Abstract
Aims
To assess the proportion of women with gestational diabetes (GDM) by performing postpartum Oral Glucose Tolerance Test (OGTT) and to identify GDM phenotypes at high-risk of postpartum dysglycemia (PPD).
Methods
Observational, retrospective, multicenter study involving consecutive GDM women. Recursive partitioning (RECPAM) analysis was used to identify distinct and homogeneous subgroups of women at different PPD risk.
Results
From a sample of 2,736 women, OGTT was performed in 941 (34.4%) women, of whom 217 (23.0%) developed PPD. Insulin-treated women having family history of diabetes represented the subgroup with the highest PPD risk (OR 5.57, 95% CI 3.60–8.63) compared to the reference class (women on diet with pre-pregnancy BMI < = 28.1 kg/m2). Insulin-treated women without family diabetes history and women on diet with pre-pregnancy BMI > 28.1 kg/m2 showed a two-fold PPD risk. Previous GDM and socioeconomic status represent additional predictors. Fasting more than post-prandial glycemia plays a predictive role, with values of 81–87 mg/dl (4.5–4.8 mmol/l) (lower than the current diagnostic GDM threshold) being associated with PPD risk.
Conclusions
Increasing compliance to postpartum OGTT to prevent/delay PPD is a priority. Easily available characteristics identify subgroups of women more likely to benefit from preventive strategies. Fasting BG values during pregnancy lower than those usually considered deserve attention.
Keywords: Gestational diabetes, Postpartum dysglycemia, Risk factors, Type 2 diabetes, Obesity
Introduction
Gestational diabetes mellitus (GDM) represents a pathological condition for the mother and the fetus during pregnancy, at delivery and in the follow-up period [1]. Prevalence varies in the different countries and in Italy it is estimated that 11% of pregnancies are aggravated by GDM [2, 3].
Women with GDM have an increased risk of adverse obstetric events and adverse neonatal outcomes compared to women with physiological pregnancy, including fetal macrosomia, shoulder dystocia, neonatal trauma, neonatal jaundice, respiratory distress, and neonatal hypoglycemia [4–6].
GDM is also associated with an increased risk of dysglycemia and type 2 diabetes development after delivery compared to normal pregnancy [7, 8]. Women with gestational diabetes have a 7–12 times higher risk of developing type 2 diabetes compared to women with normoglycemic pregnancy [7].
Recent evidence underlines the importance of early identification of GDM and its subsequent treatment to promote maternal–fetal health [9]. Based on the HAPO (Hyperglycemia and Adverse Pregnancy Outcomes) study, the IADPSG panel defined new guidelines for GDM, improving criteria for screening, diagnosis and treatment of GDM [10]. In Italy, the ‘Guideline on physiological pregnancy’’ was also developed, to disseminate specific recommendation on GDM, describing screening and diagnostic procedures. A selective screening, based on the presence of specific risk factors, is recommended after the exclusion of overt diabetes; this guideline also recommends a follow-up OGTT to be performed not before 6 weeks from the delivery [11, 12].
A previous publication based on the STRONG study aimed to assess the risk of adverse neonatal outcomes in women with GDM and to identify subgroups of women at high risk for adverse neonatal outcomes [13]. This secondary analysis, based on the same data, aimed at evaluating compliance with follow-up OGTT and identifying subgroups of women at high risk for developing postpartum dysglycemia (PPD).
Methods
This was an observational, retrospective, multicenter study. Details on materials and methods of the study protocol have been published elsewhere [13].
The study involved consecutive women with pregnancy complicated by GDM cared for by a network of Italian outpatient diabetes clinics.
Women with the following characteristics were eligible for this study: age ≥ 18 years, GDM, delivery by the end of the planned study period (May 2015), signature of informed consent.
Exclusion criteria were: diagnosis of pre-gestational diabetes, twin pregnancy.
According to current Italian recommendations [11, 12], all women with a physiologic pregnancy receive an evaluation of fasting plasma glucose during the first trimester. Women with at least one risk factor (age > = 35 years, first-degree relative with diabetes, BMI > 25 kg/m2, Asian, Middle Eastern, or Caribbean ethnicity) perform the screening for GDM at 24–28th weeks of gestation. Nevertheless, it is recommended that women with at least one of the following conditions should be screened at 16–18 weeks of gestation: previous GDM, pre-pregnancy body mass index (BMI) ≥ 30 kg/m2, plasma glucose values at the beginning of pregnancy (within the first trimester) between 100 and 125 mg/dl (5.6–6.9 mmol/l).
An oral glucose tolerance test with 75 g of glucose (OGTT-75 g) is performed.
Women with one or more plasma glucose values above the established thresholds (≥ 92 mg/dl/5.1 mmol/l at baseline (T0′), ≥ 180 mg/dl/10.0 mmol/l after 1 h (T60′) from the load, ≥ 153 mg/dl/8.5 mmol/l after 2 h (T120′) from the load) are diagnosed as affected by GDM. After the diagnosis, according to usual care, women with GDM are invited to perform self-monitoring of blood glucose, to follow a balanced diet, and to do regular physical activity. If blood glucose (BG) is not in target, a pharmacological therapy (in Italy the only approved treatment is insulin) is started.
Follow-up OGTT is recommended within 12 weeks after delivery. Based on follow-up OGTT results, dysglycemia was defined as Impaired Fasting Glucose (IFG, BG levels between 100 and 125 mg/dl/5.6 and 6.9 mmol/l at T0′), Impaired Glucose Tolerance (IGT, BG levels between 140 and 199 mg/dl/7.8 and 11.1 mmol/l at T120′), or type 2 diabetes (BG levels > = 126 mg/dl/7.0 mmol/l at T0′ or > = 200 mg/dl/11.1 at T120′).
Data collected included sociodemographic information, clinical characteristics related to the period before and during pregnancy, and information on maternal–fetal outcomes (i.e., adequate, large or small fetal growth for gestational age, macrosomia, minor and major malformations, neonatal intensive care need, neonatal hypoglycemia needing i.v. treatment, neonatal hypocalcemia, neonatal hyperbilirubinemia, shoulder distocia, respiratory distress, type of delivery, stillbirths, maternal mortality, neonatal mortality) [13].
All information was collected on electronic case report forms and data were anonymous.
The study was conducted in accordance with the Helsinki Declaration on Medical Research on Humans and with the Good Clinical Practice (GCP). The study was approved by the Ethics Committees of all participating Centers. Participant patients gave informed consent before taking part in the study.
Statistical analysis
Descriptive data were summarized as mean and standard deviation or percentages. Characteristics of the study population were assessed overall and by the development of dysglycemia after the delivery.
Groups were compared using Student’s test (continuous, normally distributed variables), Mann–Whitney U-test (continuous, not normally distributed variables), or chi-square test (categorical variables).
