Abstract
Celiac disease is an immune-mediated systemic disease triggered by intake of gluten in genetically susceptible individuals. The prevalence of celiac disease in the general population is estimated to be 1% in the world. Its prevalence differs depending on geographical and ethnic variations. The prevalence of celiac disease has increased significantly in the last 30 years due to the increased knowledge and awareness of physicians and the widespread use of highly sensitive and specific diagnostic tests for celiac disease. Despite increased awareness and knowledge about celiac disease, up to 95% of celiac patients still remain undiagnosed. The presentations of celiac disease have significantly changed in the last few decades. Classical symptoms of celiac disease occur in a minority of celiac patients, while older children have either minimal or atypical symptoms. Serologic tests for celiac disease should be done in patients with unexplained chronic or intermittent diarrhea, failure to thrive, weight loss, delayed puberty, short stature, amenorrhea, iron deficiency anemia, nausea, vomiting, chronic abdominal pain, abdominal distension, chronic constipation, recurrent aphthous stomatitis, and abnormal liver enzyme elevation, and in children who belong to specific groups at risk. Early diagnosis of celiac disease is very important to prevent long-term complications. Currently, the only effective treatment is a lifelong gluten-free diet. In this review, we will discuss the epidemiology, clinical findings, diagnostic tests, and treatment of celiac disease in the light of the latest literature.
Keywords: Celiac disease, Children, Intestinal biopsy
Core Tip: Celiac disease is a systemic lifelong disease. The prevalence of celiac disease has increased significantly in the last three decades due to the increased awareness of physicians and widespread use of highly sensitive and specific diagnostic tests for celiac disease. Despite increased awareness and widespread use of diagnostic tests, up to 95% of celiac patients still remain undiagnosed. Early diagnosis is very important to prevent long-term complications. The only effective treatment is still a lifelong gluten-free diet. In this review, we will discuss the epidemiology, clinical findings, diagnostic tests, and treatment of celiac disease in the light of the latest literature.
INTRODUCTION
Celiac disease is an immune-mediated systemic disease triggered by intake of gluten and related prolamines in genetically susceptible individuals, characterized by presence of various combinations of small intestinal damages, celiac specific antibodies, human leukocyte antigen (HLA)-DQ2 or HLA-DQ8, and gluten-dependent clinical manifestations[1]. Gluten is found in wheat, barley, rye, and oats[2].
PATHOGENESIS
The key elements of the celiac disease, an autoimmune disease, are genetics HLA-DQ2 and HLA-DQ8 genotypes, environmental factors (gluten intake), and autoantigen to tissue transglutaminase (tTG), which are known to play an important role in the pathogenesis[3]. In addition to genetic susceptibility and gluten exposure, loss of intestinal barrier function, gluten-induced proinflammatory innate immune response, inappropriate adaptive immune response, and unbalanced gut microbiome all seem to be components of the celiac disease autoimmunity[3]. More than 99% of celiac patients have HLA-DQ2 or HLA-DQ8 compared to 40% in the general population[4].
It has been suggested that breast milk, mode of delivery, and the age of gluten intake in infants are a risk for developing celiac disease and may affect the incidence of celiac disease. However, there is a limited information in retrospective studies that those factors affect the risk of developing celiac disease[5-7].
Furthermore, it has been suggested that gastrointestinal system (GIS) infections such as rotavirus may increase the risk of developing celiac disease, and therefore rotavirus vaccine may significantly reduce the risk of celiac disease especially in infants with gluten intake before 6 mo[8].
EPIDEMIOLOGY
The prevalence of celiac disease in the general population is estimated to be 1% in the world[9]. The seroprevalence of celiac disease and a biopsy-proven prevalence of celiac disease in the world is 1.4% and 0.7%, respectively[10]. Its prevalence varies depending on geographical and ethnic variations. The highest prevalence is in Europe (0.8%) and Ocenia (0.8%), and the lowest prevalence is in South America (0.4%). The biopsy-proven prevalence of celiac disease was found to be 1.5 times higher in women than men, and approximately two times higher in children than adults. The reason for this difference may be genetic factors [human leukocyte antigen (HLA) and non-HLA genes], environmental factors such as wheat consumption, age at gluten intake, gastrointestinal infections, proton pump inhibitor and antibiotic use, and the rate of cesarean section[10-12].
Celiac disease can occur at any age from early childhood to old age. It has two peaks; the first peak occurs after gluten intake within the first 2 years of life, the second is seen in the second or third decade of life. The diagnosis of celiac disease is difficult because symptoms vary from patient to patient[13].
The prevalence of celiac disease has increased significantly in the last 30 years, the reason for this is not only the increased knowledge and awareness of physicians about celiac disease but also due to the widespread use of highly sensitive and specific diagnostic tests for celiac disease[14,15]. For example, the incidence of pediatric celiac disease in Canada has increased 3-fold after the use of the endomysial antibody (EMA) test[16]. Despite increased awareness and knowledge about celiac disease, up to 95% of celiac patients still remain undiagnosed[17-19]. The delay in celiac disease diagnosis is reported to be 4-10 years in some studies[20,21]. There are many undiagnosed cases even in developed countries. Very few patients have clinically significant signs of celiac disease. The majority of cases have atypical signs or vague symptoms, so the diagnosis could not be made or diagnosis is delayed[22,23]. The reason for delayed or overlooked diagnosis may be the limited accessibility to serological diagnostic tests in developing countries and the lack of experienced specialists in this field[24].
The risk of developing celiac disease is higher in first- and second-degree relatives of celiac patients, Down syndrome, type 1 diabetes mellitus (DM), selective immunoglobulin (Ig)A deficiency, autoimmune thyroiditis, Turner syndrome, and Williams syndrome (Table 1)[25-28]. Screening tests for celiac disease at risk groups such as type 1 DM, autoimmune thyroid diseases, and first degree relatives of celiac patients also contributed to the increase in prevalence of celiac disease[27,29,30].
Table 1.
Groups with higher risk of developing celiac disease
|
Groups with higher risk of developing celiacdisease
|
| First-degree relatives of celiac patients |
| Second-degree relatives of celiac patients |
| Type 1 diabetes mellitus |
| Autoimmune thyroid disease |
| Autoimmune liver disease |
| Down syndrome |
| Turner syndrome |
| Williams syndrome |
| Selective IgA deficiency |
| Systemic lupus eryhtematosus |
| Juvenile chronic arthritis |
The prevalence of celiac disease in first degree relatives of celiac patients is as high as 10%-20%[1,31]. In a recent study of Sahin et al[32] the prevalence of celiac disease (CD) in siblings of pediatric celiac patients is reported to be 3.9%. The prevalence of CD in monozygotic twins has been found as high as 75%-80%[33,34].
In recent years, there has been a marked increase in the number of people having gluten-free diet. Furthermore, it has been observed that first-degree relatives of celiac patients start on a gluten-free diet before serologic tests for celiac disease were performed[35]. Therefore, before performing a serological test for celiac disease, it should be paid attention to whether they are on a gluten-free diet. Otherwise, the result of serological tests may be negative, and it would be difficult to diagnose celiac disease. Patients should take gluten-containing foods for 2-8 wk before serological tests[36].
CLINICAL MANIFESTATIONS
Symptoms usually occur in children after ingestion of gluten containing grains between 4 and 24 mo. There may be a delay or latent period between gluten intake and the onset of symptoms[37].
GIS and extra-intestinal manifestations are common in celiac disease[38]. The main GIS manifestations of celiac disease are chronic diarrhea, recurrent abdominal pain, nausea, vomiting, and abdominal distension. Common extra-intestinal manifestations are failure to thrive, short stature, chronic anemia, osteopenia, osteoporosis, delayed puberty, dental enamel defect, irritability, chronic fatigue, neuropathy, arthritis, arthralgia, amenorrhea, and increased liver enzymes[1,38].
Symptoms are usually different in infants than older children. Diarrhea, anorexia, abdominal distension, and abdominal pain are usually seen in younger children. If the diagnosis is delayed, failure to thrive, irritability, and severe malnutrition can be seen. GIS symptoms such as diarrhea, nausea, vomiting, abdominal pain, abdominal distension, weight loss, and constipation may occur in older children depending on the amount of gluten intake[28,37]. GIS signs of celiac disease such as diarrhea are seen in approximately 50% of patients[39-41].
The presentations of CD have significantly changed in the last few decades[41-48]. Classical symptoms of celiac disease occur in a minority of celiac patients, while older children have either minimal or atypical symptoms. GIS symptoms are mild or nonspecific[48,49].
It has been shown that pediatric patients diagnosed with celiac disease who are younger age at the diagnosis have less severe symptoms in the last 20 years. Also, it has been reported that the rate of asymptomatic patients, closer follow up, and strict adherence to gluten-free diet is higher in the last 10 years and that normalization of serological tests is faster than in the last decade[42].
Recently, the clinical symptoms of children with celiac disease are observed to change from GIS symptoms to extra-intestinal symptoms[39,50]. The exact reason for this is unclear, but it has been suggested that there may be increased awareness and widespread use of highly sensitive and specific serologic tests. It has been reported that isolated short stature is seen in up to 47.5% of celiac patients[41,51].
EXTRA-INTESTINAL MANIFESTATIONS
Extra-intestinal findings are seen in up to 60% of pediatric celiac patients (Table 2)[52]. Short stature is the most common finding in children[52-54]. It has been reported that 10%-47.5% of pediatric celiac patients have short stature at the time of diagnosis[41,54-57]. Nineteen percent to 59% of the non-endocrinologic causes of short stature are reported to be celiac disease[55,56,58-60]. Starting a gluten-free diet in the early period causes rapid growth and weight catch up, especially in the first 6 mo. The target height is usually reached within 3 years after diagnosis. If the target height is not reached despite a strict gluten-free diet, endocrinological evaluation should be done to rule out growth hormone deficiency[55,61-63].
Table 2.
Extra-intestinal manifestations of celiac disease
|
Extra-intestinal manifestations of celiac disease
|
| Short stature |
| Anemia |
| Osteopenia/osteoporosis |
| Delayed puberty |
| Dental enamel defects |
| Dermatitis herpetiformis |
| Recurrent aphtous stomatitis |
| Neurological manifestations; peripheral neuropathy, epilepsy, ataxia, headache |
| Arthritis, arthralgia |
| Infertility |
| Amenorrhea |
| Elevated liver enzymes |
| Alopecia |
| Anxiety, depression |
Hypogonadism in girls and delayed puberty in boys due to androgen resistance is a common finding in undiagnosed or untreated pediatric celiac patients[55,64,65]. Delayed puberty is seen in 10%-20% of celiac patients[52,66]. Generally, the development of puberty occurs within 6-8 mo after starting a gluten-free diet. If delayed puberty persists, the patient should be referred to pediatric endocrinology for further evaluation of other disorders of the reproductive system[55,67].
Iron deficiency anemia is seen in up to 40% of pediatric celiac patients[52,53,68,69]. Since iron is absorbed from the first part of the duodenum, which is mainly affected by celiac disease, iron deficiency anemia is common in celiac patients. It has been reported that 84% of pediatric celiac patients have the complete recovery of iron deficiency anemia with a strict gluten-free diet and iron supplementation therapy within 12-24 mo[52].
