Abstract
Recent discoveries have improved our understanding of the physiological and pathological roles of the dopamine transporter (DAT); however, only a few drugs are clinically available for DAT-implicated disorders. Among those drugs, modafinil (MOD) and its (R)-enantiomer (R-MOD) have been used off-label as therapies for psychostimulant use disorders, but they have shown limited effectiveness in clinical trials. Recent preclinical studies on MOD and R-MOD have led to chemically modified structures aimed toward improving their neurobiological properties that might lead to more effective therapeutics for stimulant use disorders. This study examines three MOD analogs (JJC8-016, JJC8-088, and JJC8-091) with improved DAT affinities compared to their parent compound. These compounds were investigated for their effects on the neurochemistry (brain microdialysis and FSCV) and behavior (ambulatory activity) of male Swiss-Webster mice. Our data indicate that these compounds have dissimilar effects on tonic and phasic dopamine in the nucleus accumbens shell and variability in producing ambulatory activity. These results suggest that small changes in the chemical structure of a DAT inhibitor can cause compounds such as JJC8-088 to produce effects similar to abused psychostimulants like cocaine. In contrast, other compounds like JJC8-091 do not share cocaine-like effects and have a more atypical DAT-inhibitor profile, which may prove to be an advancement in the treatment of psychostimulant use disorders.
Keywords: cocaine use disorders, addiction, dopamine microdialysis, FSCV, nucleus accumbens shell, Modafinil
Graphical Abstract
INTRODUCTION
Recent discoveries on the dopamine (DA) transporter (DAT) have improved not only our understanding of its physiological functions but also of how several neuropsychiatric disorders occur as a result of its altered functionality1–3. Nonetheless, only a few drugs are clinically available that show therapeutic actions through inhibition of DA uptake at the DAT. Among them, D- and L-amphetamine (Adderall) and methylphenidate (Ritalin) are used to treat attention deficit hyperactivity disorder, bupropion (Wellbutrin and Zyban) is approved for treatment of nicotine dependence, and modafinil (Provigil, MOD) and its (R)-enantiomer (Nuvigil, R-MOD) are used for the treatment of sleep disorders. One of the factors limiting the clinical development of this class of drugs lies in its potential for abuse, and possibly the development of dependence. A reduction in or blockade of DAT function results in increased synaptic levels of DA in selected brain areas4, which is the main pharmacological action of abused psychostimulants like cocaine and methamphetamine.
Though an initial screening of DAT inhibitors predicted that the entire class of drugs would produce cocaine-like reinforcing effects5, 6, several “atypical” DAT blockers have been discovered and show reduced - if any - cocaine-like reinforcing actions in preclinical models7, 8. Interestingly, while atypical DAT blockers and cocaine share the same molecular target, their pharmacological and behavioral effects, including abuse liability, vary greatly. Recent studies have suggested that different chemical classes of DAT inhibitors prefer distinct conformations of the DAT protein, which appears to be related to their pharmacological actions9, 10. For example, cocaine and other typical (or cocaine-like) DAT inhibitors have been shown to stabilize an outward-facing conformation of DAT, while atypical DAT blockers prefer a more occluded and inward-facing conformation10–12. This difference in binding modes may have implications on the behavioral and pharmacological effects of these two types of reuptake inhibitors8, 13–15.
MOD and R-MOD are among the DAT blockers that promote a conformation of the DAT that is dissimilar to that preferred by cocaine and are FDA-approved wakefulness-promoting drugs used primarily for the treatment of sleep disorders16, 17. The abuse potential for either MOD or its (R)-enantiomer is low18, 19, despite their ability to increase extracellular DA levels by inhibiting reuptake via the DAT10, 20. MOD has been evaluated in clinical trials and has shown promising results as a treatment for psychostimulant use disorders21–23, though in only a selected patient population24, 25. Although the reasons for its limited success are unknown, attempts have been made to improve therapeutic efficacy for psychostimulant use disorders by conducting structure-activity relationship (SAR) studies on the parent compound to increase water solubility and DAT affinity26–28. Hence, several MOD analogs were recently designed, synthesized and evaluated in rodent models of psychostimulant abuse29, 30.
In this manuscript we further the in-vivo assessment in Swiss Webster mice of three previously identified lead compounds27, 28, JJC8-016, JJC8-088, and JJC8-091 (Figure 1), which have increased affinity for the DAT compared to MOD. Here, we assess each compound for its effects on locomotor activity, brain nucleus accumbens shell (NAS) DA dynamics (phasic changes, and reuptake) via fast scan cyclic voltammetry (FSCV), and extracellular NAS DA levels via brain microdialysis. These results provide further guidance towards the discovery of therapeutic strategies to treat psychostimulant use disorders.
Figure 1.
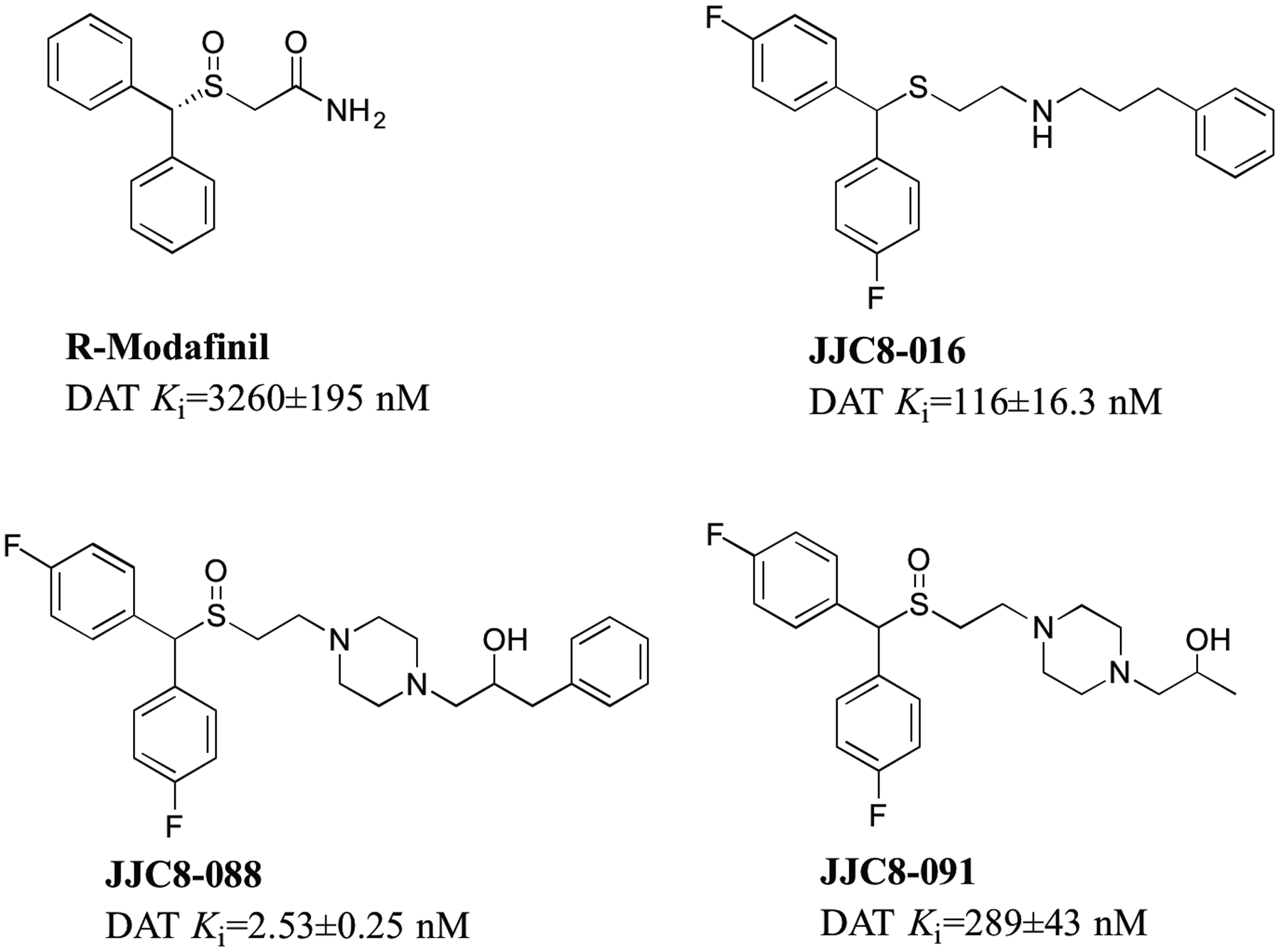
Chemical structures and DAT binding affinities (nM ± SEM) for R-MOD and MOD analogs (from Cao et al 2011, and 2016) tested in this report.
