Abstract
Rationale
The development of addiction is accompanied by a shift in the mechanisms motivating cocaine use from nucleus accumbens (NAc) dopamine D1 receptor (D1R) signaling to glutamate AMPA-kainate receptor (AMPA-R) signaling.
Objective
Here we determined whether similar shifts occur for NAc-D2R signaling and following systemic manipulation of D1R, D2R, and AMPA-R signaling.
Methods
Male rats were given short-access (20 infusions/day) or extended-access to cocaine (24-hr/day, 96 infusions/day, 10 days). Motivation for cocaine was assessed following 14-days of abstinence using a progressive-ratio schedule. Once responding stabilized, the effects of NAc-D2R antagonism (eticlopride; 0-10.0 μg/side) and systemic D1R (SCH-23390; 0-1.0 mg/kg), D2R (eticlopride; 0-0.1 mg/kg), and AMPA-R (CNQX; 0-1.5 mg/kg) antagonism, and NAc-dopamine-R gene expression (Drd1/2/3) were examined.
Results
Motivation for cocaine was markedly higher in the extended- versus short-access group confirming the development of an addiction-like phenotype in the extended-access group. NAc-infused eticlopride decreased motivation for cocaine in both the short- and extended-access groups although low doses (0.1-0.3-μg) were more effective in the short-access group and high doses (3-10-μg/side) tended to be more effective in the extended-access group. Systemic administration of eticlopride (0.1 mg/kg) was more effective in the extended-access group, and systemic administration of CNQX was effective in the extended-, but not short-access group. NAc-Drd2 expression was decreased in both the short- and extended-access groups.
Conclusion
These findings indicate that in contrast to NAc-D1R, D2R remain critical for motivating cocaine use with the development of an addiction-like phenotype. These findings also indicate that shifts in the mechanisms motivating cocaine use impact the response to both site-specific and systemic pharmacological treatment.
Keywords: Extended Access, Cocaine, Dopamine, Gene Expression, Glutamate, Motivation, Self-Administration
Introduction
Despite a decrease in the number of individuals in the US with a cocaine use disorder in the past several years (2012-2018; Substance Abuse and Mental Health Services Administration, 2019), cocaine-related overdose deaths have increased by 52.7% within this time-frame (Seth et al. 2018). While several factors likely contribute to overdose deaths, individuals with prolonged abstinence appear to be at higher risk (Binswanger et al. 2012; Binswanger et al. 2007; Seymour et al. 2000), perhaps due to the combined effects of precipitated cue-induced craving over abstinence and a lowered physiological tolerance to the drug (Parvaz et al. 2016). Considerable attention and resources have been allocated towards developing pharmacological compounds to reduce craving and prevent relapse; however, these efforts have been met with limited success.
Part of the problem may be that the models used for medications development still typically use either non-contingent administration or short-access (ShA) self-administration (e.g. Bornebusch et al. 2019; Gentile et al. 2018; Hammad et al. 2017; Neisewander et al. 2014) which, while useful for determining vulnerability, generally do not capture critical features of human cocaine addiction (Allain and Samaha 2019; Lynch 2018; Oleson and Roberts 2008; 2009; Zimmer et al. 2012). For example, when cocaine is available during short daily sessions (1-2 hr/day), intake is relatively stable, and animals take roughly the same amount of drug every day (Lynch and Carroll 2001; Roberts et al. 2007) and show low-to-moderate levels of motivation for the drug and drug-seeking (Deroche-Gamonet et al. 2004; Doyle et al. 2014; Piazza et al. 2000; Ramôa et al. 2014; Vranjkovic et al. 2018). Yet in humans, cocaine use disorder is characterized by compulsive use and high levels of craving and motivation for the drug (American Psychiatric Association 2013; O’Brien 2005). These characteristics can be captured in animals, particularly when access to the drug is extended to six or more hours/day (for review see Koob and Kreek 2007; Lynch 2018; Negus and Banks 2018; Yenniro et al. 2016). For example, animals given extended-access (ExA) to cocaine (6-24/hr/day) under either continuous or intermittent-access schedules self-administer cocaine in escalating or binge-abstinent patterns, and following protracted abstinence, show marked increases in drug-seeking and motivation for the drug (for review see Koob and Kreek 2007; Lynch 2018; Negus and Banks 2018; Venniro et al. 2016). While these procedures are used extensively to determine neurobiological correlates of addiction-like behaviors, particularly for drug-seeking/relapse (for review see Loweth et al. 2014; Pickens et al. 2011; Wolf and Tseng 2012), few studies have used them to screen potential medications or to determine neurobiological correlates of motivational aspects of addiction (e.g. Doyle et al. 2014; Ducret et al. 2016; James et al. 2019; Pelloux et al. 2018; Ramôa et al. 2014; Schmeichel et al. 2017; Valenza et al. 2017). This is critical since the neurobiological changes are different following ShA versus ExA self-administration (e.g. Ben-Shahar et al. 2007; Boudreau and Wolf 2005; Briand et al. 2008; Conrad et al. 2008; Fischer et al. 2013; Greenwell et al. 2009; Hao et al. 2010; Nicolas et al. 2017; Zimmer et al. 2013; Zorrilla et al. 2012).
Considerable evidence from ShA self-administration studies indicates that dopamine (DA) signaling in the nucleus accumbens (NAc) is critical for the reinforcing effects of cocaine (Bibb et al. 1999; Kalivas and Volkow 2005; Willuhn et al. 2010). Signaling at the D1 receptor (D1R), in particular, is implicated as a marker for initial vulnerability to cocaine (Bibb et al. 1999; Zhang et al. 2006) and results show that blockade of these receptors prevents acquisition of cocaine self-administration, disrupts maintenance intake, and decreases motivation for the drug (Bari and Pierce 2005; McGregor and Roberts 1993; Pierce and Kumaresan 2006). In contrast, evidence from studies using ExA self-administration indicate that the role of NAc-D1R signaling becomes diminished with the development of an addiction-like phenotype (Doyle et al. 2014; Ramôa et al. 2014). Specifically, we and others have shown that motivation for cocaine, as assessed under a progressive-ratio (PR) schedule, markedly increases following protracted abstinence from ExA, but not ShA cocaine self-administration (Doyle et al. 2014; Morgan et al. 2002; Morgan and Roberts 2004; Morgan et al. 2005; Ramôa et al. 2014). While NAc-D1R antagonism dose-dependently reduced motivation for cocaine following ShA self-administration, it was not effective following ExA self-administration (Ramôa et al. 2014). Notably, with the development of an enhanced motivation for cocaine, which has been used to define the addiction-like phenotype, there is also a strengthening of the role of NAc glutamatergic signaling (Doyle et al. 2014; Ramôa et al. 2014). Specifically, we previously showed that while NAc-AMPA-kainate receptor (AMPA-R) antagonism was without effect following ShA self-administration, it dose-dependently reduced motivation for cocaine following ExA self-administration (Doyle et al. 2014). These findings indicate that with the development of an addiction-like phenotype, there is a shift in the mechanisms motivating cocaine use from NAc-D1R to AMPA-R signaling. Findings in humans also indicate that alterations in glutamatergic, rather than DAergic, signaling differentiate individuals with cocaine use disorder from controls (Hemby et al. 2005; Martinez et al. 2009b; Meador-Woodruff et al. 1993; Tang et al. 2003).
While the studies described above provide evidence for a diminished role of DA signaling with the development of cocaine addiction, it is possible that DA signaling at D2Rs remains involved. This view is supported by preclinical findings showing that following prolonged ShA self-administration (4-hr/day for 3 weeks), the balance between D1R and D2Rs becomes dysregulated (Edwards et al. 2007), with alterations in D2Rs showing a more persistent time-course (Conrad et al. 2010). Similarly, human imaging studies show that NAc-D2R availability is persistently decreased in cocaine users as compared to controls (Volkow et al. 1993; Volkow et al. 1990; Volkow et al. 1997). However, other studies in animals show no change in NAc-D2R as a result of ExA cocaine self-administration (Ben-Shahar et al. 2007), and post-mortem tissue studies show no difference in D2R levels between people who died from cocaine overdose and controls (Meador-Woodruff et al. 1993; Tang et al. 2003). Thus, the goal of the current study was to determine the role of NAc-D2R in motivating cocaine use with the development of an addiction-like phenotype. To do so, we examined the effects of intra-NAc infusions of the D2R antagonist eticlopride on motivation for cocaine (as assessed under a PR schedule) in male rats tested following ExA (versus ShA) cocaine self-administration and 14-days of abstinence, conditions known to induce an enhanced motivation for cocaine and heightened drug-seeking (Abel et al. 2019; Ben-Shahar et al. 2008; Doyle et al. 2014; Ramôa et al. 2014; Venniro et al. 2016). Based on our previous findings on NAc-D1R signaling (Ramôa et al. 2014), we predicted that the role of NAc-D2Rs would become diminished with the development of an addiction-like phenotype. We also examined changes in NAc-D2R, as well as D1R and D3R, at a molecular level (gene expression).
A second goal of the current study was to determine whether similar mechanistic changes can also be observed following systemic manipulation. This is important since medications for substance use disorder are typically administered systemically in humans (i.e., oral administration, de Lima et al. 2002). To address this question, we compared the effects of systemic administration of D1R, D2R, and AMPA-R antagonists on motivation for cocaine following ShA versus ExA self-administration. Based on findings in humans with a cocaine use disorder suggesting better efficacy for glutamate versus dopamine-based pharmacological treatments (Haile et al. 2013), we predicted that systemic administration of DA and glutamate receptor antagonists would mirror effects observed following site-specific modulation in the NAc (i.e. greater effects of DA receptor antagonism in the ShA versus ExA group, and greater efficacy of AMPA-R antagonism in the ExA versus ShA group; Doyle et al. 2014; Ramôa et al. 2014).
Materials and Methods
Subjects.
