Abstract
Objectives
To evaluate, in a cohort of adults aged ≥80 years, the overlapping effect of clinical severity, comorbidities, cognitive impairment, and frailty, for the in-hospital death risk stratification of COVID-19 older patients since emergency department (ED) admission.
Design
Single-center prospective observational cohort study.
Setting and Participants
The study was conducted in the ED of a teaching hospital that is a referral center for COVID-19 in central Italy. We enrolled all patients with aged ≥80 years old consecutively admitted to the ED between April 2020 and March 2021.
Methods
Clinical variables assessed in the ED were evaluated for the association with all-cause in-hospital death. Evaluated parameters were severity of disease, frailty, comorbidities, cognitive impairment, delirium, and dependency in daily life activities. Cox regression analysis was used to identify independent risk factors for poor outcomes.
Results
A total of 729 patients aged ≥80 years were enrolled [median age 85 years (interquartile range 82-89); 346 were males (47.3%)]. According to the Clinical Frailty Scale, 61 (8.4%) were classified as fit, 417 (57.2%) as vulnerable, and 251 (34.4%) as frail. Severe disease [hazard ratio (HR) 1.87, 95% confidence interval (CI) 1.31-2.59], ≥3 comorbidities (HR 1.54, 95% CI 1.11-2.13), male sex (HR 1.46, 95% CI 1.14-1.87), and frailty (HR 6.93, 95% CI 1.69-28.27) for vulnerable and an overall HR of 12.55 (95% CI 2.96-53.21) for frail were independent risk factors for in-hospital death.
Conclusions and Implications
The ED approach to older patients with COVID-19 should take into account the functional and clinical characteristics of patients being admitted. A sole evaluation based on the clinical severity and the presence of comorbidities does not reflect the complexity of this population. A comprehensive evaluation based on clinical severity, multimorbidity, and frailty could effectively predict the clinical risk of in-hospital death for patients with COVID-19 aged ≥80 years at the time of ED presentation.
Keywords: COVID-19, Clinical Frailty Scale, multimorbidity, dementia, older adults
Since December 2019, the novel coronavirus designated as SARS-CoV-2 has determined the tragic pandemic of the respiratory illness named COVID-19.1 , 2 Vaccination campaigns had started in most countries of the world, however, the number of affected patients and the death toll is still increasing.3 Italy faces one of the worst clusters of COVID-19, and the mortality rate and death toll are particularly high.4
The clinical course of COVID-19 is various, ranging from possible asymptomatic patients to severe progressive pneumonia leading to death.5 Overall, the prevalence of respiratory failure in patients hospitalized with COVID-19 was estimated to be about 19%, with up to 12% of patients requiring mechanical ventilation.1 , 2 , 6 In this context of an overflow of critically ill patients, the emergency department (ED) physician must establish clear and objective criteria to stratify COVID-19 death risk.
Patients ≥80 years old are the most at risk of death for COVID-19.7, 8, 9, 10, 11, 12, 13 Most of the current research focuses on the presence of multiple comorbidities in these populations to explain for the disproportionate death rate that has characterized the clinical course of these patients.1 , 2 , 5, 6, 7, 8, 9, 10, 11, 12, 13 However, it has been argued that these conditions cannot comprehensively predict the extremely poor outcomes observed in older patients with COVID-19.13
Older adults have heterogeneous baseline clinical conditions. Often, chronological age and comorbidities do not truly reflect the overall health status of older patients. To overcome these issues, the frailty syndrome was introduced to include several dimensions of physical fitness and autonomy. Frailty is defined as a condition characterized by a progressive declined physiologic function and diminished strength leading to vulnerability and reduced resilience to stressors that led to an increased risk of adverse outcomes.14 Frailty was found to be an independent predictor for death in hospitalized patients with several clinical conditions as well as COVID-19.15, 16, 17, 18, 19, 20, 21 Furthermore, several other conditions such as cognitive impairment9 , 22 , 23 and occurrence of delirium could play a significant prognostic role for hospitalized older adults, including those with COVID-19.24 , 25
This study aims to evaluate, in a cohort of adults aged ≥80 years, the overlapping effect of clinical severity, comorbidities, cognitive impairment, frailty, and occurrence of delirium for the risk stratification of COVID-19 older patients since ED admission.
Methods
Study Design
This is a single-center, prospective observational cohort study, conducted in the ED of an urban teaching hospital, which is a referral center for COVID-19, in central Italy. The study enrolled all the patients aged ≥80 years consecutively admitted to our ED from April 2020 to March 2021. COVID-19 was diagnosed based on the WHO interim guidance. We included in the analysis only patients with a positive result of real-time reverse transcriptase–polymerase chain reaction assay of nasal and pharyngeal swab specimens.26 Patients who did not receive a complete frailty assessment in the ED and patients who refused to participate in the study were excluded from the analysis.
Study Variables
All patients were assessed in the ED to retrieve the following clinical and demographic data:
-
•
Age, gender
-
•
Overall frailty: assessed using the Clinical Frailty Scale,27 patients were further categorized as fit, for scores 1 to 3 (corresponding to fit and mild vulnerability); vulnerable, for scores 4 to 6 (corresponding to vulnerable or mild frail); and frail, for scores 7 to 9 (corresponding to moderate to severe frailty)
-
•
Presence of cognitive impairment, based on an established dementia diagnosis before SARS-CoV-2 infection
-
•
Dependency in activities of daily living (ADL), based on the clinical status before SARS-CoV-2 infection
-
•
Delirium occurrence: established based on the Richmond Agitation-Sedation Scale28 at 24 hours since ED admission
-
•
Clinical presentation symptoms including fever, dyspnea, cough, diarrhea, abdominal pain, neurologic symptoms (including headache, ageusia/anosmia, and confusion), myalgia/asthenia, and syncope/presyncope
-
•
Physiological parameters, including body temperature, heart rate, respiratory rate, blood pressure, Glasgow Coma Scale, and peripheral oxygen saturation: based on these measures, the National Early Warning Score was calculated for each patient29
-
•
Classification according to WHO guidelines in severe or nonsevere presentations: defined as severe COVID-19 for respiratory distress, respiratory frequency ≥30 times/min, peripheral oxygen saturation at rest ≤92%, or oxygenation index (Pao 2/Fio 2) ≤300 mm Hg; a National Early Warning Score >5 at presentation was also considered a severe disease
-
•
Clinical history and comorbidities: hypertension, severe obesity (defined as body mass index >40), history of coronary artery disease, congestive heart failure, cerebrovascular disease, dementia, diabetes, chronic obstructive pulmonary disease, chronic kidney disease, and malignancy. The overall comorbidity presence was assessed by the Charlson Comorbidity index for each patient.30
-
•
Radiologic findings: based on chest radiographs, patients were categorized as either negative/interstitial involvement or positive for consolidative pneumonia
-
•
A comprehensive laboratory evaluation and a blood gas determination of all patients in the ED
-
•
Time of infection: patients admitted in the first wave of infections in Italy (from April 1 to August 31, 2020) were compared with those infected later (September 1, 2020, to March 30, 2021).
