Abstract
Objective
To explore the association between three-dimensional (3D) cardiac magnetic resonance (CMR) feature tracking (FT) right ventricular peak global longitudinal strain (RVpGLS) and major adverse cardiovascular events (MACEs) in patients with stage C or D heart failure (HF) with non-ischaemic dilated cardiomyopathy (NIDCM) but without atrial fibrillation (AF).
Methods
Patients with dilated cardiomyopathy were enrolled in this prospective cohort study. Comprehensive clinical and biochemical analysis and CMR imaging were performed. All patients were followed up for MACEs.
Results
A total of 192 patients (age 53±14 years) were eligible for this study. A combination of cardiovascular death and cardiac transplantation occurred in 18 subjects during the median follow-up of 567 (311, 920) days. Brain natriuretic peptide, creatinine, left ventricular (LV) end-diastolic volume, LV end-systolic volume, right ventricular (RV) end-diastolic volume and RVpGLS from CMR were associated with the outcomes. The multivariate Cox regression model adjusting for traditional risk factors and CMR variables detected a significant association between RVpGLS and MACEs in patients with stage C or D HF with NIDCM without AF. Kaplan-Meier analysis based on RVpGLS cut-off value revealed that patients with RVpGLS <−8.5% showed more favourable clinical outcomes than those with RVpGLS ≥−8.5% (p=0.0037). Subanalysis found that this association remained unchanged.
Conclusions
RVpGLS-derived from 3D CMR FT is associated with a significant prognostic impact in patients with NIDCM with stage C or D HF and without AF.
Keywords: cardiac imaging and diagnostics, advanced cardiac imaging, cardiac magnetic resonance (CMR) imaging, heart failure, myocardial disease
Introduction
Non-ischaemic dilated cardiomyopathy (NIDCM) is defined as a disease of myocardium characterised by systolic dysfunction, either of the left ventricle (LV) alone or biventricular and LV dilatation that cannot be explained exclusively by abnormal loading or coronary artery disease.1 In patients with NIDCM, the functional assessment of the LV has been under intensive focus. However, in patients with advanced HF with NIDCM, the degree of impaired LV systolic function loses its ability of survival prediction.2 3 On the other hand, such patients might also have the right ventricle (RV) dysfunction because NIDCM affects the myocardium of both ventricles.4 The RV dysfunction is a robust predictor in patients with NIDCM.5 6 Therefore, the comprehensive evaluation of RV function is crucial in patients with HF and NIDCM.7
Unlike LV measurement, the estimation of RV function is challenging due to its complex geometry, different myocytes arrangement and motion.8 CMR has a high diagnostic accuracy, which demonstrates cardiac morphology and function. CMR is a gold standard for assessing the complex geometry of the RV.9 The analysis of strain is one of the several indexes for the assessment of RV systolic function according to the current international guidelines.10 Some recent studies have shown that CMR RV feature-tracking (FT) myocardial strain analysis is an independent prognostic factor in many cardiovascular diseases.11 Also, the three-dimensional (3D) cardiac magnetic resonance (CMR) FT strain analysis has been suggested as a putative breakthrough for an accurate evaluation of the strain.12
Herein, we aimed to assess the correlation between right ventricular peak global longitudinal strain (RVpGLS) from 3D CMR FT and the prognosis of patients with NIDCM with advanced HF and without atrial fibrillation (AF). We hypothesised that the 3D FT RVpGLS is associated with a high risk of adverse cardiac outcomes in patients with stage C or D HF and NIDCM but without AF.
Methods
Study population and study design
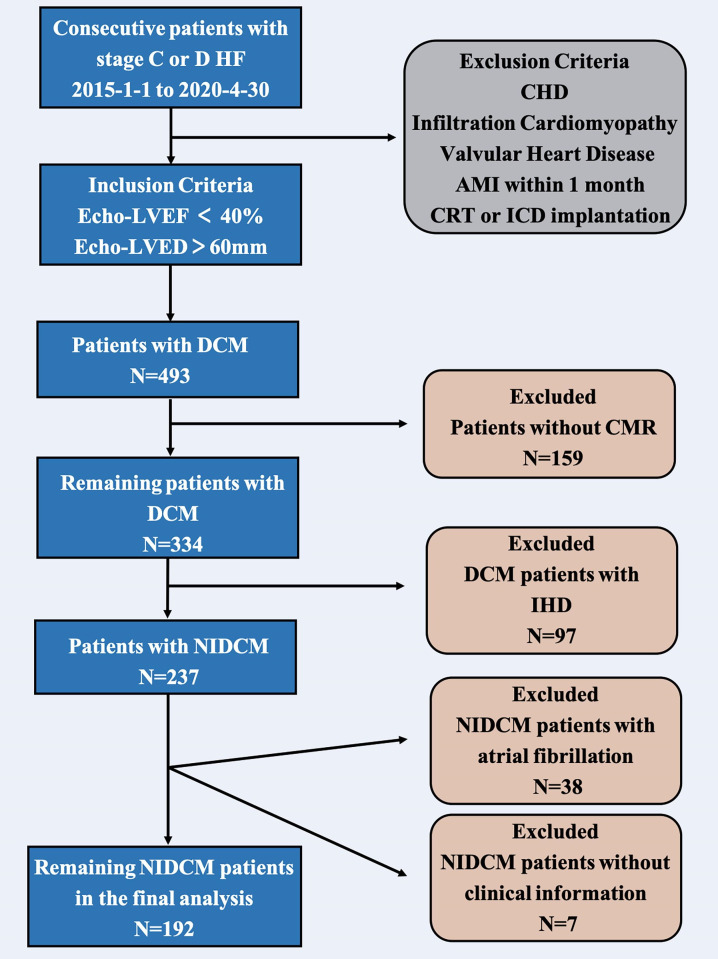
Between 1 January 2015 and 30 April 2020, consecutive patients with stage C or D HF were identified and enrolled into this observational cohort study in the Department of Heart Failure Program at Anzhen Hospital (Capital Medical University of Beijing, China).13 The current study enrolled 493 patients with dilated cardiomyopathy (DCM) that was diagnosed according to the guidelines of the WHO/International Society and the Federation of Cardiology. According to the inclusion criteria of the study, patients with stage C or D HF,14 LV ejection fraction (LVEF) <40% and LV end-diastolic diameter >60 mm on echocardiography. Patients with congenital heart disease, infiltrative cardiomyopathy (ie, sarcoidosis and amyloidosis), significant valvular heart disease, acute myocardial infarction (MI) within 1 month or a history of cardiac resynchronisation therapy and implantable cardioverter-defibrillator implantation were excluded. However, 159 patients who had not undergone a CMR, were excluded from this study. All patients with DCM underwent coronary angiography, and 97 patients with ischaemic heart disease (IHD) showed >50% narrowing of the coronary artery lumen on coronary angiography or a history of MI or stent implantation. Next, 38 patients with NIDCM and AF were excluded from this study because of unclear CMR images. Seven patients were excluded due to the lack of complete clinical data. The final analysis included 192 patients with stage C or D HF with NIDCM and without AF (figure 1).
