Key Points
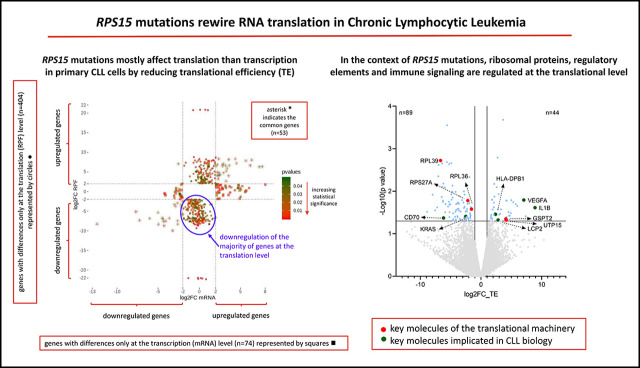
RPS15 mutations rewire the translation program of primary CLL cells by reducing their translational efficiency.
In the context of RPS15 mutations, ribosomal proteins, regulatory elements, and immune signaling are regulated at the translational level.
Visual Abstract
Abstract
Recent studies of chronic lymphocytic leukemia (CLL) have reported recurrent mutations in the RPS15 gene, which encodes the ribosomal protein S15 (RPS15), a component of the 40S ribosomal subunit. Despite some evidence about the role of mutant RPS15 (mostly obtained from the analysis of cell lines), the precise impact of RPS15 mutations on the translational program in primary CLL cells remains largely unexplored. Here, using RNA sequencing and ribosome profiling, a technique that involves measuring translational efficiency, we sought to obtain global insight into changes in translation induced by RPS15 mutations in CLL cells. To this end, we evaluated primary CLL cells from patients with wild-type or mutant RPS15 as well as MEC1 CLL cells transfected with mutant or wild-type RPS15. Our data indicate that RPS15 mutations rewire the translation program of primary CLL cells by reducing their translational efficiency, an effect not seen in MEC1 cells. In detail, RPS15 mutant primary CLL cells displayed altered translation efficiency of other ribosomal proteins and regulatory elements that affect key cell processes, such as the translational machinery and immune signaling, as well as genes known to be implicated in CLL, hence highlighting a relevant role for RPS15 in the natural history of CLL.
Introduction
In chronic lymphocytic leukemia (CLL), we and others have reported recurrent mutations in the RPS15 gene,1-4 which encodes a component of the 40S ribosomal subunit.1,2 RPS15 mutations are infrequent (∼4%) in untreated patients,2,3 albeit enriched (∼20%) within aggressive CLL, particularly in patients relapsing after treatment with fludarabine-cyclophosphamide-rituximab.1 Interestingly, RPS15 mutations were clonal before treatment and remained stable at relapse, pointing to an early event in CLL pathogenesis. In addition to the established role of ribosomal protein S15 (RPS15) in protein translation, evidence exists that RPS15 binds MDM2/MDMX, while its mutants reduce stabilization and increase p53 degradation, indicating a novel molecular mechanism involved in CLL pathobiology.1
A recent study reported that certain RPS15 mutations affected both protein stability and cell proliferation rates in the fibroblast HEK293T and CLL MEC1 cell lines and that RPS15 variants induced proteome-wide changes.5 Here, using ribosome sequencing (Ribo-seq), we investigated the possibility that defects in the synthesis or function of the ribosomes induced by RPS15 mutations could also affect the pattern of translated messenger RNAs (mRNAs) in both primary CLL cells and CLL cell lines.
Methods
Patient samples and generation of RPS15 mutant MEC1 cell lines
Blood samples were collected under informed consent from patients with CLL (supplemental Table 1). Additionally, transformed MEC1 CLL cells that overexpress mutant or wild-type RPS15 were generated as described in supplemental Methods.
The study was approved by the local ethics review committee of the participating institutions.
Ribosome profiling (Ribo-seq) and mRNA sequencing
Ribosome protected fragment (RPF) generation and library preparation for Ribo-seq and mRNA sequencing was performed using the TruSeq Ribo Profile (Mammalian) kit (Illumina) (supplemental methods). All samples were sequenced using Illumina’s NextSeq-500 (with 75 nt read length). Each sequencing run resulted in 38 to 113 million raw reads/sample, of which 5 to 33 million and 13 to 79 million reads were used for subsequent analysis for RPFs and mRNAs, respectively (supplemental Table 2), following a bioinformatics pipeline described in supplemental Methods.
