ABSTRACT
Introduction: Despite the availability of new vaccines for SARS-CoV-2, there has been slow uptake and problems with supply in some parts of the world. Hence, there is still a necessity for drugs that can prevent hospitalization of patients and reduce the strain on health care systems. Drugs with sigma affinity potentially provide protection against the most severe symptoms of SARS-COV-2 and could prevent mortality via interactions with the sigma-1 receptor.
Areas covered: This review examines the role of the sigma-1 receptor and autophagy in SARS-CoV-2 infections and how they may be linked. The authors reveal how sigma ligands may reduce the symptoms, complications, and deaths resulting from SARS-CoV-2 and offer insights on those patient cohorts that may benefit most from these drugs.
Expert opinion: Drugs with sigma affinity potentially offer protection against the most severe symptoms of SARS-CoV-2 via interactions with the sigma-1 receptor. Agonists of the sigma-1 receptor may provide protection of the mitochondria, activate mitophagy to remove damaged and leaking mitochondria, prevent ER stress, manage calcium ion transport, and induce autophagy to prevent cell death in response to infection.
KEYWORDS: COVID-19, fluvoxamine, fluoxetine, donepezil, chlorpromazine, SARS-CoV-2, sigma-1, autophagy, critical care, long COVID
1. Introduction
The SARS-CoV-2 (also known as COVID-19) pandemic has become a significant health burden globally, which has affected the social and political landscape and takes a considerable toll on the field of medicine, putting healthcare systems around the world under enormous pressure [1]. The common symptoms observed are cough, fever, and breathlessness [2,3]. In many cases, the symptoms will be mild or non-existent; however, the symptoms will be severe in a significant minority of cases (around 5%). There may be acute lung injury and respiratory distress, heart failure, and death within these severe cases due to cardiac arrest. Despite the constant increase in positive cases and mortality, the development of vaccines gives hope. However, vaccination programs take time and resources to implement, and in the meantime, infection rates continue to rise with the risk of new mutants and variants that could be resistant to the current vaccines. As such, there is still a need for effective treatments that keep patients out of intensive care units and reduce pressure on healthcare systems, especially in those developing nations that have yet to gain access to a vaccine.
Researchers are finding evidence of neurological manifestations of SARS-CoV-2, which may be of prognostic significance [4]. A recent study of 236 379 patients diagnosed with SARS-CoV-2 infection showed that there was a significant increase in risk of developing a neurological disease in the following six months after infection. For patients admitted to an intensive therapy unit, the incidence of subsequent neurological disease diagnosis was over 40%, with more than 25% having their first such diagnosis. These diseases identified included intracranial hemorrhage, ischemic stroke, parkinsonism, dementia, anxiety and mood disorders, and other psychiatric conditions [5]. The neuropathological effects of SARS-CoV-2 infection majorly constitute (i) direct viral injury, (ii) neural or vascular pathology arising secondary to a hyperinflammatory state, (iii) vasculopathy/coagulopathy, (iv) post-infectious autoimmune, and (v) neurological consequence of severe illness (sepsis, hypoxia) [4,6], which can affect both central as well as the peripheral nervous system. However, the evidence so far suggests that the neurological diseases developed after the infection might be because of the reactions inside the body and not by the direct invasion of virus into the nervous system [3,7].
SARS-CoV-2 can impact the CNS, thereby leading to ischemic stroke, manifested by raised inflammatory markers and endothelial dysfunction [7–9]. Encephalopathy, encephalitis, acute demyelinating encephalomyelitis with and without hemorrhage, and transverse myelitis are also reported CNS syndromes [4]. Guillain-Barre syndrome (GBS), brachial plexopathy, polycranial neuropathies, and myopathies are reported syndromes in the CNS [2,6]. However, in the present recovery phase from the pandemic, preference should also be given for post-infectious fatigue by planning strategies for long-term care of recovered patients [10,11].
The development of new drugs is costly and time-consuming; leading chemists estimate that each new chemical entity requires seven or more synthetic steps in their production [12]. Furthermore, these new drugs need to be tested for safety and efficacy before entering the market. Drugs that have already passed critical safety studies could save lives and resources, especially in developing countries without access to the newly developed vaccines. An example of such drugs would be the administration of anti-depressants in patients, which showed a reduced risk of hospitalization, intubation, and death caused by SARS-CoV-2 as reported from early observational studies [13].
The present review addresses the possibility of reducing the hospitalization and mortality rate due to SARS-CoV-2 infection using readily available drugs and speculates on the possible sigma-1 receptor-related mechanisms behind their protective effects.
2. SARS-CoV-2 infection and autophagy
Generally, coronaviruses enter the cells via one of two mechanisms: (1) entry of viral RNA into the cytosol through fusion pore in the plasma membrane (2) endocytosis [14,15]. In the former mechanism, SARS-CoV-2 binds to the cell surface receptor, Angiotensin-converting enzyme 2 (ACE-2) through spike protein (S), resulting in the activation through the protease activity by transmembrane serine protease 2 (TMPRSS2). The released fragment S2 of the proteolytic cleavage binds to the cell membrane leading to the transfer of the viral genome into the cytosol of the host [16]. The presence of TMPRSS2 favors the mechanism, and as the expression of the protease is absent, viral entry is mediated by endocytic machinery, where the spike protein is processed and activated by cathepsin L in the lysosome [17]. Once inside the cell, the viral RNA starts replicating and producing the necessary proteins by controlling the cell replication machinery, subsequently assembled in the endoplasmic reticulum (ER) and Golgi body and released out of the cell through exocytosis.
SARS-CoV-2 hijacks the ER functions to process and pack secretory and structural proteins compromising the hosts’ functions. The buildup of the viral proteins in the ER can result in ER stress, and to maintain cellular homeostasis, unfolded protein response (UPR) is activated [18]. Binding-immunoglobulin protein/78-kDa glucose-regulated protein (BiP/Grp78) acts as a master regulator of the UPR mechanism that keeps the downstream effectors inositol-requiring protein 1α (IRE-1α), protein kinase RNA-like endoplasmic reticulum kinase (PERK) and activating transcription factor 6 (ATF6) in check. ER stress releases Grp78 leading to the activation of the three transmembrane receptors that send signals for reprogramming the cell’s machinery for a particular time to ease the burden [19]. However, if the stress conditions persist for a longer period, it leads to autophagy and apoptosis [20]. HEK293T cells overexpressing SARS-CoV-2 spike protein has been shown to induce the expression of Grp78 with further increase in expression of phosphorylated eIF2α through PERK activation to relieve the stress conditions. Likewise, in a case-control study, a significant increase in Grp78 and PERK without any change in CHOP expression was observed in individuals with SARS-CoV-2 compared to the healthy individuals indicating the activation of the UPR mechanism during infection [21]. Using a bioinformatics approach, potential human microRNA (miRNA) interactions with the SARS-CoV-2’s genome have been predicted [22]. miRNAs are involved in the host’s antiviral cellular defense that could inhibit viral protein translation and regulate immune response [23,24]. Exposure to ER stress-inducing agent tunicamycin has been reported to reduce miRNA expression in human lung epithelial cells indicating ER stress induced by SARS-Cov-2 could also result in the same [25]. The bioinformatics study revealed that reduction of host miR-34a-3p and miR-495-5p could increase the expression of XBP-1s, Grp78, and promote cell survival. Further, interaction with miR-376b-3p could possibly modulate mTOR and autophagic mechanisms spilling on the hypothesis that SARS-CoV-2 could modulate host miRNAs to favorably hijack ER machinery of the infected cells and evade themselves from the host immune system [22].
Autophagy is a dynamic, evolutionarily conserved process among eukaryotic cells involved in the removal of damaged organelles, bacteria, viral particles, and misfolded proteins for maintaining cellular homeostasis. The process begins with the engulfment of damaged organelles by autophagosomes, which in turn fuse with lysosomes, where the contents are degraded with the help of proteolytic enzymes [26]. Studies have pointed out that viral infection can either activate or inhibit autophagy and acts as a double-edged sword depending on the type of virus and host cell to evade the immune system. For example, herpes simplex virus 1, coxsackievirus B, cytomegalovirus [27–29] inhibit autophagy, while human immunodeficiency virus, influenza virus, hepatitis C virus induces autophagy for their replication and propagation [30–32]. Growing evidence suggests that SARS-CoV-2 impairs the autophagic process for their propagation by interfering with several cellular processes. Network profiling and transcriptomic analysis reported that SARS-CoV-2 causes perturbations in autophagy and impedes autophagic flux either by GSK3β upregulation or downregulation of autophagy-related genes SNAP29, lysosome acidification genes contributing to viral escape and propagation [33,34]. Gassen et al. (2020) reported that SARS-CoV-2 infection showed a reduction in glycolysis and protein translation by AMPK, mTORC1 inhibition. Further, downregulation of spermidine, degradation of Beclin-1 via AKT1/SKP2, reduction of autophagic flux, and increase in the level of P62 was also observed, indicating inhibition of autophagy. The subsequent decrease follows the downregulation of Beclin-1 in ATG-14, which controls STX17-SNAP29-VAMP8 SNARE-mediated autophagosome-lysosome fusion indicating disruption of the process.
An increase in autophagosome formation with reduced maturation and autophagic flux in Vero-6 cells upon SARS-CoV-2 infection was also reported by [35], where the authors have demonstrated that expression of the accessory protein ORF3a alone can trigger incomplete autophagy. ORF3a localizes in the late endosome/lysosomes, interacts with homotypic fusion and protein sorting (HOPS) complex (consisting of VPS11, VPS16, VPS18, VPS33, VPS39, and VPS41), and affects the formation of VPS39-containing functional HOPS complex by inducing the accumulation of VPS39 in the late endosomes/lysosomes. HOPS complex is involved in mediating autophagosome-lysosome fusion by interacting with syntaxin 17 (STX17) [36]. ORF3a inhibits the interaction of the HOPS complex with STX17 and prevents STX17-SNAP29-VAMP8 SNARE complex formation and causes a defect in autophagosome maturation [37]. In another study, ORF3a has been shown to induce the Beclin-1-Vps34-Atg14 complex, involved in autophagosome formation, and inhibit the Beclin-1-VPS34-UVRAG complex, which facilitates autophagosome maturation. The inhibition of the later complex prevents autophagosome-lysosome fusion, which helps the viral particles from degradation. Though, UV resistance-associated gene (UVRAG) and the class III phosphatidylinositol 3-kinase VPS34 compete for the same binding site with Beclin-1 to form the respective complexes, Beclin-1-UVRAG complex is more functionally significant and competitive than ATG14, facilitating increased degradation [38]. However, ORF3a decreased UVRAG-Beclin-1 complex formation making ATG14 compete more for the binding site promoting incomplete autophagy [35]. In line with the observation, Yuen et al. (2020) have shown that targeting VPS34 using the inhibitor VPS34-IN1 resulted in significant suppression of SARS-CoV-2 infection in Vero-6 cells rather than using inhibitor SBI0206965 against ULK1 (upstream of VPS34) or 3-methyladenine that inhibits class I PI3-K [39]. These results indicate the possibility that SARS-CoV-2 does not use the complete autophagic machinery; instead, it uses any of the components involved in the process. Preliminary studies using autophagy modulators chloroquine, hydroxychloroquine, clomipramine, mefloquine, ROC-325 have shown to reduce the cytopathic effects of SARS-CoV-2 [40]. Thus, with reports on the link between autophagy and SARS-CoV-2 propagation, treatment strategies using autophagy-modulating drugs can be of potential use in the fight against the virus.
