Abstract
Purpose of the Review
This review aims to summarize the current knowledge of the extracellular matrix remodeling during hepatic fibrosis. We discuss the diverse interactions of the extracellular matrix with hepatic cells and the surrounding matrix in liver fibrosis, with the focus on the molecular pathways and the mechanisms that regulate extracellular matrix remodeling.
Recent Findings
The extracellular matrix not only provides structure and support for the cells, but also controls cell behavior by providing adhesion signals and by acting as a reservoir of growth factors and cytokines.
Summary
Hepatic fibrosis is characterized by an excessive accumulation of extracellular matrix. During fibrogenesis, the natural remodeling process of the extracellular matrix varies, resulting in the excessive accumulation of its components, mainly collagens. Signals released by the extracellular matrix induce the activation of hepatic stellate cells, which are the major source of extracellular matrix and most abundant myofibroblasts in the liver.
Graphical abstract
Keywords: Liver fibrosis, Extracellular matrix, Hepatic stellate cell, Collagen, Metalloproteinases, TGF-β1
Introduction
The extracellular matrix (ECM) is a complex cross-linked network of macromolecular proteins that not only provide structural support, but also play an essential role in the development and maintenance of tissue homeostasis [1]. In addition, the interaction between cells and the ECM is bi-directional. Cells are constantly receiving and accepting information from the ECM and, in turn, remodel the ECM in which old proteins are degraded and replaced by new ones in order to maintain the tissue homeostasis [2]. This interaction is mainly mediated by cell surface receptors such as integrins. Integrins are a large family of surface receptors that can signal through the cell membrane in either direction [3]. As a consequence, these integrins transmit signals that regulate cell adhesion, migration, proliferation, apoptosis, survival, or differentiation [4–6]. The ECM proteins contain sites responsible for binding to collagen, the most abundant protein and main structural element of the ECM, as well as for the cross-linking to other ECM proteins such elastin and fibronectin, allowing the degradation by proteases. The common feature of fibrotic diseases is a dysregulation of the ECM composition due to an unbalanced chronic wound-healing process, affecting its structure and biophysical properties [7, 8]. As a consequence, scar formation and tissue fibrosis develop [9].
The liver has a high regenerative potential; however, when the damage becomes persistent, this regeneration turns into chronic diseases, such as fibrosis [10, 11]. Liver fibrosis is usually preceded by inflammation, followed by the activation of the main fibrotic cell type in the liver, the hepatic stellate cells (HSCs) [12]. It has become evident that the ECM proteins represent important mediators of the gain-of-function properties of the HSC during the progression of liver fibrosis [13].
Progressive liver fibrosis can be caused by chronic viral hepatitis, alcohol abuse, non-alcoholic steatohepatitis (NASH), autoimmune or cholestatic disorders, and metabolic diseases [14, 15]. Cirrhosis is the terminal stage of progressive liver fibrosis [16–18]. Hepatic fibrosis is characterized by excess accumulation of ECM [19]. Although initially beneficial, the excessive accumulation of several extracellular proteins leads to an unbalance in the wound-healing process, causing fibrosis [20]. It is important to notice that liver fibrosis is not a unidirectional process, which ultimately will lead to organ failure, but is in principle reversible [21]. In order to monitor the progression of liver fibrosis, there is a need to understand the cellular and molecular mechanisms that shift the balance from healthy to fibrotic liver and, therefore, to develop new antifibrotic therapies in the near future. In this review, we discuss the role of the ECM during liver fibrosis, including the main hepatic cell types and molecular pathways involved in this process, as well as the enzymes and ECM components that contribute and regulate the remodeling and the physical properties of ECM.
Cell Types Involved in Liver Fibrosis
Understanding liver fibrosis implies knowing how the ECM proteins change during this process, as well as how the cellular players interact with each other. The main cell types in the liver are hepatocytes, Kupffer cells, HSC, liver sinusoidal endothelial cells (LSECs), and cholangiocytes (Figure 1).
Figure 1.
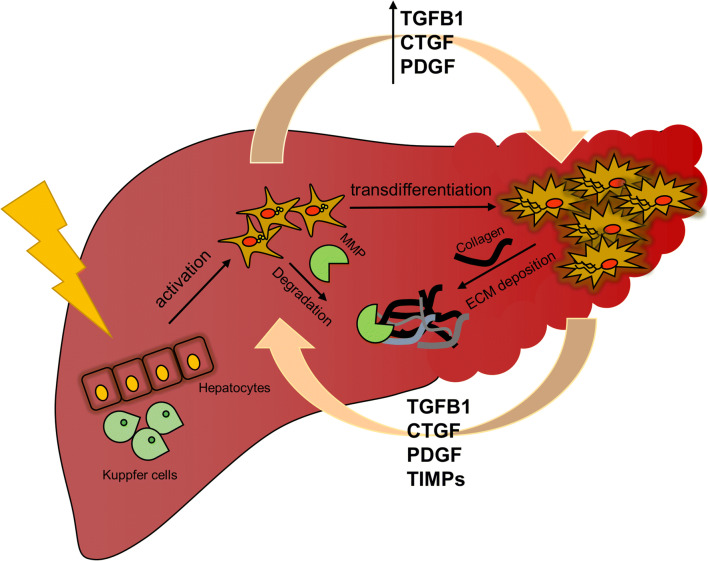
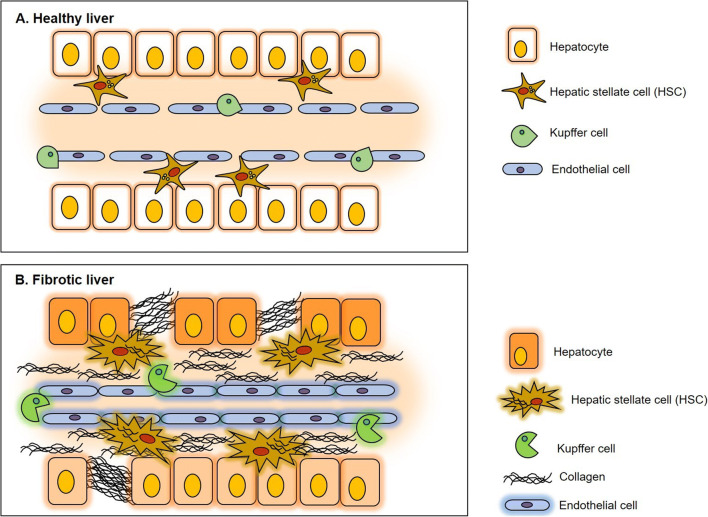
Cellular alterations in liver fibrosis. The main hepatic cell types in the liver are the hepatocytes, the hepatic stellate cells (HSCs), Kupffer cells, and the fenestrated endothelial cells. a In a healthy liver, the space between the hepatocytes and endothelial cells is known as the space of Disse, which are located the HSCs. b Upon injury, the HSCs become activated and secrete a large amount of extracellular matrix (ECM), which results in gradual thickening of the space. The large amount of ECM, mostly collagen, produced by the activated HSCs leads to the loss of hepatocyte and endothelial fenestrations causing an increase in the portal pressure. The imbalance caused by ECM production and the gain-function of the HSCs cause liver fibrosis.
Hepatocytes are the major parenchymal cell type in the liver and account for 80–90% of the cells in the liver. As such, they perform the majority of liver functions, including nutrient metabolism and detoxification [22, 23]. Cholangiocytes are a small epithelial cell population (3–5%) that lines up the bile duct system [24]. Their role involves the secretion and absorption of water, electrolytes, and organic solutes [25, 26]. Kupffer cells are the resident macrophages of the liver and largest resident macrophage population in the body (80–90%) [27, 28]. They have a high endocytic and phagocytic capacity, and they play a crucial homeostatic role in the hepatic immune system [29]. LSECs are fenestrated and form a permeable barrier between the blood and the hepatocytes and HSC which facilitates the passage of molecules from the sinusoidal endothelium to the liver parenchyma and contributes to the maintenance of the cellular and hemodynamic homeostasis [30]. HSCs are resident liver cells located in the so-called space of Disse between LSECs and hepatocytes [31••]. In the healthy liver, HSCs are present in a quiescent state, are a reservoir of retinoic acid (vitamin A), and represent approximately 10% of the liver cell population [31••, 32]. LSECs have an important role in the maintenance of the quiescent state of HSCs [33, 34]. In a healthy liver, tissue homeostasis is maintained by intracellular communication between HSCs, hepatocytes, cholangiocytes, Kupffer cells, and LSECs, mainly via cytokines/chemokines. This mechanism is critical for maintenance of the distinct functions of the liver resident cells [35].
Liver injury disturbs tissue homeostasis and causes cell damage in hepatocytes, LSECs, and cholangiocytes leading to necrosis and the release of inflammatory markers, growth factors, reactive oxygen species, and cytokines [17, 29, 36]. Thus, tissue inflammation promotes the attraction and activation of Kupffer cells and HSCs. Although hepatocytes, Kupffer cells, LSECs, and cholangiocytes can contribute to the fibrogenic processes, the primary drivers of fibrosis are the HSCs. Activated HSCs acquire proliferative, migratory, and contractile properties and contribute to 90% of ECM while they develop a myofibroblast-like phenotype and release inflammatory molecules [37].
Portal fibroblasts are another non-parenchymal hepatic cell population and are similar to hepatic stellate cells in some characteristics, since they are also activated in chronic liver injury and contribute to ECM production in liver fibrosis. However, there are also differences between these two cell types. Portal fibroblasts play an important role in cholangitis, since they are located close to the bile ducts [38]. Hepatic stellate cells and portal fibroblasts can be differentiated by their expression profiles. While hepatic stellate cells highly express desmin and scarcely elastin, portal fibroblasts show the opposite expression pattern [39]. The differences in cell marker expression may be used for the research of portal fibroblasts, which play an inferior role in current research efforts when compared to hepatic stellate cells [38].
ECM Components Implicated in Remodeling
The ECM consists of multiple proteins, such as collagens, elastins, fibronectins, and laminins, that control the cellular phenotype and function [40]. During the transition from healthy to fibrotic liver, the homeostasis between all the ECM proteins and their specific interacting partners is shifted to cause cell inflammation and, finally, contribute to progression of liver fibrosis which in turn is associated with high morbidity and mortality [41].