Flow cytometry
The protein expression of BCL2L11 and CXCR4 was assessed by flow cytometry as detailed in supplemental Methods.
Results and discussion
Ribo-seq involves measuring translational efficiency (TE), by comparing the levels of ribosome-associated mRNA footprints to the total mRNA for each gene.6,7 Here, we applied Ribo-seq and RNA sequencing (RNA-seq) for studying primary CLL cells with wild-type RPS15 (CLL_RPS15wt) and mutant RPS15 (CLL_RPS15mut) (3 cases each), as well as mutant MEC1 (RPS15P131S transfected MEC1_RPS15mut) and wild-type MEC1 (RPS15wt transfected MEC1_RPS15wt) cells.
Ribo-seq revealed that (1) RPF libraries had the expected 28-nt size; (2) the reads from the RPF libraries were low at 3' untranslated regions, and a few RPFs mapped to annotated 5' untranslated regions, suggesting the presence of ribosomes at upstream open reading frames, while the majority mapped to coding regions; and (3) triplet periodicity was observed around the transcription start and stop sites. In contrast, the mRNA reads mapped uniformly across the length of the mRNA, as expected for randomly fragmented mRNA, with no triplet periodicity (supplemental Figure 1).
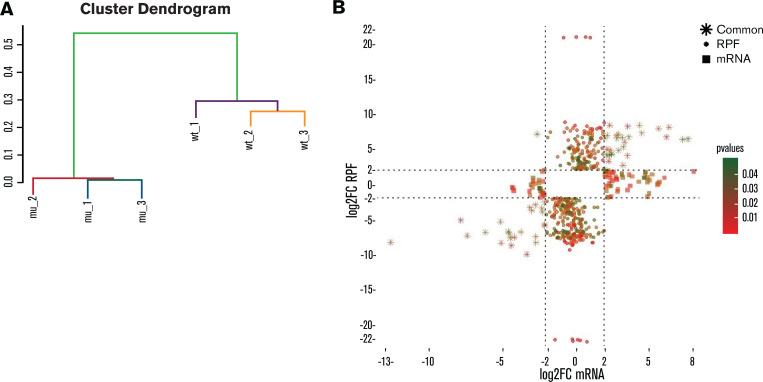
Unsupervised agglomerative hierarchical clustering analysis, based on the RPF normalized counts of the 4000 most variable genes, could distinguish RPS15mut from RPS15wt CLL cases (Figure 1A). In order to distinguish translational from transcriptional regulation of gene expression, we analyzed the Ribo-seq and mRNA-seq datasets separately and compared differences (log2 fold change [log2FC] ≥ |2|, P ≤ .05) in mRNA levels to variation in RPF abundance between CLL_RPS15wt and CLL_RPS15mut. We identified 3 groups of collectively 531 genes that represent different modes of regulation: (1) 404 genes with differences only at the translation (RPF) level, thus identified only by Ribo-seq analysis; (2) 74 genes with differences only at the transcription (mRNA) level, thus identified only by RNA-seq analysis; and (3) 53 genes with differences at both the transcription (mRNA) and translation (RPF) level, thus identified by both RNA-seq and Ribo-seq (Figure 1B). Altogether, these findings suggest that Ribo-seq can reveal additional information compared with mRNA-seq alone while also indicating that the predominant disruptive effect of RPS15 mutations in CLL is exerted at the translation level.
Figure 1.
RPS15 mutations mostly affect translation rather than transcription. (A) Cluster dendrogram based on unsupervised agglomerative hierarchical clustering analysis of the 4000 most variable genes (based on standard deviation). (B) Scatterplot indicating differentially expressed genes (log2FC≥|2|; P < .05) at mRNA and RPF level in RPS15mut vs RPS15wt CLL cases. The x-axis represents the log2FC of mRNAs, while the y-axis represents the log2FC of RPFs. The color code (green to red) indicates the statistical significance of the observed differences. Squares indicate genes that show differential expression at mRNA level only, circles indicate the genes that show differential expression at RPF level only, and stars indicate the genes that show differential expression at mRNA and RPF level.
