Abstract
Hypercoagulability related to SARS-CoV-2 infection is one of the main extrapulmonary complications of COVID-19. We present three cases of intrabdominal thrombotic complications related to the state of hypercoagulability of COVID-19 and its tomographic features. Hypercoagulability state should be taking into account in the interpretation of radiological images in all infected patients with COVID-19.
Keywords: COVID- 19, thrombosis, complications, mesenteric ischemia, computed tomography
Introduction
The viral infection of the new coronavirus [SARS-CoV-2] can produce a variety of extra pulmonary complications, and the hypercoagulability state is one of the most important and serious sequela [1]. The physiopathology of this complication is unknown, however, the suggested mechanism associated could be: endothelial injury by direct invasion of the virus to endothelial cells [2], activation of the intravascular complement system, and endothelia known as "cytokine storm" [3,4]. Other mechanisms could be increased coagulation factors such as factor VIII and fibrinogen, hyperviscosity of the blood [5], thrombotic microangiopathy, and venous stasis [6,7].
D-dimer elevation and hypercoagulability are prognostic factors of morbidity [6]. In patients bearing these characteristics, arterial thrombosis risk appears to be lower than the risk of venous thrombosis.
Although percutaneous endovascular is considered the first choice of treatment in patients with mesenteric and/or visceral abdominal ischemia. In our experience, we have observed that patients with COVID-19 infection might have generalized or vascular access thrombosis, possibly because the endothelium manipulation enhances the demonstrated prothrombotic state.
This report presents three cases of intra-abdominal ischemic pathology associated with a prothrombotic state triggered by COVID-19 infection.
Clinical case 1
We attended in the urgency room a 62 year-old male patient with a history of high blood pressure with a 2 hour evolution and colic-type abdominal pain after food intake, along with several emetic episodes. Five days before admission, the patient had a molecular test positive for SARS-CoV-2, managed at home because he had only mild symptoms.
Clinical parameters at admission were cardiac frequency, 88 lats/min, respiratory frequency, 16/min, arterial pressure, 140/90 mm/Hg, temperature, 36.5°C, and 96% oxygen saturation. Hearth noises were rhythmic without murmurs, vesicular murmurs preserved without over grouped noises. Depressible soft abdomen, painful on palpation in the mesogastrium without signs of peritoneal irritation.
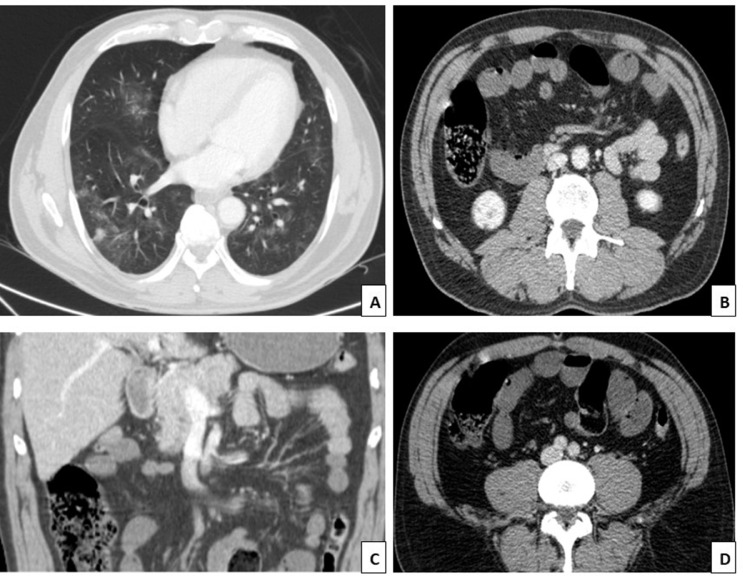
In the abdominal computed tomography (CT) with contrast, multiple peripheral frosted glass spotlights, compatible with viral pneumonia, were observed at both pulmonary bases. In addition, there was an absence of opacification of the superior mesenteric artery in its distal part, suggesting the presence of thrombus, likewise, dilation of the thin intestinal loops was identified (distal jejunum and proximal ileum) (Fig. 1).
