Abstract
Background
Observational data has indicated improved survival after radical prostatectomy (RP) compared with definitive radiotherapy (RT) in men with high-risk prostate cancer (PCa).
Objective
To compare PCa-specific mortality (PCSM) and overall mortality (OM) in men with high-risk PCa treated with RP or RT, providing information on target doses and fractionations.
Design, setting, and participants
This is an observational study from the Cancer Registry of Norway. Patients were diagnosed with high-risk PCa during 2006–2015, treated with RP ≤12 mo or RT ≤15 mo after diagnosis, and stratified according to RP or RT modality; external beam radiotherapy (EBRT; 70–<74, 74–<78, or 78 Gy), hypofractionated RT or EBRT combined with brachytherapy (BT-RT).
Outcome measurements and statistical analysis
Competing risk and Kaplan-Meier methods estimated PCSM and OM, respectively. Multivariable Cox regression models evaluated hazard ratios (HRs) for PCSM and OM.
Results and limitations
In total, 9254 patients were included (RP 47%, RT 53%). RT patients were older, had poorer performance status and more unfavorable disease characteristics. With a median follow-up time of seven and eight yrs, the overall 10-yr PCSM was 7.2% (95% confidence interval [CI] 6.4–8.0) and OM was 22.9% (95% CI 21.8–24.1). Compared with RP, EBRT 70–<74 Gy was associated with increased (HR 1.88, 95% CI 1.33–2.65, p < 0.001) and BT-RT with decreased (HR 0.49, 95% CI 0.24–0.96, p = 0.039) 10-yr PCSM. Patients treated with EBRT 70–78 Gy had higher adjusted 10-yr OM than those treated with RP.
Conclusions
In men with high-risk PCa, treatment with EBRT <74 Gy was associated with increased adjusted 10-yr PCSM and OM, and BT-RT with decreased 10-yr PCSM, compared with RP.
Patient summary
In this study, we compared mortality after radical prostatectomy (RP) and radiotherapy (RT) in men with high-risk prostate cancer (PCa); the results suggest that men receiving lower-dose RT have higher, and patients receiving brachytherapy may have lower, risk of death from PCa than patients treated with prostatectomy.
Keywords: High risk, Mortality, Prostate cancer, Prostatectomy, Radiotherapy, Treatment
Take Home Message
In this observational population-based study, curatively intended treatment with external beam radiotherapy (EBRT) <74 Gy was associated with increased, and EBRT combined with brachytherapy with decreased, prostate cancer–specific mortality compared with radical prostatectomy in men diagnosed with high-risk prostate cancer.
1. Introduction
Men diagnosed with high-risk prostate cancer (PCa) have a substantial risk of death from PCa, even after curatively intended treatment [1]. In the primary setting, curative treatment options include radical prostatectomy (RP) with extended pelvic lymph node dissection, in selected patients followed by adjuvant radiotherapy (aRT), or definitive radiotherapy (RT) combined with long-term (neo-)adjuvant androgen deprivation therapy (ADT) [2], [3].
Based on observational data, RP has been associated with increased survival compared with RT in men with high-risk PCa [4], [5], [6], [7], [8], [9], [10], [11]. External beam RT (EBRT) combined with brachytherapy (BT-RT) has, however, indicated comparable results to RP [7], [8], [12], particularly in patients with Gleason score (GS) 9–10 tumors [13]. In observational studies, adjustments for pretreatment clinical parameters attenuate survival differences between treatment groups [4], [6], but residual confounding, incomplete information on ADT, different surgical and RT techniques and application of multimodal therapy limit the interpretation and clinical application of such results [9]. Dose-escalated RT (≥74 Gy) and BT-RT have been associated with improved PCa-specific and overall survival compared with lower-dose RT in men with intermediate- to high-risk disease [14], [15], [16], [17], [18]. The majority of registry-based studies comparing the effectiveness of RP versus RT, however, do not include information on target doses and dose fractionation. In patients with locally and regionally advanced disease, RP followed by pelvic RT has been associated with increased disease-specific and overall survival compared with RP alone, with comparable results to the traditional treatment with combined RT and ADT [19], [20]. Not until the completion of the ongoing SPCG-15 trial, randomized evidence will be available on survival outcomes in men with T3 tumors treated with RP +/− aRT compared with dose-escalated RT combined with ADT (NCT02102477). Further, even in patients with high-risk PCa, survival differences among curative treatment groups may become evident only after long-term follow-up [21].
With this background, the aim of this cohort study is to compare PCa-specific (PCSM) and overall (OM) mortality in men with high-risk PCa treated with RP or definitive RT regimens, providing information on RT techniques/modalities and adjusting for available clinical variables recorded in a population-based cancer registry.
2. Patients and methods
2.1. Data sources
PCa cases were identified by the Cancer Registry of Norway (CRN), which supplied date of and age at diagnosis, as well as information on prior cancers. Clinically relevant data were obtained from the Norwegian Prostate Cancer Registry (NoPCR), a clinical registry with national coverage administered by the CRN. The following variables were extracted: Eastern Cooperative Oncology Group (ECOG) performance status (PS), prostate-specific antigen (PSA) level, TNM categories, GS and date of RP. The National Radiotherapy Database, also administered by the CRN, provided data on RT (treatment intention, target dose to the prostate and start date) [22]. Information on the date and cause of death was collected from the Cause of Death Registry. The study was approved by the Regional Committee for Medical and Health Research Ethics (2011/1746).
