Central Message.
Various techniques are available for aortic cusp and valve repair. An individualized approach should be taken owing to the diverse valvular morphology and pathophysiology.
Common aortic repair techniques for bicuspid and tricuspid aortic valves.
See Commentaries on pages 117 and 119.
Aortic regurgitation (AR) is one of the most prevalent cardiac valvular diseases, affecting up to 13% of the population.1 The pathophysiology of AR is dependent on the acuity of the disease and the dysfunction of the aortic valvular apparatus.2 Functionally, AR can be classified into type I, with normal cusp motion but dilated functional aortic annulus (FAA) or cusp perforation; type II, with cusp prolapse due to excess cusp tissue or commissural disruption3; or type III, with cusp restriction typically seen in bicuspid aortic valve (BAV), degenerative, or rheumatic valvular disease.2 This functional AR classification allows a systematic approach to the surgical treatment options for AR. On the transcriptional level, AR valves are associated with unique gene expression patterns compared with normal aortic valves.4 These findings may help guide the development of novel gene editing therapies for patients with AR.
The specific indications for aortic valve repair have not been standardized. Rather, the current guidelines from the American College of Cardiology and the American Heart Association focus on overall aortic valve intervention.5 Although aortic valve replacement has been a mainstay of therapy for decades, choosing between a mechanical or bioprosthetic replacement valve involves committing patients either to lifelong anticoagulation and its associated risk of thromboembolic or hemorrhagic sequelae, or to future reoperation for structural valve deterioration and its associated perioperative risks.6 Nonetheless, aortic valve repair has been shown to be a safe and highly effective strategy that avoids the disadvantages associated with valve replacement.7, 8, 9
Surgical Anatomy
Understanding the dynamics and anatomic structure of the FAA is critical to the success of AR elimination and ensuring long-term durability of the repair. Any geometric alternation in the FAA, destruction of the aortic cusps, or dysfunction in leaflet motion and coaptation can result in AR.2 Importantly, the subcommissural triangles that directly underlie the 3 commissures consist of a thin section of aortic wall and important anatomic structures, such as the conduction system underneath the right/non commissure and the aortomitral curtain below the left/non commissure. The sinuses of Valsalva play host to the coronary artery ostia, but the precise function is unclear. Although evidence suggests their role in maintaining proper flow vortices and leaflet hemodynamics, the absence of neosinuses is not associated with worsened cusp biomechanics or clinical outcomes in valve-sparing root replacement (VSRR) repairs.10,11
BAV is one of the most common congenital defects,12 and its morphology varies depending on the cusp fusion pattern and the presence of raphe.13 The most commonly used BAV classification is the Sievers classification, in which the morphology types are determined by the number of raphe. The type I BAV with left–right coronary cusp fusion is the most prevalent morphology.13 More recently, de Kerchove and colleagues14 demonstrated that BAV morphology follows a continuous spectrum with wide variations in commissural orientation, fusion length, and nonfunctional commissure height, extending from type A or Sievers type 0 BAV to type C or Sievers type I BAV. Of note, as the fusion length and the cusp geometric height decrease from type A to type C, the nonfunctional commissure height increases, posing important considerations when determining valve repair techniques.14
Surgical Approach
The standard approach for aortic valve repair is via a median sternotomy. A mini-sternotomy and right anterior thoracotomy also have been adopted in experienced centers for aortic valve interventions15; however, the durability of repair with these approaches is unclear. Arterial cannulation is typically performed in the ascending aorta, distal to any diseased segments, and 2-stage venous cannulation can be obtained through the right atrial appendage. Alternatively, peripheral cannulation can be achieved, especially when using a minimally invasive approach. Following administration of retrograde cardioplegia, an aortotomy is made approximately 1 cm above the sinotubular junction (STJ) superior to the noncoronary sinus and extended circumferentially.
