± 1.095 | 12.765 ± 2.313 (99.7%) |
| GJ7 | ND | ND | 0.380 ± 0.121 | 0.174 ± 0.077 | 0.554 ± 0.197 (100.0%) |
| GJ8 | ND | ND | 6.980 ± 0.377 | 1.208 ± 0.087 | 8.188 ± 0.460 (100.0%) |
| GJ9 | ND | ND | 1.277 ± 0.352 | 2.960 ± 0.446 | 4.237 ± 0.790 (100.0%) |
| GJ10 | 0.324 ± 0.006 | 0.442 ± 0.008 | 3.032 ± 0.053 | 1.948 ± 0.020 | 5.746 ± 0.077 (86.7%) |
| GJ11 | 0.018 ± 0.000 | 0.015 ± 0.000 | 9.206 ± 0.060 | 0.855 ± 0.016 | 1.828 ± 0.018 (89.2%) |
| GJ12 | 0.030 ± 0.000 | 0.024 ± 0.000 | 10.615 ± 0.057 | 2.977 ± 0.010 | 10.094 ± 0.051 (99.7%) |
| GJ13 | 0.070 ± 0.001 | 0.128 ± 0.001 | 0.882 ± 0.011 | 0.748 ± 0.006 | 13.645 ± 0.059 (99.6%) |
| GJ14 | ND | ND | 2.999 ± 0.525 | 3.240 ± 0.483 | 6.240 ± 1.004 (100.0%) |
| GJ15 | 0.709 ± 0.040 | 0.690 ± 0.039 | 4.006 ± 0.197 | 6.040 ± 0.251 | 11.445 ± 0.524 (87.8%) |
Herb codes (GJ1-15) in Part B correspond to the patient codes (P1-15) in Part A, respectively.
ND not detectable.
aALT normal reference range: <40 U/L; AST normal reference range: <35 U/L; total bilirubin normal reference range: 5–28 µmol/L.
bBlood level of pyrrole-protein adducts and the total contents of PA + PA N-oxide of GJ1-15 were reported by our group [20].
cClinical outcome was classified into recovery (signs and symptoms disappeared and drug therapies were stopped at 100 days after the onset), chronicity (signs and symptoms remained for 100 days and therapeutic drugs, such as diuretics, were needed), and severe outcome (liver injury caused the death or liver transplantation was performed within 100 days after the onset).
Pyrrole-protein adducts were unequivocally determined in the blood samples from all patients. A wide range of concentrations was measured; the median value was 10.70 nM (Table 4, Part A). It is worth noting that the blood pyrrole-protein adduct level was not necessarily correlated with the severity of the Tu-San-Qi-induced HSOS patient outcome because the level of the adducts could be affected by many factors, such as (i) the amount of Tu-San-Qi consumed, (ii) the duration of consumption, (iii) the blood sampling time, and (iv) the Tu-San-Qi withdrawal time. However, not all of these factors can be accurately determined due to the lack of availability of adequate information. Nevertheless, analysis of our developed mechanism-based biomarker revealed that it is specific for PA exposure; in combination with the identification of PAs/PA N-oxides in the ingested Tu-San-Qi, this unequivocal detection of the blood pyrrole-protein adducts in the suspected patients should undoubtedly help the confirmative diagnosis of PA-producing Tu-San-Qi-induced HSOS.
Discussion
Other PA-producing herbs used in China
In China, it is common for more than one plant species to be used as the plant source for the same TCM herb, which often confuses consumers and even TCM practitioners. As a result, unauthorized medicinal herbs account for a large proportion of HILIs in China [62]. For instance, notorious aristolochic acid-induced nephrotoxicity results from the erroneous use of the aristolochic acid-producing Aristolochia fangchi as a substitute for the nontoxic Stephania tetrandra. Unlike the aristolochic acids that are present primarily in plants in the Aristolochiaceae family, hepatotoxic PAs are widely distributed across the plant kingdom in up to 13 distantly related angiosperm families, rendering PA-induced hepatotoxicity more pronounced and difficult to avoid. Based on the currently available data, it is clear that PA-producing G. japonica, when used as the plant source of Tu-San-Qi, is a major culprit of PA-induced HSOS in China.