RESULTS AND DISCUSSION
The present study was aimed to assess the behavioral and neurochemical features of selected MOD analogs that may provide insight into the search for pharmacological therapies for psychostimulant use disorders. In our FSCV experiments, R-MOD, the MOD derivatives (JJC8-016, JJC8-088, and JJC8-091), and cocaine were assessed in the NAS of mice. Cumulative dosing curves were created to demonstrate the effect of each compound on the DA peak produced by stimulus (DAMax) and the rate of DA removal from the electrode (clearance), which is mainly driven by reuptake of DA through the DAT. The dose range used for each compound was based on its DAT affinity (Figure 1) as previously reported27, 28. Experiments were performed in urethane anesthetized mice and doses are represented as mg/kg (i.p.). The NAS was selected for these tests as it was shown that acute administration of abused psychostimulants resulted in changes in DA dynamics related to their potential reinforcing effects31, 32. All the statistical analysis results are reported in the supplementary information (SI, Tables 1–8).
An example of color-plot and concentration vs time plot representations of the effects of administration of each compound on DA dynamics is presented in Figure 2. Each compound and the vehicle are scaled to the same z-axis for comparison, and demonstrated various degrees of efficacy in their effects on the clearance of DA and DAMax. Administration of the vehicle did not produce significant changes in the rate of DA clearance (Figure 2A, SI Table 2). Each of the tested compounds reduced the rate of DA clearance, creating a color-plot where the extracellular DA level remained elevated as shown on Figure 2. Additionally, R-MOD (Figure 2B), JJC8-088 (Figure 2D), and JJC8-091 (Figure 2E) increased the DAMax, while JJC8-016 (Figure 2C) did not.
Figure 2.

Representative colorplots demonstrating the effects of (A) vehicle (10% DMSO, 15% Tween 80); (B) 32 mg/kg R-MOD; (C) 32 mg/kg JJC8-016; (D) 32 mg/kg JJC8-088; and (E) 32 mg/kg JJC8-091 on the elicited DA peak (DAMax) and clearance rate of NAS DA from the electrode during FSCV experiments. Inset above each colorplot is the corresponding DA concentration vs time plot for the same measurement.
The cumulative dosing analysis for each compound tested is shown in Figure 3, where data are reported as percentage of baseline (see SI table 1 for absolute baseline values) obtained for each animal during a 20 min data collection period before the administration of the tested compounds. The vehicle (10% DMSO, 15% Tween80) was found to have no significant effect on the clearance rate (see SI table 2). Cocaine, a typical DAT inhibitor, has been reported in several publications to reduce DA reuptake by directly blocking the DAT33–35. In agreement with these previous reports, cocaine (0.5–56 mg/kg i.p.) was found to dose dependently decrease the clearance rate of DA measured here by k36, first order rate constant, from Equation 1. R-MOD and each of the analogs also dose-dependently (SI Table 3) decreased DA clearance rate to varying degrees (Figure 3A). Even though R-MOD (5–100 mg/kg i.p.) has a very low affinity for the DAT, about 3.1 μM, the lowest in the compounds tested here, it dose-dependently blocked DA reuptake, reducing the clearance rate of DA down to about 40% baseline. JJC8-088 (0.5–56 mg/kg i.p.), the compound with the highest DAT affinity in this series, produced a significant shift in the clearance rate of DA down to about 30% of baseline. JJC8-091 (5–56 mg/kg i.p.) and JJC8-016 (0.5–56 mg/kg i.p.) have higher affinity for DAT than R-MOD and both produced significant reductions in DA clearance rate (Figure 3A, SI Table 3).
Figure 3.
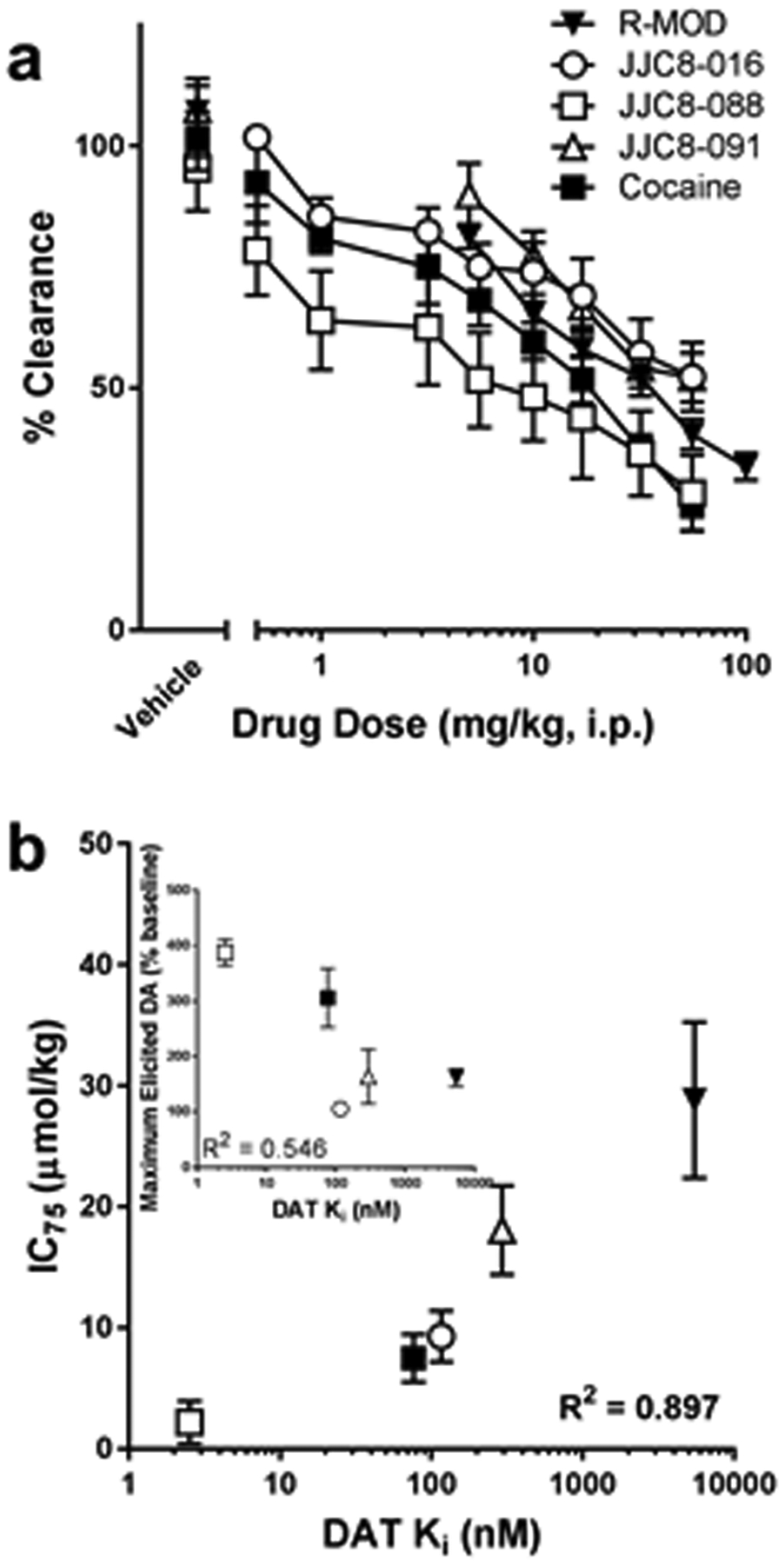
(A) Effects of administration of cumulative doses, in mg/kg, of R-MOD and each analog compound and cocaine on the rate of DA reuptake (clearance) at DA nerve terminals in the NAS. Baseline values: Cocaine, 1.61 ± 0.26 s−1 (n=6); R-MOD, 1.73 ± 0.21 s−1 (n=4); JJC8-016, 1.61 ± 0.22 s−1 (n=5); JJC8-088 2.17 ± 0.28 s−1 (n=4); JJC8-091 2.20 ± 0.08 s−1 (n=4). Baseline values were not found to be significantly different between groups or from vehicle treatments (SI Tables 1 and 2). (B) A relationship was found between the DAT affinity for the drugs tested (data from Cao et al, 2016 27) and the IC75, expressed as μmol/kg, measured in the present report. (B, inset) A similar relationship was not apparent between the maximal elicited DA peak and DAT affinity. Data represent mean ± SEM.
To determine if DAT affinity plays a role in each compound’s effectiveness at blocking DA reuptake, the IC75 calculated from the clearance rates from dose response curves on Figure 3A were used to determine inhibition constants that could be compared to the DAT affinity(Figure 3B). C75, expressed as μmol/kg for each drug, is a measure of the ability of each compound to block DA reuptake after stimulus; the 75th percentile was used instead of the more common 50th percentile because some of the compounds tested here were unable to produce an effect that large even at high doses. As seen in Figure 3B, there is a good relationship, R2 = 0.897, between the log DAT affinity of the compounds and their IC75, indicating that higher affinity compounds, such as JJC8-088, block reuptake with lower doses than lower affinity compounds, such as R-MOD. This suggests a link between DAT affinity and each compound’s ability to inhibit DA reuptake in-vivo.
