Abstract
Inhaled corticosteroids (ICS) suppress eosinophilic airway inflammation in asthma, but patients may not adhere to prescribed use. Mean adherence—averaging total doses taken over prescribed—fails to capture many aspects of adherence. Patients with difficult-to-treat asthma underwent electronic monitoring of ICS, with data collected over 50 days. These were used to calculate entropy (H) a measure of irregular inhaler use over this period, defined in terms of transitional probabilities between different levels of adherence, further partitioned into increasing (Hinc) or decreasing (Hdec) adherence. Mean adherence, time between actuations (Gapmax), and cumulative time- and dose-based variability (area-under-the-curve) were measured. Associations between adherence metrics and 6-month asthma status and attacks were assessed. Only H and Hdec were associated with poor baseline status and 6-month outcomes: H and Hdec correlated negatively with baseline quality of life (H:Spearman rS = − 0·330, p = 0·019, Hdec:rS = − 0·385, p = 0·006) and symptom control (H:rS = − 0·288, p = 0·041, Hdec: rS = − 0·351, p = 0·012). H was associated with subsequent asthma attacks requiring hospitalisation (Wilcoxon Z-statistic = − 2.34, p = 0·019), and Hdec with subsequent asthma attacks of other severities. Significant associations were maintained in multivariable analyses, except when adjusted for blood eosinophils. Entropy analysis may provide insight into adherence behavior, and guide assessment and improvement of adherence in uncontrolled asthma.
Subject terms: Asthma, Therapeutics
Introduction
In asthma, regular inhaled corticosteroid (ICS) controller use suppresses eosinophilic airway inflammation and reduces airway hyperresponsiveness, reducing symptoms and protecting patients from potentially life-threatening attacks1,2. Asthma that remains uncontrolled despite the use of high-dose ICS-based controller is regarded as ‘difficult-to-treat’, and presents a complex clinical challenge3.
Medication adherence describes the extent to which patients use medication as prescribed4,5. In difficult-to-treat asthma, patients frequently deviate from prescribed use6,7. This can be tracked objectively by attaching electronic monitoring devices (EMDs) to a patient’s inhaler, recording the date and time of each actuation8. In future, electronic monitors may be routinely integrated into inhalers during manufacture9.
Clarifying the interplay between poor adherence and adverse outcomes could help improve adherence and enhance patient health. So far, work has focused on time-averaged metrics, typically mean adherence (total doses taken/total doses prescribed) and it has been difficult to demonstrate a relationship between asthma outcomes and mean adherence10,11. This averaged metric fails to capture potentially important variations in medication-taking behaviour, e.g. a mean adherence rate of 50% cannot distinguish between one patient consistently taking half the prescribed dose daily and another taking the full prescribed dose, but for only half the required period. Other metrics do take into account either the interval between doses, or the time above a minimum dose threshold, and some have shown a relationship to attack rates in airways diseases10,12. However, these strategies still only represent summative time-averaged metrics, and do not describe day-to-day deviations from regular prescribed usage.
We designed adherence metrics to capture via EMD the extent to which patients with difficult-to-treat asthma deviate from regular controller usage, by measuring the entropy—irregularity, or disorder—with which daily medication doses are taken. The concept of entropy is derived from information theory where it is used to quantify the ‘information’ in a process. Entropy has been previously applied to respiratory symptoms13, breathing patterns, and lung function14. We examined whether these entropy measures of adherence related to specific patient characteristics or predicted subsequent asthma-related clinical outcomes. For comparison, we also measured conventional mean adherence, time- and dose-based variability (using additional metrics reflecting missed days and incomplete doses respectively), and the duration of gaps in which patients completely forwent medication.
We hypothesised that the degree of irregularity of ICS controller usage may be more relevant in difficult-to-treat asthma, and better predict poor outcomes. Highly disordered medication-taking behaviour may place patients at higher clinical risk and may be associated with poorer outcomes.
Methods
Study participants
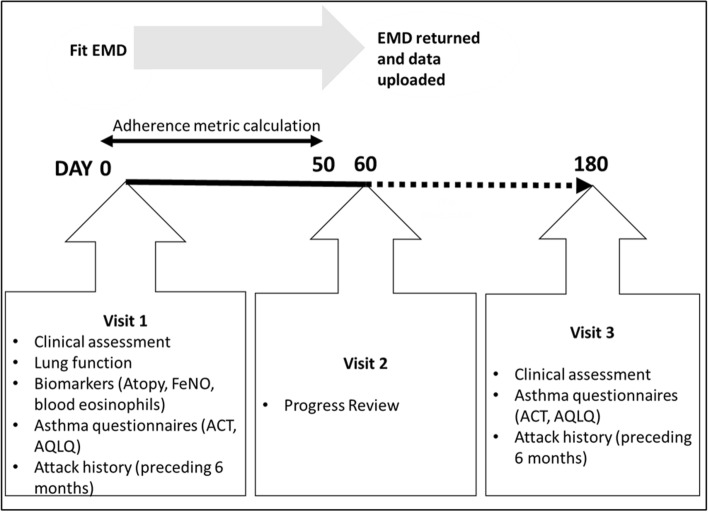
Our tertiary centre receives referrals of adults with difficult-to-treat asthma from specialists in secondary care17. Patients underwent multidisciplinary assessment according to a pre-specified protocol over three visits over six months—previously reported in detail18–22—to confirm asthma diagnosis, address comorbidities, and optimise treatment (Fig. 1).
Figure 1.