Fig. 1.
(A) Lung bases with multiple peripheral ground glass foci compatible with viral pneumonia. (B and C) Distal thrombus in the superior mesenteric artery with the occlusion of this and its branches. (D) Dilation of thin intestinal loops
The laboratory studies showed leukocytosis (20100 u/L), increased D-dimer (58608 ng/ml), LDH (534u/L), and serum ferritin (1543 u/L), while other studies were within normal limits.
The medical treatment given was anticoagulation with non-fractional heparin (HNF). Thrombectomy was not considered due to the distal location of the thrombus. The endovascular manipulation of the endothelium increases the risk of generalized thrombosis due to the hypercoagulability of the patients. At 48 hours after admission, the patient had increased pain and abdominal distention progressively increased his oxygen requirements and received mechanical ventilation.
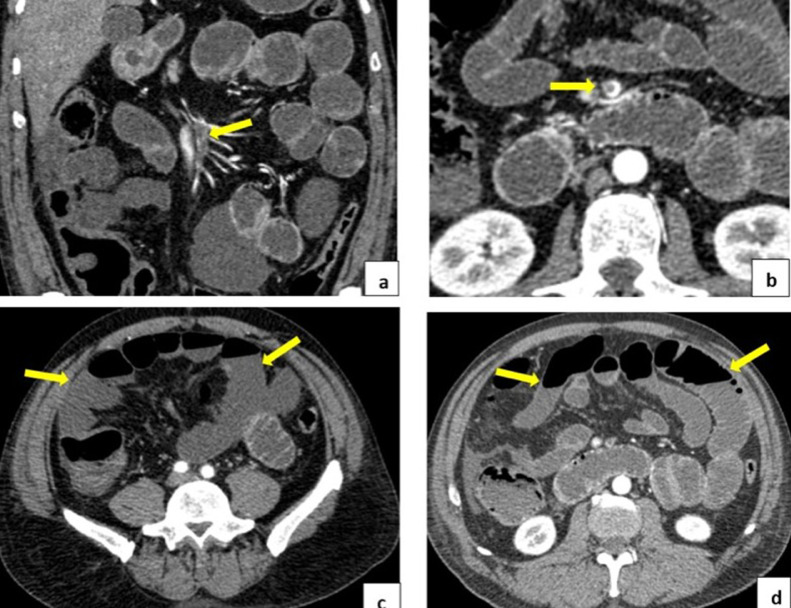
Abdominal computed tomography angiography showed a sub-occlusive thrombus-filling defect in the distal third of the upper mesenteric artery and its distal branches. It was associated with the diffuse absence of enhancement in the ileum wall due to the evolving ischemic events. Adjacent free fluid was observed as a sign of loop distress, diffuse dilation of intestinal loops, and formation of air-fluid levels due to secondary ileums. (Fig. 2).
Fig. 2.
(A and B) Subocclusive thrombus in the distal third of the superior mesenteric artery. Absence of enhancement in the walls of thin intestinal loops in the relationship of signs of evolving ischemia. (D) Diffuse dilation of intestinal loops and presence of air-fluid levels
Due to the tomographic findings, we indicated an exploratory laparotomy and were observed that a moderate amount of hemorrhagic fluid throughout the abdominal cavity. Areas of necrosis and segmental ischemia throughout the jejunum and ileum because of their wide extension did not benefit from the surgical resection. Subsequently, the patient presented a torpid evolution, given the intestinal ischemia associated with complications from COVID-19 infection.
Clinical case 2
A 22-year-old female patient, without significant past medical history, presented a 20-days history of abdominal pain mainly located in the mesogastrium. The patient had multiple admissions to the emergency room. In her last visit, 8 days before admittance, she presented with an increase in appendix size. It was shown in the abdominal CT the patient underwent an appendectomy. The pain reappeared on the first postoperative day and was associated with fever for 3 days. The patient was readmitted without gastrointestinal symptoms. Moreover, the patient referred she had close contact with relatives with respiratory symptoms who had not been tested for COVID-19.