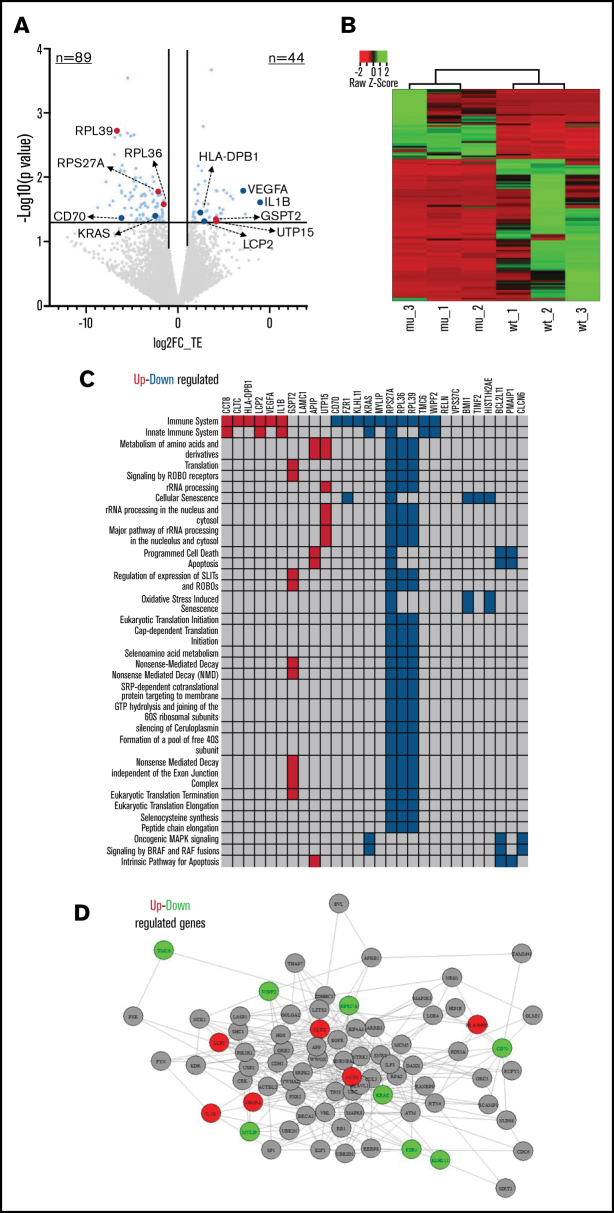
In order to detect the major patterns of translational regulation, we calculated the differences in TE between RPS15mut and RPS15wt CLL cases. We found 133 genes with altered TE (log2TE>|1|, P < .05) of which 89 (66.9%) were downregulated in RPS15mut CLL cases (Figure 2A; supplemental Table 3), implying that RPS15 mutations mainly repress mRNA translation. Supervised hierarchical clustering analysis based on the differentially translated genes could discriminate the RPS15mut from the RPS15wt CLL cases (Figure 2B).
Figure 2.
RPS15 mutations rewire CLL translational program. (A) Volcano plot indicating the results from differential TE analysis in RPS15mut vs RPS15wt CLL cases. The x-axis represents the log2FC of TE, while the y-axis represents P values. Genes that show statistically significant differential TE (log2FC ≥ |1|; P < .05) are highlighted in light blue. Genes highlighted in red are those implicated in the translation machinery, while genes in dark blue have already been implicated in CLL biology. (B) Supervised hierarchical clustering analysis based on the differentially translated genes between RPS15mut and RPS15wt CLL cases. (C) The figure shows enriched pathways from the Reactome database (false discovery rate < 0.05) and the differentially translated genes between RPS15mut and RPS15wt CLL cases. Upregulated genes in RPS15mut compared with RPS15wt cases are shown in red, while downregulated genes are shown in blue. (D) An example of the enriched immune signaling pathway. The interactions are confirmed by Biogrid. Upregulated genes in RPS15mut compared with RPS15wt cases are shown in red, while downregulated genes are shown in green.
