Abstract
In the last few decades, the thinning of the ozone layer due to increased atmospheric pollution has exacerbated the negative effects of excessive exposure to solar ultraviolet radiation (UVR), and skin cancer has become a major public health concern. In order to prevent skin damage, public health advice mainly focuses on the use of sunscreens, along with wearing protective clothing and avoiding sun exposure during peak hours. Sunscreens present on the market are topical formulations that contain a number of different synthetic, organic, and inorganic UVR filters with different absorbance profiles, which, when combined, provide broad UVR spectrum protection. However, increased evidence suggests that some of these compounds cause subtle damage to marine ecosystems. One alternative may be the use of natural products that are produced in a wide range of marine species and are mainly thought to act as a defense against UVR-mediated damage. However, their potential for human photoprotection is largely under-investigated. In this review, attention has been placed on the molecular strategies adopted by marine organisms to counteract UVR-induced negative effects and we provide a broad portrayal of the recent literature concerning marine-derived natural products having potential as natural sunscreens/photoprotectants for human skin. Their chemical structure, UVR absorption properties, and their pleiotropic role as bioactive molecules are discussed. Most studies strongly suggest that these natural products could be promising for use in biocompatible sunscreens and may represent an alternative eco-friendly approach to protect humans against UV-induced skin damage.
Keywords: photoprotection, skin, marine natural products, solar radiation, antioxidants
1. Introduction
Solar energy fuels life on Earth and, in part, in the oceans. Nevertheless, excessive sunlight can cause photodamage in photosynthetic organisms. In addition, 5% of the solar spectrum is constituted by ultraviolet radiation (UVR), which is partly shielded from gases in the atmosphere. Indeed, the ozone layer acts as a filter for UVR because it completely absorbs the UV-C component and 90% of UV-B rays. Therefore, only part of UV-B and UV-A reach the Earth and the sea and consequently can be deleterious for both terrestrial and aquatic organisms.
The damaging effects of UVR can be mediated by the direct interaction of UV photons with DNA or indirectly by the production of reactive oxygen species (ROS), which can cause oxidative modifications to DNA and other macromolecules such as proteins and lipids [1].
Marine organisms living in the photic zone of the sea are constantly exposed to changes in light intensity and spectral composition; hence, they have evolved the ability to use solar radiation for survival but also to protect themselves from UVR-induced insults—for example, by triggering repair mechanisms after damage.
Humans can take advantage and inspiration from strategies adopted by nature to counteract the negative effects of solar radiation, especially UVR. In particular, UVR can cause sunburn and erythema as a result of acute exposure, or even skin aging and cancer, along with harmful effects on the eyes and the immune system in the long term [2]. Indeed, although humans are endowed with endogenous mechanisms of defense against light-induced damage, such as melanin production, these mechanisms are insufficient to provide full protection from UVR. Moreover, in the last few decades, the thinning of the ozone layer due to increased atmospheric pollution has favored the penetration of UV-B rays in the atmosphere, thus increasing the exposure of humans to these rays.
Consequently, the World Health Organization (WHO) has laid out a set of recommendations regarding sun protection that include: (1) limiting sun exposure from 10 a.m. to 4 p.m., when UVR rays are stronger; (2) checking the UV index before planning outdoor activities; (3) staying under shady structures, such as trees or umbrellas, during the most intense hours, taking into consideration that they do not provide complete protection; (4) wearing protective clothes and accessories, such as hats and sunglasses; (5) using sunscreens; (6) avoiding sunlamps [3]. Thus, using sunscreens is one of the six key strategies to avoid UVR-induced damage.
However, consumers are becoming increasingly aware of where the products they use come from, whether of synthetic or natural origin, and whether they are eco-friendly and eco-sustainable. Despite the knowledge that sunscreens are an undeniably important tool in the fight against skin cancer, their formulations may need to be improved to contain safer ingredients and to make them more water-resistant, particularly in view of the concerns raised on the potential eco-toxicity of some sunscreens to the marine environment as well as to human health. For instance, some sunscreens containing UV synthetic filters such as benzophenone-3 (oxybenzone) and octyl methoxycinnamate (octinoxate) have now been banned from sale and distribution in Hawaii as of 1st January 2021 for their suspected harm to coral reefs and marine life, and other USA locations have followed suit (Key West, U.S. Virgin Islands). Other nations, such as Palau, parts of Mexico, and the Caribbean islands of Bonaire and Aruba, have also put in place similar enforcements [4]. These UV filters, along with others, have also fallen under scrutiny for their potential toxicity concerning reproduction and development in various organisms [5]. Therefore, discovering and approving new sunscreen compounds with fewer downsides could alleviate concerns from all corners and increase sunscreen use.
Turning to nature to replace synthetic chemicals is a growing trend in the pharmaceutical/cosmeceutical sector, where there is a surge in demand for valid alternatives of natural origin [6]. In this context, attention has recently been devoted to the photoprotective chemicals produced by the marine environment, either by extracting UV-filtering compounds from their natural sources or by engineering yeast or other organisms to produce them. Indeed, the huge and still underexploited marine biodiversity represents a yet untapped source of a wide range of naturally occurring UVR screening compounds, which can be used for cosmeceutical applications as eco-friendly and safer alternatives to synthetic UV filters.
The aim of this review is therefore to summarize the most recent literature on the molecular mechanisms evolved by marine organisms to counteract light-induced damage and to give a panorama on the diversity of photoprotective marine-derived compounds from which mankind could take inspiration to develop natural-based products that could be safer and more environmentally friendly.
2. The Photic Zone: Where Light Fuels Life in the Ocean
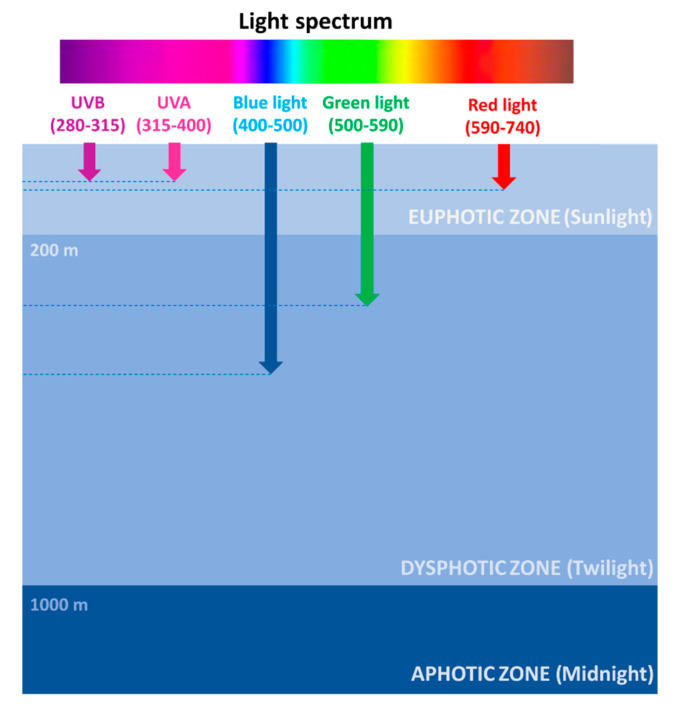
The photic zone is the uppermost layer of the sea, distinguishable in two main areas: the euphotic (or sunlight) zone, approximately 200 m deep, where sunlight allows phytoplankton to accomplish photosynthesis, and the dysphotic (or twilight) zone (200–1000 m deep), where sunlight rapidly decreases with depth and where photosynthesis cannot occur (Figure 1).
Figure 1.
Light spectrum penetration at sea.
However, the depth of the euphotic zone depends on the transparency of the water. If the water is very clear, light can penetrate deeper, while if the water is murky, the depth of the euphotic zone is reduced to only 50 feet (15 m). Ninety-five percent of photosynthesis in the ocean occurs in this zone, where the photosynthesis rate exceeds the respiration rate. Therefore, the high oxygen concentration fuels the life of 90% of marine organisms, including phytoplankton, such as dinoflagellates, diatoms, and cyanobacteria; zooplankton, such as copepods; and nekton, such as fish, squids, and crabs.
In the ocean, exposure to sunlight is extremely variable along the water column, both in terms of photon flux density and spectral composition (Figure 1). Several factors are responsible for light intensity and spectrum variability at sea, including the geographical location, the day hours/season, the weather conditions, and the occurrence of organic matter. Red wavelengths (589–656 nm) are absorbed in the euphotic zone and are rapidly attenuated in the first water layers, thus reaching approximately 100 m in depth. Green wavelengths (479–579 nm) can penetrate until a depth of approximately 300 m and are used by algae for photosynthesis. Blue wavelengths (422–496 nm) can penetrate farther, being the only ones to reach the deep ocean, approximately 500 m in depth, and are mostly responsible for the color of the oceans [7,8]. The prevalence of blue/green light at sea has prompted marine organisms to sense and respond prevalently to these wavelengths, even though red/far-red sensing systems have also been described in diatoms [9].
3. Light Perception and Light-Driven Physiological Processes at Sea
Photosynthetic organisms mainly rely on light for their survival and they have evolved complex systems of photoperception and photoresponse. Blue light in particular is a key regulator of growth, development, and photosynthesis. It is sensed by blue light photoreceptors, consisting of flavoproteins belonging to the cryptochrome/photolyases family. They are known to mediate several light-induced responses, such as DNA repair, light perception, and the circadian clock [10,11]. However, several additional photosensors can be found in photosynthetic organisms, differing in the light absorption range, domain architecture, and bound chromophore. They are involved in the regulation of metabolism, cell cycle, and other important physiological processes [12].
However, light is not only an essential source of energy for photosynthetic organisms and, indirectly, for all life on Earth, but it can also be perceived and used by non-photosynthetic organisms. Marine animals, for example, use it for vision in the photic zone of the sea to escape predators or catch preys [13]. In most vertebrates, rod and cone photoreceptors mediate vision in low and high light, respectively [14]. These photoreceptors possess light-sensitive pigments, called opsins, whose number and type are extremely variable depending on the light habitat of the organism, allowing vision both in shallow waters, characterized by high light exposure, and in the dim light typical of deep oceans [15,16,17].
As mentioned above, the most dangerous portion of light belongs to the UVR spectrum (280–400 nm), which is able to reach the water column and induce responses in sea-inhabiting organisms. The degree of UVR penetration in water mainly depends on the optical properties of dissolved and suspended organic matter; hence, it is higher in clear open oceans than in turbid coastal waters [18,19]. Prolonged exposure to UVR can cause a variety of damage in living organisms, discussed in the next section. However, low levels have been reported to increase photosynthetic carbon fixation or even enhance primary production in certain conditions, and to influence the metabolism in photosynthetic organisms [20,21]. Furthermore, UV light is used for vision by some marine animals, such as coral fish, improving their ability to catch prey [22,23].