Figure 1.
Flow chart of the observational study. AMI, acute myocardial infarction; CHD, coronary heart disease; CMR, cardiac magnetic resonance; CRT, cardiac resynchronisation therapy; DCM, dilated cardiomyopathy; HF, heart failure; ICD, implantable cardioverter-defibrillator; IHD, ischaemic heart disease; NIDCM, non-ischaemic dilated cardiomyopathy.
CMR data acquisition
CMR studies were conducted on a 3.0 T scanner (Magnetom Verio; Siemens AG Healthcare, Erlangen, Germany or MR750W, General Electric Healthcare, Waukesha, Wisconsin, USA) with retrospective EKG gating and 32-channel phased-array coil. The standardised imaging protocol consisted of steady-state free precession breath-hold cine images and late-enhanced images (LGE) using gadolinium.15 16 Whole LV and RV from the annulus of the AV valves to the apex were encompassed in contiguous short-axis slices (8 mm slice thickness without intersection gap) with 25 phases per cardiac cycle. Long axis planes (2-chamber, 4-chamber and 3-chamber views) with 5 mm slice thickness had no spacing intersection gap. LGE images were acquired using a prospectively EKG-gated gradient echo sequence with an inversion prepulse at 10 min after intravenous infusion with gadolinium chelate contrast agent. The inversion time of LGE images was optimised to null normal myocardium, and the imaging parameters were as follows: repetition time/echo time, 4.1/1.6 ms; flip angle, 20°; image matrix, 256×130.
CMR analysis
The LV and RV function and masses were analysed on a commercially available workstation CVI42 software (Version 5.6.3 Circle Cardiovascular Imaging, Calgary, Canada). Short-axis cine and 4-chamber or 2-chamber or 3-chamber images were used for semi-automated analysis. Endocardial and epicardial borders were drawn automatically and amended by a radiologist on the short-axis cine images with papillary muscles excluded from volumes, and the RV insertion sites were marked to indicate the outer anteroseptal and infraseptal borders. The LV and RV range were marked on 4-chamber images. LV and RV volumes, masses, and EF were quantified from cine images automatically. The epicardial and endocardial contours were placed manually on LGE images that were present in any areas with a signal intensity of 5SD above that of normal myocardium.17
CMR analysis of standard cardiac parameters
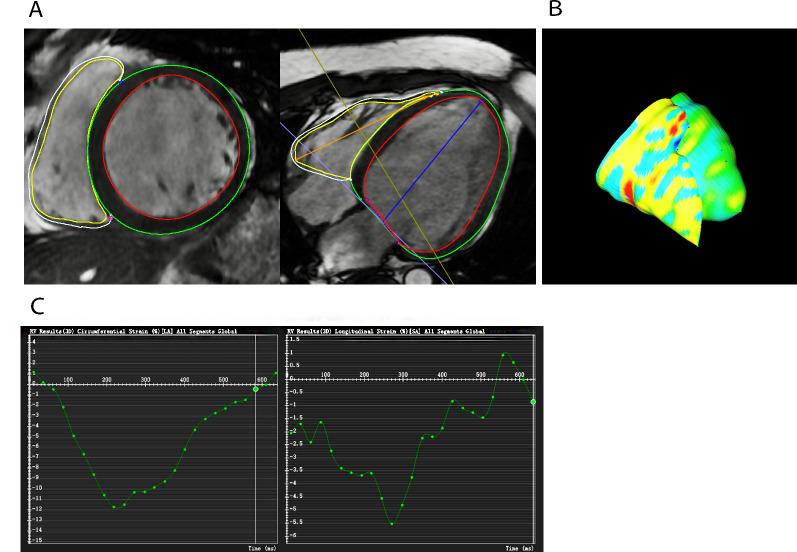
Three-dimensional FT global circumferential strain (GCS) and global longitudinal strain (GLS) strain of LV and RV were obtained using CVI42 (figure 2). The end-diastolic phase was chosen as a reference. The endocardial and epicardial short-axis contours in all slices that encompass the LV and RV were defined, and blood volume was excluded. The endocardial and epicardial biventricular borders were drawn in the same end-diastolic phase in the 4-chamber and 2-chamber tissue, while the anterior and inferior insertion points were drawn manually.
Figure 2.
Representative example of right ventricular peak global longitudinal strain (RVpGLS)%. (A) Endocardial and epicardial borders are drawn on the 4-chamber long-axis and short-axis images in the same end-diastolic phase for RVpGLS and right ventricular peak global circumferential strain (RVpGCS) measurement. (B) Three-dimensional (3D) model of the myocardium in diastole. (C) Software automatically constructs the curve.
Biochemical evaluation
Blood samples of patients were withdrawn to evaluate the liver and kidney function, blood routine and the level of brain natriuretic peptide (BNP) in Anzhen Hospital Central Laboratory.
Follow-up and study end points
The attending physician of the patients had to fill out clinical information and the results of biochemical and imaging were obtained via the Empower DataWeb data collection system (X&Y Solutions, Boston, Massachusetts, USA). The patients were followed up at a 3-month interval with the outpatient visit or by contact with the patient’s family members. The status of the events was updated on the Empower DataWeb each time.
The primary outcome was the composite of major adverse cardiac events (MACEs), including cardiovascular death and cardiac transplantation, based on the information obtained from the Empower DataWeb. Cardiovascular death was defined as HF death, fatal MI, sudden death, stroke, cardiovascular procedure or cardiovascular haemorrhage. The patients were followed up until the beginning of May 2020.
Statistical analysis
Continuous variables are presented as mean±SDs (normal distribution) or medians and IQRs (skewed distribution), while categorical variables are presented as number (%). Box-Cox transformation was used to handle the skewed distribution variable. We also used multiple imputations (MIs) based on five replications and a chained equation approach method in the R MI procedure, to account for missing data on the most important covariate. A p value <0.05 was deemed statistically significant.
