Abstract
Aim
To examine the association of psychological distress with all-cause, cardiovascular disease (CVD) and cancer mortality in US adults, and verified whether the associations differed between participants with and without diabetes.
Methods
A total of 485,864 adults (446,288 without diabetes and 39,576 with diabetes) who participated in the National Health Interview Survey from 1997 to 2013 were linked to the National Death Index through December 31, 2015. Psychological distress was measured by the Kessler 6 distress scale (K6). Multivariable Cox proportional hazards regression models were performed to estimate hazard ratios (HR) and 95% confidence intervals (95% CI) for the association between psychological distress and mortality.
Results
We ascertained 11,746 deaths (mean follow-up, 7. 7 years) among people with diabetes and 51,636 deaths (9.9 years) among those without diabetes. Psychological distress was associated with higher all-cause, CVD, and cancer mortality. Compared to non-diabetic adults without psychological distress, HRs (95% CI) were 1.07 (1.04 to 1.09) for mild, 1.26 (1.22 to 1.30) for moderate and 1.46 (1.38 to 1.55) for severe psychological distress. Compared to the same reference group, in diabetic participants the HRs were 1.39 (1.33 to 1.44) for no psychological distress, 1.59 (1.53 to 1.66) for mild, 1.90 (1.80 to 2.00) for moderate and 1.98 (1.82 to 2.17) for severe psychological distress. Similar associations were also observed for CVD and cancer mortality but with non-statistically significant interaction.
Conclusion
Psychological distress was associated with higher mortality, particularly in participants with diabetes. Strategies to ameliorate psychological distress may be important to reduce mortality in this population.
Keywords: diabetes mellitus, psychological distress, mortality, cohort
Introduction
The prevalence of diabetes has increased substantially over the last decades.1 In 2019, about 463 million adults had a diagnosis of diabetes, and this number is projected to increase to 578 million by 2030. Patients with diabetes are at increased risk of cardiovascular disease (CVD),2 cancer3,4 and all-cause mortality.3 Diabetes and its complications seriously affect patient’s quality of life and life expectancy.5
Due to ongoing self-management and adverse experiences of drug side effects or complications, diabetic people are at higher risk of psychological distress.6 Psychological distress is an emotional response towards adverse or unpleasant stressors,7 which is linked to an increase of all-cause and cause-specific mortality.8–12 Women with diabetes have a higher risk of psychological problems than men, particularly in those taking non-insulin glucose-lowering drugs for treatment.13 However, other studies14,15 have reported an association between psychological distress and increased all-cause mortality in men, but not in women, and participants’ age ≥65 years.16 Although studies have shown an association between psychological distress and increased mortality, it is unclear whether the association differs in participants with and without diabetes. A few studies indicated that the coexistence of diabetes and psychological distress or depression were significantly associated with greater mortality,17,18 which may indicate a potential interaction of diabetes and psychological distress on mortality risk.
In this study, we examined the association of psychological distress with all-cause, CVD and cancer mortality in a large prospective cohort of US adults; and verified whether these associations differed between participants with and without diabetes. Additionally, we specifically sought to examine these associations across age, sex, and medication status.
Methods
Participants
We used data from the National Health Interview Survey (NHIS), a cross-sectional annual health survey representative of non-institutionalized individuals in the US conducted by the National Center for Health Statistics (NCHS). NHIS uses a multistage probability sampling to select approximately 35,000 households from randomly selected clusters. For each participating household, one adult aged 18 years or older was randomly selected for a detailed interview on health status, health behavior, lifestyle risk factors and other health information. About 750 interviewers from the US Census Bureau who are well trained by health survey supervisors in the US Census Bureau Regional Offices are responsible for the interviews and data collection for NHIS. Details of the survey design and documentation are available on the NCHS website.19 All participants provided informed consent before participating in the survey. Institutional Review Board approval is considered as exempt under the ethical board review of the corresponding author’s institution because this study was based on secondary analyses of publicly available,20 de-identified data that do not include any traceable personal information.
A total of 493,365 adults who participated in the NHIS survey from 1997 to 2013 linked to the National Death Index through 31 December 2015 were eligible for the final analytical sample. Participants were included only if they had responded to all items of the K6 questionnaire and did not have missing data on psychological distress and diagnosis status of diabetes. Of these participants, 485,864 were enrolled in the final analysis after excluding missing data about psychological distress (n=7263, 1.5% of total participants) and diagnosis of diabetes at baseline (n=238, 0.1% of total participants).
Mortality Ascertainment
The mortality outcomes were ascertained by linkage to the National Death Index (NDI) records.21 Participants not matched with the NDI record were considered alive during the follow-up period. The coding of underlying causes of death before 1999 was classified according to the International Classification of Diseases (ICD-9) guidelines and from 1999 onwards according to the ICD-10 guidelines. Mortality outcomes were defined as follows: 1) all-cause mortality; 2) CVD mortality (codes I00 to I09, I11, I13, and I20 to I51, I60 to I69); 3) cancer mortality (codes C00 to C97).
Assessment of Psychological Distress
Psychological distress was measured by the Kessler 6 scale (K6).22 Respondents were asked about the frequency of experiencing the following six symptoms in the past 30 days: depression, nervousness, restlessness or fidgety, hopelessness, felt that everything was an effort, and worthlessness, rated on a 5-point Likert scale (0=none of the time, 1=a little of the time, 2=some of the time, 3=most of the time and 4=all the time). Each of the six items was summed to yield a total K6 score between 0 and 24. The validity of the K6 scale has been previously verified, and the cut-off point of ≥5 and ≥13 was suggested and widely used to categorize participants with a likelihood of moderate and severe (serious) psychological distress.23–26 In this study, we classified psychological distress status into four categories: 1) no psychological distress: K6=0 point; 2) mild psychological distress: 0< K6 <5 points; 3) moderate psychological distress: 5≤ K6 <13 points; 4) severe psychological distress: K6 ≥13 points.