4. Photo-Damage/-Protection Mechanisms in Marine Organisms
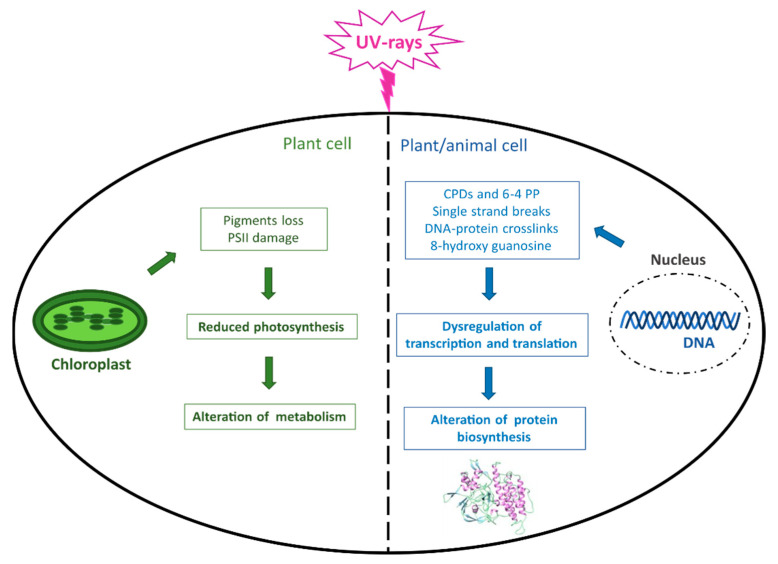
Although light is essential for life on Earth and in the oceans, photosynthetic organisms can be harmed by an excess of photons reaching the photosynthetic apparatus, whose damage can result in a decrease in photosynthesis and metabolism (Figure 2). Moreover, the major macromolecules, including proteins, lipids, and especially DNA, can be damaged by both direct UVR and consequent ROS production, which ultimately leads to altered protein biosynthesis and crucial cellular functions not only in plant but also in animal organisms (Figure 2).
Figure 2.
UVR-induced damage in living organisms. The figure summarizes the main effects caused by UVR occurring at the DNA level in both plant and animal cells and in chloroplasts in photosynthetic cells. CPDs = cyclobutane pyrimidine dimers; 6–4 PP = pyrimidine (6–4) pyrimidone photoproducts; PSII = Photosystem II.
Such an environmental constraint has forced them to evolve a complex system of photoprotective mechanisms and antioxidant enzymes and molecules, in order to counteract light-dependent stress. For instance, the first defense mechanism used by algae against high light is the dissipation of excess light into heat through non-photochemical quenching (NPQ) of chlorophyll fluorescence. This mechanism allows algae to prevent photodamage [24]. In particular, the increased proton flux leads to acidification of the thylakoid lumen inside chloroplasts, with consequent activation of the xanthophyll cycle, a key player in the NPQ process [25]. In addition to these short-term responses, cells have adopted a variety of long-term responses to counteract light stress, including several integrated signaling pathways and associated changes in gene expression. In particular, variations in the state of chloroplasts are sensed by the nucleus through a “retrograde signaling” in which the tetrapyrroles of chlorophyll seem to play a central role [26].
However, despite such protective mechanisms, high light exposure can ultimately lead to the formation of ROS. Algae possess a complex enzymatic and non-enzymatic defense system to prevent photo-oxidative damage to cellular components. Enzymatic antioxidant protection includes three types of superoxide dismutases possessing different metal cofactors (CuZn-SOD, Fe-SOD, Mn-SOD), catalase, ascorbate peroxidase, glutathione peroxidase, while the non-enzymatic defense system includes ascorbic acid, glutathione, tocopherols, carotenoids, polyphenols, phycobilin proteins, dimethylsulfide/dimethylsulfoxide, sulfated polysaccharides [27], and sulfur-containing histidines such as ovothiols [28,29].
As mentioned earlier, the UV portion of light is able to reach water bodies and can represent a source of damage for their inhabitants. UVB (280–315 nm) in particular can induce damage to DNA, through the generation of cyclobutane pyrimidine dimers (CPDs) and pyrimidine (6–4) pyrimidone photoproducts (6–4 PP), while UVA (315–400) mainly induces indirect oxidative damage to DNA in terms of single-strand breaks, DNA–protein crosslinks, and 8-hydroxy-guanosine (8-oxoG) [30]. These photoproducts can inhibit DNA replication and cell cycle progression, threatening, for example, phytoplankton growth and photosynthesis, although the sensitivity to UVR can vary among different species [31,32,33,34]. UVR are not only dangerous for photosynthetic organisms but they can cause damage to a wide range of living organisms, from bacteria to higher vertebrates, and their effects can change depending on other environmental conditions [35]. For instance, UVR negatively affects the viability and development of invertebrates and fish, whose early developmental stages are generally more sensitive [36,37]. Indeed, CPDs formation in sea urchin embryos and larvae inhibits their development, decreasing population survival and fitness [38].
In addition to the above-mentioned enzymatic and non-enzymatic antioxidant systems used by photosynthetic organisms to counteract light stress, marine animals have adopted some UVR-avoiding strategies, such as living in deeper water. Several organisms have also adapted by synthesizing natural UV filters, such as melanin and mycosporine-like amino acids (MAAs) [39,40], or by acquiring screening compounds, such as scytonemin, flavonoids, and carotenoids from other organisms via their diet or by symbiosis.
5. UVR Detrimental Effects on Humans
All living organisms can be exposed to the harmful effects of UVR and humans are not an exception. Indeed, even though UVB rays are useful for vitamin D biosynthesis in human skin [41], they can lead to a variety of dysfunctions, especially in certain human organs that are mostly exposed to UVR, such as the eyes and skin. While the only clinically relevant effect of excessive UVR exposure on the eyes is represented by a temporary inflammation of the corneal epithelium, generally appearing 6–12 h after exposure and resolving in around 48 h without long-term effects, the UVR-induced photodamage in the skin is quite complex [42].
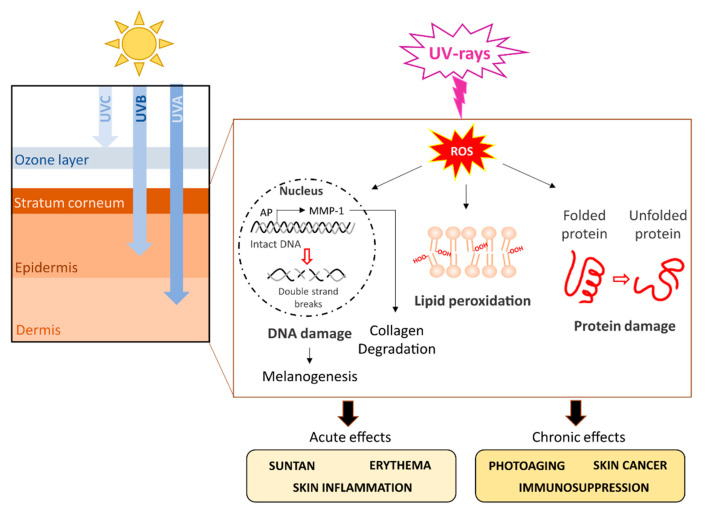
The degree of skin penetration of UVR depends on the wavelength (WL). In particular, UVB rays, having a shorter WL (280–320 nm), are absorbed by keratinocytes in the stratum corneum of the epidermis, whereas UVA rays, having a longer WL (320–400 nm), are able to penetrate deeper into the dermis [1] (Figure 3).
Figure 3.
Major UVR-induced damage in humans. The figure summarizes the main direct and ROS-mediated effects caused by UVR in human skin. Abbreviations: AP = activator protein; MMP-1 = matrix metalloproteinase-1.
The first acute reaction to UVR is a suntan due to DNA photodamage and the consequent stimulation of repair mechanisms and melanogenesis [43,44]. High levels of UVR (especially UVB) can cause skin inflammation, erythema, edemas, blisters, and sunburn cells, which undergo apoptosis [45]. Moreover, UVR can lead to a suppression of local and systemic immunity, thus exposing the body to several types of infections [46]. Chronic UVR effects include skin cancer, photoaging, and immunosuppression [47,48,49] (Figure 3).
Although humans produce protective molecules such as melanin, which is, to some extent, able to protect DNA from photodamage, this is often not enough for providing sufficient protection against prolonged or excessive exposure to UVR. To help the skin to protect the body against excessive UVR, the cosmetic and pharmaceutical industries have developed several products mainly for topical application, such as sunscreen formulations.
6. UV Filter Properties
Typically, a sunscreen is conceived as a formulation containing UV chemical filters, which can be either inorganic, such as zinc oxide (ZnO) and titanium dioxide (TiO2), or organic, such as cinnamates and benzophenones [50,51]. Conceptually, the absorption of the UVR photons’ energy causes a photochemical excitation of the filter molecule to a higher energy excited state. As the excited molecule returns to its ground state, the excess energy is partially dissipated as heat. In some cases, the absorbed energy may be transferred to a receptor molecule, or emitted in the visible region and perceived as fluorescence, or it may be emitted in the longwave UVR (380–450 nm), which may cause a photochemical reaction such as cis-trans or keto-enol isomerizations in a fraction of the filter molecules. These photochemical reactions, in some cases, may give rise to instability in the filter molecule, with chemical bond cleavage and formation of photodegradation products. This is a highly undesirable property for a UV filter since the loss in absorption efficacy defeats the purpose of a UV filter for sunscreen use. An efficient UV filter should be able to return to its ground state in its original form such that it may be available to absorb another photon. This guarantees photoprotection throughout the entire duration of UV exposure [52].
Organic UV filters used in sunscreens are powerful photochemicals whose behavior is closely related to their molecular structure. They typically contain a chromophore, which is usually an aromatic molecule conjugated to carbonyl groups. In general, an increased number of conjugated double bonds and resonance structures stabilizes the excited state, shifting the absorption spectrum of the compound to longer wavelengths. Instead, inorganic filters such as ZnO and TiO2, commonly used in sunscreen formulations, besides absorbing UV light, also possess scattering and reflecting properties. They are generally considered more natural and benign than organic UV filters, despite the fact that they contain a wide array of coatings, additives, predispersants, and dispersion enhancers. Furthermore, because of their particulate nature, which causes the so-called “whitening” phenomenon upon application, they are nowadays generally micronized to improve their cosmetic appearance [53].
There is a growing body of evidence suggesting that the synthetic UVR filters such as octocrylene, benzophenone-4, ethyl 4-aminobenzoate, 3-benzylidene camphor, and TiO2 nanoparticles may cause damage to the marine environment, due to their widespread use on sea resorts. Several negative effects include the bioaccumulation of filters in many different species, hormonal changes and endocrine disruption in fish, abnormal development in sea urchins, and the bleaching of corals [54,55,56,57].
In this context, sunscreen compounds of natural origin may provide an eco-friendly alternative to synthetic products. In particular, the marine ecosystem offers an extremely rich biodiversity of life forms, most of which are still underexploited as natural sources for cosmeceutical products [58]. Several classes of marine-derived molecules will be here discussed, including their chemistry, occurrence, natural/physiological functions, and bioactivity in in vitro mammalian cells, with a special focus on their photoprotective abilities.