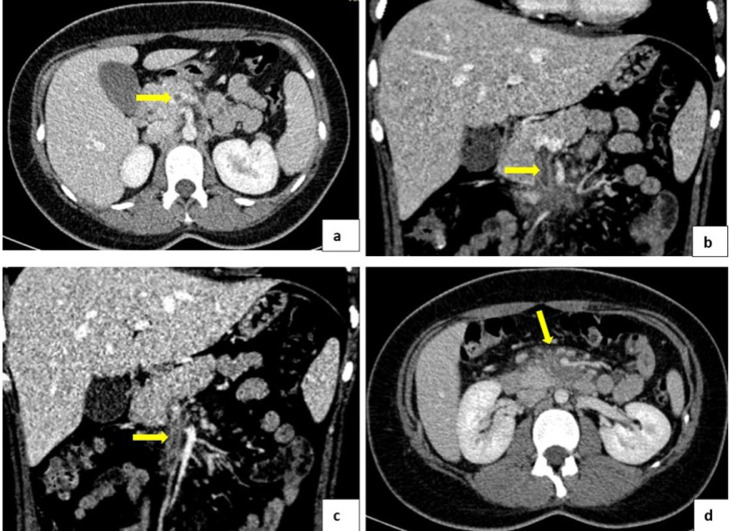
In the physical examination, at admission, the patient had O2 Sat 96%, soft depressive abdomen, healthy appendectomy, surgical wound without signs of infection, with pain on palpation of the transverse and descending colic frame without signs of peritoneal irritation. The abdominal angiography was indicated and acute and/or subacute thrombosis of the superior mesenteric vein with edematous changes of the adjacent fat was visualized (Fig. 3).
Fig. 3.
(A, B, and C) Obstructive thrombus is evidenced in the superior mesenteric vein. (D) Diffuse increase in the density of the fatty planes of the root of the mesentery to inflammatory changes
Thrombophilia and autoimmune pathology were excluded. The management was carried out with anticoagulation, analgesia, and antibiotic therapy due to the patient presenting feverish peaks during hospitalization. She had a favorable evolution.
Clinical case 3
A 65 year-old female patient came to our center with a history of left nephrectomy, 15 day history of respiratory symptoms, a positive PCR study for COVID-19 (8 days before admission), 7 days of evolution of abdominal pain predominantly in the upper hemiabdomen, with back irradiation, associated with hyporexia.
In the physical examination, at admittance, we found FC: 86/min, FR: 15/min, PA: 162/97, T: 36°C, Sat O2: 94%, visual analog pain scale: 8/10. Rhythmic heart sounds, non-aggregated ventilated lungs, depressible soft abdomen, pain on palpation of the upper hemiabdomen, and no signs of peritoneal irritation.
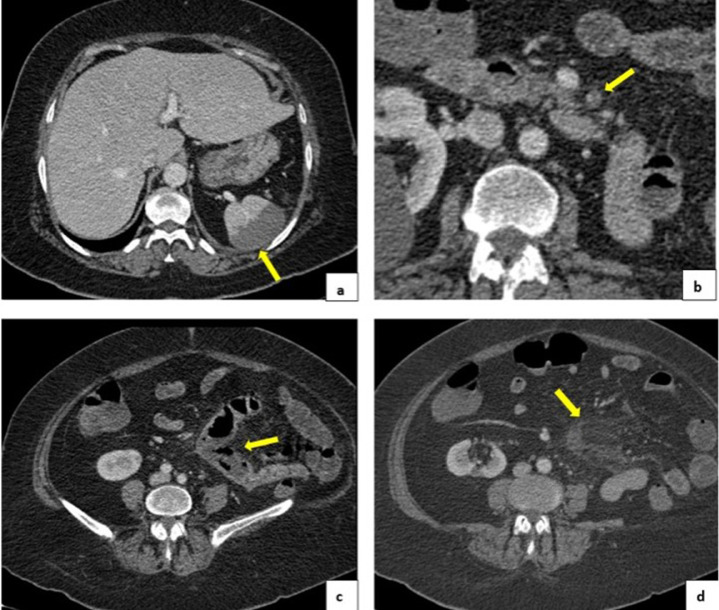
Contrast abdominal tomography was performed, where we found thrombi in the left jejunal artery branch with infarction of the corresponding jejunal loops. In addition, we observed edema of the mesenteric fat and hypo attenuation of the intestinal wall with greater dilatation of the compromised intestinal segment. It showed signs of splenic infarction and pleural parenchymal bands in both lung bases (Fig. 4).
