Abstract
Pattern separation, the ability to differentiate new information from previously experienced similar information, is highly sensitive to hippocampal structure and function and declines with age. Functional MRI studies have demonstrated hippocampal hyperactivation in older adults compared to young, with greater task-related activation associated with worse pattern separation performance. The current study was designed to determine whether pattern separation was sensitive to differences in task-free hippocampal cerebral blood flow (CBF) in 130 functionally intact older adults. Given prior evidence that apolipoprotein E e4 (APOE e4) status moderates the relationship between CBF and episodic memory, we predicted a stronger negative relationship between hippocampal CBF and pattern separation in APOE e4 carriers. An interaction between APOE group and right hippocampal CBF was present, such that greater right hippocampal CBF was related to better lure discrimination in non-carriers, whereas the effect reversed directionality in e4 carriers. These findings suggest that neurovascular changes in the medial temporal lobe may underlie memory deficits in cognitively normal older adults who are APOE e4 carriers.
Keywords: APOE, ASL perfusion, hippocampus, pattern separation
1 |. INTRODUCTION
Aging is associated with a decline in some aspects of episodic memory, including pattern separation, which represents the ability to differentiate new information from previously encountered similar information. That is, older adults require a greater degree of dissimilarity between items before new information is correctly differentiated from previously encountered similar information (Stark et al., 2019; Yassa et al., 2011), increasing false recognition of novel stimuli. Pattern separation relies heavily on the hippocampus and more extensive medial temporal regions (Bakker et al., 2008; Stevenson et al., 2020), which decline in structure and function during aging (Bettio et al., 2017; Lafosse et al., 1997; Raz et al., 2005; Raz et al., 2010; Wilson et al., 2006).
Computational models of pattern separation suggest that overlapping afferent inputs from entorhinal cortex are differentiated by granule cells of the dentate gyrus (DG) and projected via mossy fibers to the CA3 subfield of the hippocampus (Kassab & Alexandre, 2018; Yassa et al., 2011). The granule cells of DG have low firing rates, with only 1–2% exhibiting activity, and are thought to sparsely recode entorhinal cortex inputs in order to create distinct representations for similar but novel inputs (Chawla et al., 2005; Kassab & Alexandre, 2018). The CA3 receives additional input directly from layer II of entorhinal cortex (perforant pathway) and recurrent collateral input from CA3 neurons (Amaral et al., 2007; Witter, 1993). The recurrent collateral network of the CA3 is thought to function as an auto-associative pattern completion network biasing the system to retrieve a preexisting representation (old memory) rather than encoding new information when presented with a partial or degraded cue. Depending on inputs from DG and ERC, CA3 activity can result in either pattern separation or completion (Lee & Kesner, 2004; Leutgeb et al., 2007; Vazdarjanova & Guzowski, 2004). In rats, aging is associated with “rigid” and elevated place cell firing in the CA3 and a failure to distinguish between similar environments (Wilson et al., 2005; Wilson et al., 2006). Further, electrophysiological studies identified reduced excitatory presynaptic fiber potentials at the perforant path-DG synapse (Barnes et al., 2000; Dieguez & Barea-Rodriguez, 2004) and postsynaptic potentials in DG (Barnes, 1979; Barnes & McNaughton, 1980) in aged rats, potentially biasing the response in CA3 toward pattern completion.
Task-based fMRI studies comparing functional activation patterns during pattern separation performance between young and older adults identified greater right hippocampal activation in the CA3/DG subfields of older adults compared to young during successful pattern separation (Yassa et al., 2011). This “hyperactivation” was present during both the initial encoding of subsequently differentiated objects (e.g., items subsequently tested with a lure), and during successful discrimination of lures from previously viewed similar objects. Further, greater right CA3/DG activity during correct rejection of lures was associated with worse pattern separation performance. These findings were considered in light of prior work by Miller et al. (2008) in healthy older adults that demonstrated greater right hippocampal activity in low performing older adults compared to high performing older adults or young adults during successful encoding of face-name pairs (Miller et al., 2008). Taken together, hippocampal hyperactivation was interpreted as an indicator of medial temporal lobe network dysfunction, or at least an inefficient or faulty compensatory mechanism.
Additional support for hyperactivation as an indicator of network dysfunction stems from studies of those at increased risk for neurodegenerative disorders, including carriers of the apolipoprotein E e4 (APOE e4) allele (Bookheimer et al., 2000; Dennis et al., 2010; Filippini et al., 2009; Sinha et al., 2018; Trivedi et al., 2008), and individuals with amnestic mild cognitive impairment (Miller et al., 2008; O’Brien et al., 2010; Putcha et al., 2011; Tran et al., 2017; Yassa et al., 2010). Across these studies, greater resting and task-related hippocampal activation was associated with worse performance on measures of pattern separation and episodic memory. Furthermore, Bakker et al. (2012) demonstrated an improvement in memory performance in individuals with amnestic mild cognitive impairment when hippocampal activation was dampened through the administration of an antiepileptic medication (Bakker et al., 2012).
While the relationship between hippocampal hyperactivation and memory performance is well established, less is known about the impact of hippocampal perfusion on memory performance during healthy aging. The fMRI blood-oxygen-level-dependent (BOLD) signal serves as a useful proxy for neural activity. Task-based fMRI studies relying on the BOLD signal have proven particularly valuable for understanding the unique processes (e.g., pattern separation vs. pattern completion) subserved by hippocampal subfields (Bakker et al., 2008; Berron et al., 2016; Lacy et al., 2011; Yassa et al., 2010). However, the BOLD signal stems from a complex interplay of physiological factors, including changes in cerebral blood flow (CBF), blood volume and metabolic rate of oxygen uptake associated with neural activity, and can only be expressed by means of percent signal change. This leaves questions as to which physiological component of the BOLD signal accounts for hippocampal “hyperactivation” in at-risk groups, and whether these differences reflect changes in the neural vasculature or metabolism at rest, increased neural engagement relative to the same baseline during task completion, or some combination of these alternatives. ASL perfusion complements fMRI BOLD studies by providing a quantifiable measure of the rate of arterial blood flow to a capillary bed in a specific volume of tissue (Petcharunpaisan et al., 2010)—a more direct measure of neurovascular function. Therefore, ASL perfusion can address whether or not changes in the rate of blood flow to the hippocampus at rest contribute to the complex interplay of responses that produce BOLD “hyperactivation” during mnemonic discrimination tasks. Age is associated with global reductions in CBF (Bentourkia et al., 2000; Parkes et al., 2004; Staffaroni et al., 2019), with average declines in gray matter perfusion at a rate of around 0.45% annually in healthy adults aged 20–67 (Clement et al., 2018; Parkes et al., 2004), and the greatest reductions in older age groups (Chen et al., 2011).
Focusing on hippocampal CBF, Heo et al. (2010) identified age-related reductions in perfusion (using flow-enhanced signal intensity imaging) that were associated with slower reaction times, but not accuracy, on a spatial memory task (Heo et al., 2010). Other studies examining age effects of medial temporal perfusion on memory performance using ASL perfusion have focused on cognitively normal older adults at increased risk for Alzheimer’s disease. Among these studies, Bangen et al. (2012) identified greater resting CBF in the medial temporal lobes (hippocampus and parahippocampal cortex) in APOE e4 carriers compared to noncarriers (Bangen et al., 2012). In a later study, their group identified an interaction between CBF and amyloid-β accumulation, such that greater CBF in the hippocampus, posterior cingulate, and precuneus (core regions affected by AD pathology) was associated with poorer verbal memory amongst Aβ positive but not Aβ negative older adults (Bangen et al., 2017). Similar findings have been observed in those at increased genetic risk for Alzheimer’s disease, with an association between greater hippocampal blood flow and worse verbal memory in APOE e4 carriers, but not noncarriers (Hays et al., 2019). Paralleling fMRI findings, greater perfusion of the hippocampus was associated with worse memory performance, specifically in cognitively normal older adults at increased risk for Alzheimer’s disease. Together, these studies provide converging evidence that greater blood flow and metabolic activity in the hippocampus are associated with worse cognition and may serve as early indicators of individuals at increased risk for memory decline.
The aim of the current study was to characterize the contribution of hippocampal blood flow to pattern separation performance, and determine whether genetic risk for Alzheimer’s disease moderates this relationship. In order to do so, we relied on a visual mnemonic similarity task that is highly sensitive to hippocampal structure and function (Stark et al., 2019; Yassa & Stark, 2011), particularly in the CA3/DG subfields. This measure was specifically designed to tax hippocampal pattern separation processes and may contribute to more refined early detection of cognitively normal older adults at risk for cognitive decline. Based on prior associations between hippocampal hyperactivation and pattern separation performance (Yassa et al., 2011), we predicted an inverse relationship between hippocampal perfusion and the critical metric of pattern separation performance, the lure discrimination index (LDI). Further, we predicted that genetic risk for Alzheimer’s disease, conferred through the presence of at least one APOE e4 allele, would interact with hippocampal blood flow to impact LDI, such that a negative relationship would be more pronounced in e4 carriers compared to noncarriers. Given prior lateralizing findings with right hippocampus exhibiting age-related hyperactivation, we examined left and right hippocampal CBF separately, and predicted a stronger association between right hippocampal CBF and LDI.