7. Mycosporine-Like Amino Acids (MAAs)
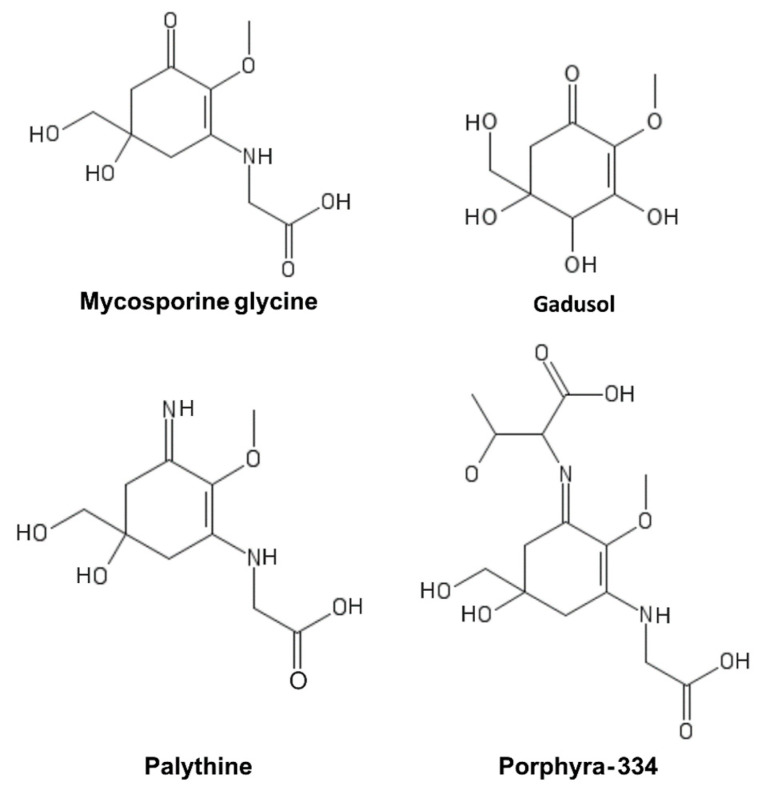
MAAs are a heterogeneous group of hydrophilic molecules (<400 Da). Their structure consists of a cyclohexanone or cyclohexenimine ring system conjugated to an amino acid or its imino alcohol [59]. Representative MAAs are reported in Figure 4.
Figure 4.
Mycosporine-like amino acids (MAAs).
They absorb in the 310–362 nm range with a high molar extinction coefficient and are produced by organisms that are particularly exposed to UV light, such as cyanobacteria [60], micro- and macroalgae [61,62], dinoflagellates [63], and fungi [64]. Their biosynthesis is mainly ascribed to the shikimate pathway, although the pentose-phosphate pathway has also been suggested to be involved in their biosynthesis [65,66]. Aromatic amino acids and MAAs in marine organisms have been generally hypothesized to be of dietary origin since most metazoans lack genes coding for the enzymes of the shikimic acid pathway [64,67,68]. However, previous studies have suggested that cnidarians, including nonsymbiotic corals and anemones, are capable of synthesizing these essential amino acids [68,69,70,71]. Starcevic and colleagues [70] provided evidence for the horizontal transfer of both bacterial and dinoflagellate ancestral genes of the shikimic acid pathway into the genome of Nematostella. Moreover, besides corals, the endogenous synthesis of gadusol, the main MAAs precursor, has been discovered to occur in fish through an alternative pathway, which is also present in amphibians, reptiles, and birds [72,73].
MAAs play an important sunscreen role in producer organisms due to their properties, which include strong UVA absorbance, photostability, and widespread distribution in cell cytoplasm [68]. In addition, MAAs possess high stability under different pH and temperature values, representing a key advantage for their use as UV filters [74]. Furthermore, MAAs also function as strong antioxidants and additional roles have been suggested in cellular osmotic regulation during salt stress or as nitrogen reservoirs during nitrogen limitation [63,75]. Several studies have reported the ability of MAAs to protect against DNA damage induced directly by UVR and indirectly by ROS production triggered either chemically or by UVA radiation [76,77,78]. MAAs have also shown promising antioxidant, anti-inflammatory, and anti-aging activities in in vitro studies on human cells. In particular, their anti-aging properties were shown to be partially mediated by a direct inhibitory activity on protein glycation and collagenase enzymes [79]. To date, only a few products that include MAAs in their formulations, such as Helioguard®365 and Helionori®, have been released [80,81]. Among them, palythine, extracted from the red alga Chondrus yendoi has recently been incorporated as an active ingredient in the skin care product line Aethic®, and launched on the market in 2021 as Dermagie. These algae can be harvested year-round and can protect human skin cells from UVA and UVB damage under artificial sunlight as intense as at noon during summer in Arizona [78]. Nevertheless, further studies are necessary to fully assess the efficacy of MAAs for application in topical formulations [81,82].
8. Scytonemin
Scytonemin is a hydrophobic dimeric molecule composed of an indolic and phenolic subunit, linked by a carbon–carbon bond [83]. Its complex ring structure (Figure 5) allows a wide absorption range not only in the UVB but also in the UVA range, reaching a maximum of 370 nm [83].
Figure 5.
Scytonemin.
It is produced by some species of cyanobacteria, such as Scytonema sp., having the ability to produce extracellular polysaccharides (EPS) and living in harsh environments typically characterized by desiccation periods, high temperatures, and nutrient limitation [84,85,86,87]. Scytonemin-producing cyanobacteria secrete this molecule in the extracellular matrix, where it acts as a physical barrier against UVA and as an antioxidant molecule against ROS produced following UVA exposure or several other abiotic stressors [86,88,89]. Scytonemin biosynthesis includes the involvement of enzymes from the shikimate pathway, tyrosine and tryptophan biosynthesis, and two additional specific enzymes called ScyB and ScyA [90].
The high extinction coefficient (ε = 250 Lg−1cm−1 at 384 nm) and the exceptionally high photostability make scytonemin an efficient photoprotective chemical [91]. The UV-protective abilities of scytonemin extracted from cyanobacterial biofilm have been demonstrated in UV-irradiated E. coli cells [92].
In addition, scytonemin is also endowed with other biological activities, acting as an antioxidant [86], anti-inflammatory [93], and antiproliferative agent [94,95,96], and as a calcium-channel blocker [97]. Moreover, due to its presence in organisms able to survive in extreme environments, it has been proposed as a biomarker for the identification of life on extraterrestrial planets [98,99].
9. Polyphenols
Polyphenols are a large class of secondary metabolites characterized by the presence of several phenol rings. Among them, phenolic acids and flavonoids are the most abundant [100]. They are mainly found in plants, including fruits, vegetables, tea, and other plant-derived beverages. Due to their pleiotropic properties, they are endowed with numerous beneficial properties for human health, ranging from the prevention of cardiovascular diseases, metabolic syndrome, and neurodegenerative disorders, to anti-aging, anticancer, and antimicrobial activities [101,102,103]. They have also recently been proposed as an alternative co-adjuvant therapy against coronavirus infections, or in combination with classical antiviral drugs [104].
Besides terrestrial sources, marine organisms are also able to produce polyphenols, especially flavonoids. Most marine flavonoids have been found in seagrasses and halophytes but they have been identified also in mangroves, bacteria, algae, fungi, and corals. Some of these molecules present unique substituents compared to their terrestrial counterparts, such as the occurrence of sulphate, methyl, chlorine, amino acids, and aminodeoxy sugar groups [105]. Marine polyphenols have been shown to have the classical pharmacological activities ascribed to those of terrestrial origin, such as antioxidant, antidiabetic, anticoagulant, antimicrobial, and anticancer properties, and, in addition, they possess antifouling and antifeedant activities [105].
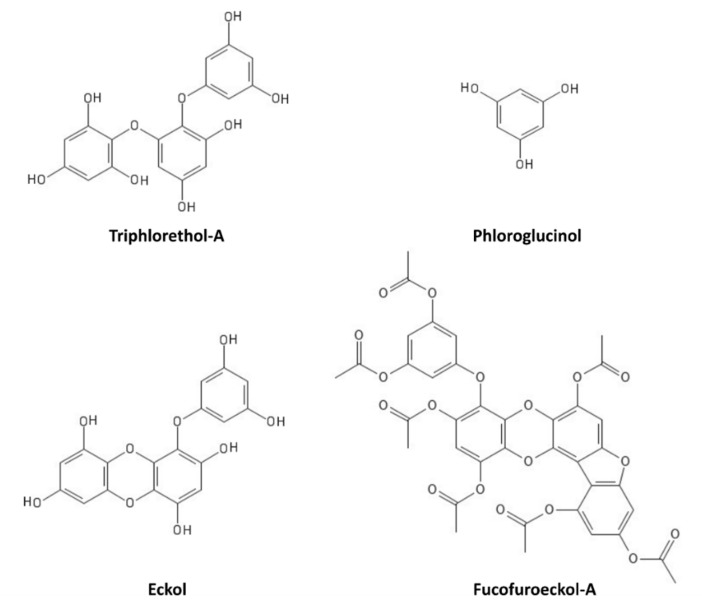
Photoprotective activities have also been described for polyphenols, whose action is either direct, due to their UV absorption ability, or indirect, thanks to their antioxidant/scavenging function and consequent regulation of different signaling pathways [106,107]. For instance, polyphenols extracted from brown algae have been reported to exert protection against UVB-induced damage in human cells and in an in vivo zebrafish model [108,109,110,111]. In particular, triphlorethol-A (Figure 6) protects human keratinocytes (HaCat) against UVB-induced damage, by exerting a UV-filtering and antioxidant function, as well as by inhibiting the caspase pathway [108]. Phloroglucinol (Figure 6) also showed UVB protection properties in HaCat cells, through the inhibition of key molecular mediators of UVB-induced photoaging, including MMP-1 (matrix metalloproteinase-1) activity, Ca2+ levels increase, MAPK (mitogen-activated protein kinase) phosphorylation, c-Fos and phospho c-Jun expression, and AP-1 (activator protein-1) binding to the MMP-1 promoter [109]. Other polyphenols, such as the phlorotannin eckol (Figure 6), protect human keratinocytes against UVB-induced oxidative stress by scavenging ROS and reducing apoptosis by inhibiting mitochondrial membrane disruption, thus ameliorating injury to cellular components [110]. Dieckol (composed of two molecules of eckol) showed a particularly strong UVB-protective ability both in HaCat cells and in live zebrafish, where it reduced ROS, NO (nitric oxide), and cell death [111]. Another derivative of eckol, fucofuroeckol-A (Figure 6), showed photoprotective properties since it reduced the UVB-induced allergic reaction in RBL-2H3 mast cells, leading to the suppression of mast cell degranulation and decreasing histamine release and Ca2+ levels [112]. Because of these photoprotective properties, polyphenols could hence be repurposed for cosmetics development in the sunscreen sector [113].
Figure 6.
Marine polyphenols with UVR-protective properties.
10. Carotenoids
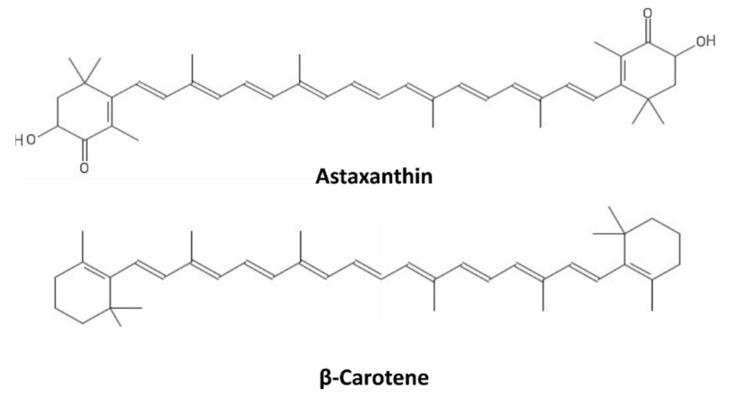
Carotenoids are a broad class of tetraterpenoids formed by 5-carbon isoprene units, polymerized to generate 40-carbon structures containing up to 15 conjugated double bonds. They include two major categories, carotenes and xanthophylls, with the latter possessing oxygen atoms in their structures. Some carotenoids consist of 6-carbon rings at one or both ends of the molecule; in the case of the xanthophylls, they possess oxo, hydroxy, or epoxy substitutions [114]. The structures of β-carotene and astaxanthin, as representatives of carotenes and xanthophylls, respectively, are shown in Figure 7.
Figure 7.
Carotenoids.
