Abstract
Transitional cell carcinoma of the bladder is a common malignancy with an estimated 549 393 new cases occurring in 2018 alone. Both non–muscle-invasive bladder cancer (NMIBC) and muscle-invasive bladder cancer (MIBC) show high recurrence and progression rates, and therefore impose a great burden on patients and health care systems. Current risk stratification and therapy strategies are predominantly based on clinical and histopathological findings for tumor stage and grade. The chemoresistance and metastasis of low-grade tumors suggest an incomplete understanding of disease mechanisms, despite numerous studies on differentiating molecular subtypes of bladder cancer to identify tumor drivers and potential therapeutic targets. We present a highly unusual course for a low-grade bladder tumor leading to metastasis and death, for which we used postmortem histopathological and molecular analyses to evaluate targetable alterations in key signaling pathways driving the underlying tumor biology.
Keywords: Non–muscle-invasive bladder cancer, Luminal subtype, FGFR3, ERBB2, KRT20, Metastasis, Death
1. Case series
In November 2018, a healthy 70-yr-old male underwent transurethral resection of the bladder (TURB) in our clinic after an episode of gross hematuria and a bladder sonogram showing a tumor mass of 3.5 cm. The patient had a history of smoking and hypotension and no family history of malignancies. A prophylactic stent was implanted in the right ureter as its orifice was close to the resected area.
Histopathology revealed a single noninvasive papillary urothelial cell cancer with good differentiation (pTa; World Health Organization [WHO]/International Society of Urological Pathology: low grade; WHO 1973: grade 2). A second TURB and ureteroscopy were performed at 6 wk, but no tumor residue was microscopically detected.
After 7 mo, a tumor recurrence with the same histopathological characteristics in a second location was treated, including postoperative installation of mitomycin C.
After 2 wk, the patient was readmitted to hospital, presenting progressive neck pain, dyspnea, a cough, and abdominal discomfort. Computed tomography images revealed multiple solid liver lesions (Fig. 1 shows corresponding autopsy results), pulmonary and multiple bone metastases, and enlarged retroperitoneal and para-aortic lymph nodes. A biopsy taken from a bone lesion confirmed the same immunhistological characteristics as the previous bladder specimens.
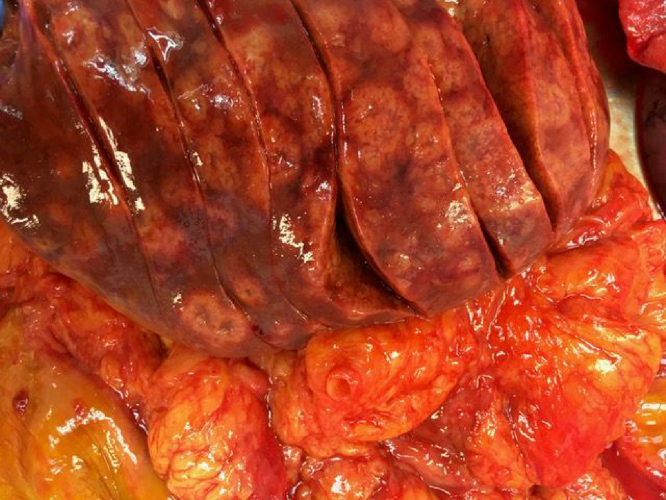
Fig. 1.
Liver metastases.
According to the tumor board recommendation, palliative platin-based chemotherapy was about to be implemented when rapid deterioration of the patient’s general condition was observed and the patient died within days from cancer-related liver failure.
Consent was obtained to perform an autopsy to investigate the cause of the patient’s rapid death. No muscle-invasive tumor was found in bladder, ureter, or kidney specimens. Apart from the known sites of metastases, pathology findings included an intact basement membrane in the bladder, but extensive lymphangitic and hemangiotic carcinomatosis in the bladder, lungs, right ureter, and adjacent tissue.
Further analysis was performed in collaboration with a second institute of pathology to investigate the tumor biology at a molecular level and identify tumor markers and drivers (Supplementary material). mRNA expression levels of the genes investigated are listed in Table 1, Table 2; CALM2 (encoding calmodulin) was used as reference.