First, generalised additive models (GAMs) were used to identify the correlations between RVpGLS and outcomes because RVpGLS was a continuous variable. Second, univariate Cox regression analysis was used to evaluate the association between each variable and MACEs in patients with NIDCM within 5 years of follow-up. Third, multivariate Cox regression models were used to examine whether 3D CMR FT RVpGLS had an independent effect on MACEs in patients with stage C or D HF with NIDCM within 5-year follow-up. An initial multivariable Cox regression model included demographic factors, clinical factors, laboratory tests and CMR variables. The covariates, when added to this model, altered the effect estimate of >10%,18 and covariates of known clinical importance were adjusted. Owing to 28.1% of patients missing BNP, we applied MIs for the models with BNP, five sets of imputed data were used and results were pooled to account for the variance among the data. In addition, we also used the Cox model with the inverse probability weighting of a marginal structural model to calculate the weight used for the whole set of covariates.19 Finally, the subgroup analysis was conducted to find whether potential risk factors had an impact on the results. The interactions among the subgroups were examined using multivariate-adjusted Cox regression model.
Time-dependent receiver operating characteristic (ROC) curves were used to determine the optimal cut-off of 3D CMR FT RVpGLS for the 5-year primary composite end point based on the Youden Index. The best cut-off values of RVpGLS for the analysed events were based on the highest sum of sensitivity and specificity. The event-free curves were based on Kaplan-Meier analysis stratified by RVpGLS optimal cutoffs and compared using the log-rank test. All analyses were performed using Empower (R) (http://www.empowerstats.com, X&Y Solutions) and R (http://www.R-project.org).
Patient and public involvement
It was not possible to involve patients or the public in the design, conduct, reporting or dissemination plans of our research.
Results
Clinical and CMR characteristics of stage C or D HF patients with NIDCM without AF
The final analysis included 192 patients with NIDCM with stage C or D HF without AF (figure 1). The average age was 53 years (53±14) and 72.92% males. The mean left ventricular end-diastolic volume (LVEDV), left ventricular end-systolic volume (LVESV) and LVEF was 293±110 mL, 233±104 mL and 22.37%±9.75%, respectively. The mean RVEF on CMR was 30.35%±14.11%; 78 (41.27%) patients were diagnosed with hypertension, 28 (14.81%) were diagnosed with diabetes mellitus and left bundle branch block (LBBB) was observed on the ECG in 17.65% of patients with NIDCM. The mean of QRS duration was 124.18±31.62 ms. Furthermore, 91.57% of the cohort was prescribed either ACE inhibitor or angiotensin II receptor blocker or angiotensin receptor neprilysin inhibitor, 95.83% was prescribed a beta-blocker and 89.58% was prescribed an aldosterone antagonist (table 1).
Table 1.
Baseline characteristics of patients with stage C or D heart failure with non-ischaemic dilated cardiomyopathy but without AF
| Mean+SD/Median and IQRs | All patients (n=192) |
| Age, mean (SD) (years) | 53±14 |
| Male (%) | 140 (72.92%) |
| BMI, mean (SD) (kg/m2) | 26.09±5.18 |
| Hypertension (%) | 78 (41.27%) |
| Diabetes mellitus (%) | 28 (14.81%) |
| CKD (%) | 2 (1.06%) |
| LBBB on EKG (%) | 33 (17.65%) |
| RBBB on EKG (%) | 9 (4.81%) |
| Intraventricular block on EKG (%) | 13 (6.91%) |
| QRS duration mean (SD) (ms) | 124.18±31.62 |
| Alanine aminotransferase (ALT) (U/L) | 33.39±33.63 |
| Aspartate aminotransferase (AST) (U/L) | 28.90±24.46 |
| Creatinine mean (SD) (mmol/L) | 82.53±25.22 |
| Na+ (SD) (mmol/L) | 139.83±2.59 |
| Cl− (SD) (mmol/L) | 102.76±4.60 |
| hs-CRP mean (SD) (mmol/L) | 3.53±5.42 |
| Homocysteine (μmol/L) | 23.43±32.48 |
| White blood cell (G/L) | 6.83±1.83 |
| Haemoglobin (G/L) | 147.76±19.72 |
| Platelets (G/L) | 210.08±64.24 |
| BNP (pg/mL) | 245.50 (129.75–514.25) |
| CMR-LVEDV (mL) | 293±110 |
| CMR-LVESV (mL) | 233±104 |
| CMR-LVEF, (%) | 22.37±9.75 |
| CMR-LVMASS (g) | 150.54±49.02 |
| CMR-RVEDV (mL) | 135±44 |
| CMR-RVESV (mL) | 96±41 |
| CMR-RVEF (%) | 30.35±14.11 |
| CMR-LVpGLS (%) | −6.12±2.78 |
| CMR-LVpGCS (%) | −7.19±2.89 |
| CMR-RVpGCS (%) | −7.71±3.76 |
| CMR-RVpGLS (%) | −10.49±5.16 |
| CMR-LVLGE (%) | 64 (33.51%) |
| Medication | |
| ACEI/ARB/ARNI (%) | 176 (91.57%) |
| Beta-blocker (%) | 184 (95.83%) |
| Spironolactone (%) | 172 (89.58%) |
ACEI, ACE inhibitor; AF, atrial fibrillation; ARB, angiotensin II receptor blocker; ARNI, angiotensin receptor neprilysin inhibitor; BMI, body mass index; BNP, brain natriuretic peptide; CKD, chronic kidney disease; CMR, cardiac magnetic resonance; hs-CRP, high-sensitivity C reactive protein; LBBB, left bundle branch block; LVEDV, left ventricular end-diastolic volume; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; LVLGE, left ventricular late gadolinium enhancement; LVMASS, left ventricular mass; LVpGCS, left ventricular peak global circumferential strain; LVpGLS, left ventricular peak global longitudinal strain; RBBB, right bundle branch block; RVEDV, right ventricular end-diastolic volume; RVEF, right ventricular ejection fraction; RVESV, right ventricular end-systolic volume; RVpGCS, right ventricular peak global circumferential strain; RVpGLS, right ventricular peak global longitudinal strain.
