Abstract
The purpose of this study was to compare the polyphenol, antioxidant and antidiabetic potential of powdered Matcha and bagged Sencha tea during in vitro digestion. Total phenols (TP), flavonoids (TF), flavanols (TFLA), antioxidant and antidiabetic (α-glucosidase inhibition) activity were higher in Matcha tea before and in most in vitro digestion phases. Upon gastric digestion, in Matcha tea TP, TF, TFLA were 2.6, 1.4 and 1.2 times significantly higher (p ≤ 0.05), respectively; gallic acid, gallocatechin, epigallocatechin, quercetin and kaempferol 1.5, 1.6, 1.8, 1.7, 1.2 times, respectively; whereas antioxidant activity was significantly (p ≤ 0.05) higher 3.2 and 1.1 times with ABTS and FRAP and α-glucosidase inhibition 1.8 times. After the intestinal phase, TP and TFLA were 3.4 and 1.7 times significantly (p ≤ 0.05) higher, respectively, antioxidant activity was significantly (p ≤ 0.05) higher 2.4 and 2.0 times with ABTS and FRAP, respectively, while inhibition of α-glucosidase was 1.7 time significantly (p ≤ 0.05) higher in Matcha tea, but the differences in TF, TP and identified phenolics (with the exception of gallic acid) between Matcha and Sencha tea were neutralized. Our results are the first to demonstrate that, during digestion of Matcha powder together with its water extract, Matcha polyphenols are more bioavailable and possess higher antioxidant and antidiabetic activity compared to Sencha.
Supplementary Information
The online version contains supplementary material available at 10.1007/s13197-021-05086-5.
Keywords: Camellia sinensis, Flavonoids, Phenolic acids, RP-HPLC, Antioxidant activity, Antidiabetic activity
Introduction
Tea, an infusion of Camellia sinensis dried leaves, is one of the most popular beverages worldwide, with a long tradition of consumption, especially in the East. It has aroused great interest from scientists ever since the first relevant scientific data about its beneficial health effects were published. In the last two decades, tea has been investigated from different points of view. Moreover, many different forms of Camellia sinensis have appeared on the market. All of them have been subjected to extensive scientific investigations and their biochemical composition and impacts on human health have been compared (Rusak et al. 2008; Fu et al. 2017; Nayyar et al. 2017; Meng et al. 2019; Corrêa et al. 2020). The most familiar teas are green, black, oolong and white tea, which differ from one another in the processing of the leaves, i.e. in the degree of oxidation. In the manufacturing process of green tea, the leaves of C. sinensis are harvested, withered, rolled and quickly heated or steamed with the aim to prevent the enzymatic degradation of polyphenols. Disruption of cellular compartments after harvesting brings polyphenols into contact with polyphenol oxidases. Heating inhibits the enzymes involved in enzymatic oxidation of polyphenols but also other enzymes such as the esterases, glycosidases and decarbocxylases involved in polyphenol transformation. The predominant bioactive compounds in tea are phenolics, secondary metabolites involved in a wide range of physiological processes in plants as well as substances with significant implications for human health. The major phenolics in tea are flavan-3-ols, also known as catechins, which constitute up to 30% of the dry weight of tea leaves. In a smaller amount, flavonols (up to 4%) and flavones (in traces) are also present in tea leaves (Rusak et al. 2008).
A broad spectrum of beneficial effects of bioactive substances from tea on human health has been proven including anticancer, anticollagenase, antidiabetic, antihypertensive, antimicrobial, antiobesity and antioxidant activities (Nayyar et al. 2017). According to Corrêa et al. (2020), metabolic syndrome involves a cluster of interrelated prejudicial conditions (hyperglycemia, abdominal/visceral obesity, dyslipidemia, and hypertension) that increase risk of type 2 diabetes and cardiovascular diseases. Recent in vitro, in vivo and clinical studies have confirmed that green tea catechins, mainly epigallocatechin-gallate (EGCG) have various antidiabetic activities on type 2 diabetes mellitus (Fu et al. 2017, Corrêa et al. 2020). According to Meng et al. (2019), the possible mechanisms of tea to manage diabetes mellitus are amelioration of insulin resistance, activation of the insulin signaling pathway, playing an insulin-like role, scavenging free radicals and alleviating inflammatory response. An epidemiological study based on a follow-up investigation of 82 369 Japanese people over 13 years showed that green tea consumption lowers the risk of cardiovascular diseases and stroke (Kokubo et al. 2013). Moreover, a follow-up study on 90 914 Japanese people across 18.7 years revealed that green tea reduces the risk of total mortality and some of the leading causes of death such as heart, cerebrovascular and respiratory diseases (Saito et al. 2015).
Evidence regarding the health benefits of green tea consumption therefore appeals to health-conscious people. The appearance of any new form of tea on the market opens the question if this particular variation possesses more powerful effects on human health than the ones before it. That same question was raised once Matcha tea appeared on the market. To produce Matcha tea, three to four weeks before harvesting C. sinensis, plants are 90% shaded which increases the maturing period. After harvesting, leaves undergo steaming and drying, as would a common green tea grown in sunlight (Sencha tea). Lately, Matcha tea has become a very popular beverage despite the fact that its price is significantly higher in comparison with Sencha tea. There is no scientific answer to whether the high price is justified by any kind of significantly higher bioactive potential of Matcha compared to Sencha tea. There is a significant difference in the way how Matcha and Sencha tea are consumed; Matcha tea consumption implies taking tea powder together with its water extract, whereas ingestion of Sencha tea includes a water extract only.
So far, only the bioactive compounds and antioxidant activity of Matcha and Sencha tea water extracts have been compared (Komes et al. 2010); however a much more relevant insight into the bioactive potential of these two green tea forms could be obtained upon digestion. There are no available data on the polyphenol composition and antioxidant and antidiabetic activity of Matcha and Sencha tea during digestion. Therefore, the goal of our study was to determine and compare the polyphenol composition and in vitro digestive stability as well as the effect of digestion on antioxidant and antidiabetic activity of polyphenols from two different types of green tea, powdered Matcha and bagged Sencha tea. To achieve this, we performed: a) a spectrophotometric determination and comparison of total phenols, flavonoids, as well as flavanols in Matcha and Sencha tea before and after salivary, gastric and intestinal digestion; b) a determination and comparison of antioxidant activity of Matcha and Sencha tea before and after salivary, gastric and intestinal digestion; c) a quantification of phenols and their comparison in the investigated tea forms before and after salivary, gastric and intestinal digestion using an already developed HPLC method; d) a determination and comparison of the antidiabetic activity of Matcha and Sencha tea before and after gastric and intestinal digestion in order to yield a better complementary strategy to control glucose absorption in the treatment of type 2 diabetes.