Functional enrichment analysis revealed that deregulated genes are implicated in processes relevant to translation such as translation initiation, elongation, and termination as well as ribosomal RNA processing, indicating that RPS15 mutations affect key molecules of the translational machinery (Figure 2A). Moreover, immune processes were also affected; key deregulated molecules, already implicated in CLL biology, include CD70,8 IL1B,9 VEGFA,10 KRAS,11 HLA-DPB112 and LCP213 (Figure 2A,C-D; supplemental Table 4). Significant enrichment was found in processes related to protein binding, nucleic acid binding and catalytic activity on proteins (supplemental Table 5). Hence, many genes and processes associated with CLL and cancer at large are primarily translationally rather than transcriptionally deregulated in a setting of RPS15 mutations.
We next performed similar analyses in MEC1 cells where more genes were found to show differences at the mRNA level only (n = 94), while fewer genes (n = 32) differed at the translation level. Relevant to mention, TE was induced in the majority of the genes (65.6%) in MEC1_RPS15mut vs MEC1_RPS15wt (supplemental Tables 6-7). The overlap between the differentially translated genes in primary CLL cells and the MEC1 cell line was minimal (0.7%) (supplemental Figure 2). The observed differences, may be attributed to the following reasons: (1) MEC1 cells are TP53 aberrant; (2) MEC1_RPS15wt cells overexpress the RPS15 gene compared with primary CLL cells; and (3) MEC1 cells are highly proliferative, thus contrasting most circulating CLL cells and, moreover, are CD5 negative. Hence, results from CLL-derived cell lines, as those recently reported,5 cannot be a priori considered as equivalent to those in primary CLL cells, since they appear dependent on the cell model used.
In order to validate Ribo-seq results, using flow cytometry, we assessed BCL2L11 protein expression in both RPS15mut and RPS15wt primary CLL cells (n = 13 and n = 8 cases, respectively), as it was found to exhibit remarkably reduced TE in the former. BCL2L11 was significantly downregulated in RPS15mut samples (fold difference: 1.3; P < .05), in keeping with the reduced TE revealed by Ribo-seq in RPS15mut vs RPS15wt CLL cases (supplemental Figure 3A). We extended the validation experiments in RPS15mut and RPS15wt MEC1 cell lines, where we assessed the expression of CXCR4 by flow cytometry, found suppressed at the mRNA level in the former, and report concordant decreased CXCR4 protein expression in MEC1_RPS15mut cells (fold difference: 1.95; P = .1) (supplemental Figure 3B).
In conclusion, our data indicate that RPS15 mutations rewire the translation program of CLL cells by reducing their TE, likely due to altering the TE of other ribosomal proteins and regulatory elements, inducing substantial changes in several proteins implicated in CLL pathobiology. As the equation of transcript abundances to protein levels is still under consideration14,15 we argue that the analysis of the translated genome in CLL will shed light into the genes and regulatory pathways implicated in disease phenotypes.
Supplementary Material
The full-text version of this article contains a data supplement.
Acknowledgments
This project was supported in part by TRANSCAN JTC 2014, Horizon 2020 project NEoteRIC (871330), the Swedish Cancer Society, the Swedish Research Council, the Knut and Alice Wallenberg Foundation, the Karolinska Institutet, Karolinska University Hospital, Radiumhemmets Forskningsfonder Stockholm (projects RVO 65269705 and AZV NV19-03-00091), and grant NU21-08-00237 from the Czech Ministry of Health. This work has also been supported by the project ODYSSEAS (Intelligent and Automated Systems for enabling the Design, Simulation and Development of Integrated Processes and Products) implemented under the “Action for the Strategic Development on the Research and Technological Sector”, funded by the operational program “Competitiveness, Entrepreneurship and Innovation” (NSRF 2014-2020) and co-financed by Greece and the European Union, with grant agreement no: MIS 5002462”.
Authorship
Contribution: S.N. designed the research, performed the study and wrote the manuscript; S.L., M.G., G.T. assisted in research; F.P., T.M., N.P., L.M. assisted in data analysis, and interpretation; A.A., N.S., S.P., K.P. provided patient samples; A.M. assisted in data interpretation and revised the manuscript; R.R. and K.S. designed and supervised the research and wrote the manuscript.