Study Endpoint
The primary study endpoint was all-cause in-hospital death.
Statistical Analysis
Continuous variables were reported as median [interquartile range (IQR)] and are compared at univariate analysis by Mann-Whitney U test or Kruskal-Wallis test in case of 3 or more groups. Categorical variables were reported as absolute number (percentage) and are compared by chi-square test (with Fisher test if appropriate).
Receiver operating characteristic (ROC) curve analysis was used to evaluate the overall performance of the Clinical Frailty Scale in predicting in-hospital death. Follow-up and length of hospital stay were calculated from the time of ED admission to discharge or death. Survival curves were estimated by the Kaplan-Meier methods.
The study variables were assessed for the association to all-cause in-hospital death by a univariate Cox regression analysis. Significant variables at univariate analysis were entered into a multivariate Cox regression model to identify independent risk factors for survival. For a better model fit and hazard estimation, we categorized the continuous variables (National Early Warning Score, Charlson Comorbidity index) into dichotomous parameters (ie, low/high). For each variable, we obtained the optimal dividing cutoff by the Youden index, performing an ROC curve analysis for the association with death. To avoid model redundancy or overfitting, single items composing derived variables (like Charlson Comorbidity index and National Early Warning Score) were excluded from multivariate analysis. Multivariate association of factors to the risk of in-hospital death was expressed as hazard ratios (HRs) and 95% confidence intervals (CIs).
Further multivariate Cox regression models were performed to obtain the adjusted HR for death comparing the patients admitted in the first months of COVID-19 with the later waves of infection. Similarly, we estimated the age-adjusted survival rates for the patients divided into 5-year age groups (80-84, 85-89, 90-94, and ≥95 years old). A 2-sided P ≤.05 was considered significant. Data were analyzed by SPSS, version 25 (IBM, Armonk, NY, USA).
Statement of Ethics
The study was conducted according to the Declaration of Helsinki and its later amendments and was approved by the local institutional review board (IRB 001705520). Each patient gave informed consent to be included in the analysis.
Results
Study Cohort and Baseline Characteristics
Overall, 7742 patients aged ≥80 years were evaluated in the ED in the study period. Among them, 843 had a positive swab for SARS-CoV-2. After excluding patients who did not complete the frailty assessment and those who refused to be enrolled, we included in the study cohort 729 patients.
Enrolled patients had a median age of 85 years (IQR 82-89) and males were 346 (47.3%). According to the Clinical Frailty Scale scores, 61 (8.4%) aged 84 years (IQR 81-85) were classified as fit, 417 (57.2%) aged 84 years (IQR 81-87) were classified as vulnerable, and 251 (34.4%) aged 88 years (IQR 85-92) were classified as frail. Overall, 192 (26.3%) patients had an established cognitive impairment diagnosed, and 400 (54.9%) were dependent for ADL.
Clinical Characteristics at Presentation According to Frailty Group
Clinical presentation was similar across frailty groups. Fit patients reported more fever, cough, diarrhea, and neurologic symptoms. Frail and vulnerable patients were generally referred to the ED by their caregivers, mainly for dyspnea and fever (Table 1 ).
Table 1.
Clinical Characteristics of Enrolled Patients According to Frailty Status
| All Cases (N = 729) | Fit (n = 61) | Vulnerable (n = 417) | Frail (n = 251) |
P Value |
|
|---|---|---|---|---|---|
| Age, y, median (IQR) | 85 (82-89) | 84 (81-85) | 84 (81-87) | 88 (85-92) | <.001 |
| Sex: male | 345 (47.3) | 30 (49.2) | 204 (48.9) | 111 (44.2) | .48 |
| ED presentation symptoms | |||||
| Fever | 494 (67.8) | 54 (88.5) | 278 (66.7) | 162 (64.5) | .001 |
| Dyspnea | 461 (63.2) | 37 (60.7) | 279 (66.9) | 145 (57.8) | .06 |
| Cough | 63 (8.6) | 9 (14.8) | 39 (9.4) | 15 (6.0) | .07 |
| Diarrhea | 21 (2.9) | 6 (9.8) | 10 (2.4) | 5 (2.0) | .003 |
| Abdominal pain | 28 (3.8) | 0 (0) | 20 (4.8) | 8 (3.2) | .15 |
| Neurologic symptoms | 69 (9.5) | 10 (16.4) | 41 (9.8) | 18 (7.2) | .08 |
| Malaise | 49 (6.7) | 3 (4.9) | 31 (7.4) | 15 (6.0) | .65 |