Thus, results from FSCV experiments show that all tested MOD analogs reduced the clearance rate of extracellular DA in the NAS. The potency of this effect was significantly related to the in vitro DAT binding affinities of these compounds. In contrast, the analogs’ effects on DAMax proved to be more variable. Administration of each compound or the vehicle demonstrated a different degree of effects on DAMax as shown by the color-plot examples in Figure 2. The drugs’ vehicle did not significantly modify DAMax as compared to baseline (see SI Table 4, baseline values, and SI Table 5, t test values for vehicle vs baseline for all treatment groups). The cumulative dosing administration of R-MOD, up to 100 mg/kg i.p., produced a small but significant increase in DAMax, about 60% over baseline (Figure 4, SI Table 6). The 2-OH-propyl piperazine analog, JJC8-091, produced an increase in DAMax, about 60%, comparable to that elicited by R-MOD at a similar dose range, but that did not reach statistical significance. However, the 2-OH-3-phenylpropyl piperazine analog, JJC8-088, significantly (see SI Table 6) increased the DAMax to nearly 400% of baseline at the highest dose. JJC8-016, which unlike JJC8-088 and JJC8-091 does not contain a piperazine ring, did not increase but actually decreased the DAMax by 10% at high doses. Thus, only JJC8-088 produced effects at levels similar to those elicited by cocaine, which also significantly increased DAMax to nearly 300% of baseline at the highest doses (Figure 4, SI Table 6). A similar relationship to that found for DAT affinity and IC75 clearance rate was not found between the maximal effect each compound has on DAMax and DAT affinity, R2 = 0.546 (Figure 3B inset). This suggests that DAT affinity may not be the only factor playing a significant role in the ability of a DAT inhibitor to influence elicited DA release. Previous studies reported increases in the DA peak caused by an electrical stimulus in freely moving and urethane anesthetized mice and rats treated with DAT blockers37–39. However, in the present study JJC8-016 significantly and dose-dependently reduced DA clearance rate but did not produce a significant increase in the DA peak at any dose.
Figure 4.
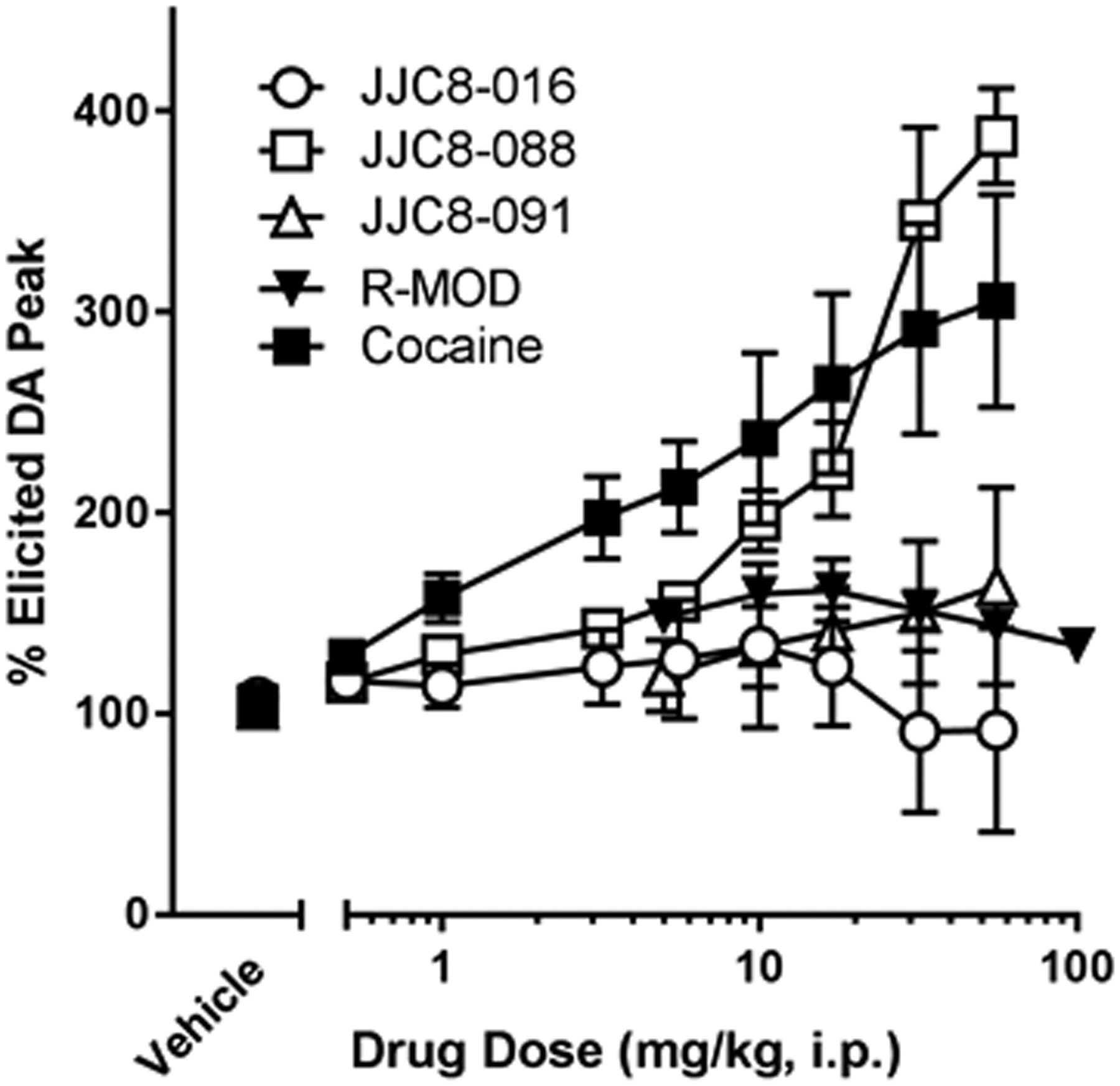
Effects of administration of cumulative doses of R-MOD, each derivative compound and cocaine on the maximum elicited DA peak (DAMax), reported as a percentage of baseline DAMax. Data represents mean ± SEM (n = 4–6). Baseline values: Cocaine, 0.13 ± 0.02 μM (n=6); R-MOD, 0.38 ± 0.08 μM (n=4); JJC8-016, 0.43 ± 0.14 μM (n=5); JJC8-088, 0.29 ± 0.13 μM (n=4); JJC8-091, 0.26 ± 0.06 μM (n=4). DAMax baseline values were not found to be significantly different between groups or from vehicle treatments (SI Tables 4 and 5).
R-MOD and each of its analogs have been shown to bind to the DAT27; however, differences in their binding modalities, which could induce different DAT conformations or differences in off-target interactions may result in different effects on DAMax. R-MOD significantly increased DAMAX, while the effects elicited by JJC8-091on DAMAX did not reach statistical significance though with a 10-fold greater DAT affinity. This suggests that further research into its potential as a treatment for psychostimulant use disorders may be warranted. At the same time JJC8-088, which is structurally very similar to JJC8-091 significantly enhanced DAMax, as has previously been demonstrated with cocaine in multiple rodent species33, 34.
In vivo brain microdialysis tests provide a good assessment of drug-induced changes in extracellular DA levels as a function of dose and time. Our tests were performed in freely-moving mice implanted with probes in the NAS. Basal levels of NAS DA (41.87 ± 3.61 fmol/sample, n=55) were not significantly different between experimental groups (ANOVA, F 9,45 = 0.694, p = 0.71).
The acute administration of JJC8-016 did not elicit significant changes in DA levels in dialysates after the lowest doses, 10 and 30 mg/kg i.p. Larger doses of JJC8-016 (50 and 100 mg/kg i.p.) modestly decreased extracellular DA levels below basal values, an effect that lasted over 2 hours but did not reach significance (p>0.05) as compared to vehicle treated mice (Figure 5A, SI Table 7). The administration of JJC8-088 (10, 32, 56 mg/kg i.p.) elicited a rapid, significant increase in DA levels in dialysates, which reached their maxima (about 570% of basal DA values) about 20–30 min after injection of the highest doses (Figure 5B, SI Table 7). All doses of JJC8-088 produced a long-lasting stimulation of DA levels with values still reaching about 200% of basal levels 2 hours after drug administration (Figure 5B). Administration of JJC8-091 produced a slow increase in extracellular DA levels in dialysates from the NAS with maxima (168 to 220% of basal values, depending on dose) reached at the 50–60 minute time points. The effects of JJC8-091 on DA levels were long lasting, with DA values still above basal levels (50 to 90%) 2 hours after drug administration (Figure 5C, SI Table 7).
Figure 5.