Technical Considerations
Successful valve preservation demands thorough investigation of the aortic valvular apparatus using echocardiography and direct visual inspection. Calcification typically precludes a durable repair, and valve replacement should be considered. In contrast, valves with mild cusp thickening can be repaired if the cusps are mobile and pliable. Depending on the location and leaflet geometry, cusp fenestration can be repaired with leading edge reinforcement or pericardial reconstruction. After determining the adequacy of the cusp for repair, measurements of the cusp depth and length, commissural height, and diameters of the annulus, sinuses of Valsalva, and STJ should be made. Close attention should be paid to the root geometry, as asymmetric sinuses and commissure positions are commonly observed, especially in patients with BAV. The native geometry should be carefully considered when implementing cusp repair and valve implantation techniques to provide an individualized strategy for each patient.16 After repair completion, valve competency should be tested by administering antegrade cold blood into the reconstructed aortic root. An endoscope can also be used to directly evaluate valve geometry under physiologic pressure before aortotomy closure, providing an opportunity to refine cusp repair until satisfying results are achieved.16 Intraoperative echocardiography should always be obtained to assess valve anatomy and any residual valve dysfunction after repair. If necessary, further aortic repair should be reattempted after cardiac arrest. Aortic valve repair techniques generally can be divided into 3 categories: aortic cusp repair, annular and STJ stabilization, and aortic valve reimplantation. A flowchart outlining the key indication criteria and technical steps for each repair technique is shown in Figure 1.
Figure 1.
A flowchart outlining the key indication criteria and technical steps for each repair technique.
Aortic Cusp Repair
Free Margin Plication
The free margin plication technique, which aims to equalize the cusp free margin lengths to recreate an adequate leaflet coaptation area, is most useful in the presence of only a small discrepancy in leaflet lengths (Figure 2, A). A 7-0 polypropylene suture is passed through the noduli of the nonprolapsing cusps, and gentle axial traction is applied to create a reference length. Next, the prolapsing cusp is pulled parallel to the reference cusp to estimate the length of free margin to be plicated. A CV-6 polytetrafluoroethylene (PTFE) suture enters from the aortic side and reenters from the ventricular side, creating a small fold in the prolapsed cusp. The suture is then tied on the aortic side. The plication can be further extended toward the leaflet attachment sites if indicated. This repair technique also can be performed close to the commissure to generate paracentral plication (Figure 2, B). The central plication should be used only when the noduli are not prominent or thickened.
Figure 2.
A, The central free margin plication technique is used to recreate an adequate leaflet coaptation area by plicating the prolapsed cusp toward the annulus. B, A paracentral plication technique can also be performed by plicating the cusp closer to the commissure. C, Commissuroplasty can be performed asymmetrically by pulling the plication toward one of the commissures to realign the free margin of the prolapsed cusp.
Commissuroplasty
The commissuroplasty repair technique effectively reduces the aortic root dimension at the commissure level and shortens the free margin. The commissures are plicated with pledgeted, horizontal mattress stitches. Central plication can be performed concomitantly. Alternatively, asymmetric commissuroplasty can be performed by pulling the plication toward one of the commissures, realigning the free margin of the prolapsed cusp (Figure 2, C). The subcommissuroplasty repair technique reduces the width of the interleaflet triangle, improves cusp coaptation, and helps stabilize the ventriculoaortic junction. Compared with commissuroplasty, subcommissuroplasty is typically completed at the mid-commissure level, except at the non-right commissure, where the membranous septum should be avoided. Care should be taken during knot tying to avoid tearing the septum. Both repair techniques are relatively easy to perform and add very little time to the operation, but these techniques abolish the role of the interleaflet triangle and may affect leaflet hemodynamics.