RECursive Partitioning and AMalgamation (RECPAM) analysis, a tree-based statistical method that integrates standard regression and tree-growing techniques, was used to detect potential interactions among the different variables in predicting development of postpartum dysglycemia and identify homogeneous and distinct subgroups of patients with increased likelihood of reaching the endpoint [14]. At each partitioning step, the RECPAM method automatically choses the covariate and its best binary split to maximize the difference in risk of experiencing the outcome. The algorithm stops when user-defined stopping rules are met. In this case, each final class was required to have at least 100 patients in total and 30 patients with the target endpoint. The set of variables tested in the RECPAM analysis included: age, ethnicity, education level, occupation, pre-pregnancy BMI, previous GDM, family history (first-degree relatives) of diabetes, physical activity before pregnancy, first pregnancy, smoke and GDM treatment. Continuous variables were not categorized so as to allow the algorithm to choose the natural cut-off points when identifying distinct subgroups of patients. For each subgroup or class, the proportion (%) of patients reaching the endpoint and the likelihood (OR and 95% CI) to reach the endpoint versus the reference subgroup were obtained.
Additional RECPAM analyses were performed to test the role of fasting BG at first trimester of pregnancy and BG values at T0′, T60′, and T120′ of diagnostic OGTT (at 16–18 or 24–28 weeks, separately) in the subgroups of women with available values.
All analyses were performed using the SAS version9.3 (SAS Institute Inc.) program.
Results
From a total sample of 2,736 women, postpartum OGTT was performed within 6–12 weeks from the delivery in 941 (34.4%) women. Median proportion of women with a follow-up OGTT largely varied among the 42 participating centers (median 42.0%, interquartile range 22.0–80.0%).
Compared to women not performing follow-up OGTT, those performing the test were less often Caucasian, were less likely to be at their first pregnancy, had more frequently had a previous GDM, had lower levels of diastolic blood pressure, total cholesterol and triglycerides, and a more frequent use of insulin for their GDM (Table 1).
Table 1.
Sociodemographic and clinical characteristics of patients, overall, by availability of follow-up OGTT within 12 weeks from the delivery and by development of postpartum dysglycemia
| Women without follow-up OGTT | Women with follow-up OGTT | p value* | No postpartum dysglycemia | Postpartum dysglycemia | p value* | |
|---|---|---|---|---|---|---|
| N | 1795 | 941 | 724 | 217 | ||
| Age (years) | 36.5 ± 5.2 | 36.7 ± 4.8 | 0.51 | 36.8 ± 4.7 | 36.6 ± 5.4 | 0.62 |
| School education (%) | ||||||
| Low | 26.3 | 25.5 | 0.41 | 22.6 | 35.3 | 0.002 |
| Median | 47.3 | 50.6 | 50.9 | 49.6 | ||
| High | 26.3 | 23.9 | 26.4 | 15.1 | ||
| Occupation (%) | 0.12 | |||||
| Housewife | 36.8 | 33.2 | 28.7 | 48.2 | < 0.0001 | |
| Employed | 62.8 | 65.7 | 70.1 | 51.1 | ||
| Student | 0.4 | 1.1 | 1.3 | 0.7 | ||
| Physical activity before pregnancy (%) | 24.6 | 26 | 0.52 | 28.8 | 16.7 | 0.002 |
| Smoke (%) | 0.27 | |||||
| No | 77.4 | 80.2 | 80 | 81.1 | 0.06 | |
| Yes | 9.9 | 8.9 | 8 | 11.7 | ||
| Ex | 12.6 | 10.9 | 12 | 7.3 | ||
| Caucasian ethnicity (%) | 48.5 | 37.8 | < 0.0001 | 36.2 | 42.9 | 0.08 |
| Family history of diabetes (%) | 41.3 | 42.4 | 0.58 | 38.8 | 54.7 | < 0.0001 |
| First pregnancy (%) | 47.5 | 41.1 | 0.002 | 43.9 | 31.9 | 0.002 |
| Previous GDM (%) | 12.3 | 17.3 | 0.0004 | 14.8 | 25.7 | 0.0002 |
| Pre-pregnancy BMI (kg/m2) | 26.0 ± 6.0 | 26.0 ± 5.5 | 0.57 | 25.6 ± 5.5 | 27.4 ± 5.1 | < 0.0001 |
| Pre-pregnancy BMI in classes (%) | ||||||
| < 25 | 52.1 | 51.0 | 0.10 | 55.6 | 35.4 | < 0.0001 |
| 25–30 | 26.5 | 30.0 | 27.9 | 37.4 | ||
| > 30 | 21.4 | 19.0 | 16.5 | 27.2 | ||
| Weight gain (kg) | 9.8 ± 5.9 | 9.9 ± 5.3 | 0.98 | 10.2 ± 4.9 | 8.8 ± 6.2 | 0.0002 |
| Blood glucose at first trimester (mg/dl) | 88.9 ± 11.7 | 88.7 ± 11.4 | 0.86 | 86.8 ± 10.5 | 94.6 ± 12.1 | < 0.0001 |
| Diagnostic OGTT (%) | ||||||
| 16–17th week | 12.4 | 16.3 | 0.02 | 12.2 | 32.6 | < 0.0001 |
| 18–24th week | 87.6 | 83.7 | 87.8 | 67.4 | ||
| HbA1c at diagnosis % (mmol/mol) | 32.7 ± 8.3 | 32.7 ± 8.6 | 0.29 | 32.3 ± 7.9 | 34.0 ± 10.5 | < 0.0001 |
| Systolic blood pressure (mmHg) | 112.7 ± 13.8 | 111.2 ± 12.8 | 0.06 | 111.3 ± 12.8 | 111.1 ± 12.7 | 0.75 |
| Diastolic blood pressure (mmHg) | 71.0 ± 9.9 | 69.2 ± 9.2 | < 0.0001 | 69.4 ± 9.2 | 68.7 ± 9.0 | 0.30 |
| Total cholesterol (mg/dl) | 254.5 ± 51.2 | 247.0 ± 47.8 | 0.04 | 248.1 ± 47.1 | 242.6 ± 50.6 | 0.33 |
| HDL cholesterol (mg/dl) | 68.1 ± 15.1 | 69.3 ± 15.8 | 0.32 | 69.5 ± 15.9 | 68.1 ± 15.4 | 0.52 |
| LDL cholesterol (mg/dl) | 142.0 ± 45.6 | 142.8 ± 44.1 | 0.74 | 143.9 ± 42.1 | 137.6 ± 52.5 | 0.14 |
| Triglycerides (mg/dl) | 220.6 ± 102.3 | 186.6 ± 83.1 | < 0.0001 | 183.3 ± 79.7 | 199.8 ± 94.8 | 0.21 |
| Glucose lowering treatment (%) | ||||||
| Diet | 63.2 | 50.5 | < 0.0001 | 56.3 | 31.5 | < 0.0001 |
| Insulin | 36.8 | 49.5 | 43.7 | 68.5 | ||
| Gestational week at delivery | 38.4 ± 1.7 | 38.6 ± 1.4 | 0.03 | 38.7 ± 1.4 | 38.3 ± 1.5 | < 0.0001 |
| Adverse neonatal outcome (%) | 30.3 | 29.1 | 0.52 | 27 | 36.1 | 0.009 |
*p values < 0.05 are in bold and expresses statistically significant between-group differences. Student’s test (for continuous, normally distributed variables), Mann–Whitney test (for continuous, not normally distributed variables) or Chi-square test (for categorical variables)
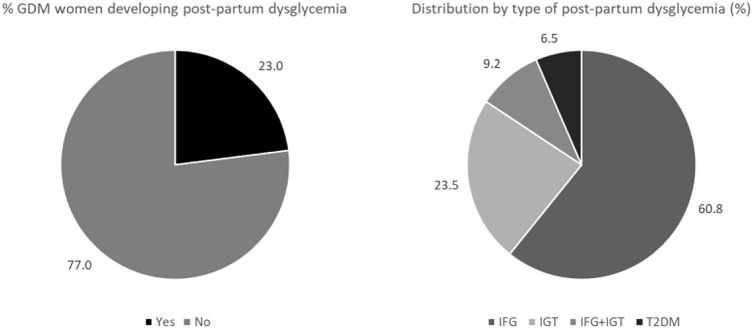
Among women performing follow-up OGTT, 217 (23.0%) developed postpartum dysglycemia (Table 1). In particular, 132 (60.8%) women developed IFG, 51 (23.5%) developed IGT, 20 (9.2%) developed both IFG and IGT, and 14 (6.5%) developed type 2 diabetes (Fig. 1).