2.2. Patients
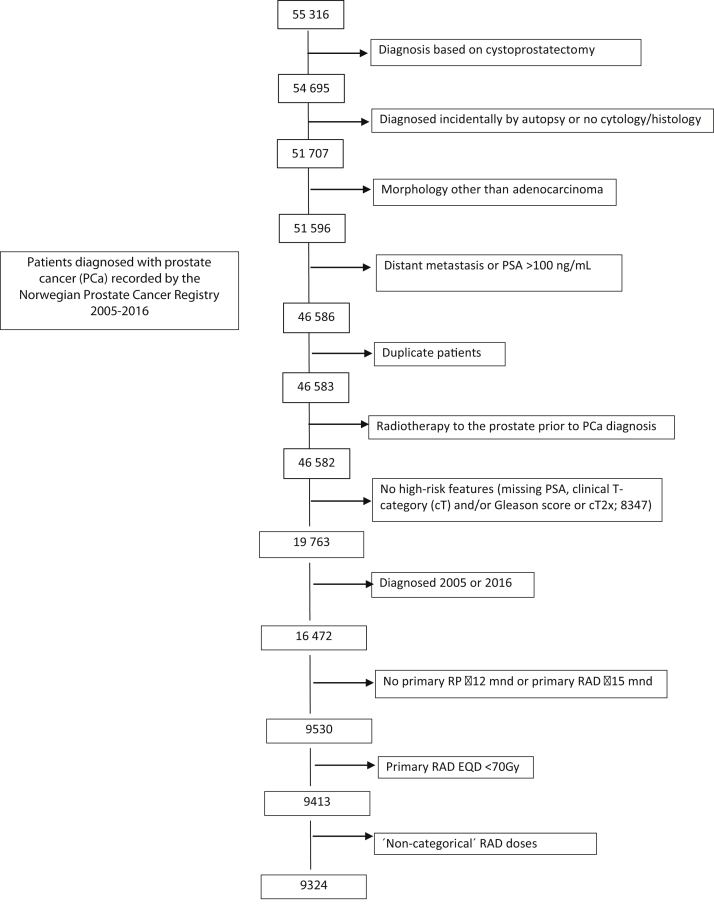
Eligible patients were diagnosed with PCa in Norway from 2006 to 2015 and classified with high risk disease according to the European Association of Urology (PSA >20 ng/ml or GS 8–10 or clinical (c)T category ≥2c) [2]. Complete information on clinical N category was not available. The following exclusion criteria were applied:
-
1
No cytological/histological verification of cancer, diagnosis based on cystoprostatectomy or morphology other than adenocarcinoma
-
2
Evidence of distant metastases or PSA >100 ng/ml
-
3
No curative treatment
Curative treatment was defined as RP performed ≤12 mo after diagnosis, with or without aRT initiated within 4 mo of RP [23], or RT ≥70 Gy started ≤15 mo after diagnosis. Performance of pelvic lymph node dissection, the use of pelvic lymph node–directed RT or (neo-)adjuvant ADT were not documented reliably in the registry. Long-term ADT (2–3 yrs) was, however, routine practice in high-risk RT patients in Norway during the study period. Patients receiving RT were stratified into the following subgroups: conventionally fractionated EBRT 70–<74 Gy or dose-escalated EBRT 74–<78 Gy or 78 Gy. We also identified patients who received ≥2.7 Gy/d × 25 (HYPO-RT) and high-dose-rate brachytherapy (BT) combined with EBRT (BT-RT) [24]. Patients who received other RT regimens were excluded (89 patients).
2.3. Statistical methods
Descriptive statistics were presented as means or medians for continuous variables, and as frequencies and percentages for categorical variables. For survival analyses, patients were followed from the date of primary treatment to the date of the event of interest (death from PCa or death from all causes), date of emigration or date of administrative censoring, whichever came first. Based on complete information on vital status and cause of death, administrative censoring occurred on December 31, 2019 when analyzing OM and December 31, 2018 when analyzing PCSM.
The standard Kaplan-Meier method estimated OM, whereas PCSM was estimated using the Aalen-Johansen estimator, incorporating the competing risk of death from other causes. Differences in OM curves were tested using standard log-rank tests. Differences in PCSM curves were tested by estimating a univariable Fine-Gray regression model. Independent-sample t test and standard chi-square test were used for differences in clinical parameters among treatment groups.
As we were interested in estimating the effects of treatment on PCSM and OM, which could facilitate some causal interpretation, we estimated cause-specific hazard ratios (CSHRs) from multivariable Cox regressions. When estimating CSHRs for PCSM, this meant censoring all individuals on the date of death if they died of other causes. To better understand the effects of confounding, we first estimated univariable Cox regressions and compared the CSHR estimates with estimates from two multivariable models. The first multivariable model adjusted only for basic confounders such as age at diagnosis (<60, 60–69, 70–79, and 80+ yrs), cT category (T1–2 vs T3–4), PSA level (<10, 10–20, and >20 ng/ml), and GS (6–10). In the second model, we added diagnostic period (2006–2010 vs 2011–2015), health region (southeast, west, middle and north), prior cancer diagnosis (yes/no), and ECOG PS (0, 1, and ≥2). A p value of <0.05 was considered statistically significant. All analyses were done using SPSS v25.1 and Stata v16.1.
3. Results
In total, 9254 patients were eligible for the present study, of whom 4306 (47%) underwent RP and 4948 (53%) underwent RT (Table 1 and Supplementary Table 1). The use of RP increased during the study period, comprising 29% of all curatively treated high-risk patients in 2006 and 54% in 2015 (Supplementary Table 1). In total, 300 RP patients received aRT. The majority of patients who underwent RT received EBRT 74–<78 Gy (18%) or EBRT 78 Gy (18%). There were significant regional differences according to RT modalities applied (Table 1).
Table 1.