Covariates
Covariates included as potential confounders were age, sex, race (Hispanic, non-Hispanic White, non-Hispanic black, and others), education level (less than high school degree, high school degree, and more than high school degree), household income, body mass index (BMI: normal weight or underweight: <25 kg/m2, pre-obese: 25 to ≤30 kg/m2, obese: >30 kg/m2), physical activity (0, 1–149 min/week, ≥150 min/week, according to physical activity guidelines27), smoking status (non-smoker, former smoker, current smoker), alcohol intake status (lifetime abstainer, former drinker, light drinker, moderate drinker, heavy drinker), self-reported diagnosis of hypertension, CVD and cancer. We also collected the medication treatment status among participants with diabetes and stratified into two groups: no medication treatment, with glucose-lowering medication treatment (oral glucose-lowering drugs and/or insulin injection).
Statistical Analysis
Descriptive statistics were used to report the distribution of participants’ baseline characteristics by psychological distress. Categorical variables were displayed as frequency and percentage (%). The mortality rates (per 10,000 person-years) were age standardized for age groups of 18 to 44 years, 45 to 64 years, and 65 years or older and computed using the direct method and the overall NHIS sample from 1997 to 2013 (n = 493,365) as the standard population. Multivariable Cox proportional hazards regression models were performed to estimate hazard ratios (HRs) and 95% confidence intervals (95% CI) for the association between psychological distress and mortality. Two models were fitted: Model 1 included age and sex; Model 2: model 1 plus race, education level, household income, BMI, physical activity, smoking, alcohol drinking, diagnosis of hypertension, CVD, stroke and cancer at baseline. We analyzed the associations of psychological distress status with mortality in participants with and without diabetes and further stratified by age, sex and medication treatment status. We also assessed the joint association of psychological distress (a) and presence of diabetes (b) on mortality by generating eight subgroups of participants: non-diabetes with no (reference group, HR0), mild (HRa1), moderate (HRa2) and severe (HRa3) psychological distress, respectively; and diabetes with no (HRb), mild (HRa1b), moderate (HRa2b) and severe (HRa3b) psychological distress, respectively. The relative excess risk due to interaction (RERI) of psychological distress and diabetes on mortality were calculated.28 A RERI >0 and the lower limit of 95% CI > 0 suggests a synergistic (biological) interaction.29,30 Generally, the effect of baseline exposure on mortality lags for several years. Therefore, we conducted sensitivity analysis by excluding participants with <2, <5, and <10 yrs of follow-up times to examine the influence of reverse causation/confounding due to pre-existing diseases in our main findings. Also, we excluded participants with a history of CVD and cancer at baseline to control for confounding due to pre-existing disease.31 All statistical analyses were performed with STATA version 15.0 (Stata Corp, College Station, TX, USA), a two-sided P value <0.05 was considered statistically significant.
Results
Participant Characteristics
A total of 51,636 deaths were ascertained during a mean follow-up of 9.9 years among 446,288 participants without diabetes. A total of 11,746 deaths occurred during a mean follow-up of 7.7 years among 39,576 participants with diabetes. Baseline characteristics of participants according to psychological distress status displayed in Table 1. Among participants without diabetes, 69,604 (15.6%) and 14,598 (3.3%) adults had moderate (5≤ K6 scale <13) to severe (K6 ≥13 points) psychological stress, whereas the numbers were 8575 (21.7%) and 2761 (7.0%) among participants with diabetes. Participants with moderate or severe psychological distress were more likely to be women, age <65 years, Hispanic, less than high school degree, with low or middle household income, obese, lacking of physical activity, current smoker, former alcohol drinker and heavy drinker, and have a history of hypertension, CVD and cancer at baseline (Table 1).
Table 1.
Baseline Characteristics of the Study Population According to Psychological Distress Status
| Baseline Characteristics | Psychological Distress Status | |||
|---|---|---|---|---|
| No | Mild | Moderate | Severe | |
| Sample size, n(%) | 16,345 (42.3) | 15,192 (38.6) | 5278 (12.7) | 2761 (6.5) |
| Mean age, (years) | 60.95 | 59.27 | 57.81 | 55.97 |
| Age (years) | ||||
| 18–64 | 8412 (56.7) | 6740 (61.6) | 5378 (65.9) | 1991 (75.5) |
| ≥65 | 7933 (43.3) | 5155 (38.4) | 3197 (34.1) | 770 (24.5) |
| Sex, n(%) | ||||
| Male | 8393 (55.9) | 4988 (47) | 3153 (41.2) | 980 (40.5) |
| Female | 7952 (44.1) | 6907 (53.1) | 5422 (58.9) | 1781 (59.5) |
| Race, n(%) | ||||
| Hispanic | 2701 (12.5) | 1935 (12.3) | 1688 (14.2) | 731 (18.1) |
| Non-Hispanic White | 9111 (64.7) | 7178 (68.8) | 4695 (65.6) | 1441 (64.4) |
| Non-Hispanic Black | 3664 (17.2) | 2292 (14.5) | 1836 (16) | 489 (13.5) |
| Non-Hispanic Other | 869 (5.6) | 490 (4.4) | 356 (4.2) | 100 (3.9) |
| Education level, n(%) | ||||
| Less than high school degree | 4302 (22.5) | 3153 (22.7) | 3101 (32) | 1187 (38.8) |
| High school degree | 4851 (31.1) | 3586 (30.9) | 2564 (32) | 804 (31.6) |
| More than high school degree | 7065 (45.7) | 5097 (46) | 2846 (35.2) | 751 (29) |
| Income, n(%) | ||||
| Low | 2573 (11.3) | 2162 (13) | 2441 (21.8) | 1144 (34.3) |
| Middle | 9093 (54.2) | 6721 (55.8) | 4915 (60) | 1406 (55.7) |
| High | 4679 (34.5) | 3012 (31.2) | 1219 (18.2) | 211 (10) |
| BMI (kg/m2), n(%) | ||||
| Normal weight/Underweight, <25 kg/m2 | 3037 (17.8) | 2087 (16.9) | 1391 (15.6) | 438 (14.3) |
| Pre-obesity, 25–29.9 kg/m2 | 5569 (33.7) | 3589 (30) | 2422 (28) | 706 (25.7) |
| Obesity, ≥30 kg/m2 | 7157 (44.8) | 5806 (49.6) | 4443 (52.7) | 1527 (57) |
| Physical activity, n(%) | ||||
| 0 min/week | 8229 (47.9) | 5537 (43) | 5081 (57.7) | 1921 (68.3) |
| 1–149 min/week | 2916 (18.1) | 2519 (22.1) | 1580 (18.8) | 407 (15.6) |
| ≥150 min/week | 4803 (31.3) | 3532 (32) | 1740 (21.1) | 393 (14.5) |
| Smoking status, n(%) | ||||
| Non-smoker | 8542 (51.3) | 5959 (49.2) | 3967 (44.8) | 1133 (38.9) |
| Former smoker | 5579 (35.2) | 4031 (34.9) | 2759 (32.9) | 815 (31.6) |
| Current smoker | 2137 (13) | 1838 (15.4) | 1800 (21.7) | 802 (29) |
| Alcohol intake status, n(%) | ||||
| Lifetime abstainer | 5289 (29.8) | 3151 (23.9) | 2499 (27.8) | 829 (27.8) |
| Former drinker | 4597 (27.3) | 3615 (29.4) | 2822 (32.7) | 964 (36) |
| Light drinker | 4670 (31.2) | 3953 (36.4) | 2504 (30.9) | 938 (27.4) |
| Moderate drinker | 1179 (7.8) | 756 (6.8) | 425 (5.5) | 114 (4.2) |
| Heavy drinker | 298 (2.0) | 269 (2.3) | 191 (2.1) | 84 (2.8) |
| Hypertension, n(%) | 10,757 (64.3) | 8223 (67.5) | 6296 (72.2) | 2131 (76.3) |
| CVD, n(%) | 4910 (29.7) | 4077 (33.5) | 3778 (44.1) | 1405 (50.2) |
| Cancer, n(%) | 2181 (13.3) | 1295 (15.5) | 410 (14.5) | 410 (14.5) |
Note: Values are n (percentage).