Hypertransaminasemia is seen in 9%-14% of celiac patients[70]. Mostly, liver damage is reversible, and liver failure rarely occurs[71]. It has been suggested that as a result of exposure to more hepatotoxins through the portal circulation due to the altered intestinal permeability, inflammation and liver damage may occur[54,72]. The response to a strict gluten-free diet is excellent. The increased liver enzymes return to normal by the rate of 75%-90% within 12-24 mo with a strict gluten-free diet[73].
Osteopenia and osteoporosis are usually seen in patients with celiac disease. Approximately 75% of celiac patients have osteopenia and 10%-30% have osteoporosis[74]. Secondary hyperparathyroidism occurs due to the insufficient absorption of vitamin D and calcium from the damaged duodenal mucosa. It is commonly seen in 12%-54% of celiac patients[75]. Normal blood levels of vitamin D and calcium is observed within the first year after a strict gluten-free diet[76,77].
The most common joint and muscle disorders seen in celiac disease are myopathy, arthralgia, and non-erosive arthritis[55,78]. Since arthralgia is mostly seen after the age of 12, the most common finding in pediatric celiac patients is subclinical synovitis. It is most commonly seen in the knee joint. Its incidence is 5%-10%[54]. Since symptoms are mild, ultrasonography is important in the diagnosis of joint disorders.
The most common finding of neurological manifestations is headache, which is seen in up to 20% of celiac patients. More rarely, ataxia and neuropathy (0.1%-7.4%) are seen[79,80]. The prevalence of epilepsy is reported to be 1.43 times higher in children with celiac disease compared to the general population[81]. The relationship between epilepsy and CD is still unclear.
The exact prevalence of enamel defects in celiac disease is unknown. In recent studies, it has been reported that enamel defects are seen in 55%-64% of celiac patients[82,83].
Aphthous stomatitis is seen in up to 46% of celiac patients[84]. Although its mechanism is not known exactly, it is usually completely cured with a strict gluten-free diet[52].
Dermatitis herpetiformis is thought to be an extra-intestinal manifestation of celiac disease, but it is relatively rare in pediatric celiac patients in Finland[85]. Unlike celiac disease, its annual incidence is decreasing. The reason for this is unknown exactly[85]. In contrast to that study, it has been reported that it is more common in childhood[86].
ASSOCIATED DISEASES WITH CELIAC DISEASE
The risk of another autoimmune disease is three to 10 times higher in patients with celiac disease compared to the general population[87,88].
The most common accompanying disease is type 1 DM since it has common genetic factors and pathogenic mechanisms with celiac disease[89]. HLA-DQ2 is present in approximately 90%-95% of celiac patients and 50% of type 1 DM patients, but HLA-DQ8 is detected in approximately 10% of celiac patients and approximately 70% of type 1 DM patients[90]. In a systematic review, the prevalence of celiac disease in patients with type 1 DM was reported to be approximately six times higher than in the general population[91]. The prevalence of celiac disease was reported to be 2.4%-16.4% in children with type 1 DM[92-95]. There is consensus about initial screening for celiac disease in newly diagnosed DM patients, but it is not clear when and how often to screen for celiac disease and initiate a gluten-free diet in asymptomatic patients[93]. It has been recommended that screening test for CD should be done at the time of type 1 DM diagnosis and then every 2 years[96]. In another study, it was recommended that children diagnosed with type 1 DM should be screened for celiac disease once a year for the first 5 years[92]. In other studies, it has been recommended that serological screening tests for celiac disease should be done within the first 2 years when the diagnosis is made, then 5 years after the diagnosis and if there is any symptom suggestive of CD[93,97]. Since 58%-85% of type 1 DM patients diagnosed with CD are asymptomatic, early diagnosis of CD is very important to prevent long-term complications such as failure to thrive, osteopenia, infertility, and malignancy[29,77,92,93,98,99].
There is good evidence that autoimmune thyroid diseases are associated with celiac disease[1,100]. The prevalence of celiac disease in patients with autoimmune thyroid disease is found to be 3.0%-4.8%[30,101,102].
Also, the prevalence of celiac disease in patients with selective IgA deficiency is reported to be 10-20 times higher than in the general population[103].
There is a close relationship between Down syndrome and celiac disease. The prevalence of celiac disease in patients diagnosed with Down syndrome is reported to be 5%-12%[104-108]. The North American Society for Paediatric Gastroenterology, Hepatology and Nutrition and The European Society for Paediatric Gastroenterology, Hepatology and Nutrition (ESPGHAN) recommend screening tests for celiac disease in children with Down syndrome due to the increased risk of developing celiac disease[28]. In a study conducted in 2020, involving 1317 pediatric patients with Down syndrome aged 3 and over, the prevalence of celiac disease was found to be 9.8% in children with Down syndrome[109]. If screening test for celiac disease is not done, the diagnosis of celiac disease is either overlooked or delayed in 82% of the patients with Down syndrome, thus causing increased morbidity[109].
The increased prevalence of celiac disease is also seen in autoimmune liver disease, Turner syndrome, and Williams syndrome[1,110-116].
THE DEFINITONS RELATED TO CELIAC DISEASE
Silent celiac disease
Silent celiac disease is defined by the presence of celiac antibodies and HLA-DQ2 or HLA-DQ8 and small intestinal biopsy findings compatible with celiac disease especially in patients with autoimmune disease or a genetic disorder or relatives of celiac disease but without any symptoms suggestive of CD[1].
Potential celiac disease
Potential celiac disease is defined by the presence of celiac antibodies, HLA-DQ2 or HLA-DQ8, but intestinal biopsy is not compatible with celiac disease. Marsh classification score 0 or 1 is detected in intestinal biopsy, and the risk of developing celiac disease is increased[117].
Clinical symptoms and signs of the celiac disease are not always seen. Even if there are clinical findings, they are usually mild. The diagnosis of potential CD has increased significantly in recent years due to increased use of serological screening for celiac disease in the general population. A lower prevalence of HLA-DQ2 and a higher prevalence of HLA-DQ8 are detected in potential celiac patients compared to active celiac patients[118].
It should be considered that the cause of negative intestinal biopsy may be the patchy involvement of the small intestinal mucosa, low gluten intake, and inappropriate biopsy orientation[119].
Its treatment is still uncertain and controversial. There is no consensus about how often celiac serological tests should be performed in potential celiac patients on a gluten-containing diet, and how often they should be evaluated clinically[120]. It has been reported that villous atrophy is observed in 33% of symptomatic potential celiac patients after 3 years[121]. Therefore, it has been suggested that symptomatic patients should be given a gluten free diet.
Refractory celiac disease
Refractory celiac disease is characterized by the persistence of symptoms and intestinal villous atrophy despite a strict gluten-free diet for at least 12 mo. Generally, celiac antibodies are negative in most patients at the time of diagnosis, but the presence of high-titer antibodies does not rule out the refractory celiac disease. In all cases, dietary adherence should be carefully questioned. It can cause complications such as ulcerative jejunoileitis, collagenous sprue, and intestinal lymphoma[117].
Seronegative celiac disease
It is characterized by the presence of clinical signs of severe malabsorption and intestinal villous atrophy and negative celiac antibodies[122]. It constitutes approximately 2%-3% of celiac patients. Seronegative celiac disease can be confirmed with improvement in both symptoms and histology 1 year after starting a gluten-free diet[122]. Compared with classical celiac disease, seronegative celiac patients are associated with a higher rate of autoimmune disease, and these patients have a higher risk of developing refractory celiac disease[122].
In this form of celiac disease, genetic analysis is the key step for the diagnosis, because if it is found as negative, celiac disease is ruled out. Other diseases causing villous atrophy are parasitic infections (e.g., Giardia lamblia), autoimmune enteropathy, small intestinal bacterial overgrowth, common variable immunodeficiency, eosinophilic gastroenteritis, drug induced enteropathy (e.g., olmesartan, mycophenolate), intestinal lymphoma, Crohn's disease, tropical sprue, human immunodeficiency virus enteropathy, and Whipple disease should be considered in the differential diagnosis (Table 3)[122-124].
Table 3.
Other diseases causing villous atrophy
|
Other diseases causing villous atrophy
|
| Parasitic infections (Giardia lamblia) |
| Autoimmune enteropathy |
| Small intestinal bacterial overgrowth |
| Common variable immunodeficiency |
| Cow's milk or soya protein hypersensitivity |
| Intractable diarrhea of infancy |
| Eosinophilic gastroenteritis |
| Drug induced enteropathy (e.g., olmesartan, mycophenolate) |
| Intestinal lymphoma |
| Crohn's disease |
| Human immunodeficiency virus enteropathy |
| Tropical disease |
Non-responsive celiac disease
Non-responsive celiac disease is defined by the persistence of GI symptoms more than 12 mo despite a strict gluten-free diet. The most common causes of non-responsive celiac disease are persistent gluten ingestion and incorrect diagnosis[125,126]. It needs to be differentiated from active celiac disease and other conditions associated with celiac disease.
DIAGNOSIS
The clinical symptoms of celiac disease are very diverse. Celiac patients may present with symptoms of GIS or extra-intestinal symptoms or no symptoms at all. Therefore, serologic tests for celiac disease should be done in patients with unexplained chronic or intermittent diarrhea, failure to thrive, weight loss, delayed puberty, short stature, amenorrhea, iron deficiency anemia, nausea, vomiting, chronic abdominal pain, abdominal distension, chronic constipation, recurrent aphthous stomatitis, and abnormal liver enzyme elevation[1].
Furthermore, celiac disease should be investigated in patients with high risk of developing celiac disease, such as type 1 DM, Down syndrome, autoimmune thyroid disease, Turner syndrome, selective IgA deficiency, autoimmune liver disease, and first-degree relatives of celiac patients, even if they are asymptomatic[1].
Celiac disease is diagnosed by a variable combination of symptoms, positive celiac antibodies, presence of HLA-DQ2/DQ8, and duodenal histology[1].
ESPGHAN guidelines from 2012 recommend tissue tTG-IgA test, which is highly sensitive and specific and less costly compared to EMA IgA antibody test, as an initial screening test for suspected celiac disease, and the total IgA test to rule out selective IgA deficiency. The analysis of deamidated gliadin peptide (DGP) IgA test is recommended for children under 2 years of age. If there is IgA deficiency, the tTG-IgG test or the EMA-IgG test or the DGP-IgG test should be performed[1].
If serological tests are negative for tTG-IgA and total IgA level is normal, celiac disease is unlikely. In this condition, the reasons leading to the false negative tTG result should be considered. Those are low gluten intake, protein-losing enteropathy, use of immunosuppressive drugs, and patients under 2 years of age. If the tTG is found as positive [lower than 10 times upper limit of normal (ULN)], gastroduodenoscopy and multiple biopsies of the small intestine should be performed to confirm the diagnosis[1].
If the tTG is higher than 10 times ULN in a symptomatic patient, it should be discussed with the parents in order to make a diagnosis of celiac disease without biopsy. If the parents agree, EMA test and HLA-DQ2/DQ8 analysis are performed. To rule out false positivity of the tTG test, an EMA test is performed from a second blood sample. If EMA and HLA-DQ2 or HLA-DQ8 are positive, celiac disease is diagnosed without biopsy[1]. In practice, it has been reported that this reduces the need for endoscopy by 30%-50%[127].