CMR-RVpGLS levels and 5-year MACEs in patients with stage C or D HF with NIDCM without AF
A total of 18 patients developed 5-year (the median follow-up of 567 days) MACEs, consisting of 16 patient deaths and 2 heart transplantation patients. In the present study, we established a linear correlation between RVpGLS and MACEs (online supplemental figure 1).
heartjnl-2020-317949supp001.pdf (57.6KB, pdf)
The results of univariate analyses of 5-year MACEs are summarised in table 2. Univariate analyses showed that creatinine, BNP, Na+, Cl−, CMR-LVEDV, CMR-LVESV, CMR -RVEDV and CMR-RVpGLS were associated with a significant increase in the incidence of 5-year MACEs. We performed a multivariate Cox regression analysis to explore CMR-RVpGLS as a prognostic marker. In the multivariable analysis shown in table 3, CMR-RVpGLS level was the independent risk factor for 5-year MACEs in model I (HR adjusted (HRadj) 1.16, 95% CI: 1.02 to 1.31; p=0.02) after adjusting for age, sex and body mass index (BMI). This was also true in model II (HRadj 1.19, 95% CI: 1.02 to 1.38; p=0.02) after adjusting for sex, age, BMI and LBBB on EKG, QRS duration, Na+, Cl−, BNP, haemoglobin (Hb) and creatinine; model III (HRadj 1.18, 95% CI: 1.01 to 1.37; p=0.04) after adjusting for sex, age, BMI, LGE, LVEDV, RVEDV, LVpGLS, LVMASS, LVpGCS and RVpGCS and model IV (HRadj 1.19, 95% CI: 0.98 to 1.43; p=0.06), after adjusting for sex, age, BMI, LBBB on EKG, QRS duration, Na+, Cl−, BNP, Hb, creatinine, LGE, LVEDV, RVEDV, LVpGLS, LVMASS, LVpGCS and RVpGCS. Model V (HRadj 1.18, 95% CI: 1.05 to 1.33; p=0.01) was obtained from the Cox model with the inverse probability weighting of a marginal structural model to calculate the weight used for the whole set of covariates.
Table 2.
Univariate analysis for major adverse events in patients with stage C or D heart failure with non-ischaemic dilated cardiomyopathy during a 5-year follow-up
| HR | 95% CI | P value | |
| Age (years) | 1.03 | 1.00 to 1.07 | 0.0787 |
| Male | 2.77 | 0.64 to 12.04 | 0.175 |
| BMI (kg/m2) | 0.96 | 0.94 to 1.03 | 0.160 |
| Hypertension (%) | 0.81 | 0.31 to 2.10 | 0.666 |
| Diabetes mellitus (%) | 1.09 | 0.31 to 3.76 | 0.896 |
| QRS duration (ms) | 1.01 | 0.99 to 1.02 | 0.475 |
| LBBB on EKG (%) | 1.20 | 0.39 to 3.65 | 0.752 |
| RBBB on EKG (%) | 0.96 | 0.13 to 7.26 | 0.971 |
| Intraventricular block (%) | _* | _* | _* |
| ALT (U/L) | 0.98 | 0.96 to 1.01 | 0.257 |
| AST (U/L) | 0.98 | 0.94 to 1.02 | 0.344 |
| Creatinine (μmol/L) | 1.01 | 1.00 to 1.02 | 0.019 |
| Na+ (SD) (mmol/L) | 0.86 | 0.75 to 0.98 | 0.0245 |
| Cl− (SD) (mmol/L) | 0.85 | 0.76 to 0.95 | 0.005 |
| hs-CRP (mmol/L) | 1.05 | 0.98 to 1.11 | 0.150 |
| Hcy (μmol/L) | 1.00 | 0.98 to 1.01 | 0.764 |
| WBC (G/L) | 0.85 | 0.63 to 1.14 | 0.279 |
| Hb (G/L) | 1.00 | 0.98 to 1.02 | 0.724 |
| PLT (G/L) | 1.00 | 0.99 to 1.01 | 0.951 |
| BNP (Box-Cox transform) (pg/mL) | 1.57 | 1.20 to 2.04 | 0.001 |
| CMR-LVEDV (mL) | 1.00 | 1.00 to 1.01 | 0.002 |
| CMR-LVESV (mL) | 1.01 | 1.00 to 1.01 | 0.001 |
| CMR-LVEF % | 0.96 | 0.91 to 1.02 | 0.169 |
| CMR-LVMASS (g) | 1.01 | 1.00 to 1.01 | 0.161 |
| CMR-RVEDV (mL) | 1.01 | 1.00 to 1.02 | 0.045 |
| CMR-RVESV (mL) | 1.01 | 1.00 to 1.02 | 0.149 |
| CMR-RVEF % | 0.99 | 0.96 to 1.02 | 0.686 |
| CMR-LVpGLS (%) | 1.10 | 0.91 to 1.32 | 0.335 |
| CMR-LVpGCS (%) | 1.09 | 0.91 to 1.30 | 0.339 |
| CMR-RVpGCS (%) | 1.05 | 0.93 to 1.19 | 0.456 |
| CMR-RVpGLS (%) | 1.14 | 1.01 to 1.28 | 0.035 |
| CMR-LVLGE (%) | 2.57 | 0.99 to 6.68 | 0.052 |
| Medication | |||
| ACEI/ARB/ARNI (%) | 0.36 | 0.10 to 1.24 | 0.1047 |
| Beta-blocker (%) | 3.33 | 0.76 to 14.53 | 0.1092 |
| Spironolactone (%) | 0.52 | 0.07 to 3.92 | 0.5258 |
BNP has done the Box-Cox transformation whose formula is (BNPˆ0.1418–1)/0.1418.
*The model failed because of the small sample size.
ACEI, ACE inhibitor; ALT, alanine transaminase; ARB, angiotensin II receptor blocker; ARNI, angiotensin receptor neprilysin inhibitor; AST, aspartate transaminase; BMI, body mass index; BNP, brain natriuretic peptide; CMR, cardiac magnetic resonance; Hb, haemoglobin; Hcy, homocysteine; hs-CRP, high-sensitivity C reactive protein; LBBB, left bundle branch block; LVEDV, left ventricular end-diastolic volume; LVEF, left ventricular ejection fraction; LVESV, left ventricular end-systolic volume; LVLGE, left ventricular late gadolinium enhancement; LVMASS, left ventricular mass; LVpGCS, left ventricular peak global circumferential strain; LVpGLS, left ventricular peak global longitudinal strain; PLT, platelets; RBBB, right bundle branch block; RVEDV, right ventricular end-diastolic volume; RVESV, right ventricular end-systolic volume; RVpGCS, right ventricular peak global circumferential strain; RVpGLS, right ventricular peak global longitudinal strain; WBC, white blood cell.