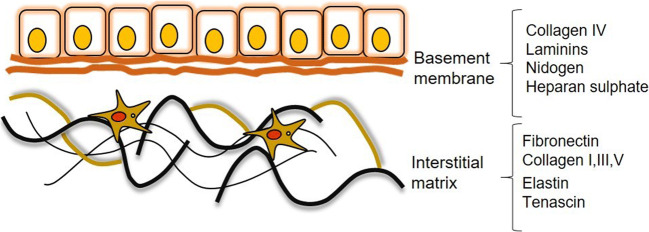
There are two types of ECM [40]. The first is the basement membrane (BM) which separates the epithelium from the mesenchyme and the interstitial matrix (IM), produced by fibroblasts and surrounds cells, making up the bulk of the ECM in the body (Figure 2). Laminins, nidogen/entactin, heparan sulfate proteoglycans, and the non-fibrillar collagens, like collagen type IV, are the most abundant components of the BM [42]. In this regard, BM is a highly specialized type of ECM that serves as a reservoir of growth factors that direct cellular functions, provide cell adhesion, and control cell organization and differentiation [40, 43]. Types I, III, and V are the most abundant fibrillary collagens that form the IM, together with elastin, fibronectin, and tenascin [41, 44•]. Type I collagen is the most abundant collagen that is mainly associated with collagen III [45]. Although type V collagen is also found together with collagen I forming heterofibrils, it is not one of the major components of this complex, but it is essential for the structure of tissue [46]. This last characteristic of collagen V makes it unique, since it has been shown that it enhances the stability of collagen fibrils and its gene expression is regulated by TGF-β, ending up in activation of HSCs [47].
Figure 2.
Schematic representation of the two types of extracellular matrix (ECM). The ECM is divided into the loose basement membrane and a compact interstitial matrix. The basement membrane consists in collagen IV, laminins, nicoden, and heparin sulfate proteoglycans. The interstitial matrix is more compact and contain the fibrillary collagens I, III, and V, elastin, fibronectin and tenascin.
The cross-links of the different collagens are mainly involved in the progression of liver fibrosis in the BM and the IM [48]. Due to this cross-link, an increase of up to tenfold in the collagens I, III, and V is detected [49, 50]. Type IV collagen is also increased during liver fibrogenesis, together with laminin and nidogen [51]. The collagen formation observed during fibrosis favors myofibroblast activation, and, although myofibroblasts are the main ECM-producers, other hepatic cell types, such hepatocytes and macrophages, are important regulators of hepatic fibrogenesis and direct effectors of fibrosis progression. The secretion of pro-fibrotic cytokines, chemokines, growth factors, or signaling peptides derived from collagens has been identified as important regulators during liver fibrosis [52].
Endostatin is a potent signaling peptide derived from the C terminus of collagen XVIII that is located in the BM. Collagen XVIII is primarily produced by hepatocytes and is associated to advanced liver fibrosis [53]. In order to reverse the fibrotic phenotype, it has been shown that endostatins ameliorate fibrosis by inhibiting HSC activation [54].
Type IV collagen fragments, from the BM, have been found to have important signaling properties [55]. Six different collagen IV chains have been described up to now (α1–α6), arrestin, canstatin, tumstatin, tetrastatin, pentastatin, and hexastatin [44•, 56–60]. The first three fragments have been shown to inhibit angiogenesis in liver disease as well as endothelial cell proliferation via inhibition of MAPK pathways signaling and inducing apoptosis in endothelial cells [61]. The remaining type IV collagen chains are more limited in distribution than the first ones [51]. Tetrastatin and pentastatin possess similar anti-angiogenic activity and show inhibition of endothelial cell migration [59]; hexastatin was found to regulate endothelial cell adhesion, migration, and proliferation [62]. All together, these signaling peptides are attractive candidates for potential liver fibrosis therapy.
Restin, a signaling peptide of type XV collagen, another BM collagen, is highly abundant in the portal ECM of the liver [63, 64] and shows inhibitory effects on endothelial cell migration but not on their proliferation when the purified protein was assayed in vitro with different endothelial cell lines [65].
Type VI collagen is a microfibrillar collagen found between the IM and the BM that can stimulate the proliferation of mesenchymal cells [66, 67]. During liver fibrosis, it has been found that collagen VI is up to tenfold induced in liver fibrosis [68], and its signaling peptide endotrophin plays a crucial role in fibrosis. In vivo studies showed that in CCl4-intoxicated mice, endotrophin was upregulated in injured hepatocytes contributing further to their apoptosis. The inflammatory signal released from injured hepatocytes activates HSCs, resulting in further aggravation of liver fibrosis [69, 70].
Lysyl oxidases (LOX) are the one of the family enzymes that modify the ECM [71, 72]. LOX family enzymes catalyze the cross-linking of collagens through oxidative deamination of lysine residues for the maintenance of the tensile strength and structural integrity of the ECM [73]. At least four different LOX-like (LOXL) proteins (LOXL1, LOXL2, LOXL3, and LOXL4) have been described [71]. LOXL proteins are highly controlled during normal tissue development; however, their aberrant expressions have been reported in liver disease [73]. In the liver, HSCs and portal fibroblasts are the main producers of LOX proteins [74]. During liver fibrosis specifically, LOX and LOXL2, which are absent in healthy tissues but strongly induced in liver fibrosis, have been shown to be upregulated, promoting collagen I cross-linking and its stabilization increasing its resistance to proteolytic degradation, maintaining HSC in an activated state [75]. Increased LOX activity has been detected in sera of patients with hepatic diseases [76, 77], suggesting the LOX family of proteins as a potential biomarker for liver fibrosis. Different approaches have been performed in order to inhibit the activity of this enzyme. For instance, inhibition of LOX with b-aminopropionitrile (BAPN), a potent inhibitor of cross-linking enzymes in the LOX family, has been shown to affect collagen cross-linking making the progression of fibrosis more reversible and delaying the effects of CCl4-intoxication [78]. In mouse models of mild liver fibrosis, it has been shown that inhibition of LOXL2 with a specific monoclonal antibody (AB0023) prevents fibrosis [79]. Further analysis using AB0023 showed efficient inhibition of collagen cross-linking, suppression of fibrosis progression [80, 81•].
Transglutaminases (TGs), in addition to LOX, are a well-characterized family of proteins that are able to covalent cross-link several collagen types from the BM and other ECM proteins like fibronectin or nicogen, increasing their resistance to proteolytic degradation, consequently losing tissue functionality [82–84]. TGs are calcium-dependent enzymes that catalyze the cross-linking, transamidation, or deamidation of proteins [85]. TG2 is the most abundant member and the most studied of the nine members of the TG family that is expressed ubiquitously in many types of tissue, cell membrane, nucleus, and extracellular [86]. During liver inflammation and fibrosis, TG2 is tightly associated with soluble integrins and fibronectins in both, covalently and non-covalently manner, promoting the fibronectin deposition into ECM, and forming stable complexes with both fibronectin and integrins [87]. This association stabilizes the integrin-ECM-fibronectin interaction, making the ECM polymers resistant to proteolytic degradation and therefore, contributing to the progression of fibrosis [88, 89]. In addition, it has been shown that TGF-β enhances the binding of integrin-TG2-fibronectin in fibroblasts and elevates ECM formation during fibrosis [90]. To explore the possibility of TG2 as a target to decrease liver fibrosis, TG2 knockout mice after CCl4-induced liver damage did not show a significant decrease in liver fibrosis when compared to wild-type mice; however, these animals were more susceptible to liver inflammation, which suggest a role of TG2 in control of both inflammation and fibrosis progression [91].
Molecular Signaling Pathways Involve in Liver Fibrosis
Growth factors, such as transforming growth factor-β (TGF-β), platelet-derived growth factor (PDGF) and connective tissue growth factor (CTGF), as well as oxidative stress are the most potent mediators of inflammatory signals to induce fibrogenesis.
The transforming growth factor-β (TGF-β) signaling is known as the pathway that most hepatic cell types are susceptible to. Three TGF-β isoforms are known in humans, two are fibrogenic, and one seems to have antagonistic properties. While TGF-β1, the major isoform in the liver, is associated with HSC activation and ECM production in liver fibrosis in general and TGF-β2 is associated with biliary liver disease, TGF-β3 might inhibit TGF-β1 and TGF-β2 expression [92–94]. However, data on the role of TGF-β3 in liver fibrosis is scarce.
TGF-β1 signaling is considered one of the main pathways driving HSC activation and the most potent fibrogenic cytokine in the liver [95, 96]. Furthermore, the correlation of TGF-β1 with the severity of liver fibrogenesis demonstrates its importance for liver fibrosis [97]. TGF-β1 is synthesized as a latent precursor with its prodomain and stored in the ECM as part of a large complex [98]. It is activated by mechanical force to induce the conformational changes of the latent complex and release of active TGF-β. Increased contractility by activated HSCs and increased mechanical resistance by higher liver stiffness due to accumulated ECM are the two necessary components to promote TGF-β activation and release [99]. Integrins transmit the force of actin cytoskeleton contractility to the prodomain of the large latent TGF-β complex in the ECM. The linkage of ECM and cytoskeleton via integrins favors the release of TGF-β from the latent TGF-β binding protein complex [100]. Integrins are composed of two subunits, α and β, and each combination has its own binding specificity and signaling properties [95, 101–103]. Although in general the integrins αvβ3, αvβ5, αvβ6, and αvβ8 can bind this specific sequence of the latent TGF-β1, in liver fibrosis, mainly αvβ3 and αvβ6 play a role [104].
While αvβ6 is barely expressed in normal liver, it is highly expressed in fibrosis [105]. Chemical inhibition of αvβ6 results in downregulation of pro-fibrotic and upregulation of fibrolytic genes in experimental fibrosis [106]. Several studies have shown in vitro that cells expressing αvβ6 integrin activate TGF-β1, and this interaction can be inhibited by blocking the integrins expressed in the myofibroblasts with antibodies, and thus reducing the fibrotic process [104]. Activated HSCs express αvβ3, which seems to regulate cell proliferation [107]. Pharmacological inhibition of this integrin resulted in significant collagen reduction mediated by decreased HSC activity [108].
Once activated, TGF-β1 signals by binding to the transmembrane TGF-β type II receptor (TβRII), which recruits and phosphorylates TGF-β type I receptor (TβRI). This process transmits the extracellular TGF-β1 signal towards the intracellular receptor part where the substrates SMAD2 and SMAD3 can bind. SMAD2/3 is activated by phosphorylation and translocates to the nucleus to regulate the transcription of genes maintaining the fibrotic and contractile state of HSCs [109]. TGF-β activation ultimately drives fibrogenesis and ECM production. Specifically, ECM components fibronectin and collagen types I, III, and IV are regulated by TGF-β1 signaling [97, 110]. Increased activation of HSC mediated by TGF-β1 results in proliferation of activated cells and thereby increases the amount of contractile cells which, in turn, promote activation and release of TGF-β1. Therefore, the interaction between HSC and TGF-β1 in fibrogenesis can lead to a vicious cycle of a paracrine activation of HSC. The situation in portal fibroblasts is contrary to HSC. TGF-β1 and TGF-β2 are also produced by portal fibroblasts [111, 112]. While TGF-β1 and TGF-β2 promote HSC proliferation, they inhibit proliferation of portal fibroblast. Overall, this mechanism might provide a growth advantage to HSC over portal fibroblasts [94].