Assessment protocol timeline, visits and clinical measures. EMD electronic monitoring device, ACT asthma control test, AQLQ asthma quality of life. (Microsoft Powerpoint, version 2101, https://office.live.com/start/powerpoint.aspx).
Inhaler technique was reviewed and optimised.
Electronic devices
A compatible EMD (Adherium, Auckland, New Zealand) was fitted to the patient’s ICS-containing controller inhaler at visit one (day 0) and EMD-collected ICS data were uploaded at visit two (approximately day 60, Fig. 1). Outcomes were assessed at visit 3 (~ 180 days). EMDs were available for budesonide/formoterol (Turbuhaler and Rapihaler) and fluticasone proprionate/salmeterol (metered dose inhaler and Accuhaler). To allow uninterrupted monitoring, participants were instructed how to move the EMD if they were to change their inhaler. Audiovisual reminders were not activated during the study period but participants could access 7-day EMD data on their devices.
Clinical outcomes
Evaluation included the Asthma Control Test23 and Asthma Quality of Life Questionnaire24 (with permission) at baseline (visit one) and 6 months (visit three) (Fig. 1). Patients were asked to recall the number of attacks in the 6 months prior to visit one, and again, in the 6-month period prior to visit three. Attacks were then confirmed by medical and prescription records where possible. Attacks were also categorised by severity, defined by worsening asthma symptoms requiring: a visit to the general practitioner (least severe); a course (or an increased dose) of oral corticosteroids (OCS; more severe); or hospitalisation (most severe). It was also noted if hospitalization required intensive care admission. Frequency of short acting bronchodilator use over the past four weeks prior to visit one were self-reported and recorded in terms of days and nights per week, as well as number of puffs per day and night.
Patients who completed three visits between August 2015 and February 2018 were eligible for study inclusion. All study protocols and data analysis were approved by the Alfred Health Ethics Committee (285/15) and the Monash University Human Research Ethics Committee (MUHREC). As data were collected as part of routine clinical care and audit, the requirement for signed informed consent was waived by the Alfred Health Ethics Committee. All methods were carried out in accordance with relevant guidelines and regulations as governed by the Australian Health Practitioner Regulation Agency (AHPRA).
Adherence metrics
Metrics were quantified using Python (Python Software Foundation, version 3·6). For standardisation, the first 50 days of available data were extracted for each patient (Fig. 1); this excluded any days with missing data (defined as days when inhaler was not attached—logged by the device as distinct to zero adherence). As the EMD was returned at visit two, adherence data were not available to day 180. Last observation carried forward was not performed to minimise the risk of introducing bias into the adherence metrics, particularly entropy.
Entropy (H), a measure of disorder, was adapted to the adherence data to reflect the various ways in which the patients changed their ICS-taking behavior from day to day. In information theory, a ‘Markov chain’ can be used to describe the sequence of occurrences of certain ‘states’ and the probabilities of transitioning from one state to another given the previous state (e.g. the appearance of specific sequences of letters in a message). H is then used to quantify the complexity of the information, in terms of the transitional probabilities between states, for all possible states observed. Here, we classified adherence into different levels, which are analogous to the different states of a Markov chain, similar to an approach previously applied to respiratory symptoms13. Given an adherence time series x, where x is the dose taken/prescribed dose expressed as a percentage, we obtained a state-based series by mapping each element of xi to the state space S = {1,2,3,4,5,6,7} as follows:
| State, si | Dose range |
|---|---|
| 1 | xi = 0% |
| 2 | 0 < xi ≤ 50% |
| 3 | 50% < xi < 100% |
| 4 | xi = 100% |
| 5 | 100 < xi ≤ 200% |
| 6 | 200 < xi ≤ 300% |
| 7 | xi > 300% |
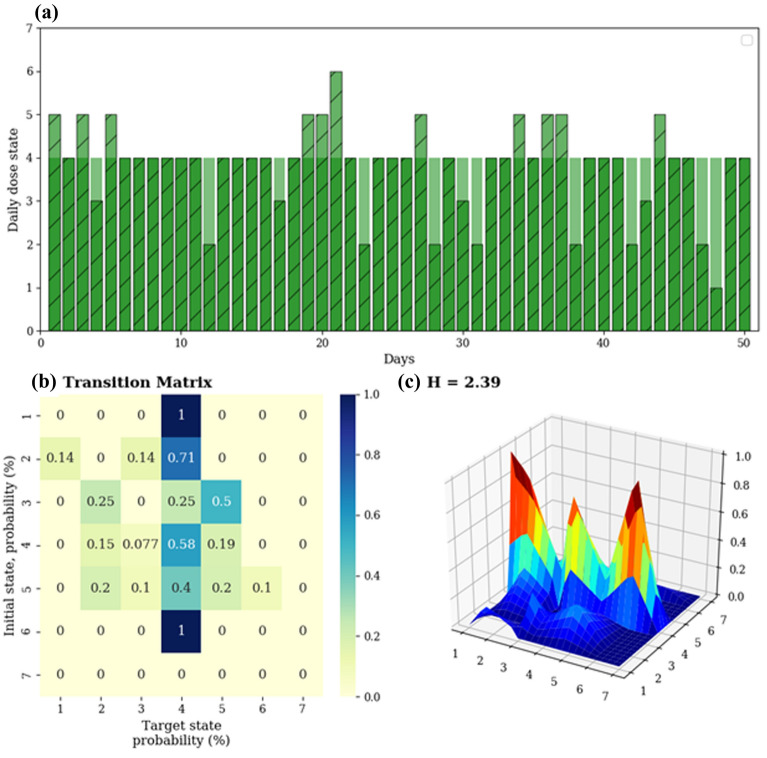
We then computed the 7 × 7 transitional probability matrix, which comprised probabilities Pi,j denoting the probability of transitioning from state j, given an initial state i, for every combination of states i,j. The entropy (H) of the system was determined as , representing the disorder of transitions between daily dose states. An example of H calculation is shown in Fig. 2.