Fig. 1.
Distribution by development and type of postpartum dysglycemia. GDM gestational diabetes mellitus, IFG impaired fasting glucose, IGT impaired glucose tolerance, T2DM type 2 diabetes
Compared to women not developing dysglycemia, those developing the condition were more likely to report a low level of school education and being not employed, and less likely to practice physical activity. Pre-pregnancy BMI was significantly higher in women developing dysglycemia, although their weight gain during pregnancy was significantly lower according to IOM guidelines [15]. Furthermore, they more frequently reported family history of diabetes and history of previous GDM. In these women HbA1c levels and BG levels during pregnancy were higher, a higher proportion performed the diagnostic OGTT at 16-18th weeks, and they were treated more often with insulin for GDM. Adverse neonatal outcomes had occurred more frequently in women with postpartum dysglycemia.
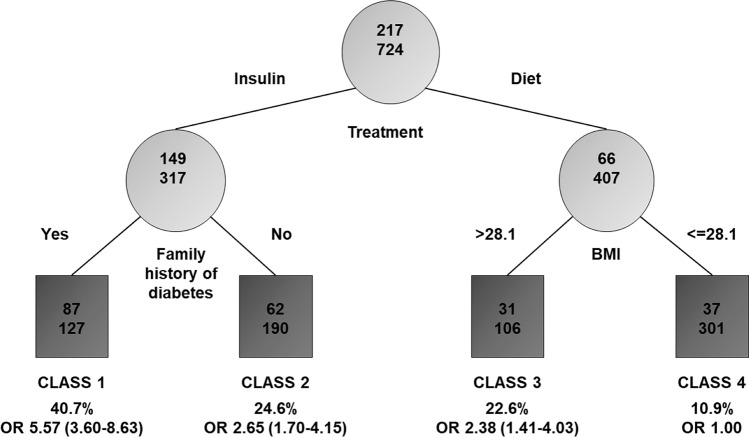
RECPAM analysis led to the identification of four classes at different risks of developing postpartum dysglycemia (Fig. 2). The most important variables differentiating the risk of developing postpartum dysglycemia were insulin GDM treatment and pre-pregnancy BMI, with patients treated with diet and pre-pregnancy BMI levels < = 28.1 kg/m2 having the lowest prevalence (10.9%). Therefore, this group served as the reference category.
Fig. 2.
Results of the RECPAM analysis: identification of subgroups at different risks of developing postpartum dysglycemia. The tree-growing algorithm modeled odds ratios (ORs) following a logistic regression model with age, education, occupation, ethnicity, previous GDM, pre-pregnancy BMI, family history of diabetes, first pregnancy, physical activity before pregnancy, smoke, treatment, total cholesterol, HDL, LDL, triglycerides, systolic blood pressure, diastolic blood pressure and composite of adverse neonatal outcomes as covariates. Splitting variables are shown between branches, whereas the condition sending patients to the left or right sibling is on the relative branch. Circles indicate subgroups of patients, squares indicate the final RECPAM classes. Numbers inside circles and squares represent the number of events (top) and the number of nonevents (bottom), respectively. An OR with the corresponding 95% CI (in parentheses) is shown for each class. Class 4, with the lowest risk of developing dysglycemia, is placed at the extreme right and is the reference category (OR = 1)
On the opposite side of the regression tree, patients treated with insulin and having family history of diabetes represented the subgroup with the highest prevalence (40.7%) and the highest risk of dysglycemia (OR = 5.57; 95%CI 3.60–8.63).
Women treated with insulin for GDM and no family history of diabetes (class 2) also had a significant risk of adverse outcome compared with the reference category (OR = 2.65; 95% CI 1.70–4.15); similarly, women not treated with insulin and with pre-pregnancy BMI levels > 28.1 kg/m2 (class 3) showed a twofold increased risk of dysglycemia (OR = 2.38; 95% CI 1.41–4.03).
The four RECPAM classes differed for other characteristics (Table 2): women with the highest risk of dysglycemia were more likely to have a low level of school education. Average pre-pregnancy BMI substantially differed among classes, ranging from 22.1 to 30.4 kg/m2. Results of OGTT performed during pregnancy also varied among classes. Relevantly, in class 4 OGTT test at 16–18 weeks showed levels of BG at T0′ and T60′ markedly lower than the other 3 classes. The four classes also had different levels of systolic and diastolic blood pressure, without a clear trend. Triglycerides mean levels were of 212 mg/dl in class 1, 186 mg/dl in class 2 and 3, and 141 in class 4 (p < 0.0001). Levels of follow-up OGTT at T0′ and T120′ decreased from class 1 to class 4. From class 1 to class 4, adverse neonatal outcomes occurred in 37.9%, 31.0%, 24.8%, and 23.8%, respectively (p = 0.003).
Table 2.