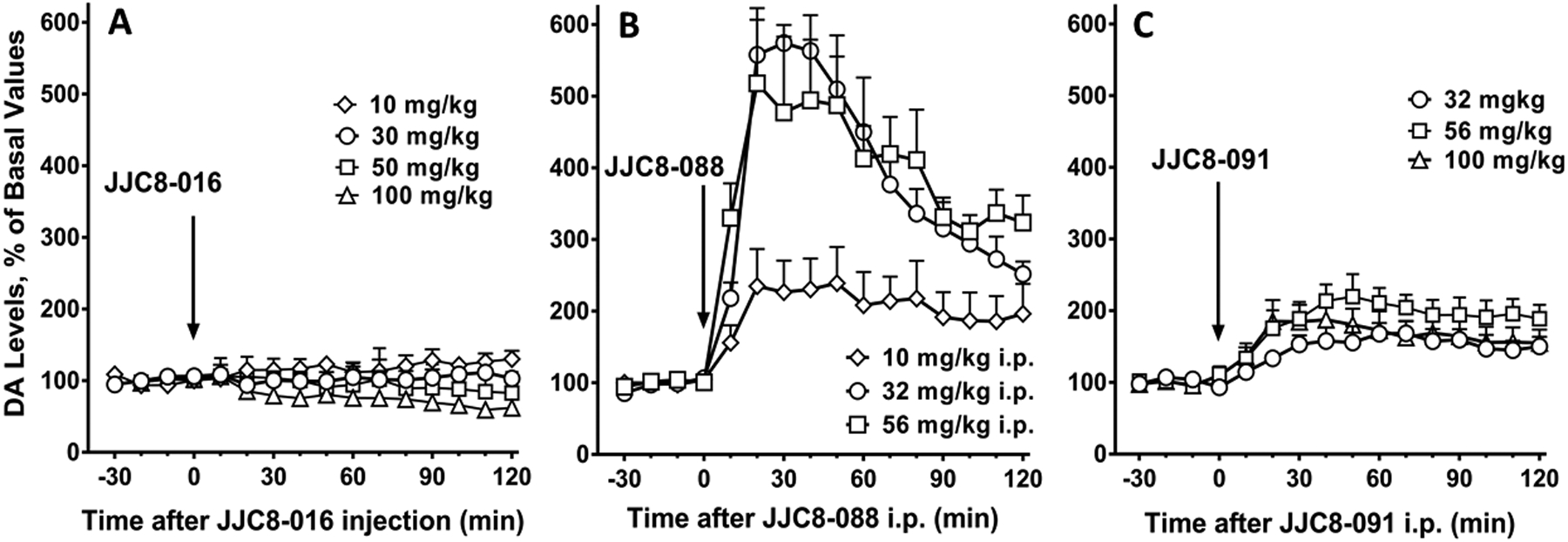
Time course of effects of administration of JJC8-016 (A) (10, 30, 50, 100 mg/kg i.p., n= 5–7), JJC8-088 (B) (10, 32, 56 mg/kg i.p., n= 4–6) and JJC8-091 (C) (10, 32, 56 mg/kg i.p., n= 5–6) on DA levels in dialysates from the NAS in Swiss Webster mice. Results are means, with vertical bars representing SEM, of the amount of DA in 10-min dialysate samples, expressed as percentage of basal values, uncorrected for probe recovery.
JJC8-088 elicited a significant increase in DAMax (about 400%) in FSCV tests, and it also produced a large stimulation of extracellular NAS DA levels. JJC8-091, like JJC8-088, produced a long lasting (>2 hours) though less efficacious stimulation of NAS DA levels, but did not significantly increase DAMAX in FSCV tests. On the other hand, JJC8-016 did not produce significant stimulation in DAMax in FSCV tests or in extracellular NAS DA levels at any of the doses tested, but administration of the highest doses induced a reduction in extracellular levels of DA that, though not reaching statistical significance, was not expected for from a drug that significantly inhibits DA reuptake.
The reduction in clearance rate of NAS DA measured by FSCV was statistically significant for all of the MOD analogs and directly related to their DAT binding affinities. This demonstrates that these MOD analogs block the reuptake of DA via the DAT, resulting in increased extracellular levels of DA. However, a significant increase in NAS levels of DA was obtained after administration of JJC8-088 and JJC8-091, while we did not observe an increase in DA levels after administration of JJC8-016. As stated previously, administration of JJC8-016 produced a reduction in extracellular DA levels, which is difficult to reconcile with a significant blockade of the DAT. We suggest that off-target actions of JJC8-016, unrelated to blockade of the DAT, may play a role in this observation27, 30. In addition, phase I metabolic instability reduced its concentrations to less than 10 percent remaining after 30 min incubation in rat liver microsomes, which might provide an alternative explanation for the effects of JJC8-01629. This rapid metabolism could affect its efficacy as a DAT blocker, and could also result in an increased presence of metabolites, which may provide the off-target effects related to reduction in DA levels at high doses.
It has been established previously that both FSCV and microdialysis report the changes in DA that correlate with the kinetics of pharmacological manipulations in the nucleus accumbens of rats40. While the two techniques generally measure events on different time scales (FSCV in tenths of seconds, microdialysis in minutes), measurements of DA in the NAS have been linked to behavioral and reinforcing effects as well as linked to changes in neuronal structure and function. A previous report41 used both microdialysis and in vitro FSCV to assess DA functions on locomotor activity, allowing researchers to see changes in DA uptake rate due to genetic alteration that correlated with differences found in extracellular DA levels and locomotion between wildtype and DAT KO mice41. In this study, the use of urethane anesthetized mice allowed for the evaluation of the DA uptake rate and elicited DA peak in a preparation where neuronal and physiological feedbacks might also play a role in the responses to the drugs tested.
In freely moving animals, behaviors can be studied in relation to DA dynamics. For instance, rats trained to self-administer cocaine do so in a manner that maintains a threshold extracellular DA concentration as measured by microdialysis in the core and shell of the nucleus accumbens42. In the same brain areas, intravenous infusions of cocaine have reported to produce an increased amplitude and frequency of spontaneous DA release42. This work illustrates one of the causes behind the rise in extracellular DA when cocaine is present43, 44 in addition to blockade of the DAT. Further studies involving FSCV have indicated that DA release can be elicited by the cue that was associated with availability instead of the delivery of the reinforcer45, 46. Taken together the results from microdialysis and FSCV studies have brought light to insights in the DA signaling that could not have been discovered using another method.
Previous studies have attempted to clarify how tonic and phasic modalities of DA neuronal firing and the related release of DA would contribute to the extracellular levels of DA in nerve terminals during resting conditions47–49 or in response to external stimuli32, 43, 44. In their study, Owesson-White and colleagues49 compared the microdialysis and FSCV data obtained from awake freely moving rats, proposing that the extracellular level of DA largely reflects DA transients produced by the phasic firing of DA neurons. Therefore, using microdialysis and FSCV together allows us to measure both the basal DA concentration in the synapse, as well as the role the DAT is playing in those changes. Although there are differences in the tissue surrounding a microdialysis probe and a carbon fiber microelectrode50, the combination of techniques can be highly informative. For instance, each of the MOD analogs tested in the present study significantly reduced the DAT clearance rate, but the changes in NAS DA concentrations were at about 500 to 600 % of basal values for JJC8-088 at the highest doses and only at about 200 % for JJC8-091, or did not show significant changes in DA levels at all for JJC8-016. This suggests that not just the blockade of DAT but also pharmacokinetic and/or brain penetration differences27, 29 and possibly other actions at the synaptic level, may account for their varying efficacy in increasing extracellular DA levels. The affinity of R-MOD and its analogs for DA D2 and σ1 receptors, and that for the serotonin and norepinephrine transporters has recently been reported27. As discussed in that report, even though some of the compounds showed affinity for D2 receptors, their potency in inhibiting quinpirole-stimulated D2 receptor activation was very low. These effects suggest that even though some of the behavioral effects of MOD could be mediated by actions at D2 receptors, this is likely due to an indirect action resulting from an increase in DA levels51. Another possible off-target activity of R-MOD and its analogs is their potential affinity for σ1 receptors27. Interactions between DAT and σ1 receptors have been suggested as a potential mechanism for the attenuation of cocaine reinforcing effects by atypical DAT blockers52, 53. However, acute cocaine-induced stimulation of DA levels in the NAS was not significantly affected by antagonists of σ1 receptors54, suggesting that the dopaminergic effects of acute administration of the DAT blockers tested in the present experiments are not likely mediated by σ1 modulatory actions. Finally, off-target effects due to interactions with SERT or NET seem unlikely due to the high selectivity of these MOD analogs for DAT27. Even though the DAT is the main pharmacological target of cocaine related to its reinforcing effects, several studies have shown that off-target effects may also contribute to its psycho-emotional actions55, 56. While our study was not aimed to address the central or peripheral contribution of off-targets effects to the actions of cocaine or these MOD analogs, further investigation may be required to address off-target mechanism/s of action if, for example, JJC8-091 is moved toward the clinic.
Acute administration of standard DAT inhibitors (i.e. cocaine) in rodents produces a dose-dependent stimulation of behaviors, like ambulatory or stereotypy activities57, which may be different from the stimulation produced by non-standard, atypical DAT blockers7, 8. These different levels of behavioral activation may be related to distinct modalities of interaction with the DAT, or the presence of potential off-target effects. Thus, results from these behavioral tests could help clarify the pharmacological profiles of novel DAT blockers.
In the present behavioral tests, when animals received vehicle, the ambulatory activity, expressed as distance traveled (cm/10 min), peaked in the first 10 min and then gradually decreased to its minimum at 120 min. This effect could be explained by the increase in exploratory behavior elicited by placing mice in a novel environment. A dose of 3 mg/kg JJC8-016 produced a small but significant increase of ambulatory activity only at the 10 min time point after administration, whereas larger doses (10, 30, and 50 mg/kg i.p.) had no significant effect or decreased activity as compared with vehicle (Figure 6A; SI Table 8). In contrast, administration of JJC8-088 (10, 32, and 56 mg/kg i.p.) induced a dose-dependent stimulation of distance traveled that peaked at 30–40 min and remained elevated, compared to vehicle effects, throughout the experimental session, 120 min (Figure 6B; SI Table 8). Intermediate and small doses of JJC8-091 (3, 10, and 32 mg/kg i.p.) minimally increased ambulatory activity at 10 and 30 min whereas a larger dose (56 mg/kg i.p.) reduced it below levels observed after vehicle administration in the first 30 min of the session (Figure 6C; SI Table 8). Cocaine (3, 10, 17, and 30 mg/kg i.p.) induced a rapid and high stimulation of ambulatory activity that then decreased in a time- and dose-dependent fashion to vehicle levels at the end of the session (Figure 6D; SI Table 8).