Free Margin Resuspension
The free margin resuspension technique removes cusp redundancy and returns the coaptation surfaces to the same level to reestablish valve competency (Figure 3). This technique is particularly useful when the prolapsed cusp has a fragile free margin or fenestration. A pericardial patch used for cusp augmentation also can be repositioned using this technique. The nonprolapsed cusps are aligned using the same technique described above. Next, a double-armed CV-6 PTFE suture is woven over the free margin of the prolapsed cusp. The free margin length can be adjusted by applying gentle traction on each arm of the suture until the same free margin length is achieved compared with that of the reference cusp. The sutures can then be tied at the commissures. This repair technique has been evaluated biomechanically in an ex vivo BAV model and showed its efficacy in eliminating AR without impairing valve hemodynamics.17
Figure 3.
The free margin resuspension technique can reestablish valvular competency by weaving a suture along the cusp-free margin.
Decalcification and Triangular Resection
For aortic valves with mild calcifications and especially for type I BAVs, the calcification and BAV raphe should be assessed and shaved if it is relatively mobile. While the use of devices such as the Cavitron Ultrasonic Surgical Aspirator is a potential approach,18,19 we prefer to use sharp dissection. It is also not uncommon to see a calcified bicuspid or tricuspid aortic valve with leaflet restriction. Calcified leaflet fusion restricts mobility of the entire fused cusp. In this situation, a triangular resection should be performed (Figure 4). This can be achieved by excising the central portion and then suturing the edges back together. A continuous suture is the preferred option over interrupted sutures, because it decreases the likelihood of thrombosis and residual AR.20
Figure 4.
Leaflet decalcification and resection followed by pericardial patch reconstruction repair for a bicuspid aortic valve. Specifically, an oval-shaped pericardial patch of an appropriate size is sutured in with a running suture to repair the resected aortic leaflet or raphe.
Pericardial Reconstruction and Augmentation
In the case of a large leaflet resection, pericardial patch reconstruction may be indicated (Figure 4). In this technique, an oval-shaped autologous or bovine pericardial patch that is slightly larger than the leaflet defect is prepared. Note that the entire aortic cusp could be replaced by pericardium, such as in the Ozaki procedure, but the extent of pericardial patch use should depend on the defect size, location, and its impact on valvular motion, hemodynamics, and coaptation. The pericardial patch is sewn to the leaflet defect using a running CV-6 PTFE suture. This will typically result in a type 0 BAV transformed from a type I BAV with excellent leaflet hemodynamics. Alternatively, when inadequate cusp tissue remains, a pericardial patch may be used for leaflet augmentation. This valve extension technique is completed by sewing a strip of pericardium to the free edge of the cusp to recreate a coaptation surface. The final cusp margin should be assessed using the alignment technique described previously.
Inverted Neochordal Repair
The innovative inverted neochordal repair technique represents another creative way to recreate a proper coaptation plane.21 A polytetrafluoroethylene suture is placed at the fused cusp margin and sewn toward the malformed commissure. The suture tails are tied and then anchored to the aorta at the malformed commissure apex, forming a suspensory neochord. The length of the neochord can be gently adjusted before securing it with knots to ensure the that proper coaptation plane is achieved.
Annular and STJ Stabilization
Annuloplasty
The annuloplasty repair technique aims to eliminate AR secondary to annular dilation without structural valve defects. In addition to VSRR, circular and external aortic annuloplasty can be adopted. The circular annuloplasty technique uses a continuous mattress suture that passes through the aortic wall at the leaflet attachment sites around the entire circumference of the valve. The suture is pulled tight at the ends to imbricate the aortic wall. The external aortic annuloplasty uses a flexible ring to correct ventriculoaortic junction dilation. This technique can be used as an adjunct to VSRR or in isolation. Owing to the difficulty of external dissection, this technique is typically limited to reducing the aortic dimension at the level of the ventriculoaortic junction, whereas the circular annuloplasty technique effectively reduces the aortic dimension at the level of the annulus. Both approaches may be appropriate for selected patients with mild aortic root dilation. Alternatively, internal annuloplasty rings have been developed that can be implanted just below the leaflet attachment sites inside the aorta. However, these internal annuloplasty rings do not address leaflet asymmetry, especially for BAV, require extensive manipulation of the leaflet and root structure, and can potentially alter the flow hemodynamics in the left ventricular outflow tract.