Table 3.
Multivariate Cox regression analysis with CMR-RVpGLS for major adverse cardiac events in patients with stage C or D heart failure with non-ischaemic dilated cardiomyopathy during a 5-year follow-up
| CMR-RVpGLS (%) | HR | 95% CI | P value |
| Model I | 1.16 | 1.02 to 1.31 | 0.02 |
| Model II* | 1.19 | 1.02 to 1.38 | 0.02 |
| Model III | 1.18 | 1.01 to 1.37 | 0.04 |
| Model IV* | 1.19 | 0.98 to 1.43 | 0.06 |
| Model V† with inverse probability weighting | 1.17 | 1.04 to 1.32 | 0.01 |
Model I adjust for: sex, age, BMI.
Model II adjust for: sex, age, BMI, LBBB on EKG, QRS duration, Na+, Cl−, BNP (Box-Cox transformation), Hb, creatinine.
Model III adjust for: sex, age, BMI, LGE, LVEDV, RVEDV, LVpGLS, LVMASS, LVpGCS, RVpGCS.
Model IV adjust for: sex, age, BMI, LBBB on EKG, QRS duration, Na+, Cl−, BNP (Box-Cox transformation), Hb, creatinine, LGE, LVEDV, RVEDV, LVpGLS, LVMASS, LVpGCS, RVpGCS.
*Data on the BNP, age, BMI, creatinine, Na+, Cl−, LBBB on EKG, QRS duration and Hb were missing for 54 (28.1%), 1 (0.5%), 5 (2.6%), 16 (8.3%), 21 (10.9%), 22 (11.4%), 5 (2.6%), 11 (5.7%) and 27 (14.0%) patients, respectively. Multiple imputation was used to account for missing data in the model II and model IV.
†Model V is inverse probability weighting of marginal structure model that the weight account for the covariates same as model IV.
BMI, body mass index; BNP, brain natriuretic peptide; CMR, cardiac magnetic resonance; Hb, haemoglobin; LBBB, left bundle branch block; LGE, late gadolinium enhancement; LVEDV, left ventricular end-diastolic volume; LVMASS, left ventricular mass; LVpGCS, left ventricular peak global circumferential strain; LVpGLS, left ventricular peak global longitudinal strain; RVEDV, right ventricular end-diastolic volume; RVpGCS, right ventricular peak global circumferential strain; RVpGLS, right ventricular peak global longitudinal strain.
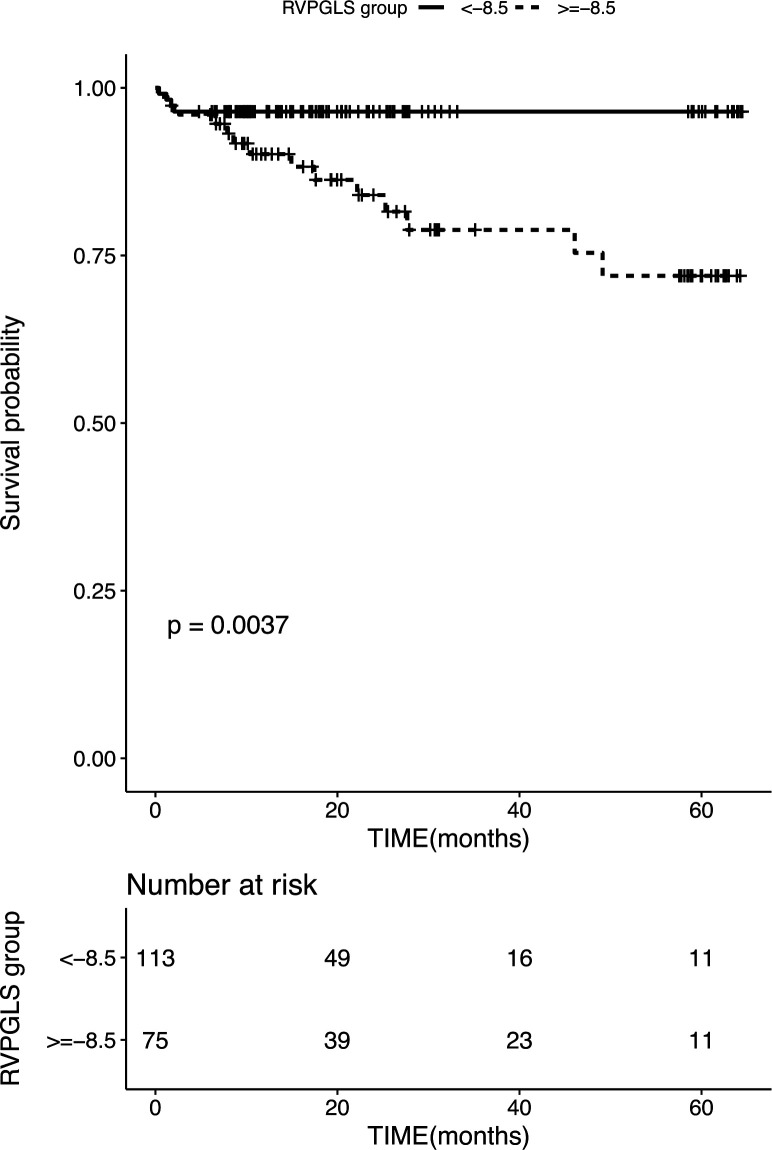
Time-dependent ROC curves were used to determine the cut-off of CMR-RVpGLS for the 5-year primary composite with −8.5% end point (online supplemental figure 2). The dichotomised RVpGLS value by −8.5% was used in Kaplan-Meier survival analysis (figure 3), which showed a significant difference among patients stratified by the RVpGLS value by −8.5%. Specifically, RVpGLS values ≥−8.5% exhibited 5-year MACEs as compared with those with <−8.5% (p=0.0037).
Figure 3.
Kaplan-Meier event-free survival curve. Kaplan-Meier analysis of freedom from major adverse cardiac events based on CMR-RVpGLS (log rank, p=0.0037). Numbers that do not add up to 192 are attributed to the missing data for RVpGLS. CMR, cardiac magnetic resonance; RVpGLS, right ventricular peak global longitudinal strain.
heartjnl-2020-317949supp002.pdf (65.5KB, pdf)
Subgroup analysis between CMR-RVpGLS levels and 5-year MACEs in patients with stage C or D HF with NIDCM without AF
To evaluate the potential influence of other factors, a subanalysis was conducted stratifying patients by age, sex, BMI, hypertension, diabetes mellitus, LBBB on EKG and CMR-LVLGE (table 4). Notably, all subgroups demonstrated a similar overall correlation between CMR-RVpGLS levels and 5-year MACEs in patients with stage C or D heart failure without AF.