| Syncope/presyncope | 27 (3.7) | 1 (1.6) | 18 (4.3) | 8 (3.2) | .51 |
| Clinical evaluation and physiological parameters | |||||
| Severe COVID-19∗ | 64 (8.8) | 1 (1.6) | 32 (7.7) | 31 (12.4) | .014 |
| SaO2 | 94 (90-96) | 94 (90-96) | 94 (91-96) | 93 (88-95) | .006 |
| Heart rate | 85 (75-95) | 88 (75-97) | 83 (73-91) | 86 (75-99) | .022 |
| Respiratory rate | 24 (20-28) | 22 (14-26) | 24 (20-28) | 25 (19-29) | .18 |
| Maximum BP | 129 (114-145) | 125 (116-144) | 130 (119-148) | 125 (109-140) | .004 |
| Minimum BP | 75 (65-83) | 75 (66-81) | 76 (67-85) | 71 (63-82) | .15 |
| Pao2/Fio2 | 270 (212-319) | 311 (263-386) | 261 (214-321) | 271 (195-314) | .13 |
| NEWS | 6 (4-7) | 5 (4-6) | 5 (4-7) | 6 (4-7.25) | .17 |
| Consolidation at radiography | 454 (62.3) | 42 (68.9) | 267 (64.0) | 145 (57.8) | .15 |
| Delirium | 82 (11.2) | 0 (0) | 44 (10.6) | 38 (15.1) | .003 |
| Laboratory values | |||||
| Neutrophil, cells/mm3 | 7190 (4990-10,100) | 8300 (6237-10,605) | 7510 (5475-10,545) | 6335 (4820-9928) | .14 |
| Lymphocyte, cells/mm3 | 940 (670-1280) | 990 (795-1417) | 950 (670-1286) | 915 (627-1270) | .52 |
| Creatinine, mg/dL | 0.98 (0.76-1.40) | 0.83 (0.63-1.07) | 1.13 (0.80-1.70) | 0.94 (0.71-1.38) | .06 |
| BUN, mg/dL | 25 (19-38) | 16 (12-20) | 24 (16-39) | 27 (20-40) | .001 |
| LDH, IU/L | 283 (219-377) | 238 (203-343) | 309 (233-431) | 263 (203-338) | .04 |
| C-reactive protein, mg/L | 65 (28-132) | 52 (26-86) | 59 (24-140) | 73 (31-132) | .50 |
| Clinical history—comorbidities | |||||
| Dependent in ADL | 400 (54.9) | 0 (0) | 149 (35.7) | 251 (100) | <.001 |
| Hypertension | 318 (43.6) | 30 (49.2) | 194 (46.5) | 94 (37.5) | .048 |
| Severe obesity | 7 (1.0) | 0 (0) | 6 (1.4) | 1 (0.4) | .30 |
| Comorbidities in Charlson Comorbidity Index | |||||
| Dementia | 192 (26.3) | 0 (0) | 49 (11.8) | 143 (57.0) | <.001 |
| History of CAD | 110 (15.1) | 5 (8.2) | 73 (17.5) | 32 (12.7) | .07 |
| Congestive heart failure | 116 (15.9) | 4 (6.6) | 65 (15.6) | 47 (18.7) | .06 |
| Cerebrovascular disease | 40 (5.5) | 1 (1.6) | 15 (3.6) | 24 (9.6) | .002 |
| COPD | 106 (14.5) | 3 (4.9) | 68 (16.3) | 35 (13.9) | .06 |
| Diabetes | 167 (22.9) | 11 (18.0) | 94 (22.5) | 62 (24.7) | .52 |
| Chronic kidney disease | 75 (10.3) | 1 (1.6) | 49 (11.8) | 25 (10.0) | .05 |
| Malignancy | 27 (3.7) | 1 (1.6) | 17 (4.1) | 9 (3.6) | .64 |
| Other in CCI | 24 (3.3) | 0 (0) | 17 (4.1) | 7 (2.8) | .21 |
| CCI | 5 (4-6) | 4 (4-5) | 5 (4-6) | 5 (4-6) | <.001 |
| CCI comorbidities ≥3 | 72 (9.9) | 1 (1.6) | 36 (8.6) | 35 (13.9) | .007 |
ADL, activities of daily living; BUN, blood urea nitrogen; CAD, coronary artery disease; CCI, Charlson Comorbidity Index; COPD, chronic obstructive pulmonary disease; LDH, lactate dehydrogenase; NEWS, National Early Warning Score.
Values are median (IQR) or n (%). Frailty was defined as fit for Clinical Frailty Scale (CFS) scores 1 to 3, vulnerable for CFS scores 4 to 6, and frail for CFS scores 7 to 9.
Severe COVID-19 was defined as respiratory rate ≥30 times/min, Pao2 at rest ≤92%, Pao2/Fio2 ≤300 mm Hg, or NEWS >5.
Physiological parameters were slightly worse in frail patients, and as a result, both the National Early Warning Score and the rate of presentation with severe disease were higher in frailer patients. The rate of pulmonary involvement, reflected by consolidation at chest radiograph was similar among the 3 groups. Interestingly, but not unexpectedly, delirium occurred only in vulnerable and frail patients (Table 1).
Most of the enrolled patients had comorbidities. Frail and vulnerable patients had more comorbidities, as shown by a higher rate of patients having 3 or more major comorbidities. As largely expected, the frailer patients were more dependent in ADL and had a higher rate of cognitive impairment (Table 1).
Factors Associated With In-Hospital Death
In line with several COVID-19 reports, the deceased patients in our cohort were significantly older and were more frequently male (Table 2 ). Although the main symptoms were similar for the deceased and survived groups, physiological parameters at admission were significantly out of range in the deceased group, particularly for lower peripheral oxygen saturation (Table 2). As a result, the National Early Warning Score was higher in the deceased, and the number of patients with severe disease was higher in the deceased group (Table 2).
Table 2.