STJ Stabilization
STJ stabilization and remodeling effectively reduce aortic dilation at the level of the STJ. Therefore, patients with type I AR may be good candidates for STJ remodeling repairs, such as ascending aorta replacement using a Dacron graft. A modified technique for ascending aorta replacement with STJ stabilization provides additional benefit in terms of operation time and hemostasis.22 This modified repair can be achieved by first suspending the 3 commissures, then invaginating the Dacron graft toward the ventricle, followed by sewing the proximal anastomosis using a running 4-0 polypropylene suture. The intussuscepted graft is finally pulled outside of the ventricle for distal anastomosis.
Valve Reimplantation
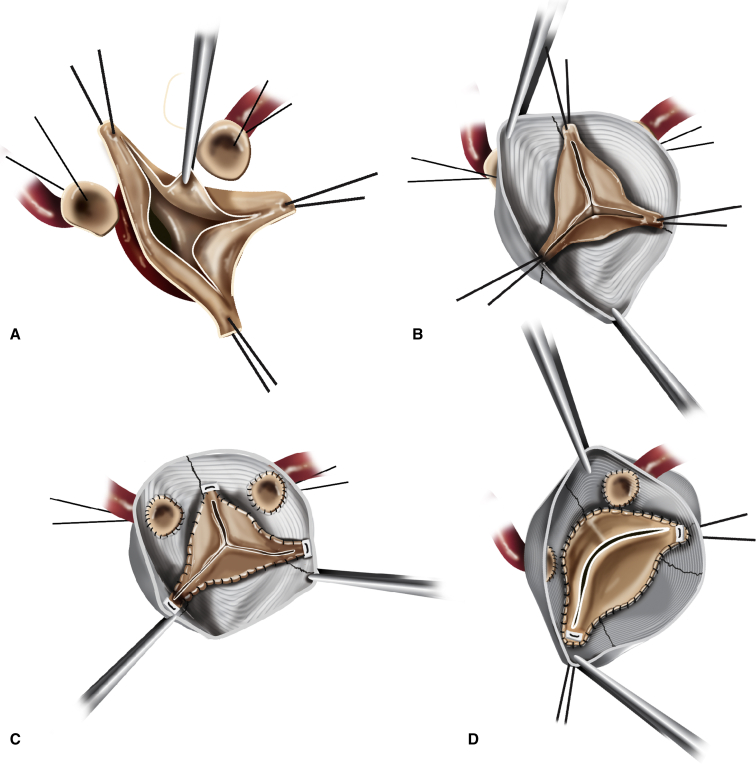
Aortic valve reimplantation techniques, such as the VSRR repair, can simultaneously address anomalies in the aortic cusp, aortic annulus, and STJ. The Dacron graft provides excellent external support for the aortic annulus and the STJ, making it an excellent option for patients with type I AR with otherwise normal aortic leaflets. This is also an attractive repair technique for patients with connective tissue diseases. However, aortic valves with severe calcification or leaflet destruction would likely not be successfully repaired using the reimplantation technique. The concomitant use of aortic cusp repair techniques with the VSRR repair is not uncommon. Finally, the VSRR technique allows for individual, asymmetric adjustment of the height and orientation of each commissure for the optimal leaflet coaptation geometry (Figure 5, A–C). In fact, commissure malalignment in height or orientation has been biomechanically shown to critically contribute to cusp prolapse in both bicuspid and tricuspid aortic valve regurgitation.3,17 VSRR is a highly versatile technique and has been adopted to address a wide variety of aortic and even pulmonary autograft valve dysfunctions.7,23, 24, 25 Although VSRR repair has undergone several iterations, the detailed repair techniques remain largely similar. In brief, the aortic root is first dissected to the natural anatomic limitation of the root insertion into the ventricular muscle. The pathologic aortic root is resected while preserving a thin strip of native aortic wall tissue along the leaflet attachment sites. The coronary buttons are harvested and retracted. The commissures are aligned using traction sutures, and a Dacron graft of the appropriate size is chosen. The commissures are reimplanted into the graft while suspending the commissures as high as possible. Pledgeted nonabsorbable sutures are placed underneath the cusps through the aorta and then passed through the base of the graft, following a scalloped pattern. Next, running sutures are placed along the native aortic wall tissue to reanastomose the valvular apparatus onto the Dacron graft. Finally, the coronary buttons are reattached in their respective locations above the right and left coronary cusps. Clinically, many graft configurations have been used for VSRR repair with some specifically focused on recapitulating the neosinus geometry. Using a 3D-printed ex vivo left heart simulator, it has been shown that straight grafts are associated with the lowest root distensibility, cusp