Table 1.
mRNA expression of immune-associated genes in bladder tissue samples from the patient
| Specimen | CALM2 | PD-L1 | PD-1 | CXCL9 | CD3 | CD8 |
|---|---|---|---|---|---|---|
| Healthy bladder tissue | 26.84 | 35.75 | 32.50 | 33.46 | 35.00 | 34.27 |
| Primary bladder tumor | 25.42 | 33.35 | 26.85 | 32.76 | 28.17 | 30.84 |
Bold values represent tumor associated overexpressions among investigated genes and were used to emphazise this aspect.
Table 2.
mRNA expression of investigated genes in tissue samples from the patient
| Specimen | CALM2 | KRT5 | KRT20 | ERBB2 | FGFR1 | FGFER3 |
|---|---|---|---|---|---|---|
| Healthy bladder tissue | 26.82 | 34.47 | 35.98 | 37.39 | 35.75 | 36.95 |
| Primary bladder tumor | 26.57 | 36.52 | 40.79 | 39.33 | 32.36 | 41.02 |
| Recurrent bladder tumor | 25.32 | 37.56 | 40.33 | 39.78 | 32.64 | 41.26 |
| Liver metastasisa | 34.49 | 40.31 | 38.41 | 38.94 | 35.28 | 41.03 |
| Bone metastasisa | 34.79 | 36.20 | 40.16 | 39.53 | ND | 39.59 |
| Lymph nodea | 33.19 | 35.27 | 39.72 | 35.76 | 34.91 | 39.86 |
| Right uretera | 34.45 | 38.55 | ND | 37.13 | 35.15 | 40.03 |
ND = not detected.
Bold values represent tumor associated overexpressions among investigated genes and were used to emphazise this aspect.
Obtained from autopsy.
Strong overexpression of FGFR3 and ERBB2 was found across primary and metastatic lesions. A negative association was found between KRT5 and KRT20 expression levels. A reverse association between these two markers and FGFR1 and FGFR2 was demonstrated on Pearson correlation analysis (Table 3). Furthermore a positive association was found between ERBB2 expression levels and FGFR2–FGFR4, but not FGFR1.
Table 3.
Pearson correlation of mRNA expression of investigated genes
| KRT5 | KRT20 | ERBB2 | FGFR1 | FGFR2 | FGFER3 | FGFR4 | |
|---|---|---|---|---|---|---|---|
| KRT5 | 1.0000 | −0.3712 | 0.2592 | 0.3913 | −0.2815 | 0.4767 | 0.5763 |
| KRT20 | −0.3712 | 1.0000 | 0.4097 | −0.3095 | 0.9928 | 0.2942 | 0.4088 |
| ERBB2 | 0.2592 | 0.4097 | 1.0000 | −0.4610 | 0.4282 | 0.5351 | 0.5955 |
| FGFR1 | 0.3913 | −0.3095 | −0.4610 | 1.0000 | −0.3127 | 0.4777 | 0.3540 |
| FGFR2 | −0.2815 | 0.9928 | 0.4282 | −0.3127 | 1.0000 | 0.2982 | 0.4462 |
| FGRF3 | 0.4767 | 0.2942 | 0.5351 | 0.4777 | 0.2982 | 1.0000 | 0.9394 |
| FGFR4 | 0.5763 | 0.4088 | 0.5955 | 0.3540 | 0.4462 | 0.9394 | 1.0000 |
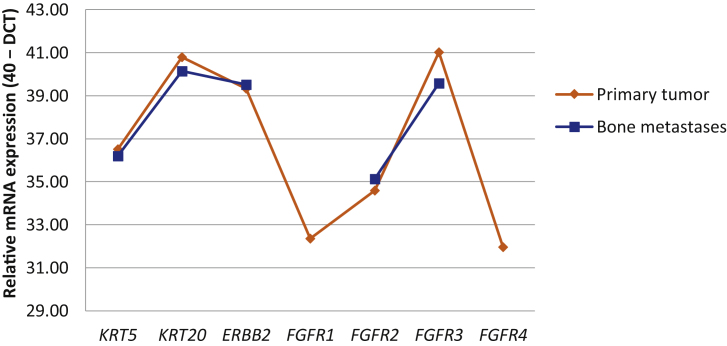
The FGFR3 mutation p.S249C was identified in all specimens and confirmed by independent methods. The stability of mRNA overexpression in primary tumor and bone lesions is illustrated in Figure 2.
Fig. 2.
Comparison of the relative mRNA expression of candidate genes in primary tumor and bone metastasis tissue.