3. Sigma-1 receptor activation and autophagy
In recent years, there have been several studies linking the sigma-1 receptor to the regulation of autophagic activity. The most compelling of which is that knockout of the sigma-1 receptor results in incomplete autophagy, resulting in a buildup of autophagosomes [41]. Furthermore, transfection of sigma-1 negative cells with a full-length copy of the sigma-1 gene (but not truncated versions) restores autophagic function [41]. The evidence for the involvement of the sigma-1 receptor in autophagy is further bolstered by studies showing PRE-084 and ANAVEX2-73 (sigma-1 agonists) can induce the biogenesis of autophagosomes with an increase in UKL-1 phosphorylation [42]. Moreover, many anti-depressant drugs with sigma-1 receptor affinity, such as fluoxetine, fluvoxamine, and imipramine, have been shown to induce an autophagic response in various studies [43–46]. Autophagy dysregulation is known as a key component of several neurodegenerative diseases including Alzheimer’s disease [47] and in recent years the sigma-1 receptor has been shown to be a target that can directly and indirectly affect autophagy as well as other cellular mechanisms that could be involved in neurodegeneration [48]. Recent research has shown that sigma-1 receptor stimulation using fluvoxamine activates autophagy and protects against cardiac fibrosis by inhibiting the IRE1 pathway [49].
4. Sigma-1 receptors and SARS-CoV-2
The sigma-1 receptor is a chaperone protein that is expressed in a vast number of tissues and resides mainly ER or associated mitochondrial membranes (MEM), where it interacts with various receptors [50], ion channels [51–53] and G-proteins [54,55]. The sigma-1 receptor may translocate to the plasma membrane (PM) on activation, dissociating with other proteins from which it forms a complex such as binding immunoglobulin protein (BiP) [56]. The sigma-1 receptor plays a role in managing cellular stress, and its activation prevents ER and mitochondrial stress likely through the shuttling of calcium to the mitochondria [57]. As such sigma-1 receptor agonists tend to have cytoprotective properties, leading to the development of new drugs for the treatment of neurodegenerative diseases such as Alzheimer’s disease [58–60].
A protein interaction mapping study identified 66 druggable human proteins or host factors targeted by 69 compounds (29 FDA-approved drugs, 12 drugs in clinical trials, and 28 pre-clinical compounds). Of particular interest were the sigma receptors (Sigma-1 and TMEM97/Sigma-2), with sigma ligands such as PB28, haloperidol, cloperastine, PD144418, and hydroxychloroquine (Figure 1) having inhibitory activities on viral infectivity in the 10–100 nM range [61]. Knockdown and knockout studies of the sigma-1 receptor showed significant reductions in SARS-CoV-2 replication [62]. Moreover, genome analysis focusing on the sigma-1 receptor locus at chromosome 9p13.3, identified that the minor homozygous TT allele at the single nucleotide polymorphism (SNP) rs17775810 (an SNP associated with loss of function and motor neuron disease) had reduced mortality rates (0%) compared to the Heterozygous TC allele and homozygous CC allele (16.2%), further suggesting a role for the sigma-1 receptor in the viral replication of SARS-CoV-2 [63].
Figure 1.
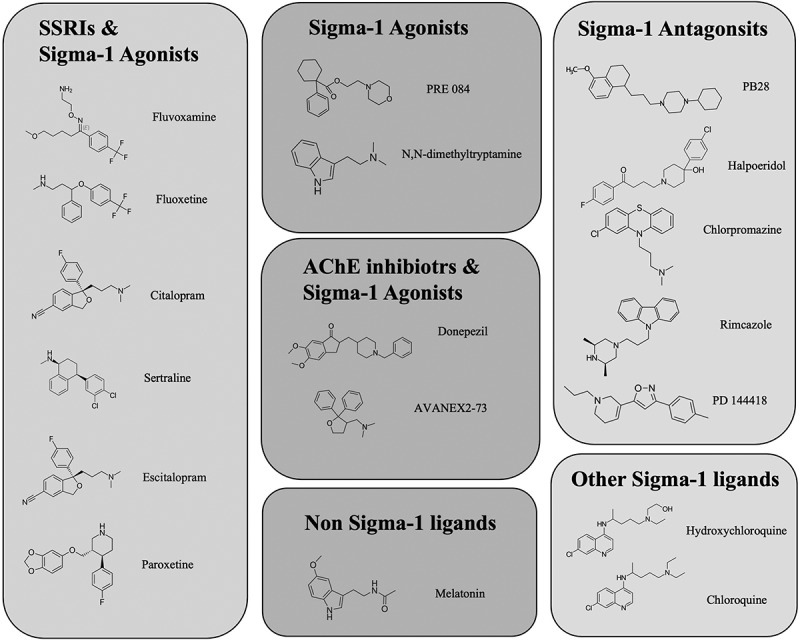
The structures of various compounds that have activities related to the sigma-1 receptor
5. The role of autophagy, the Sigma-1 receptor, and its ligands, in other pandemics
Autophagy plays a crucial role in the viral evasion of range of virus from host immune system. Studies show that genetic or pharmacological inhibition of S-phase kinase-associated protein 2 (SKP2) decreases BECN1 ubiquitination and enhances autophagic flux to reduce the viral replication of MERS-CoV [64]. Hepatitis C virus (HCV) can directly activate autophagy by inducing ER stress [65] or by the interaction, overexpression of the viral proteins (p7 ion channel protein, NS5A) with Beclin-1 [66,67]. Dreuex et al. (2009) has reported that autophagic machinery is not needed for the entry or secretion of HCV viral particles, instead require for replicon replication and life cycle [68]. In the case of influenza A virus (H1N1), binding of viral hemagglutinin glycoprotein to heat shock protein 90AA1, located on the host cell membrane inhibits Akt/mTOR pathway to induce autophagy [69]. The viral proteins of Zika virus (NS4A, NS4B) together suppress Akt/mTOR pathway and promote autophagy, which could be noted from the increase in LC3-II level [70]. Sigma-1 receptor activation by small molecules have been reported to induce autophagy [42]. The role of sigma-1 receptor in other viral infections have not been well studied and reported except for a report showing the regulation of early steps of hepatitis C viral (HCV) RNA replication by sigma-1 receptor [71], and that sigma-1 receptor agonism with cocaine increases the expression of HIV-1 expression in microglial cells [72]. Furthermore, using a sigma-1 receptor antagonist prior to cocaine treatment, reduced the cathepsin B expression and reduced apoptosis signaling in HIV-1 infected macrophages [73].
Sigma-1 receptor ligands have often been tested against various viral infections. For instance, chloroquine shows activity against a number of viruses. Chloroquine administration significantly attenuated the vertical transmission of chronic Zika virus infection in interferon signaling-competent pregnant SJL mice, and attenuated mortality upon acute ZIKV infection in AG129 mice with lower side effects [74]. Hydroxychloroquine inhibits the protease activity of NS2B-NS3, which helps in the hydrolysis and maturation of Zika virus and decreases the infection in placental cells [75]. Derivatives of chloroquine could inhibit the Zika virus replication in cervical cells [76]. Chloroquine being weak bases can accumulate within the acidic organelles such as endosomes, Golgi vesicles, lysosomes, on which the virus depends for entry, replication, and maturation [77]. A randomized, triple-blind, placebo-controlled pilot trial reports that treatment with chloroquine reduces the HCV viral RNA levels in non-responders to other viral treatments [78]. Chloroquine treatment to Huh-7 cells transfected with HCV replicon, inhibited viral replication by targeting virus-associated autophagy [79]. Likewise, Chlorpromazine and Trifluoperazine strongly inhibited MERS-CoV and SARS-CoV infection with EC50 values of 5.76 and 12.9 μM, respectively, in Vero E6 cell lines [80]. Treatment with chlorpromazine significantly inhibited the clathrin-dependent entry of SARS-CoV in caveolin-1-negative HepG2 cells [81] and moderately inhibited MERS-CoV replication and cell–cell fusion [82]. Pretreatment of MDCK cells with chlorpromazine inhibits clathrin-mediated entry of H1N1 virus [83]. Fluoxetine inhibits Coxsackie virus replication by binding to the non-structural viral protein 2 C in a stereospecific manner [84,85]. Given the role of sigma-1 receptor in viral entry, replication, maturation and modulation of autophagy inside the host cells, in-depth study on the range of viral infections in relation to sigma-1 receptor would provide better insight and open more possible mode of action of the drugs used.
6. Sigma receptor drugs and SARS-CoV-2
6.1. Fluvoxamine
Fluvoxamine (Figure 1), sold under the brand name Luvox is a selective serotonin reuptake inhibitor, a known anti-depressant, which can also activate sigma-1 receptors (Ki 36 nM) [60] at the ER, thereby decreasing cytokine production [86]. To date, there have been two double-blind, randomized placebo-controlled studies into the protective effects of fluvoxamine against hospitalization as a result of SARS-CoV-2 infection, and there are more extensive studies currently underway around the world. Of the completed studies, one randomized, double-blind clinical trial with 152 subjects reported that fluvoxamine decreased clinical progression, defined as hypoxia coupled with either shortness of breath or hospitalization, from 8% with observation alone to 0% with fluvoxamine at up to 300 mg daily [87]. In a second study with 65 patients taking fluvoxamine and 48 patients without, the incidence of hospitalization was 0% and 12.5%, respectively, and at 14 days, the persistence of residual symptoms was at 0% and 60% for fluvoxamine and no treatment, respectively [88]. While both these studies are small, and the second does not include any placebo (only untreated patients), they both show very promising initial data suggesting that fluvoxamine can prevent the required hospitalization, intubation, and death. Furthermore, it appears that fluvoxamine may even prevent the long-term symptoms (past 2 weeks). However, we must wait for larger randomized placebo-controlled trials to be completed before recommending its use as a SARS-CoV-2 treatment.
The replication of the coronavirus is associated with the ER [89–91] and has been shown to induce ER stress and activate the unfolded protein response [92,93]. Therefore, as a sigma-1 receptor agonist, fluvoxamine can reduce cellular stress by reducing ER stress and cytokine secretion without inhibiting conventional inflammatory pathways. This cytoprotective activity may help prevent cardiac injury due to cytokine storm syndrome brought on by SARS-CoV-2 infection [94,95]. The sigma-1 receptor has been shown to modulate IRE-1α activity, reduce ER stress, and studies in rodents have shown that sigma-1 receptor agonists are cardioprotective and enhance survival in pre-clinical models of sepsis [86]. IRE-1 is essential to the induction of autophagy resulting from coronavirus infection [92]. Therefore, IRE-1 inhibition by sigma-1 receptor activation may prevent SARS-CoV-2 from hijacking the autophagy pathway as part of its replication. Fluvoxamine has also been shown to increase the bioavailability of the hormone melatonin [96,97], which itself may have a role to play in SARS-CoV-2 infection (discussed below) [98–101].
The SARS-CoV-2 virus hijacks the mitochondria as part of its replication process and evades detection [102]. This results in mitochondrial depolarization and the release of mitochondrial DNA into the cytoplasm along with other mitochondrial components such as cytochrome-C and calcium ions. The release of these components triggers apoptosis and cell death [103]. This is a particular problem in lung and heart tissue, as damage here can have severe consequences for the infected patient. Stimulation of the sigma-1 receptor with fluvoxamine has been shown to restore mitochondrial calcium mobilization and rescue ATP production in cardiomyocytes [104] and has other beneficial effects (including antioxidant effects) on heart tissues [105,106]. Furthermore, sigma-1 KO mice appear to have cardiac dysfunction associated with mitochondria energy dynamics [107].