Characteristics of patients receiving primary curative treatment for high-risk prostate cancer in Norway during 2006–2015
| Treatment group | RP | Def-RT | EBRT 70–<74 Gy | EBRT 74–<78 Gy | EBRT 78 Gy | HYPO-RT | BT-RT | All |
|---|---|---|---|---|---|---|---|---|
| Number of patients | 4306 | 4948 | 606 | 1651 | 1692 | 565 | 434 | 9254 |
| Diagnostic period | ||||||||
| 2006–2010 | 1443 (34) | 2393 (48) | 514 (85) | 579 (30) | 849 (50) | 181 (32) | 270 (62) | 3836 (42) |
| 2011–2015 | 2863 (66) | 2555 (52) | 92 (15) | 1072 (65) | 843 (50) | 384 (68) | 164 (38) | 5418 (59) |
| Health region | ||||||||
| Southeast | 2611 (61) | 2644 (53) | 485 (80) | 1578 (96) | 148 (9) | 14 (3) | 419 (97) | 5255 (57) |
| West | 534 (12) | 872 (18) | 56 (9) | 19 (1) | 243 (14) | 548 (97) | 6 (1) | 1406 (15) |
| Middle | 766 (18) | 954 (19) | 34 (6) | 10 (<1) | 907 (54) | 0 | 3 (<1) | 1720 (19) |
| North | 380 (9) | 459 (9) | 25 (4) | 38 (2) | 390 (23) | 0 | 6 (1) | 839 (9) |
| Unknown | 15 (<1) | 19 (<1) | 6 (1) | 6 (<1) | 4 (<1) | 3 (<1) | 0 | 34 (<1) |
| Age group (yrs), mean | 64 | 69 | 68 | 70 | 69 | 68 | 67 | 66 |
| <60 | 1197 (28) | 542 (11) | 84 (14) | 135 (8) | 177 (11) | 70 (12) | 76 (18) | 1739 (19) |
| 60–<70 | 2615 (61) | 2282 (46) | 305 (50) | 718 (44) | 775 (46) | 257 (46) | 227 (52) | 4897 (53) |
| 70–<80 | 492 (11) | 2049 (41) | 211 (35) | 762 (46) | 719 (43) | 226 (40) | 131 (30) | 2541 (28) |
| 80+ | 2 (<1) | 75 (2) | 6 (1) | 36 (2) | 21 (1) | 12 (2) | 0 | 77 (<1) |
| ECOG PS | ||||||||
| 0 | 3259 (76) | 3250 (66) | 423 (70) | 965 (58) | 1114 (66) | 421 (75) | 327 (75) | 6509 (70) |
| 1 | 304 (7) | 853 (17) | 90 (15) | 319 (19) | 322 (19) | 68 (12) | 54 (12) | 1157 (13) |
| ≥2 | 52 (1) | 212 (4) | 35 (6) | 94 (6) | 64 (4) | 8 (1) | 11 (3) | 264 (3) |
| Missing | 691 (16) | 633 (13) | 58 (10) | 273 (17) | 192 (11) | 68 (12) | 42 (10) | 1324 (14) |
| Prior cancer | ||||||||
| No | 4062 (94) | 4514 (91) | 560 (92) | 1497 (91) | 1539 (91) | 510 (90) | 408 (94) | 8576 (93) |
| Yes | 244 (6) | 434 (9) | 46 (8) | 154 (9) | 153 (9) | 55 (10) | 26 (6) | 678 (7) |
| PSA (ng/ml), median | 10 | 15 | 17 | 15 | 15 | 20 | 14 | 12 |
| <10 | 2183 (51) | 1407 (28) | 167 (28) | 495 (30) | 482 (29) | 113 (20) | 150 (35) | 3590 (39) |
| 10–20 | 1019 (24) | 1434 (29) | 154 (25) | 467 (28) | 532 (31) | 154 (27) | 127 (29) | 2453 (27) |
| >20 | 772 (18) | 1779 (36) | 241 (40) | 529 (32) | 630 (37) | 251 (44) | 128 (30) | 2551 (28) |
| Missing | 332 (8) | 4620 (93) | 44 (7) | 160 (10) | 48 (3) | 47 (8) | 29 (7) | 660 (7) |
| Gleason score | ||||||||
| 6 | 539 (13) | 440 (9) | 88 (15) | 101 (6) | 172 (10) | 45 (8) | 34 (8) | 979 (11) |
| 7a | 1064 (25) | 1009 (20) | 138 (23) | 286 (17) | 373 (22) | 99 (18) | 113 (26) | 2073 (22) |
| 7b | 639 (15) | 880 (18) | 104 (17) | 266 (16) | 317 (19) | 107 (19) | 86 (20) | 1519 (16) |
| 8 | 1417 (33) | 1572 (32) | 179 (30) | 630 (38) | 443 (26) | 183 (32) | 137 (32) | 2989 (32) |
| 9–10 | 567 (13) | 977 (20) | 76 (13) | 350 (21) | 367 (22) | 126 (22) | 58 (13) | 1544 (17) |
| Missing | 80 (2) | 70 (1) | 21 (4) | 18 (1) | 20 (1) | 5 (<1) | 6 (1) | 150 (2) |
| cT category | ||||||||
| 1–2 | 2673 (62) | 2025 (41) | 267 (44) | 631 (38) | 712 (42) | 245 (43) | 170 (39) | 4698 (51) |
| 3a | 919 (21) | 1617 (33) | 175 (29) | 521 (32) | 538 (32) | 196 (35) | 187 (43) | 2536 (27) |
| 3b | 238 (6) | 518 (11) | 34 (6) | 206 (13) | 212 (13) | 51 (9) | 15 (4) | 756 (8) |
| 3x | 175 (4) | 417 (8) | 94 (16) | 119 (7) | 147 (9) | 22 (4) | 35 (8) | 592 (6) |
| 4 | 17 (<1) | 60 (1) | 6 (1) | 25 (2) | 25 (2) | 2 (<1) | 2 (<1) | 77 (<1) |
| Missing | 284 (7) | 311 (6) | 30 (5) | 149 (9) | 58 (3) | 49 (9) | 25 (6) | 595 (6) |
BT-RT = brachytherapy combined with EBRT; cT category = clinical tumor category; Def-RT = definitive radiotherapy; EBRT = external beam radiotherapy; ECOG PS = Eastern Cooperative Oncology Group performance status; HYPO-RT = hypofractionated radiotherapy; PSA = prostate specific antigen; RP = radical prostatectomy.
