Abstract
A female patient presented with severe, symptomatic aortic stenosis after distant surgical aortic valve replacement and subsequent transcatheter aortic valve implantation 6 years prior. A 23-mm CoreValve Evolut (Medtronic, Dublin, Ireland) was implanted with excellent symptomatic and hemodynamic results. (Level of Difficulty: Advanced.)
Key Words: aortic stenosis, transcatheter aortic valve implantation, valve disease
Abbreviations and Acronyms: TAVI, transcatheter aortic valve implantation
Graphical abstract
A female patient presented with severe, symptomatic aortic stenosis after distant surgical aortic valve replacement and subsequent transcatheter…
Aortic stenosis is a chronic and progressive disease and the most common valve disorder in Western countries, with a prevalence of 2% in the general population (1). Bioprosthetic valve failure rates are reported to be low, with an average reoperation rate of 5% to 15% over a 10-year follow-up (2). Transcatheter bioprosthetic aortic valve implants have less long-term study, but failure rates are similarly low (3). Valve-in-valve percutaneous treatment is an evolving method for the treatment of degenerated bioprosthetic valves (4). We present a case of a patient managed with an initial surgical valve followed by 2 separate transcatheter valve implantations for bioprosthetic valve stenosis.
Case Description
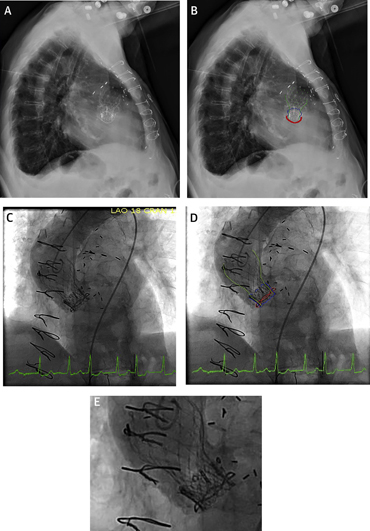
We present a case of a 70-year-old woman. Her cardiac history was significant for bacterial endocarditis 16 years prior, which necessitated a surgical aortic valve replacement; this was accomplished with a 23-mm Hancock bioprosthetic valve (Medtronic, Dublin, Ireland). Six years ago, she developed symptomatic bioprosthetic valve stenosis, and a valve-in-valve transcatheter aortic valve implantation (TAVI) was performed using a 23-mm Sapien valve (Edwards Lifesciences, Irvine, California). The patient now returns with symptoms of chest pain and New York Heart Association functional class IV heart failure symptoms. Transthoracic echocardiography revealed an aortic peak velocity of 5.4 m/s with a mean gradient of 68 mm Hg. A valve-in-valve-in-valve TAVI was performed successfully using a Medtronic 23-mm CoreValve Evolut. The patient was seen 30 days after her latest TAVI and was doing well without symptoms of aortic stenosis and was able to return to work. The post-implantation transaortic velocity was measured at 3 m/s. Figure 1 shows this interesting valve configuration from a routine lateral chest x-ray as well as fluoroscopy imaging from the time of implantation.
Figure 1.
Valve in Valve in Valve
(A) Lateral chest-x-ray, (B) lateral chest x-ray with the aortic bioprosthesis highlighted, (C) fluoroscopy at the time of implantation, (D) fluoroscopy with the aortic bioprosthesis highlighted, and (E) a close-up of the valve configuration. Red outline = Surgical Hancock II aortic valve ring; blue outline = Edwards-Sapien transcatheter aortic valve implantation; green outline = Medtronic CoreValve Evolut transcatheter aortic valve implantation.
Procedures of valve in valve in valve have been reported elsewhere, although most are related to immediate complications of perivalvular leak, aortic insufficiency, or valve recoil. This case is unique in that the Medtronic Hancock valve is not fracturable. We felt the best option was to use a supra-annular self-expanding valve and deploy it as high as possible to maximize the valve area. Pre-procedural planning was undertaken with consideration for risk of coronary artery occlusion with valve deployment, although it is uncommon in routine practice (5). On computed tomography, the sinuses were both large in diameter and tall in height with ample room to accommodate the valve leaflets. The sinus of Valsalva measured 29 mm. The left main coronary artery and the right coronary artery were high above the aortic annulus (11.5 mm and 10 mm, respectively). During the implantation, an aortic root angiogram was performed with the valve 80% deployed, which showed filling of both coronaries. An echocardiogram to assess wall motion was also performed with the valve 80% deployed and was normal.
Footnotes
Dr. Grondahl has reported that he has no relationships relevant to the contents of this paper to disclose. Dr. Dhar is a speaker for Boston Scientific; and is a proctor for Abbott Laboratories and Edwards Lifesciences.
Informed consent was obtained for this case.
References
- 1.Fried L.P., Borhani N.O., Enright P. The Cardiovascular Health Study: design and rationale. Ann Epidemiol. 1991;1:263–276. doi: 10.1016/1047-2797(91)90005-w. [DOI] [PubMed] [Google Scholar]
- 2.Rodriguez-Gabella T., Voisine P., Puri R., Pibarot P., Rodés-Cabau J. Aortic bioprosthetic valve durability: incidence, mechanisms, predictors, and management of surgical and transcatheter valve degeneration. J Am Coll Cardiol. 2017;70:1013–1028. doi: 10.1016/j.jacc.2017.07.715. [DOI] [PubMed] [Google Scholar]
- 3.Babanti M., Costa G., Zappula P. Incidence of long-term structural valve dysfunction and bioprosthetic valve failure after transcatheter aortic valve replacement. J Am Heart Assoc. 2018;7 doi: 10.1161/JAHA.117.008440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Webb J.G., Mack M.J., White J.M. Transcatheter aortic valve implantation within degenerated aortic surgical bioprostheses: PARTNER 2 valve-in-valve registry. J Am Coll Cardiol. 2017;69:2253–2262. doi: 10.1016/j.jacc.2017.02.057. [DOI] [PubMed] [Google Scholar]
- 5.Ribeiro H.B., Webb J.G., Makkar R.R. Predictive factors, management, and clinical outcomes of coronary obstruction following transcatheter aortic valve implantation: insights from a large multicenter registry. J Am Coll Cardiol. 2013;62:1552–1562. doi: 10.1016/j.jacc.2013.07.040. [DOI] [PubMed] [Google Scholar]