Fig. 4.
(A) Splenic infarction. (B) Thrombus in left jejunal arterial branches. (C) Signs of intestinal infarction: hypoattenuation of the intestinal wall with greater dilation of the compromised intestinal segment. (D) Mesenteric fat edema
The patient underwent an exploratory laparotomy with an intestinal resection of the compromised segment, intraperitoneal collection, drainage, and cavity lavage were performed.
Discussion
Currently, tested computed tomography is considered as the imaging study of choice for the evaluation of patients with clinical suspicion of arterial or venous thrombosis. It allows direct visualization of the thrombus as a defect within the affected vessel. Due to its high sensitivity (93.3%) and specificity (95.9%), it has displaced angiography [8], [9], [10]. In addition, it allows characterizing the complications derived such as mesenteric ischemia, splenic infarction, liver, renal, portal thrombosis, Bud Chiari, among others.
For the evaluation of intra-abdominal thrombotic pathology, it suggested to perform an angiotomography with an iodinated contrast volume of 80 to 100 ml (320-350) through a 20-gauge antecubital intravenous catheter, at an injection speed of 4 ml/s, with a slice thickness of 0.625 mm and multiplanar reconstructions that allow adequate characterization of the findings [8].
The most common intra-abdominal thrombotic complications are described below.
Mesenteric ischemia
Within abdominal vessel thrombosis, the most common presentation is mesenteric ischemia, which usually appears acutely in up to 95% of cases. Its etiology corresponds to arterial obstruction, both embolic and thrombotic in 60% to 70% of cases, venous obstruction in 5% to 10%, and non-occlusive associated with pathologies such as acute myocardial infarction, low cardiac output, and hypovolemia in 20% [9].
Acute mesenteric ischemia has classically presented in adults over 60 years of age due to comorbidities such as atrial fibrillation, atherosclerosis, arterial hypertension, heart failure, valve disease, and portal hypertension. However, in the current context of the COVID-19 pandemic, both acute mesenteric ischemia and complications associated with thrombosis of abdominal visceral vessels are a cause that must rule out in patients infected by the virus, who present with acute abdominal pain, regardless of age group.
The severity of mesenteric ischemia varies from mild, where generally transient superficial changes of the intestinal mucosa occur, to severe, producing transmural necrosis of the intestinal wall, more dangerous and potentially fatal [10].
The natural evolution of the disease leads to successive complications that begin with bacterial proliferation in the intestinal wall (pneumatosis of the wall) favored by its necrosis, subsequently, the gas passes to the mesenteric vessels and from there to the portal vein (portal pneumatosis). Finally, sepsis and/or intestinal perforation are commonly the causes of death. It is important to mention that ischemia of thrombotic origin can lead to reactive mesenteric vasoconstriction, reducing compensatory collateral flow, worsening ischemic damage [11].
Regardless of the clinic, abdominal pain is a common characteristic in patients with this pathology. Usually of rapid onset and high intensity could be associated with nausea, vomiting, abdominal distension, and diarrhea [9,10].
The findings observed in tomography include identification of the arterial and/or venous vascular filling defect. In addition, visceral findings such as parietal thickening, a more marked mural of the affected loop wall with a “target” appearance, dilation of intestinal loops, and hydro levels in the airways. In more severe cases, when there is intestinal infarction, thinning, and hypo enhancement of the intestinal wall with or without pneumatosis, the air in the mesenteric and/or portal veins. Other findings include diffuse increased attenuation of mesenteric fat, interested fluid, and pneumoperitoneum [10], [11], [12], [13].