Compared with RP patients, men treated with RT were older and had higher ECOG PS, PSA levels, GSs (RT: 52% GS 8–10 vs RP: 46%), and cT categories (RT: 53% cT3–4 vs RP: 31%; Table 1). Comparing RP and BT-RT patients, similar differences emerged. Within the RT subgroups, BT-RT patients were younger and had lower ECOG scores than the average RT patient, and patients who received EBRT 74–<78 Gy and HYPO-RT had the highest GSs (Table 1).
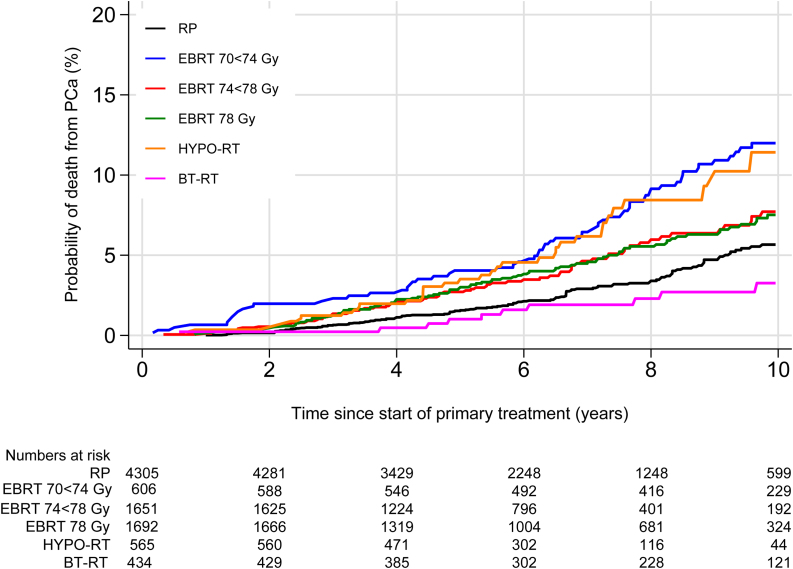
The median follow-up time was seven yrs for the evaluation of PCSM and eight yrs for OM. For all patients, the 5- and 10-yr PCSM rates were 2.3% (95% confidence interval [CI] 2.0–2.6) and 7.2% (95% CI 6.4–8.0), and OM 8.1% (95% CI 7.5–8.7) and 22.9% (95% CI 21.8–24.1), respectively (Table 2). Patients treated with RP and BT-RT had the lowest, and those treated with EBRT 70–<74 Gy had the highest 10-yr PCSM rates (Table 2 and Fig. 1). Unadjusted 10-yr PCSM was significantly higher in patients receiving EBRT 70–<78 Gy and HYPO-RT than in patients treated with RP (Supplementary Table 2 and Fig. 1). When restricting the analysis to patients with GS 8–10 disease, the risk of PCa death was higher after RP than after BT-RT (Supplementary Fig. 1). BT-RT was not superior to RP in subanalyses of patients with localized or locally advanced disease, and only EBRT 70–<74 Gy was inferior to RP in patients with cT3–4 tumors (Supplementary Fig. 2A and 2B).
Table 2.
Five-year and 10-yr prostate cancer–specific mortality (PCSM) and overall mortality (OM) in men with high-risk prostate cancer according to primary treatment
| Treatment | 5-yr PCSM | 10-yr PCSM |
|---|---|---|
| RP | 1.5 (1.2–2.0) | 5.7 (4.6–6.9) |
| EBRT 70–<74 Gy | 4.0 (2.7–5.9) | 12.0 (9.4–15.0) |
| EBRT 74–<78 Gy | 2.7 (2.0–3.6) | 7.7 (5.9–9.8) |
| EBRT 78 Gy | 2.9 (2.1–3.8) | 7.5 (6.0–9.3) |
| HYPO-RT | 3.5 (2.2–5.4) | 11.4 (7.5–16.3) |
| BT-RT | 1.0 (0.3–2.5) | 3.3 (1.6–5.8) |
| Total | 2.3 (2.0–2.6) | 7.2 (6.4–8.0) |
| 5-yr OM | 10-yr OM | |
|---|---|---|
| RP | 5.2 (4.6–5.9) | 15.5 (14.0–17.1) |
| EBRT 70–<74 Gy | 11.9 (9.5–14.7) | 32.4 (28.5–36.6) |
| EBRT 74–<78 Gy | 11.3 (9.8–12.9) | 30.9 (27.7–34.3) |
| EBRT 78 Gy | 10.6 (9.2–12.3) | 28.6 (26.0–31.4) |
| HYPO-RT | 11.1 (8.7–14.0) | 28.6 (23.4–34.6) |
| BT-RT | 5.4 (3.6–8.1) | 15.4 (11.9–19.8) |
| Total | 8.1 (7.5–8.7) | 22.9 (21.8–24.1) |
BT-RT = brachytherapy combined with EBRT; EBRT = external beam radiotherapy; HYPO-RT = hypofractionated radiotherapy; OM = overall mortality; PCSM = prostate cancer–specific mortality; RP = radical prostatectomy.
Fig. 1.