2 |. METHODS
2.1 |. Participants
Participants were recruited at the University of California San Francisco’s (UCSF) Memory and Aging Center as part of the Hillblom Aging Network. All participants provided written informed consent and the study was approved by the UCSF Committee on Human Research. Participants completed neuroimaging, neuropsychological testing, neurological examination, and select genetic testing. Participants were characterized as clinically and functionally intact based on consensus conference in which neurological and neuropsychological findings were reviewed by a neurologist and neuropsychologist. From this cohort, we included all participants who completed the mnemonic similarity task and an arterial spin labeling MRI scan. This resulted in 130 older adults (60 females, 70 males) with a mean age of 75 (SD = 6.2; range = 58–92 years) and 18 years of education (SD = 2.1; range = 12–20). Of the 130 participants, 36 were carriers of the APOE e4 allele and 94 were noncarriers (see Table 1). The sample was 88% white/Caucasian, 4% Chinese, 2% Japanese, 2% other races, and <1% Asian Indian and Filipino.
TABLE 1.
Participant demographics, cognition, neuroimaging and health characteristics
| e4 carriers (N = 36) Means (SD) | Noncarriers (N = 94) | t-Statistic/chi-square | Confidence interval | |
|---|---|---|---|---|
| Age | 74.20 (5.20) | 75.42 (6.47) | −1.02 | [−3.61, 1.16] |
| Sex (M/F) | 19/17 | 41/53 | 0.88 | – |
| Education | 17.64 (2.28) | 17.48 (2.09) | 0.38 | [−0.67, 0.99] |
| Precentral CBF | 21.67 (6.05) | 20.33 (6.46) | 1.08 | [−1.12, 3.81] |
| Left | 21.89 (6.57) | 20.78 (7.04) | 0.82 | [−1.57, 3.79] |
| Right | 21.46 (6.02) | 19.88 (6.40) | 1.28 | [−0.86, 4.02] |
| Hippocampal CBF | 27.36 (6.80) | 25.96 (6.37) | 1.11 | [−1.11, 3.92] |
| Left | 26.75 (6.93) | 25.46 (6.14) | 1.03 | [−1.18, 3.76] |
| Right | 27.97 (7.02) | 26.45 (7.41) | 1.06 | [−1.31, 4.35] |
| BMI | 25.32 (3.71) | 25.43 (4.76) | −0.12 | [−1.94, 1.72] |
| Diastolic BP | 71.91 (10.06) | 73.54 (7.76) | −0.95 | [−5.05, 1.79] |
| Systolic BP | 133.49 (17.96) | 133.39 (17.88) | −0.61 | [−9.06, 4.81] |
| MMSE | 28.73 (1.33) | 28.66 (1.50) | 0.22 | [−0.52, 0.65] |
Note: Carriers and noncarriers did not differ on any of these variables (p’s > .05).
Abbreviation: CBF, cerebral blood flow.
2.2 |. Mnemonic similarity task
Participants completed a version of the mnemonic similarity task described in Stark et al. (2013). During incidental encoding, participants viewed pictures of 80 common objects. In order to enhance encoding/attention, they were asked to judge whether the objects were most commonly found indoors or outdoors. Immediately following encoding, they were shown 40 of the studied objects, 40 novel foil objects, and 40 lure objects that were similar but not identical to studied objects. Participants were asked to classify items as “old,” “similar” or “new.” The critical metric of pattern separation is the ability to discriminate similar lure items from old items. In order to account for response biases, the LDI was calculated, which is the difference between the probability of giving a “similar” response to lure items and the probability of giving a “similar” response to a new item.
2.3 |. APOE genotyping
Genomic DNA was extracted from peripheral blood using standard protocols (Gentra PureGene Blood Kit, Qiagen). Genotyping was performed using either TaqMan or Sequenom genotyping. TaqMan Allelic Discrimination Assay was used for APOE genotyping (rs429358 and rs7412), and was conducted on an ABI 7900HT Fast Real-Time PCR system (Applied Biosystems) according to the manufacturer’s instructions. The Spectro Aquire and MassARRAY Typer Software packages (Sequenom) were used for interpretation and Typer analyzer (v3.4.0.18) was used to review and analyze data.
2.4 |. Neuroimaging
2.4.1 |. Scanner
Scanner magnetic resonance imaging data were acquired on a 3 T Trio scanner (Siemens Medical Systems, Erlangen, Germany). Volumetric MPRAGE sequences at UCSF was used to acquire T1-weighted images of the entire brain (Sagittal slice orientation; slice thickness = 1.0 mm; slices per slab = 160; in-plane resolution = 1.0 × 1.0 mm; matrix = 240 × 256; TR = 2300 ms; TE = 2.98 ms; TI = 900 ms; flip angle = 9). Pulsed ASL imaging was acquired using QUIPSSII with Thin-slice TI1 Periodic Saturation sequence incorporated in a proximal inversion with control of off-resonance effects labeling scheme (Luh et al., 1999). The periodic saturation pulses started at the post labeling delay inversion time TI1 = 700 ms after the in-plan pre saturation radio infrequency pulse; the readout started at the post labeling delay inversion time TI2 = 1800 ms. The repetition and echo time were TR/TE = 2500/11 ms. We acquired 16 slices, each 6 mm thick with a 7.2 mm center to center distance and a matrix 64 × 56 of 4 × 4 mm2 in-plane voxel resolution.
2.4.2 |. ASL perfusion
ASL data were processed to obtain partial volume corrected (PVC) maps of gray matter perfusion as previously described (Du et al., 2006; Hayasaka et al., 2006; Johnson et al., 2005). Frames of the ASL acquisition were corrected for motion and co-registered with the first frame (M0) using FSL (Jenkinson et al., 2012). The effect of motion contamination was reduced by applying DVAR algorithm (Tanenbaum et al., 2015). Differential perfusion images were created by subtracting unlabeled from adjacent labeled frames and averaging these subtraction images (Aguirre et al., 2002). Susceptibility artifacts along the phase-encoding direction were corrected in the M0 frame and perfusion map using ANTs SyN (Avants et al., 2008) restricted to the coronal axis. An automatic quality control process removed tagged/untagged pair of frames when the relative root mean square (RMS) distance value between two consecutive frames was higher than 0.5 mm. The participant was dropped if this RMS value was higher than 1 mm. CBF was calculated by applying the Buxton kinetic model to the perfusion map (Buxton et al., 1998; Wang et al., 2003). Partial volume correction was based on the tissue segmentation maps from MPRAGE using the transformation matrix from T1 to M0 (Du et al., 2006; Müller-Gärtner et al., 1992).
All CBF images were visually inspected in the native and study-specific template; analyses were conducted in a study-specific template. Finally, for the purposes of visualization, images were transformed into MNI space to verify anatomical localization because most standard atlases and templates are in MNI space. Poor quality images that were out of the field of view, or contained large susceptibility or motion artifacts were removed from the study (N = 12). Ten participants were excluded due to motion artifacts, and three participants were excluded due to images out of the field of view (two inferior temporal cutoff, one inferior occipital cutoff). Twenty-three percent of the excluded participants were e4 carriers, consistent with the percentage of e4 carriers in the included sample (21%).
The CBF maps were PVC in the gray matter at each time point in their native space. After applying the structural registration transformations to the CBF PVC maps, the maps were masked using voxels from the group template with ≥50% probability of being gray matter; cerebellar voxels were excluded. A global CBF value was calculated that averaged all voxels in this mask. Hippocampal and precentral gyrus ROIs were selected after registering the Desikan anatomically parcellated brain atlas (Desikan et al., 2006) to our group template. The ROI gray matter volume was obtained by integrating all the voxels from the gray matter probability map in a specific ROI; the CBF was calculated by averaging the values of all the voxels from the CBF map in a specific ROI (see Figure S1, supplementary materials). Hippocampal and precentral gyrus ROIs were examined separately by hemisphere, based on prior evidence of hemispheric differences in pattern separation effects (Miller et al., 2008; Yassa et al., 2011).
2.5 |. Statistical analyses
Linear regressions were conducted using SPSS PROCESS (Hayes, 2012) to examine the effects of hippocampal CBF, APOE e4 status, and the interaction between hippocampal CBF and APOE e4 status on pattern separation performance (LDI). LDI was entered as the dependent variable, with age, sex, education, precentral gyrus CBF, hippocampal CBF, APOE status, and the interaction between hippocampal CBF and APOE status entered as predictors. All continuous independent variables were mean centered prior to analyses. Precentral gyrus CBF was used as a control region for CBF due to its relative resistance to neurodegenerative effects (for similar methods, see Bangen et al., 2017; Yew & Nation, 2017). Separate models were conducted to examine hemisphere-specific effects.