Similar to the use of PA-producing G. japonica as the plant source of Tu-San-Qi, three other PA-producing herbs, namely, Shan-Zi-Wan, Pei-Lan and Dong-Bei-He-Shi, have also been reported as substitutes for three non-PA-producing TCM herbs (Table 5, Part A) [63]. Moreover, in China, ~49 herbal plants have been reported to contain PAs [64], and six of them are documented in the current edition of the Chinese pharmacopoeia (Table 5, Part B) [64, 65]. Apparently, erroneous ingestion of these PA-producing herbs may also increase the risk of PA poisoning. More importantly, many unrecognized cases of liver disorders induced by prolonged exposure to a relatively low dose of PAs may not show typical liver injury symptoms. Long-term chronic or intermittent low-level intake of PAs may imperceptibly induce chronic liver diseases such as liver fibrosis and cancer, which may not show overt symptoms in the early stage of disease development. Therefore, public education on the misuse of PA-producing herbal plants is a practical and important step in reducing the incidence of HSOS. A reporting system for adverse events should also be emphasized and established. Instructions for the diagnosis and treatment of PA-induced liver injury should also be promoted and popularized to physicians and TCM practitioners.
Table 5.
Pyrrolizidine alkaloids (PA)-producing herb used as the substitute for non-PA-producing TCM herbs documented in Chinese pharmacopoeia (2015 edition) (Part A) and PA-producing TCM herbs documented in Chinese pharmacopoeia (2015 edition) (Part B).
| Part A | |||||||
|---|---|---|---|---|---|---|---|
| TCM herb documented in Chinese pharmacopoeia | Substitute | ||||||
| Chinese name | Latin name | Origin | Used in TCM herbal formulation | PA | Chinese name | Origin | PA identified |
|
紫菀 Zi-Wan |
Asteris Radix et Rhizoma | Roots or rhizome of Aster tataricus L. f. | Ermu Ansou Pills; Zhikebao Tablets; Zhike Juhong Mixture; Zhike Juhong Pills; Qingyu Biwen Pills; Ruyi Dingchuan Tablets; Kechuanshun Pills; Fufang Haqing Tablets; Jizhi Syrup; Runfei Zhisou Pills; Gejie Dingchuan Pills; Gejie Dingchuan Capsules; Juhong Pills; Juhong Granules | Nil |
山紫菀 Shan-Zi-Wan |
Roots or rhizome of Ligularia genus, such as L. fischeri Turcz. (硬紫菀), L. hodgsonii Hook. (川紫菀/鹿蹄橐吾), L. duciformis (C. Winkl.) Hand.-Mazz. (大黄橐吾), etc. | Clivorine, Ligularine, Isoline. |
|
泽兰 Ze-Lan |
Lycopi Herba |
Aerial parts of Lycopus lucidus Tilrcz. var. hirtus Rege |
Yigan Yangyin Huoxue Granules; Sizhi Xiangfu Pills; Qianliexin Gapsules; Qianlietong Tablets; Tiaojing Zhitong Tablets | Nil |
佩兰 Pei-Lan |
Aerial parts of Eupatorium fortunei Turcz. (佩兰) and E. heterophyllum DC. (单叶佩兰) | Amabiline, Supinine, Echinatine, Rinderine, Intermedine, lycopsamine. |
|
鹤虱 He-Shi |
Carpesii Fructus | Fruits of Carpesium abrotanoides L. | Huaji Mixture | Nil |
东北鹤虱 Dong-Bei-He-Shi |
Fruits of Lappula myositis V. Wolf, L. (鹤虱), L. redowskii (Hornem.) Greene. (东北鹤虱) | Lycopsamine, Intermedines, Acetylintermiedine, Acetyllycopsamine, Lasiocarpine. |
| Part B | ||||
|---|---|---|---|---|
| Chinese name | Latin name | Origin | Formulations | PA |
|
千里光 Qian-Li-Guang |
Senecionis Scandentis Herba | Aerial parts of Senecio scandens Buch. Ham. Ex D. Don | Qianbai Biyan Tablets | Senecionine, Seneciphylline, Adonifoline |
|
款冬花 Kuan-Dong-Hua |