Prostate cancer–specific mortality according to treatment modality in men with high-risk prostate cancer. BT-RT = external beam radiotherapy combined with brachytherapy; EBRT = external beam radiotherapy; HYPO-RT = hypofractionated radiotherapy; PCa = prostate cancer; RP = radical prostatectomy.
When adjusted for clinical covariates, including age and tumor risk factors (GS, PSA, and cT category), EBRT 70–<74 Gy was associated with higher 10-yr PCSM than RP (hazard ratio [HR] 1.88, 95% CI 1.33–2.65, p < 0.001; Table 3). Using RP as a reference, the risk of PCa death was reduced by half in the BT-RT group (HR 0.49, 95% CI 0.24–0.96, p = 0.039). Inclusion of diagnostic period, health region, prior cancer diagnosis and ECOG PS in the analysis did not alter these results (Table 3). No differences in adjusted HR for PCa death emerged according to treatment at 5 yrs follow-up (Supplementary Table 3). There was no significant difference in 10-yr PCSM between RP patients treated with or without aRT (Supplementary Table 4).
Table 3.
Adjusted Cox regression of 10-yr prostate cancer–specific (PCSM) and overall (OM) mortality in men with high-risk prostate cancer
| PCSM |
OM |
|||||||
|---|---|---|---|---|---|---|---|---|
| Model 1 |
Model 2 |
Model 1 |
Model 2 |
|||||
| HR | p value | HR | p value | HR | p value | HR | p value | |
| Treatment | ||||||||
| RP | 1 | 1 | 1 | 1 | ||||
| EBRT 70–<74 Gy | 1.88 | 0.000 | 1.54 | 0.023 | 1.75 | 0.000 | 1.56 | 0.000 |
| EBRT 74–<78 Gy | 1.09 | 0.627 | 0.95 | 0.795 | 1.55 | 0.000 | 1.40 | 0.001 |
| EBRT 78 Gy | 1.08 | 0.615 | 1.10 | 0.617 | 1.50 | 0.000 | 1.50 | 0.000 |
| HYPO-RT | 1.24 | 0.340 | 1.04 | 0.882 | 1.32 | 0.023 | 1.14 | 0.377 |
| BT-RT | 0.49 | 0.039 | 0.43 | 0.019 | 0.87 | 0.372 | 0.81 | 0.173 |
| Age (yrs) | ||||||||
| <60 | 1 | 1 | 1 | 1 | ||||
| 60–69 | 0.93 | 0.598 | 0.86 | 0.316 | 1.40 | 0.000 | 1.33 | 0.002 |
| 70–79 | 1.06 | 0.704 | 1.05 | 0.777 | 2.20 | 0.000 | 2.00 | 0.000 |
| 80+ | 1.16 | 0.798 | 1.01 | 0.993 | 2.60 | 0.000 | 1.96 | 0.023 |
| Gleason score | ||||||||
| 6 | 1 | 1 | 1 | 1 | ||||
| 7a | 2.46 | 0.007 | 2.46 | 0.008 | 1.56 | 0.001 | 1.52 | 0.001 |
| 7b | 4.71 | 0.000 | 4.71 | 0.000 | 1.82 | 0.000 | 1.79 | 0.000 |
| 8 | 5.13 | 0.000 | 4.94 | 0.000 | 1.93 | 0.000 | 1.85 | 0.000 |
| 9–10 | 13.66 | 0.000 | 13.97 | 0.000 | 2.78 | 0.000 | 2.75 | 0.000 |
| PSA (ng/ml) | ||||||||
| <10 | 1 | 1 | 1 | 1 | ||||
| 10–20 | 1.20 | 0.188 | 1.21 | 0.189 | 1.30 | 0.000 | 1.31 | 0.000 |
| >20 | 1.48 | 0.004 | 1.53 | 0.003 | 1.53 | 0.000 | 1.49 | 0.000 |
| cT category | ||||||||
| T1–2 | 1 | 1 | 1 | 1 | ||||
| T3–4 | 1.35 | 0.007 | 1.40 | 0.004 | 1.08 | 0.200 | 1.13 | 0.048 |
| Diagnostic period | ||||||||
| 2006–2010 | 1 | 1 | ||||||
| 2011–2015 | 0.68 | 0.008 | 0.96 | 0.593 | ||||
| Health region | ||||||||
| Southeast | 1 | 1 | ||||||
| West | 0.99 | 0.956 | 1.06 | 0.609 | ||||
| Middle | 0.83 | 0.340 | 0.82 | 0.062 | ||||
| North | 0.88 | 0.552 | 0.91 | 0.394 | ||||
| Prior cancer | ||||||||
| No | 1 | 1 | ||||||
| Yes | 1.15 | 0.502 | 1.66 | 0.000 | ||||
| ECOG PS | ||||||||
| 0 | 1 | 1 | ||||||
| 1 | 1.04 | 0.786 | 1.52 | 0.000 | ||||
| ≥2 | 0.81 | 0.491 | 1.86 | 0.000 | ||||
BT-RT = brachytherapy combined with EBRT; cT category = clinical tumor category; EBRT = external beam radiotherapy; ECOG PS = Eastern Cooperative Oncology Group performance status; HR = hazard ratio; HYPO-RT = hypofractionated radiotherapy; OM = overall mortality, PCSM = prostate cancer–specific mortality, PSA = prostate-specific antigen; RP = radical prostatectomy.