opening and closing velocities, and relative cusp closing forces compared with other conduit configurations such as Valsalva grafts, grafts with Stanford modification, and grafts with anticommissural plication.11 Although the VSRR technique was originally described for tricuspid aortic valve, as the global experience with VSRR increases, this repair technique has been adopted for BAV patients with similar repair indications as tricuspid aortic valves, and excellent outcomes have been reported.26 Typically for type 0 BAV, the native commissure orientation will be respected during implantation. Otherwise, a 170°/190° commissural orientation in VSRR produces excellent valvular hemodynamics both clinically and through ex vivo biomechanical analyses (Figure 5, D).16,27
Figure 5.
A, The aortic root is dissected to the root insertion plane into the ventricular muscle. The pathologic aortic root is resected with the coronary buttons harvested and retracted. B, Symmetric suspension of the commissures may reveal a prolapsed cusp. C, The commissures can be subsequently rotated, with simultaneous height adjustment to correct cusp prolapse. D, Type I bicuspid aortic valve can be successfully repaired using the valve-sparing root replacement technique by creating a 170°/190° commissural orientation.
Conclusions
Aortic valve repair is an appealing technique for restoring valve competency without the risks and complications associated with aortic valve replacement. An individualized approach should be taken when repairing aortic valves. A systematic evaluation of all aortic valve repair techniques is warranted to further understand the biomechanics and clinical outcomes associated with each repair strategy.
Conflict of Interest Statement
The authors reported no conflicts of interest.
The Journal policy requires editors and reviewers to disclose conflicts of interest and to decline handling or reviewing manuscripts for which they may have a conflict of interest. The editors and reviewers of this article have no conflicts of interest.
References
- 1.Singh J.P., Evans J.C., Levy D., Larson M.G., Freed L.A., Fuller D.L. Prevalence and clinical determinants of mitral, tricuspid, and aortic regurgitation (The Framingham Heart Study) Am J Cardiol. 1999;83:897–902. doi: 10.1016/s0002-9149(98)01064-9. [DOI] [PubMed] [Google Scholar]
- 2.Boodhwani M., de Kerchove L., Glineur D., Poncelet A., Rubay J., Astarci P. Repair-oriented classification of aortic insufficiency: impact on surgical techniques and clinical outcomes. J Thorac Cardiovasc Surg. 2009;137:286–294. doi: 10.1016/j.jtcvs.2008.08.054. [DOI] [PubMed] [Google Scholar]
- 3.Zhu Y., Imbrie-Moore A.M., Paulsen M.J., Priromprintr B., Park M.H., Wang H. A novel aortic regurgitation model from cusp prolapse with hemodynamic validation using an ex vivo left heart simulator. J Cardiovasc Transl Res. June 3, 2020 doi: 10.1007/s12265-020-10038-z. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Greene C.L., Jaatinen K.J., Wang H., Koyano T.K., Bilbao M.S., Woo Y.J. Transcriptional profiling of normal, stenotic, and regurgitant human aortic valves. Genes (Basel) 2020;11:789. doi: 10.3390/genes11070789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nishimura R.A., Otto C.M., Bonow R.O., Carabello B.A., Erwin J.P., Fleisher L.A. 2017 AHA/ACC focused update of the 2014 AHA/ACC Guideline for the Management of Patients With Valvular Heart Disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2017;70:252–289. doi: 10.1016/j.jacc.2017.03.011. [DOI] [PubMed] [Google Scholar]