Farfarae Flos | Flowers of Tussilago farfara L. | Ermu Ansou Pills; Chuanbei Xueli Concentrated Decoction; Zhike Juhong Mixture; Zhike Juhong Pills; Zhisou Huatan Pills; Kechuanshun Pills; Runfei Zhisou Pills; Juhong Pills; Juhong Granules | Senecionine, Senkirkine |
|
佩兰 Pei-Lan |
Eupatorii Herba | Aerial parts of Eupatorium fortunei Turcz. | Nil | Supinine, Rinderine, 7-Acetylrinderine |
|
一点红 Yi-Dian-Hong (included in Appendix) |
Emiliae Herba | Whole plant of Emilia sonchifolia (L.) DC. | Huahong Tablets; Huahong Granules | Doronine, Senkirkine |
|
紫草 Zi-Cao |
Arnebiae Radix | Roots of Arnebia euchroma (Royle) Johns. | Xiao’er Feireping Capsules; Baishi Pills; Baidianfeng Capsules; Guogong Wine; Fufang Qingdai Pills; Xiaocuo Pills; Xiaomi Suppositories; Tangshang Oil; Kangfu Xiaoyan Suppositories; Zihua Shaoshang Ointments; Zicao Ointment | O7-Angeloylretronecine, O9-Angeloylretronecine |
|
返魂草 Fan-Hun-Cao (included in Appendix) |
Senecionis Cannabifolii Herba | Aerial parts of Senecio cannabifolius Less. | Aotaile Granules | Senecicannabine |
The important contribution of PA N-oxides to PA-induced HSOS
Our findings on PA N-oxide-induced hepatotoxicity in humans and rodents revealed the important contribution of PA N-oxides to the generally defined and currently well-accepted PA-induced HSOS. It is also worth mentioning that PA N-oxides are more water soluble than the corresponding PAs, and thus, a much higher quantity of PA N-oxides would be present in an herbal decoction and ingested by a patient. Therefore, for the future risk assessment of PA-induced HSOS and the establishment of regulations for PA exposure thresholds, PA N-oxides should also be included when determining the total PA content and evaluating the toxicity of PA-producing herbs/natural products and PA-contaminated foodstuffs. In addition, the biotransformation of PA N-oxides into the corresponding PAs is the first step in the development of PA N-oxide-induced hepatotoxicity and is mainly mediated by the intestinal microbiome [21, 49, 51]. Compared with PA intoxication, the susceptibility to PA N-oxide-induced hepatotoxicity and its toxic potency are much more difficult to predict because the intestinal micorbiome is readily altered by diet, medications, and/or lifestyle. Therefore, the public should be aware of and pay greater attention to the potentially remarkable variations in PA N-oxide-induced hepatotoxicity among individual subjects who are exposed to PA N-oxides under different living environments and/or with different lifestyles.
Conclusion
The misuse of PA-producing Tu-San-Qi (G. japonica) has resulted in numerous incidences of PA-induced HSOS in China. Our established mechanism-based specific biomarker, blood pyrrole-protein adducts, is of great clinical importance in the confirmative diagnosis of PA-induced HSOS. Only with the accurate identification of the etiology is it possible to aid the patients to cease consumption of the offending agents, including PA-producing Tu-San-Qi and G. japonica, and to provide an appropriate therapeutic intervention. The authors hope that this review article can improve the awareness of PA-producing Tu-San-Qi (G. japonica) and other medicinal herb-induced HSOS among the public, herbal specialists, TCM practitioners, physicians, and healthcare policy makers and facilitate future prevention and better clinical management of this severe HILI. In conclusion, prevention is always more effective than the use of therapy after the onset of injury.