DCT = Delta for the cycle threshold.
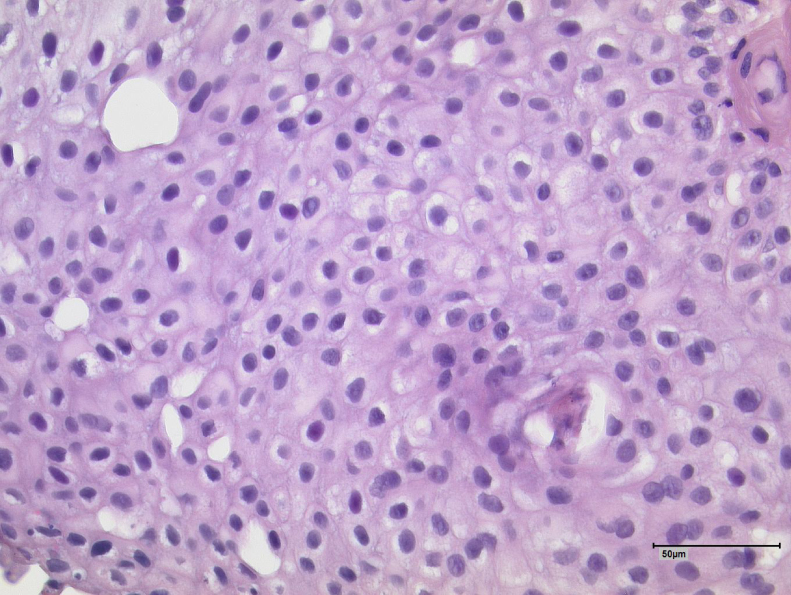
Hematoxylin and eosin stains of the primary tumor samples were reanalyzed and a consistent picture of a low-grade tumor without stroma invasion was confirmed (Fig. 3).
Fig. 3.
Hematoxylin and eosin staining of a sample of the primary bladder tumor.
2. Discussion
Bladder cancer is a common malignancy worldwide. In approximately three-quarters of patients, NMIBC is diagnosed, and of the remaining quarter with MIBC, metastatic stage is found in 5% of cases. Imaging beyond ultrasound is recommended in selected patients with multiple, high-risk or trigonum tumors, as well as in MIBC. Current therapy strategies are mainly based on histopathological tumor stage and grading, with local management for NMIBC and systematic treatment and radical cystectomy for MIBC. However, even after radical therapy, local recurrences and distant metastasis occur in up to 50% of cases [1].
In 2006, the European Organization for Research and Treatment of Cancer developed a risk calculator for NMIBC that is frequently used to predict the risk of recurrence and progression [2]. In our case the risk of recurrence was 24% within the first year (46% within 5 yr) and the risk of progression was 1% (6% within 5 yr).
An incomplete understanding of tumor biology and responsiveness to current therapy strategies has led to numerous studies on differentiating molecular subtypes of bladder cancer. Tumor heterogeneity at the molecular level has been identified and two major subtypes as well as subclasses have been described for NMIBC and MIBC. There are similarities to breast cancer subtypes, for which targeted therapies are well established [3], [4]. Subclassification appears to be necessary for targeted therapies, as they show distinct differences in biological behavior and chemotherapy sensitivity [3], [5]. However, intratumoral heterogeneity and variant classification sets in MIBC further complicate patient therapy approaches. A consensus on classification systems in MIBC has only recently been proposed [6], [7].
Our patient had expression of luminal subtype markers with high mRNA expression of KRT20 and immunohistochemical expression of GATA3 within the primary tumor and all metastatic lesions. Interestingly, overexpression of the luminal marker KRT20 was stable across all lesions, while expression of the basal marker KRT5 varied to some extent between sites, possibly indicating that the phenotypic basal subtype contributions are less stable than the genotypic target overexpression (Table 2).
As previously reported, luminal tumors with high KRT20 mRNA expressions are negatively associated with T-cell infiltration (CD3, CD8, PD-1) and chemokines that attract T cells (CXCL9) [8]. Low expressions of all immune markers may indicate the capacity of the initial tumor to prevent immune cell infiltration by T cells and therefore might reflect a higher risk of recurrence and progression [8].