- 6.Goldstone A.B., Chiu P., Baiocchi M., Lingala B., Patrick W.L., Fischbein M.P. Mechanical or biologic prostheses for aortic-valve and mitral-valve replacement. N Engl J Med. 2017;377:1847–1857. doi: 10.1056/NEJMoa1613792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zhu Y., Cohen J.E., Ma M., Woo Y.J. Redo valve-sparing root replacement for delayed cusp derangement from ventricular septal defect. Ann Thorac Surg. 2019;108:e295–e296. doi: 10.1016/j.athoracsur.2019.03.040. [DOI] [PubMed] [Google Scholar]
- 8.Svensson L.G., Al Kindi A.H., Vivacqua A., Pettersson G.B., Gillinov A.M., Mihaljevic T. Long-term durability of bicuspid aortic valve repair. Ann Thorac Surg. 2014;97:1539–1548. doi: 10.1016/j.athoracsur.2013.11.036. [DOI] [PubMed] [Google Scholar]
- 9.Tanaka H., Takahashi H., Inoue T., Matsueda T., Oda T., Abe N. Which technique of cusp repair is durable in reimplantation procedure? Eur J Cardiothorac Surg. 2017;52:112–117. doi: 10.1093/ejcts/ezx034. [DOI] [PubMed] [Google Scholar]
- 10.Paulsen M.J., Kasinpila P., Imbrie-Moore A.M., Wang H., Hironaka C.E., Koyano T.K. Modeling conduit choice for valve-sparing aortic root replacement on biomechanics with a 3-dimensional-printed heart simulator. J Thorac Cardiovasc Surg. 2019;158:392–403. doi: 10.1016/j.jtcvs.2018.10.145. [DOI] [PubMed] [Google Scholar]
- 11.Paulsen M.J., Imbrie-Moore A.M., Baiocchi M., Wang H., Hironaka C.E., Lucian H.J. Comprehensive ex vivo comparison of 5 clinically used conduit configurations for valve-sparing aortic root replacement using a 3-dimensional–printed heart simulator. Circulation. 2020;142:1361–1373. doi: 10.1161/CIRCULATIONAHA.120.046612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhu Y., Roselli E.E., Idrees J.J., Wojnarski C.M., Griffin B., Kalahasti V. Outcomes after operations for unicuspid aortic valve with or without ascending repair in adults. Ann Thorac Surg. 2016;101:613–619. doi: 10.1016/j.athoracsur.2015.07.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ridley C.H., Vallabhajosyula P., Bavaria J.E., Patel P.A., Gutsche J.T., Shah R. The Sievers classification of the bicuspid aortic valve for the perioperative echocardiographer: the importance of valve phenotype for aortic valve repair in the era of the functional aortic annulus. J Cardiothorac Vasc Anesth. 2016;30:1142–1151. doi: 10.1053/j.jvca.2016.02.009. [DOI] [PubMed] [Google Scholar]
- 14.de Kerchove L., Mastrobuoni S., Froede L., Tamer S., Boodhwani M., van Dyck M. Variability of repairable bicuspid aortic valve phenotypes: towards an anatomical and repair-oriented classification. Eur J Cardiothorac Surg. February 20, 2019 doi: 10.1093/ejcts/ezz033. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 15.Nair S.K., Sudarshan C.D., Thorpe B.S., Singh J., Pillay T., Catarino P. Mini-Stern trial: a randomized trial comparing mini-sternotomy to full median sternotomy for aortic valve replacement. J Thorac Cardiovasc Surg. 2018;156:2124–2132.e31. doi: 10.1016/j.jtcvs.2018.05.057. [DOI] [PubMed] [Google Scholar]