Acknowledgements
The present study was supported by the Research Grant Council of Hong Kong (GRF Grant no. 14111816 and 14106318), the CUHK Project Impact Enhancement Fund (PIEF Grant No. 3133029) and the CUHK Direct Grant (Grant No. 4054503). This article is not an official USA Food and Drug Administration (FDA) guidance or policy statement. No official support or endorsement by the USA FDA is intended or should be inferred.
Competing interests
The authors declare no competing interests.
Footnotes
These authors contributed equally: Lin Zhu, Chun-yuan Zhang
References
- 1.Chalasani NP, Hayashi PH, Bonkovsky HL, Herbert L, Navarro VJ, Lee WM, et al. ACG clinical guideline: the diagnosis and management of idiosyncratic drug-induced liver injury. Am J Gastroenterol. 2014;109:950–66. doi: 10.1038/ajg.2014.131. [DOI] [PubMed] [Google Scholar]
- 2.Kaplowitz N. Idiosyncratic drug hepatotoxicity. Nat Rev Drug Discov. 2005;4:489–99. doi: 10.1038/nrd1750. [DOI] [PubMed] [Google Scholar]
- 3.Navarro VJ, Senior JR. Drug-related hepatotoxicity. N. Engl J Med. 2006;354:731–9. doi: 10.1056/NEJMra052270. [DOI] [PubMed] [Google Scholar]
- 4.Shen T, Liu YX, Shang J, Xie Q, Li J, Yan M, et al. Incidence and etiology of drug-induced liver injury in mainland China. Gastroenterology. 2019;156:2230–41. doi: 10.1053/j.gastro.2019.02.002. [DOI] [PubMed] [Google Scholar]
- 5.Valla DC, Cazals-Hatem D. Sinusoidal obstruction syndrome. Clin Res Hepatol Gastroenterol. 2016;40:378–85. doi: 10.1016/j.clinre.2016.01.006. [DOI] [PubMed] [Google Scholar]
- 6.Wang Y, Qiao D, Li Y, Xu F. Risk factors for hepatic veno-occlusive disease caused by Gynura segetum: a retrospective study. BMC Gastroenterol. 2018;18:156. doi: 10.1186/s12876-018-0879-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang JY, Gao H. Tusanqi and hepatic sinusoidal obstruction syndrome. J Dig Dis. 2014;15:105–7. doi: 10.1111/1751-2980.12112. [DOI] [PubMed] [Google Scholar]
- 8.Edgar JA, Molyneux RJ, Colegate SM. Pyrrolizidine alkaloids: potential role in the etiology of cancers, pulmonary hypertension, congenital anomalies, and liver disease. Chem Res Toxicol. 2015;28:4–20. doi: 10.1021/tx500403t. [DOI] [PubMed] [Google Scholar]
- 9.Lin G, Wang JY, Li N, Li M, Gao H, Ji Y, et al. Hepatic sinusoidal obstruction syndrome associated with consumption of Gynura segetum. J Hepatol. 2011;54:666–73. doi: 10.1016/j.jhep.2010.07.031. [DOI] [PubMed] [Google Scholar]
- 10.Fu PP, Xia QS, Lin G, Chou MW. Pyrrolizidine alkaloids–genotoxicity, metabolism enzymes, metabolic activation, and mechanisms. Drug Metab Rev. 2004;36:1–55. doi: 10.1081/DMR-120028426. [DOI] [PubMed] [Google Scholar]
- 11.Mattocks AR. Chemistry and toxicology of pyrrolizidine alkaloids. London: Academic Press; 1986.