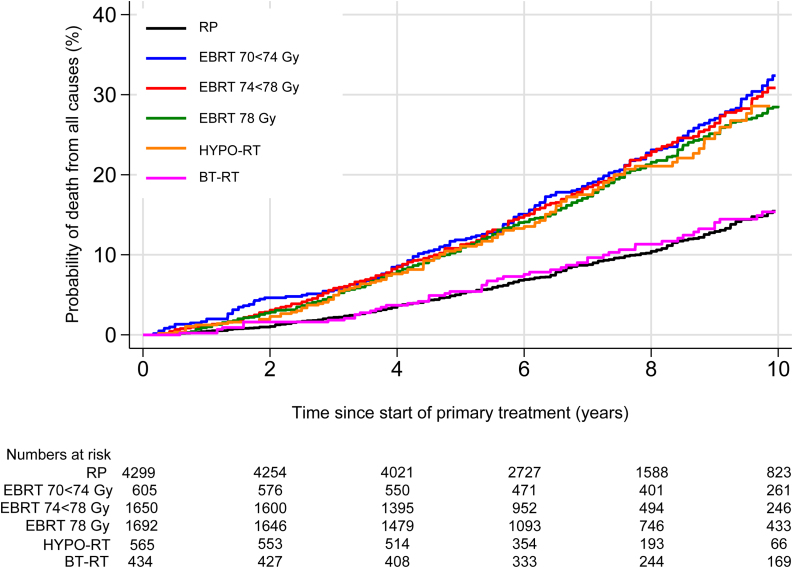
Similar to PCSM, the 10-yr OM rates were lowest in the RP and BT-RT groups, and highest among patients treated with EBRT 70–<74 Gy (Table 2, Supplementary Table 2 and Fig. 2). Adjusted for covariates, patients receiving EBRT 70–78 Gy had an increased risk of overall death after 5 and 10 yrs compared with RP patients (Table 3 and Supplementary Table 3). Increasing age, ECOG PS and prior cancer, along with increasing GS, PSA levels, and locally advanced disease, increased the risk of overall death (Table 3).
Fig. 2.
Overall mortality according to treatment modality in men with high-risk prostate cancer. BT-RT = external beam radiotherapy combined with brachytherapy; EBRT = external beam radiotherapy; HYPO-RT = hypofractionated radiotherapy; RP = radical prostatectomy.
4. Discussion
In this population-based historical prospective cohort study, comprising patients with high-risk PCa only, primary treatment with EBRT 70–<74 Gy was associated with increased 10-yr PCSM and EBRT 70–78 Gy with increased 10-yr OM, compared with RP. The risk of death due to PCa was lowest in men treated with BT-RT. The survival differences emerged beyond 5 yrs of follow-up.
Definitions of high-risk PCa comprise heterogeneous subgroups of patients with multiple combinations of prognostic clinical parameters (PSA, GS, and cT category) influencing selection and outcomes of treatment. In this study, the proportion of patients with extraprostatic tumors ranged from 32% in the RP group to 56% in the BT-RT group (the majority having cT3a tumors in both groups). Crude survival rates according to treatment are therefore of limited value in comparative effectiveness studies, even in patients within the same risk category. Although the availability is limited in public registries, adjustments for clinical covariates impact the associations between curative treatment modality and mortality, in most cases by attenuating survival differences, and more so when analyzing PCSM compared with OM.
In the absence of randomized evidence, registry-based studies have previously demonstrated survival benefits with RP over RT in patients with high-risk PCa. However, in the current study, providing details on RT modalities, the increase in adjusted HR for PCa death with RT compared with RP was limited to patients receiving EBRT doses 70–<74 Gy. Similarly, dose-escalated RT (≥74 Gy) has been associated with lower 10-yr PCSM and OM in high-risk patients than with lower RT doses [14], [15], [16]. Our findings of reduced PCSM in men treated with BT-RT compared with those treated with RP, are in agreement with the conclusion of a recent systematic review by Greenberger et al [7]. Moreover, in line with our findings, Kishan et al [13] demonstrated significantly better PCSM with BT-RT than with EBRT and RP in patients with GS 9–10 tumors. In the present study, a survival benefit with BT-RT compared with RP in men with locally advanced disease approached the level of significance (p = 0.059).
There was no difference in OM between the RP and BT-RT groups, and we can only speculate whether unrecognized residual confounding by comorbidity not covered by ECOG performance status affects RT patients more than RP patients. This may also contribute to the increased risk of all-cause death in patients receiving EBRT compared with those treated with RP.
With adjustments for pretreatment clinical factors, RP followed by aRT was not associated with a reduction in PCSM compared with RP alone in this dataset; however, patients with more aggressive tumors, based on pathological examination of the RP specimen, would have been more likely to receive aRT in the study period. Excluding the aRT patients from analysis did not significantly alter the treatment-related HRs for 10-yr PCSM (data not shown).
Our findings justify already well-established trends and contemporary guidelines defining RP, dose-escalated EBRT (≥74 Gy), and BT-RT with long-term ADT as valid curative treatment options for men with high-risk PCa [2]. Whether modernly hypofractionated, in particular ultrafractionated, RT is non-inferior to curative treatment modalities in relation to 10-yr PCSM, is so far unknown [25]. Although results are significant in large cohorts, during counselling of individual patients, any survival benefit of a treatment in a population must be balanced against the risk of toxicity and altered health-related quality of life [8].
Besides the limitations related to use of registry data for the purpose of comparing treatments, there are other limitations to this study. First, regarding patient characteristics, we did not have detailed information on comorbidity and socioeconomic status, nor did we have complete data of clinical N category. The performance of pelvic lymph node dissection or pelvic lymph node–directed RT was not documented reliably in this study, although neither procedure has been proved to increase survival in PCa patients [26]. Considering long-term (neo-)adjuvant ADT reducing disease-specific mortality and OM in RT patients with a high risk of recurrence [7], [15], [19], [27], complete information on ADT was unavailable. Further, no information on disease progression and second-line PCa treatments was available in the CRN. Finally, residual confounding in the treatment groups, even with adjustments for all parameters available in the registry, and the potential excess registration of death from PCa in old patients, must be acknowledged [28].