Study Variables in Survived vs Deceased Patients
| Survived (n = 442) | Deceased (n = 287) | P Value | |
|---|---|---|---|
| Age, y | 85 (82-88) | 86 (83-90) | .001 |
| Sex: male | 194 (43.9) | 151 (52.6) | .021 |
| ED presentation symptoms | |||
| Fever | 320 (72.4) | 174 (60.6) | .001 |
| Dyspnea | 297 (67.2) | 164 (57.1) | .006 |
| Cough | 43 (9.7) | 20 (7.0) | .20 |
| Diarrhea | 16 (3.6) | 5 (1.7) | .14 |
| Abdominal pain | 18 (4.1) | 10 (3.5) | .69 |
| Neurologic symptoms | 47 (10.6) | 22 (7.7) | .18 |
| Malaise | 29 (6.6) | 20 (7.0) | .83 |
| Syncope/presyncope | 18 (4.1) | 9 (3.1) | .51 |
| Clinical evaluation | |||
| Severe COVID-19∗ | 19 (4.3) | 45 (15.7) | <.001 |
| Sao2 | 94 (91-97) | 92 (88-95) | <.001 |
| Heart rate | 83 (74-92) | 85 (77-99) | .020 |
| Respiratory rate | 23 (19-28) | 25 (21-28) | .06 |
| Maximum BP | 130 (119-145) | 125 (110-144) | .004 |
| Minimum BP | 77 (68-85) | 70 (60-81) | .005 |
| Pao2/Fio2 | 291 (228-347) | 238 (181-300) | <.001 |
| NEWS | 5 (4-6) | 6 (5-7) | .030 |
| Consolidation at chest radiography | 281 (63.6) | 173 (60.3) | .37 |
| Delirium | 41 (9.3) | 41 (14.3) | .036 |
| Frailty assessment | |||
| Fit (CFS scores 1-3) | 59 (96.7) | 2 (3.3) | <.001 |
| Vulnerable (CFS scores 4-6) | 289 (69.3) | 128 (30.7) | <.001 |
| Frail (CFS scores 7-9) | 94 (37.5) | 157 (62.5) | <.001 |
| Laboratory values | |||
| Neutrophil, cells/mm3 | 7220 (5020-9850) | 7030 (4897-10,115) | .78 |
| Lymphocyte, cells/mm3 | 1050 (770-1370) | 785 (585-1110) | .002 |
| Creatinine, mg/dL | 0.93 (0.69-1.30) | 1.10 (0.80-1.77) | .050 |
| BUN, mg/dL | 21 (16-30) | 29 (22-42) | <.001 |
| LDH, IU/L | 284 (213-373) | 276 (220-429) | .41 |
| C-reactive protein, mg/L | 48 (24-93) | 98 (51-155) | <.001 |
| Clinical history—comorbidities | |||
| Dependent in ADL | 189 (42.8) | 211 (73.5) | <.001 |
| Hypertension | 203 (45.9) | 115 (40.1) | .12 |
| Severe obesity | 4 (0.9) | 3 (1.0) | .85 |
| Comorbidities included in CCI | |||
| Dementia | 89 (20.1) | 103 (35.9) | <.001 |
| History of CAD | 64 (14.5) | 46 (16.0) | .57 |
| Congestive heart failure | 61 (13.8) | 55 (19.2) | .05 |
| Cerebrovascular disease | 17 (3.8) | 23 (8.0) | .016 |
| COPD | 68 (15.4) | 38 (13.2) | .42 |
| Diabetes | 102 (23.1) | 65 (22.6) | .89 |
| Chronic kidney disease | 37 (8.4) | 38 (13.2) | .034 |
| Malignancy | 14 (3.2) | 13 (4.5) | .31 |
| Other in CCI | 11 (2.5) | 13 (4.5) | .13 |
| Charlson Comorbidity Index | 5 (3-6) | 5 (4-7) | .010 |
| CCI comorbidities ≥3 | 26 (5.9) | 46 (16.0) | <.001 |
ADL, activities of daily living; BP, blood pressure; BUN, blood urea nitrogen; CAD, coronary artery disease; CCI, Charlson Comorbidity Index; CFS, Clinical Frailty Scale; COPD, chronic obstructive pulmonary disease; LDH, lactate dehydrogenase; NEWS, National Early Warning Score.
Values are median (IQR) or n (%). All-cause in-hospital death was considered.
Severe COVID-19 was defined as respiratory rate ≥30 times/min, Pao2 at rest ≤92%, Pao2/Fio2 ≤300 mm Hg, or NEWS >5.
Survival was significantly different for different grades of frailty, with deceased patients mostly classified as vulnerable or frail according to the Clinical Frailty Scale (Table 2). Deceased patients had a higher rate of delirium occurrence, a higher rate of dependency in ADL, cognitive impairment, and had ≥3 comorbidities (Table 2). Overall, the Clinical Frailty Scale had a fair predicting ability for in-hospital poor outcome, with an area under the ROC curve for death of 0.743 (range 0.708-0.779).
Multivariate Analysis for In-Hospital Death
When entered into a multivariate Cox regression analysis, several factors emerged as independent predictors of poor outcomes in our cohort. Among these, frailty was a significant risk factor for death, with the HR for vulnerable patients being 7 times higher than fit ones and a further doubling of the risk for frail patients (Table 3 ). Interestingly, though predictably, once adjusted for baseline covariates and frailty, both dementia and dependency in ADL were not independent risk factors for death. Similarly, the occurrence of delirium was not associated with an increased adjusted HR for death in this cohort (Table 3).
Table 3.
Multivariate Analysis (Cox Regression Model) of Significant Factor Associated With Survival at Univariate Analysis
| Factor | Wald | Hazard Ratio (95% Confidence Interval) | Multivariate P Value |
|---|---|---|---|
| Frailty | |||
| Fit (CFS scores 1-3) | 19.649 | Reference | <.001 |
| Vulnerable (CFS scores 4-6) | 7.277 | 6.93 (1.69-28.27) | .007 |
| Frail (CFS scores 7-9) | 11.774 | 12.55 (2.96-53.21) | .001 |
| Sex: male | 8.869 | 1.46 (1.14-1.87) | .003 |
| Age ≥85 y | 1.291 | 1.16 (0.89-1.51) | .26 |
| Severe COVID-19 at ED admission | 13.967 | 1.87 (1.34-2.59) | <.001 |
| CCI comorbidities ≥3 | 6.676 | 1.54 (1.11-2.13) | .010 |
| Dependent in ADL | 1.333 | 1.24 (0.86-1.80) | .25 |
| Dementia | 0.015 | 1.02 (0.77-1.35) | .90 |
| Delirium in ED | 1.698 | 0.79 (0.56-1.12) | .19 |
ADL, activities of daily living; CCI, Charlson Comorbidity Index; ED, emergency department; NEWS, National Early Warning Score.
Cutoff values for continuous variables were chosen according to receiver operating characteristic (ROC) curve analysis Youden index J. Time was calculated from ED admission to discharge/death.
As expected, the clinical severity at admission significantly increased the overall death risk as well as the presence of ≥3 comorbidities. The analysis also found an increased risk for male sex (Table 3). Overall, the death risk progressively increased for severe disease, comorbidity, and frailty, being almost 100% in patients combining all these factors (Figure 1 ).
