Abstract
Iron is a micronutrient essential for a wide range of metabolic processes in virtually all living organisms. During infections, a battle for iron takes place between the human host and the invading pathogens. The liver peptide hepcidin, which is phylogenetically and structurally linked to defensins (antimicrobial peptides of the innate immunity), plays a pivotal role by subtracting iron to pathogens through its sequestration into host cells, mainly macrophages. While this phenomenon is well studied in certain bacterial infections, much less is known regarding viral infections. Iron metabolism also has implications on the functionality of cells of the immune system. Once primed by the contact with antigen presenting cells, lymphocytes need iron to sustain the metabolic burst required for mounting an effective cellular and humoral response. The COVID-19 pandemic has boosted an amount of clinical and translational research over the possible influences of nutrients on SARS-CoV-2 infection, in terms of either susceptibility or clinical course. Here we review the intersections between iron metabolism and COVID-19, belonging to the wider domain of the so-called “nutritional immunity”. A better understanding of such connections has potential broad implications, either from a mechanistic standpoint, or for the development of more effective strategies for managing COVID-19 and possible future pandemics.
Keywords: COVID-19, Iron, Ferritin, Hepcidin, Immunity
Abbreviations: COVID-19, coronavirus disease 2019; IQR, interquartile range; MIS-C, multisystem inflammatory syndrome in children (MIS-C); MLA, machine learning approaches; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; TFr1, transferrin receptor 1; TSAT, transferrin saturation; WMD, weighted mean difference
Introduction
Iron is an essential micronutrient for virtually all living organisms [1], with very few exceptions including Borrelia burgdorferi [2] and certain lactobacilli [3]. It is needed for the synthesis of heme and iron sulfur clusters, which in turn are key functional components of many proteins and enzymes involved in vital cellular processes, including oxygen transport, energy production, and DNA synthesis. The last two decades have witnessed tremendous advance in the knowledge of the pathophysiology of iron metabolism, particularly after the discovery of hepcidin, the master regulator of systemic iron homeostasis [4]. Hepcidin, mainly produced by the liver, acts by binding and inactivating ferroportin [5], the ubiquitous and only known cell membrane iron exporter in mammalian cells [6]. Ferroportin is particularly expressed by iron absorbing enterocytes and by macrophages recycling iron from phagocytosis of senescent erythrocytes [7]. In physiological conditions, hepcidin production is strictly regulated in order to match iron absorption and recycling with daily body iron losses (1-2 mg daily) and the need of erythroid bone marrow precursors (20-25 mg/daily), respectively [8]. Of note, hepcidin also stands at the crossroad between iron metabolism and host defense mechanisms [9]. During infections, a competition for iron takes place between the host and the invading pathogen [10]. Microbes use an array of siderophores to acquire iron, while the host in turn uses different strategies to limit iron availability to microbes. The latter are part of a complex repertoire of mechanisms aimed at depriving microbes of essential trace minerals (not only iron, but also zinc and manganese), collectively termed as “nutritional immunity” [11]. As a small, disulfide-stabilized cationic peptide, hepcidin is structurally similar to defensins, a large family of antimicrobial peptides of innate immunity mainly produced by neutrophils and epithelial cells [12]. Hepcidin is rapidly and potently stimulated by pro-inflammatory cytokines, particularly interleukin 6 (IL-6) [13], leading to iron trapping into macrophages, hypoferremia, and starvation of invading pathogens. While hepcidin also retains some direct antimicrobial activity [14], this mechanism accounts for an important indirect antimicrobial activity, and represents a major determinant of the so-called anemia of inflammation [15]. Beyond this direct involvement of hepcidin in innate immunity, it is intriguing to note that the HFE protein, which is part of the complex cell membrane iron sensing machinery in hepatocytes controlling hepcidin production and is mutated in the majority of patients with hemochromatosis[16], belongs to HLA family proteins involved in antigen recognition [17]. The awareness on the multiple and important intersections between iron metabolism and immunity has been further reinforced by the recent discovery of a novel type of combined immunodeficiency with functional impairment of both T and B lymphocytes due to mutations in TFRC [18]. Indeed, TFRC encode for the main protein involved in cellular iron uptake, transferrin receptor 1 (TfR1). Of note, patients with TFRC mutations shows a surprisingly mild anemia but severe childhood infections, highlighting the requirement of intact iron transport for proper lymphocyte function and adaptative immunity [19]. Ferroportin has also been recently involved in nutritional immunity, particularly by controlling iron loading of macrophage phagosomes during experimental challenge with several infectious agents [20].
The COVID-19 pandemic has led to a major worldwide health crisis with multiple socioeconomical and medical aspects [21], boosting an unprecedented expansion of research [22]. This has especially focused on the extraordinarily complex immunological response to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [23], [24], [25], [26], [27], which is central in the pathogenesis of the widely variable clinical manifestations of COVID-19 (see below), as well as for the development of effective COVID-19 vaccines.
Due to the growing evidence of the importance of iron status for immunity, it is not surprising that biomarkers of iron metabolism have been evaluated in an array of studies on COVID-19 patients.
Here we summarize the main results, highlighting intersections between iron metabolism and COVID-19. A better understanding of such connections has potential broad implications, either from a mechanistic standpoint, or for the development of more effective strategies for managing COVID-19 and possible future pandemics.