Adult male Sprague-Dawley rats (Charles River Laboratories, ME), weighing 380-410 g at the start of the experiments, were used as subjects (N=131). Upon arrival, rats were housed in individual operant chambers (Med Associates Inc, VT) in a temperature (20-22° C) and humidity (40-70%) controlled vivarium and maintained on a 12-hr light/dark cycle (room/house lights on at 7AM) with ad libitum access to food and water. After a 2-day acclimation period, rats were pre-trained to lever-press for sucrose pellets using methods previously described (fixed-ratio 1, FR1, ≥50 pellets/day, 2 consecutive days; Abel et al. 2019; Doyle et al. 2014; Ramôa et al. 2014). Following lever pre-training, rats underwent a jugular catheterization surgery using methods previously described (Abel et al. 2019; Doyle et al. 2014; Ramôa et al. 2014). Catheters were maintained as described previously (Abel et al. 2019; Doyle et al. 2014; Ramôa et al. 2014), and if no longer patent, a new catheter was implanted into the left jugular vein with testing resuming after 2-days of recovery. All the procedures were approved by the University of Virginia Animal Care and Use Committee and were conducted within the guidelines set by the NIH.
Procedures
Cocaine Self-Administration and Abstinence.
Following recovery from surgery, rats were trained to self-administer cocaine (1.5 mg/kg/infusion) under an FR1 schedule as previously described (20 infusions/day; Abel et al. 2019; Doyle et al. 2014; Ramôa et al. 2014). Following acquisition (20 infusions/day for two consecutive sessions), rats were randomly assigned to either a ShA (n=60) or ExA group (n=55). Rats in the ShA group were given three additional FR1 sessions, and rats in the ExA group were given 24-hr/day intermittent-access to cocaine (1.5 mg/kg/infusion) for 10 days using a discrete trial procedure (4 trials/hour; 96 infusions/day, Doyle et al. 2014; Ramôa et al. 2014). These ExA conditions are known to induce binge-abstinence patterns of self-administration as well as high levels of drug-seeking and motivation for the drug when assessed following 14-days of abstinence (Abel et al. 2019; Doyle et al. 2014; Ramôa et al. 2014). Importantly, motivation for cocaine does not change from baseline levels following abstinence from self-administration under these ShA conditions (Doyle et al. 2014; Ramôa et al. 2014). Rats in the ExA group were then given two additional FR1 sessions to confirm catheter patency and to minimize between-group differences in cocaine levels before abstinence. A 14-day abstinence period began after the last FR1 session, wherein rats remained in their chambers with the active lever retracted. The main experimental procedure is also summarized in Fig. 1a–b.
Fig. 1. Summary of the experimental design as well as cannulae track placement for Experiment 1.
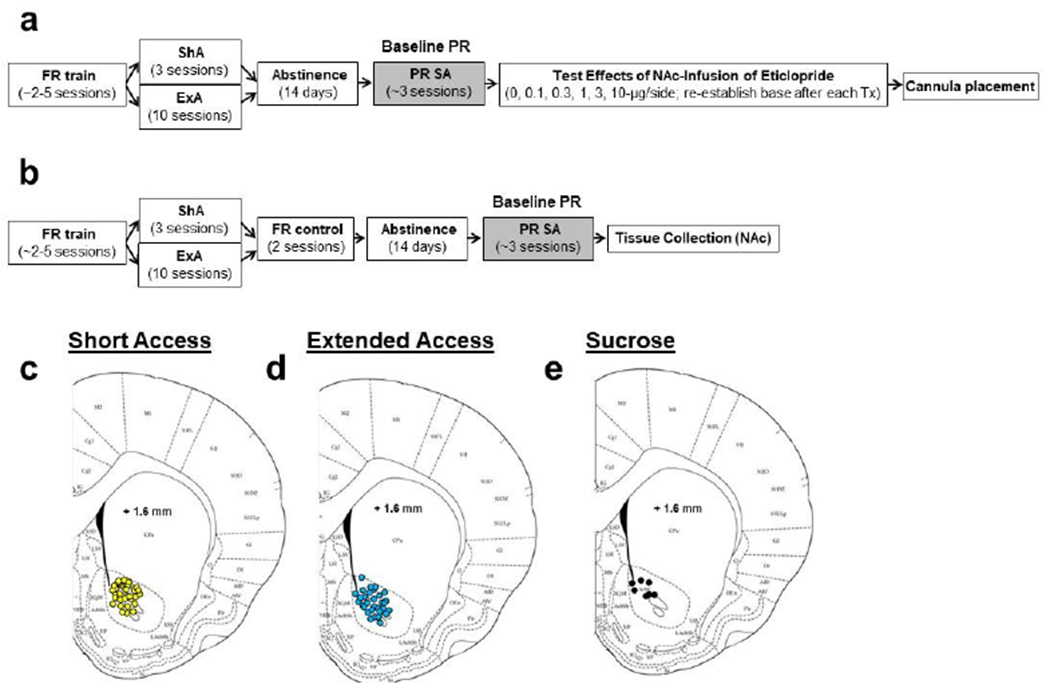
(a) Experiment 1a-b. Following acquisition under a fixed-ratio 1 (FR-1) schedule (FR train), rats were given either short-access (ShA; FR1, maximum of 20 infusions/day, 3 days) or extended-access (ExA) to cocaine (4 discrete trials/hr, maximum of 96 infusions/day, 10 days). Rats in the extended access group were then given access to cocaine in two consecutive FR1 sessions (maximum of 20 infusions/day) in order to verify patency and to equate intake between the ShA and ExA groups prior to abstinence. Motivation to obtain cocaine was assessed following a 14-day abstinence period using a progressive-ratio (PR) schedule (PR SA, self-administration). Testing for the effects of NAc infusions of eticlopride began once baseline was established and a minimum of three stable PR sessions separated each test session. The effects of NAc infusions of eticlopride were also examined in additional groups of rats tested following sucrose self-administration training, short-access sucrose self-administration and 14-days of abstinence using the same experimental procedures. Following study completion, tissue was collected for cannulation placement verification. (b) Experiment 1c. Following acquisition, short- or extended-access cocaine self-administration, and 14 days of abstinence, rats were given access to cocaine infusions under a PR schedule. Brain tissue was obtained from the NAc following the third stable PR session (~18 hours after the last infusion). Histological cannulae placements in the NAc for each of the rats in the (c) short-access (d) extended-access and (e) sucrose control groups. Schematics adapted from the atlas of Paxinos and Watson.
Experiment 1a: Effect of NAc-D2R Antagonism on Motivation for Cocaine.
We targeted the core region of the NAc for this experiment since this region is critical for motivating cocaine use following both ShA and ExA self-administration (Doyle et al. 2014; Ramôa et al. 2014), and an important point of interaction between DAergic and glutamatergic signaling (Kalivas and McFarland 2003). Following ShA (n=37) or ExA (n=31) self-administration, rats were implanted with a bilateral infusion cannula aimed at the NAc core (+1.2 mm anterior-posterior, ±1.5 mm mediolateral, −5.7 mm dorsoventral) on abstinence day 8 or 9 (Doyle et al. 2014; Ramôa et al. 2014). Motivation for cocaine was examined following the 14th day of abstinence using a PR schedule and methods previously described (Doyle et al. 2014; Ramôa et al. 2014). A moderate-to-high cocaine dose (0.5 mg/kg/infusion) was selected based on our findings showing that motivation markedly increases for this dose following abstinence from ExA self-administration (Doyle et al. 2014; Ramôa et al. 2014).
Effects of NAc-D2R antagonism were examined once PR responding stabilized (no increasing or decreasing trend in the number of infusions obtained over three consecutive sessions) using a within-subject design (see Fig. 1a). Doses (0.3, 1,3, 10-μg/side) were based on previous studies using intra-NAc infusion and selected to induce threshold versus maximal, but selective effects (i.e. no effects on locomotion or non-specific responding, Bachtell et al. 2005; Bari and Pierce 2005). However, because our initial analysis revealed a decrease in responding for both the ShA and ExA groups at the lowest dose (0.3-μg/side), we expanded the dose range to include a lower dose (0.1-μg/side). Based on a power analysis using data from our initial analysis (α=0.05), this dose was examined in 10 ShA and 9 ExA rats, and in order to compare this dose to the others, it was run contemporaneously with the other doses with dose order counter-balanced between subjects. To do so, it was necessary to use a larger n for the other doses (0, 0.3, 1.0, 3.0, and 10-μg; ShA, n=21, 23, 21, 20, 20, respectively; ExA, n=15, 16, 16, 18, 19, respectively).
Infusions were administered immediately before the PR test session (0.5 μl/side; 2-min), and a minimum of three stable PR sessions separated each test session (Doyle et al. 2014; Ramôa et al. 2014). Due to catheter patency issues, not all rats received all six doses (average treatments rat, 3.1±0.2 for the ShA group and 3.0 ±0.3 for the ExA group). Cannula placement was confirmed using methods previously described (Doyle et al. 2014; Ramôa et al. 2014). Placement was within the NAc core (Fig. 1c–d) for all but six rats who were excluded from subject numbers and analyses.
Experiment 1b: Effect of NAc-D2R Antagonism on Motivation for Sucrose.
As a test of selectivity for the effects of the intra-NAc infusion of eticlopride, we examined its effect on motivation for sucrose using methods previous described (Doyle et al. 2014). Briefly, following sucrose lever-press self-administration training as described above, rats underwent a 14-day “abstinence” period with stereotaxic surgery occurring on abstinence day 8 or 9 as described above. PR responding for sucrose began on abstinence day 15, and once stable, effects of eticlopride were examined as described above for cocaine except that, in this experiment, all rats received all doses of eticlopride (0, 0.3, 1, 3, 10-μg/side) except the lowest dose (0,1-μg/side), which was not examined in this experiment. Based on a power analysis and our previous studies with sucrose self-administration (Doyle et al. 2014), effects were examined in 7 rats. As with the cocaine groups, sucrose controls had ad libitum access to food. Cannula placement was within the NAc core for all subjects (Fig. 1e).