Figure 6.
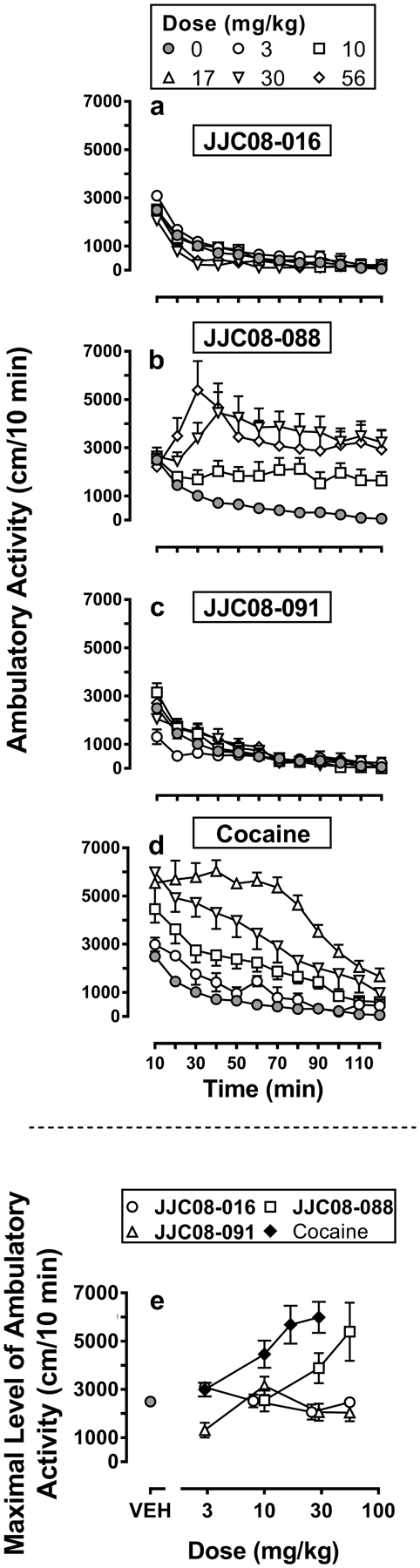
Time course (panels A-D) of ambulatory activity expressed as distance traveled, cm/10 min, obtained after administration of JJC8-016 (panel A; 3, 10, 30, 56 mg/kg i.p.); JJC8-088 (panel B; 10, 30, 56 mg/kg i.p.); JJC8-091 (panel C; 10, 30, 56 mg/kg i.p.); and cocaine (panel D; 3, 10, 17, 30 mg/kg i.p.) in Swiss Webster mice. The legend on the top panel indicates the doses of drugs reported on panels A to D. The maximal level of ambulatory activity obtained for each drug as a function of dose is reported on the bottom panel (E). Each point represents the group mean ± SEM (vertical bars) of the distance traveled (cm) in 10 min. Group size was at least six subjects for all groups, except for the group administered with the lowest cocaine dose (3 mg/kg, n=4).
Dose-effect curves revealed comparable distance traveled maxima produced by cocaine and JJC8-088, with the latter approximately 10-fold less potent than cocaine. This difference may be the result of pharmacokinetics/brain penetrance, since JJC8-088 has higher affinity (Ki = 2.5 nM) for the DAT than cocaine (Ki = 72 nM), but poorer brain penetration29. The R-MOD analogs, JJC8-016 and JJC8-091, did not stimulate locomotor activity at levels that exceeded that of the ineffective dose of cocaine (3 mg/kg) (Figure 6E). Even though a direct comparison of ambulatory effects elicited by R-MOD and its analogs is not shown here, the ambulatory activity of R-MOD has been previously reported28 and compared to that elicited by cocaine. While the maximal effect of R-MOD was obtained at the 100 mg/kg dose, it was noted that such activity was lower than that produced by cocaine. In that report, the equipment was the same and experimental conditions by Cao et al were comparable to those used for the present experiments. Thus, taking into consideration those previously reported results, among the drugs tested only JJC8-088 shows a cocaine-like stimulation of maximal ambulatory activity, which appears larger than that originally reported for R-MOD28. The maximal stimulation of ambulatory activity shown by the other MOD analogs was at levels similar to those obtained by vehicle administration. The limited stimulation of ambulatory activity obtained with JJC8-016 and JJC8-091 was not due to an increase in the frequency of other behaviors not compatible with locomotion (i.e. stereotypies and rearing; data not shown). As both compounds have affinity for the DAT (Ki = 116 nM and 289 nM for JJC8-016 and JJC8-091, respectively), it is possible that they interact with the DAT differently, potentially in divergent binding modes than cocaine and JJC8-088. Indeed, distinct interactions with the DAT have been suggested to play a role in behaviors associated with administration of typical (cocaine-like) or atypical DA uptake inhibitors10, 13, 14.
Recently, these compounds have been tested in intravenous self-administration procedures in rats to provide data on their potential actions against the reinforcing effects of methamphetamine29. Pretreatments with JJC8-088 did not significantly reduce the behavioral effects of methamphetamine related to its reinforcing actions in both short (1 hour) and long (6 hour) access sessions of methamphetamine self-administration. JJC8-016 and JJC8-091, instead, significantly and dose-dependently reduced the number of methamphetamine infusions during both the short and long access sessions29. JJC8-016 pretreatments also reduced the reinforcing actions of cocaine in a separate group of experiments30. Thus, our data provide further information on the pharmacological profiles and potential therapeutic actions of these MOD analogs, exhibiting the cocaine-like stimulant actions of JJC8-088 and the atypical DAT blocker profile of JJC8-016 and JJC8-091 through independent experiments.
CONCLUSIONS
Modifications to the MOD structure have resulted in analogs with significantly higher affinity for the DAT than the parent compound27, 28. The present results show that these MOD analogs have distinct behavioral and neurochemical profiles from the parent drug and from each other. Among the compounds tested in this study, JJC8-088, the analog with the highest binding affinity for the DAT, demonstrated a behavioral profile and DA dynamics that were similar to those of cocaine or typical DAT blockers. The other analogs tested here, JJC8-016 and JJC8-091, have lower DAT affinities than JJC8-088 but much higher (10–20 times) than their parent drug and close to the DAT affinity of cocaine28. In contrast, they showed effects on DA dynamics, time course of changes in extracellular DA levels, and behavioral stimulant activities, which have been described for atypical DAT blockers7, 8. Our results have shown various degrees of effects among the MOD-analogs and their parent drug on ambulatory activity, DA dynamics and time-course of extracellular levels that are likely the result of differences in their DAT affinity, pharmacokinetics and brain permeability27, 29. Further, binding modes that induce conformational changes at the DAT protein level may also be involved and are currently being investigated.
MATERIALS AND METHODS
Subjects.
Male, Swiss-Webster mice (Charles River, Wilmington, MA), weighing 25–30 g and experimentally naïve at the start of the study, were group housed (four mice per cage) in temperature- and humidity-controlled rooms on a 12-h light/dark cycle (lights on from 0700–1900h). Mice had free access to food and water at all times except during test sessions performed between 09:00 and 18:00 hours. Mice were kept in the animal facility for at least a week before being assigned to experimental groups or to undergo any behavioral or surgical procedure. The present study was approved by the Animal Care and Use Committee of the National Institute on Drug Abuse, Intramural Research Program, Baltimore MD, USA. Care of the subjects was in accordance with the guidelines of the National Institutes of Health and the National Institute on Drug Abuse Intramural Research Program, Animal Care and use Program, which is fully accredited by AAALAC International.
Compounds.
(R)-MOD, JJC8-016, JJC8-088, and JJC8-091 were synthesized in the Medicinal Chemistry Section, NIDA-IRP, according to published procedures27, 28, 58, and dissolved in a vehicle containing DMSO 10%, Tween-80 15%, and sterile saline 75% (V/V/V). Cocaine (Mallinckrodt, Saint Louis, MO) was dissolved in saline. Pretreatment times and doses of drugs used in the present study are described below and were chosen based on preliminary data obtained in this laboratory.