Abbreviations: BMI, body mass index; CVD, cardiovascular disease.
Psychological Distress and Mortality Among Adults with and without Diabetes
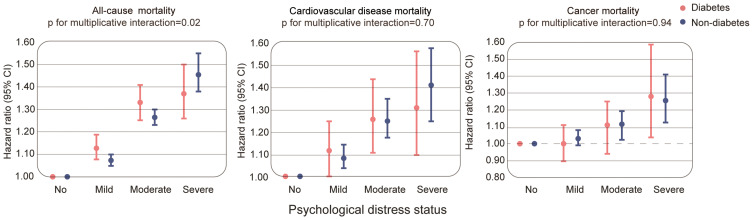
In the fully adjusted model, we observed a stronger association between severe psychological distress and all-cause mortality in participants without diabetes compared to those with diabetes. Among participants with diabetes, the HRs (95% CI) for all-cause mortality were 1.12 (1.07 to 1.18) for mild, 1.33 (1.25 to 1.41) for moderate and 1.37 (1.26 to 1.50) for severe vs no psychological distress. For participants without diabetes, the respective HRs (95% CI) for all-cause mortality were 1.07 (1.05 to 1.10), 1.26 (1.23 to 1.30) and 1.46 (1.38 to 1.55). There was a significant multiplicative interaction between psychological distress and the presence of diabetes (pinteraction=0.02). We found no evidence of multiplicative interaction between psychological distress and diabetes for CVD (pinteraction= 0.70) and cancer mortality (pinteraction = 0.94) (Figure 1, Sup Table 1).
Figure 1.
Psychological distress and the risk of mortality according to the presence of diabetes. The HRs (95% CI) are based on Cox regression models adjusted for age, sex, race/ethnicity, education, income, body mass index, smoking, alcohol intake, physical activity, history of CVD and cancer. The psychological distress score was measured by the Kessler 6 nonspecific distress scale.
Joint Association Between Psychological Distress and Diabetes with Mortality
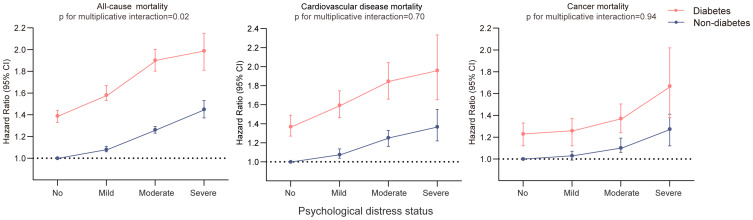
The mortality rates were higher in participants with diabetes and higher psychological distress. The absolute rate of all-cause mortality from no to severe psychological distress was 94.5 to 195.8 deaths per 10,000 person-years for participants without diabetes and 173.8 to 319.7 deaths per 10,000 person-years for participants with diabetes (Table 2).
Table 2.