Figure 2.
Calculation of entropy. Panel (a) shows a perfectly-adherent time series (green) in the background comprising 100% of prescribed puffs for 100% of the time, and an example patient time series (blue) overlaid atop the perfectly-adherent series, with instances of under- and over-adherence, both mapped to the state-based series. Panel (b) displays the corresponding transitional probability matrix, while panel (c) allows us to visualise the same matrix (and the “disorder”) in a 3-dimensional graph. The entropy of the transitional probability matrix is then calculated as . (Python Software Foundation, version 3·6 http://www.python.org).
We further partitioned the probability matrix into “increasing” (Hinc) or “decreasing” (Hdec) adherence, by only considering transitions that moved from lower to higher adherence states on the next available day, or vice versa, respectively, i.e. splitting the transitional probabilities along the diagonal of the matrix. Hinc was determined as , where i < j. Similarly, Hdec was defined as , where i > j. Thus, while H represents the day to day changes in adherence levels in general, Hinc represents the different ways in which a patient may increase their adherence level, and conversely Hdec the different ways in which decreases in adherence may occur.
Figure 3 illustrates how different adherence time series with the same conventional mean adherence (PTmean, see below) may vary in Hinc and Hdec.
Figure 3.
Sample adherence time series from 4 different patients over the study period. All patients had ‘good adherence’ as defined by mean adherence PTmean (note not capped at 100% for the purpose of demonstrating variability), however with different increasing and decreasing entropy (Hinc and Hdec) measures, which better reflect the variability in patient inhaler adherence behaviour. Panel (a) demonstrates a patient who took their inhaled controller on average 84% of prescribed doses with calculated entropy 2.24 and increasing entropy 1.61. Panel (b) demonstrates a second patient who took their controller 90% of prescribed doses, with calculated entropy of 2.32 and increasing entropy of 0.92. Panel (c) demonstrates a third patient who took their controller 102.5% of prescribe doses, with calculated entropy of 1.85 and decreasing entropy of 0.95. Panel (d) demonstrates a fourth patient who took their controller 102.5% of prescribed doses, with calculated entropy of 1.86 and decreasing entropy of 0.59. (Python Software Foundation, version 3·6 http://www.python.org).
Conventional mean adherence was described as PTmean, expressed as a percentage of the prescribed number of puffs per day. This was capped at 100%—all higher daily instances were converted to 100% to allow comparison of the mean (PTmean,cap) with other published studies. PTSD and PTCV represented the standard deviation and coefficient of variation of PTmean respectively, based on uncapped data to capture the full variability in adherence.
Area under the curve (AUC) measures were inspired by methods previously described10 to investigate both time- and dose-based adherence variability. In brief, perfect time adherence was first defined as medication taken daily (over the first 50 days), regardless of dosage. The time adherence curve was then defined as the cumulative sum of every day when medication was taken. Thus, perfect time adherence corresponded to a straight line with an area under the curve normalised to 100%. The time-based AUC (T-AUC) for an individual patient was taken as the difference between their time adherence curve and the perfect curve, expressed as a percentage deviation from 100%. In this way, the T-AUC described how consistently the patient took any medication over time. The use of the cumulative sum meant that earlier and/or larger gaps have greater effects on the T-AUC. Similarly, the dose-based area AUC (D-AUC) described the cumulative deviation from the patient’s total prescribed dose over the same 50-day period. We also multiplied the time-based deviation and the dose-based adherence for each day, to construct a composite curve. The Prod-AUC was then calculated as the cumulative deviation from the product of the perfect time x dose curves. This metric thus reflects adherence behavior in terms of both time and doses taken over the given time period.
The Gapmax metric described the maximum length of gaps between days when medication was last taken, regardless of number of puffs within a day, during the 50-day period.
Statistical analysis
Relationships between adherence metrics and clinical characteristics (at baseline) and asthma outcomes (at/over six months) were examined using Spearman rank correlation (rs) for continuous variables, and Wilcoxon rank sum or Kruskal–Wallis tests for comparisons between 2 groups or > 2 groups, respectively. Multivariable regression was performed to confirm if any significant associations between adherence metrics and clinical characteristics and asthma outcomes identified from univariate analyses were still independent predictors after adjusting for the potential confounders of age, sex, baseline eosinophils and baseline lung function. Adjustment for baseline asthma questionnaire scores (AQLQ and ACT) was undertaken where the respective asthma questionnaire score was the outcome.
Statistical analysis was undertaken in R version 3·325. Descriptive statistics are presented as proportions for categorical variables, means and standard deviations for normally distributed continuous variables, or medians and interquartile ranges otherwise.
Results
Participants
Systematic assessment was undertaken by 108 patients. Forty (37%) did not receive a monitoring device: four (3.7%) from physician choice, two (1.9%) declined, two (1.9%) did not have asthma (and were diagnosed with vocal cord dysfunction), and 32 (29%) had inhalers with no compatible EMDs available in Australia. Among 68 patients who underwent inhaler monitoring, 11 (16.2%) devices had less than 50 days of data due to device detachment, and 4 (5.9%) devices malfunctioned (Fig. 4). These patients were excluded from the analysis. Of the 11 devices with missing data, the mean number of days monitored was 37, (SD 69, range 3–220 days).