FSCV
FSCV Surgery
Swiss Webster mice were anesthetized with 1.2 g/kg urethane in sterile saline and given boosters of one third the original dose until properly anesthetized. Mice were placed in a stereotaxic apparatus where the skull was exposed, and four small holes were drilled to expose the dura. Mice were then implanted with a bipolar tungsten stimulation electrode into the Medial Forebrain Bundle (MFB) [uncorrected coordinates from bregma59, posterior −1.5 mm, lateral, ±1.0 mm, and ventral −4.5 mm]; the stimulation electrode was tested by applying a train of 24 pulses of 180 μA, 60 Hz, 4 ms in duration which produced a detectable movement of the whiskers. A carbon fiber microelectrode (CFME)60 working electrode was slowly lowered to its final position in the NAS of the ipsilateral hemisphere (anterior +1.5 mm, lateral ±1.3 mm, ventral −4.8 to −5.0 mm from bregma) while testing the DA response to stimulus to insure a location with a robust DA signal was found. Only the final CFME location was used as the recording site for data collection before and after vehicle and drug treatments. In each subject a baseline period and a vehicle treatment were collected before a cumulative dose curve for one of the test compounds was collected. An Ag/AgCl reference electrode secured by a screw was implanted in the contralateral hemisphere. At the conclusion of the experiment the electrode placement was marked with an electrolytic lesion by applying 10 V cathodically for 30 s to the working electrode creating a small recognizable landmark in the NAS.
Electrochemistry
DA was detected with 100 μm long cylindrical glass sealed carbon-fiber microelectrodes60 as previously described61, and pre-calibrated in vitro with known concentrations of DA (0.5–3 μM) using a custom built flow cell. DA was identified by oxidation and reduction peaks of the CV with fast-scan cyclic voltammetry with a voltage scan from −0.4 to 1.3 V at 400 V/s. During the experiment a stimulus of 24 pulses of 180 μA, 60 Hz, 4 ms in duration was applied at five-minute intervals. Data was collected using a UEI potentiostat and breakout box running Tarheel CV (University of North Carolina, Chapel Hill electronics shop) using a pair of Digitimer Neurolog NL800A (Digitimer North America, Ft. Lauderdale, FL, USA) current stimulus isolators to control the stimulation. The presence of DA was confirmed by cyclic voltammogram produced by the peak after stimulation.
Histology
At the end of the experiment mice were euthanized with an excess of urethane and an electrolytic lesion was created, as described above; brains were removed and fixed in a 4% formalin solution. Following fixation brains were transferred to a 30% sucrose solution for several days before being sectioned (30 μm slices) with a cryostat (Leica Biosystems, Richmond, Il). Sections were mounted and stained with Cresyl Violet, placement in the NAS was confirmed by the location of the electrolytic lesion in relation to anatomical landmarks.
FSCV Data Analysis
Data recorded by each stimulus was analyzed using HDCV (University of North Carolina, Chapel Hill) to determine the concentrations of the chemical components contributing to the stimulated peak62. The DAMax and DA clearance rate were determined using a custom macro written in Igor Carbon Pro which identified peaks greater than 3 X root mean square noise and fit the descending portion of the peak to equation 1, which defines the changes in DA concentration (μM) as a function of t, time (s), where DAMax is the peak DA concentration after stimulus, and k is first-order rate constant, which is used as an estimate as the clearance rate of DA from the electrode36, 63.
| (1) |
A 20 min of baseline data was collected for each animal before the tested compounds were administered. Data are reported as percentage of baseline. Differences in baseline clearance rate of DA or in baseline stimulated DA between different experimental groups were analyzed with one-way ANOVA with a Bonferroni post-hoc analysis; no significant baseline differences between groups was found (SI tables 1 and 3). Potential changes in baselines due to vehicle administration were analyzed with a paired t test comparing baseline to vehicle measurements for DA clearance rate and DAMax in each group (SI Tables 1 and 4). Cumulative dosing effects for each drug were analyzed for significance using a repeated measures one-way ANOVA with a Bonferroni post-hoc analysis. Changes were considered to be significant when p<0.05. All FSCV related statistical analysis results are reported in tables in the supplementary information. The IC75 values on Figure 3B were calculated as the drug dose, expressed in μg/kg, that reduced the DA clearance to 75% of baseline by using Prism 7.0 software. Maximal elicited DA is defined as the greatest change in stimulated DAMax from baseline measured for each compound tested. Data are presented as mean ± SEM for each group. Experimental group size is indicated in the figure legends.
Microdialysis studies.
Concentric dialysis probes were prepared using AN69 dialyzing membranes (Hospal Dasco, Bologna, Italy) as described previously15, 64. The exposed dialyzing surface of the membrane, i.e. not covered by glue, was limited to the lowest 1.0 mm portion of the probes that were less than 18 mm in total length. Mice were anesthetized and then placed in a stereotaxic frame (Kopf Instruments, CA, USA); the skull was surgically exposed, and a hole was drilled to expose the dura. Concentric dialysis probes were implanted randomly in the right or the left NAS (Uncorrected coordinates from Paxinos and Franklin59 expressed in mm from bregma were: Anterior = +1.5, Lateral = ±0.6, Vertical = −5.2) as described previously64. After surgery, subjects were given a subcutaneous injection of saline to replenish body fluids and were allowed to recover overnight in square Plexiglas cages (Med Associates). Cages were equipped with overhead quartz-lined fluid swivels (Instech Laboratories Inc., Plymouth Meeting, PA) for connections to the dialysis probes. All subsequent studies were conducted in these cages64.
Experiments were performed in freely moving animals in the same cages in which they recovered from surgery. Microdialysis test sessions started at 9:00 a.m., approximately 42–47 hours after the surgical procedures. Probes were connected to fluid with swivels (375/D/22QM; Instech Laboratories, Plymouth Meeting, PA, USA) and perfused with Ringer’s solution (147.0 mM NaCl, 2.2 mM CaCl2, and 4.0 mM KCl) delivered by a 1.0 ml syringe, operated by a BAS Bee Syringe Pump Controller (BAS West Lafayette, IN, USA), through the dialysis probes at a constant flow rate of 1 μl/min. Collection of dialysate samples (10 μl) started after about 30 min, and samples collected every 10 min were immediately analyzed for DA content.
After establishment of a DA baseline, 2–4 samples differing no more than 15% (approximately after 1 hour), different groups of naïve mice were injected with one dose of the test compounds. After administration of the compounds, sample collection continued every 10 min typically during the first 2 hours after treatment, and every 20 min thereafter (but only 10 of the 20 μl collected were analyzed).
Dialysate samples (10 μl) were injected without purification into a high-performance liquid chromatography apparatus equipped with a MD 150 × 3.2 mm column, particle size 3.0 μm (ESA, Chelmsford, MA) and a coulometric detector (5200a Coulochem II, or Coulochem III, ESA) to quantify DA. Potentials for the oxidation and reduction electrodes of the analytical cell (5014B; ESA) were set at +125 mV and −125 mV, respectively. The mobile phase, containing 100 mM NaH2PO4, 0.1 mM Na2EDTA, 0.5 mM n-octyl sulfate, and 18% (v/v) methanol (pH adjusted to 5.5 with Na2HPO4), was pumped at a flow-rate of 0.50 ml/min by an ESA 582 solvent delivery module. Assay sensitivity for DA was 2 fmoles per sample.
At the end of the experiment, mice were euthanized by pentobarbital overdose, brains were removed and left to fix in 4% formaldehyde in saline solution15. Brains were sliced, using a vibratome (Vibratome Plus, The Vibratome Company, St. Louis, MO), in serial coronal slices oriented according to the mouse brain atlas59, in order to identify the location of the probes. Data were only used from subjects for which probe tracks were within the correct NAS boundaries.
Microdialysis data are shown as percentage changes in basal DA values, calculated as the mean of 2–4 consecutive samples immediately preceding the first drug or vehicle injection. All results are presented as group means (± SEM). Statistical analysis (see SI table 7) was carried out with Statistica Software (Tulsa, Oklahoma) using one-way ANOVA or two-way ANOVA for repeated measures over time, with drug dose and time as factors. Results from treatments showing overall changes were subjected to post-hoc Tukey`s test. Changes were considered to be significant when p<0.05.
Behavioral activity
Drug effects on behavior in mice, ambulatory activity (expressed as distance traveled in cm/10 min), were assessed in open field cages, clear acrylic testing chambers (40 cm3) (Med Associates, St. Albans, VT). Chambers were equipped with infrared light sources directed at light sensitive detectors (mounted on the opposing walls), spaced 2.5 cm apart along two perpendicular walls. Activity counts were recorded each time the subject interrupted a single light beam. Mice were transferred from the animal facility vivarium to the behavioral activity testing room about one to two hours before the test. Mice were injected i.p. with test compounds or vehicle and immediately placed into the testing chamber. For each animal and dose of compound, behavioral activity counts were summed in 10 minutes bins and then averaged to obtain the group mean and corresponding SEM. Data of each drug and vehicle were analyzed by two-way repeated measure ANOVA with “Dose” and “Time” as independent factors (Prism 7, GraphPad software, La Jolla, CA, USA) (SI table 8). Dunnett’s post-hoc test was employed to compare the activity observed after doses of test compounds with that after vehicle; q values indicate the difference of the two means divided by the standard error of that difference. Details of statistical analysis that indicated a significant drug-induced stimulation of behavior were reported. Dose-effect curves were obtained plotting the value of the bin with maximal activity counts as function of the dose of drug administered. Group size was at least six subjects for all groups, except for the group administered with the lowest cocaine dose (3 mg/kg, n=4). Mice were used only once.
Supplementary Material
Acknowledgements.