3 |. RESULTS
3.1 |. Behavioral results
Consistent with the performance of cognitively normal older adults in other samples (Sinha et al., 2018; Yassa et al., 2011), average lure discrimination was 0.24 (SD = 0.22) and ranged from −0.25 to 0.87 with higher scores indicating better discrimination. Participants successfully identified 33% (SD = 19) of the lure items as “similar,” 84% (SD = 14) of old items as “old,” and 86% (SD = 11) of foil items as “new.” Participants falsely classified foil items as “similar” on 10% (SD = 9) of the trials.
APOE groups did not statistically differ in performance across any of these pattern separation metrics (t-values <1.16, p-values >.25), including the critical metric of lure discrimination. While noncarriers identified 32% of the lure items as “similar,” e4 carriers identified 36%. False alarm rate (classifying similar items as “old”) was 55% in noncarriers compared to 54% in e4 carriers. Noncarriers classified 10% of foil items as “similar” compared to 9% in e4 carriers. Results are depicted in Figure 1.
FIGURE 1.
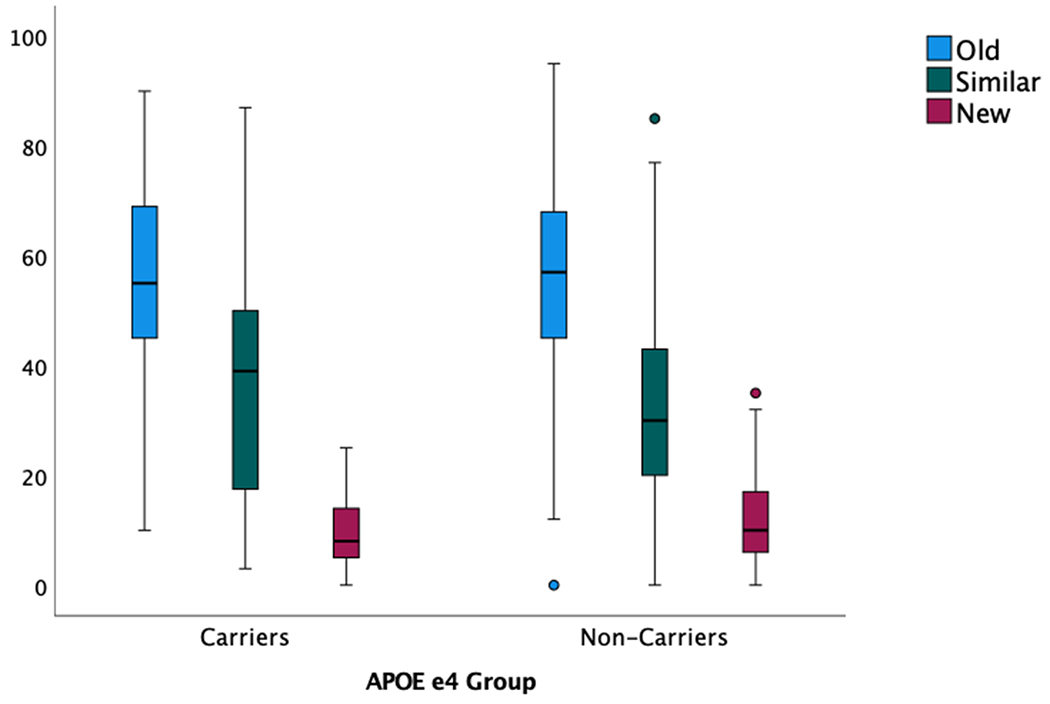
Behavioral results—Proportions of responses to similar objects (e.g., Old = proportion of similar objects classified as “old”) depicted separately for APOE groups. Performance did not statistically differ between groups [Color figure can be viewed at wileyonlinelibrary.com]
3.2 |. Hippocampal blood flow and LDI
In models examining the main effects of hippocampal CBF and APOE e4 status on LDI, controlling for age, sex, education, and precentral gyrus CBF, neither left nor right hippocampal CBF was associated with LDI (left: B = −0.17, SE = 0.36, p = .639, 95% CI [−0.88, 0.54]; right: B = 0.13, SE = 0.05, p = .643, 95% CI [−0.43, 0.70]). APOE e4 group was not associated with LDI (left: B = −5.30, SE = 4.13, p = .203, 95% CI [−13.47, 2.89]; right = B = −5.55, SE = 4.14, p = .183, 95% CI [−13.75, 2.65]). Older age was associated with worse LDI (left: B = −0.83, SE = 0.31, p = .008, 95% CI [−1.43, −0.22]; right: B = −0.81, SE = 0.30, p = .008, 95% CI [−1.41, −0.21]). Women exhibited better LDI than men (left: B = 9.18, SE = 3.92, p = .021, 95% CI [1.41, 16.94]; right: B = 10.49, SE = 4.02, p = .010, 95% CI [2.54, 18.44]). Blood flow in our control region, the precentral gyrus, was not associated with LDI (left: B = −0.12, SE = 0.34, p = .719, 95% CI [−0.79, 0.55]; right: B = −0.43, SE = 0.35, p = .215, 95% CI [−1.11, 0.25]).
3.3 |. Effect of APOE on the relationship between hippocampal CBF and pattern separation
In order to address our primary hypothesis regarding the interaction between APOE e4 status and hippocampal blood flow, the interaction term was added to the above models. The model examining the effect of right hippocampal CBF was significant, F(7,122) = 3.40, p = .002, and explained 16% of the variance in LDI (see Table 2). The interaction between APOE status and right hippocampal CBF was significant (B = 1.24, SE = 0.58, p = .035, 95% CI [0.091, 2.38]). The model examining the effect of left hippocampal CBF was significant, F(7,122) = 3.13, p = .005, and explained 15% of the variance in LDI (see Table 2). The interaction between APOE status and left hippocampal CBF was marginal (B = 1.17, SE = 0.61, p = .058, 95% CI [−0.04, 2.39]). The main effects of sex (right: B = 11.15, SE = 3.97, p = .005, 95% CI [3.28, 19.01]; left: B = 9.50, SE = 3.88, p = .016, 95% CI [1.81, 17.19]) and age (right: B = −0.82, SE = 0.30, p = .007, 95% CI [−1.41, −0.23]; left: B = −0.80, SE = 0.30, p = .009, 95% CI [−1.40, −0.20]) remained significant in both models.
TABLE 2.
Main and interaction effects of APOE e4 and CBF on pattern separation
| Variables | Coefficient (SE) | Standardized effects | p | LLCI | ULCI | |
|---|---|---|---|---|---|---|
| Right | Constant | 13.66 (8.05) | .092 | −2.27 | 29.59 | |
| Sex | 11.15 (3.97) | 0.25 | .006 | 3.28 | 19.01 | |
| Age | −0.82 (0.30) | −0.23 | .007 | −1.41 | −0.23 | |
| Education | 1.83 (0.89) | 0.18 | .043 | 0.06 | 3.59 | |
| Precentral CBF | −0.34 (0.34) | −0.10 | .329 | −1.02 | 0.34 | |
| Hippocampal CBF | 0.07 (0.28) | −0.28 | .808 | −0.49 | 0.63 | |
| APOE status (+ vs. −) | −6.38 (4.10) | −0.13 | .122 | −14.50 | 1.74 | |
| APOE × hippocampal CBF | 1.24 (0.58) | 0.36 | .035 | 0.09 | 2.38 | |
| Left | Constant | 12.25 (8.51) | .15 | −4.60 | 29.11 | |
| Sex | 9.50 (3.88) | 0.22 | .015 | 1.81 | 17.19 | |
| Age | −0.80 (0.30) | −0.23 | .009 | −1.40 | −0.20 | |
| Education | 1.65 (0.90) | 0.16 | .071 | −0.14 | 3.43 | |
| Precentral CBF | −0.14 (0.34) | −0.05 | .676 | −0.80 | 0.52 | |
| Hippocampal CBF | −0.10 (0.36) | −0.28 | .772 | −0.81 | 0.60 | |
| APOE status (+ vs. −) | −5.98 (4.10) | −0.12 | .148 | −14.10 | 2.15 | |
| APOE × hippocampal CBF | 1.17(0.61) | 0.28 | .058 | −0.04 | 2.39 | |
Note: Significant effects (p < .05) are shown in bold.
Abbreviation: CBF, cerebral blood flow.
In order to probe the directionality of the interaction, we examined the effect of right hippocampal CBF on LDI separately in APOE e4 carriers and noncarriers. Greater right hippocampal CBF was associated with better LDI in noncarriers (B = 0.41), but worse LDI in e4 carriers (B = −0.83) (see Figure 2). The directionality of the effects was similar for left hippocampus (noncarriers: B = 0.22; e4 carriers: B = −0.95).