Experiment 1c: Effect of ExA versus ShA Self-Administration on NAc Gene Expression.
Changes in NAc-DA receptor gene expression were examined for D1R (Drd1), as well as D2R and D3R (Drd2/3), in additional groups of rats tested on PR responding for cocaine following ShA versus ExA cocaine self-administration (see Fig. 1b). In order to establish cocaine-induced changes, additional groups of rats were given access to saline under the same conditions as used for ShA (n=5) and ExA (n=4) self-administration; however, since no behavioral or molecular differences were observed, data were combined. Based on a power analysis and our previous data (Abel et al. 2019), effects were examined in a minimum of 7 rats/group (n=9, 8, and 8 for the saline, ShA, and ExA group, respectively). Motivation for cocaine/saline was assessed following 14-days of abstinence as described above with brain tissue obtained following the third stable PR session (~18 hours after the last infusion) using methods previously described (Abel et al. 2019). The NAc core was dissected from whole frozen brains and stored at −80 °C until processed for RT-qPCR.
RNA extraction, cDNA transcription, and RT-qPCR were performed as previously described (Smith et al. 2018). Oligonucleotide primers were chosen from prior publications and synthesized by Invitrogen (Carlsbad, CA). cDNA templates were normalized with Gapdh, which was prescreened to assure similar expression between groups. One data point from the ShA group for the Drd1 expression was not detected and one data point from the ExA group for Drd1 expression was as an extreme outlier (>2.5 standard deviations from the mean); these data were excluded from the Drd1 analysis (n=7/group).
Experiment 2: Effect of Systemic D1R and D2R and AMPA-R Antagonism on Motivation for Cocaine.
As with the site-specific study, in this study using systemic manipulation, effects were examined following abstinence from ExA (n=16) versus ShA (n=13) self-administration once PR responding stabilized using a within-subject design (Fig. 1a–b). Doses of eticlopride (0.05 and 0.1 mg/kg), the D1R antagonist SCH-23390 (0.5 and 1.0 mg/kg), and the AMPA-R receptor antagonist CNQX (1.5 mg/kg) were based on previous studies (Backstrom and Hyytia 2003; 2006; Baker et al. 1998; Murschall and Hauber 2005; Palmatier et al. 2014; Schenk and Gittings 2003) and were selected to modestly (SCH-23390 and eticlopride) versus robustly, but selectively (SCH-23390, eticlopride, and CNQX), impact cocaine self-administration. Although only one dose of CNQX was tested, the moderate-to-high dose selected has been reported to maximally, but selectively, impact behavior; it was not possible to test a higher dose since these doses have non-specific effects (e.g. locomotor behavior, Backstrom and Hyytia 2003; 2006). Effects were compared to an equal volume of vehicle treatment, and based on a power analysis and our previous data (Doyle et al. 2014), were examined in a minimum of 8 rats/treatment-dose (vehicle, 0.05 and 0.1 eticlopride, 0.5 and 1.0 SCH-23390, and 1.5 CNQX; ShA, n=10, 9 and 11, 10 and 9, and 12, respectively; ExA, n=8, 9 and 12, 12 and 11, and 11, respectively). Treatments were administered intraperitoneally 15-min prior to the test session. A minimum of three stable PR sessions separated each test session. The order of drug/dose administration was counterbalanced between subjects. Due to challenges in maintaining catheter patency, not all rats received all six treatments doses (average treatments/rat, 4.1±0.4 for the ShA group and 4.0 ±0.4 for the ExA group.
Drugs.
Cocaine ([−]-cocaine hydrochloride) was obtained from the National Institute on Drug Abuse (NIDA; Research Triangle Park, NC), dissolved in sterile saline, filtered, and stored at 4°C. The infusion duration was adjusted three times/week based on body weight (2-sec/100 g/kg). SCH-23390, eticlopride, and CNQX were purchased from Sigma (St. Louis, MO) and dissolved in sterile water (vehicle).
Data Analysis.
The development of an addiction-like phenotype in the ExA group, as defined by an enhanced motivation for cocaine relative to the ShA group, was examined by comparing the number of infusions obtained during the first three stable PR sessions between groups using repeated measures ANOVA. The effect of NAc-D2R antagonism on motivation for cocaine was assessed by comparing the number of cocaine infusions obtained at baseline (averaged across the three stable PR sessions that proceeded the treatment session) to those obtained during the treatment session using repeated measures ANOVA with group and dose between-subject factors. A similar repeated measures ANOVA analysis was used to examine the effect of eticlopride on motivation for sucrose in Experiment 1b and the effect of systemic eticlopride, SCH-23390, and CNQX treatment in Experiment 2 with treatment/dose replacing dose as a between-subjects factor. Given baseline differences in the number of cocaine infusions obtained between the ExA and ShA groups, the effects of NAc-eticlopride and systemic eticlopride, SCH-23390, and CNQX were also examined as percent change from the baseline number of infusions using univariate ANOYA with group and dose as between subject factors (Experiment 1a) and group, drug, and dose (0, low, high, CNQX; Experiment 2). Univariate ANOYA was also used to assess group differences in DA receptor gene expression in Experiment 1c. Post-hoc comparisons to baseline/0 (no change) or vehicle were made using either one-tailed (for stated predicted differences) or two-tailed Bonferroni-corrected t-tests. All data were normally distributed and analyzed using SPSS-25. Differences were considered significant at P<0.05.
Results
Experiment 1a: Effect of NAC-D2R Antagonism on Motivation for Cocaine
Motivation for Cocaine.
Rats in the ExA group self-administered a high level of cocaine (68.3±1.1 infusions/day versus 20 infusions/day in the ShA), and as expected, following abstinence, obtained more infusions of cocaine under the PR schedule as compared to ShA controls (Fig. 2a; group: F(1,66)=22.8, P<0.001); this was a stable difference and similar for each of the first three stable sessions (session and session-by-group, P’s>0.05). Thus, following ExA self-administration and abstinence, motivation for cocaine was markedly higher (~35%) in the ExA versus ShA group confirming the development of an addiction-like phenotype in the ExA group.
Fig. 2. NAc-D2R antagonism decreased motivation for cocaine in both the extended-access and short-access groups.
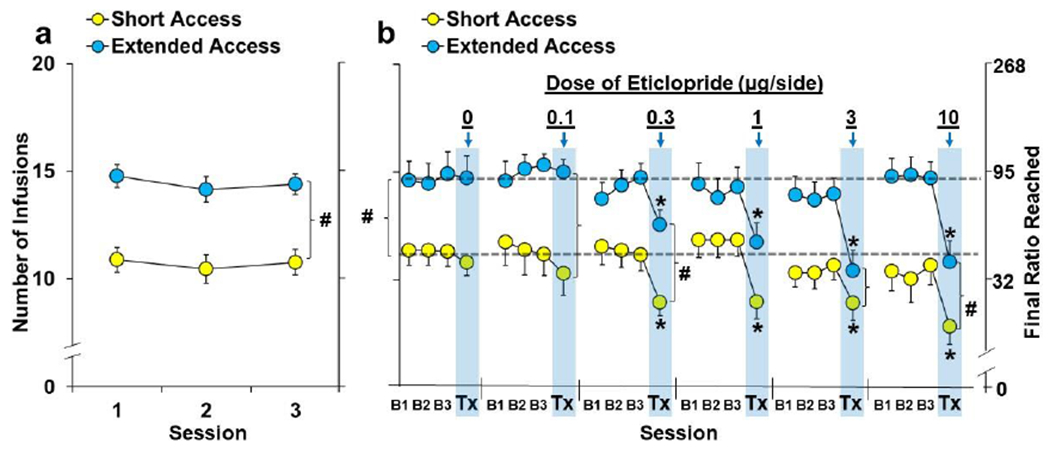
Data are plotted as mean (± SEM) number of infusions obtained and the corresponding final ratios completed for the short-access (n=37) and extended-access (n=31) groups for (a) the first three stable PR sessions and (b) the three baseline sessions prior treatment (B1, B2, B3) and the day of treatment (Tx; arrows and shaded bars) as a function of eticlopride dose (0, 0.1, 0.3, 1, 3, 10-μg/side; short-access, n=21, 10, 23, 21, 20, 20, respectively; extended-access, n=15, 9, 16, 16, 18, 19, respectively). A (#) indicates a significant difference between the extended- versus short-access groups during the first three baseline sessions (a), averaged across the pretreatment baselines (b; represented by dotted lines), and following treatment with low (0.1-0.3-μg) or high doses (3-10-μg) of eticlopride (b). An (*) indicates a significant difference from baseline (b).
Effect of NAc-D2R Antagonism.
As with the first three stable sessions, the average number of infusions obtained wider the PR schedule at baseline prior to each of the eticlopride treatments was higher in the ExA versus ShA group (14.4±0.3 versus 11.2±0.3; P<0.001). Contrary to our hypothesis, NAc-D2R antagonism impacted PR responding for cocaine for both the ShA and ExA groups although some group differences were observed (Fig. 2b). A repeated-measures ANOVA comparing the number of infusions obtained at baseline to the treatment session revealed significant effects of session (F(1,196)=199.6, P<0.001), group (F(1,196)=48.0, P<0.001), and session-by-dose (F(5,196)=13.8, P<0.001). We also observed a significant interaction of session-by-dose-by-group (F(5,196)=3.6, P<0.01), which appears to be due greater decreases in the ShA group following treatment with low doses of eticlopride (0.1-0.03 μg side: session-by-group, F(1,56)=4.0, P<0.05) and to greater decreases in the ExA group following treatment with high doses of eticlopride (3-10 μg/side; session-by-group, F(1,75)=9.0, P<0.01). However, subsequent analysis within each of the groups revealed similar dose-dependent changes following eticlopride treatment. Specifically, analysis within the ShA group revealed significant effects of session (F(1,109)=110.3, P<0.001) and session-by-dose (F(5,109)=5.1, P<0.001), and post-hoc comparisons to baseline within each dose revealed a significant difference for each of the treatments (P’s<0.05) except vehicle (P>0.05) and the lowest dose (0.1 μg/side; P>0.05). Similarly, analysis within the ExA group revealed significant effects of session (F(1,90)=95.7, P<0.001) and session-by-dose (F(5,90)=21.2, P<0.001), and post-hoc comparisons to baseline within each dose revealed significant effects for each of the treatments (P’s<0.05) except vehicle (P>0.05) and the lowest dose (0.1-μg side; P>0.05).