Fig. 1.
Number of patients and mortality rate (%) according to the presence of a severe clinical presentation, Charlson comorbidities ≥3, and frailty assessed by the Clinical Frailty Scale (CFS). Frailty was defined as Fit for CFS scores 1 to 3, vulnerable for CFS scores 4 to 6, and frail for CFS scores 7 to 9. Green is for mortality 0% to 33%, yellow for mortality 33% to 66%, and red for mortality >66%.
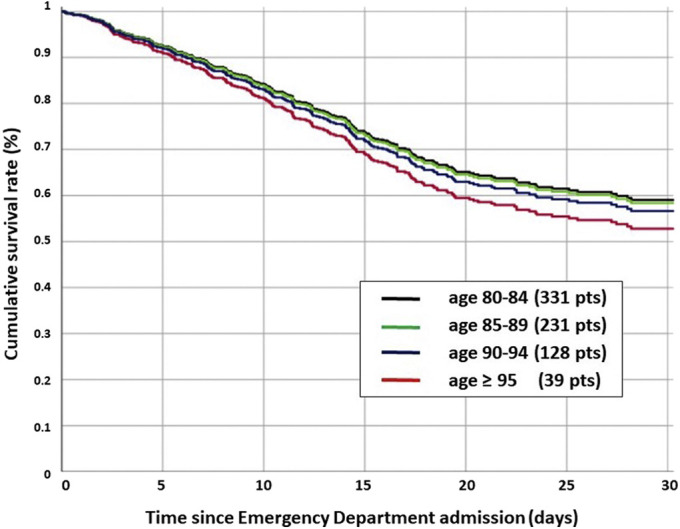
Age-Related Survival Analysis
Dividing the patients into 5-year groups, we observed a progressive increase in the crude death rate for older ages. Overall, 111 of 331 patients (34.4%) died in the 80-84-year age group, 97 of 231 (42.0%) in the 85-89-year age group, 52 of 128 (40.6%) in the 90-94-year age group, and 24 of 39 (61.5%) in the ≥95-year age group (P = .007). However, when the age group was combined with frailty, comorbidity, sex, and disease severity in a Cox multivariate model, the adjusted hazard risk for death did not significantly differ among the groups. Compared with the 80-84-year age group, the hazard ratio for death was 0.99 (IQR 0.69-1.40) for 85-89 years, 1.19 (IQR 0.89-1.57) for 90-94 years, and 1.37 (0.86-2.19) for those aged ≥95 years (Supplementary Figure 1).
Supplementary Fig. 1.

Adjusted age-related survival was obtained by dividing the patients into 5-year groups. Crude death rate was 111 of 331 (34.4%) in the 80-84-year group, 97 of 231 (42.0%) in the 85-89-year group, 52 of 128 (40.6%) for the 90-94-year group, and 24 of 39 (61.5%) in the ≥95-year group (P = .007). Compared with the 80-84-year group, the Hazard for death was 0.99 (0.69-1.40) for 85-89 years, 1.19 (0.89-1.57) for 90-94 years, and 1.37 (0.86-2.19) for those ≥95 years. The frailty, comorbidity, and severity-adjusted hazard risk for death did not significantly differ among the groups.
Death Rate According to the Period of the Pandemic
Considering the 203 patients admitted in the first phase of the pandemic (April to August 2020) and the 526 patients admitted from September 2020 to March 2021, we observed 89 deaths (43.8%) in the first period and 198 (37.6%) deaths in the successive months. Although the crude mortality was lower, the adjusted hazard risk for death was not significantly different [0.79 (IQR 0.61-1.02); P = .072) (Supplementary Figure 2).
Supplementary Fig. 2.
Adjusted period-related survival was obtained for patients admitted from April to August 2020 and patients admitted from September 2020 and March 2021. Overall, 203 patients were admitted in the first phase with 89 deaths (43.8%), and 526 patients were admitted in the second phase with 198 deaths (37.6%). The adjusted hazard risk for death was not significantly different [second wave HR = 0.79 (0.61-1.02); P = .072].
Discussion
The main finding of the present study is that in patients aged ≥80 years, frailty assessment in the ED could accurately recognize patients at increased risk for in-hospital death for COVID-19. The frailty evaluation identifies patients at risk independently from other well-known risk factors for COVID-19, such as clinical severity of the disease, presence of comorbidities, and male sex.1 , 2 , 6, 7, 8, 9, 10
At the same time, the comprehensive frailty assessment includes by definition other known factors that have been associated with poor COVID-19 prognosis in older adults, including dementia, dependency in ADL, and delirium.12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25 Hence, the adjusted HR for these latter factors did not reach statistical significance in our cohort.
An optimal screening tool for frailty in the ED setting should be practical, simple, and accurate. Among the existing clinical score, we adopted the Clinical Frailty Scale, which is already widely used and is particularly efficient for the emergency setting because there are only 5 patient domains that need to be assessed.31 The CSF was already found to be linearly correlated with death in a meta-analysis on a pooled sample of 3817 patients with COVID-19. However, the analysis included patients of different age groups, and the pooled analysis was not fully adjusted for disease severity or comorbidities.18
The concept of frailty is often confused in clinical practice with multimorbidity. However, contrary to general perception, multimorbidity does not necessarily imply the onset of frailty.32 , 33 Nevertheless, these conditions share several aspects, and chronic diseases are often a key component of the frailty status.34 Indeed, both conditions are associated with an increased risk of poor outcomes in hospitalized patients.35
Since the first COVID-19 clinical reports, comorbidities have been identified as the crucial factors to define the risk of poor outcome in COVID-19.1, 2, 3, 4, 5, 6, 7, 8, 9, 10 Several factors have been advocated as the main determinant of risk, including older age, cardiovascular comorbidities, obesity, diabetes, and chronic kidney disease.6, 7, 8, 9, 10 , 19 In the present analysis, the considered comorbidities were those included in the well-established Charlson Comorbidity index. Nevertheless, >95% of comorbidities reported in our cohort consisted of dementia, history of coronary artery disease, congestive heart failure, cerebrovascular disease, diabetes, chronic obstructive pulmonary disease, chronic kidney disease, and malignancy. This finding is consistent with current literature6, 7, 8, 9, 10 , 19 , 36 , 37 and suggests that a simplified comorbidity assessment including these 7 factors could be sufficient for the risk stratification of patients with COVID-19 aged ≥80 years.