FIGURE 2.

Scatterplot of the interaction between APOE status and right hippocampal blood flow on lure discrimination index. Dotted lines represent 95% confidence intervals [Color figure can be viewed at wileyonlinelibrary.com]
3.4 |. Post hoc analyses: Sex as a biological variable
Growing evidence indicates a more substantial negative impact of APOE e4 status on cognition (Wang et al., 2019), brain structure (Liu et al., 2019; Shen et al., 2019), and function (Damoiseaux et al., 2012; Sampedro et al., 2015) in female e4 carriers compared to males. In order to determine whether the interaction between hippocampal blood flow and APOE status differed based on sex, we stratified the sample by sex and evaluated the interactive effect between APOE status and hippocampal blood flow on LDI, separately by group. Effects were considered significant if they survived correction for multiple comparisons (p-values <.25). Supporting the vulnerability of female e4 carriers to negative effects on cognition and brain health, an interaction between APOE status and left hippocampal blood flow was observed in the female sample (B = 2.28, SE = 0.82, p = .007, 95% CI [0.63, 3.92]), but not the male sample (B = −0.30, SE = 0.94, p = .754, 95% CI [−2.19, 1.59]). In females, greater left hippocampal blood flow was associated with worse LDI in APOE e4 carriers (B = −1.66, SE = 0.74, p = .029, 95%% CI [−3.15, −0.18]), but not noncarriers (B = 0.61, SE = 0.56, p = .28, 95% CI [−0.51, 1.73]). The interaction between APOE status and right hippocampal blood flow did not reach significance in females (B = 1.64, SE = 0.82, p = .052, 95% CI [−0.01, 3.28]) or males (B = 0.84, SE = 0.89, p = .350, 95% CI [−0.95, 2.63]). Results represented in Figure 3.
FIGURE 3.

Interaction between APOE status and left hippocampal blood flow on lure discrimination index stratified by sex. Dotted lines represent 95% confidence intervals [Color figure can be viewed at wileyonlinelibrary.com]
4 |. DISCUSSION
Using a pattern separation task that is highly sensitive to hippocampal structure and function, the current study provided evidence that the association between hippocampal blood flow and pattern separation performance may be modified by APOE status. Specifically, greater right hippocampal blood flow was associated with worse LDI in e4 carriers, whereas the directionality of this effect was reversed in non-carriers, despite equivalent behavioral performance across groups. These findings are consistent with previous studies showing that APOE genotype (Hays et al., 2019) and amyloid burden (Bangen et al., 2017) moderate the effect of CBF on verbal memory performance. Taken together, these findings suggest that greater perfusion of the hippocampus is associated with worse memory, and may be an early indicator of neurovascular dysfunction in those at increased risk for Alzheimer’s disease.
The negative association between hippocampal resting blood flow and pattern separation performance in cognitively normal APOE e4 carriers may be indicative of subtle changes in hippocampal structure and function that predict future cognitive decline. Consistent with this interpretation, changes in frontal, parietal, and posterior temporal CBF have been linked to progression from normal cognition to cognitive impairment (Beason-Held et al., 2013) and from mild cognitive impairment to Alzheimer’s disease (Chao et al., 2010). Similar to the findings from our main effects model, hippocampal blood flow did not predict cognitive decline in the aforementioned studies. However, APOE interactions were not included in the models, potentially missing differences between groups. Given the effects observed in our at-risk but clinically normal cohort, the predictive utility of longitudinal changes in hippocampal blood flow, particularly in APOE e4 carriers, warrants further investigation in future works.
Negative relationships between hippocampal hyperactivity and episodic memory have been identified across a range of functional neuroimaging studies (Miller et al., 2008; Sinha et al., 2018; Yassa et al., 2011), particularly in those with cognitive impairment (Miller et al., 2008; O’Brien et al., 2010; Putcha et al., 2011; Tran et al., 2017; Yassa et al., 2010). However, only a handful of studies have identified associations between task-free quantification of hippocampal CBF and cognition, with mixed results. Although several studies have identified an inverse relationship between hippocampal blood flow and verbal memory (Bangen et al., 2017; Hays et al., 2019; Zlatar et al., 2016), positive relationships have also been identified. For example, Heo et al. (2010) found greater hippocampal blood flow was associated with faster reaction time on a spatial memory task in a sample of cognitively normal older adults. However, Heo et al. (2010) did not characterize participants based on Alzheimer’s disease risk factors, which our data and others suggest interact with perfusion to drive memory performance. Further, their spatial memory task was significantly easier than our mnemonic discrimination task, with an average accuracy of 80% compared to 33% on our critical measure of lure discrimination, potentially minimizing sensitivity to detect subtle differences in hippocampal structure and function.
Surprisingly, in our sample, pattern separation did not vary based solely on APOE e4 status. This differs from a recent study examining the effect of APOE e4 status on pattern separation performance in healthy older African Americans (Sinha et al., 2018), which identified worse lure discrimination in older adults with at least one APOE e4 allele, driven by a greater likelihood of generating false alarms to lure items (e.g., classifying lures as “old” rather than similar). One possible explanation for this difference is that the APOE e4 carriers in Sinha et al. (2018) also performed worse on measures of verbal list learning (RAVLT Immediate and Delayed Recall), and prior work has demonstrated a moderating effect of verbal memory performance on the relationship between APOE e4 status and pattern separation ability (Sheppard et al., 2016), perhaps suggesting that the effects of APOE on pattern separation ability are more pronounced in participants who are more cognitively impaired.
Consistent with the robust literature on sex differences in episodic memory (Asperholm et al., 2019), females were more accurate than males on our measure of lure discrimination. Our findings are not surprising given the female advantage on memory tasks with verbal stimuli (words, sentences, prose) or nameable images, with male advantages on more spatially-oriented tasks (Asperholm et al., 2019). Prior work investigating the relationship between sex differences and APOE status on memory performance identified baseline advantages for females and accelerated verbal memory decline in APOE e4 carriers, but no difference in the rate of decline based on sex (Caselli et al., 2015). In the current sample, the interaction between left hippocampal blood flow and APOE status was only significant in females, such that greater left hippocampal blood flow was associated with worse lure discrimination in e4 carriers, whereas the reverse was true for noncarriers. These results provide preliminary evidence that female APOE e4 carriers may be more vulnerable to neurovascular changes impacting hippocampal function and memory performance. Future examination of this effect is needed, and it would be particularly beneficial to understand whether certain APOE genotypes convey neuroprotective effects in females, but not males.
Although the APOE e4 allele has been associated with substantial variation in the structure (Cohen et al., 2001; Den Heijer et al., 2002; Haller et al., 2020; Jak et al., 2007; Lind et al., 2006; Montagne et al., 2020; Plassman et al., 1997; Weintraub et al., 2020) and function of the medial temporal lobes (Bookheimer et al., 2000; De Marco et al., 2017; Filippini et al., 2009; Fleisher et al., 2009; Han et al., 2007; Nichols et al., 2012; Suri et al., 2015; Suthana et al., 2010; Zheng et al., 2018), our APOE e4 groups did not differ in resting hippocampal blood flow. Prior investigations of MTL blood flow in APOE e4 carriers have been mixed, with some identifying no difference in hippocampal blood flow between groups (Bangen et al., 2017; Hays et al., 2019), and others demonstrating altered resting CBF across widespread medial temporal, frontal and parietal regions (Tai et al., 2016; Wierenga et al., 2014). It is possible that these differences are attributable to the time point at which data were sampled, as higher resting CBF is often observed in APOE e4 carriers during early adulthood and middle age, with lower resting CBF in older age compared to noncarriers (Thambisetty et al., 2010; Wierenga et al., 2013), resulting in mixed findings in cross-sectional analyses (Bangen et al., 2009; Filippini et al., 2011; Thambisetty et al., 2010; Wierenga et al., 2013; Zlatar et al., 2016). This curvilinear pattern of hyperperfusion followed by hypoperfusion in APOE e4 carriers compared to noncarriers, has been likened to functional hyperactivation, and is thought to reflect faltering compensation due to deteriorating cerebrovascular function. Recent findings of blood brain barrier breakdown in the hippocampi and medial temporal lobes of APOE e4 carriers provides further support for this interpretation (Montagne et al., 2020).