- 16.Zhu Y., Imbrie-Moore A.M., Park M.H., Paulsen M.J., Wang H., MacArthur J.W. Ex vivo analysis of a porcine bicuspid aortic valve and aneurysm disease model. Ann Thorac Surg. 2021;111:e113–e115. doi: 10.1016/j.athoracsur.2020.05.086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhu Y., Imbrie-Moore A.M., Paulsen M.J., Priromprintr B., Wang H., Lucian H.J. Novel bicuspid aortic valve model with aortic regurgitation for hemodynamic status analysis using an ex vivo simulator. J Thorac Cardiovasc Surg. June 29, 2020 doi: 10.1016/j.jtcvs.2020.06.028. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Baumgartner F., Pearsall P., Omari K., Robertson J. Aortic and mitral annular remodeling using ultrasonic decalcification. Herz. 1996;21:179–182. [PubMed] [Google Scholar]
- 19.Kellner H.J., Pracki P., Hildebrandt A., Binner C., Eisele G., Struck E. Aortic valve debridement by ultrasonic surgical aspirator in degenerative, aortic valve stenosis: follow-up with Doppler echocardiography. Eur J Cardiothorac Surg. 1996;10:498–504. doi: 10.1016/s1010-7940(96)80414-3. [DOI] [PubMed] [Google Scholar]
- 20.Carr J.A., Savage E.B. Aortic valve repair for aortic insufficiency in adults: a contemporary review and comparison with replacement techniques. Eur J Cardiothorac Surg. 2004;25:6–15. doi: 10.1016/j.ejcts.2003.09.018. [DOI] [PubMed] [Google Scholar]
- 21.Woo Y.J., Frederick J.R. Valve-sparing aortic root replacement and neochordal repair of complex aortic leaflet pathology for ruptured sinus of Valsalva aneurysm fistulizing to the right ventricle. Ann Thorac Surg. 2013;96:1891–1893. doi: 10.1016/j.athoracsur.2013.05.008. [DOI] [PubMed] [Google Scholar]
- 22.Moscarelli M., Bonifazi R., Nasso G., Di Bari N., Speziale G. A modified technique for ascending aorta replacement with sinotubular junction stabilization. Ann Thorac Surg. 2020;110:e335–e337. doi: 10.1016/j.athoracsur.2020.03.121. [DOI] [PubMed] [Google Scholar]
- 23.Goldstone A.B., Jensen C.W., Bilbao M.S., Woo Y.J. Autograft valve-sparing root replacement for late Ross failure during quadruple-valve surgery. Ann Thorac Cardiovasc Surg. 2017;23:313–315. doi: 10.5761/atcs.cr.16-00264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kaczorowski D.J., Woo Y.J. Valve-sparing aortic root replacement with translocation of anomalous left coronary artery. Ann Thorac Surg. 2013;96:1466–1469. doi: 10.1016/j.athoracsur.2013.01.090. [DOI] [PubMed] [Google Scholar]
- 25.Goldstone A.B., Woo Y.J. Pulmonary autograft leaflet repair and valve sparing root replacement to correct late failure of the Ross procedure. J Card Surg. 2013;28:496–499. doi: 10.1111/jocs.12150. [DOI] [PubMed] [Google Scholar]
- 26.Beckmann E., Martens A., Krüger H., Korte W., Kaufeld T., Stettinger A. Aortic valve-sparing root replacement in patients with bicuspid aortic valve: long-term outcome with the David I procedure over 20 years. Eur J Cardiothorac Surg. 2020;58:86–93. doi: 10.1093/ejcts/ezaa083. [DOI] [PubMed] [Google Scholar]
- 27.Siki M.A., Habertheuer A., Bavaria J.E., Komlo C., Hung M., Freas M.A. Two different geometric orientations for aortic neoroot creation in bicuspid aortic valve repair with root reimplantation. J Thorac Cardiovasc Surg. 2020;160:45–57. doi: 10.1016/j.jtcvs.2019.06.119. [DOI] [PubMed] [Google Scholar]