Sociodemographic and clinical characteristics of patients by RECPAM classes
| CLASS1 | CLASS2 | CLASS3 | CLASS4 | p value* | |
|---|---|---|---|---|---|
| N | 214 | 252 | 137 | 338 | |
| Age (years) | 37.0 ± 5.4 | 36.5 ± 4.5 | 37.0 ± 5.1 | 36.6 ± 4.6 | 0.59 |
| Education (%) | |||||
| Low | 36.2 | 28.3 | 29.2 | 16.2 | 0.001 |
| Median | 46.5 | 47.6 | 51.7 | 54.7 | |
| High | 17.3 | 24.1 | 19.1 | 29.1 | |
| Occupation (%) | |||||
| Housewife | 39.2 | 38.9 | 33.7 | 25.5 | 0.06 |
| Employed | 60.8 | 59.9 | 65.2 | 72.7 | |
| Student | 0 | 1.2 | 1.1 | 1.7 | |
| Physical activity before pregnancy (%) | 19.9 | 22.3 | 20.2 | 34.4 | 0.002 |
| Smoke (%) | |||||
| No | 75.6 | 78.1 | 83.8 | 83.2 | 0.01 |
| Yes | 15.0 | 9.9 | 6.9 | 5.1 | |
| Ex | 9.3 | 12.0 | 9.2 | 11.7 | |
| Ethnicity (%) | |||||
| Caucasian | 48.8 | 36.1 | 37.5 | 32.2 | 0.002 |
| Other | 51.2 | 63.9 | 62.5 | 67.8 | |
| Family history of diabetes (%) | 100 | 0 | 41.0 | 38.6 | < 0.0001 |
| First pregnancy (%) | 26.9 | 39.7 | 37.2 | 52.7 | < 0.0001 |
| Previous GDM (%) | 19.9 | 22.1 | 16.9 | 12.3 | 0.01 |
| Pre-pregnancy BMI (kg/m2) | 27.8 ± 5.8 | 27.0 ± 5.7 | 30.4 ± 4.1 | 22.1 ± 2.3 | < 0.0001 |
| Weight gain (kg) | 9.8 ± 5.7 | 10.1 ± 5.6 | 7.7 ± 5.7 | 10.7 ± 4.2 | < 0.0001 |
| Blood glucose at first trimester (mg/dl) | 92.2 ± 11.3 | 91.0 ± 11.8 | 87.4 ± 8.5 | 85.1 ± 11.1 | < 0.0001 |
| HbA1c at diagnosis (mmol/mol) | 34.5 ± 8.9 | 34.6 ± 6.6 | 32.5 ± 8.1 | 30.2 ± 9.2 | < 0.0001 |
| Diagnostic OGTT (%) | |||||
| 16–17th week | 25.0 | 22.7 | 17.0 | 7.1 | < 0.0001 |
| 18–24th week | 75.0 | 77.3 | 83.0 | 92.9 | |
| 16–18 weeks OGTT blood glucose T0′ (mg/dl) | 98.8 ± 9.4 | 95.9 ± 9.4 | 92.6 ± 9.0 | 90.0 ± 9.6 | 0.002 |
| 16–18 weeks OGTT blood glucose T60′ (mg/dl) | 174.0 ± 34.3 | 170.1 ± 37.3 | 179.3 ± 30.2 | 143.6 ± 35.9 | 0.02 |
| 16–18 weeks OGTT blood glucose T120′ (mg/dl) | 141.2 ± 35.1 | 142.5 ± 33.1 | 143.0 ± 33.0 | 123.8 ± 30.2 | 0.16 |
| 24–28 weeks OGTT blood glucose T0′ (mg/dl) | 79.8 ± 35.6 | 80.7 ± 31.9 | 84.5 ± 20.5 | 82.3 ± 19.3 | < 0.0001 |
| 24–28 weeks OGTT blood glucose T60′ (mg/dl) | 179.6 ± 27.5 | 174.9 ± 30.4 | 176.1 ± 28.8 | 176.1 ± 30.5 | 0.82 |
| 24–28 weeks OGTT blood glucose T120′ (mg/dl) | 146.9 ± 29.2 | 149.4 ± 31.5 | 151.6 ± 33.5 | 147.1 ± 31.1 | 0.61 |
| Follow-up OGTT T0′ (mg/dl) | 95.6 ± 12.9 | 92.1 ± 10.1 | 90.6 ± 8.6 | 87.6 ± 9.0 | < 0.0001 |
| Follow-up OGTT T120′ (mg/dl) | 110.3 ± 33.1 | 101.8 ± 30.1 | 104.4 ± 24.2 | 95.8 ± 24.7 | < 0.0001 |
| Systolic blood pressure (mmHg) | 111.8 ± 12.4 | 109.9 ± 13.0 | 116.7 ± 13.1 | 109.5 ± 12.0 | < 0.0001 |
| Diastolic blood pressure (mmHg) | 69.1 ± 8.9 | 68.9 ± 9.2 | 72.2 ± 9.8 | 68.3 ± 8.8 | 0.002 |
| Total cholesterol (mg/dl) | 249.7 ± 46.3 | 251.3 ± 53.3 | 245.7 ± 56.1 | 243.1 ± 40.4 | 0.66 |
| HDL cholesterol (mg/dl) | 68.7 ± 16.0 | 70.1 ± 16.1 | 68.6 ± 16.7 | 69.5 ± 14.9 | 0.76 |
| LDL Cholesterol (mg/dl) | 143.5 ± 44.0 | 146.8 ± 50.1 | 141.5 ± 49.9 | 140.5 ± 36.2 | 0.97 |
| Triglycerides (mg/dl) | 212.9 ± 81.5 | 185.5 ± 73.3 | 186.7 ± 75.7 | 168.7 ± 89.0 | < 0.0001 |
| Glucose lowering treatment (%) | |||||
| Diet | 0 | 0 | 100 | 100 | < 0.0001 |
| Insulin | 100 | 100 | 0 | 0 | |
| Gestational week at delivery | 38.2 ± 1.7 | 38.6 ± 1.2 | 38.8 ± 1.3 | 38.9 ± 1.4 | < 0.0001 |
| Adverse neonatal outcome (%) | 37.9 | 31.0 | 24.8 | 23.8 | 0.003 |
Student’s test (for continuous, normally distributed variables), Mann–Whitney test (for continuous, not normally distributed variables) or Chi-square test (for categorical variables)
*p values < 0.05 are in bold and expresses statistically significant between-group differences
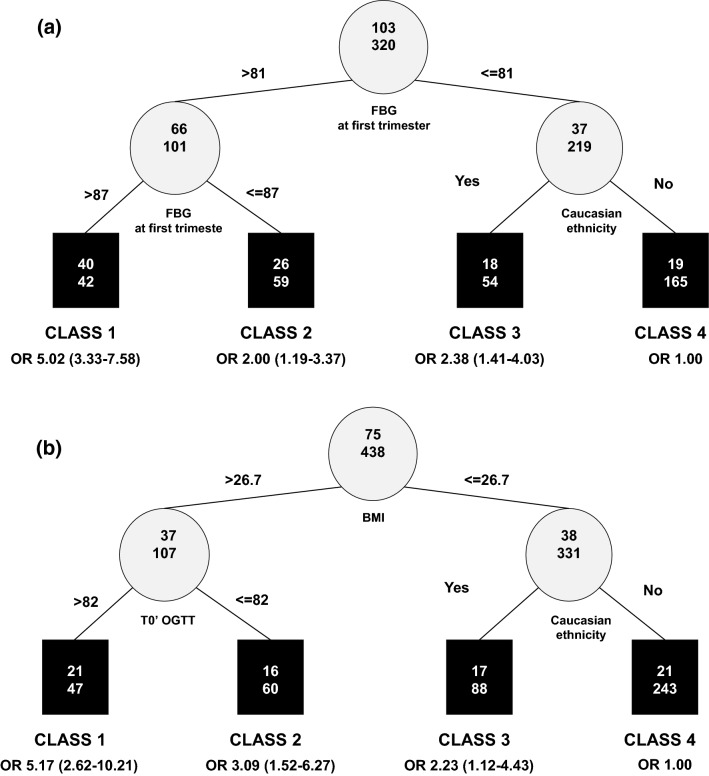
Data on fasting BG in the first trimester, OGTT at 16–18 weeks and OGTT at 24–28 weeks were available for 423 women (44.9%), 96 (10.2%) and 514 (54.6%), respectively. Additional exploratory RECPAM analyses in the two subgroups with adequate sample size (the subgroup with OGTT at 16–18 weeks was too small to allow additional analyses) were performed to assess the role of these BG values on the risk of postpartum dysglycemia (Fig. 3). In the two RECPAM models, fasting BG at first trimester represented the first selected splitting variable, while BG at T0′ of OGTT was selected as the second splitting variable after BMI. In both cases, the cut-offs associated with increased risk ranged between 81 and 87 mg/dl, therefore lower than 92 mg/dl, which is the usually accepted threshold for the diagnosis of GDM.