Support for this research was provided by the National Institute on Drug Abuse - Intramural Research Program, NIH/DHHS (Z1A DA000389 and Z1A DA000611).
List of nonstandard abbreviations:
- ANOVA
analysis of variance
- CFME
carbon-fiber microelectrode
- DA
dopamine
- DAT
dopamine transporter
- FSCV
fast scan cyclic voltammetry
- i.p.
intraperitoneally
- i.v.
intravenously
- MOD
modafinil
- NS
non-significant
- NAS
nucleus accumbens shell
- R-MOD
(R)-modafinil
- SEM
standard error of the mean
- SAR
Structure Activity Relationships
Footnotes
Conflict of interest
AHN and JJC are inventors on the licensed US patent E-073-2013/0-US-06 - NIH0072US2 in which JJC8-088 and JJC8-091 are disclosed. The NIH owns all rights to this patent. All authors declare that there are no competing financial interests of any kind in relation to the work described in the present manuscript.
Supporting Information: Absolute baseline values for DA clearance rate and DAMAX; Statistical analysis.
REFERENCES
- 1.German CL, Baladi MG, McFadden LM, Hanson GR, and Fleckenstein AE (2015) Regulation of the Dopamine and Vesicular Monoamine Transporters: Pharmacological Targets and Implications for Disease, Pharmacol Rev 67, 1005–1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Salatino-Oliveira A, Rohde LA, and Hutz MH (2018) The dopamine transporter role in psychiatric phenotypes, Am J Med Genet B Neuropsychiatr Genet 177, 211–231. [DOI] [PubMed] [Google Scholar]
- 3.Sulzer D, Cragg SJ, and Rice ME (2016) Striatal dopamine neurotransmission: regulation of release and uptake, Basal Ganglia 6, 123–148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pontieri FE, Tanda G, and Di Chiara G (1995) Intravenous cocaine, morphine, and amphetamine preferentially increase extracellular dopamine in the “shell” as compared with the “core” of the rat nucleus accumbens, Proc Natl Acad Sci U S A 92, 12304–12308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kuhar MJ, Ritz MC, and Boja JW (1991) The dopamine hypothesis of the reinforcing properties of cocaine, Trends Neurosci 14, 299–302. [DOI] [PubMed] [Google Scholar]
- 6.Ritz MC, Boja JW, George FR, and Kuhar MJ (1989a) Cocaine binding sites related to drug self-administration, NIDA Res Monogr 95, 239–246. [PubMed] [Google Scholar]
- 7.Reith ME, Blough BE, Hong WC, Jones KT, Schmitt KC, Baumann MH, Partilla JS, Rothman RB, and Katz JL (2015) Behavioral, biological, and chemical perspectives on atypical agents targeting the dopamine transporter, Drug Alcohol Depend 147, 1–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tanda G, Newman AH, and Katz JL (2009) Discovery of drugs to treat cocaine dependence: behavioral and neurochemical effects of atypical dopamine transport inhibitors, Adv Pharmacol 57, 253–289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Beuming T, Kniazeff J, Bergmann ML, Shi L, Gracia L, Raniszewska K, Newman AH, Javitch JA, Weinstein H, Gether U, and Loland CJ (2008) The binding sites for cocaine and dopamine in the dopamine transporter overlap, Nat Neurosci 11, 780–789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Loland CJ, Mereu M, Okunola OM, Cao J, Prisinzano TE, Mazier S, Kopajtic T, Shi L, Katz JL, Tanda G, and Newman AH (2012) R-modafinil (armodafinil): a unique dopamine uptake inhibitor and potential medication for psychostimulant abuse, Biol Psychiatry 72, 405–413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Loland CJ, Desai RI, Zou MF, Cao J, Grundt P, Gerstbrein K, Sitte HH, Newman AH, Katz JL, and Gether U (2008) Relationship between conformational changes in the dopamine transporter and cocaine-like subjective effects of uptake inhibitors, Mol Pharmacol 73, 813–823. [DOI] [PubMed] [Google Scholar]
- 12.Schmitt KC, and Reith ME (2011) The atypical stimulant and nootropic modafinil interacts with the dopamine transporter in a different manner than classical cocaine-like inhibitors, PLoS One 6, e25790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kohut SJ, Hiranita T, Hong SK, Ebbs AL, Tronci V, Green J, Garces-Ramirez L, Chun LE, Mereu M, Newman AH, Katz JL, and Tanda G (2014) Preference for distinct functional conformations of the dopamine transporter alters the relationship between subjective effects of cocaine and stimulation of mesolimbic dopamine, Biol Psychiatry 76, 802–809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tanda G, Li SM, Mereu M, Thomas AM, Ebbs AL, Chun LE, Tronci V, Green JL, Zou MF, Kopajtic TA, Newman AH, and Katz JL (2013) Relations between stimulation of mesolimbic dopamine and place conditioning in rats produced by cocaine or drugs that are tolerant to dopamine transporter conformational change, Psychopharmacology (Berl) 229, 307–321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tanda G, Newman AH, Ebbs AL, Tronci V, Green JL, Tallarida RJ, and Katz JL (2009) Combinations of cocaine with other dopamine uptake inhibitors: assessment of additivity, J Pharmacol Exp Ther 330, 802–809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mereu M, Bonci A, Newman AH, and Tanda G (2013) The neurobiology of modafinil as an enhancer of cognitive performance and a potential treatment for substance use disorders, Psychopharmacology (Berl) 229, 415–434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rodrigues TM, Castro Caldas A, and Ferreira JJ (2016) Pharmacological interventions for daytime sleepiness and sleep disorders in Parkinson’s disease: Systematic review and meta-analysis, Parkinsonism Relat Disord 27, 25–34. [DOI] [PubMed] [Google Scholar]
- 18.Krishnan R, and Chary KV (2015) A rare case modafinil dependence, J Pharmacol Pharmacother 6, 49–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ozturk A, and Deveci E (2014) Drug Abuse of Modafinil by a Cannabis User, Bulletin of Clinical Psychopharmacology 24, 405–407. [Google Scholar]
- 20.Mereu M, Chun LE, Prisinzano TE, Newman AH, Katz JL, and Tanda G (2017) The unique psychostimulant profile of (+/−)-modafinil: investigation of behavioral and neurochemical effects in mice, Eur J Neurosci 45, 167–174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dackis CA, Lynch KG, Yu E, Samaha FF, Kampman KM, Cornish JW, Rowan A, Poole S, White L, and O’Brien CP (2003) Modafinil and cocaine: a double-blind, placebo-controlled drug interaction study, Drug Alcohol Depend 70, 29–37. [DOI] [PubMed] [Google Scholar]
- 22.Hart CL, Haney M, Vosburg SK, Rubin E, and Foltin RW (2008) Smoked cocaine self-administration is decreased by modafinil, Neuropsychopharmacology 33, 761–768. [DOI] [PubMed] [Google Scholar]
- 23.Mahoney JJ, Jackson BJ, Kalechstein AD, De La Garza R, Chang LC, and Newton TF (2012) Acute modafinil exposure reduces daytime sleepiness in abstinent methamphetamine-dependent volunteers, Int J Neuropsychopharmacol 15, 1241–1249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Anderson AL, Li SH, Biswas K, McSherry F, Holmes T, Iturriaga E, Kahn R, Chiang N, Beresford T, Campbell J, Haning W, Mawhinney J, McCann M, Rawson R, Stock C, Weis D, Yu E, and Elkashef AM (2012) Modafinil for the treatment of methamphetamine dependence, Drug Alcohol Depend 120, 135–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kampman KM, Lynch KG, Pettinati HM, Spratt K, Wierzbicki MR, Dackis C, and O’Brien CP (2015) A double blind, placebo controlled trial of modafinil for the treatment of cocaine dependence without co-morbid alcohol dependence, Drug Alcohol Depend 155, 105–110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Okunola-Bakare OM, Cao J, Kopajtic T, Katz JL, Loland CJ, Shi L, and Newman AH (2014) Elucidation of structural elements for selectivity across monoamine transporters: novel 2-[(diphenylmethyl)sulfinyl]acetamide (modafinil) analogues, J Med Chem 57, 1000–1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cao J, Slack RD, Bakare OM, Burzynski C, Rais R, Slusher BS, Kopajtic T, Bonifazi A, Ellenberger MP, Yano H, He Y, Bi GH, Xi ZX, Loland CJ, and Newman AH (2016) Novel and High Affinity 2-[(Diphenylmethyl)sulfinyl]acetamide (Modafinil) Analogues as Atypical Dopamine Transporter Inhibitors, J Med Chem 59, 10676–10691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cao J, Prisinzano TE, Okunola OM, Kopajtic T, Shook M, Katz JL, and Newman AH (2011) Structure-Activity Relationships at the Monoamine Transporters for a Novel Series of Modafinil (2-[(diphenylmethyl)sulfinyl]acetamide) Analogues, ACS Med Chem Lett 2, 48–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tunstall BJ, Ho CP, Cao J, Vendruscolo JCM, Schmeichel BE, Slack RD, Tanda G, Gadiano AJ, Rais R, Slusher BS, Koob GF, Newman AH, and Vendruscolo LF (2018) Atypical dopamine transporter inhibitors attenuate compulsive-like methamphetamine self-administration in rats, Neuropharmacology 131, 96–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Zhang HY, Bi GH, Yang HJ, He Y, Xue G, Cao J, Tanda G, Gardner EL, Newman AH, and Xi ZX (2017) The Novel Modafinil Analog, JJC8-016, as a Potential Cocaine Abuse Pharmacotherapeutic, Neuropsychopharmacology 42, 1871–1883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Aragona BJ, Cleaveland NA, Stuber GD, Day JJ, Carelli RM, and Wightman RM (2008) Preferential enhancement of dopamine transmission within the nucleus accumbens shell by cocaine is attributable to a direct increase in phasic dopamine release events, J Neurosci 28, 8821–8831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Owesson-White CA, Ariansen J, Stuber GD, Cleaveland NA, Cheer JF, Wightman RM, and Carelli RM (2009) Neural encoding of cocaine-seeking behavior is coincident with phasic dopamine release in the accumbens core and shell, Eur J Neurosci 30, 1117–1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Greco PG, Meisel RL, Heidenreich BA, and Garris PA (2006) Voltammetric measurement of electrically evoked dopamine levels in the striatum of the anesthetized Syrian hamster, J Neurosci Methods 152, 55–64. [DOI] [PubMed] [Google Scholar]