We presumed that digestion diminishes the difference between Matcha and Sencha tea biopotential. The difference in how Matcha (water extract together with powder) and Sencha tea (water extract only) are taken has an impact on the bioavailability of phenolics and the antioxidative and antidiabetic potential of these two teas. These differences could be proven using an in vitro digestion method.
Material and methods
Chemicals and materials
Enzymes were purchased from Sigma Aldrich GmbH (Taufkirchen, Germany). All chemicals and reagents were of analytical grade and supplied by Sigma Aldrich GmbH (Taufkirchen, Germany) or Kemika (Zagreb, Croatia). Standards were HPLC grade and purchased from Sigma Aldrich (GmbH (Taufkirchen, Germany) or from Extrasynthese (Genay, France).
Matcha powdered green tea and Sencha bagged tea (Suppl. Figure 1.) were purchased at a local store. Matcha powdered green tea was prepared at a concentration of 10 g/L as follows: 100 mL of deionized water (80 °C) was poured over powder (1 g) and whisked vigorously with a glass stick for 3 min. Bagged Sencha green tea was prepared at the same concentration (10 g/L) as Matcha tea: 100 mL of deionized water (80 °C) was poured over a bag (1 g) of tea and incubated for 3 min without whisking. Tea extracts were cooled down on 25 °C under running water flow before the in vitro digestion experiment and stored at − 20 °C until spectrophotometric and HPLC analysis.
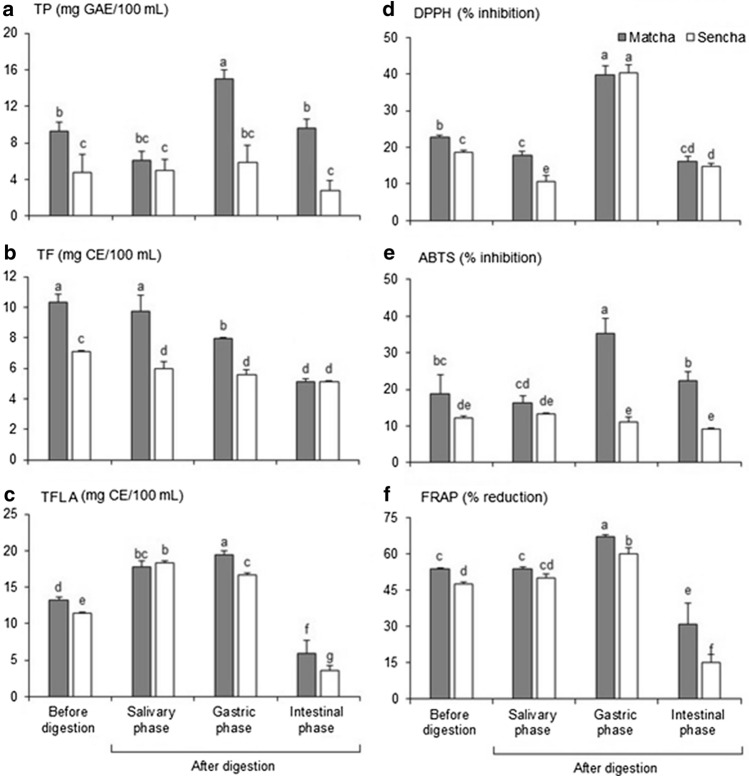
Fig. 1.
Polyphenol content: a total phenolics (TP), b total flavonoids (TF) and c total flavanols (TFL) and antioxidant activity (d DPPH; % inhibition, e ABTS; % inhibition and f FRAP; % reduction) in Matcha and Sencha green tea before and after exposure to in vitro digestive condition. Values represent mean ± SD of 3 replicates. Different letters indicate significant difference at p < 0.05. GAE = gallic acid equivalent, CE = catechin equivalent
In vitro digestion
The in vitro digestion model was performed as described in Šola et al. (2020a, b) with slight modification. First, a volume of 0.75 mL Matcha or Sencha tea was mixed with the same volume of 20 mM phosphate buffer pH 7.0. To initialize the salivary phase of digestion, 25 μL of amylase (0.48 mg/mL in 20 mM phosphate buffer pH 7.0) was added and incubated for 5 min at 37 °C in a shaking water bath at 150 rpm. For simulating stomach digestion, a volume of 1 mL of porcine pepsin solution (3 mg/mL in 0.1 M HCl) was added and acidified with 0.5 M HCl (pH 2.0). Samples were incubated in a shaking water bath for 1 h at 37 °C at 150 rpm. The upper intestinal phase of digestion was mimicked first by adding sodium bicarbonate (1 M NaHCO3) to adjust the pH to 5.3. After pH adjustment, a volume of 2.25 mL of pancreatic juices (2.4 mg bile acids/mL, 0.2 mg porcine lipase/mL, 0.4 mg pancreatin/mL in 20 mM phosphate buffer pH 7.0) was added. The final total volume of each intestinal phase sample was brought to 5 mL with 20 mM phosphate buffer (pH 7.0). The final pH was adjusted additionally to 7.0 with 1 M NaOH. Samples were then incubated for 2 h at 37 °C in a shaking water bath at 150 rpm. The final volume of each sample, both before and after digestion, was brought to 5 mL with 20 mM phosphate buffer (pH 7.0). Samples were centrifuged at 11 000 rpm for 10 min at 4 °C and supernatants were stored at − 20 °C until spectrophotometric and HPLC analyses.
Phytochemical analysis
The polyphenolic compounds were assessed in powdered Matcha and bagged Sencha predigested tea at a final concentration of 10 g/L and in tea samples before (tea and 20 mM phosphate buffer pH 7.0) and after the salivary, gastric and intestinal phases of digestion at a final tea concentration of 1.5 g/L.
Total soluble phenols (TP) of predigested tea and tea samples before and after the salivary, gastric and intestinal phases of digestion were determined with Foline-Ciocalteau reagent according to Zhishen et al. (1999). A volume 2 μL of tested solution was diluted with 158 μL of distilled water and then 10 μL of Foline-Ciocalteau reagent was added. Afterwards, 30 μL Na2CO3 (1.88 M) was added and the mixture was incubated for 30 min at 45 °C. The absorbance of the mixture was measured at 740 nm. The TP content was calculated from the calibration curve and expressed as gallic acid equivalents (GAE).