Fig. 3.
Additional exploratory RECPAM analyses on the subgroups of women with available data on FBG at first trimester and OGTT 24–28 weeks. a Identification of subgroups at different risks of developing postpartum dysglycemia including FBG (mg/dl) at first trimester of pregnancy as additional covariate (N = 423). b Identification of subgroups at different risks of developing postpartum dysglycemia including OGTT at 24–28 weeks (mg/dl) as additional covariate (N = 513)
Discussion
Main findings
This Italian observational study shows that among women with a recent history of GDM only about one in three is compliant with the recommended follow-up OGTT within 12 weeks, despite the high risk of postpartum dysglycemia related to GDM.
Our study identified a specific phenotype of women more likely to be compliant with postpartum OGTT, i.e., women with more severe GDM (as documented by more frequent use of insulin) and attitude to be adherent to the overall care (as documented by the better control of all parameters). However, our study also clarifies that compliance with follow-up OGTT is largely dependent from organizational aspects of each center, as documented by the different proportions of women who performed it in the different centers.
Furthermore, our study documents that among women with follow-up OGTT, about 1/4 developed postpartum dysglycemia. IFG was the most common glucose metabolism abnormality, being present in 60.8% of the women as the only alteration. RECPAM analyses show that fasting BG more than post-prandial BG deserve particular consideration: fasting values during pregnancy ranging between 81 and 87 mg/dl (4.5 and 4.8 mmol/l), lower than the current diagnostic threshold of 92 mg/dl (5.1 mmol/l) for GDM at T0′, are associated with increased risk of postpartum dysglycemia.
In addition, our study documents that, among a set of known risk factors, insulin treatment (as a proxy of more severe GDM), pre-pregnancy BMI, and family history of diabetes are the key variables for identifying subgroups of women with a from twofold to fivefold higher risk of developing dysglycemia compared to the reference class (women with BMI lower than 28 kg/m2 not requiring insulin for GDM).
Comparison with existing data
In other countries, among women with GDM, postpartum IGT prevalence ranged between 17 and 23% and diabetes prevalence between 5 and 14% [16–18], and rates of postpartum follow-up was low in most parts of the world [17, 18].
Prevalence of postpartum dysglycemia and distribution of IFG/IGT after postpartum varied according to different ethnic populations [19]. It is also known that traditional cardiovascular risk factors (especially high levels of BMI) have a stronger association with isolated IFG than with isolated IGT in women with prior GDM [20].
It is noteworthy that many prominent barriers for non-compliance with follow-up OGTT could be overcome through appropriate educational sessions [21, 22] and reminder/engagement systems [23, 24]. In this respect, previous studies have documented that fasting BG and/or HbA1c are not sensitive enough to replace the OGTT in early postpartum [25].
Recently, Waters et al. documented in women with GDM that no significant correlations were present between longitudinal changes in maternal lipids, body weight, and inflammatory markers and changes in insulin sensitivity, insulin response and disposition index from late pregnancy and during the early postpartum period. Therefore, owing to difficulties in completing an OGTT in the later postpartum period (6–12 weeks), screening within 1–5 days postpartum is proposed as a viable option [24].
Established risk factors for GDM include ethnicity, obesity, and family history of diabetes [26, 27], while lower insulin sensitivity, use of insulin therapy, pre-pregnancy obesity, severity of GDM and high HbA1c levels during pregnancy were identified as independent predictors of subsequent diabetes in previous observational studies [28–30], in line with our data. Our study adds novel information about their interaction and their priority in identifying specific phenotypes. In addition, while the predictive role of first trimester BG values is recognized [31], only another study identified a cut-off of fasting BG lower than 92 mg/dl (5.1 mmol/l) and near to 80 mg/dl (4.4 mmol/l) as a risk factor in this population, although the outcome was GDM and not postpartum dysglycemia, in line with our data [32].
Implications for research and clinical practice
Increasing the awareness of the risk of dysglycemia after GDM is a priority. Early diagnosis of postpartum dysglycemia provides an opportunity to use dietary, lifestyle, and pharmacological interventions that might prevent or delay the onset of type 2 diabetes in GDM women [33–38]. A return to pre-pregnancy weight within 1 year postpartum should be the goal [39–41].
Timing of follow-up OGTT should be more clearly defined. Some guidelines recommend that after delivery all women with GDM have to be re-evaluated by a 75 g OGTT (WHO criteria) 4–12 weeks postpartum to reclassify the glucose tolerance and every 2 years in cases of normal glucose tolerance, but evidence level is B [42].
The reliability of follow-up OGTT within few days after delivery deserves further investigation [24].
New research is focused on genetic, immunologic and biological markers to predict future GDM or T2DM [43]. However, in clinical practice it remains important to “phenotyping” the women at risk on the basis of easily available information.
Strengths and limitations
As the main strength, this is a large national multicenter study giving a national picture of the care and the outcome of pregnancies complicated by GDM. Study limitations were the retrospective design, not having planned a longer mother and children follow-up and not having collected information about specific ethnicities of women.
Conclusions
Increasing compliance to postpartum OGTT to prevent or delay type 2 diabetes onset after GDM is a priority. Easily available patient characteristics such as socioeconomic status, family history of diabetes, previous GDM, pre-pregnancy BMI, need for insulin for GDM treatment, and fasting BG in the first phase of pregnancy at lower thresholds than those currently utilized, can help focusing on those women who are more likely to benefit from preventive strategies.