The platelet-derived growth factor (PDGF) signaling pathway functions include regulation of cellular proliferation, cell migration, and stimulation of synthesis of the major components of the ECMs, such as collagen [113]. During liver fibrosis, the PDGF signaling plays an important role in activating HSCs and portal fibroblasts [114]. Among the four secreted cellular PDGF ligands, A-D, PDGF-B, and –D are the most effective in stimulating HSCs and portal myofibroblast proliferation [115]. The biological effects of PDGF are exerted through its ligands, which bind to their receptors, PDGFR-α and PDGFR-β, and therefore inducing proliferation, migration, and cell survival [116]. It has been shown that HSCs express both PDGF receptors; however, only β is upregulated during its activation in vivo and in vitro specifically in liver injury [114]. In contrast to the β isoform, the α isoform expression remained unchanged after liver injury. Given the importance of PDFG signaling in HSC activation during liver fibrosis, much efforts have been taken in order to pharmacologically inhibit the interaction between the ligand and the receptor [117]. There are multikinase inhibitors, sorafenib, which targets tyrosine kinase associated with PDGFR-β, showing downregulation of collagen expression in the livers of fibrotic rats [118, 119].
The connective tissue growth factor (CTGF) is a multi-functional protein which is highly overexpressed during liver fibrosis. CTGF regulates many cellular functions including increased cell proliferation, differentiation, migration, adhesion, and ECM synthesis, and it plays a direct role by interacting with matrix components [120, 121]. CTGF binds integrins, heparin sulfate proteoglycans, and tyrosine kinase receptors that may be located in the matrix or on the cell surface modulating signal transduction into cells [122, 123]. CTGF is a downstream effector of TGF-β [124], and its inhibition has been shown to suppress the TGF-β-dependent induction of ECM proteins, such as collagen and fibronectin [125, 126]. Activated HSCs are the most important source of CTGF, although hepatocytes, portal fibroblast, and cholangiocytes can also contribute to its production [127, 128]. The role of CTGF in liver fibrosis has been studied in vitro using human biopsies from various chronic liver diseases, as well as in activation of HSCs, showing in both cases a strong correlation of CTGF production during liver fibrosis. These results were further confirmed in vivo in CCl4-treated and bile duct–ligated (BDL) animals to study liver fibrosis [128]. BDL is an experimental model for cholestatic liver disease which mimics the human primary biliary cirrhosis, and CCl4 induces toxic liver fibrosis [129].
Reactive oxygen species (ROS), mediating oxidative stress, are potent pro-fibrotic mediators released mostly by hepatocytes, HSCs and Kupffer cells, that stimulate the production of collagen, acting as a mediator of the fibrogenic action of TGF-β [130, 131]. In liver fibrosis, NADPH oxidases (NOX) are described to be the major sources of ROS [132]. Studies have shown in vivo that NOX1, NOX2, and NOX4 are increased in two models of fibrotic mice, the BDL treatment and the CCl4 intoxication. The knockout of NOX1 and NOX4 in these animals resulted in decreased ECM synthesis and overall ROS production. As a consequence, these knockout animals showed a reduction of liver fibrosis, inflammation, and HSC proliferation [133, 134].
The renin-angiotensin system (RAS) is one of the main drivers of HSC activation [135–137, 138•]. RAS is known to be one of the most complex hormonal systems and interplay among its multiple enzymatic peptide and receptor constituents [139]. In the classical RAS, the enzyme renin cleaves its substrate angiotensinogen forming angiotensin I that is in turn, cleaved by angiotensin-converting enzyme (ACE) to produce the angiotensin II (Ang II), the biologically active peptide of the system [139]. In the classical RAS, Ang II activates the Ang II type I receptor (AT1R) to induce, among other biological processes, vasoconstriction. However, an alternative arm of the RAS cleaves Ang II by ACE homologue ACE2 to Angiotensin- [1–7] and stimulates the proto-oncogen Mas receptor cascade (MasR) causing vasodilation that generally opposes the actions of Ang II via AT1R [140, 141]. It is well-known that Ang II is the central effector that stimulates HSCs to produce pro-fibrotic cytokines, such as TGF-β1 and CTGF, which further increases hepatic resistance to portal flow and enhanced matrix formation [138•, 142••, 143–145]. However, ACE2 is also the major cellular entry receptor for Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) as a cellular receptor to infect alveolar epithelial cells, causing the severe respiratory disease Coronavirus disease 2019 (COVID-19) in humans [146, 147].
Given the high expression of ACE2 in the liver, specifically in cholangiocytes, resident liver cells located in the bile duct, and at lower levels in hepatocytes, it has been found that a significant number of COVID-19 patients showed abnormal liver test results. Using human organoids as a tool to investigate the SARS-CoV-2 infection, it confirmed that cholangiocytes are the liver cells that express the receptor markers for being infected [148]. Downregulation of ACE2 upon binding of SARS-CoV-2 increases Ang II levels and, consequently, ECM synthesis by activated HSCs. Since SARS viruses are already known to promote pulmonary fibrosis [149], it seems very likely that the same mechanisms can lead to fibrogenesis in the liver too. In this regard, a recently published article supports this hypothesis by analysis in different infected patients and showed that the death of cholangiocytes is induced by SARS-CoV-2. As a consequence, hepatocytes release proinflammatory cytokines that can easily produce liver injury [150, 151].
Matrix Stiffness and Inflammation
One of the characteristic events as the liver becomes fibrotic is that the ECM stiffness increases by an extensive deposition of their extracellular proteins, including fibrillar and membrane collagens, affecting the cellular behavior [152]. The concept of stiffness has been analyzed years ago [153] and, nowadays, it is well-known that it can contribute to the mechanical properties of the ECM proteins. Matrix stiffness affects the behavior of the HSCs, including growth, motility, adhesion, and differentiation into myofibroblasts [154].
Fibrosis is associated with an increased matrix stiffness as a consequence of excessive collagen deposition and cross-linking [155]. This stiffness is detected by HSC surface receptors, the integrins, allowing the HSC activation [156]. It has been shown that the ECM components are responsible for increased matrix stiffness and, as a consequence, promotes HSC activation via cytoskeleton modulation [157•]. An important aspect concerning matrix stiffness during liver fibrosis progression is the imbalance of the main enzymes implicated in the ECM degradation, such as the matrix metalloproteinases (MMPs) and their inhibitors. This family of proteins includes the tissue inhibitors of metalloproteinases (TIMPs) [158, 159]. Up to now, 25 different MMPs have been identified that regulate the degradation of most ECM proteins [160]. In a normal liver, MMPs are capable to degrade any protein from the ECM in order to maintain tissue homeostasis [161]. However, when HSCs are activated and collagen accumulates excessively, the matrix regeneration fails, leading to an increase in the stiffness [162]. In this regard, in vitro studies using HSCs cultured on different substrates of varying rigidity, that mimics healthy and fibrotic liver tissue, have shown that increasing fibrotic matrix stiffness downregulates MMP-9 gene expression. As a consequence, this increase in rigidity promotes the secretion of TIMP-1 which inhibits permanently the MMP activity to degrade ECM, promoting fibrosis perpetuation [152]. In addition, other in vitro studies using activated HSCs showed that activation of the HSCs is associated as well with increased MMP-2 and MMP-14 protein expression [163].
Inflammation is a crucial mechanism promoting liver fibrosis by initiation of a protective response to tissue injury [164, 165]. The components of the ECM are the main regulators of tissue inflammation [166]. More specifically, the ECM-derived damage-associated molecular patterns (DAMPs) activate Toll-like receptors (TLRs) and the inflammasome in order to induce tissue inflammation [167, 168]. ECM proteoglycans and their fragments, such as biglycan and decorin, are the most prominent and well-characterized ECM-derived DAMPs [169–171]. Both can interact with TLR2 and TLR4 inducing pro- and anti-inflammatory effects and recruiting macrophages [172, 173]. Macrophages sense changes in matrix stiffness through mechanotransduction and respond by regulating the TLR-mediated inflammatory signal. In this context, the release of the proinflammatory cytokine TNF-α, mainly involved in liver inflammation leading to fibrosis, is increased in response to TLR4 and TLR9 stimulation [174].
Biophysical Mechanisms of ECM Stiffness Sensing
ECM stiffness does not remain unrecognized by adjacent cells. It has been reported that fibroblasts tend to move from softer to stiffer regions [175]. This directed migration is called durotaxis, and the underlying mechanisms are less well-understood than in chemotaxis in which specific cell membrane receptor sense is a gradient in a soluble factor. To detect a gradient in stiffness, cells need to actively apply a pulling force to the substrate. Pulling forces are generated by the actomyosin cytoskeleton, and they can be transmitted to integrins through specific protein complexes called focal adhesions [176]. Focal adhesions contain mechanosensitive proteins such as talin and vinculin. Talin links the cytoskeleton to ECM via intregrins and vinculin shows increased binding to focal adhesion complexes with applied force. The interaction of both proteins is important to sense forces. Under force, the talin structure unfolds to expose multiple binding sites for vinculin. Without an applied force, the talin structure contains less vinculin binding site [177]. The dynamics of the linear physical connection between the actomyosin cytoskeleton and the ECM has been understood through the so-called clutch models [178]. In their simplest form, these models include the deformability of the matrix, the on and off rates of the different integrins and adaptor molecules that connect the actomyosin cytoskeleton and the ECM, and the physical relation that links force and velocity generated by myosin motors [179]. The force generated by the actomyosin cytoskeleton is contractile and results in a flow of actin towards the center of the cell, called retrograde flow. In general, this flow is inversely proportional to the traction force exerted by the cell migrating on the ECM [178].
When actomyosin-generated forces build up at focal adhesions, two main outcomes are possible depending on the rigidity of the substrate [180•]. On soft substrates, force builds up slowly and integrins unbind from the ECM before talin unfolds, resulting in rapid actin retrograde flow. By contrast, on stiff substrates force builds up fast and talin unfolds before integrins unbind, resulting in slow actin flow. Talin unfolding on stiff substrates triggers a reinforcement feedback loop by which vinculin is recruited to focal adhesions. Downstream of vinculin binding to talin, both actin and integrins are recruited to focal adhesions and mediate their reinforcement [180•]. Clutch models thus provide a conceptual framework that explains how rigidity sensing can be tuned through the regulation of vinculin and talin binding rates, activation of mechanochemical switches, and changes in cellular contractility. This mechanism has also been suggested to explain durotaxis, i.e., the ability of single cells and cell collective to migrate from soft to rigid ECM [181].