Joint Association of Diabetes and Psychological Distress Status with All-Cause, Cardiovascular Disease and Cancer Mortality
| Psychological Distress Status | No. of Participants | No. of Deaths | Age-Standardized Mortality Rate per 10,000 (95% CI) | Model 1 | Model 2 | p for Multiplicative Interaction† |
|---|---|---|---|---|---|---|
| HR (95% CI) | HR (95% CI) | |||||
| All-cause mortality | 0.02 | |||||
| Non-diabetes | ||||||
| No | 216,538 | 24,576 | 94.5 (94.4,94.5) | 1.00 (Reference) | 1.00 (Reference) | |
| Mild | 145,548 | 15,632 | 105.3 (105.3,105.3) | 1.11 (1.08,1.13) | 1.07 (1.04,1.09) | |
| Moderate | 69,604 | 9041 | 154.1 (154.1,154.2) | 1.61 (1.56,1.65) | 1.26 (1.22,1.30) | |
| Severe | 14,598 | 2387 | 195.8 (195.7,195.9) | 2.32 (2.19,2.46) | 1.46 (1.38,1.55) | |
| Diabetes | ||||||
| No | 16,345 | 4581 | 173.8 (173.7,173.9) | 1.59 (1.53,1.65) | 1.39 (1.33,1.44) | |
| Mild | 11,895 | 3447 | 209.3 (209.2,209.4) | 1.90 (1.83,1.98) | 1.59 (1.53,1.66) | |
| Moderate | 8575 | 2808 | 269.0 (268.8,269.2) | 2.72 (2.59,2.87) | 1.90 (1.80,2.00) | |
| Severe | 2761 | 910 | 319.7 (319.3,320.0) | 3.30 (3.02,3.61) | 1.98 (1.82,2.17) | |
| CVD mortality | 0.70 | |||||
| Non-diabetes | ||||||
| No | 216,538 | 5410 | 19.7 (19.7,19.7) | 1.00 (Reference) | 1.00 (Reference) | |
| Mild | 145,548 | 3425 | 23.2 (23.1,23.2) | 1.15 (1.09,1.21) | 1.09 (1.03,1.15) | |
| Moderate | 69,604 | 1975 | 31.3 (31.2,31.3) | 1.67 (1.57,1.79) | 1.25 (1.17,1.34) | |
| Severe | 14,598 | 485 | 41.4 (41.4,41.5) | 2.34 (2.09,2.62) | 1.39 (1.24,1.56) | |
| Diabetes | ||||||
| No | 16,345 | 1157 | 40.3 (40.2,40.3) | 1.72 (1.60,1.86) | 1.38 (1.27,1.49) | |
| Mild | 11,895 | 872 | 46.9 (46.8,46.9) | 2.11 (1.93,2.30) | 1.60 (1.46,1.75) | |
| Moderate | 8575 | 710 | 54.1 (54.1,54.2) | 3.04 (2.74,3.38) | 1.84 (1.65,2.04) | |
| Severe | 2761 | 226 | 73.4 (73.2,73.5) | 3.86 (3.25,4.58) | 1.96 (1.66,2.33) | |
| Cancer mortality | 0.94 | |||||
| Non-diabetes | ||||||
| No | 216,538 | 6339 | 25.8 (25.8,25.8) | 1.00 (Reference) | 1.00 (Reference) | |
| Mild | 145,548 | 3976 | 27.2 (27.2,27.2) | 1.07 (1.02,1.11) | 1.02 (0.98,1.07) | |
| Moderate | 69,604 | 2168 | 36.0 (35.9,36.0) | 1.39 (1.31,1.48) | 1.12 (1.05,1.19) | |
| Severe | 14,598 | 546 | 42.9 (42.9,43.0) | 1.91 (1.71,2.13) | 1.26 (1.13,1.41) | |
| Diabetes | ||||||
| No | 16,345 | 935 | 36.8 (36.8,36.9) | 1.34 (1.23,1.45) | 1.22 (1.13,1.33) | |
| Mild | 11,895 | 643 | 40.5 (40.5,40.6) | 1.41 (1.29,1.54) | 1.25 (1.13,1.37) | |
| Moderate | 8575 | 462 | 46.0 (46.0,46.1) | 1.76 (1.56,1.98) | 1.38 (1.22,1.55) | |
| Severe | 2761 | 164 | 59.6 (59.5,59.8) | 2.35 (1.93,2.86) | 1.66 (1.37,2.02) |
Notes: Model 1: adjusted for age and sex at baseline. Model 2: Model 1 plus additionally adjusted for race/ethnicity, education, income, body mass index, smoking, alcohol intake, physical activity, hypertension, CVD and cancer at baseline. †The p for multiplicative interaction between psychological distress and presence of diabetes were obtained using Wald testing.
Abbreviations: CI, confidence interval; HR, hazard ratio.
Compared to non-diabetic adults without psychological distress, HRs (95% CI) were 1.07 (1.04 to 1.09) for mild, 1.26 (1.22 to 1.30) for moderate and 1.46 (1.38 to 1.55) for severe psychological distress in non-diabetic participants. Compared to the same reference group, in diabetic participants the HRs were 1.39 (1.33 to 1.44) for no psychological distress, 1.59 (1.53 to 1.66) for mild, 1.90 (1.80 to 2.00) for moderate and 1.98 (1.82 to 2.17) for severe psychological distress. For CVD and cancer mortality, we found higher magnitude of associations with psychological distress in participants with diabetes versus without diabetes (Table 2, Figure 2). There was significant interaction between psychological distress (mild/moderate vs no) and presence of diabetes, but did not reach a statistically significant threshold for CVD and cancer mortality (Sup Table 2).
Figure 2.
Joint association of diabetes and psychological distress with all-cause mortality. The HRs (95% CI) are based on Cox regression models adjusted for age, sex, race/ethnicity, education, income, body mass index, smoking, alcohol intake, physical activity, history of CVD and cancer. The psychological distress score was measured by the Kessler 6 nonspecific distress scale. The P for interaction between the presence of diabetes and psychological distress on mortality were obtained using Wald testing.
Subgroup Analysis Among Adults with Diabetes
There were no significant differences in associations of psychological distress with all-cause, CVD and cancer mortality across age subgroup (all pinteraction >0.05). However, the associations were stronger in men (HRs [95%] for mild, moderate and severe psychological distress, respectively, were: 1.16 [1.08 to 1.25]; 1.41 [1.29 to 1.53]; 1.62 [1.41 to 1.87]) than in women (HRs [95%] were 1.07 [0.99 to 1.15], 1.25 [1.15 to 1.35], 1.18 [1.04 to 1.34], respectively; p for multiplicative interaction =0.004) (Table 3). Similar patterns were observed for CVD mortality (pinteraction =0.02) (Sup Tables 3, 4).
Table 3.
Subgroup Analysis of the Association of Psychological Distress Status with All-Cause Mortality Among Participants with Diabetes
| Subgroup | Psychological Distress Status | p for Multiplicative Interaction | |||
|---|---|---|---|---|---|
| No | Mild | Moderate | Severe | ||
| Age | 0.63 | ||||
| <65 | 1 (Reference) | 1.14 (1.02,1.26) | 1.32 (1.19,1.46) | 1.34 (1.18,1.52) | |
| ≥65 | 1 (Reference) | 1.10 (1.04,1.17) | 1.32 (1.22,1.42) | 1.35 (1.17,1.55) | |
| Sex | 0.004 | ||||
| Female | 1 (Reference) | 1.07 (0.99,1.15) | 1.25 (1.15,1.35) | 1.18 (1.04,1.34) | |
| Male | 1 (Reference) | 1.16 (1.08,1.25) | 1.41 (1.29,1.53) | 1.62 (1.41,1.87) | |
| Medication treatment status | 0.02 | ||||
| No | 1 (Reference) | 1.00 (0.86,1.16) | 1.23 (1.03,1.46) | 1.02 (0.75,1.39) | |
| Yes | 1 (Reference) | 1.14 (1.07,1.21) | 1.34 (1.25,1.44) | 1.48 (1.32,1.65) | |
Notes: Values are HR (95% CI). Adjusted for age, sex race/ethnicity, education, income, body mass index, smoking, alcohol intake, physical activity, hypertension, CVD and cancer at baseline. The p for interaction between psychological distress and age/sex/medication treatment status were obtained using Wald testing.