- 34.Hoffman AF, Spivak CE, and Lupica CR (2016) Enhanced Dopamine Release by Dopamine Transport Inhibitors Described by a Restricted Diffusion Model and Fast-Scan Cyclic Voltammetry, ACS Chem Neurosci 7, 700–709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yorgason JT, Jones SR, and Espana RA (2011) Low and high affinity dopamine transporter inhibitors block dopamine uptake within 5 sec of intravenous injection, Neuroscience 182, 125–132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sabeti J, Adams CE, Burmeister J, Gerhardt GA, and Zahniser NR (2002) Kinetic analysis of striatal clearance of exogenous dopamine recorded by chronoamperometry in freely-moving rats, J Neurosci Methods 121, 41–52. [DOI] [PubMed] [Google Scholar]
- 37.Oleson EB, Salek J, Bonin KD, Jones SR, and Budygin EA (2009) Real-time voltammetric detection of cocaine-induced dopamine changes in the striatum of freely moving mice, Neurosci Lett 467, 144–146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pattison LP, Bonin KD, Hemby SE, and Budygin EA (2011) Speedball induced changes in electrically stimulated dopamine overflow in rat nucleus accumbens, Neuropharmacology 60, 312–317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Venton BJ, Seipel AT, Phillips PE, Wetsel WC, Gitler D, Greengard P, Augustine GJ, and Wightman RM (2006) Cocaine increases dopamine release by mobilization of a synapsin-dependent reserve pool, J Neurosci 26, 3206–3209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Budygin EA, Kilpatrick MR, Gainetdinov RR, and Wightman RM (2000) Correlation between behavior and extracellular dopamine levels in rat striatum: comparison of microdialysis and fast-scan cyclic voltammetry, Neurosci Lett 281, 9–12. [DOI] [PubMed] [Google Scholar]
- 41.Jones SR, Gainetdinov RR, and Caron MG (1999) Application of microdialysis and voltammetry to assess dopamine functions in genetically altered mice: correlation with locomotor activity, Psychopharmacology (Berl) 147, 30–32. [DOI] [PubMed] [Google Scholar]
- 42.Suto N, Ecke LE, You ZB, and Wise RA (2010) Extracellular fluctuations of dopamine and glutamate in the nucleus accumbens core and shell associated with lever-pressing during cocaine self-administration, extinction, and yoked cocaine administration, Psychopharmacology (Berl) 211, 267–275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Stuber GD, Roitman MF, Phillips PE, Carelli RM, and Wightman RM (2005) Rapid dopamine signaling in the nucleus accumbens during contingent and noncontingent cocaine administration, Neuropsychopharmacology 30, 853–863. [DOI] [PubMed] [Google Scholar]
- 44.Heien ML, Khan AS, Ariansen JL, Cheer JF, Phillips PE, Wassum KM, and Wightman RM (2005) Real-time measurement of dopamine fluctuations after cocaine in the brain of behaving rats, Proc Natl Acad Sci U S A 102, 10023–10028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cacciapaglia F, Saddoris MP, Wightman RM, and Carelli RM (2012) Differential dopamine release dynamics in the nucleus accumbens core and shell track distinct aspects of goal-directed behavior for sucrose, Neuropharmacology 62, 2050–2056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Stuber GD, Wightman RM, and Carelli RM (2005) Extinction of cocaine self-administration reveals functionally and temporally distinct dopaminergic signals in the nucleus accumbens, Neuron 46, 661–669. [DOI] [PubMed] [Google Scholar]
- 47.Grace AA, and Bunney BS (1984) The control of firing pattern in nigral dopamine neurons: burst firing, J Neurosci 4, 2877–2890. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Grace AA, and Bunney BS (1984) The control of firing pattern in nigral dopamine neurons: single spike firing, J Neurosci 4, 2866–2876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Owesson-White CA, Roitman MF, Sombers LA, Belle AM, Keithley RB, Peele JL, Carelli RM, and Wightman RM (2012) Sources contributing to the average extracellular concentration of dopamine in the nucleus accumbens, J Neurochem 121, 252–262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jaquins-Gerstl A, and Michael AC (2015) A review of the effects of FSCV and microdialysis measurements on dopamine release in the surrounding tissue, Analyst 140, 3696–3708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Avelar AJ, Cao J, Newman AH, and Beckstead MJ (2017) Atypical dopamine transporter inhibitors R-modafinil and JHW 007 differentially affect D2 autoreceptor neurotransmission and the firing rate of midbrain dopamine neurons, Neuropharmacology 123, 410–419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hiranita T, Soto PL, Kohut SJ, Kopajtic T, Cao J, Newman AH, Tanda G, and Katz JL (2011) Decreases in cocaine self-administration with dual inhibition of the dopamine transporter and sigma receptors, J Pharmacol Exp Ther 339, 662–677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Katz JL, Su TP, Hiranita T, Hayashi T, Tanda G, Kopajtic T, and Tsai SY (2011) A Role for Sigma Receptors in Stimulant Self Administration and Addiction, Pharmaceuticals (Basel) 4, 880–914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Garces-Ramirez L, Green JL, Hiranita T, Kopajtic TA, Mereu M, Thomas AM, Mesangeau C, Narayanan S, McCurdy CR, Katz JL, and Tanda G (2011) Sigma receptor agonists: receptor binding and effects on mesolimbic dopamine neurotransmission assessed by microdialysis, Biol Psychiatry 69, 208–217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Kiyatkin EA, and Brown PL (2007) I.v. cocaine induces rapid, transient excitation of striatal neurons via its action on peripheral neural elements: single-cell, iontophoretic study in awake and anesthetized rats, Neuroscience 148, 978–995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Kiyatkin EA, and Rebec GV (2000) Dopamine-independent action of cocaine on striatal and accumbal neurons, Eur J Neurosci 12, 1789–1800. [DOI] [PubMed] [Google Scholar]
- 57.Desai RI, Kopajtic TA, French D, Newman AH, and Katz JL (2005) Relationship between in vivo occupancy at the dopamine transporter and behavioral effects of cocaine, GBR 12909 [1-{2-[bis-(4-fluorophenyl)methoxy]ethyl}−4-(3-phenylpropyl)piperazine], and benztropine analogs, J Pharmacol Exp Ther 315, 397–404. [DOI] [PubMed] [Google Scholar]
- 58.Prisinzano TE, Tidgewell K, Podobinski J, Luo M, and Swenson D (2004) Synthesis and determination of the absolute configuration of the enantiomers of modafinil., Tetrahedron-Asymmetr 15, 1053–1058. [Google Scholar]
- 59.Paxinos G, and Franklin KBJ (2001) The mouse brain in stereotaxic coordinates, Second ed., Academic Press, New York, New York. [Google Scholar]
- 60.Huffman ML, and Venton BJ (2009) Carbon-fiber microelectrodes for in vivo applications, Analyst 134, 18–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cheer JF, Wassum KM, Heien ML, Phillips PE, and Wightman RM (2004) Cannabinoids enhance subsecond dopamine release in the nucleus accumbens of awake rats, J Neurosci 24, 4393–4400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Bucher ES, Brooks K, Verber MD, Keithley RB, Owesson-White C, Carroll S, Takmakov P, McKinney CJ, and Wightman RM (2013) Flexible software platform for fast-scan cyclic voltammetry data acquisition and analysis, Anal Chem 85, 10344–10353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Berglund EC, Makos MA, Keighron JD, Phan N, Heien ML, and Ewing AG (2013) Oral administration of methylphenidate blocks the effect of cocaine on uptake at the Drosophila dopamine transporter, ACS Chem Neurosci 4, 566–574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Mereu M, Tronci V, Chun LE, Thomas AM, Green JL, Katz JL, and Tanda G (2015) Cocaine-induced endocannabinoid release modulates behavioral and neurochemical sensitization in mice, Addict Biol 20, 91–103. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