Apart from specific diseases and syndromes, neurologic comorbidities and dementia have been identified as specific risk factors for poor prognosis in older patients.9, 10, 11 In the present study, dementia was confirmed to be significantly associated with poor outcomes; however, when the analysis was adjusted for frailty status, it did not emerge as an independent factor for death. This latter finding could help to clarify the role of cognitive impairment for COVID-19 prognosis. Based on a meta-analysis of available data, dementia seems to be associated with an enhanced risk of mortality from COVID-19 infection.38
Several explanations have been advocated to explain this result. First, it was suggested that most of the patients with dementia were old and had several comorbidities, and as such dementia was only a marker of these conditions.38 Furthermore, it has been speculated that older patients with dementia and COVID-19 infection may present with atypical symptoms, such as delirium or isolated functional decline, impeding the early recognition of the disease.39 Other hypotheses included the association of ApoE e4 genotype with dementia and the modulation of pro- and anti-inflammatory phenotypes and the expression of ACE2 receptors.40
Our data suggest that cognitive impairment could be just a marker of increased frailty, and certainly it constitutes one of its major determinants. In this way, a comprehensive frailty assessment includes in the frail group those with cognitive impairment. Hence, it is frailty and not dementia in itself that justifies the increased mortality. This point of view could easily be extended to other medical conditions, and explain the association between dementia and mortality that could be seen even for non–COVID-19 pneumonia-associated mortality.41
Similar to dementia, the dependency in ADL shares some aspects with overall frailty and constitutes one of its main manifestations. Although associated with poor outcomes in COVID-19 older patients,7 , 8 , 15, 16, 17 , 20 , 42 when the analysis was adjusted for frailty assessment, dependency in ADL alone did not result as an independent risk factor for death. Once again, it can be speculated that dependency of ADL constitutes a marker of frailty instead of a risk determinant by itself.
The same explanation could be extended to delirium, which occurs most frequently in frailer patients.43 Disease severity at presentation is an obvious prognostic determinant for patients with COVID-19. Although several aspects could contribute to define the clinical severity of a given patient, the ED determination of physiological parameters could successfully resume the overall status by a single variable.44 , 45 Several tools have been proposed for this purpose.10
In this study, the clinical severity was defined according to WHO guidelines and the National Early Warning Score evaluation, which demonstrated effectiveness in predicting both death and intensive care unit admission in the general population with COVID-19 and in older adults.10 , 44 , 45 Indeed, because respiratory illness is the key determinant of COVID-19 morbidity, a rapid assessment of disease severity could be obtained just by evaluating the respiratory parameters (SaO2, respiratory rate, and Pao 2/Fio 2). The present analysis confirmed that patients defined as clinically severe had a worse prognosis and, not unexpectedly, this was independent of other conditions such as comorbidities and frailty.
The correlation between older age and the risk of death for COVID-19 has been widely recognized. This often led to resources allocation strategies and guidelines that could directly or indirectly result in discrimination based on age.46 , 47 However, most of the current data on the correlation between age and mortality for COVID-19 do not take into account the specific subset of patients aged ≥80 years, which are mostly considered as a single group.48
The covariate-adjusted analysis in the present study demonstrates that the simple increase of chronological age is not an independent predictor of poor outcomes in patients aged ≥80 years. This was true both when stratifying patients by the 85-year cutoff chosen by ROC analysis and when dividing the patients into 5-year subgroups. In both the analyses, we observed a crude mortality rate higher for older cohorts; however, when the analysis was adjusted for disease severity and frailty, the difference was not statistically significant (Supplementary Figure 2). This already emerged in some studies on patients with COVID-199 and was demonstrated also for the non-COVID hospitalized population ≥80 years old.49
The clinical complexity of the geriatric patients and the overlap between multimorbidity, cognitive impairment, dependency in ADL, and increasing age often make difficult the clinical assessment of older patients. This is particularly true in the ED where the already limited time for an effective clinical evaluation is made even more difficult by the wearing of personal protective equipment for COVID-19. The present research suggests that although in general patients with COVID-19 an effective and quick risk stratification could be obtained in the ED by evaluating the clinical severity and by assessing the presence of comorbidities, this could not be sufficient for patients aged ≥80 years. In these latter patients, a further component of clinical evaluation should include the assessment of frailty, which is different from the simple assessment of multimorbidity.50
As a final clue emerging from our investigation, we evaluated the changes in mortality rate between the first phase of the pandemic and the later “waves” of infection. Some authors reported a decreased mortality over time in the geriatric population, particularly during the first wave.51
Although not completely explained, this observation could be ascribed to a general increase in the awareness for the disease, associated with precocious diagnoses, and to a general improvement in the hospital care for COVID-19. However, available data were not corrected for frailty, and thus it could be also speculated that the high mortality rates at the very beginning of the pandemic could be just due to a “harvest” effect on the frailer part of the geriatric population. This latter hypothesis could be supported by the present study. Indeed, although the crude mortality rate was higher in the early phase of the pandemic, the adjusted survival rates were similar when corrected for disease severity, comorbidities, and frailty (Supplementary Figure 1).
Study Limitations
Our research presents some limitations. First, it is conducted in a single institution, which is also a referral center for COVID-19, and for this reason it could not be generalizable to all EDs. Moreover, our ED has a dedicated geriatric unit for the early identification of frail patients, and our comprehensive frailty assessment could be more accurate compared with those of general ED physicians.
Conclusions and Implications
The emergency physician approach to older patients with COVID-19 should take into account the peculiar clinical and functional characteristics of this population and should not be simply conditioned by the chronological age. The common evaluation based on the assessment of clinical severity and presence of comorbidities should be enriched by a further evaluation based on a frailty assessment. The comprehensive evaluation based on severity, multimorbidity, and frailty could effectively predict the clinical risk of in-hospital death for patients with COVID-19 aged ≥80 years after ED admission.