Table 4.
Effects of CMR-RVpGLS on 5-year major adverse cardiac events in patients with stage C or D heart failure with non-ischaemic dilated cardiomyopathy in each subgroup by multivariable Cox model
| CMR-RVpGLS | HR (95% CI) | P value | P for interaction |
| Age (years) | 0.522 | ||
| ≤56 | 1.07 (0.92 to 1.25) | 0.353 | |
| >56 | 1.25 (1.00 to 1.57) | 0.047 | |
| Sex | 0.359 | ||
| Female | 0.97 (0.78 to 1.20) | 0.763 | |
| Male | 1.19 (1.03 to 1.38) | 0.017 | |
| BMI | 0.441 | ||
| ≤26 | 1.12 (0.98 to 1.29) | 0.089 | |
| >26 | 1.22 (0.93 to 1.59) | 0.153 | |
| Hypertension | 0.144 | ||
| No | 1.08 (0.94 to 1.25) | 0.253 | |
| Yes | 1.31 (0.99 to 1.73) | 0.058 | |
| Diabetes mellitus | 0.240 | ||
| No | 1.21 (1.03 to 1.42) | 0.018 | |
| Yes | 1.02 (0.86 to 1.22) | 0.784 | |
| LBBB on EKG | 0.279 | ||
| No | 1.11 (0.97 to 1.27) | 0.118 | |
| Yes | 1.27 (0.92 to 1.75) | 0.142 |
*Adjusted for age, sex, BMI, hypertension, diabetes mellitus, LBBB on EKG and CMR LVLGE except the subgroup variable.
BMI, body mass index; CMR, cardiac magnetic resonance; LBBB, left bundle branch block; LVLGE, left ventricular late gadolinium enhancement; RVpGLS, right ventricular peak global longitudinal strain.
Discussion
The results of the present study are summarised as follows: (1) all patients with stage C or D HF with NIDCM without AF had impaired RV systolic function; (2) RVpGLS from 3D CMR FT was also independently related to MACEs, even after adjusting for clinical, laboratory and CMR variables, in patients with NIDCM and biventricular dysfunction without AF during the 5-year follow-up; (3) patients with RVpGLS from 3D CMR FT values >−8.5% were at maximal risk for MACEs.
The LV dysfunction affected the RV function in patients with stage C or D HF with NIDCM primarily through elevated LV end-diastolic pressure, which increased the RV afterload and decreased the ventricular septal. The RVEF <45% is defined as RV dysfunction20 and is the most common indicator of RV function.20 The current study suggested that 100% of patients with HF have RV systolic dysfunction. But a previous study reported that RV systolic dysfunction is about 65% of DCM, which is a biventricular disease.4 The reason why our results are different might be that the previous study population suffered from NIDCM and ischaemic cardiomyopathy,4 while the population in this study was from NIDCM. As was testified by La Vecchia et al, the RVEF generally tends to be lower in patients with NIDCM compared with patients with ischaemic cardiomyopathy, suggesting that the underlying pathophysiological process might be more localised in ischaemic cardiomyopathy.4 Even in some cases, the RVEF remains normal in patients with ischaemic cardiomyopathy despite severely abnormal LVEF.6
Many studies have recently shown that myocardial strain analysis, especially GLS,21–24 might serve as an independent prognostic factor in patients with HF.5 11 25 26 Park et al 23 conducted a prospective observational study of >4000 patients enrolled over 8 years in three tertiary centres. In the heterogeneous group of patients with HF, LVpGLS-speckle tracking from echocardiography showed strong predictive value among known predictors of all-cause death. Conversely, Hamada-Harimura et al 24 evaluated both LV and RV strain with speckle tracking echocardiography in patients with acute decompensated HF in a multicentre study. During the median follow-up of 427 days, the RV longitudinal strain was identified as an independent predictor of cardiovascular death and readmission for HF in acute decompensated HF. Moreover, LV strain parameters failed to provide the prognostic value of adverse outcomes. Another study recruited 143 stable patients with HF due to NIDCM (age: 64.6±15.3 years, 35% female, LVEF 27.4%±7.6%) during a median follow-up of 40 months. Seo et al 22 found that RV free wall longitudinal strain was associated with a prognostic impact in patients with NIDCM, which did not differ markedly from the LV function assessed by LVEF or LVpGLS. In these studies, the myocardial strain measurements of speckle tracking echocardiography were contradictory. CMR is increasingly used as a standard tool in the evaluation of cardiac structure and function.27 Theoretically, it provides reproducible data on myocardial deformation, especially on RV longitudinal strain.11 25 27 The advantage of FT-CMR, which uses the steady-state free precession cine images, is that it does not require additional sequences and offers rapid post-processing.28 However, limited data are available on the RV 3D CMR FT strain. In a previous study, Buss et al 21 found that LV longitudinal strain is an independent predictor of survival in NIDCM, as assessed by CMR. Nonetheless, the study21 did not evaluate RV strain in NIDCM, and LVpGLS employed 2D FT methods of CMR. Such an FT strain analysis-based two-dimensional algorithm suffers from loss of features in the 3D, which could be adversely influenced by imperfect tracking within the chosen slice to reduce reproducibility.12 29 Furthermore, the assessment of strain from a limited number of short-axis and long-axis may not be an appropriate representation of the global myocardial function. A recent algorithm has been developed that allows 3D CMR FT of cine images,29 but there are no data to investigate the association between RVpGLS and prognosis in patients with stage C or D HF with NIDCM. To the best of our knowledge, the present study might be first to illustrate the independent value of RVpGLS from 3D CMR FT in patients with NIDCM and biventricular dysfunction. The study found that RV longitudinal strain assessed using 3D CMR FT is an independent predictor of cardiovascular death and cardiac transplantation in NIDCM with biventricular dysfunction. Compared with patients with RVpGLS <−8.5%, patients with RVpGLS >−8.5% have a higher risk of experiencing the composite of MACEs.