To further assess differential effects of D2R antagonism between the ShA and ExA groups, we examined effects relative to baseline (i.e., percent change from baseline; Fig. 3). As with the previous analysis, this analysis revealed a significant effect of dose F(5,196)=16.8, P<0.05) as well as a dose-by-group interaction F(5,196)=2.4, P<0.05) that appears to be due to greater decreases in the ShA group following treatment with low doses of eticlopride (0.1-0.3 μg/side; p<0.01). However, in contrast to the previous analysis, effects at higher doses (3-10 μg/side; p<0.01) did not differ significantly between the ShA and ExA groups indicating that differences at higher doses are modest and variable. The effects of the moderate dose (1 μg/side) did not differ between the groups (P>0.05). Analysis within the ShA group revealed an overall effect of dose (F(5,109)=5.9, P<0.001), and post-hoc comparisons to vehicle were significant for each of the treatments (P’s<0.05) except the lowest dose (0.1 μg/side; P<0.05). Post-hoc comparisons to the lowest dose (0.1 μg/side) revealed a significant difference for the 1 and 10-μg doses (P’s<0.05), but not for the 0.3 or 3-μg dose (P>0.05). Each of the higher doses were statistically similar (P’s>0.05) indicating that increases in eticlopride dose beyond 0.3-μg did not further enhance its effects. Analysis within the ExA group also revealed a significant effect of dose (F(5, 87)=10.57, P<0.001), with post-hoc comparisons to vehicle revealing a significant difference for the 0.3, 1, 3, and 10-μg doses (P’s<0.05), but not for the 0.1-μg dose (P>0.05). Post-hoc comparisons to the 0.1-μg dose revealed a significant difference for the 1, 3, and 10-μg doses (P’s<0.05), but not the 0.3-μg dose (P>0.05). The 3 and 10-μg doses also differed from the 0.3-μg dose (P’s<0.05). However, no differences were observed between the three highest doses (1,3, and 10-μg; P’s>0.05), indicating that increases in eticlopride dose beyond 1.0-μg did not further enhance its effects. Thus, intra-NAcc infusion of eticlopride decreased PR responding for cocaine in the both ShA and ExA groups although effects at low doses were more pronounced in the ShA versus ExA groups. Higher doses of eticlopride also tended to induce greater effects in the ExA versus ShA group, but these effects were variable. Increases beyond the 0,3-μg dose also further enhanced the efficacy of eticlopride within the ExA, but not ShA group.
Fig. 3. Differential effects of NAc-D2R antagonism on motivation for cocaine between the extended- and short-access groups.
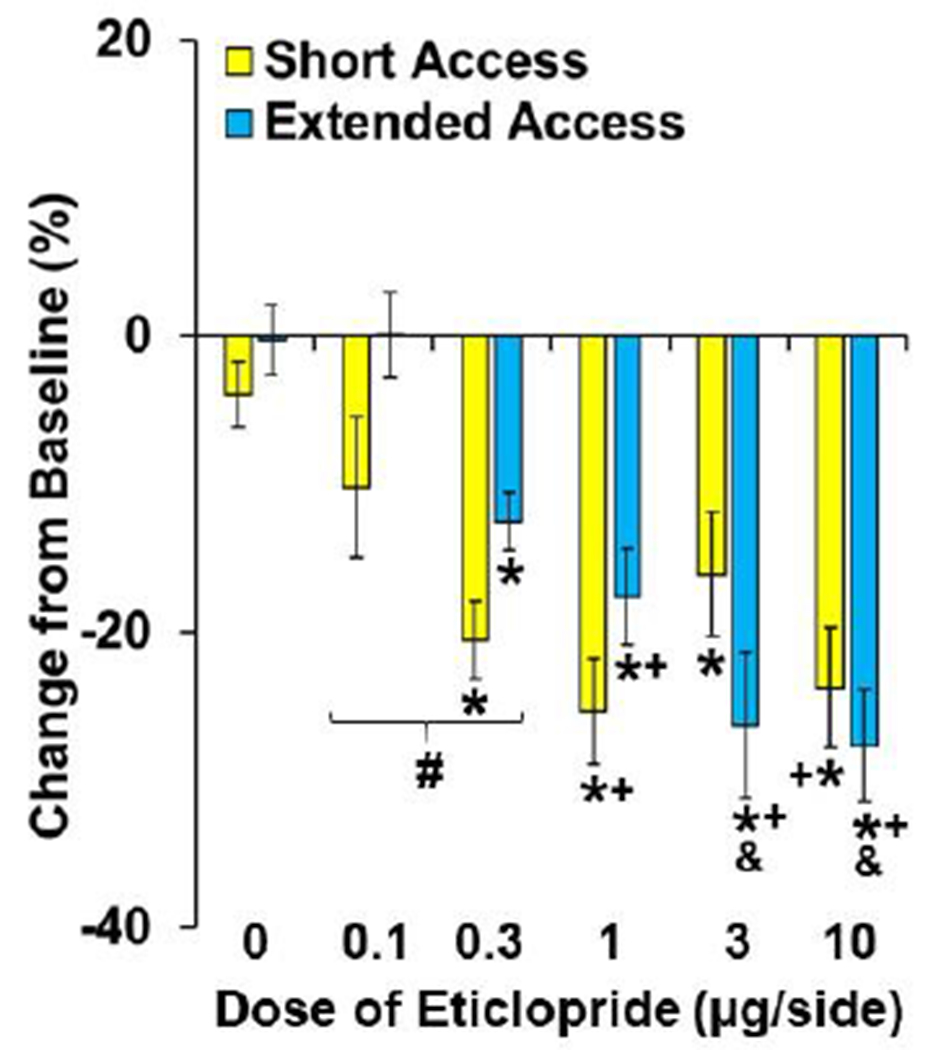
Data are plotted as percent change (± SEM) from the baseline number of infusions obtained by the short- and extended-access groups following treatment with eticlopride (0, 0.1, 0.3, 1, 3, and 10-μg/side; short-access, n=21, 10, 23, 21, 20, 20, respectively; extended-access, n=15, 9, 16, 16, 18, 19, respectively). A (#) indicates a significant difference between the extended- versus short-access groups following treatment with low doses (0.1-0.3-μg) of eticlopride. An (*) indicates a significant difference from vehicle (0-μg), a (+) indicates a significant difference from the 0.1-μg dose, and (&) indicates a significant difference from the 0.3-μg dose.
Experiment 1b: Effect of NAc-D2R Antagonism on Motivation for Sucrose.
No significant overall or interactive effects of eticlopride were observed for PR responding for sucrose (P’s>0.05) when analyzed as number of deliveries at baseline versus the treatment session (Fig. 4) or change from baseline deliveries. Thus, eticlopride did not affect general motivational behaviors.
Fig. 4. No effect of NAc-D2R antagonism on motivation for sucrose pellets.
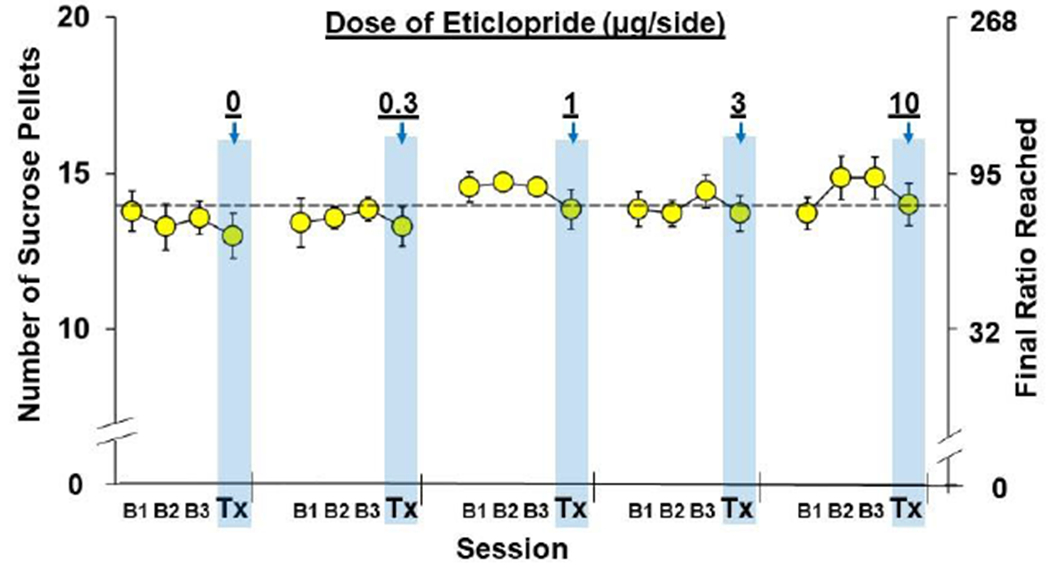
(a) Data are plotted as mean (± SEM) number of sucrose pellets obtained and the corresponding final ratios completed for the three baseline sessions prior treatment (B1, B2, B3) and the day of treatment (Tx; arrows and shaded bars) as a function of eticlopride dose (0, 0.3, 1, 3, 10-μg/side; n=7).
Experiment 1c: Effect of ExA versus ShA Self-Administration on NAc Gene Expression.
Motivation for Cocaine.