5. Conclusions
This population-based cohort study including men with high-risk PCa demonstrates increased PCSM and OM with nonescalated dose RT (<74 Gy) and suggests decreased PCSM with BT-RT compared with RP.
To obtain high-level comparative evidence on mortality outcomes in patients with high-risk PCa undergoing contemporary multimodal curative therapies, including RP and BT-RT, studies documenting details of RP and RT techniques, (neo-)adjuvant systemic treatments and long-term follow-up are called for.
Author contributions: Kirsti Aas had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study concept and design: Aas, Fosså, Myklebust.
Acquisition of data: Aas, Myklebust.
Analysis and interpretation of data: Aas, Berge, Fosså, Myklebust.
Drafting of the manuscript: Aas, Fosså, Myklebust.
Critical revision of the manuscript for important intellectual content: Aas, Berge, Fosså, Myklebust.
Statistical analysis: Aas, Myklebust.
Obtaining funding: Aas, Fosså.
Administrative, technical, or material support: Aas, Myklebust.
Supervision: Aas, Fosså, Myklebust.
Other: None.
Financial disclosures: Kirsti Aas certifies that all conflicts of interest, including specific financial interests and relationships and affiliations relevant to the subject matter or materials discussed in the manuscript (eg, employment/affiliation, grants or funding, consultancies, honoraria, stock ownership or options, expert testimony, royalties, or patents filed, received, or pending), are the following: None.
Funding/Support and role of the sponsor: The Vestre Viken Hospital Trust (project no. 2503002), the Norwegian Cancer Society (fund no. 5781243), and the Radium Hospital Foundation (project no. 335007) have provided financial support.
Data sharing: The data that support the findings of this study are available from the Cancer Registry of Norway. Restrictions apply to the availability of these data, which were used under license for this study. Data are available from the authors with the permission of the Cancer Registry of Norway.
CRediT authorship contribution statement
Kirsti Aas: Conceptualization, Methodology, Formal analysis, Investigation, Writing - original draft, Writing - review & editing, Visualization, Project administration, Funding acquisition. Viktor Berge: Writing - review & editing. Tor Åge Myklebust: Methodology, Formal analysis, Investigation, Writing - review & editing, Visualization. Sophie Dorothea Fosså: Conceptualization, Data curation, Investigation, Methodology, Project administration, Writing - review & editing.
Associate Editor: Guillaume Ploussard
Footnotes
Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.euros.2021.01.011.
Appendix A. Patient selection
Appendix B. Supplementary data
The following are Supplementary data to this article:
References
- 1.Fossa S.D., Wiklund F., Klepp O. Ten- and 15-yr prostate cancer-specific mortality in patients with nonmetastatic locally advanced or aggressive intermediate prostate cancer, randomized to lifelong endocrine treatment alone or combined with radiotherapy: final results of the Scandinavian Prostate Cancer Group-7. Eur Urol. 2016;70:684–691. doi: 10.1016/j.eururo.2016.03.021. [DOI] [PubMed] [Google Scholar]
- 2.Mottet N., Bellmunt J., Bolla M. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2017;71:618–629. doi: 10.1016/j.eururo.2016.08.003. [DOI] [PubMed] [Google Scholar]
- 3.Moris L., Cumberbatch M.G., Van den Broeck T. Benefits and risks of primary treatments for high-risk localized and locally advanced prostate cancer: an international multidisciplinary systematic review. Eur Urol. 2020;77:614–627. doi: 10.1016/j.eururo.2020.01.033. [DOI] [PubMed] [Google Scholar]
- 4.Sooriakumaran P., Nyberg T., Akre O. Comparative effectiveness of radical prostatectomy and radiotherapy in prostate cancer: observational study of mortality outcomes. BMJ. 2014;348:g1502. doi: 10.1136/bmj.g1502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wallis C.J.D., Saskin R., Choo R. Surgery versus radiotherapy for clinically-localized prostate cancer: a systematic review and meta-analysis. Eur Urol. 2016;70:21–30. doi: 10.1016/j.eururo.2015.11.010. [DOI] [PubMed] [Google Scholar]
- 6.Robinson D., Garmo H., Lissbrant I.F. Prostate cancer death after radiotherapy or radical prostatectomy: a nationwide population-based observational study. Eur Urol. 2018;73:502–511. doi: 10.1016/j.eururo.2017.11.039. [DOI] [PubMed] [Google Scholar]
- 7.Greenberger B.A., Zaorsky N.G., Den R.B. Comparison of radical prostatectomy versus radiation and androgen deprivation therapy strategies as primary treatment for high-risk localized prostate cancer: a systematic review and meta-analysis. Eur Urol Focus. 2020;6:404–418. doi: 10.1016/j.euf.2019.11.007. [DOI] [PubMed] [Google Scholar]
- 8.Wang Z., Ni Y., Chen J. The efficacy and safety of radical prostatectomy and radiotherapy in high-risk prostate cancer: a systematic review and meta-analysis. World J Surg Oncol. 2020;18:42. doi: 10.1186/s12957-020-01824-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Williams S.B., Huo J., Chamie K. Discerning the survival advantage among patients with prostate cancer who undergo radical prostatectomy or radiotherapy: the limitations of cancer registry data. Cancer. 2017;123:1617–1624. doi: 10.1002/cncr.30506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gu X., Gao X., Cui M. Survival outcomes of radical prostatectomy and external beam radiotherapy in clinically localized high-risk prostate cancer: a population-based, propensity score matched study. Cancer Manag Res. 2018;10:1061–1067. doi: 10.2147/CMAR.S157442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Petrelli F., Vavassori I., Coinu A., Borgonovo K., Sarti E., Barni S. Radical prostatectomy or radiotherapy in high-risk prostate cancer: a systematic review and metaanalysis. Clin Genitourin Cancer. 2014;12:215–224. doi: 10.1016/j.clgc.2014.01.010. [DOI] [PubMed] [Google Scholar]