Since celiac disease causes patchy involvement in the small intestine, at least four biopsies from the duodenum and at least one biopsy from the bulbus should be performed by gastroduodenoscopy. Biopsies are evaluated according to modified Marsh-Oberhuber classification (Table 4)[128]. Since the lesion of celiac disease can only be seen in the bulb, at least one biopsy should be taken from the bulb[129].
Table 4.
The modified Marsh classification
| IEL | Crypts | Villi | |
| Type 0 | < 40 | Normal | Normal |
| Type 1 | > 40 | Normal | Normal |
| Type 2 | > 40 | Hypertrophic | Normal |
| Type 3a | > 40 | Hypertrophic | Mild atrophy |
| Type 3b | > 40 | Hypertrophic | Marked atrophy |
| Type 3c | > 40 | Hypertrophic | Absent |
IEL: Intraepithelial lymphocyte count/100 epithelial cells.
While interpreting the serological test results of celiac disease, serum total IgA levels, the amount of gluten consumption, use of immunosuppressive drugs, and age of the patient should be considered[1]. IgG class celiac antibody tests should be performed in patients with low serum IgA levels (total serum IgA < 0.2 g/L)[1].
If the patient has the gluten-free diet for a long time or gluten-free diet for a short time before testing, false negative results may occur[130]. Therefore, patients should take definitely gluten-containing foods before the test. Gluten challenge test should be performed for patients with a gluten-free diet before serological tests, 3-7.5 g/d gluten-containing diet (approximately two slices of bread) is recommended for 2 wk[131].
If the patient is strongly suspected of celiac disease, multiple intestinal biopsy and HLA-DQ2/DQ8 analysis are recommended, even if the serological tests for celiac disease are negative. If the histology is compatible with celiac disease but HLA-DQ2/8 negative, celiac disease is unlikely and other causes of enteropathy should be investigated (Table 3)[1]. Celiac disease is diagnosed if the celiac serological tests are positive and the biopsy is compatible with celiac disease.
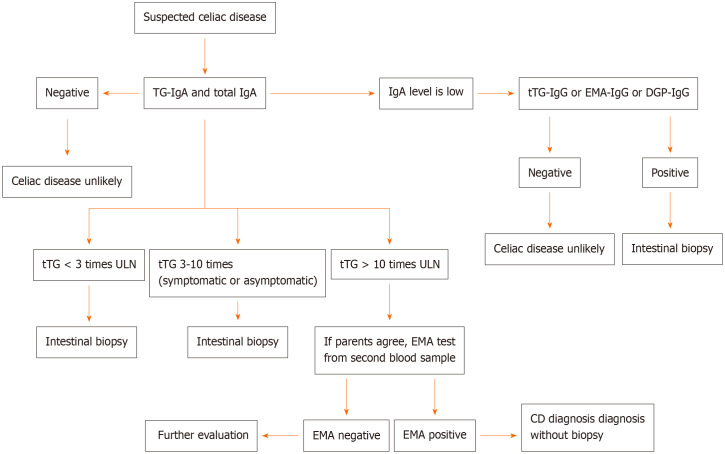
ESPGHAN guidelines from 2020 report that the tTG-IgA test and total IgA test combination give more accurate results than other test combinations as the initial test for suspected celiac disease regardless of age. If total IgA level is found to be low, tTG-IgG test or EMA-IgG test or DGP-IgG test should be performed (Figure 1)[119].
Figure 1.
Algorithm for diagnosis of celiac disease. CD: Celiac disease; DGP: Deamidated gliadin peptide; EMA: Endomysial antibody; tTG: Tissue transglutaminase antibody; ULN: Upper limit of normal.
If the tTG test is found as positive (> 10 times ULN), HLA-DQ2/8 analysis is not recommended in the ESPGHAN 2020 guidelines even if the patient is asymptomatic. It has been suggested that the EMA test should be checked in a second blood sample and if the EMA test is detected positive and the family agrees, celiac disease can be diagnosed without biopsy. In other words, the presence of HLA-DQ2/8 analysis and clinical symptoms are not mandatory for celiac diagnosis in last guideline in 2020 (Figure 1)[119].
If HLA-DQ2/DQ8 test is negative, the probability of celiac disease is low, but a positive HLA-DQ2/DQ8 test does not confirm the diagnosis of celiac disease[132]. If the tTG test is detected positive (< 10 times ULN), multiple intestinal biopsy is recommended to rule out false positivity. It is not recommended to diagnose without biopsy in patients with selective IgA deficiency even if IgG-based antibody positivity is detected[119].
It has been considered that villous atrophy may be seen in other GIS diseases such as parasitic infections, autoimmune diseases, bacterial overgrowth in the small intestine, and Crohn's disease (Table 3)[133].
It has been reported that the pooled sensitivity and specificity of tTG or DGP or tTG + antigliadin antibodies for diagnosing celiac disease is 94.0% and 94.4%, respectively, in a systematic review[134]. It has been suggested that those tests can be used in places where access to laboratory tests is limited.
MANAGEMENT
Currently, the only effective treatment is a lifelong gluten-free diet. Significant improvements in symptoms, normalization of biochemical tests, and improvement in quality of life with a strict gluten-free diet are seen[135].
Rapid improvement in clinical symptoms is observed within 2-4 wk in children. Serological and histological responses are slower compared to clinical symptoms[136]. Although histological response in children is observed within 2 years by a rate of 95%, this rate is 60% in adults[137].
The amount of tolerable gluten varies from patient to patient. As little as 50 mg of gluten, present in a few amounts of bread crumbs or a small piece of cake or traces of contamination, may cause symptoms and/or enteropathy in asymptomatic patients[135,138]. It is unlikely that a gluten intake of less than 10 mg/d will cause significant histological abnormality[139].
Adherence to the gluten-free diet is better in children diagnosed with CD at an early age and those who continue to follow up regularly. It is less in adolescents compared to adults[135].
It has been reported that there is a direct relationship between the duration of exposure to the gluten-free diet and increased autoimmune disorders[140].
In a multicenter prospective study involving 6605 children with the HLA genotype associated with celiac disease, it was shown that the amount of gluten exposure in the first 5 years of life is associated with the development of celiac disease and celiac autoimmunity[141]. Since celiac disease is a multisytemic disease that affects multiple organs, a lifelong gluten-free diet may reduce malignant and non-malignant complications[142].
FOLLOW-UP
Currently, there are no standard evidence-based recommendations for the follow-up of pediatric celiac disease[143].
Patients with celiac disease should be followed up 6 mo after diagnosis and every 6 mo in terms of improvement in symptoms, compliance with the gluten-free diet, quality of life, and progressive normalization of celiac-associated antibodies. Screening tests should be done in terms of autoimmune thyroid disease. A control duodenal biopsy is not required after a gluten-free diet. However, if there is a partial or no response to the gluten-free diet, careful examination should be done for involuntary gluten contamination or poor compliance with the gluten-free diet. If the response to a strict gluten-free diet is poor, duodenal biopsy can be performed[135,143,144].
Earlier diagnosis of celiac disease in asymptomatic patients is associated with better quality of life as well as better compliance with the gluten-free diet[42,145,146].
It has been shown that pediatric patients who are lost to follow up are less adherent to the gluten-free diet and have positive celiac serological antibodies[147]. It has been shown that the regular control is very important.
Routine testing for vitamin and mineral deficiency is reported to be unnecessary in the vast majority of children who follow up to regular controls and have normal growth and development and have no symptoms[148].
The essential marker of the success of the gluten-free diet is still satisfactory height and weight gain in children and adolescents[135].
The best marker of proper follow-up and management is the decline in the antibody levels and the return of antibody levels to normal in follow-up. The presence of persistent positive antibodies usually indicates ongoing intestinal damage and gluten exposure. Serological follow-up should be done within 6 mo and 12 mo after diagnosis and then once a year[149].
tTG-IgA test is reported to be best test in follow up[150]. It has been shown that the average time to return to normal levels of the tTG test in patients with strictly adherent to the gluten-free diet is 1 year[151].
It has been detected that there is no correlation between symptoms and mucosal healing[152]. Gluten challenge test can be performed in cases when there is a doubt about the initial diagnosis of celiac disease. However, HLA typing should be done before evaluation of mucosal damage. Gluten challenge is not recommended under 5 years of age and during pubertal development[1].
In recent studies, it has been reported that gluten consumption can be shown in symptomatic and asymptomatic patients who are unaware of gluten intake by gluten immunogenic peptide tests in stool and urine[153,154]. Gluten intake of more than 50 mg/d for stool test and more than 25 mg/d for urine test seems to be necessary for the sensitivity of the test[153]. Dietary adherence to the gluten-free diet can be evaluated with this test. It can replace serological tests in follow-up, but its use in routine practice is still uncertain and further studies are needed.
DIETS AND NEW TREATMENTS
Currently, the only effective treatment is still to avoid gluten completely for life. The adherence to the gluten-free diet has some disadvantages; negative impact on quality of life, psychological problems, involuntary gluten contamination, possible vitamin and mineral deficiencies, metabolic syndrome, increased cardiovascular risk, and severe constipation[153,155-157].
Approximately 40% of celiac patients are not satisfied with the gluten-free diet due to the negative effect on their quality of life and seek alternative treatments[158,159].
Clinical studies are still ongoing in the treatment of celiac disease. Larazotide acetate is a zonulin antagonist that blocks the tight junction, thus restricting the passage of gluten through the permeable intestinal mucosa[160]. This drug is shown to be effective in controlling gluten-related symptoms[160]. There is also limited information that larazotide may allow patients to tolerate minimal amounts of gluten (involuntary gluten contamination or short-term feeding with a small amount of gluten).
ALV003 (latiglutenase) reduces gluten into small pieces in the stomach before it passes into the duodenum[161]. In a study involving 494 celiac patients, latiglutenase was compared with placebo. It has been shown that latiglutenase did not improve histological findings or symptoms[162]. Further studies are needed.
Vaccination (Nexvax2) is another therapeutic option intended to be used for desensitization in celiac patients against gliadin peptides. Although its major side effects are abdominal pain and vomiting, it passed phase 1. Given the effectiveness of vaccines, it can be a definitive cure for celiac disease[163].
COMPLICATIONS
Complication are usually manifested in late-diagnosed celiac patients (after the age of 50) and in patients not adhering to a strict gluten-free diet. These patients have a higher mortality than the general population[164], but complications are rare (< 1%)[165].
Complications of celiac disease include hyposplenism, refractory celiac disease, intestinal lymphoma, small bowel adenocarcinoma, and ulcerative jejunoileitis[166].
Despite adhering to a gluten-free diet and having complaints that cannot be explained by any other reason, complications should be considered in every patient whose symptoms persist.
CONCLUSION
Celiac disease is a lifelong multi-systemic disease triggered by intake of gluten in genetically susceptible individuals.
Serologic tests for CD should be done in patients with unexplained chronic or intermittent diarrhea, failure to thrive, weight loss, delayed puberty, short stature, amenorrhea, iron deficiency anemia, nausea, vomiting, chronic abdominal pain, abdominal distension, chronic constipation, recurrent aphthous stomatitis, and abnormal liver enzyme elevation.
Since tTG-IgA test and total IgA test combination give more accurate results than other test combinations, ESPGHAN 2020 guideline recommends this combination as the initial test for suspected celiac disease regardless of age. While interpreting the serological test results of celiac disease, serum total IgA levels, the amount of gluten consumption, use of immunosuppressive drugs, and age of the patient should be considered.