Footnotes
The authors declare no conflicts of interest.
Supplementary Data
References
- 1.Guan W.J., Ni Z.Y., Hu Y. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Huang C., Wang Y., Li X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.World Health Organization WHO Coronavirus Disease (COVID-19) Dashboard. https://covid19.who.int/ Available at:
- 4.Ministero della Salute COVID-19 Situazione in Italia. http://www.salute.gov.it/portale/nuovocoronavirus/ Available at:
- 5.Weiss P., Murdoch D.R. Clinical course and mortality risk of severe COVID-19. Lancet. 2020;395:1014–1015. doi: 10.1016/S0140-6736(20)30633-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Grasselli G., Greco M., Zanella A., COVID-19 Lombardy ICU network Risk factors associated with mortality among patients with COVID-19 in intensive care units in Lombardy, Italy. JAMA Intern Med. 2020;180:1345–1355. doi: 10.1001/jamainternmed.2020.3539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hwang J., Ryu H.S., Kim H.A. Prognostic factors of COVID-19 infection in elderly patients: A multicenter study. J Clin Med. 2020;9:3932. doi: 10.3390/jcm9123932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Becerra-Muñoz V.M., Núñez-Gil I.J., Eid C.M. Clinical profile and predictors of in-hospital mortality among older patients hospitalised for COVID-19. Age Ageing. 2021;50:326–334. doi: 10.1093/ageing/afaa258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Covino M., De Matteis G., Santoro M. Clinical characteristics and prognostic factors in COVID-19 patients aged ≥80 years. Geriatr Gerontol Int. 2020;20:704–708. doi: 10.1111/ggi.13960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Covino M., De Matteis G., Burzo M.L. Predicting in-hospital mortality in COVID-19 older patients with specifically developed scores. J Am Geriatr Soc. 2021;69:37–43. doi: 10.1111/jgs.16956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Blagosklonny M.V. From causes of aging to death from COVID-19. Aging (Albany NY) 2020;12:10004–10021. doi: 10.18632/aging.103493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zuccaro V., Celsa C., Sambo M. Competing-risk analysis of coronavirus disease 2019 in-hospital mortality in a Northern Italian centre from SMAtteo COvid19 REgistry (SMACORE) Sci Rep. 2021;11:1137. doi: 10.1038/s41598-020-80679-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mueller A.L., McNamara M.S., Sinclair D.A. Why does COVID-19 disproportionately affect older people? Aging (Albany NY) 2020;12:9959–9981. doi: 10.18632/aging.103344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vermeiren S., Vella-Azzopardi R., Beckwée D., Gerontopole Brussels Study group Frailty and the prediction of negative health outcomes: A meta-analysis. J Am Med Dir Assoc. 2016;17:1163.e1–1163.e17. doi: 10.1016/j.jamda.2016.09.010. [DOI] [PubMed] [Google Scholar]
- 15.Zhang X.M., Jiao J., Cao J. Frailty as a predictor of mortality among patients with COVID-19: A systematic review and meta-analysis. BMC Geriatr. 2021;21:186. doi: 10.1186/s12877-021-02138-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Aliberti M.G.R., Covinsky K.E., Barreto Garcez F. A fuller picture of COVID-19 prognosis: The added value of vulnerability measures to predict mortality in hospitalised older adults Age. Ageing. 2021;5:32–39. doi: 10.1093/ageing/afaa240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Blomaard L.C., van der Linden C.M.J., van der Bol J.M. Frailty is associated with in- hospital mortality in older hospitalised COVID-19 patients in the Netherlands: The COVID-OLD study. Age Ageing. 2021;50:631–640. doi: 10.1093/ageing/afab018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pranata R., Henrina J., Lim M.A. Clinical frailty scale and mortality in COVID-19: A systematic review and dose-response meta-analysis. Arch Gerontol Geriatr. 2021;93:104324. doi: 10.1016/j.archger.2020.104324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hägg S., Jylhävä J., Wang Y. Age, frailty, and comorbidity as prognostic factors for short-term outcomes in patients with coronavirus disease 2019 in geriatric care. J Am Med Dir Assoc. 2020;21:1555–1559.e2. doi: 10.1016/j.jamda.2020.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Laosa O., Pedraza L., Álvarez-Bustos A. Rapid assessment at hospital admission of mortality risk from COVID-19: The role of functional status. J Am Med Dir Assoc. 2020;21:1798–1802.e2. doi: 10.1016/j.jamda.2020.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.De Smet R., Mellaerts B., Vandewinckele H. Frailty and mortality in hospitalized older adults with COVID-19: Retrospective observational study. J Am Med Dir Assoc. 2020;21:928–932.e1. doi: 10.1016/j.jamda.2020.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Foley N.C., Affoo R.H., Martin R.E. A systematic review and meta-analysis examining pneumonia-associated mortality in dementia. Dement Geriatr Cogn Disord. 2015;39:52–67. doi: 10.1159/000367783. [DOI] [PubMed] [Google Scholar]
- 23.Hariyanto T.I., Putri C., Arisa J. Dementia and outcomes from coronavirus disease 2019 (COVID-19) pneumonia: A systematic review and meta-analysis. Arch Gerontol Geriatr. 2021;93:104299. doi: 10.1016/j.archger.2020.104299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang Z., Pan L., Ni H. Impact of delirium on clinical outcome in critically ill patients: A meta-analysis. Gen Hosp Psychiatry. 2013;35:105–111. doi: 10.1016/j.genhosppsych.2012.11.003. [DOI] [PubMed] [Google Scholar]
- 25.Garcez F.B., Aliberti M.J.R., Poco P.C.E. Delirium and adverse outcomes in hospitalized patients with COVID-19. J Am Geriatr Soc. 2020;68:2440–2446. doi: 10.1111/jgs.16803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.World Health Organization Clinical management of severe acute respiratory infection when novel coronavirus (2019-nCoV) infection is suspected: Interim guidance. 2020. https://www.who.int/docs/default-source/coronaviruse/clinical-management-of-novel-cov.pdf Available at:
- 27.Rockwood K., Song X., MacKnight C. A global clinical measure of fitness and frailty in elderly people. CMAJ. 2005;173:489–495. doi: 10.1503/cmaj.050051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sessler C.N., Gosnell M.S., Grap M.J. The Richmond Agitation-Sedation Scale: Validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166:1338–1344. doi: 10.1164/rccm.2107138. [DOI] [PubMed] [Google Scholar]
- 29.Smith G.B., Prytherch D.R., Meredith P. The ability of the National Early Warning Score (NEWS) to discriminate patients at risk of early cardiac arrest, unanticipated intensive care unit admission, and death. Resuscitation. 2013;84:465–470. doi: 10.1016/j.resuscitation.2012.12.016. [DOI] [PubMed] [Google Scholar]
- 30.Charlson M., Szatrowski T.P., Peterson J., Gold J. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47:1245–1251. doi: 10.1016/0895-4356(94)90129-5. [DOI] [PubMed] [Google Scholar]
- 31.Juma S., Taabazuing M.M., Montero-Odasso M. Clinical frailty scale in an acute medicine unit: A simple tool that predicts length of stay. Can Geriatrics J. 2016;19:34–39. doi: 10.5770/cgj.19.196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Onder G., Cesari M., Maggio M., Palmer K. Defining a care pathway for patients with multimorbidity or frailty. Eur J Intern Med. 2017;38:1–2. doi: 10.1016/j.ejim.2017.01.013. [DOI] [PubMed] [Google Scholar]
- 33.BritainThinks Frailty, language and perceptions: a report prepared by BritainThinks on behalf of Age UK and the British Geriatrics. Society. 2015 https://www.ageuk.org.uk/globalassets/age-uk/documents/reports-and-publications/reports-and-briefings/health--wellbeing/rb_june15_frailty_language_and_perceptions.pdf 2015. [Google Scholar]
- 34.Fried L.P., Tangen C.M., Walston J. Frailty in older adults: Evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56:146–156. doi: 10.1093/gerona/56.3.m146. [DOI] [PubMed] [Google Scholar]
- 35.Kojima G. Frailty defined by FRAIL scale as a predictor of mortality: A systematic review and meta-analysis. J Am Med Dir Assoc. 2018;19:480–483. doi: 10.1016/j.jamda.2018.04.006. [DOI] [PubMed] [Google Scholar]
- 36.Li Y., Ashcroft T., Chung A. Risk factors for poor outcomes in hospitalised COVID-19 patients: A systematic review and meta-analysis. J Glob Health. 2021;11:10001. doi: 10.7189/jogh.11.10001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cheng S., Zhao Y., Wang F. Comorbidities' potential impacts on severe and non-severe patients with COVID-19: A systematic review and meta-analysis. Medicine (Baltimore) 2021;100:e24971. doi: 10.1097/MD.0000000000024971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hariyanto T.I., Putri C., Situmeang R.F.V., Kurniawan A. Dementia is a predictor for mortality outcome from coronavirus disease 2019 (COVID-19) infection. Eur Arch Psychiatry Clin Neurosci. 2021;271:393–395. doi: 10.1007/s00406-020-01205-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Numbers K., Brodaty H. The effects of the COVID-19 pandemic on people with dementia. Nat Rev Neurol. 2021;6:1–2. doi: 10.1038/s41582-020-00450-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kuo C.L., Pilling L.C., Atkins J.L. APOE e4 genotype predicts severe COVID-19 in the UK Biobank Community Cohort. J Gerontol A Biol Sci Med Sci. 2020;75:2231–2232. doi: 10.1093/gerona/glaa131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Manabe T., Fujikura Y., Mizukami K. Pneumonia-associated death in patients with dementia: A systematic review and meta-analysis. PLoS One. 2019;14:e0213825. doi: 10.1371/journal.pone.0213825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Heras E., Garibaldi P., Boix M. COVID-19 mortality risk factors in older people in a long-term care center. Eur Geriatr Med. 2020;27:1–7. doi: 10.1007/s41999-020-00432-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Rebora P., Rozzini R., Bianchetti A., CoViD-19 Lombardia Team Delirium in patients with SARS-CoV-2 infection: a multicenter study. J Am Geriatr Soc. 2021;69:293–299. doi: 10.1111/jgs.16969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kostakis I., Smith G.B., Prytherch D., Portsmouth Academic ConsortIum For Investigating COVID-19 (PACIFIC-19) The performance of the National Early Warning Score and National Early Warning Score 2 in hospitalised patients infected by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Resuscitation. 2021;159:150–157. doi: 10.1016/j.resuscitation.2020.10.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Covino M., Sandroni C., Santoro M. Predicting intensive care unit admission and death for COVID-19 patients in the emergency department using early warning scores. Resuscitation. 2020;156:84–91. doi: 10.1016/j.resuscitation.2020.08.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Savulescu J., Cameron J., Wilkinson D. Equality or utility? Ethics and law of rationing ventilators. Br J Anaesth. 2020;125:10–15. doi: 10.1016/j.bja.2020.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.NICE Guidelines . National Institute for Health and Care Excellence (UK); London: 2020. COVID-19 rapid guideline: Arranging planned care in hospitals and diagnostic services. [PubMed] [Google Scholar]
- 48.Bonanad C., García-Blas S., Tarazona-Santabalbina F. The effect of age on mortality in patients with COVID-19: A meta-analysis with 611,583 subjects. J Am Med Dir Assoc. 2020;21:915–918. doi: 10.1016/j.jamda.2020.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Landi F., Liperoti R., Russo A. Disability, more than multimorbidity, was predictive of mortality among older persons aged 80 years and older. J Clin Epidemiol. 2010;63:752–759. doi: 10.1016/j.jclinepi.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 50.Aliberti M.J.R., Szlejf C., Avelino-Silva V.I., COVID HCFMUSP Study Group COVID-19 is not over and age is not enough: Using frailty for prognostication in hospitalized patients. J Am Geriatr Soc. 2021;69:1116–1127. doi: 10.1111/jgs.17146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Xu H., Garcia-Ptacek S., Annetorp M. Decreased mortality over time during the first wave in patients with COVID-19 in geriatric care: Data from the Stockholm GeroCovid Study. J Am Med Dir Assoc. 2021;22:1565–1573.e4. doi: 10.1016/j.jamda.2021.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]