Nevertheless, the study has some limitations. First, this is a single-centre study with a limited number of patients that might restrict the generalisability of these results. Second, we focused only on 3D CMR FT biventricular global myocardial strain and evaluated neither the biventricular regional or segmental myocardial strain nor the bi-atrial strain. However, regional or segmental strain analysis is not the main aim of the study, which is focused on the evaluation of RV global myocardial strain analysis. Reportedly, the FT techniques are more robust and reproducible for global rather than regional values.25 Third, the data of the BNP covariate was missing in 28% of the participants. However, statistical techniques employed MI to address the issue of missing data, and sensitivity analyses indicated that our assumptions regarding the patterns of missing data were reasonable (online supplemental table 1). Finally, other existing independent precipitating factors, such as infections, arrhythmias and treatment differences, were not considered that may be clinically important and potentially related to MACEs.
heartjnl-2020-317949supp003.pdf (60.5KB, pdf)
Despite the limitations, this is the first study to assess the association between RV strain from 3D CMR FT and the long-term outcomes in NIDCM with biventricular dysfunction without AF. The finding has potential clinical implications that may have applications in the reclassification of patient groups suitable for advanced therapy (ie, transplantation, LV assist device implantation) with end-stage HF. Further research efforts should include a prospectively cohort to determine whether CMR in combination with novel markers could help identify patients with biventricular dysfunction without AF who may benefit from advanced treatment.
Key messages.
What is already known on this subject?
Many studies have recently shown that myocardial strain analysis, especially global longitudinal strain, might serve as an independent prognostic significance factor in patients with heart failure (HF).
It is unknown that the association between three-dimensional (3D) cardiac magnetic resonance (CMR) feature tracking (FT) right ventricular peak global longitudinal strain (RVpGLS) and major adverse cardiovascular events (MACEs) in patients with stage C or D HF with non-ischaemic dilated cardiomyopathy (NIDCM) but without atrial fibrillation (AF).
What might this study add?
We performed the observation cohort study to explore the association between 3D CMR FT RVpGLS and MACEs in patients with stage C or D HF.
Our study showed that RVpGLS-derived from 3D CMR FT is associated with a significant prognostic impact in patients with NIDCM with stage C or D HF and without AF.
How might this impact on clinical practice?
Our study showed that RVpGLS-derived 3D CMR FT is associated with a significant prognostic impact in patients with NIDCM with stage C or D HF and without AF.
The finding has potential clinical implications that may have applications in the reclassification of patient groups suitable for advanced therapy (ie, transplantation, left ventricular assist device implantation) with end-stage HF.
Further research efforts should include a prospectively enrolled cohort to determine whether CMR in combination with other novel markers could help identify patients with biventricular dysfunction without AF who may benefit from advanced treatment.
Acknowledgments
The authors would like to thank Xinlin Chen and Changzhong Chen (Department of Epidemiology and Biostatistics, X&Y Solutions in Boston) for their helpful review and comments about the manuscript and Jumatay Biekan (Circle Cardiovascular Imaging, Calgary, Alberta, Canada) for his assistance in image postprocessing. The authors would also like to thank all of the participating physicians who contributed patients to the project and assisted with the clinical follow-up.
Footnotes
Correction notice: This article has been corrected since it was published Online First. The funding statement was added.
Contributors: JD and LX are the guarantors of the integrity of the entire study. TL, JD and LX are responsible for the study concepts and design. TL, YG, HW, ZZ, RW, S-SC, YL, YS and HR performed the experimental studies. TL, YG and HW analysed and interpreted the data. TL and YG performed the statistical analysis and all the results were checked by JD and LX. TL, YG, HW, ZZ, RW, S-SC, YL, YS and HR prepared the first draft of the manuscript, which was critically revised by GY, DF, JD and LX.
Funding: This work was supported by grant from the National Key Research and Development Program of China (2016YFC1300300 and 2016YFC1301002), with additional grant supports provided by the National Natural Science Foundation of China (U1908211), and the Capital Medical Development Research Foundation of China (PXM2020_026272_000013). This work also supported in part by the British Heart Foundation (Grant Number:PG/16/78/32402).
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available on reasonable request. The datasets used and analysed during the current study are available from the corresponding author on reasonable request.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
This study protocol was approved by the Human Subjects Review Committee at Beijing Anzhen Hospital (Approval No. 2013007X).
References
- 1. Halliday BP, Cleland JGF, Goldberger JJ, et al. Personalizing risk stratification for sudden death in dilated cardiomyopathy: the past, present, and future. Circulation 2017;136:215–31. 10.1161/CIRCULATIONAHA.116.027134 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Liu T, Ma X, Liu W, et al. Late gadolinium enhancement amount as an independent risk factor for the incidence of adverse cardiovascular events in patients with stage C or D heart failure. Front Physiol 2016;7:484. 10.3389/fphys.2016.00484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Liu T, Song D, Dong J, et al. Current understanding of the pathophysiology of myocardial fibrosis and its quantitative assessment in heart failure. Front Physiol 2017;8:238. 10.3389/fphys.2017.00238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. La Vecchia L, Zanolla L, Varotto L, et al. Reduced right ventricular ejection fraction as a marker for idiopathic dilated cardiomyopathy compared with ischemic left ventricular dysfunction. Am Heart J 2001;142:181–9. 10.1067/mhj.2001.116071 [DOI] [PubMed] [Google Scholar]