Early diagnosis of CD is very important to prevent long-term complications such as failure to thrive, osteopenia, infertility, and malignancy.
Currently, the only effective treatment is a lifelong gluten-free diet.
Footnotes
Conflict-of-interest statement: Author has nothing to disclose.
Manuscript source: Invited manuscript
Corresponding Author's Membership in Professional Societies: Turkish Society of Gastroenterology
Peer-review started: February 2, 2021
First decision: March 17, 2021
Article in press: May 22, 2021
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Turkey
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B, B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Pavlovic M, Wierzbicka A S-Editor: Liu M L-Editor: Filipodia P-Editor: Wang LYT
References
- 1.Husby S, Koletzko S, Korponay-Szabó IR, Mearin ML, Phillips A, Shamir R, Troncone R, Giersiepen K, Branski D, Catassi C, Lelgeman M, Mäki M, Ribes-Koninckx C, Ventura A, Zimmer KP ESPGHAN Working Group on Coeliac Disease Diagnosis; ESPGHAN Gastroenterology Committee; European Society for Pediatric Gastroenterology. Hepatology , and Nutrition. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. J Pediatr Gastroenterol Nutr. 2012;54:136–160. doi: 10.1097/MPG.0b013e31821a23d0. [DOI] [PubMed] [Google Scholar]
- 2.Fasano A, Catassi C. Clinical practice. Celiac disease. N Engl J Med. 2012;367:2419–2426. doi: 10.1056/NEJMcp1113994. [DOI] [PubMed] [Google Scholar]
- 3.Caio G, Volta U, Sapone A, Leffler DA, De Giorgio R, Catassi C, Fasano A. Celiac disease: a comprehensive current review. BMC Med. 2019;17:142. doi: 10.1186/s12916-019-1380-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hadithi M, von Blomberg BM, Crusius JB, Bloemena E, Kostense PJ, Meijer JW, Mulder CJ, Stehouwer CD, Peña AS. Accuracy of serologic tests and HLA-DQ typing for diagnosing celiac disease. Ann Intern Med. 2007;147:294–302. doi: 10.7326/0003-4819-147-5-200709040-00003. [DOI] [PubMed] [Google Scholar]
- 5.Lionetti E, Castellaneta S, Francavilla R, Pulvirenti A, Catassi C SIGENP Working Group of Weaning and CD Risk. Mode of Delivery and Risk of Celiac Disease: Risk of Celiac Disease and Age at Gluten Introduction Cohort Study. J Pediatr. 2017;184:81–86.e2. doi: 10.1016/j.jpeds.2017.01.023. [DOI] [PubMed] [Google Scholar]
- 6.Koletzko S, Lee HS, Beyerlein A, Aronsson CA, Hummel M, Liu E, Simell V, Kurppa K, Lernmark Å, Hagopian W, Rewers M, She JX, Simell O, Toppari J, Ziegler AG, Krischer J, Agardh D TEDDY Study Group. Cesarean Section on the Risk of Celiac Disease in the Offspring: The Teddy Study. J Pediatr Gastroenterol Nutr. 2018;66:417–424. doi: 10.1097/MPG.0000000000001682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dydensborg Sander S, Hansen AV, Størdal K, Andersen AN, Murray JA, Husby S. Mode of delivery is not associated with celiac disease. Clin Epidemiol. 2018;10:323–332. doi: 10.2147/CLEP.S152168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Silvester JA, Leffler DA. Is Autoimmunity Infectious? Clin Gastroenterol Hepatol. 2017;15:703–705. doi: 10.1016/j.cgh.2016.12.014. [DOI] [PubMed] [Google Scholar]
- 9.Lindfors K, Ciacci C, Kurppa K, Lundin KEA, Makharia GK, Mearin ML, Murray JA, Verdu EF, Kaukinen K. Coeliac disease. Nat Rev Dis Primers. 2019;5:3. doi: 10.1038/s41572-018-0054-z. [DOI] [PubMed] [Google Scholar]
- 10.Singh P, Arora A, Strand TA, Leffler DA, Catassi C, Green PH, Kelly CP, Ahuja V, Makharia GK. Global Prevalence of Celiac Disease: Systematic Review and Meta-analysis. Clin Gastroenterol Hepatol. 2018;16:823–836.e2. doi: 10.1016/j.cgh.2017.06.037. [DOI] [PubMed] [Google Scholar]
- 11.Lebwohl B, Murray JA, Verdú EF, Crowe SE, Dennis M, Fasano A, Green PH, Guandalini S, Khosla C. Gluten Introduction, Breastfeeding, and Celiac Disease: Back to the Drawing Board. Am J Gastroenterol. 2016;111:12–14. doi: 10.1038/ajg.2015.219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Choung RS, Ditah IC, Nadeau AM, Rubio-Tapia A, Marietta EV, Brantner TL, Camilleri MJ, Rajkumar SV, Landgren O, Everhart JE, Murray JA. Trends and racial/ethnic disparities in gluten-sensitive problems in the United States: findings from the National Health and Nutrition Examination Surveys from 1988 to 2012. Am J Gastroenterol. 2015;110:455–461. doi: 10.1038/ajg.2015.8. [DOI] [PubMed] [Google Scholar]
- 13.Fasano A. Celiac disease--how to handle a clinical chameleon. N Engl J Med. 2003;348:2568–2570. doi: 10.1056/NEJMe030050. [DOI] [PubMed] [Google Scholar]
- 14.Liu E, Dong F, Barón AE, Taki I, Norris JM, Frohnert BI, Hoffenberg EJ, Rewers M. High Incidence of Celiac Disease in a Long-term Study of Adolescents With Susceptibility Genotypes. Gastroenterology. 2017;152:1329–1336.e1. doi: 10.1053/j.gastro.2017.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.King JA, Jeong J, Underwood FE, Quan J, Panaccione N, Windsor JW, Coward S, deBruyn J, Ronksley PE, Shaheen AA, Quan H, Godley J, Veldhuyzen van Zanten S, Lebwohl B, Ng SC, Ludvigsson JF, Kaplan GG. Incidence of Celiac Disease Is Increasing Over Time: A Systematic Review and Meta-analysis. Am J Gastroenterol. 2020;115:507–525. doi: 10.14309/ajg.0000000000000523. [DOI] [PubMed] [Google Scholar]
- 16.McGowan KE, Castiglione DA, Butzner JD. The changing face of childhood celiac disease in north america: impact of serological testing. Pediatrics. 2009;124:1572–1578. doi: 10.1542/peds.2008-2373. [DOI] [PubMed] [Google Scholar]
- 17.Hujoel IA, Van Dyke CT, Brantner T, Larson J, King KS, Sharma A, Murray JA, Rubio-Tapia A. Natural history and clinical detection of undiagnosed coeliac disease in a North American community. Aliment Pharmacol Ther. 2018;47:1358–1366. doi: 10.1111/apt.14625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Murray JA, Van Dyke C, Plevak MF, Dierkhising RA, Zinsmeister AR, Melton LJ 3rd. Trends in the identification and clinical features of celiac disease in a North American community, 1950-2001. Clin Gastroenterol Hepatol. 2003;1:19–27. doi: 10.1053/jcgh.2003.50004. [DOI] [PubMed] [Google Scholar]
- 19.Lebwohl B, Rubio-Tapia A, Assiri A, Newland C, Guandalini S. Diagnosis of celiac disease. Gastrointest Endosc Clin N Am. 2012;22:661–677. doi: 10.1016/j.giec.2012.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sanders DS, Hurlstone DP, Stokes RO, Rashid F, Milford-Ward A, Hadjivassiliou M, Lobo AJ. Changing face of adult coeliac disease: experience of a single university hospital in South Yorkshire. Postgrad Med J. 2002;78:31–33. doi: 10.1136/pmj.78.915.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lo W, Sano K, Lebwohl B, Diamond B, Green PH. Changing presentation of adult celiac disease. Dig Dis Sci. 2003;48:395–398. doi: 10.1023/a:1021956200382. [DOI] [PubMed] [Google Scholar]
- 22.Lebwohl B, Sanders DS, Green PHR. Coeliac disease. Lancet. 2018;391:70–81. doi: 10.1016/S0140-6736(17)31796-8. [DOI] [PubMed] [Google Scholar]
- 23.Singh P, Wadhwa N, Chaturvedi MK, Bhatia V, Saini S, Tandon N, Makharia GK, Maki M, Not T, Phillips A, Bhatnagar S. Validation of point-of-care testing for coeliac disease in children in a tertiary hospital in north India. Arch Dis Child. 2014;99:1004–1008. doi: 10.1136/archdischild-2013-305567. [DOI] [PubMed] [Google Scholar]
- 24.Nenna R, Tiberti C, Petrarca L, Lucantoni F, Mennini M, Luparia RP, Panimolle F, Mastrogiorgio G, Pietropaoli N, Magliocca FM, Bonamico M. The celiac iceberg: characterization of the disease in primary schoolchildren. J Pediatr Gastroenterol Nutr. 2013;56:416–421. doi: 10.1097/MPG.0b013e31827b7f64. [DOI] [PubMed] [Google Scholar]
- 25.Nellikkal SS, Hafed Y, Larson JJ, Murray JA, Absah I. High Prevalence of Celiac Disease Among Screened First-Degree Relatives. Mayo Clin Proc. 2019;94:1807–1813. doi: 10.1016/j.mayocp.2019.03.027. [DOI] [PubMed] [Google Scholar]
- 26.Fasano A, Berti I, Gerarduzzi T, Not T, Colletti RB, Drago S, Elitsur Y, Green PH, Guandalini S, Hill ID, Pietzak M, Ventura A, Thorpe M, Kryszak D, Fornaroli F, Wasserman SS, Murray JA, Horvath K. Prevalence of celiac disease in at-risk and not-at-risk groups in the United States: a large multicenter study. Arch Intern Med. 2003;163:286–292. doi: 10.1001/archinte.163.3.286. [DOI] [PubMed] [Google Scholar]
- 27.Singh P, Arora S, Lal S, Strand TA, Makharia GK. Risk of Celiac Disease in the First- and Second-Degree Relatives of Patients With Celiac Disease: A Systematic Review and Meta-Analysis. Am J Gastroenterol. 2015;110:1539–1548. doi: 10.1038/ajg.2015.296. [DOI] [PubMed] [Google Scholar]
- 28.Hill ID, Dirks MH, Liptak GS, Colletti RB, Fasano A, Guandalini S, Hoffenberg EJ, Horvath K, Murray JA, Pivor M, Seidman EG North American Society for Pediatric Gastroenterology. Hepatology and Nutrition. Guideline for the diagnosis and treatment of celiac disease in children: recommendations of the North American Society for Pediatric Gastroenterology, Hepatology and Nutrition. J Pediatr Gastroenterol Nutr. 2005;40:1–19. doi: 10.1097/00005176-200501000-00001. [DOI] [PubMed] [Google Scholar]
- 29.Pham-Short A, Donaghue KC, Ambler G, Phelan H, Twigg S, Craig ME. Screening for Celiac Disease in Type 1 Diabetes: A Systematic Review. Pediatrics. 2015;136:e170–e176. doi: 10.1542/peds.2014-2883. [DOI] [PubMed] [Google Scholar]