Iron biomarkers in severe COVID-19
SARS-CoV-2 infection is a protean condition with a wide spectrum of clinical manifestations [28], ranging from absence of any symptoms [29] to a fatal disease [30]. In COVID-19 symptomatic patients the infection is mostly mild, with no or minimal pneumonia reported in 81 percent [31]. Severe disease, defined as dyspnea with respiratory rate >30 breaths per minute and hypoxia, or >50 percent lung involvement on imaging, has been reported in 14 percent. In 5 percent of patients the disease become critical, with respiratory and multi-organ failure [31], and a fatality rate at this stage as high as 49 percent [32]. The disease severity is dictated mostly by increasing age and comorbidities [33], but genetic host factors also play a role [34,35]. Severe COVID-19 is characterized by hyperinflammation due to a marked increase of proinflammatory cytokines [36], which, however, appears lower than that reported in other conditions like in response to T cell immunotherapy (the so-called true cytokine release syndrome), as well as in patients with bacterial sepsis [37]. Nonetheless, the term “viral sepsis” has been coined for severe COVID-19, also pointing out the multi-systemic involvement with typical manifestations of shock even without overt hypotension [38].
As a general note, when comparing studies on different biomarkers in COVID-19 it is important to consider the wide heterogeneity of the selected clinical cases, with apparently discordant results being often attributable to this point.
Serum ferritin
Ferritin is generally considered as a marker of iron stores, but also long known as an acute phase reactant due to either transcriptional or post-transcriptional regulation by pro-inflammatory cytokines [39]. Thus, ferritin has been extensively studied in COVID-19, with marked hyperferritinemia being a nearly constant finding in severe disease. In clinical practice, ferritin has been also frequently included in routine evaluation of COVID-19 at hospital admission [40]. Several meta-analyses have evaluated the performance of ferritin as a prognostic index in COVID-19 [41,42].
The first meta-analysis by Henry and colleagues in March 2020 evaluated 3,377 patients from 21 studies [41]. They found that serum ferritin was significantly higher in non-survivors versus survivors, as well as in patients with severe versus non-severe disease, with weighted mean differences (WMD) of 760.2 and 408.3 µg/L, respectively. The Authors recommended the inclusion of ferritin among the laboratory parameters to be closely monitored in hospitalized patients as markers for potential progression to critical illness.
Later, Cheng and colleagues evaluated 10,614 patients from 52 studies, substantially confirming the earlier findings [42]. Serum ferritin was significantly higher in patients with severe COVID-19 as compared to patients with mild disease, with a WMD of 397.77 (95% CI 306.51-489.02; P < .001). Ferritin was also significantly higher in non-survivors as compared to survivors, with a WMD of 677.17 (95% CI 391.01-963.33; P < .001).
The significant association of ferritin with disease severity and mortality was confirmed also in the comprehensive meta-analysis by Mahat and colleagues, which included a wide array of laboratory parameters [43]. Serum ferritin has been also shown as a marker of severity in a meta-analysis restricted to children with COVID-19 related multisystem inflammatory syndrome (MIS-C) [44].
Altogether, these results supported the usefulness of ferritin testing as a screening test to evaluate the presence of hyperinflammation and to predict worsening and mortality in COVID-19 hospitalized patients.
As expected, consistent reduction of ferritin has been reported in COVID-19 patients treated with anti-cytokine agents like tocilizumab, a monoclonal antibody that targets the interleukin 6 (IL-6) receptor [45,46] and represents one of the few reliable therapeutic options in critically ill COVID-19 patients [47]. In particular, Ramiro and colleagues showed that patients with ferritin levels higher that the median value of 1,419 µg/L had the better benefit from tocilizumab plus corticosteroids [46]. Despite such robust evidence, ferritin has been rarely incorporated in COVID-19 simple multiparameter prognostic scores [48], with notable exceptions represented by complex algorithms obtained through machine learning approaches (MLA) [49,50]. For example, Kar and colleagues applied MLA to 1,393 hospitalized patients, obtaining a multivariable mortality risk score that was prospectively validated in further 977 patients [50]. Of note, of the 23 parameters that entered the final score (available at http://20.44.39.47/covid19v2/page1.php), ferritin ranked in the fourth position in terms of adjusted hazard ratio (HR 2.48, 95% CI 1.32-4.74; P < .005), only preceded by lactate dehydrogenase (HR 4.02, 95% CI 2.66-6.07; P < .005) and certain comorbidities like chronic liver disease (3.95, 95% CI 1.16-13.42; P = .02), and chronic kidney disease (HR 3.04, 95% CI 1.72-5.38; P < .005). In this MLA-based study the discriminant cut-off for ferritin was >450 µg/L, which performed better than well-established clinical risk factors (ie, age and cardiovascular comorbidities) and other inflammatory parameters like C-Reactive Protein [50].
Finally, persisting hyperferritinemia has been reported in a fraction of COVID-19 up to two months after the acute infection [51]. It was closely associated with non-resolving lung pathology and impaired physical performance. Due to the tremendous impact of the so-called “long-COVID-19″ [52,53] on public health [54], the role of ferritin as long prognostic marker deserves further studies.