Carotenoids absorb in the blue-green region of the light spectrum (450–550 nm), and in photosynthetic organisms, they act as both light-harvesting molecules and as photoprotective agents through their ROS scavenging activity, thus contributing to most of the NPQ [115]. While photosynthetic organisms, including unicellular cyanobacteria and microalgae or multicellular algae and plants, are natural producers of carotenoids, marine animals can only acquire them through the diet or symbiotic relationships, such that they can also benefit from the valuable properties of carotenoids, including photoprotective action [116]. Carotenoids are mainly known as precursors of vitamin A biosynthesis in humans; however, they are endowed with multiple beneficial activities against neurodegenerative diseases, cancer, and cardiovascular pathologies [117]. Several studies have reported promising photoprotective activities of carotenoids against UV light-induced erythema [118]. In this process, the singlet oxygen (1O2) quenching activity is crucial, but they also activate different signaling pathways. β-Carotene, for instance, induces gene expression changes in human keratinocytes, resulting in decreased extracellular matrix degradation and increased cell differentiation. Both these effects were enhanced following keratinocyte irradiation with UVA light [119]. These mechanisms of action highlight the potentialities of β-carotene as a protective agent against photoaging and skin diseases, such as skin cancer and psoriasis. Two marine carotenoids, astaxanthin and fucoxanthin, also revealed promising anti-inflammatory and immunostimulant activity in addition to their protective properties against DNA damage and oxidative stress [120]. Astaxanthin, especially, has been tested in in vivo studies and clinical trials. Indeed, the application of a liposomal preparation of astaxanthin on the dorsal skin of Hos:HR-1 hairless mice prevented wrinkle formation induced by UV exposure [121] and its effects were confirmed by additional studies on animal models [122,123]. Moreover, astaxanthin supplementation in humans significantly increased their minimal erythema dose [124], defined as “the smallest UV dose that produces perceptible redness of the skin (erythema) with clearly defined borders at 16 to 24 h after UV exposure”, and different other clinical trials highlighted the prevention and curing abilities of astaxanthin against photoaging [125,126,127,128].
11. Sulfated Polysaccharides
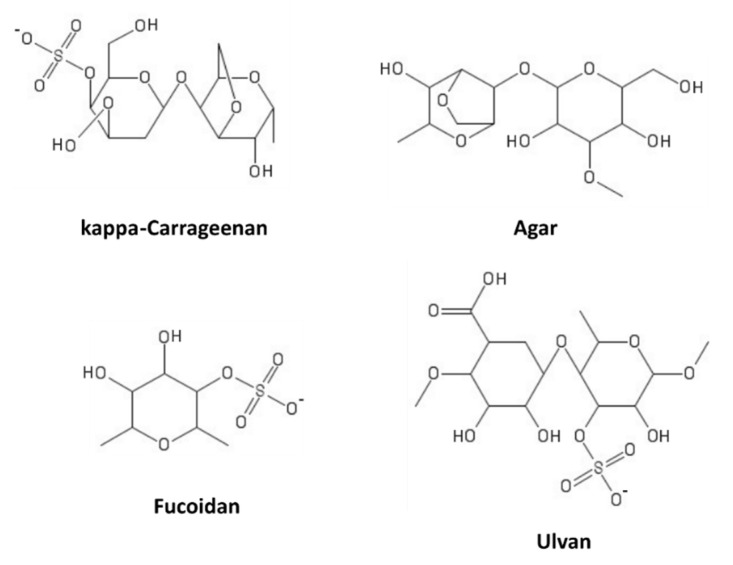
Sulfated polysaccharides (SPs) consist of more than 10 monosaccharides, possessing sulfate groups on the hydroxyls of sugar units. They are present in several natural sources, such as plants, or can be obtained by chemical substitution of non-sulfated polysaccharides [129]. Marine macroalgae (seaweeds) are rich sources of these compounds. Based on their sugar composition, stereochemistry, and glycosidic linkages, algal SPs can be distinguished in sulfated galactans (carrageenans and agarans, produced by red algae), ulvans (in green algae), and fucans (in brown algae) [130]. Some representatives of these classes are shown in Figure 8.
Figure 8.
Marine sulfated polysaccharides.
Some of them play structural roles in the cell wall composition, while others represent a sort of energy storage. Little is known about other physiological functions of SPs [131,132]; for example, they have been suggested to confer resistance against desiccation and osmotic stress in marine plants [133]. In addition, SPs have also been found in marine sponges, where they are presumably involved in species-specific cell aggregation and structural integrity, resembling the structural function of glycosaminoglycans in the connective tissues of mammals [134]. For their gelling properties, SPs are used in drug delivery systems, while, for their numerous bioactivities, including their ability to modulate metabolism and the immune system, and their anticancer, antimicrobial, and anticoagulant properties, they possess many applications in the cosmetic, nutraceutical, and pharmaceutical industries [135,136].
SPs do not absorb in the UV region; nevertheless, they showed strong UV-protective activities in in vitro and in vivo studies [137,138,139]. The protective effects of SPs against UVB-induced skin damage were tested in vitro in human dermal fibroblasts. SPs significantly reduced intracellular ROS levels and improved the viability of UVB-irradiated cells in a dose-dependent manner. Furthermore, they significantly inhibited intracellular collagenase and elastase activities, prevented collagen synthesis, and reduced MMP expression by the NF-κB and MAPK signaling pathways [138]. In addition, in vivo tests also demonstrated that SPs significantly reduce intracellular ROS levels, cell death, NO production, and lipid peroxidation levels in UVB-irradiated zebrafish in a dose-dependent manner [139]. These results pointed to SPs as potential ingredients in the cosmeceutical industry.
12. Other Potential Photoprotective Compounds of Marine Origin
Melanin-related compounds, such as eumelanin from sepia ink, have been proposed as natural UV filters and antioxidants, although their actual use in topical formulations is limited by the unaesthetic dark color conferred to the skin by any product containing eumelanin [140]. Another compound whose photoprotective abilities have recently been investigated is topsentin, a bis(indolyl)imidazole alkaloid, isolated from the marine sponge Spongosorites genitrix [141]. It has been shown to inhibit the expression of cyclooxygenase-2, an important enzyme involved in prostaglandin E2 (PGE2) synthesis, and miR-4485, a newly identified inflammation marker in UVB-irradiated human keratinocytes, and lowered PGE2 formation in a reconstructed human skin model [141].
Worthy of note is that some cyanobacteria, microalgae, and several marine invertebrates produce other amino acid derivatives such as sulfur-containing histidine endowed with antioxidant properties, and anti-inflammatory and antiproliferative activities in human cells [28,142,143,144,145,146]. They are named ovothiols due to their high concentration in sea urchin eggs [147] and exhibit a characteristic absorbance spectrum (200–320 nm) with a maximum around 257 nm [148]. In addition, the gene expression of the key enzyme involved in their biosynthesis is modulated by several environmental factors, including exposure to metals and toxins in sea urchin embryos [149] and to high light in microalgae [29]. The light-induced modulation of ovothiol biosynthesis suggests their possible role in the protection of microalgae from light-induced stress. This, together with ovothiols’ UV absorption properties, might point to them as potential candidates for photoprotective activities.
13. Concluding Remarks and Future Perspectives
The effective dissemination of scientific research through social media has led to increased awareness in the population about the risk of using certain chemicals in drugs and cosmetics as well as the health benefits associated with compounds obtained from natural resources. This has contributed to the expansion of the “blue biotechnology” from the marine environment as opposed to the “green technology” from the terrestrial one. Oceans occupy approximately 71% of the Earth's surface and they represent the “special” environment where the first living forms originated and evolved. Nevertheless, due to their vast extension, human knowledge concerning the sea is limited to a small part of marine environments and organisms, which still provide an extremely rich hotspot of biodiversity in terms of species and bioactive compounds, including natural sunscreens. A few products of marine origin with photoprotective properties have already appeared on the market, as mentioned in this review, but the number of these products is still very minimal considering the vastness of the sea; hence, there is great potential for breakthroughs in the not-too-distant future.
To exploit the potential of the marine environment, more efforts in research and development investments should be dedicated to the isolation and characterization of products from this natural resource, to discover the UV chromophores, and to evaluate their photoprotective properties and skin safety aspects. After all, it is reasonable to benefit from and mimic the sun-screening mechanisms that nature has optimized and refined over thousands of years to withstand sun overexposure. Marine-derived UV filters possess molar absorption coefficient (ε) values typically around or greater than 20,000, compared to the values of synthetic commercial UV filters that usually range from 10,000–30,000 [52], making nature-derived sunscreens particularly appealing and comparable with the man-made counterparts, currently available on the market. Furthermore, some natural compounds also possess broad UVA/UVB spectrum coverage [150] which would reduce the need for blending a series of filters to achieve this purpose. An additional feature in favor of marine-derived photoprotectants is that they are generally regarded as safe, since they are naturally found in seaweed and marine animals without any toxicity, and they have been part of human diets for hundreds of centuries, thus unlikely to be harmful. Moreover, marine-derived sunscreens may represent a possible biodegradable solution to curb environmental pollution coming from the use of all kinds of synthetic cosmetic products, not only sunscreens, which, once disposed and released into the environment, may contaminate ground and sea waters, destabilizing the health of marine habitats and wildlife such as coral reefs. UV filters washed off from sunscreens is another potential source of sea and freshwater pollution since sunscreen formulations are never 100% waterproof.
Some of the limitations preventing the full exploitation of marine resources for sun-screening purposes are those linked to the low concentrations of these compounds in natural producers, and whose biosynthesis is often influenced by environmental fluctuations, thus offering an overall low yield compared to that obtainable for synthetic products. Another limitation concerns large-scale sourcing or non-sustainable production methods that could disrupt already threatened marine ecosystems. These drawbacks could be overcome by resorting to sustainable, modern aquaculture farming techniques of UV-filter-producing marine sources, coupled with the optimization of growing and harvesting conditions for maximum yields of bioactive compounds for cosmeceutical purposes. Indeed, photosynthetic marine organisms have the inherent ability to self-renew and reproduce, ensuring sustainable supplies. In addition, exciting breakthroughs in this direction are coming from new developments and technical improvements for the cultivation of photosynthetic microorganisms in photobioreactors, such as the high-density long-term cultivation of the green alga Chlorella vulgaris [151]. Furthermore, engineering bacteria, yeast or other emerging industrial hosts, such as microalgae, to efficiently produce large quantities of natural sunscreens is another avenue that is being explored to meet market demands [72]. As appealing as this may seem, the downside of engineering microorganisms is that consumers may remain skeptical on using photoprotectants labeled as “natural” that have been obtained through genetic modification of living organisms.
Regardless of these issues, nature-derived sunscreen agents for sun-care products must first and foremost be both safe and have a beneficial photoprotective effect on human skin; hence, their concentration, compatibility, and stability in a sunscreen formulation must be carefully evaluated. Additionally, they must confer comparable protection from harmful UV rays over the entire exposure time as the currently available organic and inorganic UV filters present on the market. Furthermore, instead of fully replacing current UV filters in a formulation, natural marine-derived photoprotectants could at first be concomitantly added alongside with them, so that the concentrations of current UV filters may be reduced. This solution might lead to optimal synergistic effects since different nature-derived compounds can exert different UV defense mechanisms and combine them with those of synthetic counterparts.
In conclusion, as we move into an era where compounds derived from natural sources are gaining momentum for use in the cosmetic industry for improving skin protection in an effective and eco-conscious way, marine-derived photoprotectants may be a possible solution. In this review, we have outlined the different marine-derived compounds discovered so far, that could potentially be used for this purpose. They would address issues related to their impact on the health of marine habitats whilst providing the essential sun protection that human skin requires to protect it against damage caused by excessive sun exposure. However, further research is greatly needed before marine-derived compounds become effective and biocompatible mainstream ingredients in sun-care products that indeed attract consumers’ interest.