The content of total flavonoids (TF) of predigested and tea samples before and after all phases of digestion was determined with AlCl3 according to Zhishen et al. (1999). To dilute the tested solution (2 μL in 80 μL of dH2O), a volume of 6 μL NaNO2 (5%) was added. After 5 min incubation, volume of 6 μL AlCl3 (10%) was added and mixture was incubated at room temperature for additional 6 min. Afterwards, 40 μL NaOH (1 M) and distilled water were added to final volume of 200 μL. The absorbance of the reaction mixture was read at 520 nm. The TF content was calculated from the calibration curve and expressed as catechin equivalents (CE).
The total flavanol (TFL) content was determined using p-dimethylaminocinnamaldehyde (DMACA) according to Kusznierewicz et al. (2008). A volume of 100 μL of tested solution was mixed with 150 μL of DMACA solution (0.1% in 1 M HCl in MeOH). After 10 min of incubation at room temperature, absorbance at 595 nm was measured. TFL content was calculated from the calibration curve and expressed as catechin equivalents (CE).
RP-HPLC analysis of polyphenol compounds
Before HPLC analysis, samples were hydrolyzed as follows: 125 µL of each extract was mixed with 1.2 M HCl and incubated for 2 h at 80 °C and 300 rpm. The solutions were centrifuged three times (13,000 rpm, 5 min) and the supernatants stored at -20 °C until analysis.
Qualitative and quantitative RP-HPLC analysis was performed using the Agilent 1100 Series system equipped with a quaternary pump, multiwave UV/Vis detector, autosampler, fraction collector, Zorbax SB C-18 analytical guard column (12.5 × 4.6 mm, 5 µm particle size) and Poroshell 120 EC-C18 column (100 × 4.6 mm, 4 µm particle size) (Agilent Technologies, Waldbronn, Germany). The solvents used were: (A) 0.2% (v/v) glacial acetic acid, (B) 40% acetonitrile, 40% methanol and 0.2% glacial acetic acid. The gradient profile was (A/B): 100/0 at 0 min, 50/50 at 25 min, 50/50 at 30 min, 0/100 at 35 min. The injection volume was 50 µL, the constant flow rate 1.0 mL/min, and the column temperature 30 °C. For quantification, the multiwave UV/Vis detector was set at 210 nm for flavanols, 280 nm for gallic acid, 310 nm for p-coumaric acid and 360 nm for flavonoid determination. Phenolic compounds were characterized according to their retention times and UV spectra compared with commercial standards. For quantitative analyses, calibration curves were obtained by an injection of 5 known concentrations (in the range 1–250 µg/mL) of the mixed 96% EtOH standard solution in triplicate. The injection volume was 5 µL. Phenolic quantification was made by an integration of peak areas with reference to the calibration curves made using known amounts of available pure standard compounds. The results were expressed as mg/L of tea ± SD.
Antioxidant activity
The same Matcha and Sencha predigested tea and tea samples before (tea and 20 mM phosphate buffer pH 7.0) and after the salivary, gastric and intestinal phase were used for antioxidant activity assays. DPPH and ABTS radical scavenging and ferric reducing/antioxidant power (FRAP) assay was performed as reported by Šola et al. (2020a) adapted to small volumes. The results are expressed in percentage of inhibition. Trolox was used as a positive control for all three methods.
Antidiabetic activity
The antidiabetic properties of Matcha and Sencha tea samples through the inhibition of porcine pancreatic α-amylase were tested using the pre-incubation method as described by Šola et al. (2020b) and through the inhibition of α-glucosidase from Saccharomyces cerevisiae using the pre-incubation method as described by Salahuddin et al. (2020) with slight modification. In brief, the application of the modified method included mixing tea samples (10 μL) with 10 μL α-glucosidase (1.5 U/mL in phosphate buffer) and 130 μL phosphate buffer (50 mM, pH 6.5) and then pre-incubating for 15 min at 37 °C. A volume of 50 μL p-nitrophenyl-α-D-glucopyranoside (1 mM in phosphate buffer) was added and re-incubated for 5 min at 37 °C. The enzyme reaction activity was terminated by an addition of 50 μL Na2CO3 (0.1 M, pH 10). The absorbance was measured at 405 nm. Appropriate blanks and controls were carried out for both methods. Enzyme inhibitory activity was calculated from the equation: where At was the absorbance of the test (with enzyme), Atb was the absorbance of test blank (without enzyme), Ac was the absorbance of control (with enzyme) and Acb was the absorbance of the control blank (without enzyme). Acarbose (10 and 1.5 g/L) was used as a positive control for both methods.
All absorbance measurements were performed with a Fluostar Optima microplate reader (BMG Labtech GmbH, Offenburg, Germany).
Statistical analysis
All results were evaluated using Statistica 12.0 software package (Stat Soft Inc., USA). RP-HPLC and results from polyphenolics and antioxidative assays were subjected to one-way ANOVA for comparison of means. Significant differences were calculated according to Duncan's multiple range test. Pearson correlation coefficients between the polyphenolic content, antioxidant and antidiabetic activity were calculated. Data were considered statistically significant at p ≤ 0.05.
Results and discussion
Matcha and Sencha tea grow in different conditions (Sencha in intensive sun light, Matcha in 90% shade for 3–4 weeks). The processing procedures of Matcha and Sencha tea are also different. Sencha is rolled and packed in bags, while Matcha is finely grounded in a stone mill and vacuum packed in small containers. Furthermore, their preparation and manner of consumption are also different. Powdered Matcha is whisked vigorously with a whisk for 3 min and suspended leaves are consumed. Sencha bagged tea is incubated for 3 min without whisking and filtered water extract is consumed (Weiss and Anderton 2003). All these differences may affect the content of bioactive compounds and impact the bioavailability of phenolics and antioxidative and antidiabetic during in vitro digestion.