Possible pathophysiological meaning: Due to analogies with secondary hemophagocytic lymphohistiocytosis (sHLH) syndromes long recognized as life-threatening hyperinflammatory and possibly fatal complication of viral infections [55], severe COVID-19 has been included in the category of the “hyperferritinemic syndromes” [56], [57], [58]. Indeed, a simple “hemophagocytosis score” including very high ferritin (>1922.58 µg/L), low platelets (<101 × 109/L) and high triglycerides (>2.28 mmol/L) significantly predicted multi-organ failure and mortality in COVID-19 patients [59].
Ferritin is a ubiquitous intracellular protein which serves to store iron safely within its shell composed by 24 subunits of two main types (Heavy – H- or Light – L - chains) [60]. It is present at very low concentration in the circulation (µg/L) as compared with transferrin, the iron transporter protein in plasma (g/L). Serum ferritin derives essentially from macrophages through two distinct nonclassical vesicular pathways [61]. The physiological meaning of circulating ferritin is unknown, as well as the meaning of the marked increase of ferritin during the hyperinflammatory syndromes. Nonetheless it has been recently demonstrated that ferritin, particularly the H subunit, is able to stimulate the expression of pro-inflammatory cytokines by macrophages (IL-1β, IL-6, IL-12, and TNF-α) and also the key inflammasome player NOD-, LRR and pyrin domain-containing protein 3 (NLRP3) [62,63]. Thus, secreted ferritin could act as a further pro-inflammatory enhancer, by perpetuating a vicious pathogenic loop. Further studies are required to confirm this interesting hypothesis (reviewed in [58]), which could theoretically pave the way to novel therapeutic approaches.
Serum iron and transferrin saturation
While less frequently studied than ferritin, marked hypoferremia and low transferrin saturation (TSAT) have been consistently reported in severe COVID-19, and associated with a worse outcome [64], [65], [66], [67].
In multivariate regression analysis involving a total of 308 COVID-19 patients, Hippchen and colleagues found that hypoferremia (serum iron concentration <6 μmol/l) was a significant predictor of hospitalization (area under the curve - AUC - of 0.894, with a sensitivity of 94.7% and a specificity of 67.9%) [65]. Hospitalized patients had very low TSAT (median 7% with interquartile range – IQR – 5%-11%) as compared to patients with mild disease who showed only a marginal reduction (median 19% with IQR 12-28%; P < .01). Surprisingly, hypoferremia poorly correlated with serum hepcidin, which nonetheless was increased as expected.
Similar results were reported by Shah and colleagues, who found marked hypoferremia (median 2.3, IQR 2.2-2.5 μmol/L) and reduced TSAT (7%, IQR 6%-12%) in critically ill COVID-19 patients requiring admission in intensive care unit (ICU) [67].
Possible pathophysiological meaning: Interestingly, both the studies mentioned above found a relationship between hypoferremia and hypoxia in COVID-19 patients. In the study by Hippchen and colleagues, serum iron levels were lower at hospital admission in patients with high oxygen demand as compared to those with low oxygen demand. Serial measurements during hospitalization further showed an association between low serum iron and worsening clinical course [65]. Using linear regression, Shah and colleagues found an association between serum iron and the ratio of partial pressure arterial oxygen to fractional inspired oxygen [67], that is the PaO2/FiO2 ratio universally recognized as a major prognostic factor in COVID-19 [32]. Of note, increasing evidence support multiple interactions between iron metabolism and lung respiratory function [68,69], especially in hypoxemic disorders [70]. Just as an example, it has been elegantly documented that the cellular response to hypoxia is impaired in subjects with absolute iron deficiency [71]. This could be due, at least partly, to the strict iron dependency of the prolyl-hydroxylase domain (PHD) enzymes, known as the key regulators of Hypoxia Inducible Factors (HIFs) [72]. Thus, the marked functional iron deficiency seen in hyperinflammatory syndromes could also impair cellular oxygen sensing, and further contribute to a detrimental vicious circle in severe hypoxia of critically ill COVID-19 patients. Further studies are needed in this direction, as functional iron deficiency is theoretically manageable by using novel anti-hepcidin agents [73,74].
Another possible detrimental effect of marked hypoferremia is linked to altered lymphocyte function. Indeed, upon antigen activation either B or T lymphocytes undergo to proliferation and requires iron to sustain a high metabolic demand [75]. As mentioned above, iron acquisition via TfR1 is required for B and T cell responses [18], and recent experimental models by Frost and colleagues have shown that hypoferremia impairs effector and memory responses to vaccinations [76]. Noteworthy, Shah and colleagues found a significant association between low serum iron and low lymphocyte count in critically ill COVID-19 patients [67]. Thus, the marked hypoferremia that seems to characterize severe COVID-19 as compared to other critical conditions [67,77] may contribute to worsening prognosis by impairing not only response to hypoxia but also immune function.
Finally, it has to be taken into account that low iron status could theoretically impair the efficacy of COVID-19 vaccination, which remains the cornerstone to eventually control the pandemic [78,79]. Beyond the experimental model by Frost and colleagues, where hepcidin-induced hypoferremia blunted neutralizing antibody response to influenza virus vaccination [76], iron deficiency has been found to impair response to diphtheria, pertussis, and pneumococcal vaccines in Kenyan infants [80]. However, at present there is insufficient evidence to make recommendations on correcting iron deficiency before vaccinations. Readers interested on this point are referred to a recent perspective [81].