Figure 4.
Consort diagram demonstrating flow of participants through the study. EMD electronic monitoring device. †Due to EMD detachment. (Microsoft Powerpoint, version 2101, https://office.live.com/start/powerpoint.aspx).
Data were analysed from 53 patients (Table 1).
Table 1.
Baseline patient characteristics.
| Total n = 53 | |
|---|---|
| Demographics | |
| Age years, mean (range, SD) | 51 (19–77, 15) |
| Gender Female, n (%) | 29 (55) |
| Body mass index kg/m2, mean (SD) | 32 (8) |
| Smoking status | |
| Never | 33 (62.3) |
| Ex-smoker | 17 (32.1) |
| Current smoker | 2 (3.8) |
| Asthma medications, n(%) | |
| Short acting muscarinic antagonist | 4 (7.5) |
| Long acting beta agonist | 1 (1.9%) |
| Inhaled corticosteroid | 20 (37.7%) |
| Inhaled corticosteroid/long acting beta agonist combination | 52 (98.1%) |
| Leukotriene receptor antagonist | 10 (18.9%) |
| Long acting muscarinic antagonist | 19 (35.8%) |
| Oral corticosteroids | 10 (18.9%) |
| Theophylline | 2 (3.8%) |
| Omalizumab (anti-IgE monoclonal antibody) | 2 (3.8%) |
| Total number asthma medications, mean, range (SD) | 2.3, 1–6 (1.2) |
| Total daily ICS dose mcg, mean, range (SD) | 969, 200–2000 (475) |
| Asthma severity | |
| Pre-bronchodilator FEV1% predicted, mean (SD) | 64 (21) |
| FEV1/FVC ratio | 61 (16) |
| ACT score at visit one, median (IQR) (23) | 11 (9–16·5) |
| AQLQ score at visit one, mean (SD) (24) | 4 (1·2) |
| On high dose inhaled corticosteroids*, n(%) | 44 (83) |
| Asthma attack rate | |
| Baseline attack number in the six months prior to visit one (mean, SD) | |
| Requiring oral corticosteroids | 2.5 (2) |
| Requiring GP visit | 2.4 (3.7) |
| Requiring ED presentation | 0.8 (1.3) |
| Requiring hospital admission | 0.4 (1) |
| Attack rate in the six months prior to visit three (mean, SD) | |
| Requiring oral corticosteroids | 1.7 (2.9) |
| Requiring GP visit | 2 (5.8) |
| Requiring ED presentation | 0.4 (1) |
| Requiring hospital admission | 0.3 (0.8) |
| Asthma phenotype | |
| FeNO result ppb, mean (range, SD) | 35 (5–137, 32) |
| IgE kU/L, mean (range, SD) | 528 (4–4304, 913) |
| Atopic (positive skin prick test or serum specific IgE to commonly tested aeroallergens), n(%) | 37 (70) |
| Blood eosinophils × 109/L, mean (range, SD) | 0·37 (0–1·18, 0·31) |
FEV1:Forced expiratory volume in one second, FVC forced vital capacity, ACT Asthma Control Test (scores range from 5 (poor asthma control) to 25 (complete asthma control), scores > 19 indicate well controlled asthma), AQLQ asthma quality of life questionnaire (out of 7, high score indicating better quality of life). FeNO fraction of expired nitric oxide, IgE immunoglobulin E, GP General practitioner, ED emergency department.
*Fluticasone propionate equivalent ≥ 500mcg daily.
Adherence metrics and baseline clinical characteristics
Summary statistics of adherence metrics calculated for the first 50 days in all patients are reported in supplemental Table S1. No adherence metric was related to age, sex or baseline lung function on correlation testing (Supplemental Table S2). PTmean,cap correlated with baseline asthma quality of life as measured by AQLQ (Spearman correlation, rS = 0·284, p = 0·046). Large gaps in inhaler use (Gapmax) and lower T-AUC were associated with a greater likelihood of previous intensive care or hospitalisation for an asthma attack in the six-month period prior to visit one (Wilcoxon rank sum test), Gapmax: Z = − 2.068, p = 0·039 and Z = − 2.08, p = 0·037 respectively, T-AUC: Z = − 2.065, p = 0·039 and Z = − 2.042, p = 0·041 respectively).
Regarding entropy, higher H correlated negatively with baseline AQLQ and ACT scores (rS = − 0·330, p = 0·019 and rS = − 0·288, p = 0·041 respectively). Higher Hdec similarly correlated negatively with baseline AQLQ and ACT (rS = -0·385, p = 0·006 and rS = − 0·351, p = 0·012 respectively), and was further associated with higher SABA reliever use in terms of puffs and days per week (rS = 0·318, p = 0·02 and rS = 0·286, p = 0·04 respectively).
The relationships between entropy measures (H and Hdec) and baseline ACT and reliever use remained significant following multivariable regression models adjusting for age, sex, baseline eosinophil count and baseline FEV, while all other measures did not (Table 2).
Table 2.
Multivariable analysis relating adherence metrics to baseline clinical characteristics.
| Baseline measures (adjusted for age, sex, peripheral blood eosinophils and FEV1) | |||
|---|---|---|---|
| Adherence metric | Baseline measure | Coefficient [SE] | p value |
| Mean adherence (PTmean,cap) | AQLQ | 0·19 [0.20] | 0·36 |
| Entropy (H) | ACT | − 0·49 [0·17] | 0·008 |
| AQLQ | − 0·29 [0·15] | 0·065 | |
| Decreasing Entropy (Hdec) | ACT | − 0·51 [0·21] | 0·026 |
| AQLQ | − 0·35 [0·19] | 0·068 | |
| Reliever use, puffs per week | 0·60 [0·28] | 0·04 | |
Adherence metrics and baseline measures reported here are those which showed significant associations in univariate analyses.