- 30.Sahin Y, Evliyaoglu O, Erkan T, Cokugras FC, Ercan O, Kutlu T. The frequency of celiac disease in children with autoimmune thyroiditis. Acta Gastroenterol Belg. 2018;81:5–8. [PubMed] [Google Scholar]
- 31.Rubio-Tapia A, Van Dyke CT, Lahr BD, Zinsmeister AR, El-Youssef M, Moore SB, Bowman M, Burgart LJ, Melton LJ 3rd, Murray JA. Predictors of family risk for celiac disease: a population-based study. Clin Gastroenterol Hepatol. 2008;6:983–987. doi: 10.1016/j.cgh.2008.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sahin Y. The Frequency of Celiac Disease in Siblings of Celiac Patients. EC Paediatrics. 2019;2:154–157. [Google Scholar]
- 33.Greco L, Romino R, Coto I, Di Cosmo N, Percopo S, Maglio M, Paparo F, Gasperi V, Limongelli MG, Cotichini R, D'Agate C, Tinto N, Sacchetti L, Tosi R, Stazi MA. The first large population based twin study of coeliac disease. Gut. 2002;50:624–628. doi: 10.1136/gut.50.5.624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lundin KE, Wijmenga C. Coeliac disease and autoimmune disease-genetic overlap and screening. Nat Rev Gastroenterol Hepatol. 2015;12:507–515. doi: 10.1038/nrgastro.2015.136. [DOI] [PubMed] [Google Scholar]
- 35.Kim HS, Patel KG, Orosz E, Kothari N, Demyen MF, Pyrsopoulos N, Ahlawat SK. Time Trends in the Prevalence of Celiac Disease and Gluten-Free Diet in the US Population: Results From the National Health and Nutrition Examination Surveys 2009-2014. JAMA Intern Med. 2016;176:1716–1717. doi: 10.1001/jamainternmed.2016.5254. [DOI] [PubMed] [Google Scholar]
- 36.Rubio-Tapia A, Hill ID, Kelly CP, Calderwood AH, Murray JA American College of Gastroenterology. ACG clinical guidelines: diagnosis and management of celiac disease. Am J Gastroenterol. 2013;108:656–76; quiz 677. doi: 10.1038/ajg.2013.79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gallegos C, Merkel R. Current Evidence in the Diagnosis and Treatment of Children With Celiac Disease. Gastroenterol Nurs. 2019;42:41–48. doi: 10.1097/SGA.0000000000000365. [DOI] [PubMed] [Google Scholar]
- 38.Van Kalleveen MW, de Meij T, Plötz FB. Clinical spectrum of paediatric coeliac disease: a 10-year single-centre experience. Eur J Pediatr. 2018;177:593–602. doi: 10.1007/s00431-018-3103-4. [DOI] [PubMed] [Google Scholar]
- 39.Garampazzi A, Rapa A, Mura S, Capelli A, Valori A, Boldorini R, Oderda G. Clinical pattern of celiac disease is still changing. J Pediatr Gastroenterol Nutr. 2007;45:611–614. doi: 10.1097/MPG.0b013e31814c3d79. [DOI] [PubMed] [Google Scholar]
- 40.Simmons JH, Klingensmith GJ, McFann K, Rewers M, Taylor J, Emery LM, Taki I, Vanyi S, Liu E, Hoffenberg EJ. Impact of celiac autoimmunity on children with type 1 diabetes. J Pediatr. 2007;150:461–466. doi: 10.1016/j.jpeds.2006.12.046. [DOI] [PubMed] [Google Scholar]
- 41.Sahin Y. Clinical evaluation of children with celiac disease: a single-center experience. Arch Clin Gastroenterol. 2020;6:26–30. [Google Scholar]
- 42.Krauthammer A, Guz-Mark A, Zevit N, Marderfeld L, Waisbourd-Zinman O, Silbermintz A, Mozer-Glassberg Y, Nachmias Friedler V, Rozenfeld Bar Lev M, Matar M, Assa A, Shamir R. Two decades of pediatric celiac disease in a tertiary referral center: What has changed? Dig Liver Dis. 2020;52:457–461. doi: 10.1016/j.dld.2020.02.001. [DOI] [PubMed] [Google Scholar]
- 43.Fasano A. Clinical presentation of celiac disease in the pediatric population. Gastroenterology. 2005;128:S68–S73. doi: 10.1053/j.gastro.2005.02.015. [DOI] [PubMed] [Google Scholar]
- 44.Beattie RM. The changing face of coeliac disease. Arch Dis Child. 2006;91:955–956. doi: 10.1136/adc.2006.099671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ravikumara M, Tuthill DP, Jenkins HR. The changing clinical presentation of coeliac disease. Arch Dis Child. 2006;91:969–971. doi: 10.1136/adc.2006.094045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lebenthal E, Shteyer E, Branski D. The changing clinical presentation of celiac disease. In: Fasano A, Troncone R, Branski D. Frontiers in celiac disease. Pediatr Adolesc Med Basel: Karger, 2008: 18-22. [Google Scholar]
- 47.Khatib M, Baker RD, Ly EK, Kozielski R, Baker SS. Presenting Pattern of Pediatric Celiac Disease. J Pediatr Gastroenterol Nutr. 2016;62:60–63. doi: 10.1097/MPG.0000000000000887. [DOI] [PubMed] [Google Scholar]
- 48.Kivelä L, Kaukinen K, Lähdeaho ML, Huhtala H, Ashorn M, Ruuska T, Hiltunen P, Visakorpi J, Mäki M, Kurppa K. Presentation of Celiac Disease in Finnish Children Is No Longer Changing: A 50-Year Perspective. J Pediatr. 2015;167:1109–15.e1. doi: 10.1016/j.jpeds.2015.07.057. [DOI] [PubMed] [Google Scholar]
- 49.Almallouhi E, King KS, Patel B, Wi C, Juhn YJ, Murray JA, Absah I. Increasing Incidence and Altered Presentation in a Population-based Study of Pediatric Celiac Disease in North America. J Pediatr Gastroenterol Nutr. 2017;65:432–437. doi: 10.1097/MPG.0000000000001532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bottaro G, Cataldo F, Rotolo N, Spina M, Corazza GR. The clinical pattern of subclinical/silent celiac disease: an analysis on 1026 consecutive cases. Am J Gastroenterol. 1999;94:691–696. doi: 10.1111/j.1572-0241.1999.00938.x. [DOI] [PubMed] [Google Scholar]
- 51.van Rijn JC, Grote FK, Oostdijk W, Wit JM. Short stature and the probability of coeliac disease, in the absence of gastrointestinal symptoms. Arch Dis Child. 2004;89:882–883. doi: 10.1136/adc.2004.057851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jericho H, Sansotta N, Guandalini S. Extraintestinal Manifestations of Celiac Disease: Effectiveness of the Gluten-Free Diet. J Pediatr Gastroenterol Nutr. 2017;65:75–79. doi: 10.1097/MPG.0000000000001420. [DOI] [PubMed] [Google Scholar]
- 53.Nurminen S, Kivelä L, Huhtala H, Kaukinen K, Kurppa K. Extraintestinal manifestations were common in children with coeliac disease and were more prevalent in patients with more severe clinical and histological presentation. Acta Paediatr. 2019;108:681–687. doi: 10.1111/apa.14324. [DOI] [PubMed] [Google Scholar]
- 54.Jericho H, Guandalini S. Extra-Intestinal Manifestation of Celiac Disease in Children. Nutrients. 2018;10:755. doi: 10.3390/nu10060755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Nardecchia S, Auricchio R, Discepolo V, Troncone R. Extra-Intestinal Manifestations of Coeliac Disease in Children: Clinical Features and Mechanisms. Front Pediatr. 2019;7:56. doi: 10.3389/fped.2019.00056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bonamico M, Sciré G, Mariani P, Pasquino AM, Triglione P, Scaccia S, Ballati G, Boscherini B. Short stature as the primary manifestation of monosymptomatic celiac disease. J Pediatr Gastroenterol Nutr. 1992;14:12–16. doi: 10.1097/00005176-199201000-00003. [DOI] [PubMed] [Google Scholar]
- 57.Gokce S, Arslantas E. Changing face and clinical features of celiac disease in children. Pediatr Int. 2015;57:107–112. doi: 10.1111/ped.12448. [DOI] [PubMed] [Google Scholar]
- 58.Hyer W, Cotterill AM, Savage MO. Common causes of short stature detectable by a height surveillance programme. J Med Screen. 1995;2:150–153. doi: 10.1177/096914139500200310. [DOI] [PubMed] [Google Scholar]
- 59.Saari A, Harju S, Mäkitie O, Saha MT, Dunkel L, Sankilampi U. Systematic growth monitoring for the early detection of celiac disease in children. JAMA Pediatr. 2015;169:e1525. doi: 10.1001/jamapediatrics.2015.25. [DOI] [PubMed] [Google Scholar]
- 60.Singh P, Sharma PK, Agnihotri A, Jyotsna VP, Das P, Gupta SD, Makharia GK, Khadgawat R. Coeliac disease in patients with short stature: A tertiary care centre experience. Natl Med J India. 2015;28:176–180. [PubMed] [Google Scholar]
- 61.Troncone R, Kosova R. Short stature and catch-up growth in celiac disease. J Pediatr Gastroenterol Nutr. 2010;51 Suppl 3:S137–S138. doi: 10.1097/MPG.0b013e3181f1dd66. [DOI] [PubMed] [Google Scholar]
- 62.Patwari AK, Kapur G, Satyanarayana L, Anand VK, Jain A, Gangil A, Balani B. Catch-up growth in children with late-diagnosed coeliac disease. Br J Nutr. 2005;94:437–442. doi: 10.1079/bjn20051479. [DOI] [PubMed] [Google Scholar]
- 63.Giovenale D, Meazza C, Cardinale GM, Sposito M, Mastrangelo C, Messini B, Citro G, Delvecchio M, Di Maio S, Bozzola M. The prevalence of growth hormone deficiency and celiac disease in short children. Clin Med Res. 2006;4:180–183. doi: 10.3121/cmr.4.3.180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Leffler DA, Green PH, Fasano A. Extraintestinal manifestations of coeliac disease. Nat Rev Gastroenterol Hepatol. 2015;12:561–571. doi: 10.1038/nrgastro.2015.131. [DOI] [PubMed] [Google Scholar]
- 65.Abaci A, Esen I, Unuvar T, Arslan N, Bober E. Two cases presenting with pubertal delay and diagnosed as Celiac disease. Clin Pediatr (Phila) 2008;47:607–609. doi: 10.1177/0009922808316185. [DOI] [PubMed] [Google Scholar]
- 66.Philip R, Patidar P, Saran S, Agarwal P, Arya T, Gupta K. Endocrine manifestations of celiac disease. Indian J Endocrinol Metab. 2012;16:S506–S508. doi: 10.4103/2230-8210.104149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Traggiai C, Stanhope R. Disorders of pubertal development. Best Pract Res Clin Obstet Gynaecol. 2003;17:41–56. doi: 10.1053/ybeog.2003.0360. [DOI] [PubMed] [Google Scholar]