Abbreviations: CI, confidence interval; CVD, cardiovascular disease; HR, hazard ratio.
We further conducted subgroup analyses stratified by medication treatment status among participants with diabetes. In adults who were treated with glucose-lowering medication, greater HRs were observed for the associations of psychological distress with all-cause, CVD and cancer mortality(Table 3, Sup Tables 3, 4). For those not treated with glucose-lowering medication, significant higher HR for all-cause mortality was observed only in those with moderate psychological distress (HR=1.23, 95% CI= [1.03 to 1.46]). The interaction between psychological distress and medication treatment status was only significant for all-cause mortality (pinteraction=0.02) (Table 3).
Sensitivity Analysis
We performed sensitivity analyses excluding participants with <2, <5, and <10 years of follow-up and with a history of CVD and cancer at baseline and observed minor changes in the magnitude of the associations of psychological distress with all-cause, CVD and cancer mortality (Sup table 5, 6).
Discussion
This large prospective study of US adults showed that psychological distress was associated with increased risk of all-cause and cause-specific mortality among adults with and without diabetes. The joint association of psychological distress and diabetes on mortality suggested that the presence of diabetes may lead to a higher mortality risk among adults with psychological distress. Given the major burden of diabetes and psychological distress in the US population, such disparities may have a major effect on public health.
The higher HR for severe psychological distress on mortality observed in the non-diabetic participants versus diabetic participants may be attributed to lower mortality in the non-diabetes population. A possible explanation is that diabetic adults may attain more medical attention and could be beneficial consequences in terms of enhancing multidisciplinary care access;32,33 this diabetes intervention may restrict the disparity in mortality risk compared to the general population. Although disparities in relative risk for all-cause mortality are weaker in participants with versus without diabetes, the absolute mortality risk for diabetic adults with psychological distress is greater. The all-cause mortality risk further increased due to the synergistic effect of psychological distress and diabetes on mortality, which could contribute to a major mortality burden.
It is well known that individuals with diabetes have higher mortality than the general population.33 Diabetes that coexists with severe psychological distress may suppress the patient’s self-management,34,35 and has been associated with worse glycemic control,36,37 and elevated risk of CVD and death,14,18,38 the coexistence of diabetes and psychological distress may interact and further increase mortality. However, to our knowledge, few studies have focused on the synergistic association between psychological distress and diagnosis of diabetes on mortality outcomes. A previous study18 indicated an interaction between diabetes and psychological distress with regard to mortality: adults with diabetes and psychological distress had higher mortality than those without diabetes, but with psychological distress. This synergistic effect on mortality among people with diabetes can be explained by the potential effect of psychological distress on poor glycemic control and higher risk of diabetic complications.39,40 A study conducted among Chinese Type 2 diabetes patients indicated that diabetes distress (measured by the 15-item Chinese version of Diabetes Distress Scale, CDDS-15) was associated with poor glycemic control, obesity, depressive symptoms, and quality of life;39 however, this study included a small sample size. Another study assessing diabetes distress and clinical depression among type 2 diabetes patients found that poor glycemic control contributed to diabetes distress but not depression.37 A prospective study including 985 patients who initiated insulin therapy found that diabetes-related distress was associated with higher levels of HbA1c and risk of microvascular complications.40 The increased mortality among adults with diabetes and psychological distress may be mediated by poor sleep. Sleep disturbance is a prominent symptom in patients with psychological disorder41 and coexisting sleep disturbances and severe psychological distress have a high prevalence.42 Persons with poor sleep quality have been found to have a 40% lower glucose clearance rate, higher sympathetic activation,43 and poorer self-care behaviors.44 In addition, diabetic people with sleep disturbances have an increased all-cause and cause-specific mortality burden.45
Among participants with diabetes, we found an association between levels of psychological distress and increased risk of CVD mortality. This finding is consistent with a previous study conducted in diabetic patients, indicating that patients with psychological distress (measured by Mental Health Inventory 5) were at increased risk of all-cause mortality and CVD events.15 Another study found a 1.76-fold higher risk of mortality and a 1.69-fold higher incidence of CVD events with psychological distress in participants with vs those without diabetes.38 Psychological distress is already acknowledged as an important risk factor for CVD events. Adverse psychological factors are associated with the increased sympathetic nervous system, immune dysregulation, altered vascular reactivity, heightened coagulation and chronic downstream endothelial dysfunction, and heightened inflammation, which may ultimately increase the risk of fatal and non-fatal CVD events.46 In our study, we also found that participants with diabetes and severe psychological distress had a greater risk of mortality from cancer, which corroborate with previous studies.8,11 Psychosocial factors such as depression have been associated immune dysregulation which compromised the repair capacity of the cells and created a chronic inflammatory environment. This chronic inflammatory response and immune dysregulation can compromise the repair capacity of the exposed cells, potentially modulate fundamental processes in cancer growth, invasion, and metastasis,47,48 and further contributed to greater risk of cancer mortality.
Moreover, our study revealed that men with psychological distress had greater HRs for CVD, cancer and all-cause mortality than women. However, our results are inconsistent with one recent Japanese study of type 2 diabetes patients.14 This study used the Japanese version of Problem Areas In Diabetes (PAID) questionnaire to access diabetes-related emotional distress of participants and revealed that participants with a high level of diabetes-related distress (PAID score ≥40) had a 1.56-fold (95% CI 1.17 to 2.08) and 1.76-fold (95% CI 1.26 to 2.46) increased risk of all-cause mortality in the total population and men, respectively, but this association was not observed in women (HR=1.09, 95% CI= 0.60 to 2.00, P>0.05). On the other hand, a recent cohort study assessed the psychological distress among 662 individuals with coronary artery disease and only found an association between psychological distress and increased risk of CVD events in women.49 However, this study was limited by a relatively small sample size and short follow-up time (from 2011 to 2014). Age-stratified subgroup analysis in our study demonstrated a similar association of psychological distress on mortality in participants with diabetes when analyses were stratified by age <65 and ≥65 years. These findings are inconsistent with a study conducted among the Japanese type 2 diabetes population that found no association between diabetes distress and mortality among participants’ age ≥65 years.14 Whether the association between psychological distress and mortality differ by age and sex is still inconclusive, and further studies are needed to clarify this issue.