AQLQ Asthma quality of life questionnaire, ACT asthma control test.
Adherence metrics and subsequent outcomes at six months
Among all adherence metrics, only measures of entropy, measured in the first 50 days, were associated with asthma outcomes at six months (Supplemental Table S3). Higher H was associated with more asthma attacks requiring hospitalisation over six months prior to visit three (Z = − 2.34, p = 0·019, Fig. 5a).Higher Hdec was associated with more asthma attacks over the six months prior to visit three, requiring a visit to a general practitioner (Z = − 2.43, p = 0·015), oral corticosteroids (Z = − 2.508, p = 0·012), or hospitalisation (Z = − 2.07, p = 0·038, Fig. 5b–d). (All comparisons performed by Wilcoxon Rank Sums Test).
Figure 5.
Entropy metrics (over day 0–50) predict asthma outcomes (over days 0–180). Panel (a): Entropy (H) and attacks requiring hospitalisation. Panels (b–d): Decreasing entropy (Hdec) and attacks requiring general practitioner (GP) visit, oral corticosteroids or hospitalisation respectively. The boxes depict the 25th, 50th, and 75th percentiles while the whiskers depict the minimum and maximum values in the data. The individual data points are also shown as dots. (R version 3·3 https://www.R-project.org/).
For regression analysis, it was only possible to adjust for one confounder at a time, due to the amount of patient data available at 6 months. Relationships between H and asthma attacks requiring hospitalisation remained significant regardless of adjustment for age, sex and baseline FEV1 (Table 3). Similarly, relationships between Hdec and attacks requiring oral corticosteroids or general practitioner visits also remained significant with these adjustments (Table 3) and also when adjusted for baseline number of attacks in the six months prior to visit one requiring oral corticosteroids (0.92, SE 0.38, p = 0.017), GP visit (1.06, SE 0.45, p = 0.018) or hospitalization (1.39, SE 0.68 p = 0.041). (Spearman coefficients reported).
Table 3.
Multivariable analysis relating adherence metrics to 6 month outcomes.
| Six-month outcomes (adjusted for age, sex, peripheral blood eosinophils and FEV1) | |||||
|---|---|---|---|---|---|
| Adherence metric | Outcome measure at 6 months | Coefficient [SE] (p value) when adjusted for | |||
| Age | Sex | Blood Eosinophils | FEV1 | ||
| Entropy (H) | Attacks requiring hospitalisation |
1·35 [0·68] (p = 0·046) |
1·34 [0·68] (p = 0·047) |
2·37 [1·50] (p = 0·113) |
1·4 [0·69] (p = 0·044) |
| Decreasing Entropy (Hdec) | Attacks requiring GP visit |
0·98 [0·49] (p = 0·045) |
0·98 [0·43] (p = 0·021) |
0·91 [0·48] (p = 0·059) |
1·03 [0·43] (p = 0·017) |
| Attacks requiring oral corticosteroids |
0·83 [0·39] (p = 0·034) |
0·91 [0·38] (p = 0·017) |
0·50 [0·46] (p = 0·284) |
0·99 [0·40] (p = 0·014) |
|
| Attacks requiring hospitalisation |
1·11 [0·60] (p = 0·064) |
1·07 [0·59] (p = 0·068) |
0·82 [0·69] (p = 0·231) |
1·10 [0·58] (p = 0·058) |
|
GP general practitioner. Adherence metrics and outcome measures reported here are those which showed significant associations in univariate analyses.
However, after adjustment for baseline eosinophil count, the relationships between H or Hdec and attacks at six months were no longer significant. Further examination showed that H (but not Hdec) was correlated with baseline eosinophil count (rS = 0·352, p = 0·045) suggesting collinearity between baseline eosinophils and H.
Discussion
With increasing emphasis on inhaler adherence monitoring in airways diseases, particularly in the era of biologic therapies for severe asthma, there is a pressing need to identify the optimal metrics with which to measure inhaled controller adherence7,26,27. We showed that disordered controller use in difficult-to-treat asthma—as reflected by entropy analysis—reflected poor baseline asthma control and were associated with subsequent attacks of any severity. This could potentially be mediated through unchecked eosinophilic inflammation.
Entropy measures have previously been used to describe respiratory symptoms and breathing patterns, with higher entropy associated with adverse outcomes13,28. We designed entropy measures (H, Hdec and Hinc) to measure the irregularity of day-to-day dose-taking behaviour, analogous to the original use of H in information theory to quantify the complexity in strings of text29. We used it to describe the diversity in patterns in observed transitions in adherence, choosing to also examine irregularity or diversity in increases and decreases in adherence, as they may be clinically relevant. In considering all (or a subset in the case of Hdec and Hinc) of the elements in the transitional probability matrix, our method of calculating H differs from that of Usemann et al., where entropy was calculated from rows of elements and then averaged13. Nevertheless, H calculated using our method was highly correlated with their method when applied to this dataset (r = 0·912, p < 0·001, data available on request).
To accommodate the original study design, we chose a period of 50 days to maximise participants with sufficient data. This proof-of-concept study justifies validation in larger cohorts and the development of more dynamic measures of entropy, similar to our previous work on peak flows to predict attacks30.