- 5. Park JH, Park JJ, Park JB, et al. Prognostic Value of Biventricular Strain in Risk Stratifying in Patients With Acute Heart Failure. Journal of the American Heart Association 2018;7:e009331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Brieke A, DeNofrio D. Right ventricular dysfunction in chronic dilated cardiomyopathy and heart failure. Coron Artery Dis 2005;16:5–11. 10.1097/00019501-200502000-00002 [DOI] [PubMed] [Google Scholar]
- 7. Konstam MA, Kiernan MS, Bernstein D, et al. Evaluation and management of right-sided heart failure: a scientific statement from the American heart association. Circulation 2018;137:e578–622. 10.1161/CIR.0000000000000560 [DOI] [PubMed] [Google Scholar]
- 8. Gulati A, Ismail TF, Jabbour A, et al. The prevalence and prognostic significance of right ventricular systolic dysfunction in nonischemic dilated cardiomyopathy. Circulation 2013;128:1623–33. 10.1161/CIRCULATIONAHA.113.002518 [DOI] [PubMed] [Google Scholar]
- 9. Schulz-Menger J, Bluemke DA, Bremerich J, et al. Standardized image interpretation and post-processing in cardiovascular magnetic resonance - 2020 update : Society for Cardiovascular Magnetic Resonance (SCMR): Board of Trustees Task Force on Standardized Post-Processing. J Cardiovasc Magn Reson 2020;22:19. 10.1186/s12968-020-00610-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of echocardiography and the European association of cardiovascular imaging. Eur Heart J Cardiovasc Imaging 2015;16:233–71. 10.1093/ehjci/jev014 [DOI] [PubMed] [Google Scholar]
- 11. Voigt J-U, Cvijic M. 2- and 3-dimensional myocardial strain in cardiac health and disease. JACC Cardiovasc Imaging 2019;12:1849–63. 10.1016/j.jcmg.2019.01.044 [DOI] [PubMed] [Google Scholar]
- 12. Gatti M, Palmisano A, Faletti R, et al. Two-Dimensional and three-dimensional cardiac magnetic resonance feature-tracking myocardial strain analysis in acute myocarditis patients with preserved ejection fraction. Int J Cardiovasc Imaging 2019;35:1101–9. 10.1007/s10554-019-01588-8 [DOI] [PubMed] [Google Scholar]
- 13. Liu W, Wang Y, Zheng J, et al. Syndecan-1 as an independent risk factor for the incidence of adverse cardiovascular events in patients having stage C and D heart failure with non-ischemic dilated cardiomyopathy. Clinica Chimica Acta 2019;490:63–8. 10.1016/j.cca.2018.12.022 [DOI] [PubMed] [Google Scholar]
- 14. Jessup M, Abraham WT, Casey DE, et al. 2009 focused update: ACCF/AHA guidelines for the diagnosis and management of heart failure in adults: a report of the American College of cardiology Foundation/American heart association Task force on practice guidelines: developed in collaboration with the International Society for heart and lung transplantation. Circulation 2009;119:1977–2016. 10.1161/CIRCULATIONAHA.109.192064 [DOI] [PubMed] [Google Scholar]
- 15. Kramer CM, Barkhausen J, Flamm SD, et al. Standardized cardiovascular magnetic resonance imaging (CMR) protocols, Society for cardiovascular magnetic resonance: board of trustees Task force on standardized protocols. J Cardiovasc Magn Reson 2008;10:35. 10.1186/1532-429X-10-35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kramer CM, Barkhausen J, Flamm SD, et al. Standardized cardiovascular magnetic resonance (CMR) protocols 2013 update. J Cardiovasc Magn Reson 2013;15:91. 10.1186/1532-429X-15-91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Mordi I, Bezerra H, Carrick D, et al. The combined incremental prognostic value of LVEF, late gadolinium enhancement, and global circumferential strain assessed by CMR. JACC Cardiovasc Imaging 2015;8:540–9. 10.1016/j.jcmg.2015.02.005 [DOI] [PubMed] [Google Scholar]
- 18. Jaddoe VWV, de Jonge LL, Hofman A, et al. First trimester fetal growth restriction and cardiovascular risk factors in school age children: population based cohort study. BMJ 2014;348:14. 10.1136/bmj.g14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Cole SR, Hernán MA. Constructing inverse probability weights for marginal structural models. Am J Epidemiol 2008;168:656–64. 10.1093/aje/kwn164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Del Rio JM, Grecu L, Nicoara A. Right ventricular function in left heart disease. Semin Cardiothorac Vasc Anesth 2019;23:88–107. 10.1177/1089253218799345 [DOI] [PubMed] [Google Scholar]
- 21. Buss SJ, Breuninger K, Lehrke S, et al. Assessment of myocardial deformation with cardiac magnetic resonance strain imaging improves risk stratification in patients with dilated cardiomyopathy. Eur Heart J Cardiovasc Imaging 2015;16:307–15. 10.1093/ehjci/jeu181 [DOI] [PubMed] [Google Scholar]
- 22. Seo J, Jung IH, Park JH, et al. The prognostic value of 2D strain in assessment of the right ventricle in patients with dilated cardiomyopathy. Eur Heart J Cardiovasc Imaging 2019;20:1043–50. 10.1093/ehjci/jez015 [DOI] [PubMed] [Google Scholar]
- 23. Park JJ, Park J-B, Park J-H, et al. Global Longitudinal Strain to Predict Mortality in Patients With Acute Heart Failure. J Am Coll Cardiol 2018;71:1947–57. 10.1016/j.jacc.2018.02.064 [DOI] [PubMed] [Google Scholar]
- 24. Hamada-Harimura Y, Seo Y, Ishizu T, et al. Incremental prognostic value of right ventricular strain in patients with acute decompensated heart failure. Circ Cardiovasc Imaging 2018;11:e007249. 10.1161/CIRCIMAGING.117.007249 [DOI] [PubMed] [Google Scholar]
- 25. Claus P, Omar AMS, Pedrizzetti G, et al. Tissue tracking technology for assessing cardiac mechanics: principles, normal values, and clinical applications. JACC Cardiovasc Imaging 2015;8:1444–60. 10.1016/j.jcmg.2015.11.001 [DOI] [PubMed] [Google Scholar]
- 26. Edvardsen T, Klaeboe LG. Imaging and heart failure: myocardial strain. Curr Opin Cardiol 2019;34:490–4. 10.1097/HCO.0000000000000653 [DOI] [PubMed] [Google Scholar]
- 27. Haddad F, Hunt SA, Rosenthal DN, et al. Right ventricular function in cardiovascular disease, part I: anatomy, physiology, aging, and functional assessment of the right ventricle. Circulation 2008;117:1436–48. 10.1161/CIRCULATIONAHA.107.653576 [DOI] [PubMed] [Google Scholar]
- 28. Salerno M. Feature Tracking by CMR: A "Double Feature"? JACC Cardiovasc Imaging 2018;11:206–8. 10.1016/j.jcmg.2017.01.024 [DOI] [PubMed] [Google Scholar]
- 29. Liu B, Dardeer AM, Moody WE, et al. Reference ranges for three-dimensional feature tracking cardiac magnetic resonance: comparison with two-dimensional methodology and relevance of age and gender. Int J Cardiovasc Imaging 2018;34:761–75. 10.1007/s10554-017-1277-x [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
heartjnl-2020-317949supp001.pdf (57.6KB, pdf)
heartjnl-2020-317949supp002.pdf (65.5KB, pdf)
heartjnl-2020-317949supp003.pdf (60.5KB, pdf)
Data Availability Statement
Data are available on reasonable request. The datasets used and analysed during the current study are available from the corresponding author on reasonable request.