Serum hepcidin
Serum hepcidin measurement represents a promising tool for evaluating iron status, particularly in certain conditions when absolute and functional iron deficiency may coexist [82,83], but it has not yet widely implemented in clinical practice [7]. Recent studies have documented its potential usefulness in critically ill patients, where detecting iron deficiency is particularly difficult due to inflammation and multiple opposing stimuli [84]. Moreover, hepcidin has been proven as a useful independent prognostic marker of either poor physical recovery or mortality in ICU patients [85].
Hepcidin has been rarely measured in COVID-19 patients [65,86,87]. As expected in severe illness with hyperinflammation, it was reported consistently high. Nai and colleagues evaluated 111 hospitalized patients, the majority of whom showed an inflammation-dependent upregulation of hepcidin levels of variable degree [87]. Of note, hepcidin correlated negatively with the PaO2/FiO2 ratio, and predicted mortality in ICU patients independently of age, lung function, and other inflammatory biomarkers.
Such intriguing finding needs confirmation in wider cohorts, as well as further studies should better clarify the relationship between hypoferremia and hepcidin in COVID-19. As pointed out by Hippchen and colleagues, still unidentified hepcidin-independent mechanisms could play an important role [65]. Finally, COVID-19 could represent a useful model to study hepcidin upregulation by different pro-inflammatory cytokines in humans, as most of data in this sense are derived from cellular and animal models [13,88].
Conclusions
The COVID-19 pandemic has boosted research on multiple aspects of immunology, including nutritional immunity. Iron is emerging as an important player to ensure an adequate immune response, and studies on iron metabolism in COVID-19 have highlighted a number of interesting intersections. While several studies have established the functional implications of the battle for iron between bacteria, certain parasites, and the host, the scenario is less clear in viral infections [89]. Indeed, distinct patterns of hepcidin and iron regulation have been reported to occur during different viral infections [90].
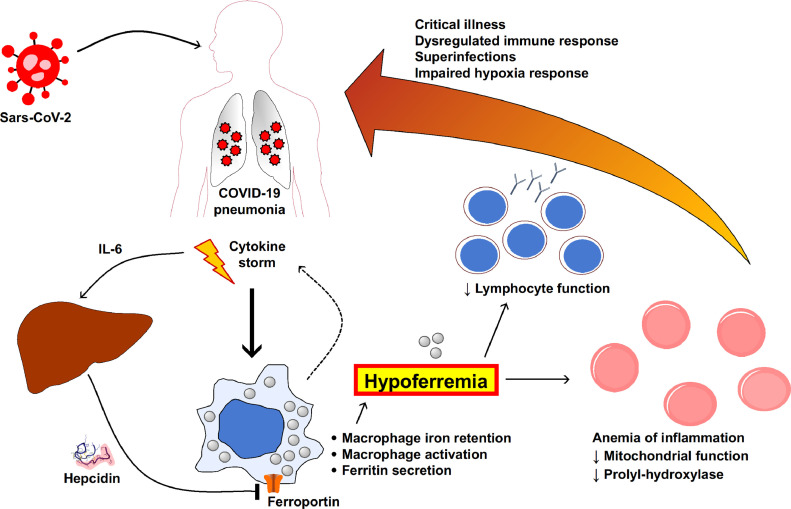
Severe COVID-19 appears to be characterized by high hepcidin and marked functional iron deficiency, the latter being possibly related to impaired response to hypoxia and lymphocyte function. The multiple possible functional intersection between SARS-CoV-2 infection and iron metabolism discussed above are summarized in Fig. 1 . Assuming COVID-19 as a valuable model, further studies are needed to better clarify the role of altered iron metabolism in severe viral infections and hyperinflammatory syndromes, possibly paving the way to new therapeutic approaches.
Fig. 1.
Intersections between iron metabolism and severe COVID-19, according to suggestions from studies described and referenced in the text. Marked hypoferremia has been consistently reported in severe COVID-19, and might play a substantial role as contributing factor to a worsening clinical course. Hypoferremia is associated to the marked increase of pro-inflammatory cytokines, including IL-6 which is a major stimulator of hepcidin production by the liver. Hepcidin contribute to hypoferremia by inhibiting iron export from macrophages due to ferroportin internalization and degradation. Other still unknown mechanisms could contribute to hepcidin-independent hypoferremia. Activation of macrophages can mimic hemophagocytic syndromes with marked hyperferritinemia and further contribute to the cytokine storm. The functional iron deficiency in turn is a major determinant of the anemia of inflammation, and can alter either oxygen sensing and lymphocyte function. Altogether these mechanisms could contribute to disease severity.
Disclosure
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Footnotes
Supported by the grant “ENACT” from the Cariverona Foundation.