- 12.Smith LW, Culvenor CC. Plant sources of hepatotoxic pyrrolizidine alkaloids. J Nat Prod. 1981;44:129–52. doi: 10.1021/np50014a001. [DOI] [PubMed] [Google Scholar]
- 13.Kakar F, Akbarian Z, Leslie T, Mustafa ML, Watson J, Egmond HPV, et al. An outbreak of hepatic veno-occlusive disease in western Afghanistan associated with exposure to wheat flour contaminated with pyrrolizidine alkaloids. J Toxicol. 2010;2010:313280. doi: 10.1155/2010/313280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Edgar JA, Smith LW. Transfer of pyrrolizidine alkaloids into eggs: food safety implications. ACS Symp Ser. 1999;8:118–28. doi: 10.1021/bk-2000-0745.ch008. [DOI] [Google Scholar]
- 15.Kempf M, Heil S, Hasslauer I, Schmidt L, Ohe Kvd, Theuring C, et al. Pyrrolizidine alkaloids in pollen and pollen products. Mol Nutr Food Res. 2010;54:292–300. doi: 10.1002/mnfr.200900289. [DOI] [PubMed] [Google Scholar]
- 16.Panter KE, James LF. Natural plant toxicants in milk: a review. J Anim Sci. 1990;68:892–904. doi: 10.2527/1990.683892x. [DOI] [PubMed] [Google Scholar]
- 17.Willmot FC, Robertson GW. Senecio disease or cirrhosis of the liver due to Senecio poisoning. Lancet. 1920;2:848. doi: 10.1016/S0140-6736(01)00020-4. [DOI] [Google Scholar]
- 18.Ma J, Ruan J, Chen X, Li D, Yao S, Fu PP, et al. Pyrrole-hemoglobin adducts, a more feasible potential biomarker of pyrrolizidine alkaloid exposure. Chem Res Toxicol. 2019;32:1027–39. doi: 10.1021/acs.chemrestox.8b00369. [DOI] [PubMed] [Google Scholar]
- 19.Cooperative Group for Health and Gall Diseases, Chinese Society of Gastroenterology, Chinese Medical Association. Expert consensus on diagnosis and treatment of pyrrolizidine alkaloids-related sinusoidal obstruction syndrome (2017, Nanjing) J Clin Hepatol. 2017;33:1627–37. [Google Scholar]
- 20.Ruan JQ, Gao H, Li N, Xue JY, Chen J, Ke CQ, et al. Blood pyrrole-protein adducts-a biomarker of pyrrolizidine alkaloid-induced liver injury in humans. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2015;33:404–21. doi: 10.1080/10590501.2015.1096882. [DOI] [PubMed] [Google Scholar]
- 21.Yang MB, Ruan JQ, Gao H, Li N, Ma J, Xue JY, et al. First evidence of pyrrolizidine alkaloid N-oxide-induced hepatic sinusoidal obstruction syndrome in humans. Arch Toxicol. 2017;91:3913–2925. doi: 10.1007/s00204-017-2013-y. [DOI] [PubMed] [Google Scholar]
- 22.Wang XX, Qi XS, Guo XZ. Tusanqi-related sinusoidal obstruction syndrome in China: a systematic review of the literatures. Medicine. 2015;94:e942. doi: 10.1097/MD.0000000000000942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Commission of Chinese Materia Medica. Materia Medica of China. Shanghai (China): Shanghai Scientific and Technical Publishers; 1999a; 3: p 765–7.
- 24.Commission of Chinese Materia Medica. Materia Medica of China. Shanghai (China): Shanghai Scientific and Technical Publishers; 1999b; 5: p 839–50.
- 25.Commission of Chinese Materia Medica. Materia Medica of China. Shanghai (China): Shanghai Scientific and Technical Publishers; 1999c; 7: p 855–7.
- 26.Flora of China Editorial Committee. Beijing (China): Flora of China. Science Press; 1999a; 78: p 128.