- 68.Kalayci AG, Kanber Y, Birinci A, Yildiz L, Albayrak D. The prevalence of coeliac disease as detected by screening in children with iron deficiency anaemia. Acta Paediatr. 2005;94:678–681. doi: 10.1111/j.1651-2227.2005.tb01964.x. [DOI] [PubMed] [Google Scholar]
- 69.Baydoun A, Maakaron JE, Halawi H, Abou Rahal J, Taher AT. Hematological manifestations of celiac disease. Scand J Gastroenterol. 2012;47:1401–1411. doi: 10.3109/00365521.2012.706828. [DOI] [PubMed] [Google Scholar]
- 70.Äärelä L, Nurminen S, Kivelä L, Huhtala H, Mäki M, Viitasalo A, Kaukinen K, Lakka T, Kurppa K. Prevalence and associated factors of abnormal liver values in children with celiac disease. Dig Liver Dis. 2016;48:1023–1029. doi: 10.1016/j.dld.2016.05.022. [DOI] [PubMed] [Google Scholar]
- 71.Kaukinen K, Halme L, Collin P, Färkkilä M, Mäki M, Vehmanen P, Partanen J, Höckerstedt K. Celiac disease in patients with severe liver disease: gluten-free diet may reverse hepatic failure. Gastroenterology. 2002;122:881–888. doi: 10.1053/gast.2002.32416. [DOI] [PubMed] [Google Scholar]
- 72.Anania C, De Luca E, De Castro G, Chiesa C, Pacifico L. Liver involvement in pediatric celiac disease. World J Gastroenterol. 2015;21:5813–5822. doi: 10.3748/wjg.v21.i19.5813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lee GJ, Boyle B, Ediger T, Hill I. Hypertransaminasemia in Newly Diagnosed Pediatric Patients With Celiac Disease. J Pediatr Gastroenterol Nutr. 2016;63:340–343. doi: 10.1097/MPG.0000000000001153. [DOI] [PubMed] [Google Scholar]
- 74.Pantaleoni S, Luchino M, Adriani A, Pellicano R, Stradella D, Ribaldone DG, Sapone N, Isaia GC, Di Stefano M, Astegiano M. Bone mineral density at diagnosis of celiac disease and after 1 year of gluten-free diet. ScientificWorldJournal. 2014;2014:173082. doi: 10.1155/2014/173082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Keaveny AP, Freaney R, McKenna MJ, Masterson J, O'Donoghue DP. Bone remodeling indices and secondary hyperparathyroidism in celiac disease. Am J Gastroenterol. 1996;91:1226–1231. [PubMed] [Google Scholar]
- 76.Margoni D, Chouliaras G, Duscas G, Voskaki I, Voutsas N, Papadopoulou A, Panayiotou J, Roma E. Bone health in children with celiac disease assessed by dual x-ray absorptiometry: effect of gluten-free diet and predictive value of serum biochemical indices. J Pediatr Gastroenterol Nutr. 2012;54:680–684. doi: 10.1097/MPG.0b013e31823f5fc5. [DOI] [PubMed] [Google Scholar]
- 77.Björck S, Brundin C, Karlsson M, Agardh D. Reduced Bone Mineral Density in Children With Screening-detected Celiac Disease. J Pediatr Gastroenterol Nutr. 2017;65:526–532. doi: 10.1097/MPG.0000000000001568. [DOI] [PubMed] [Google Scholar]
- 78.Garg K, Agarwal P, Gupta RK, Sitaraman S. Joint Involvement in Children with Celiac Disease. Indian Pediatr. 2017;54:946–948. doi: 10.1007/s13312-017-1188-x. [DOI] [PubMed] [Google Scholar]
- 79.Casella G, Bordo BM, Schalling R, Villanacci V, Salemme M, Di Bella C, Baldini V, Bassotti G. Neurological disorders and celiac disease. Minerva Gastroenterol Dietol. 2016;62:197–206. [PubMed] [Google Scholar]
- 80.Lionetti E, Francavilla R, Pavone P, Pavone L, Francavilla T, Pulvirenti A, Giugno R, Ruggieri M. The neurology of coeliac disease in childhood: what is the evidence? Dev Med Child Neurol. 2010;52:700–707. doi: 10.1111/j.1469-8749.2010.03647.x. [DOI] [PubMed] [Google Scholar]
- 81.Ludvigsson JF, Zingone F, Tomson T, Ekbom A, Ciacci C. Increased risk of epilepsy in biopsy-verified celiac disease: a population-based cohort study. Neurology. 2012;78:1401–1407. doi: 10.1212/WNL.0b013e3182544728. [DOI] [PubMed] [Google Scholar]
- 82.Zoumpoulakis M, Fotoulaki M, Topitsoglou V, Lazidou P, Zouloumis L, Kotsanos N. Prevalence of Dental Enamel Defects, Aphthous-Like Ulcers and Other Oral Manifestations in Celiac Children and Adolescents: A Comparative Study. J Clin Pediatr Dent. 2019;43:274–280. doi: 10.17796/1053-4625-43.4.9. [DOI] [PubMed] [Google Scholar]
- 83.Macho VMP, de Barros Menéres Manso MCA, E Silva DMV, de Andrade DJC. The difference in symmetry of the enamel defects in celiac disease versus non-celiac pediatric population. J Dent Sci. 2020;15:345–350. doi: 10.1016/j.jds.2020.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Bucci P, Carile F, Sangianantoni A, D'Angiò F, Santarelli A, Lo Muzio L. Oral aphthous ulcers and dental enamel defects in children with coeliac disease. Acta Paediatr. 2006;95:203–207. doi: 10.1080/08035250500355022. [DOI] [PubMed] [Google Scholar]
- 85.Graziano M, Rossi M. An update on the cutaneous manifestations of coeliac disease and non-coeliac gluten sensitivity. Int Rev Immunol. 2018;37:291–300. doi: 10.1080/08830185.2018.1533008. [DOI] [PubMed] [Google Scholar]
- 86.Reunala T, Salmi TT, Hervonen K, Kaukinen K, Collin P. Dermatitis Herpetiformis: A Common Extraintestinal Manifestation of Coeliac Disease. Nutrients. 2018;10 doi: 10.3390/nu10050602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Kahaly GJ, Frommer L, Schuppan D. Celiac Disease and Glandular Autoimmunity. Nutrients. 2018;10:814. doi: 10.3390/nu10070814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Assa A, Frenkel-Nir Y, Tzur D, Katz LH, Shamir R. Large population study shows that adolescents with celiac disease have an increased risk of multiple autoimmune and nonautoimmune comorbidities. Acta Paediatr. 2017;106:967–972. doi: 10.1111/apa.13808. [DOI] [PubMed] [Google Scholar]
- 89.Akirov A, Pinhas-Hamiel O. Co-occurrence of type 1 diabetes mellitus and celiac disease. World J Diabetes. 2015;6:707–714. doi: 10.4239/wjd.v6.i5.707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hermann R, Turpeinen H, Laine AP, Veijola R, Knip M, Simell O, Sipilä I, Akerblom HK, Ilonen J. HLA DR-DQ-encoded genetic determinants of childhood-onset type 1 diabetes in Finland: an analysis of 622 nuclear families. Tissue Antigens. 2003;62:162–169. doi: 10.1034/j.1399-0039.2003.00071.x. [DOI] [PubMed] [Google Scholar]
- 91.Elfström P, Sundström J, Ludvigsson JF. Systematic review with meta-analysis: associations between coeliac disease and type 1 diabetes. Aliment Pharmacol Ther. 2014;40:1123–1132. doi: 10.1111/apt.12973. [DOI] [PubMed] [Google Scholar]
- 92.Sahin Y, Cakir MD, Isakoca M, Sahin DA. Prevalence of Celiac Disease in Children with Type 1 Diabetes Mellitus in the South of Turkey. Iran J Ped. 2020;30:e97306. [Google Scholar]
- 93.Weiss B, Pinhas-Hamiel O. Celiac Disease and Diabetes: When to Test and Treat. J Pediatr Gastroenterol Nutr. 2017;64:175–179. doi: 10.1097/MPG.0000000000001388. [DOI] [PubMed] [Google Scholar]
- 94.Hansen D, Brock-Jacobsen B, Lund E, Bjørn C, Hansen LP, Nielsen C, Fenger C, Lillevang ST, Husby S. Clinical benefit of a gluten-free diet in type 1 diabetic children with screening-detected celiac disease: a population-based screening study with 2 years' follow-up. Diabetes Care. 2006;29:2452–2456. doi: 10.2337/dc06-0990. [DOI] [PubMed] [Google Scholar]
- 95.Salardi S, Volta U, Zucchini S, Fiorini E, Maltoni G, Vaira B, Cicognani A. Prevalence of celiac disease in children with type 1 diabetes mellitus increased in the mid-1990 s: an 18-year longitudinal study based on anti-endomysial antibodies. J Pediatr Gastroenterol Nutr. 2008;46:612–614. doi: 10.1097/MPG.0b013e31815d697e. [DOI] [PubMed] [Google Scholar]
- 96.Pham-Short A, Donaghue KC, Ambler G, Chan AK, Craig ME. Coeliac disease in Type 1 diabetes from 1990 to 2009: higher incidence in young children after longer diabetes duration. Diabet Med. 2012;29:e286–e289. doi: 10.1111/j.1464-5491.2012.03720.x. [DOI] [PubMed] [Google Scholar]
- 97.Chiang JL, Maahs DM, Garvey KC, Hood KK, Laffel LM, Weinzimer SA, Wolfsdorf JI, Schatz D. Type 1 Diabetes in Children and Adolescents: A Position Statement by the American Diabetes Association. Diabetes Care. 2018;41:2026–2044. doi: 10.2337/dci18-0023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.van der Pals M, Myléus A, Norström F, Hammarroth S, Högberg L, Rosén A, Ivarsson A, Carlsson A. Body mass index is not a reliable tool in predicting celiac disease in children. BMC Pediatr. 2014;14:165. doi: 10.1186/1471-2431-14-165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Poulain C, Johanet C, Delcroix C, Lévy-Marchal C, Tubiana-Rufi N. Prevalence and clinical features of celiac disease in 950 children with type 1 diabetes in France. Diabetes Metab. 2007;33:453–458. doi: 10.1016/j.diabet.2007.06.004. [DOI] [PubMed] [Google Scholar]
- 100.Midhagen G, Järnerot G, Kraaz W. Adult coeliac disease within a defined geographic area in Sweden. A study of prevalence and associated diseases. Scand J Gastroenterol. 1988;23:1000–1004. doi: 10.3109/00365528809090160. [DOI] [PubMed] [Google Scholar]
- 101.Collin P, Salmi J, Hällström O, Reunala T, Pasternack A. Autoimmune thyroid disorders and coeliac disease. Eur J Endocrinol. 1994;130:137–140. doi: 10.1530/eje.0.1300137. [DOI] [PubMed] [Google Scholar]
- 102.Sategna-Guidetti C, Bruno M, Mazza E, Carlino A, Predebon S, Tagliabue M, Brossa C. Autoimmune thyroid diseases and coeliac disease. Eur J Gastroenterol Hepatol. 1998;10:927–931. doi: 10.1097/00042737-199811000-00005. [DOI] [PubMed] [Google Scholar]