6.2. Fluoxetine
Fluoxetine is a selective serotonin reuptake inhibitor (SSRI) anti-depressant and sigma-1 receptor agonist (Ki 214–240 nM) [108] (sold under the brand name Prozac) that has several immunomodulatory effects [109] as well as being an inducer of autophagy [110]. In vitro studies of fluoxetine have shown it to inhibit SARS-CoV-2, but not other viruses such as the rabies virus, human respiratory syncytial virus replication, the human herpesvirus-8, or Herpes simplex virus type 1. This suggests that fluoxetine acts specifically to inhibit SARS-CoV-2 rather than activating or inhibiting a more generalized pathway. The effective dose is reported to be around 1–50 µM [111,112], with concentrations below 50 µM being nontoxic in cell culture models of SARS-CoV-2 infection [112].
6.3. Donepezil
Donepezil is a neuroprotective agent used in the treatment of Alzheimer’s disease. It is a sigma-1 receptor agonist as well as having anti-acetylcholinesterase activity. In rat studies, donepezil has been shown to dose-dependently occupy sigma-1R sites in rat brains [113]. PET studies in humans using [11C]SA4503 showed that oral administration of donepezil resulted in 60–75% occupancy of sigma-1 receptor sites in the brain [114]. Donepezil has been shown to induce neuroprotective effects in retinal ganglion cells by promoting neurite outgrowth and inducing autophagic flux [115].
Furthermore, donepezil has also reported activation of AMPK, indicating it can induce autophagy by mTOR-independent mechanism [116]. Moreover, multiple studies have investigated the beneficial effects of donepezil on mitophagy and autophagy concerning cardiac ischemia and reperfusion injury [117,118]. Taken together, all this evidence for donepezil’s potential sigma-1 agonist activity, cytoprotective properties, and auto/mitophagic control could be of benefit when treating patients infected with SARS-CoV-2. Further studies will be required to ascertain whether there is indeed any potential benefit from donepezil treatment.
6.4. Melatonin
Melatonin (MN) has a structure similar to that of the putative sigma-1 receptor agonist N, N-dimethyltryptamine (DMT) (Figure 1). Although similar in structure to DMT, MN is an amide (unlike DMT, which is an amine), and there is no current evidence that MN binds to or activates the sigma-1 receptor. However, studies with the sigma-1 receptor agonist in rats have shown that (+)-N-allylnormetazocine can potentiate MN production at night (but not during the day) [119]. MN is a hormone released by the pineal gland at night and has primarily been associated with the regulation of sleep cycles. Fluvoxamine has been shown to increase the bioavailability of the hormone melatonin [96,97], and there are also ongoing studies into the supplementation of melatonin and SARS-CoV-2 infection. This would appear to correlate with the fact that aged patients have reduced melatonin levels and are more vulnerable to SARS-CoV-2-related death. Furthermore, melatonin acts as an antioxidant and an anti-inflammatory agent. There is much evidence that the mitochondria can synthesize melatonin in tissues throughout the body [120,121], where it can act to mitigate oxidative stress and act as a target for mitochondrial autophagy (mitophagy). As discussed above, SARS-CoV-2 causes mitochondrial depolarization, cytochrome-C release, and oxidative stress. Dysregulation of mitophagy could result in changes in melatonin production and further oxidative stress in the cells as the damaged mitochondria are not removed.
6.5. Chloroquine and hydroxychloroquine
Chloroquine and hydroxychloroquine are anti-malarial medications and are sometimes used to treat autoimmune diseases with moderate affinities for the sigma-1 receptor. Chloroquine has been shown previously to provide cytoprotective properties in a sigma-1 receptor-dependent fashion at concentrations below that at which it inhibits autophagy [122], making it a potential candidate as a SARS-CoV-2 treatment. In the initial stages of the SARS-CoV-2 outbreak, there was a scramble to find treatments to slow or prevent symptoms of infection. Hydroxychloroquine was initially recommended by various health authorities as treatments for SARS-CoV-2 infection (including India, China, Korea, and Italy), as in vitro data as well as some small uncontrolled studies appear to suggest its efficacy in preventing SARS-CoV-2 infection plus it has a tolerable safety profile [123]. Hydroxychloroquine has numerous and potentially life-threatening side effects, including retinopathy, hypoglycemia, life-threatening arrhythmia, and cardiomyopathy. To reduce the risk of sudden cardiac death, it was prescribed along with azithromycin [124]. Hydroxychloroquine was even endorsed by the then President of the United States of America, Donald J. Trump, in March 2020, leading to high demand for the drugs [125]. However, as early as June 2020, clinical studies showed little benefit to hospitalized patients treated with hydroxychloroquine [126–129], and even more alarming was a study that indicated that hydroxychloroquine could increase the risk of death in SARS-CoV-2 hospitalized patients [130]. Subsequently, there were also retractions of published studies due to data irregularities and the validity of the studies supporting hydroxychloroquine use for the treatment of SARS-CoV-2 hospitalized patients [131]. Subsequently, due to safety concerns and lack of efficacy, the World Health Organization (WHO) and the National Institutes of Health (NIH) have recommended against hydroxychloroquine to treat SARS-CoV-2 infection, and their clinical trials stopped.
6.6. PB28
PB28 has an affinity for both the sigma-1 receptor and sigma-2/TMEM97 (0.38 and 0.68 nM, respectively) [132], acting as an antagonist of the sigma-1 and agonist of the sigma-2. PB28 has been identified as a drug that can lessen the severity of SARS-CoV-2 infection [61]. PB28 has been reviewed in much detail regarding its role in preventing the severity of SARS-CoV-2 infection [133]. One compelling hypothesis that could explain PB28’s SARS-CoV-2 protective properties involves the InsP3 receptor and the SARS-CoV-2 NSP6 protein. The NSP6 protein locates at the ER and modulates autophagy [134], favoring further infection by preventing the delivery of viral components to the lysosome by the autophagosome. The binding of PB28 to the Sigma-InsP3 receptor complex results in modulation of NSP6 activity at the ER. PB28 could induce NSP6 binding while also inducing oxidative stress via lysosomal leakage, which in turn activates cytoprotective autophagosome activation [135].
6.7. Haloperidol
Haloperidol is an antipsychotic drug with an affinity for both the sigma-1 receptor and sigma-2/TMEM97. It is a sigma-1 receptor antagonist. Gordon et al., has identified haloperidol as a drug that could limit the severity of SARS-CoV-2 infection, inhibiting SARS-CoV-2 with Ki of 2–12 nM [61]. Furthermore, docking analysis of haloperidol with the NSP6 protein shows haloperidol binds with an affinity score of −7.7 kcal/mol, which predicts a KD in the nano to the micromolar range [136]. However, it should be noted that an initial observational clinical study has indicated that haloperidol treatment does not change the associated risk of intubation or death compared to patients not treated with haloperidol, nor does it affect time to discharge [137].
6.8. Chlorpromazine
Chlorpromazine is a neuroleptic drug with an affinity for the sigma-1 receptor and is a sigma-1 receptor antagonist [138]. Chlorpromazine has been shown in monkey and human cells (Vero and A549) to prevent SARS-CoV-2 infection, with IC50s of 8.2 µM and 11.3 8.2 µM, respectively [139]. Once again, as with haloperidol, observational studies of patients admitted to hospital with SARS-CoV-2 infection indicated that patients who had taken chlorpromazine compared to patients who had not shown any association with chlorpromazine treatment and reduced death from SARS-CoV-2 infection [139]. However, these observational studies do have their limitations, and very few SARS-CoV-2 patients have taken chlorpromazine compared to those who have not.
7. The role of the Sigma-1 receptor in neurological manifestations and post-infection related neurological disease
7.1. Stroke
SARS-CoV-2 is an independent risk factor for acute ischemic stroke, with studies showing a statistically significant increase in ischemic stroke cases among patients with SARS-CoV-2 [140] and systematic reviews showing of the clinical data showing the proportions of patients with SARS-CoV-2 and stroke was almost 2% and the risk of death while in hospital was approximately 35% and patients over the age of 70 years being most at risk [141]. SARS-CoV-2 infected patients were statistically more likely than patients with influenza or other respiratory infection to suffer a first-time stroke with odds ratios of 1.7 for each. Furthermore, patients who were admitted to the ITU had a greater risk of first-time stroke with an odds ratio of 3.5 compared to those who did not undergo ITU treatment [5]. The sigma-1 receptor has been shown to be involved in protective mechanisms that are beneficial during the recovery from ischemic stroke. Studies have shown that the activation of the sigma-1 receptor can modulate inflammatory responses involved in cerebral ischemia [142]. Treatment with sigma-1 receptor agonist dimemorfan at the onset of reperfusion reduced the expression of proinflammatory cytokines MCP-1 and IL-1β via inhibition of the NF-κB pathway [143]. Furthermore, treatment with PRE-084 after the embolic stroke onset (3 and 24 hours) reduced the infarct volume significantly, inhibited proinflammatory cytokines, promoted anti-inflammatory cytokines (such as IL-10 and IL-4) [144]. Activation of the Nrf-2 antioxidant pathway another mechanism that benefits patients suffering with ischemic stroke [145], and activating it via the sigma-1 receptor using a sigma-1 receptor agonist such as fluvoxamine or cutamesine (SA4503) [146] could aid in stroke recovery as well as preventing further SARS-CoV-2 symptoms. Interestingly, the use of a sigma-1 receptor antagonist S1RA has also been shown to reduce damage caused by stroke and ameliorate post-stroke neurological deficits [147].
7.2. Encephalopathy
Encephalopathy and encephalitis are complications of SARS-CoV-2 infection, and those who suffer from encephalopathy or encephalitis usually critically ill with a poor prognosis. Encephalopathy is characterized by diffuse inflammation in the brain causing acute clinical symptoms including fever, headache, seizure, focal neurological deficits, and altered consciousness (which may manifest as mental confusion, drowsiness, or a deep coma) [148]. Patients with SARS-CoV-2 compared to patients with influenza or other respiratory infection had statistically significantly increased odds ratios for developing encephalitis of 1.8 and 2.3, respectively. There is little evidence so far that shows the sigma-1 receptor can directly treat encephalopathies but given the sigma-1 receptors ability to reduce oxidative stress and activate many anti-inflammatory pathways there is a possibility that targeting the sigma-1 receptor may be beneficial, especially if using sigma-1 ligands to treat SARS-CoV-2 infection before the infection crosses the blood brain barrier (BBB) and leads to encephalopathy.
7.3. Dementia
Dementia has been shown to be one of the neurological sequelae that result from SARS-CoV-2 infection with the odds ratios vs influenza infection and other respiratory infections being 2.33 and 1.71 respectively [5]. SARS-CoV-2 can enter the CNS in various ways [149], possibly by infected lymphocytes crossing the BBB [150] during this BBB disruption or via the infection of the olfactory and enteric neurons [151]. As people age there is a loss of integrity of the BBB, and thus making older people more susceptible to SARS-CoV-2 neuroinvasion [152]. There is a growing body of evidence that SARS-CoV-2 infection can increase the risk of developing Alzheimer’s disease. It is thought that the infection with SARS-CoV-2 induces neuroinflammation, via the activation of microglia and astrocytes, which produce the proinflammatory cytokines IL-1β, IL-6, IL-12, TNF-α, studies of which have shown to be involved in neurodegenerative disease [153]. Furthermore, in cases of severe inflammation as a result of SARS-CoV-2 infection the BBB integrity may be damaged by cytokine storm, causing damage to neuronal and glial cells, which could be part of the underlying pathology of the sequelae that lead to Alzheimer’s disease.