- 27.Flora of China Editorial Committee. Beijing (China): Flora of China. Science Press; Flora of China. Science Press; 1999b; 78: p 312–3.
- 28.Gynura japonica (Thunb.) Juel. World Flora Online. 2020. http://www.worldfloraonline.org/taxon/wfo-0000011089.
- 29.Xiong AZ, Shao YL, Fang LX, Yang X, Zhang SC, Zheng J, et al. Comparative analysis of toxic components in different medicinal parts of Gynura japonica and its toxicity assessment on mice. Phytomedicine. 2019;54:77–88. doi: 10.1016/j.phymed.2018.06.015. [DOI] [PubMed] [Google Scholar]
- 30.Hou JG. Veno-occlusive disease of the liver with report of 2 cases. Zhonghua Nei Ke Za Zhi. 1980;19:187–91. [PubMed] [Google Scholar]
- 31.Zhang M, Zhuge YZ, Chen J. Hepatic venous pressure gradient and transjugular liver biopsy confirms a case of atypical hepatic venoocclusive disease. Chin J Gastroenterol. 2013;33:636–7. [Google Scholar]
- 32.Zhuge YZ, Liu YL, Xie WF, Zou XP, Xu JM, Wang JY, et al. Expert consensus on the clinical management of pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome. J Gastroenterol Hepatol. 2019;34:634–42. doi: 10.1111/jgh.14612. [DOI] [PubMed] [Google Scholar]
- 33.Beihany AA, Omar HA, Sahovic E, Chaudhri N, Mohareb FA, Sharif FA, et al. Successful treatment of hepatic veno-occlusive disease after myeloablative allogeneic hematopoietic stem cell transplantation by early administration of a short course of methylprednisolone. Bone Marrow Transpl. 2008;41:287–91. doi: 10.1038/sj.bmt.1705896. [DOI] [PubMed] [Google Scholar]
- 34.Narita M, Hatano E, Tamaki N, Yamanaka K, Yanagida A, Nagata H, et al. Dai-kenchu-to attenuates rat sinusoidal obstruction syndrome by inhibiting the accumulation of neutrophils in the liver. J Gastroenterol Hepatol. 2009;24:1051–7. doi: 10.1111/j.1440-1746.2009.05795.x. [DOI] [PubMed] [Google Scholar]
- 35.Zhu HY, Chu Y, Huo JR, Chen Z, Yang L. Effect of prednisone on transforming growth factor-β1, connective tissue growth factor, nuclear factor-κB p65 and tumor necrosis factor-α expression in a murine model of hepatic sinusoidal obstruction syndrome induced by Gynura segetum. Hepatol Res. 2011;41:795–803. doi: 10.1111/j.1872-034X.2011.00830.x. [DOI] [PubMed] [Google Scholar]
- 36.Zhuge YZ, Wang Y, Zhang F, Zhu CK, Zhang W, Zhang M, et al. Clinical characteristics and treatment of pyrrolizidine alkaloid-related hepatic vein occlusive disease. Liver Int. 2018;38:1867–74. doi: 10.1111/liv.13684. [DOI] [PubMed] [Google Scholar]
- 37.Deleve LD, Valla DC, Garcia-Tsao G. Vascular disorders of the liver. Hepatology. 2009;49:1729–64. doi: 10.1002/hep.22772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rubia J, Carral A, Montes H, Urquijo JJ, Sanz GF, Sanz MA. Successful treatment of hepatic veno-occlusive disease in a peripheral blood progenitor cell transplant patient with a transjugular intrahepatic portosystemic stent-shunt (TIPS) Haematologica. 1996;81:536–9. [PubMed] [Google Scholar]
- 39.Valla DC. Budd-Chiari syndrome and veno-occlusive disease/sinusoidal obstruction syndrome. Gut. 2008;57:1469–78. doi: 10.1136/gut.2007.133637. [DOI] [PubMed] [Google Scholar]