- 103.Korponay-Szabó IR, Dahlbom I, Laurila K, Koskinen S, Woolley N, Partanen J, Kovács JB, Mäki M, Hansson T. Elevation of IgG antibodies against tissue transglutaminase as a diagnostic tool for coeliac disease in selective IgA deficiency. Gut. 2003;52:1567–1571. doi: 10.1136/gut.52.11.1567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Carlsson A, Axelsson I, Borulf S, Bredberg A, Forslund M, Lindberg B, Sjöberg K, Ivarsson SA. Prevalence of IgA-antigliadin antibodies and IgA-antiendomysium antibodies related to celiac disease in children with Down syndrome. Pediatrics. 1998;101:272–275. doi: 10.1542/peds.101.2.272. [DOI] [PubMed] [Google Scholar]
- 105.Gale L, Wimalaratna H, Brotodiharjo A, Duggan JM. Down's syndrome is strongly associated with coeliac disease. Gut. 1997;40:492–496. doi: 10.1136/gut.40.4.492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Bonamico M, Mariani P, Danesi HM, Crisogianni M, Failla P, Gemme G, Quartino AR, Giannotti A, Castro M, Balli F, Lecora M, Andria G, Guariso G, Gabrielli O, Catassi C, Lazzari R, Balocco NA, De Virgiliis S, Culasso F, Romano C SIGEP (Italian Society of Pediatric Gastroenterology and Hepatology) and Medical Genetic Group. Prevalence and clinical picture of celiac disease in italian down syndrome patients: a multicenter study. J Pediatr Gastroenterol Nutr. 2001;33:139–143. doi: 10.1097/00005176-200108000-00008. [DOI] [PubMed] [Google Scholar]
- 107.Book L, Hart A, Black J, Feolo M, Zone JJ, Neuhausen SL. Prevalence and clinical characteristics of celiac disease in Downs syndrome in a US study. Am J Med Genet. 2001;98:70–74. [PubMed] [Google Scholar]
- 108.Zachor DA, Mroczek-Musulman E, Brown P. Prevalence of celiac disease in Down syndrome in the United States. J Pediatr Gastroenterol Nutr. 2000;31:275–279. doi: 10.1097/00005176-200009000-00014. [DOI] [PubMed] [Google Scholar]
- 109.Liu E, Wolter-Warmerdam K, Marmolejo J, Daniels D, Prince G, Hickey F. Routine Screening for Celiac Disease in Children With Down Syndrome Improves Case Finding. J Pediatr Gastroenterol Nutr. 2020;71:252–256. doi: 10.1097/MPG.0000000000002742. [DOI] [PubMed] [Google Scholar]
- 110.Bonamico M, Pasquino AM, Mariani P, Danesi HM, Culasso F, Mazzanti L, Petri A, Bona G Italian Society Of Pediatric Gastroenterology Hepatology (SIGEP); Italian Study Group for Turner Syndrom (ISGTS) Prevalence and clinical picture of celiac disease in Turner syndrome. J Clin Endocrinol Metab. 2002;87:5495–5498. doi: 10.1210/jc.2002-020855. [DOI] [PubMed] [Google Scholar]
- 111.Gillett PM, Gillett HR, Israel DM, Metzger DL, Stewart L, Chanoine JP, Freeman HJ. Increased prevalence of celiac disease in girls with Turner syndrome detected using antibodies to endomysium and tissue transglutaminase. Can J Gastroenterol. 2000;14:915–918. doi: 10.1155/2000/172914. [DOI] [PubMed] [Google Scholar]
- 112.Ivarsson SA, Carlsson A, Bredberg A, Alm J, Aronsson S, Gustafsson J, Hagenäs L, Häger A, Kriström B, Marcus C, Moëll C, Nilsson KO, Tuvemo T, Westphal O, Albertsson-Wikland K, Aman J. Prevalence of coeliac disease in Turner syndrome. Acta Paediatr. 1999;88:933–936. doi: 10.1080/08035259950168397. [DOI] [PubMed] [Google Scholar]
- 113.Rujner J, Wisniewski A, Gregorek H, Wozniewicz B, Młynarski W, Witas HW. Coeliac disease and HLA-DQ 2 (DQA1* 0501 and DQB1* 0201) in patients with Turner syndrome. J Pediatr Gastroenterol Nutr. 2001;32:114–115. doi: 10.1097/00005176-200101000-00033. [DOI] [PubMed] [Google Scholar]
- 114.Nadeem M, Roche EF. Coeliac disease in Turner syndrome. Arch Dis Child. 2013;98:649–650. doi: 10.1136/archdischild-2013-304126. [DOI] [PubMed] [Google Scholar]
- 115.Mårild K, Størdal K, Hagman A, Ludvigsson JF. Turner Syndrome and Celiac Disease: A Case-Control Study. Pediatrics. 2016;137:e20152232. doi: 10.1542/peds.2015-2232. [DOI] [PubMed] [Google Scholar]
- 116.Giannotti A, Tiberio G, Castro M, Virgilii F, Colistro F, Ferretti F, Digilio MC, Gambarara M, Dallapiccola B. Coeliac disease in Williams syndrome. J Med Genet. 2001;38:767–768. doi: 10.1136/jmg.38.11.767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Ludvigsson JF, Leffler DA, Bai JC, Biagi F, Fasano A, Green PH, Hadjivassiliou M, Kaukinen K, Kelly CP, Leonard JN, Lundin KE, Murray JA, Sanders DS, Walker MM, Zingone F, Ciacci C. The Oslo definitions for coeliac disease and related terms. Gut. 2013;62:43–52. doi: 10.1136/gutjnl-2011-301346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Biagi F, Bianchi PI, Vattiato C, Marchese A, Trotta L, Badulli C, De Silvestri A, Martinetti M, Corazza GR. Influence of HLA-DQ2 and DQ8 on severity in celiac Disease. J Clin Gastroenterol. 2012;46:46–50. doi: 10.1097/MCG.0b013e318221077e. [DOI] [PubMed] [Google Scholar]
- 119.Husby S, Koletzko S, Korponay-Szabó I, Kurppa K, Mearin ML, Ribes-Koninckx C, Shamir R, Troncone R, Auricchio R, Castillejo G, Christensen R, Dolinsek J, Gillett P, Hróbjartsson A, Koltai T, Maki M, Nielsen SM, Popp A, Størdal K, Werkstetter K, Wessels M. European Society Paediatric Gastroenterology, Hepatology and Nutrition Guidelines for Diagnosing Coeliac Disease 2020. J Pediatr Gastroenterol Nutr. 2020;70:141–156. doi: 10.1097/MPG.0000000000002497. [DOI] [PubMed] [Google Scholar]
- 120.Trovato CM, Montuori M, Valitutti F, Leter B, Cucchiara S, Oliva S. The Challenge of Treatment in Potential Celiac Disease. Gastroenterol Res Pract. 2019;2019:8974751. doi: 10.1155/2019/8974751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Tosco A, Salvati VM, Auricchio R, Maglio M, Borrelli M, Coruzzo A, Paparo F, Boffardi M, Esposito A, D'Adamo G, Malamisura B, Greco L, Troncone R. Natural history of potential celiac disease in children. Clin Gastroenterol Hepatol. 2011;9:320–5; quiz e36. doi: 10.1016/j.cgh.2010.09.006. [DOI] [PubMed] [Google Scholar]
- 122.Volta U, Caio G, Boschetti E, Giancola F, Rhoden KJ, Ruggeri E, Paterini P, De Giorgio R. Seronegative celiac disease: Shedding light on an obscure clinical entity. Dig Liver Dis. 2016;48:1018–1022. doi: 10.1016/j.dld.2016.05.024. [DOI] [PubMed] [Google Scholar]
- 123.Shah VH, Rotterdam H, Kotler DP, Fasano A, Green PH. All that scallops is not celiac disease. Gastrointest Endosc. 2000;51:717–720. doi: 10.1067/mge.2000.104977. [DOI] [PubMed] [Google Scholar]
- 124.Greenson JK. The biopsy pathology of non-coeliac enteropathy. Histopathology. 2015;66:29–36. doi: 10.1111/his.12522. [DOI] [PubMed] [Google Scholar]
- 125.Mooney PD, Evans KE, Singh S, Sanders DS. Treatment failure in coeliac disease: a practical guide to investigation and treatment of non-responsive and refractory coeliac disease. J Gastrointestin Liver Dis. 2012;21:197–203. [PubMed] [Google Scholar]
- 126.Dewar DH, Donnelly SC, McLaughlin SD, Johnson MW, Ellis HJ, Ciclitira PJ. Celiac disease: management of persistent symptoms in patients on a gluten-free diet. World J Gastroenterol. 2012;18:1348–1356. doi: 10.3748/wjg.v18.i12.1348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Werkstetter KJ, Korponay-Szabó IR, Popp A, Villanacci V, Salemme M, Heilig G, Lillevang ST, Mearin ML, Ribes-Koninckx C, Thomas A, Troncone R, Filipiak B, Mäki M, Gyimesi J, Najafi M, Dolinšek J, Dydensborg Sander S, Auricchio R, Papadopoulou A, Vécsei A, Szitanyi P, Donat E, Nenna R, Alliet P, Penagini F, Garnier-Lengliné H, Castillejo G, Kurppa K, Shamir R, Hauer AC, Smets F, Corujeira S, van Winckel M, Buderus S, Chong S, Husby S, Koletzko S ProCeDE study group. Accuracy in Diagnosis of Celiac Disease Without Biopsies in Clinical Practice. Gastroenterology. 2017;153:924–935. doi: 10.1053/j.gastro.2017.06.002. [DOI] [PubMed] [Google Scholar]
- 128.Oberhuber G, Granditsch G, Vogelsang H. The histopathology of coeliac disease: time for a standardized report scheme for pathologists. Eur J Gastroenterol Hepatol. 1999;11:1185–1194. doi: 10.1097/00042737-199910000-00019. [DOI] [PubMed] [Google Scholar]
- 129.Rashid M, MacDonald A. Importance of duodenal bulb biopsies in children for diagnosis of celiac disease in clinical practice. BMC Gastroenterol. 2009;9:78. doi: 10.1186/1471-230X-9-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Hill ID, Fasano A, Guandalini S, Hoffenberg E, Levy J, Reilly N, Verma R. NASPGHAN Clinical Report on the Diagnosis and Treatment of Gluten-related Disorders. J Pediatr Gastroenterol Nutr. 2016;63:156–165. doi: 10.1097/MPG.0000000000001216. [DOI] [PubMed] [Google Scholar]
- 131.Bascuñán KA, Roncoroni L, Branchi F, Doneda L, Scricciolo A, Ferretti F, Araya M, Elli L. The 5 Ws of a gluten challenge for gluten-related disorders. Nutr Rev. 2018;76:79–87. doi: 10.1093/nutrit/nux068. [DOI] [PubMed] [Google Scholar]
- 132.Poddighe D, Turganbekova A, Baymukasheva D, Saduakas Z, Zhanzakova Z, Abdrakhmanova S. Genetic predisposition to celiac disease in Kazakhstan: Potential impact on the clinical practice in Central Asia. PLoS One. 2020;15:e0226546. doi: 10.1371/journal.pone.0226546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Volta U, Caio G, De Giorgio R. Mistakes in coeliac disease diagnosis and how to avoid them. UEG Education. 2016;16:1–3. [Google Scholar]