ERBB2 overexpression is often found in high-grade tumors and seems to be associated with high recurrence rates and worse progression-free survival for T1 NMBC; it also serves as a discriminating factor between high- and low-risk tumors [9]. Despite well-established agents targeting Her2 in breast and gastric cancers, trials of different agents to treat MIBC have not yet shown sufficient efficacy [10]. A possible explanation for this might be that studies have primarily been based on gene amplification rather than mRNA expression of ERBB2, with the latter appearing less consistent in bladder cancer than in breast cancer.
For many carcinomas, the FGFR pathway seems to play a role in tumor growth, invasion, and progression. Dysregulated expression and alteration of FGFR1 and FGFR3 are often found in bladder cancer, with a missense mutation leading to p.S249C in FGFR3, one of three major alterations [10]. This p.S249C mutation in FGFR3 and Her2/neu overexpression could be understood as concerted action of two tumor drivers and could possibly serve as a starting point for targeted therapy.
The combination of dual tyrosine kinase therapy targeting FGFR3 and ERBB2 has not yet been investigated. Given the endogenous interplay and correlations of both pathways, this combination could provide synergistic effects and overcome the limited activity of single-agent treatment.
In conclusion, it is unlikely that our patient would have improved under the planned cisplatin-based therapy. However, an early molecular analysis and determination of the extent of the cancer might have led to an experimental therapy approach via dual blockade of FGFR and ERBB signaling.
There is a great need for further investigations of molecular markers for BC in the context of risk stratification and for developing combined targeted therapy options to prevent progression and cancer-related death. Therefore, we have started a study on FGFR3 mutation analysis among bladder cancer patients, specifically including NMIBC and MIBC.
CME question
Which of the following markers is NOT related to bladder cancer?
A) FGFR3
B) HER-2
C) CA19-9
D) TP53
Compliance with ethical standards: Informed consent was obtained from the deceased’s family.
Conflicts of interest: The authors have nothing to disclose.
Associate Editor: Guillaume Ploussard
Footnotes
Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.euros.2021.02.006.
Appendix A. Supplementary data
The following is Supplementary data to this article:
References
- 1.Foth M., Ismail N.F.B., Kung J.S.C. FGFR3 mutation increases bladder tumourgenesis by suppressing acute inflammation. J Pathol. 2018;246:3331–3343. doi: 10.1002/path.5143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sylvester R.J., van der Meijden A.P.M., Oosterlinck W. Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials. Eur Urol. 2006;49:466–477. doi: 10.1016/j.eururo.2005.12.031. [DOI] [PubMed] [Google Scholar]
- 3.Cancer Genome Atlas Research Network Comprehensive molecular characterization of urothelial bladder carcinoma. Nature. 2014;507:315–322. doi: 10.1038/nature12965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Choi W., Porter S., Kim S. Identification of distinct basal und luminal subtypes of muscle-invasive bladder cancer with different sensitivities to frontline chemotherapy. Cancer Cell. 2014;25:152–165. doi: 10.1016/j.ccr.2014.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.McConkey D.J., Choi W. Molecular subtypes of bladder cancer. Curr Oncol Rep. 2018;20:77. doi: 10.1007/s11912-018-0727-5. [DOI] [PubMed] [Google Scholar]
- 6.Warrick J.I., Sjödahl G., Kaag M. Intratumoral heterogeneity of bladder cancer by molecular subtypes and histologic variants. Eur Urol. 2019;75:18–22. doi: 10.1016/j.eururo.2018.09.003. [DOI] [PubMed] [Google Scholar]
- 7.Kamoun A., de Reyniès A., Allory Y. A consensus molecular classification of muscle-invasive bladder cancer. Eur Urol. 2020;77:420–433. doi: 10.1016/j.eururo.2019.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kubon J., Sikic D., Eckstein M. Analysis of CXCL9, PD1 and PD-L1 mRNA in stage T1 non-muscle invasive bladder cancer and their association with prognosis. Cancers. 2020;12:2794. doi: 10.3390/cancers12102794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Breyer J., Wirtz R.M., Laible M. ESR1, ERBB2, and Ki67 mRNA expression predicts stage and grade of non-muscle-invasive bladder carcinoma (NMIBC) Virchows Arch. 2016;469:547–552. doi: 10.1007/s00428-016-2002-1. [DOI] [PubMed] [Google Scholar]
- 10.Koshkin V.S., O’Donnell P., Yu E.Y., Grivas P. Systematic review: targeting HER2 in bladder cancer. Bladder Cancer. 2019;5:1–12. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.