Patients infected with SARS-CoV-2 have shown hypoxic alterations and demyelinating lesions, alterations of functional brain integrity, especially in the hippocampus up to 3 months following infection [154,155]. The hippocampus region of the brain is particularly vulnerable to infection [156], and atrophy of the hippocampus results in neurocognitive decline commonly seen in Alzheimer’s disease patients [157,158]. Treatment with SSRIs, many of which have moderate to high sigma-1 affinity such as fluoxetine, has been shown to prevent stress induced atrophy [159] and even induce neurogenesis in the hippocampus [160].
Another interesting hypothesis (although yet to be proved) is the antimicrobial activities of Amyloid-β (Aβ) lead to increased Aβ expression in response to SARS-CoV-2 infection as part of the immune response thus leading to more Aβ deposition in the brain [161].
The sigma-1 receptor has been studied extensively with regard to dementia and in particular Alzheimer’s disease [48], with several new and upcoming Alzheimer’s disease drugs having sigma-1 receptor affinity and acting as sigma-1 receptor agonists. SARS-CoV-2 patients may benefit from sigma-1 drugs as they may alleviate many of the stresses and neuroinflammatory pathways that are part of Alzheimer’s disease’s pathology.
7.4. Parkinson’s disease
It is unclear whether Parkinson’s disease results in vulnerability to SARS-CoV-2, but Parkinson’s disease is an often observed consequence of several viral encephalitis [162]. While there is currently little evidence that sigma-1 receptor drugs may treat encephalitis, they may prevent the neuroinflammatory pathways that lead to encephalitis, and thus prevent the downstream effects that lead to the development of Parkinson’s disease. Furthermore, the sigma-1 receptor is a target for a number of drugs aimed at treating Parkinson’s disease [163], such as pridopidine [164] and PRE-084 [165]. Furthermore, the sigma-1 receptor has been shown to be expressed at lower levels in the early stages of Parkinson’s disease [166]. Several studies have indicated that dopamine induced cell death of various cultured cell lines can be prevented by sigma-1 receptor ligands [58,167], with many of these activities revolving around preventing oxidative stress and preventing endoplasmic reticulum and mitochondrial stress.
7.5. Mental health
The SARS-CoV-2 pandemic has resulted in a large increase in cases of depression, anxiety, and suicide. It is thought that the possible causes of this increase could be related to the stress and fear of catching the disease [168], social isolation [169], the economic burden of unemployment, the possible exacerbation of preexisting neurological conditions, or profound changes to the brain caused by SARS-CoV-2 infection [170].
The sigma-1 receptor has a close relationship with many neurological conditions [60]; many sigma-1 receptor agonists have antidepressant-like activities in animals [59,171], and sigma-1 KO mice show a depressive like phenotype [172].
8. The link between anxiety and depression with mortality
The link between anxiety and depression with mortality has been well studied, particularly in cardiology [173] and oncology. Numerous studies have been constantly searched for the correlation between mental disorders and mortality to evaluate whether these psychiatric illnesses are substantial causes of death worldwide and preventable. Walker et al. Walker, McGee [174] conducted a meta-analysis of 203 relevant articles and found that the chance of death in people with mental disorders was 2-fold greater than that in people with good mental health [174]. Inpatients’ mortality rates were also significantly higher than outpatients due to the general tendency to have more advanced disease stages and comorbid conditions [174]. Depression, one of the most common mental illnesses, was proved to be associated with excess mortality [175], and the risks among different disease-related depression groups (such as cancer, heart, and kidney diseases) were comparable, except chronic obstructive pulmonary disease (COPD) [176]. There has been shown to be a link between cardiovascular disease, depression, and sigma-1 receptor expression, with fluvoxamine showing cardioprotective and antidepressant effects in rodents [177,178]. Given the risk of death from SARS-CoV-2 infection is greatly increased in patients with cardiovascular disease, this could be of interest regarding possible therapies. The SARS-CoV-2 outbreak has led to vast numbers of deaths and adverse mental health consequences, including the uncertainty of treatment and availability of a vaccine, limited social communication, quarantine, and strict lockdown measures. This immediate stress considerably aggravated levels of post-traumatic stress disorder (PTSD) symptoms, depression, anxiety, stress, insomnia, emotional management, and suicidal ideation in psychiatric patients [179]. Patients with a psychiatric history together with SARS-CoV-2 diagnosis also showed a higher mortality rate than those infected patients without psychiatric condition [180], and patients with a severe psychiatric disorder (SPD) were reported to die from SARS-CoV-2 at a younger age than patients with a common mental disorder (CMD) [181]. It is then assuming that psychiatric history and severity can be essential parts of mortality risk factors. The impact of SARS-CoV-2 was even left to survivors after the recovery by being at a higher risk of psychiatric morbidity afterward [182]. For the general population, the SARS-CoV-2 pandemic could enhance levels of depression and anxiety especially in females, students, low-income and low educational level respondents [183]. One of the main reasons is a lack in social connection and loneliness, which contributes to depression and subsequently leads to premature mortality, and this risk factor was indicated to be equivalent to grades 2 and 3 obesity [184]. Another source of pronounced depression and anxiety is economic damage that could further develop to long-term implications on mental health, resulting in shortened longevity and even suicide [185]. Not surprisingly, a prevalence of psychological and mental disorders in global level has been constantly increased following the pandemic with around 30% in each stress, anxiety, and depression category [186]. Therefore, consideration in psychological strategies for prevention and therapeutic rehabilitation is necessary. Sigma-1 agonists have antidepressant activity; furthermore, many commonly used antidepressants have an affinity for the sigma-1 receptor [59,60]. This could be of great interest when it comes to treating the symptoms of SARS-CoV-2 infection, preventing depressive symptoms, and at the same time providing protection to the heart and lungs [187].
9. Conclusions
Despite the roll out of safe and effective vaccines, there has been suspicion and unwillingness by certain sections of the population to take the vaccine. Furthermore, some nations have found difficulty securing a supply of vaccines, while doses in developed nations go to waste due to lack of demand. Without a comprehensive and cooperative worldwide effort to vaccinate enough of the population such that herd immunity takes effect, SARS-CoV-2 will continue to spread, and we will continue to see mutant strains, such as the alpha (B.1.1.7), beta (B.1.351), delta (B.1617.2), and gamma (P.1) variants, which in a worst-case scenario may evade vaccination or prove even more virulent and deadly. For this reason, drugs that lessen the severity of the infection and keep patients out of the ITU will take the strain from healthcare systems worldwide and save thousands of lives. Drugs like fluvoxamine have been identified as drugs that may prevent the worst symptoms of SARS-CoV-2 infection, and small clinical studies have backed this up (Table 1). We wait for more extensive ongoing clinical study results before we can say for sure whether fluvoxamine is indeed as effective as the initial study has indicated.
Table 1.
Sigma-1 receptor mediated drugs that may act against SARS CoV-2
| Name of the Drug | Properties | Advantages | Mode of Action | Disadvantages | References |
|---|---|---|---|---|---|
| Fluvoxamine | A selective serotonin reuptake inhibitor, and an anti-depressant. Sigma-1 receptor agonist |
Fluvoxamine decreased clinical progression, coupled with shortness of breath or hospitalization, with fluvoxamine at up to 300 mg per day. Another study reported the incidence of hospitalization and the persistence of residual symptoms was at 0% |
Fluvoxamine can reduce cellular stress by reducing ER stress and cytokine secretion without inhibiting conventional inflammatory pathways, thereby prevent cardiac injury. Fluvoxamine can increase the bioavailability of melatonin |
Larger, randomized and placebo-controlled trials are required | [87,88,94,95] |
| Fluoxetine | SSRI and sigma-1 receptor agonist Anti-depressant which can induce autophagy | Can inhibit the growth of SARS CoV-2 in vitro | Fluoxetine acts specifically to inhibit SARS CoV-2 and it could not inhibit the growth of other viruses | In vivo and clinical trials should be done to confirm the activity | [108,110–112] |
| Donepezil | Neuroprotective agent with anti-acetylcholinesterase and sigma-1 receptor activity | Donepezil could occupy sigma-1 receptor sites in the brain, induce neuroprotective effects by promoting neurite outgrowth and inducing autophagic flux, along with activation of AMPK |
In vitro, In vivo and clinical trials should be done to confirm the activity | [114–116] | |
| Melatonin | Hormone associated with sleep cycles | Fluvoxamine has been reported to increase the bioavailability of the melatonin | Melatonin, majorly synthesized in mitochondria, acts as an antioxidant and an anti-inflammatory agent and activate mitophagy | SARS CoV-2 can damage mitophagy. In vitro, In vivo and clinical trials should be done to confirm the activity |
[96,97,120,121] |
| Hydroxychloroquine | Anti-malarial drug with sigma-1 receptor agonist activity |
In vitro and some small uncontrolled studies suggested its efficacy in preventing SARS CoV-2 infection | N/A | Clinical studies showed little benefit to hospitalized patients treated with hydroxychloroquine which, however, could increase the risk of death in SARs CoV-2 hospitalized patients | [123,126–130] |
| PB28 | Antagonist of the sigma-1 receptor | Prevent the severity of SARS-CoV-2 infection | PB28 binds to the Sigma-InsP3 receptor complex results in modulation of NSP6 activity leading to autophagosome activation | In vitro, In vivo and clinical trials should be done to confirm the activity | [61,133–135] |
| Haloperidol | Antipsychotic drug with sigma-1 (antagonist) and sigma-2 activity (agonist) |
Limit the severity of SARS-CoV-2 infection. Docking studies suggest haloperidol could effectively bind with NSP6 |
Activation of NSP6 could lead to activation of autophagy | Clinical study has indicated that haloperidol treatment does not change the associated risk of intubation or death compared to patients not treated with haloperidol, nor does it affect time to discharge | [61,136,137] |
| Chlorpromazine | Neuroleptic drug with sigma-1 receptor activity |
Prevent the severity of SARS-CoV-2 infection in monkey and human cells and in some patients | Unknown | Very few SARS-CoV-2 patients have taken chlorpromazine and more studies should be done | [138,139] |
10. Expert opinion
The SARS-CoV-2 outbreak has put tremendous pressure on healthcare systems worldwide, with wave after wave of infections and new strains appearing with different infection rates and different responses to the newly developed vaccines. There is a great effort to vaccinate the world population to bring an end to what has become the worst pandemic since the 1918 influenza (Spanish flu), and vaccination programs worldwide are ongoing at different rates. There are obstacles in the way of vaccinating the world, including access to vaccines, particularly in developing nations with fewer resources, misinformation, and fear of vaccinations, partially since the apparent (although not conclusive and rare) risk of blood clots with the AstraZeneca vaccine. The priority is to save lives and reduce the pressure on health care systems, and drugs that prevent hospitalization and the need for ITU care can do both. Secondly, there are the long-term effects of SARS-CoV-2 (long-haul symptoms). Limiting these would have a positive impact on people’s health and wellbeing. Thirdly, there is recent evidence that SARS-CoV-2 infection increases the risk of developing the neurological disease. Sigma-1 receptor ligands such as fluvoxamine may be of benefit to all three aspects of SARS-CoV-2 infection. Early clinical studies have shown it to prevent hospitalization and death and possibly limit some of the long-term (long haul) symptoms, which may prevent some of the neurological symptoms either directly or indirectly.