Concluding Remarks
In this review, we clearly point out the prime importance of the complex interplay between distinct cells and the ECM for health and disease in the liver, where TGF-β occupies an outstanding position as a key mediator of this interaction. ECM not only plays an important role as the framework of the liver, but it also participates actively in the cellular processes as well as in the cell-cell communication. ECM components and properties modulate physiological and pathological processes in the liver. ECM research remains a dynamic field, and recent findings might help to fight contemporary health threats like COVID-19. The complex interaction of ECM with the surrounding cells and compartments needs to be taken into account when designing diagnostic tools and therapeutical antifibrotic strategies.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any original data on human or animal subjects performed by any of the authors, but only review of published evidence.
Footnotes
This article is part of the Topical Collection on Chronic Liver Disease
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
- 1.Bedossa P, Paradis V. Liver extracellular matrix in health and disease. J Pathol. 2003;200(4):504–515. doi: 10.1002/path.1397. [DOI] [PubMed] [Google Scholar]
- 2.Rozario T, DeSimone DW. The extracellular matrix in development and morphogenesis: a dynamic view. Dev Biol. 2010;341(1):126–140. doi: 10.1016/j.ydbio.2009.10.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mezu-Ndubuisi OJ, Maheshwari A. The role of integrins in inflammation and angiogenesis. Pediatr Res. 2020;7:1–8. doi: 10.1038/s41390-020-01177-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bökel C, Brown NH. Integrins in development: moving on, responding to, and sticking to the extracellular matrix. Dev Cell. 2002;3(3):311–321. doi: 10.1016/S1534-5807(02)00265-4. [DOI] [PubMed] [Google Scholar]
- 5.Bonnans C, Chou J, Werb Z. Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol. 2014;15(12):786–801. doi: 10.1038/nrm3904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Harburger DS, Calderwood DA. Integrin signalling at a glance. J Cell Sci. 2009;122(Pt 2):159–163. doi: 10.1242/jcs.018093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rockey DC, Bell PD, Hill JA. Fibrosis--a common pathway to organ injury and failure. N Engl J Med. 2015;373(1):96. doi: 10.1056/NEJMc1504848. [DOI] [PubMed] [Google Scholar]
- 8.Walraven M, Hinz B. Therapeutic approaches to control tissue repair and fibrosis: extracellular matrix as a game changer. Matrix Biol. 2018;71–72:205–224. doi: 10.1016/j.matbio.2018.02.020. [DOI] [PubMed] [Google Scholar]
- 9.Harvey A, Montezano AC, Lopes RA, Rios F, Touyz RM. Vascular fibrosis in aging and hypertension: molecular mechanisms and clinical implications. Can J Cardiol. 2016;32(5):659–668. doi: 10.1016/j.cjca.2016.02.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cordero-Espinoza L, Huch M. The balancing act of the liver: tissue regeneration versus fibrosis. J Clin Invest. 2018 Jan 2;128(1):85–96. [DOI] [PMC free article] [PubMed]
- 11.Michalopoulos GK. Liver regeneration. J Cell Physiol. 2007;213(2):286–300. doi: 10.1002/jcp.21172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Reeves HL, Friedman SL. Activation of hepatic stellate cells--a key issue in liver fibrosis. Front Biosci. 2002;7:d808–d826. doi: 10.2741/reeves. [DOI] [PubMed] [Google Scholar]
- 13.Puche JE, Saiman Y, Friedman SL. Hepatic stellate cells and liver fibrosis. Compr Physiol. 2013;3(4):1473–1492. doi: 10.1002/cphy.c120035. [DOI] [PubMed] [Google Scholar]
- 14.Hernandez-Gea V, Friedman SL. Pathogenesis of liver fibrosis. Annu Rev Pathol. 2011;6:425–456. doi: 10.1146/annurev-pathol-011110-130246. [DOI] [PubMed] [Google Scholar]
- 15.Wiegand J, Berg T. The etiology, diagnosis and prevention of liver cirrhosis: part 1 of a series on liver cirrhosis. Dtsch Arztebl Int. 2013;110(6):85–91. doi: 10.3238/arztebl.2013.0085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Koyama Y, Brenner DA. Liver inflammation and fibrosis. J Clin Invest. 2017;127(1):55–64. doi: 10.1172/JCI88881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pellicoro A, Ramachandran P, Iredale JP, Fallowfield JA. Liver fibrosis and repair: immune regulation of wound healing in a solid organ. Nat Rev Immunol. 2014;14(3):181–194. doi: 10.1038/nri3623. [DOI] [PubMed] [Google Scholar]
- 18.Schuppan D, Afdhal NH. Liver cirrhosis. Lancet. 2008;371(9615):838–851. doi: 10.1016/S0140-6736(08)60383-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee UE, Friedman SL. Mechanisms of hepatic fibrogenesis. Best Pract Res Clin Gastroenterol. 2011;25(2):195–206. doi: 10.1016/j.bpg.2011.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Iredale JP, Thompson A, Henderson NC. Extracellular matrix degradation in liver fibrosis: biochemistry and regulation. Biochim Biophys Acta (BBA) - Mol Basis Dis. 2013;1832(7):876–883. doi: 10.1016/j.bbadis.2012.11.002. [DOI] [PubMed] [Google Scholar]
- 21.Iredale JP, Benyon RC, Pickering J, McCullen M, Northrop M, Pawley S, Hovell C, Arthur MJ. Mechanisms of spontaneous resolution of rat liver fibrosis. Hepatic stellate cell apoptosis and reduced hepatic expression of metalloproteinase inhibitors. J Clin Invest. 1998;102(3):538–549. doi: 10.1172/JCI1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Schulze RJ, Schott MB, Casey CA, Tuma PL, McNiven MA. The cell biology of the hepatocyte: a membrane trafficking machine. J Cell Biol. 2019;218(7):2096–2112. doi: 10.1083/jcb.201903090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Treyer A, Müsch A. Hepatocyte polarity. Compr Physiol. 2013;3(1):243–287. doi: 10.1002/cphy.c120009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.LeSage G, Glaser S, Alpini G. Regulation of cholangiocyte proliferation. Liver. 2001;21(2):73–80. doi: 10.1034/j.1600-0676.2001.021002073.x. [DOI] [PubMed] [Google Scholar]
- 25.Banales JM, Huebert RC, Karlsen T, Strazzabosco M, LaRusso NF, Gores GJ. Cholangiocyte pathobiology. Nat Rev Gastroenterol Hepatol. 2019;16(5):269–281. doi: 10.1038/s41575-019-0125-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tam PKH, Yiu RS, Lendahl U, Andersson ER. Cholangiopathies - Towards a molecular understanding. EBioMedicine. 2018;35:381–393. doi: 10.1016/j.ebiom.2018.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Blériot C, Ginhoux F. Understanding the heterogeneity of resident liver macrophages. Front Immunol. 2019;10:2694. doi: 10.3389/fimmu.2019.02694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Dixon LJ, Barnes M, Tang H, Pritchard MT, Nagy LE. Kupffer cells in the liver. Compr Physiol. 2013;3(2):785–797. doi: 10.1002/cphy.c120026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Vollmar B, Menger MD. The hepatic microcirculation: mechanistic contributions and therapeutic targets in liver injury and repair. Physiol Rev. 2009;89(4):1269–1339. doi: 10.1152/physrev.00027.2008. [DOI] [PubMed] [Google Scholar]
- 30.Rockey D. The cellular pathogenesis of portal hypertension: stellate cell contractility, endothelin, and nitric oxide. Hepatology. 1997;25(1):2–5. doi: 10.1002/hep.510250102. [DOI] [PubMed] [Google Scholar]
- 31.Friedman SL. Hepatic stellate cells: protean, multifunctional, and enigmatic cells of the liver. Physiol Rev. 2008;88(1):125–172. doi: 10.1152/physrev.00013.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wake K. ‘Sternzellen’ in the liver: perisinusoidal cells with special reference to storage of vitamin A. Am J Anat. 1971;132(4):429–462. doi: 10.1002/aja.1001320404. [DOI] [PubMed] [Google Scholar]
- 33.Shetty S, Lalor PF, Adams DH. Liver sinusoidal endothelial cells - gatekeepers of hepatic immunity. Nat Rev Gastroenterol Hepatol. 2018;15(9):555–567. doi: 10.1038/s41575-018-0020-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sørensen KK, Simon-Santamaria J, McCuskey RS, Smedsrød B. Liver sinusoidal endothelial cells. Compr Physiol. 2015;5(4):1751–1774. doi: 10.1002/cphy.c140078. [DOI] [PubMed] [Google Scholar]
- 35.Heymann F, Tacke F. Immunology in the liver--from homeostasis to disease. Nat Rev Gastroenterol Hepatol. 2016;13(2):88–110. doi: 10.1038/nrgastro.2015.200. [DOI] [PubMed] [Google Scholar]
- 36.Jenne CN, Kubes P. Immune surveillance by the liver. Nat Immunol. 2013;14(10):996–1006. doi: 10.1038/ni.2691. [DOI] [PubMed] [Google Scholar]