- 12.Ennis R.D., Hu L., Ryemon S.N., Lin J., Mazumdar M. Brachytherapy-based radiotherapy and radical prostatectomy are associated with similar survival in high-risk localized prostate cancer. J Clin Oncol. 2018;36:1192–1198. doi: 10.1200/JCO.2017.75.9134. [DOI] [PubMed] [Google Scholar]
- 13.Kishan A.U., Cook R.R., Ciezki J.P. Radical prostatectomy, external beam radiotherapy, or external beam radiotherapy with brachytherapy boost and disease progression and mortality in patients with Gleason score 9-10 prostate cancer. JAMA. 2018;319:896–905. doi: 10.1001/jama.2018.0587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kuban D.A., Levy L.B., Cheung M.R. Long-term failure patterns and survival in a randomized dose-escalation trial for prostate cancer. Who dies of disease? Int J Radiat Oncol Biol Phys. 2011;79:1310–1317. doi: 10.1016/j.ijrobp.2010.01.006. [DOI] [PubMed] [Google Scholar]
- 15.Kalbasi A., Li J., Berman A. Dose-escalated irradiation and overall survival in men with nonmetastatic prostate cancer. JAMA Oncol. 2015;1:897–906. doi: 10.1001/jamaoncol.2015.2316. [DOI] [PubMed] [Google Scholar]
- 16.Pettersson A., Alm D., Garmo H. Comparative effectiveness of different radical radiotherapy treatment regimens for prostate cancer: a population-based cohort study. JNCI Cancer Spectr. 2020;4:pkaa006. doi: 10.1093/jncics/pkaa006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Morris W.J., Tyldesley S., Rodda S. Androgen suppression combined with elective nodal and dose escalated radiation therapy (the ASCENDE-RT trial): an analysis of survival endpoints for a randomized trial comparing a low-dose-rate brachytherapy boost to a dose-escalated external beam boost for high- and intermediate-risk prostate cancer. Int J Radiat Oncol Biol Phys. 2017;98:275–285. doi: 10.1016/j.ijrobp.2016.11.026. [DOI] [PubMed] [Google Scholar]
- 18.Pasalic D., Kuban D.A., Allen P.K. Dose escalation for prostate adenocarcinoma: a long-term update on the outcomes of a phase 3, single institution randomized clinical trial. Int J Radiat Oncol Biol Phys. 2019;104:790–797. doi: 10.1016/j.ijrobp.2019.02.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fahmy O., Khairul-Asri M.G., Hadi S., Gakis G., Stenzl A. The role of radical prostatectomy and radiotherapy in treatment of locally advanced prostate cancer: a systematic review and meta-analysis. Urol Int. 2017;99:249–256. doi: 10.1159/000478789. [DOI] [PubMed] [Google Scholar]
- 20.Jang T.L., Patel N., Faiena I. Comparative effectiveness of radical prostatectomy with adjuvant radiotherapy versus radiotherapy plus androgen deprivation therapy for men with advanced prostate cancer. Cancer. 2018;124:4010–4022. doi: 10.1002/cncr.31726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ekanger C., Helle S.I., Heinrich D. Ten-year results from a phase II study on image guided, intensity modulated radiation therapy with simultaneous integrated boost in high-risk prostate cancer. Adv Radiat Oncol. 2020;5:396–403. doi: 10.1016/j.adro.2019.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Asli L.M., Kvaloy S.O., Jetne V. Utilization of radiation therapy in Norway after the implementation of the national cancer plan—a national, population-based study. Int J Radiat Oncol Biol Phys. 2014;90:707–714. doi: 10.1016/j.ijrobp.2014.06.059. [DOI] [PubMed] [Google Scholar]
- 23.Vatne K., Stensvold A., Myklebust T.A. Pre- and post-prostatectomy variables associated with pelvic post-operative radiotherapy in prostate cancer patients: a national registry-based study. Acta Oncol. 2017;56:1295–1301. doi: 10.1080/0284186X.2017.1314006. [DOI] [PubMed] [Google Scholar]
- 24.Fossa S.D.F., Aas K., Müller C. Definitive radiotherapy in Norway 2006–2015: temporal trends, performance and survival. Radiother Oncol. 2020;155:33–41. doi: 10.1016/j.radonc.2020.10.022. [DOI] [PubMed] [Google Scholar]
- 25.Widmark A., Gunnlaugsson A., Beckman L. Ultra-hypofractionated versus conventionally fractionated radiotherapy for prostate cancer: 5-year outcomes of the HYPO-RT-PC randomised, non-inferiority, phase 3 trial. Lancet. 2019;394:385–395. doi: 10.1016/S0140-6736(19)31131-6. [DOI] [PubMed] [Google Scholar]
- 26.Fossati N., Willemse P.M., Van den Broeck T. The benefits and harms of different extents of lymph node dissection during radical prostatectomy for prostate cancer: a systematic review. Eur Urol. 2017;72:84–109. doi: 10.1016/j.eururo.2016.12.003. [DOI] [PubMed] [Google Scholar]
- 27.Bolla M., Van Tienhoven G., Warde P. External irradiation with or without long-term androgen suppression for prostate cancer with high metastatic risk: 10-year results of an EORTC randomised study. Lancet Oncol. 2010;11:1066–1073. doi: 10.1016/S1470-2045(10)70223-0. [DOI] [PubMed] [Google Scholar]
- 28.Loffeler S., Halland A., Weedon-Fekjaer H., Nikitenko A., Ellingsen C.L., Haug E.S. High Norwegian prostate cancer mortality: evidence of over-reporting. Scand J Urol. 2018;52:122–128. doi: 10.1080/21681805.2017.1421260. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.