In any case, the success of the treatment is obtained by influencing the reversible phase of ischemia to avoid complete necrosis of the intestinal loop. On the other, endovascular management is based on quickly restoring blood flow in the affected vessel using techniques such as aspiration, thrombectomy, thrombolysis, and angioplasty with or without stenting, surgical exploration of the intestine, and resection of necrotic intestinal segments, and anticoagulation [14].
Portal thrombosis
It is a rare complication in the general population with a prevalence of 1%, relative to most cases due to hepatic cirrhosis. However, it can also appear as a primarily vascular disorder, which is a hereditary or acquired prothrombotic state, associated with tumor invasion of the carrier or pylephlebitis [15,16]. It can also spread to the splenic vein and upper mesenteric vein [17].
Tomographic findings are the filling defect that partially or completely occludes the light of the vessel and where enhances the edge of the vessel wall, alterations, and heterogeneity in the liver parenchyma highlight this pattern. In chronic cases, there can be seen a decreased diameter of the carrier with collateral circulation around it, which can in some cases be seen as a solid mass in the liver hilum with progressive enhancement, where it is not possible to individualize the national veins, which is known as "portalcavernomatosis" [18]. When thrombosis is acute, a higher attenuation content can sometimes be identified inside the portal vein and an increase in cup caliber.
Splenic infarction
For the evaluation, it is recommended to carry out the tomographic study in the venous phase, to avoid confusion with the “brindle” the enhancement characteristic of the splenic parenchyma in the arterial phase. Wedge hypodensity is one of the typical characteristics of this pathology, as well as multiple hypodense or hypo enhanced areas of the entire spleen, in cases of multiple infarcts or global infarction respectively [19,20].
It should be taken into account that in the hyperacute phase there may be hyperdense areas in the simple phase due to hemorrhagic infarcts. While in the chronic phase, it is common to find areas of fibrotic retraction of the parenchyma or cystic areas due to tissue liquefaction [20, 21]. Associated complications are rupture and abscess [22].
Bud Chiari syndrome
It comprises a group of conditions characterized by a partial or complete blockage of the outlet flow of the hepatic veins. If the blockage is severe and not corrected early, it can lead to necrosis of hepatocytes and progress to liver fibrosis and cirrhosis [23,24].
The most common cause is an underlying hypercoagulability or prothrombotic state. Clinical presentation ranges from mild symptoms to fulminant hepatic impairment, and most patients manifest as a chronic liver disease [24]. Acutely, patients have predominant pain in the hemiabdomen, ascites, hepatomegaly, renal failure, and jaundice [25].
In tomography, the most common direct sign is the absence of visualization of the hepatic veins. If thrombosis is acute, a filling defect will be evident inside the hepatic veins, which are usually distended, with the presence of a hypodense thrombus in light [26].
There are also indirect tomographic findings characterized by morphological changes in the patterns of parenchymal enhancer and collateral circulation. In the acute stage, there is uniform hepatomegaly with a smooth contour of the liver, a heterogeneous liver enhanced in the portal phase, and hypo-attentive areas setting up a mosaic pattern, due to venous congestion. More than in subacute and chronic stages, when intrahepatic collateral has developed and hepatic mass fibrosis has been established, it is common to find a liver with nodular contour is, with volume redistribution to signs of cirrhosis and differences in the pattern of energy become more subtle [25,26].
Conclusion
Hypercoagulability is 1 of the most important extra-pulmonary complications in patients with SARS CoV-2. In the current context of the COVID-19 pandemic, complications associated with thrombosis of abdominal visceral vessels are a cause that must rule out in infected patients with the virus, who had acute abdominal pain independent of the age group.
Currently, contrast computed tomography is considered the imaging study of choice for the evaluation of patients with clinical suspicion of arterial or venous thrombosis. When abdominal vascular occlusion is suspected, it is important to carry out the study with an angiography technique that includes at least 1 arterial and portal venous phase.