Phytochemical analysis
TP and TF content were significantly higher in predigested Matcha than Sencha tea samples (Table 1), which is in accordance with the results of Komes et al. (2010). In our experiment, TFLA (Table 1) in Matcha tea was 1.1 times higher than in Sencha tea, but this was not statistically significant (p ≤ 0.05). DMACA spectrophotometric method is less sensitive for TFLA determination with catechin as a standard (Khoddami et al. 2013), so HPLC analysis was also performed. The highest TP and TFLA content in the in vitro digestion model was detected in Matcha tea samples after the gastric phase of digestion (Fig. 1). We assumed that an acid environment (pH 2.0) in gastric digestion can increase the availability of captured polyphenols in suspended leaves through chemical degradation of cell walls. Also, in acidic solutions tea polyphenols (Friedman et al. 2009), especially tea flavanols (catechins), are very stable (Zhou et al. 2002). Wootton-Beard et al. (2011) also detected an increase in TP content after the gastric phase of vegetable juice digestion. TP content in different plant infusions was enhanced also following the gastric digestion phase in Coe et al. (2013). The TP and TFLA values of Matcha tea samples were statistically higher than those of Sencha samples before digestion and after the gastric and intestinal phase. This could be explained with a more efficient extraction from finely grounded leaf powder during the vigorous whisking of Matcha powder. A low pH in gastric environments can induce hydrolysis of polyphenols and make it, in the form of aglycones, more available for absorption at the gastric level and intestinum (Manach et al. 2004). The highest TF content was detected in Matcha tea before and after the salivary phase of digestion. TF content was significantly higher in Matcha tea before and after digestion (salivary and gastric phase). TF content in Matcha tea significantly dropped after the gastric (1.3 times) and intestinal phase (2 times). In Sencha tea samples, TF significantly dropped after all three phases of digestion in comparison to the initial phase. After the intestinal phase, the TP contents of Matcha and Sencha tea samples dropped to their initial level before digestion, while the TF and TFLA of Matcha and Sencha tea samples dropped significantly below their initial values. Coe et al. (2013) and Wootton-Beard et al. (2011) also detected a decrease in TP content after the intestinal phase in the four tested juices and in green tea relative to the gastric phase. After the salivary and gastric phase, TFLA increased significantly in Matcha and Sencha tea samples and decreased after the intestinal phase compared to the initial phase of digestion. Recovery of green tea TP content after the gastric phase was 90.7% and after the intestinal 35.2% in Okello et al. (2011). This great recovery of TP agreed with the highest TP content in Matcha tea samples after the gastric phase as well as with the decrease in TP content after the intestinal phase of digestion.
Table 1.
Polyphenol content (total phenolics (TP), total flavonoids (TF) and total flavanols (TFLA)), antioxidant activity (DPPH, ABTS and FRAP) and antidiabetic activity (α-glucosidase and α-amylase inhibitory activity) in Matcha and Sencha pure green tea
| TP (mg GAE/100 mL) | TF (mg CE/100 mL) | TFLA (mg CE/100 mL) | α-glucosidase (% inhibition) | |
|---|---|---|---|---|
| Matcha | 129.02 ± 13.20a | 22.95 ± 2.08a | 53.6 ± 52.07a | 99.67 ± 0.88a |
| Sencha | 66.76 ± 5.66 b | 15.29 ± 1.06b | 51.19 ± 5.31a | 102.72 ± 8.83a |
| DPPH (% inhibition) | ABTS (% inhibition) | FRAP (% reduction) | α-amylase (% inhibition) | |
|---|---|---|---|---|
| Matcha | 79.90 ± 0.68a | 78.98 ± 1.16a | 94.06 ± 0.17a | ND |
| Sencha | 81.59 ± 0.12a | 72.43 ± 2.67b | 93.92 ± 0.09a | ND |
| Trolox | 81.95 ± 0.11 | 81.37 ± 1.06 | 97.21 ± 0.24 |
Values represent mean ± standard deviation of 3 replicates. Different letters indicate significant difference at p ≤ 0.05.
GAE-gallic acid equivalent, CE-catechin equivalent, ND-non detected
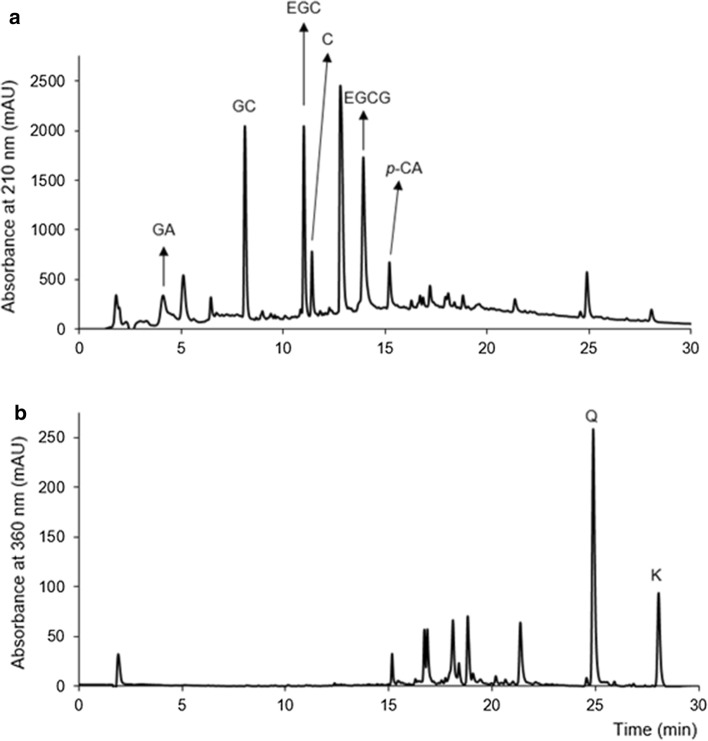
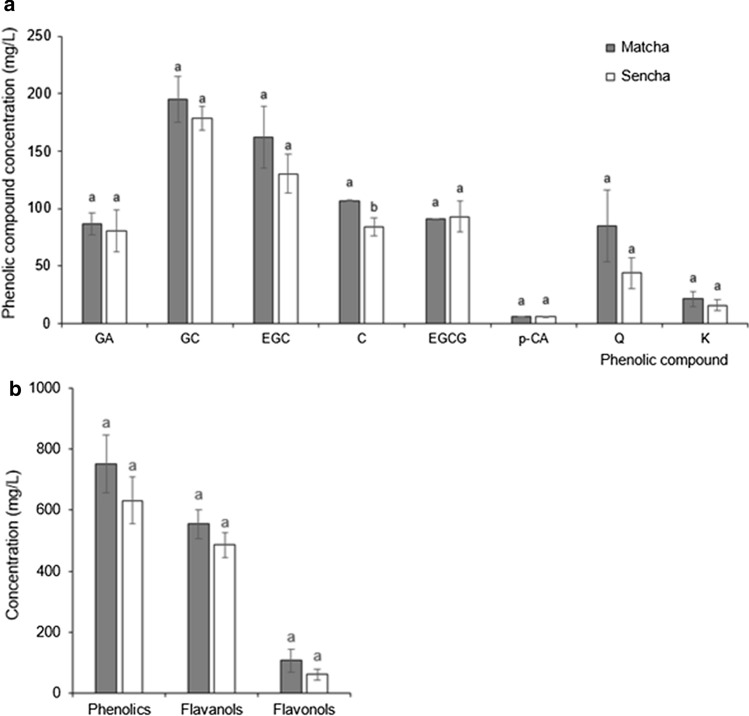
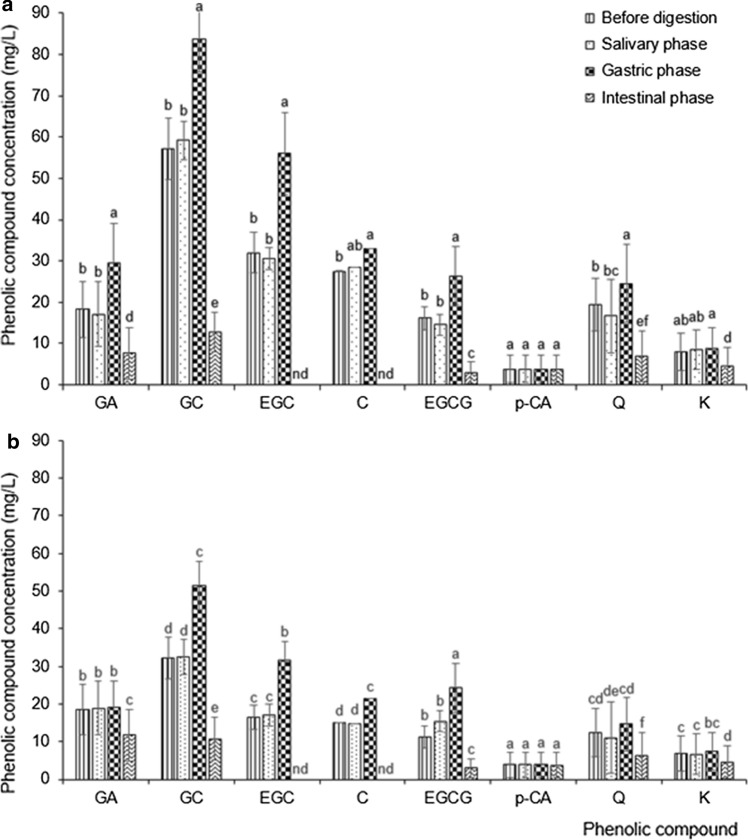