References
- 1.Sheftel AD, Mason AB, Ponka P. The long history of iron in the Universe and in health and disease. Biochim Biophys Acta. 2012;1820(3):161–187. doi: 10.1016/j.bbagen.2011.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Posey JE, Gherardini FC. Lack of a role for iron in the Lyme disease pathogen. Science. 2000;288(5471):1651–1653. doi: 10.1126/science.288.5471.1651. [DOI] [PubMed] [Google Scholar]
- 3.Ganz T. Iron and infection. Int J Hematol. 2018;107(1):7–15. doi: 10.1007/s12185-017-2366-2. [DOI] [PubMed] [Google Scholar]
- 4.Camaschella C, Nai A, Silvestri L. Iron metabolism and iron disorders revisited in the hepcidin era. Haematologica. 2020;105(2):260–272. doi: 10.3324/haematol.2019.232124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aschemeyer S, Qiao B, Stefanova D, et al. Structure-function analysis of ferroportin defines the binding site and an alternative mechanism of action of hepcidin. Blood. 2018;131(8):899–910. doi: 10.1182/blood-2017-05-786590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pan Y, Ren Z, Gao S, et al. Structural basis of ion transport and inhibition in ferroportin. Nat Commun. 2020;11(1):5686. doi: 10.1038/s41467-020-19458-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Girelli D, Nemeth E, Swinkels DW. Hepcidin in the diagnosis of iron disorders. Blood. 2016;127(23):2809–2813. doi: 10.1182/blood-2015-12-639112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Girelli D, Ugolini S, Busti F, Marchi G, Castagna A. Modern iron replacement therapy: clinical and pathophysiological insights. Int J Hematol. 2018;107(1):16–30. doi: 10.1007/s12185-017-2373-3. [DOI] [PubMed] [Google Scholar]
- 9.Ganz T, Nemeth E. Iron homeostasis in host defence and inflammation. Nat Rev Immunol. 2015;15(8):500–510. doi: 10.1038/nri3863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Drakesmith H, Prentice AM. Hepcidin and the iron-infection axis. Science. 2012;338(6108):768–772. doi: 10.1126/science.1224577. [DOI] [PubMed] [Google Scholar]
- 11.Hood MI, Skaar EP. Nutritional immunity: transition metals at the pathogen-host interface. Nat Rev Microbiol. 2012;10(8):525–537. doi: 10.1038/nrmicro2836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ganz T. Defensins: antimicrobial peptides of innate immunity. Nat Rev Immunol. 2003;3(9):710–720. doi: 10.1038/nri1180. [DOI] [PubMed] [Google Scholar]
- 13.Nemeth E, Rivera S, Gabayan V, et al. IL-6 mediates hypoferremia of inflammation by inducing the synthesis of the iron regulatory hormone hepcidin. J Clin Investig. 2004;113(9):1271–1276. doi: 10.1172/JCI20945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maisetta G, Petruzzelli R, Brancatisano FL, et al. Antimicrobial activity of human hepcidin 20 and 25 against clinically relevant bacterial strains: effect of copper and acidic pH. Peptides. 2010;31(11):1995–2002. doi: 10.1016/j.peptides.2010.08.007. [DOI] [PubMed] [Google Scholar]
- 15.Ganz T. Anemia of inflammation. N Engl J Med. 2019;381(12):1148–1157. doi: 10.1056/NEJMra1804281. [DOI] [PubMed] [Google Scholar]
- 16.Piperno A, Sampietro M, Pietrangelo A, et al. Heterogeneity of hemochromatosis in Italy. Gastroenterology. 1998;114(5):996–1002. doi: 10.1016/s0016-5085(98)70319-1. [DOI] [PubMed] [Google Scholar]
- 17.Carella M, D'Ambrosio L, Totaro A, et al. Mutation analysis of the HLA-H gene in Italian hemochromatosis patients. Am J Hum Genet. 1997;60(4):828–832. [PMC free article] [PubMed] [Google Scholar]
- 18.Jabara HH, Boyden SE, Chou J, et al. A missense mutation in TFRC, encoding transferrin receptor 1, causes combined immunodeficiency. Nat Genet. 2016;48(1):74–78. doi: 10.1038/ng.3465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lo B. The requirement of iron transport for lymphocyte function. Nat Genet. 2016;48(1):10–11. doi: 10.1038/ng.3478. [DOI] [PubMed] [Google Scholar]
- 20.Flannagan RS, Farrell TJ, Trothen SM, Dikeakos JD, Heinrichs DE. Rapid removal of phagosomal ferroportin in macrophages contributes to nutritional immunity. Blood Adv. 2021;5(2):459–474. doi: 10.1182/bloodadvances.2020002833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Blumenthal D, Fowler EJ, Abrams M, Collins SR. Covid-19 - implications for the health care system. N Engl J Med. 2020;383(15):1483–1488. doi: 10.1056/NEJMsb2021088. [DOI] [PubMed] [Google Scholar]
- 22.Else H. How a torrent of COVID science changed research publishing - in seven charts. Nature. 2020;588(7839):553. doi: 10.1038/d41586-020-03564-y. [DOI] [PubMed] [Google Scholar]
- 23.Wilk AJ, Rustagi A, Zhao NQ, et al. A single-cell atlas of the peripheral immune response in patients with severe COVID-19. Nat Med. 2020;26(7):1070–1076. doi: 10.1038/s41591-020-0944-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Giamarellos-Bourboulis EJ, Netea MG, Rovina N, et al. Complex immune dysregulation in COVID-19 patients with severe respiratory failure. Cell Host Microbe. 2020;27(6):992–1000. doi: 10.1016/j.chom.2020.04.009. e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schultze JL, Aschenbrenner AC. COVID-19 and the human innate immune system. Cell. 2021;184(7):1671–1692. doi: 10.1016/j.cell.2021.02.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol. 2020;20(5):269–270. doi: 10.1038/s41577-020-0308-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Brodin P. Immune determinants of COVID-19 disease presentation and severity. Nat Med. 2021;27(1):28–33. doi: 10.1038/s41591-020-01202-8. [DOI] [PubMed] [Google Scholar]