- 40.Corbacioglu S, Kernan N, Lehmann L, Brochstein J, Revta C, Grupp S, et al. Defibrotide for the treatment of hepatic veno-occlusive disease in children after hematopoietic stem cell transplantation. Expert Rev Hematol. 2012;5:291–302. doi: 10.1586/ehm.12.18. [DOI] [PubMed] [Google Scholar]
- 41.Dignan FL, Wynn RF, Hadzic N, Karani J, Quaglia A, Pagliuca A, et al. BCSH/BSBMT guideline: diagnosis and management of veno-occlusive disease (sinusoidal obstruction syndrome) following haematopoietic stem cell transplantation. Br J Haematol. 2013;163:444–57. doi: 10.1111/bjh.12558. [DOI] [PubMed] [Google Scholar]
- 42.Ho VT, Revta C, Richardson PG. Hepatic veno-occlusive disease after hematopoietic stem cell transplantation: update on defibrotide and other current investigational therapies. Bone Marrow Transpl. 2008;41:229–37. doi: 10.1038/sj.bmt.1705899. [DOI] [PubMed] [Google Scholar]
- 43.Cao Y, Colegate SM, Edgar JA. Safety assessment of food and herbal products containing hepatotoxic pyrrolizidine alkaloids: interlaboratory consistency and the importance of N-oxide determination. Phytochem Anal. 2008;19:526–33. doi: 10.1002/pca.1079. [DOI] [PubMed] [Google Scholar]
- 44.Molyneux RJ, Gardner DL, Colegate SM, Edgar JA. Pyrrolizidine alkaloid toxicity in livestock: a paradigm for human poisoning? Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2011;28:293–307. doi: 10.1080/19440049.2010.547519. [DOI] [PubMed] [Google Scholar]
- 45.Williams MT, Warnock BJ, Betz JM, Beck JJ, Gardner DR, Lee ST, et al. Detection of high levels of pyrrolizidine-N-oxides in the endangered plant Cryptantha crassipes (Terlingua Creek cat’s-eye) using HPLC-ESI-MS. Phytochem Anal. 2011;22:532–40. doi: 10.1002/pca.1314. [DOI] [PubMed] [Google Scholar]
- 46.Edgar JA, Roeder E, Molyneux RJ. Honey from plants containing pyrrolizidine alkaloids: a potential threat to health. J Agric Food Chem. 2002;50:2719–30. doi: 10.1021/jf0114482. [DOI] [PubMed] [Google Scholar]
- 47.Fu PP, Xia QS, Chou MW, Lin G. Detection, hepatotoxicity, and tumorigenicity of pyrrolizidine alkaloids in Chinese herbal plants and herbal dietary supplements. J Food Drug Anal. 2007;15:400–15. [Google Scholar]
- 48.Stegelmeier BL, Edgar JA, Colegate SM, Gardner DR, Schoch TK, Coulombe RA, et al. Pyrrolizidine alkaloid plants, metabolism and toxicity. J Nat Toxins. 1999;8:95–116. [PubMed] [Google Scholar]
- 49.Yang MB, Ma J, Ruan JQ, Ye Y, Fu PP, Lin G. Intestinal and hepatic biotransformation of pyrrolizidine alkaloid N-oxides to toxic pyrrolizidine alkaloids. Arch Toxicol. 2019;93:2197–209. doi: 10.1007/s00204-019-02499-2. [DOI] [PubMed] [Google Scholar]
- 50.Lu Y, Ma J, Song ZJ, Ye Y, Fu PP, Lin G. The role of formation of pyrrole-ATP synthase subunit beta adduct in pyrrolizidine alkaloid-induced hepatotoxicity. Arch Toxicol. 2018;92:3403–14. doi: 10.1007/s00204-018-2309-6. [DOI] [PubMed] [Google Scholar]
- 51.Yang MB, Ma J, Ruan JQ, Zhang CY, Ye Y, Fu PP, et al. Absorption difference between hepatotoxic pyrrolizidine alkaloids and their N-oxides-mechanism and its potential toxic impact. J Ethnopharmacol. 2020;249:112421. doi: 10.1016/j.jep.2019.112421. [DOI] [PubMed] [Google Scholar]