We developed a new short and reliable RP-HPLC method for tea polyphenol separation, identification and quantification (Fig. 2). Considering the available standards, we identified four flavanols (GC, EGC, cC and EGCG), two flavonols (Q and K), one hydroxybenzoic acid (GA) and one hydroxycinnamic acid (p-CA) in both forms of pre-digested tea (Fig. 2), which is in accordance with the results of other authors (Del Rio et al. 2004). The prevalence of flavanols was expected based on current knowledge, namely ( −)‐epigallocatechin, ( −)‐epicatechin, ( −)‐epigallocatechin gallate and ( −)‐epicatechin gallate are the most abundant phenolics in green tea (Del Rio et al. 2004). The percentile composition of flavanols in Matcha tea was 73.6% and in Sencha tea 76.9%, while flavonols were represented with 14.1 and 9.4%, respectively. Such a result perfectly matches that of Del Rio et al. (2004). Only catechin was present at a significantly higher concentration in pre-digested Matcha than in Sencha tea; all other identified compounds occurred at similar concentrations (Fig. 3a). Even though a tendency of higher concentrations in Matcha than Sencha tea was present, the concentrations of total identified phenolics (TiP), flavanols (TiFLA) or flavonols (TiFLO) did not differ significantly (p ≤ 0.05) between Matcha and Sencha tea (Fig. 3b). More precisely, we recorded 16.1% more TiP in undigested Matcha than in Sencha tea (Fig. 3b), which was in accordance with Zhang et al. (2018) who found up to 15% more polyphenols in small tea leaves compared to larger leaves. Curiously, we recorded a significantly higher amount of TiP, TiFLA and TiFLO in Matcha than in Sencha buffered tea upon the salivary and gastric digestion phase (Fig. 5). The difference between Matcha and Sencha tea at the level of phenolic compounds would not be significant when served in a cup, but does come to the fore under digestion conditions (Figs. 3b and 5, respectively). Accordingly, we concluded that the stability of the compounds depends on the interaction of their matrix and digestion conditions. Digestion conditions obviously emphasize differences in the stability between the phenolic compounds originating from tea forms which differ in the degree of leaf fragmentation (Matcha or Sencha). Moreover, the Matcha matrix enhanced the release of gallic acid and quercetin after the gastric phase of digestion, while the Sencha matrix did not (Fig. 4). Obviously, the fragmentation degree of leaves is crucial during the gastric phase release of these two compounds from green tea. After the intestinal phase, the amount of identified phenolic compounds was significantly reduced and no difference (p ≤ 0.05) could be observed in the amount of total and individual identified polyphenol compounds between Matcha and Sencha tea (the only exception was gallic acid, which was present at a higher amount in Sencha than in Matcha tea) (Fig. 5). The susceptibility of phenolics to alkaline conditions has already been reported (Green et al. 2007; Record and Lane 2001), therefore such a result was expected. We further observed that both Matcha and Sencha tea released their highest concentrations of flavanols after the gastric phase of digestion, i.e. after treatment with low pH and pepsin (Fig. 5b). More precisely, in Matcha tea the flavanol concentration was higher by 50%, while in Sencha tea by 72%. One exception was catechin in Matcha tea, whose concentration was not significantly higher, even though such a tendency was present (Fig. 4b). Our results are in discrepancy with those published by Okello et al. (2011) and Shim et al. (2012). As the model of digestion was quite similar to those of other authors, but the form and geographical origin of green tea were not, we presumed this could have been one of the main reasons behind this discrepancy. Moreover, as a low pH (2) was used in the gastric phase, and it is well known that conjugated phenolics release their free forms in acidic media, this supports our finding of higher phenolic concentrations upon the gastric phase. After the intestinal phase, the concentration of TiP, TiFLA or TiFLO was significantly lower in both forms of tea (Fig. 5), moreover the highest degree of degradation was observed for flavanols. For example, catechin and epigallocatechin could not be detected after the intestinal digestion phase at all (Fig. 4). Interestingly, their gallated forms were still present, suggesting galloylation as a protection of flavanols under intestinal conditions. We also recorded that the concentration of TiP was reduced by 79% in Matcha and 68% in Sencha tea, TiFLA were reduced by 88% in Matcha and 83% in Sencha tea, and TiFLO by 58% in Matcha and 51% in Sencha tea (Fig. 5). Okello et al. (2011) and Record and Lane (2001) also found a decrease of phenolic compounds concentration upon intestinal digestion. Green et al. (2007) revealed that more than 80% of the TFLA was degraded after human digestion. The only identified phenolic compound whose concentration did not change during digestion and was low from the very start was p-CA (Fig. 4). We hypothesize that one of the possible reasons could be the co-crystallization of p-CA with alkaloids caffeine and/or theophylline, as has already been chemically shown by Schultheiss et al. (2011).