As in Experiment 1a, in this experiment rats in the ExA group self-administered high levels of cocaine (average of 70.3±1.6 infusions/day versus 20 infusion/day in the ShA), and following abstinence, obtained significantly more infusions of cocaine under the PR schedule than rats in the ShA group (Fig. 5a; group: F(1,14)=6.3, P<0.05); this difference was similar for each of the three stable baseline sessions (session and session-by-group, P’s>0.05). Thus, following ExA self-administration and abstinence, motivation for cocaine was markedly higher (~36%) in the ExA versus ShA group.
Fig. 5. Effect of short- versus extended-access cocaine self-administration on NAc-dopamine receptor gene expression.
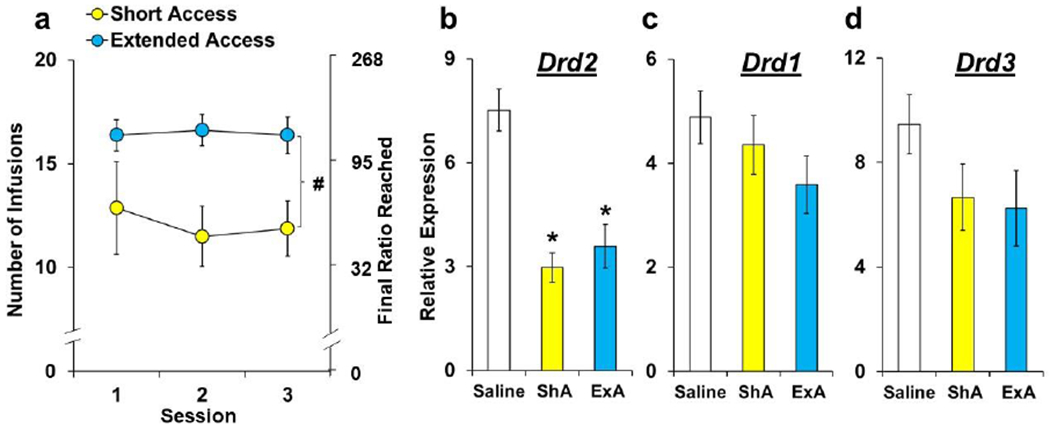
(a) Data are plotted as mean (± SEM) number of cocaine infusions obtained and the corresponding final ratios completed during the first three stable PR sessions following short- (n=8) versus extended-access (n=8) cocaine self-administration and 14-days of abstinence. Data are plotted as mean (± SEM) relative expression levels of Drd1 (b), Drd2 (c), and Drd3 (d) in the NAc of rats in the saline (n=9), short-access (ShA; n=7-8), and extended-access (ExA; n=7-8) self-administration groups. A (#) indicates a significant difference between the extended- and short-access groups. An (*) indicates a significant difference from saline controls.
Gene Expression Changes.
NAc-D2 receptor gene expression (Drd2) was decreased in rats tested following saline versus ShA and ExA cocaine self-administration (Fig. 5b; group, F(2,22)=19.4, P<0.001; versus saline, P’s<0.001). NAc-D1 receptor gene expression (Drd1) also tended to be decreased following cocaine versus saline self-administration, particularly following ExA self-administration (Fig. 5c); however, expression levels were more variable and the overall effect of group did not reach statistical significance (p=0.062). No significant differences were observed for NAc-D3 receptor gene expression (Drd3; Fig. 5d).
Experiment 2: Effect of Systemic D1R and D2R and AMPA-R Antagonism on Motivation for Cocaine
Motivation for Cocaine.
As in Experiment 1, in this experiment, the ExA group self-administered a high level of cocaine (average of 65.9±1.5 infusions/day versus 20 infusion/day in the ShA), and following abstinence, obtained significantly more infusions of cocaine under the PR schedule as compared to the ShA group (Fig. 6a; group: F(1,29)=11.5, P<0.01). This difference was similar for each of the first three stable sessions (session-by-group, P>0.05). Thus, following ExA self-administration and abstinence, motivation for cocaine was markedly higher (~31%) in the ExA versus ShA group.
Fig. 6. Systemic D1R, D2R, and AMPA-R antagonism differentially impact motivation for cocaine in the extended- versus short-access groups.
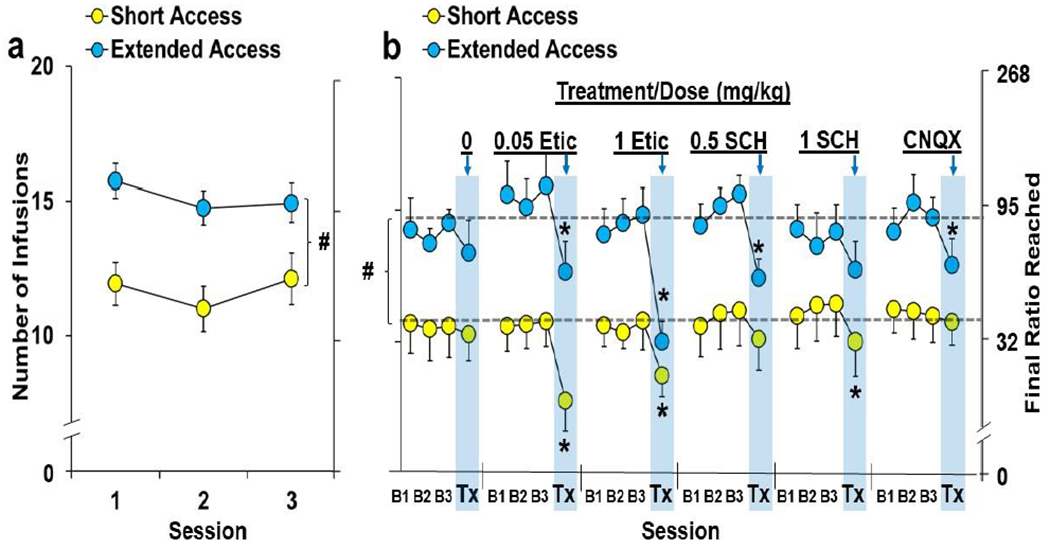
Data are plotted as mean (± SEM) number of infusions obtained and the corresponding final ratios completed for the short-access (n=15) and extended-access (n=16) groups for (a) the first three stable PR sessions and (b) the three baseline sessions prior treatment (B1, B2, B3) and the day of treatment (Tx; arrows and shaded bars) as a function of dose (mg/kg)/treatment (0/Vehicle, 0.05 and 0.1 eticlopride/Etic, 0.5 and 1.0 SCH-23390/SCH, and 1.5 CNQX; short-access, n=10, 9 and 11, 10 and 9 and 12, respectively; extended-access, n=8, 9 and 12, 12 and 11, and 11, respectively). A (#) indicates a significant difference between the extended- versus short-access groups during the first three baseline sessions (a) and averaged across the pretreatment baselines (b; represented by dotted lines). An (*) indicates a significant difference from baseline (b).
Effects of Systemic D1R and D2R and AMPA-R Antagonism.
As with the first three stable sessions, baseline PR responding prior to each of the systemic treatments was higher in the ExA versus ShA group (14.7±0.4 versus 10.9±0.4; P<0.001; Fig. 6b). As expected, systemic administration of eticlopride, SCH-23390, and CNQX differentially impacted motivation for cocaine in the ShA versus ExA groups. A repeated-measures ANOVA comparing the number of infusions obtained at baseline versus the treatment session revealed significant overall effects of session (F(1,112)=105.5, P<0.001) and group (F(1,112)=34.5, P<0.001), as well as significant interactions of session-by-drug/dose (F(5,112)=5.8, P<0.001) and session-by-group (F(1,112)=8.7, P<0.01). Analysis within the ShA group revealed significant effects of session (F(1,55)=35.1, P<0.001) and session-by-drug/dose (F(5,55)=3.4, P<0.01), with post-hoc comparison to baseline revealing a significant difference for the low and high dose of eticlopride and the high dose of SCH-23390 (P’s<0.05). Analysis within the ExA group also revealed significant effects of session (F(1,57)=71.4, P<0.001), and session-by-drug/dose (F(5,57)=4.9, P<0.01), with post-hoc comparison to baseline revealing a significant decrease for each of the treatments (P’s<0.05) except vehicle and the high dose of SCH-23390 (P>0.05).
Group differences in the effects of systemic administration of eticlopride, SCH-23390, and CNQX were further examined by assessing changes relative to the baseline number of infusions obtained (Fig. 7). This analysis revealed a significant effect of drug (F(1,112)=12.5, P<0.01) as well as a significant interaction of group-by-drug-by-dose (F(1,112)=7.4, P<0.01). Subsequent analysis within each drug revealed a significant interaction of group-by-dose for eticlopride (F(1,37)=5.1, P<0.05), which appears to be due to a greater effect of the high dose in the ExA versus ShA group (P<0.05), and a significant effect of group for CNQX (t21=3.8, P<0.05), which decreased the number of infusions obtained in the ExA group (versus 0; P<0.05) but not the ShA group (P>0.05). While the effects of the high dose of eticlopride were more pronounced in the ExA versus ShA group, both the low and the high dose decreased PR cocaine self-administration in both groups (versus 0; P’s<0.05). SCH-22390 also decreased the number of cocaine infusions obtained (versus 0; P<0.05) although effects were modest and did not differ between groups or doses. No significant effects were observed for vehicle. Thus, while systemic administration of eticlopride, SCH-23390, and CNQX decreased motivation for cocaine, effects of eticlopride were more pronounced in the ExA versus ShA group (at the high dose), effects of SCH-23390 were modest, and effects of CNQX were apparent in the ExA, but not ShA, group.
Fig. 7. Differential effects of systemic D2R and AMPA-R antagonism on motivation for cocaine between the extended- and short-access groups.