Evidence from animal models provides an alternate explanation to compensation. Compared to unimpaired aged rats, memory impaired rats exhibit reduced expression of an ensemble of genes in the CA3 subfield of the hippocampus involved in synaptic plasticity, inhibitory control, and memory (Haberman et al., 2013), suggesting that reduced inhibitory control in hippocampal neurons contributes to memory deficits. Supporting this hypothesis, administration of the atypical antiepileptic levetiracetam improves spatial memory (Koh et al., 2013) and restores somatostatin protein expression among DG interneurons in aged memory-impaired rats (Spiegel et al., 2013). In human amyloid precursor protein transgenic mice, memory deficits and network hypersynchrony on electroencephalographic recordings are associated with reduced gamma oscillatory activity due to inhibitory interneuron dysfunction in parvalbumin cells (Palop & Mucke, 2016; Verret et al., 2012), identifying this pathway as another source of global hyperactivity in memory impaired rodents. In rodent models of APOE, learning and memory deficits are associated with age-dependent decreases in GABAergic interneurons in the DG of the hippocampus (Andrews-Zwilling et al., 2010), identifying inhibitory interneuron dysfunction and DG neuronal loss as potential contributors to memory impairment in rodent models of APOE and aging.
Given the relatively low spatial resolution of our ASL sequence, which captures blood flow in the hippocampus as a whole rather than subfield-specific perfusion, inferences from subfield specific rodent findings should be made with caution. Nevertheless, inhibitory interneuron dysfunction in the DG and CA3 subfields of the hippocampus would directly impact pattern separation abilities by shifting computational processes in the MTL toward pattern completion. In order to create distinct, orthogonal representations that support successful pattern separation, the DG relies on a sparse coding scheme (Bakker et al., 2008; Kitamura et al., 2015; Marr, 1971). With reductions in inhibitory interneuron function, the coding scheme in DG becomes less sparse and more widely distributed, resulting in fuzzier representations and an increased likelihood of pattern completion (Espinoza et al., 2018; Guo et al., 2018). Further downstream, inhibitory interneurons play a critical role in regulating the recurrent excitatory collaterals of the CA3 auto-associative system, which bias the system toward pattern completion under normal circumstances (Treves & Rolls, 1994). Disinhibition across these two hippocampal subfields results in an additive bias toward pattern completion, thereby reducing mnemonic discrimination, and increasing the likelihood that similar lures are falsely classified as “old” on behavioral measures.
Although several studies have identified similar associations between medial temporal hyperperfusion and memory decrements in APOE e4 carriers, future work should seek to replicate these findings in a larger and more diverse sample in order to carry out in-depth examination of the complex relationship amongst multiple interacting factors (e.g., sex, cardiovascular health). Our sample was highly educated and primarily Caucasian, limiting generalizability to the more diverse population. Further, it would be ideal to examine the relationship between CBF, Alzheimer’s pathology (PET measures of amyloid and tau), and cerebrovascular reactivity within a large and well-characterized sample. Finally, longitudinal measures of CBF and memory in middle-aged to older adults would help clarify the time course of APOE effects and aid in determining how these factors can lead to increased risk for progression to Alzheimer’s disease.
Although several steps were taken to minimize the effects of partial voluming on estimates of hippocampal CBF, the low spatial resolution of ASL makes it particularly challenging to draw inferences from subfield specific animal models and contextualize results from high-resolution fMRI studies in humans, although efforts are underway to improve ASL resolution. A more fine-grained imaging approach is needed to understand the relationship between hippocampal perfusion and CA3/DG “hyperactivation” in older adults and those at increased risk for Alzheimer’s disease.
5 |. CONCLUSIONS
Our findings demonstrate a differential association between hippocampal perfusion and lure discrimination based on APOE genotype. A negative association between right hippocampal CBF and lure discrimination was identified in e4 carriers, an effect that reversed directionality in noncarriers. This work complements prior fMRI studies on hippocampal hyperactivation by establishing hippocampal hyperperfusion as an indicator of medial temporal network dysfunction in cognitively normal older adults at increased risk for Alzheimer’s disease. Further, it highlights the importance of APOE status as a modifying factor in neurovascular models of cognitive aging. Future work focused on longitudinal changes in hippocampal blood flow and metabolism may help identify patterns that differentiate between successful agers and those who progress to develop a neurodegenerative process. Additional high-resolution imaging is needed to understand the mechanisms underlying hippocampal contributions to age-related pattern separation deficits.
Supplementary Material
ACKNOWLEDGMENTS
This work was supported by NIH-NIA grants K23AG058752 (PI: K. B. C.), 1R01AG032289 (PI: J. H. K.), R01AG048234 (PI: J. H. K.), K23AG061253 (PI: A. M. S.), and UCSF ADRC P50 AG023501. This work was also supported by Larry L. Hillblom Network Grant (PI: J. H. K.; 2014-A-004-NET) and Fellowship Grant (PI: A. M. S., 2018-A-025-FEL).
Funding information
Larry Hillblom Network Grant, Grant/Award Numbers: 2014-A-004-NET, 2018-A-025-FEL; NIH-NIA, Grant/Award Numbers: 1R01AG032289, K23AG058752, K23AG061253, R01AG048234
Footnotes
CONFLICT OF INTEREST
The authors declare no conflict of interest.
SUPPORTING INFORMATION
Additional supporting information may be found online in the Supporting Information section at the end of this article.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- Aguirre GK, Detre JA, Zarahn E, & Alsop DC (2002). Experimental design and the relative sensitivity of BOLD and perfusion fMRI. NeuroImage, 15, 488–500. 10.1006/nimg.2001.0990 [DOI] [PubMed] [Google Scholar]
- Amaral DG, Scharfman HE, & Lavenex P (2007). The dentate gyrus: Fundamental neuroanatomical organization (dentate gyrus for dummies). Progress in Brain Research, 163, 3–22. 10.1016/S0079-6123(07)63001-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews-Zwilling Y, Bien-Ly N, Xu Q, Li G, Bernardo A, Yoon SY, … Huang Y (2010). Apolipoprotein E4 causes age- and Tau-dependent impairment of GABAergic interneurons, leading to learning and memory deficits in mice. Journal of Neuroscience, 30, 13707–13717. 10.1523/JNEUROSCI.4040-10.2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asperholm M, Högman N, Rafi J, & Herlitz A (2019). What did you do yesterday? A meta-analysis of sex differences in episodic memory. Psychological Bulletin, 145(8), 785–821. 10.1037/bul0000197 [DOI] [PubMed] [Google Scholar]
- Avants BB, Epstein CL, Grossman M, & Gee JC (2008). Symmetric diffeomorphic image registration with cross-correlation: Evaluating automated labeling of elderly and neurodegenerative brain. Medical Image Analysis, 12, 26–41. 10.1016/j.media.2007.06.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bakker A, Kirwan CB, Miller M, & Stark CEL (2008). Pattern separation in the human hippocampal CA3 and dentate gyrus. Science, 319, 1640–1642. 10.1126/science.1152882 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bakker A, Krauss GL, Albert MS, Speck CL, Jones LR, Stark CE, … Gallagher M (2012). Reduction of hippocampal hyperactivity improves cognition in amnestic mild cognitive impairment. Neuron, 74, 467–474. 10.1016/j.neuron.2012.03.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bangen KJ, Clark AL, Edmonds EC, Evangelista ND, Werhane ML, Thomas KR, … Delano-Wood L (2017). Cerebral blood flow and amyloid-β interact to affect memory performance in cognitively normal older adults. Frontiers in Aging Neuroscience, 9, 181. 10.3389/fnagi.2017.00181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bangen KJ, Restom K, Liu TT, Jak AJ, Wierenga CE, Salmon DP, & Bondi MW (2009). Differential age effects on cerebral blood flow and BOLD response to encoding: Associations with cognition and stroke risk. Neurobiology of Aging, 30, 1276–1287. 10.1016/j.neurobiolaging.2007.11.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bangen KJ, Restom K, Liu TT, Wierenga CE, Jak AJ, Salmon DP, & Bondi MW (2012). Assessment of Alzheimer’s disease risk with functional magnetic resonance imaging: An arterial spin labeling study. Journal of Alzheimer’s Disease, 31, S59–S74. 10.3233/JAD-2012-120292 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes CA (1979). Memory deficits associated with senescence: A neurophysiological and behavioral study in the rat. Journal of Comparative and Physiological Psychology, 93, 74–104. 10.1037/h0077579 [DOI] [PubMed] [Google Scholar]