Fig. 2.
HPLC profiles of a flavanols recorded at 210 nm, gallic acid at 280 nm and p-coumaric acid recorded at 310 nm, and b flavonols recorded at 360 nm in Matcha tea after gastric phase of digestion. GA gallic acid, GC gallocatechin, EGC epigallocatechin, C catechin, EGCG epigallocatechin-gallate, p-CA p-coumaric acid, Q quercetin, K kaempferol
Fig. 3.
Concentration of a individual phenolic compounds and b total identified compounds in predigested Matcha and Sencha tea. Data are means of three replicates ± SD. GA gallic acid, GC gallocatechin, EGC epigallocatechin, C catechin, EGCG epigallocatechin-gallate, p-CA p-coumaric acid, Q quercetin, K kaempferol. Different small letters indicate significant difference for a each individual component and b groups of identified components between Matcha and Sencha tea at p ≤ 0.05
Fig. 5.
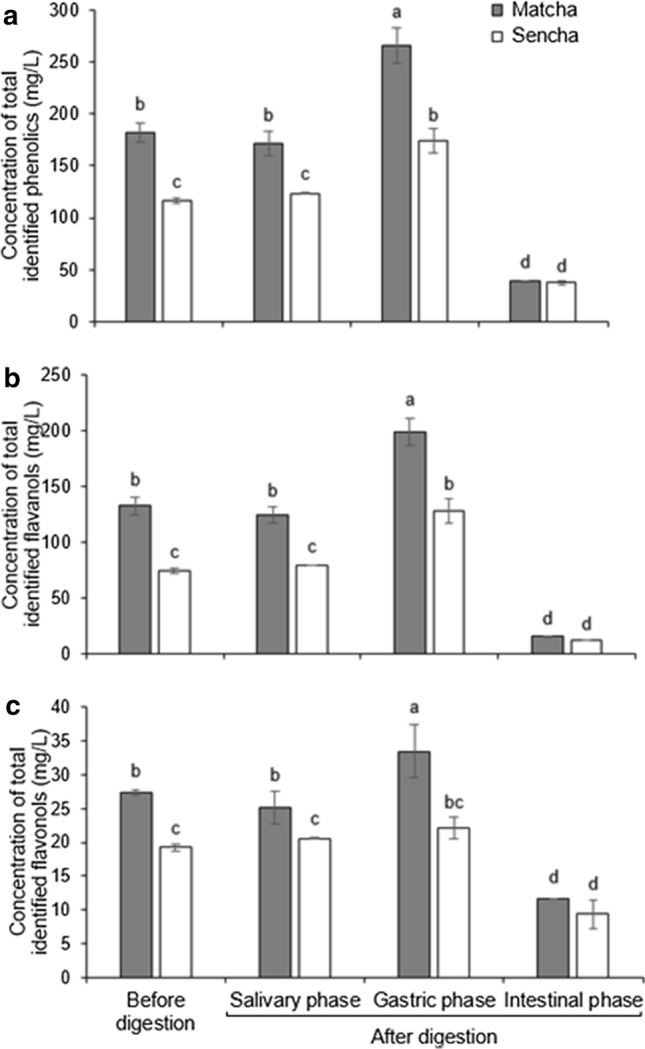
The concentration of a total identified phenolic compounds, b total identified flavanols and c total identified flavonols before and after in vitro digestion phases of Matcha and Sencha green tea. Data are means of three replicates ± SD. Different small letters indicate significant difference between Matcha and Sencha digestion phases at p ≤ 0.05
Fig. 4.
Effect of in vitro digestion on the concentration of individual phenolic compounds in a Matcha and b Sencha tea. Data are means of three replicates ± SD. GA gallic acid, GC gallocatechin, EGC epigallocatechin, C catechin, EGCG epigallocatechin-gallate, p-CA p-coumaric acid, Q quercetin, K kaempferol, nd not detected. Different small letters indicate significant difference between Matcha and Sencha digestion phases for each component separately at p ≤ 0.05
Antioxidant activity
All Matcha and Sencha predigested tea samples (10 g/L) have a strong antioxidant activity in relation to Trolox (Table 1). A significant increase in the inhibition percentage of ABTS radical was detected for Matcha tea. There was no significant difference between undigested Matcha and Sencha tea samples in the DPPH and FRAP methods. Komes et al. (2010) also measured antioxidant activity in the same concentration of powered Matcha and loose leaf Sencha tea by ABTS, DPPH and FRAP method. They reported a higher antioxidant activity of Matcha tea with all three methods. The differences in our results can be explained by the fact that we used bagged Sencha tea which had shredded leaf parts. Shredded leaf parts in tea bags could be better for short extraction of phytochemicals with antioxidant properties. Komes et al. (2010) also reported that short extraction (3 min, 80° C) of hand-rolled tea leaf is most likely insufficient to extract a higher antioxidant content.
DPPH, ABTS and FRAP methods were also used for antioxidant measurements in Matcha and Sencha green tea samples (1.5 g/L) before and after exposure to in vitro digestive conditions and the results are presented in Fig. 1 (d-f). Moderate antioxidant activity was measured in Matcha and Sencha samples by DPPH method (39.87 and 40.28%) and in Matcha samples by ABTS method (35.41%) after gastric digestion in comparison to the standard compound. The inhibition percentage of Trolox for the DPPH method was 82.4% and for ABTS 82.1%. With FRAP, all Matcha and Sencha samples before and after digestion (mouth and stomach) showed moderate (47.49–66.98%) antioxidant activity in relation to Trolox (97.6%). In Matcha samples, significantly higher antioxidant activity in almost all antioxidant methods before and after digestion was detected, with the exception of the gastric phase for DPPH and salivary phase for ABTS and FRAP methods.
After the initial and salivary phase, a significant increase in antioxidant activity in Matcha and Sencha tea samples was detected in the gastric phase by ABTS, FRAP and DPPH methods, with the exception of Sencha tea samples and the DPPH method. After the gastric phase, a significant decrease in antioxidant activity of Matcha and Sencha tea samples was detected in the intestinal phase, also with the exception of Sencha tea samples and the DPPH method.