- 28.Gupta A, Madhavan MV, Sehgal K, et al. Extrapulmonary manifestations of COVID-19. Nat Med. 2020;26(7):1017–1032. doi: 10.1038/s41591-020-0968-3. [DOI] [PubMed] [Google Scholar]
- 29.Oran DP, Topol EJ. Prevalence of asymptomatic SARS-CoV-2 infection: a narrative review. Ann Intern Med. 2020;173(5):362–367. doi: 10.7326/M20-3012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Hu B, Guo H, Zhou P, Shi ZL. Characteristics of SARS-CoV-2 and COVID-19. Nat Rev Microbiol. 2021;19(3):141–154. doi: 10.1038/s41579-020-00459-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in china: summary of a report of 72314 cases from the chinese center for disease control and prevention. JAMA. 2020;323(13):1239–1242. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 32.Berlin DA, Gulick RM, Martinez FJ. Severe Covid-19. N Engl J Med. 2020;383(25):2451–2460. doi: 10.1056/NEJMcp2009575. [DOI] [PubMed] [Google Scholar]
- 33.Williamson EJ, Walker AJ, Bhaskaran K, et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature. 2020;584(7821):430–436. doi: 10.1038/s41586-020-2521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Severe Covid GG, Ellinghaus D, Degenhardt F, et al. Genomewide association study of severe Covid-19 with respiratory failure. N Engl J Med. 2020;383(16):1522–1534. doi: 10.1056/NEJMoa2020283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pairo-Castineira E, Clohisey S, Klaric L, et al. Genetic mechanisms of critical illness in COVID-19. Nature. 2021;591(7848):92–98. doi: 10.1038/s41586-020-03065-y. [DOI] [PubMed] [Google Scholar]
- 36.Huang C, Wang Y, Li X, Ren L, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Leisman DE, Ronner L, Pinotti R, et al. Cytokine elevation in severe and critical COVID-19: a rapid systematic review, meta-analysis, and comparison with other inflammatory syndromes. Lancet Respir Med. 2020;8(12):1233–1244. doi: 10.1016/S2213-2600(20)30404-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Li H, Liu L, Zhang D, et al. SARS-CoV-2 and viral sepsis: observations and hypotheses. Lancet. 2020;395(10235):1517–1520. doi: 10.1016/S0140-6736(20)30920-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Torti FM, Torti SV. Regulation of ferritin genes and protein. Blood. 2002;99(10):3505–3516. doi: 10.1182/blood.v99.10.3505. [DOI] [PubMed] [Google Scholar]
- 40.Chen Z, Xu W, Ma W, et al. Clinical laboratory evaluation of COVID-19. Clin Chim Acta. 2021;519:172–182. doi: 10.1016/j.cca.2021.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Henry BM, de Oliveira MHS, Benoit S, Plebani M, Lippi G. Hematologic, biochemical and immune biomarker abnormalities associated with severe illness and mortality in coronavirus disease 2019 (COVID-19): a meta-analysis. Clin Chem Lab Med. 2020;58(7):1021–1028. doi: 10.1515/cclm-2020-0369. [DOI] [PubMed] [Google Scholar]
- 42.Cheng L, Li H, Li L, Liu C, et al. Ferritin in the coronavirus disease 2019 (COVID-19): A systematic review and meta-analysis. J Clin Lab Anal. 2020;34(10):e23618. doi: 10.1002/jcla.23618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mahat RK, Panda S, Rathore V, Swain S, Yadav L, Sah SP. The dynamics of inflammatory markers in coronavirus disease-2019 (COVID-19) patients: A systematic review and meta-analysis. Clin Epidemiol Glob Health. 2021;11 doi: 10.1016/j.cegh.2021.100727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhao Y, Yin L, Patel J, Tang L, Huang Y. The inflammatory markers of multisystem inflammatory syndrome in children (MIS-C) and adolescents associated with COVID-19: A meta-analysis. J Med Virol. 2021;93(7):4358–4369. doi: 10.1002/jmv.26951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Toniati P, Piva S, Cattalini M, et al. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: A single center study of 100 patients in Brescia, Italy. Autoimmun Rev. 2020;19(7) doi: 10.1016/j.autrev.2020.102568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ramiro S, Mostard RLM, Magro-Checa C, et al. Historically controlled comparison of glucocorticoids with or without tocilizumab versus supportive care only in patients with COVID-19-associated cytokine storm syndrome: results of the CHIC study. Ann Rheum Dis. 2020;79(9):1143–1151. doi: 10.1136/annrheumdis-2020-218479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Investigators R-C, Gordon AC, Mouncey PR, et al. Interleukin-6 receptor antagonists in critically Ill patients with Covid-19. N Engl J Med. 2021;384(16):1491–1502. doi: 10.1056/NEJMoa2100433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bats ML, Rucheton B, Fleur T, et al. Covichem: A biochemical severity risk score of COVID-19 upon hospital admission. PloS one. 2021;16(5) doi: 10.1371/journal.pone.0250956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Guan X, Zhang