The present study revealed a greater association of psychological distress with increased risk of all-cause, CVD and cancer mortality in participants using glucose-lowering medication. For diabetic adults without medication use, a statistically significantly increased all-cause mortality was observed only in moderate psychological distress. Some evidence showed that glucose-lowering agents add no risk for psychological issues and may even have a protective role in depression;50,51 however, few studies revealed that diabetes patients treated with insulin therapy were at increased risk of depressive symptoms52 and diabetes distress.53 These insulin-treated patients may negatively affect insulin therapy and be compounded by a sense of guilt or failure for the additional medication treatment.54 Nevertheless, patients with diabetes using glucose-lowering medication may have poorer glucose metabolism, glycemic control and worse health status than those not using these medications, which contributed to a higher incidence of mortality.
Our study has some strengths, including the relatively large sample size of US adults and long length of follow-up. Thus, we were able to conduct subgroup analyses and to test for interactions by age, sex, and medication treatment status, and we conducted sensitivity analyses to examine the robustness of our main results. However, our study also has several limitations. First, although we controlled for several sociodemographic, lifestyle and health status variables, residual confounding cannot be ruled out. Second, the data on diabetes diagnoses and medication use were self-reported, which may underestimate the total number of adults with diabetes; however, studies55,56 have shown acceptable or even good validity for risk assessment in cohort studies. Third, NHIS assessed psychological distress by self-reported non-specific questionnaire. Although K6 has been validated and there is support for its use in a preliminary screening of mental distress,23,57 it still cannot serve as diagnostic criteria of clinical psychological distress.
In conclusion, we found a dose-response relationship between psychological distress and increased risk of all-cause and cause-specific mortality in adults with and without diabetes. The joint analysis showed that diabetic participants coexisting with psychological distress have a greater mortality compared to those without a diagnosis of diabetes with psychological distress. The associations between psychological distress and all-cause and CVD mortality were stronger among diabetic men than in women, and also stronger among diabetic patients treated with medication, compared with those without medication treatment. If confirmed causal, our results underscore the significance of awareness, prevention and treatment of psychological distress in a population with diabetes as a potential strategy for reducing adverse health outcomes and mortality.
Acknowledgments
The authors thank the NCHS of the Centers for Disease Control and Prevention for sharing the NHIS data.
Funding Statement
There is no funding to report.
Data Sharing Statement
The NHIS data are available from www.cdc.gov/nchs/nhis/index.htm.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
None of the authors has a conflict of interest.
References
- 1.Zhou B, Lu Y, Hajifathalian K, Collaboration NCDRF. Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4·4 million participants. Lancet (London, England). 2016;387(10027):1513–1530. doi: 10.1016/S0140-6736(16)00618-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ramezankhani A, Azizi F, Hadaegh F, et al. Diabetes and number of years of life lost with and without cardiovascular disease: a multi-state homogeneous semi-Markov model. Acta Diabetol. 2018;55(3):253–262. doi: 10.1007/s00592-017-1083-x [DOI] [PubMed] [Google Scholar]
- 3.Campbell PT, Newton CC, Patel AV, et al. Diabetes and cause-specific mortality in a prospective cohort of one million U.S. adults. Diabetes Care. 2012;35(9):1835–1844. doi: 10.2337/dc12-0002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chen Y, Wu F, Saito E, et al. Association between type 2 diabetes and risk of cancer mortality: a pooled analysis of over 771,000 individuals in the Asia Cohort Consortium. Diabetologia. 2017;60(6):1022–1032. doi: 10.1007/s00125-017-4229-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.International Diabetes Federation. IDF Diabetes Atlas, 9th edn. Brussels, Belgium: 2019. Available at: https://www.diabetesatlas.org. Accessed July 1, 2021. [Google Scholar]
- 6.Perrin NE, Davies MJ, Robertson N, et al. The prevalence of diabetes-specific emotional distress in people with Type 2 diabetes: a systematic review and meta-analysis. Diabetic Med. 2017;34(11):1508–1520. doi: 10.1111/dme.13448 [DOI] [PubMed] [Google Scholar]
- 7.Snoek FJ, Bremmer MA, Hermanns N. Constructs of depression and distress in diabetes: time for an appraisal. Lancet Diabetes Endocrinol. 2015;3(6):450–460. doi: 10.1016/s2213-8587(15)00135-7 [DOI] [PubMed] [Google Scholar]
- 8.Russ TC, Stamatakis E, Hamer M, et al. Association between psychological distress and mortality: individual participant pooled analysis of 10 prospective cohort studies. BMJ. 2012;345:e4933. doi: 10.1136/bmj.e4933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ponizovsky AM, Haklai Z, Goldberger N. Association between psychological distress and mortality: the case of Israel. J Epidemiol Community Health. 2018;72(8):726–732. doi: 10.1136/jech-2017-210356 [DOI] [PubMed] [Google Scholar]
- 10.Wang YH, Li JQ, Shi JF, et al. Depression and anxiety in relation to cancer incidence and mortality: a systematic review and meta-analysis of cohort studies. Mol Psychiatry. 2020;25(7):1487–1499. doi: 10.1038/s41380-019-0595-x [DOI] [PubMed] [Google Scholar]