There is a strong connection between the sigma-1 receptor and SARS-CoV-2 infection and replication, with studies showing that sigma-1 receptor KO reduces replication and that sigma-1 receptor ligands such as fluvoxamine prevent hospitalization and death. Drugs that occupy the sigma-1 receptor may be enough to prevent some of the negative aspects of SARS-CoV-2 infection since both agonists and antagonists appear beneficial. However, there is an argument for selecting the sigma-1 receptor agonists when choosing drugs that are protective against SARS-CoV-2 symptoms. Agonists of the sigma-1 receptor may also provide protection to the mitochondria, activate mitophagy to remove damaged and leaking mitochondria, prevent ER stress, manage calcium ion transport, and induce autophagy to prevent cell death in response to infection. This may provide the much-needed protection in the heart and lungs that prevents cellular/tissue damage that can lead to long-term symptoms or death. Furthermore, the recent identification of the increased risk of neurological disease for those who required ITU care during SARS-CoV-2 points to the potential for sigma-1 ligands as a neuroprotective strategy during and after SARS-CoV-2 infection. Especially as a large number of sigma-1 agonists are showing great potential in the treatment of neurological diseases such as donepezil, T-817MA, ANAVEX2-73, and AF710B for Alzheimer’s disease, pridopidine for Amyotrophic lateral sclerosis, and multiple sclerosis, and a host of drugs for depression including fluvoxamine, imipramine, opipramol, igmesine, and sertraline.
It may be of great importance to consider the neurological effects of SARS-CoV-2 infection and evaluate the future risk of the increased neurological disease brought about by the SARS-CoV-2 pandemic. Measures may need to be put into place to screen for these neurological diseases in people who have been previously infected, especially as early detection in these neurological diseases can have more beneficial treatment outcomes.
The patient cohorts most at risk of death when infected with SARS-CoV-2 are people over 60 years of age, and those with comorbidities such as cardiovascular disease and respiratory diseases. Furthermore, men are at a greater risk of death from SARS-CoV-2 than women [188]. Since the sigma-1 receptor activation has been shown to be beneficial in the treatment of cardiovascular and respiratory diseases (providing protection in preclinical studies to the heart and lungs at times of stress) [107,177,189], it would appear that males over the age of 60 with comorbidities such as cardiovascular disease would benefit most from treatment with drugs such as fluvoxamine. Other at-risk groups such as women over the age of 60 with comorbidities, and those younger individuals who suffer from cardiovascular and respiratory diseases could also benefit from sigma-1 receptor drugs potentially preventing the requirement for intensive treatment and reducing the risk of death in these cohorts. This would also reduce the pressure on health care systems allowing them to care for other patients who are not suffering with SARS-CoV-2 infection.
The link between anxiety and other mental disorders and mortality should also be a concern for treating SARS-CoV-2, especially with social distancing and lockdown measures potentially bringing about an increase in these cases. Anti-depressant drugs such as fluvoxamine, with their sigma-1 receptor activity, may reduce mortality in many ways, including reducing anxiety and depression that could come about from a SARS-CoV-2 diagnosis.
The way out of the pandemic will be via vaccination and social distancing, thus preventing the further spread of the virus; however, in the meantime, there is a need for drugs that prevent hospitalization, reduce mortality, and prevent the long-term symptoms of SARS-CoV-2 infection. The sigma-1 receptor could be a target that kills two birds with one stone preventing infection and replication of the virus and mitigating damage caused by the virus to important tissues in the body, including heart, lung, and brain tissues.
Funding Statement
J.M.B. and M.I.P. were funded by the Rachadapisek Sompote Fund for Postdoctoral Fellowship, Chulalongkorn University. D.S.M. was funded by The Second Century Fund (C2F) for Postdoctoral Fellowship, Chulalongkorn University.
Article highlights
There appears to be a link between sigma-1 receptor ligands and the attenuation of the effects of SARS-CoV-2 infection.
The role of the sigma-1 receptor in autophagic functions and cell survival may explain why drugs with sigma affinity have a positive effect in SARS-CoV-2 infections.
Fluvoxamine has been demonstrated [in at least one small study] to offer significant protection against severe symptoms of SARS-CoV-2 infection.
Fluvoxamine may also prevent the long-term effects of SARS-CoV-2 infection and prevent the neurological problems post SARS-CoV-2 infection.
Many sigma-1 receptor agonists have antidepressant-like activities in animals and sigma-1 knock our mice show a depressive-like phenotype.
This box summarizes key points contained in the article.
Declaration of interest
The author(s) have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Reviewer disclosures
Peer reviewers on this manuscript have no relevant financial or other relationships to disclose
Author contributions
J.M.B, S.B, and T.T, conceived the idea for the manuscript, MIP, DSM, PT, and JMB drafted the manuscript. J.M.B, S.B, P.T, and T.T edited the final draft. All authors had in put to the final draft of the manuscript and agree to its publication.
References
Papers of special note have been highlighted as either of interest (•) or of considerable interest (••) to readers.
- 1.Dodds K, Broto VC, Detterbeck K, et al. The COVID-19 pandemic: territorial, political and governance dimensions of the crisis. Territory, Politics, Governance. 2020. May 26;8(3):289–298. [Google Scholar]
- 2.Ellul MA, Benjamin L, Singh B, et al. Neurological associations of COVID-19. Lancet Neurol. 2020. September 01;19(9):767–783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Garg RK, Paliwal VK, Gupta A.. Encephalopathy in patients with COVID-19: a review. J Med Virol. 2021;93(1):206–222. [DOI] [PubMed] [Google Scholar]
- 4.Tan L, Lin ZC, Ray J, et al. Neurological implications of COVID-19: a review of the science and clinical guidance. BMJ Neurol Open. 2020;2(2):e000101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Taquet M, Geddes JR, Husain M, et al. 66-month neurological and psychiatric outcomes in 236 379 survivors of COVID-19: a retrospective cohort study using electronic health records. Lancet Psychiatry. 2021;8(5): 416–427. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Indication of the need for treatment with neuroprotective drugs in COVID 19 patients.
- 6.Paterson RW, Brown RL, Benjamin L, et al. The emerging spectrum of COVID-19 neurology: clinical, radiological and laboratory findings. Brain. 2020;143(10):3104–3120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ghannam M, Alshaer Q, Al-Chalabi M, et al. Neurological involvement of coronavirus disease 2019: a systematic review. J Neurol. 2020. November 01;267(11):3135–3153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li Y, Li M, Wang M, et al. Acute cerebrovascular disease following COVID-19: a single center, retrospective, observational study. Stroke Vasc Neurol. 2020;5(3):279–284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Oxley TJ, Mocco J, Majidi S, et al. Large-vessel stroke as a presenting feature of Covid-19 in the young. N Engl J Med. 2020;382(20):e60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moro E, Priori A, Beghi E, et al. The international European academy of neurology survey on neurological symptoms in patients with COVID-19 infection. Eur J Neurol. 2020;27(9):1727–1737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lam MH-B, Wing Y-K, Yu -MW-M, et al. Mental morbidities and chronic fatigue in severe acute respiratory syndrome survivors: long-term follow-up. Arch Internal Med. 2009;169(22):2142–2147. [DOI] [PubMed] [Google Scholar]
- 12.Baxendale IR, Hayward JJ, Ley SV, et al. Pharmaceutical strategy and innovation: an academics perspective. ChemMedChem. 2007;2(6):768–788. [DOI] [PubMed] [Google Scholar]
- 13.Hoertel N, Sánchez-Rico M, Vernet R, et al. Association between antidepressant use and reduced risk of intubation or death in hospitalized patients with COVID-19: results from an observational study. Mol Psychiatry. 2021. DOI: 10.1038/s41380-021-01021-4. [DOI] [PubMed] [Google Scholar]
- 14.Tang T, Bidon M, Jaimes JA, et al. Coronavirus membrane fusion mechanism offers a potential target for antiviral development. Antiviral Res. 2020;178:104792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wang H, Yang P, Liu K, et al. SARS coronavirus entry into host cells through a novel clathrin-and caveolae-independent endocytic pathway. Cell Res. 2008;18(2):290–301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tang T, Jaimes JA, Bidon MK, et al. Proteolytic activation of SARS-CoV-2 spike at the S1/S2 boundary: potential role of proteases beyond furin. ACS Infect Dis. 2021;7(2):264–272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kawase M, Shirato K, van der Hoek L, et al. Simultaneous treatment of human bronchial epithelial cells with serine and cysteine protease inhibitors prevents severe acute respiratory syndrome coronavirus entry. J Virol. 2012;86(12):6537–6545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.He B. Viruses, endoplasmic reticulum stress, and interferon responses. Cell Death Differ. 2006;13(3):393–403. [DOI] [PubMed] [Google Scholar]
- 19.Hetz CA, Soto C. Stressing out the ER: a role of the unfolded protein response in prion-related disorders. Curr Mol Med. 2006;6(1):37–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sano R, Reed JC. ER stress-induced cell death mechanisms. Biochim Biophys Acta-Mol Cell Res. 2013;1833(12):3460–3470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Köseler A, Sabirli R, Gören T, et al. Endoplasmic reticulum stress markers in SARS-COV-2 infection and pneumonia: case-control study. in vivo. 2020;34(3 suppl):1645–1650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bartoszewski R, Dabrowski M, Jakiela B, et al. SARS-CoV-2 may regulate cellular responses through depletion of specific host miRNAs. Am J Physiol Lung Cell Mol Physiol. 2020;319(3):L444–L455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Peng S, Wang J, Wei S, et al. Endogenous cellular microRNAs mediate antiviral defense against influenza A virus. Mol Ther Nucleic Acids. 2018;10:361–375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tsitsiou E, Lindsay MA. microRNAs and the immune response. Curr Opin Pharmacol. 2009;9(4):514–520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gebert M, Bartoszewska S, Janaszak-Jasiecka A, et al. PIWI proteins contribute to apoptosis during the UPR in human airway epithelial cells. Sci Rep. 2018;8(1):1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Morishita H, Mizushima N. Diverse cellular roles of autophagy. Annu Rev Cell Dev Biol. 2019;35(1):453–475. [DOI] [PubMed] [Google Scholar]
- 27.Liu X, Matrenec R, Gack MU, et al. Disassembly of the TRIM23-TBK1 complex by the Us11 protein of herpes simplex virus 1 impairs autophagy. J Virol. 2019;93(17). DOI: 10.1128/JVI.00497-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tian L, Yang Y, Li C, et al. The cytotoxicity of coxsackievirus B3 is associated with a blockage of autophagic flux mediated by reduced syntaxin 17 expression. Cell Death Dis. 2018;9(2):1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chaumorcel M, Lussignol M, Mouna L, et al. The human cytomegalovirus protein TRS1 inhibits autophagy via its interaction with Beclin 1. J Virol. 2012;86(5):2571–2584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bruno AP, De Simone FI, Iorio V, et al. HIV-1 Tat protein induces glial cell autophagy through enhancement of BAG3 protein levels. Cell Cycle. 2014;13(23):3640–3644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yeganeh B, Ghavami S, Rahim MN, et al. Autophagy activation is required for influenza A virus-induced apoptosis and replication. Biochim Biophys Acta-Mol Cell Res. 2018;1865(2):364–378. [DOI] [PubMed] [Google Scholar]