Acknowledgements
STRONG Study Collaborators: Angela Napoli, Olimpia Bitterman, Camilla Festa, Chiara Giuliani, Matteo Bonomo, Basilio Pintaudi, Elena Cimino, Elena Mion, Teresa Marcone, Cristina Lencioni, Graziano Di Cianni, Laura Sciacca, Andrea Tumminia, Agostino Milluzzo, Ester Vitacolonna, Federica Fraticelli, Marica Franzago, Alessandro Roberto Dodesini, Elena Ciriello, Mariagrazia Dalfrà, Annunziata Lapolla, Raffaella Fresa, Aurora Grassi, Paolo Limone, Annamaria Nuzzi, Andi Masha, Laura Grimaldi, Sara Biglino, Egle Ansaldi, Maurizia Battezzati, Giancarla Meregalli, Valentina De Mori, Denise Berzi, Antonio Bossi, Viviana Baggi, Elisabetta Lovati, Lara Quarleri, Tiziana Romanelli, Silvia Clementi, Ilaria Nicolao, Francesca Zambotti, Simonetta Lombardi, Silvana Costa, Chiara Tommasi, Silvia Rancan, Giovanna Lisato, Paola Bordon, Daniela Turazzi, Francesco Mollo, Franco Grimaldi, Laura Tonutti, Sandra Agus, Maria Rosaria Falivene, Giorgio Versari, Laura Corsi, Maria Delucchi, Luisa Ratto, Maria Grazia Magotti, Tiziana Frusca, Silvia Haddoub, Alice Suprani, Mary Mori, Maria Grazia Vita, Nicolina Di Biase, Alessandra Bertolotto, Aragona Michele, Bianchi Cristina, Emilia Lacaria, Elisa Guarino, Federica Monaci, Francesco Dotta, Elisabetta Torlone, Carlo Lalli, Chiara di Loreto, Maura Scarponi, Angela Del Prete, Sergio Leotta, Iolanda Coletta, Santina Abbruzzese, Valeria Montani, Emanuela Cannarsa, Pierpaolo Contini, Raffaella Vero, Rosa Oliverio, Marina Scavini, Nicoletta Dozio, Maria Pia Imbergamo, Renzo Cordera, Laura Affinito, Davide Maggi, Caterina Bordone, Elena Fochesato, Alessandra Pissarelli, Eros Libera, Susanna Morano, Tiziana Filardi, Mara Fallarino.
Abbreviations
- 95% CI
95% Confidence intervals
- BG
Blood Glucose
- BMI
Body mass index
- GCP
Good Clinical Practice
- GDM
Gestational diabetes mellitus
- IFG
Impaired Fasting Glucose
- IGT
Impaired Glucose Tolerance
- OGTT
Oral Glucose Tolerance Test
- OR
Odds Ratio
- RECPAM
RECursive Partitioning and AMalgamation
- T0′
OGTT-baseline value
- T120′
OGTT-value after 2 h
Authors' contributions
AN, BP, AN were responsible for study conception and design; AN, LS, BP, AT, MD, CF, RF, CL, MS, EV, MB, ET collected data; GG, AN, MCR analyzed data; AN, BP, MCR, AN drafted the article; LS, AT, MD, OB, CF, GF, RF, CG, CL, ES, MAS, MS, EV, MB, ET were responsible for critical revision for important intellectual content. All authors approved the version to be published.
Funding
Open access funding provided by Università degli Studi di Roma La Sapienza within the CRUI-CARE Agreement. The study was promoted and funded by the AMD-SID Diabetes and Pregnancy Study Group, Rome, Italy. The editorial assistance was provided by Airon Communication through a Novo Nordisk S.p.A. unconditional grant. The authors of the publication are fully responsible for the contents and conclusions. Novo Nordisk S.p.A. did not influence and has not been involved in the data interpretation and statistical analysis presented in the manuscript.
Availability of data and materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Compliance with ethical standards
Conflict of interest
None.
Ethical Standard Statement
The study was conducted in accordance with the Helsinki Declaration on Medical Research on Humans and with the Good Clinical Practice (GCP). The study was approved by the Ethics Committees of all participating Centers.
Informed consent
Participant patients gave informed consent before taking part in the study.
Footnotes
The original online version of this article was revised: The correct version of figure 3 updated and the collaborator author names updated as Study group in Acknowledgements section.
This article belongs to the topical collection Pregnancy and Diabetes, managed by Antonio Secchi and Marina Scavini.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
6/11/2021
A Correction to this paper has been published: 10.1007/s00592-021-01730-w
Contributor Information
Angela Napoli, Email: angela.napoli@uniroma1.it.
the STRONG Study Collaborators and the AMD-SID Diabetes and Pregnancy Study Group:
Angela Napoli, Olimpia Bitterman, Camilla Festa, Chiara Giuliani, Matteo Bonomo, Basilio Pintaudi, Elena Cimino, Elena Mion, Teresa Marcone, Cristina Lencioni, Graziano Di Cianni, Laura Sciacca, Andrea Tumminia, Agostino Milluzzo, Ester Vitacolonna, Federica Fraticelli, Marica Franzago, Alessandro Roberto Dodesini, Elena Ciriello, Mariagrazia Dalfrà, Annunziata Lapolla, Raffaella Fresa, Aurora Grassi, Paolo Limone, Annamaria Nuzzi, Andi Masha, Laura Grimaldi, Sara Biglino, Egle Ansaldi, Maurizia Battezzati, Giancarla Meregalli, Valentina De Mori, Denise Berzi, Antonio Bossi, Viviana Baggi, Elisabetta Lovati, Lara Quarleri, Tiziana Romanelli, Silvia Clementi, Ilaria Nicolao, Francesca Zambotti, Simonetta Lombardi, Silvana Costa, Chiara Tommasi, Silvia Rancan, Giovanna Lisato, Paola Bordon, Daniela Turazzi, Francesco Mollo, Franco Grimaldi, Laura Tonutti, Sandra Agus, Maria Rosaria Falivene, Giorgio Versari, Laura Corsi, Maria Delucchi, Luisa Ratto, Maria Grazia Magotti, Tiziana Frusca, Silvia Haddoub, Alice Suprani, Mary Mori, Maria Grazia Vita, Nicolina Di Biase, Alessandra Bertolotto, Michele Aragona, Cristina Bianchi, Emilia Lacaria, Elisa Guarino, Federica Monaci, Francesco Dotta, Elisabetta Torlone, Carlo Lalli, Chiara Di Loreto, Maura Scarponi, Angela Del Prete, Sergio Leotta, Iolanda Coletta, Santina Abbruzzese, Valeria Montani, Emanuela Cannarsa, Pierpaolo Contini, Raffaella Vero, Rosa Oliverio, Marina Scavini, Nicoletta Dozio, Maria Pia Imbergamo, Renzo Cordera, Laura Affinito, Davide Maggi, Caterina Bordone, Elena Fochesato, Alessandra Pissarelli, Eros Libera, Susanna Morano, Tiziana Filardi, and Mara Fallarino
References
- 1.American Diabetes Association Standards of medical care in diabetes 2020. Diabetes Care. 2020;43(Supplement 1):S1. doi: 10.2337/dc20-Sint. [DOI] [PubMed] [Google Scholar]
- 2.Buckley BS, Harreiter J, Damm P, et al. Gestational diabetes mellitus in Europe: prevalence, current screening practice and barriers to screening. A review. Diabetes Med. 2012;29:844–854. doi: 10.1111/j.1464-5491.2011.03541.x. [DOI] [PubMed] [Google Scholar]