- 37.Mederacke I, Hsu CC, Troeger JS, Huebener P, Mu X, Dapito DH, Pradere JP, Schwabe RF. Fate tracing reveals hepatic stellate cells as dominant contributors to liver fibrosis independent of its aetiology. Nat Commun. 2013;4:2823. doi: 10.1038/ncomms3823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dranoff JA, Wells RG. Portal fibroblasts: underappreciated mediators of biliary fibrosis. Hepatology. 2010;51(4):1438–1444. doi: 10.1002/hep.23405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Li Z, Dranoff JA, Chan EP, Uemura M, Sévigny J, Wells RG. Transforming growth factor-β and substrate stiffness regulate portal fibroblast activation in culture. Hepatology. 2007;46(4):1246–1256. doi: 10.1002/hep.21792. [DOI] [PubMed] [Google Scholar]
- 40.Karsdal MA, Nielsen MJ, Sand JM, Henriksen K, Genovese F, Bay-Jensen A-C, Smith V, Adamkewicz JI, Christiansen C, Leeming DJ. Extracellular matrix remodeling: the common denominator in connective tissue diseases. Possibilities for evaluation and current understanding of the matrix as more than a passive architecture, but a key player in tissue failure. Assay Drug Dev Technol. 2013;11(2):70–92. doi: 10.1089/adt.2012.474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Karsdal MA, Manon-Jensen T, Genovese F, Kristensen JH, Nielsen MJ, Sand JMB, Hansen NUB, Bay-Jensen AC, Bager CL, Krag A, Blanchard A, Krarup H, Leeming DJ, Schuppan D. Novel insights into the function and dynamics of extracellular matrix in liver fibrosis. Am J Physiol Gastrointest Liver Physiol. 2015;308(10):G807–G830. doi: 10.1152/ajpgi.00447.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Pozzi A, Yurchenco PD, Iozzo RV. The nature and biology of basement membranes. Matrix Biol. 2017;57–58:1–11. doi: 10.1016/j.matbio.2016.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yurchenco PD. Basement membranes: cell scaffoldings and signaling platforms. Cold Spring Harb Perspect Biol. 2011;1:3(2). doi: 10.1101/cshperspect.a004911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44•.Karsdal MA, Nielsen SH, Leeming DJ, Langholm LL, Nielsen MJ, Manon-Jensen T, et al. The good and the bad collagens of fibrosis - their role in signaling and organ function. Adv Drug Deliv Rev. 2017;121:43–56. doi: 10.1016/j.addr.2017.07.014. [DOI] [PubMed] [Google Scholar]
- 45.Hinz B, Phan SH, Thannickal VJ, Galli A, Bochaton-Piallat M-L, Gabbiani G. The myofibroblast: one function, multiple origins. Am J Pathol. 2007;170(6):1807–1816. doi: 10.2353/ajpath.2007.070112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gelse K, Pöschl E, Aigner T. Collagens--structure, function, and biosynthesis. Adv Drug Deliv Rev. 2003;55(12):1531–1546. doi: 10.1016/j.addr.2003.08.002. [DOI] [PubMed] [Google Scholar]
- 47.Mak KM, Png CYM, Lee DJ. Type V collagen in health, disease, and fibrosis. Anat Rec (Hoboken) 2016;299(5):613–629. doi: 10.1002/ar.23330. [DOI] [PubMed] [Google Scholar]
- 48.Brown B, Lindberg K, Reing J, Stolz DB, Badylak SF. The basement membrane component of biologic scaffolds derived from extracellular matrix. Tissue Eng. 2006;12(3):519–526. doi: 10.1089/ten.2006.12.519. [DOI] [PubMed] [Google Scholar]
- 49.MacKenna D, Summerour SR, Villarreal FJ. Role of mechanical factors in modulating cardiac fibroblast function and extracellular matrix synthesis. Cardiovasc Res. 2000;46(2):257–263. doi: 10.1016/S0008-6363(00)00030-4. [DOI] [PubMed] [Google Scholar]
- 50.Rojkind M, Giambrone MA, Biempica L. Collagen types in normal and cirrhotic liver. Gastroenterology. 1979;76(4):710–719. doi: 10.1016/S0016-5085(79)80170-5. [DOI] [PubMed] [Google Scholar]
- 51.Hudson BG, Tryggvason K, Sundaramoorthy M, Neilson EG. Alport’s syndrome, Goodpasture’s syndrome, and type IV collagen. N Engl J Med. 2003;348(25):2543–2556. doi: 10.1056/NEJMra022296. [DOI] [PubMed] [Google Scholar]
- 52.Wynn TA. Cellular and molecular mechanisms of fibrosis. J Pathol. 2008;214(2):199–210. doi: 10.1002/path.2277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Schuppan D, Cramer T, Bauer M, Strefeld T, Hahn EG, Herbst H. Hepatocytes as a source of collagen type XVIII endostatin. Lancet. 1998;352(9131):879–880. doi: 10.1016/S0140-6736(05)60006-2. [DOI] [PubMed] [Google Scholar]
- 54.Ren H, Li Y, Chen Y, Wang L. Endostatin attenuates PDGF-BB- or TGF-β1-induced HSCs activation via suppressing RhoA/ROCK1 signal pathways. Drug Des Devel Ther. 2019;13:285–290. doi: 10.2147/DDDT.S191617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zhou J, Ding M, Zhao Z, Reeders ST. Complete primary structure of the sixth chain of human basement membrane collagen, alpha 6(IV). Isolation of the cDNAs for alpha 6(IV) and comparison with five other type IV collagen chains. J Biol Chem. 1994;269(18):13193–13199. doi: 10.1016/S0021-9258(17)36818-7. [DOI] [PubMed] [Google Scholar]
- 56.Kamphaus GD, Colorado PC, Panka DJ, Hopfer H, Ramchandran R, Torre A, Maeshima Y, Mier JW, Sukhatme VP, Kalluri R. Canstatin, a novel matrix-derived inhibitor of angiogenesis and tumor growth. J Biol Chem. 2000;275(2):1209–1215. doi: 10.1074/jbc.275.2.1209. [DOI] [PubMed] [Google Scholar]
- 57.Hamano Y, Zeisberg M, Sugimoto H, Lively JC, Maeshima Y, Yang C, Hynes RO, Werb Z, Sudhakar A, Kalluri R. Physiological levels of tumstatin, a fragment of collagen IV alpha3 chain, are generated by MMP-9 proteolysis and suppress angiogenesis via alphaV beta3 integrin. Cancer Cell. 2003;3(6):589–601. doi: 10.1016/S1535-6108(03)00133-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Colorado PC, Torre A, Kamphaus G, Maeshima Y, Hopfer H, Takahashi K, Volk R, Zamborsky ED, Herman S, Sarkar PK, Ericksen MB, Dhanabal M, Simons M, Post M, Kufe DW, Weichselbaum RR, Sukhatme VP, Kalluri R. Anti-angiogenic cues from vascular basement membrane collagen. Cancer Res. 2000;60(9):2520–2526. [PubMed] [Google Scholar]
- 59.Karagiannis ED, Popel AS. Identification of novel short peptides derived from the alpha 4, alpha 5, and alpha 6 fibrils of type IV collagen with anti-angiogenic properties. Biochem Biophys Res Commun. 2007;354(2):434–439. doi: 10.1016/j.bbrc.2006.12.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mundel TM, Kalluri R. Type IV collagen-derived angiogenesis inhibitors. Microvasc Res. 2007;74(2–3):85–89. doi: 10.1016/j.mvr.2007.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Cooke VG, Kalluri R. Chapter 1. Molecular mechanism of type IV collagen-derived endogenous inhibitors of angiogenesis. Methods Enzymol. 2008;444:1–19. doi: 10.1016/S0076-6879(08)02801-2. [DOI] [PubMed] [Google Scholar]
- 62.Sund M, Nyberg P, Eikesdal HP. Endogenous matrix-derived inhibitors of angiogenesis. Pharmaceuticals (Basel) 2010;3(10):3021–3039. doi: 10.3390/ph3103021. [DOI] [Google Scholar]
- 63.Hägg PM, Hägg PO, Peltonen S, Autio-Harmainen H, Pihlajaniemi T. Location of type XV collagen in human tissues and its accumulation in the interstitial matrix of the fibrotic kidney. Am J Pathol. 1997;150(6):2075–2086. [PMC free article] [PubMed] [Google Scholar]
- 64.Harrison SA, Rossi SJ, Paredes AH, Trotter JF, Bashir MR, Guy CD, Banerjee R, Jaros MJ, Owers S, Baxter BA, Ling L, DePaoli AM. NGM282 improves liver fibrosis and histology in 12 weeks in patients with nonalcoholic steatohepatitis. Hepatology. 2020;71(4):1198–1212. doi: 10.1002/hep.30590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ramchandran R, Dhanabal M, Volk R, Waterman MJ, Segal M, Lu H, et al. Antiangiogenic activity of restin, NC10 domain of human collagen XV: comparison to endostatin. Biochem Biophys Res Commun. 1999;255(3):735–739. doi: 10.1006/bbrc.1999.0248. [DOI] [PubMed] [Google Scholar]
- 66.Atkinson JC, Rühl M, Becker J, Ackermann R, Schuppan D. Collagen VI regulates normal and transformed mesenchymal cell proliferation in vitro. Exp Cell Res. 1996;228(2):283–291. doi: 10.1006/excr.1996.0328. [DOI] [PubMed] [Google Scholar]
- 67.Villesen IF, Daniels SJ, Leeming DJ, Karsdal MA, Nielsen MJ. Review article: the signalling and functional role of the extracellular matrix in the development of liver fibrosis. Aliment Pharmacol Ther. 2020;52(1):85–97. doi: 10.1111/apt.15773. [DOI] [PubMed] [Google Scholar]
- 68.Schuppan D, Rühlmann T, Hahn EG. Radioimmunoassay for human type VI collagen and its application to tissue and body fluids. Anal Biochem. 1985;149(1):238–247. doi: 10.1016/0003-2697(85)90501-9. [DOI] [PubMed] [Google Scholar]
- 69.Karsdal M, Reese-Petersen A, Nielsen MJ, Krag A, Thiele M, Gudmann N, et al. The pro-peptide of type VI collagen (endotrophin) is equipotent to Tgf-B in inducing fibrosis: identification of a self-perpetuating vicious cycle driving fibrosis mediated by collagen signals. In: Hepatology [Internet]. JohnWiley & Sons, Inc.; 2019;84A–85A. Availablefrom: https://portal.findresearcher.sdu.dk/en/publications/the-pro-peptide-of-type-vi-collagen-endotrophin-is-equipotent-to-. Accessed 16 Dec 2020.