- 134.Singh P, Arora A, Strand TA, Leffler DA, Mäki M, Kelly CP, Ahuja V, Makharia GK. Diagnostic Accuracy of Point of Care Tests for Diagnosing Celiac Disease: A Systematic Review and Meta-Analysis. J Clin Gastroenterol. 2019;53:535–542. doi: 10.1097/MCG.0000000000001081. [DOI] [PubMed] [Google Scholar]
- 135.Husby S, Bai JC. Follow-up of Celiac Disease. Gastroenterol Clin North Am. 2019;48:127–136. doi: 10.1016/j.gtc.2018.09.009. [DOI] [PubMed] [Google Scholar]
- 136.Bishop J, Ravikumara M. Coeliac disease in childhood: An overview. J Paediatr Child Health. 2020;56:1685–1693. doi: 10.1111/jpc.14674. [DOI] [PubMed] [Google Scholar]
- 137.Newton KP, Singer SA. Celiac disease in children and adolescents: special considerations. Semin Immunopathol. 2012;34:479–496. doi: 10.1007/s00281-012-0313-0. [DOI] [PubMed] [Google Scholar]
- 138.Catassi C, Fabiani E, Iacono G, D'Agate C, Francavilla R, Biagi F, Volta U, Accomando S, Picarelli A, De Vitis I, Pianelli G, Gesuita R, Carle F, Mandolesi A, Bearzi I, Fasano A. A prospective, double-blind, placebo-controlled trial to establish a safe gluten threshold for patients with celiac disease. Am J Clin Nutr. 2007;85:160–166. doi: 10.1093/ajcn/85.1.160. [DOI] [PubMed] [Google Scholar]
- 139.Akobeng AK, Thomas AG. Systematic review: tolerable amount of gluten for people with coeliac disease. Aliment Pharmacol Ther. 2008;27:1044–1052. doi: 10.1111/j.1365-2036.2008.03669.x. [DOI] [PubMed] [Google Scholar]
- 140.Ventura A, Magazzù G, Greco L. Duration of exposure to gluten and risk for autoimmune disorders in patients with celiac disease. SIGEP Study Group for Autoimmune Disorders in Celiac Disease. Gastroenterology. 1999;117:297–303. doi: 10.1053/gast.1999.0029900297. [DOI] [PubMed] [Google Scholar]
- 141.Andrén Aronsson C, Lee HS, Hård Af Segerstad EM, Uusitalo U, Yang J, Koletzko S, Liu E, Kurppa K, Bingley PJ, Toppari J, Ziegler AG, She JX, Hagopian WA, Rewers M, Akolkar B, Krischer JP, Virtanen SM, Norris JM, Agardh D TEDDY Study Group. Association of Gluten Intake During the First 5 Years of Life With Incidence of Celiac Disease Autoimmunity and Celiac Disease Among Children at Increased Risk. JAMA. 2019;322:514–523. doi: 10.1001/jama.2019.10329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Ludvigsson JF. Mortality and malignancy in celiac disease. Gastrointest Endosc Clin N Am. 2012;22:705–722. doi: 10.1016/j.giec.2012.07.005. [DOI] [PubMed] [Google Scholar]
- 143.Valitutti F, Trovato CM, Montuori M, Cucchiara S. Pediatric Celiac Disease: Follow-Up in the Spotlight. Adv Nutr. 2017;8:356–361. doi: 10.3945/an.116.013292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Leonard MM, Fasano A. Zero, One, or Two Endoscopies to Diagnose and Monitor Pediatric Celiac Disease? J Pediatr Gastroenterol Nutr. 2017;65:270–271. doi: 10.1097/MPG.0000000000001666. [DOI] [PubMed] [Google Scholar]
- 145.Mahadev S, Gardner R, Lewis SK, Lebwohl B, Green PH. Quality of Life in Screen-detected Celiac Disease Patients in the United States. J Clin Gastroenterol. 2016;50:393–397. doi: 10.1097/MCG.0000000000000433. [DOI] [PubMed] [Google Scholar]
- 146.Webb C, Myléus A, Norström F, Hammarroth S, Högberg L, Lagerqvist C, Rosén A, Sandström O, Stenhammar L, Ivarsson A, Carlsson A. High adherence to a gluten-free diet in adolescents with screening-detected celiac disease. J Pediatr Gastroenterol Nutr. 2015;60:54–59. doi: 10.1097/MPG.0000000000000571. [DOI] [PubMed] [Google Scholar]
- 147.Barnea L, Mozer-Glassberg Y, Hojsak I, Hartman C, Shamir R. Pediatric celiac disease patients who are lost to follow-up have a poorly controlled disease. Digestion. 2014;90:248–253. doi: 10.1159/000368395. [DOI] [PubMed] [Google Scholar]
- 148.Wessels MM, van Veen II, Vriezinga SL, Putter H, Rings EH, Mearin ML. Complementary Serologic Investigations in Children with Celiac Disease Is Unnecessary during Follow-Up. J Pediatr. 2016;169:55–60. doi: 10.1016/j.jpeds.2015.09.078. [DOI] [PubMed] [Google Scholar]
- 149.Husby S, Murray JA, Katzka DA. AGA Clinical Practice Update on Diagnosis and Monitoring of Celiac Disease-Changing Utility of Serology and Histologic Measures: Expert Review. Gastroenterology. 2019;156:885–889. doi: 10.1053/j.gastro.2018.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Dipper CR, Maitra S, Thomas R, Lamb CA, McLean-Tooke AP, Ward R, Smith D, Spickett G, Mansfield JC. Anti-tissue transglutaminase antibodies in the follow-up of adult coeliac disease. Aliment Pharmacol Ther. 2009;30:236–244. doi: 10.1111/j.1365-2036.2009.04039.x. [DOI] [PubMed] [Google Scholar]
- 151.Isaac DM, Rajani S, Yaskina M, Huynh HQ, Turner JM. Antitissue Transglutaminase Normalization Postdiagnosis in Children With Celiac Disease. J Pediatr Gastroenterol Nutr. 2017;65:195–199. doi: 10.1097/MPG.0000000000001480. [DOI] [PubMed] [Google Scholar]
- 152.Mahadev S, Murray JA, Wu TT, Chandan VS, Torbenson MS, Kelly CP, Maki M, Green PH, Adelman D, Lebwohl B. Factors associated with villus atrophy in symptomatic coeliac disease patients on a gluten-free diet. Aliment Pharmacol Ther. 2017;45:1084–1093. doi: 10.1111/apt.13988. [DOI] [PubMed] [Google Scholar]
- 153.Moreno ML, Cebolla Á, Muñoz-Suano A, Carrillo-Carrion C, Comino I, Pizarro Á, León F, Rodríguez-Herrera A, Sousa C. Detection of gluten immunogenic peptides in the urine of patients with coeliac disease reveals transgressions in the gluten-free diet and incomplete mucosal healing. Gut. 2017;66:250–257. doi: 10.1136/gutjnl-2015-310148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Syage JA, Kelly CP, Dickason MA, Ramirez AC, Leon F, Dominguez R, Sealey-Voyksner JA. Determination of gluten consumption in celiac disease patients on a gluten-free diet. Am J Clin Nutr. 2018;107:201–207. doi: 10.1093/ajcn/nqx049. [DOI] [PubMed] [Google Scholar]
- 155.West J, Logan RF, Card TR, Smith C, Hubbard R. Risk of vascular disease in adults with diagnosed coeliac disease: a population-based study. Aliment Pharmacol Ther. 2004;20:73–79. doi: 10.1111/j.1365-2036.2004.02008.x. [DOI] [PubMed] [Google Scholar]
- 156.Hallert C, Grant C, Grehn S, Grännö C, Hultén S, Midhagen G, Ström M, Svensson H, Valdimarsson T. Evidence of poor vitamin status in coeliac patients on a gluten-free diet for 10 years. Aliment Pharmacol Ther. 2002;16:1333–1339. doi: 10.1046/j.1365-2036.2002.01283.x. [DOI] [PubMed] [Google Scholar]
- 157.Midhagen G, Hallert C. High rate of gastrointestinal symptoms in celiac patients living on a gluten-free diet: controlled study. Am J Gastroenterol. 2003;98:2023–2026. doi: 10.1111/j.1572-0241.2003.07632.x. [DOI] [PubMed] [Google Scholar]
- 158.Aziz I, Evans KE, Papageorgiou V, Sanders DS. Are patients with coeliac disease seeking alternative therapies to a gluten-free diet? J Gastrointestin Liver Dis. 2011;20:27–31. [PubMed] [Google Scholar]
- 159.Rashid M, Cranney A, Zarkadas M, Graham ID, Switzer C, Case S, Molloy M, Warren RE, Burrows V, Butzner JD. Celiac disease: evaluation of the diagnosis and dietary compliance in Canadian children. Pediatrics. 2005;116:e754–e759. doi: 10.1542/peds.2005-0904. [DOI] [PubMed] [Google Scholar]
- 160.Leffler DA, Kelly CP, Green PH, Fedorak RN, DiMarino A, Perrow W, Rasmussen H, Wang C, Bercik P, Bachir NM, Murray JA. Larazotide acetate for persistent symptoms of celiac disease despite a gluten-free diet: a randomized controlled trial. Gastroenterology. 2015;148:1311–9.e6. doi: 10.1053/j.gastro.2015.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Lähdeaho ML, Kaukinen K, Laurila K, Vuotikka P, Koivurova OP, Kärjä-Lahdensuu T, Marcantonio A, Adelman DC, Mäki M. Glutenase ALV003 attenuates gluten-induced mucosal injury in patients with celiac disease. Gastroenterology. 2014;146:1649–1658. doi: 10.1053/j.gastro.2014.02.031. [DOI] [PubMed] [Google Scholar]
- 162.Murray JA, Kelly CP, Green PHR, Marcantonio A, Wu TT, Mäki M, Adelman DC CeliAction Study Group of Investigators. No Difference Between Latiglutenase and Placebo in Reducing Villous Atrophy or Improving Symptoms in Patients With Symptomatic Celiac Disease. Gastroenterology. 2017;152:787–798.e2. doi: 10.1053/j.gastro.2016.11.004. [DOI] [PubMed] [Google Scholar]
- 163.Anderson RP, Jabri B. Vaccine against autoimmune disease: antigen-specific immunotherapy. Curr Opin Immunol. 2013;25:410–417. doi: 10.1016/j.coi.2013.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Rubio-Tapia A, Ludvigsson JF, Choung RS, Brantner TL, Rajkumar SV, Landgren O, Murray JA. Increased mortality among men aged 50 years old or above with elevated IgA anti-transglutaminase antibodies: NHANES III. BMC Gastroenterol. 2016;16:136. doi: 10.1186/s12876-016-0547-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Biagi F, Gobbi P, Marchese A, Borsotti E, Zingone F, Ciacci C, Volta U, Caio G, Carroccio A, Ambrosiano G, Mansueto P, Corazza GR. Low incidence but poor prognosis of complicated coeliac disease: a retrospective multicentre study. Dig Liver Dis. 2014;46:227–230. doi: 10.1016/j.dld.2013.10.010. [DOI] [PubMed] [Google Scholar]
- 166.Al-Toma A, Goerres MS, Meijer JW, Peña AS, Crusius JB, Mulder CJ. Human leukocyte antigen-DQ2 homozygosity and the development of refractory celiac disease and enteropathy-associated T-cell lymphoma. Clin Gastroenterol Hepatol. 2006;4:315–319. doi: 10.1016/j.cgh.2005.12.011. [DOI] [PubMed] [Google Scholar]