Author Contributions
Conceptualization, I.C., A.M. and E.D.; Writing—Original Draft Preparation, A.M.; Figures Preparation, A.M.; Writing—Review and Editing, I.C. and E.D.; Supervision, I.C. and E.D. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations All authors have read and agreed to the published version of the manuscript..
References
- 1.D’Orazio J., Jarrett S., Amaro-Ortiz A., Scott T. UV Radiation and the Skin. Int. J. Mol. Sci. 2013;14:12222–12248. doi: 10.3390/ijms140612222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Narayanan D.L., Saladi R.N., Fox J.L. Ultraviolet radiation and skin cancer. Int. J. Dermatol. 2010;49:978–986. doi: 10.1111/j.1365-4632.2010.04474.x. [DOI] [PubMed] [Google Scholar]
- 3.WHO. [(accessed on 10 May 2021)]; Available online: https://www.who.int/news-room/q-a-detail/radiation-sun-protection.
- 4.Zen Life and Travel. [(accessed on 10 May 2021)]; Available online: https://www.zenlifeandtravel.com/sunscreen-bans/
- 5.Schlumpf M., Schmid P., Durrer S., Conscience M., Maerkel K., Henseler M., Gruetter M., Herzog I., Reolon S., Ceccatelli R., et al. Endocrine activity and developmental toxicity of cosmetic UV filters—An update. Toxicology. 2004;205:113–122. doi: 10.1016/j.tox.2004.06.043. [DOI] [PubMed] [Google Scholar]
- 6.He H., Li A., Li S., Tang J., Li L., Xiong L. Natural components in sunscreens: Topical formulations with sun protection factor (SPF) Biomed. Pharmacother. 2020;134:111161. doi: 10.1016/j.biopha.2020.111161. [DOI] [PubMed] [Google Scholar]
- 7.Kirk J.T.O. Light and Photosynthesis in Aquatic Ecosystems. 3rd ed. Cambridge University Press; Cambridge, UK: 2011. [Google Scholar]
- 8.Brunet C., Chandrasekaran R., Barra L., Giovagnetti V., Corato F., Ruban A.V. Spectral radiation dependent photoprotective mechanism in the diatom Pseudo-nitzschia multistriata. PLoS ONE. 2014;9:e87015. doi: 10.1371/journal.pone.0087015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fortunato A.E., Jaubert M., Enomoto G., Bouly J.P., Raniello R., Thaler M., Malviya S., Bernardes J.S., Rappaport F., Gentili B., et al. Diatom Phytochromes Reveal the Existence of Far-Red-Light-Based Sensing in the Ocean. Plant Cell. 2016;28:616–628. doi: 10.1105/tpc.15.00928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Thompson C.L., Sancar A. Photolyase/cryptochrome blue-light photoreceptors use photon energy to repair DNA and reset the circadian clock. Oncogene. 2002;21:9043–9056. doi: 10.1038/sj.onc.1205958. [DOI] [PubMed] [Google Scholar]
- 11.Fortunato A.E., Annunziata R., Jaubert M., Bouly J.P., Falciatore A. Dealing with light: The widespread and multitasking cryptochrome/photolyase family in photosynthetic organisms. J. Plant Physiol. 2015;172:42–54. doi: 10.1016/j.jplph.2014.06.011. [DOI] [PubMed] [Google Scholar]
- 12.Jaubert M., Bouly J.P., Ribera d’ Alcalà M., Falciatore A. Light sensing and responses in marine microalgae. Curr. Opin. Plant Biol. 2017;37:70–77. doi: 10.1016/j.pbi.2017.03.005. [DOI] [PubMed] [Google Scholar]
- 13.Marshall J. Vision and lack of vision in the ocean. Curr. Biol. 2017;27:R494–R502. doi: 10.1016/j.cub.2017.03.012. [DOI] [PubMed] [Google Scholar]
- 14.Baylor D. How photons start vision. Proc. Natl. Acad. Sci. USA. 1996;93:560–565. doi: 10.1073/pnas.93.2.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yokoyama S. Evolution of Dim-Light and Color Vision Pigments. Annu. Rev. Genomics Hum. Genet. 2008;9:259–282. doi: 10.1146/annurev.genom.9.081307.164228. [DOI] [PubMed] [Google Scholar]
- 16.Porter M.L., Blasic J.R., Bok M.J., Cameron E.G., Pringle T., Cronin T.W., Robinson P.R. Shedding new light on opsin evolution. Proc. Biol. Sci. 2012;279:3–14. doi: 10.1098/rspb.2011.1819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Musilova Z., Cortesi F., Matschiner M., Davies W.I.L., Patel J.S., Stieb S.M., de Busserolles F., Malmstrøm M., Tørresen O.K., Brown C.J., et al. Vision using multiple distinct rod opsins in deep-sea fishes. Science. 2019;364:588–592. doi: 10.1126/science.aav4632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hargreaves B.R. Water column optics and penetration of UVR. In: Helbling E.W., Zagarese H., editors. UV Effects in Aquatic Organisms and Ecosystems. Volume 1. The Royal Society of Chemistry; London, UK: 2003. pp. 59–106. [Google Scholar]
- 19.Morris D.P., Zagarese H., Williamson C.E., Balseiro E.G., Hargreaves B.R., Modenutti B., Moeller R., Queimalinos C. The attenuation of solar UV radiation in lakes and the role of dissolved organic carbon. Limnol. Oceanogr. 1995;40:1381–1391. doi: 10.4319/lo.1995.40.8.1381. [DOI] [Google Scholar]
- 20.Dring M.J. Photocontrol of development in algae. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1988;39:157–174. doi: 10.1146/annurev.pp.39.060188.001105. [DOI] [Google Scholar]
- 21.Gao K., Wu Y., Li G., Wu H., Villafañe V.E., Helbling E.W. Solar UV radiation drives CO2 fixation in marine phytoplankton: A double-edged sword. Plant Physiol. 2007;144:54–59. doi: 10.1104/pp.107.098491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Johnsen S., Widder E. Ultraviolet absorption in transparent zooplankton and its implications for depth distribution and visual predation. Mar. Biol. 2001;138:717–730. doi: 10.1007/s002270000499. [DOI] [Google Scholar]
- 23.Cronin T.W., Bok M.J. Photoreception and vision in the ultraviolet. J. Exp. Biol. 2016;219:2790–2801. doi: 10.1242/jeb.128769. [DOI] [PubMed] [Google Scholar]
- 24.Niyogi K.K., Truong T.B. Evolution of flexible non-photochemical quenching mechanisms that regulate light harvesting in oxygenic photosynthesis. Curr. Opin. Plant Biol. 2013;16:307–314. doi: 10.1016/j.pbi.2013.03.011. [DOI] [PubMed] [Google Scholar]
- 25.Demmig-Adams B., Adams W.-W. Photoprotection and other responses of plants to high light stress. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1992;43:599–626. doi: 10.1146/annurev.pp.43.060192.003123. [DOI] [Google Scholar]
- 26.Rochaix J.-D. The Dynamics of the Photosynthetic Apparatus in Algae. In: Larkum A., Grossman A., Raven J., editors. Photosynthesis in Algae: Biochemical and Physiological Mechanisms. Volume 45. Springer International Publishing; Cham, Switzerland: 2020. pp. 57–82. [Google Scholar]
- 27.Goiris K., Muylaert K., De Cooman L. Microalgae as a Novel Source of Antioxidants for Nutritional Applications. In: Kim S.-K., editor. Handbook of Marine Microalgae. Academic Press; Cambridge, MA, USA: 2015. pp. 269–280. Chapter 17. [Google Scholar]
- 28.Milito A., Castellano I., Burn R., Seebeck F.P., Brunet C., Palumbo A. First evidence of ovothiol biosynthesis in marine diatoms. Free Rad. Biol. Med. 2020;152:680–688. doi: 10.1016/j.freeradbiomed.2020.01.010. [DOI] [PubMed] [Google Scholar]
- 29.Milito A., Orefice I., Smerilli A., Castellano I., Napolitano A., Brunet C., Palumbo A. Insights into the Light Response of Skeletonema marinoi: Involvement of Ovothiol. Mar. Drugs. 2020;18:477. doi: 10.3390/md18090477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Buma A.G.J., Boelen P., Jeffrey W.H. UVR- induced DNA damage in aquatic organisms. In: Helbling E.W., Zagarese H.E., editors. UV Effects in Aquatic Organisms and Ecosystems. Volume 1. The Royal Society of Chemistry; Cambridge, UK: 2003. pp. 289–327. [Google Scholar]
- 31.Heraud P., Beardall J. Changes in chlorophyll fluorescence during exposure of Dunaliella tertiolecta to UV radiation indicate a dynamic interaction between damage and repair processes. Photosynth. Res. 2000;63:123–134. doi: 10.1023/A:1006319802047. [DOI] [PubMed] [Google Scholar]
- 32.Gao K.S., Guan W.C., Helbling E.W. Effects of solar ultraviolet radiation on photosynthesis of the marine red tide alga Heterosigma akashiwo (Raphidophyceae) J. Photochem. Photobiol. B Biol. 2007;86:140–148. doi: 10.1016/j.jphotobiol.2006.05.007. [DOI] [PubMed] [Google Scholar]
- 33.Harrison J.W., Smith R.E. Effects of ultraviolet radiation on the productivity and composition of freshwater phytoplankton communities. Photochem. Photobiol. Sci. 2009;8:1218–1232. doi: 10.1039/b902604e. [DOI] [PubMed] [Google Scholar]
- 34.Jiang H.B., Qiu B.S. Inhibition of photosynthesis by UV-B exposure and its repair in the bloom-forming cyanobacterium Microcystis aeruginosa. J. Appl. Phycol. 2011;23:691–696. doi: 10.1007/s10811-010-9562-2. [DOI] [Google Scholar]
- 35.Häder D.P., Williamson C.E., Wängberg S.Å., Rautio M., Rose K.C., Gao K., Helbling E.W., Sinha R.P., Worrest R. Effects of UV radiation on aquatic ecosystems and interactions with other environmental factors. Photochem. Photobiol. Sci. 2015;14:108–126. doi: 10.1039/C4PP90035A. [DOI] [PubMed] [Google Scholar]
- 36.Dahms H.-U., Lee J.-S. UV radiation in marine ectotherms: Molecular effects and responses. Aquat. Toxicol. 2010;97:3–14. doi: 10.1016/j.aquatox.2009.12.002. [DOI] [PubMed] [Google Scholar]
- 37.Alves R.N., Agustí S. Effect of ultraviolet radiation (UVR) on the life stages of fish. Rev. Fish Biol. Fish. 2020;30:335–372. doi: 10.1007/s11160-020-09603-1. [DOI] [Google Scholar]
- 38.Lesser M.P., Lamare M.D., Barker M.F. Transmission of ultraviolet radiation through the Antarctic annual sea ice and its biological effects on sea urchin embryos. Limnol. Oceanogr. 2004;49:1957–1963. doi: 10.4319/lo.2004.49.6.1957. [DOI] [Google Scholar]
- 39.Figueroa F.L., Escassi L., Pérez-Rodríguez E., Korbee-Peinado N., Giles A.D., Johnsen G. Effects of short-term irradiation on photoinhibition and accumulation of mycosporine-like amino acids in sun and shade species of the red algal genus Porphyra. J. Photochem. Photobiol. B Biol. 2003;69:21–30. doi: 10.1016/S1011-1344(02)00388-3. [DOI] [PubMed] [Google Scholar]