- 32.Ait-Goughoulte M, Kanda T, Meyer K, et al. Hepatitis C virus genotype 1a growth and induction of autophagy. J Virol. 2008;82(5):2241–2249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gassen NC, Papies J, Bajaj T, et al. SARS-CoV-2-mediated dysregulation of metabolism and autophagy uncovers host-targeting antivirals. Nature Communications. 2021;12(1):3818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Singh K, Chen Y-C, Hassanzadeh S, et al. Network analysis and transcriptome profiling identify autophagic and mitochondrial dysfunctions in SARS-CoV-2 infection. Front Genet. 2021;12:289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Qu Y, Wang X, Zhu Y, et al. ORF3a mediated-incomplete autophagy facilitates SARS-CoV-2 replication. Preprint;2020. doi: 10.1101/2020.11.12.380709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jiang P, Nishimura T, Sakamaki Y, et al. The HOPS complex mediates autophagosome–lysosome fusion through interaction with syntaxin 17. Mol Biol Cell. 2014;25(8):1327–1337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Miao G, Zhao H, Li Y, et al. ORF3a of the COVID-19 virus SARS-CoV-2 blocks HOPS complex-mediated assembly of the SNARE complex required for autolysosome formation. Dev Cell. 2021;56(4):427–442. e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wu S, He Y, Qiu X, et al. Targeting the potent Beclin 1–UVRAG coiled-coil interaction with designed peptides enhances autophagy and endolysosomal trafficking. Proc Nat Acad Sci. 2018;115(25):E5669–E5678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yuen CK, Wong WM, Mak LF, et al. Suppression of SARS‐CoV‐2 infection in ex‐vivo human lung tissues by targeting class III phosphoinositide 3‐kinase. J Med Virol. 2021;93(4):2076–2083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gorshkov K, Chen CZ, Bostwick R, et al. The SARS-CoV-2 Cytopathic Effect Is Blocked by Lysosome Alkalizing Small Molecules. ACS Infectious Diseases. 2021;7(6):1389–1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yang H, Shen H, Li J, et al. Sigma-1 receptor ablation impairs autophagosome clearance. Autophagy. 2019;15(8):1492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Christ MG, Huesmann H, Nagel H, et al. Sigma-1 receptor activation induces autophagy and increases proteostasis capacity in vitro and in vivo. Cells. 2019;8(3):211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jeon S-H, Kim SH, Kim Y, et al. The tricyclic antidepressant imipramine induces autophagic cell death in U-87MG glioma cells. Biochem Biophys Res Commun. 2011;413(2):311–317. [DOI] [PubMed] [Google Scholar]
- 44.Li J-R, Xu H-Z, Nie S, et al. Fluoxetine-enhanced autophagy ameliorates early brain injury via inhibition of NLRP3 inflammasome activation following subarachnoid hemorrhage in rats. J Neuroinflammation. 2017;14(1):1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shu X, Sun Y, Sun X, et al. The effect of fluoxetine on astrocyte autophagy flux and injured mitochondria clearance in a mouse model of depression. Cell Death Dis. 2019;10(8):1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cho YS, Yen C-N, Shim JS, et al. Antidepressant indatraline induces autophagy and inhibits restenosis via suppression of mTOR/S6 kinase signaling pathway. Sci Rep. 2016;6(1):34655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Liu J, Li L. Targeting autophagy for the treatment of Alzheimer’s disease: challenges and opportunities. Front Mol Neurosci. 2019;12:203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Prasanth MI, Malar D, Tencomnao T, et al. The emerging role of the sigma-1 receptor in autophagy: hand-in-hand targets for the treatment of Alzheimer’s disease. Expert Opinion on Therapeutic Targets. 2021; Online ahead of print; doi. 10.1080/14728222.2021.1939681 [DOI] [PubMed] [Google Scholar]
- 49.Qu J, Li M, Li D, et al. Stimulation of sigma-1 receptor protects against cardiac fibrosis by alleviating IRE1 pathway and autophagy impairment. Oxid Med Cell Longev. 2021. Jan:8836818. DOI: 10.1155/2021/8836818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kourrich S, Su T-P, Fujimoto M, et al. The sigma-1 receptor: roles in neuronal plasticity and disease. Trends Neurosci. 2012;35(12):762–771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Morales-Lázaro SL, González-Ramírez R, Rosenbaum T. Molecular interplay between the sigma-1 receptor, steroids, and ion channels. Front Pharmacol. 2019;10:419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tchedre KT, Huang R-Q, Dibas A, et al. Sigma-1 receptor regulation of voltage-gated calcium channels involves a direct interaction. Invest Ophthalmol Vis Sci. 2008;49(11):4993–5002. [DOI] [PubMed] [Google Scholar]
- 53.Kinoshita M, Matsuoka Y, Suzuki T, et al. Sigma-1 receptor alters the kinetics of Kv1. 3 voltage gated potassium channels but not the sensitivity to receptor ligands. Brain Res. 2012;1452:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Brimson J, Brown C, Safrany S. Antagonists show GTP-sensitive high affinity binding to the sigma-1 receptor. Br J Pharmacol. 2011;164(2b):772–780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ueda H, Yoshida A, Tokuyama S, et al. Neurosteroids stimulate G protein-coupled sigma receptors in mouse brain synaptic membrane. Neurosci Res. 2001;41(1):33–40. [DOI] [PubMed] [Google Scholar]
- 56.Hayashi T. The sigma-1 receptor in cellular stress signaling [mini review]. Front Neurosci. 2019. July 16;13(733). DOI: 10.3389/fnins.2019.00733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Weng T-Y, Tsai S-YA, Su T-P. Roles of sigma-1 receptors on mitochondrial functions relevant to neurodegenerative diseases. J Biomed Sci. 2017;24(1):1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Brimson JM, Safrany ST, Qassam H, et al. Dipentylammonium binds to the sigma-1 receptor and protects against glutamate toxicity, attenuates dopamine toxicity and potentiates neurite outgrowth in various cultured cell lines. Neurotox Res. 2018;34(2):263–272. [DOI] [PubMed] [Google Scholar]
- 59.Brimson JM, Akula KK, Abbas H, et al. Simple ammonium salts acting on sigma-1 receptors yield potential treatments for cancer and depression. Sci Rep. 2020;10(1):9251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Brimson JM, Brimson S, Chomchoei C, et al. Using Sigma-ligands as part of a multi-receptor approach to target diseases of the brain. Expert Opin Ther Targets. 2020;24(10):1009–1028. [DOI] [PubMed] [Google Scholar]
- 61.Gordon DE, Jang GM, Bouhaddou M, et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature. 2020;583(7816): 459–468. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Simga-1 ligands identified for COVID 19 treatment including fluvoxamine.
- 62.Gordon DE, Hiatt J, Bouhaddou M, et al. Comparative host-coronavirus protein interaction networks reveal pan-viral disease mechanisms. Science (New York, NY). 2020;370(6521): eabe9403. [DOI] [PMC free article] [PubMed] [Google Scholar]; • Simga-1 ligands validated the biological and clinical relevance to COVID 19.
- 63.Lehrer S, Rheinstein PH. Homozygosity for rs17775810 Minor Allele associated with reduced mortality of COVID-19 in the UK Biobank Cohort. in vivo. 2021;35(2):965–968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Gassen NC, Niemeyer D, Muth D, et al. SKP2 attenuates autophagy through Beclin1-ubiquitination and its inhibition reduces MERS-Coronavirus infection. Nat Commun. 2019;10(1):1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Jheng J-R, Ho J-Y, Horng J-T. ER stress, autophagy, and RNA viruses. Front Microbiol. 2014;5:388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Aweya JJ, Mak TM, Lim SG, et al. The p7 protein of the hepatitis C virus induces cell death differently from the influenza A virus viroporin M2. Virus Res. 2013;172(1–2):24–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Shrivastava S, Bhanja Chowdhury J, Steele R, et al. Hepatitis C virus upregulates Beclin1 for induction of autophagy and activates mTOR signaling. J Virol. 2012;86(16):8705–8712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Dreux M, Gastaminza P, Wieland SF, et al. The autophagy machinery is required to initiate hepatitis C virus replication. Proc Nat Acad Sci. 2009;106(33):14046–14051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wang X, Zheng T, Lin L, et al. Influenza A virus induces autophagy by its hemagglutinin binding to cell surface heat shock protein 90AA1. Front Microbiol. 2020;11:2399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Liang Q, Luo Z, Zeng J, et al. Zika virus NS4A and NS4B proteins deregulate Akt-mTOR signaling in human fetal neural stem cells to inhibit neurogenesis and induce autophagy. Cell Stem Cell. 2016;19(5):663–671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Friesland M, Mingorance L, Chung J, et al. Sigma-1 receptor regulates early steps of viral RNA replication at the onset of hepatitis C virus infection. J Virol. 2013;87(11):6377–6390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Gekker G, Hu S, Sheng WS, et al. Cocaine-induced HIV-1 expression in microglia involves sigma-1 receptors and transforming growth factor-β1. Int Immunopharmacol. 2006. June 01;6(6):1029–1033. [DOI] [PubMed] [Google Scholar]
- 73.López OV, Gorantla S, Segarra AC, et al. Sigma-1 receptor antagonist (BD1047) decreases Cathepsin B secretion in HIV-infected macrophages exposed to cocaine. J Neuroimmune Pharmacol. 2019. June 01;14(2):226–240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Shiryaev SA, Mesci P, Pinto A, et al. Repurposing of the anti-malaria drug chloroquine for Zika Virus treatment and prophylaxis. Sci Rep. 2017;7(1):1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kumar A, Liang B, Aarthy M, et al. Hydroxychloroquine inhibits Zika virus NS2B-NS3 protease. ACS Omega. 2018;3(12):18132–18141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.de Souza AAA, Torres LR, Capobianco LRPL, et al. Chloroquine and sulfadoxine derivatives inhibit ZIKV replication in cervical cells. Viruses. 2021;13(1):36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Savarino A, Boelaert JR, Cassone A, et al. Effects of chloroquine on viral infections: an old drug against today’s diseases. Lancet Infect Dis. 2003;3(11):722–727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Peymani P, Yeganeh B, Sabour S, et al. New use of an old drug: chloroquine reduces viral and ALT levels in HCV non-responders (a randomized, triple-blind, placebo-controlled pilot trial). Can J Physiol Pharmacol. 2016;94(6):613–619. [DOI] [PubMed] [Google Scholar]
- 79.Mizui T, Yamashina S, Tanida I, et al. Inhibition of hepatitis C virus replication by chloroquine targeting virus-associated autophagy. J Gastroenterol. 2010;45(2):195–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Dyall J, Coleman CM, Venkataraman T, et al. Repurposing of clinically developed drugs for treatment of Middle East respiratory syndrome coronavirus infection. Antimicrob Agents Chemother. 2014;58(8):4885–4893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Inoue Y, Tanaka N, Tanaka Y, et al. Clathrin-dependent entry of severe acute respiratory syndrome coronavirus into target cells expressing ACE2 with the cytoplasmic tail deleted. J Virol. 2007;81(16):8722–8729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Liu Q, Xia S, Sun Z, et al. Testing of Middle East respiratory syndrome coronavirus replication inhibitors for the ability to block viral entry. Antimicrob Agents Chemother. 2015;59(1):742–744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Nunes-Correia I, Eulálio A, Nir S, et al. Caveolae as an additional route for influenza virus endocytosis in MDCK cells. Cell Mol Biol Lett. 2004;9(1):47–60. [PubMed] [Google Scholar]
- 84.Zuo J, Quinn KK, Kye S, et al. Fluoxetine is a potent inhibitor of coxsackievirus replication. Antimicrob Agents Chemother. 2012;56(9):4838–4844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bauer L, Manganaro R, Zonsics B, et al. Fluoxetine inhibits enterovirus replication by targeting the viral 2C protein in a stereospecific manner. ACS Infect Dis. 2019;5(9):1609–1623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Rosen DA, Seki SM, Fernández-Castañeda A, et al. Modulation of the sigma-1 receptor–IRE1 pathway is beneficial in preclinical models of inflammation and sepsis. Sci Transl Med. 2019;11(478):eaau5266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lenze EJ, Mattar C, Zorumski CF, et al. Fluvoxamine vs Placebo and clinical deterioration in outpatients with symptomatic COVID-19: a randomized clinical trial. JAMA. 2020;324(22): 2292–2300. [DOI] [PMC free article] [PubMed] [Google Scholar]; •• Clinical study identifying Fluvoxmine as potentially useful in COVID-19 treatment.