- Barnes CA, & McNaughton BL (1980). Physiological compensation for loss of afferent synapses in rat hippocampal granule cells during senescence. The Journal of Physiology, 309, 473–485. 10.1113/jphysiol.1980.sp013521 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes CA, Rao G, & Houston FP (2000). LTP induction threshold change in old rats at the perforant path-granule cell synapse. Neurobiology of Aging, 21, 613–620. 10.1016/S0197-4580(00)00163-9 [DOI] [PubMed] [Google Scholar]
- Beason-Held LL, Goh JO, An Y, Kraut MA, O’Brien RJ, Ferrucci L, & Resnick SM (2013). Changes in brain function occur years before the onset of cognitive impairment. Journal of Neuroscience, 33, 18008–18014. 10.1523/JNEUROSCI.1402-13.2013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bentourkia M, Bol A, Ivanoiu A, Labar D, Sibomana M, Coppens A, … De Volder AG (2000). Comparison of regional cerebral blood flow and glucose metabolism in the normal brain: Effect of aging. Journal of the Neurological Sciences, 181(1-2), 19–28. 10.1016/S0022-510X(00)00396-8 [DOI] [PubMed] [Google Scholar]
- Berron D, Schütze H, Maass A, Cardenas-Blanco A, Kuijf HJ, Kumaran D, & Düzel E (2016). Strong evidence for pattern separation in human dentate gyrus. Journal of Neuroscience, 36, 7569–7579. 10.1523/JNEUROSCI.0518-16.2016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bettio LEB, Rajendran L, & Gil-Mohapel J (2017). The effects of aging in the hippocampus and cognitive decline. Neuroscience and Biobehavioral Reviews, 79, 66–86. 10.1016/j.neubiorev.2017.04.030 [DOI] [PubMed] [Google Scholar]
- Bookheimer SY, Strojwas MH, Cohen MS, Saunders AM, Pericak-Vance MA, Mazziotta JC, & Small GW (2000). Patterns of brain activation in people at risk for Alzheimer’s disease. New England Journal of Medicine, 343, 450–456. 10.1056/nejm200008173430701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buxton RB, Frank LR, Wong EC, Siewert B, Warach S, & Edelman RR (1998). A general kinetic model for quantitative perfusion imaging with arterial spin labeling. Magnetic Resonance in Medicine, 40, 383–396. 10.1002/mrm.1910400308 [DOI] [PubMed] [Google Scholar]
- Caselli RJ, Dueck AC, Locke DEC, Baxter LC, Woodruff BK, & Geda YE (2015). Sex-based memory advantages and cognitive aging: A challenge to the cognitive reserve construct? Journal of the International Neuropsychological Society, 21, 95–104. 10.1017/S1355617715000016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chao LL, Buckley ST, Kornak J, Schuff N, Madison C, Yaffe K, … Weiner MW (2010). ASL perfusion MRI predicts cognitive decline and conversion from MCI to dementia. Alzheimer Disease and Associated Disorders, 24, 19–27. 10.1097/WAD.0b013e3181b4f736 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chawla MK, Guzowski JF, Ramirez-Amaya V, Lipa P, Hoffman KL, Marriott LK, … Barnes CA (2005). Sparse, environmentally selective expression of arc RNA in the upper blade of the rodent fascia dentata by brief spatial experience. Hippocampus, 15, 579–586. 10.1002/hipo.20091 [DOI] [PubMed] [Google Scholar]
- Chen JJ, Rosas HD, & Salat DH (2011). Age-associated reductions in cerebral blood flow are independent from regional atrophy. NeuroImage, 55, 468–478. 10.1016/j.neuroimage.2010.12.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clement P, Mutsaerts HJ, Václavů L, Ghariq E, Pizzini FB, Smits M, … Achten E (2018). Variability of physiological brain perfusion in healthy subjects – A systematic review of modifiers. Considerations for multi-center ASL studies. Journal of Cerebral Blood Flow and Metabolism, 38, 1418–1437. 10.1177/0271678X17702156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen RM, Small C, Lalonde F, Friz J, & Sunderland T (2001). Effect of apolipoprotein E genotype on hippocampal volume loss in aging healthy women. Neurology, 57, 2223–2228. 10.1212/WNL.57.12.2223 [DOI] [PubMed] [Google Scholar]
- Damoiseaux JS, Seeley WW, Zhou J, Shirer WR, Coppola G, Karydas A, … Greicius MD (2012). Gender modulates the APOE ε4 effect in healthy older adults: Convergent evidence from functional brain connectivity and spinal fluid tau levels. Journal of Neuroscience, 32, 8254–8262. 10.1523/JNEUROSCI.0305-12.2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Marco M, Vallelunga A, Meneghello F, Varma S, Frangi AF, & Venneri A (2017). ApoE ε4 allele related alterations in hippocampal connectivity in early Alzheimer’s disease support memory performance. Current Alzheimer Research, 14, 766–777. 10.2174/1567205014666170206113528 [DOI] [PubMed] [Google Scholar]
- Den Heijer T, Oudkerk M, Launer LJ, Van Duijn CM, Hofman A, & Breteler MMB (2002). Hippocampal, amygdalar, and global brain atrophy in different apolipoprotein E genotypes. Neurology, 59, 746–748. 10.1212/WNL.59.5.746 [DOI] [PubMed] [Google Scholar]
- Dennis NA, Browndyke JN, Stokes J, Need A, Burke JR, Welsh-Bohmer KA, & Cabeza R (2010). Temporal lobe functional activity and connectivity in young adult APOE ε4 carriers. Alzheimer’s and Dementia, 6, 303–311. 10.1016/j.jalz.2009.07.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desikan RS, Ségonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, … Killiany RJ (2006). An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. NeuroImage, 31, 968–980. 10.1016/j.neuroimage.2006.01.021 [DOI] [PubMed] [Google Scholar]
- Dieguez D, & Barea-Rodriguez EJ (2004). Aging impairs the late phase of long-term potentiation at the medial perforant path-CA3 synapse in awake rats. Synapse, 52, 53–61. 10.1002/syn.20004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du AT, Jahng GH, Hayasaka S, Kramer JH, Rosen HJ, Gorno-Tempini ML, … Schuff N (2006). Hypoperfusion in frontotemporal dementia and Alzheimer disease by arterial spin labeling MRI. Neurology, 67, 1215–1220. 10.1212/01.wnl.0000238163.71349.78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Espinoza C, Guzman SJ, Zhang X, & Jonas P (2018). Parvalbumin + interneurons obey unique connectivity rules and establish a powerful lateral-inhibition microcircuit in dentate gyrus. Nature Communications, 9, 4605. 10.1038/s41467-018-06899-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Filippini N, Ebmeier KP, MacIntosh BJ, Trachtenberg AJ, Frisoni GB, Wilcock GK, … Mackay CE (2011). Differential effects of the APOE genotype on brain function across the lifespan. NeuroImage, 54, 602–610. 10.1016/j.neuroimage.2010.08.009 [DOI] [PubMed] [Google Scholar]
- Filippini N, MacIntosh BJ, Hough MG, Goodwin GM, Frisoni GB, Smith SM, … Mackay CE (2009). Distinct patterns of brain activity in young carriers of the APOE-ε4 allele. Proceedings of the National Academy of Sciences of the United States of America, 106, 7209–7214. 10.1073/pnas.0811879106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleisher AS, Podraza KM, Bangen KJ, Taylor C, Sherzai A, Sidhar K, … Buxton RB (2009). Cerebral perfusion and oxygenation differences in Alzheimer’s disease risk. Neurobiology of Aging, 30, 1737–1748. 10.1016/j.neurobiolaging.2008.01.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo N, Soden ME, Herber C, Kim MTW, Besnard A, Lin P, … Sahay A (2018). Dentate granule cell recruitment of feedforward inhibition governs engram maintenance and remote memory generalization. Nature Medicine, 24, 438–449. 10.1038/nm.4491 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haberman RP, Colantuoni C, Koh MT, & Gallagher M (2013). Behaviorally activated mRNA expression profiles produce signatures of learning and enhanced inhibition in aged rats with preserved memory. PLoS One, 8, e83674. 10.1371/journal.pone.0083674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haller S, Montandon ML, Rodriguez C, Garibotto V, Herrmann FR, & Giannakopoulos P (2020). Hippocampal volume loss, brain amyloid accumulation, and APOE status in cognitively intact elderly subjects. Neurodegenerative Diseases, 19, 139–147. 10.1159/000504302 [DOI] [PubMed] [Google Scholar]