- 70.Lee C, Kim M, Lee JH, Oh J, Shin H-H, Lee SM, Scherer PE, Kwon HM, Choi JH, Park J. COL6A3-derived endotrophin links reciprocal interactions among hepatic cells in the pathology of chronic liver disease. J Pathol. 2019;247(1):99–109. doi: 10.1002/path.5172. [DOI] [PubMed] [Google Scholar]
- 71.Csiszar K. Lysyl oxidases: a novel multifunctional amine oxidase family. Prog Nucleic Acid Res Mol Biol. 2001;70:1–32. doi: 10.1016/S0079-6603(01)70012-8. [DOI] [PubMed] [Google Scholar]
- 72.Lucero HA, Kagan HM. Lysyl oxidase: an oxidative enzyme and effector of cell function. Cell Mol Life Sci. 2006;63(19–20):2304–2316. doi: 10.1007/s00018-006-6149-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Kagan HM. Lysyl oxidase: mechanism, regulation and relationship to liver fibrosis. Pathol Res Pract. 1994;190(9–10):910–919. doi: 10.1016/S0344-0338(11)80995-7. [DOI] [PubMed] [Google Scholar]
- 74.Perepelyuk M, Terajima M, Wang AY, Georges PC, Janmey PA, Yamauchi M, Wells RG. Hepatic stellate cells and portal fibroblasts are the major cellular sources of collagens and lysyl oxidases in normal liver and early after injury. Am J Physiol Gastrointest Liver Physiol. 2013;304(6):G605–G614. doi: 10.1152/ajpgi.00222.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Kagan HM, Li W. Lysyl oxidase: properties, specificity, and biological roles inside and outside of the cell. J Cell Biochem. 2003;88(4):660–672. doi: 10.1002/jcb.10413. [DOI] [PubMed] [Google Scholar]
- 76.Mesarwi OA, Shin M-K, Drager LF, Bevans-Fonti S, Jun JC, Putcha N, Torbenson MS, Pedrosa RP, Lorenzi-Filho G, Steele KE, Schweitzer MA, Magnuson TH, Lidor AO, Schwartz AR, Polotsky VY. Lysyl oxidase as a serum biomarker of liver fibrosis in patients with severe obesity and obstructive sleep apnea. Sleep. 2015;38(10):1583–1591. doi: 10.5665/sleep.5052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Murawaki Y, Kusakabe Y, Hirayama C. Serum lysyl oxidase activity in chronic liver disease in comparison with serum levels of prolyl hydroxylase and laminin. Hepatology. 1991;14(6):1167–1173. doi: 10.1002/hep.1840140635. [DOI] [PubMed] [Google Scholar]
- 78.Liu SB, Ikenaga N, Peng Z-W, Sverdlov DY, Greenstein A, Smith V, Schuppan D, Popov Y. Lysyl oxidase activity contributes to collagen stabilization during liver fibrosis progression and limits spontaneous fibrosis reversal in mice. FASEB J. 2016;30(4):1599–1609. doi: 10.1096/fj.14-268425. [DOI] [PubMed] [Google Scholar]
- 79.Barry-Hamilton V, Spangler R, Marshall D, McCauley S, Rodriguez HM, Oyasu M, Mikels A, Vaysberg M, Ghermazien H, Wai C, Garcia CA, Velayo AC, Jorgensen B, Biermann D, Tsai D, Green J, Zaffryar-Eilot S, Holzer A, Ogg S, Thai D, Neufeld G, van Vlasselaer P, Smith V. Allosteric inhibition of lysyl oxidase–like-2 impedes the development of a pathologic microenvironment. Nat Med. 2010;16(9):1009–1017. doi: 10.1038/nm.2208. [DOI] [PubMed] [Google Scholar]
- 80.Ikenaga N, Peng Z-W, Vaid KA, Liu SB, Yoshida S, Sverdlov DY, Mikels-Vigdal A, Smith V, Schuppan D, Popov YV. Selective targeting of lysyl oxidase-like 2 (LOXL2) suppresses hepatic fibrosis progression and accelerates its reversal. Gut. 2017;66(9):1697–1708. doi: 10.1136/gutjnl-2016-312473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Magdaleno F, Schierwagen R, Uschner FE, Trebicka J. ‘Tipping’ extracellular matrix remodeling towards regression of liver fibrosis: novel concepts. Minerva Gastroenterol Dietol. 2018;64(1):51–61. doi: 10.23736/S1121-421X.17.02442-4. [DOI] [PubMed] [Google Scholar]
- 82.Brown-Augsburger P, Broekelmann T, Mecham L, Mercer R, Gibson MA, Cleary EG, Abrams WR, Rosenbloom J, Mecham RP. Microfibril-associated glycoprotein binds to the carboxyl-terminal domain of tropoelastin and is a substrate for transglutaminase. J Biol Chem. 1994;269(45):28443–28449. doi: 10.1016/S0021-9258(18)46947-5. [DOI] [PubMed] [Google Scholar]
- 83.Aeschlimann D, Paulsson M. Transglutaminases: protein cross-linking enzymes in tissues and body fluids. Thromb Haemost. 1994;71(4):402–415. doi: 10.1055/s-0038-1642451. [DOI] [PubMed] [Google Scholar]
- 84.Kadler KE, Hill A, Canty-Laird EG. Collagen fibrillogenesis: fibronectin, integrins, and minor collagens as organizers and nucleators. Curr Opin Cell Biol. 2008;20(5):495–501. doi: 10.1016/j.ceb.2008.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Griffin M, Casadio R, Bergamini CM. Transglutaminases: nature’s biological glues. Biochem J. 2002;368(Pt 2):377–396. doi: 10.1042/bj20021234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Odii BO, Coussons P. Biological functionalities of transglutaminase 2 and the possibility of its compensation by other members of the transglutaminase family. ScientificWorldJournal [Internet]. 2014 Mar 23. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3981525/. Accessed 25 Nov 2020. [DOI] [PMC free article] [PubMed]
- 87.Belkin AM. Extracellular TG2: emerging functions and regulation. FEBS J. 2011;278(24):4704–4716. doi: 10.1111/j.1742-4658.2011.08346.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Lorand L, Graham RM. Transglutaminases: crosslinking enzymes with pleiotropic functions. Nat Rev Mol Cell Biol. 2003;4(2):140–156. doi: 10.1038/nrm1014. [DOI] [PubMed] [Google Scholar]
- 89.Zemskov EA, Janiak A, Hang J, Waghray A, Belkin AM. The role of tissue transglutaminase in cell-matrix interactions. Front Biosci. 2006;11:1057–1076. doi: 10.2741/1863. [DOI] [PubMed] [Google Scholar]
- 90.Elli L, Bergamini CM, Bardella MT, Schuppan D. Transglutaminases in inflammation and fibrosis of the gastrointestinal tract and the liver. Dig Liver Dis. 2009;41(8):541–550. doi: 10.1016/j.dld.2008.12.095. [DOI] [PubMed] [Google Scholar]
- 91.Popov Y, Sverdlov DY, Sharma AK, Bhaskar KR, Li S, Freitag TL, Lee J, Dieterich W, Melino G, Schuppan D. Tissue transglutaminase does not affect fibrotic matrix stability or regression of liver fibrosis in mice. Gastroenterology. 2011;140(5):1642–1652. doi: 10.1053/j.gastro.2011.01.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wang L, Tu L, Zhang J, Xu K, Qian W. Stellate cell activation and imbalanced expression of TGF-β1/TGF-β3 in acute autoimmune liver lesions induced by ConA in mice. Biomed Res Int [Internet]. 2017. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5303577/. Accessed 24 Mar 2021. [DOI] [PMC free article] [PubMed]
- 93.Fabregat I, Moreno-Càceres J, Sánchez A, Dooley S, Dewidar B, Giannelli G, ten Dijke P, the IT-LIVER Consortium TGF-β signalling and liver disease. FEBS J. 2016;283(12):2219–2232. doi: 10.1111/febs.13665. [DOI] [PubMed] [Google Scholar]
- 94.Wells RG, Kruglov E, Dranoff JA. Autocrine release of TGF-beta by portal fibroblasts regulates cell growth. FEBS Lett. 2004;559(1–3):107–110. doi: 10.1016/S0014-5793(04)00037-7. [DOI] [PubMed] [Google Scholar]
- 95.Dewidar B, Meyer C, Dooley S, Meindl-Beinker AN. TGF-β in hepatic stellate cell activation and liver fibrogenesis-updated Cells. 2019 Nov 11;8(11):1419. [DOI] [PMC free article] [PubMed]
- 96.Inagaki Y, Okazaki I. Emerging insights into transforming growth factor beta Smad signal in hepatic fibrogenesis. Gut. 2007;56(2):284–292. doi: 10.1136/gut.2005.088690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kanzler S, Lohse AW, Keil A, Henninger J, Dienes HP, Schirmacher P, Rose-John S, zum Büschenfelde K, Blessing M. TGF-beta1 in liver fibrosis: an inducible transgenic mouse model to study liver fibrogenesis. Am J Phys. 1999;276(4):G1059–G1068. doi: 10.1152/ajpgi.1999.276.4.G1059. [DOI] [PubMed] [Google Scholar]
- 98.Shi M, Zhu J, Wang R, Chen X, Mi L, Walz T, Springer TA. Latent TGF-β structure and activation. Nature. 2011;474(7351):343–349. doi: 10.1038/nature10152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Hinz B. The extracellular matrix and transforming growth factor-β1: tale of a strained relationship. Matrix Biol. 2015;47:54–65. doi: 10.1016/j.matbio.2015.05.006. [DOI] [PubMed] [Google Scholar]
- 100.Conroy KP, Kitto LJ, Henderson NC. αv integrins: key regulators of tissue fibrosis. Cell Tissue Res. 2016;365(3):511–519. doi: 10.1007/s00441-016-2407-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Fan W, Liu T, Chen W, Hammad S, Longerich T, Hausser I, et al. ECM1 prevents activation of transforming growth factor β, hepatic stellate cells, and fibrogenesis in mice. Gastroenterology. 2019;157(5):1352–1367.e13. doi: 10.1053/j.gastro.2019.07.036. [DOI] [PubMed] [Google Scholar]
- 102.Nishimura SL. Integrin-mediated transforming growth factor-beta activation, a potential therapeutic target in fibrogenic disorders. Am J Pathol. 2009;175(4):1362–1370. doi: 10.2353/ajpath.2009.090393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Giancotti FG, Ruoslahti E. Integrin signaling. Science. 1999;285(5430):1028–1032. doi: 10.1126/science.285.5430.1028. [DOI] [PubMed] [Google Scholar]
- 104.Henderson NC, Sheppard D. Integrin-mediated regulation of TGFβ in fibrosis. Biochim Biophys Acta (BBA) - Mol Basis Dis. 2013;1832(7):891–896. doi: 10.1016/j.bbadis.2012.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Popov Y, Patsenker E, Stickel F, Zaks J, Bhaskar KR, Niedobitek G, Kolb A, Friess H, Schuppan D. Integrin alphavbeta6 is a marker of the progression of biliary and portal liver fibrosis and a novel target for antifibrotic therapies. J Hepatol. 2008;48(3):453–464. doi: 10.1016/j.jhep.2007.11.021. [DOI] [PubMed] [Google Scholar]