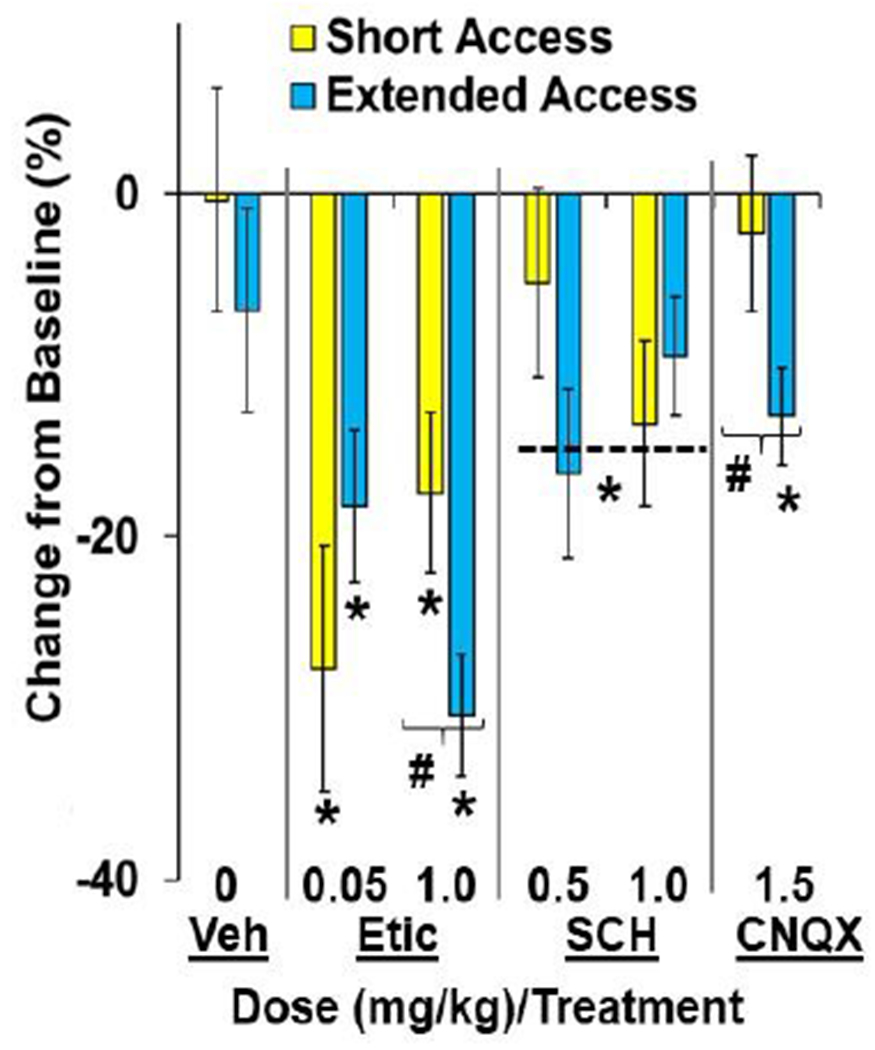
Data are plotted as percent change (± SEM) from the baseline number of infusions obtained by the short- and extended-access groups following systemic treatment as a function of dose (mg/kg)/treatment (0/Vehicle, 0.05 and 0.1 eticlopride, 0.5 and 1.0 SCH-23390, and 1.5 mg/kg CNQX; short-access, n=10, 9 and 11, 10 and 9, and 12, respectively; extended-access, n=8, 9 and 12, 12 and 11, and 11, respectively). A (#) indicates a significant difference between the extended- versus short-access groups following treatment with the high dose of eticlopride (1 mg/kg) and CNQX. An (*) indicates a significant difference from baseline/0 (no change).
Discussion
We previously showed that an enhanced motivation for cocaine develops over abstinence following ExA self-administration and corresponds to a shift in the mechanisms motivating cocaine use from NAc-D1R signaling to AMPA-R signaling (Doyle et al. 2014; Ramôa et al. 2014). In the present study, we built on these findings with the prediction that the role of NAc-D2R signaling in motivating cocaine use would also diminish with the development of the addiction-like phenotype. In contrast to this hypothesis, we found that NAc-D2R blockade decreased motivation to cocaine in both the ShA and ExA groups. We did, however, observe some group differences in the effects of D2R antagonism, in that low doses of eticlopride were more effective at decreasing motivation for cocaine in the ShA versus ExA group, and high doses of eticlopride tended to be more effective in the ExA versus ShA group. The effects of eticlopride appear to be specific to cocaine since no effects were observed in PR responding for sucrose. Together, these results indicate that unlike NAc-D1Rs, NAc-D2Rs continue to modulate motivation for cocaine following the development of an addiction-like phenotype. This interpretation is also consistent with our molecular findings showing that NAC-Drd2 is decreased from saline control levels in both the ShA and ExA groups. Our second objective was to determine whether mechanistic shifts impact the response to systemically administered treatments. The results with AMPA-R receptor antagonism manipulation are consistent with our previous findings following NAc-specific manipulation and show decreased PR responding for cocaine in the ExA, but not ShA group. Like NAc-specific manipulation, systemic D2R antagonism decreased motivation for cocaine in both the ShA and ExA groups and its efficacy differed between the groups. In contrast to effects observed previous with NAc-specific manipulation, systemic manipulation of D2R similarly decreased motivation for cocaine in the ShA and ExA groups. These findings indicate that the mechanisms motivating cocaine use change with the development of an addiction-like phenotype and impact the response to both site-specific and systemic drug administration.
Effect of NAc-D2R Antagonism on Motivation for Cocaine.
While NAc-infused eticlopride decreased motivation for cocaine in both ShA and ExA groups, low doses of NAc-infused eticlopride were less effective in the ExA versus ShA group. Based on the assumption that low doses are more selective and induce fewer side effects than high doses, it is possible that this finding reflects a general decrease in sensitivity to D2R antagonism in the ExA group. This interpretation is also consistent with our findings showing that a maximal, or “ceiling” effect of eticlopride occurred at a lower dose in the ShA versus ExA group (0.3 versus 1.0-μg/side). It is also consistent with previous results showing that NAc-DA terminals and autoreceptors are less sensitive following ExA versus ShA self-administration (Mateo et al. 2005). One caveat to this conclusion, however, is that the high dose of NAc-infused eticlopride tended to induce an even greater decrease in PR responding for cocaine in the ExA versus ShA group although these effects were variable.
None-the-less, given that multiple doses of NAc-infused eticlopride decreased PR responding for cocaine in the ExA group, it is likely that, unlike NAc-D1Rs (Ramôa et al. 2014), D2Rs remain involved following the development of an addiction-like phenotype. This interpretation is supported by our gene expression data showing that NAc-Drd2 expression is decreased in both ShA and ExA groups, and is consistent with results showing lasting alterations in NAc-D2R availability in individuals with a substance use disorder, including cocaine (Martinez et al. 2009a; Volkow et al. 1993; Volkow et al. 1990; Volkow et al. 1997).
Effect of Systemic DA D1R, D2R, AMPA-R Antagonism on Motivation for Cocaine.
Systemic treatment with eticlopride and CNQX differentially impacted motivation for cocaine in the ExA versus ShA groups. These differential effects are consistent with several recent studies showing that neurobiological changes in discrete brain regions are different following the ShA versus ExA self-administration and translate to differential effects following systemic administration (Ahmed and Koob 2004; Allain and Samaha 2019; Ben-Shahar et al. 2007; Boudreau and Wolf 2005; Briand et al. 2008; Conrad et al. 2008; Fischer et al. 2013; Greenwell et al. 2009; Hao et al. 2010; Nicolas et al. 2017). As expected, systemic administration of the AMPA-R antagonist CNQX decreased PR responding in the ExA, but not the ShA group. This finding is consistent with effects following NAc manipulation (Doyle et al. 2014), and with a large body of research indicating that changes in the role of AMPA-R signaling with the development of an addiction-like phenotype reflect a general shift (for review see Amato et al. 2007). Our findings with eticlopride also show similar patterns of changes following NAc-specific and systemic manipulation in that both routes of administration decreased motivation for cocaine in both the ShA and ExA groups; differential effects were also observed between the ShA and ExA groups following both NAc-specific and systemic manipulation. While these findings are surprising considering the lack of efficacy of DA receptor antagonists in humans with a cocaine use disorder, it is important to note that many of these compounds lack efficacy due to issues with compliance (Amato et al. 2007). In contrast, our findings showing that systemic D1R manipulation similarly affected motivation for cocaine in the ShA and ExA groups is in contrast to our previous findings showing that NAc-specific manipulation impacts motivation for cocaine in the ShA, but not ExA group. These results suggest that changes in other brain regions (e.g., prefrontal cortex, ventral tegmental area) may counter shifts in NAc-D1R signaling with the development of an addiction-like phenotype. Future research is necessary to investigate this possibility.
Translational Implications and Conclusions.
These findings demonstrate differential effects of site-specific and systemic pharmacological manipulations following ShA versus ExA self-administration and strongly indicate the need for the use of animal models that induce an addiction-like phenotype for studies examining mechanistic changes and potential treatments for cocaine use disorder. Studies are currently underway to determine whether similar effects also occur in females tested following ShA versus ExA cocaine self-administration. This is important since women and female animals develop addiction/an addiction-like phenotype more rapidly than do men and male animals.
Acknowledgements:
We would like to acknowledge Lasya Pidaparthi for technical assistance. This study was supported by the National Institute on Drug Abuse (2R01DA024716; WJL).
Footnotes
Publisher's Disclaimer: This Author Accepted Manuscript is a PDF file of a an unedited peer-reviewed manuscript that has been accepted for publication but has not been copyedited or corrected. The official version of record that is published in the journal is kept up to date and so may therefore differ from this version.
Conflict of Interest:
The authors declare that they have no conflict of interest.