Although endovascular management is considered of choice in patients with mesenteric ischemia to influence the reversible phase of intestinal ischemia. We consider that for patients with COVID-19 infection, the possible complications and advantages of the procedure should be taken into account, because of the manipulation of the endothelium. Especially in these patients, endovascular approach devices can increase the risk of generalized thrombosis or in the approach route, adding to the already established state of hypercoagulability.
Patient consent
Our case reports only used tomography images. As said in Guide Authors, the patient's consent is not required for this type of image. I cited bellowed:
Non-identifiable images
● Formal consents are not required for the use of entirely anonymized images from which the individual cannot be identified- for example, X-rays, ultrasound images, pathology slides, or laparoscopic images, provided that these do not contain any identifying marks and are not accompanied by text that might identify the individual concerned.
Authors contributions
LP, AP, JG, SE, GA, and DM were involved in the histopathological assessment and diagnosis of the cases, patients treatments and examination. JP and AG made signifcant contributions in designing and drafting the manuscript, as well as reviewing the literature. All the authors contributed to the manuscript revision and gave fnal approval for publication. Moreover, the authors read and approved the fnal manuscript.
Footnotes
Competing Interests: None
References
- 1.Abou-Ismail MY, Diamond A, Kapoor S, Arafah Y, Nayak L. The hypercoagulable state in COVID-19: incidence, pathophysiology, and management. Thromb Res. 2020;194:101–115. doi: 10.1016/j.thromres.2020.06.029. Doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Teuwen L-A, Geldhof V, Pasut A, Carmeliet P. COVID-19: the vasculature unleashed. Nat Rev Immunol. 2020;20:389–391. doi: 10.1038/s41577-020-0343-0. Doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guan W, Ni Z, Hu Y, Liang W, Ou C, He J. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. Doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. Doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Maier CL, Truong AD, Auld SC, Polly DM, Tanksley C-L, Duncan A. COVID-19-associated hyperviscosity: a link between inflammation and thrombophilia? Lancet (London, England) 2020;395:1758–1759. doi: 10.1016/S0140-6736(20)31209-5. Doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Levolger S, Bokkers R, Wille J, Kropman R, Vries JP. Arterial thrombotic complications in COVID-19 patients. J Vasc Surg Cases Innov Tech. 2020 doi: 10.1016/j.jvscit.2020.06.012. Doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Motta-Ramírez GA, Sánchez-García JC, Ontiveros-Rodríguez A. Acute mesenteric ischemia: urgency that requires a comprehensive diagnostic approach. An Radiol Mexico. 2015;14:66–88. [Google Scholar]
- 8.Cademartiri F, Raaijmakers RHJM, Kuiper JW, van Dijk LC, Pattynama PMT, Krestin GP. Multi–Detector Row CT angiography in patients with abdominal angina. RadioGraphics. 2004;24:969–984. doi: 10.1148/rg.244035166. Doi: [DOI] [PubMed] [Google Scholar]
- 9.Wiesner W, Khurana B, Ji H, Ros PR. CT of acute bowel ischemia. Radiology. 2003;226:635–650. doi: 10.1148/radiol.2263011540. Doi: [DOI] [PubMed] [Google Scholar]
- 10.Schieda N, Fasih N, Shabana W. Triphasic CT in the diagnosis of acute mesenteric ischaemia. Eur Radiol. 2013;23:1891–1900. doi: 10.1007/s00330-013-2797-y. Doi: [DOI] [PubMed] [Google Scholar]