- Han SD, Houston WS, Jak AJ, Eyler LT, Nagel BJ, Fleisher AS, … Bondi MW (2007). Verbal paired-associate learning by APOE genotype in non-demented older adults: fMRI evidence of a right hemispheric compensatory response. Neurobiology of Aging, 28, 238–247. 10.1016/j.neurobiolaging.2005.12.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayasaka S, Du AT, Duarte A, Kornak J, Jahng GH, Weiner MW, & Schuff N (2006). A non-parametric approach for co-analysis of multi-modal brain imaging data: Application to Alzheimer’s disease. NeuroImage, 30, 768–779. 10.1016/j.neuroimage.2005.10.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayes AF (2012). PROCESS: A versatile computational tool for observed variable mediation, moderation, and conditional process modeling [White paper]. Retrieved from http://www.afhayes.com/public/process2012.pdf
- Hays CC, Zlatar ZZ, Meloy MJ, Bondi MW, Gilbert PE, Liu TT, … Wierenga CE (2019). APOE modifies the interaction of entorhinal cerebral blood flow and cortical thickness on memory function in cognitively normal older adults. NeuroImage, 202, 116162. 10.1016/j.neuroimage.2019.116162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heo S, Prakash RS, Voss MW, Erickson KI, Ouyang C, Sutton BP, & Kramer AF (2010). Resting hippocampal blood flow, spatial memory and aging. Brain Research, 1315, 119–127. 10.1016/j.brainres.2009.12.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jak AJ, Houston WS, Nagel BJ, Corey-Bloom J, & Bondi MW (2007). Differential cross-sectional and longitudinal impact of APOE genotype on hippocampal volumes in nondemented older adults. Dementia and Geriatric Cognitive Disorders, 23, 382–389. 10.1159/000101340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkinson M, Beckmann CF, Behrens TEJ, Woolrich MW, & Smith SM (2012). Review FSL. NeuroImage, 62, 782–790. [DOI] [PubMed] [Google Scholar]
- Johnson NA, Jahng GH, Weiner MW, Miller BL, Chui HC, Jagust WJ, … Schuff N (2005). Pattern of cerebral hypoperfusion in Alzheimer disease and mild cognitive impairment measured with arterial spin-labeling MR imaging: Initial experience. Radiology, 234, 851–859. 10.1148/radiol.2343040197 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kassab R, & Alexandre F (2018). Pattern separation in the hippocampus: Distinct circuits under different conditions. Brain Structure and Function, 223, 2785–2808. 10.1007/s00429-018-1659-4 [DOI] [PubMed] [Google Scholar]
- Kitamura T, Sun C, Martin J, Kitch LJ, Schnitzer MJ, & Tonegawa S (2015). Entorhinal cortical ocean cells encode specific contexts and drive context-specific fear memory. Neuron, 87, 1317–1331. 10.1016/j.neuron.2015.08.036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koh MT, Rosenzweig-Lipson S, & Gallagher M (2013). Selective GABAA α5 positive allosteric modulators improve cognitive function in aged rats with memory impairment. Neuropharmacology, 64, 145–152. 10.1016/j.neuropharm.2012.06.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lacy JW, Yassa MA, Stark SM, Muftuler LT, & Stark CEL (2011). Distinct pattern separation related transfer functions in human CA3/dentate and CA1 revealed using high resolution fMRI and variable mnemonic similarity. Learning and Memory, 18, 15–18. 10.1101/lm.1971111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lafosse JM, Reed BR, Mungas D, Sterling SB, Wahbeh H, & Jagust WJ (1997). Fluency and memory differences between ischemic vascular dementia and Alzheimer’s disease. Neuropsychology, 11, 514–522. 10.1037/0894-4105.11.4.514 [DOI] [PubMed] [Google Scholar]
- Lee I, & Kesner RP (2004). Encoding versus retrieval of spatial memory: Double dissociation between the dentate gyrus and the perforant path inputs into CA3 in the dorsal hippocampus. Hippocampus, 14, 66–76. 10.1002/hipo.10167 [DOI] [PubMed] [Google Scholar]
- Leutgeb JK, Leutgeb S, Moser MB, & Moser EI (2007). Pattern separation in the dentate gyrus and CA3 of the hippocampus. Science, 315, 961–966. 10.1126/science.1135801 [DOI] [PubMed] [Google Scholar]
- Lind J, Larsson A, Persson J, Ingvar M, Nilsson LG, Bäckman L, … Nyberg L (2006). Reduced hippocampal volume in non-demented carriers of the apolipoprotein E ε4: Relation to chronological age and recognition memory. Neuroscience Letters, 396, 23–27. 10.1016/j.neulet.2005.11.070 [DOI] [PubMed] [Google Scholar]
- Liu M, Paranjpe MD, Zhou X, Duy PQ, Goyal MS, Benzinger TLS, … Zhou Y (2019). Sex modulates the ApoE ε4 effect on brain tau deposition measured by 18F-AV-1451 PET in individuals with mild cognitive impairment. Theranostics, 9, 4959–4970. 10.7150/thno.35366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luh WM, Wong EC, Bandettini PA, & Hyde JS (1999). QUIPSS II with thin-slice TI1 periodic saturation: A method for improving accuracy of quantitative perfusion imaging using pulsed arterial spin labeling. Magnetic Resonance in Medicine, 41, 1246–1254. [DOI] [PubMed] [Google Scholar]
- Marr D (1971). Simple memory: A theory for archicortex. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 262(841), 23–81. 10.1098/rstb.1971.0078 [DOI] [PubMed] [Google Scholar]
- Miller SL, Celone K, DePeau K, Diamond E, Dickerson BC, Rentz D, … Sperling RA (2008). Age-related memory impairment associated with loss of parietal deactivation but preserved hippocampal activation. Proceedings of the National Academy of Sciences of the United States of America, 105, 2181–2186. 10.1073/pnas.0706818105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montagne A, Nation DA, Sagare AP, Barisano G, Sweeney MD, Chakhoyan A, … Zlokovic BV (2020). APOE4 leads to blood–brain barrier dysfunction predicting cognitive decline. Nature, 581, 71–76. 10.1038/s41586-020-2247-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller-Gärtner HW, Links JM, Prince JL, Bryan RN, McVeigh E, Leal JP, … Frost JJ (1992). Measurement of radiotracer concentration in brain gray matter using positron emission tomography: MRI-based correction for partial volume effects. Journal of Cerebral Blood Flow and Metabolism, 12, 571–583. 10.1038/jcbfm.1992.81 [DOI] [PubMed] [Google Scholar]
- Nichols LM, Masdeu JC, Mattay VS, Kohn P, Emery M, Sambataro F, … Berman KF (2012). Interactive effect of apolipoprotein E genotype and age on hippocampal activation during memory processing in healthy adults. Archives of General Psychiatry, 69, 804–813. 10.1001/archgenpsychiatry.2011.1893 [DOI] [PubMed] [Google Scholar]
- O’Brien JL, O’Keefe KM, Laviolette PS, Deluca AN, Blacker D, Dickerson BC, & Sperling RA (2010). Longitudinal fMRI in elderly reveals loss of hippocampal activation with clinical decline. Neurology, 74, 1969–1976. 10.1212/WNL.0b013e3181e3966e [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palop JJ, & Mucke L (2016). Network abnormalities and interneuron dysfunction in Alzheimer disease. Nature Reviews Neuroscience, 17, 777–792. 10.1038/nrn.2016.141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parkes LM, Rashid W, Chard DT, & Tofts PS (2004). Normal cerebral perfusion measurements using arterial spin labeling: Reproducibility, stability, and age and gender effects. Magnetic Resonance in Medicine, 51(4), 736–743. 10.1002/mrm.20023 [DOI] [PubMed] [Google Scholar]
- Petcharunpaisan S, Ramalho J, & Castillo M (2010). Arterial spin labeling in neuroimaging arterial spin labeling: Basic principles, emerging techniques and clinical applications in neuroimaging. World Journal of Radiology, 2(10), 384–398. 10.4329/wjr.v2.i10.384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plassman BL, Welsh-Bohmer KA, Bigler ED, Johnson MA, Anderson CV, Helms MJ, … Breitner JCS (1997). Apolipoprotein E ε4 allele and hippocampal volume in twins with normal cognition. Neurology, 48, 985–988. 10.1212/wnl.48.4.985 [DOI] [PubMed] [Google Scholar]
- Putcha D, Brickhouse M, O’Keefe K, Sullivan C, Rentz D, Marshall G, … Sperling R (2011). Hippocampal hyperactivation associated with cortical thinning in Alzheimer’s disease signature regions in non-demented elderly adults. Journal of Neuroscience, 31, 17680–17688. 10.1523/JNEUROSCI.4740-11.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raz N, Ghisletta P, Rodrigue KM, Kennedy KM, & Lindenberger U (2010). Trajectories of brain aging in middle-aged and older adults: Regional and individual differences. NeuroImage, 51, 501–511. 10.1016/j.neuroimage.2010.03.020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raz N, Lindenberger U, Rodrigue KM, Kennedy KM, Head D, Williamson A, … Acker JD (2005). Regional brain changes in aging healthy adults: General trends, individual differences and modifiers. Cerebral Cortex, 15, 1676–1689. 10.1093/cercor/bhi044 [DOI] [PubMed] [Google Scholar]
- Sampedro F, Vilaplana E, de Leon MJ, Alcolea D, Pegueroles J, Montal V, … Fortea J (2015). APOE-by-sex interactions on brain structure and metabolism in healthy elderly controls. Oncotarget, 6, 26663–26674. 10.18632/oncotarget.5185 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen S, Zhou W, Chen X, & Zhang J (2019). Sex differences in the association of APOE ε4 genotype with longitudinal hippocampal atrophy in cognitively normal older people. European Journal of Neurology, 26, 1362–1369. 10.1111/ene.13987 [DOI] [PubMed] [Google Scholar]