- 11.Batty GD, Russ TC, Stamatakis E, et al. Psychological distress in relation to site specific cancer mortality: pooling of unpublished data from 16 prospective cohort studies. BMJ. 2017;356:j108. doi: 10.1136/bmj.j108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lee H, Singh GK. Psychological Distress and Heart Disease Mortality in the United States: results from the 1997–2014 NHIS-NDI Record Linkage Study. Int j MCH AIDS. 2020;9(3):260–273. doi: 10.21106/ijma.391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bo A, Pouwer F, Juul L, et al. Prevalence and correlates of diabetes distress, perceived stress and depressive symptoms among adults with early-onset Type 2 diabetes: cross-sectional survey results from the Danish DD2 study. Diabetic Med. 2020;37(10):1679–1687. doi: 10.1111/dme.14087 [DOI] [PubMed] [Google Scholar]
- 14.Hayashino Y, Okamura S, Tsujii S, et al. Association between diabetes distress and all-cause mortality in Japanese individuals with type 2 diabetes: a prospective cohort study (Diabetes Distress and Care Registry in Tenri [DDCRT 18]). Diabetologia. 2018;61(9):1978–1984. doi: 10.1007/s00125-018-4657-4 [DOI] [PubMed] [Google Scholar]
- 15.Hayashino Y, Okamura S, Tsujii S, et al. The joint association of diabetes distress and depressive symptoms with all-cause mortality in Japanese individuals with type 2 diabetes: a prospective cohort study (Diabetes Distress and Care Registry in Tenri [DDCRT 20]). Diabetologia. 2020;63(12):2595–2604. doi: 10.1007/s00125-020-05274-6 [DOI] [PubMed] [Google Scholar]
- 16.Lindekilde N, Pouwer F. More work is needed to better understand diabetes distress as a predictor of all-cause mortality in type 2 diabetes. Diabetologia. 2018;61(10):2247–2248. doi: 10.1007/s00125-018-4714-z [DOI] [PubMed] [Google Scholar]
- 17.Egede LE, Nietert PJ, Zheng D. Depression and all-cause and coronary heart disease mortality among adults with and without diabetes. Diabetes Care. 2005;28(6):1339–1345. doi: 10.2337/diacare.28.6.1339 [DOI] [PubMed] [Google Scholar]
- 18.Hamer M, Stamatakis E, Kivimäki M, et al. Psychological distress, glycated hemoglobin, and mortality in adults with and without diabetes. Psychosom Med. 2010;72(9):882–886. doi: 10.1097/PSY.0b013e3181f6696e [DOI] [PubMed] [Google Scholar]
- 19.Centers for Disease Control and Prevention. Available from: www.cdc.gov/nchs/nhis/index.htm. Accessed July1, 2021.
- 20.Lynn A, Blewett JARD, King ML, Williams KCW. IPUMS Health Surveys: National Health Interview Survey, Version 6.4 [dataset].
- 21.The Linkage of National Center for Health Statistics Survey Data to the National Death Index-2015 Linked Mortality File (LMF): methodology overview and analytic considerations; 2019. Available from: www.cdc.gov/nchs/data/datalinkage/lmf2015_methodology_analytic_considerations.pdf. Accessed July1, 2021.
- 22.Kessler RC, Barker PR, Colpe LJ, et al. Screening for serious mental illness in the general population. Arch Gen Psychiatry. 2003;60(2):184–189. doi: 10.1001/archpsyc.60.2.184 [DOI] [PubMed] [Google Scholar]
- 23.Prochaska JJ, Sung HY, Max W, et al. Validity study of the K6 scale as a measure of moderate mental distress based on mental health treatment need and utilization. Int J Methods Psychiatr Res. 2012;21(2):88–97. doi: 10.1002/mpr.1349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pratt LA. Serious psychological distress, as measured by the K6, and mortality. Ann Epidemiol. 2009;19(3):202–209. doi: 10.1016/j.annepidem.2008.12.005 [DOI] [PubMed] [Google Scholar]
- 25.Venkataramani AS, Shah SJ, O’Brien R, et al. Health consequences of the US Deferred Action for Childhood Arrivals (DACA) immigration programme: a quasi-experimental study. Lancet Public Health. 2017;2(4):e175–e81. doi: 10.1016/s2468-2667(17)30047-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nishi D, Susukida R, Usuda K, et al. Trends in the prevalence of psychological distress and the use of mental health services from 2007 to 2016 in Japan. J Affect Disord. 2018;239:208–213. doi: 10.1016/j.jad.2018.07.016 [DOI] [PubMed] [Google Scholar]
- 27.Piercy KL, Troiano RP, Ballard RM, et al. The physical activity guidelines for Americans. JAMA. 2018;320(19):2020–2028. doi: 10.1001/jama.2018.14854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Andersson T, Alfredsson L, Källberg H, et al. Calculating measures of biological interaction. Eur J Epidemiol. 2005;20(7):575–579. doi: 10.1007/s10654-005-7835-x [DOI] [PubMed] [Google Scholar]
- 29.Zhang YB, Chen C, Pan XF, et al. Associations of healthy lifestyle and socioeconomic status with mortality and incident cardiovascular disease: two prospective cohort studies. BMJ. 2021;373:n604. doi: 10.1136/bmj.n604 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Su X, Zhao Y, Yang Y, et al. Correlation between exposure to fine particulate matter and hypertensive disorders of pregnancy in Shanghai, China. Environ Health. 2020;19(1):101. doi: 10.1186/s12940-020-00655-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Singh PN, Wang X. Simulation study of the effect of the early mortality exclusion on confounding of the exposure-mortality relation by preexisting disease. Am J Epidemiol. 2001;154(10):963–971. doi: 10.1093/aje/154.10.963 [DOI] [PubMed] [Google Scholar]
- 32.Johnson SB, Marrero D. Innovations in healthcare delivery and policy: implications for the role of the psychologist in preventing and treating diabetes. Am Psychol. 2016;71(7):628–637. doi: 10.1037/a0040439 [DOI] [PubMed] [Google Scholar]
- 33.Gregg EW, Cheng YJ, Srinivasan M, et al. Trends in cause-specific mortality among adults with and without diagnosed diabetes in the USA: an epidemiological analysis of linked national survey and vital statistics data. Lancet. 2018;391(10138):2430–2440. doi: 10.1016/s0140-6736(18)30314-3 [DOI] [PubMed] [Google Scholar]
- 34.Dirmaier J, Watzke B, Koch U, et al. Diabetes in primary care: prospective associations between depression, nonadherence and glycemic control. Psychother Psychosom. 2010;79(3):172–178. doi: 10.1159/000296135 [DOI] [PubMed] [Google Scholar]