Entropy metrics, specifically in relation to decreasing states (Hdec) over a 50-day period, related to worse asthma control and increased short-acting reliever use at baseline. Notably, greater Hdec also predicted subsequent risk of attacks of any severity, whether requiring general practitioner visit, increase in oral steroids, or hospitalisation (the latter also predicted by H). That Hdec, rather than Hinc, has these relationships suggests that irregular drops in adherence may have more clinical impact than over-adherence. These relationships were no longer significant when adjusting for baseline peripheral eosinophil levels. The correlation between H (though not Hdec) and peripheral eosinophils suggests that higher baseline eosinophil counts may represent previous poor adherence. We hypothesise that the same pattern of behaviour may then have continued during the period of monitoring, with unchecked eosinophilic inflammation subsequently leading to asthma attacks. Previous studies have demonstrated that peripheral eosinophils are an independent predictor for asthma attacks31. Within this small study, baseline blood eosinophils did not predict asthma attacks at 6 months, nor was FeNO related to any adherence measures (supplemental data).
While non-adherence can be intentional due to issues such as mistrust, lack of medication understanding, fixed beliefs and cost, unintentional disordered medication use may also indicate a corresponding degree of chaos in patients’ lives. In asthma, poor family routines accompanied diminished inhaler adherence in children32. In post-myocardial infarct patients, ‘life-chaos’—a highly variable daily routine with an inability to plan and anticipate the future, paralleled poor adherence to cardiac medication15. Similar life-chaos among patients with HIV was associated with increased health care use and missed clinic appointments16. We speculate that the extent of entropy in controller use in difficult-to-treat asthma may also reflect overall life-chaos. Measurement of entropy in inhaled controller use could be used in the clinic setting to target patients particularly with high Hdec for adherence interventions. Such patients may have otherwise been missed if conventional averaged adherence measures were used (Fig. 3). Entropy measures may also prompt the clinician to review the wider social situation of the patient for other indicators of ‘life chaos’.
As anticipated, conventional mean adherence (PTmean,cap) in our study (following adjustment for potential confounders) was not related to baseline asthma status, nor predicted longitudinal outcomes.
Similarly, neither variability in dosage nor timing metric was associated with clinically important outcomes (Supplemental tables S2 and S3). In a previous analysis of a clinical trial in moderate asthma, the use of AUC-based metrics did relate to asthma-related quality of life and lung function by peak flow measurement10. Note that our AUC metrics were based upon, but were not directly comparable to previously-published methods10, which accounted for technique/device errors using a specialised INCA device. Furthermore, our study cohort included consecutive patients drawn from clinical practice.
Our patient population had significant disease with poorly controlled symptoms and high exacerbation rates, despite having previously been assessed by respiratory specialists. We have previously demonstrated that this population still has high non-adherence rates despite specialist intervention. Our results are likely to be representative of difficult-to-treat asthma patients encountered in the ‘real world’, but may not represent less severe patients. The association of entropy with other behaviour that can affect adherence such as mistrust of medication, financial barriers, and not attending a pharmacy access to refill prescriptions would be worth pursuing with future research.
Limitations
Given the complexity of difficult-to-treat asthma, poor disease control may relate to a wide range of disease and patient factors, e.g. biological severity, corticosteroid insensitivity, multimorbidity, poor self-management skills—all addressed in our clinic’s systematic protocol33–35. Notwithstanding the presence of such confounding issues, a significant effect of disordered controller use on risk of asthma attack remained detectable. However, it is possible our single-center study had insufficient statistical power from a reduction in data available due to device incompatibility device malfunction, missing data, small sample size and short duration of data collection, to detect weaker associations. We also relied on patient recollection for asthma attack history which could be inaccurate, although these data were verified when available in medical records. We explored a range of metrics, baseline characteristics, and asthma outcomes, so increasing the likelihood of a chance finding. However, the consistent pattern of results and their persistence following adjustment for confounding both support a true result. We only collected adherence data between visit one and two of our study (most consistently for 50 days), and analysed outcomes at day 180 (visit three). It is possible that adherence would have improved beyond 50 days, however we wished to analyse the impact of the patient’s initial adherence behaviour on future asthma outcomes. It is likely other aspects of adherence behaviour would add to the predictive power of entropy measures; larger validation datasets would enable further exploration as well as control for other possible confounders in the same model. Future studies could also explore the impact of patient socioeconomic status or device polypharmacy on entropy of inhaled controller use as well as examine aspects of ‘life chaos’ more qualitatively.
Conclusions
We showed higher irregularity assessed by entropy in controller use of patients with difficult-to-treat asthma, with effects that appear mediated through eosinophilic inflammation, and were associated with an increased risk of future attacks. Entropy may reflect the ‘life chaos’ experienced by people with difficult-to-treat asthma, a possible target for appropriate intervention. Entropy analysis could guide future approaches to improve adherence and enhance patient health, potentially applicable to other domains of respiratory or other chronic disease.
Supplementary Information
Acknowledgements
We gratefully acknowledge the assistance of Fiona Hore-Lacy for patient coordination, and the Alfred difficult asthma clinic nursing staff (Anna Mackay, Anita Hazard, Eddie Weber) for patient education.