B, Fu M, et al. Clinical and inflammatory features based machine learning model for fatal risk prediction of hospitalized COVID-19 patients: results from a retrospective cohort study. Ann Med. 2021;53(1):257–266. doi: 10.1080/07853890.2020.1868564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kar S, Chawla R, Haranath SP, et al. Multivariable mortality risk prediction using machine learning for COVID-19 patients at admission (AICOVID) Sci Rep. 2021;11(1):12801. doi: 10.1038/s41598-021-92146-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Sonnweber T, Boehm A, Sahanic S, et al. Persisting alterations of iron homeostasis in COVID-19 are associated with non-resolving lung pathologies and poor patients' performance: a prospective observational cohort study. Respir Res. 2020;21(1):276. doi: 10.1186/s12931-020-01546-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Nalbandian A, Sehgal K, Gupta A, et al. Post-acute COVID-19 syndrome. Nat Med. 2021;27(4):601–615. doi: 10.1038/s41591-021-01283-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Del Rio C, Collins LF, Malani P. Long-term Health Consequences of COVID-19. JAMA. 2020;324(17):1723–1724. doi: 10.1001/jama.2020.19719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Al-Aly Z, Xie Y, Bowe B. High-dimensional characterization of post-acute sequelae of COVID-19. Nature. 2021;594(7862):259–264. doi: 10.1038/s41586-021-03553-9. [DOI] [PubMed] [Google Scholar]
- 55.Al-Samkari H, Berliner N. Hemophagocytic Lymphohistiocytosis. Annu Rev Pathol. 2018;13:27–49. doi: 10.1146/annurev-pathol-020117-043625. [DOI] [PubMed] [Google Scholar]
- 56.Rosario C, Zandman-Goddard G, Meyron-Holtz EG, D'Cruz DP, Shoenfeld Y. The hyperferritinemic syndrome: macrophage activation syndrome, Still's disease, septic shock and catastrophic antiphospholipid syndrome. BMC Med. 2013;11:185. doi: 10.1186/1741-7015-11-185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ruscitti P, Giacomelli R. Ferritin and Severe COVID-19, from Clinical Observations to Pathogenic Implications and Therapeutic Perspectives. Isr Med Assoc J. 2020;22(8):516–518. [PubMed] [Google Scholar]
- 58.Ruscitti P, Berardicurti O, Di Benedetto P, et al. Severe COVID-19, Another Piece in the Puzzle of the Hyperferritinemic Syndrome. An Immunomodulatory Perspective to Alleviate the Storm. Front Immunol. 2020;11:1130. doi: 10.3389/fimmu.2020.01130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Meng M, Chen L, Zhang S, et al. Risk factors for secondary hemophagocytic lymphohistiocytosis in severe coronavirus disease 2019 adult patients. BMC Infect Dis. 2021;21(1):398. doi: 10.1186/s12879-021-06094-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Harrison PM, Arosio P. The ferritins: molecular properties, iron storage function and cellular regulation. Biochim Biophys Acta. 1996;1275(3):161–203. doi: 10.1016/0005-2728(96)00022-9. [DOI] [PubMed] [Google Scholar]
- 61.Truman-Rosentsvit M, Berenbaum D, Spektor L, et al. Ferritin is secreted via 2 distinct nonclassical vesicular pathways. Blood. 2018;131(3):342–352. doi: 10.1182/blood-2017-02-768580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ruscitti P, Di Benedetto P, Berardicurti O, et al. Pro-inflammatory properties of H-ferritin on human macrophages, ex vivo and in vitro observations. Sci Rep. 2020;10(1):12232. doi: 10.1038/s41598-020-69031-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Broz P, Dixit VM. Inflammasomes: mechanism of assembly, regulation and signalling. Nat Rev Immunol. 2016;16(7):407–420. doi: 10.1038/nri.2016.58. [DOI] [PubMed] [Google Scholar]
- 64.Bolondi G, Russo E, Gamberini E, et al. Iron metabolism and lymphocyte characterisation during Covid-19 infection in ICU patients: an observational cohort study. World J Emerg Surg. 2020;15(1):41. doi: 10.1186/s13017-020-00323-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hippchen T, Altamura S, Muckenthaler MU, Merle U. Hypoferremia is associated with increased hospitalization and oxygen demand in COVID-19 patients. Hemasphere. 2020;4(6):e492. doi: 10.1097/HS9.0000000000000492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhao K, Huang J, Dai D, Feng Y, Liu L, Nie S. Serum iron level as a potential predictor of coronavirus disease 2019 severity and mortality: a retrospective study. Open Forum Infect Dis. 2020;7(7):ofaa250. doi: 10.1093/ofid/ofaa250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Shah A, Frost JN, Aaron L, Donovan K, Drakesmith H, Collaborators Systemic hypoferremia and severity of hypoxemic respiratory failure in COVID-19. Crit Care. 2020;24(1):320. doi: 10.1186/s13054-020-03051-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Neves J, Haider T, Gassmann M, Muckenthaler MU. Iron homeostasis in the lungs-A balance between health and disease. Pharmaceuticals (Basel) 2019;12(1):5. doi: 10.3390/ph12010005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhang V, Nemeth E, Kim A. Iron in Lung Pathology. Pharmaceuticals (Basel) 2019;12(1):30. doi: 10.3390/ph12010030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Cloonan SM, Mumby S, Adcock IM, Choi AMK, Chung KF, Quinlan GJ. The "Iron"-y of Iron Overload and Iron Deficiency in Chronic Obstructive Pulmonary Disease. Am J Respir Crit Care Med. 2017;196(9):1103–1112. doi: 10.1164/rccm.201702-0311PP. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Frise MC, Cheng HY, Nickol AH, et al. Clinical iron deficiency disturbs normal human responses to hypoxia. J Clin Investig. 2016;126(6):2139–2150. doi: 10.1172/JCI85715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Busti F, Marchi G, Vianello A, Girelli D. Practical implications of the 2019 Nobel Prize in Physiology or Medicine: from molecular adaptation to hypoxia to novel anti-anemic drugs in the clinic. Intern Emerg Med. 2020;15(6):911–915. doi: 10.1007/s11739-020-02417-w. [DOI] [PubMed] [Google Scholar]