- 52.Gao H, Ruan JQ, Chen J, Li N, Ke CQ, Ye Y, et al. Blood pyrrole-protein adducts as a diagnostic and prognostic index in pyrrolizidine alkaloid-hepatic sinusoidal obstruction syndrome. Drug Des Devel Ther. 2015;9:4861–8. doi: 10.2147/DDDT.S87858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Gao H, Li N, Wang JY, Zhang SC, Lin G. Definitive diagnosis of hepatic sinusoidal obstruction syndrome induced by pyrrolizidine alkaloids. J Dig Dis. 2012;13:33–39. doi: 10.1111/j.1751-2980.2011.00552.x. [DOI] [PubMed] [Google Scholar]
- 54.European Association for the Study of the Liver (EASL), Clinical Practice Guideline Panel. EASL clinical practice guidelines: drug-induced liver injury. J Hepatol. 2019;70:1222–61. doi: 10.1016/j.jhep.2019.02.014. [DOI] [PubMed] [Google Scholar]
- 55.Ou PC, Chen Y, Li BZ, Zhang M, Liu XY, Li FF, et al. Causes, clinical features and outcomes of drug-induced liver injury in hospitalized patients in a Chinese tertiary care hospital. Springerplus. 2015;4:1–8. doi: 10.1186/s40064-015-1600-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yu YC, Mao YM, Chen CW, Chen JJ, Chen J, Cong WM, et al. CSH guidelines for the diagnosis and treatment of drug-induced liver injury. Hepatol Int. 2017;11:221–41. doi: 10.1007/s12072-017-9793-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Drucker E, Krapfenbauer K. Pitfalls and limitations in translation from biomarker discovery to clinical utility in predictive and personalised medicine. EPMA J. 2013;4:1–10. doi: 10.1186/1878-5085-4-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Meunier L, Larrey D. Drug-induced liver injury: bomarkers, requirements, candidates, and validation. Front Pharmacol. 2019;10:1482. doi: 10.3389/fphar.2019.01482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang C, Jiang M, Zhang G, Bian ZX, Lu AP. Progress and perspectives of biomarker discovery in Chinese medicine research. Chin J Integr Med. 2014;9:1–9. doi: 10.1007/s11655-014-1848-9. [DOI] [PubMed] [Google Scholar]
- 60.Ma J, Xia QS, Fu PP, Lin G. Pyrrole-protein adducts–A biomarker of pyrrolizidine alkaloid-induced hepatotoxicity. J Food Drug Anal. 2018;26:965–72. doi: 10.1016/j.jfda.2018.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.National Center for Environmental Health, 2012. https://www.cdc.gov/nceh/stories/ethiopia.html
- 62.Cong WH, Xin QQ, Gao YQ. RE: Incidence and etiology of drug-induced liver injury in mainland China. Gastroenterology. 2019;157:1438–9. doi: 10.1053/j.gastro.2019.05.076. [DOI] [PubMed] [Google Scholar]
- 63.Tang J, Hattori M. Pyrrolizidine alkaloids-containing Chinese medicines in the Chinese Pharmacopoeia and related safety concerns. Yao Xue Xue Bao. 2011;46:762–72. [PubMed] [Google Scholar]
- 64.Fu PP, Yang YC, Xia QS, Chou MW, Cui YY, Lin G. Pyrrolizidine alkaloids-tumorigenic components in Chinese herbal medicines and dietary supplements. J Food Drug Anal. 2002;10:198–211. [Google Scholar]
- 65.Chinese Pharmacopoeia Commission. Pharmacopoeia of the People’s Republic of China. Beijing: Chinese Medical Science and Technology Press; Volume 1, 2015.