- 88.Seftel D, Boulware DR. Prospective cohort of fluvoxamine for early treatment of Coronavirus disease 19. Open Forum Infect Dis. 2021. Feb 8;8(2):ofab050. [DOI] [PMC free article] [PubMed] [Google Scholar]; • Clinical study identifying Fluvoxmine as potentially useful in COVID-19 treatment.
- 89.Knoops K, Kikkert M, Van Den Worm SH, et al. SARS-coronavirus replication is supported by a reticulovesicular network of modified endoplasmic reticulum. PLoS Biol. 2008;6(9):e226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Maier HJ, Hawes PC, Cottam EM, et al. Infectious bronchitis virus generates spherules from zippered endoplasmic reticulum membranes. MBio. 2013;4(5). DOI: 10.1128/mBio.00801-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Reggiori F, Monastyrska I, Verheije MH, et al. Coronaviruses Hijack the LC3-I-positive EDEMosomes, ER-derived vesicles exporting short-lived ERAD regulators, for replication. Cell Host Microbe. 2010;7(6):500–508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Fung TS, Liu DX. The ER stress sensor IRE1 and MAP kinase ERK modulate autophagy induction in cells infected with coronavirus infectious bronchitis virus. Virology. 2019. Jul;533:34–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Fung TS, Liu DX. Coronavirus infection, ER stress, apoptosis and innate immunity. Front Microbiol. 2014;5:296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Song P, Li W, Xie J, et al. Cytokine storm induced by SARS-CoV-2. Clin Chim Acta. 2020;509:280–287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Pedersen SF, Ho Y-C. SARS-CoV-2: a storm is raging. J Clin Invest. 2020;130(5):2202–2205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Härtter S, Grözinger M, Weigmann H, et al. Increased bioavailability of oral melatonin after fluvoxamine coadministration. Clin Pharmacol Ther. 2000;67(1):1–6. [DOI] [PubMed] [Google Scholar]
- 97.Demisch K, Demisch L, Bochnik HJ, et al. Melatonin and cortisol increase after fluvoxamine. Br J Clin Pharmacol. 1986;22(5):620–622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Anderson GM. Fluvoxamine, melatonin and COVID-19. Psychopharmacology (Berl). 2021. February 01;238(2):611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Pandi-Perumal SR, Cardinali DP, Reiter RJ, et al. Low melatonin as a contributor to SARS-CoV-2 disease. Melatonin Res. 2020;3(4):558–576. [Google Scholar]
- 100.Tesarik J. Melatonin attenuates growth factor receptor signaling required for SARS-CoV-2 replication. Melatonin Res. 2020;3(4):534–537. [Google Scholar]
- 101.Boga JA, Coto-Montes A. ER stress and autophagy induced by SARS-CoV-2: the targets for melatonin treatment. Melatonin Res. 2020;3(3):346–361. [Google Scholar]
- 102.Singh KK, Chaubey G, Chen JY, et al. Decoding SARS-CoV-2 hijacking of host mitochondria in COVID-19 pathogenesis. Am J Physiol Cell Physiol. 2020;319(2):C258–C267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Gatti P, Ilamathi HS, Todkar K, et al. Mitochondria targeted viral replication and survival strategies—prospective on SARS-CoV-2. Front Pharmacol. 2020;11:578599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Tagashira H, Bhuiyan MS, Shioda N, et al. Fluvoxamine rescues mitochondrial Ca2+ transport and ATP production through σ1-receptor in hypertrophic cardiomyocytes. Life Sci. 2014;95(2):89–100. [DOI] [PubMed] [Google Scholar]
- 105.Braz GRF, Freitas CM, Nascimento L, et al. Neonatal SSRI exposure improves mitochondrial function and antioxidant defense in rat heart. Appl Physiol Nutr Metab. 2016;41(4):362–369. [DOI] [PubMed] [Google Scholar]
- 106.Tagashira H, Bhuiyan S, Shioda N, et al. σ1-receptor stimulation with fluvoxamine ameliorates transverse aortic constriction-induced myocardial hypertrophy and dysfunction in mice. Am J Physiol Heart Circ Physiol. 2010;299(5):H1535–H1545. [DOI] [PubMed] [Google Scholar]
- 107.Abdullah CS, Alam S, Aishwarya R, et al. Cardiac dysfunction in the sigma 1 receptor knockout mouse associated with impaired mitochondrial dynamics and bioenergetics. J Am Heart Assoc. 2018;7(20):e009775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Safrany ST, Brimson JM. Are fluoxetine’s effects due to sigma-1 receptor agonism? Pharmacol Res. 2016;113:707–708. [DOI] [PubMed] [Google Scholar]
- 109.Emilia M, Rosso D, Laura M, et al. Immunomodulatory effects of fluoxetine: a new potential pharmacological action for a classic antidepressant drug? Pharmacol Res. 2016;109:101–107. [DOI] [PubMed] [Google Scholar]
- 110.Gulbins A, Schumacher F, Becker KA, et al. Antidepressants act by inducing autophagy controlled by sphingomyelin–ceramide. Mol Psychiatry. 2018;23(12):2324–2346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Zimniak M, Kirschner L, Hilpert H, et al. The serotonin reuptake inhibitor fluoxetine inhibits SARS-CoV-2 in human lung tissue. Sci Rep. 2021;11(1):1–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Dechaumes A, Nekoua MP, Belouzard S, et al. Fluoxetine can inhibit SARS-CoV-2 in vitro. Microorganisms. 2021;9(2):339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Ramakrishnan NK, Visser AK, Schepers M, et al. Dose-dependent sigma-1 receptor occupancy by donepezil in rat brain can be assessed with 11 C-SA4503 and microPET. Psychopharmacology (Berl). 2014;231(20):3997–4006. [DOI] [PubMed] [Google Scholar]
- 114.Ishikawa M, Hashimoto K. The role of sigma-1 receptors in the pathophysiology of neuropsychiatric diseases. J Receptor Ligand Channel. 2010;3:25–36. [Google Scholar]
- 115.Inagaki S, Funato M, Nakamura S, et al. Donepezil, an anti-Alzheimer’s disease drug has the neuroprotective effect on RGCs derived from familial glaucoma patients’ iPS cells. Invest Ophthalmol Vis Sci. 2019;60(9):626. [Google Scholar]
- 116.Kim E, Park M, Jeong J, et al. Cholinesterase inhibitor donepezil increases mitochondrial biogenesis through AMP-activated protein kinase in the hippocampus. Neuropsychobiology. 2016;73(2):81–91. [DOI] [PubMed] [Google Scholar]
- 117.Khuanjing T, Palee S, Kerdphoo S, et al. Donepezil attenuated cardiac ischemia/reperfusion injury through balancing mitochondrial dynamics, mitophagy, and autophagy. Transl Res. 2021;230:82–97. [DOI] [PubMed] [Google Scholar]
- 118.Khuanjing T, Palee S, Chattipakorn S, et al. Acetylcholinesterase inhibitor protects against cardiac ischaemia/reperfusion injury via enhancing mitophagy and rebalancing mitochondrial dynamics. Eur Heart J. 2020;41(Supplement_2):ehaa946.3639. [Google Scholar]
- 119.Steardo L, Monteleone P, d’Istria M, et al. Sigma receptor modulation of noradrenergic-stimulated pineal melatonin biosynthesis in rats. J Neurochem. 1996. Jul;67(1):287–293. [DOI] [PubMed] [Google Scholar]
- 120.Pourhanifeh MH, Dehdashtian E, Hosseinzadeh A, et al. Clinical application of melatonin in the treatment of cardiovascular diseases: current evidence and new insights into the cardioprotective and cardiotherapeutic properties. Cardiovasc Drugs Ther. 2020;1–25. DOI: 10.1007/s10557-020-07052-3. [DOI] [PubMed] [Google Scholar]
- 121.Mehrzadi S, Karimi MY, Fatemi A, et al. SARS-CoV-2 and other coronaviruses negatively influence mitochondrial quality control: beneficial effects of melatonin. Pharmacol Ther. 2021. Jul;224:107825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Hirata Y, Yamamoto H, Atta MSM, et al. Chloroquine inhibits glutamate‐induced death of a neuronal cell line by reducing reactive oxygen species through sigma‐1 receptor. J Neurochem. 2011;119(4):839–847. [DOI] [PubMed] [Google Scholar]
- 123.Yao X, Ye F, Zhang M, et al. In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome Coronavirus 2 (SARS-CoV-2). Clinl Infect Dis. 2020;71(15):732–739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Echeverría-Esnal D, Martin-Ontiyuelo C, Navarrete-Rouco ME, et al. Azithromycin in the treatment of COVID-19: a review. Expert Rev Anti Infect Ther. 2021. February 01;19(2):147–163. [DOI] [PubMed] [Google Scholar]
- 125.Liu M, Caputi TL, Dredze M, et al. Internet searches for unproven COVID-19 therapies in the United States. JAMA Intern Med. 2020;180(8):1116–1118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Horby P, Mafham M, Linsell L, et al. Effect of hydroxychloroquine in hospitalized patients with Covid-19. N Engl J Med. 2020;383(21):2030–2040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Geleris J, Sun Y, Platt J, et al. Observational study of hydroxychloroquine in hospitalized patients with Covid-19. N Engl J Med. 2020;382(25):2411–2418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Chen Z, Hu J, Zhang Z, et al. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. United States: medrxiv. Preprint;2020. doi: 10.1101/2020.03.22.20040758. [DOI] [Google Scholar]
- 129.Mahévas M, Tran V-T, Roumier M, et al. Clinical efficacy of hydroxychloroquine in patients with covid-19 pneumonia who require oxygen: observational comparative study using routine care data. BMJ. 2020;369:m1844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Rosenberg ES, Dufort EM, Udo T, et al. Association of treatment with hydroxychloroquine or azithromycin with in-hospital mortality in patients with COVID-19 in New York State. JAMA. 2020;323(24):2493–2502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Mehra MR, Ruschitzka F, Patel AN. Retraction-hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis. Lancet. 2020;395(10240):1820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Berardi F, Abate C, Ferorelli S, et al. 1-Cyclohexylpiperazine and 3, 3-Dimethylpiperidine derivatives as sigma-1 (sig1) and sigma-2 (sig2) receptor ligands: a review. Central Nervous System Agents Med Chem. 2009;9(3):205–219. [DOI] [PubMed] [Google Scholar]
- 133.Abate C, Niso M, Abatematteo FS, et al. PB28, the sigma-1 and sigma-2 receptors modulator with potent anti–SARS-CoV-2 activity: a review about its pharmacological properties and structure affinity relationships. Front Pharmacol. 2020;11. DOI: 10.3389/fphar.2020.589810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Benvenuto D, Angeletti S, Giovanetti M, et al. Evolutionary analysis of SARS-CoV-2: how mutation of non-structural protein 6 (NSP6) could affect viral autophagy. J Infect. 2020;81(1):e24–e27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Colabufo NA, Leopoldo M, Ferorelli S, et al. Why PB28 could be a Covid 2019 game changer? ACS Med Chem Lett. 2020;11(10):2048–2050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Pandey P, Prasad K, Prakash A, et al. Insights into the biased activity of dextromethorphan and haloperidol towards SARS-CoV-2 NSP6: in silico binding mechanistic analysis. J Mol Med. 2020;98(12):1659–1673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Hoertel N, Sánchez-Rico M, Vernet R, et al. Observational study of haloperidol in hospitalized patients with COVID-19. PloS One. 2021;16(2):e0247122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Su T-P, Hayashi T. Understanding the molecular mechanism of sigma-1 receptors: towards a hypothesis that sigma-1 receptors are intracellular amplifiers for signal transduction. Curr Med Chem. 2003;10(20):2073–2080. [DOI] [PubMed] [Google Scholar]