- 11.Lourenco PDM, Rawski R, Mohammed MF, Khosa F, Nicolaou S, McLaughlin P. Dual-Energy CT Iodine Mapping and 40-keV monoenergetic applications in the diagnosis of acute bowel ischemia. Am J Roentgenol. 2018;211:564–570. doi: 10.2214/AJR.18.19554. Doi: [DOI] [PubMed] [Google Scholar]
- 12.Menke J. Diagnostic accuracy of multidetector CT in acute mesenteric ischemia: systematic review and meta-analysis. Radiology. 2010;256:93–101. doi: 10.1148/radiol.10091938. Doi: [DOI] [PubMed] [Google Scholar]
- 13.Fitzpatrick LA, Rivers-Bowerman MD, Thipphavong S, Clarke SE, Rowe JA, Costa AF. Pearls, pitfalls, and conditions that mimic mesenteric ischemia at CT. radiogr a. Rev Publ Radiol Soc North Am Inc. 2020;40:545–561. doi: 10.1148/rg.2020190122. Doi: [DOI] [PubMed] [Google Scholar]
- 14.Ierardi AM, Tsetis D, Sbaraini S, Angileri SA, Galanakis N, Petrillo M. The role of endovascular therapy in acute mesenteric ischemia. Ann Gastroenterol. 2017;30:526–533. doi: 10.20524/aog.2017.0164. Doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lee H-K, Park SJ, Yi B-H, Yeon E-K, Kim JH, Hong H-S. Portal vein thrombosis: CT features. Abdom Imaging. 2008;33:72–79. doi: 10.1007/s00261-007-9200-x. Doi: [DOI] [PubMed] [Google Scholar]
- 16.Denninger MH, Chat Y, Casadevall N, Hillaire S, Guillin MC, Bezeaud A. Cause of portal or hepatic venous thrombosis in adults: the role of multiple concurrent factors. Hepatology. 2000;31:587–591. doi: 10.1002/hep.510310307. Doi: [DOI] [PubMed] [Google Scholar]
- 17.Dabén RP, Ten ADD, Wolf VF, Lorenzo JDLC, Del Pozo JC, Sánchez FG. Radiological approach to portal thrombosis. Seram. 2018:0. [Google Scholar]
- 18.Rossini S., Haberman D., Perez J., Capiel C., Costantino S., Landi M., Bouzas C. Portal Vein Thrombosis: Findings in MDCT and MRI. 2014 https://www.webcir.org/revistavirtual/articulos/2015/junio/argentina/RARD/portal_vein_thrombosis.pdf. [Google Scholar]
- 19.Nores M, Phillips EH, Morgenstern L, Hiatt JR. The clinical spectrum of splenic infarction. Am Surg. 1998;64:182–188. [PubMed] [Google Scholar]
- 20.Rabushka LS, Kawashima A, Fishman EK. Imaging of the spleen: CT with supplemental MR examination. Radiogr a Rev Publ Radiol Soc North Am Inc. 1994;14:307–332. doi: 10.1148/radiographics.14.2.8190956. Doi: [DOI] [PubMed] [Google Scholar]
- 21.Collie DA, Ferguson JM, Allan PL, Redhead DN. Case report: computed tomography features of complete splenic infarction, cavitation and spontaneous decompression complicating pancreatitis. Br J Radiol. 1995;68:662–664. doi: 10.1259/0007-1285-68-810-662. Doi: [DOI] [PubMed] [Google Scholar]
- 22.Goerg C, Schwerk WB. Splenic infarction: sonographic patterns, diagnosis, follow-up, and complications. Radiology. 1990;174:803–807. doi: 10.1148/radiology.174.3.2406785. Doi: [DOI] [PubMed] [Google Scholar]
- 23.Goel RM, Johnston EL, Patel K V, Wong T. Budd-Chiari syndrome: investigation, treatment and outcomes. Postgraduate Med J. 2015;91:692–697. doi: 10.1136/postgradmedj-2015-133402. Doi: [DOI] [PubMed] [Google Scholar]
- 24.Bansal V, Gupta P, Sinha S, Dhaka N, Kalra N, Vijayvergiya R. Budd-Chiari syndrome: imaging review. Br J Radiol. 2018;91 doi: 10.1259/bjr.20180441. Doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Loomes DE, Chang A, Webber D, Scudamore CH, Yoshida EM. Acute Budd-Chiari syndrome. Can J Gastroenterol. 2011;25:302–303. doi: 10.1155/2011/756425. Doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Faraoun SA, Boudjella MEA, Debzi N, Benidir N, Afredj N, Guerrache Y. Budd-Chiari syndrome: an update on imaging features. Clin Imaging. 2016;40:637–646. doi: 10.1016/j.clinimag.2016.01.006. Doi: [DOI] [PubMed] [Google Scholar]