- 40.Lalegerie F., Lajili S., Bedoux G., Taupin L., Stiger-Pouvreau V., Connan S. Photoprotective compounds in red macroalgae from Brittany: Considerable diversity in mycosporine-like amino acids (MAAs) Mar. Environ. Res. 2019;147:37–48. doi: 10.1016/j.marenvres.2019.04.001. [DOI] [PubMed] [Google Scholar]
- 41.Nair R., Maseeh A. Vitamin D: The “sunshine” vitamin. J. Pharmacol. Pharm. 2012;3:118–126. doi: 10.4103/0976-500X.95506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Young A.R. Acute effects of UVR on human eyes and skin. Prog. Biophys. Mol. Biol. 2006;92:80–85. doi: 10.1016/j.pbiomolbio.2006.02.005. [DOI] [PubMed] [Google Scholar]
- 43.Schulman J.M., Fisher D.E. Indoor ultraviolet tanning and skin cancer: Health risks and opportunities. Curr. Opin. Oncol. 2009;21:144–149. doi: 10.1097/CCO.0b013e3283252fc5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Agar N., Young A.R. Melanogenesis: A photoprotective response to DNA damage? Mutat. Res. 2005;571:121–132. doi: 10.1016/j.mrfmmm.2004.11.016. [DOI] [PubMed] [Google Scholar]
- 45.Brenner M., Hearing V.J. The protective role of melanin against UV damage in human skin. Photochem. Photobiol. 2008;84:539–549. doi: 10.1111/j.1751-1097.2007.00226.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gallagher R.P., Lee T.K. Adverse effects of ultraviolet radiation: A brief review. Prog. Biophys. Mol. Biol. 2006;92:119–131. doi: 10.1016/j.pbiomolbio.2006.02.011. [DOI] [PubMed] [Google Scholar]
- 47.Agarwal N. UVR and Role of Pigmentation in Skin Aging and Cancer. In: Dwivedi A., Agarwal N., Ray L., Tripathi A., editors. Skin Aging & Cancer. Springer; Singapore: 2019. pp. 59–69. [Google Scholar]
- 48.Hart P.H., Norval M. Ultraviolet radiation-induced immunosuppression and its relevance for skin carcinogenesis. Photochem. Photobiol. Sci. 2018;17:1872–1884. doi: 10.1039/C7PP00312A. [DOI] [PubMed] [Google Scholar]
- 49.Damiani E., Ullrich S.E. Understanding the connection between platelet-activating factor, a UV-induced lipid mediator of inflammation, immune suppression and skin cancer. Prog. Lipid Res. 2016;63:14–27. doi: 10.1016/j.plipres.2016.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Osterwalder U., Sohn M., Herzog B. Global state of sunscreens. Photodermatol. Photoimmunol. Photomed. 2014;30:62–80. doi: 10.1111/phpp.12112. [DOI] [PubMed] [Google Scholar]
- 51.Sánchez-Quiles D., Tovar-Sánchez A. Sunscreens as a source of hydrogen peroxide production in coastal waters. Environ. Sci. Technol. 2014;48:9037–9042. doi: 10.1021/es5020696. [DOI] [PubMed] [Google Scholar]
- 52.Shaath N. Sunscreens: Regulations and Commercial Development. 3rd ed. Taylor & Francis; Boca Raton, FL, USA: 2005. p. 222. Chapter 13. [Google Scholar]
- 53.Shaath N.A. Ultraviolet filters. Photochem. Photobiol. Sci. 2010;9:464–469. doi: 10.1039/b9pp00174c. [DOI] [PubMed] [Google Scholar]
- 54.Sendra M., Sánchez-Quiles D., Blasco J., Moreno-Garrido I., Lubián L.M., Pérez-García S., Tovar-Sánchez A. Effects of TiO2 nanoparticles and sunscreens on coastal marine microalgae: Ultraviolet radiation is key variable for toxicity assessment. Environ. Int. 2017;98:62–68. doi: 10.1016/j.envint.2016.09.024. [DOI] [PubMed] [Google Scholar]
- 55.Corinaldesi C., Damiani E., Marcellini F., Falugi C., Tiano L., Brugè F., Danovaro R. Sunscreen products impair the early developmental stages of the sea urchin Paracentrotus lividus. Sci. Rep. 2017;7:7815. doi: 10.1038/s41598-017-08013-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Corinaldesi C., Marcellini F., Nepote E., Damiani E., Danovaro R. Impact of inorganic UV filters contained in sunscreen products on tropical stony corals (Acropora spp.) Sci. Total Environ. 2018;637:1279–1285. doi: 10.1016/j.scitotenv.2018.05.108. [DOI] [PubMed] [Google Scholar]
- 57.Danovaro R., Bongiorni L., Corinaldesi C., Giovannelli D., Damiani E., Astolfi P., Greci L., Pusceddu A. Sunscreens cause coral bleaching by promoting viral infections. Environ. Health Perspect. 2008;116:441–447. doi: 10.1289/ehp.10966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Amador-Castro F., Rodriguez-Martinez V., Carrillo-Nieves D. Robust natural ultraviolet filters from marine ecosystems for the formulation of environmental friendlier bio-sunscreens. Sci. Total Environ. 2020;749:141576. doi: 10.1016/j.scitotenv.2020.141576. [DOI] [PubMed] [Google Scholar]
- 59.Carreto J.I., Carignan M.O. Mycosporine-Like Amino Acids: Relevant Secondary Metabolites. Chemical and Ecological Aspects. Mar. Drugs. 2011;9:387–446. doi: 10.3390/md9030387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Singh D.K., Pathak J., Pandey A., Singh V., Rajneesh H.A., Kumar D., Sinha R.P. Ultraviolet-screening compound mycosporine-like amino acids in cyanobacteria: Biosynthesis, functions, and applications. In: Singh P.K., Kumar A., Singh V.K., Shrivastava A.K., editors. Advances in Cyanobacterial Biology. Academic Press; Cambridge, MA, USA: 2020. pp. 219–233. Chapter 15. [Google Scholar]
- 61.Xiong F., Kopecky J., Nedbal L. The occurrence of UV-B absorbing mycosporine-like amino acids in freshwater and terrestrial microalgae (Chlorophyta) Aquat. Bot. 1999;63:37–49. doi: 10.1016/S0304-3770(98)00106-5. [DOI] [Google Scholar]
- 62.Sun Y., Zhang N., Zhou J., Dong S., Zhang X., Guo L., Guo G. Distribution, Contents, and Types of Mycosporine-Like Amino Acids (MAAs) in Marine Macroalgae and a Database for MAAs Based on These Characteristics. Mar. Drugs. 2020;18:43. doi: 10.3390/md18010043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Rosic N.N., Dove S. Mycosporine-Like Amino Acids from Coral Dinoflagellates. Appl. Environ. Microbiol. 2011;77:8478–8486. doi: 10.1128/AEM.05870-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sinha R.P., Singh S.P., Häder D.-P. Database on mycosporines and mycosporine-like amino acids (MAAs) in fungi, cyanobacteria, macroalgae, phytoplankton and animals. J. Photochem. Photobiol. B. 2007;89:29–35. doi: 10.1016/j.jphotobiol.2007.07.006. [DOI] [PubMed] [Google Scholar]
- 65.Shick J.M., Romaine-Lioud S., Ferrier-Pages C., Gattuso J.P. Ultraviolet-B radiation stimulates shikimate pathway-dependent accumulation of mycosporine-like amino acids in the coral Stylophora pistillata despite decreases in its population of symbiotic dinoflagellates. Limnol. Oceanogr. 1999;44:1667–1682. doi: 10.4319/lo.1999.44.7.1667. [DOI] [Google Scholar]
- 66.Balskus E.P., Walsh C.T. The genetic and molecular basis for sunscreen biosynthesis in cyanobacteria. Science. 2010;329:1653–1656. doi: 10.1126/science.1193637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Newman S.J., Dunlap W.C., Nicol S., Ritz D. Antarctic krill (Euphausia superba) acquire a UV-absorbing mycosporine-like amino acid from dietary algae. J. Exp. Mar. Biol. Ecol. 2000;255:93–110. doi: 10.1016/S0022-0981(00)00293-8. [DOI] [PubMed] [Google Scholar]
- 68.Shick J.M., Dunlap W.C. Mycosporine-like amino acids and related gadusols: Biosynthesis, accumulation, and UV-protective functions in aquatic organisms. Annu. Rev. Physiol. 2002;64:223–262. doi: 10.1146/annurev.physiol.64.081501.155802. [DOI] [PubMed] [Google Scholar]
- 69.Fitzgerald L.M., Szmant A.M. Biosynthesis of ‘essential’ amino acids by scleractinian corals. Biochem. J. 1997;322:213–221. doi: 10.1042/bj3220213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Starcevic A., Akthar S., Dunlap W.C., Shick J.M., Hranueli D., Cullum J., Long P.F. Enzymes of the shikimic acid pathway encoded in the genome of a basal metazoan, Nematostella vectensis, have microbial origins. Proc. Natl. Acad. Sci. USA. 2008;105:2533–2537. doi: 10.1073/pnas.0707388105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Shinzato C., Shoguchi E., Kawashima T., Hamada M., Hisata K., Tanaka M., Fujie M., Fujiwara M., Koyanagi R., Ikuta T., et al. Using the Acropora digitifera genome to understand coral responses to environmental change. Nature. 2011;476:320–323. doi: 10.1038/nature10249. [DOI] [PubMed] [Google Scholar]
- 72.Osborn A.R., Almabruk K.H., Holzwarth G., Asamizu S., LaDu J., Kean K.M., Karplus P.A., Tanguay R.L., Bakalinsky A.T., Mahmud T. De novo synthesis of a sunscreen compound in vertebrates. eLife. 2015;4:e05919. doi: 10.7554/eLife.05919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Brotherton C.A., Balskus E.P. Shedding light on sunscreen biosynthesis in zebrafish. eLife. 2015;4:e07961. doi: 10.7554/eLife.07961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.De la Coba F., Aguilera J., Korbee N., de Gálvez M.V., Herrera-Ceballos E., Álvarez-Gómez F., Figueroa F.L. UVA and UVB Photoprotective Capabilities of Topical Formulations Containing Mycosporine-like Amino Acids (MAAs) through Different Biological Effective Protection Factors (BEPFs) Mar. Drugs. 2019;17:55. doi: 10.3390/md17010055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Oren A., Gunde-Cimerman N. Mycosporines and mycosporine-like amino acids: UV protectants or multipurpose secondary metabolites? FEMS Microbiol. Lett. 2007;269:1–10. doi: 10.1111/j.1574-6968.2007.00650.x. [DOI] [PubMed] [Google Scholar]
- 76.Schmid D., Schürch C., Zülli F. Mycosporine-like amino acids from red algae protect against premature skin-aging. Euro Cosmet. 2006;9:1–4. [Google Scholar]
- 77.Cheewinthamrongrod V., Kageyama H., Palaga T., Takabe T., Waditee-Sirisattha R. DNA damage protecting and free radical scavenging properties of mycosporine-2-glycine from the Dead Sea cyanobacterium in A375 human melanoma cell lines. J. Photochem. Photobiol. B Biol. 2016;164:289–295. doi: 10.1016/j.jphotobiol.2016.09.037. [DOI] [PubMed] [Google Scholar]