- 106.Patsenker E, Popov Y, Stickel F, Jonczyk A, Goodman SL, Schuppan D. Inhibition of integrin alphavbeta6 on cholangiocytes blocks transforming growth factor-beta activation and retards biliary fibrosis progression. Gastroenterology. 2008;135(2):660–670. doi: 10.1053/j.gastro.2008.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Zhou X, Murphy FR, Gehdu N, Zhang J, Iredale JP, Benyon RC. Engagement of alphavbeta3 integrin regulates proliferation and apoptosis of hepatic stellate cells. J Biol Chem. 2004;279(23):23996–24006. doi: 10.1074/jbc.M311668200. [DOI] [PubMed] [Google Scholar]
- 108.Patsenker E, Popov Y, Stickel F, Schneider V, Ledermann M, Sägesser H, Niedobitek G, Goodman SL, Schuppan D. Pharmacological inhibition of integrin alphavbeta3 aggravates experimental liver fibrosis and suppresses hepatic angiogenesis. Hepatology. 2009;50(5):1501–1511. doi: 10.1002/hep.23144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Derynck R, Budi EH. Specificity, versatility, and control of TGF-β family signaling. Sci Signal. 2019 Feb26;12(570):eaav5183. [DOI] [PMC free article] [PubMed]
- 110.Knittel T, Janneck T, Müller L, Fellmer P, Ramadori G. Transforming growth factor beta 1-regulated gene expression of Ito cells. Hepatology. 1996;24(2):352–360. doi: 10.1002/hep.510240211. [DOI] [PubMed] [Google Scholar]
- 111.Liu C, Gaça MDA, Swenson ES, Vellucci VF, Reiss M, Wells RG. Smads 2 and 3 are differentially activated by transforming growth factor-beta (TGF-beta) in quiescent and activated hepatic stellate cells. Constitutive nuclear localization of Smads in activated cells is TGF-beta-independent. J Biol Chem. 2003;278(13):11721–11728. doi: 10.1074/jbc.M207728200. [DOI] [PubMed] [Google Scholar]
- 112.Milani S, Herbst H, Schuppan D, Stein H, Surrenti C. Transforming growth factors beta 1 and beta 2 are differentially expressed in fibrotic liver disease. Am J Pathol. 1991;139(6):1221–1229. [PMC free article] [PubMed] [Google Scholar]
- 113.Heldin C-H, Westermark B. Mechanism of action and in vivo role of platelet-derived growth factor. Physiol Rev. 1999;79(4):1283–1316. doi: 10.1152/physrev.1999.79.4.1283. [DOI] [PubMed] [Google Scholar]
- 114.Wong L, Yamasaki G, Johnson RJ, Friedman SL. Induction of beta-platelet-derived growth factor receptor in rat hepatic lipocytes during cellular activation in vivo and in culture. J Clin Invest. 1994;94(4):1563–1569. doi: 10.1172/JCI117497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ying H-Z, Chen Q, Zhang W-Y, Zhang H-H, Ma Y, Zhang S-Z, Fang J, Yu CH. PDGF signaling pathway in hepatic fibrosis pathogenesis and therapeutics. Mol Med Rep. 2017;16(6):7879–7889. doi: 10.3892/mmr.2017.7641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Kelly JD, Haldeman BA, Grant FJ, Murray MJ, Seifert RA, Bowen-Pope DF, Cooper JA, Kazlauskas A. Platelet-derived growth factor (PDGF) stimulates PDGF receptor subunit dimerization and intersubunit trans-phosphorylation. J Biol Chem. 1991;266(14):8987–8992. doi: 10.1016/S0021-9258(18)31541-2. [DOI] [PubMed] [Google Scholar]
- 117.Borkham-Kamphorst E, Stoll D, Gressner AM, Weiskirchen R. Antisense strategy against PDGF B-chain proves effective in preventing experimental liver fibrogenesis. Biochem Biophys Res Commun. 2004;321(2):413–423. doi: 10.1016/j.bbrc.2004.06.153. [DOI] [PubMed] [Google Scholar]
- 118.Ogawa S, Ochi T, Shimada H, Inagaki K, Fujita I, Nii A, Moffat MA, Katragadda M, Violand BN, Arch RH, Masferrer JL. Anti-PDGF-B monoclonal antibody reduces liver fibrosis development. Hepatol Res. 2010;40(11):1128–1141. doi: 10.1111/j.1872-034X.2010.00718.x. [DOI] [PubMed] [Google Scholar]
- 119.Wang Y, Gao J, Zhang D, Zhang J, Ma J, Jiang H. New insights into the antifibrotic effects of sorafenib on hepatic stellate cells and liver fibrosis. J Hepatol. 2010;53(1):132–144. doi: 10.1016/j.jhep.2010.02.027. [DOI] [PubMed] [Google Scholar]
- 120.Rachfal AW, Brigstock DR. Connective tissue growth factor (CTGF/CCN2) in hepatic fibrosis. Hepatol Res. 2003;26(1):1–9. doi: 10.1016/S1386-6346(03)00115-3. [DOI] [PubMed] [Google Scholar]
- 121.Yoshida K, Munakata H. Connective tissue growth factor binds to fibronectin through the type I repeat modules and enhances the affinity of fibronectin to fibrin. Biochim Biophys Acta Gen Subj. 2007;1770(4):672–680. doi: 10.1016/j.bbagen.2006.11.010. [DOI] [PubMed] [Google Scholar]
- 122.Jun J-I, Lau LF. Taking aim at the extracellular matrix: CCN proteins as emerging therapeutic targets. Nat Rev Drug Discov. 2011;10(12):945–963. doi: 10.1038/nrd3599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Gao R, Brigstock DR. Connective tissue growth factor (CCN2) induces adhesion of rat activated hepatic stellate cells by binding of its C-terminal domain to integrin alpha(v)beta(3) and heparan sulfate proteoglycan. J Biol Chem. 2004;279(10):8848–8855. doi: 10.1074/jbc.M313204200. [DOI] [PubMed] [Google Scholar]
- 124.Igarashi A, Okochi H, Bradham DM, Grotendorst GR. Regulation of connective tissue growth factor gene expression in human skin fibroblasts and during wound repair. Mol Biol Cell. 1993;4(6):637–645. doi: 10.1091/mbc.4.6.637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Kothapalli D, Frazier KS, Welply A, Segarini PR, Grotendorst GR. Transforming growth factor beta induces anchorage-independent growth of NRK fibroblasts via a connective tissue growth factor-dependent signaling pathway. Cell Growth Differ. 1997;8(1):61–68. [PubMed] [Google Scholar]
- 126.Duncan MR, Frazier KS, Abramson S, Williams S, Klapper H, Huang X, Grotendorst GR. Connective tissue growth factor mediates transforming growth factor beta-induced collagen synthesis: down-regulation by cAMP. FASEB J. 1999;13(13):1774–1786. doi: 10.1096/fasebj.13.13.1774. [DOI] [PubMed] [Google Scholar]
- 127.Abou-Shady M, Friess H, Zimmermann A, di Mola FF, Guo XZ, Baer HU, Büchler MW. Connective tissue growth factor in human liver cirrhosis. Liver. 2000;20(4):296–304. doi: 10.1034/j.1600-0676.2000.020004296.x. [DOI] [PubMed] [Google Scholar]
- 128.Paradis V, Dargere D, Vidaud M, De Gouville AC, Huet S, Martinez V, et al. Expression of connective tissue growth factor in experimental rat and human liver fibrosis. Hepatology. 1999;30(4):968–976. doi: 10.1002/hep.510300425. [DOI] [PubMed] [Google Scholar]
- 129.Klein S, Schierwagen R, Uschner FE, Trebicka J. Mouse and rat models of induction of hepatic fibrosis and assessment of portal hypertension. Methods Mol Biol. 1627;2017:91–116. doi: 10.1007/978-1-4939-7113-8_7. [DOI] [PubMed] [Google Scholar]
- 130.Luedde T, Kaplowitz N, Schwabe RF. Cell death and cell death responses in liver disease: mechanisms and clinical relevance. Gastroenterology. 2014;147(4):765–783.e4. doi: 10.1053/j.gastro.2014.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Tsuchida T, Friedman SL. Mechanisms of hepatic stellate cell activation. Nat Rev Gastroenterol Hepatol. 2017;14(7):397–411. doi: 10.1038/nrgastro.2017.38. [DOI] [PubMed] [Google Scholar]
- 132.Liang S, Kisseleva T, Brenner DA. The role of NADPH oxidases (NOXs) in liver fibrosis and the activation of myofibroblasts. Front Physiol [Internet]. 2016 Feb 2;7:17. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4735448/. Accessed 20 Nov 2020. [DOI] [PMC free article] [PubMed]
- 133.Lan T, Kisseleva T, Brenner DA. Deficiency of NOX1 or NOX4 prevents liver inflammation and fibrosis in mice through inhibition of hepatic stellate cell activation. PLoS One. 2015;10(7):e0129743. doi: 10.1371/journal.pone.0129743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Paik Y-H, Iwaisako K, Seki E, Inokuchi S, Schnabl B, Österreicher CH, Kisseleva T, Brenner DA. The nicotinamide adenine dinucleotide phosphate oxidase homologues NOX1 and NOX2/gp91phox mediate hepatic fibrosis in mice. Hepatology. 2011;53(5):1730–1741. doi: 10.1002/hep.24281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Bataller R, Ginès P, Nicolás JM, Görbig MN, Garcia-Ramallo E, Gasull X, et al. Angiotensin II induces contraction and proliferation of human hepatic stellate cells. Gastroenterology. 2000;118(6):1149–1156. doi: 10.1016/S0016-5085(00)70368-4. [DOI] [PubMed] [Google Scholar]
- 136.Heller J, Shiozawa T, Trebicka J, Hennenberg M, Schepke M, Neef M, Sauerbruch T. Acute haemodynamic effects of losartan in anaesthetized cirrhotic rats. Eur J Clin Investig. 2003;33(11):1006–1012. doi: 10.1046/j.1365-2362.2003.01251.x. [DOI] [PubMed] [Google Scholar]
- 137.Heller J, Trebicka J, Shiozawa T, Schepke M, Neef M, Hennenberg M, Sauerbruch T. Vascular, hemodynamic and renal effects of low-dose losartan in rats with secondary biliary cirrhosis. Liver Int. 2005;25(3):657–666. doi: 10.1111/j.1478-3231.2005.01053.x. [DOI] [PubMed] [Google Scholar]
- 138•.Granzow M, Schierwagen R, Klein S, Kowallick B, Huss S, Linhart M, et al. Angiotensin-II type 1 receptor-mediated Janus kinase 2 activation induces liver fibrosis. Hepatology. 2014;60(1):334–348. doi: 10.1002/hep.27117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Carey RM. Newly discovered components and actions of the renin–angiotensin system. Hypertension. 2013;62(5):818–822. doi: 10.1161/HYPERTENSIONAHA.113.01111. [DOI] [PubMed] [Google Scholar]
- 140.Tipnis SR, Hooper NM, Hyde R, Karran E, Christie G, Turner AJ. A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J Biol Chem. 2000;275(43):33238–43. doi: 10.1074/jbc.M002615200. [DOI] [PubMed] [Google Scholar]
- 141.Donoghue M, Hsieh F, Baronas E, Godbout K, Gosselin M, Stagliano N, Donovan M, Woolf B, Robison K, Jeyaseelan R, Breitbart RE, Acton S. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1-9. Circ Res. 2000;87(5):E1–E9. doi: 10.1161/01.RES.87.5.e1. [DOI] [PubMed] [Google Scholar]
- 142.Klein S, Rick J, Lehmann J, Schierwagen R, Schierwagen IG, Verbeke L, et al. Janus-kinase-2 relates directly to portal hypertension and to complications in rodent and human cirrhosis. Gut. 2017;66(1):145–155. doi: 10.1136/gutjnl-2015-309600. [DOI] [PubMed] [Google Scholar]