Conflict-of-interest disclosure: K.S. has received research support and honoraria from Janssen Pharmaceutica, Gilead Sciences, and AbbVie SA. R.R. has received honoraria from AbbVie, AstraZeneca, Illumina, Janssen, and Roche. The remaining authors declare no competing financial interests.
Correspondence: Kostas Stamatopoulos, Institute of Applied Biosciences, Centre for Research and Technology Hellas, 6th km Charilaou-Thermi Rd, GR57001, Thessaloniki, Greece; e-mail: kostas.stamatopoulos@certh.gr.
References
- 1.Ljungström V, Cortese D, Young E, et al. Whole-exome sequencing in relapsing chronic lymphocytic leukemia: clinical impact of recurrent RPS15 mutations. Blood. 2016;127(8):1007-1016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Landau DA, Tausch E, Taylor-Weiner AN, et al. Mutations driving CLL and their evolution in progression and relapse. Nature. 2015;526(7574):525-530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tausch E, Beck P, Schlenk RF, et al. Prognostic and predictive role of gene mutations in chronic lymphocytic leukemia: results from the pivotal phase III study COMPLEMENT1. Haematologica. 2020;105(10):2440-2447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ljungström V, Rosenquist R.. Not so lost in translation: RPS15 mutations in CLL. Blood. 2018;132(22):2317-2319. [DOI] [PubMed] [Google Scholar]
- 5.Bretones G, Álvarez MG, Arango JR, et al. Altered patterns of global protein synthesis and translational fidelity in RPS15-mutated chronic lymphocytic leukemia. Blood. 2018;132(22):2375-2388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ingolia NT, Ghaemmaghami S, Newman JRS, Weissman JS.. Genome-wide analysis in vivo of translation with nucleotide resolution using ribosome profiling. Science. 2009;324(5924):218-223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ingolia NT. Ribosome profiling: new views of translation, from single codons to genome scale. Nat Rev Genet. 2014;15(3):205-213. [DOI] [PubMed] [Google Scholar]
- 8.Ranheim EA, Cantwell MJ, Kipps TJ.. Expression of CD27 and its ligand, CD70, on chronic lymphocytic leukemia B cells. Blood. 1995;85(12):3556-3565. [PubMed] [Google Scholar]
- 9.Ennas MG, Moore PS, Zucca M, et al. Interleukin-1B (IL1B) and interleukin-6 (IL6) gene polymorphisms are associated with risk of chronic lymphocytic leukaemia. Hematol Oncol. 2008;26(2):98-103. [DOI] [PubMed] [Google Scholar]
- 10.Lozano-Santos C, Martinez-Velasquez J, Fernandez-Cuevas B, et al. Vascular endothelial growth factor A (VEGFA) gene polymorphisms have an impact on survival in a subgroup of indolent patients with chronic lymphocytic leukemia. PLoS One. 2014;9(6):e101063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Herling CD, Klaumünzer M, Rocha CK, et al. Complex karyotypes and KRAS and POT1 mutations impact outcome in CLL after chlorambucil-based chemotherapy or chemoimmunotherapy. Blood. 2016;128(3):395-404. [DOI] [PubMed] [Google Scholar]
- 12.Machulla HKG, Müller LP, Schaaf A, Kujat G, Schönermarck U, Langner J.. Association of chronic lymphocytic leukemia with specific alleles of the HLA-DR4:DR53:DQ8 haplotype in German patients. Int J Cancer. 2001;92(2):203-207. [DOI] [PubMed] [Google Scholar]
- 13.Aalto Y, El-Rifa W, Vilpo L, et al. Distinct gene expression profiling in chronic lymphocytic leukemia with 11q23 deletion. Leukemia. 2001;15(11):1721-1728. [DOI] [PubMed] [Google Scholar]
- 14.Schwanhäusser B, Busse D, Li N, et al. Global quantification of mammalian gene expression control [published correction appears in Nature. 2013;495:126-127]. Nature. 2011;473(7347):337-342. [DOI] [PubMed] [Google Scholar]
- 15.Battle A, Khan Z, Wang SH, et al. Genomic variation. Impact of regulatory variation from RNA to protein. Science. 2015;347(6222):664-667. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.