- 35.McKellar JD, Humphreys K, Piette JD. Depression increases diabetes symptoms by complicating patients’ self-care adherence. Diabetes Educ. 2004;30(3):485–492. doi: 10.1177/014572170403000320 [DOI] [PubMed] [Google Scholar]
- 36.Aikens JE. Prospective associations between emotional distress and poor outcomes in type 2 diabetes. Diabetes Care. 2012;35(12):2472–2478. doi: 10.2337/dc12-0181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fisher L, Mullan JT, Arean P, et al. Diabetes distress but not clinical depression or depressive symptoms is associated with glycemic control in both cross-sectional and longitudinal analyses. Diabetes Care. 2010;33(1):23–28. doi: 10.2337/dc09-1238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dalsgaard EM, Vestergaard M, Skriver MV, et al. Psychological distress, cardiovascular complications and mortality among people with screen-detected type 2 diabetes: follow-up of the ADDITION-Denmark trial. Diabetologia. 2014;57(4):710–717. doi: 10.1007/s00125-014-3165-4 [DOI] [PubMed] [Google Scholar]
- 39.Ting RZ, Nan H, Yu MW, et al. Diabetes-related distress and physical and psychological health in Chinese type 2 diabetic patients. Diabetes Care. 2011;34(5):1094–1096. doi: 10.2337/dc10-1612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ascher-Svanum H, Zagar A, Jiang D, et al. Associations between glycemic control, depressed mood, clinical depression, and diabetes distress before and after insulin initiation: an exploratory, post hoc analysis. Diabetes Ther. 2015;6(3):303–316. doi: 10.1007/s13300-015-0118-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fang H, Tu S, Sheng J, et al. Depression in sleep disturbance: a review on a bidirectional relationship, mechanisms and treatment. J Cell Mol Med. 2019;23(4):2324–2332. doi: 10.1111/jcmm.14170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Huang YC, Zuñiga JA, García AA. Association between sleep and serious psychological distress in patients with diabetes. Psychol Health Med. 2019;24(8):925–935. doi: 10.1080/13548506.2019.1612075 [DOI] [PubMed] [Google Scholar]
- 43.Spiegel K, Leproult R, Van Cauter E. Impact of sleep debt on metabolic and endocrine function. Lancet (London, England). 1999;354(9188):1435–1439. doi: 10.1016/s0140-6736(99)01376-8 [DOI] [PubMed] [Google Scholar]
- 44.Zhu B, Quinn L, Kapella MC, et al. Relationship between sleep disturbance and self-care in adults with type 2 diabetes. Acta Diabetol. 2018;55(9):963–970. doi: 10.1007/s00592-018-1181-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wang Y, Huang W, O’Neil A, et al. Association between sleep duration and mortality risk among adults with type 2 diabetes: a prospective cohort study. Diabetologia. 2020. doi: 10.1007/s00125-020-05214-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Osborne MT, Shin LM, Mehta NN, et al. Disentangling the links between psychosocial stress and cardiovascular disease. Circ Cardiovasc Imaging. 2020;13(8):e010931. doi: 10.1161/circimaging.120.010931 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lutgendorf SK, Andersen BL. Biobehavioral approaches to cancer progression and survival: mechanisms and interventions. Am Psychol. 2015;70(2):186–197. doi: 10.1037/a0035730 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Fedeles BI, Freudenthal BD, Yau E, et al. Intrinsic mutagenic properties of 5-chlorocytosine: a mechanistic connection between chronic inflammation and cancer. Proc Natl Acad Sci U S A. 2015;112(33):E4571–80. doi: 10.1073/pnas.1507709112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pimple P, Lima BB, Hammadah M, et al. Psychological distress and subsequent cardiovascular events in individuals with coronary artery disease. J Am Heart Assoc. 2019;8(9):e011866. doi: 10.1161/jaha.118.011866 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gamble JM, Chibrikov E, Midodzi WK, et al. Examining the risk of depression or self-harm associated with incretin-based therapies used to manage hyperglycaemia in patients with type 2 diabetes: a cohort study using the UK Clinical Practice Research Datalink. BMJ Open. 2018;8(10):e023830. doi: 10.1136/bmjopen-2018-023830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Shivavedi N, Kumar M, Tej G, et al. Metformin and ascorbic acid combination therapy ameliorates type 2 diabetes mellitus and comorbid depression in rats. Brain Res. 2017;1674:1–9. doi: 10.1016/j.brainres.2017.08.019 [DOI] [PubMed] [Google Scholar]
- 52.Bai X, Liu Z, Li Z, et al. The association between insulin therapy and depression in patients with type 2 diabetes mellitus: a meta-analysis. BMJ Open. 2018;8(11):e020062. doi: 10.1136/bmjopen-2017-020062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Nanayakkara N, Pease A, Ranasinha S, et al. Depression and diabetes distress in adults with type 2 diabetes: results from the Australian National Diabetes Audit (ANDA) 2016. Sci Rep. 2018;8(1):7846. doi: 10.1038/s41598-018-26138-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Benroubi M. Fear, guilt feelings and misconceptions: barriers to effective insulin treatment in type 2 diabetes. Diabetes Res Clin Pract. 2011;93 Suppl 1:S97–9. doi: 10.1016/s0168-8227(11)70021-3 [DOI] [PubMed] [Google Scholar]
- 55.Day HR, Parker JD. Self-report of diabetes and claims-based identification of diabetes among Medicare beneficiaries. Natl Health Stat Report. 2013;(69):1–14. [PubMed] [Google Scholar]
- 56.Margolis KL, Lihong Q, Brzyski R, et al. Validity of diabetes self-reports in the Women’s Health Initiative: comparison with medication inventories and fasting glucose measurements. Clin Trials. 2008;5(3):240–247. doi: 10.1177/1740774508091749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Furukawa TA, Kessler RC, Slade T, et al. The performance of the K6 and K10 screening scales for psychological distress in the Australian National Survey of Mental Health and Well-Being. Psychol Med. 2003;33(2):357–362. doi: 10.1017/s0033291702006700 [DOI] [PubMed] [Google Scholar]