- 143.Bataller R, Gäbele E, Schoonhoven R, Morris T, Lehnert M, Yang L, Brenner DA, Rippe RA. Prolonged infusion of angiotensin II into normal rats induces stellate cell activation and proinflammatory events in liver. Am J Physiol Gastrointest Liver Physiol. 2003;285(3):G642–G651. doi: 10.1152/ajpgi.00037.2003. [DOI] [PubMed] [Google Scholar]
- 144.Bataller R, Sancho-Bru P, Ginès P, Lora JM, Al-Garawi A, Solé M, et al. Activated human hepatic stellate cells express the renin-angiotensin system and synthesize angiotensin II. Gastroenterology. 2003;125(1):117–125. doi: 10.1016/S0016-5085(03)00695-4. [DOI] [PubMed] [Google Scholar]
- 145.Paizis G, Cooper ME, Schembri JM, Tikellis C, Burrell LM, Angus PW. Up-regulation of components of the renin-angiotensin system in the bile duct-ligated rat liver. Gastroenterology. 2002;123(5):1667–1676. doi: 10.1053/gast.2002.36561. [DOI] [PubMed] [Google Scholar]
- 146.Warner FJ, Rajapaksha H, Shackel N, Herath CB. ACE2: from protection of liver disease to propagation of COVID-19. Clin Sci (Lond) 2020;134(23):3137–3158. doi: 10.1042/CS20201268. [DOI] [PubMed] [Google Scholar]
- 147.Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–280.e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Zhao B, Ni C, Gao R, Wang Y, Yang L, Wei J, Lv T, Liang J, Zhang Q, Xu W, Xie Y, Wang X, Yuan Z, Liang J, Zhang R, Lin X. Recapitulation of SARS-CoV-2 infection and cholangiocyte damage with human liver ductal organoids. Protein Cell. 2020;11(10):771–775. doi: 10.1007/s13238-020-00718-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Wigén J, Löfdahl A, Bjermer L, Elowsson-Rendin L, Westergren-Thorsson G. Converging pathways in pulmonary fibrosis and Covid-19 - the fibrotic link to disease severity. Respir Med X. 2020;2:100023. doi: 10.1016/j.yrmex.2020.100023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Lopez-Mendez I, Aquino-Matus J, Gall SM-B, Prieto-Nava JD, Juarez-Hernandez E, Uribe M, et al. Association of liver steatosis and fibrosis with clinical outcomes in patients with SARS-CoV-2 infection (COVID-19) Ann Hepatol. 2020;20:100271. doi: 10.1016/j.aohep.2020.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Saviano A, Wrensch F, Ghany MG, Baumert TF. Liver disease and COVID-19: from pathogenesis to clinical care. Hepatology. 10.1002/hep.31684. [DOI] [PMC free article] [PubMed]
- 152.Lachowski D, Cortes E, Rice A, Pinato D, Rombouts K, del Rio Hernandez A. Matrix stiffness modulates the activity of MMP-9 and TIMP-1 in hepatic stellate cells to perpetuate fibrosis. Sci Rep. 2019;9(1):7299. doi: 10.1038/s41598-019-43759-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Carrel A, Hartmann A. Cicatrization of wounds I. The relation between the size of a wound and the rate of its cicatrization. J Exp Med. 1916;24(5):429–450. doi: 10.1084/jem.24.5.429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Wells RG. The role of matrix stiffness in regulating cell behavior. Hepatology. 2008;47(4):1394–1400. doi: 10.1002/hep.22193. [DOI] [PubMed] [Google Scholar]
- 155.Wells RG. Tissue mechanics and fibrosis. Biochim Biophys Acta (BBA) - Mol Basis Dis. 2013;1832(7):884–890. doi: 10.1016/j.bbadis.2013.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Caliari SR, Perepelyuk M, Cosgrove BD, Tsai SJ, Lee GY, Mauck RL, Wells RG, Burdick JA. Stiffening hydrogels for investigating the dynamics of hepatic stellate cell mechanotransduction during myofibroblast activation. Sci Rep. 2016;6(1):21387. doi: 10.1038/srep21387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Görtzen J, Schierwagen R, Bierwolf J, Klein S, Uschner FE, van der Ven PF, et al. Interplay of matrix stiffness and c-SRC in hepatic fibrosis. Front Physiol. 2015;6:359. doi: 10.3389/fphys.2015.00359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Hemmann S, Graf J, Roderfeld M, Roeb E. Expression of MMPs and TIMPs in liver fibrosis - a systematic review with special emphasis on anti-fibrotic strategies. J Hepatol. 2007;46(5):955–975. doi: 10.1016/j.jhep.2007.02.003. [DOI] [PubMed] [Google Scholar]
- 159.Martinez-Hernandez A, Amenta PS. The extracellular matrix in hepatic regeneration. FASEB J. 1995;9(14):1401–1410. doi: 10.1096/fasebj.9.14.7589981. [DOI] [PubMed] [Google Scholar]
- 160.Campana L, Iredale JP. Regression of liver fibrosis. Semin Liver Dis. 2017;58(1):1–10. doi: 10.1055/s-0036-1597816. [DOI] [PubMed] [Google Scholar]
- 161.Kessenbrock K, Plaks V, Werb Z. Matrix metalloproteinases: regulators of the tumor microenvironment. Cell. 2010;141(1):52–67. doi: 10.1016/j.cell.2010.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Moreira RK. Hepatic stellate cells and liver fibrosis. Arch Pathol Lab Med. 2007;131(11):1728–1734. doi: 10.5858/2007-131-1728-HSCALF. [DOI] [PubMed] [Google Scholar]
- 163.Benyon RC, Hovell CJ, Da Gaça M, Jones EH, Iredale JP, Arthur MJ. Progelatinase A is produced and activated by rat hepatic stellate cells and promotes their proliferation. Hepatology. 1999;30(4):977–986. doi: 10.1002/hep.510300431. [DOI] [PubMed] [Google Scholar]
- 164.Anders H-J, Schaefer L. Beyond tissue injury—damage-associated molecular patterns, toll-like receptors, and inflammasomes also drive regeneration and fibrosis. J Am Soc Nephrol. 2014;25(7):1387–1400. doi: 10.1681/ASN.2014010117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Iozzo RV, Schaefer L. Proteoglycan form and function: a comprehensive nomenclature of proteoglycans. Matrix Biol. 2015;42:11–55. doi: 10.1016/j.matbio.2015.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Schaefer L. Complexity of danger: the diverse nature of damage-associated molecular patterns. J Biol Chem. 2014;289(51):35237–35245. doi: 10.1074/jbc.R114.619304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Moreth K, Iozzo RV, Schaefer L. Small leucine-rich proteoglycans orchestrate receptor crosstalk during inflammation. Cell Cycle. 2012;11(11):2084–2091. doi: 10.4161/cc.20316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Piccinini AM, Midwood KS. DAMPening inflammation by modulating TLR signalling. Mediators Inflamm [Internet]. 2010;2010:672395. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2913853/. Accessed 11 Jan 2021. [DOI] [PMC free article] [PubMed]
- 169.Frey H, Schroeder N, Manon-Jensen T, Iozzo RV, Schaefer L. Biological interplay between proteoglycans and their innate immune receptors in inflammation. FEBS J. 2013;280(10):2165–2179. doi: 10.1111/febs.12145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Schaefer L, Babelova A, Kiss E, Hausser H-J, Baliova M, Krzyzankova M, Marsche G, Young MF, Mihalik D, Götte M, Malle E, Schaefer RM, Gröne HJ. The matrix component biglycan is proinflammatory and signals through Toll-like receptors 4 and 2 in macrophages. J Clin Invest. 2005;115(8):2223–2233. doi: 10.1172/JCI23755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Nastase MV, Young MF, Schaefer L. Biglycan. J Histochem Cytochem. 2012;60(12):963–975. doi: 10.1369/0022155412456380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Hsieh LT-H, Nastase M-V, Zeng-Brouwers J, Iozzo RV, Schaefer L. Soluble biglycan as a biomarker of inflammatory renal diseases. Int J Biochem Cell Biol. 2014;54:223–235. doi: 10.1016/j.biocel.2014.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Merline R, Moreth K, Beckmann J, Nastase MV, Zeng-Brouwers J, Tralhão JG, et al. Signaling by the matrix proteoglycan decorin controls inflammation and cancer through PDCD4 and MicroRNA-21. Sci Signal. 2011;4(199):ra75. doi: 10.1126/scisignal.2001868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Gruber E, Heyward C, Cameron J, Leifer C. Toll-like receptor signaling in macrophages is regulated by extracellular substrate stiffness and Rho-associated coiled-coil kinase (ROCK1/2) Int Immunol. 2018;30(6):267–278. doi: 10.1093/intimm/dxy027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Lo C-M, Wang H-B, Dembo M, Wang Y. Cell movement is guided by the rigidity of the substrate. Biophys J. 2000;79(1):144–152. doi: 10.1016/S0006-3495(00)76279-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Riveline D, Zamir E, Balaban NQ, Schwarz US, Ishizaki T, Narumiya S, Kam Z, Geiger B, Bershadsky AD. Focal contacts as mechanosensors. J Cell Biol. 2001;153(6):1175–1186. doi: 10.1083/jcb.153.6.1175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.del Rio A, Perez-Jimenez R, Liu R, Roca-Cusachs P, Fernandez JM, Sheetz MP. Stretching single talin rod molecules activates vinculin binding. Science. 2009;323(5914):638–641. doi: 10.1126/science.1162912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Chan CE, Odde DJ. Traction dynamics of filopodia on compliant substrates. Science. 2008;322(5908):1687–1691. doi: 10.1126/science.1163595. [DOI] [PubMed] [Google Scholar]
- 179.Elosegui-Artola A, Trepat X, Roca-Cusachs P. Control of mechanotransduction by molecular clutch dynamics. Trends Cell Biol. 2018;28(5):356–367. doi: 10.1016/j.tcb.2018.01.008. [DOI] [PubMed] [Google Scholar]
- 180.Elosegui-Artola A, Oria R, Chen Y, Kosmalska A, Pérez-González C, Castro N, et al. Mechanical regulation of a molecular clutch defines force transmission and transduction in response to matrix rigidity. Nat Cell Biol. 2016;18(5):540–548. doi: 10.1038/ncb3336. [DOI] [PubMed] [Google Scholar]
- 181.Sunyer R, Conte V, Escribano J, Elosegui-Artola A, Labernadie A, Valon L, Navajas D, Garcia-Aznar JM, Munoz JJ, Roca-Cusachs P, Trepat X. Collective cell durotaxis emerges from long-range intercellular force transmission. Science. 2016;353(6304):1157–1161. doi: 10.1126/science.aaf7119. [DOI] [PubMed] [Google Scholar]