References
- Abel JM, Nesil T, Bakhti-Suroosh A, Grant PA, Lynch WJ (2019) Mechanisms underlying the efficacy of exercise as an intervention for cocaine relapse: a focus on mGlu5 in the dorsal medial prefrontal cortex. Psychopharmacology (Berl) 236: 2155–2171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmed SH, Koob GF (2004) Changes in response to a dopamine receptor antagonist in rats with escalating cocaine intake. Psychopharmacology (Berl) 172: 450–4. [DOI] [PubMed] [Google Scholar]
- Allain F, Samaha AN (2019) Revisiting long-access versus short-access cocaine self-administration in rats: intermittent intake promotes addiction symptoms independent of session length. Addict Biol 24: 641–651. [DOI] [PubMed] [Google Scholar]
- Amato L, Minozzi S, Pani PP, Davoli M (2007) Antipsychotic medications for cocaine dependence. Cochrane Database Syst Rev: CD006306. [DOI] [PubMed] [Google Scholar]
- American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders. (5th ed.). American Psychiatric Publishing, Arlington VA. [Google Scholar]
- Bachtell RK, Whisler K, Karanian D, Self DW (2005) Effects of intra-nucleus accumbens shell administration of dopamine agonists and antagonists on cocaine-taking and cocaine-seeking behaviors in the rat. Psychopharmacology (Berl) 183: 41–53. [DOI] [PubMed] [Google Scholar]
- Backstrom P, Hyytia P (2003) Attenuation of cocaine-seeking behaviour by the AMPA/kainate receptor antagonist CNQX in rats. Psychopharmacology (Berl) 166: 69–76. [DOI] [PubMed] [Google Scholar]
- Backstrom P, Hyytia P (2006) Ionotropic and metabotropic glutamate receptor antagonism attenuates cue-induced cocaine seeking. Neuropsychopharmacology 31: 778–86. [DOI] [PubMed] [Google Scholar]
- Baker DA, Fuchs RA, Specio SE, Khroyan TV, Neisewander JL (1998) Effects of intraaccumbens administration of SCH-23390 on cocaine-induced locomotion and conditioned place preference. Synapse 30: 181–93. [DOI] [PubMed] [Google Scholar]
- Bari AA, Pierce RC (2005) D1-like and D2 dopamine receptor antagonists administered into the shell subregion of the rat nucleus accumbens decrease cocaine, but not food, reinforcement. Neuroscience 135: 959–68. [DOI] [PubMed] [Google Scholar]
- Ben-Shahar O, Keeley P, Cook M, Brake W, Joyce M, Nyffeler M, Heston R, Ettenberg A (2007) Changes in levels of D1, D2, or NMDA receptors during withdrawal from brief or extended daily access to IV cocaine. Brain Res 1131: 220–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben-Shahar O, Posthumus EJ, Waldroup SA, Ettenberg A (2008) Heightened drug-seeking motivation following extended daily access to self-administered cocaine. Prog Neuropsychopharmacol Biol Psychiatry 32: 863–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bibb JA, Snyder GL, Nishi A, Yan Z, Meijer L, Fienberg AA, Tsai LH, Kwon YT, Girault JA, Czernik AJ, Huganir RL, Hemmings HC Jr., Nairn AC, Greengard P (1999) Phosphorylation of DARPP-32 by Cdk5 modulates dopamine signalling in neurons. Nature 402: 669–71. [DOI] [PubMed] [Google Scholar]
- Binswanger IA, Stern MF, Deyo RA, Heagerty PJ, Cheadle A, Elmore JG, Koepsell TD (2007) Release from prison--a high risk of death for former inmates. N Engl J Med 356: 157–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Binswanger IA, Nowels C, Corsi KF, Glanz J, Long J, Booth RE, Steiner JF (2012) Return to drug use and overdose after release from prison: a qualitative study of risk and protective factors. Addict Sci Clin Pract 7: 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bornebusch AB, Fink-Jensen A, Wortwein G, Seeley RJ, Thomsen M (2019) Glucagon-Like Peptide-1 Receptor Agonist Treatment Does Not Reduce Abuse-Related Effects of Opioid Drugs. eNeuro 6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boudreau AC, Wolf ME (2005) Behavioral sensitization to cocaine is associated with increased AMPA receptor surface expression in the nucleus accumbens. J Neurosci 25: 9144–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briand LA, Flagel SB, Garcia-Fuster MJ, Watson SJ, Akil H, Sarter M, Robinson TE (2008) Persistent alterations in cognitive function and prefrontal dopamine D2 receptors following extended, but not limited, access to self-administered cocaine. Neuropsychopharmacology 33: 2969–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conrad KL, Tseng KY, Uejima JL, Reimers JM, Heng LJ, Shaham Y, Marinelli M, Wolf ME (2008) Formation of accumbens GluR2-lacking AMPA receptors mediates incubation of cocaine craving. Nature 454: 118–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conrad KL, Ford K, Marinelli M, Wolf ME (2010) Dopamine receptor expression and distribution dynamically change in the rat nucleus accumbens after withdrawal from cocaine self-administration. Neuroscience 169: 182–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Lima MS, de Oliveira Soares BG, Reisser AA, Farrell M (2002) Pharmacological treatment of cocaine dependence: a systematic review. Addiction 97: 931–49. [DOI] [PubMed] [Google Scholar]
- Deroche-Gamonet V, Belin D, Piazza PV (2004) Evidence for addiction-like behavior in the rat. Science 305: 1014–7. [DOI] [PubMed] [Google Scholar]
- Doyle SE, Ramôa C, Garber G, Newman J, Toor Z, Lynch WJ (2014) A shift in the role of glutamatergic signaling in the nucleus accumbens core with the development of an addicted phenotype. Biol Psychiatry 76: 810–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ducret E, Puaud M, Lacoste J, Belin-Rauscent A, Fouyssac M, Dugast E, Murray JE, Everitt BJ, Houeto JL, Belin D (2016) N-acetylcysteine Facilitates Self-Imposed Abstinence After Escalation of Cocaine Intake. Biol Psychiatry 80: 226–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwards S, Whisler KN, Fuller DC, Orsulak PJ, Self DW (2007) Addiction-related alterations in D1 and D2 dopamine receptor behavioral responses following chronic cocaine self-administration. Neuropsychopharmacology 32: 354–66. [DOI] [PubMed] [Google Scholar]
- Fischer KD, Houston AC, Rebec GV (2013) Role of the major glutamate transporter GLT1 in nucleus accumbens core versus shell in cue-induced cocaine-seeking behavior. J Neurosci 33: 9319–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gentile TA, Simmons SJ, Barker DJ, Shaw JK, Espana RA, Muschamp JW (2018) Suvorexant, an orexin/hypocretin receptor antagonist, attenuates motivational and hedonic properties of cocaine. Addict Biol 23: 247–255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenwell TN, Funk CK, Cottone P, Richardson HN, Chen SA, Rice KC, Zorrilla EP, Koob GF (2009) Corticotropin-releasing factor-1 receptor antagonists decrease heroin self-administration in long-but not short-access rats. Addict Biol 14: 130–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haile CN, Kosten TR (2013) Pharmacotherapy for stimulant-related disorders. Curr Psychiatry Rep 15: 415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammad AM, Alasmari F, Althobaiti YS, Sari Y (2017) Modulatory effects of Ampicillin/Sulbactam on glial glutamate transporters and metabotropic glutamate receptor 1 as well as reinstatement to cocaine-seeking behavior. Behav Brain Res 332: 288–298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hao Y, Martin-Fardon R, Weiss F (2010) Behavioral and functional evidence of metabotropic glutamate receptor 2/3 and metabotropic glutamate receptor 5 dysregulation in cocaine-escalated rats: factor in the transition to dependence. Biol Psychiatry 68: 240–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hemby SE, Tang W, Muly EC, Kuhar MJ, Howell L, Mash DC (2005) Cocaine-induced alterations in nucleus accumbens ionotropic glutamate receptor subunits in human and non-human primates. J Neurochem 95: 1785–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- James MH, Stopper CM, Zimmer BA, Koll NE, Bowrey HE, Aston-Jones G (2019) Increased Number and Activity of a Lateral Subpopulation of Hypothalamic Orexin/Hypocretin Neurons Underlies the Expression of an Addicted State in Rats. Biol Psychiatry 85: 925–935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalivas PW, McFarland K (2003) Brain circuitry and the reinstatement of cocaine-seeking behavior. Psychopharmacology (Berl) 168: 44–56. [DOI] [PubMed] [Google Scholar]
- Kalivas PW, Volkow ND (2005) The neural basis of addiction: a pathology of motivation and choice. Am J Psychiatry 162: 1403–13. [DOI] [PubMed] [Google Scholar]
- Koob G, Kreek MJ (2007) Stress, dysregulation of drug reward pathways, and the transition to drug dependence. Am J Psychiatry 164: 1149–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loweth JA, Tseng KY, Wolf ME (2014) Adaptations in AMPA receptor transmission in the nucleus accumbens contributing to incubation of cocaine craving. Neuropharmacology 76 Pt B: 287–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynch WJ, Carroll ME (2001) Regulation of drug intake. Exp Clin Psychopharmacol 9: 131–43. [DOI] [PubMed] [Google Scholar]
- Lynch WJ (2018) Modeling the development of drug addiction in male and female animals. Pharmacol Biochem Behav 164: 50–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez D, Greene K, Broft A, Kumar D, Liu F, Narendran R, Slifstein M, Van Heertum R, Kleber HD (2009a) Lower level of endogenous dopamine in patients with cocaine dependence: findings from PET imaging of D(2)/D(3) receptors following acute dopamine depletion. Am J Psychiatry 166: 1170–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez D, Slifstein M, Narendran R, Foltin RW, Broft A, Hwang DR, Perez A, Abi-Dargham A, Fischman MW, Kleber HD, Laruelle M (2009b) Dopamine D1 receptors in cocaine dependence measured with PET and the choice to self-administer cocaine. Neuropsychopharmacology 34: 1774–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mateo Y, Lack CM, Morgan D, Roberts DC, Jones SR (2005) Reduced dopamine terminal function and insensitivity to cocaine following cocaine binge self-administration and deprivation. Neuropsychopharmacology 30: 1455–63. [DOI] [PubMed] [Google Scholar]