- Sheppard DP, Graves LV, Holden HM, Delano-Wood L, Bondi MW, & Gilbert PE (2016). Spatial pattern separation differences in older adult carriers and non-carriers for the apolipoprotein E epsilon 4 allele. Neurobiology of Learning and Memory, 129, 113–119. 10.1016/j.nlm.2015.04.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha N, Berg CN, Tustison NJ, Shaw A, Hill D, Yassa MA, & Gluck MA (2018). APOE ε4 status in healthy older African Americans is associated with deficits in pattern separation and hippocampal hyperactivation. Neurobiology of Aging, 69, 221–229. 10.1016/j.neurobiolaging.2018.05.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiegel AM, Koh MT, Vogt NM, Rapp PR, & Gallagher M (2013). Hilar interneuron vulnerability distinguishes aged rats with memory impairment. Journal of Comparative Neurology, 521, 3508–3523. 10.1002/cne.23367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staffaroni AM, Cobigo Y, Elahi FM, Casaletto KB, Walters SM, Wolf A, … Kramer JH (2019). A longitudinal characterization of perfusion in the aging brain and associations with cognition and neural structure. Human Brain Mapping, 40(12), 3522–3533. 10.1002/hbm.24613 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stark SM, Kirwan CB, & Stark CEL (2019). Mnemonic similarity task: A tool for assessing hippocampal integrity. Trends in Cognitive Sciences, 23(11), 938–951. 10.1016/j.tics.2019.08.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stark SM, Yassa MA, Lacy JW, & Stark CE (2013). A task to assess behavioral pattern separation (BPS) in humans: Data from healthy aging and mild cognitive impairment. Neuropsychologia, 51(12), 2442–2449. 10.1016/j.neuropsychologia.2012.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevenson RF, Reagh ZM, Chun AP, Murray EA, & Yassa MA (2020). Pattern separation and source memory engage distinct hippocampal and neocortical regions during retrieval. Journal of Neuroscience, 40, 843–851. 10.1523/JNEUROSCI.0564-19.2019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suri S, Mackay CE, Kelly ME, Germuska M, Tunbridge EM, Frisoni GB, … Filippini N (2015). Reduced cerebrovascular reactivity in young adults carrying the APOE ε4 allele. Alzheimer’s and Dementia, 11, 648–657.e1. 10.1016/j.jalz.2014.05.1755 [DOI] [PubMed] [Google Scholar]
- Suthana NA, Krupa A, Donix M, Burggren A, Ekstrom AD, Jones M, … Bookheimer SY (2010). Reduced hippocampal CA2, CA3, and dentate gyrus activity in asymptomatic people at genetic risk for Alzheimer’s disease. NeuroImage, 53, 1077–1084. 10.1016/j.neuroimage.2009.12.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tai LM, Thomas R, Marottoli FM, Koster KP, Kanekiyo T, Morris AWJ, & Bu G (2016). The role of APOE in cerebrovascular dysfunction. Acta Neuropathologica, 131, 709–723. 10.1007/s00401-016-1547-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanenbaum AB, Snyder AZ, Brier MR, & Ances BM (2015). A method for reducing the effects of motion contamination in arterial spin labeling magnetic resonance imaging. Journal of Cerebral Blood Flow and Metabolism, 35, 1697–1702. 10.1038/jcbfm.2015.124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thambisetty M, Beason-Held L, An Y, Kraut MA, & Resnick SM (2010). APOE ε4 genotype and longitudinal changes in cerebral blood flow in normal aging. Archives of Neurology, 67, 93–98. 10.1001/archneurol.2009.913 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tran TT, Speck CL, Pisupati A, Gallagher M, & Bakker A (2017). Increased hippocampal activation in ApoE-4 carriers and non-carriers with amnestic mild cognitive impairment. NeuroImage: Clinical, 13, 237–245. 10.1016/j.nicl.2016.12.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Treves A, & Rolls ET (1994). Computational analysis of the role of the hippocampus in memory. Hippocampus, 4, 374–391. 10.1002/hipo.450040319 [DOI] [PubMed] [Google Scholar]
- Trivedi MA, Schmitz TW, Ries ML, Hess TM, Fitzgerald ME, Atwood CS, … Johnson SC (2008). fMRI activation during episodic encoding and metacognitive appraisal across the lifespan: Risk factors for Alzheimer’s disease. Neuropsychologia, 46, 1667–1678. 10.1016/j.neuropsychologia.2007.11.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vazdarjanova A, & Guzowski JF (2004). Differences in hippocampal neuronal population responses to modifications of an environmental context: Evidence for distinct, yet complementary, functions of CA3 and CA1 ensembles. Journal of Neuroscience, 24, 6489–6496. 10.1523/JNEUROSCI.0350-04.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verret L, Mann EO, Hang GB, Barth AMI, Cobos I, Ho K, … Palop JJ (2012). Inhibitory interneuron deficit links altered network activity and cognitive dysfunction in Alzheimer model. Cell, 149, 708–721. 10.1016/j.cell.2012.02.046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Aguirre GK, Kimberg DY, Roc AC, Li L, & Detre JA (2003). Arterial spin labeling perfusion fMRI with very low task frequency. Magnetic Resonance in Medicine, 49, 796–802. 10.1002/mrm.10437 [DOI] [PubMed] [Google Scholar]
- Wang X, Zhou W, Ye T, Lin X, & Zhang J (2019). Sex difference in the association of APOE4 with memory decline in mild cognitive impairment. Journal of Alzheimer’s Disease, 69, 1161–1169. 10.3233/JAD-181234 [DOI] [PubMed] [Google Scholar]
- Weintraub S, Teylan M, Rader B, Chan KCG, Bollenbeck M, Kukull WA, … Mesulam MM (2020). APOE is a correlate of phenotypic heterogeneity in Alzheimer disease in a national cohort. Neurology, 94, e607–e612. 10.1212/WNL.0000000000008666 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wierenga CE, Clark LR, Dev SI, Shin DD, Jurick SM, Rissman RA, … Bondi MW (2013). Interaction of age and APOE genotype on cerebral blood flow at rest. Journal of Alzheimer’s Disease, 34, 921–935. 10.3233/JAD-121897 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wierenga CE, Hays CC, & Zlatar ZZ (2014). Cerebral blood flow measured by arterial spin labeling MRI as a preclinical marker of Alzheimer’s disease. Journal of Alzheimer’s Disease, 42, S411–S419. 10.3233/JAD-141467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson IA, Gallagher M, Eichenbaum H, & Tanila H (2006). Neurocognitive aging: Prior memories hinder new hippocampal encoding. Trends in Neurosciences, 29, 662–670. 10.1016/j.tins.2006.10.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson IA, Ikonen S, Gallagher M, Eichenbaum H, & Tanila H (2005). Age-associated alterations of hippocampal place cells are subregion specific. Journal of Neuroscience, 25, 6877–6886. 10.1523/JNEUROSCI.1744-05.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Witter MP (1993). Organization of the entorhinal—Hippocampal system: A review of current anatomical data. Hippocampus, 3, 33–44. 10.1002/hipo.1993.4500030707 [DOI] [PubMed] [Google Scholar]
- Yassa MA, Lacy JW, Stark SM, Albert MS, Gallagher M, & Stark CEL (2011). Pattern separation deficits associated with increased hippocampal CA3 and dentate gyrus activity in nondemented older adults. Hippocampus, 21(9), 968–979. 10.1002/hipo.20808 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yassa MA, & Stark CEL (2011). Pattern separation in the hippocampus. Trends in Neurosciences, 34, 515–525. 10.1016/j.tins.2011.06.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yassa MA, Stark SM, Bakker A, Albert MS, Gallagher M, & Stark CEL (2010). High-resolution structural and functional MRI of hippocampal CA3 and dentate gyrus in patients with amnestic mild cognitive impairment. NeuroImage, 51, 1242–1252. 10.1016/j.neuroimage.2010.03.040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yew B, & Nation DA (2017). Cerebrovascular resistance: Effects on cognitive decline, cortical atrophy, and progression to dementia. Brain, 140, 1987–2001. 10.1093/brain/awx112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng LJ, Su YY, Wang YF, Schoepf UJ, Varga-Szemes A, Pannell J, … Zhang LJ (2018). Different hippocampus functional connectivity patterns in healthy young adults with mutations of APP/-Presenilin-1/2 and APOEε4. Molecular Neurobiology, 55, 3439–3450. 10.1007/s12035-017-0540-4 [DOI] [PubMed] [Google Scholar]
- Zlatar ZZ, Bischoff-Grethe A, Hays CC, Liu TT, Meloy MJ, Rissman RA, … Wierenga CE (2016). Higher brain perfusion may not support memory functions in cognitively normal carriers of the ApoE ε4 allele compared to non-carriers. Frontiers in Aging Neuroscience, 8, 151. 10.3389/fnagi.2016.00151 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