- 3.Lapolla A, Dalfra MG, Lencioni C, Di Cianni G. Epidemiology of diabetes in pregnancy: a review of Italian data. Diabetes Nutr Metab. 2004;17:358–367. [PubMed] [Google Scholar]
- 4.HAPO Study Cooperative Research Group Hyperglycemia and adverse pregnancy outcomes. N Engl J Med. 2008;358:1991–2002. doi: 10.1056/NEJMoa0707943. [DOI] [PubMed] [Google Scholar]
- 5.O’Sullivan EP, Avalos G, O’Reilly M, Dennedy MC, Gaffney G, Dunne F. Atlantic DIP Collaborators. Atlantic Diabetes in pregnancy (DIP): the prevalence and outcomes of gestational diabetes mellitus using new diagnostic criteria. Diabetologia. 2011;54:1670–1675. doi: 10.1007/s00125-011-2150-4. [DOI] [PubMed] [Google Scholar]
- 6.Watson D, Rowan J, Neale L, Battin MR. Admissions to neonatal intensive care unit following pregnancies complicated by gestational or by gestational or type 2 diabetes. Aust N Z J Obstet Gynaecol. 2003;43:429–432. doi: 10.1046/j.0004-8666.2003.00116.x. [DOI] [PubMed] [Google Scholar]
- 7.Bellamy L, Casas JP, Hingorani AD, Williams D. Type 2 diabetes mellitus after gestational diabetes: a systematic review and meta-analysis. Lancet. 2009;373:1773–1779. doi: 10.1016/S0140-6736(09)60731-5. [DOI] [PubMed] [Google Scholar]
- 8.Goueslard K, Cottenet J, Mariet AS, Sagot P, Petit JM, Quantin C. Early screening for type 2 diabetes following gestational diabetes mellitus in France: hardly any impact of the 2010 guidelines. Acta Diabetol. 2017;54:645–651. doi: 10.1007/s00592-017-0986-x. [DOI] [PubMed] [Google Scholar]
- 9.Reece EA, Leguizamo G, Wiznitzer A. Gestational diabetes: the need for a common ground. Lancet. 2009;373:1789–1797. doi: 10.1016/S0140-6736(09)60515-8. [DOI] [PubMed] [Google Scholar]
- 10.International Association of Diabetes and Pregnancy Study Groups Consensus Panel. Metzger BE, Gabbe SG, Persson B, Buchanan TA, Catalano PA, Damm P, Dyer AR, Leiva Ad, Hod M, Kitzmiler JL, Lowe LP, McIntyre HD, Oats JJ, Omori Y, Schmidt MI. International Association of Diabetes and Pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care. 2010;33:676–682. doi: 10.2337/dc09-1848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Linea guida gravidanza fisiologica. Sistema Nazionale per le Linee Guida dell’Istituto Superiore di Sanità. http://www.salute.gov.it/imgs/C_17_pubblicazioni_1436_allegato.pdf. Accessed 14 Apr 2020
- 12.Pintaudi B, Fresa R, Dalfrà M, Marcone T, Dodesini AR, Napoli A, Bonomo M. Level of implementation of guidelines on screening and diagnosis of gestational diabetes: a national survey. Diabetes Res Clin Pract. 2016;113:48–52. doi: 10.1016/j.diabres.2015.12.008. [DOI] [PubMed] [Google Scholar]
- 13.Pintaudi B, Fresa R, Dalfrà M, Dodesini AR, Vitacolonna E, Tumminia A, Sciacca L, Lencioni C, Marcone T, Lucisano G, Nicolucci A, Bonomo M, Napoli A. The risk stratification of adverse neonatal outcomes in women with gestational diabetes (STRONG) study. Acta Diabetol. 2018;55:1261–1273. doi: 10.1007/s00592-018-1208-x. [DOI] [PubMed] [Google Scholar]
- 14.Ciampi A (1992) Constructing prediction trees from data: the RECPAM approach. In: Proceedings from the Prague 1991 summer school on computational aspects of model choice. Physica Verlag, Heidelberg, pp165–178
- 15.Rasmussen K, Yaktine AL (eds) (2009) Institute of Medicine; National Research Council. Weight Gain During Pregnancy: Reexamining the Guidelines. National Academies Press, Washington DC [PubMed]
- 16.de Gennaro G, Bianchi C, Aragona M, Battini L, Baronti W, Brocchi A, Del Prato S, Bertolotto A. Postpartum screening for Type 2 Diabetes Mellitus in women with Gestational Diabetes: is it really performed? Diabetes Res Clin Pract. 2020;166:108309. doi: 10.1016/j.diabres.2020.108309. [DOI] [PubMed] [Google Scholar]
- 17.Kitzmiller JL, Dang-Kilduff L, Taslimi MM. Gestational diabetes after delivery. Short-term management and long-term risks. Diabetes Care. 2007;30:S225–S235. doi: 10.2337/dc07-s221. [DOI] [PubMed] [Google Scholar]
- 18.Catalano PM, Vargo KM, Bernstein IM, Amini SB. Incidence and risk factors associated with abnormal postpartum glucose tolerance in women with gestational diabetes. Am J Obstet Gynecol. 1991;165:914–919. doi: 10.1016/0002-9378(91)90438-w. [DOI] [PubMed] [Google Scholar]
- 19.Pastore I, Chiefari E, Vero R, Brunetti A. Postpartum glucose intolerance: an updated overview. Endocrine. 2018;59:481–494. doi: 10.1007/s12020-017-1388-0]. [DOI] [PubMed] [Google Scholar]
- 20.Pallardo LF, Herranz L, Martin-Vaquero P, Garcia-Ingelmo T, Grande C, Jañez M. Impaired fasting glucose and impaired glucose tolerance in women with prior gestational diabetes are associated with a different cardiovascular profile. Diabetes Care. 2003;2003(26):2318–2322. doi: 10.2337/diacare.26.8.2318). [DOI] [PubMed] [Google Scholar]
- 21.Quaresima P, Visconti F, Chiefari E, Puccio L, Foti DP, Venturella R, Vero R, Brunetti A, Di Carlo C. Barriers to postpartum glucose intolerance screening in an Italian population. Int J Environ Res Public Health. 2018;15:2853. doi: 10.3390/ijerph15122853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Balaji B, RanjitMohan A, Rajendra P, Mohan D, Ram U, Viswanathan M. Gestational diabetes mellitus postpartum follow-up testing: challenges and solutions. Can J Diabetes. 2019;43:641–646. doi: 10.1016/j.jcjd.2019.04.011. [DOI] [PubMed] [Google Scholar]
- 23.O'Reilly SL, Dunbar JA, Versace V, Janus E, Best JD, Carter R, Oats JJ, Skinner T, Ackland M, Phillips PA, Ebeling PR, Reynolds J, Shih ST, Hagger V, Coates M, Wildey C. Mothers after gestational diabetes in Australia (MAGDA): a randomised controlled trial of a postnatal diabetes prevention program. PLoS Med. 2016;13:e1002092. doi: 10.1371/journal.pmed.1002092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Waters TP, Kim SY, Sharma AJ, Schnellinger P, Bobo JK, Woodruff RT, Cubbins LA, Haghiac M, Minium J, Presley L, Wolfe H, Hauguel-de Mouzon S, Adams W, Catalano PM. Longitudinal changes in glucose metabolism in women with gestational diabetes, from late pregnancy to the postpartum period. Diabetologia. 2000;63:385–394. doi: 10.1007/s00125-019-05051-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Metzger BE, Buchanan TA, Coustan DR, de Leiva A, Dunger DB, Hadden DR, Hod M, Kitzmiller JL, Kjos SL, Oats JN, Pettitt DJ, Sacks DA, Zoupas C. Summary and recommendations of the Fifth International Workshop-Conference on Gestational Diabetes Mellitus. Diabetes Care. 2007;30 Suppl 2:S251–S260. doi: 10.2337/dc07-s225. [DOI] [PubMed] [Google Scholar]