- 73.Hawula ZJ, Wallace DF, Subramaniam VN, Rishi G. Therapeutic advances in regulating the hepcidin/ferroportin axis. Pharmaceuticals (Basel) 2019;12(4):170. doi: 10.3390/ph12040170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Ruchala P, Nemeth E. The pathophysiology and pharmacology of hepcidin. Trends Pharmacol Sci. 2014;35(3):155–161. doi: 10.1016/j.tips.2014.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Jiang Y, Li C, Wu Q, An P, et al. Iron-dependent histone 3 lysine 9 demethylation controls B cell proliferation and humoral immune responses. Nat Commun. 2019;10(1):2935. doi: 10.1038/s41467-019-11002-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Frost JN, Tan TK, Abbas M, et al. Hepcidin-mediated hypoferremia disrupts immune responses to vaccination and infection. Med (N Y) 2021;2(2):164–179. doi: 10.1016/j.medj.2020.10.004. e12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Tacke F, Nuraldeen R, Koch A, et al. Iron parameters determine the prognosis of critically Ill patients. Crit Care Med. 2016;44(6):1049–1058. doi: 10.1097/CCM.0000000000001607. [DOI] [PubMed] [Google Scholar]
- 78.Haas EJ, Angulo FJ, McLaughlin JM, et al. Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: an observational study using national surveillance data. Lancet. 2021;397(10287):1819–1829. doi: 10.1016/S0140-6736(21)00947-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Leshem E, Wilder-Smith A. COVID-19 vaccine impact in Israel and a way out of the pandemic. Lancet. 2021;397(10287):1783–1785. doi: 10.1016/S0140-6736(21)01018-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Stoffel NU, Uyoga MA, Mutuku FM, et al. Iron Deficiency anemia at time of vaccination predicts decreased vaccine response and iron supplementation at time of vaccination increases humoral vaccine response: a birth cohort study and a randomized trial follow-up study in kenyan infants. Front Immunol. 2020;11:1313. doi: 10.3389/fimmu.2020.01313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.H Drakesmith, et al. Viewpoint: Vaccine efficacy and iron deficiency: an intertwined pair? Lancet Haematol. 2021 doi: 10.1016/S2352-3026(21)00201-5. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Stoffel NU, Lazrak M, Bellitir S, et al. The opposing effects of acute inflammation and iron deficiency anemia on serum hepcidin and iron absorption in young women. Haematologica. 2019;104(6):1143–1149. doi: 10.3324/haematol.2018.208645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Bergamaschi G, Di Sabatino A, Albertini R, et al. Serum hepcidin in inflammatory bowel diseases: biological and clinical significance. Inflamm Bowel Dis. 2013;19(10):2166–2172. doi: 10.1097/MIB.0b013e31829a6e43. [DOI] [PubMed] [Google Scholar]
- 84.Lasocki S, Baron G, Driss F, et al. Diagnostic accuracy of serum hepcidin for iron deficiency in critically ill patients with anemia. Intensive Care Med. 2010;36(6):1044–1048. doi: 10.1007/s00134-010-1794-8. [DOI] [PubMed] [Google Scholar]
- 85.Lasocki S, Lefebvre T, Mayeur C, et al. Iron deficiency diagnosed using hepcidin on critical care discharge is an independent risk factor for death and poor quality of life at one year: an observational prospective study on 1161 patients. Crit Care. 2018;22(1):314. doi: 10.1186/s13054-018-2253-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Zhou C, Chen Y, Ji Y, He X, Xue D. Increased serum levels of hepcidin and ferritin are associated with severity of COVID-19. Med Sci Monit. 2020;26 doi: 10.12659/MSM.926178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Nai A, Lore NI, Pagani A, et al. Hepcidin levels predict Covid-19 severity and mortality in a cohort of hospitalized Italian patients. Am J Hematol. 2021;96(1):E32–E35. doi: 10.1002/ajh.26027. [DOI] [PubMed] [Google Scholar]
- 88.Roth MP, Meynard D, Coppin H. Regulators of hepcidin expression. Vitam Horm. 2019;110:101–129. doi: 10.1016/bs.vh.2019.01.005. [DOI] [PubMed] [Google Scholar]
- 89.Drakesmith H, Prentice A. Viral infection and iron metabolism. Nat Rev Microbiol. 2008;6(7):541–552. doi: 10.1038/nrmicro1930. [DOI] [PubMed] [Google Scholar]
- 90.Armitage AE, Stacey AR, Giannoulatou E, et al. Distinct patterns of hepcidin and iron regulation during HIV-1, HBV, and HCV infections. Proc Natl Acad Sci U S A. 2014;111(33):12187–12192. doi: 10.1073/pnas.1402351111. [DOI] [PMC free article] [PubMed] [Google Scholar]