- 78.Lawrence K.P., Gacesa R., Long P.F., Young A.R. Molecular photoprotection of human keratinocytes in vitro by the naturally occurring mycosporine-like amino acid palythine. Br. J. Dermatol. 2018;178:1353–1363. doi: 10.1111/bjd.16125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kageyama H., Waditee-Sirisattha R. Antioxidative, Anti-Inflammatory, and Anti-Aging Properties of Mycosporine-Like Amino Acids: Molecular and Cellular Mechanisms in the Protection of Skin-Aging. Mar. Drugs. 2019;17:222. doi: 10.3390/md17040222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Schmidt E.W. An Enzymatic Route to Sunscreens. ChemBioChem. 2011;12:363–365. doi: 10.1002/cbic.201000709. [DOI] [PubMed] [Google Scholar]
- 81.Chrapusta E., Kaminski A., Duchnik K., Bober B., Adamski M., Bialczyk J. Mycosporine-Like Amino Acids: Potential Health and Beauty Ingredients. Mar. Drugs. 2017;15:326. doi: 10.3390/md15100326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Rosic N.N. Mycosporine-Like Amino Acids: Making the Foundation for Organic Personalised Sunscreens. Mar. Drugs. 2019;17:638. doi: 10.3390/md17110638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Proteau P.J., Gerwick W.H., Garcia-Pichel F., Castenholz R. The structure of scytonemin, an ultraviolet sunscreen pigment from the sheaths of cyanobacteria. Experientia. 1993;49:825–829. doi: 10.1007/BF01923559. [DOI] [PubMed] [Google Scholar]
- 84.Büdel B., Karsten U., Garcia-Pichel F. Ultraviolet-absorbing scytonemin and mycosporine-like amino acid derivatives in exposed, rock-inhabiting cyanobacterial lichens. Oecologia. 1997;112:165–172. doi: 10.1007/s004420050296. [DOI] [PubMed] [Google Scholar]
- 85.Balskus E.P., Case R.J., Walsh C.T. The biosynthesis of cyanobacterial sunscreen scytonemin in microbial mat communities. FEMS Microbiol. Ecol. 2011;77:322–332. doi: 10.1111/j.1574-6941.2011.01113.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Matsui K., Nazifi E., Hirai Y., Wada N., Matsugo S., Sakamoto T. The cyanobacterial UVabsorbing pigment scytonemin displays radical scavenging activity. J. Gen. Appl. Microbiol. 2012;58:137–144. doi: 10.2323/jgam.58.137. [DOI] [PubMed] [Google Scholar]
- 87.Pathak J., Sonker A.S., Richa R., Rajneesh R., Kannaujiya V.K., Singh V., Ahmed H., Sinha R.P. Screening and partial purification of photoprotective pigment, scytonemin from cyanobacteria dominated biological crusts dwelling on the historical monuments in and around Varanasi, India. Microbiol. Res. 2017;8:6559. doi: 10.4081/mr.2017.6559. [DOI] [Google Scholar]
- 88.Simeonov A., Michaelian K. Properties of cyanobacterial UV-absorbing pigments suggest their evolution was driven by optimizing photon dissipation rather than photoprotection. arXiv Prepr. 20171702.03588 [Google Scholar]
- 89.Pandey A., Pathak J., Singh D.K., Ahmed H., Singh V., Kumar D., Sinha R.P. Photoprotective role of UV-screening pigment scytonemin against UV-B-induced damages in the heterocyst-forming cyanobacterium Nostoc sp. strain HKAR-2. Braz. J. Bot. 2020;43:67–80. doi: 10.1007/s40415-020-00589-5. [DOI] [Google Scholar]
- 90.Pathak J., Pandey A., Maurya P.K., Rajneesh R., Sinha R.P., Singh S.P. Cyanobacterial Secondary Metabolite Scytonemin: A Potential Photoprotective and Pharmaceutical Compound. Proc. Natl. Acad. Sci. India Sect. B Biol. Sci. 2020;90:467–481. doi: 10.1007/s40011-019-01134-5. [DOI] [Google Scholar]
- 91.Sinha R.P., Pathak J., Ahmed R.H., Pandey A., Singh P.R., Mishra S., Häder D.-P. Cyanobacterial photoprotective compounds: Characterization and utilization in human welfare. In: Sinha R.P., Häder D.-P., editors. Natural Bioactive Compounds. Academic Press; Cambridge, MA, USA: 2021. pp. 83–114. Chapter 5. [Google Scholar]
- 92.Pandey A., Sinha R.P. Biophysical Characterization of Scytonemin: A Sunsreening Pigment in Cyanobacterial Biofilms. JIAPS. 2019;23:411–424. [Google Scholar]
- 93.Kang M.R., Jo S.A., Lee H., Yoon Y.D., Kwon J.-H., Yang J.-W., Choi B.J., Park K.H., Lee M.Y., Lee C.W., et al. Inhibition of Skin Inflammation by Scytonemin, an Ultraviolet Sunscreen Pigment. Mar. Drugs. 2020;18:300. doi: 10.3390/md18060300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Stevenson C.S., Capper E.A., Roshak A.K., Marquez B., Eichman C., Jackson J.R., Mattern M., Gerwick W.H., Jacobs R.S., Marshall L.A. The identification and characterization of the marine natural product scytonemin as a novel antiproliferative pharmacophore. J. Pharm. Exp. Ther. 2002;303:858–866. doi: 10.1124/jpet.102.036350. [DOI] [PubMed] [Google Scholar]
- 95.Itoh T., Tsuzuki R., Tanaka T., Ninomiya M., Yamaguchi Y., Takenaka H., Ando M., Tsukamasa Y., Koketsu M. Reduced scytonemin isolated from Nostoc commune induces autophagic cell death in human T-lymphoid cell line Jurkat cells. Food Chem. Toxicol. 2013;60:76–82. doi: 10.1016/j.fct.2013.07.016. [DOI] [PubMed] [Google Scholar]
- 96.Itoh T., Koketsu M., Yokota N., Touho S., Ando M., Tsukamasa Y. Reduced scytonemin isolated from Nostoc commune suppresses LPS/IFNc-induced NO production in murine macrophage RAW264 cells by inducing hemeoxygenase-1 expression via the Nrf2/ARE pathway. Food Chem. Toxicol. 2014;69:330–338. doi: 10.1016/j.fct.2014.04.019. [DOI] [PubMed] [Google Scholar]
- 97.Helms G.L., Moore R.E., Niemczura W.P., Patterson G.M.L., Tomer K.B., Gross M.L. Scytonemin A, a novel calcium antagonist from a blue-green alga. J. Org. Chem. 1988;53:1298–1307. doi: 10.1021/jo00241a033. [DOI] [Google Scholar]
- 98.Edwards H.G., Sadooni F., Vítek P., Jehlička J. Raman spectroscopy of the Dukhan sabkha: Identification of geological and biogeological molecules in an extreme environment. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2010;368:3099–3107. doi: 10.1098/rsta.2010.0101. [DOI] [PubMed] [Google Scholar]
- 99.Varnali T., Edwards H.G.M. Reduced and oxidised scytonemin: Theoretical protocol for Raman spectroscopic Cyanobacterial Secondary Metabolite Scytonemin: A Potential Photoprotective and identification of potential key biomolecules for astrobiology. Spectrochim. Acta A. 2014;117:72–77. doi: 10.1016/j.saa.2013.08.002. [DOI] [PubMed] [Google Scholar]
- 100.Ignat I., Volf I., Popa V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011;126:1821–1835. doi: 10.1016/j.foodchem.2010.12.026. [DOI] [PubMed] [Google Scholar]
- 101.Durazzo A., Lucarini M., Souto E.B., Cicala C., Caiazzo E., Izzo A.A., Novellino E., Santini A. Polyphenols: A concise overview on the chemistry, occurrence, and human health. Phytother. Res. 2019;33:2221–2243. doi: 10.1002/ptr.6419. [DOI] [PubMed] [Google Scholar]
- 102.Spagnuolo C., Moccia S., Russo G.L. Anti-inflammatory effects of flavonoids in neurodegenerative disorders. Eur. J. Med. Chem. 2018;153:105–115. doi: 10.1016/j.ejmech.2017.09.001. [DOI] [PubMed] [Google Scholar]
- 103.Russo G.L., Spagnuolo C., Russo M., Tedesco I., Moccia S., Cervellera C. Mechanisms of aging and potential role of selected polyphenols in extending healthspan. Biochem. Pharmacol. 2020;173:113719. doi: 10.1016/j.bcp.2019.113719. [DOI] [PubMed] [Google Scholar]
- 104.Russo M., Moccia S., Spagnuolo C., Tedesco I., Russo G.L. Roles of flavonoids against coronavirus infection. Chem. Biol. Interact. 2020;328:109211. doi: 10.1016/j.cbi.2020.109211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Martins B.T., Correia da Silva M., Pinto M., Cidade H., Kijjoa A. Marine natural flavonoids: Chemistry and biological activities. Nat. Prod. Res. 2019;33:3260–3272. doi: 10.1080/14786419.2018.1470514. [DOI] [PubMed] [Google Scholar]
- 106.Agati G., Brunetti C., Di Ferdinando M., Ferrini F., Pollastri S., Tattini M. Functional roles of flavonoids in photoprotection: New evidence, lessons from the past. Plant Physiol. Biochem. 2013;72:35–45. doi: 10.1016/j.plaphy.2013.03.014. [DOI] [PubMed] [Google Scholar]
- 107.Saewan N., Jimtaisong A. Photoprotection of natural flavonoids. J. Appl. Pharm. Sci. 2013;3:129–141. [Google Scholar]
- 108.Piao M.J., Zhang R., Lee N.H., Hyun J.W. Protective effect of triphlorethol-A against ultraviolet B-mediated damage of human keratinocytes. J. Photochem. Photobiol. B. 2012;106:74–80. doi: 10.1016/j.jphotobiol.2011.10.007. [DOI] [PubMed] [Google Scholar]
- 109.Piao M.J., Zhang R., Lee N.H., Hyun J.W. Phloroglucinol attenuates ultraviolet B radiation-induced matrix metalloproteinase-1 production in human keratinocytes via inhibitory actions against mitogen-activated protein kinases and activator protein-1. Photochem. Photobiol. 2012;88:381–388. doi: 10.1111/j.1751-1097.2012.01074.x. [DOI] [PubMed] [Google Scholar]
- 110.Piao M.J., Lee N.H., Chae S., Hyun J.W. Eckol inhibits ultraviolet B-induced cell damage in human keratinocytes via a decrease in oxidative stress. Biol. Pharm. Bull. 2012;35:873–880. doi: 10.1248/bpb.35.873. [DOI] [PubMed] [Google Scholar]
- 111.Ko S.-C., Cha S.-H., Heo S.-J., Lee S.-H., Kang S.-M., Jeon Y.-J. Protective effect of Ecklonia cava on UVB-induced oxidative stress: In vitro and in vivo zebrafish model. J. Appl. Phycol. 2011;23:697–708. doi: 10.1007/s10811-010-9565-z. [DOI] [Google Scholar]
- 112.Vo T.S., Kim S.-K., Ryu B., Ngo D.H., Yoon N.-Y., Bach L.G., Hang N.T.N., Ngo D.N. The Suppressive Activity of Fucofuroeckol-A Derived from Brown Algal Ecklonia stolonifera Okamura on UVB-Induced Mast Cell Degranulation. Mar. Drugs. 2018;16:1. doi: 10.3390/md16010001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Sotiropoulou G., Zingkou E., Pampalakis G. Redirecting drug repositioning to discover innovative cosmeceuticals. Exp. Dermatol. 2021;30:628–644. doi: 10.1111/exd.14299. [DOI] [PubMed] [Google Scholar]
- 114.Merhan O. The Biochemistry and Antioxidant Properties of Carotenoids. In: Cvetkovic D.J., Nikolic G.S., editors. Carotenoids. IntechOpen Limited; London, UK: 2017. [Google Scholar]
- 115.Hashimoto H., Uragami C., Cogdell R.J. Subcellular Biochemistry. Volume 79. Springer; Berlin/Heidelberg, Germany: 2016. Carotenoids and Photosynthesis; pp. 111–139. [DOI] [PubMed] [Google Scholar]