Wootton-Beard et al. (2011) detected a slight increase in antioxidant activity (ABTS, FRAP, DPPH) following the gastric phase and a slight decrease following the duodenal phase in most of the tested juices. Record and Lane (2001) investigated antioxidant activity (FRAP) and TP content after exposure to gastric pH for 60 min and intestinal pH for 15, 30 and 60 min. They observed a 10% decrease in antioxidant activity (FRAP) and in TP content after gastric phase as well as a 27–28% decrease in antioxidant activity (FRAP) and TP content after the intestinal phase. Wu et al. (2015) observed a significant decrease in total phenol content (6.12%) and antioxidant activity (55.9%) in green tea samples after in vitro simulated gastro-intestinal digestion with DPPH, and no significant decrease (10.7%) with ABTS. Both of the groups used undigested tea as the control; if we compare our digestion results with those for undigested tea, we can also detect a decrease in antioxidant activity with all three of the used methods.
Antidiabetic activity
Controlling the increase in postprandial blood glucose is possible with dietary supplementation with tea rich in α-glucosidase and α-amylase inhibitors (Fu et al. 2017). So far, the inhibitions of these enzymes have been mainly tested in predigested teas (Kwon et al. 2008; Quesille-Villalobos et al. 2013), so there is a need to examine the inhibition of enzymes during digestion to obtain a better and more effective complementary therapy for postprandial hyperglycemia. Both predigested Matcha and Sencha tea samples (10 g/L) had high α-glucosidase inhibitory activity 99.67 ± 0.88 and 102.72 ± 8.83, respectively in relation to acarbose (Table 1). A significant increase in the inhibition percentage of α-glucosidase was detected by Matcha tea in the initial, gastric and intestinal phase (70.91 ± 2.99, 62.90 ± 1.44 and 113.46 ± 6.09) of digestion compared to Sencha tea (56.85 ± 9.67, 34.21 ± 3.44 and 66.64 ± 10.50) (Suppl. Tab. 1). After the intestinal phase, Matcha had strong and Sencha tea moderate α-glucosidase inhibitory activity. α-amylase inhibitory activity was not detected in predigested and digested Matcha and Sencha tea samples (Table 1 and Suppl. Tab. 1). Kwon et al. (2008) also detected high α-glucosidase inhibitory activity and low α-amylase inhibitory activity in green tea. According to Quesille-Villalobos et al. (2013), α-amylase inhibition was low in most of green teas and was not detected in Sencha green tea.
Pearson correlations
All polyphenols, antioxidant and antidiabetic activity methods correlated (Suppl. Tab. 2.) with one another and had very strong (0.80–1) or strong correlations (0.76–0.79). α-glucosidase inhibition method had the highest correlation (≥ 0.90) with catechin and kaempferol. ABTS had the highest correlation (≥ 0.90) with all total and individual polyphenols, α-glucosidase, FRAP and DPPH. Accordingly, antioxidant and antidiabetic activity depends on the concentration of total and individual polyphenols before and during in vitro digestion.
Conclusion
So far there have been no available data on the effect of digestion on the polyphenol composition and antioxidant and antidiabetic activity of powdered Matcha and bagged Sencha tea. Our findings have shown that the concentration of Matcha and Sencha tea polyphenols and antioxidant activity during in vitro digestion did not reflect the phenolic concentration and antioxidant activity of predigested tea. The difference between Matcha and Sencha tea after gastric digestion phase was emphasized, but neutralized upon intestinal digestion. The fragmentation degree of leaves and digestion conditions (acid or alkaline conditions, microbiota) are crucial for the stability and digestive availability of Matcha and Sencha polyphenols. According to the results of antidiabetic measurements after in vitro digestion, Matcha tea is potentially better than Sencha tea for effective complementary therapy of postprandial hyperglycemia with less side effects (e.g. abnormal bacterial fermentation of carbohydrates in the colon by pancreatic amylase).
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
N/A
Abbreviations
- C
Catechin
- EGC
Epigallocatechin
- EGCG
Epigallocatechin-gallate
- GA
Gallic acid
- GC
Gallocatechin
- K
Kaempferol
- p-CA
p-Coumaric acid
- Q
Quercetin
- TF
Total flavonoids
- TFLA
Total flavanols
- TP
Total phenols
Authors' contributions
GR conceived, supervised the work and wrote the MS; IŠ and VVB carried out analysis, wrote and edited the MS.
Funding
This work was supported by the University of Zagreb, Croatia [Grant No. 20283112].
Availability of data and material
Upon request.
Declarations
Conflicts of interest
The authors declare that they have no conflict of interest
Ethics approval
N/A.
Consent to participate
N/A.
Consent for publication
All authors have read and approved the MS and all co-authors are aware of its submission to JFST including the concerned authorities.
Code availability
N/A.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Gordana Rusak, Email: gordana.rusak@biol.pmf.hr.
Ivana Šola, Email: ivana.sola@biol.pmf.hr.
Valerija Vujčić Bok, Email: valerija.vujcic@biol.pmf.hr.