- 26.Farahvar S, Walfisch A, Sheiner E. Gestational diabetes risk factors and long-term consequences for both mother and offspring: a literature review. Expert Rev Endocrinol Metab. 2019;14:63–74. doi: 10.1080/17446651.2018.1476135. [DOI] [PubMed] [Google Scholar]
- 27.Eppel D, Feichtinger M, Lindner T, Kotzaeridi G, Rosicky I, Yerlikaya-Schatten G, Eppel W, Husslein P, Tura A, Göbl CS. Association between maternal triglycerides and disturbed glucose metabolism in pregnancy. Acta Diabetol. 2021 doi: 10.1007/s00592-020-01644-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kugishima Y, Yasuhi I, Yamashita H, Fukuda M, Kuzume A, Sugimi S, Umezaki Y, Suga S, Kusuda N. Risk factors associated with abnormal glucose tolerance in the early postpartum period among Japanese women with gestational diabetes. Int J Gynaecol Obstet. 2015;129:42–45. doi: 10.1016/j.ijgo.2014.09.030. [DOI] [PubMed] [Google Scholar]
- 29.Pallardo F, Herranz L, Garcia-Ingelmo T, Grande C, Martin-Vaquero P, Jañez M, Gonzalez A. Early postpartum metabolic assessment in women with prior gestational diabetes. Diabetes Care. 1999;22:1053–1058. doi: 10.2337/diacare.22.7.1053. [DOI] [PubMed] [Google Scholar]
- 30.Katon J, Reiber G, Williams MA, Yanez D, Miller E. Hemoglobin A1c and postpartum abnormal glucose tolerance among women with gestational diabetes mellitus. Obstet Gynecol. 2012;119:566–574. doi: 10.1097/AOG.0b013e3182475ac2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sesmilo G, Prats P, Garcia S, Rodríguez I, Rodríguez-Melcón A, Berges I, Serra B. First-trimester fasting glycemia as a predictor of gestational diabetes (GDM) and adverse pregnancy outcomes. Acta Diabetol. 2020;57(6):697–703. doi: 10.1007/s00592-019-01474-8. [DOI] [PubMed] [Google Scholar]
- 32.Pintaudi B, Di Vieste G, Corrado F, Lucisano G, Pellegrini F, Giunta L, Nicolucci A, D'Anna R, Di Benedetto A. Improvement of selective screening strategy for gestational diabetes through a more accurate definition of high-risk groups. Eur J Endocrinol. 2013;170:87–93. doi: 10.1530/EJE-13-0759. [DOI] [PubMed] [Google Scholar]
- 33.Colatrella A, Loguercio V, Mattei L, Trappolini M, Festa C, Stoppo M, Napoli A. Hypertension in diabetic pregnancy: impact and long-term outlook. Best Pract Res Clin Endocrinol Metab. 2010;24:635–651. doi: 10.1016/j.beem.2010.05.003. [DOI] [PubMed] [Google Scholar]
- 34.Festa C, Mattei L, Bitterman O, Pintaudi B, Framarino Dei Malatesta M, Bianchi P, Trappolini M, Colatrella A, Napoli A. Hypertensive disorders during pregnancy and 3 years after delivery in women with gestational hyperglycemia. J Endocrinol Investig. 2018;41:1075–1082. doi: 10.1007/s40618-018-0833-0. [DOI] [PubMed] [Google Scholar]
- 35.Colatrella A, Braucci S, Festa C, Bianchi P, Fallucca F, Mattei L, Trappolini M, Napoli A. Hypertensive disorders in normal/over-weight and obese type 2 diabetic pregnant women. Exp Clin Endocrinol Diabetes. 2009;117:373–377. doi: 10.1055/s-0029-1220763. [DOI] [PubMed] [Google Scholar]
- 36.Aroda VR, Christophi CA, Edelstein SL, Zhang P, Herman WH, Barrett-Connor E, Delahanty LM, Montez MG, Ackermann RT, Zhuo X, Knowler WC, Ratner RE. The effect of lifestyle intervention and metformin on preventing or delaying diabetes among women with and without gestational diabetes: the diabetes prevention program outcomes study 10-year follow-up. J Clin Endocrinol Metab. 2015;100:1646–1653. doi: 10.1210/jc.2014-3761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Aroda VR, Knowler WC, Crandall JP, Perreault L, Edelstein SL, Jeffries SL, Molitch ME, Pi-Sunyer X, Darwin C, Heckman-Stoddard BM, Temprosa M, Kahn SE, Nathan DM. Metformin for diabetes prevention: insights gained from the diabetes prevention program/diabetes prevention program outcomes study. Diabetologia. 2017;60:1601–1611. doi: 10.1007/s00125-017-4361-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tumminia A, Milluzzo A, Cinti F, Parisi M, Tata F, Frasca F, Frittitta L, Vigneri R, Sciacca L. Abnormal 1-hour post-load glycemia during pregnancy impairs postpartum metabolic status: a single-center experience. J Endocrinol Investig. 2018;41:567–573. doi: 10.1007/s40618-017-0774-z. [DOI] [PubMed] [Google Scholar]
- 39.Berggren EK, Presley L, Amini SB, Hauguel-de Mouzon S, Catalano PM. Are the metabolic changes of pregnancy reversible in the first year postpartum? Diabetologia. 2015;58:1561–1568. doi: 10.1007/s00125-015-3604-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Di Biase N, Balducci S, Lencioni C, Bertolotto A, Tumminia A, Dodesini AR, Pintaudi B, Marcone T, Vitacolonna E, Napoli A. Review of general suggestions on physical activity to prevent and treat gestational and pre-existing diabetes during pregnancy and in postpartum. Nutr Metab Cardiovasc Dis. 2019;29:115–126. doi: 10.1016/j.numecd.2018.10.013. [DOI] [PubMed] [Google Scholar]
- 41.Bogdanet D, Reddin C, Macken E, Griffin TP, Fhelelboom N, Biesty L, Thangaratinam S, Dempsey E, Crowther C, Galjaard S, Maresh M, Loeken MR, Napoli A, Anastasiou E, Noctor E, de Valk HW, van Poppel MNM, Agostini A, Clarson C, Egan AM, O'Shea PM, Devane D, Dunne FP. Follow-up at 1 year and beyond of women with gestational diabetes treated with insulin and/or oral glucose-lowering agents: a core outcome set using a Delphi survey. Diabetologia. 2019;62:2007–2016. doi: 10.1007/s00125-019-4935-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kautzky-Willer A, Harreiter J, Winhofer-Stöckl Y, Bancher-Todesca D, Berger A, Repa A, Lechleitner M, Weitgasser R. Gestational diabetes mellitus (update 2019) Wien Klin Wochenschr. 2019;131:91–102. doi: 10.1007/s00508-018-1419-8. [DOI] [PubMed] [Google Scholar]
- 43.Rodrigo N, Glastras SJ. The emerging role of biomarkers in the diagnosis of gestational diabetes mellitus. J Clin Med. 2018;7:120. doi: 10.3390/jcm7060120. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Not applicable.