- 116.Rastogi R.P., Richa R., Sinha R.P., Singh S.P., Häder D.-P. Photoprotective compounds from marine organisms. J. Ind. Microbiol. Biotechnol. 2010;37:537–558. doi: 10.1007/s10295-010-0718-5. [DOI] [PubMed] [Google Scholar]
- 117.Vílchez C., Forján E., Cuaresma M., Bédmar F., Garbayo I., Vega J.M. Marine Carotenoids: Biological Functions and Commercial Applications. Mar. Drugs. 2011;9:319–333. doi: 10.3390/md9030319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Stahl W., Sies H. Photoprotection by dietary carotenoids: Concept, mechanisms, evidence and future development. Mol. Nutr. Food Res. 2012;56:287–295. doi: 10.1002/mnfr.201100232. [DOI] [PubMed] [Google Scholar]
- 119.Wertz K., Hunziker P.B., Seifert N., Riss G., Neeb M., Steiner G., Hunziker W., Goralczyk R. beta-Carotene interferes with ultraviolet light A-induced gene expression by multiple pathways. J. Investig. Dermatol. 2005;124:428–434. doi: 10.1111/j.0022-202X.2004.23593.x. [DOI] [PubMed] [Google Scholar]
- 120.Catanzaro E., Bishayee A., Fimognari C. On a Beam of Light: Photoprotective Activities of the Marine Carotenoids Astaxanthin and Fucoxanthin in Suppression of Inflammation and Cancer. Mar. Drugs. 2020;18:544. doi: 10.3390/md18110544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Hama S., Takahashi K., Inai Y., Shiota K., Sakamoto R., Yamada A., Tsuchiya H., Kanamura K., Yamashita E., Kogure K. Protective effects of topical application of a poorly soluble antioxidant astaxanthin liposomal formulation on ultraviolet-induced skin damage. J. Pharm. Sci. 2012;101:2909–2916. doi: 10.1002/jps.23216. [DOI] [PubMed] [Google Scholar]
- 122.Komatsu T., Sasaki S., Manabe Y., Hirata T., Sugawara T. Preventive effect of dietary astaxanthin on UVA-induced skin photoaging in hairless mice. PLoS ONE. 2017;12:e0171178. doi: 10.1371/journal.pone.0171178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Li X., Matsumoto T., Takuwa M., Saeed Ebrahim Shaiku Ali M., Hirabashi T., Kondo H., Fujino H. Protective effects of astaxanthin supplementation against ultraviolet-induced photoaging in hairless mice. Biomedicines. 2020;8:18. doi: 10.3390/biomedicines8020018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Ito N., Seki S., Ueda F. The protective role of Astaxanthin for UV-induced skin deterioration in healthy people-a randomized, double-blind, placebo-controlled trial. Nutrients. 2018;10:817. doi: 10.3390/nu10070817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Tominaga K., Hongo N., Karato M., Yamashita E. Cosmetic benefits of astaxanthin on humans subjects. Acta Biochim. Pol. 2012;59:43–47. doi: 10.18388/abp.2012_2168. [DOI] [PubMed] [Google Scholar]
- 126.Chalyk N.E., Klochkov V.A., Bandaletova T.Y., Kyle N.H., Petyaev I.M. Continuous Astaxanthin intake reduces oxidative stress and reverses age-related morphological changes of residual skin surface components in middle-aged volunteers. Nutr. Res. 2017;48:40–48. doi: 10.1016/j.nutres.2017.10.006. [DOI] [PubMed] [Google Scholar]
- 127.Yoon H.-S., Cho H.H., Cho S., Lee S.-R., Shin M.-H., Chung J.H. Supplementing with dietary astaxanthin combined with collagen hydrolysate improves facial elasticity and decreases matrix metalloproteinase-1 and -12 expression: A comparative study with placebo. J. Med. Food. 2014;17:810–816. doi: 10.1089/jmf.2013.3060. [DOI] [PubMed] [Google Scholar]
- 128.Ng Q.X., De Deyn M.L.Z.Q., Loke W., Foo N.X., Chan H.W., Yeo W.S. Effects of Astaxanthin supplementation on skin health: A systematic review of clinical studies. J. Diet. Suppl. 2020;18:169–182. doi: 10.1080/19390211.2020.1739187. [DOI] [PubMed] [Google Scholar]
- 129.Wang Z., Xie J., Shen M., Nie S., Xie M. Sulfated modification of polysaccharides: Synthesis, characterization and bioactivities. Trends Food Sci. Technol. 2018;74:147–157. doi: 10.1016/j.tifs.2018.02.010. [DOI] [Google Scholar]
- 130.Jiao G., Yu G., Zhang J., Ewart H.S. Chemical Structures and Bioactivities of Sulfated Polysaccharides from Marine Algae. Mar. Drugs. 2011;9:196–223. doi: 10.3390/md9020196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Percival E. The polysaccharides of green, red and brown seaweeds: Their basic structure, biosynthesis and function. Br. Phycol. J. 1979;14:103–117. doi: 10.1080/00071617900650121. [DOI] [Google Scholar]
- 132.Winberg P.C., Fitton H.J., Stringer D., Karpiniec S.S., Gardiner V.-A. Controlling Seaweed Biology, Physiology and Metabolic Traits in Production for Commercially Relevant Bioactives in Glycobiology. Adv. Bot. Res. 2014;71:221–252. [Google Scholar]
- 133.Aquino R.S., Grativol C., Mourão P.A.S. Rising from the Sea: Correlations between Sulfated Polysaccharides and Salinity in Plants. PLoS ONE. 2011;6:e18862. doi: 10.1371/journal.pone.0018862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Zierer M.S., Mourão P.A.S. A wide diversity of sulfated polysaccharides are synthesized by different species of marine sponges. Carbohydr. Res. 2000;328:209–216. doi: 10.1016/S0008-6215(00)00076-8. [DOI] [PubMed] [Google Scholar]
- 135.Sanjeewa K.K.A., Kang N., Ahn G., Jee Y., Kim Y., Jeon Y. Bioactive potentials of sulfated polysaccharides isolated from brown seaweed Sargassum spp in related to human health applications: A review. Food Hydrocoll. 2018;81:200–208. doi: 10.1016/j.foodhyd.2018.02.040. [DOI] [Google Scholar]
- 136.Tanna B., Mishra A. Nutraceutical Potential of Seaweed Polysaccharides: Structure, Bioactivity, Safety, and Toxicity. Compr. Rev. Food Sci. Food Saf. 2019;18:817–831. doi: 10.1111/1541-4337.12441. [DOI] [PubMed] [Google Scholar]
- 137.Thevanayagam H., Mohamed S.M., Chu W. Assessment of UVB-photoprotective and antioxidative activities of carrageenan in keratinocytes. J. Appl. Phycol. 2014;26:1813–1821. doi: 10.1007/s10811-013-0207-0. [DOI] [Google Scholar]
- 138.Wang L., Lee W., Oh J.Y., Cui Y.R., Ryu B., Jeon Y.-J. Protective Effect of Sulfated Polysaccharides from Celluclast-Assisted Extract of Hizikia fusiforme Against Ultraviolet B-Induced Skin Damage by Regulating NF-κB, AP-1, and MAPKs Signaling Pathways In Vitro in Human Dermal Fibroblasts. Mar. Drugs. 2018;16:239. doi: 10.3390/md16070239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Wang L., Oh J.Y., Yang H., Kim H.S., Jeon Y.-J. Protective effect of sulfated polysaccharides from a Celluclast-assisted extract of Hizikia fusiforme against ultraviolet B-induced photoaging in vitro in human keratinocytes and in vivo in zebrafish. Mar. Life Sci. Technol. 2019;1:104–111. doi: 10.1007/s42995-019-00006-1. [DOI] [Google Scholar]
- 140.Solano F. Photoprotection and Skin Pigmentation: Melanin-Related Molecules and Some Other New Agents Obtained from Natural Sources. Molecules. 2020;25:1537. doi: 10.3390/molecules25071537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Hwang J., Kim D., Park J.S., Park H.J., Shin J., Lee S.K. Photoprotective Activity of Topsentin, A Bis(Indole) Alkaloid from the Marine Sponge Spongosorites genitrix, by Regulation of COX-2 and Mir-4485 Expression in UVB-Irradiated Human Keratinocyte Cells. Mar. Drugs. 2020;18:87. doi: 10.3390/md18020087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Castellano I., Seebeck F. On ovothiol biosynthesis and biological roles: From life in the ocean to therapeutic potential. Nat. Prod. Rep. 2018;35:1241–1250. doi: 10.1039/C8NP00045J. [DOI] [PubMed] [Google Scholar]
- 143.Gerdol M., Sollitto M., Pallavicini A., Castellano I. The complex evolutionary history of sulfoxide synthase in ovothiol biosynthesis. Proc. R. Soc. B. 2019;286:20191812. doi: 10.1098/rspb.2019.1812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Brancaccio M., Russo M., Masullo M., Palumbo A., Russo G.L., Castellano I. Sulfur-containing histidine compounds inhibit γ-glutamyl transpeptidase activity in human cancer cells. J. Biol. Chem. 2019;294:14603–14614. doi: 10.1074/jbc.RA119.009304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Milito A., Brancaccio M., Lisurek M., Masullo M., Palumbo A., Castellano I. Probing the Interactions of Sulfur-Containing Histidine Compounds with Human Gamma-Glutamyl Transpeptidase. Mar. Drugs. 2019;17:650. doi: 10.3390/md17120650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Milito A., Brancaccio M., D’ Argenio G., Castellano I. Natural Sulfur-Containing Compounds: An Alternative Therapeutic Strategy against Liver Fibrosis. Cells. 2019;8:1356. doi: 10.3390/cells8111356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Palumbo A., Castellano I., Napolitano A. Ovothiol: A potent natural antioxidant from marine organisms. In: Barre S., Bates S.S., editors. Blue Biotechnology: Production and Use of Marine Molecules. Wiley VCH; Weinheim, Germany: 2018. pp. 583–610. [Google Scholar]
- 148.Russo G.L., Russo M., Castellano I., Napolitano A., Palumbo A. Ovothiol isolated from sea urchin oocytes induces autophagy in the Hep-G2 cell line. Mar. Drugs. 2014;12:4069–4085. doi: 10.3390/md12074069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Castellano I., Migliaccio O., D’Aniello S., Merlino A., Napolitano A., Palumbo A. Shedding light on ovothiol biosynthesis in marine metazoans. Sci. Rep. 2016;6:21506. doi: 10.1038/srep21506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Lawrence K.P., Long P.F., Young A.R. Mycosporine-Like Amino Acids for Skin Photoprotection. Curr. Med. Chem. 2018;25:5512–5527. doi: 10.2174/0929867324666170529124237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Helisch H., Keppler J., Detrell G., Belz S., Ewald R., Fasoulas S., Heyer A.G. High density long-term cultivation of Chlorella vulgaris SAG 211-12 in a novel microgravity-capable membrane raceway photobioreactor for future bioregenerative life support in SPACE. Life Sci. Space Res. 2020;24:91–107. doi: 10.1016/j.lssr.2019.08.001. [DOI] [PubMed] [Google Scholar]