References
- Coe S, Fraser A, Ryan L. Polyphenol bioaccessibility and sugar reducing capacity of black, green, and white teas. Int J Food Sci. 2013;2013:1–6. doi: 10.1155/2013/238216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corrêa TAF, Rozenbaum AC, Rogero MM. Role of tea polyphenols in metabolic syndrome. IntechOpen. 2020 [Google Scholar]
- Del Rio D, Stewart AJ, Mullen W, Burns J, Lean MEJ, Brighenti F, Crozier A. HPLC-MS n analysis of phenolic compounds and purine alkaloids in green and black tea. J Agric Food Chem. 2004;52:2807–2815. doi: 10.1021/jf0354848. [DOI] [PubMed] [Google Scholar]
- Fu QY, Li QS, Lin XM, Qiao RY, Yang R, Li XM, Dong ZB, Xiang LP, Zheng XQ, Lu JL, Yuan CB, Ye JH, Liang YR. Antidiabetic effects of tea. Mol. 2017;22:849. doi: 10.3390/molecules22050849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman M, Levin CE, Lee S-U, Kozukue N. Stability of green tea catechins in commercial tea leaves during storage for 6 months. J Food Sci. 2009;74:47–51. doi: 10.1111/j.1750-3841.2008.01033.x. [DOI] [PubMed] [Google Scholar]
- Green RJ, Murphy AS, Schulz B, Watkins BA, Ferruzzi MG. Common tea formulations modulate in vitro digestive recovery of green tea catechins. Mol Nutr Food Res. 2007;51:1152–1162. doi: 10.1002/mnfr.200700086. [DOI] [PubMed] [Google Scholar]
- Khoddami A, Wilkes M, Roberts T. Techniques for analysis of plant phenolic compounds. Molecules. 2013;18:2328–2375. doi: 10.3390/molecules18022328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kokubo Y, Iso H, Saito I, Yamagishi K, Yatsuya H, Ishihara J, Inoue M, Tsugane S. The impact of green tea and coffee consumption on the reduced risk of stroke incidence in Japanese population. Stroke. 2013;44:1369–1374. doi: 10.1161/STROKEAHA.111.677500. [DOI] [PubMed] [Google Scholar]
- Komes D, Horžić D, Belščak A, Kovačević Ganić K, Vulić I. Green tea preparation and its influence on the content of bioactive compounds. Food Res Int. 2010;43:167–176. [Google Scholar]
- Kusznierewicz B, Bartoszek A, Wolska L, et al. Partial characterization of white cabbages (Brassica oleracea var. capitata f. alba) from different regions by glucosinolates, bioactive compounds, total antioxidant activities and proteins. LWT Food Sci Technol. 2008;41:1–9. [Google Scholar]
- Kwon Y-I, Apostolidis E, Shetty K. Inhibitory potential of wine and tea against α-amylase and α-glucosidase for management of hyperglycemia linked to type 2 diabetes. J Food Biochem. 2008;32:15–31. [Google Scholar]
- Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L. Polyphenols: food sources and bioavailability. Am J Clin Nutr. 2004;79:727–747. doi: 10.1093/ajcn/79.5.727. [DOI] [PubMed] [Google Scholar]
- Meng JM, Cao SY, Wei XL, Gan RY, Wang YF, Cai SX, Xu XY, Zhang PZ, Li HB. Effects and mechanisms of tea for the prevention and management of diabetes mellitus and diabetic complications: an updated review. Antioxid. 2019;8:170. doi: 10.3390/antiox8060170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nayyar AS, Das M, Deosarkar B, Deosarkar SB, Karan A, Sinh P, Paraye S. Green tea in medicine: a brief overview. J Int Dent Medical Res. 2017;5:191. [Google Scholar]
- Shim SM, Yoo SH, Ra CS, Kim YK, Chung JO, Lee SJ. Digestive stability and absorption of green tea polyphenols: Influence of acid and xylitol addition. Food Res Int. 2012;45:204–210. [Google Scholar]
- Okello EJ, McDougall GJ, Kumar S, Seal CJ. In vitro protective effects of colon-available extract of Camellia sinensis (tea) against hydrogen peroxide and beta-amyloid (Aβ(1–42)) induced cytotoxicity in differentiated PC12 cells. Phytomedicine. 2011;18:691–696. doi: 10.1016/j.phymed.2010.11.004. [DOI] [PubMed] [Google Scholar]
- Quesille-Villalobos AM, Torrico JS, Ranilla LG. Phenolic compounds, antioxidant capacity, and in vitro α-amylase inhibitory potential of tea infusio. CyTA-J Food. 2013;11:60–67. [Google Scholar]
- Record IR, Lane JM. Simulated intestinal digestion of green and black teas. Food Chem. 2001;73:481–486. [Google Scholar]
- Rusak G, Komes D, Likić S, Horžić D, Kovač M. Phenolic content and antioxidative capacity of green and white tea extracts depending on extraction conditions and the solvent used. Food Chem. 2008;110:852–858. doi: 10.1016/j.foodchem.2008.02.072. [DOI] [PubMed] [Google Scholar]
- Saito E, Inoue M, Sawada N, Shimazu T, Yamaji T, Iwasaki M, Sasazuki S, Noda M, Iso H, Tsugane S, JPHC Study Group Association of green tea consumption with mortality due to all causes and major causes of death in a Japanese population: the Japan Public Health Center-based Prospective Study (JPHC Study) Ann Epidemiol. 2015;25:512–518.e3. doi: 10.1016/j.annepidem.2015.03.007. [DOI] [PubMed] [Google Scholar]
- Salahuddin MAH, Ismail A, Kassim NK, Hamid M, Ali MSM. Phenolic profiling and evaluation of in vitro antioxidant, α-glucosidase and α-amylase inhibitory activities of Lepisanthes fruticosa (Roxb) Leenh fruit extracts. Food Chem. 2020;331:127240. doi: 10.1016/j.foodchem.2020.127240. [DOI] [PubMed] [Google Scholar]
- Schultheiss N, Roe M, Boerrigter SXM. Cocrystals of nutraceuticalp-coumaric acid with caffeine and theophylline: polymorphism and solid-state stability explored in detail using their crystal graphs. CrystEngComm. 2011;13:611–619. [Google Scholar]
- Šola I, Vujčić Bok V, Dujmović M, Rusak G. Developmentally-related changes in phenolic and L-ascorbic acid content and antioxidant capacity of Chinese cabbage sprouts. J Food Sci Technol. 2020;57:702–712. doi: 10.1007/s13197-019-04103-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Šola I, Vujčić Bok V, Pinterić M, Auer S, Ludwig-Müller J, Rusak G. Improving the phytochemical profile and bioactivity of Chinese cabbage sprouts by interspecific transfer of metabolites. Food Res Int. 2020;137:109726. doi: 10.1016/j.foodres.2020.109726. [DOI] [PubMed] [Google Scholar]
- Weiss DJ, Anderton CR. Determination of catechins in matcha green tea by micellar electrokinetic chromatography. J Chromatogr A. 2003;1011:173–180. doi: 10.1016/s0021-9673(03)01133-6. [DOI] [PubMed] [Google Scholar]
- Wootton-Beard PC, Moran A, Ryan L. Stability of the total antioxidant capacity and total polyphenol content of 23 commercially available vegetable juices before and after in vitro digestion measured by FRAP, DPPH, ABTS and Folin-Ciocalteu methods. Food Res Int. 2011;44:217–224. [Google Scholar]
- Wu Z, Teng J, Huang L, Xia N, Wei B. Stability, antioxidant activity and in vitro bile acid-binding of green, black and dark tea polyphenols during simulated in vitro gastrointestinal digestion. RSC Adv. 2015;5:92089–92095. [Google Scholar]
- Zhang C, Suen CL-C, Yang C, Quek SY. Antioxidant capacity and major polyphenol composition of teas as affected by geographical location, plantation elevation and leaf grade. Food Chem. 2018;244:109–119. doi: 10.1016/j.foodchem.2017.09.126. [DOI] [PubMed] [Google Scholar]
- Zhishen J, Mengcheng T, Jianming W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999;64:555–559. [Google Scholar]
- Zhou Q, Chiang H, Portocarrero C, Zhu Y, Hill S, Heppert K, Jayaratna H, Davies M, Janle E, Kissinger P. Investigating the stability of EGCg in aqueous media. Curr Sep. 2002;20:83–86. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Upon request.