- McGregor A, Roberts DC (1993) Dopaminergic antagonism within the nucleus accumbens or the amygdala produces differential effects on intravenous cocaine self-administration under fixed and progressive ratio schedules of reinforcement. Brain Res 624: 245–52. [DOI] [PubMed] [Google Scholar]
- Meador-Woodruff JH, Little KY, Damask SP, Mansour A, Watson SJ (1993) Effects of cocaine on dopamine receptor gene expression: a study in the postmortem human brain. Biol Psychiatry 34: 348–55. [DOI] [PubMed] [Google Scholar]
- Morgan D, Brebner K, Lynch WJ, Roberts DC (2002) Increases in the reinforcing efficacy of cocaine after particular histories of reinforcement. Behav Pharmacol 13: 389–96. [DOI] [PubMed] [Google Scholar]
- Morgan D, Roberts DC (2004) Sensitization to the reinforcing effects of cocaine following binge-abstinent self-administration. Neurosci Biobehav Rev 27: 803–12. [DOI] [PubMed] [Google Scholar]
- Morgan D, Smith MA, Roberts DC (2005) Binge self-administration and deprivation produces sensitization to the reinforcing effects of cocaine in rats. Psychopharmacology (Berl) 178: 309–16. [DOI] [PubMed] [Google Scholar]
- Murschall A, Hauber W (2005) Effects of a systemic AMPA/KA and NMDA receptor blockade on pavlovian-instrumental transfer. Psychopharmacology (Berl) 182: 290–6. [DOI] [PubMed] [Google Scholar]
- Negus SS, Banks ML (2018) Modulation of drug choice by extended drug access and withdrawal in rhesus monkeys: Implications for negative reinforcement as a driver of addiction and target for medications development. Pharmacol Biochem Behav 164: 32–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neisewander JL, Cheung TH, Pentkowski NS (2014) Dopamine D3 and 5-HT1B receptor dysregulation as a result of psychostimulant intake and forced abstinence: Implications for medications development. Neuropharmacology 76 Pt B: 301–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicolas C, Tauber C, Lepelletier FX, Chalon S, Belujon P, Galineau L, Solinas M (2017) Longitudinal Changes in Brain Metabolic Activity after Withdrawal from Escalation of Cocaine Self-Administration. Neuropsychopharmacology 42: 1981–1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Brien C (2005) Drug addiction and drug abuse. In: Brunton LL, Lazo JS, Parker K (eds). Goodman and Gilman’s: The pharmacological basis of therapeutics. 2005. 11th edition. McGraw-Hill: New York, USA. pp 607–627. [Google Scholar]
- Oleson EB, Roberts DC (2008) Parsing the Addiction Phenomenon: Self-Administration Procedures Modeling Enhanced Motivation for Drug and Escalation of Drug Intake. Drug Discov Today Dis Models 5: 217–226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oleson EB, Roberts DC (2009) Behavioral economic assessment of price and cocaine consumption following self-administration histories that produce escalation of either final ratios or intake. Neuropsychopharmacology 34: 796–804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmatier MI, Kellicut MR, Brianna Sheppard A, Brown RW, Robinson DL (2014) The incentive amplifying effects of nicotine are reduced by selective and non-selective dopamine antagonists in rats. Pharmacol Biochem Behav 126: 50–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parvaz MA, Moeller SJ, Goldstein RZ (2016) Incubation of Cue-Induced Craving in Adults Addicted to Cocaine Measured by Electroencephalography. JAMA Psychiatry 73: 1127–1134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelloux Y, Degoulet M, Tiran-Cappello A, Cohen C, Lardeux S, George O, Koob GF, Ahmed SH, Baunez C (2018) Subthalamic nucleus high frequency stimulation prevents and reverses escalated cocaine use. Mol Psychiatry 23: 2266–2276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piazza PV, Deroche-Gamonent V, Rouge-Pont F, Le Moal M (2000) Vertical shifts in self-administration dose-response functions predict a drug-vulnerable phenotype predisposed to addiction. J Neurosci 20: 4226–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pickens CL, Airavaara M, Theberge F, Fanous S, Hope BT, Shaham Y (2011) Neurobiology of the incubation of drug craving. Trends Neurosci 34: 411–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce RC, Kumaresan V (2006) The mesolimbic dopamine system: the final common pathway for the reinforcing effect of drugs of abuse? Neurosci Biobehav Rev 30: 215–38. [DOI] [PubMed] [Google Scholar]
- Ramôa CP, Doyle SE, Lycas MD, Chernau AK, Lynch WJ (2014) Diminished role of dopamine D1-receptor signaling with the development of an addicted phenotype in rats. Biol Psychiatry 76: 8–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts DC, Morgan D, Liu Y (2007) How to make a rat addicted to cocaine. Prog Neuropsychopharmacol Biol Psychiatry 31: 1614–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schenk S, Gittings D (2003) Effects of SCH 23390 and eticlopride on cocaine-seeking produced by cocaine and WIN 35,428 in rats. Psychopharmacology (Berl) 168: 118–123. [DOI] [PubMed] [Google Scholar]
- Schmeichel BE, Herman MA, Roberto M, Koob GF (2017) Hypocretin Neurotransmission Within the Central Amygdala Mediates Escalated Cocaine Self-administration and Stress-Induced Reinstatement in Rats. Biol Psychiatry 81: 606–615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seth P, Scholl L, Rudd RA, Bacon S (2018) Overdose Deaths Involving Opioids, Cocaine, and Psychostimulants-United States, 2015-2016. MMWR Morb Mortal Wkly Rep 67: 349–358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seymour A, Black M, Simpson K, Oliver JS (2000) Drug related deaths amongst Glasgow city hostel dwellers. J Clin Forensic Med 7: 183–7. [DOI] [PubMed] [Google Scholar]
- Smith MA, Fronk GE, Abel JM, Lacy RT, Bills SE, Lynch WJ (2018) Resistance exercise decreases heroin self-administration and alters gene expression in the nucleus accumbens of heroin-exposed rats. Psychopharmacology (Berl) 235: 1245–1255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Substance Abuse and Mental Health Services Administration (2019) Key substance use and mental health indicators in the United States: Results from the 2018 National Survey on Drug Use and Health (HHS Publication No. PEP19-5068, NSDUH Series H-54). Rockville, MD: Center for Behavioral Health Statistics and Quality, Substance Abuse and Mental Health Services Administration. [Google Scholar]
- Tang WX, Fasulo WH, Mash DC, Hemby SE (2003) Molecular profiling of midbrain dopamine regions in cocaine overdose victims. J Neurochem 85: 911–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valenza M, Butelman ER, Kreek MJ (2017) “Effects of the novel relatively short-acting kappa opioid receptor antagonist LY2444296 in behaviors observed after chronic extended-access cocaine self-administration in rats”. Psychopharmacology (Berl) 234: 2219–2231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venniro M, Caprioli D, Shaham Y (2016) Animal models of drug relapse and craving: From drug priming-induced reinstatement to incubation of craving after voluntary abstinence. Prog Brain Res 224: 25–52. [DOI] [PubMed] [Google Scholar]
- Volkow ND, Fowler JS, Wolf AP, Schlyer D, Shiue CY, Alpert R, Dewey SL, Logan J, Bendriem B, Christman D, et al. (1990) Effects of chronic cocaine abuse on postsynaptic dopamine receptors. Am J Psychiatry 147: 719–24. [DOI] [PubMed] [Google Scholar]
- Volkow ND, Fowler JS, Wang GJ, Hitzemann R, Logan J, Schlyer DJ, Dewey SL, Wolf AP (1993) Decreased dopamine D2 receptor availability is associated with reduced frontal metabolism in cocaine abusers. Synapse 14: 169–77. [DOI] [PubMed] [Google Scholar]
- Volkow ND, Wang GJ, Fowler JS, Logan J, Gatley SJ, Hitzemann R, Chen AD, Dewey SL, Pappas N (1997) Decreased striatal dopaminergic responsiveness in detoxified cocaine-dependent subjects. Nature 386: 830–3. [DOI] [PubMed] [Google Scholar]
- Vranjkovic O, Van Newenhizen EC, Nordness ME, Blacktop JM, Urbanik LA, Mathy JC, McReynolds JR, Miller AM, Doncheck EM, Kloehn TM, Stinnett GS, Gerndt CH, Ketchesin KD, Baker DA, Seasholtz AF, Mantsch JR (2018) Enhanced CRFR1-Dependent Regulation of a Ventral Tegmental Area to Prelimbic Cortex Projection Establishes Susceptibility to Stress-Induced Cocaine Seeking. J Neurosci 38: 10657–10671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willuhn I, Wanat MJ, Clark JJ, Phillips PE (2010) Dopamine signaling in the nucleus accumbens of animals self-administering drugs of abuse. Curr Top Behav Neurosci 3: 29–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf ME, Tseng KY (2012) Calcium-permeable AMPA receptors in the VTA and nucleus accumbens after cocaine exposure: when, how, and why? Front Mol Neurosci 5: 72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Svenningsson P, Picetti R, Schlussman SD, Nairn AC, Ho A, Greengard P, Kreek MJ (2006) Cocaine self-administration in mice is inversely related to phosphorylation at Thr34 (protein kinase A site) and Ser130 (kinase CK1 site) of DARPP-32. J Neurosci 26: 2645–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmer BA, Oleson EB, Roberts DC (2012) The motivation to self-administer is increased after a history of spiking brain levels of cocaine. Neuropsychopharmacology 37: 1901–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmer BA, Dobrin CV, Roberts DC (2013) Examination of behavioral strategies regulating cocaine intake in rats. Psychopharmacology (Berl) 225: 935–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zorrilla EP, Wee S, Zhao Y, Specio S, Boutrel B, Koob GF, Weiss F (2012) Extended access cocaine self-administration differentially activates dorsal raphe and amygdala corticotropin-releasing factor systems in rats. Addict Biol 17: 300–8. [DOI] [PMC free article] [PubMed] [Google Scholar]