Abbreviations
- ACT
Asthma control test
- AQLQ
Asthma quality of life questionnaire
- AUC
Area under the curve
- EMD
Electronic monitoring device
- FEV1
Forced expiratory volume in one second
- FeNO
Fraction of exhaled nitric oxide
- FVC
Forced vital capacity
- GP
General practitioner
- H
Entropy
- Hdec
Decreasing entropy states
- Hinc
Increasing entropy states
- ICS
Inhaled corticosteroids
- IgE
Immunoglobulin E
- OCS
Oral corticosteroids
- PT mean
Conventional mean adherence
- SABA
Short acting bronchodilator
Author contributions
The study was conceived by J.L., M.H., J.F. and C.T. Clinical data was collected by J.L. and M.H. Data, statistical analysis and entropy calculations were performed by J.H. and C.T. Data interpretation was performed by all authors. Figures were prepared by J.L., J.H. and C.T. Following input by all authors, the first draft was written by M.H., J.L. and C.T. All authors participated in editing and discussion. Study supervision was by M.H. and C.T.
Competing interests
MJA holds investigator-initiated grants for unrelated research from Pfizer and Boehringer-Ingelheim. He has undertaken an unrelated consultancy for and received assistance with conference attendance from Sanofi. He has also received a speaker’s fee from GSK. HKR or her institute has received fees for providing independent medical advice on advisory boards for AstraZeneca, GlaxoSmithKline, Boehringer Ingelheim, GlaxoSmithKline, Novartis and Sanofi/Genzyme, for providing independent medical education at symposia funded by AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Mundipharma, Novartis and Teva, and research grants from AstraZeneca, GlaxoSmithKline and Novartis, all unrelated to this research. MH has received grants-in-aid, speaker fees, and fees for serving on the advisory boards of GlaxoSmithKline, AstraZeneca, Novartis, Teva, Sanofi, and Seqirus, all unrelated to the current manuscript, all paid to his institutional employer Alfred Health. JL has received fees for providing unrelated independent medical advice for GlaxoSmithKline and has provided speaker fees for medical education purposes from Boehringer Ingelheim, GlaxoSmithKline and AstraZeneca. HR reports grants and personal fees from AstraZeneca, grants and personal fees from GlaxoSmithKline, personal fees from Merck, grants and personal fees from Novartis, personal fees from Teva, personal fees from Boehringer Ingelheim, personal fees from Sanofi Genzyme, outside the submitted work. CT is a NHMRC Career Development Fellow (Level 1). JL received support through an Australian Government Research Training Program Scholarship. Both funding sources had no role in study design, collection, analysis, interpretation of data, writing of the report of decision to submit this manuscript for publication. JH and JF have no competing interests to disclose.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Cindy Thamrin and Mark Hew.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-94219-z.
References
- 1.Barnes PJ. Efficacy of inhaled corticosteroids in asthma. J. Allergy Clin. Immunol. 1998;102:531–538. doi: 10.1016/S0091-6749(98)70268-4. [DOI] [PubMed] [Google Scholar]
- 2.Demarche SF, et al. Effectiveness of inhaled corticosteroids in real life on clinical outcomes, sputum cells and systemic inflammation in asthmatics: A retrospective cohort study in a secondary care centre. BMJ Open. 2017;7:e018186. doi: 10.1136/bmjopen-2017-018186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.GINA. Diagnosis and management of difficult-to-treat and severe asthma in adolescent and adult patients. (2019). Available online at https://ginasthma.org/wp-content/uploads/2018/11/GINA-SA-FINAL-wms.pdf.
- 4.Vrijens B, et al. What we mean when we talk about adherence in respiratory medicine. J. Allergy Clin. Immunol. Pract. 2016;4:802–812. doi: 10.1016/j.jaip.2016.05.019. [DOI] [PubMed] [Google Scholar]
- 5.Kini V, Ho PM. Interventions to improve medication adherence. JAMA. 2018;320:2461. doi: 10.1001/jama.2018.19271. [DOI] [PubMed] [Google Scholar]
- 6.Gamble J, Stevenson M, McClean E, Heaney LG. The prevalence of nonadherence in difficult asthma. Am. J. Respir. Crit. Care Med. 2009;180:817–822. doi: 10.1164/rccm.200902-0166oc. [DOI] [PubMed] [Google Scholar]
- 7.Lee J, et al. Non-adherence in the era of severe asthma biologics and thermoplasty. Eur. Respir. J. 2018;2:2. doi: 10.1183/13993003.01836-2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Foster JM, et al. The reliability and patient acceptability of the SmartTrack device: A new electronic monitor and reminder device for metered dose inhalers. J. Asthma. 2012;49:657–662. doi: 10.3109/02770903.2012.684253. [DOI] [PubMed] [Google Scholar]
- 9.Hew M, Reddel HK. Integrated adherence monitoring for inhaler medications. JAMA. 2019;321:1045–1046. doi: 10.1001/jama.2019.1289. [DOI] [PubMed] [Google Scholar]
- 10.Sulaiman I, et al. A method to calculate adherence to inhaled therapy that reflects the changes in clinical features of asthma. Ann. Am. Thorac. Soc. 2016;13:1894–1903. doi: 10.1513/annalsats.201603-222oc. [DOI] [PubMed] [Google Scholar]
- 11.Foster JM, et al. Inhaler reminders improve adherence with controller treatment in primary care patients with asthma. J. Allergy Clin. Immunol. 2014;134:1260–1268.e1263. doi: 10.1016/j.jaci.2014.05.041. [DOI] [PubMed] [Google Scholar]