- 139.Plaze M, Attali D, Prot M, et al. Inhibition of the replication of SARS-CoV-2 in human cells by the FDA-approved drug chlorpromazine. Int J Antimicrob Agents. 2021;57(3):106274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Belani P, Schefflein J, Kihira S, et al. COVID-19 is an independent risk factor for acute ischemic stroke. Am J Neuroradiol. 2020;41(8):1361–1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Fridman S, Bullrich MB, Jimenez-Ruiz A, et al. Stroke risk, phenotypes, and death in COVID-19: systematic review and newly reported cases. Neurology. 2020;95(24):e3373–e3385. [DOI] [PubMed] [Google Scholar]
- 142.Jia J, Cheng J, Wang C, et al. Sigma-1 receptor-modulated neuroinflammation in neurological diseases [mini review]. Front Cell Neurosci. 2018. September 20;12(314). DOI: 10.3389/fncel.2018.00314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Shen YC, Wang YH, Chou YC, et al. Dimemorfan protects rats against ischemic stroke through activation of sigma‐1 receptor‐mediated mechanisms by decreasing glutamate accumulation. J Neurochem. 2008;104(2):558–572. [DOI] [PubMed] [Google Scholar]
- 144.Allahtavakoli M, Jarrott B. Sigma-1 receptor ligand PRE-084 reduced infarct volume, neurological deficits, pro-inflammatory cytokines and enhanced anti-inflammatory cytokines after embolic stroke in rats. Brain Res Bull. 2011;85(3–4):219–224. [DOI] [PubMed] [Google Scholar]
- 145.Mazur A, Fangman M, Ashouri R, et al. Nrf2 as a therapeutic target in ischemic stroke. Expert Opin Ther Targets. 2021. March 04;25(3):163–166. [DOI] [PubMed] [Google Scholar]
- 146.Prasanth MI, Tencomnao T, Brimson JM. Letter to the editor, the sigma-1 receptors role in neuroprotection: comment on Nrf2 as a therapeutic target in ischemic stroke. Expert Opinion on Therapeutic Targets. 2021; Online ahead of print. doi. 10.1080/14728222.2021.1948016 [DOI] [PubMed] [Google Scholar]
- 147.Sánchez-Blázquez P, Pozo-Rodrigálvarez A, Merlos M, et al. The sigma-1 receptor antagonist, S1RA, reduces stroke damage, ameliorates post-stroke neurological deficits and suppresses the overexpression of MMP-9. Mol Neurobiol. 2018. June 01;55(6):4940–4951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Garg RK, Paliwal VK, Gupta A. Encephalopathy in patients with COVID‐19: a review. J Med Virol. 2020;93(1):206–222. [DOI] [PubMed] [Google Scholar]
- 149.Bougakov D, Podell K, Goldberg E. Multiple neuroinvasive pathways in COVID-19. Mol Neurobiol. 2021;58(2):564–575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Hascup ER, Hascup KN. Does SARS-CoV-2 infection cause chronic neurological complications? GeroScience. 2020;42(4):1083–1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Desforges M, Le Coupanec A, Dubeau P, et al. Human coronaviruses and other respiratory viruses: underestimated opportunistic pathogens of the central nervous system? Viruses. 2020;12(1):14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Ciaccio M, Lo Sasso B, Scazzone C, et al. COVID-19 and Alzheimer’s disease. Brain Sci. 2021;11(3):305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Mohammadi S, Moosaie F, Aarabi MH. Understanding the immunologic characteristics of neurologic manifestations of SARS-CoV-2 and potential immunological mechanisms. Mol Neurobiol. 2020;57(12):5263–5275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Coolen T, Lolli V, Sadeghi N, et al. Early postmortem brain MRI findings in COVID-19 non-survivors. Neurology. 2020;95(14):e2016–e2027. [DOI] [PubMed] [Google Scholar]
- 155.Reichard RR, Kashani KB, Boire NA, et al. Neuropathology of COVID-19: a spectrum of vascular and acute disseminated encephalomyelitis (ADEM)-like pathology. Acta Neuropathol. 2020;140(1):1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Jacomy H, Fragoso G, Almazan G, et al. Human coronavirus OC43 infection induces chronic encephalitis leading to disabilities in BALB/C mice. Virology. 2006;349(2):335–346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Lu Y, Li X, Geng D, et al. Cerebral micro-structural changes in COVID-19 patients–an MRI-based 3-month follow-up study. EClinicalMedicine. 2020;25:100484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Love S, Miners JS. Cerebrovascular disease in ageing and Alzheimer’s disease. Acta Neuropathol. 2016;131(5):645–658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Luo L, Tan R-X. Fluoxetine inhibits dendrite atrophy of hippocampal neurons by decreasing nitric oxide synthase expression in rat depression model. Acta Pharmacol Sin. 2001;22(10):865–870. [PubMed] [Google Scholar]
- 160.Santarelli L, Saxe M, Gross C, et al. Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. science. 2003;301(5634):805–809. [DOI] [PubMed] [Google Scholar]
- 161.Soscia SJ, Kirby JE, Washicosky KJ, et al. The Alzheimer’s disease-associated amyloid β-protein is an antimicrobial peptide. PloS One. 2010;5(3):e9505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Tipton PW, Wszolek ZK. What can Parkinson’s disease teach us about COVID-19? Neurol Neurochir Pol. 2020;54(2):204–206. [DOI] [PubMed] [Google Scholar]
- 163.Francardo V. Sigma-1 receptor: a potential new target for Parkinson’s disease? Neural Regen Res. 2014;9(21):1882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Francardo V, Geva M, Bez F, et al. Pridopidine induces functional neurorestoration via the sigma-1 receptor in a mouse model of Parkinson’s disease. Neurotherapeutics. 2019;16(2):465–479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Guo C-H, Cao T, Zheng L-T, et al. Development and characterization of an inducible Dicer conditional knockout mouse model of Parkinson’s disease: validation of the antiparkinsonian effects of a sigma-1 receptor agonist and dihydromyricetin. Acta Pharmacol Sin. 2020;41(4):499–507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Mishina M, Ishiwata K, Ishii K, et al. Function of sigma 1 receptors in Parkinson’s disease. Acta Neurol Scand. 2005;112(2):103–107. [DOI] [PubMed] [Google Scholar]
- 167.Mori T, Hayashi T, Su TP. Compromising σ-1 receptors at the endoplasmic reticulum render cytotoxicity to physiologically relevant concentrations of dopamine in a nuclear factor-κB/Bcl-2-dependent mechanism: potential relevance to Parkinson’s disease. J Pharmacol Exp Ther. 2012;341(3):663–671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Ahorsu DK, Lin CY, Imani V, et al. The fear of COVID-19 scale: development and initial validation. Int J Ment Health Addict. 2020;27:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Mamun MA, Ullah I. COVID-19 suicides in Pakistan, dying off not COVID-19 fear but poverty? - The forthcoming economic challenges for a developing country. Brain Behav Immun. 2020. Jul;87:163–166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Troyer EA, Kohn JN, Hong S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav Immun. 2020. Jul;87:34–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Bermack JE, Debonnel G. The role of sigma receptors in depression. J Pharmacol Sci. 2005;336:317–336. [DOI] [PubMed] [Google Scholar]
- 172.Sabino V, Cottone P, Parylak SL, et al. Sigma-1 receptor knockout mice display a depressive-like phenotype. Behav Brain Res. 2009;198(2):472–476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Celano CM, Millstein RA, Bedoya CA, et al. Association between anxiety and mortality in patients with coronary artery disease: a meta-analysis. Am Heart J. 2015;170(6):1105–1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Walker ER, McGee RE, Druss BG. Mortality in mental disorders and global disease burden implications: a systematic review and meta-analysis. JAMA Psychiatry. 2015;72(4):334–341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Gilman SE, Sucha E, Kingsbury M, et al. Depression and mortality in a longitudinal study: 1952-2011. CMAJ. 2017;189(42):E1304–E1310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Cuijpers P, Vogelzangs N, Twisk J, et al. Comprehensive meta-analysis of excess mortality in depression in the general community versus patients with specific illnesses. Am J Psychiatry. 2014. Apr;171(4):453–462. [DOI] [PubMed] [Google Scholar]
- 177.Hashimoto K. Sigma-1 receptor chaperone and brain-derived neurotrophic factor: emerging links between cardiovascular disease and depression. Prog Neurobiol. 2013;100:15–29. [DOI] [PubMed] [Google Scholar]
- 178.Ito K, Hirooka Y, Matsukawa R, et al. Decreased brain sigma-1 receptor contributes to the relationship between heart failure and depression. Cardiovasc Res. 2012;93(1):33–40. [DOI] [PubMed] [Google Scholar]
- 179.Hao F, Tan W, Jiang L, et al. Do psychiatric patients experience more psychiatric symptoms during COVID-19 pandemic and lockdown? A case-control study with service and research implications for immunopsychiatry. Brain Behav Immun. 2020;87:100–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Li L, Li F, Fortunati F, et al. Association of a prior psychiatric diagnosis with mortality among hospitalized patients with coronavirus disease 2019 (COVID-19) infection. JAMA Network Open. 2020;3(9):e2023282–e2023282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Lega I, Nisticò L, Palmieri L, et al. Psychiatric disorders among hospitalized patients deceased with COVID-19 in Italy. EClinicalMedicine. 2021. Apr;23:100854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Taquet M, Luciano S, Geddes JR, et al. Bidirectional associations between COVID-19 and psychiatric disorder: retrospective cohort studies of 62 354 COVID-19 cases in the USA. Lancet Psychiatry. 2021. Feb;8(2):130–140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Alyami HS, Naser AY, Dahmash EZ, et al. Depression and anxiety during the COVID-19 pandemic in Saudi Arabia: a cross-sectional study. Int J Clin Pract. 2021;20:e14244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Holt-Lunstad J, Smith TB, Baker M, et al. Loneliness and social isolation as risk factors for mortality: a meta-analytic review. Perspect Psychol Sci. 2015. Mar;10(2):227–237. [DOI] [PubMed] [Google Scholar]
- 185.Brenner MH, Bhugra D. Acceleration of anxiety, depression, and suicide: secondary effects of economicdisruption related to COVID-19. Front Psychiatry. 2020;11:592467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Salari N, Hosseinian-Far A, Jalali R, et al. Prevalence of stress, anxiety, depression among the general population during the COVID-19 pandemic: a systematic review and meta-analysis. Global Health. 2020. Jul 6;16(1):57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Couly S, Goguadze N, Yasui Y, et al. Knocking out sigma-1 receptors reveals diverse health problems. Cell Mol Neurobiol. 2020. Oct:1–24. DOI: 10.1007/s10571-020-00983-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Caramelo F, Ferreira N, Oliveiros B. Estimation of risk factors for COVID-19 mortality-preliminary results. United States:MedRxiv. Preprint; 2020. DOI: 10.1101/2020.02.24.20027268. [DOI] [Google Scholar]
- 189.Kourrich S, Su T-P, Fujimoto M, et al. The sigma-1 receptor: roles in neuronal plasticity and disease. Trends Neurosci. 2012;35(12):1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]