- 12.Greene G, et al. A novel statistical method for assessing effective adherence to medication and calculating optimal drug dosages. PLoS ONE. 2018;13:e0195663–e0195663. doi: 10.1371/journal.pone.0195663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Usemann J, et al. Dynamics of respiratory symptoms during infancy and associations with wheezing at school age. ERJ Open Res. 2018;4:00037–02018. doi: 10.1183/23120541.00037-2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bravi A, Longtin A, Seely AJE. Review and classification of variability analysis techniques with clinical applications. Biomed. Eng. Online. 2011;10:90. doi: 10.1186/1475-925X-10-90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zullig Leah L, et al. Association between perceived life chaos and medication adherence in a postmyocardial infarction population. Circ. Cardiovasc. Qual. Outcomes. 2013;6:619–625. doi: 10.1161/CIRCOUTCOMES.113.000435. [DOI] [PubMed] [Google Scholar]
- 16.Wong MD, Sarkisian CA, Davis C, Kinsler J, Cunningham WE. The association between life chaos, health care use, and health status among HIV-infected persons. J. Gen. Intern. Med. 2007;22:1286–1291. doi: 10.1007/s11606-007-0265-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Radhakrishna N, et al. Profile of difficult to treat asthma patients referred for systematic assessment. Respir. Med. 2016;117:166–173. doi: 10.1016/j.rmed.2016.06.012. [DOI] [PubMed] [Google Scholar]
- 18.Denton E, et al. Systematic assessment for difficult and severe asthma improves outcomes and halves oral corticosteroid burden independent of monoclonal biologic use. J. Allergy Clin. Immunol. 2020 doi: 10.1016/j.jaip.2019.12.037. [DOI] [PubMed] [Google Scholar]
- 19.Denton E, et al. Factors associated with dysfunctional breathing in patients with difficult to treat asthma. J. Allergy Clin. Immunol. Pract. 2018;7:1471–1476. doi: 10.1016/j.jaip.2018.11.037. [DOI] [PubMed] [Google Scholar]
- 20.Lee J, et al. Paradoxical vocal fold motion in difficult asthma is associated with dysfunctional breathing and preserved lung function. J. Allergy Clin. Immunol. Pract. 2020;8:2256–2262. doi: 10.1016/j.jaip.2020.02.037. [DOI] [PubMed] [Google Scholar]
- 21.Tay TR, et al. A structured approach to specialist-referred difficult asthma patients improves control of comorbidities and enhances asthma outcomes. J. Allergy Clin. Immunol. Pract. 2017 doi: 10.1016/j.jaip.2016.12.030. [DOI] [PubMed] [Google Scholar]
- 22.Denton E, et al. Severe asthma global evaluation (SAGE): An electronic platform for severe asthma. J. Allergy Clin. Immunol. Pract. 2019;7:1440–1449. doi: 10.1016/j.jaip.2019.02.042. [DOI] [PubMed] [Google Scholar]
- 23.Nathan RA, et al. Development of the asthma control test☆A survey for assessing asthma control. J. Allergy Clin. Immunol. 2004;113:59–65. doi: 10.1016/j.jaci.2003.09.008. [DOI] [PubMed] [Google Scholar]
- 24.Juniper EF, et al. Evaluation of impairment of health related quality of life in asthma: Development of a questionnaire for use in clinical trials. Thorax. 1992;47:76–83. doi: 10.1136/thx.47.2.76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.The R Project for Statistical Computing (2017).
- 26.Costello RW, Cushen B. Looking back to go forward: adherence to inhaled therapy before biologic therapy in severe asthma. Eur. Respir. J. 2020;55:2000954. doi: 10.1183/13993003.00954-2020. [DOI] [PubMed] [Google Scholar]
- 27.Ancona G, et al. Adherence to corticosteroids and clinical outcomes in mepolizumab therapy for severe asthma. Eur. Respir. J. 2020;55:1902259. doi: 10.1183/13993003.02259-2019. [DOI] [PubMed] [Google Scholar]
- 28.Engoren M. Approximate entropy of respiratory rate and tidal volume during weaning from mechanical ventilation. Crit. Care Med. 1998;26:2. doi: 10.1097/00003246-199811000-00021. [DOI] [PubMed] [Google Scholar]
- 29.Shannon CE. A mathematical theory of communication. Bell Syst. Tech. J. 1948;27:379–423. doi: 10.1002/j.1538-7305.1948.tb01338.x. [DOI] [Google Scholar]
- 30.Thamrin C, et al. Predicting future risk of asthma exacerbations using individual conditional probabilities. J. Allergy Clin. Immunol. 2011 doi: 10.1016/j.jaci.2011.01.018. [DOI] [PubMed] [Google Scholar]
- 31.Zeiger RS, et al. High blood eosinophil count is a risk factor for future asthma exacerbations in adult persistent asthma. J. Allergy Clin. Immunol. Pract. 2014;2:741–750. doi: 10.1016/j.jaip.2014.06.005. [DOI] [PubMed] [Google Scholar]
- 32.Fiese BH, Wamboldt FS, Anbar RD. Family asthma management routines: Connections to medical adherence and quality of life. J. Pediatr. 2005;146:171–176. doi: 10.1016/j.jpeds.2004.08.083. [DOI] [PubMed] [Google Scholar]
- 33.Hew M, et al. Relative corticosteroid insensitivity of peripheral blood mononuclear cells in severe asthma. Am. J. Respir. Crit. Care Med. 2006;174:134–141. doi: 10.1164/rccm.200512-1930OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hew M, Heaney LG. In: Severe Asthma [ERS Monograph] Chung KF, Israel E, Gibson PG, editors. European Respiratory Society; 2019. pp. 30–48. [Google Scholar]
- 35.Hew M, et al. Systematic assessment of difficult-to-treat asthma: Principles and Perspectives. J. Allergy Clin. Immunol. Pract. 2020;8:2222–2233. doi: 10.1016/j.jaip.2020.02.036. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.